Abstract
Amphibian skin secretions are a source of potential new drugs with medical and biotechnological applications. Rich in peptides produced by holocrine-type serous glands in the integument, these secretions play different roles, either in the regulation of physiological skin functions or in the defense against predators or microorganisms. The aim of the present work was to identify novel peptides with bradykinin-like structure and/or activity present in the skin of Phyllomedusa nordestina. In order to achieve this goal, the crude skin secretion of this frog was pre-fractionated by solid phase extraction and separated by reversed-phase chromatography. The fractions were screened for low-molecular-mass peptides and sequenced by mass spectrometry. It was possible to identify three novel bradykinin-related peptides, namely: KPLWRL-NH2 (Pnor 3), RPLSWLPK (Pnor 5) and VPPKGVSM (Pnor 7) presenting vascular activities as assessed by intravital microscopy. Pnor 3 and Pnor 7 were able to induce vasodilation. On the other hand, Pnor 5 was a potent vasoconstrictor. These effects were reproduced by their synthetic analogues.
Phyllomedusa nordestina; bradykinin; mass spectrometry; natural peptides
Identification of bradykinin-related peptides from Phyllomedusa nordestina skin secretion using electrospray ionization tandem mass spectrometry after a single-step liquid chromatography
Conceição KI; Bruni FMI; Sciani JMIII; Konno KI; Melo RLI; Antoniazzi MMII; Jared CII; Lopes-Ferreira MI; Pimenta DCIII
ISpecial Laboratory of Applied Toxinology, Center for Applied Toxinology, CAT/CEPID, Butantan Institute, São Paulo, São Paulo State, Brazil
IILaboratory of Cell Biology, Center for Applied Toxinology, CAT/CEPID, Butantan Institute, São Paulo, São Paulo State, Brazil
IIILaboratory of Biochemistry and Biophysics, Butantan Institute, São Paulo, São Paulo State, Brazil
Correspondence to Correspondence to: Daniel Carvalho Pimenta Avenida Vital Brazil, 1500, São Paulo, SP, 05503-900, Brasil. Phone/fax: + 55 11 3726 72 22, ext. 2101. Email: dcpimenta@butantan.gov.br.
ABSTRACT
Amphibian skin secretions are a source of potential new drugs with medical and biotechnological applications. Rich in peptides produced by holocrine-type serous glands in the integument, these secretions play different roles, either in the regulation of physiological skin functions or in the defense against predators or microorganisms. The aim of the present work was to identify novel peptides with bradykinin-like structure and/or activity present in the skin of Phyllomedusa nordestina. In order to achieve this goal, the crude skin secretion of this frog was pre-fractionated by solid phase extraction and separated by reversed-phase chromatography. The fractions were screened for low-molecular-mass peptides and sequenced by mass spectrometry. It was possible to identify three novel bradykinin-related peptides, namely: KPLWRL-NH2 (Pnor 3), RPLSWLPK (Pnor 5) and VPPKGVSM (Pnor 7) presenting vascular activities as assessed by intravital microscopy. Pnor 3 and Pnor 7 were able to induce vasodilation. On the other hand, Pnor 5 was a potent vasoconstrictor. These effects were reproduced by their synthetic analogues.
Keywords: Phyllomedusa nordestina, bradykinin, mass spectrometry, natural peptides.
INTRODUCTION
Amphibian skin secretions contain a large number of biologically active molecules that function not only as a protection against microorganisms, but also as an anti-predator system (1, 2-4). Moreover, some amphibian secretions contain a number of small proteins and a large number of peptide components whose biological functions are still undetermined. Several peptides from skin secretions of amphibians, more particularly tree-frogs, have been purified in recent years, including antimicrobial peptides, bradykinin related peptides and one bradykinin-potentiating peptide (BPP) (5-9).
The generation of bradykinin (BK) in the blood by the action of the kallikreinkinin system has been studied intensively in mammals, but the system has received relatively little attention in non-mammalian vertebrates. Amphibian skin has proven a remarkably rich storehouse of regulatory peptides including kinins, while the occurrence of BK-related peptides in skin secretions of frogs has been investigated by several groups (1, 10-12). Yet, the search for the existence of at least one component of the kallikreinkinin system in anuran plasma has been unsuccessful, for instance one in the form of a biosynthetic precursor (e.g., pre-pro-BK) (13).
