Abstract
Scorpionism is a common problem that occurs in tropical and subtropical countries and assumes great medical-sanitary importance due to its fatal effect on sensitive individuals, being able to lead children and aged people to death. The envenomation lethal potential is responsible for the serious cardiopulmonary alterations the scorpion toxin produces in its victims. The present research evaluated the effects of Tityus serrulatus venom on dogs, using two distinct doses: a dose that simulates natural envenomation (0.4 mg/total dose), and an experimental dose (0.25 mg/kg). General clinical signs were observed at different moments after envenomation, and specific data related to the cardiopulmonary system were evaluated by systemic arterial pressure measurement, CK-MB enzymatic activity dosage, and radiographic, electrocardiographic and echocardiographic examinations. Results demonstrated that the scorpion venom, in experimental doses, was able to cause acute and reversible cardiac injury in few days, and, in the dose that simulated natural accident, it produced clinical signs of light envenomation, such as local pain, hyperesthesia, sialorrhea, vomiting, diarrhea, sneeze and prostration.
scorpion; dogs; cardiopulmonary alterations; Tityus serrulatus
ORIGINAL PAPER
Clinical and cardiovascular alterations produced by scorpion envenomation in dogs
Cordeiro F. F.I; Sakate M.I; Fernandes V.II; Cuyumjian P. R.III
IDepartment of Clinical Veterinary Medicine, School of Veterinary Medicine and Animal Husbandry, São Paulo State University, UNESP, Botucatu, São Paulo, Brazil
IIClinical Laboratory, School of Veterinary Medicine and Animal Husbandry, São Paulo State University, UNESP, Botucatu, São Paulo, Brazil
IIIVeterinary Diagnostic, São Paulo, Brazil
Correspondence to Correspondence to: Fernanda F. Cordeiro Departamento de Clínica Veterinária Faculdade de Medicina Veterinária e Zootecnia- UNESP Distrito de Rubião Júnior, s/n 18.618-000, Botucatu, SP, Brasil Email: fercord@uol.com.br
ABSTRACT
Scorpionism is a common problem that occurs in tropical and subtropical countries and assumes great medical-sanitary importance due to its fatal effect on sensitive individuals, being able to lead children and aged people to death. The envenomation lethal potential is responsible for the serious cardiopulmonary alterations the scorpion toxin produces in its victims. The present research evaluated the effects of Tityus serrulatus venom on dogs, using two distinct doses: a dose that simulates natural envenomation (0.4 mg/total dose), and an experimental dose (0.25 mg/kg). General clinical signs were observed at different moments after envenomation, and specific data related to the cardiopulmonary system were evaluated by systemic arterial pressure measurement, CK-MB enzymatic activity dosage, and radiographic, electrocardiographic and echocardiographic examinations. Results demonstrated that the scorpion venom, in experimental doses, was able to cause acute and reversible cardiac injury in few days, and, in the dose that simulated natural accident, it produced clinical signs of light envenomation, such as local pain, hyperesthesia, sialorrhea, vomiting, diarrhea, sneeze and prostration.
KEY WORDS: scorpion, dogs, cardiopulmonary alterations, Tityus serrulatus.
INTRODUCTION
Scorpion envenomation is a common accident in tropical and subtropical regions. In Human Medicine, these accidents are considered a medical-sanitary problem of great importance, with the potential to cause serious clinical signs and to be fatal, especially for children (13, 22, 27). So far, studies on the syndrome caused by scorpion accidents, despite the efforts of some researchers, have not been complete. In Veterinary Medicine, there are no researches on scorpion envenomations, neither of natural nor of experimental occurrence.
In Brazil, the scorpion species most frequently related to the accidents is Tityus serrulatus (11). It is also known that the venom toxicity is related to the scorpion species involved in the accident; the most severe cases are related to the above- mentioned scorpion. Tityus serrulatus venom is composed of a toxic fraction called tityustoxin, a neurotoxin that acts on the peripheral nervous terminations and is capable of stimulating the sympathetic and parasympathetic systems with acetylcholine, adrenalin and noradrenaline release, which is responsible for most of the clinical signs in scorpion envenomation (44). The victims of accidents with arachnids develop clinical signs such as pain at the bite site, vomit, sudoresis, agitation, sinusal tachycardia or bradycardia, varied cardiac arrhythmias, respiratory disturbances, arterial hypertension or hypotension, heart failure, pulmonary shock, pulmonary edema, neurological signs, and laboratory disorders like hyperglycemia, increase in the amylase activity (17, 22), and leukocytosis due to neutrophilia (9). Increased serum levels of creatine kinase, CK (10), aspartate aminotransferase, AST, and lactate dehydrogenase, LDH, are described together with myoglobinemia and myoglobinuria, which suggests cardiac injury (26, 44). It is known that the severity of clinical signs is related to the venom plasmatic concentration (42) and to the individual susceptibility (30).
In dogs, alterations caused by envenomation of natural occurrence are related to the central nervous system and include hyperactivity, hypermotility, disorientation, hypertension, pulmonary edema and seizures, besides local pain (36). Experimentally, intravenous inoculation of venom at lethal doses (0.5-1.0 mg/kg) in dogs caused salivation, lacrimation, dyspnea, evacuation, vomiting, micturition, tremors and prostration, and these dogs died in less than six hours (30).
Some researchers have made great efforts to study the cardiovascular effect manifested by the cardiac failure and pulmonary edema present in severe scorpion envenomations, once these alterations reveal themselves potentially lethal (13, 24, 25).
Myocardic involvement in these situations has raised many controversies and has been attributed to the venom action on the sympathetic nervous system and to the massive release of catecholamines and/or to the direct action of the venom on cardiomyocytes, which may lead to a reversible "scorpionic cardiomyopathy" (13, 18, 23, 24, 43).
