Abstract
The cumulative actions of scorpion neurotoxins are complex and may be traced to activation of different ion channels with subsequent release of various transmitters and modulators including inflammatory mediators. This could lead to various pathological manifestations such as acute respiratory distress syndrome (ARDS), systemic inflammatory response syndrome (SIRS), and multiple organ failure (MOF). Several approaches have been advocated to treat the multitude of scorpion-venom-elicited pathological changes. However, few have tried to combat the venom-induced effects on the inflammatory process, which manifest as ARDS, SIDS and MOF. Thus, the aim of this study was to determine the capability of inhibitors of different steps of the inflammatory sequence of events in scorpion envenomation to ameliorate the detrimental action of the venom and prolong survival of mice injected with Leiurus quinquestriatus quinquestriatus (LQQ) venom. Animals were divided into groups (n = 10) and given montelukast (10 or 20 mg.kg-1, orally), hydrocortisone (5 or 10 mg.kg-1, intravenously) or indomethacin (10 or 20 mg kg-1, intravenously). Then, all animals were subcutaneously injected with either 0.25 or 0.3 mg.kg-1 LQQ venom. Signs and symptoms of envenomation were recorded and survival percentages after 24 hours as well as survival time were determined in each group. To analyze data, we utilized Covariance Wilcoxon survival statistics and survival distribution curves. In general, when compared to venom alone, administration of montelukast (p<0.001), hydrocortisone (p<0.05) and indomethacin (p<0.05) prolonged survival time and increased the percentage of surviving animals per group, with montelukast exhibiting the greatest protecting power. Thus, anti-inflammatory drugs may play an important role in protection against the lethal effects of scorpion venoms.
Leiurus quinquestriatus quinquestriatus; scorpion venom; acute respiratory distress syndrome; systemic inflammatory response syndrome; multiple organ failure; montelukast; hydrocortisone; indomethacin
ORIGINAL PAPER
Effect of selected anti-inflammatory drugs on the lethal actions of Leiurus quinquestriatus venom
Abdoon N. A.I; Ali A. A.II; Alnema A. A.II; Hag-Ali M.III; Fatani A. J.IV
INational Antivenom and Vaccine Production Center, King Fahad National Guard Hospital, Riyadh, Kingdom of Saudi Arabia
IICollege of Pharmacy, University of Khartoum, Khartoum, Sudan
IIIZayed Military Hospital, Abu Dhabi, United Arab Emirates
IVCollege of Pharmacy, King Saud University, Riyadh, Kingdom of Saudi Arabia
Correspondence to Correspondence to: Nuzha A. Abdoon National Antivenom and Vaccine Production Center, King Fahad National Guard Hospital P.O. Box 22490, Riyadh, 11426, Kingdom of Saudi Arabia Email: nozadoon@hotmail.com or amfatani@yahoo.com
ABSTRACT
The cumulative actions of scorpion neurotoxins are complex and may be traced to activation of different ion channels with subsequent release of various transmitters and modulators including inflammatory mediators. This could lead to various pathological manifestations such as acute respiratory distress syndrome (ARDS), systemic inflammatory response syndrome (SIRS), and multiple organ failure (MOF). Several approaches have been advocated to treat the multitude of scorpion-venom-elicited pathological changes. However, few have tried to combat the venom-induced effects on the inflammatory process, which manifest as ARDS, SIDS and MOF. Thus, the aim of this study was to determine the capability of inhibitors of different steps of the inflammatory sequence of events in scorpion envenomation to ameliorate the detrimental action of the venom and prolong survival of mice injected with Leiurus quinquestriatus quinquestriatus (LQQ) venom. Animals were divided into groups (n = 10) and given montelukast (10 or 20 mg.kg-1, orally), hydrocortisone (5 or 10 mg.kg-1, intravenously) or indomethacin (10 or 20 mg kg-1, intravenously). Then, all animals were subcutaneously injected with either 0.25 or 0.3 mg.kg-1LQQ venom. Signs and symptoms of envenomation were recorded and survival percentages after 24 hours as well as survival time were determined in each group. To analyze data, we utilized Covariance Wilcoxon survival statistics and survival distribution curves. In general, when compared to venom alone, administration of montelukast (p<0.001), hydrocortisone (p<0.05) and indomethacin (p<0.05) prolonged survival time and increased the percentage of surviving animals per group, with montelukast exhibiting the greatest protecting power. Thus, anti-inflammatory drugs may play an important role in protection against the lethal effects of scorpion venoms.