The hylid genus Phyllomedusa contains 32 species distributed throughout southern Central America and much of South America (14, 15). Phyllomedusa nordestina, formely comprised into Phyllomedusa hypochondrialis, is a typical Brazilian genus, inhabiting the semi-arid region of northeastern Brazilian (15-17). Peptides weighing less than 5 kDa are the predominant molecules in the secretions of many tree frogs of the sub-family Phyllomedusinae, whereas bradykinin and bradykinin-related peptides (BRP) have been identified in the skin secretions of these animals. The fact that such analogues have been found in these skin secretions supports the idea that BRP are produced as part of a diverse defense system.
The aim of the present work was to investigate directly by RP-HPLC and mass spectrometry the low-molecular-mass peptides from P. nordestina skin secretion. A single chromatographic step was chosen in order to avoid the loss of the minor components. Matrix assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF/MS) and orthogonal quadrupole time-of-flight electrospray ionization tandem mass spectrometry (ESI-Q-TOF-MS/MS) de novo sequencing analyses yielded three new sequences with vascular activities, as assessed by intravital microscopy, that were reproduced by the synthetic analogues. Taken together, these results indicate the usefulness of this sole chromatographic step approach that, combined with specific biological assays, was able to successfully identify low-abundance small bioactive peptides from complex samples such as this anuran skin secretion.
MATHERIALS AND METHODS
Animals
Groups of five male Swiss mice weighing 18 to 22 g were used throughout. The animals, provided by Instituto Butantan animal house, were kept in temperature and humidity-controlled rooms, and received food and water ad libitum. All the procedures involving mice were in accordance with the guidelines provided by Ethics Committee on Animal Use of Butantan Institute (n. 378/07).
Drugs and Reagents
Sep-Pak C18® cartridges were purchased from Waters Corporation (USA); Fmoc-amino acids from Calbiochem-Novabiochem Corporation (USA); and acetonitrile (HPLC grade) was purchased from J. T. Baker (USA). Alpha-cyano-4-hydroxycinnamic acid, iodoacetamide, NaI, molecular mass standards (ProteoMass Kit®) and acetylcholine hydrochloride were all obtained from Sigma Co. (USA). Sodium pentobarbital and sodium heparin were acquired from Roche Laboratories (Brazil).
Collection of Specimens
Specimens (n = 12; non-sexed) of Phyllomedusa nordestina were collected at Angicos, in the state of Rio Grande do Norte, Brazil. The tree frogs were kept alive in the animal house of the Department of Cellular Biology, Butantan Institute, São Paulo, Brazil. The tree frogs were collected according to the Brazilian environmental agency (Brazilian Institute of Environment and Renewable Natural Resources IBAMA) under the license 02027.023238/03-91.
Purification Procedures
Skin secretion obtainment and solid phase extraction
Glandular secretions were obtained from adult specimens of P. nordestina, submerged in a beaker containing deionized water, that were manually and gently compressed. This solution was lyophilized and stored at 20°C. Pooled lyophilized secretions from twelve animals were dissolved in deionized water and centrifuged at 5000 x g for 20 minutes (room temperature). The supernatant was pre-purified by solid phase extraction (SPE) using Sep-Pak C18® cartridges (Waters Corporation, USA). A single aliquot of 2 mg diluted in 2 mL of 0.1% TFA was loaded; then 80% acetonitrile containing 0.1% TFA was eluted. This sample was lyophilized to prior analysis.
Reversed phase chromatography
A reversed-phase binary HPLC system (Äkta, Sweden) was used for sample separation. The SPE-eluted fraction was loaded in a Shimadzu C18 column (Shim-Pack® 5µ, 4.6 x 250 mm, Japan) in a two-solvent system: (A) trifluoroacetic acid (TFA)/H2O (1:1000) and (B) TFA/acetonitrile (ACN)/H2O (1:900:100). The column was eluted at a flow rate of 1.0 mL/min with a 10 to 80% gradient of solvent B over 60 minutes. The HPLC column eluents were monitored by their UV absorbance at 214 nm.
Mass spectrometry analyses
Peptide mass analyses were performed on a Q-TOF Ultima API® (Micromass, UK), under positive ionization mode and/or by MALDI-TOF mass spectrometry on an Ettan MALDI-TOF/Pro system® (Amersham Biosciences, Sweden) using α-cyano-4-hydroxycinnamic acid as matrix.