Some researchers have evaluated the action of scorpion venom on cardiovascular systems of dogs, in an attempt to elucidate its pathogenesis, and they have observed bradyarrhythmias and tachyarrhythmias, as well as increase in the systemic arterial pressure and in the left ventricular end-diastolic pressure. Those authors have concluded that some hemodynamic factors could explain the occurrence of cardiac failure and pulmonary edema in severe scorpion envenomation. These factors include a sudden decrease in the left ventricular compliance, a sudden increase in the systemic pressure due to release of vasoactive amines with consequent injury of the left ventricular emptying, and additive effects of bradyarrhythmia and tachyarrhythmia, which also contribute to alterations in the ventricular emptying (19).
Evidences of myocardial insult include electrocardiographic, echocardiographic and laboratory alterations that simulate acute myocardial infarction (26). The main electrocardiographic findings described in the literature are sinusal tachycardia with premature ventricular complexes, wandering pacemaker, depression of ST segment, and atrial-ventricular blockade, among others (3, 10, 11, 13, 17, 23, 25, 43, 44). Alterations observed in the QRS complex morphology and in the T wave are explained by the electrolytic imbalances caused by the scorpion venom. The main electrolytic disturbances described in association with scorpionism are hyperkalemia and hypocalcaemia (27, 39).
Echocardiographic studies reveal impaired ventricular function and ejection fraction with variable degrees of hypokinesis, resembling the clinical signs of dilated cardiomyopathy (3, 10, 11, 13, 25, 26, 29, 44). Ventricular dysfunction is clearly demonstrated by radioisotopic ventriculography, evidencing reduced values of ventricular ejection fraction associated with severe alteration in the regional parietal movement (21). In the same way, scintigraphic evaluations of myocardial perfusion with Thallium-201 demonstrate that disturbance of myocardial coronary perfusion exists (21, 32) and that coronary vasoconstriction can be considered a consequence of the excessive release of catecholamines (39).
Several mechanisms are presented as responsible for the cardiac injury, among them is the venom direct action on the cardiac cell, which causes great Ca++ influx in cardiomyocytes, producing increased inotropic activity and structural alterations (48, 55). The initial positive inotropic effect, associated with the high oxygen demand due to the massive release of catecholamines, might be responsible for the cardiac function deterioration that occurs lately (48). Another explanation relates the increase of free fatty acid levels in the blood due to lipolyses caused by the great release of catecholamines, glucagons and cortisol to the decrease of insulin levels. High concentrations of fatty acids cause greater O2 consumption by the myocardium, which does aggravate the ischemic state and produces electric instability. This occurrence is associated with the massive release of catecholamines, which also increases the cardiac O2 consumption (39), although a previous study considered improbable that the cardiac injury caused by the scorpion venom could develop due tohypoxia or anoxia (54). It has also been demonstrated degenerative injuries in the cardiac muscle fiber and necrotic areas with polymorphonuclear infiltrations (54), hemorrhagic subendocardial and subpericardial injuries, and presence of microthrombi in capillaries due to disseminated intravascular coagulation - DIC (40). Increased free fatty acid levels, due to alteration in the platelets function, may augment the tendency for intravascular thrombus and DIC (39).
Cardiac failure can also be explained by the increase in the plasmatic renin and angiotensin II levels, which can aggravate the effects caused by the excess of circulating catecholamines, once it causes coronary and peripheral vasoconstriction, increasing the afterload (39). Some authors attribute the occurrence of pulmonary edema, at least partly, to a myocardial dysfunction (1). Other researchers have related its incidence to diverse other factors, such as acute arterial hypertension of adrenergic nature, release of substances that increase the vascular permeability like cytokines and epinephrine, increase in the venous return also related to the release of catecholamines, and presence of serious sinusal tachycardia of adrenergic origin (16). Increased permeability of the alveolus-capillary membrane is demonstrated by a high concentration of plasmatic proteins in tracheobronchial wash, as well as in electronic microscopy of the pulmonary parenchyma of a victim of acute pulmonary edema due to scorpion envenomation (2).
Recent publications proved the effectiveness of platelet-activating factor and tachykinin inhibitors in suppressing the pulmonary edema caused by scorpion envenomation, suggesting the involvement of inflammatory mediators in its beginning (34), and demonstrated the prominent importance of mast cells in the development of pulmonary injury in rats inoculated with scorpion venom (14).
Thus, many researches are carried out in human medicine in an attempt to improve the therapeutic measures related to accidents by scorpions and to control the venom deleterious effects on the cardiopulmonary system. Briefly, the aim of the therapy is to suppress the envenomation signs and symptoms, to neutralize the circulating venom by means of scorpion antivenom (16, 17, 44), and to institute supportive therapy for the vital functions (4), in which vasodilatator (5, 6, 7, 20, 27, 28, 45), antiemetic, anticonvulsive and analgesic drugs are also indicated (36).
Despite the antivenom therapy, other authors also suggest the treatment with insulin-glucose infusion, which successfully reverses cardiovascular and hemodynamic changes and pulmonary edema in victims of scorpion stings (39, 56).
MATERIALS AND METHODS
Animals
Twelve adult dogs were used. Distinctions between sexes or races were not made, however the use of too much young or aged animals, those weighing more than 10 kg, and pregnant or female dogs in estrus phase was avoided. Among these animals, nine were females and three were males. They were supplied by the Central Kennel - UNESP, Botucatu, São Paulo, Brazil.
Scorpion Venom
Tityus serrulatus venom was gently supplied by the Butantan Institute, São Paulo, Brazil.