Key words:Leiurus quinquestriatus quinquestriatus, scorpion venom, acute respiratory distress syndrome, systemic inflammatory response syndrome, multiple organ failure, montelukast, hydrocortisone, indomethacin.
INTRODUCTION
Human envenomation by scorpions is a life-threatening hazard and fatal accidents are commonly reported in many regions of the world, especially with children (21, 25, 28). Neuro plus cardio-toxins are present in the majority of scorpion venoms (18), and from a pharmacological point of view, these two toxins are their most important components. Cumulative actions of scorpion neurotoxins may be traced to activation of different ion channels. The venom action on sodium channels of neuronal terminals may lead to depolarization of the axonal membrane and release of several neurotransmitters, which eventually affect various systems including the gastrointestinal tract, and the respiratory, cardiovascular and nervous systems (12, 17, 18, 22, 23, 26). In addition to neurotransmitters, other mediators such as those affecting inflammatory processes may be released after scorpion envenomation including kinins, ecosanoids, cytokines, platelet activating factor, permeability increasing factor, and nitric oxide (11, 16, 22, 32, 34). This release of cytokines and other mediators may account for several of the inflammatory manifestations observed such as acute respiratory distress syndrome [ARDS], systemic inflammatory responses syndrome [SIRS], and multiple organ failure [MOF] (18, 35, 36, 37). The ARDS, a syndrome that is related to the uncontrolled production and release of cytokines and other products of activated macrophage, lymphocytes and tissue resident cells, has been reported in children stung by scorpions (12, 48, 50) and may encompass the venom elicited non-cardiogenic pulmonary edema observed by several investigators (10, 33, 37, 48, 50). Clinical signs and symptoms of SIRS, a condition that may be caused by the massive release of cytokines and may be involved in the pathogenesis of shock, cardiac dysfunction and pulmonary edema (6), have been documented in severely envenomed victims (3, 48, 50). Moreover, it was stated that cytokines in conjunction with the neuro-endocrine axis might play a major role in the response to venom-evoked tissue injury, eventually leading to MOF (35, 48).
In general, treatment of scorpion-stung victims involves antivenom although its use remains controversial and symptomatic therapies including vasodilators, autonomic blockers, glucose-insulin infusion and inotropic drugs (8, 13, 24, 31, 45). Even though investigators have mentioned increased levels of selected cytokines after scorpion envenomation, few have tried to combat their effect and thus attempted to ameliorate venom-induced ARDS, SIRS, and MOF.
Ismail et al. (30) demonstrated the effectiveness of indomethacin and hydrocortisone anti-inflammatory agents in prolonging the survival of LQQ envenomed rats. In addition, Fatani et al. (16) showed that administration of aprotinin, a kallikren-kinin inhibitor, or icatibant, a B2 bradykinin antagonist, significantly attenuated venom-elicited cardio-respiratory abnormalities and increased survival of envenomed rabbits. This suggests that inflammatory mediators, prostaglandins or kinins play a role in venom-evoked effects such as cardio-respiratory changes, ARDS, SIRS, and MOF.
Aim of the work
Over the decades, much work has been performed to study the various aspects of scorpion envenomation and to discover how to alleviate the suffering of its victims; however, more studies are still needed. Scorpion venoms, by their capability to act mainly on Na+ channels, increase the release of various transmitters and modulators leading to a cascade of pathological events in different systems of the body. Recent reports have examined venom-induced non-cardiogenic pulmonary edema, ARDS, SIRS, and MOF. Investigators have related these actions to venom-evoked increased release and activation of endogenous immunological and inflammatory mediators (11, 16, 34, 36). However, it seems that the mechanisms involved in venom-elicited immunological and inflammatory responses have not been sufficiently investigated. Thus, the aim of this study is to discern whether blockers of different steps in the inflammatory sequence of events of scorpion envenomation could reduce venom-elicited inflammatory responses and prolong survival. This may lead to the emergence of new treatment modalities that could help save lives.
MATERIALS AND METHODS
Chemicals
Leiurus quinquestriatus quinquestriatus (LQQ) venom was obtained from mature scorpions collected from Aswan, Egypt, or Dongola, Sudan, by electrically stimulating their telsons, as described by Ismail et al. (29). Venom was then extracted by distilled water, centrifuged, and the supernatant was freeze-dried using HTRO SICC lyophilizer (Germany). The dried venom was kept at -20°C; when required, it was reconstituted by addition of 0.9% NaCl solution.
