Abstract
Leptospirosis is a worldwide infection that affects many species, including wild animals. The present study aimed to detect Leptospira spp. antibodies in 17 captive coatis (Nasua nasua) by the microscopic agglutination test (MAT). Nine (52.94%) animals tested positive and the following serovars were identified: Copenhageni (22.22%), Shermani (22.22%), Andamana (11.11%), Wolfii (11.11%) and Pyrogenes (11.11%). Two samples presented coagglutination, one (11.11%) for Hebdomadis and Wolfii, and another (11.11%) for Hebdomadis, Hardjo and Wolfii. The current study revealed the presence of the infection even in animals without any clinical signs, reinforcing the possibility that wild animals in captivity may be infected by leptospire serovars, thus enabling reservoirs.
Nasua nasua; Leptospira spp.; captive coatis; antibodies; microscopic agglutination test
Leptospira spp. antibodies in captive coatis (Nasua nasua Storr, 1780) (Carnivora: Procyonidae)
Langoni HI; Kawaguchi MFI; Oshika JCII; Da Silva RCI; Teixeira CRII
IZoonosis Research Center, NUPEZO, Veterinary Medicine and Animal Husbandry School, São Paulo State University, UNESP, Botucatu, São Paulo State, Brazil
IIDepartment of Veterinary Surgery and Anesthesiology, Veterinary Medicine and Animal Husbandry School, São Paulo State University, UNESP, Botucatu, São Paulo State, Brazil
Correspondence to Correspondence to: Heliio Langoni Departamento de Higiene Veterinária e Saúde Pública Faculdade de Medicina Veterinária e Zootecnia, UNESP Distrito de Rubião Jr, s/n, Botucatu, SP, 18618-000, Brasil. Phone: + 55 14 38116270. Fax: + 55 14 38116075. Email: hlangoni@fmvz.unesp.br.
ABSTRACT
Leptospirosis is a worldwide infection that affects many species, including wild animals. The present study aimed to detect Leptospira spp. antibodies in 17 captive coatis (Nasua nasua) by the microscopic agglutination test (MAT). Nine (52.94%) animals tested positive and the following serovars were identified: Copenhageni (22.22%), Shermani (22.22%), Andamana (11.11%), Wolfii (11.11%) and Pyrogenes (11.11%). Two samples presented coagglutination, one (11.11%) for Hebdomadis and Wolfii, and another (11.11%) for Hebdomadis, Hardjo and Wolfii. The current study revealed the presence of the infection even in animals without any clinical signs, reinforcing the possibility that wild animals in captivity may be infected by leptospire serovars, thus enabling reservoirs.
Keywords:Nasua nasua, Leptospira spp., captive coatis, antibodies, microscopic agglutination test.
INTRODUCTION
In South America, the family Procyonidae, order Carnivora, is represented by 18 species distributed among six genera, including Nasua spp. In the rest of the world, coatis thrive in different environments, such as deciduous and tropical forests, deserts and chaparral regions. Although these animals are classified as carnivores, they may also feed on fruits (1).
Leptospirosis, a zoonosis present throughout the globe, is naturally transmitted between humans and other animals (2). The etiologic agent responsible for the disease is a spirochete, Sphirochaetales order, Leptospiraceae family, that presents three genera: Leptospira, Leptonema and Turneria. Currently, leptospires are classified into more than 250 serovars. This disease assumes an important epidemiological role because it affects domestic and wild species. Some of them develop subclinical infection and maintain the agent in their kidneys with intermittent elimination through urine, constituting a serious public health problem (3).
Serological studies based on leptospirosis had been performed in many wild species, including coatis (Nasua nasua) (4). In some places, such as zoos and parks, several species must live in restricted areas. This condition can disseminate numerous infectious agents that may cause a wide variety of zoonotic diseases (5).
In order to ascertain important epidemiological aspects of the disease in wild animals, this study aimed to verify the occurrence of Leptospira spp. antibodies in captive coatis and to establish sanitary measures for its control.
