Abstract
Oxidative stress and proteases have been implicated in several diseases and extensive evidence indicates that antioxidants and protease inhibitors help prevent organ functional damage. Leiurus quinquestriatus (LQQ) scorpion venom causes cellular injuries that may lead to multiple organ failure. Thus, the capability of the antioxidant "natural standardized extract of Gingko biloba leaves (Gin, EGb 761)" and the non-selective protease inhibitor, aprotinin, in ameliorating venom-induced biochemical alterations indicative of cellular injury and oxidative stress was studied to determine their effectiveness in protecting rats from venom-evoked cellular damages. Thus, in this study, rats were treated with LQQ venom (0.3mg.kg-1, subcutaneously) alone or after Gin (150mg.kg-1, orally, daily for 2 weeks before venom) and/or aprotinin (Apr, 46000 KIU.kg-1, intraperitoneally, 30 min before venom). Control groups were injected with saline or treatment modalities. Lungs and hearts were excised after decapitating rats (n=8/group) 60 min after venom injection and the following activities were measured: reduced glutathione (GSH), malondialdehyde (MDA) - an index of lipid peroxidation, glutathione peroxidase (GPx), glucose-6-phosphate dehydrogenase (G6PD), and lactate dehydrogenase (LDH). Our findings demonstrate that LQQ venomsignificantly elevated GSH (p<0.05 vs. control), MDA (p<0.05), G6PD (p<0.05), and LDH activities (p<0.001) in hearts of envenomed rats. The venom also elevated MDA (p<0.05 vs. control) and reduced GSH and GPx (p<0.05) in the lungs of envenomed rats. In general, pretreatment with EGb761 attenuated LQQ venom-evoked increases in GSH (p<0.05 vs. venom), MDA in rat hearts and lungs (p<0.05 vs. venom), plus LDH in the heart (p<0.01). Aprotinin alone significantly reduced the venom-elicited increase in G6PD and LDH activities and the decrease in GPx levels (p<0.05). In general, these protective effects of EGb761 on GSH, MDA (p<0.01 vs. venom) and LDH (p<0.001) in the heart and/or lung were potentiated when combined with aprotinin. We concluded that the effectiveness of EGb761 and Apr in ameliorating venom-evoked biochemical changes indicative of necrosis and free radical generation point out the involvement of oxidative stress and proteases in venom-evoked cellular damages seen in this study in isolated rat hearts and lungs.
Leiurusquinquestriatus; ginkgo; aprotinin; scorpion venoms
ORIGINAL PAPER
Protective effects of the antioxidant Ginkgo biloba extract and the protease inhibitor aprotinin against Leiurus quinquestriatus venom-induced tissue damage in rats
Fatani A. J.I; Al-Zuhair H. A.I; Yaquob H. I.I; Abdel-Fattah A. A.II; El-Sayed M. I.I; El-Sayed F. A.II
IDepartment of Pharmacology, King Saud University, Riyadh, Saudi Arabia
IIDepartment of Biochemistry, College of Pharmacy, King Saud University, Riyadh, Saudi Arabia
Correspondence to Correspondence to: Fairouz El-Sayed Department of Biochemistry College of Pharmacy, King Saud University P.O. Box 616, Riyadh, 11421, Saudi Arabia. Mobile Phone: 00 9 66 50897 5933. Phone/Fax: 00 9 66 1403 1214. Email: rosa_bio_2004@hotmail.com
ABSTRACT
Oxidative stress and proteases have been implicated in several diseases and extensive evidence indicates that antioxidants and protease inhibitors help prevent organ functional damage. Leiurus quinquestriatus (LQQ) scorpion venom causes cellular injuries that may lead to multiple organ failure. Thus, the capability of the antioxidant "natural standardized extract of Gingko biloba leaves (Gin, EGb 761)" and the non-selective protease inhibitor, aprotinin, in ameliorating venom-induced biochemical alterations indicative of cellular injury and oxidative stress was studied to determine their effectiveness in protecting rats from venom-evoked cellular damages. Thus, in this study, rats were treated with LQQ venom (0.3mg.kg-1, subcutaneously) alone or after Gin (150mg.kg-1, orally, daily for 2 weeks before venom) and/or aprotinin (Apr, 46000 KIU.kg-1, intraperitoneally, 30 min before venom). Control groups were injected with saline or treatment modalities. Lungs and hearts were excised after decapitating rats (n=8/group) 60 min after venom injection and the following activities were measured: reduced glutathione (GSH), malondialdehyde (MDA) an index of lipid peroxidation, glutathione peroxidase (GPx), glucose-6-phosphate dehydrogenase (G6PD), and lactate dehydrogenase (LDH). Our findings demonstrate that LQQ venomsignificantly elevated GSH (p<0.05 vs. control), MDA (p<0.05), G6PD (p<0.05), and LDH activities (p<0.001) in hearts of envenomed rats. The venom also elevated MDA (p<0.05 vs. control) and reduced GSH and GPx (p<0.05) in the lungs of envenomed rats. In general, pretreatment with EGb761 attenuated LQQ venom-evoked increases in GSH (p<0.05 vs. venom), MDA in rat hearts and lungs (p<0.05 vs. venom), plus LDH in the heart (p<0.01). Aprotinin alone significantly reduced the venom-elicited increase in G6PD and LDH activities and the decrease in GPx levels (p<0.05). In general, these protective effects of EGb761 on GSH, MDA (p<0.01 vs. venom) and LDH (p<0.001) in the heart and/or lung were potentiated when combined with aprotinin. We concluded that the effectiveness of EGb761 and Apr in ameliorating venom-evoked biochemical changes indicative of necrosis and free radical generation point out the involvement of oxidative stress and proteases in venom-evoked cellular damages seen in this study in isolated rat hearts and lungs.
