Abstract
Red scorpion (Mesobuthus tamulus or Buthus tamulus) venom samples were collected at different regions of India: western (Chiplun and Ahmednagar from Maharashtra State) and southern (Ratnagiri and Chennai from Tamil Nadu State). The action of whole venoms on the blood sodium levels of mice was assessed using flame photometry. Seven peptides were common to all venom samples. They were separated using the native polyacrylamide gel electrophoresis (PAGE) technique and their activities were also studied using flame photometry. There was a decrease in the concentration of sodium ions in the serum, which suggested the blockage of such ions by scorpion venom toxins. Among the 10 protein bands isolated, the band at 79.6 kDa presented maximum activity in decreasing serum sodium ions concentration. Whole venom from Chiplun region also showed maximum activity. The western blotting technique demonstrated that the anti-scorpion venom sera produced by Haffkine Biopharmaceuticals Corporation Ltd., India, neutralized all four venom samples.
Red scorpion; Buthus tamulus; venom activity; sodium ion concentration; venom protein fraction; pharmacological action
ORIGINAL PAPER
The action of red scorpion (Mesobuthus tamulus coconsis, pocock) venom and its isolated protein fractions on blood sodium levels
Badhe R. V.I; Thomas A. B.I; Deshpande A. D.I, Salvi N.II; Waghmare A.II
IDepartment of Pharmaceutical Chemistry, Dr. D. Y. Patil Institute of Pharmaceutical Sciences and Research, Pimpri, Pune, Maharashtra, India
IIHaffkine Biopharmaceutical Corporation Ltd., Pimpri, Pune, Maharashtra, India
Correspondence to Correspondence to: Ravindra V. Badhe Department of Pharmaceutical Chemistry Dr. D. Y. Patil Institute of Pharmaceutical Sciences and Research Pimpri, Pune, 411018, Maharashtra, India Phone: +9194 2243 2038 Email: badheravi@yahoo.co.in
ABSTRACT
Red scorpion (Mesobuthus tamulus or Buthus tamulus) venom samples were collected at different regions of India: western (Chiplun and Ahmednagar from Maharashtra State) and southern (Ratnagiri and Chennai from Tamil Nadu State). The action of whole venoms on the blood sodium levels of mice was assessed using flame photometry. Seven peptides were common to all venom samples. They were separated using the native polyacrylamide gel electrophoresis (PAGE) technique and their activities were also studied using flame photometry. There was a decrease in the concentration of sodium ions in the serum, which suggested the blockage of such ions by scorpion venom toxins. Among the 10 protein bands isolated, the band at 79.6 kDa presented maximum activity in decreasing serum sodium ions concentration. Whole venom from Chiplun region also showed maximum activity. The western blotting technique demonstrated that the anti-scorpion venom sera produced by Haffkine Biopharmaceuticals Corporation Ltd., India, neutralized all four venom samples.
Key words: Red scorpion, Buthus tamulus, venom activity, sodium ion concentration, venom protein fraction, pharmacological action.
INTRODUCTION
Scorpion venom consists of numerous polypeptides (23), many of which interfere with the activity of ion channels and modulate their functional properties. These polypeptides have been used as tools to isolate ion channels and to study their pharmacology (18) and molecular mechanism of action. Sodium-channel inactivating toxins have similar active sites in rats and insects, in spite of their structural and pharmacological differences (9). Venom polypeptides have different physiological and pharmacological activities (22). They are specific to mammals, insects (32) and crustaceans (14), and have been used for insect control (11, 19). Scorpions of the Buthidae family are medicinally important and more toxic than any other scorpion species.
Scorpion venom toxins can be divided into four classes according to their physiological and pharmacological activities. The first class comprises the toxins acting on sodium channels, which consists of 60-70 amino-acid residues and four inter-molecular disulphide bonds. These long-chain toxins modulate the mechanism of activation or inactivation of sodium channels and are classified into ten subfamilies (21). The second class includes the toxins that act on potassium channels (10, 15, 28); the third class is represented by the toxins that act on chloride channels (3); and the fourth class of toxins comprises the toxins acting on calcium channels (30,31).
