Abstract
The lethal and enzymatic activities of venom from Naja sumatrana (Equatorial spitting cobra) were determined and compared to venoms from three other Southeast Asian cobras (Naja sputatrix, Naja siamensis and Naja kaouthia). All four venoms exhibited the common characteristic enzymatic activities of Asiatic cobra venoms: low protease, phosphodiesterase, alkaline phosphomonoesterase and L-amino acid oxidase activities, moderately high acetylcholinesterase and hyaluronidase activities and high phospholipase A2. Fractionation of N. sumatrana venom by Resource® S cation exchange chromatography (GE Healthcare, USA) yielded nine major protein peaks, with all except the acidic protein peak being lethal to mice. Most of the protein peaks exhibit enzymatic activities, and L-amino acid oxidase, alkaline phosphomonoesterase, acetylcholinesterase, 5'-nucleotidase and hyaluronidase exist in multiple forms. Comparison of the Resource® S chromatograms of the four cobra venoms clearly indicates that the protein composition of N. sumatrana venom is distinct from venoms of the other two spitting cobras, N. sputatrix (Javan spitting cobra) and N. siamensis (Indochinese spitting cobra). The results support the revised systematics of the Asiatic cobra based on multivariate analysis of morphological characters. The three spitting cobra venoms exhibit two common features: the presence of basic, potentially pharmacologically active phospholipases A2 and a high content of polypeptide cardiotoxin, suggesting that the pathophysiological actions of the three spitting cobra venoms may be similar.
Naja; cobra venom; enzyme activity; cardiotoxin
ORIGINAL PAPERS
Biochemical and toxinological characterization of Naja sumatrana (Equatorial spitting cobra) venom
Yap MKK; Tan NH; Fung SY
Department of Molecular Medicine, Center for Natural Products and Drug Research (CENAR), Faculty of Medicine, University of Malaya, Kuala Lumpur, Malaysia
Correspondence to Correspondence to: Tan Nget Hong Department of Molecular Medicine Faculty of Medicine University of Malaya Kuala Lumpur, Malaysia Phone: +603 7967 4912. Fax: + 603 7967 4957 Email: tanngethong@yahoo.com.sg.
ABSTRACT
The lethal and enzymatic activities of venom from Naja sumatrana (Equatorial spitting cobra) were determined and compared to venoms from three other Southeast Asian cobras (Naja sputatrix, Naja siamensis and Naja kaouthia). All four venoms exhibited the common characteristic enzymatic activities of Asiatic cobra venoms: low protease, phosphodiesterase, alkaline phosphomonoesterase and L-amino acid oxidase activities, moderately high acetylcholinesterase and hyaluronidase activities and high phospholipase A2. Fractionation of N. sumatrana venom by Resource® S cation exchange chromatography (GE Healthcare, USA) yielded nine major protein peaks, with all except the acidic protein peak being lethal to mice. Most of the protein peaks exhibit enzymatic activities, and L-amino acid oxidase, alkaline phosphomonoesterase, acetylcholinesterase, 5'-nucleotidase and hyaluronidase exist in multiple forms. Comparison of the Resource® S chromatograms of the four cobra venoms clearly indicates that the protein composition of N. sumatrana venom is distinct from venoms of the other two spitting cobras, N. sputatrix (Javan spitting cobra) and N. siamensis (Indochinese spitting cobra). The results support the revised systematics of the Asiatic cobra based on multivariate analysis of morphological characters. The three spitting cobra venoms exhibit two common features: the presence of basic, potentially pharmacologically active phospholipases A2 and a high content of polypeptide cardiotoxin, suggesting that the pathophysiological actions of the three spitting cobra venoms may be similar.
Key words:Naja, cobra venom, enzyme activity, cardiotoxin.
