Abstract
Background:
Here, we described the presence of a neurotoxin with phospholipase A2 activity isolated from Micrurus lemniscatus venom (Mlx-8) with affinity for muscarinic acetylcholine receptors (mAChRs).
Methods:
The purification, molecular mass determination, partial amino acid sequencing, phospholipase A2 activity determination, inhibition of the binding of the selective muscarinic ligand [3H]QNB and inhibition of the total [3H]inositol phosphate accumulation in rat hippocampus of the Mlx-8 were determined.
Results:
Thirty-one fractions were collected from HPLC chromatography, and the Mlx-8 toxin was used in this work. The molecular mass of Mlx-8 is 13.628 Da. Edman degradation yielded the following sequence: NLYQFKNMIQCTNTRSWL-DFADYG-CYCGRGGSGT. The Mlx-8 had phospholipase A2 enzymatic activity. The pKi values were determined for Mlx-8 toxin and the M1 selective muscarinic antagonist pirenzepine in hippocampus membranes via [3H]QNB competition binding assays. The pKi values obtained from the analysis of Mlx-8 and pirenzepine displacement curves were 7.32 ± 0.15, n = 4 and 5.84 ± 0.18, n = 4, respectively. These results indicate that Mlx-8 has affinity for mAChRs. There was no effect on the inhibition ability of the [3H]QNB binding in hippocampus membranes when 1 µM Mlx-8 was incubated with 200 µM DEDA, an inhibitor of phospholipase A2. This suggests that the inhibition of the phospholipase A2 activity of the venom did not alter its ability to bind to displace [3H]QNB binding. In addition, the Mlx-8 toxin caused a blockade of 43.31 ± 8.86%, n = 3 and 97.42 ± 2.02%, n = 3 for 0.1 and 1 µM Mlx-8, respectively, on the total [3H]inositol phosphate content induced by 10 µM carbachol. This suggests that Mlx-8 inhibits the intracellular signaling pathway linked to activation of mAChRs in hippocampus.
Conclusion:
The results of the present work show, for the first time, that muscarinic receptors are also affected by the Mlx-8 toxin, a muscarinic ligand with phospholipase A2 characteristics, obtained from the venom of the Elapidae snake Micrurus lemniscatus, since this toxin was able to compete with muscarinic ligand [3H]QNB in hippocampus of rats. In addition, Mlx-8 also blocked the accumulation of total [3H]inositol phosphate induced by muscarinic agonist carbachol. Thus, Mlx-8 may be a new pharmacological tool for examining muscarinic cholinergic function.
Keywords
Muscarinic receptors; Hippocampus;
Micrurus lemniscatus
; Inositol phosphate; Phopholipase A2
Background
In the Americas, the Elapidae family is represented by coralsnakes that comprise 120 species and subspecies belonging to the genera Micruroides, Leptomicrurus and Micrurus [11. Bucaretchi F, Capitani EM, Vieira RJ, Rodrigues CK, Zannin M, da Silva NJ Jr, et al. Coral snake bites (Micrurus spp.) in Brazil: a review of literature reports. Clin Toxicol (Phila). 2016 Mar;54(3):222-34., 22. Zheng Y, Wiens JJ. Combining phylogenomic and supermatrix approaches, and a time-calibrated phylogeny for squamate reptiles (lizards and snakes) based on 52 genes and 4162 species. Mol Phylogenet Evol. 2016 Jan;94(Pt B):537-47.]. Micrurus is the most abundant and diverse genus with many species found in South and Central America and the Southern United States [33. Lomonte B, Rey-Suárez P, Fernández J, Sasa M, Pla D, Vargas N, et al. Venoms of Micrurus coral snakes: evolutionary trends in compositional patterns emerging from proteomic analyses. Toxicon. 2016 Nov;122:7-25.-66. da SilvaJr NJ. As Cobras-Corais do Brasil: Biologia, Taxonomia, Venenos e Envenenamentos. 1nd ed. da Silva Jr NJ, editor. Editora da PUC Goiás, Goiânia; 2016. ]. However, the biochemistry and pharmacology of components from coralsnake venoms have not yet been thoroughly studied.
Currently, Micrurus lemniscatus is a species composed of three subspecies (M. l. carvalhoi, M. l. helleri and M. l. lemniscatus). Particularly, M. l. carvalhoi is distributed along the Brazilian east coast from the northeast to southeast of the country and in parts of central, central-western, southeastern and southern Brazil, as well as eastern Paraguay and northeastern Argentina [77. Sanz L, Quesada-Bernat S, Ramos T, Casais-e-Silva LL, Corrêa-Netto C, Silva-Haad JJ, et al. New insights into the phylogeographic distribution of the 3FTx/PLA2 venom dichotomy across genus Micrurus in South America. J Proteomics. 2019 May 30;200:90-101., 88. Floriano RS, Schezaro‑Ramos R, da Silva JrNJ, Bucaretchi F, Rowan EG, Hyslop S. Neurotoxicity of Micrurus lemniscatus lemniscatus (South American coralsnake) venom in vertebrate neuromuscular preparations in vitro and neutralization by antivenom. Arch Toxicol. 2019 Jul;93(7):2065-86.]. Moreover, the venom of this animal is composed of approximately 70% three-finger toxins (3FTxs) and 10% phospholipase A2 (PLA2) toxins [99. Aird SD, da Silva JrNJ, Qiu L, Villar-Briones A, Saddi VA, Telles MPC, et al. Coralsnake venomics: analyses of venom gland transcriptomes and proteomes of six brazilian taxa. Toxins (Basel). 2017 Jun;9(6):pii: E187.]. While enzymatic toxins contribute mainly to slow immobilization and digestion of prey, the non-enzymatic toxins stimulate rapid immobilization through their neurotoxic or cardiotoxic effects [1010. Kessler P, Marchot P, Silva M, Servent D. The three‐finger toxin fold: a multifunctional structural scaffold able to modulate cholinergic functions. J Neurochem. 2017 Aug;142(Suppl 2):7-18.].
In the elapid envenomation the presynaptic neurotoxins or β-neurotoxins and postsynaptic neurotoxins or (-neurotoxins are recognized as major and most important components of these venoms [1111. Ray-Suarez P, Nunez V, Gutiérrez JM, Lomonte B. Proteomic and biological characterization of the venom of the redtail coral snake, Micrurus mipartitus (Elapidae), from Colombia and Costa Rica. J Proteomics. 2011 Dec 21;75(2):655-67.-1313. Vergara I, Pedraza-Escalona M, Paniagua D, Restano-Cassulini R, Zamudio F, Batista CVF, et al. Eastern coral Micrurus fulvius venom toxicity in mice is mainly determined by neurotoxic phospholipases A2. J Proteomics. 2014 Jun 13;105:295-306.]. β-neurotoxins are characterized by their PLA2 activity while (-neurotoxins can be characterized as 3FTx enzymatic-free proteins that interact with cholinergic nicotinic receptors and others that interact with muscarinic acetylcholine receptors (mAChRs).
Secreted PLA2, found in mammals and animal venoms, have a molecular weight between 12 and 19 kDa, have five to eight disulfide bridges and need millimolar calcium concentrations for its catalytic activity [1414. Hanasaki K, Arita H. Phospholipase A2 receptor: a regulator of biological functions of secretory phospholipase A2. Prostaglandins Other Lipid Mediat. 2002 Aug; 68-69: 71-82.]. Among the main components of animal venoms are the secreted PLA₂ that belong to distinct PLA₂s groups. Snake venom PLA₂s from Elapidae and Viperidae families belong, respectively, to the IA and IIA/IIB groups [1515. de Carvalho ND, Garcia RCT, Ferreira AK, Batista DR, Cassola AC, Maria D, et al. Neurotoxicity of coral snake phospholipases A2 in cultured rat hippocampal neurons. Brain Res. 2014 Mar 13;1552:1-16. , 1616. Zambelli VO, Picolo G, Fernandes CAH, Fontes MRM, Cury Y. Secreted phospholipases A₂ from animal venoms in pain and analgesia. Toxins (Basel). 2017 Dec 19;9(12):pii: E406.]. For instance, snake venoms are rich sources of PLA2 enzymes that are frequently found as a large number of isozymes [1717. Ciscotto PH, Rates B, Silva DA, Richardson M, Silva LP, Andrade H, et al. Venomic analysis and evaluation of antivenom cross-reactivity of South American Micrurus species. J Proteomics. 2011 Aug 24;74(9):1810-25.].