Key words: Leiurusquinquestriatus, ginkgo, aprotinin, scorpion venoms
INTRODUCTION
Scorpion venom toxins from different species, such as Leiurus quinquestriatus, bind to voltage-sensitive sodium channels causing exaggerated release of several neurotransmitters and modulators. This usually produces similar consequences in humans and experimental animals afflicting the nervous, cardiovascular, respiratory, skeletal, and gastro-intestinal systems. Moreover, these toxins appear to cause multiple organ dysfunction (MOD) in several organs in the body, especially in children, with death usually credited to cardiovascular and respiratory failure (7, 15, 24, 28, 40, 41). In the latter-mentioned studies, the authors demonstrated an increase in markers indicative of apoptosis and lipid peroxides following scorpion envenomation, suggesting their possible involvement in venom-elicited cellular damages. It has been stated that specific pathophysiological conditions may trigger the onset of MOD, such as generation of oxygen free radicals as well as hypoxia, increased levels of cytokines and nitric oxide, the latter three being observed following scorpion envenomation (17, 38, 40, 41). Further studies are needed, however, to assess the involvement of oxygen free radicals and oxidative stress in scorpion envenomation.
During the past decade, the reactive oxygen species (ROS) generation and oxidative stress have been implicated in the development of many diverse diseases including hypertension, cardiac dysrhythmia, and myocardial damage (53), all of which are present in scorpion envenomation (7, 15, 24, 28). These ROS are highly reactive species that have the potential to oxidize biological molecules including proteins, lipids, and DNA (5). Several researchers have shown that natural or synthetic antioxidants could decrease the risk of cardiovascular diseases, and could also act as membrane stabilizers preventing functional damage of the membranes (6, 9, 37). Thus, it would be beneficial to discover if scorpion venoms enhance oxidative stress and ROS production and to assess whether antioxidants can ameliorate the cellular toxic damages caused by scorpion venoms, stabilize membranes, and possibly prevent leakage of cellular substances that ultimately could trigger the cascade of events usually seen in the scorpion envenoming syndrome (7, 15, 24, 28, 40).
One such agent is the natural antioxidant extracted from the leaves of the Ginkgo biloba L. plant (Salisburia adiantifolia Smith), which contains flavonoids, including ginkgo-flavone glycosides. In addition, it also contains terpene lactones, such as ginkgolides A, B, C, J and M, plus bilobalide, proanthrocyanidine and ginkgolic acid (13, 16, 31). This standardized plant extract (EGb 761) appears to participate in ameliorating certain diseases such as peripheral artery insufficiency (39, 53), pancreatic cancer (27), and diseases associated with free radical generation such as cerebral and myocardial ischemia (25). Scorpion venoms may cause myocardial ischemia plus infarction, left ventricular heart failure, and pulmonary edema (7, 24, 28).
Another facet that has been investigated is the role of the kallikrein-kinin system in several scorpion envenomation. Plasma kinins were found to be released by Heterometrus bengalensis scorpion venom (34) and appear to contribute to the pulmonary edema induced by scorpion toxins in rats (8). Moreover, Fatani et al. (21) showed that pretreatment of rabbits with the kallikrein-kinin inhibitor, aprotinin, attenuated L. quinquestriatus venom-induced pulmonary edema, arrhythmia, and terminal hypotension. Aprotinin is a polyvalent non-selective protease inhibitor that inhibits many of the enzymes involved in inflammatory and hemostatic processes in the scorpion envenoming syndrome (22, 49). On the other hand, icatibant, a B2 bradykinin antagonist, although capable of decreasing the venom-evoked actions, was not as effective as aprotinin, pointing to the possibility that its protease activity may have contributed to its efficacy against scorpion venom deleterious effects.
Although knowledge about the detrimental effects of scorpion envenomation in humans and experimental animals has grown over the years, much more studies are still needed. Full understanding of the complex aspects of scorpion envenomation can set forth an appropriate treatment protocol that will take all mechanisms and complications involved into consideration. Thus, this study was an attempt to uncover the cellular mechanism of the venom-induced pathological changes, and to test the effectiveness of the natural antioxidant, Ginkgo biloba extract, and the protease inhibitor, aprotinin, in blocking LQQ venom-elicited biochemical changes indicative of cellular damage and necrosis in commonly affected organs such as the heart and lung in a rat experimental model.
Materials and Methods
Venom
Leiurus quinquestriatus venom was obtained from mature scorpions, collected by the Antivenom Center, King Fahad National Guard Hospital. The venom was then suspended in water, centrifuged and the supernatant was freeze-dried before storing at -20°C (28). When required, the venom was reconstituted by the addition of 0.9% sodium chloride solution. Aprotinin (100,000 KIU/10ml ampoules) was purchased from Bayer (Germany), and Ginkgo biloba leave extract (EGb 761) was obtained from Schwabe (Germany). All other chemicals utilized were of analytical grade and purchased from Sigma (UK).