Mass spectrometric de novo peptide sequencing was carried out in positive ionization mode on a Q-TOF Ultima API® fitted with an electrospray ion source (Micromass, UK). Samples were dissolved into a mobile phase of 50% acetonitrile, containing 0.1% formic acid, and directly injected (10 µL) using a Rheodyne 7010® sample loop (USA) coupled to a LC-10A VP® Shimadzu (Japan) pump at a constant 20 µL/minute flow rate. The instrument control and data acquisition were conducted by MassLynx 4.0® data system (Micromass, UK) and experiments were performed by scanning from a mass-to-charge ratio (m/z) of 50 to 1800 using a scan time of 2 seconds applied during the whole infusion. The mass spectra corresponding to each signal from the total ion current (TIC) chromatogram were averaged, allowing an accurate molecular mass determination. External calibration of the mass scale was performed with NaI. For the MS/MS analysis, collision energy ranged from 18 to 45 eV and the precursor ions were selected under a 1-m/z window. All MS/MS spectra were analyzed manually; the sequences were determined by precise mass differences between adjacent b ions (confirmed by y ions).
Peptide synthesis
Synthetic peptides were obtained by an automated benchtop simultaneous multiple solid-phase synthesizer (PSSM 8® system from Shimadzu, Japan) using solid phase peptides synthesis by the Fmoc procedure (18). The peptides were purified by reversed-phase chromatography (Shim-pack Prep-ODS®, 5µ, 20 x 250 mm, Shimadzu, Japan) in a binary semi-preparative system and their purity and identity were confirmed by MALDI-TOF and/or ESI-Q-TOF mass.
Intravital microscopy
The dynamic of alterations in the microcirculatory network was determined using intravital microscopy by transillumination of mouse cremaster muscle after topical application of the peptide. Sterile saline was used as control. In three independent experiments (n = 5, for each experiment) mice were anaesthetized with sodium pentobarbital (Hypnol®, Cristália, Brazil; 50 mg/kg, intraperitoneal route) and the cremaster muscle was exposed for microscopic examination