Abstract
Total RNA from lipopolysaccharide (LPS)-stimulated rat macrophages used to treat protoplasts from an Aspergillus nidulans strain originated the RT2 regenerated strain, whose culture supernatant showed anti-inflammatory activity in Wistar rats. The protein fraction presenting such anti-inflammatory activity was purified and biochemically identified. The screening of the fraction responsible for such anti-inflammatory property was performed by evaluating the inhibition of carrageenan-induced paw edema in male Swiss mice. Biochemical analyses of the anti-inflammatory protein used chromatography, carbohydrates quantification of the protein sample, amino acids content analysis and sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). Total sugar quantification revealed 32% glycosylation of the protein fraction. Amino acid analysis of such fraction showed a peculiar pattern presenting 29% valine. SDS-PAGE revealed that the protein sample is pure and its molecular weight is about 40kDa. Intravenous injection of the isolated substance into mice significantly inhibited carrageenan-induced paw edema. The isolated glycoprotein decreased carrageenan-induced paw edema in a prostaglandin-dependent phase, suggesting an inhibitory effect of the isolated glycoprotein on prostaglandin synthesis.
Aspergillus nidulans; protein purification and characterization; glycoprotein; anti-inflammatory activity
ORIGINAL PAPER
A glycoprotein with anti-inflammatory properties secreted by an Aspergillus nidulans modified strain
Queiroz J. C. F.I; Teixeira C. F. P.II; Lebrun I.III; Spencer P. J.IV; Zucchi T. M. A. D.I
IDepartment of Parasitology and Biotechnology Research Center, Institute of Biomedical Sciences, University of São Paulo, São Paulo, São Paulo State, Brazil
IILaboratory of Pharmacology, Butantan Institute, São Paulo, São Paulo State, Brazil
IIIBiochemistry and Biophysics Laboratorty, Butantan Institute, São Paulo, São Paulo State, Brazil
IVBiotechnology Center, Nuclear Energy Research Institute (IPEN/CNEN/SP), São Paulo, São Paulo State, Brazil
Correspondence to Correspondence to: Jean César Farias de Queiroz Av. Prof. Lineu Prestes, 1374 Cidade Universitária 05508-900, São Paulo, SP, Brasil Phone: 11 3091 7264 Email: queiroz.jcf@gmail.com
ABSTRACT
Total RNA from lipopolysaccharide (LPS)-stimulated rat macrophages used to treat protoplasts from an Aspergillus nidulans strain originated the RT2 regenerated strain, whose culture supernatant showed anti-inflammatory activity in Wistar rats. The protein fraction presenting such anti-inflammatory activity was purified and biochemically identified. The screening of the fraction responsible for such anti-inflammatory property was performed by evaluating the inhibition of carrageenan-induced paw edema in male Swiss mice. Biochemical analyses of the anti-inflammatory protein used chromatography, carbohydrates quantification of the protein sample, amino acids content analysis and sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). Total sugar quantification revealed 32% glycosylation of the protein fraction. Amino acid analysis of such fraction showed a peculiar pattern presenting 29% valine. SDS-PAGE revealed that the protein sample is pure and its molecular weight is about 40kDa. Intravenous injection of the isolated substance into mice significantly inhibited carrageenan-induced paw edema. The isolated glycoprotein decreased carrageenan-induced paw edema in a prostaglandin-dependent phase, suggesting an inhibitory effect of the isolated glycoprotein on prostaglandin synthesis.
Key words:Aspergillus nidulans, protein purification and characterization, glycoprotein, anti-inflammatory activity.
INTRODUCTION
To degrade substrate and absorb nutrients, fungi secrete a large spectrum of powerful enzymes into its environment (4). They also interact with the human environment by producing diseases in plants and animals and are used as food as well as for antibiotics production. Presently, these lower eukaryotes have been used as bio-factories to produce proteins with pharmacological and commercial properties (4, 11, 18). Such industrial values led Bennett (4) to evaluate the importance of fungi in Biotechnology and Mycotechnology and the results were very positive.
