Abstract
The red seaweed Galaxaura marginata (Ellis & Solander) Lamouroux, well known by the antibacterial activity of its polar extract and the cytotoxic activity of its oxygenated desmosterol, showed anti-inflammatory action in its apolar fraction. Topical anti-inflammatory activity was observed in samples collected at São Sebastião channel, northern littoral of São Paulo State, Brazil. The apolar extract and its fractions obtained through Thin-Layer Chromatography (TLC) reduced the topical inflammation produced by croton oil in mouse ear. Such data indicated that the apolar extract from the marine red alga G. marginata displayed anti-inflammatory activity (since 1mg/ear extract reduced 95±0.5% inflammation), which could be the result of the synergic activity of the four fractions present in the apolar extract.
seaweed; Galaxaura marginata; inflammation; Croton oil; edema; anti-inflammatory agents
SHORT COMMUNICATION
Anti-inflammatory activity of the apolar extract from the seaweed Galaxaura marginata (Rhodophyta, Nemaliales)
Rozas E.; Freitas J. C.
Department of Physiology, Biosciences Institute, University of São Paulo, São Paulo, São Paulo State, Brazil
Correspondence to Correspondence to: Enrique Rozas Departamento de Fisiologia Instituto de Biociências Universidade de São Paulo Rua do Matão, Travessa 14, 101 05508-900, São Paulo, SP, Brasil Fax: (5511) 38187568 Email: enrique.sanchez@lycos.com
ABSTRACT
The red seaweed Galaxaura marginata (Ellis & Solander) Lamouroux, well known by the antibacterial activity of its polar extract and the cytotoxic activity of its oxygenated desmosterol, showed anti-inflammatory action in its apolar fraction. Topical anti-inflammatory activity was observed in samples collected at São Sebastião channel, northern littoral of São Paulo State, Brazil. The apolar extract and its fractions obtained through Thin-Layer Chromatography (TLC) reduced the topical inflammation produced by croton oil in mouse ear. Such data indicated that the apolar extract from the marine red alga G. marginata displayed anti-inflammatory activity (since 1mg/ear extract reduced 95±0.5% inflammation), which could be the result of the synergic activity of the four fractions present in the apolar extract.
Key words: seaweed, Galaxaura marginata, inflammation, Croton oil, edema, anti-inflammatory agents.
INTRODUCTION
In the last decades, seaweed metabolites presenting biological activities have been increasingly discovered. Such compounds have shown antibacterial, cytotoxic and anticoagulant activities; capability of agglutinating red blood cells and stimulating cell migration; anticancer properties; effects on the immune response; and anti-inflammatory activity (10). The latter has indicated those metabolites as a viable alternative to replace traditional drugs. Also, 6-n-tridecylsalicylic acid, isolated from Caulocystis cephalornithos, is an active anti-inflammatory agent in both acute and chronic animal models of inflammation and is chemically similar to the salicylic acid but less ulcerogenic a common side effect of salicylic acid (2).
Acute and chronic inflammations are complex processes that can be induced by several means, and anti-inflammatory agents exert their effects through different modes of action (1, 4).
For the screening of new anti-inflammatory compounds, the croton oil-induced mouse ear edema test is widely used together with the in vitro phospholipase A2 assay (5, 3). An acetylene-containing fatty acid derivative, isolated from the red seaweed Liagora farinosa, showed activity in both tests mentioned above, reducing the induced mouse ear edema and inhibiting the bee-venom phospholipase A2 (PLA2) activity (7, 6). Other compounds isolated from red algae Phacelocarpus labillardieri (Mertens) J. Agardh, Sphaerococcus coronopifolius Stackhouse, and Phacelocarpus labillardieri also showed bee-venom PLA2 inhibitory activity (6, 11). Extracts from brown seaweed Padina boergesenii and Hypnea valentia also inhibited the action by Naja nigricollis venom, when inoculated into mice and when in vitro assayed (12).