in situ as described by Lomonte et al. (19). The animals were maintained on a special board thermostatically controlled at 37°C, which included a transparent platform on which the tissue to be transilluminated was placed. Images of the microcirculation were simultaneously visualized on TV and computer monitors using a color video camera (TK-C600®, JVC, Japan) incorporated to a triocular microscope (Axioskope®, Carl-Zeiss, Germany). Images obtained on the TV monitor were recorded on a video recorder and computer digitized images were analyzed using standard imaging software (KS300®, Kontron, Germany). The images were obtained using a ×10/025 longitudinal distance objective/numeric aperture and ×1.6 optovar.
Statistical Analyses
One-way analysis of variance (ANOVA), followed by Newman-Keuls test, was performed to determine significance of differences. The significance level was set at p < 0.05.
RESULTS
Peptide Pooling
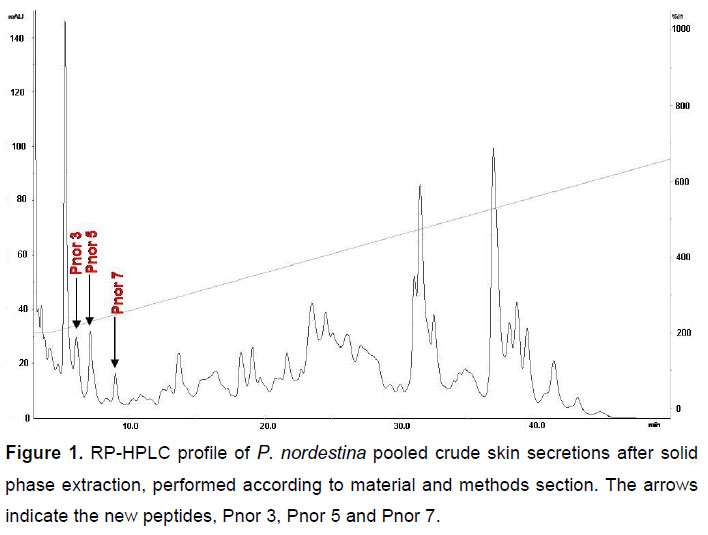
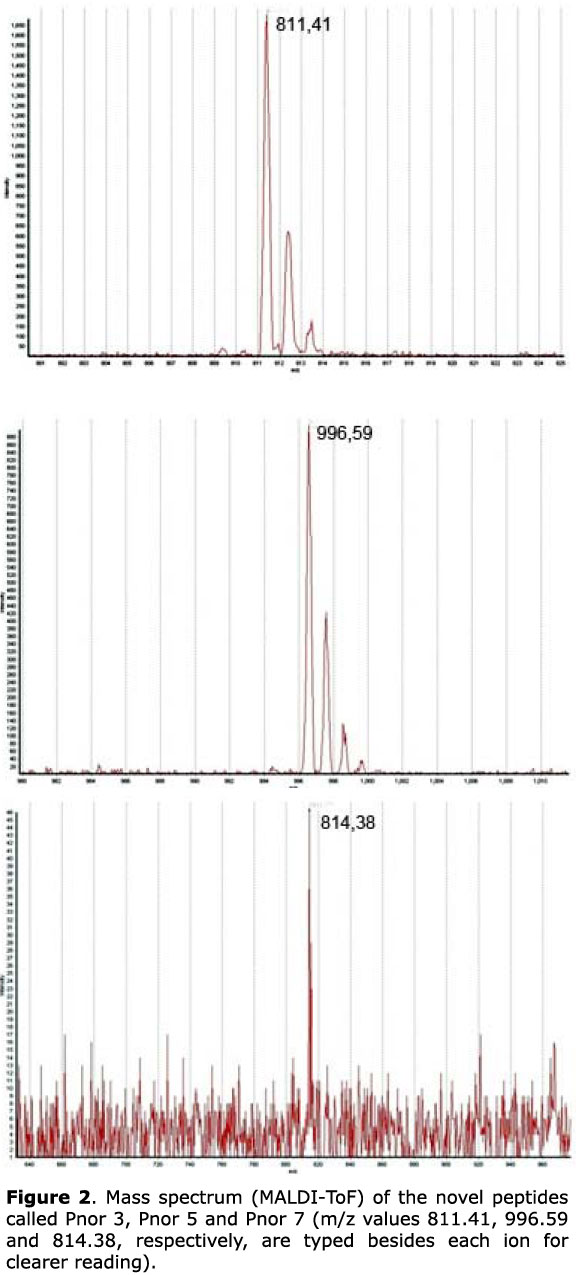
The lyophilized SPE-Phyllomedusa nordestina crude skin secretion was fractionated based only on peak shape and distribution, according to the analytical RP-HPLC profile presented in Figure 1. All pooled peptides were tested for vascular effects and those pools that were active had their molecular mass distribution profiled by mass spectrometry (Figure 2) (MALDI/TOF and/or ESI-Q-TOF). This work has focused solely on bioactive peptides ranging from 700 to 1100 Da; therefore, the arrowed peaks in Figure 1 were the only ones falling into this profile: small vasoactive peptides. These pools were subsequently submitted to de novo MS/MS sequencing.
De Novo Peptide Sequencing
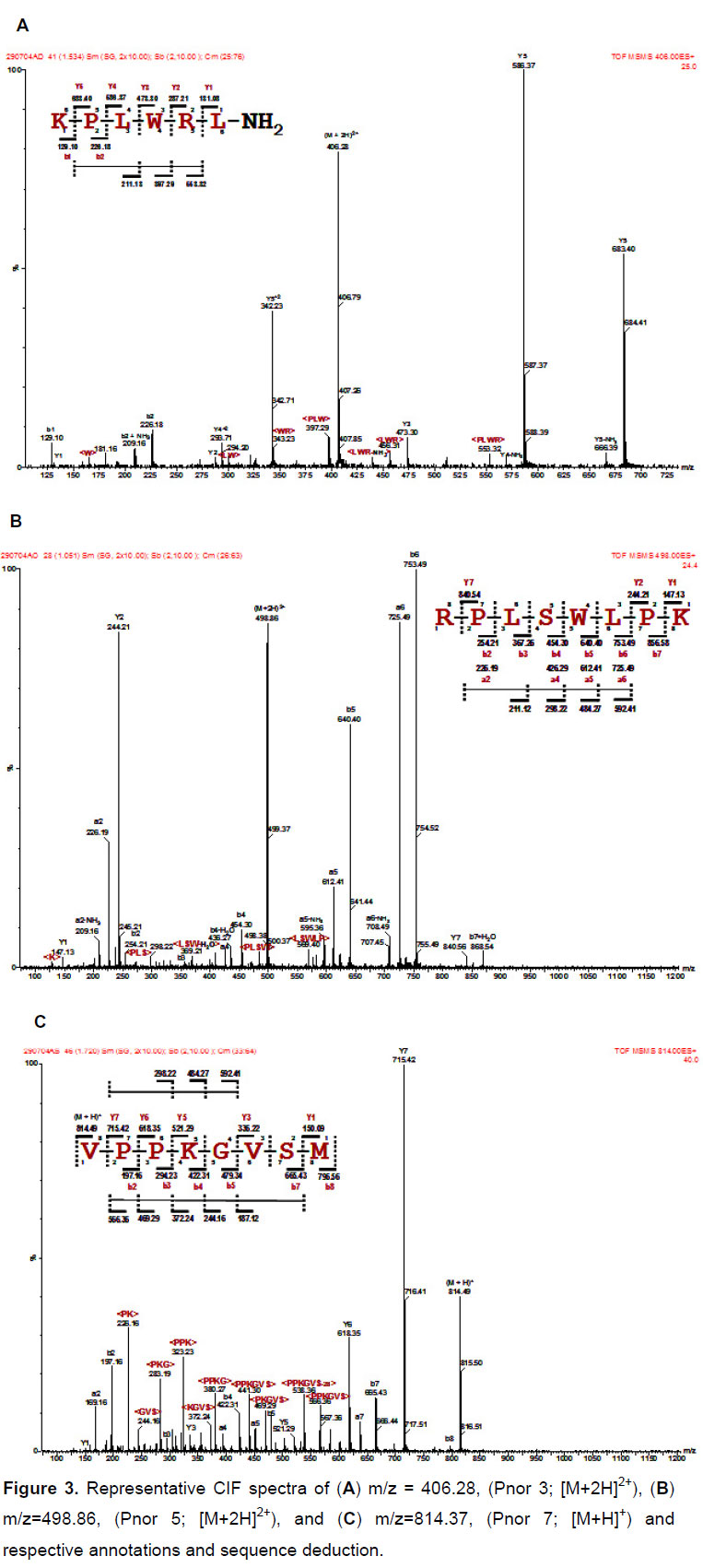
Those pools selected for de novo (or collision-induced fragmentation analysis by Q-TOF-MS/MS) sequencing were processed accordingly. Briefly, after cystein bridge reduction and alkylation by iodoacetamide, the obtained peptides were individually selected for MS/MS analysis and fragmented by collision with argon (CIF), yielding daughter ion spectra as presented in Figure 3. This