Based in transcriptomic data it can be observed that Micrurus species are arranged in an approximately northwestern to southeastern sequence, the high PLA2 and low 3FTx concentrations in the North to high 3FTx and low PLA2 concentration in the South [99. Aird SD, da Silva JrNJ, Qiu L, Villar-Briones A, Saddi VA, Telles MPC, et al. Coralsnake venomics: analyses of venom gland transcriptomes and proteomes of six brazilian taxa. Toxins (Basel). 2017 Jun;9(6):pii: E187.]. In this way, the proteomics of the Micrurus venoms present a great diversity concerning the PLA2 composition. M. surinamensis and M. l. carvalhoi venoms show relatively little PLA2 activity. However, activity does not necessarily reflect the amount of PLA2 present. Structure determination of new micrurine PLA2 illustrates their great structural diversity. Of 121 PLA2s with partial or complete structures, the majority are apparently catalytic, having the requisite H48, D49, Y52, and D101 in their active sites. The remains are apparently non-catalytic [see 9, for review].
Quantitative differences in the content of 3FTx and PLA2 might reflect directly in the pharmacological and biological activities of Micrurus venoms. On the other hand, Tanaka et al. [1212. Tanaka GD, Furtado MF, Portaro FCV, Sant'Anna OA, Tambourgi DV. Diversity of Micrurus snake species related to their venom toxic effects and the prospective of antivenom neutralization. PLoS Negl Trop Dis. 2010 Mar 9;4(3):e622.] showed that M. frontalis, M. ibiboboca and M. lemniscatus venoms contain different levels of PLA2 activity, although the venom of M. frontalis seems to have a lower hydrolytic activity when compared to M. lemniscatus and M. ibiboboca venoms.
Ciscotto et al. [1717. Ciscotto PH, Rates B, Silva DA, Richardson M, Silva LP, Andrade H, et al. Venomic analysis and evaluation of antivenom cross-reactivity of South American Micrurus species. J Proteomics. 2011 Aug 24;74(9):1810-25.] identified that most proteins (12-14 kDa) that were found are similar to PLA2 and indicated the presence of both acidic and basic PLA2 in M. frontalis, M. ibiboboca, M. lemniscatus and M. spixii. In general, basic PLA2 enzymes are more toxic and exhibit higher pharmacological potency than their neutral and acidic counterparts, being the basic residues responsible for such potency and lethality [1818. Kini RM, Evans HJ. Structure-function relationships of phospholipases. The anticoagulant region of phospholipases A2. J Biol Chem. 1987 Oct 25;262(30):14402-7.]. Aside from displaying enzymatic activities, some vPLA2 possess a wide range of toxic effects, including neurotoxicity, myotoxicity, cardiotoxicity, cytotoxicity, and may provoke convulsion and hypotension or affect blood coagulation and platelet aggregation [1717. Ciscotto PH, Rates B, Silva DA, Richardson M, Silva LP, Andrade H, et al. Venomic analysis and evaluation of antivenom cross-reactivity of South American Micrurus species. J Proteomics. 2011 Aug 24;74(9):1810-25.].
Toxins from Elapid snake venoms play an important role in the characterization and function of mAChRs in muscle and in the identification of muscarinic and nicotinic subtypes of receptors in the central and peripheral nervous system. The venom of Elapid snakes of the genus Dendroaspis (mambas) and Naja contain 3FTx muscarinic neurotoxins with activity in mAChRs. Moreover, these have a high affinity for a specific receptor subtype. In addition, muscarinic toxins isolated from these venoms with agonist and antagonist features have also been described [1919. Bradley KN, Rowan EG, Harvey AL. Effects of muscarinic toxins MT2 and MT7, from green mamba venom, on m1, m3 and m5 muscarinic receptors expressed in Chinese hamster ovary cells. Toxicon. 2003 Feb;41(2):207-15.-2525. Servent D, Fruchart-Gaillard C. Muscarinic toxins: tools for the study of the pharmacological and functional properties of muscarinic receptors. J Neurochem. 2009 Jun;109:1193-202.]. In this way, we previously characterized the biochemical and pharmacological features of a 3FTx, MT-Mlα, isolated from Micrurus lemmiscatus venom. This toxin could displace the binding of the selective muscarinic ligand [3H]quinuclidinyl benzilate ([3H]QNB) in rat hippocampus. Furthermore, studying pathways of second messengers that can be involved in the effects of the MT-Mlα, our results demonstrated that this toxin inhibited the total [3H]inositol phosphate accumulation induced by muscarinic agonist carbachol [2626. da Silva DC, de Medeiros WA, Batista IFC, Pimenta DC, Lebrun I, Abdalla FMF, et al. Characterization of a new muscarinic toxin from the venom of the Brazilian coral snake Micrurus lemniscatus in rat hippocampus. Life Sci. 2011 Dec 19;89(25-26):931-8.].
A new class of muscarinic neurotoxins has also been described. Thus, elapid PLA2 neurotoxins isolated from Naja naja sputatrix [2727. Miyoshi S, Tu AT. Phospholipase A2 from Naja naja sputatrix venom is a muscarinic acetylcholine receptor inhibitor. Arch Biochem Biophys. 1996 Apr 1;328(1):17-25., 2828. Miyoshi S, Tu AT. Muscarinic acetylcholine receptor (mAChR) inhibitor from snake venom: interaction with subtypes of human mAChR. Arch Biochem Biophys. 1999 Sep 1;369(1):114-8.] and Naja atra [2929. Huang LF, Zheng JB, Xu Y, Song HT, Yu CX. A snake venom phospholipase A2 with high affinity for muscarinic acetylcholine receptors acts on guinea pig ileum. Toxicon. 2008 May;51(6):1008-16.] venoms have a muscarinic inhibitor activity. In addition, previously studies from our laboratory showed the neurotoxicity of four PLA2 (Mlx-8, 9, 11, and 12) isolated from the elapid Micrurus lemniscatus snake venom after microinjection into the brain [3030. Oliveira DA, Harasawa C, Seibert CS, Casais e Silva LL, Pimenta DC, Lebrun I, et al. Phospholipases A2 isolated from Micrurus lemniscatus coral snake venom: behavioral, electroencephalographic, and neuropathological aspects. Brain Res Bull. 2008 Mar 28;75(5):629-39. ]. Those studies showed the presence of isolated and clustered spikes on EEG records. These behavioral alterations were characterized mainly by forelimb clonus, compulsive scratching, and severe neuronal damage. A recent study investigated in detail the neurotoxic effects of two PLA2 toxins (Mlx-8 and Mlx-9) isolated from Micrurus lemniscatus venom on cultured primary hippocampal neurons. These data demonstrated that the PLA2 toxins Mlx-8 and Mlx-9 induce an early increase in free cytosolic calcium concentration and mitochondrial function impairment, which would lead to structural changes and could explain the toxicity to hippocampal neurons. Furthermore, the morphological approaches showed features of hybrid cell death with apoptotic, autophagic, and necrotic signs [1515. de Carvalho ND, Garcia RCT, Ferreira AK, Batista DR, Cassola AC, Maria D, et al. Neurotoxicity of coral snake phospholipases A2 in cultured rat hippocampal neurons. Brain Res. 2014 Mar 13;1552:1-16. ]. Interestingly, a recent isoform of the Mlx-8 toxin named Lemnitoxin has PLA2 activity was also isolated from Micrurus lemniscatus venom. This was cytotoxic to differentiated myotubes in vitro and muscle fibers in vivo. A pro-inflammatory activity was also described [3131. Casais-e-Silva LL, Teixeira CF, Lebrun I, Lomonte B, Alape-Giron A, Gutierrez JM. Lemnitoxin, the major component of Micrurus lemniscatus coral snake venom, is a myotoxic and pro-inflammatory phospholipase A2. Toxicol Lett. 2016 Aug 22;257:60-71.].