INTRODUCTION
Snake envenomation is a major public health problem in tropical and subtropical countries. Recent studies indicated that globally, there are more than 400,000 envenomation cases annually of which at least 20,000 are fatal (1). Cobra (Naja sp.) is one of the commonest causes of snake envenomations in Asia, including Malaysia (2). Envenomation by Naja sp. is characterized by local necrosis, neurological paralysis and cardiotoxicity (3). Snake venoms are complex mixtures containing predominantly proteins and polypeptides and small amount of organic compounds and minerals. Many of the proteins exhibit enzymatic activities, whereas the polypeptides include neurotoxins, cardiotoxins, myotoxins and cytotoxins.
The systematics of the Asiatic cobra (formerly Naja naja) has recently been revised and many subspecies have been elevated to the status of full species (4). A number of the cobras have also been more appropriately renamed. According to WŰster (4), the spitting cobras of Southeast Asia, once known collectively as Naja sputatrix (formerly known as Malayan cobra) actually consist of three separate species: Naja siamensis (Indochinese spitting cobra) in Thailand, Naja sumatrana (Equatorial spitting cobra) in Peninsular Malaysia and Sumatra, and Naja sputatrix (Javan spitting cobra) in Java and southern Indonesia.
Despite some early reports on the toxinological properties of the spitting cobra in Malaysia, it is not known whether the venom samples used for the investigations, labeled as N. sputatrix, were from N. sumatrana or N. sputatrix (5). This was due to insufficient understanding of the systematics of the Southeast Asian spitting cobra prior to the 1990's and it was assumed at that time that N. sputatrix refers to the Malaysian spitting cobra. In view of the medical importance of spitting cobras in Southeast Asia, it is important to re-investigate their venom properties. In this paper, we report the biochemical and toxinological properties of venom from N. sumatrana captured in central Malaysia, and compared with the biochemical and toxinological properties of venoms from the other two Southeast Asian spitting cobras (N. sputatrix and N. siamensis) as well as N. kaouthia (monocellate cobra), also a common cobra in Malaysia. Knowledge of the toxinological properties of these venoms is important in order to understand the pathophysiological effects of cobra bites in Southeast Asia.
MATERIALS AND METHODS
N. sumatrana venom was a pooled venom sample obtained from spitting cobras captured in central Malaysia (Snake Valley, Malaysia) and identified by one of the authors (Tan NH). Venoms of N. sputatrix, N. siamensis and N. kaouthia (Thailand) were purchased from Latoxan (France). All chemicals and reagents used in this study were of analytical grade and purchased from Sigma-Aldrich (USA). Resource® S ion exchange column (1 mL) was purchased from GE Healthcare (USA). Albino mice (ICR strain) were supplied from Laboratory Animal Center, Faculty of Medicine, University of Malaya. The animals were handled according to the CIOMS guidelines (6).
Fractionation of Naja sp. Venoms by Resource® S Ion Exchange Chromatography
Ten milligram of the venom dissolved in 200 µL of start buffer (20 mM MES, pH 6.0) was subjected to Resource® S cation exchange chromatography using the Shimadzu LC-20AD HPLC system (Japan). The column was first pre-equilibrated with the same start buffer, followed by elution by a linear gradient (0.0-0.8 M NaCl) of 0-30% from 5 to 30 minutes, followed by 30-100% from 30-55 minutes, at a flow rate of 1 mL/minute. Absorbance at 280 nm was measured.
Determination of Protein Concentration by Bradford Method
Protein content was determined according to the Bradford method (7). A standard curve was constructed using bovine serum albumin (0-10 µg of protein).
Enzyme Assays
Venom was dissolved in physiological saline (1 mg/mL) and used for enzyme assays and lethality determinations.
Protease assay
Protease activity was determined according to the method described by Kunitz (8) with slight modifications. One milliliter of 1% casein in 0.25 M sodium phosphate buffer, pH 7.75, and 50 µL of venom solution/fraction were incubated at 37°C for 30 minutes. The enzyme reaction was stopped and undigested casein was precipitated by adding 1 mL of 5% trichloroacetic acid. The mixture was then centrifuged at 10,000 x g for 10 minutes. The absorbance of the supernatant at 280 nm was then measured. Protease activity was arbitrarily defined as an increase of one absorbance unit at 280 nm per hour.
