Abstract
The neurotoxin purified from the venom of Bungarus caeruleus causes a neuromuscular blockade on acetylcholine-induced muscle twitch response in isolated frog rectus abdominis muscle preparation. Neuromuscular blockade produced by d-tubocurarine on acetylcholine-induced muscle twitch response in an isolated frog rectus abdominis muscle preparation was reversed to normal muscle twitch response in presence of neostigmine. Whereas the purified neurotoxin produced an irreversible neuromuscular blockade in presence of the same strength of neostigmine. As it is already known, botulinum toxin, which also brings about neuromuscular blockade, is effectively used as a drug in the treatment of painful movement disorders. Since the purified toxin also causes paralysis of the muscle, we propose its possible efficacy in the treatment of neuromuscular disorders.
neuromuscular block; Bungarus caeruleus
ORIGINAL PAPER
Preliminary studies with a neurotoxin obtained from Bungarus caeruleus venom
Mirajkar K. K.I; More S.I; Gadag J. R.
IDepartment of Biochemistry, Karnatak University Dharwad, Karnataka, India
Correspondence to Correspondence to: Kiran K. Mirajkar Plot no. 1633, Anjaneya Nagar, Opposite Ganesha Temple, Belgaum 590016, Karnataka, India Email: drkiran_km@yahoo.com.in
ABSTRACT
The neurotoxin purified from the venom of Bungarus caeruleus causes a neuromuscular blockade on acetylcholine-induced muscle twitch response in isolated frog rectus abdominis muscle preparation. Neuromuscular blockade produced by d-tubocurarine on acetylcholine-induced muscle twitch response in an isolated frog rectus abdominis muscle preparation was reversed to normal muscle twitch response in presence of neostigmine. Whereas the purified neurotoxin produced an irreversible neuromuscular blockade in presence of the same strength of neostigmine. As it is already known, botulinum toxin, which also brings about neuromuscular blockade, is effectively used as a drug in the treatment of painful movement disorders. Since the purified toxin also causes paralysis of the muscle, we propose its possible efficacy in the treatment of neuromuscular disorders.
KEY WORDS: neuromuscular block, Bungarus caeruleus.
INTRODUCTION
Toxins isolated from the venoms of Elapidae and Crotalidae snakes exert potent neurotoxic action by inhibiting the release of acetylcholine from nerve terminals in neuromuscular junctions. Such toxins so far isolated from snake venoms are bungarotoxin (18), from Bungarus multicinctus; notexin (15), from Notechis scutatus scutatus; and taipoxin (11), from Oxyuranus scutellatus.
Almost all vital biological processes are open to disruption from snake toxins. Many species of snake contain toxins that act either to destroy muscle tissue or to interfere with the function of the neuromuscular junction. They are classified according to their pharmacological activity (12, 13, 27, 28,) as presynaptic and postsynaptic toxins. Postsynaptically active toxins bind to the acetylcholine receptors at the postsynaptic membrane of the neuromuscular junction and prevent the binding of acetylcholine. The effect is to produce a non-depolarizing type of neuromuscular blockade. The classical example is a-bungarotoxin from the venom of Bungarus multicinctus. Presynaptically active toxins act on motor end terminals to either facilitate or block neurotransmitter release, e.g. b-bungarotoxin, also from the venom of Bungarus multicinctus (6, 7).
Earlier work on Bungarus caeruleus (4) included the biochemical and pharmacological characterization of neurotoxic phospholipase A2. The toxin b-bungarotoxin from krait (24) has been isolated, purified, crystallized, and preliminary x-ray analyzed.