Evaluation of the animals
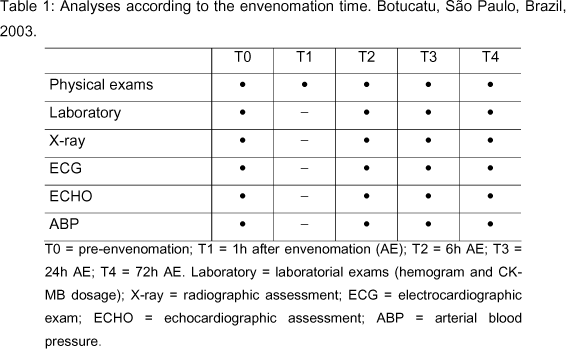
Before the beginning of the experiment, the animals were subjected to the evaluations mentioned below, and these individuals were controls of themselves. This stage was considered as T0 - pre-envenomation - and included clinical evaluation by means of physical examination of the animals; laboratory evaluation by determining the hematological profile and blood levels of the myocardium-specific isoenzyme creatine-kinase-MB (CK-MB); radiographic examination of the dogs, emphasizing the lungs; electrocardiographic evaluation; echocardiographic examination; and arterial systemic pressure measure.
Experimental Protocol
Animals were divided into two groups of six animals each: in group 1 (G1) the scorpion venom was administered subcutaneously at the dose of 0.25 mg/kg (30), and in group 2 (G2) the inoculation of the scorpion venom was also by subcutaneous injection of 0.4 mg/animal, since this is the maximum volume that can be extracted from a Tityus serrulatus scorpion by means of electrical stimulation according to Dr. Irene Knysak from the Arthropods Laboratory, Butantan Institute, São Paulo, Brazil, 2002.
After inoculation, all the animals were more incisively examined during the first 24 hours (44) and were monitored until the third day after envenomation, since the alterations disappeared in approximately 72 hours, in agreement with a pilot study. Table 1 demonstrates the moments of analyses of the twelve above-described animals.
Clinical Evaluation
This evaluation consisted of physical and laboratorial assessment, including hemogram and biochemical tests; radiographic, electrocardiographic and echocardiographic analyses; as well as arterial blood pressure measurement by the non-invasive oscillometric method (Dixtal - model 2710).
Physical Examination
The data collected consisted of corporal temperature, mucosa coloration, time of capillary filling, and cardiac and respiratory frequencies (35). Other apparent clinical signs were enumerated by observing the animals. During the physical examination, dogs were subjected to blood pressure measure and determination of the systolic and diastolic pressures (8).
Laboratorial Exams
Blood samples were collected from the external jugular vein or cephalic vein for hemogram, which was performed by means of an automatic cell counter (CELM CC 510), and leukocytes differential counting, performed by the manual method. Determination of the myocardium-specific creatine-kinase isoenzyme (CK-MB) blood levels was carried out by using a specific kit (Diagnóstica - Merck) (38).
Radiographic Examination
Thoracic radiographies (TUR 800) of the right lateral and ventrodorsal recumbence of the thorax were carried out for pulmonary evaluation and search for pulmonary edema evidences (12).
Echocardiographic Evaluation
Animals were subjected to an echocardiographic study (Hitachi - Portable 405 Model), in which the following parameters were determined: left ventricular function (DD%), ejection fraction (EF%), final left ventricle diastolic diameter (FLVDD), and final left ventricle systolic diameter (FLVSD). All these parameters were obtained by calculating an average value from three subsequent measures. The methodology used is recommended by the "Echocardiography Committee of the Cardiology Specialty" (49).
Electrocardiographic Examination
The electrocardiographic exam was recorded in limb leads and the analysis was made in DII lead, where we observed the cardiac frequency and rhythm, duration of P waves, QRS complex, PR and QT intervals in seconds (s), P and R waves amplitudes in millivolts (mV), depression or elevation of ST segment, and T wave polarity and amplitude (50). Dogs were monitored for one minute and a half in each evaluation. The computerized electrocardiography (TEB-ECG PC) was programmed to record in the speed of 50 mm/s (1 cm = 1 mV), and the normal values were those used for computerized methods (53). The cardiac frequency values used for analysis were resultant from the arithmetic mean between the cardiac frequency obtained at the electrocardiographic examination and the cardiac frequency obtained by means of physical examination.
Therapeutical Protocol
All the animals received infiltrative local anesthetic blockade without vasoconstrictor (Xylocaine® 2% - Chemical Astra and Pharmaceutical Ltda.) respecting the allowed maximum dose of 7 mg/kg (33) at the moment T1 (one hour after the venom inoculation).
Statistical Analysis
Quantitative characteristics were evaluated by analysis of variance according to the different doses of venom inoculated. Qualitative characteristics were evaluated by Q-square test for determination of the dose-effect relationship (47). Analyses were carried out using SAS-94 program, and values of p<0.05 were considered statistically significant.
RESULTS
Physical Examination
Among the 12 animals studied, the most evident clinical sign was pain at the site of venom inoculation, and it was more evident in the group that received the highest dose of the toxin (G1). In both groups, pain occurred at the injection site immediately after inoculation, spreading throughout the body in G1 animals and remaining during the first 24 hours after envenomation. During this period, the animals became stiffness and hypersensitive to touch, developing hyperaesthesia. Other clinical signs observed were sialorrhea, vomiting, diarrhea, sneezing and prostration, and these symptoms were more severe in G1 when compared to G2. Table 2 demonstrates the clinical signs observed in the different groups at the four moments after envenomation; Figure 1 demonstrates the difference between the effects of the two doses related to the clinical signs presented at T1 (1h after envenomation).
There was no statistically significant variation on the other surveyed physical parameters between the moments studied, such as corporal temperature, mucosa coloration, and cardiac and respiratory frequencies. Although the values of cardiac frequency did not demonstrate statistical difference between the moments studied, there was an increase in this parameter in four animals of G1 at the moments T2 and T3 (6h and 24h after envenomation, respectively). Differences in the cardiac frequency values among G1 animals at the moments studied and the comparison between the two groups analyzed are shown in Figures 2 and 3. As an isolated finding, we observed the occurrence of systolic murmur in the animal number 2 of G1 six hours after the venom inoculation (T2).
Local reactions at the venom inoculation site such as erythema or edema were not observed.