The Indian red scorpion (Mesobuthus tamulus or Buthus tamulus) is a dangerously venomous species, especially to children (24). Active fractions of Buthus tamulus venom have been characterized to act not only on various ion channels but also as protease inhibitors and histamine releasers (2). Iberiotoxin, isolated from B. tamulus venom, is a unique and highly specific blocker of the high-conductance calcium-activated potassium channel and has been completely characterized (7). Tamulus toxin is another toxin isolated from B. tamulus venom. It acts on potassium channels (27). Bt-IT, a mammalian-specific toxin (12), and Buta-IT and BtITx3, insect-specific toxins, were also isolated from B. tamulus (4, 29).
The present paper describes the isolation of peptides, using SDS-PAGE, from whole venoms of scorpions from different regions of India (Ratnagiri, Chiplun, Ahmednagar, and Tamil Nadu) as well as the characterization of their molecular weights followed by native PAGE to obtain nondenatured peptide fractions. Western blotting was also carried out in order to observe the efficiency of the anti-scorpion venom sera produced by Haffkine Biopharmaceuticals Corporation Ltd., Pune, India.
Seven peptides, common to scorpion venoms from all four regions, were isolated by extraction following native PAGE and then subjected to animal studies to assess their effects, together with the effects of the whole venom samples, on Na+ channels.
MATERIALS AND METHODS
Materials
All materials used for SDS-PAGE and native PAGE were of electrophoresis grade.
Mice were obtained from Serum Institute, Pune, M.S., India, and the Flame photometer, from Elico Make, India. Molecular weight markers and silver staining kit were obtained from Banglore Gennie, India.
Samples Preparation
Venom from the scorpion B. tamulus from different areas of India (Ratnagiri, Chiplun, Ahmednagar and Tamil Nadu) was utilized in lyophilized form. Crude venom (1mg) was weighed, dissolved in 0.5ml normal saline plus 0.5ml of 50% (v/v) glycerin solution, and stirred for 1h at 4°C. The solution was then centrifuged at 4,000Xg for 20 min at 4°C to separate the mucous material. This was a standard method set by Haffkine Biopharmaceuticals Corporation Ltd., Pune, India. The clear supernatant was separated and filtered through a 0.2µ filter. Protein concentration was assessed by the Lowry method, using bovine serum albumin as standard (16), and the solution was stored at 2-4°C and used for further processing.
Separation of Proteins using Gel Electrophoresis
Sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE) was carried out according to the method of Lammeli (13) using 15% polyacrylamide gel concentration. Molecular weight markers (205-3kDa) as well as Coomassie blue and silver staining were used.
Western blotting (6) was carried out to determine the reactivity of the horse anti-scorpion venom sera towards the four whole scorpion venoms. About 20µg of protein was separated by SDS-PAGE and transferred to a 0.45µm Immobilone-P membrane (Millipore) at 150mA for 3h in a mini-transblot cell (Biorad) using Tris-glycine buffer. Proteins on the membrane were reversibly stained with Ponceau S to check transfer efficiency. The gel was blocked with fat-free milk and incubated with anti-scorpion venom sera manufactured by Haffkine Biopharmaceuticals Corporation Ltd., Pune, India, Batch No. 402. Bound antibodies were detected with rabbit anti-horse IgG conjugated to horseradish peroxidase, using tetramethyl benzidine / hydrogen peroxide (TMB / H2O2) as substrates.
Native PAGE (6) was performed with 15% acrylamide concentration. To visualize nondenatured bands, Coomassie blue staining was used. Strips of the separated protein bands were cut out, crushed and kept in normal saline for 24h for extraction by ultrasonification. The extracted fractions contained the peptides equivalent to the whole venom.
Data Analysis
Similarity among scorpion strains from different regions was measured using the Jaccard Index [J] (17).
a = bands shared between two strains;
b = total number of bands in strain 1;
c = total number of bands in strain 2.
Animal Studies using Flame Photometer
A total of eleven groups of Swiss Albino mice, weighed about 28-30g, were used for the animal studies. The experiment was carried out in triplicate for each group. Animals received intraperitoneal injection of normal saline (blank) followed by injections of three whole venom samples (Ratnagiri, Ahmednagar and Tamil Nadu) at 100µg/ml concentration. Seven venom fractions (VF1-7) common to all venom samples, isolated from separated protein bands and extracted using native PAGE, were also intraperitoneally injected. Blood samples were collected after 2h by the retro-orbital route from all groups and centrifuged at 4,000Xg. The separated serum samples (200µl) were diluted with double distilled water and sodium ions concentration was determined using flame photometry.