Montelukast and indomethacin were purchased from Merck Sharp and Dohm (UK), hydrocortisone from Upjohn (USA), and heparin from B-Breun (Germany).
Animals
Swiss albino white male mice weighing 20±0.1 g were obtained from the Experimental Animal Unit, National Antivenom and Vaccine Production Center, Riyadh, Kingdom of Saudi Arabia. Mice were kept at an ambient temperature and given free access to food and water. During the experiment, we followed the Institution's guide for the care and use of laboratory animals.
Methods
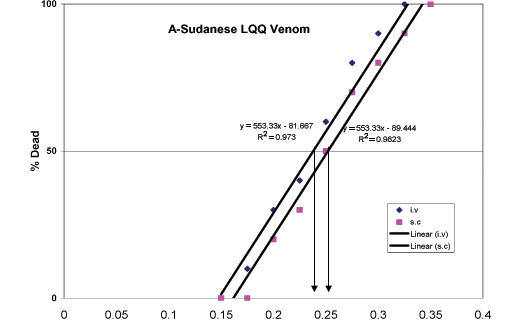
Mice were divided into different sets to determine both the intravenous and subcutaneous doses that would kill 50% of the animals (LD50) and the minimum lethal dose (MLD) for LQQ venoms collected from Egypt and Sudan, utilizing the method described by Miller and Tainter (38). Briefly, each group of 20 animals from both sets was subcutaneously injected with one of the following doses: 0.175, 0.2, 0.225, 0.25, 0.275, 0.3, 0.325, or 0.35 mg.kg-1 of Egyptian or Sudanese LQQ venom. The experiment was then repeated in a similar way, with the venom injected via the tail vein (0.15, 0.175, 0.2, 0.225, 0.25, 0.275. 0.3 or 0.325 mg.kg-1), for both types of venom. Signs and symptoms of envenomation as well as the number of dead animals after 24 hours were recorded. LD50 and MLD values were calculated using a special logarithmic graph paper.
In another set of experiments, animals were also divided into groups (n = 20) and given montelukast (10 or 20 mg.kg-1, orally, 2h before venom), hydrocortisone (5 or 10 mg.kg-1, intravenously, 30 min before venom), or indomethacin (10 or 20 mg.kg-1, intravenously, 30 min before venom); all doses according to Eum et al. (14), Burkovskaia (7), and Pakulska et al. (44), respectively. Then, 0.25 or 0.3mg.kg-1 of LQQ venom was subcutaneously injected into all animals. In control groups, mice were treated with 0.2 ml/mouse of 0.9% NaCl, montelukast, hydrocortisone or indomethacin. Moreover, two groups of animals were given LQQ venom alone, 0.25 or 0.3 mg.kg-1, as a positive control. Signs and symptoms of envenomation were recorded and mortality rate was determined in each group. Covariance Wilcoxon survival statistics was used to analyze data and survival distribution curves were drawn utilizing the SAS program with the aid of the Biomedical Statistics Department at the King Faisal Specialist Hospital Research Center. All values were presented as mean ± SEM with values of p<0.05 considered significant.
RESULTS
Effect of intravenous and subcutaneous Leiurus quinquestriatus quinquestriatus venom injection on mice survival
When Egyptian LQQ venom was injected into mice at gradually increasing doses ranging from 0.15 to 0.35 mg.kg-1, by intravenous (i.v.) or subcutaneous (s.c.) route, the LD50 of both routes were similar [i.v. route 0.23+0.02 mg.kg-1; s.c. route 0.255+0.018 mg.kg-1] (Figure 1B). In addition, the MLD was 0.325 mg.kg-1 for the i.v. route and 0.35 mg.kg-1 for the s.c. route. On the other hand, LD50 of Sudanese LQQ venom was 0.225+0.018 mg.kg-1 for the i.v. route and 0.245+0.02 mg.kg-1 for the s.c. route (Figure 1A), and its MLD was 0.325 mg.kg-1 for the i.v. route and 0.35 mg.kg-1 for the s.c. route. All animals injected with both venoms showed signs of scorpion envenomation, which included fighting behavior, lachrymation, hypersalivation, micturition, defecation, increased respiration, tremors, and occasional convulsions. Before death, the animals exhibited decreased motor activity, depressed respiration, gasping, and convulsions. Postmortem macroscopical examination of the hearts and lungs showed cardiac arrest in systole with congestion of the heart plus hemorrhagic patches in the lung.
