Abstract
Wasp venom is mixture of complex proteins that have several physical and pharmacological properties. The photochemical detoxification of Vespa orientalis venom is expected to generate photooxidized venom sac extract (PVSE). Antigenically active PVSE is obtained by exposing the venom sac extract (VSE) of Vespa orientalis to ultraviolet radiation in the presence of methylene blue. The aim of the present work was to evaluate the effect of PVSE on learning and memory of rats. Detoxification of PVSE was evident since treated mice had longer survival time than the group of mice treated with VSE. Photooxidized VSE of V. orientalis revealed enhancement on learning and memory by shortening the time to reach food (TRF) in T-maze. In a 28-day study with rats, we observed that PVSE significantly decreased transfer latency (TL) in elevated plus maze (EPM), significantly increased step down latency (SDL), diminished step down errors (SDE) and time spent in shock zone (TSS) in step down avoidance test. Thus, we concluded that although there is a possibility of employing PVSE in the treatment of Alzheimer, dementia or neurodegenerative illness as a non-herbal and non-synthetic alternative for patients who do not respond to available therapy, further investigation is still required.
Vespa venom; photochemical processes; learning; memory; maze learning
ORIGINAL PAPERS
Evaluation of effects of photooxidized Vespa orientalis venom on memory and learning in rats
Mukund HI; Gawade SPII
IDepartment of Pharmacology, PES College of Pharmacy, Bangalore, Karnataka, India
IISVERI's College of Pharmacy, Sholapur, Maharashtra, India
Correspondence to Correspondence to: Mukund Handral Department of Pharmacology PES College of Pharmacy Hanumanthanagar, 50 feet road Bangalore, 560050, India Phone: +91 8026600741. Fax: +91 8026506928 Email: Mukund_handral@yahoo.com.
ABSTRACT
Wasp venom is mixture of complex proteins that have several physical and pharmacological properties. The photochemical detoxification of Vespa orientalis venom is expected to generate photooxidized venom sac extract (PVSE). Antigenically active PVSE is obtained by exposing the venom sac extract (VSE) of Vespa orientalis to ultraviolet radiation in the presence of methylene blue. The aim of the present work was to evaluate the effect of PVSE on learning and memory of rats. Detoxification of PVSE was evident since treated mice had longer survival time than the group of mice treated with VSE. Photooxidized VSE of V. orientalis revealed enhancement on learning and memory by shortening the time to reach food (TRF) in T-maze. In a 28-day study with rats, we observed that PVSE significantly decreased transfer latency (TL) in elevated plus maze (EPM), significantly increased step down latency (SDL), diminished step down errors (SDE) and time spent in shock zone (TSS) in step down avoidance test. Thus, we concluded that although there is a possibility of employing PVSE in the treatment of Alzheimer, dementia or neurodegenerative illness as a non-herbal and non-synthetic alternative for patients who do not respond to available therapy, further investigation is still required.
Key words:Vespa venom, photochemical processes, learning, memory, maze learning.
INTRODUCTION
The venoms of arthropods have attracted considerable interest as potential sources of pharmacological substances. Over the past decades, studies have focused on bioactive compounds present in wasp venoms. These compounds include amines, small peptides, and high and low molecular weight proteins such as enzymes, allergens and toxins (1). Vespa orientalis is one of the species of vespine found in most parts of India. The venom of Vespa orientalis (Vo) is a mixture of proteins and chemical constituents with several physical and pharmacological properties produced by the venom gland and injected into prey to paralyze it or employed for defense purposes. The reactions produced by Vespa orientalis stings range from severe pain, local irritation and urticarial papules to serious symptoms. The chemical constituents present in this venom are acetylcholine, serotonin, adrenaline, nor-adrenaline, dopamine, kinins, and high molecular weight compounds such as phospholipase-A2; hyaluronidase; histidine decarboxylase; acid, alkaline, and neutral DNAse; poly and disaccharidase; and several polycationic peptide and protein that act together to produce biological effects (2).
Previous studies demonstrated that photooxidation of snake venoms by exposure to ultraviolet (UV) radiation at different time intervals in the presence of sensitizer methylene blue resulted in detoxification of venom, and led to significant changes with no loss of antigenicity (3). Photooxidized Echis carinatus venom demonstrated antidepressant and central nervous system (CNS) stimulant activities, and enhancement of memory and learning in rats (4, 5).