Phosphodiesterase assay
Phosphodiesterase activity was determined by a method modified from Lo et al. (9). First, 0.1 mL of venom solution/fraction was added to an assay mixture containing 0.5 mL of 0.0025 M Ca-bis-p-nitrophenyl phosphate, 0.3 mL of 0.01 M MgSO4 and 0.5 mL of 0.17 M Veronal buffer, pH 9.0. The reaction was monitored by absorbance measurement at 400 nm. Phosphodiesterase activity was expressed in nanomole of product released/minute. Molar extinction coefficient at 400 nm was 8100 cm-1M-1.
Alkaline phosphomonoesterase assay
Alkaline phosphomonoesterase assay was carried out by a method modified from Lo et al. (9). A substrate mixture consisting of 0.5 mL of 0.5 M glycine buffer, pH 8.5; 0.5 mL of 0.01 M p-nitrophenylphosphate and 0.3 mL of 0.01 M MgSO4 was prepared. Then, 0.1 mL of venom solution/fraction was added to the substrate mixture, and incubated at 37°C for 30 minutes. After 20 minutes, 2 mL of 0.2 M sodium hydroxide was added, and absorbance at 400 nm was measured. Alkaline phosphomonoesterase activity was expressed as nanomole of product released/minute. The molar extinction coefficient at 400 nm was 18500 cm-1M-1.
5'-Nucleotidase assay
The 5'-nucleotidase activity was measured according to Heppel and Hilmore (10). First, 0.1 mL of venom solution/fraction was added to an assay mixture containing 0.5 mL of 0.02 M 5'-AMP (pre-adjusted to pH 8.5), 0.5 mL of 0.2 M glycine buffer, pH 8.5 and 0.1 mL of 0.1 M MgSO4. The mixture was incubated at 37°C for 10 minutes, and the reaction was terminated by adding 1.5 mL of 10% trifluoroacetic acid. The quantity of inorganic phosphate released was determined by ascorbic acid method (11). Briefly, 1 mL of ascorbic acid reagent (equal parts of 3 M sulfuric acid, 2.5 % ammonium molibdate, 10% ascorbic acid and water) was added to the above mixture. The mixture was left at room temperature for 30 minutes and the absorbance at 820 nm was then determined. A standard curve of known phosphate concentrations was constructed. The enzyme activity was expressed as nanomole of phosphate/minute.
Hyaluronidase assay
Hyaluronidase activity was determined turbidimetrically as described by Dorfman (12). First, 0.1 mL of venom solution/fraction was added to an assay mixture containing 1 mL of 0.03 % (w/v) hyaluronic acid in 0.3 M sodium phosphate pH 5.35, and 1 mL of 0.02 M sodium phosphate, pH 7.0 containing 0.077 M sodium chloride. The mixture was incubated at 37°C for 45 minutes. Then 2.5 mL of acid albumin reagent [0.024 M sodium acetate, pH 3.75 containing 0.1% (w/v) bovine serum albumin] was added to 0.5 mL of the above reaction mixture and incubated at room temperature for another 10 minutes. The absorbance at 600 nm was then measured. A standard curve was constructed using a standard hyaluronidase (760 National Formulary units/ mg solid). Hyaluronidase activity was expressed in National Formulary Unit/mg (NFU/mg).
Phospholipase A2 assay
Phospholipase A2 assay was determined according to the acidimetric method of Tan and Tan (13). Briefly, an egg yolk suspension was prepared by mixing three equal parts consisting of one part chicken egg yolk, one part 18 mM calcium chloride, and one part 8.1 mM sodium deoxycholate. The pH of the egg yolk suspension was adjusted to 8.0 with 1 M sodium hydroxide, and stirred for 10 minutes to ensure homogenous mixing. Next, 0.1 mL of venom solution/fraction was added to 15 mL of egg yolk suspension to initiate the hydrolysis. The initial decrease in pH was measured by a pH meter. A decrease of 1 pH unit corresponds to 133 µmoles of fatty acid released. Enzyme activity was expressed as µmoles of fatty acid released/minute.