In this study we intend to demonstrate the neuromuscular blocking action of a toxin purified from Bungarus caeruleus venom and also propose the use of this toxin for treatment of neuromuscular disorders. Bungarus caeruleus (Indian krait) neurotoxin is suggested as a possible alternative for the treatment of painful movement disorders (focal dystonias) because botulinum toxin (14), which is currently used, is regarded as the most toxic substance and is also known to have many side effects. Botulinum toxin is a large peptide eliciting the production of high antibody titers in patients, making subsequent injections of the same botulinum toxin subtype ineffective (8) and thus compelling the patient to receive different subtypes of this toxin. Botulinum toxin can also enter the spinal cord region and has the ability to bind to the brain synaptosomes. Botulinum toxin paralysis results from a permanent blockade (9) of neurotransmission at the motor end plate. This blockade is caused by the inhibition of acetylcholine release from nerve endings. As a consequence of neuroparalysis, secondary changes occur (e.g. limited atrophy of affected muscle fibers and sprouting of the terminal nerve fibers). The toxin also interferes with the uptake of cytoplasmic acetylcholine by the presynaptic vesicles in the motor end plate (10). Localized injection of minute doses of Botox (botulinum toxin) has been shown to alleviate muscle spasms in dystonia and other movement disorders.
MATERIALS AND METHODS
Materials used:
- Lyophilized Bungarus caeruleus snake venom, a generous gift from Haffkine Institute, Mumbai, India;
- Albino mice (Mus musculus), 20-40g body weight, 10 months old;
- Frog's isolated rectus abdominis muscle preparation, Dale's organ bath, kymograph with recording drum, aerator and writing lever, and Ringer's solution (0.6%NaCl + 0.14%KCl + 0.012%CaCl2 + 0.02%NaHCO3);
- Acetylcholine, d-tubocurarine and neostigmine, Bungarus caeruleus crude venom, and purified toxin.
Isolation and purification of toxin
Bungarus caeruleus crude venom was fractionated on a CM-Sephadex C-25 column (17, 21). Venom (100mg) dissolved in 5ml of 0.05M ammonium acetate buffer, pH 5.8, was loaded on a previously equilibrated CM Sephadex C-25 column (1.5x30cm). After washing the column with 500ml of ammonium acetate buffer, pH 5.8, venom components were eluted using ammonium acetate buffer linear gradient (0.05M, pH 5.8, to 0.95M, pH 6.8) at a flow rate of 40ml/h and 4ml-fractions were collected. Protein elution profile was monitored at 280nm on a spectrophotometer. Fractions showing enzymatic activity were pooled, desalted, and lyophilized.
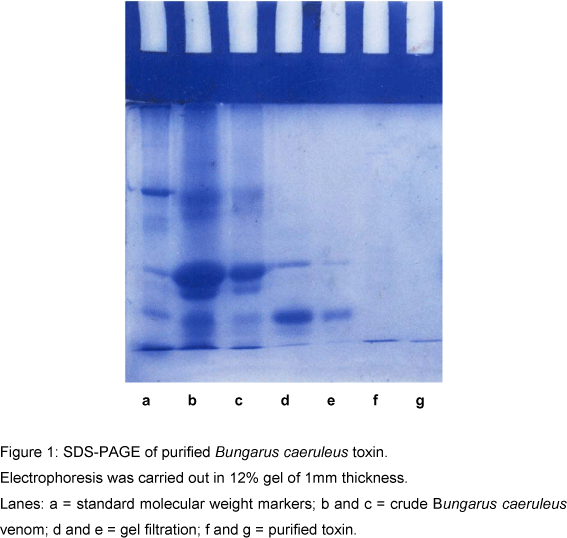
The major fraction was subsequently subjected to gel filtration on a Sephadex G-50 column (1.5x90cm) and eluted with 0.1M ammonium acetate (pH 7.0). The protein was eluted at a flow rate of 20ml/h. Fractions (3ml) were collected and elution was monitored at 280nm on a UV-spectrophotometer. The major protein peak eluted from the column was next subjected to SDS-PAGE, as shown in Figure 1, and further characterized.
Protein concentrations (19) were determined by measuring their absorbance at 280nm using bovine serum albumin as standard.
Determination of LD50 of crude venom and purified toxin
The LD50 determination (22) was carried out for both Bungarus caeruleus crude venom and its purified toxin.
Mice were maintained on standard food pellets overnight, however fasting animals were used in the present study. Six groups, each group comprising of six overnight fasting animals were treated as follows:
Group 1: Saline administered animals (control group).
Groups 2-6: Crude venom / purified toxin administered animals.