We have launched a search for components associated with mAChRs in the venom of the Brazilian snake Micrurus lemniscatus. We examined different peaks isolated from this venom (named earlier Mlx-1, Mlx-2, Mlx-3, Mlx-4, Mlx-5, MT-Mlα and Mlx-8). These were obtained from the analytical RP-HPLC profile of Micrurus lemniscatus venom on a C8 column. The components were also examined for their ability to compete with [3H]QNB for its binding sites. However, only MT-Mlα (a 3FTx; [2626. da Silva DC, de Medeiros WA, Batista IFC, Pimenta DC, Lebrun I, Abdalla FMF, et al. Characterization of a new muscarinic toxin from the venom of the Brazilian coral snake Micrurus lemniscatus in rat hippocampus. Life Sci. 2011 Dec 19;89(25-26):931-8.]) and Mlx-8 (a PLA2-neurotoxin; unpublished data) could displace the binding of the muscarinic ligand. In addition, partial amino acid sequences were determined for MT-Mlα [2626. da Silva DC, de Medeiros WA, Batista IFC, Pimenta DC, Lebrun I, Abdalla FMF, et al. Characterization of a new muscarinic toxin from the venom of the Brazilian coral snake Micrurus lemniscatus in rat hippocampus. Life Sci. 2011 Dec 19;89(25-26):931-8.] and Mlx-8 (unpublished data). Based on these previous results, the present study investigated the biochemical and pharmacological features of Mlx-8 isolated from Micrurus lemniscatus venom with affinity for mAChRs. Thus, this work describes the purification, molecular mass determination, partial amino acid sequencing, and phospholipase A2 activity determination of Mlx-8. Furthermore, we characterize its effects on the inhibition of the binding of the selective muscarinic ligand [3H]QNB as well as inhibition of the total [3H]inositol phosphate accumulation in male rat hippocampus.
Methods
Venom
Micrurus lemniscatus crude venom was obtained from the Laboratory of Herpetology, Butantan Institute, São Paulo, Brazil. The venom was a pool of several specimens collected in the Southeast region of Brazil. It was lyophilized and stored dry at -20°C until use.
Animals
The conduct and procedures involving animal experiments were approved by the Butantan Institute Committee for Ethics in Animal Experiments (license number CEUAIB 1100/13) in compliance with the recommendations of the National Council for the Control of Animal Experimentation of Brazil (CONCEA). All efforts were made to minimize animal suffering.
Male Wistar rats (90 day old; 324.8 ± 3.1 g, n = 52), coming from the Central Animal Laboratory of the Butantan Institute, were housed in a polypropylene box (inside length × width × height = 56 cm × 35 cm × 19 cm) (5 animals/cage containing wood shavings) within a ventilated container (Alesco Ind. Com Ltda, Brazil) under controlled temperature (23 ± 2oC), relative humidity (65 ± 1%) and 12-h light/12-h dark cycle (lights on at 6:00 a.m.). The animals were allowed to feed and to drink water ad libitum.
Drugs and Radiochemicals
Carbachol (carbamylcholine chloride), lithium chloride, myo-inositol, pirenzepine (pirenzepine hydrochloride), HPLC grade acetonitrile, and trifluoroacetic acid were obtained from Sigma Chemical Co. (USA). The 7,7-dimethyl-5,8-eicosadienoic acid (DEDA) was obtained from Abcam Laboratories (USA). The [3H]quinuclidinyl benzylate (specific activity 47 Ci/mmol) was obtained from New England Nuclear (USA). Myo-[1,2-3H] inositol (specific activity 18 Ci/mmol) was purchased from Amersham (UK). The OptiPhase HiSafe 3 was obtained from Perkin Elmer (UK). The AG( 1-X8 (200 - 400 mesh) resin was purchased from Bio Rad Laboratories (Richmond, CA, USA). All other drugs and reagents were obtained from Merck (Darmstadt, Germany) or Sigma Chemical Co.
RP-HPLC Purification of Micrurus lemniscatus Venom
Micrurus lemniscatus crude venom (30 mg) was diluted in 3 mL of Milli-Q water and purified as described by da Silva et al. [2626. da Silva DC, de Medeiros WA, Batista IFC, Pimenta DC, Lebrun I, Abdalla FMF, et al. Characterization of a new muscarinic toxin from the venom of the Brazilian coral snake Micrurus lemniscatus in rat hippocampus. Life Sci. 2011 Dec 19;89(25-26):931-8.]. Briefly, after filtration in a 0.45-μm filter (Millipore), 800-μL samples (400 μg) were applied to a C8 reversed-phase column (Shim-Pack; 4.6 mm× 250 mm, 5-μm particle) coupled to a HP 1100 series HPLC system. The elution used a flow rate of 1 mL.min-1, and this was monitored at 214 nm. The proteins were eluted with a linear gradient of trifluoroacetic acid (TFA) (solvent A) (0.1% TFA in water) and acetonitrile (solvent B) (90% acetonitrile + 10% A) from 10% to 35% of B over 80 min. Thirty-one fractions were manually collected according to their absorbance. Fractions that contained the Mlx-8 were purified in a C18 RP-HPLC column (SUPELCOSIL-LC-18-DB 15 cm × 4.6 mm cat. no. 58348) eluted with a gradient of 0 to 90% acetonitrile (ACN JT Baker) containing 0.1% of TFA. Solvent A was 0.1% TFA (in Milli-Q water), and solvent B was 90% ACN with 0.1% TFA. The purified Mlx-8 toxin was assayed for its ability to inhibit the binding of selective muscarinic ligand [3H]QNB. Moreover, the total [3H]inositol phosphate was also determined for pharmacological performance.
Mass Spectrometry
The samples were mixed in a saturated aqueous solution containing sulfuric acid (1:1 v/v) and synergistic acid (90% of 2,5-dihydroxybenzoic acid and 10% of α-cyano-4-hydroxycarnamic acid) as described by da Silva et al. [2626. da Silva DC, de Medeiros WA, Batista IFC, Pimenta DC, Lebrun I, Abdalla FMF, et al. Characterization of a new muscarinic toxin from the venom of the Brazilian coral snake Micrurus lemniscatus in rat hippocampus. Life Sci. 2011 Dec 19;89(25-26):931-8.]. Briefly, a cation exchange step was added immediately before the analysis on an AnchorChip 600/384 MTP plates. This was co-crystallized at ambient temperature, and the samples were processed with reagents from Sigma-Aldrich (USA). The α-cyano-4-hydroxycinnamic acid MALDI matrix was processed with Millipore® C18 Ziptips. MALDI-TOF mass spectrometry was performed on an Axima Performance MALDI-TOF/TOF (Shimadzu, Japan) using an α-cyano-4-hydroxycinnamic acid as the matrix. The peptide profile was acquired in linear mode with 75 V laser power.
N-terminal Sequence Determination
The purified protein (500 pmol) was dissolved in ACN 37% to determine the N-terminus sequence as described by da Silva et al. [2626. da Silva DC, de Medeiros WA, Batista IFC, Pimenta DC, Lebrun I, Abdalla FMF, et al. Characterization of a new muscarinic toxin from the venom of the Brazilian coral snake Micrurus lemniscatus in rat hippocampus. Life Sci. 2011 Dec 19;89(25-26):931-8.]. Briefly, this was processed with Edman degradation using a PPSQ-21A Protein Sequencer following the manufacturer’s instructions and protocols (Shimadzu, Japan). The N-terminal sequence was analyzed with the Expert Protein Analysis System (http://www.expasy.org/) and the Blast platform was adopted to perform the sequence search (<https://web.expasy.org/tmp/1week/blastf25027.html>). The sequences alignments were performed with ClustalW (http://www.ebi.ac.uk/clustalw/).
Phospholipase A2 Activity
The purified Mlx-8 toxin and crude venom were obtained from Micrurus lemniscatus and were assayed for phospholipase A2 activity using 4-nitro-3 (octanoloxy) benzoic acid (NOBA) as the substrate [3232. Holzer M, Mackessy SP. An aqueous endpoint assay of snake venom phospholipase A2. Toxicon. 1996 Oct;34(10):1149-55.]. Different protein concentrations in 20 µL of 150 mM NaCl were incubated with 20 µL of 3 mM NOBA in acetonitrile and 100 µL of a buffer containing 10 mM Tris, 10 mM CaCl2, and 100 mM NaCl, pH 8. Plates were incubated for 30, 40, and 60 min at 37°C, and absorbance was recorded at 425 nm using a Spectra Max 190 plate reader (Molecular Devices, USA) after addition of 20 µL of 2.5% Triton X-100. The results were expressed as mmol/min/mg of protein of one experiment performed in triplicate.
In another series of experiments, 200 μM DEDA, a PLA2 inhibitor, was incubated in the presence of 2.8 μg Mlx-8 for 60 min and the phospholipase A2 activity was determined as described above.