Laboratorial Exams
Blood analysis of the animals studied evidenced a statistical significant (p<0.05) leukocytosis and an increase in the red blood cells count, together with an increase in the hematocrit (Ht) and plasmatic protein (Pt) values at T2 (6h after envenomation) in G1. At the moment T4 (72h after envenomation), there was a significant reduction (p<0.05) in the number of red blood cells, also accompanied by the reduction in the Ht and Pt values (Figure 4). Although G2 animals demonstrated gradual reduction in the red blood cells count at the four moments studied, it was not statistically important.
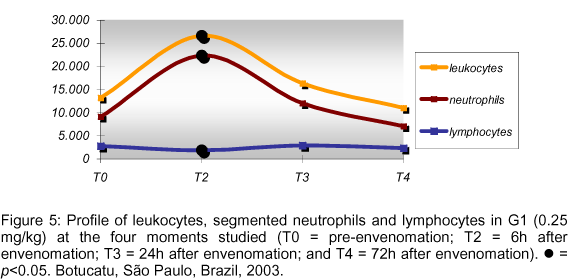
The increased number of leukocytes at T2 (6h after envenomation) was also statistically significant; for instance, leukocytosis, or a number of leukocytes higher than 18,000 leukocytes/ml, was observed in seven individuals. Out of these, five were from G1 and the other two from G2, and presented leukocytosis with neutrophilia and lymphopenia. Figure 5 demonstrates the counting of leukocytes, segmented neutrophils and lymphocytes in G1 at the moments studied.
Dosage of myocardium-specific creatine kinase isoenzyme (CK-MB) was higher at T2 (6h after envenomation) when compared to the pre-envenomation moment (T0) and similar to the other parameters; this alteration was statistically significant (p<0.05) only in G1 (Figure 6).
Radiographic Examination
There were no noteworthy radiographic alterations in the pulmonary fields of the ten animals studied; only individuals number 1 and 4 from G1 presented radiological images of diffuse interstitial opacification of the pulmonary parenchyma at T2 and T3 (6h and 24h after envenomation, respectively).
Echocardiographic Evaluation
Significant echocardiographic alterations between groups or moments were not observed, and the dimensions of cardiac chambers, such as the index of ventricular function, remained constant.
Electrocardiographic Examination
The cardiac rhythms observed in this experiment were sinusal rhythm and sinusal arrhythmia with periods of sinus arrest and did not show variation during the moments studied. Parameters such as cardiac frequency, P wave duration, PR interval, QRS complex, and QT interval, as well as ST segment, remained without remarkable changes. However, inversion of the T wave polarity and/or increase in its amplitude to values above 25% of the R wave at T2 and T3 (6h and 24h after-envenomation, respectively) in both groups were findings of electrocardiographic statistic relevance. Increase in the P and R waves amplitude in G1 animals at T2 (6h after-envenomation) also assumed statistical significance. Figure 7 illustrates the inversion of T wave polarity and the increased amplitude of R wave in a G1 animal at the four moments studied. Separately, animal number 6 from G1 presented increase in the Q wave amplitude at T2 (6h after-envenomation).
Systemic Arterial Blood Pressure
Similar to the other parameters studied, the arterial systolic and diastolic pressures were not significantly modified between the moments and neither between the groups studied.
DISCUSSION
In a general way, the results of this study showed there is a direct relationship between the amount of scorpion venom inoculated and the clinical signs observed (42), demonstrating that these signs concentrate during the first 24 hours after envenomation (44). Therefore, the most severe physical alterations were clearly observed in those animals that received 0.25 mg/kg (G1) at the moments T1, T2 and T3 (1h, 6h and 24h after envenomation).
Pain occurred at the inoculation site of 100% G1 individuals, irradiating to the body and remaining for 24 hours, although all the dogs received local anesthetic infiltrative blockade by using Xilocaína® 2% one hour after the inoculation, as previously described. From the six animals that received the dose of 0.4 mg/animal, only five demonstrated painful sensitivity at the inoculation site, which was easily controlled by infiltrative anesthesia. This denotes that, in natural scorpion accidents with adult dogs, pain is significant and a clinical finding easy to control. The painful sensitivity presented by G2 animals was slighter and disappeared quickly. Hyperesthesia with intense response to any manipulation at the inoculation site is described only in dogs (36) and, in this study, was observed in the animals of G1.
Other clinical signs evidenced were similar to those described in the literature: vomiting, prostration, tremors (3, 11, 13, 17), diarrhea (40), and excessive salivation (13, 25, 44). Only animal number 2 from G1 presented systolic apical murmur six hours after the envenomation (T2), suggesting mitral valve incompetence due to myocardial injury (3, 6, 19, 21, 24). Although occurrence of murmur in the other animals studied could not be detected by physical examination, we cannot deny a possible occurrence of valvar regurgitation in these individuals once the analysis of the blood flow by means of echodopplercardiographic examination was not carried out. Myocardial injury in the animal number 2 from G1 is also confirmed by an evident increase in the cardiac frequency at T2 and T3 (6h and 24h after envenomation) associated with a considerable increase in the values of CK-MB enzymatic activity at these two moments and alteration in the R wave amplitude at T2 (6h after envenomation). Although alterations in the ventricular function were not evidenced in any of the animals studied, myocardial injuries might have been local or little and insufficient to cause alterations in the global function.
Although there are no previous descriptions, sneezes were a very consistent sign in the envenomed animals of G1, which seems to be a late-triggered disturbance, since there was a greater incidence at the moments T2 and T3 (6h and 24h after envenomation), affecting five dogs of this group. It is known that severe scorpion envenomation causes the release of several cytokines (31), which could indicate a possible allergic reaction, justifying the sneezes observed in the animals of this study.
According to the classification presented by the literature consulted (31, 44), the animals from this study presented moderate and severe degrees of envenomation (local pain followed by at least one of the next symptoms: vomiting, prostration, salivation, and tremors), and G2 animals showed mild envenomation (only mild pain at the inoculation site).