Filamentous fungi, particularly Aspergillus nidulans, have been extensively studied due to its capability to produce high levels of extracellular proteins in a very inexpensive way. Since most human proteins with therapeutic potential require post-translation modification to present biological properties, the eukaryotic expression-secretion system has become an interesting subject. Recent research on DNA-mediated transformation systems allowed the use of A. nidulans strains as host-cells for the production of recombinant proteins (10, 13, 14). Several well-characterized features and the availability of different mutants made A. nidulans the target organism to develop many expression-secretion systems. In mitotically stable multicopies, recombinant strains frequently present integrated expression cassettes. According to Archer and Peberdy (2) and Gouka et al. (10), Aspergillus fungi are excellent as receptor cells to express and secrete proteins of therapeutic importance.
Based on this potential, Zucchi et al. (27) used total RNA from LPS-stimulated rat (Wistar) macrophages to treat protoplasts from an A. nidulans strain (UT448). After protoplasts regeneration, colonies presenting clear anti-inflammatory factor in the culture supernant composed the RT2 strain that was genetically selected and pharmacologically analyzed. Recently, its epigenetic behavior has been studied.
The characterization of the factor produced by RT2 Aspergillus nidulans strain is very important to understand how Aspergillus fungi can cause invasive pulmonary infection without an immune response by the patients. In the present work, the biochemical properties of the anti-inflammatory factor found by Zucchi et al. (27) were purified and identified, and its activity in carrageenan-induced paw edema was tested in mice to verify which inflammatory mediator is blocked by this factor.
MATERIALS AND METHODS
Aspergillus nidulans Strains and Procedures
Originally from Glasgow, UK, the strains used were obtained from stocks from Utrecht, Holland, (UT448). The RT2 strain was obtained after total RNA treatment of UT448 protoplasts. RNA was extracted from LPS-stimulated rat (Wistar) macrophages (26). The mutant loci were described as: UT448 wA2 (II), white conidia; riboA1, pabaA124, biA1 (I), with need for riboflavin, p-aminobenzoic acid and biotin, respectively; AcrA1 (II), resistant to acriflavine. RT2 This strain was produced from UT448. It is prototrophic for all the genetic markers and acriflavine-resistant. The conidia color is green (26).
The minimum (MM) and complete (CM) media were prepared according to Van Arman et al. (25). Incubation temperature was 37ºC. Solid CM was used to maintain stocks at 4ºC or for the colonies development; mycelia of fungi were developed in liquid CM.
Conidia (106) from the RT2 strain were grown in 100ml liquid CM for 20h at 37ºC using an orbital shaker. After this period, mycelia were pelleted by centrifugation and aseptically filtered in cellulose acetate membranes (0.45µm), and 50mM EDTA was added to supernant in order to stop protease activities. The clarified supernant was frozen, liophylized and kept in freezer (-20ºC) until the next steps.
Gel Filtration
Gel filtration was carried out in a molecular exclusion Sephadex G100 column (60 X 2.5cm, Pharmacia, Upsalla, Sweden), previously equilibrated with 50mM ammonium bicarbonate, pH 7.0, and eluted with the same buffer at 8ml/h flow rate. One ml fractions were collected.
In vivo Assay for Detection of Anti-inflammatory Effects
To detect anti-inflammatory effects of protein fractions, in vivo assays were performed according to the method described by Garcia Leme et al. (9). Groups of six adult male Swiss mice were intravenously (tail vein) injected with selected concentrations of protein samples. Fifteen minutes after such treatments, animals were injected into the subplantar surface of one hind paw with 50µl of 1% carrageenan solution. The contralateral paw received the same volume of sterile saline. A control group received bovine serum albumin (BSA) intravenously, at the same concentration as the test substances. The increase in paw volume (edema) was measured through plethysmography, according to the method of Van Arman et al. (25), at 15, 30, 60, 90, 180, 240 and 360min intervals following carrageenan injection. Edema values were calculated as the difference between the values obtained in both paws and expressed as percentage (%) of increase in the paw volume. Results obtained for protein-treated animals were compared to those obtained for BSA-treated (control) animals.