[3H]Quinuclidinyl Benzilate ([3H]QNB) Binding Assay
The hippocampus membrane was collected from six animals per experiment and was prepared as described previously [3333. Cardoso CC, Pereira RT, Koyama CA, Porto CS, Abdalla FMF. Effects of estrogen on muscarinic acetylcholine receptors in the rat hippocampus. Neuroendocrinology. 2004;80(6):379-86.]. Briefly, the hippocampi were isolated from rats, minced, and homogenized in 25 mM Tris-HCl, pH 7.4 (containing 0.3 M sucrose, 5 mM MgCl2, 1 mM EDTA, and 1 mM phenylmethylsulfonyl fluoride) with a Ultra-Turrax homogenizer (T-25, Ika Labortechnik, Staufen, Germany). The homogenate was centrifuged at 1,000 × g for 10 min. The supernatant was then filtered through two layers of gauze and centrifuged at 100,000 × g for 60 min. The final 100,000 × g pellet was re-suspended in 1 mL of 25 mM Tris-HCl, pH 7.4 (containing 5 mM MgCl2, 1 mM EDTA, and 1 mM phenylmethylsulfonyl fluoride) using a Dounce homogenizer and stored at -80oC. All procedures were performed at 4oC, and all solutions contained freshly added 1 mM phenylmethylsulfonyl fluoride to inhibit proteolysis. The total protein concentration in the membrane preparations was determined with a protein reagent assay (Bio Rad Laboratories Inc., USA).
Competition binding experiments were performed as previously described [3434. Abdalla FMF, Abreu LC, Porto CS. Effect of estrogen on intracellular signaling pathways linked to activation of M2- and M3-muscarinic acetylcholine receptors in the rat myometrium. Mol Cell Endocrinol. 2000 Feb 25;160(1):17-24.]. Briefly, the hippocampus membrane solution (80 µg protein/mL) was incubated with [3H]QNB (concentration near the KD values) [3333. Cardoso CC, Pereira RT, Koyama CA, Porto CS, Abdalla FMF. Effects of estrogen on muscarinic acetylcholine receptors in the rat hippocampus. Neuroendocrinology. 2004;80(6):379-86.] for 1 h at 30oC in the absence and presence of increasing concentrations of Mlx-8 toxin or muscarinic antagonist pirenzepine (control). In another series of experiments, 1 μM Mlx-8 toxin was incubated with [3H]QNB in the absence or presence of 200 μM DEDA [2727. Miyoshi S, Tu AT. Phospholipase A2 from Naja naja sputatrix venom is a muscarinic acetylcholine receptor inhibitor. Arch Biochem Biophys. 1996 Apr 1;328(1):17-25., 3535. Chen M, Xiao CY, Hashizume H, Abiko Y. Phospholipase A2 is not responsible for lysophosphatidylcholine-induced damage in cardiomyocytes. Am J Physiol. 1998 Nov;275(5):H1782-7., 3636. Fatehi M, Rowan EG, Harvey AL. The effects of two phospholipase A2 inhibitors on the neuromuscular blocking activities of homologous phospholipases A2 from the venom of Pseudechis australis, the Australian king brown snake. Toxicon. 1995 Dec;33(12):1633-43.] as described above.
Competition binding data were analyzed using a weighted nonlinear least-squares interactive curve-fitting program GraphPad Prism (GraphPad Prism Software Inc, USA). A mathematical model for one or two binding sites was applied. The inhibition constant (Ki) was determined from competition curves using the Cheng and Prusoff equation [3737. Cheng Y, Prusoff WH. Relationship between the inhibition constant (K1) and the concentration of inhibitor which causes 50 per cent inhibition (I50) of an enzymatic reaction. Biochem Pharmacol. 1973 Dec 1;22:3099-108.]. The potency of the antagonist was expressed via the negative logarithm of their Ki value (pKi).
Measurement of Total [3H]inositol Phosphate
Hippocampi were isolated from rats and washed with a nutrient solution of the following composition (mM): NaCl 118.00; KCl 4.78; CaCl2 2.43; MgSO4 1.16; NaHCO3 23.80; KH2PO4 1.17; and glucose 2.92 (pH 7.4). Hippocampus slices (100 mg of tissue) were allowed to equilibrate for 10 min in nutrient solution at 37oC under constant shaking. The slices were incubated for 40 min with 1 µCi myo-[3H]inositol and for an additional 30 min with 10 mM lithium chloride with myo-[3H]inositol. Tissues were then incubated in the absence (basal level) or presence of carbachol (CCh, 10-8 to 10-3 M) for 40 min. Mlx-8 toxin (10-7 and 10-6 M) was added 5 min prior to incubation with CCh (10-5 M). Tissues were washed three times with nutrient solution, transferred to 2 mL of methanol:chloroform (2:1 v/v) at 4oC, and homogenized with a Ultra-Turrax T25 homogenizer at 9,500 rpm. Chloroform (0.62 mL) and H2O (0.93 mL) were added to the homogenate, and the solution was centrifuged for 10 min at 2,000 x g and 4oC to separate the aqueous and organic phases [3838. Fox AW, Abel PW, Minneman KP. Activation of alpha 1-adrenoceptors increases [3H]inositol metabolism in rat vas deferens and caudal artery. Eur J Pharmacol. 1985 Oct 8;116(1-2):145-52., 3939. Pereira RT, Porto CS, Godinho RO, Abdalla FMF. Effects of estrogen on intracellular signaling pathways linked to activation of muscarinic acetylcholine receptors and on acetylcholinesterase activity in rat hippocampus. Biochem Pharmacol. 2008 May1;75(9):1827-34.].
Total [3H]inositol phosphate was measured as previously described [4040. Ascoli M, Pignataro OP, Segaloff DL. The inositol phosphate/diacylglycerol pathway in MA-10 Leydig tumor cells. Activation by arginine vasopressin and lack of effect of epidermal growth factor and human choriogonadotropin. J Biol Chem. 1989 Apr 25;264(12):6674-81.] with the following modification: the aqueous layer was mixed with 1 mL anion-exchange resin (Dowex AG-X8, formate form, 200-400 mesh) allowed to equilibrate for 30 min at room temperature. It was then centrifuged at 1,000 × g for 5 min at 4oC. The resin was sequentially washed with myo-inositol (4 mL) and 5 mM sodium tetraborate/60 mM sodium formate (2 mL). Subsequently, the resin was incubated for 30 min at room temperature with 2 mL of 0.1M formic acid/1M ammonium formate. The total [3H]inositol phosphate was eluted and placed in scintillation vials containing OptiPhase HiSafe 3. The amount of radioactivity was determined in a scintillation β-counter (LS 6500 IC, Beckman). Total [3H]inositol phosphate was expressed as dpm/mg tissue.
Statistical Analysis
Data were expressed as the mean ± S.E.M. Data were analyzed by ANOVA followed by Newman-Keuls test for multiple comparisons or via a two-tailed Student’s t-test to compare a response between the two groups [4141. Snedecor GW, Cochran WG. Statistical Methods. 7th edn. Iowa State University Press Ames USA,. Statistical methods, p. 89-233; 1980.]. P values < 0.05 were considered to be significant.
Results
Biochemical Characterization of the Mlx-8 Toxin
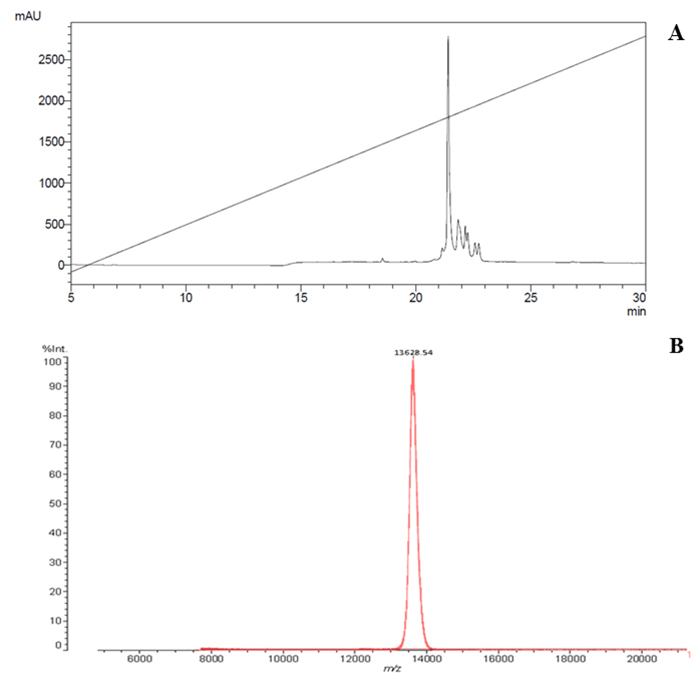
Figure 1 presents the RP-HPLC profile of the Micrurus lemniscatus venom. The fraction that contains the Mlx-8 was purified in a C18 RP-HPLC column (Fig. 2A): 180 µg of Mlx-8 toxin was obtained from 30 mg of the crude venom. Mlx-8 toxin was collected and had its molecular mass verified by MALDI-TOF. The MS profile was 13,628 as shown for the peak in Figure 2B.