Vomiting is described as an indicative of the scorpion envenomation severity (44) and, in fact, it was prominent in G1 during the first 24 hours after envenomation. Some studies discuss the causes of emesis in scorpionism as the occurrence of early acute pancreatitis after envenomation in dogs and rats (30, 37) presenting exocrine pancreatic hypersecretion (41). It is also associated with pancreatic dysfunction, and inhibition of the gastric emptying and intestinal transit (52) accompanied by increase in the gastrin seric levels (51).
Although there are many descriptions of clinical signs, such as tachypnea and alterations of the corporal temperature (3, 17, 44), no variation on these parameters were observed at the moments after envenomation, even in G1 individuals. Some animals of this study presented increased values of respiratory frequency at the moments observed, since hyperventilation was frequently observed in these individuals, which was clearly determined by the temperature of the place where the experiment was carried out. Similarly, none of the animals studied developed systemic arterial hypertension or hypotension, opposing to the data from literature, which report alterations in the blood pressure in human victims of scorpionism by Tityus serrulatus (3, 17, 25, 26) and in dogs experimentally subjected to Leiurus quinquestriatus scorpion envenomation (19). Severe hypertension, however, is considered less prevalent in accidents by Tityus serrulatus when compared to the hypertension caused by other species of scorpions (4). In fact, the oscillometric method cannot be considered the most sensitive in measuring the arterial pressure (8), however, the method considered "gold standard" demands the implantation of an intra-arterial catheter, which would be invasive and unfeasible considering that the individuals had their pressure measured until 72 hours after the venom inoculation. In previous studies with dogs that had developed arterial hypertension, the animals were observed under general anesthesia and for a short time, which evidently allows the modification of all the methodology employed (19, 54). Since these same studies used Leiurus quinquestriatus scorpion toxin to produce envenomation, it must be also considered that tityustoxin is not capable of causing hypertension in dogs, differently of what is described in humans.
Increase in the number of red blood cells, hematocrit, and plasmatic proteins in G1 individuals at T2 (6h after envenomation) demonstrates the effects of dehydration on the hematologic picture, coinciding with the moments in which the animals suffered the greatest corporeal liquids depletion by means of vomiting, hypersalivation, and diarrhea. Reduction in the number of red blood cells during the 72 hours after envenomation (T4) indicates the action the scorpion toxin exerts on the red blood cell membrane causing hemolysis (39). This decrease could also be justified by the disseminated intravascular coagulation caused by the venom, even though this action has been studied with the toxin of Buthus tamulus, a scorpion of another species but that belongs to the same family of Tityus serrulatus (40). These possible mechanisms could be demonstrated by the reduction in the number of red blood cells in G2 during the three moments observed after envenomation, since these animals did not present clinically significant dehydration. Leukocytosis in victims of scorpionism is reported in many studies (2, 3, 10, 11, 13, 23, 25, 31), being justified by neutrophilia due to mobilization of neutrophils from the bone marrow to the blood compartment (9). The animals studied demonstrated leukocytosis with neutrophilia and lymphopenia, which was sufficiently evident in G1 and mild in G2, characterizing the occurrence of "stress leucogram", which could be caused by the release of catecholamines described in scorpionism.
The animals from G1 also presented expressive alterations in the activity of the myocardium-specific isoenzyme CK-MB, corroborating the findings of a previous research (3, 11, 13, 44). This fact evidences the high sensitivity of the enzymatic test in identifying myocardial injury, once the animals from this experiment, even those from G1, did not demonstrate apparent cardiac injury at radiographic and echocardiographic examinations, except for electrocardiographic alterations. There was not failure of the cardiac system, although the occurrence of injury in cardiac cells was evidenced by the electrocardiographic evaluation as well as by the biochemical test (46). The use of this laboratorial evaluation in Veterinary Medicine could be useful to an early investigation of myocardial injury, being necessary more studies about the subject.
The aim of the radiographic analyses in this study was to assess the pulmonary patterns of the animals studied. Pulmonary edema in severe scorpionism of natural or experimental occurrence is commonly described (1, 2, 11, 13, 17, 19, 23, 25) and is closely related to the deaths caused by scorpion envenomation (24). Pulmonary edema either by cardiac mechanisms or by humoral mechanisms due to release of vasoactive substances (27), as a consequence of the inflammatory cascade activation (14, 31, 34) or because of the increase in the alveolus-capillary permeability (2), was not shown by the radiographic analysis in any of the animals from this study. Perhaps with the use of bronchoalveolar lavage (14), a more specific method of pulmonary analysis, alterations in the pulmonary condition after envenomation could have been early detected.
Echocardiographic analyses did not show increase in the diastolic or systolic diameters of the left ventricle in the animals studied, consequently there was no alteration in the left ventricular function, unequally to what other authors reported (3, 10, 25, 26, 29). Although myocardial necrosis was clearly manifested by the increase of the CK-MB activity in G1 animals, myocardial injuries were probably small or local and insufficient to modify the global cardiac function (46). Previous researches evaluating the coronary perfusion by scintigraphy had noted regional perfusion alterations (21, 32); also, echocardiographic studies had detected alterations in the ventricular regional wall motion (15). In the present study, we did not perform an evaluation of the ventricular regional wall motion, which could have shown discrete regional wall dysfunctionsunable to modify the global cardiac function. For example,one of the G1 animals developed transitory mitral incompetence, which could be explained by the reduction in the papillary muscles contraction or by the alteration in the mitral valve apparatus geometry and functional integrity (3).