Hornet envenomation caused specific CNS effects in dogs, cats, and mice, which suggests that anticholinesterase-like components are present in basic venom fractions (6). Numerous studies have been carried out to test the properties of wasp venom as a pharmacological agent. However, no attempts have been made to investigate the effects of photooxidized venom sac extract (PVSE) of Vo on cognition and dementia in animal models as an alternative mechanism that may create lead compounds for the treatment of neurodegenerative diseases. Thus, the present work aimed to evaluate the psychopharmacological effects of PVSE using suitable animal models for memory and learning.
MATERIAL AND METHODS
Animals
Male albino Wistar mice (20 -25 g) were used in toxicity studies, male albino Wistar rats (180-220 g) were employed in psychopharmacological studies, and albino rabbits (2-2.5 kg) were utilized in hyperimmunization studies. The Institutional Animal Ethics Committee (IAEC) of Al-Ameen College of Pharmacy, Bangalore, India, approved all animal experiments. All procedures were conducted in accordance with the guidelines of the Committee for the Purpose of Control and Supervision on Experiments on Animals (CPCSEA), Government of India, under recommended temperature and relative humidity.
Drugs and Chemicals
Piracetam was received as a gift sample from UCB India Private Ltd. (India), Freund's complete and incomplete adjuvants were purchased from Genie Bioscience (India), diethylaminoethyl cellulose, methylene blue (MB - Qualigens Fine Chem Ltd, India) and all other chemicals used in the study were of analytical grade.
Venom Collection
About 3,190 Vespa orientalis wasps were collected from the Belavanki, India, during summer season (March to June) and were authenticated by the entomologist Dr. Chandrashekhar, University of Agricultural Sciences, Bangalore, India. A specimen wasp is maintained in the Department of Entomology.
Preparation of Venom Sac Extract
The preparation of venom sac extract followed the procedure suggested by Friedman et al. (7) with some modifications. The collected wasps were immediately frozen at -20°C for 20 minutes. From the frozen wasps, the stinger apparatus at the tip of abdomen was gently pulled with help of fine forceps. Along with stinger a small white colored venom gland about 0.5 to 1 mm was collected in a tube containing ice-cold saline. The whole content was pooled, homogenized for ten minutes followed by centrifugation at 10,000 g for 15 minutes at -20°C. The supernatant was lyophilized and stored in desiccators under reduced pressure for further use. The total weight of the lyophilized VSE was 5.312 g. From each venom gland 1.6 mg of VSE was obtained.
Determination of Minimum Lethal Dose
Groups of three mice each were injected (via intraperitoneal and intravenous routes) with doses ranging from 75-85 mg/kg and 70-85 mg/ kg of VSE dissolved in phosphate buffer. Control group animals were treated with phosphate buffer only. Following treatment with VSE, the signs of toxicity and behavioral changes were observed 1, 2, 4, 24 and 72 hours after the administration (8).
Hyperimmunization in Rabbits
Three rabbits weighing 2.5 kg each were immunized with 5 mg of VSE diluted with 5 mL of phosphate buffer, and mixed with equal volume of complete and incomplete Freund's adjuvant (Genie Bioscience, India). Five-milliliter doses of the resulting emulsion were inoculated intradermally at 16 sites along the animals' spine. Two weeks later, the animals were boosted with 7 mg VSE in 1 mL of phosphate buffer via intraperitoneal (IP) route. The same procedure was repeated twice, three and four weeks later; then animals were bled and their sera were pooled. During the immunization schedule, a sample serum was continuously applied on to the immunodiffusion gel for appearance of precipitin lines (9).
Isolation of Immunoglobulin from Antisera
The isolation of immunoglobulin (IgE) from rabbit antisera was carried out by ion-exchange method using diethylaminoethyl cellulose (DEAE) (10). The purified immunoglobulins were lyophilized at -10°C and stored under reduced pressure until dried.