Crude venom purified on high performance liquid chromatography (HPLC) using a C8 column on a Prominence binary system (Shimadzu). The venom components were eluted with a flow rate of 1 mL/min with solvents A [0.1% trifluoroacetic acid (TFA) in deionized H2O] and B (90% acetonitrile, 0.1% TFA in deionized H2O) with gradient from 10 to 35% of solution B, represented by the trace. The absorbance was read at 214 nm. In the highlighted area, the arrow indicates the fraction that contains the Mlx-8 with a retention time of 61 minutes in 75 minutes run.
(A) The fraction that contains the Mlx-8 was purified on HPLC-RF using a C18 column eluted under a flow rate of 1 mL/min with solvents A and B from 0 to 100% acetonitrile in 0.1% TFA aqueous solution represented by the trace. The absorbance was read at 214 nm. Only the highest peak was collected, thus removing the contaminants from the sample and guaranteeing its purity. (B) Mass spectrum of the Mlx-8 toxin obtained via mass spectrometry technique in MALDI-TOF ionization mode. The toxin was analyzed using the saturated sinapinic acid matrix solution (1:1 v/v) and deposited directly onto MCP AnchorChip 600/384 plates. This was co-crystallized at room temperature. After ionization, the Mlx-8 toxin molecules were transformed into ions and counted by the detector as a function of their mass/charge (m/z) and their molecular mass was identified.
The Mlx-8 N-terminal sequence was determined by Edman degradation and the following sequence: NLYQFKNMIQCTNTRSWLDFADYGCYCGRGGSGT (Fig. 3) was obtained. The sequence determination showed that the Mlx-8 presents high similarity to other toxins from Elapidae such as the PLA2 from Micrurus lemniscatus carvalhoi [99. Aird SD, da Silva JrNJ, Qiu L, Villar-Briones A, Saddi VA, Telles MPC, et al. Coralsnake venomics: analyses of venom gland transcriptomes and proteomes of six brazilian taxa. Toxins (Basel). 2017 Jun;9(6):pii: E187.] and from Lemnitoxin from Micrurus lemniscatus [3131. Casais-e-Silva LL, Teixeira CF, Lebrun I, Lomonte B, Alape-Giron A, Gutierrez JM. Lemnitoxin, the major component of Micrurus lemniscatus coral snake venom, is a myotoxic and pro-inflammatory phospholipase A2. Toxicol Lett. 2016 Aug 22;257:60-71.]. In addition, this sequence was analyzed against the public protein data bank to check for similarities with known proteins. Besides that, matches were identified with toxins from Naja kaouthia, N. sagittifera, N. atra, N. sputatrix, M. tener, Pseudechis australis, P. papuanus and M. nigrocintus venoms (Fig. 3).
Amino acid multiple sequence alignment. The Mlx-8 toxin obtained from Micrurus lemniscatus venom was aligned with other PLA2 from Micrurus lemniscatus carvalhoi [99. Aird SD, da Silva JrNJ, Qiu L, Villar-Briones A, Saddi VA, Telles MPC, et al. Coralsnake venomics: analyses of venom gland transcriptomes and proteomes of six brazilian taxa. Toxins (Basel). 2017 Jun;9(6):pii: E187.] and from Lemnitoxin from Micrurus lemniscatus [3131. Casais-e-Silva LL, Teixeira CF, Lebrun I, Lomonte B, Alape-Giron A, Gutierrez JM. Lemnitoxin, the major component of Micrurus lemniscatus coral snake venom, is a myotoxic and pro-inflammatory phospholipase A2. Toxicol Lett. 2016 Aug 22;257:60-71.]. In addition, this sequence was analyzed against the public protein data bank to check for similarities with known proteins. Besides that, matches were identified with toxins from Naja Kaouthia, N. sagittifera, N. atra, N. sputatrix, M. tener, Pseudechis australis, P. papuanus and M. nigrocintus venoms.
Phospolipase A2 Activity of the Mlx-8 Toxin
The phospholipase A2 enzymatic activity of 2.8 μg Mlx-8 toxin was determined at different time periods (30, 40 and 60 minutes). The Mlx-8 could hydrolyze the substrate to phospholipase A2, 4-NOBA at all times. The 60-minute time point revealed a greater activity (229.4 ± 14.33, 345.5 ± 9.12, and 582.8 ± 29.3 mmol/min/mg, respectively, 30, 40 and 60 minutes).
The amount of 2.8 μg Mlx-8 in the presence of 200 μM DEDA for 60 min decreased 51% (285.57 ± 7.47 mmol/min/mg) the phospholipase A2 enzymatic activity.
Effect of Mlx-8 Toxin on [3H]QNB Binding in Hippocampus Membranes
Figure 4A shows the displacement curves of [3H]QNB bound to hippocampus membranes induced by Mlx-8 toxin and pirezenpine (M1 selective antagonist) [4242. Eglen RM. Muscarinic receptor subtypes in neuronal and non-neuronal cholinergic function. Auton Autacoid Pharmacol. 2006 Jul;26(3):219-33.]. Analysis of the displacement curves induced by Mlx-8 toxin and pirenzepine indicated a statistical preference for a one-site rather than a two-site fit (F-test, GraphPad Prism program). The pKi values obtained from the analysis of Mlx-8 and antagonist displacement curves via one-site fit and their respective Hill slopes (nH) were 7.32 ± 0.15, n = 4 (nH = 1.14 ± 0.13) and 5.84 ± 0.18, n = 4 (nH = 0.94 ± 0.15) for Mlx-8 and pirenzepine, respectively.
(A) Displacement curves of [3H]QNB bound to hippocampus membranes from male rats induced by Mlx-8 toxin (⯀) and muscarinic acetylcholine receptor antagonist pirezenpine (●). (B) Displacement of [3H]QNB bound to hippocampus membrane by 1 µM Mlx-8 toxin obtained from Micrurus lemniscatus venom in the absence and presence of 200 µM DEDA, an inhibitor of phospholipase A2. The data are plotted as percentages of the binding in the absence of Mlx-8 or muscarinic acetylcholine receptor antagonists. Each point and vertical line represents the mean ± S.E.M. of n = 4, performed in duplicate. Different letters indicate statistical significance (p < 0.05; ANOVA, Newman–Keuls test).
The 200 µM DEDA had no effect on the inhibition of [3H]QNB binding in hippocampus membranes when using 1 µM Mlx-8 (Fig. 4B).
Effects of Carbachol and Mlx-8 Toxin on total [3H]inositol Phosphate Accumulation
The basal level of the total [3H]inositol phosphate in rat hippocampus was 69.12 ± 5.80 dpm/mg tissue, n = 12. The cholinergic agonist carbachol (CCh, 10-8 M to 10-3 M) caused a concentration-dependent increase in the hippocampal total [3H]inositol phosphate accumulation (Fig. 5A). The maximum total [3H]inositol phosphate accumulation was obtained with 10-5 M CCh (38.80 ± 4.50% above basal, n = 4) (Fig. 5A).
(A) Concentration-effect curve of carbachol (CCh) on total [3H]inositol phosphate accumulation. (B) Effect of the Mlx-8 toxin on total [3H]inositol phosphate accumulation induced by 10-5 M CCh in the hippocampi from male rats. Each point and vertical line represent the mean ± S.E.M. of n = 3. Different letters indicate statistical significance (p < 0.05; ANOVA, Newman-Keuls test).
The purified Mlx-8 toxin (10-7 and 10-6 M) obtained from Micrurus lemniscatus snake venom did alter the total [3H]inositol phosphate accumulation induced by 10-5 M CCh in a concentration-dependent manner. The Mlx-8 toxin caused a blockade of 43.31 ± 8.86% (n = 3) and 97.42 ± 2.02% (n = 3) at 10-7 and 10-6 M, respectively, on [3H]inositol phosphate accumulation induced by 10-5 M CCh in the rat hippocampus (Fig. 5B). In the absence of CCh, the Mlx-8 toxin (10-6 M) did not alter the total [3H]inositol phosphate accumulation in the hippocampus (Fig. 5B) (p < 0.05; ANOVA, Newman-Keuls test).