Reversed-Phase Chromatography
For High-Performance Liquid Chromatography (HPLC), a C8 column (Shimadzu, 250 X 4.6mm, 5µm) was used, with the following buffers for elution: A) 0.1% orthophosphoric acid, and B) 10% buffer A plus 90% acetonitrile. The flow rate was 1ml/min and the gradient of buffer B was from 0% to 70% in 15min, kept at 70% for 2min, and decreased to 0% in 3min. The absorbance of the eluate was monitored at 214nm and the peaks were manually collected.
Trypsinization of the Collected Sample
Trypsin (666g/µl, Sigma) was diluted in Tris, 50mM, pH 8.5, at 1:1 (sample:trypsin) ratio; the solution was incubated at 37ºC under constant agitation. Samples were collected in triplicate at 0, 30, 60, 120 and 180min. Protein degradation of the purified and concentrated sample was determined using the Bradford method and observed at 495nm. Results corresponded to the average of triplicate readings.
Amino Acid Analysis using Acidic Hydrolysis
The amino acids concentration and composition were determined using sample hydrolysis in 6N HCl during 12h at 110ºC, after which the acid was removed by vacuum. The amino acids were derivatized with phenylisothiocyanate and identified using reversed-phase C18 column chromatography. Their composition and concentration were compared to an amino acid standard (Sigma), according to retention time and area of corresponding peaks. The procedure was adapted from Fullmer and Wasserman (8).
Total Sugar Quantification
Protein samples were diluted in 200µl distilled water. The glucose quantification standard curve was performed according to Chaplin and Kennedy method (5), in which 2, 4, 6, 8, 10, 20, 30, 40, 50, and 60µg glucose are diluted with 200µl of distilled water and used to establish a standard curve to quantify sugar. Results from samples were compared to the standard quantification curve.
SDS-PAGE
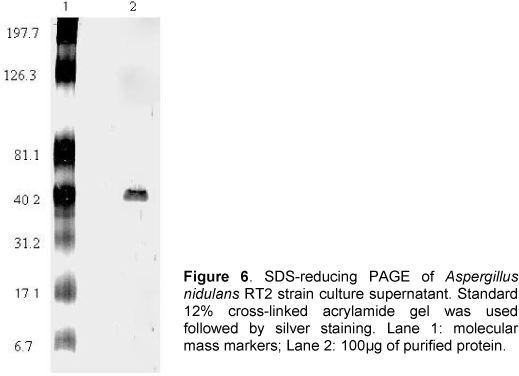
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) was performed as described by Laemmli (12) on one-dimensional 12% polyacrylamide slab gels containing 0.1% SDS, followed by standard silver staining. The subunit apparent molecular weight was determined by measuring and comparing relative motilities with the following prestained SDS-PAGE molecular weight standards (BIO-RAD): myosin (197.7kDa), b-galactosidase (126.3kDa), BSA (81.1kDa), carbonic anhydrase (40.2kDa), soybean trypsin inhibitor (31.2kDa), lysozyme (17.1kDa), and aprotinin (6.7kDa).
Statistical Analysis
Results were presented as the mean ± standard error of mean (S.E.M.) of six animals. The percentages of inhibition were reported as mean ± S.E.M. of the inhibitions obtained in each individual experiment during the edema peak. Data were statistically compared using analysis of variance (ANOVA) followed by Student's unpaired t-test. Values of p<0.05 were considered significant.
RESULTS
Protein Purification
Since large amounts of different proteins were expected, the collected samples were organized in six pools, I to VI, as presented in the first chromatogram (Fig. 1). Each pool was individually tested for paw edema development in Swiss male mice (Fig. 2).
The tests with samples from the first purification (Sephadex S100) were performed after 1, 3, and 6 hours (Fig. 2). This approach allowed the determination of the kinetics of the proteins effects on paw edema induced by carrageenan (1%) in male mice. Pools I, II, IV and VI did not cause any significant reduction in the paw volume at the times observed, which indicates that such pools do not contain any potent anti-inflammatory factor. Alternatively, Pool V caused paw volume reduction 3h after the carrageenan injection, although after 6h the anti-inflammatory potential was similar to those of the other pools. Pool II caused progressive reduction in the paw volume after the first hour of treatment, and after 3h, anti-inflammatory results similar to those of other pools were found.