Photooxidation of VSE of Vespa orientalis
The reaction mixture containing 2 mL of VSE (25 mg in 2 mL of 0.05 M phosphate buffer at pH 6.8) and 2 mL of methylene blue (0.003% w/v in phosphate buffer at pH 6.8) solution was kept on a magnetic stirrer in a photooxidation chamber exposed to UV light (tubular ultraviolet 15 W lamp, 615, T8, Philips, Holland, UV output 4.8 W, 49 µW/ cm2) at about 10-cm distance and gently stirred in different time intervals (15, 30, 45, and 60 minutes) at 37°C. Then, 200 µL of activated charcoal (1% w/v in phosphate buffer at pH 6.8) was added to it, the mixture was stirred five more minutes without light exposure. The photooxidized venom sac extract (PVSE) of V. orientalis venom was filtered through a 0.2-µm filter using a syringe filtering unit (Minisart®, Sartorius, India). Similarly, the absorbance of the control mixture (2 mL of VSE and MB without UV exposure maintained in similar conditions) was measured separately at 200-400 nm using Shimadzu spectrophotometer (Japan) (4).
Total Protein Estimation
Hartree (11) method was used to estimate protein concentration of both VSE and PVSE using BSA as a standard.
Antigenicity Test
The antigenicity test of PVSE submitted to different UV time exposure was carried out using immunogel diffusion method, in which antigens and antibodies migrate through an agarose gel and react, resulting precipitin lines (12).
In vitro Neutralization Studies of Immunoglobulins
A constant amount of VSE (1 MLD) was allowed to react with 1% and 0.1% dilutions of isolated immunoglobulin and the mixtures were incubated at 37°C for 30 minutes. The protection or death (if any), was determined after intraperitoneal injection (0.2 mL) into mice (n = 3) and observed for seven days. (13)
Detoxification Test in Mice
The detoxification of PVSE exposed to UV light for 45 minutes (which preserved antigencity as tested out by immunogel diffusion method) was intraperitoneally injected into a group of mice (n = 3) at a dose based on body weight (0.1 mL/25 g or 25 mg/mL) and observed up to seven days (4).
Psychopharmacological Studies Learning and memory in T-maze
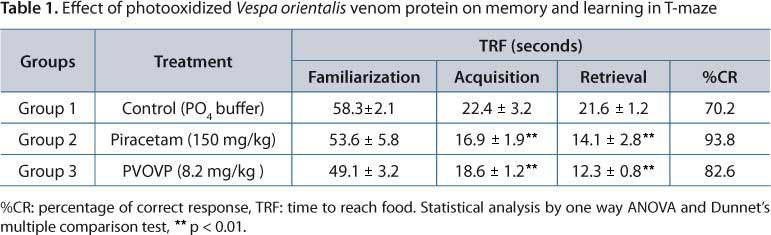
T-maze was used to evaluate spatial working memory in rats. The apparatus is made up of Perspex glass, has a starting arm box (86.5 x 18 cm) and two right and left arms (62 x 18 cm) and the whole maze was placed at the height of 87 cm from the ground. The food pellets were kept at both ends of the arm. Three groups of six rats were used for the experiment. Group 1 (control) received phosphate buffer for 28 days; Group 2 (standard) received piracetam (150 mg/kg IP) for 28 days; Group 3 (test) PVSE (8.2 mg/kg IP) for 28 days.
Training of the animals was carried out for three consecutive days. On the first day, each rat placed on the maze completed 15 trials to become familiarized with the maze and access food. Then, animals were deprived of food for 24 hours. On the second day, 60 minutes before experiment, each group was treated as describe above. Rats were trained until they attained nine correct arm choices out of 10 consecutive trials. The number of correct responses and time needed to reach the food (TRF) in each trial were recorded. The percentage of correct responses (%CR) was calculated. TL was recorded on completion of the fourth week of treatment (14).
Transfer latency in elevated plus maze (EPM)
The EPM is made up of black painted wooden with two open arms (47 x 14 cm) and two closed arms with a wall (47 x 14 x 37 cm) and is placed 47 cm above the ground. Three groups of six rats were used for the experiment. Group 1 (control) received phosphate buffer for 28 days; Group 2 (standard) received piracetam (150 mg/kg IP) for 28 days; Group 3 (test) received PVSE (8.2 mg/kg IP) for 28 days. The rats were placed individually at end of the open arm facing away from the central platform. The time taken to enter any closed arm with all four paws was taken as transfer latency (TL). On the first day, rats were allowed to explore the maze for five minutes and TL was recorded (acquisition) and on the second day, 60 minutes prior to experiment, animals were treated as described above and retention of the learned task was examined. TL was recorded on completion of the fourth week of treatment (15).