Discussion
The results show for the first time that the mAChRs function is drastically affected by Mlx-8 toxin, a muscarinic ligand with phospholipase A2 activity obtained from Micrurus lemniscatus venom. This species is in the Elapidae family, and its toxin can inhibit binding of the selective muscarinic ligand [3H]QNB in rat membranes from the hippocampus. Furthermore, the toxin also inhibited [3H]inositol phosphate accumulation in the hippocampus.
Muscarinic toxins that affect ligand binding to mAChR have been isolated from mamba venom [see 4343. Lahiani A, Yavin E, Lazarovici P. The molecular basis of toxins interactions with intracellular signaling via discrete portals. Toxins. 2017 Mar 16;9(3): pii: E107.-4545. Servent D, Blanchet G, Mourier G, Marquer C, Marcon E, Fruchart-Gaillard C. Muscarinic toxins. Toxicon. 2011 Nov;58(6-7):455-63., for review]. The structures of this group of toxins are somewhat similar to the postsynaptic neurotoxins and consist of three polypeptide loops (3FTx). They all share roughly the same number of amino acids (63-66 AA) and molecular weight (about 7 kDa). However, the molecular mass of Mlx-8 (13.6 kDa) from the venom of Micrurus lemniscatus seen here is clearly different from muscarinic toxins. In this way, a similar molecular mass of Mlx-8 was observed versus muscarinic toxins with phospholipase A2 activity obtained from Naja naja sputatrix (13.6 kDa) [2727. Miyoshi S, Tu AT. Phospholipase A2 from Naja naja sputatrix venom is a muscarinic acetylcholine receptor inhibitor. Arch Biochem Biophys. 1996 Apr 1;328(1):17-25.] and Naja atra (13.3 kDa) [2929. Huang LF, Zheng JB, Xu Y, Song HT, Yu CX. A snake venom phospholipase A2 with high affinity for muscarinic acetylcholine receptors acts on guinea pig ileum. Toxicon. 2008 May;51(6):1008-16.]. This indicates that Mlx-8 may belong to a group of snake PLA2-toxins.
Indeed, when N-terminal analysis and alignment of Mlx-8 (NLYQFKNMIQCTNTRSWL-DFADYG-CYCGRGGSGT) was determined and compared to other proteins with muscarinic activity, the data revealed a high similarity to Elapidae venom proteins including a neural phospholipase A2 muscarinic inhibitor from Naja naja sputatrix (NLYQFKNMIQCTVPNR) [2727. Miyoshi S, Tu AT. Phospholipase A2 from Naja naja sputatrix venom is a muscarinic acetylcholine receptor inhibitor. Arch Biochem Biophys. 1996 Apr 1;328(1):17-25.] and Naja atra (NLYQFKNMIQCTVPSR) [2929. Huang LF, Zheng JB, Xu Y, Song HT, Yu CX. A snake venom phospholipase A2 with high affinity for muscarinic acetylcholine receptors acts on guinea pig ileum. Toxicon. 2008 May;51(6):1008-16.]. Recently, a toxin named Lemnitoxin was isolated from Micrurus lemniscatus venom and shown to be a PLA2 with myotoxic and pro-inflammatory activity [3131. Casais-e-Silva LL, Teixeira CF, Lebrun I, Lomonte B, Alape-Giron A, Gutierrez JM. Lemnitoxin, the major component of Micrurus lemniscatus coral snake venom, is a myotoxic and pro-inflammatory phospholipase A2. Toxicol Lett. 2016 Aug 22;257:60-71.]. The N-terminal comparison of the Mlx-8 toxin with Lemnitoxin (NLYQFKNMIQCTNTRSWL-DFADYG-CYCGYGGSGT) revealed an almost identical amino acid sequence between both toxins suggesting either a very similar toxin or an isoform. Other studies are needed to prove this issue. The Mlx-8 toxin was strongly expected to have phospholipase A2 activity in view of the biochemical properties described above. In fact, Mlx-8 shows phospholipase A2 enzymatic activity.
The mAChRs mediate a wide range of functions of the parasympathetic nervous system both centrally and peripherally. Different experimental approaches have shown that mAChRs are present in all organs, tissues, or cell types [see 4646. Lebois EP, Thorn C, Edgerton JR, Popiolek M, Xi S. Muscarinic receptor subtype distribution in the central nervous system and relevance to aging and Alzheimer's disease. Neuropharmacology. 2018 Jul 1;136(Pt C):362-73., for review]. The muscarinic actions of acetylcholine are mediated by five distinct mAChR subtypes (M1 to M5) [4747. Caulfield MP. Muscarinic receptors--characterization, coupling and function. Pharmacol Ther. 1993 Jun;58(3):319-79.-4949. Wess J. Molecular biology of muscarinic acetylcholine receptors. Crit Rev Neurobiol. 1996;10(1):69-99.]. The M1, M3, and M5 subtypes couple primarily to phospholipase C-mediated phosphoinositide hydrolysis. On the other hand, the M2 and M4 subtypes couple primarily to adenylyl cyclase inhibition [see 5050. Ballinger EC, Ananth M, Talmage DA, Role LW. Basal forebrain cholinergic circuits and signaling in cognition and cognitive decline. Neuron. 2016 Sep 21;91(6):1199-218., for review]. To characterize the effect of Mlx-8 toxin on mAChRs at the protein level, Mlx-8 and the M1 selective muscarinic antagonist pirenzepine were examined for their ability to compete with [3H]QNB for binding sites in the hippocampus membrane. The pKi of the Mlx-8 (7.32) was higher than that obtained by pirenzepine (5.84). Moreover, the Hill slope coefficients calculated for Mlx-8 and pirenzepine did not differ from unity. These data support the idea that Mlx-8 has affinity for mAChRs. Further experimental approaches are needed to clarify the mechanisms involved and the functional significance of Mlx-8 on mAChRs.
This study focused only on the phospholipase C-mediated phosphoinositide hydrolysis in hippocampal tissue because the population of M1 receptors is predominant in the rat hippocampus [see 4646. Lebois EP, Thorn C, Edgerton JR, Popiolek M, Xi S. Muscarinic receptor subtype distribution in the central nervous system and relevance to aging and Alzheimer's disease. Neuropharmacology. 2018 Jul 1;136(Pt C):362-73., for review]. The Mlx-8 toxin obtained from Micrurus lemniscatus venom reduced the response to carbachol on total [3H]inositol phosphate accumulation in a concentration-dependent manner. In the absence of carbachol, 1 µM Mlx-8 did not alter the level of total [3H]inositol phosphate. These studies collectively indicate that the Mlx-8 toxin blocked the intracellular signaling pathway linked to activation of mAChRs in rat hippocampus. Interestingly, the Mlx-8 toxin is quite different from the toxin obtained from Naja atra venom [2929. Huang LF, Zheng JB, Xu Y, Song HT, Yu CX. A snake venom phospholipase A2 with high affinity for muscarinic acetylcholine receptors acts on guinea pig ileum. Toxicon. 2008 May;51(6):1008-16.]. Although both exhibit similarity of the N-terminal amino acid sequence and molecular mass, Mlx-8 (1 µM) inhibits the total [3H]inositol phosphate accumulation (97%) induced by muscarinic agonist carbachol while the Naja atra venom promotes contraction in the ileum of guinea pig via mAChRs [2929. Huang LF, Zheng JB, Xu Y, Song HT, Yu CX. A snake venom phospholipase A2 with high affinity for muscarinic acetylcholine receptors acts on guinea pig ileum. Toxicon. 2008 May;51(6):1008-16.]. Whether the Mlx-8 toxin plays a role in other intracellular signaling pathways coupled to mAChRs remains to be explored.