L-amino acid oxidase assay
L- amino acid oxidase activity was determined using L-leucine as substrate as described by Decker (14). First, 50 µL of 0.0075% horseradish peroxidase (100 purpurogalin unit/mg) was added to 0.9 mL of 0.2 M triethanolamine buffer, pH 7.6, containing 0.1% L-leucine and 0.0075% o-dianisidine, and incubated for 3 minutes at room temperature. Then, 50 µL of venom solution/fraction was added and the increase in absorbance at 436 nm was measured. The molar extinction coefficient was 8.31 x 10-3 cm-1M-1. L- amino acid oxidase activity was expressed as µmole of L-leucine oxidized/minute.
Acetylcholinesterase assay
Acetylcholinesterase activity was determined according to the method described by Ellman et al. (15). One hundred milliliters of venom solution/fraction was added to an assay mixture contained 0.8 mL of 0.025 M sodium phosphate buffer, pH 7.5, 0.05 mL of 0.0125 M acetylthiocholine iodide and 0.05 mL of 6.66 mM dithionitrobenzoate (DTNB) containing 1 mg/mL sodium bicarbonate. The hydrolysis of the substrate was measured by the increase in absorbance at 412 nm. The molar extinction coefficient was 13600 cm-1M-1. The enzyme activity was expressed as µmole of product released/minute.
Determination of the Median Lethal Dose (LD50)
The median lethal dose (LD50) of the venoms and venom fractions obtained from Resource® S chromatography was determined by injecting appropriate dilutions of the sample intravenously into the caudal vein of mice (n = 4, 18-20 g). The survival ratio was determined after 24 hours. The LD50 (95% confidence interval) was then calculated by the Probit analysis (16).
Confirmation of the Identity of Fractions 8 and 9 as Cardiotoxins by Mass Spectrometry
The cardiotoxic fractions 8 and 9 isolated from Resource® S ion exchange chromatography were further purified by C-18 reverse-phase HPLC using a Shimadzu LC-20AD HPLC system (Japan), eluted with 0 to 100 % acetonitrile gradient. In both cases, there was only one major protein peak and the protein was subjected to tryptic digestion. The digested peptides were then analyzed by mass spectrometry (MALDI-TOF/TOF); ion spectra of peptides generated were interpreted using MASCOT database so that they could be assigned to known protein families. The deduced peptide ion sequence was submitted to BLAST to search for protein sequence similarity.
RESULTS AND DISCUSSION
Enzymatic and Lethal Activities of Venoms from N. sumatrana and Three Other Southeast Asian Cobras
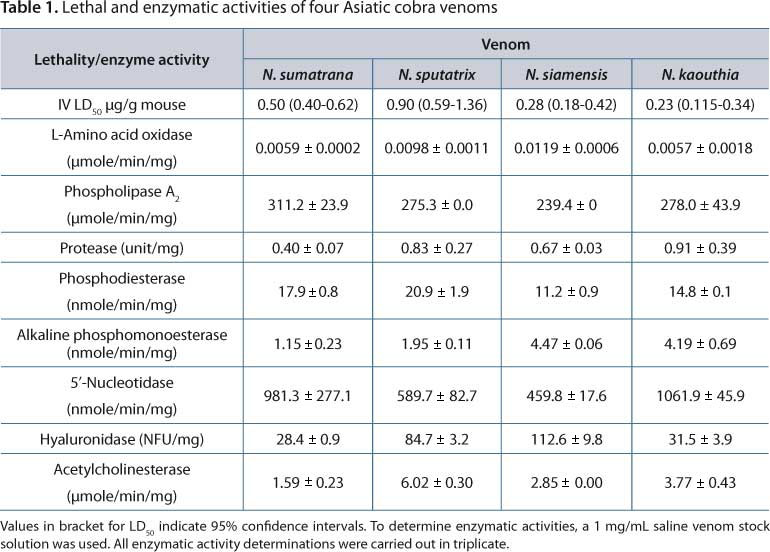
The enzymatic activities of venoms from N. sumatrana, N. sputatrix, N. siamensis and N. kaouthia are shown in Table 1. All four venoms exhibited the common characteristic enzymatic activities of Asiatic cobra venoms: low protease, phosphodiesterase, alkaline phosphomonoesterase and L-amino acid oxidase activities, moderately high acetylcholinesterase and hyaluronidase activities and high phospholipase A2 (17). Venoms from the two cobras of Thailand (N. kaouthia and N. siamensis) were highly lethal (with comparable intravenous (IV) LD50 of 0.22 µg/g mouse and 0.28 µg/g mouse respectively). N. sputatrix (Javan spitting cobra) was the least lethal (IV LD50 of 0.90 µg/g mouse) whereas the spitting cobra in Malaysia, N. sumatrana, presented a moderate IV LD50 of 0.50 µg/g mouse.