routine was repeated until sufficient information regarding the peptide sequence was gathered. The MS/MS spectra were analyzed by the BioLynx software module of MassLynx 4.0 and manually verified for accuracy in the amino acid sequence interpretation. The active peptides that had their amino acid sequences deduced were: KPLWRL-NH2; RPLSWLPK and VPPKGVSM, denominated Pnor 3, 5 and 7 respectively. Table 1 shows that Pnor peptides align fairly well with bradykinin and other kinins from other amphibians.
Bioassay
Figure 4 presents the changes in diameter in groups of arterioles in response to local application of the peptides, over time. The topical administration of 10 nmol of Pnor 3 (Figure 4A) induced vasodilatation. An increase in the diameter of large arterioles was observed after 10 minutes, compared to the initial diameter (75 ± 10 µm and 37 ± 8 µm, respectively). The relative magnitude of arteriolar vasodilatation in response to the peptide was augmented after 20 and 30 minutes (130 ± 12 µm and 175 ± 10 µm, respectively).
Also, for the peptide Pnor 7 (Figure 4C), a significant vasodilatation was observed. Ten minutes after the topical administration of 10nmol Pnor 7, the arteriolar diameter doubled (Figure 4C; i) relative to the initial diameter (42 ± 5 µm and 87.4 ± 11 µm, respectively). The vasodilatation became more pronounced after 20 minutes (125 ± 18 µm) and remained so until 30 minutes of observation.
Interestingly, Figure 4B displays the changes in diameter of the arteriole groups in response to the local application of Pnor 5. As observed, the topical administration of 10nmol of Pnor 5 provoked an accentuated arteriolar constriction. This vasoconstriction activity was evident 10 minutes after the application, when compared to the initial diameter (26 ± 7 µm and 43 ± 9 µm, respectively). The relative magnitude of arteriolar constriction in response to the peptide had not been restored by the end of 30 minutes of observation (Figure 4B, ii).
No change in rolling leukocyte velocity or venule diameter was seen over time in either vehicle or peptide-treated animals.