Galaxaura marginata, a red seaweed that belongs to the same order of L. farinosa, presents well-known significant cytotoxic activity against carcinoma and leukemia cells, showing different desmosterol structures acting on such cells and antibacterial activity (9, 8), but its anti-inflammatory action is unknown. The present study investigated the anti-inflammatory effects of G. marginata apolar substances on acute inflammation using the croton oil-induced mouse ear edema test.
To obtain the extract, 1kg sample of G. marginata was collected by free diving in shallow waters of São Sebastião channel (45º25'W; 26º49'S), northern coast of São Paulo State, Brazil, during the spring of 2000. The algae were washed with fresh water to remove epiphytes and salts. Fresh plant material was exhaustively extracted using ethanol:acetic acid (3:1 v/v) and filtered. The extract was concentrated under reduced pressure and partitioned against hexanes.
After the hexane was removed from the apolar phase, the extract was dissolved in acetone and kept at -20ºC until precipitate. A portion (5mg) of the precipitate was then separated through TLC (Si gel 60 F254) using ethyl acetate-hexane (2:3), producing four spots. Each spot was cut, scraped off and extracted with ethyl acetate-hexane; the fraction obtained from each spot was dispensed in glass vial, dried under nitrogen and weighed.
The topical anti-inflammatory activity of the apolar extract and fractions separated through TLC was tested using the mouse ear edema assay as experimental model of topical inflammation. Croton oil (Sigma-Aldrich Co.) was dissolved in acetone (20µg/ml). The mice left ears were topically treated with 20µl croton oil solution (200µg) and apolar extract (0.251.0mg/ear) or 20µl of croton oil and each fraction obtained from TLC (200µg/ear). Treatments were applied in the inner surface of the left ear of mice (Mus musculus, n=5, 25±3g), and 20µl acetone was applied in the right ear. As control, 100% inflammatory activity was induced by applying 20ml of croton-oil solution in the left ear and 20ml of pure acetone in the right ear of mice (Mus musculus, n=10, 25±3g). Ear disks of 6mm diameter were taken from the animals for analysis 3h after the treatments application. The anti-inflammatory activity (AI) was measured as follows: AI = [(WcontrolWtrial) / Wcontrol] X 100%, where Wcontrol = Left ear disk weight right ear disk weight from control animals; Wtrial = Left ear disk weight right ear disk weight from animals with the same concentration of the apolar extract and fractions tested.
Data analysis was carried out using the Graphpad Prism 4 software. The activity was quantitatively evaluated by determining the 50% Effective Concentration (EC50), using nonlinear regression with sigmoidal dose-response curve fit. Statistical analysis consisted of analysis of variance, and differences were evaluated using the Student's t-test; p<0.05 indicated statistical significance.
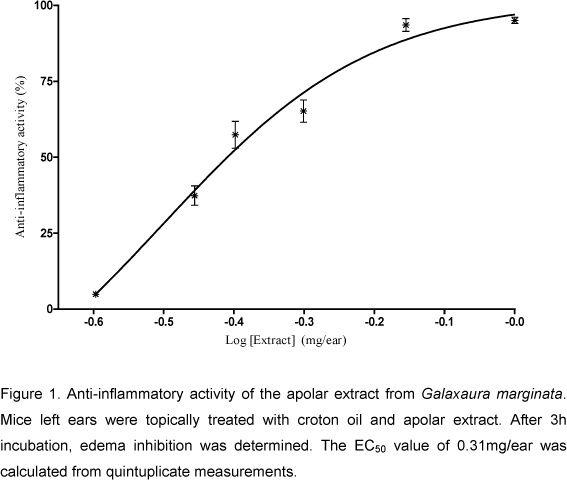
The apolar extract and the four fractions obtained from TLC separation showed inhibition of the edema induced by croton oil in mouse ear. The extract reduced 95±0.5% inflammation when administered at the highest dose (1mg/ear). The EC50 was reached when mice ears were treated with 0.31mg/ear (95% confidence intervals: 0.240.41mg/ear; Fig. 1). The four TLC substances inhibited inflammation by 55%, 75%, 100%, and 100%, respectively, considering the chromatography speed order.