To further evaluate the anti-inflammatory potential of Pools II and V, paw-induced edema tests were also carried out using saline and BSA as control. This strategy allows the exclusion of any other exogenous protein (such as albumin) also presenting anti-inflammatory response. The results are presented in Figure 3, indicating that the exogenous protein and Pool V had similar activities. Pool II showed significant anti-inflammatory potential decreasing paw volume gain from 61.85% with the albumin control to 14.37% when Pool II was used.
Based on such results, the subsequent experiments were performed using only Pool II as it clearly presented consistent and stable anti-inflammatory properties.
To evaluate the purity of Pool II, separation by a C8 reverse-phase column was carried out. The sample did not bind to the stationary phase of the column nor the retention time was altered, even when changing to acetonitrile gradient (Figure 4). Although observed at 280 and 214nm, Pool II was not adsorbed to the ion-exchange columns (anionic or cationic); thus, a confirmation of its proteic nature was needed. One-ml sample collected from reversed-phase C8 column presenting a peak with optical density of 1.1 at 280nm was added to 20µg trypsin and kept in a shaker at 37ºC for 3h. After 0, 30, 60, 120 and 180min incubation, 50µl samples were collected in triplicate.
The gradual protein decrease, as determined by the Bradford method, was presented in Figure 5. Results indicated that the molecule of interest is of proteic nature. The 90% reduction from the initial concentration showed that the protein presented few exposed lysine-arginine residues. The anti-inflammatory activity decrease was proportional to the trypsinization assay.
Amino Acid Analysis
As presented in Table 1, the test sample showed 10 peaks versus 14 presented by the standard sample (Sigma). Such fast screening does not allow discriminating glutamic acid from glutamine, alanine from threonine, and arginine from proline.
The relative amino acid proportion refers to the total of amino acids found in the protein. The highest proportions were: valine (29.39%), phenylalanine (18.57%) and histidine (17.16%); and the lowest were: aspartic acid (1.15%) and GIX (1.69%). This result agrees with the trypsinization test (Figure 5). Since the amino acid standards did not contain asparagine, this amino acid was not characterized by the screening. However, asparagine was recognized as the most N-glycosilated amino acid (Table 1).
Sugar Quantification in the Selected Protein
Protein (87.4g) was diluted in 200l distilled water. Spectrophotometric measurements revealed that the sample presented 32% glycosylation. This quite high value might explain why this protein was unable to bind to several kinds of chromatographic resins.
O-deglycosylation was performed using 25% ammonium hydroxide (NH4OH) incubated during 36h at 45º C, according to Rademaker et al. (19). The product resultant from protein deglycosylation did not have its anti-inflammatory property changed. Also, its chromatographic behavior remained the same, which suggests that this protein is a typical N-glycosylated product. Other works using glycoproteins from the genus Aspergillus supported this hypothesis, as N-glycosylation is common in such fungi (7, 16, 21).
SDS-PAGE
Figure 6 shows the results of SDS-PAGE of the purified anti-inflammatory protein from A. nidulans under reduced conditions followed by silver staining. The protein (Lane 2) showed a single band at ~40kDa, which indicates that the purification methods used were highly efficient.
In vivo Activity of the Purified Supernatant
After protein purification, the kinetics of the inflammatory process of the carrageenan-induced paw edema in mice was determined. Protein (1.0mg/kg/mice) was injected into groups of five animals. After carrageenan injections, the volumes of the animals' hind legs were evaluated at different times: zero, 15, 30, 60, 90, 180, 240 and 360min. After 30min, animals showed little differences, compared to the control group. Differences were only significant at 90 (p<0.05), 180 (p<0.02), and 240 (p<0.05) min after injections (Student's t-test). After 180min, differences between control and test groups were the highest; the protein doses caused 63.31% swelling reduction, compared to the control (Figure 7).