Animals
Adult male Wistar rats (150-200g) were used in this study. The animals were obtained from the animal care facilities of King Saud University, housed in clean cages and allowed free access to standard rat chow and water. The College of Pharmacy "International guidelines for care, handling and use of laboratory animals" were strongly adhered to throughout the study.
Experimental protocol
Rats were randomly assigned to 7 groups, each consisting of 8 animals. They were given either LQQ scorpion venom alone (0.30mg.kg-1, subcutaneously, 60 min before decapitation, Group 1), or after aprotinin (46000 K.I.U, intraperitoneally, 30 min prior to venom injection, Group 2), or Ginkgo biloba extract (150mg.kg-1, orally, once daily for 2 weeks up to the day of venom injection, Group 3), or a combination of both (ginkgo, as above and 1h after the last dose on day of experiment, and aprotinin, injected as mentioned above, Group 4). Animals of Groups 5, 6, and 7 were given normal saline, ginkgo and aprotinin, respectively, and served as controls.
The rats were then killed by decapitation 1h after venom injection to ensure absorption and commencement of the venom's deleterious actions. Chests were immediately opened and hearts and lungs were quickly excised, rinsed, blotted dry and weighed. Each organ was afterwards homogenized in ice-cold 1,15% KCl using a glass Potter Elvehjem homogenizer to produce a 20% w/v homogenate. Two portions of each homogenate were used for the estimation of both reduced glutathione (GSH) and malondialdehyde (MDA). The rest of the homogenates were then centrifuged at 105,000g for 1h; the supernatants contained the cytosolic fractions, and were used for estimation of glucose-6-phosphate dehydrogenase (G6PDH), lactate dehydrogenase (LDH), and glutathione peroxidase (GPx) activities.
Biochemical parameters measurements
Glutathione (GSH), an endogenous antioxidant, was detected spectrophotometrically at 412nm using the method of Sedlak and Lindsay (54). Malondialdehyde (MDA) was determined at 523nm utilizing the method of Uchiyama and Mihara (43); as an aldehyde formed by breakdown of lipid hydroxides in the tissue, it was used as a measure of degree of lipid peroxidation. Glutathione peroxidase (GPx)was measured by following the decrease in absorbance when oxidized glutathione is converted to glutathione via glutathione reductase utilizing a standard commercial kit (Randox, UK). Lactate dehydrogenase (LDH) activity was calculated using commercially available UV-kinetic biochemical kit (Stanbio Lab, Inc., USA). Glucose-6-phosphate dehydrogenase (G6PDH) was estimated by measuring the rate of absorbance due to the reduction of NADP+ with the aid of a standard commercial kit (Randox, UK). Cytosolic protein contentwas determined using Folin reagent based on the method of Lowry et al. (36).
Statistical analysis
Numeric variables are expressed as arithmetic mean + standard error of the mean (SEM). One Way Analysis of Variance followed by Tukey Kramer multiple comparison post tests were used to analyze the data. Values of p<0.05 were considered significant.
Results
Effect of Leiurus quinquestriatus venom on the biochemical parameters in hearts and lungs homogenates
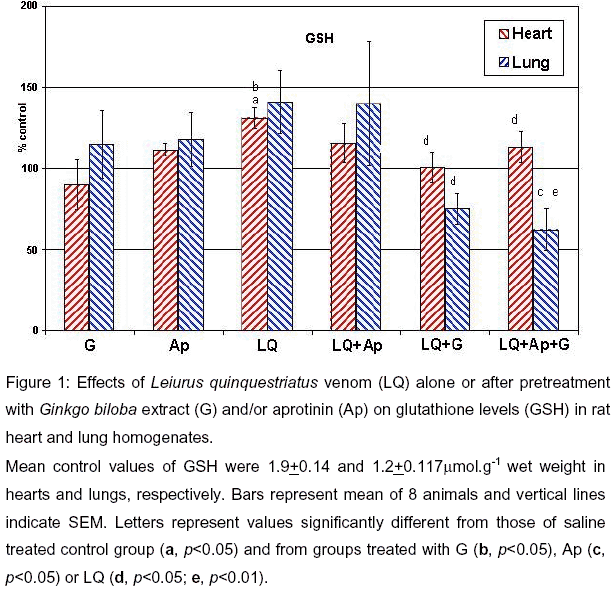
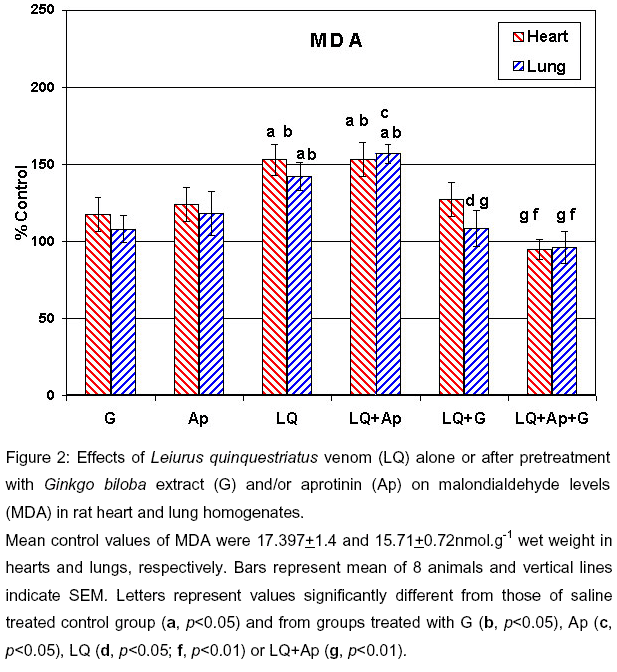
Leiurus quinquestriatus (LQQ) scorpion venom was capable of significantly elevating the GSH (p<0.05), MDA levels (p<0.05) indicative of lipid peroxidation, G6PD (p<0.05), and LDH (p<0.001) in hearts of envenomed rats (Figures 1, 2, 4 and 5 ). On the other hand, MDA levels were significantly elevated (p<0.05) while GPx was significantly reduced (p<0.05, Figures 2 and 3) in the lungs of envenomed rats. Although levels of GSH in the lungs tended to increase, the elevation was not significant when compared to the control group.