RESULTS
Separation of Proteins using Gel Electrophoresis
SDS-PAGE indicated 10 peptide bands for the venom sample from Chiplun region, 8 peptide bands for those from Ratnagiri and Ahmednagar regions, and 7 peptide bands for the venom sample from Tamil Nadu region (Figure 1 and Table 1). Seven protein bands of 170, 79.6, 60, 57, 43, 38 and 29kDa molecular weights were present in the venom samples from all four regions.
The Jaccard index, calculated to assess similarity among scorpion strains from different regions of India, suggested a significant degree of similarity [J=0.33] between strains from Ratnagiri (strain 1) and Ahmednagar (strain 2). The degree of similarity between scorpion strains from Ratnagiri (strain 1) and Tamil Nadu (strain 2) was 0.31, and the least similarity [J=0.30] occurred between strains from Ratnagiri (strain 1) and Chiplun (strain 2). It can be inferred that there exists a genetic variation among the different strains of red scorpion from Western and Southern India.
Western blotting of all four venom samples (Ratnagiri, Ahmednagar, Tamil Nadu and Chiplun) using the anti-scorpion venom sera produced by Haffkine Biopharmaceutical Corporation Ltd. showed neutralization since there was specific binding of antibodies to the venom proteins (Figure 2).
Animal Studies using Flame Photometer
The venom dose used (100µg/ml), which is the minimum effective dose chosen by Haffkine Biopharmaceutical Corporation Ltd., was intraperitoneally administered to animals. Inflammation at the injection site, restlessness, increased urination and fecal discharge were noticed. About two and a half hours after venom injections, paralysis was observed in some animals.
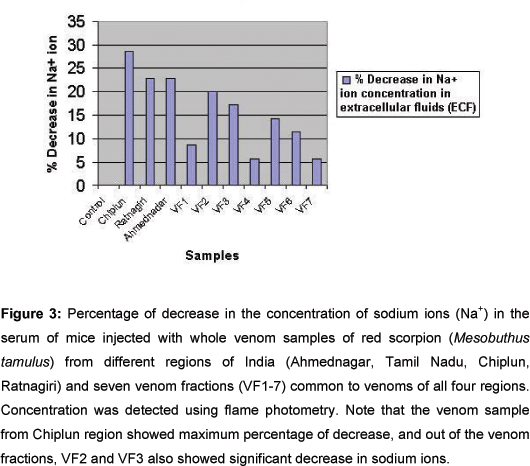
Compared with the blank (normal saline) and control samples, blood samples of animals injected with whole venom from Chiplun region plus VF2 and VF3 (79.6 and 60 kDa, respectively) showed the lowest concentration of sodium ions (Figure 3). These data corroborated the work of Gawade (8), and suggested that the venom sample of red scorpion from Chiplun region is more potent in decreasing sodium ions concentrations in the extracellular fluids (ECF) and that VF2 and VF3, present in all venom samples studied, led to maximum potency.
DISCUSSION
Results of SDS-PAGE indicated that there was a significant difference among the peptides composition of red scorpion (B. tamulus) venoms from different regions of India. Similarity index, based on the Jaccard index, suggested the existence of genetic variation among the different strains of red scorpion. Western blotting of the red scorpion venoms using anti-scorpion venom sera produced by Haffkine Biopharmaceutical Ltd. showed complete neutralization since there was antibody fixation to separated peptide bands of all four venom samples.
Sodium ions concentration in the serum samples, determined by flame photometry, demonstrated that the venom of red scorpion from Chiplun region was more effective in reducing sodium ions concentration in the blood, and that the venom fractions VF2 and VF3 (79.6 and 60kD), common to all venom samples, led to maximum potency. Flame photometry showed that the concentration of sodium ions in the serum of mice after injection of venom was lower than that after injection of normal saline (blank), which confirmed the hypothesis that scorpion venom can increase the influx of sodium ions resulting in a decreased concentration of such ions in the extracellular fluids (ECF).