Electrocardiographic alterations in this study were related to the ventricular repolarization, represented by the inversion of the T wave polarity or increase in its amplitude in animals envenomed at the moments T2 and T3 (6h and 24h after envenomation). Alterations in the T wave are commonly described in the literature, as well as modifications in the ST and QT segments, morphology of the QRS complex, and arrhythmic events (3, 6, 10, 11, 13, 19, 23, 24, 26). These alterations are justified by electrolytic disturbances associated with sympathetic overstimulation (39). Alterations in the T wave in this study were related to the inversion in its polarity (56), while data from many other researches are related to alterations in its morphology, described as tall, peaked and slender T waves occurring due to the hyperkalemia that happens in scorpionism (27), although Bucaretchi and his collaborators (11) observed the occurrence of hypokalemia in their study. Therefore, future investigation correlating electrocardiographic findings with serum electrolytes dosages in scorpion envenomation is needed. Inversion in the T wave polarity is an unspecific finding and is more frequently seen in patients with myocardial ischemia (15). In scorpion envenomation, it could be justified by the increase of O2 consumption by the cardiac muscle due to the metabolism and perfusion alterations that occur in scorpionism (39).
Although P and R waves amplitudes were within the normal parameters, the increase in these values was an electrocardiographic finding of statistical relevance in G1 animals six hours after envenomation (T2). It is knownthat increase in the P wave occurs in the right atrial overload, a fact that did not happen in this study, in which it could be related to the increased heart rate observed in a G1 animal at T2 (50). In the same way, the R wave amplitude correlates with the ventricular mass, and its value exceeds normality in myocardial concentric hypertrophy and dilations (50), another fact that was not evidenced by the echocardiographic investigation in our study. It would fit here the questioning about the sensitivity of the electrocardiographic method in detecting a ventricular wall thickening discrete enough to be confused with intraobserver variations that occur in echocardiographic mensurations, since the myocardial thickening in scorpionism can be resultant from inflammatory responses and edema of ventricular walls (10). However, in the present experiment, values of interventricular septal wall and left ventricle posterior wall were not determined, which would be interesting in future studies for the correlation with electrocardiographic findings. Alterations in the QRS complex morphology, demonstrated not only by the increase in the R wave amplitude but also by the aspect of Q wave in one of the animals in this study, suggest the occurrence of 'current lesion', which is capable of changing the cardiac depolarization route, modifying thus the QRS complex morphology (15). This fact, among those previously described, confirms the occurrence of myocardial injury in scorpionism.
CONCLUSION
Under the experimental conditions described in this study we can affirm that at doses similar to those of natural accidents, scorpion envenomation in dogs caused slight effects such as mild pain at the inoculation site, prostration and salivation, and these signs disappeared between one and six hours after the envenomation. The hematologic, pulmonary and cardiac states and the blood pressure did not demonstrate noteworthy alterations. At higher experimental doses, there was important pain at the venom inoculation site, prostration, vomiting, diarrhea, salivation, and sneezes. Hematologic analysis demonstrated important increase in the number of leukocytes after the envenomation associated with the reduction in the red blood cells count. The CK-MB levels increased significantly and the electrocardiographic evaluation evidenced alterations in the ventricular repolarization, pointing out the occurrence of myocardial injury. Other tests related to the cardiovascular system did not show expressive alterations. Results, therefore, indicate that the severity of clinical signs in scorpion envenomation in dogs becomes directly related to the dose of venom inoculated. Higher doses can cause cardiac aggression, and lower doses, which could simulate natural envenomation, can result in a clinical picture of mild envenomation. Thus, Tityus serrulatus toxin, at the dose of 0.25 mg/kg, can be used as an experimental inductive of cardiac injury in dogs. It is also suggested that adult dogs involved in natural scorpion accidents will probably develop mild symptomatology, nevertheless we must consider the individual sensitivity of victims, and also the possibility of cardiac alterations, due to the venom potential severity, on the cardiovascular system.
ACKNOWLEDGMENTS
The authors thank Butantan Institute for the scorpion venom donation; Dr. Maria Salete Sartori for the CK-MB analysis; Dr. Henrique Nunes for the statistical analysis; Dr. Salvador Luis Rocha Urtado and Dr. Gabriela S. Rodrigues for the radiographic analysis; Marcello Bulgarelli for the manuscript translation; and Dr. Benedicto Wlademir de Martin for all the opportunities offered.
Received: January 20, 2005
Accepted: July 20, 2005
Published online: February 24, 2006
- 1 ABROUG F., BOUJDARIA R., BELGHITH M., NOUIRA S., BOUCHOUCHA S. Cardiac dysfunction and pulmonary edema following scorpion envenomation. Chest, 1991, 100, 1057-9.
- 2 AMARAL CFS., BARBOSA AJA., LEITE VHR., TAFURI WL., REZENDE NA. Scorpion sting-induced pulmonary edema: evidence of increased alveolo-capillary membrane permeability. Toxicon, 1994, 32, 999-1003.
- 3 AMARAL CFS., MAGALHÃES RA., LOPES JA., REZENDE NA. Electrocardiographic, enzymatic and echocardiographic evidence of myocardial damage after Tityus serrulatus scorpion poisoning. Am. J. Cardiol., 1991, 67, 655-7.
- 4 AMARAL CFS., REZENDE NA. Treatment of scorpion envenomation should include both a potent specific antivenom and support of vital functions. Toxicon, 2000, 38, 1005-7.
- 5 BAWASKAR HS., BAWASKAR PH. Prazosin in management of cardiovascular manifestations of scorpion sting. Lancet, 1986, 1, 510-1.
- 6 BAWASKAR HS., BAWASKAR PH. Cardiovascular manifestations of severe scorpion sting in India (review of 34 children). Ann. Trop. Paediatr., 1991, 11, 381-7.
- 7 BAWASKAR HS., BAWASKAR PH. Vasodilators: scorpion envenomation and the heart (an Indian experience). Toxicon, 1994, 32, 1031-40.
- 8 BODEY AR. Systemic hypertension in the dog: fact or fiction? In: ANNUAL WALTHAM/OSU SYMPOSIUM FOR THE TREATMENT OF SMALL ANIMAL DISEASES, 18, Vernon, 1994. Vernon: Waltham, 1994. p. 44-53.