Effect of pretreatment with Ginkgo biloba extract on Leiurus quinquestriatus venom-induced changes
In general, pretreatment of scorpion-envenomed rats with the antioxidant Ginkgo biloba extract for 2 weeks prior to venom injection significantly ameliorated the venom-evoked increases in GSH levels (p<0.05) and LDH (p<0.01) activities in the rat heart compared to the envenomed group. The extract similarly attenuated (p<0.05) venom-elicited increases in lung GSH and MDA levels (Figures 1, 2 and 5 ).
Effect of pretreatment with aprotinin on Leiurus quinquestriatus venom-induced changes
Aprotinin, the non-selective protease inhibitor, significantly attenuated (p<0.05) the venom-elicited increases in LDH and G6PD activities in rat hearts (Figures 4 and 5 ). In addition, aprotinin significantly improved the venom-evoked reduction in GPx activity in the isolated lungs (p<0.05) compared to the envenomed rats (Figure 3).
When the rats were pretreated with both Ginkgo biloba extract and aprotinin, the combined therapy reduced the venom-elicited increases in MDA levels (p<0.01) and LDH activities (p<0.001) in the heart and both GSH (p<0.01 vs LQQ venom) and MDA contents in the lung (p<0.01) to a greater extent than that seen with Ginkgo biloba extract alone (Figures 1, 2 and 5 ).
Mean control values of GSH were 1.9+0.14 and 1.2+0.117mmol.g-1 wet weight in hearts and lungs, respectively. Bars represent mean of 8 animals and vertical lines indicate SEM. Letters represent values significantly different from those of saline treated control group (a, p<0.05) and from groups treated with G (b, p<0.05), Ap (c, p<0.05) or LQ (d, p<0.05; e, p<0.01).
Mean control values of MDA were 17.397+1.4 and 15.71+0.72nmol.g-1 wet weight in hearts and lungs, respectively. Bars represent mean of 8 animals and vertical lines indicate SEM. Letters represent values significantly different from those of saline treated control group (a, p<0.05) and from groups treated with G (b, p<0.05), Ap (c, p<0.05), LQ (d, p<0.05; f, p<0.01) or LQ+Ap (g, p<0.01).
Mean control values of GPx were 2.83+0.033 and 2.76+0.035mmol.min -1.g-1 wet weight in hearts and lungs, respectively. Bars represent mean of 8animals and vertical lines indicate SEM. Letters represent values significantly different from those of saline treated control group (a, p<0.05) and from groups treated with G (b, p<0.05), Ap (c, p<0.05) or LQ (d, p<0.05).
Mean control values of G6PD were 0.023+0.001 and 0.022+0.0011U.mg-1 protein in hearts and lungs, respectively. Bars represent mean of 8animalsand vertical lines indicate SEM. Letters represent values significantly different from those of saline treated control group (a, p<0.05) and from the group treated with LQ (b, p<0.05).
Mean control values of LDH were 19.6+2.3mmol.min-1.mg-1 protein. Bars represent mean of 8 animals and vertical lines indicate SEM. Letters represent values significantly different from those of saline treated control group (a, p<0.001) and from groups treated with G (b, p<0.05; c, p<0.01), Ap (d, p<0.05; e, p<0.01), LQ (f, p<0.05; g, p<0.01; h, p<0.001), LQ+Ap (i, p<0.05) or LQ+G (j, p<0.05).
Discussion
Scorpion envenomation represents an important and serious public health problem not only in Saudi Arabia, but also in different subtropical and tropical regions of the world. This is due to their potential to induce severe and sometimes terminal damages in several systems of the body, especially the cardiovascular and respiratory systems (7, 15, 24, 28, 29). Although much work has been performed to determine the exact pathophysiology that leads to these venom-evoked effects, more studies are needed to establish the exact mechanisms involved, especially at the cellular level. Therefore, this study was undertaken to assess whether oxidative stress and/or proteases were implicated in the multiple organ damages observed following scorpion envenomation. Since death due to scorpion envenoming is usually due to cardiac and respiratory complications (7, 15, 28), and since the heart and lung are readily susceptible to the deleterious effects of attacks by oxidative stress-generated free radicals (5), biochemical parameters indicative of oxidative stress were measured in these vital organs excised from deceased envenomed rats.