Saline (0.4ml) was injected into the control group, and crude venom / purified toxin was injected at varying doses, intraperitoneally, into the other groups. Animals were observed for 48h.
Physio-pharmacological studies
To demonstrate the neuromuscular blocking action (24), frog rectus abdominis muscle preparation was isolated and mounted in Dale's organ bath containing aerated frog Ringer's solution. Tissue was stabilized for 20 minutes; 0.1ml acetylcholine (80µg/ml) was added to the bath fluid, allowed to act for three minutes and the twitch response was recorded. After two minutes relaxation, 1ml d-tubocurarine (10µg/ml) was added to the bath and allowed to act for three minutes. Muscle twitch response in the presence of acetylcholine was recorded with needle electrodes (sensitivity of mv/cm) using a kymograph. Muscle preparation was washed with frog Ringer's solution and allowed to relax for two minutes.
Again, 1ml of acetylcholine (80µg/ml), 1ml of d-tubocurarine (10µg/ml), and 1ml of neostigmine (100µg/ml) were added to the bath at an interval of three minutes and the muscle twitch response was recorded. The muscle preparation was washed again with frog Ringer's solution.
Next, acetylcholine-induced muscle twitch response was recorded for increasing doses of purified Bungarus caeruleus toxin until complete neuromuscular blockade was obtained. Acetylcholine-induced muscle twitch responses were recorded at an interval of three minutes in presence of neostigmine (100µg/ml).
RESULTS
Purification of the neurotoxin
Figure 1 shows SDS-PAGE of purified Bungarus caeruleus toxin. Electrophoresis was carried out in 12% gel of 1mm thickness.
Determination of LD50 of crude venom and purified toxin
Bungarus caeruleus neurotoxin when administered intravenously demonstrated lethality in mice with typical neurotoxic symptoms and had a LD50 value of 160 µg/kg mice, as shown in Figure 2. Dosage of the purified neurotoxin was calculated on the basis of total protein content. Bungarus caeruleus crude venom had a LD50 value of 40 µg/kg mice, which died with typical neurotoxic symptoms like shivering and tremors, as shown in Figure 2.
Physio-pharmacological studies
Acetylcholine-induced muscle twitch tension of frog rectus abdominis muscle was treated as control.
Figure 3 shows the neuromuscular blockade by d-tubocurarine on the acetylcholine-induced muscle twitch response in an isolated frog rectus abdominis muscle preparation. A concentration of 10µg of d-tubocurarine produced 44% inhibition of muscle twitch response as compared to the normal response produced by acetylcholine within 3 minutes. Neuromuscular blockade produced by d-tubocurarine was reversed to normal muscle twitch response in presence of neostigmine (100µg/ml).
d-Tubocurarine-induced blockade was compared with the neuromuscular blockade by Bungarus caeruleus neurotoxin. Purified neurotoxin produced an irreversible neuromuscular blockade in presence of the same strength of neostigmine.
Figure 4 shows the neuromuscular blockade brought about by purified Bungarus caeruleus neurotoxin on acetylcholine-induced muscle twitch response in an isolated frog rectus abdominis muscle preparation at different concentrations (25µg; 50µg; 75µg; 100µg; 125µg; 150µg; 175µg; 200µg; 225µg; 225µg + 100µg neostigmine)
A concentration of 225µg purified neurotoxin produced 100% inhibition of muscle twitch response within 3 minutes, as shown in Figure 5.
DISCUSSION
Phospholipases found in some snake venoms are potent neurotoxins that target their enzymatic action on the synaptic membrane. One of these is the heterodimeric neurotoxin b-bungarotoxin, which acts by inhibiting the release of acetylcholine from motor nerve endings, one of the most investigated steps in neuromuscular transmission (1,16,18,). Six isoforms of b-bungarotoxin (b1-b6) from the venom of Bungarus multicinctus (17) and five isoforms of b-bungarotoxin (b1-b5 caeruleotoxin) from the venom of B. caeruleus (2) have been identified and characterized. Structurally, b-bungarotoxin is a heterodimeric neurotoxin (25) consisting of a phospholipase A2 (PLA2) subunit linked by a disulfide bond to a K+ channel binding subunit, which is a member of the Kunitz protease-inhibitor superfamily. It acts presynaptically by binding via the protein-inhibitor-like subunit to a presynaptic potassium channel and then blocking neurotransmission with the second subunit, which has phospholipase A2 activity, thus altering acetylcholine release in both the peripheral and central nervous systems. Although the presynaptic action of b-bungarotoxin has been established, there is also evidence for its postsynaptic action (26) in mammalian skeletal muscle.