In all subsequent experiments, Egyptian LQQ venom was utilized at doses equivalent to the LD50 presented by the i.v. and s.c. routes (0.25 mg.kg-1) and at a dose slightly lower than the MLD (0.3 mg.kg-1) found for this venom. Survival percentage of animals per group was 50% and the average survival time was 42.45+1.6 min when the lowest dose of the venom (0.25 mg.kg-1) was injected. On the other hand, the percentage of surviving animals per group was 10% and the average survival time was 28.85+1.1 min when the highest dose (0.3 mg.kg-1) was used.
Effect of montelukast oral administration on the survival of mice subcutaneously injected with Leiurus quinquestriatus quinquestriatus scorpion venom
In control groups (n = 10 animals), injection of normal saline (0.9% NaCl, 0.2 ml/mouse, intravenously), montelukast (10 and 20 mg.kg-1, orally), hydrocortisone (5 and 10 mg.kg-1, intravenously), or indomethacin (10 and 20 mg.kg-1, intravenously) caused no external signs of abnormal toxicity. All mice were alive at the end of the experiment time limit (300 min) in all the aforementioned assays.
Montelukast (MK), at the doses of 10 and 20 mg.kg-1, orally administered two hours before LQQ venom injection (0.25 mg.kg-1, subcutaneously), significantly increased the percentage of surviving animals from 50% with venom alone to 85% (10 mg.kg-1 MK) and 90% (20 mg.kg-1 MK), p<0.01. Also, the average survival time increased from 42.45+1.6 min to 193.75+1.4 (10 mg.kg-1 MK) and 217.6+0.6 min (20 mg.kg-1 MK), p<0.001. Likewise, it increased the percentage of surviving animals from 10% to 50% (10 mg.kg-1 MK) and 70% (20 mg.kg-1 MK), p<0.01, when animals were injected with 0.3 mg.kg-1LQQ venom 2 hours after. The average survival time was also increased from 28.85+1.1 min to 107.8+1.3 min (10 mg.kg-1 MK) and 115.1+1.5 min (20 mg.kg-1 MK), p<0.0001 (Table 1).
Effect of hydrocortisone intravenous injection on the survival of mice subcutaneously injected with Leirus quinquestriatus quinquestriatus venom
When hydrocortisone (HCTZ) at the doses of 5 and 10 mg.kg-1 was intravenously injected into mice half an hour before LQQ venom (0.25 mg.kg-1, subcutaneously), it increased the percentage of surviving mice from 50% with venom alone to 70% (5 mg.kg-1 HCTZ) and 75% (10 mg.kg-1 HCTZ), p<0.05. The average survival time was prolonged from 42.45+1.6 min to 108.9+1.4 min (5 mg.kg-1 HCTZ) and 101.1+0.5 min (10 mg.kg-1 HCTZ), p<0.05. When the highest dose of LQQ venom (0.3 mg.kg-1, s.c.) was utilized, the percentage of surviving mice increased from 10% to 45% with both doses of HCTZ (p<0.05), while the average survival time increased from 28.85+1.1 min to 80.45+1.4 (5 mg.kg-1 HCTZ) and 84.5+1.7 min (10 mg.kg-1 HCTZ), p<0.0001 (Table 1).
Effect of indomethacin intravenous injection on the survival of mice subcutaneously injected with Leiurus quinquestriatus quinquestriatus venom
Indomethacin (IND, 10 and 20 mg.kg-1) intravenous injection into mice half an hour before LQQ venom (0.25 mg.kg-1, subcutaneously) injection significantly increased the percentage of surviving animals from 50% with venom alone to 70% with both doses of the drug (p<0.05). The average survival time increased from 42.45+1.6 min to 100.05+0.4 min (10 mg.kg-1 IND) and 99.04+0.4 min (20 mg.kg-1 IND), p<0.05. Using the same 2 doses of IND with the highest dose of venom (0.3 mg.kg-1), the percentage of surviving animals increased from 10% with LQQ venom alone to 40% (10 mg.kg-1 IND) and 35% (20 mg.kg-1 IND), p<0.05, and the average survival time increased from 28.85+1.1 min to 80.2+2.3 (10 mg.kg-1 IND) and 75.96+1.7 min (20 mg.kg-1 IND), p<0.001 (Table 1).