Serum samples were collected from 17 captive coatis (Nasua nasua Storr, 1780) in three cities of São Paulo state, Brazil, namely Sorocaba (23º30'06''S, 47º27'29''W), Botucatu (22º53'09''S, 48º26'42''W) and São José dos Campos (23º10'46''S, 45º53'13''W). The study was approved by the Ethics Committee on Animal Experimentation of the Veterinary Medicine and Animal Husbandry School, UNESP, Botucatu, SP, Brazil. They were randomly selected, regardless of age or gender, since they were all healthy. Animals were anesthetized in their enclosures by means of an intramuscular injection with tiletamine-zolazepam (7 mg/kg Zoletil®, Virbac, Brazil) and xylazine (0.5 mg/kg Sedazine®, Fort Dodge, Brazil) employed in pre-anesthesia and induction of anesthesia, respectively.
Subsequently, 3-mL samples of intracardiac blood were collected from each animal and placed in a test tube without anticoagulant. Samples were centrifuged at 1,200 X g for ten minutes and then stored at 80ºC. Afterward, they were submitted to the microscopic agglutination test (MAT), using the following serovars as antigens: Australis, Bratislava, Autumnalis, Butembo, Castellonis, Bataviae, Canicola, Whitcombi, Cynopteri, Djasiman, Sentot, Gryppotyphosa, Hebdomadis, Copenhageni, Icterohaemorraghiae, Javanica, Panama, Pomona, Pyrogenes, Hardjo, Hardjo-prajitno, Hardjo-miniswajezak, Hardjo-C.T.G., Hardjo-bovis, Wolffi, Shermani, Tarassovi, Andamana and Patoc (6, 7). Agglutination of more than 50% leptospires per field was considered to be positive reaction.
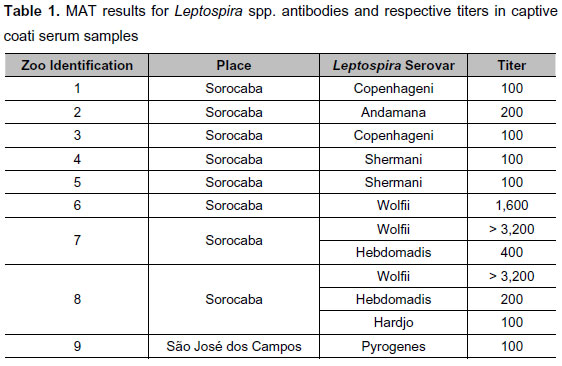
Antibodies were detected in nine of 17 (52.94%) serum samples; the results, presented in Table 1, show their identification and the specific serovars with the respective final titer. Serovar titers varied from 100 to more than 3,200.
Table 2 presents the relation among the number of samples from each zoo, seropositive samples and the prevalence of each serovar. There were eight out of 17 (61.54%) positive results from Sorocaba that reacted against six different serovars. Only one sample from São José dos Campos tested positive. From Botucatu, none of the three samples reacted.
Wolfii serovar was most frequently detected and with higher titers than the other serovars, ranging from 1,600 to more than 3,200. This serovar most commonly affects humans and other animals in urban areas of Brazil (8).
Although some studies on leptospirosis in feral animals have been carried out in the Americas, Brazil has produced few works on this topic. Therefore, more epidemiological leptospirosis analyses should be performed to develop strategies to control the disease in regions with high density of animals, forest and rivers.
There is little information about the prevalence of leptospirosis in captured animals (9, 10). In captivity, animals of different ecological origins and epidemiological backgrounds are forced to live in close proximity. This may create an opportunity for the dissemination of infectious agents. Hence, zoological gardens and parks can potentially be sources of a wide range of zoonotic diseases (5).
Lins and Lopes (11) reported a leptospiral isolate from a coati (N. nasua) whereas Roth (12) found many serovars present in a racoon (Procyon lotor) in the USA, reinforcing the importance of these species in the leptospirosis transmission chain.
A study involving seven coatis from a Rio de Janeiro zoo, Rio de Janeiro state, was performed using MAT for antibodies against Leptospira spp. Three of them reacted with Icterohemorrhagiae serovar, which characterizes the high suscebility of captive animals, compared with free-living wild animals, to infection by any serovar of Leptospira spp. This corroborates the findings of Girio et al. (4), who did not find any reactive animal among nine Brazilian free-living coatis (13). However, these animals could be infected without any humoral immune response, rendering them reservoirs of the bacterium.