Step down passive avoidance
The apparatus consisted of a rectangular wooden box (65 x 45 x 40 cm) with one wall of Plexiglas glass and an electrified grid floor. It also contained a wooden platform (shock free zone) at the center of the grid floor. The box was illuminated with a 15 W bulb during the experiment. Three groups of six rats were used for the experiment. Group 1 (control) received phosphate buffer for 28 days; Group 2 (standard) received piracetam (150 mg/kg IP) for 28 days; Group 3 (test) PVSE (8.2 mg/kg IP) for 28 days. On the first day, each rat was placed on the shock free zone, when it stepped down and placed all four paws on the grid floor, an electric shock was delivered (1 mA for 5 seconds). Rats were given 3 to 5 trials until the time spent in the shock free zone was 120 seconds, the step down latency (SDL) and time spent in the shock zone (TSS) were recorded (familiarization). Twenty four hours later, acquisition phase was recorded again. The animals were given three trials and parameters such as SDL, step down error (SDE) and time spent in shock zone were recorded. Retention was tested on the third day and all the parameters were recorded again. The drugs were administered next day of familiarization and one hour before the acquisition phase on 28th day and recorded (16).
RESULTS
About 5.212 g of lyophilized VSE was obtained from 3190 wasps Vespa orientalis with an average of 1.5 mg VSE/sac.
Determination of MLD
Administration of a 82 mg/kg VSE dose (IP) into mice showed characteristic symptoms such as dyspnea, akinesis, diarrhea, hematuria, stupor, loss righting reflex, hyperexcitation to external stimuli and death. The average time of death was 1.47 ± 0.3 hour, hence the dose of 82 mg/kg (1 MLD) was chosen for photooxidation studies to generate antigenically active detoxified PVSE.
Preparation of Immunoglobulin
The preparation of immunoglobulins against VSE was essential to determine the duration of photooxidation required to generate antigenically active detoxified venom. Immunization of rabbit with VSE emulsified with complete and incomplete Freund's adjuvant for six weeks did not confer to the sera any protection against the lethal effect of venom. Sample bleeding resulted in development of prominent precipitin lines in immunogel diffusion experiment. Isolation of immunoglobulin from rabbit antisera was carried out by ion exchange using DEAE.
Photooxidation of VSE of Vespa orientalis
Photooxidation of venom protein was previously standardized by exposed reaction mixture to UV radiation for 15, 30, 45, and 60 minutes. Antigenicity was clearly visible after 45 minutes of exposure to UV radiation, therefore this mixture was used to generate antigenically active PVSE. The UV spectrum of VSE was biphasic with maximum optical density of 1.034 at 265 nm and 3.432 at 214 nm. The UV spectrum of PVSE, the maximum wavelength (Λmax) shifted from 265 nm to 265.5 nm. The absorbance increased from 1.034 to 3.424 at 265.5 nm (data not shown).
Total Protein Estimation
Hartree (11) method was used to estimate protein concentration of VSE and PVSE using BSA as standard. A 13.5% reduction in total protein concentration was observed when VSE was photooxidized. The reduction was from 69.95% to 57.30% indicating alteration in protein concentration.
Antigencity Test
Prominent precipitin lines were showed after 45 minutes of exposure, indicating the retention of antigencity of the venom (Figure 1 - C), where precipitin lines were as reduced in intensity and number as the exposure time increased. The lines were not visible after 60 minutes of exposure.
In vitro Neutralization Studies of Purified Immunoglobulins
In vitro neutralization studies were carried out according to method of Pramanik and Banerjee (17). One MLD of VSE was incubated with 0.1% and 1% of purified immunoglobulins. One percent of Ig was able to neutralize 1 MLD of VSE and protected the mice up to seven days.
Detoxification Test in Mice
Forty-five minute UV-exposed PVSE sample did not cause death during seven days of observation period, but provoked some symptoms of CNS stimulant manifestations, circular movements and hyperexcitation to external stimuli. The same concentration of VSE injected in another group of mice exhibited continuous restlessness, respiratory abnormality, immobility, urination, defecation, dyspnea, and all animals died within 1.45 ± 0.3 hour. Based on antigencity and toxicity tests, 45-minute UV-exposed venom product was considered photooxidized venom sac extract (PVSE) of Vo venom, and it was used to study psychopharmacological activities.