Specific binding membrane receptor proteins of venom phospholipase A2 have been shown. For example, vipoxin (a minor PLA2 from Vipera russelli venom) can bind to amine receptors on rat brain [5151. Freedman JE, Snyder SH. Vipoxin. A protein from Russell's viper venom with high affinity for biogenic amine receptors. J Biol Chem. 1981 Dec 25;256(24):13172-9.]. OS2 is a single-chain PLA2 isolated from Oxyuranus scutellatus venom and associates selectively with rat brain membrane proteins termed N-type receptors [5252. Lambeau G, Barhanin J, Schweitz H, Qar J, Lazdunski M. Identification and properties of very high affinity brain membrane-binding sites for a neurotoxic phospholipase from the taipan venom. J Biol Chem. 1989 Jul 5;264(19):11503-10.]. Moreover, there is evidence suggesting that the ability to interact with nicotinic acetylcholine receptors may be a general property of several snakes PLA2 from venoms [5353. Vulfius CA, Kasheverov IE, Starkov VG, Osipov AV, Andreeva TV, Filkin SY, et al. Inhibition of nicotinic acetylcholine receptors, a novel facet in the pleiotropic activities of snake venom phospholipases A2. PLoS One. 2014 Dec 18;9(12): e115428. , 5454. Vulfius CA, Kasheverov IE, Kryukova EV, Spirova EN, Shelukhina IV, Starkov VG, et al. Pancreatic and snake venom presynaptically active phospholipases A2 inhibit nicotinic acetylcholine receptors. PLoS One. 2017 Oct 12;12(10):e0186206.]. To check the ability of the PLA2 isolated from Micrurus lemniscatus (Mlx-8) to interact with mAChRs, the inhibitor of cobra venom phospholipases A2 activity DEDA, an analogue of arachidonic acid that contains two cis double bonds as well as two methyl groups [5555. Cohen N, Weber G, Banner BL, Welton AF, Hope WC, Crowley H, et al. Analogs of arachidonic acid methylated at C-7 and C-10 as inhibitors of leukotriene biosynthesis. Prostaglandins. 1984 Apr;27(4):553-62.], was used in the present study. Indeed, the phospholipase A2 enzymatic activity of Mlx-8 in the presence of DEDA decreased 51%. Interestingly, there was no impact on inhibition of [3H]QNB binding in hippocampus membranes via DEDA, suggesting that the inhibition of the phospholipase A2 activity of the venom did not alter its ability to bind and displace [3H]QNB binding. Similarly, DEDA did not also block the mAChRs binding in muscarinic toxin with PLA2 activity obtained from Naja naja sputatrix venom [2727. Miyoshi S, Tu AT. Phospholipase A2 from Naja naja sputatrix venom is a muscarinic acetylcholine receptor inhibitor. Arch Biochem Biophys. 1996 Apr 1;328(1):17-25.]. On the other hand, the inhibitor of phospholipases A2 activity p-bromophenacyl bromide, which modifies the histidine residue in the active site of PLA2, eliminated both PLA2 activity and [3H]QNB binding [2727. Miyoshi S, Tu AT. Phospholipase A2 from Naja naja sputatrix venom is a muscarinic acetylcholine receptor inhibitor. Arch Biochem Biophys. 1996 Apr 1;328(1):17-25.]. Thus, only DEDA showed no effect on mAChRs binding when used [2828. Miyoshi S, Tu AT. Muscarinic acetylcholine receptor (mAChR) inhibitor from snake venom: interaction with subtypes of human mAChR. Arch Biochem Biophys. 1999 Sep 1;369(1):114-8.].
Micrurus venoms are natural libraries of biologically active molecules that can be used as new drug leads. However, a major obstacle to characterize the components of Micrurus venoms is the minute quantities of material obtained from specimen milking. Thus, despite the large variety of molecules with potential biotechnological application, there is still a great difficulty of their bioprospecting due to the small amount of starting material, low yield and the high cost of traditional purification strategies. In general, this alone explains the small number of animal molecules currently used as drugs. The recent development and use of “omic” tools has become increasingly prominent since they allow an overview of the composition of the venom. In addition, the transcriptome technique, associated with the cloning and heterologous expression of proteins and peptides, enables the production of molecules present in the gland or specialized tissue in sufficient quantity for their structural and functional analysis. Therefore, these studies enable the application of molecules with relevant biological activity. From this perspective, as regards Mlx-8, further studies will be required to better explore the biological potential of this toxin.
Conclusion
The results of the present work show, for the first time, that mAChRs are also affected by the Mlx-8 toxin, a muscarinic ligand with phospholipase A2 characteristics, obtained from the venom of the Elapidae snake Micrurus lemniscatus, since this toxin was able to compete with muscarinic ligand [3H]QNB in hippocampus from rats. In addition, Mlx-8 also blocked the accumulation of total [3H]inositol phosphate induced by muscarinic agonist carbachol. Thus, Mlx-8 may be a new pharmacological tool for examining muscarinic cholinergic function.
Abbreviations
[3H]QNB: [3H]quinuclidinyl benzilate; 3FTx: three finger; CCh: carbachol; DEDA: 7,7-dimethyl-5,8-eicosadienoic acid; HPLC: high performance liquid chromatography; Ki: inhibition constant; mAChRs: muscarinic acetylcholine receptors; Mlx-8 and MT-Mlα: toxins isolated from Micrurus lemniscatus venom; NOBA: 4-nitro-3 (octanoloxy) benzoic acid; PLA2: phospholipase A2; TFA: trifluoroacetic acid.
Acknowledgements
The authors are grateful to Professor Daniel Carvalho Pimenta (Laboratory of Biochemistry and Biophysics, Butantan Institute, São Paulo, SP, Brazil) for his valuable advice and support with HPLC equipment and mass spectrometry.
References
- 1. Bucaretchi F, Capitani EM, Vieira RJ, Rodrigues CK, Zannin M, da Silva NJ Jr, et al. Coral snake bites (Micrurus spp.) in Brazil: a review of literature reports. Clin Toxicol (Phila). 2016 Mar;54(3):222-34.
- 2. Zheng Y, Wiens JJ. Combining phylogenomic and supermatrix approaches, and a time-calibrated phylogeny for squamate reptiles (lizards and snakes) based on 52 genes and 4162 species. Mol Phylogenet Evol. 2016 Jan;94(Pt B):537-47.
- 3. Lomonte B, Rey-Suárez P, Fernández J, Sasa M, Pla D, Vargas N, et al. Venoms of Micrurus coral snakes: evolutionary trends in compositional patterns emerging from proteomic analyses. Toxicon. 2016 Nov;122:7-25.
- 4. Salazar AM, Vivas J, Sánchez EE, Acosta AR, Ibarra CI, Gil A, et al. Hemostatic and toxinological diversities in venom of Micrurus tener tener, Micrurus fulvius fulvius and Micrurus isozonus coral snakes. Toxicon. 2011 Jul;58(1):35-45.
- 5. Castoe TA, Smith EN, Brown RM, Parkinson CL. Higher-level phylogeny of Asian and American coralsnakes, their placement within the Elapidae (Squamata), and the systematic affinities of the enigmatic Asian coralsnake Hemibungarus calligaster (Wiegmann, 1834). Zool J Linnean Soc. 2007 Dec;151(4):809-31.
- 6. da SilvaJr NJ. As Cobras-Corais do Brasil: Biologia, Taxonomia, Venenos e Envenenamentos. 1nd ed. da Silva Jr NJ, editor. Editora da PUC Goiás, Goiânia; 2016.
- 7. Sanz L, Quesada-Bernat S, Ramos T, Casais-e-Silva LL, Corrêa-Netto C, Silva-Haad JJ, et al. New insights into the phylogeographic distribution of the 3FTx/PLA2 venom dichotomy across genus Micrurus in South America. J Proteomics. 2019 May 30;200:90-101.
- 8. Floriano RS, Schezaro‑Ramos R, da Silva JrNJ, Bucaretchi F, Rowan EG, Hyslop S. Neurotoxicity of Micrurus lemniscatus lemniscatus (South American coralsnake) venom in vertebrate neuromuscular preparations in vitro and neutralization by antivenom. Arch Toxicol. 2019 Jul;93(7):2065-86.
- 9. Aird SD, da Silva JrNJ, Qiu L, Villar-Briones A, Saddi VA, Telles MPC, et al. Coralsnake venomics: analyses of venom gland transcriptomes and proteomes of six brazilian taxa. Toxins (Basel). 2017 Jun;9(6):pii: E187.
- 10. Kessler P, Marchot P, Silva M, Servent D. The three‐finger toxin fold: a multifunctional structural scaffold able to modulate cholinergic functions. J Neurochem. 2017 Aug;142(Suppl 2):7-18.
- 11. Ray-Suarez P, Nunez V, Gutiérrez JM, Lomonte B. Proteomic and biological characterization of the venom of the redtail coral snake, Micrurus mipartitus (Elapidae), from Colombia and Costa Rica. J Proteomics. 2011 Dec 21;75(2):655-67.
- 12. Tanaka GD, Furtado MF, Portaro FCV, Sant'Anna OA, Tambourgi DV. Diversity of Micrurus snake species related to their venom toxic effects and the prospective of antivenom neutralization. PLoS Negl Trop Dis. 2010 Mar 9;4(3):e622.