DISCUSSION
In 1996, Zucchi et al. (27) developed the RT1 and RT2 Aspergillus nidulans strains, which produced an anti-inflammatory factor. The activities produced by both strainswere already compared in a previous work, which led to the knowledge of the biochemical nature of that factor. In the present study, such factor was purified and biochemically characterized; its activity was also investigated. However, it is not unmistakably known if the protein under analysis is from the fungus or from the rat; this subject continues under inquiry.
The chosen method of gel filtration, as a first step, facilitated debris removal from the clarified culture medium. Purification refinement was possible using the molecular exclusion method in HPLC systems, which allowed high protein purity. However, ion exchange and the hydrophobic interaction for purification of the final product were not efficient. The high quantity and the composition of the carbohydrates might have impaired the adsorption of proteins to the matrix of many columns.
Purification of heterologous proteins from Aspergillus niger has been reported as well as detailed studies on other host cells producing heterologous protein (lysozyme) (22). In addition, its expression and purification have been characterized. The purification method used by those authors included gel filtration in a Sephachryl HR100 column, followed by ion exchange in SP Sepharose Big Beads and yielded high and specific activity. Record et al. (20), working on the purification of a protein from Aspergillus oryzae, used Sephadex G-75 gel filtration columns and DEAE-Sepharose and Mono Q ion-exchange columns. In this case, the higher the specific activity of the protein, the lower its recovery (Mono Q); conversely, the higher its recovery, the lower its specific activity (gel filtration).
Such examples reveal that the method chosen for purification is important and depends on the aims for the protein use. Gel filtration is extensively used as an initial method for separation followed by a refinement using ion exchange and can be considered a good model, as it allowed total separation of the proteins under study. However, in the present work, this was not feasible.
In the present study, 32.03% glycosylation was verified, which was considered very high but accounted for difficulties in the purification processes. Similarly, Panchal and Wodzinski (16) found 35.29% N-glycosylation in an Aspergillus niger phytase. However, the incidence of glucose among the proteins in the sample remains unclear. The supernatant proteins may have o-glycans in their structure because they are indispensable for the exportation of proteins (17).
In Aspergillus genus, protein glycosylation has been frequently characterized as N-glycosylation (3). After the expression of pectate lyase S in A. niger, two different forms of this enzyme appeared: a non-glycosylated and an N-glycosylated enzyme. They showed similar activity.
In another study, the same pectate lyase enzyme found in A. niger (7) was outstandingly expressed in A. nidulans, indicating that the protein produced was 1200Da greater than the natural one. Studies have revealed that such extra Molecular Weight (MW) was produced through N-glycosylation using mannose. In a work using A. sojae (6), the enzyme leucine aminopeptidase, which was secreted into the culture medium, presented 3% of the total MW due to N-glycosylation.
Sorensen et al. (21) verified oligosaccharides in A. niger that were related to asparagine residues. Walmsley and Hooper (26), studying the effect of N-glycosidases, verified that their recognition site was Asn-Ile/Phe-Thr. Again, the need for asparagine was verified, although in this case it was linked to an isoleucine or phenylalanine followed by a threonine. In the present work, despite the confirmation of the presence of isoleucine, phenylalanine and threonine, the presence of asparagine could not be verified, as this amino acid was not present in the protein standard used.
Analysis of the amino acid proportions showed that the N-glycosylation site Asn-Phe-Thr, described by Walmsley and Hooper (26), was probably included in the protein, even if the presence of asparagines could not be identified. This is the key amino acid for the action of N-glucosyltransferases, as described before (6, 7, 21). SDS-PAGE showed that there were not contaminant proteins, and this protein could be purified by the methods used.
The potent anti-inflammatory activity verified in the present study demonstrated that the isolated glycoprotein had specific effect since the treatment with an equivalent molar concentration of BSA did not show anti-inflammatory activity against carrageenan-induced paw edema. It decreased the edema, which has a peak characterized by the presence of prostaglandins (24), in a similar manner as other anti-inflammatory substances isolated from natural productssuch as the extracts from Pongamia pinnata leaves (23) and ReN1869 (15), using the same experimental edema model, which has been largely used.