Received: March 21, 2006
Accepted: July 27, 2006
Abstract published online: August 2, 2006
Full paper published online: February 28, 2007
Conflicts of interest: There is no conflict.
- 1 BANKS BEC., SHIPOLINI AA. Chemistry and Pharmacology of venom. In: PIEK T. Eds. Venoms of Hymenoptera New York: Academic Press, 1986: 329-416.
- 2 CHHATWAL GS., HABERMANN E. Neurotoxins, protease inhibitors and histamine releasers in the venom of the Indian red scorpion (Buthus tamulus): isolation and partial characterization. Toxicon, 1981, 19, 807-23.
- 3 DEBIN JA., MAGGIO JE., STRICHARTZ GR. Purification and characterization of chlorotoxin, a chloride channel ligand from the venom of the scorpion. Am. J. Physiol, 1993, 264, C361-9.
- 4 DHAWAN R., JOSEPH S., STHI A., LALA AK. Purification and characterization of a short insect toxin from venom of scorpion Buthos tamulus FEBS Lett., 2002, 528, 261-6.
- 5 DIMITROV SD., NATCHAEV LA. Fractions of some bee venom components on a new type of modified cellulose. Toxicon, 1977, 15, 447-8.
- 6 DOLEY R., MUKHERJEE AK. Purification and characterization of an anticoagulant phospholipase A2 from Indian cobra (Naja kaouthia) venom. Toxicon, 2003, 41, 81-91.
- 7 GALVEZ A., GIMENEZ-GALLEGO G., RUBEN JP., ROY-CONTANCIN L., FEIGENBAUM P., KACZOROWSKI GJ. Purification and characterization of a unique, potent, peptidyl probe for the high conductance calcium-activated potassium channel from the venom of the scorpion Buthus tamulus J. Biol. Chem, 1990, 265, 11083-90.
- 8 GAWADE SP. Excitatory effect of Buthus C-56 toxin on Drosophila larva neuromuscular junction. J. Venom. Anim. Toxins incl. Trop. Dis, 2003, 9, 57-9.
- 9 GORDON D., MARTIN-EAUCLAIRE MF., CESTELE S., KOPEYAN C., CARLIER E., KHALIFA RB., PELHATE M., ROCHAT H. Scorpion toxins affecting sodium current inactivation bind to distinct homologous receptor sites on rat brain and insect sodium channels. J. Biol. Chem, 1996, 271, 8034-45.
- 10 GURROLA GB., ROSATI B., ROCCHETTI M., PIMIENTA G., ZAZA A., ARCANGELI A. A toxin to nervous, cardiac, and endocrine ERG K+ channels isolated from Centruroides noxious scorpion venom. FASEB J, 1999, 13, 953-62.
- 11 HAMMOCK BD., MCCUTCHEN BF., BEETHAM J., CHOUDARY PV., FOWLER E., ICHINOSE R. Development of recombinant viral insectides by expression of an insect-specific toxin and insect-specific enzyme in nuclear polyhedrosis viruses. Arch. Insect Biochem Physiol, 1993, 22, 315-44.
- 12 LALA K., NARAYANAN P. Purification, N-terminal sequence and structural characterization of a toxic protein from the Indian scorpion venom, Buthus tamulus Toxicon, 1994, 32, 325-38.
- 13 LAMMELI UK., Cleavage of structural proteins during the assembly of the head of the bacteriophage T4. Nature, 1970, 227, 680-5.
- 14 LEBRETON F., DELEPIERRE M., RAMIREZ AN., BALDERS C., POSSANI LD. Primary and NMR three-dimensional structure determination of a novel crustacean toxin from the venom of the scorpion Centruroides limpidus Karsch. Biochemistry, 1994, 33, 11135-49.
- 15 LEGROS C., CEARD B., BOUGIS PE., MARTIN-EAUCLAIRE MF. Evidence for a new class of scorpion toxins against K+ channels. FEBS Lett., 1998, 413, 375-80.
- 16 LOWRY OH., ROSEBROUGH NH., FARR AL., RANDALL RJ. Protein measurement with the Folin phenol reagent. J. Biol. Chem, 1951, 193, 265-75.