Following exposure of nerve muscle preparation to toxin, the frequency of miniature end plate potentials (MEPP) generally undergoes a triphasic change. The three phases are: 1) an initial reduction of MEPP frequency; 2) a phase of enhanced MEPP frequency; and 3) a decline in MEPP frequency leading to complete failure. b-bungarotoxin does not change MEPP amplitude, indicating that the sensitivity of end plates toward acetylcholine remains unchanged. Usually the muscle membrane potential and the directly elicited muscle action potential remain unchanged (23). On frog sartorius preparation (1, 3, 20), it has been reported that b-bungarotoxin causes a marked decrease in MEPP frequency in a matter of minutes after being added to the bath, and that it may last about 10 min. It has been suggested (5) that phase one is caused by the binding of b-bungarotoxin to the presynaptic membrane, but the reason why this binding results in changes in the MEPP frequency has not been explained.
Aimed at discovering and developing drugs, we initiated our efforts to isolate, purify and characterize Bungarus caeruleus toxin to treat painful neurological movement disorders, also known as dystonias, in lieu of botulinum toxin, which is highly antigenic, expensive, and nerve damaging in nature.
Though this suggestion is largely hypothetical in nature, it is sound when biochemical characteristics of both toxins are compared. Causing selective muscle paralysis is the main concern to alleviate human suffering. Development of this concept is mainly a result of intuitive empirical work by the clinicians with collaborative scientific studies generated secondarily.
While some toxins have lead directly to their use as therapeutic agents, others have provided insight into selective pharmacological actions. Although many toxins are too large or too poorly absorbed to be used as drugs, advances in techniques for analyzing three-dimensional structure and computer-aided drug design should make it likely that toxins will act as model compounds in drug discovery.
ACKNOWLEDGEMENTS
We wish to thank all our colleagues from Department of Biochemistry, Dharwad, and elsewhere, without whom it would not have been possible to carry out this work. We are especially grateful to Shri Shastri, principal of Sonia College of Pharmacy, for providing us all the facility required for this work.
Received: March 28, 2005
Accepted: May 6, 2005
Published online: February 24, 2006
- 1 ABE T., LIMBRICK AR., MILEDI R. Acute muscle denervation induced by beta-bungarotoxin. Proc. R. Soc. Lond. B Biol. Sci., 1976, 194, 545-53.
- 2 ABE T., ALEMA S., MILEDI R. Phospholipase activity in beta-bungarotoxin action. J. Physiol., 1977, 270, 55-6.
- 3 ALDERDICE MT., VOLLE RL. Multiple actions of -bungarotoxin on acetylcholine release at amphibian motor nerve terminals. Naunyn Schmiedebergs Arch. Pharmacol., 1981, 316, 126-30.
- 4 BON C., CHANGEUX JP. Chemical and pharmacological characterization of toxic polypeptides from the venom of Bungarus caeruleus Eur. J. Biochem., 1977, 74, 31-42.
- 5 CARATSCH CG., MARANDA B., MILEDI R., STRONG PN. A further study of the phospholipase-independent action of beta-bungarotoxin at frog end-plates. J. Physiol., 1981, 319, 179-91.
- 6 CHANG CC. The action of snake venoms on nerve and muscle. In: LEE CY. Ed. Snake Venoms Berlin: Springer-Verlag, 1979: 309-76.
- 7 CHANG CC. Neurotoxins with phospholipase A2 activity in snake venoms. Proc. Natl. Sci. Counc. Repub. China B, 1985, 9, 126-42.
- 8 ELSTON JS. Long-term results of treatment of idiopathic blepharospasm with botulinum toxin injections. Br. J. Ophthalmol., 1987, 71, 664-8.