The anti-inflammatory fraction, isolated from RNA-treated Aspergillus nidulans strain was demonstrated for the first time. The presence of such compounds in wild-type Aspergillus could explain why acute infection by Aspergillus sp. does not induce inflammatory responses in mammalian including humans (1). The isolated glycoprotein decreased carrageenan-induced paw edema in a prostaglandin-dependent phase, which indicates an inhibitory effect by the isolated glycoprotein on prostaglandin synthesis.
ACKNOWLEDGEMENTS
We are grateful to Professor Igor C. Almeida (University of São Paulo) for cooperation and discussion about protein glycosylation.
Received: July 19, 2006
Accepted: October 5, 2006
Abstract published online: November 16, 2006
Full paper published online: May 31, 2007
Financial Source: CNPq
Conflicts of interest: There is no conflict.
- 1 ANTACHOPOULOS C., ROILIDES E. Cytokines and fungal infections. Br. J. Haematol., 2005, 129, 583-96.
- 2 ARCHER DB., PEBERDY JF. The molecular biology of secreted enzyme production by fungi. Crit. Rev. Biotechnol., 1997, 17, 273-306.
- 3 BENEN JA., KESTER HC., PARENICOVA L., VISSER J. Characterization of Aspergillus niger pectate lyase A. Biochemistry, 2000, 39, 15563-9.
- 4 BENNETT JW. Mycotechnology: the role of fungi in biotechnology. J. Biotechnol., 1998, 11, 101-7.
- 5 CHAPLIN MF., KENNEDY JF. Carbohydrate analysis: A practical approach. 2. ed. New York: Oxford University Press, 1994.
- 6 CHIEN HC., LIN LL., CHAO SH., CHEN CC., WANG WC., SHAW CY., TSAI YC., HU HY., HSU WH. Purification, characterization, and genetic analysis of a leucine aminopeptidase from Aspergillus sojae Biochim. Biophys. Acta, 2002, 1576, 119-26.
- 7 COLANGELO J., LICON V., BENEN J., VISSER J., BERGMANN C., ORLANDO R. Characterization of the N-linked glycosylation site of recombinant pectate lyase. Rapid Commun. Mass Spectrom., 1999, 13, 2382-7.
- 8 FULMER CS., WASSERMAN RH. Analytical peptide mapping by high performance liquid chromatography. J. Biol. Chem., 1979, 254, 7208-12.
- 9 GARCIA LEME J., HAMMAMURA L., LEITE MP., ROCHA E SILVA M. Pharmacological analysis of the acute inflammatory process induced in the rat's paw by local injection of carrageenan and by heating. Br. J. Pharmacol., 1973, 48, 88-96.
- 10 GOUKA RJ., PUNT PJ., HESSING JGM., VAN DEN HONDEL CAMJJ. Efficient production of secreted proteins by Aspergillus: progress, limitations and prospects. Appl. Microbiol. Biotechnol., 1997, 47, 1-11.
- 11 IWASHITA M., HIRATA J., OGAWA K. Pseudodactylogyrus kamegaii sp. n. (Monogenea: Pseudodactylogyridae) from wild Japanese eel, Anguilla japonica Parasitol. Int., 2002, 51, 337-42. Erratum in: Parasitol. Int., 2003, 52, 185-6.
- 12 LAEMMLI U.K. Cleavage of structural proteins during assembly of the head of bacteriophage T4. Nature, 1970, 227, 6805.
- 13 LI DESTRI NICOSIA MG., BROCARD-MASSON C., DEMAIS S., HUA VAN A., Daboussi MJ., Scazzocchio C. Heterologous transposition in Aspergillus nidulans Mol. Microbiol, 2001, 39, 1330-44.
- 14 LUQUE R., OREJAS M., PEROTTI NI., RAMON D., LUCCA ME. pH control of the production of recombinant glucose oxidase in Aspergillus nidulans J. Appl. Microbiol., 2004, 97, 332-7.