In the present study, LQQ venom generally increased MDA levels, indicative of increased production of lipid peroxides, GSH contents and enhanced LDH activity in the rat lung and/or heart homogenates. This would demonstrate a role for tissue necrosis, lipid peroxidation and free radical generation in venom-induced deleterious cellular effects. Circulatory shock, hypoperfusion, ischemia, myocardial damage, activation of the cytokine network, and apoptosis, which are reported to occur in scorpion envenomation (17, 23, 28, 40, 41), favor generation of injurious oxygen free radicals, oxidative stress and lipid peroxidation (10, 35, 52).
The venom-evoked enhanced lipid peroxidation seen in this study may explain the deleterious organ defects and MOD observed following scorpion envenomation (28, 40, 41). The observed increase in lipid peroxidation was also demonstrated in scorpion-envenomed children in Egypt (40); the authors quoted the involvement of oxidative stress and apoptosis in venom-evoked multiple organ dysfunction (MOD). Enhanced production of lipid peroxides has been likewise observed in a variety of conditions such as exposure to sea anemone toxins or sulphur dioxide in mice (42, 50), in addition to cadmium (51) and chronic lead intoxication in rats (2).
Hypothetically, the mechanisms by which generation of free radicals and lipid peroxidation occurs in scorpion envenomation are complex and may depend on a multitude of interacting factors. These probably include: A) scorpion venoms contain neurotoxins that act on a number of ionic channels resulting in alteration of ionic transport and cytosolic calcium overload (26). B) This elevates AMP concentration and increases its catabolism, and subsequently leads to generation of free radicals. C). Ultimately, these free radicals attack membrane phospholipids causing their peroxidation. D) Occurrence of lipid peroxidation in biological membranes eventually causes impairment of membrane functioning, decreased fluidity, and inactivation of membrane-bound receptors, all of which may culminate in MOD (5, 10, 35), a feature encountered in scorpion envenomation (40). Alternatively, it is known that scorpion venoms, by their action mainly on sodium channels, enhance release of various neurotransmitters such as adrenaline and noradrenaline (8, 28, 40). It has been reported that catecholamines can induce the generation of free radicals which may lead to oxidative stress and apoptosis (30).
The venom-evoked increase in GSH in rat hearts (and to a lesser degree in lungs) observed in this study is an intriguing observation and is in harmony with the work of Yadav et al. (57), who observed elevated GSH concentration in the rat heart following alloxan-induced diabetes. The authors attributed its elevation to its increased activity in the heart and decreased efflux into the blood stream to neutralize the superoxide anions and counteract oxidative stress. It is known that reduced GSH is a tripeptide involved in a wide range of metabolic functions, primarily aimed at providing protection against toxic compounds and free radical species. Usually, reduced GSH is regenerated and GSH levels are maintained (5, 20, 33, 55). Both an increase and a decrease in GSH levels have been described following exposure of rats to a variety of stressors. For example, the levels of GSH were significantly decreased in several organs and body fluids following cold-restraint-elicited stress, alloxan-induced diabetes and aluminum toxicity, with depletion of GSH while combating free radical generation as its cause (1, 20, 55). Alternatively, the venom-evoked increase in GSH levels in the present study could be explained by enhanced apoptosis due to stress following venom injection (1, 18, 46, 53), which would increase availability of amino acid substrates for GSH synthesis.
Furthermore, the present study demonstrated a venom-elicited elevation in G6PDH in the rat heart, indicating an increase in the pentose phosphate pathway. This would most likely enhance NADPH production; the latter being involved in regenerating reduced GSH from its oxidized form (GSSG) (4, 32), and therefore possibly explaining the venom-evoked increased GSH concentration. It is known that GSH protects against oxidizing environments by combining with oxidants in the presence of GPx, yielding the thiol radical GSSG (32). In the present study the levels of GPx were significantly decreased in the lungs of envenomed rats. The GPx enzyme is a member of a family of peroxidases important in catalyzing the reduction of hydroperoxides and lipid peroxides (14), which would explain its depletion in this study while combating, in the presence of reduced GSH, the venom-induced enhanced lipid peroxidation.
In order to study the involvement of free radical generation and oxidative stress in venom-evoked cellular damages, the free radical scavenger natural standardized Ginkgo biloba leave extract (EGb 761) was orally administered to rats for 2 weeks prior to LQQ venom subcutaneous injection. The antioxidant EGb 761 was chosen since it is a readily available, safe natural product that has proven itself effective in combating oxidative stress resulting from different conditions such as ischemic-reperfusion injuries (25, 53), oxidative stress-induced platelet aggregation (3), and acute hypoxic stress (56). In general, pretreatment of scorpion envenomed rats with Ginkgo biloba extract significantly ameliorated the venom-evoked increases in the levels of GSH, MDA and LDH in isolated rat heart and/or lung, indicating the involvement of free radical generation, oxidative stress and organ damage in the venom's deleterious effects in the hearts and lungs. Free radicals and oxidative stress have been implicated in several conditions such as ischemia, myocardial injuries, and thought to cause direct ultrastructural alterations (44). Ginkgo biloba might also have added beneficial effects in scorpion envenomation such as its vasodilating capabilities (45), which would be useful in cases of venom-induced myocardial ischemia and infarction (29).