Fractionations of the N. sumatrana Venom by Resource® S Cation Exchange Chromatography
Figure 1 shows the elution profile of Resource® S cation exchange chromatography of N. sumatrana venom. There were nine major peaks with a number of minor peaks. The enzymatic activities, relative protein percentage and IV LD50 of the nine major fractions are shown in Table 2 and Figure 1. Of the nine major peaks, only peak 1, which contains acidic proteins, was not lethal to mice. Based on IV LD50 and relative elution volumes (slightly to moderately basic), peaks 2, 6 and 7 were probably the polypeptide neurotoxins. Peak 2 probably consisted of only polypeptide neurotoxin as it was devoid of any enzymatic activity. Their IV LD50s were < 0.1 µg/g in mice and together they constituted about 23% of venom protein. Peak 8 and 9, the two very basic protein fractions, are presumably the polypeptide cardiotoxins, as it is well established that cobra venom cardiotoxins are highly basic polypeptides (18).
The cardiotoxins (fractions 8 and 9) together constituted about 40% of the venom protein. Similarly high contents of cardiotoxins in cobra venoms have been reported (19). To confirm their identity, fractions 8 and 9 were further purified by reverse-phase HPLC and the purified polypeptides were subjected to MALDI-TOF-TOF. Both polypeptides possessed the consensus sequence of cardiotoxin RGCIDVCPK, thereby confirming that fractions 8 and 9 were indeed cardiotoxin (20). Three peaks exhibited phospholipase A2 activity: peak 1 (acidic, non-lethal), peak 4 (7% of venom protein, IV LD50 0.8 µg/g) and peak 5, the major phospholipase A2 (17% of venom protein, IV LD50 0.91 µg/g). Both the two lethal phospholipases A2 were basic proteins.
All major protein peaks except peak 2 exhibited enzymatic activities; and L-amino acid oxidase, alkaline phosphomonoesterase, acetylcholinesetrase, 5'-nucleotidase and hyaluronidase were observed in multiple forms. Figure 1 shows that all the major lethal protein fractions, except peak 2, contained two to three different types of enzymatic activities. To isolate the pure polypeptide toxins or phospholipases A2, additional chromatographic steps were therefore necessary to remove the enzymes.
Fractionation of the N. sputatrix, N. siamensis and N. kaouthia Venom by Resource® S Cation Exchange Chromatography
Figure 2 (B, C and D) show the Resource® S cation exchange chromatographic fractionation of N. sputatrix, N. siamensis and N. kaouthia venoms under the same conditions as the fractionation of N. sumatrana venom (Figure 2 - A). The phospholipase A2 activity of the peaks is also shown. It is interesting to note that the chromatographic pattern of N. sumatrana venom is similar to, yet distinct from, N. sputatrix venom, but differs greatly from that of N. kaouthia venom, in agreement with the findings of Vejayan et al. (21). On the other hand, the chromatographic pattern of N. siamensis venom is similar to that of N. kaouthia venom.
Comparison of the chromatograms clearly indicates that the composition of the N. sumatrana venom is definitely different from venoms obtained from the other two spitting cobras, N. sputatrix and N. siamensis. This supports the revised systematics of the Asiatic cobra based on multivariate analysis of morphological characteristics (22). Thus, the results and data in all previous publications on Malaysian spitting cobra (also known as Malayan cobra) based on venom labelled as N. sputatrix should be re-interpreted as properties of Javan spitting cobra venom instead.