DISCUSSION
In the present study, when a dose equivalent to either the intravenous or the subcutaneous LD50 of LQQ venom (0.25 mg.kg-1) was injected into mice, the survival percentage was approximately 50% and survival time was between 26 and 47 min. A dose slightly lower than the venom's MLD resulted in a survival percentage of 10% and survival time ranged from 17 to 36 min. These values were similar to those published by several investigators see review by Fatani (15) and attest to venom toxicity. Leirus quinquestriatus quinquestriatus is abundantly present in Middle Eastern countries such as Saudi Arabia, Egypt and Sudan. This species is known to be quite venomous, affecting both humans and experimental animals in a corresponding manner despite the zoological variation between different species (28) as shown in this study by the similarity between Egyptian and Sudanese venoms. Values of LD50 by subcutaneous and intravenous routes were similar, confirming the work of Ismail et al. (30), who showed that 70% of maximum blood concentration following LQQ venom subcutaneous injection into rats is reached within 15 min. In this work, injected mice usually showed signs of activation of the cardiovascular, respiratory, and nervous (autonomic and somatic) systems. This is in agreement with published data which showed similar signs and symptoms in different animal models injected with different scorpion venoms (16, 17, 19, 20, 28) and is comparable with those seen in clinical situations following envenomation (4, 5, 13, 25, 33).
This study demonstrated the effectiveness of montelukast in greatly protecting LQQ- envenomed mice and significantly prolonging their survival time. Montelukast is known to block the action of the cysteinyl leukotrienes (LT) C4, D4 and E4 mediated by cysteinyl leukotriene 1 receptors. These leukotrienes are products of the 5-lipooxygenase pathway of the arachidonic metabolism (27, 43). In addition, cysteinyl leukotriene receptor 1 antagonists exert part of their anti-inflammatory effects through the suppression of T helper type 2 (Th2) cells and thus through the subsequent inhibition of the production of allergen-specific IgE, mass cell degranulation, leukocyte trafficking, eosinophilia, T cell activation, release of histamines, leukotrienes, and pro-inflammatory cytokines such as interleukins 4, 5 and 13 (14, 27, 51).
Several investigators have demonstrated the capability of a number of scorpion venoms, including LQQ venom, to enhance the release of different inflammatory mediators such as cytokines IL-1B, IL-6, IL-8, IL-10 and TNF a, in addition to causing leucocytosis (22, 33, 36, 37). This venom-evoked release of cytokines and other mediators is thought to account for several inflammatory manifestations usually observed following scorpion envenomation such as acute respiratory distress syndrome [ARDS], systemic inflammatory responses syndrome [SIRS], and multiple organ failure [MOF] (5, 6, 37, 40, 42). There are now accumulating evidence to suggest a causal relationship between the overproduction of certain cytokines and both morbidity and mortality (37, 46). The efficacy of montelukast further points to the possibility that scorpion venoms release various cytokines and that they play an important role in the venom-induced pathological organ damages and subsequent lethality.
In the present study, administration of hydrocortisone significantly increased both the percentage of surviving mice and the average survival time. This result is in agreement with that obtained by Ismail et al. (30), who demonstrated hydrocortisone capability to significantly prolong survival of LQQ-envenomed mice. It is well documented that glucocorticoids, including hydrocortisone, inhibit the production of factors that are critical in generating inflammatory response. As a result, there is decreased release of vasoactive and chemoatractive factors, diminished secretion of lipolytic and proteolytic enzymes, decreased extravasation of leukocytes to areas of inflammation, and ultimately decreased fibrosis (9). The postulated beneficial effects of steroids in scorpion envenomation include modulation of inflammatory mediators, correction of putative acute adrenal insufficiency and consecutive hypovolemic shock induced by scorpion envenomation (30, 39). On the other hand, Abroug et al. (2) demonstrated a non-beneficial effect of hydrocortisone in human envenomation. A potentially important source of discrepancy between the two studies may be explained by the difference between experimental and clinical studies, or variation in the doses utilized. Further studies are needed to clarify this matter.
It was noticed that injection of indomethacin into mice half an hour before injection of LQQ venom significantly prolonged both the survival percentage and the average survival time. It is well known that indomethacin, a non-steriodal anti-inflammatory drug (NSAID), inhibits the enzymatic production of prostaglandins, which participate in the pathogenesis of inflammation and fever (41, 47). These drugs can inhibit cell migration, activation and function of neutrophils, and leukocytes adhesions, conditions that are known to accrue during the course of inflammation, perhaps by inhibiting membrane-associated processes independent of their ability to inhibit prostaglandins synthesis (1).