To determine whether proteases are involved in the cellular damages evoked by the venom, envenomed rats in this study were pretreated with the non-selective protease inhibitor, aprotinin. It significantly attenuated the venom-elicited increases in LDH in rats' hearts, suggesting the involvement of proteases in the venom-evoked cellular damages. It is possible that the venom, by its ability to act on sodium channels and to prolong depolarization (48), will subsequently enhance cellular influx of calcium, which in turn may activate proteases causing excessive protein degradation and cellular damage (12). Fatani etal. (21) showed that pretreatment of rabbits with aprotinin attenuated LQQ venom evoked pulmonary edema, arrhythmia and terminal hypotension. Morever, the protective effects of aprotinin during scorpion envenomation were also reported by Pandey and Deshpande (47) to significantly attenuate Mesobuthus tamulus venom-induced respiratory and cardiac abnormalities in rats. Eren and his collegues (19) demonstrated the ability of aprotinin to protect against ischemic-reperfusion injuries in rabbit lungs.
It must be noted that in this study aprotinin did not significantly alter the venom-evoked elevation of cardiac levels of MDA or GSH, which is not surprising since aprotinin has no hydroxyl radical, superoxide anion or H2O2 scavenger capacity (11). However, the improvement of venom-elicited changes in G6PDH and GPX activities is perplexing. It is likely that its ability to inhibit kallikrein-kinnin system would reduce bradykinin that can stimulate release of endoperoxides from different tissues (11), and possibly indirectly affect part of the process involved in cellular damages due to oxidative stress. Additional work is needed to better understand the protective role of aprotinin in scorpion envenomation, whether as an inhibitor of the kallikrein-kinin system or as a non-specific serine protease inhibitor.
In conclusion, the effectiveness of the antioxidant, Gingko biloba extract, (EGb 761) and the polyvalent protease inhibitor, aprotinin, in ameliorating venom-evoked changes indicate the involvement of oxidative stress and proteases in venom-induced cellular damages seen in the rat heart and lung tissues. It would be of value to assess whether treatment with antioxidants and/or protease inhibitors after scorpion envenomation would also display a protective effect against the venom-induced deleterious manifestation in different systems after their initiation. Additionally, use of antioxidants that could exert their protective actions within a shorter period of time would be more appropriate and should be investigated. Ultimately, protease inhibitors and antioxidants, if proven to be effective in combating scorpion venom-induced cellular damages, may have a future role in the treatment protocol of scorpion envenomation.
ACKNOWLEDGEMENTS
Special thanks to King Abdulaziz City for Science and Technology, Riyadh, Saudi Arabia for their generous grant.
The authors would like to express their gratitude to the research assistants, Mervat Al-Qutub and Farida Kader, and the technicians, Meisoon Al-Ghamdi and Eman Al-Duheim for their assistance.
- 1 ABD EL-FATTAH AA., AL-YOUSEF HM., AL-BEKAIRI AM., AL-SAWAF HA. Vitamin E protects the brain against oxidative injury stimulated by excessive aluminum intake. Biochem. Mol. Biol. Int., 1998, 46, 1175-80.
- 2 ADONAYLO VN., OTEIZA PI. Lead intoxication: antioxidant defenses and oxidative damage in rat brain. Toxicology, 1999, 135, 77-85.
- 3 AKIBA S., KAWAUCHI T., OKA T., HASHIZUME T., SATO T. Inhibitory effect of the leaf extract of Ginkgo biloba L. on oxidative stress-induced platelet aggregation. Biochem. Mol. Biol. Int., 1998, 46, 1243-8.
- 4 AL-ASHMAWY MB. The role of natural antioxidants and synthetic nonsteroidal anti-inflammatory drugs in the chemoprotection of cancer. Saudi Pharm. J., 1999, 7, 1-13.
- 5 AL-OMAR MA., BEEDHAM C., ALSARRA IA. Pathological roles of reactive oxygen species and their defense mechanisms. Saudi Pharm. J., 2004, 12, 1-18.
- 6 AL-ZUHAIR H., ABD EL-FATTAH A., EL-SAYED MI. The effect of meclofenoxate with Gingko biloba extract or zinc on lipid peroxide, some free radical scavengers and the cardiovascular system of aged rats. Pharmacol. Res., 1998, 38, 65-72.
- 7 AMITAI Y. Clinical manifestations and management of scorpion envenomation. Public Health Rev., 1998, 26, 257-63.
- 8 AZEVEDO AD., CUNHA MELO JR., FREIRE-MAIA L. Arterial hypertension and pulmonary edema induced by catecholamines in unanesthetized rats. Pharmacol. Res. Commun., 1979, 11, 157-67.
- 9 BARNES J., ANDERSON LA., PHILLIPSON JD. Herbal medicines: a guide for healthcare professionals 2.ed. London: Pharmaceutical Press, 2002. 530p.
- 10 BHAUMIK G., SRIVASTAVA KK., SELVAMURTHY W., PURKAYASTHA SS. The role of free radicals in cold injuries. Int. J. Biometeorol., 1995, 38, 171-5.
- 11 BHOOLA KD., FIGUEROA CD., WORTHY K. Bioregulation of kinins: kallikreins, kininogens, and kininases. Pharmacol. Rev., 1992, 44, 1-80.
- 12 CHOI DW. Calcium-mediated neurotoxicity: relationship to specific channel types and role in ischemic damage. Trends Neurosci., 1988, 11, 465-9.
- 13 CLOSTRE F. Ginkgo biloba extract (EGb 761). State of knowledge in the dawn of the year 2000. Ann. Pharm. Fr., 1999, 57, 1S8-88.