DISCUSSION
This work reports the purification and characterization of three novel peptides denominated Pnor 3, Pnor 5 and Pnor 7 (after Phyllomedusa nordestina). Like bradykinin, Pnor 3 provokes a strong vasodilatation as observed by the microcirculatory network assay (Figure 4A). This same effect is also induced by the administration of Pnor 7 (Figure 4C). Interestingly, Pnor 5 was the opposite effect: it is a strong arteriolar constrictor, as found in the same assay (Figure 4B).
Vasoactive peptides and inflammatory molecules tend to coexist in venoms and secretions, whereas increased leukocyte adhesion can be an indicator of an inflamed and dysfunctional endothelium (24). Previous studies by our group reported that the skin secretion of P. hypochondrialis (although we now know that we were working with P. nordestina at the time) induces increased leukocyte rolling flux followed by a gradual increase in cells firmly adherent to the endothelium in intravital experiments (25). Taking our previous results together with the present findings (characterization of BRP), one can conclude that they corroborate the previously described local inflammatory effect of P. nordestina skin administration in mice. Moreover, these skin secretion components could play a role in the initial rolling and slowing of recruited leukocytes as well as the transition from slow rolling to firm adhesion. At present, the mechanisms involved in the local inflammatory process induced by P. nordestina skin secretion is one of the relevant questions related to the complex pathophysiology induced by toxins (peptides). Kinins are rapidly produced after tissue injury and play a role in many of the components of the inflammatory response such as vasodilatation, plasma extravasation, cell migration, pain and hyperalgesia (26-31).
Peptides described in this report were initially screened for their biological activity and then for their molecular mass directly from the SPE-processed crude skin secretion. As presented in Table 1, each of these peptides possesses at least one Pro residue calculated as consensus by Multalin (20). Not surprisingly, the calculated consensus nonapeptide based on all the inputted sequences possesses six bradykinin residues at the correct positions.
Although BK and BRP are not rarely found in amphibian skin secretions, we would like to call the attention to the point that not only Val1/Thr6-BK variations can be found on these secretions. However, we were able to identify Pnor7 (VPPKGVSM), apparently a Val1-BK analogue, that was isolated through its biological effect. Moreover, Pnor7 presents a Lys residue at position 4. This insertion per se is sufficient to speculate about a whole new family of peptides, since positions 1, 2, 3, 5 and 7 are conserved (Table 1) and the presence of such a reactive amino acid should not be considered as a random event.
Also noteworthy are the opposite effects observed for Pnor3 and Pnor5. Apparently using the same amino acids in a different order, these peptides vary from vasodilatation activity (Pnor3) to vasoconstrictor activity (Pnor5).
As Harris et al. (32) described, the presence of inflammatory cells in venules may have a major influence on arteriolar constriction. So, the arteriolar constriction observed, in which venular adherent leukocytes contributed to the constriction of paired arterioles, could be attenuated by the injection of a monoclonal antibody against the adhesion molecules (33). Therefore, as seen in the work of Conceição et al. (21) on Orpotrin, in our model no change in rolling leukocyte velocity or in diameter of venules was seen over time in Pnor5-treated animals, suggesting that Pnor5 exerts a selective and direct action on arterioles.
Some authors speculate that BK and BRP are present in the skin of these tree frogs as they lack the kallikrein-kinin system (34, 35), and/or that these peptides may act on and/or potentiate the endogenous BK action on the cardiovascular and/or gastrointestinal system of their predators, thus acting as defensive peptides (13, 11). If the latter hypothesis is correct, then the best defensive peptides would not be BK or BRP by themselves, but rather, a combination of kinins and a potentiator factor. In a previous study, our group have indeed found a BPP in the skin secretion of Phyllomedusa hypochondrialis, and it was the first BPP (bearing the canonical structural motif) to be discovered not only on the frog skin but also in any natural sources other than snake venoms (9).
Investigating nature's selected molecules is a fruitful source for identifying compounds aimed at very specific targets, depending mostly on the source of the material and the biological system chosen. The present approach, i.e. mass spectrometry amino acid sequence deduction after a single-step liquid chromatography, not only requires very little sample manipulation but also has proven itself to be fast, reliable and effective in identifying bioactive peptides. It is our understanding that the combination of mass spectrometry and the biological assay allows a rapid and reliable method for screening a large number of low-molecular-mass peptides present in skin secretions.