Psychopharmacological Studies Learning and memory in T-maze
On the day of acquisition process, the TRF was significantly (p < 0.01) shortened in piracetam and PVSE treated groups by 24.5% (16.9 ± 1.9 seconds) and 16.9% (28.6 ± 0.8 seconds) respectively when compared with control (22.4 ± 1.2). In retrieval phase, the TRF was significantly decreased (p < 0.01) both for piracetam and PVSE treated animals. The percentage reduction in TRF was 34% (14.1 ± 2.8) for piracetam whereas in PVSE it was 43% (12.3 ± 0.8) when compared to control (21.6 ± 1.2). A significant improvement was observed during the retrieval phase in PVSE treated animals (p < 0.01) with increase in the correct response by 33.6% (17.4% in piracetam) as compared to control (Table 1).
Transfer latency in elevated plus maze
Animals treated with piracetam and PVSE showed a significant (p < 0.01) reduction in transfer latency in acquisition phase on first day. The percentage of reduction in TL was by 30.6% (21.3 ± 0.56) for piracetam and 36.1% (19.6 ± 0.33) for PVSE compared to control (30.7 ± 0.33). In retrieval phase, the TL was significantly reduced (p < 0.01) in both treated groups by 35.6% and 33.7% when compared to control animals. The detailed results are shown in Table 2.
Step down passive avoidance test
In step down passive avoidance test, piracetam showed significant (p < 0.01) increase in SDL by 57.3% (120.6 ± 0.8 seconds) and PVSE (p < 0.01) by 53.6% (110.9 ± 0.8) when compared to controls (51.4 ± 0.62) in acquisition phase. The effect of SDE was significantly decreased (p < 0.01) both in piracetam (80%) and in PVSE (69.3%) treated animals and also a showed a significant decrease (p < 0.01) in time spent in shock free zone compared to control (77.5 and 73.2%).
In retrieval phase, both piracetam and PVSE showed significant increase (p < 0.01) in SDL by 50.2% and 53.3% compared to controls. The effect of SDE was significantly (p < 0.01) decreased in piracetam and PVSE by 82.8% and 74.2% compared to controls. However, TSS was significantly decreased (p < 0.01) by 81.9% and 79.1% in both piracetam and PVSE treated animals compared control animals (Table 3).
DISCUSSION
All wasps belong to family Vespidae of order Hymenoptera. Wasp sting is painful to humans; it may also cause allergic reactions that are fatal in severe cases, and provokes anaphylactic shock (17). However, wasp venom toxicity varies among hornet species. In the present study, administration of VSE of Vo venom to mice caused restlessness, respiratory abnormality, immobility, urination, defecation, dyspnea, and death. Although there are many reported methods for venom collection, in the present study we obtained 1.5 mg of VSE/sac by method of milking and sac extraction (18).
The reported MLD of pure lyophilized venom was 2.5 mg/kg whereas VSE was 70 mg/kg in mice. We have obtained 82 mg/kg. Probably because the venom was obtained in crude form and due to the presence other non-protein substances in VSE, its MLD was increased. The antivenom used against Vo venom was observed to have low antigenic activity (9). Due to poor antigenicity, Freund's adjuvants were employed to increase the titer of antivenom. In the present study, we used Freund's complete and incomplete adjuvants to achieve high titer immunoglobulins. The isolated immunoglobulins obtained by ion-exchange chromatographic method were tested against 1 MLD VSE and it was found that 1% antivenom was sufficient to neutralize 1 MLD in mice.
The effects of wasp stings are provoked by the protein components (low- and high-molecular weight proteins) secreted by the venom gland (2). Different toxic components have been isolated in different laboratories and their N-terminal sequences of toxic components were analyzed (19, 20). It has been observed that the toxic components of Vespa orientalis and other vespid toxins have similar amino acid sequence.
The study of pharmacological activities of crude venom on animals is greatly impaired by the wide variety of toxic effects. Hence, photooxidation has been suggested as an alternative approach to investigate their impact when the toxophoric region is altered and in the presence of a sensitizer methylene blue (21). Many chromophoric amino acids such as histidine tryptophan, tyrosine phenylalanine are shown to be photooxidized (22). The detoxification of VSE of Vespa orientalis was carried out by photooxidation in the presence of MB at different time exposures. Based on a toxicity study and antigencity test, 45-minute exposure venom product was selected as PVSE. The presence of antigencity of PVSE is an indication of retention of intact antigenic domain while absence of toxicity reveals alteration of toxophoric groups of VSE. Any alteration due photooxidation is specific due to transitions generated in the amino acid side chain, functional group of amino acids, di-sulfide cross bridges or selective transitions generated, in particular amino acids.