Extracts as well as structurally diverse compounds obtained from marine red algae have been shown to inhibit inflammation (6, 7). In the present work, such findings were extended by reporting that marine red alga G. marginata displayed anti-inflammatory activity in its apolar extract, which was ten-fold more potent than the apolar substance obtained from L. farinosa (7). Thus, as phospholipase A2 controls inflammatory responses, the edema reduction in the mouse ear indicated that the apolar extract from G. marginata influences PLA2 activity.
ACKNOWLEDGEMENTS
The authors are grateful to Jose Eduardo Martinelli, MS, for his comments on the manuscript.
Received: September 15, 2006
Accepted: March 23, 2007
Abstract published online: March 26, 2007
Full paper published online: May 31, 2007
Financial source: FAPESP (00/07374-3).
Conflicts of interest: There is no conflict
- 1 BRUNO A., ROSSI C., MARCOLONGO G., DI LENA A., VENZO A., BERRIE Ch., CORDA D. Selective in vivo anti-inflammatory action of the galactolipid monogalactosyldiacylglycerol. Eur. J. Pharmacol., 2005, 524, 159-68.
- 2 BUCKLE P., BALDO B., TAYLOR KM. The anti-inflammatory activity of marine natural products 6-n-tridecylsalicylic acid, flexibilide and dendalone 3-hydroxybutyrate. Agents Actions, 1980, 10, 361-7.
- 3 FAROOQUI A., LITSKY M., FAROOQUI T., HORROCKS Ll. Inhibitors of intracellular phospholipase A2 activity: their neurochemical effects and therapeutical importance for neurological disorders. Brain Res. Bull., 1999, 49, 139-53.
- 4 FIERRO I., SERHAN C. Mechanisms in anti-inflammation and resolution: the role of lipoxins and aspirin-triggered lipoxins. Braz. J. Med. Biol. Res., 2001, 34, 555-66.
- 5 HAN CH., SON M., CHANG H., CHI Y., PARK H., KIM H. Inhibition of prostaglandin production by a structurally-optimized flavonoid derivative, 2,4,7-trimethoxyflavone and cellular action mechanism. Biol. Pharm. Bull., 2005, 28, 1366-70.
- 6 MAYER AM., PAUL VJ., FENICAL W., NORRIS JN., CARVALHO MS., JACOBS RS. Phospholipase A2 inhibitors from marine algae. Hydrobiologia, 1993, 260/261, 521-9.
- 7 MENDONÇA P., FREITAS JC. Topical antiedematous activity of the organic extract of Liagora farinosa algae (Rhodophyta, Nemaliales). Ciênc. Cult., 2000, 52, 175-8.
- 8 ROZAS EE., FREITAS JC. Antibacterial activity of the extract of the seaweed Galaxaura marginata (Rhodophyta, Nemaliales). J. Venom. Anim. Toxins incl. Trop. Dis., 2003, 9, 497.
- 9 SHEU J., HUANG S., DUH CH. Cytotoxic oxigenated desmosterols of the red alga Galaxaura marginata. J. Nat. Prod., 1996, 59, 23-6.
- 10 SMITH A. Medicinal and pharmaceutical uses of seaweed natural products: a review. J. Appl. Phycol., 2004, 16, 245-62.
- 11 SONG D., BLOND G., FURSTNER A. Study towards bioactive pyrone derivatives from the marine red alga Phacelocarpus labillardieri Tetrahedron, 2003, 59, 6899-904.
- 12 VASANTHI H., JASWANTH A., KRISHNARAJ V., RAJAMANICKAM G., SARASWATHY A. In vitro snake venom detoxifying action of some marine algae of Goulf of Mannar, South East of India. Phytotherapy Res., 2003, 17, 1217-9.
Publication Dates
-
Publication in this collection
13 June 2007 -
Date of issue
2007
History
-
Received
15 Sept 2006 -
Accepted
23 Mar 2007