Leptospira spp. infection may occur in Brazilian captive coatis without clinical signs, but with measurable antibodies. Most of the studied animals can be reservoirs of this bacterium and thus require serological tests for leptopsirosis and other zoonoses to ascertain the risks to the environment, park visitors and workers, and its impact on the public health.
ACKNOWLEDGEMENTS
We thank The State of São Paulo Research Foundation, FAPESP, for the scientific grant, process n. 2005/02621-6; the Ethics Committee on Animal Experimentation of the Veterinary Medicine and Animal Husbandry School, UNESP; and the animal institutions for the coatis used in the study.
Received: February 23, 2009
Accepted: July 6, 2009
Abstract published online: July 8, 2009
Full paper published online: November 30, 2009
Conflicts of interest: There is no conflict.
Financial source: FAPESP, grant 2005/02621-6.
- 1. Beisiegel BM. Notes on the coati, Nasua nasua (Carnivora: Procyonidae) in an Atlantic forest area. Braz J Biol. 2001;61(4):689-92.
- 2. Acha PN, Szyfres B. Zoonosis y enfermedades transmisibles comunes al hombre y a los animales: bacteriosis y micosis. 3rd ed. Washington: Organización Panamericana de la Salud; 2003. 480 p. 1 vol.
- 3. Corręa WM, Corręa CNM. Toxoplasmose. In: Corręa WM, Corręa CNM. Enfermidades infecciosas dos mamíferos domésticos. Rio de Janeiro: Medsi; 1992. p. 757-66.
- 4. Girio RJS, Pereira FLG, Marchiori filho M, Mathias LA, Herreira RCP, Alessi AC, et al. Investigation of antibodies to Leptospira spp. in wild and feral animals from the region of Nhecolandia, Mato Grosso do Sul, Brazil. Use of the immunohistochemistry technique for the agent detection. Cienc Rural. 2004;34(1):165-9.
- 5. Siemering H, Zoonose. In: Fowler ME, Miller RE, editors. Zoo and wild animal medicine. Califórnia: W. B. Saunders Co.; 1993. p. 64-8.
- 6. Brasil. Ministério da Saúde. Fundaçăo Nacional de Saúde. Centro Nacional de Epidemiologia. Coordenaçăo de Controle de Zoonoses e Animais Peçonhentos, 1995. Manual de Leptospirose. 2nd ed. Brasília: Fundaçăo Nacional de Saúde; 1995. 98 p.
- 7. Langoni H, da Silva AV, Pezerico SB, de Lima VY. Anti-leptospire agglutinins in equine sera, from Săo Paulo, Goiás and Mato Grosso do Sul, Brazil, 1996-2001. J Venom Anim Toxins incl Trop Dis. 2004;10(3):207-18.
- 8. Pereira MM, Korver H, Mazzonelli JM, Andrade J, Moraes G. Search for leptospires and specific antibodies in wild animals trapped in a periurban area of Rio de Janeiro, Brazil. In: Kobayashi Y, editor. Leptospirosis. Matsuyama: Proceedings of the Leptospirosis Research Conference; 1991.
- 9. Reid HAC, Herron AJ, Hines ME, Orchard EA, Altman NH. Leptospirosis in a white-lipped tamarin (Sanguinus labiatus). Lab Anim Sci. 1993;43(1):258-9.
- 10. Luna Alvarez M, Morales Cervantes LP, Torres Barranca JI, Gual Sill F. Investigación serológica de leptospirosis en fauna silvestre mantenida en cautiverio en el zoológico de Chapultepec de la ciudad de México. Vet Mex. 1996;27(3):229-34.
- 11. Lins ZC, Lopes ML. Isolation of Leptospira from wild forest animals in Amazonian Brazil. Trans R Soc Trop Med Hyg. 1984;78(1):124-6.
- 12. Roth EE. Leptospirosis in wildlife in the United States. Scient Proc Am Vet Med Assoc. 1964;101:211-8.
- 13. Lilenbaum W, Monteiro RV, Ristow P, Fraguas S, Cardoso VS, Fedullo LPL. Leptospirosis antibodies in mammals from Rio de Janeiro Zoo, Brazil. Res Vet Sci. 2002;73(3):319-21.
Publication Dates
-
Publication in this collection
27 Nov 2009 -
Date of issue
2009
History
-
Accepted
30 Nov 2009 -
Reviewed
06 July 2009 -
Received
23 Feb 2009