In the present study, there was a shift in the ODmax in PVSE absorption profile compared to VSE. An increased absorbance was reciprocated with loss of venom protein in photooxidized venom. Comparative absorbance changes in reaction mixture of VSE and PVSE revealed the loss of protein in PVSE. Similar alterations were observed in photooxidized venom product of Naja siamensis and Echis carinatus, which corroborates our hypothesis (4, 23). This may be due to unfolding of proteins in the presence of sensitizer dye.
Wasp venom is complex mixture of many enzymatic and non-enzymatic proteins. Nearly 4% of soluble proteins of Vespa orientalis are known to contain acetylcholine-like substances (7). The venom and VSE of Vo provoked CNS anticholinesterase-like activity in various animals, and may open the blood-brain barrier and cause central mediated changes in auditory and somatosensory activities in cats (6, 24). Snake venom neurotoxins and their effect on central acetylcholine receptors and various ion channels have been reported earlier (25). These facts prompt us to investigate the effect of PVSE of Vo on memory and learning in rats, since many photooxidized venom proteins have shown CNS stimulant action and improved the learning and memory functions (5, 26). In the current study, we employed T maze (for spatial working memory), elevated plus maze and step down passive avoidance (for long term memory) as models for memory and learning activity.
Administration of PVSE significantly shortened the time required to reach the food in T-maze and transfer latency in EPM, while a significant improvement was observed in step down passive avoidance test, which indicates a clear cut improvement in learning and memory function in all the models. These experimental results indicated that PVSE components might have acted directly or indirectly upon repeated administration and some water soluble components of venom responsible for boosting of blood flow in central cortex (27). However, we cannot rule out the role of other low-molecular weight protein and peptides or enzymes, which may act by anticholinesterase activities that affect CNS action (6).
ACKNOWLEDGEMENTS
The present work is part of a PhD study under Rajiv Gandhi University of Health Sciences, Bangalore, Karnataka. The authors are thankful to the principal and management of Al-Ameen College of Pharmacy and PES College of Pharmacy, Bangalore for the necessary support.
Submission status
Received: March 23, 2011.
Accepted: August 16, 2011.
Abstract:published online: August 17, 2011.
Full paper published online: November 30, 2011.
Conflicts of interest
There is no conflict.
Financial source
This work was supported by a partial grant from ICMR, New Delhi, India.
Ethics committee approval
The present study was approved by the Institutional Animal Ethics Committee (IAEC) of Al-Ameen College of Pharmacy, Bangalore, India (AACP/P-14). Moreover, all procedures were conducted in accordance with the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Government of India.
- 1. Chen W, Yang X, Yang X, Zhai L, Lu Z, Liu J, et al. Antimicrobial peptides from the venoms of Vespa bicolor Fabricius. Peptides. 2008;29(11):1887-92
- 2. Nakajima T. Biochemistry of vespid venoms. In: Tu AT, editor. Handbook of natural toxins. New York: Marcel Dekker; 1984. 109-33 p. vol 2.
- 3. Kocholoty W, Asshley BD. Detoxification of Russell's viper (Viper russelli) and water moccasin (Agkistrodon piscivorus) venom photooxidation. Toxicon. 1996;3(3):187-94.
- 4. Gawade SP, Prashar S. Pharmacodynamics of photooxidized Echis carinatus venom product using UV sensitized methylene blue. Indian J Pharma Edu. 2004; 38(2):81-5.
- 5. Reddy CM, Gawade SP. Evaluation of the effects of photooxidized Echis carinatus venom on learning, memory and stress. J Venom Anim Toxins incl Trop Dis. 2006;12(4):632-52.
- 6. Ishaya JS. Anticholinesterase-like activity by oriental hornet (Vespa orientalis) venom and venom sac extract. Cell Mol Life Sci. 1979;35(5):636-9.
- 7. Friedman J, Ishay JS. Inhibition of protein synthesis by an extract of the venom sac of the oriental hornet (Vespa orientalis). Toxicon. 1987;25(6):673-6.