- 14 COTGREAVE IA., MOLDEUS P., ORRENIUS S. Host biochemical defense mechanisms against prooxidants. Annu. Rev. Pharmacol. Toxicol., 1988, 28, 189-212.
- 15 DE ROODT AR., GARCIA SI., SALOMON OD., SEGRE L., DOLAB JA., FUNES RF., DE TITTO EH. Epidemiological and clinical aspects of scorpionism by Tityus trivittatus in Argentina. Toxicon, 2003, 41, 971-7.
- 16 DIAMOND BJ., SHIFLETT SC., FEIWEL N., MATHEIS RJ., NOSKIN O., RICHARDS JA., SCHOENBERGER NE. Ginkgo biloba extract: mechanisms and clinical indications. Arch. Phys. Med. Rehabil., 2000, 81, 668-78.
- 17 D'SUZE G., MONCADA S., GONZALEZ C., SEVCIK C., AGUILAR V., ALAGON A. Relationship between plasmatic levels of various cytokines, tumour necrosis factor, enzymes, glucose and venom concentration following Tityus scorpion sting. Toxicon, 2003, 41, 367-75.
- 18 EL-ASMAR M. Metabolic effects of scorpion venom. In: TU A. Ed. Handbook of natural toxins New York: Marcel Dekker, 1984: 551-75.
- 19 EREN S., ESME H., BALCI AE., CAKIR O., BUYUKBAYRAM H., EREN MN., ERDINC L., SATICI O. The effect of aprotinin on ischemia-reperfusion injury in an in situ normothermic ischemic lung model. Eur. J. Cardiothorac. Surg., 2003, 23, 60-5.
- 20 ERIN N., OKAR I., OKTAY S., ERCAN F., ARBAK S., YEGEN BC. Cold-restraint- and TRH-induced ulcer models demonstrate different biochemical and morphological manifestations in gastric and hepatic tissues in rats. Role of calcitonin. Dig. Dis. Sci., 1996, 41, 55-64.
- 21 FATANI AJ., FURMAN BL., ZEITLIN IJ. The involvement of plasma kinins in the cardiovascular effects of Leiurus quinquestriatus scorpion venom in anaesthetised rabbits. Toxicon, 1998, 36, 523-36.
- 22 FERREIRA SH., VANE JR. Interaction between bradykinin and prostaglandins. In: PISANO J., AUSTIN KF. Eds. Chemistry and biology of the kallikrein-kinin system in health and disease states New York: Marcel Dekker, 1976: 225-66.
- 23 FUKUHARA YD., REIS ML., DELLALIBERA-JOVILIANO R., CUNHA FQ., DONADI EA. Increased plasma levels of IL-Ibeta, IL-6, IL-8, IL-10 and TNF-alpha in patients moderately or severely envenomed by Tityus serrulatus scorpion sting. Toxicon, 2003, 41, 49-55.
- 24 HAMMOUDI-TRIKI D., FERQUEL E., ROBBE-VINCENT A., BON C., CHOUMET V., LARABA-DJEBARI F. Epidemiological data, clinical admission gradation and biological quantification by ELISA of scorpion envenomation in Algeria: effect of immunotherapy. Trans. R. Soc. Trop. Med. Hyg., 2004, 98, 240-50.
- 25 HARAMAKI N., AGGARWAL S., KAWABATA T., DROY-LEFAIX MT., PACKER L. Effects of natural antioxidant Ginkgo biloba extract (EGB 761) on myocardial ischemia-reperfusion injury. Free Radic. Biol. Med., 1994, 16, 789-94.
- 26 HARVEY AL., ANDERSON AJ., BRAGA MFM., MARSHALL DL., ROWAN EG., VATANPOUR H., CASTAÑEDA O., KARLSSON E. Toxins affecting neuronal ion channels. In: GOPALAKRISHNAKONE P., TAN CK. Eds. Recent advances in toxicology research. Singapore: National University of Singapore, 1992: 59-70.
- 27 HAUNS B., HARING B., KOHLER S., MROSS K., ROBBEN-BATHE P., UNGER C. Phase II study with 5-fluorouracil and Ginkgo biloba extract (GBE 761 ONC) in patients with pancreatic cancer. Arzneimittelforschung, 1999, 49, 1030-4.
- 28 ISMAIL M. The scorpion envenoming syndrome. Toxicon, 1995, 33, 825-58.
- 29 ISMAIL M., OSMAN OH., IBRAHIM SA., EL-ASMAR MF. Cardiovascular and respiratory responses to the venom from the scorpion Leiurus quinquestriatus East Afr. Med. J., 1972, 49, 273-81.
- 30 KHORCHID A., FRAGOSO G., SHORE G., ALMAZAN G. Catecholamine-induced oligodendrocyte cell death in culture is developmentally regulated and involves free radical generation and differential activation of caspase-3. Glia, 2002, 40, 283-99.
- 31 KLEIJNEN J., KNIPSCHILD P. Ginkgo biloba Lancet, 1992, 340, 1136-9.
- 32 KLETZIEN RF., HARRIS PK., FOELLMI LA. Glucose-6-phosphate dehydrogenase: a "housekeeping" enzyme subject to tissue-specific regulation by hormones, nutrients, and oxidant stress. FASEB J., 1994, 8, 174-81.
- 33 LANGLEY SC., KELLY FJ. Differing response of the glutathione system to fasting in neonatal and adult guinea pigs. Biochem. Pharmacol., 1992, 44, 1484-94.