Several studies have emphasized the relevance of pro-inflammatory mediators including prostaglandins in the pathophysiological manifestations of human and experimental scorpion envenomation (11, 16, 18). Freire-Maia et al. (20) reported that injection of indomethacin was capable of decreasing the severity of lung edema induced by scorpion toxins in rats. Ismail et al. (30) related terminal hypotension, resistant to hypotensive agents that were shown in the majority of children who died from scorpion stings, to a possible involvement of either prostaglandins or kinins. Furthermore, lung edema evoked by administration of scorpion venom to experimental animals was related to activation of the inflammatory cascade and release of lipid-derived mediators of inflammation, including platelet activation factor, leukotrienes and prostaglandins (11).
Although indomethacin was shown to be effective in prolonging survival of envenomed animals, it was less efficient than montelukast and hydrocortisone. It was reported that despite playing an important role in the inhibition of prostaglandins synthesis, NSAIDs generally do not inhibit the formation of eicosanoids such as leukotrienes, which also contribute to inflammation, nor do they affect the synthesis of numerous other inflammatory mediators (1, 49) that play a role in scorpion envenomation.
In conclusion, it was evident in the present study that inhibitors of different steps of the inflammatory process may play an important role in prolonging survival following scorpion envenomation. Further studies are however needed to prove the value of such agents in clinical cases.
Received: June 29, 2005
Accepted: September 12, 2005
Abstract published online: September 13, 2006
Full paper published online: August 31, 2006
- 1 ABRAMSOM SR., WEISSMAN G. The mechanism of action of nonsteroidal anti-inflammatory drugs. Arthritis Rheum, 1989, 32, 1-9.
- 2 ABROUG F., NOUIRA S., HAGUIGA H., ELATROUS S., BELGHITH M., BOUJDARIA R., TOUZI N., BOUCHOUCHA S. High-dose hydrocortisone hemisuccinate in scorpion envenomation. Ann. Emerg. Med, 1997, 30, 23-7.
- 3 AMARAL CFS., BARBOSA AJA., LEITE VHR., TAFURI WL., DE REZENDE NA. Scorpion sting-induced pulmonary oedema: evidence of increased alveolocapillary membrane permeability. Toxicon, 1994, 32, 999-1003.
- 4 BAWASKAR HS., BAWASKAR PH. Sting by red scorpion (Buthotus tamulus) in Maharashtra State, India: a clinical study. Trans. R. Soc. Trop. Med. Hyg., 1989, 83, 858-60.
- 5 BERG RA., TARANTINO MD. Envenomation by the scorpion Centruroides exilicauda (C. Sculpturatus): severe and unusual manifestations. Pediatrics, 1991, 87, 930-3.
- 6 BONE RC. Toward a theory regarding the pathogenesis of the systemic inflammatory response syndrome: what we do and don't know about cytokine regulation. Crit. Care Med, 1996, 24, 163-72.
- 7 BURKOVSKAIA TE. Effect of hydrocortisone on the osteogenetic function of the bone marrow of the mouse. Kosm. Biol. Aviakosm. Med, 1986, 20, 83-6.
- 8 CAMPOS JA., SILVA OS., LOPEZ M., FREIRE-MAIA L. Signs, symptoms and treatment of severe scorpion poisoning in children. In: EAKER D., WADSTROM T. Eds. Natural toxins. Oxford: Pergamon Press, 1980: 61-8.
- 9 CHROUSOS GP. The hypothalamic-pituitary-adrenal axis and immune-mediated inflammation. N. Engl. J. Med., 1995, 332, 1351-62.
- 10 DEITCH EA. Multiple organ failure. Pathophysiology and potential future therapy. Ann. Surg, 1992, 216, 117-34.
- 11 DE-MATOS IM., ROCHA OA., LEITE R., FREIRE-MAIA L. Lung oedema induced by Tityus serrulatus scorpion venom in the rat. Comp. Biochem. Physiol C Pharmacol. Toxicol. Endocrinol, 1997, 118, 143-8.
- 12 D' SUZE G., COMELLAS A., PESCE L., SEVCIK C., SANCHEZ-DE-LEON R. Tityus discrepans venom produces a respiratory distress syndrome in rabbits through an indirect mechanism. Toxicon, 1999, 37, 173-80.
- 13 EL-AMIN EO. Issues in management of scorpion sting in children. Toxicon, 1992, 30, 111-5.
- 14 EUM SY., MAGHNI K., HAMID Q., CAMPBELL H., EIDELMAN DH., MARTIN JG. Involvement of the cysteinyl-leukotrienes in allergen-induced airway eosinophilia and hyperresponsiveness in the mouse. Am. J. Respir. Cell. Mol. Biol, 2003, 28, 25-32.