- 15 OLSEN UB., ELTORP CT., INGVARDSEN BK., JORGESEN TK., LUNDBAEK JA., THOMSEN C., HANSEN AJ. ReN 1869, a novel tricyclic antihistamine, is active against neurogenic pain and inflammation. Eur. J. Pharmacol, 2002, 18, 435, 43-57.
- 16 PANCHAL T., WODZINSKI RJ. Comparison of glycosylation standards of phytase from Aspergillus niger (A. ficumm) NRRL 3135 and recombinant phytase. Prep. Biochem. Biotechnol., 1998, 28, 201-17.
- 17 PERLIDSKA-LENART U., KURZTKOWSKI W., JANAS P., KOPIDSKA A., PALAMARCZYK G., KRUSZEWSKA JS. Protein production and secretion in an Aspergillus nidulans mutant impaired in glycosylation. Acta Biochim. Pol., 2005, 52, 195-205.
- 18 PUNT PJ., VAN VIESEN N., CONESA A., ALBERS A., MANGNUS J., VAN DEN HONDEL C. Filamentous fungi as cell factories for heterologous protein production. TRENDS Biotechnol., 2002, 20, 200-6.
- 19 RADEMAKER GJ., PERGANTIS SA., BLOK-TIP L., LANGRIDGE JI., KLEEN A., THOMAS-OATES JE. Mass spectrometric determination of the sites of O-glycan attachment with low picomolar sensitivity. Anal. Biochem., 1998, 15, 257, 149-60.
- 20 RECORD E., ASTHER M., MARION D., ASTHER M. Purification and characterization of a novel specific phosphatidylglycerol-phosphatidylinositol transfer protein with high activity from Aspergillus oryzae Biochim. Biophys. Acta, 1995, 28, 1256, 18-24.
- 21 SORENSEN TK., DYER PS., FIERRO F., LAUBE U., PEBERDY JF. Characterization of the gtpA gene, encoding UDP N-acetylglucosamine: dolichol phosphatase N-acetylglucosaminylphosphoryl transferase, from the filamentous fungi Aspergillus niger Biochim. Biophys. Acta, 2003, 1619, 89-97.
- 22 SPENCER A., MOROZOV-ROCHE LA., NOPPE W., MACKENZIE DA., JEENES DJ., JONIAU M., DOBSON C., ARCHER DB. Expression, purification, and characterization of the recombinant calcium-binding equine lysozyme secreted by the filamentous fungus Aspergillus niger: Comparison with the production of hen and human lysozymes. Protein Expr. Purif., 1999, 16, 171-80.
- 23 SRINIVASAN K., MURUGANNANDAN S., LAL J., CHANDRA S., TANDAN SK., PRAKASH VR. Evaluation of anti-inflammatory activity of Pongamia pinata leaves in rats. J. Ethnopharmacol., 2001, 78, 151-7.
- 24 TAYLOR J., BRUNS T., WHITE T. "Fourth International Mycological Congress Abstracts," ed. by Reisinger A., Bresinsky A., Botanical Institute. Univ. of Regensburg, Regensburg - Germany, 1990, 343p.
- 25 VAN ARMAN CG., BEGANY AJ., MILLER LM., PLESS HH. Some details of inflammation caused by yeast and carrageenan. J. Pharmac. Exp. Ther., 1965, 150, 328-34.
- 26 WALMSLEY AR., HOOPER NM. Glycosylation efficiency of Asn-Xaa-Thr sequins is independent of distance from the terminus in membrane dipeptidase. Glycobiology, 2003, 13, 641-6.
- 27 ZUCCHI TMAD., CUNHA FQ., RIBEIRO RA., FERREIRA SH., DE LUCCA FL. Transformation of Aspergillus nidulans by RNA from rat macrophages stimulated with lipopolysaccharide. Cell. Mol. Biol. (Noisy-le grand), 1996, 42, 235-40.
Publication Dates
-
Publication in this collection
13 June 2007 -
Date of issue
2007
History
-
Accepted
05 Oct 2006 -
Received
19 July 2006