ACKNOWLEDGEMENTS
D. C. Pimenta is a CNPq fellow researcher (grant n. 302405/2008-9). Parts of this work were developed by D. C. Pimenta when working at CAT/CEPID, a FAPESP grant.
Received: May 15, 2009
Accepted: July 1, 2009
Abstract published online: July 7, 2009
Full paper published online: November 30, 2009
Conflicts of interest: There is no conflict.
Financial source: FAPESP, CAPES and CNPq.
- 1. Lazarus LH, Attila M. The toad, ugly and venomous, wears yet a precious jewel in his skin. Prog Neurobiol. 1993;41(4):473-507.
- 2. Daly JW. The chemistry of poisons in amphibian skin. Proc Natl Acad Sci USA. 1995;92(1):9-13.
- 3. Erspamer V. Bioactive secretions of the integument. In: Heatwole H, Barthalmus GT, editors. The integument, amphibian biology. vol. 1. Chipping Norton: Surrey Beatty & Sons; 1994. 179-350 p.
- 4. Zasloff M. Antimicrobial peptides of multicellular organisms. Nature. 2002;415:389-95.
- 5. Chen T, Zhou M, Gagliardo R, Walker B, Shaw C. Elements of the granular gland peptidome and transcriptome persist in air-dried skin of the South American orange-legged leaf frog, Phyllomedusa hypocondrialis Peptides. 2006;27(9):2129-2136.
- 6. Conceiçăo K, Konno, K, Richardson M, Antoniazzi MM, Jared C, Daffre S, Camargo ACM, Pimenta DC. Isolation and biochemical characterization of peptides presenting antimicrobial activity from the skin of Phyllomedusa hypochondrialis Peptides 2006;27(12):3092-9.
- 7. Brand GD, Krause FC, Silva LP, Leite JRSA, Melo JAT, Prates MV, Pesquero JB, Santos EL, Nakaie CR, Costa-Neto CM, Bloch C. Bradykinin-related peptides from Phyllomedusa hypochondrialis Peptides. 2006;27(9):2137-46.
- 8. Thompson AH, Bjourson AJ, Shaw C, McClean S. Bradykinin-related peptides from Phyllomedusa hypochondrialis azurea: Mass spectrometric structural characterization and cloning of precursor cDNAs. Rapid Commun Mass Spectrom. 2006;20(24):3780-8.
- 9. Conceiçăo K, Konno K, Lopes de Melo R, Antoniazzi MM, Jared C, Sciani JM, Conceiçăo IM, Prezoto BC, de Camargo ACM, Pimenta DC. Isolation and characterization of a novel bradykinin potentiating peptide (BPP) from the skin secretion of Phyllomedusa hypochondrialis Peptides. 2007; 28(3):515-23.
- 10. Conlon JM, Aronsson U. Multiple bradykinin-related peptides from the skin of the frog, Rana temporaria Peptides. 1997;18(3):361-5.
- 11. Li L, Bjourson AJ, He J, Cai G, Rao P, Shaw C. Bradykinins and their cDNA from piebald odorous frog, Odorrana schmackeri, skin. Peptides. 2003; 24(6):863-72.
- 12. Suzuki H, Iwamuro S, Ohnuma A, Coquet L, Leprince J, Jouenne T, Vaudry H, Taylor CK, Abel PW, Conlon JM. Expression of genes encoding antimicrobial and bradykinin-related peptides in skin of the stream brown frog Rana sakuraii Peptides. 2007;28(3):505-14.
- 13. Conlon JM. Bradykinin and its receptors in non-mammalian vertebrates. Regul Pept. 1999;79(2-3):71-81.
- 14. da Cruz CAG. Sobre as relaçőes intergenéricas de Phyllomedusinae da floresta atlantica (Amphibia, Anura, Hylidae). Rev Brasil Biol. 1990;50(3):709-26.
- 15. Frost DR. Amphibian species of the world: an online reference, version 5.3. Available in: http://research.amnh.org/herpetology/amphibian/index.html 2009.
- 16. Caramaschi U. Redefiniçăo do grupo de Phyllomedusa hypochondrialis, com redescriçăo de P. megacephala (Miranda-Ribeiro, 1926), revalidaçăo de P. azurea Cope, 1862 e descriçăo de uma nova espécie (Amphibia, Anura, Hylidae). Arq Mus Nac. 2006;64(2):159-79.