- 15 FATANI AJ. Pharmacological studies of the cardiovascular and related effects of scorpion envenomation: the setting up of an experimental treatment protocol Riyadh: King Saud University, 1990. 277p. [Masters Degree Thesis]
- 16 FATANI AJ., FURMAN BL., ZEITLIN IJ. The involvement of plasma kinins in the cardiovascular effects of Leiurus quinquestriatus scorpion venom in anesthetised rabbits. Toxicon, 1998, 36, 523-36.
- 17 FATANI AJ., HARVEY AL., FURMAN BL., ROWAN EG. The effects of lignocaine on actions of the venom from the yellow scorpion Leiurus quinquestriatus in vivo and in vitro Toxicon, 2000, 38, 1787-801.
- 18 FREIRE-MAIA L., ALMEIDA HO., CUNHA-MELO JR., AZEVEDO AD., BARROSO J. Mechanism of the pulmonary edema induced by intravenous injection of scorpion toxin in the rat. Agents Actions, 1978, 8, 113-8.
- 19 FREIRE-MAIA L., CAMPOS JA. Pathophysiology and treatment of scorpion poisoning. In: OWNBY CL., ODELL GV. Eds. Natural toxins: characterization, pharmacology and therapeutics Oxford: Pergamon Press, 1989: 139-59.
- 20 FREIRE-MAIA L., CAMPOS JA., AMARAL CF. Approaches to the treatment of scorpion envenoming. Toxicon, 1994, 32, 1009-14.
- 21 FREIRE-MAIA L., PINTO GI., FRANCO I. Mechanism of the cardiovascular effects produced by purified scorpion toxin in the rat. J. Pharmacol. Exp. Ther, 1974, 188, 207-13.
- 22 FUKUHARA YDM., REIS ML., DELLALIBERA-JOVILIANO R., CUNHA FQC., DONADI EA. Increased plasma levels of IL-1b, IL-6, IL-8, IL-10 and TNF-a in patients moderately or severely envenomed by Tityus serrulatus scorpion sting. Toxicon, 2003, 41, 49-55.
- 23 GUERON M., ILIA R., SOFER S. The cardiovascular system after scorpion envenomation: a review. J. Toxicol. Clin. Toxicol, 1992, 30, 245-58.
- 24 GUERON M., SOFER S. Vasodilators and calcium blocking agents as treatment of cardiovascular manifestations of human scorpion envenomation. Toxicon, 1990, 28, 127-8.
- 25 GUERON M., YAROM R. Cardiovascular manifestations of severe scorpion sting. Clinicopathologic correlations. Chest., 1970, 57, 156-62.
- 26 HARVEY AL., ANDERSON AJ., BRAGA MFM., MARSHALL DL., ROWAN EG., VATANPOUR H., CASTANEDA O., KARLSSON E. Toxins affecting neuronal ion channels. In: GOPALAKRISHNAKONE P., TAN CK. Eds. Recent advances in toxicology research Singapore: National University of Singapore, 1992: 59-70.
- 27 HENDERSON Jr. WR., TANG LO., CHU SJ., TSAO SM., CHIANG GK., JONES F., JONAS M., PAE C., WANG H., CHI EY. A role for cysteinyl leukotrienes in airway remodeling in a mouse asthma model. Am. J. Respir. Crit. Care Med., 2002, 165, 108-16.
- 28 ISMAIL M. The scorpion envenoming syndrome. Toxicon, 1995, 33, 825-58.
- 29 ISMAIL M., FATANI AJ., DABEES TT. Experimental treatment protocols for scorpion envenomation: a review of common therapies and an effect of kallikrein-kinin inhibitors. Toxicon, 1992, 30, 1257-79.
- 30 ISMAIL M., OSMAN OH., IBRAHIM SA., EL-ASMAR MF. Cardiovascular and respiratory responses to the venom from the scorpion Leiurus quinquestriatus. East Afr. Med. J, 1972, 49, 273-81.
- 31 KARNAD DR., DEO AM., APTE N., LOHE AS., THATTE S., TILVE GH. Captopril for correcting diuretic induced hypotension in pulmonary oedema after scorpion sting. Br. Med. J., 1989, 298, 1430-1.
- 32 LEME JG., PIMENTA AF., RAULINO-FLHO M., DINIZ CR. Sensory nerves and inflammation: evidence for the release of a neurogenic permeability factor by Tityustoxin J. Pathol, 1978, 124, 165-76.