Examination of the chromatograms in Figure 2 also reveals an interesting common feature of the venoms from the Southeast Asian spitting cobras - all three spitting cobra venoms contained relatively basic phospholipases A2 (peak 5 in N. sumatrana, peak 2 and 3 in N. sputatrix, peak 5 in N. siamensis), whereas there was only one (minor) neutral phospholipase A2 (peak 3) in the non-spitting N. kaouthia venom. Basic phospholipases A2 are known to be involved in many pharmacological actions of cobra venoms including myotoxicity, cytotoxicity, cardiotoxicity and edema-inducing activities (23-28). Also, we note that all three Southeast Asian spitting cobra venoms contained a substantial amount (30-40%) of polypeptide cardiotoxins (Table 3). This would suggest that the pathophysiological actions of the three spitting cobra venoms may be similar. On the other hand, Naja kaouthia venom did not possess basic phospholipase A2 and presented a much lower cardiotoxin content (18%) but very high neurotoxin content (peak 4, 40% of venom protein). This is consistent with the clinical reports that a high percentage of patients of N. kaouthia envenomation in Thailand experienced neurotoxic symptoms (29).
CONCLUSIONS
N. sumatrana venom exhibits the common characteristic enzymatic activities of Asiatic cobra venoms: The protein composition of N. sumatrana venom is distinct from venoms of the other two regional spitting cobras, N. sputatrix and N. siamensis. However, all three spitting cobra venoms contain basic phospholipases A2 and a high content of polypeptide cardiotoxin. Our results support the revised systematics of the Asiatic cobra based on multivariate analysis of morphological characteristics.
ACKNOWLEDGEMENTS
The authors are grateful to the Government of Malaysia for the financial support.
Submission status
Received: April 11, 2011.
Accepted: August 24, 2011.
Abstract:published online: August 25, 2011.
Full paper published online: November 30, 2011.
Conflicts of interest
There is no conflict.
Financial source
The Government of Malaysia provided the financial grants (PS 244/2010 B).
Ethics committee approval
The present study was approved by the Animal Care and Use Committee of the University of Malaya [PM/03/03/2010/FSY(R)]. Moreover, the study animals were handled following the CIOMS guidelines on animal experimentation.
- 1. Kasturiratne A, Wickremasinghe AR, de Silva N, Gunawardena NK, Partmeswaran A, Premaratna R, et al. Estimating the global burden snakebite: a literature analysis and modeling based on regional estimates of envenoming and deaths. PLos Med. 2008;5(11):e218.
- 2. Warrel DA. Clinical toxicology of snakebite in Asia. In: Meier J, White J, editors. Handbook of clinical toxicology of animal venoms and poisons. Boca Raton: CRC Press; 1995. p. 493-594.
- 3. Reid HA. Cobra bites. Br Med J. 1964;2(1):540-5.
- 4. Wüster W. Taxonomic changes and toxinology: systematic revisions of the Asiatic cobras (Naja naja species complex). Toxicon. 1996;34(4):399-406.
- 5. Tan NH. The biochemistry of venoms of some venomous snakes of Malaysia: a review. Trop Biomed. 1991;8(1):91-103.
- 6. Howard-Jones NA. A CIOMS ethical code for animal experimentation. WHO Chronicle. 1985;39(1):51-6.
- 7. Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72(1):248-54.
- 8. Kunitz M. Crystalline soybean trypsin inhibitor. II. General properties. J Gen Physiol. 1947;30(1):291-310.
- 9. Lo TB, Chen YH, Lee CY. Chemical studies of Formosan cobra (Naja naja atra) venom. Part 1. Chromatographic separation of crude venom on CM-Sephadex and preliminary characterization of its components. J Chinese Chem Soc. 1966;13(1):165-77.
- 10. Heppel LA, Hilmore RJ. 5'-Nucleotidase. In: Colowick SP, Kaplan NO, editors. Methods in enzymology. New York: Academic Press; 1955. p. 547-50. 2 vols.
- 11. Chen PS, Toribara TY, War H. Microdetermination of phosphorus. Anal Chem. 1956;28(1):1756-9.