- 17 LUDARING JA., RAYOLDS JF. Statistical Ecology. A primer on methods and compounds New York: John Willy and Sons, 1988: 85-103.
- 18 MACKINNON R., COHEN SL., KUO A., LEE A., CHAIT BT. Structural conservation in prokaryotic and eukaryotic potassium channels. Science, 1998, 280, 106-9.
- 19 MAEDA S., VOLRATH SL., HANZLIK TN., HARPER SA., MAJIMA K., MADDOX DW., HAMMOCK BD., FOWLER E. Insecticidal effects of an insect-specific neurotoxin expressed by a recombinant baculovirus. Virology, 1991, 184, 777-80.
- 20 MASTER RWP., RAO SS., SOMAN PD. Electrophoretic separation of biologically active constituents of scorpion venom. Biochem. Biophys. Acta., 1963, 71, 422-8.
- 21 POSSANI LD., BECERRIL B., DELEPIERRE M., TYTGAT J. Scorpion toxins specific for Na+ channels. Eur. J. Biochem, 1999, 264, 287-300.
- 22 ROCHAT H., BERNARD P., COURAUD F. Scorpion toxins: chemistry and mode of action. Adv. Cytopharmacol, 1979, 3, 325-34.
- 23 ROCHAT H., ROCHAT C., KOPEYAN C., MIRANDA F., LISSITZKY S., EDMAN P. Scorpion neurotoxins: a family of homologous proteins. FEBS Lett, 1970, 10, 349-51.
- 24 SANTHANAKRISHNAN BR., BALAGOPALA RV. Management of scorpion sting in children. J. Trop. Med. Hyg, 1974, 77, 133-5.
- 25 SCHMIDT JO. Biochemistry of insect venom. Ann. Rev. Entomol, 1982, 27, 339-68.
- 26 SOBOTKA AK., FRANKLIN RM., ADKINSON NF., VALENTINE MD., BEAR H., LICHTENSTEIN LM. Allergy to insect sting. II: The major allergen in venom. J. Allergy Clin. Immunol, 1976, 57, 29-40.
- 27 STRONG PN., CLARK GS., ARMUGAM A., ALLIE FA., JOSEPH JS., YEMUL V. Tamulustoxin: a novel potassium channel blocker from the venom of the Indian red scorpion Mesobuthus tamulus Arch. Biochem. Biophys., 2001, 385, 138-44.
- 28 TYTGAT J., CHANDY KG., GARCIA ML., GUTMAN GA., MARTIN-EAUCLAIRE MF., VAN DER WALT JJ., POSSANI LD. A unified nomenclature for short-chain peptides isolated from scorpion venoms: alpha-KTx molecular subfamilies. Trends Pharmacol. Sci., 1999, 20, 444-7.
- 29 WUDAYAGIRI R., INCEOGLU B., HERRMANN R., DERBEL M., CHOUDARY PV., HAMMOCK BD. Isolation and characterization of a novel lepidoptaran-selective toxin from the venom of South Indian red scorpion, Mesobuthus tamulus BMC Biochem, 2001, 2, 16-20.
- 30 ZAMUDIO FZ., CONDE R., AREVALO C., BECERRIL B., MARTIN BM., VALDIVIA HH., POSSANI LD. The mechanism of inhibition of ryanodine receptor channels by Imperatoxin I, a heterodimeric protein from the scorpion Pandinus imperator. J. Biol. Chem, 1997, 272, 11886-94.
- 31 ZAMUDIO FZ., GURROLA GB., AREVALO C., SREEKUMAR R., WALKER JW., VALDIVIA HN., POSSANI LD. Primary structure and synthesis of Imperatoxin A (IpTxa), a peptide activator of Ca2+ release channels/ryanodine receptors. FEBS Lett., 1997, 405, 385-9.
- 32 ZLOTKIN E., MIRANDA F., LISSITZKY S. Proteins in scorpion venoms toxic to mammals and insects. Toxicon, 1972, 10, 207-9.
Correspondence to:
Publication Dates
-
Publication in this collection
23 Apr 2007 -
Date of issue
2007
History
-
Accepted
27 July 2006 -
Received
21 Mar 2006