- 8. Turner RA. Screening methods in pharmacology. New York: Academic Press; 1965. 26 p.
- 9. Dandeu JP, Henocq E, Raffard M, Iashay J, David B. Immunochemical study of the venom from the hornet Vespa orientalis (Hymenoptera: Vespinae). Ann Allerg. 1985;54(1):236-9.
- 10. Hudson L, Hay FC. Isolation and structure of immunoglobulins. In: Hudson L, Hay FC, editors. Practical Immunology. 3rd ed. Boston: Blackwell Scientific Publication; 1989. 507 p.
- 11. Hartree EF. Determination of protein: a modification of the Lowry method that gives a linear photometric response. Anal Biochem. 1972;48(1):422-7.
- 12. Ouchterlony O. Antigen-antibody reactions in gels. Acta Pathol Microbiol Scand. 1949;26(4):507-15.
- 13. Akbari A, Rabiei H, Hedayat A, Mohammadpour N, Zolfagharian H, Teimorzadeh SH. Production of effective antivenin to treat cobra snake (Naja naja oxiana) envenoming. Arch Razi Inst. 2010;65(1):33-7.
- 14. De-Mello N, Carobrez AP. Elevated T-maze as an animal model of memory: effects of scopolamine. Behav Pharmacol. 2002;13(1):139-48.
- 15. Kulkarni SK, Verma A. Evidence for nootropic effect of BR-16A (Mentat). A herbal psychotropic preparation, in mice. Ind J Physiol Pharmacol. 1992;36(1):29.
- 16. Camacho F, Smith CP, Vargas HM, Winslow JT. Alpha2-adrenoceptor antagonists, potentiate acetylcholinesterase inhibitor effects on passive avoidance learning in rat. Psychopharmacology (Berl). 1996;124(4):347-54.
- 17. Pramanik S, Banerjee S. Wasp stings with multisystem dysfunction. Indian Pediatr. 2007;44(10):788-90.
- 18. Ring B, Ishay J, Slor H. A neutral DNase in venom sac extract of the oriental hornet Vespa orientalis: characterization, specificity and mode of action. Toxicon. 1981;19(2):241-7.
- 19. Ho CL, Lin YL, Li SF. Three toxins with phospholipase activity isolated from the yellow-legged hornet (Vespa verutina) venom. Toxicon. 1999;37(7):1015-24.
- 20. Haim B, Rimon A, Ishay JS, Rimon S. Purification, characterization and anticoagulant activity of a proteolytic enzyme from Vespa orientalis venom. Toxicon. 1999;37(5):825-9.
- 21. Gawade SP. Photodynamic studies on Aflatoxin B1 using UV radiation in the presence of methylene blue. Ind J Pharm Edu Res. 2010;44(2):142-7.
- 22. Weil L, Gordon WG, Buchert AR. Photooxidation of amino acids in the presence of methylene blue. Arch Biochem Biophys. 1951;33(1):90-109.
- 23. Gawade SP. The photodynamic action of UV sensitized methylene blue on venom of Thailand cobra Naja siamensis J Venom Anim Toxins. 2000;6(2):271-80.
- 24. Barenholz-Paniry V, Ishay JS, Freeman S, Sohmer H. Evoked potential changes in cats following injection of an extract from the venom sac of the oriental hornet (Vespa orientalis). Toxicon. 1990;28(11):1317-24.
- 25. Parmentier J, Carpenter DO. Blocking action of snake venom neurotoxins at receptor site to putative central nervous system transmitters. In: Ohsaka A, Hayashi K, Sawai Y, editors. Animal plant microbial toxins. New York: Platinum; 1976. p. 179-91. vol 2.
- 26. Kshama D, Fattepur SR, Gawade SP, Santhosh VR. Neuropharmacological studies on photo-oxidized Enhydrina schistose venom product. In: Proceeding of the 59th Indian Pharmaceutical Congress, 2007. Varanasi, India: Banaras Hindu University; 2007.
- 27. Kaspi T, Ishay JS. Cortical cerebral blood flow is enhanced by hornet venom. Pharmacol Toxicol. 1990;67(3):205-8.
Publication Dates
-
Publication in this collection
05 Dec 2011 -
Date of issue
2011
History
-
Received
23 Mar 2011 -
Accepted
16 Aug 2011