- 9 ELSTON JS. The clinical use of botulinum toxin. Semin. Ophthalmol., 1988, 3/4, 249-60.
- 10 ELSTON JS. Botulinum toxin A in clinical medicine. J. Physiol., 1990, 84, 285-9.
- 11 HABERMANN E., RÜBSAMEN K. Toxins of animal and plant origin In: DE VRIES A., KOCHVA E. Eds. London: Gordon & Breach Science, 1971: 333-41.
- 12 HARRIS JB. Polypeptides from snake venoms which act on nerve and muscle. Prog. Med. Chem., 1984, 21, 63-110.
- 13 HARRIS JB. Phospholipases in snake venoms and their effects on nerve and muscle. In: HARVEY AL. Ed. Snake Toxins New York: Pergamon Press, 1991: 91-124.
- 14 HARVEY AL. From venoms to toxins to drugs. Chem Ind., 1995, 22, 914-6.
- 15 KARLSSON E. Chemistry of protein toxins in snake venoms. In: LEE CY. Ed. Snake Venoms Berlin: Springer-Verlag, 1979: 159.
- 16 KELLY RB., BROWN FR. Biochemical and physiological properties of a purified snake venom neurotoxin which acts presynaptically. J. Neurobiol., 1974, 5, 135-50.
- 17 KONDO K., TODA H., NARITA K, LEE CY. Amino acid sequences of three beta-bungarotoxin from Bungarus multicinctus venom amino acid substitutions in the A chains. J. Biochem., 1982, 91, 1519-30.
- 18 LEE CY, CHANG CC. Modes of actions of purified toxins from elapid venoms on neuromuscular transmission. Mem. Inst. Butantan, 1966, 33, 555-72.
- 19 LOWRY OH., ROSENBROUGH NJ., FARR AL., RANDALL RJ. Protein measurement with the Folin-Phenol reagents. J. Biol. Chem., 1951, 193, 265-75.
- 20 MASUKAWA LM., LIVENGOOD DR. Alterations in spontaneous transmitter release by divalent cations after treatment of the neuromuscular junction with beta-bungarotoxin. Cell. Mol. Neurobiol., 1982, 2, 277-90.
- 21 RADVANYI F., BON C. Catalytic activity and reactivity with p-bromophenacyl bromide of the phospholipase subunit of crotoxin. Influence of dimerization and association with the non-catalytic subunit. J. Biol. Chem., 1982, 257, 12616-23.
- 22 REED LJ., MUENCH HA. A simple method for the estimated fifty percent end points. Amer. J. Hyg., 1938, 27, 493-5.
- 23 ROWAN EG. What does beta-bungarotoxin do at the neuromuscular junction? Toxicon, 2001, 39, 107-18.
- 24 SETH UK. Skeletal muscle relaxants. In: SETH UK. Ed. Selected topics in experimental pharmacology Bombay: Kothary Book Depot, 1971: 78-99.
- 25 SHARMA S., KARTHIKEYAN S., BETZEL C., SINGH TP. Isolation, purification, crystallization and preliminary X-ray analysis of beta 1-bungarotoxin from Bungarus caeruleus (Indian common krait). Acta Crystallogr. D. Biol. Crystallogr., 1999, 55, 1093-4.
- 26 STORELLA RJ., SCHOUCHOFF AL., FUJII M., HILL J., FLETCHER JE., JIANG MS., SMITH LA. Preliminary evidence for a postsynaptic action of beta-bungarotoxin in mammalian skeletal muscle. Toxicon, 1992, 30, 349-54.
- 27 TZENG MC. Interaction of presynaptically toxin phospholipases A2 with membrane receptors and other binding sites. J. Toxicol. Toxin Rev., 1993, 12, 1-62.
- 28 YANG CC., Structure-function relationship of phospholipase A2 from snake venoms. J. Toxicol. Toxin Rev., 1994, 13, 125-77.
Publication Dates
-
Publication in this collection
22 Mar 2006 -
Date of issue
2006
History
-
Accepted
06 May 2005 -
Received
28 Mar 2005