- 33 MAGALHÃES MM., PEREIRA ME., AMARAL CF., REZEDE NA., CAMPOLINA D., BUCARETCHI F., GAZZINELLI RT., CUNHA-MELO JR. Serum levels of cytokines in patients envenomed by Tityus serrulatus scorpion sting. Toxicon, 1999, 37, 1155-64.
- 34 MATOS IM., TEXEIRA MM., LEITE R., FREIRE-MAIA L. Pharmacological evidence that neuropeptides mediate part of the actions of scorpion venom on the guinea pig ileum. Eur. J. Pharmacol, 1999, 368, 231-6.
- 35 MEKI AR., HASAN HA., EL-DEEN ZM., BAKKAR S. Dysregulation of apoptosis in scorpion envenomed children: its reflection on their outcome. Toxicon, 2003, 42, 229-37.
- 36 MEKI AR., MOHAMED ZM., MOHEY EL-DEEN ZM. Significance of assessment of serum cardiac troponin I and interleukin-8 in scorpion envenomed children. Toxicon, 2003, 41, 129-37.
- 37 MEKI AR., MOHEY EL-DEAN ZM. Serum interleukin-1B, interleukin-6, nitric oxide and alpha 1-antitrypsin in scorpion envenomed children. Toxicon, 1998, 36, 1851-9.
- 38 MILLER LC., TAINTER ML. Estimation of ED50 and its error. Proc. Soc. Exp. Biol. Med., 1949, 57, 261-4.
- 39 MOHAMMED AH., ROHAYEM H., ZAKY O. The action of scorpion toxin on blood sodium and potassium. J. Trop. Med. Hyg., 1954, 57, 85-7.
- 40. MOHEY EL-DEEN ZM., MEKI AR. Scorpion envenomation in Assiut Governorate. Analysis and clinico-laboratory study. Assiut Med. J, 1996, 20, 19-33.
- 41 MONCADA S., FERREIRA SH., VANE JR. Pain and inflammatory mediators. In: VANE JR., FERREIRA SH. Eds. Handbook of experimental pharmacology Berlin: Springer-Verlag, 1978: 588-617.
- 42. MULLER GJ. Scorpionism in South Africa. A report of 42 serious scorpion envenomation. S. Afr. Med. J., 1993, 83, 405-11.
- 43 NAYAK A. A review of montelukast in the treatment of asthma and allergic rhinitis. Expert Opin. Pharmacother, 2004, 5, 679-86.
- 44 PAKULSKA W., WEJMAN I., CZARNECKA E. Influence of felbamate on the antinociceptive action of morphine. Acta Pol. Pharma, 2003, 60, 201-6.
- 45 RADHA KRISHNA MURTHY K., VAKIL AE. Elevation of angiotensin levels in dogs by Indian red scorpion (Buthus tamulus) venom and its reversal by administration of insulin and tolazoline. Indian J. Med. Res, 1988, 88, 376-9.
- 46 ROUMEN RM., HENDRIKS T., VAN DER VEN-JONGEKRIJG J., NIEUWENHUIJZEN GA., SAUERWEIN RW., VAN DER MEER JW., GORIS RJ. Cytokine patterns in patients after major vascular surgery, hemorrhagic shock, and severe blunt trauma. Relation with subsequent adult respiratory distress syndrome and multiple organ failure. Ann. Surg, 1993, 218, 769-76.
- 47 SAMUELSSON B. Leukotrienes: mediators of immediate hypersensitivity reactions and inflammation. Science, 1983, 220, 568-75.
- 48 SOFER S., GUERON M., WHITE RM., LIFSHITZ M., APTE RN. Interleukin-6 release following scorpion sting in children. Toxicon, 1996, 34, 389-92.
- 49 VANE J. Towards a better aspirin. Nature, 1994, 367, 215-6.
- 50 VORONOV E., APTE RN., SOFER S. The systemic inflammatory response syndrome related to the release of cytokines following severe envenomation. J. Venom. Anim. Toxins, 1999, 1, 5-33.
- 51 WU AY., CHIK SC., CHAN AW., LI Z., TSANG KW., LI W. Anti-inflammatory effects of high-dose montelukast in an animal model of acute asthma. Clin. Exp. Allergy, 2003, 33, 359-60.
Correspondence to:
Publication Dates
-
Publication in this collection
19 Sept 2006 -
Date of issue
2006
History
-
Accepted
12 Sept 2005 -
Received
29 June 2005