- 34 LAHIRI SC., NAG CHAUDHURI AK. Release of kinin by the scorpion (Heterometrus bengalensis). Indian J. Exp. Biol., 1983, 21, 198-202.
- 35 LOVE S. Oxidative stress in brain ischemia. Brain Pathol., 1999, 9, 119-31.
- 36 LOWRY OH., ROSEBROUGH NJ., FARR AL., RANDALL RJ. Protein measurement with Folin phenol reagent. J. Biol. Chem., 1951, 193, 265-75.
- 37 LUCY JA. Functional and structural aspects of biological membranes: a suggested structural role of vitamin E in the control of membrane permeability and stability. Ann. N. Y. Acad. Sci., 1972, 203, 4-11.
- 38 MAGALHÃES MM., PEREIRA ME, AMARAL CF., REZENDE NA., CAMPOLINA D., BUCARETCHI F., GAZZINELLI RT., CUNHA-MELO JR. Serum levels of cytokine in patients envenomed by Tityus serrulatus scorpion sting. Toxicon, 1999, 37, 1155-64.
- 39 MCKENNA DJ., JONES K., HUGHES K. Efficacy, safety and use of Ginkgo biloba in clinical and preclinical applications. Altern. Ther. Health Med., 2001, 7, 88-90.
- 40 MEKI AR., HASAN HA., EL-DEEN ZM., BAKKAR S. Dysregulation of apoptosis in scorpion envenomed children: its reflection on their outcome. Toxicon, 2003, 42, 229-37.
- 41 MEKI AR., MOHEY-EL-DEAN ZM. Serum interleukin-1beta, interleukin-6, nitric oxide and alpha1-antitrypsin in scorpion envenomed children. Toxicon, 1998, 36, 1851-9.
- 42 MENG Z. Oxidative damage of sulpur dioxide on various organs of mice: sulphur dioxide is a systemic oxidative damage agent. Inhal. Toxicol., 2003, 15, 181-95.
- 43 MIHARA M., UCHIYAMA M. Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal. Biochem., 1978, 86, 271-8.
- 44 MIKI S., ASHRAF M., SALKA S., SPERELAKIS N. Myocardial dysfunction and ultrastructural alterations mediated by oxygen metabolites. J. Mol. Cell. Cardiol., 1988, 20, 1009-24.
- 45 NISHIDA S., SATOH H. Comparative vasodilating action among terpenoids and flavonoids contained in Ginkgo biloba extract. Clin. Chim. Acta, 2004, 339, 129-33.
- 46 OMRAN MAA., ABDEL-RAHMAN MS. Effect of scorpion Leiurus quinquestriatus (H & E) venom on the clinical chemistry parameters of the rat. Toxicol. Lett., 1992, 61, 99-109.
- 47 PANDEY R., DESHPANDE SB. Protective effects of aprotinin on respiratory and cardiac abnormalities induced by Mesobuthus tamulus venom in adult rats. Toxicon, 2004, 44, 201-5.
- 48 POSSANI LD., BECERRIL B., DELEPIERRE M., TYTGAT J. Scorpion toxins specific for Na+-channels. Eur. J. Biochem., 1999, 264, 287-300.
- 49 ROYSTON D. Preventing the inflammatory response to open-heart surgery: the role of aprotinin and other protease inhibitors. Int. J. Cardiol., 1996, 53, 11-37.
- 50 SANTAMARIA A., SANCHEZ-RODRIGUEZ J., ZUGASTI A., MARTINEZ A., GALVAN-ARZATE S., SEGURA-PUERTAS L. A venom extract from the sea anemone Bartholomea anuulata produces haemolysis and lipid peroxidation in mouse erythrocytes. Toxicology, 2002, 173, 221-8.
- 51 SARKAR S., YADAV P., TRIVEDI R., BANSAL AK., BHATNAGAR D. Cadmium-induced lipid peroxidation and the status of the antioxidant system in rat tissues. J. Trace Elem. Med. Biol., 1995, 9, 144-9.
- 52 SASTRE J., PALLARDO J., GARCIA DE LA ASCUNCIÓN J., VIÑA J. Mitochondria, oxidative stress and aging. Free Rad. Res., 2000, 32, 189-98.
- 53 SATOH H., NISHIDA S. Electropharmacological actions of Ginkgo biloba extract on vascular smooth and heart muscles. Clin. Chim. Acta, 2004, 342, 13-22.
- 54 SEDLAK J., LINDSAY RH. Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman's reagent. Anal. Biochem., 1968, 25, 192-205.
- 55 SIMMONS HF., JAMES RC., HARBISON RD., ROBERTS SM. Depression of glutathione by cold-restraint in mice. Toxicology, 1990, 61, 59-71.
- 56 WELT K., FITZL G., SCHAFFRANIETZ L. Myocardium-protective effects of Ginkgo biloba extract (EGb 761) in old rats against acute isobaric hypoxia. An electron microscopic morphometric study. II. Protection of microvascular endothelium. Exp. Toxicol. Pathol., 1996, 48, 81-6.
- 57 YADAV P., SARKAR S., BHATNAGAR D. Action of Capparis decidua against alloxan-induced oxidative stress and diabetes in rat tissue. Pharmacol. Res., 1997, 36, 221-8.
Publication Dates
-
Publication in this collection
26 June 2006 -
Date of issue
Apr 2006