- 17. Daudin FM. Histoire naturelle des rainettes, des grenouilles et des crapauds: dédiée ŕ B.G.E.L. Lacépčdč. Paris: D I'lmprimerie de Bertrandet: Chez Leurault Librarie; 1802. 108 p.
- 18. Atherton E, Sheppard RC. Solid phase peptide synthesis: a practical approach. Oxford: IRL Press; 1989. 75-160 p.
- 19. Lomonte B, Lundgren L, Johansson B, Bagge U. The dynamics of local tissue damage induced by Bothrops asper snake venom and myotoxin II on the mouse cremaster muscle: an intravital and electron microscopic study. Toxicon. 1994;32(1):41-55.
- 20. Corpet F. Multiple sequence alignment with hierarchical clustering. Nucleic Acids Res. 1988;16(22):10881-90.
- 21. Conceiçăo K, Konno K, Melo RL, Marques EE, Hiruma-Lima CA, Lima C, Richardson M, Pimenta DC, Lopes-Ferreira M. Orpotrin: a novel vasoconstrictor peptide from the venom of the Brazilian stingray Potamotrygon gr. orbignyi Peptides. 2006;27(12):3039-46.
- 22. Rioli V, Gozzo FC, Heimann AS, Linardi A, Krieger JE, Shida CS, Almeida PC, Hyslop S, Eberlin MN, Ferro ES. Novel natural peptide substrates for endopeptidase 24.15, neurolysin, and angiotensin-converting enzyme. J Biol Chem. 2003;278(10):8547-55.
- 23. Piot JM, Zhao Q, Guillochon D, Ricart G, Thomas D. Isolation and characterization of two opioid peptides from a bovine hemoglobin peptic hydrolysate. Biochem Biophys Res Commun. 1992;189(1):101-10.
- 24. Ulbrich H, Eriksson EE, Lindbom L. Leukocyte and endothelial cell adhesion molecules as targets for therapeutic interventions in inflammatory disease. Trends Pharmacol Sci. 2003;24(12):640-7.
- 25. Conceiçăo K, Miriane Bruni F, Pareja-Santos A, Antoniazzi MM, Jared C, Lopes-Ferreira M, Lima C, Pimenta DC. Unusual profile of leukocyte recruitment in mice induced by a skin secretion of the tree frog Phyllomedusa hypochondrialis Toxicon. 2007;49(5):625-33.
- 26. Ahluwalia A, Perretti M. B1 receptors as a new inflammatory target. Could this B the 1? Trends Pharmacol Sci. 1999;20(3):100-4.
- 27. Calixto JB, Cabrini DA, Ferreira J, Campos MM. Kinins in pain and inflammation. Pain. 2000;87(1):1-5.
- 28. Couture R, Harrisson M, Vianna RM, Cloutier F. Kinin receptors in pain and inflammation. Eur J Pharmacol. 2001;429(1-3):161-76.
- 29. Dray A, Perkins M. Bradykinin and inflammatory pain. Trends Neurosci. 1993;16(3):99-104.
- 30. Dray A. Kinins and their receptors in hyperalgesia. Can J Physiol Pharmacol. 1997;75(6):704-12.
- 31. Perkins MN, Campbell E, Dray A. Antinociceptive activity of the bradykinin B1 and B2 receptor antagonists, des-Arg9, [Leu8]-BK and HOE 140, in two models of persistent hyperalgesia in the rat. Pain. 1993;53(2):191-7.
- 32. Harris NR, Whatley JR, Carter PR, Specian RD. Venular constriction of submucosal arterioles induced by dextran sodium sulfate. Inflamm Bowel Dis. 2005;11(9):806-13.
- 33. Zamboni WA, Roth AC, Russel RC, Graham B, Suchy H, Kucan JO. Morphologic analysis of the microcirculation during reperfusion of ischemic skeletal muscle and the effect of hyperbaric oxygen. Plast Reconstr Surg. 1993;91(6):1110-23.
- 34. Rabito SF, Binia A, Segovia R. Plasma kininogen content of toads, fowl and reptiles. Comp Biochem Physiol A Comp Physiol. 1972;41(2):281-4.
- 35. Seki T, Miwa I, Nakajima T, Erdos EG. Kallikreinkinin system in nonmammalian blood: evolutionary aspects. Am J Physiol. 1973; 224(6):1425-30.
Publication Dates
-
Publication in this collection
27 Nov 2009 -
Date of issue
2009
History
-
Accepted
30 Nov 2009 -
Reviewed
01 July 2009 -
Received
15 May 2009