- 9 BORGES CM., SILVEIRA MR., APARECIDA M., BEKER CL., FREIRE-MAIA L., TEIXEIRA MM. Scorpion venom-induced neutrophilia is inhibited by a PAF receptor antagonist in the rat. J. Leukoc. Biol., 2000, 67, 515-9.
- 10 BRAND A., KEREN A., KEREM E., REIFEN RM., BRANSKI D. Myocardial damage after a scorpion sting: long-term echocardiographic follow-up. Pediatr. Cardiol., 1988, 9, 59-61.
- 11 BUCARETCHI F., BARACAT EC., NOGUEIRA RJ., CHAVES A., ZAMBRONE FA., FONSECA MR., TOURINHO FS. A comparative study of severe scorpion envenomation in children caused by Tityus bahiensis and Tityus serrulatus Rev. Inst. Med. Trop. São Paulo, 1995, 37, 331-6.
- 12 BURK RL., ACKERMAN N. The thorax. In: BURK RL., ACKERMAN N. Eds. Small animal radiology and ultrasonography: a diagnostic atlas and text 2.ed. Philadelphia: Saunders, 1996: 196-7.
- 13 CUPO P., JURCA M., AZEVEDO-MARQUES MM., OLIVEIRA JS., HERING SE. Severe scorpion envenomation in Brazil. Clinical, laboratory and anatomopathological aspects. Rev. Inst. Med. Trop. São Paulo, 1994, 36, 67-76.
- 14 DE-MATOS IM., TALVANI A., ROCHA OO., FREIRE-MAIA L., TEIXEIRA MM. Evidence for a role of mast cells in the lung edema induced by Tityus serrulatus venom in rats. Toxicon, 2001, 39, 863-7.
- 15 FISCH C. Electrocadiography. In: BRAUNWALD E. Ed. Heart disease: a textbook of cardiovascular medicine 5.ed. Philadelphia: Saunders, 1997: 108-52.
- 16 FREIRE-MAIA L., CAMPOS JA. Response to the letter to the editor by Gueron and Ovsyscher. On the treatment of the cardiovascular manifestations of scorpion envenomation. Toxicon, 1987, 25, 125-30.
- 17 FREIRE MAIA L., CAMPOS JA., AMARAL CF. Approaches to the treatment of scorpion envenomation. Toxicon, 1994, 32, 1009-14.
- 18 FYFE A., DALY PA., DORIAN P., TOUGH J. Reversible "cardiomyopathy" after accidental adrenaline overdose. Am. J. Cardiol., 1991, 67, 318-9.
- 19 GUERON M., ADOLPH RJ., GRUPP IL., GABEL M., GRUPP G., FOWLER NO. Hemodynamic and myocardial consequences of scorpion venom. Am. J. Cardiol., 1980, 45, 979-86.
- 20 GUERON M., ILIA R., SHAHAK E., SOFER S. Renin and aldosterone levels and hypertension following envenomation in humans by the yellow scorpion Leiurus quinquestriatus Toxicon, 1992, 30, 765-7.
- 21 GUERON M., MARGULIS G., ILIA R., SOFER S. The management of scorpion envenomation. Toxicon, 1993, 31, 1071-83.
- 22 GUERON M., SOFER S. The role of the intensivist in the treatment of the cardiovascular manifestations of scorpion envenomation. Toxicon, 1994, 32, 1027-9.
- 23 GUERON M., STERN J., COHEN W. Severe myocardial damage and heart failure in scorpion sting. Report of five cases. Am. J. Cardiol., 1967, 19, 719-26.
- 24 GUERON M., YARON R.: Cardiovascular manifestations of severe scorpion sting. Clinicopathologic correlations. Chest, 1970, 57, 156-62.
- 25 HERING SE., JURCA M., VICHI FL., AZEVEDO-MARQUES MM., CUPO P. "Reversible cardiomyopathy" in patients with severe scorpion envenomation by Tityus serrulatus: evolution of enzymatic, electrocardiographic and echocardiographic alterations. Ann. Trop. Paediatr., 1993, 13, 173-82.
- 26 HERING SE., MARIN-NETO JA., MARIN NETO JA., VICHI FL., JURCA M., MARQUES MMA., CUPO P. Acidente escorpiônico (Ae) grave em adulto: miocardite grave, reversível, simulando infarto agudo do miocárdio. Arq. Bras. Cardiol., 1990, 55, 72.
- 27 ISMAIL M. The scorpion envenomation syndrome. Toxicon, 1995, 33, 825-58.
- 28 KARNAD DR., DEO AM., APTE N., LOHE AS., THATTE S., TILVE GH. Captopril for correcting diuretic induced hypotension in pulmonary oedema after scorpion sting. Br. Med. J., 1989, 298, 1430-1.
- 29 KUMAR EB., SOOMRO RS., AL HAMDANI A., EL SHIMY N. Scorpion venom cardiomyopathy. Am. Heart J., 1992, 123, 725-9.
- 30 MACHADO JC., SILVEIRA FILHO JF. Indução de pancreatite hemorrágica aguda no cão por veneno escorpiônico de T. serrutatus Mem. Inst. Butantan, 1976/1977, 40/41, 1-9.
- 31 MAGALHÃES MM., PEREIRA ME., AMARAL CF., REZENDE NA., CAMPOLINA D., BUCARETCHI F., GAZZINELLI RT., CUNHA-MELO JR. Serum levels of cytokines in patients envenomed by Tityus serrulatus scorpion sting. Toxicon, 1999, 37, 1155-64.
- 32 MARGULIS G., SOFER S., ZALSTEIN E., ZUCKER N., ILIA R., GUERON M. Abnormal coronary perfusion in experimental scorpion envenomation. Toxicon, 1994, 32, 1675-8.