- 12. Dorfman A. Mucopolysaccharidases. In: Colowick SP, Kaplan NO, editors. Methods in enzymology. New York: Academic Press; 1955. p. 166-73. 1 vol.
- 13. Tan NH, Tan CS. Acidimetric assay for phospholipase A using egg yolk suspension as substrate. Anal Biochem. 1988;170(2):282-8.
- 14. Decker A. L-amino acid oxidase, Worhington Enzyme Manual. New Jersey: Worthington Biochemical Co; 1977. 49 p.
- 15. Ellman GL, Courtney KD, Andres V Jr, Featherstone RM. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol. 1961;7(1):88-95.
- 16. Finney DJ. Probit analysis. 2nd ed. England: Cambridge University Press; 1952. 318 p.
- 17. Tan NH, Tan CS. A comparative study of cobra (Naja) venom enzymes. Comp Biochem Physiol B. 1988;90(4):745-50.
- 18. Tu AT. Venoms. Chemistry and molecular biology. New York: John Wiley & Sons; 1977. p. 151-77.
- 19. Hegde RP, Rajagopalan N, Doley R, Kini RM. Snake venom three-finger toxins. In: Mackessy SP, editor. Handbooks of venoms and toxins of reptiles. Boca Raton, FL: CRC Press; 2009. p. 287-301.
- 20. Jeyaseelan K, Armugam A, Lachumanan R, Tan CH, Tan NH. Six isoforms of cardiotoxin in malayan spitting cobra (Naja naja sputatrix) venom: cloning and characterization of cDNAs. Biochim Biophys Acta. 1998;1380(2):209-22.
- 21. Vejayan J, Shin Yee L, Ponnudurai G, Ambu S, Ibrahim I. Protein profile analysis of Malaysian snake venoms by two-dimensional gel electrophoresis. J Venom Anim Toxins incl Trop Dis. 2010;16(4):623-30.
- 22. Wuster W, Thorpe RS. Asiatic cobras: Systematics and snakebite. Experientia. 1991;47(2):205-9.
- 23. Bhat MK, Gowda TV. Purification and characterization of a myotoxic phospholipase A2 from Indian cobra (Naja naja naja) venom. Toxicon. 1989;27(8):861-73.
- 24. Bhat MK, Prasad BN, Gowda TV. Purification and characterization of a neurotoxic phospholipase A2 from Indian cobra (Naja naja naja) venom. Toxicon 1991;29(11):1345-9.
- 25. Mukherjee AK. Phospholipase A2-interacting weak neurotoxins from venom of monocled cobra Naja kaouthia display cell-specific cytotoxicity. Toxicon. 2008;51(8):1538-43.
- 26. Das T, Bhattacharya S, Halder B, Biswas A, Das Gupta S, Gomes A, et al. Cytotoxic and antioxidant property of a purified fraction (NN-32) of Indian Naja naja venom on Ehrlich ascites carcinoma in BALB/c mice. Toxicon. 2011;57(7-8):1065-72.
- 27. Fletcher JE, Yang CC, Rosenberg P. Basic phospholipase A2 from Naja nigricollis snake venom: phospholipid hydrolysis and effects of electrical and contractile activity of the rat heart. Toxicol Applied Pharmacol. 1982;66(1):39-54.
- 28. Yamaguchi Y, Shimohigashi Y, Chijiwa T, Nakai M, Ogawa T, Hattori S, et al. Characterization, amino acid sequence and evolution of edema-inducing, basic phospholipase A2 from Trimeresurus flavoviridis venom. Toxicon. 2001;39(7):1069- 76.
- 29. Viravan C, Veeravat U, Warrell MJ, Theakston RD, Warrell DA. ELISA confirmation of acute and past envenoming by the monocellate Thai Cobra (Naja kaouthia). Am J Trop Med Hyg. 1986;35(1):173-81.
Publication Dates
-
Publication in this collection
05 Dec 2011 -
Date of issue
2011
History
-
Received
11 Apr 2011 -
Accepted
24 Aug 2011