- 33 MASSONE F. Anestesia local. In: MASSONE F. Ed. Anestesiologia veterinária: farmacologia e técnicas 3.ed. Rio de Janeiro: Guanabara Koogan, 1999: 34-49.
- 34 MATOS IM., SOUZA DG., SEABRA DG., FREIRE-MAIA L., TEIXEIRA MM. Effects of tachykinin NK1 or PAF receptor blockade on the lung injury induced by scorpion venom in rats. Eur. J. Pharmacol., 1999, 376, 293-300.
- 35 McCURNIN DM., POFFENBARGER EM. Small animal physical diagnosis and clinical procedures Philadelphia: Saunders, 1991. 222p.
- 36 MICHAEL EM. Toxicologia. In: ETTINGER SJ. Ed. Tratado de medicina interna veterinária 3.ed. São Paulo: Manole, 1992: 496-7.
- 37 NOVAES G., DE QUEIROZ AC., DAS NEVES MM., CARDOZO C., RIBEIRO-FILHO L., DE CARVALHO MH., PONTE G., CHAVES A. Induction of acute and chronic pancreatitis with the use of the toxin of the scorpion Tityus serrulatus: experimental model in rats. Arq. Gastroenterol., 1998, 35, 216-22.
- 38 O'BRIEN PJ. Deficiencies of myocardial troponin-T and creatine kinase MB isoenzyme in dogs with idiopathic dilated cardiomyopathy. Am. J. Vet. Res., 1997, 58, 11-6.
- 39 RADHA KRISHNA MURTHY K. The scorpion envenomation syndrome: a different perspective. The physiological basis of the role of insulin in scorpion envenomation. J. Venom. Anim. Toxins, 2000, 6, 4-51.
- 40 REDDY CR., SUVARNAKUMARI G., DEVI CS., REDDY CN. Pathology of scorpion venom poisoning. J. Trop. Med. Hyg., 1972, 75, 98-100.
- 41 RENNER IG., PANTOJA JL., ABRAMSOM SB., DOUGLAS AP., RUSSEL FE., KOCH MK. Effects of scorpion and rattlesnake venoms on the canine pancreas following pancreaticoduodenal arterial injections. Toxicon, 1983, 21, 405-20.
- 42 REZENDE NA., CHAVÉZ-OLORTEGUI C., AMARAL CF. Is the severity of Tityus serrulatus scorpion envenomation related to plasma venom concentrations? Toxicon, 1996, 34, 820-3.
- 43 SANTHANAKRISHNAN BR., GAJALAKSHMI BS. Pathogenesis of cardiovascular complications in children following scorpion envenomation. Ann. Trop. Paediatr., 1986, 6, 117-21.
- 44 SECRETARIA DE SAÚDE DO ESTADO DE SÃO PAULO. Acidentes por escorpiões: clínica e tratamento. In: Manual de Vigilância Epidemiológica: acidentes por animais peçonhentos: identificação, diagnóstico e tratamento São Paulo, 1993: 39-42.
- 45 SOFER S., GUERON M. Vasodilators and hypertensive encephalopathy following scorpion envenomation in children. Chest, 1990, 97, 118-20.
- 46 SOFER S., SHAHAK E., SLONIM A., GUERON M. Myocardial injury without heart failure following envenomation by the scorpion Leiurus quinquestriatus in children. Toxicon, 1991, 29, 382-5.
- 47 STREINER DL., NORMAN GR. Biostatistics: the bare essentials St. Louis: Mosby, 1994. 260p.
- 48 TEIXEIRA Jr. AL., FONTOURA BF., FREIRE-MAIA L., MACHADO CR., CAMARGOS ER., TEIXEIRA MM. Evidence for a direct action of Tityus serrulatus scorpion venom on the cardiac muscle. Toxicon, 2001, 39, 703-9.
- 49 THOMAS WP., GABER CE., JACOB GJ., KAPLAN PM., LOMBARD CW., MOISE NS., MOSES BL. Recommendations for standards in transthoracic two-dimensional echocardiography in the dog and cat. Echocardiography Committee of the Specialty of Cardiology, American College of Veterinary Internal Medicine. J. Vet. Intern. Med., 1993, 7, 247-52.
- 50 TILLEY LP. Essentials of canine and feline electrocardiography: interpretation and treatment 3.ed. Philadelphia: Lea & Fegiber, 1992. 470p.
- 51 TOPPA NH., MARTINS P., COLARES CN., FREIRE-MAIA L., CUNHA-MELO JR. Effect of Tityus serrulatus scorpion toxin on serum gastrin levels in anaesthetized rat. Toxicon, 1998, 36, 1833-42.
- 52 TRONCON LEA., SANTOS AA., GARBACIO VL., SECAF M., VERCEZE AV., CUNHA-MELO JR. Inhibition of gastric emptying and intestinal transit in anaesthetized rats by a Tityus serrulatus scorpion toxin. Braz. J. Med. Biol. Res., 2000, 33, 1053-8.
- 53 WOLF R., CAMACHO AA., SOUZA RCA. Eletrocardiografia computadorizada em cães. Arq. Bras. Med. Vet. Zootec., 2000, 52, 610-5.
- 54 YAROM R., BRAUN K. Cardiovascular effects of scorpion venom, morphological changes in the myocardium. Toxicon, 1970, 8, 41-6.
- 55 YAROM R., BRAUN K. Ca2+ changes in the myocardium following scorpion venom injections J. Mol. Cell. Cardiol., 1971, 2, 177-9.
- 56 YUGANDHAR B., RADHA KRISHNA MURTHY K., SATTAR SA. Insulin administration in severe scorpion envenomation. J. Venom. Anim. Toxins, 1999, 5, 200-19.
Publication Dates
-
Publication in this collection
22 Mar 2006 -
Date of issue
2006
History
-
Accepted
20 July 2005 -
Received
20 Jan 2005