- 13. Vergara I, Pedraza-Escalona M, Paniagua D, Restano-Cassulini R, Zamudio F, Batista CVF, et al. Eastern coral Micrurus fulvius venom toxicity in mice is mainly determined by neurotoxic phospholipases A2 J Proteomics. 2014 Jun 13;105:295-306.
- 14. Hanasaki K, Arita H. Phospholipase A2 receptor: a regulator of biological functions of secretory phospholipase A2 Prostaglandins Other Lipid Mediat. 2002 Aug; 68-69: 71-82.
- 15. de Carvalho ND, Garcia RCT, Ferreira AK, Batista DR, Cassola AC, Maria D, et al. Neurotoxicity of coral snake phospholipases A2 in cultured rat hippocampal neurons. Brain Res. 2014 Mar 13;1552:1-16.
- 16. Zambelli VO, Picolo G, Fernandes CAH, Fontes MRM, Cury Y. Secreted phospholipases A₂ from animal venoms in pain and analgesia. Toxins (Basel). 2017 Dec 19;9(12):pii: E406.
- 17. Ciscotto PH, Rates B, Silva DA, Richardson M, Silva LP, Andrade H, et al. Venomic analysis and evaluation of antivenom cross-reactivity of South American Micrurus species. J Proteomics. 2011 Aug 24;74(9):1810-25.
- 18. Kini RM, Evans HJ. Structure-function relationships of phospholipases. The anticoagulant region of phospholipases A2 J Biol Chem. 1987 Oct 25;262(30):14402-7.
- 19. Bradley KN, Rowan EG, Harvey AL. Effects of muscarinic toxins MT2 and MT7, from green mamba venom, on m1, m3 and m5 muscarinic receptors expressed in Chinese hamster ovary cells. Toxicon. 2003 Feb;41(2):207-15.
- 20. Diehl F, Furstenau de Oliveira L, Sanchez G, Camboim C, de Oliveira Alvares L, Lanziotti VB, et al. Facilitatory effect of the intra-hippocampal pre-test administration of MT3 in the inhibitory avoidance task. Behav Brain Res. 2007 Feb 27;177(2):227-31.
- 21. Ferreira AR, Furstenau L, Blanco C, Kornisiuk E, Sanchez G, Daroit D, et al. Role of hippocampal M1 and M4 muscarinic receptor subtypes in memory consolidation in the rat. Pharmacol Biochem Behav. 2003 Jan;74:411-5.
- 22. Lyukmanova EN, Shenkarev ZO, Shulepko MA, Paramonov AS, Chugunov AO, Janickova H, et al. Structural insight into specificity of interactions between nonconventional three-finger weak toxin from Naja kaouthia (WTX) and Muscarinic Acetylcholine Receptors. J Biol Chem. 2015 Sep 25;290(39):23616-30.
- 23. Olianas MC, Adem A, Karlsson E, Onali P. Action of the muscarinic toxin MT7 on agonist-bound muscarinic M1 receptors. Eur J Pharmacol. 2004 Mar 8;487(1-3):65-72.
- 24. Olianas MC, Maullu C, Adem A, Mulugeta E, Karlsson E, Onali P. Inhibition of acetylcholine muscarinic M1 receptor function by the M1-selective ligand muscarinic toxin 7 (MT-7). Br J Pharmacol. 2000 Oct;131(3):447-52.
- 25. Servent D, Fruchart-Gaillard C. Muscarinic toxins: tools for the study of the pharmacological and functional properties of muscarinic receptors. J Neurochem. 2009 Jun;109:1193-202.
- 26. da Silva DC, de Medeiros WA, Batista IFC, Pimenta DC, Lebrun I, Abdalla FMF, et al. Characterization of a new muscarinic toxin from the venom of the Brazilian coral snake Micrurus lemniscatus in rat hippocampus. Life Sci. 2011 Dec 19;89(25-26):931-8.
- 27. Miyoshi S, Tu AT. Phospholipase A2 from Naja naja sputatrix venom is a muscarinic acetylcholine receptor inhibitor. Arch Biochem Biophys. 1996 Apr 1;328(1):17-25.
- 28. Miyoshi S, Tu AT. Muscarinic acetylcholine receptor (mAChR) inhibitor from snake venom: interaction with subtypes of human mAChR. Arch Biochem Biophys. 1999 Sep 1;369(1):114-8.
- 29. Huang LF, Zheng JB, Xu Y, Song HT, Yu CX. A snake venom phospholipase A2 with high affinity for muscarinic acetylcholine receptors acts on guinea pig ileum. Toxicon. 2008 May;51(6):1008-16.
- 30. Oliveira DA, Harasawa C, Seibert CS, Casais e Silva LL, Pimenta DC, Lebrun I, et al. Phospholipases A2 isolated from Micrurus lemniscatus coral snake venom: behavioral, electroencephalographic, and neuropathological aspects. Brain Res Bull. 2008 Mar 28;75(5):629-39.
- 31. Casais-e-Silva LL, Teixeira CF, Lebrun I, Lomonte B, Alape-Giron A, Gutierrez JM. Lemnitoxin, the major component of Micrurus lemniscatus coral snake venom, is a myotoxic and pro-inflammatory phospholipase A2 Toxicol Lett. 2016 Aug 22;257:60-71.
- 32. Holzer M, Mackessy SP. An aqueous endpoint assay of snake venom phospholipase A2 Toxicon. 1996 Oct;34(10):1149-55.
- 33. Cardoso CC, Pereira RT, Koyama CA, Porto CS, Abdalla FMF. Effects of estrogen on muscarinic acetylcholine receptors in the rat hippocampus. Neuroendocrinology. 2004;80(6):379-86.
- 34. Abdalla FMF, Abreu LC, Porto CS. Effect of estrogen on intracellular signaling pathways linked to activation of M2- and M3-muscarinic acetylcholine receptors in the rat myometrium. Mol Cell Endocrinol. 2000 Feb 25;160(1):17-24.
- 35. Chen M, Xiao CY, Hashizume H, Abiko Y. Phospholipase A2 is not responsible for lysophosphatidylcholine-induced damage in cardiomyocytes. Am J Physiol. 1998 Nov;275(5):H1782-7.
- 36. Fatehi M, Rowan EG, Harvey AL. The effects of two phospholipase A2 inhibitors on the neuromuscular blocking activities of homologous phospholipases A2 from the venom of Pseudechis australis, the Australian king brown snake. Toxicon. 1995 Dec;33(12):1633-43.
- 37. Cheng Y, Prusoff WH. Relationship between the inhibition constant (K1) and the concentration of inhibitor which causes 50 per cent inhibition (I50) of an enzymatic reaction. Biochem Pharmacol. 1973 Dec 1;22:3099-108.
- 38. Fox AW, Abel PW, Minneman KP. Activation of alpha 1-adrenoceptors increases [3H]inositol metabolism in rat vas deferens and caudal artery. Eur J Pharmacol. 1985 Oct 8;116(1-2):145-52.
- 39. Pereira RT, Porto CS, Godinho RO, Abdalla FMF. Effects of estrogen on intracellular signaling pathways linked to activation of muscarinic acetylcholine receptors and on acetylcholinesterase activity in rat hippocampus. Biochem Pharmacol. 2008 May1;75(9):1827-34.
- 40. Ascoli M, Pignataro OP, Segaloff DL. The inositol phosphate/diacylglycerol pathway in MA-10 Leydig tumor cells. Activation by arginine vasopressin and lack of effect of epidermal growth factor and human choriogonadotropin. J Biol Chem. 1989 Apr 25;264(12):6674-81.
- 41. Snedecor GW, Cochran WG. Statistical Methods. 7th edn. Iowa State University Press Ames USA,. Statistical methods, p. 89-233; 1980.
- 42. Eglen RM. Muscarinic receptor subtypes in neuronal and non-neuronal cholinergic function. Auton Autacoid Pharmacol. 2006 Jul;26(3):219-33.
- 43. Lahiani A, Yavin E, Lazarovici P. The molecular basis of toxins interactions with intracellular signaling via discrete portals. Toxins. 2017 Mar 16;9(3): pii: E107.
- 44. Nareoja K, Nasman J. Selective targeting of G-protein-coupled receptor subtypes with venom peptides. Acta Physiol (Oxf). 2012 Feb;204(2):186-201.
- 45. Servent D, Blanchet G, Mourier G, Marquer C, Marcon E, Fruchart-Gaillard C. Muscarinic toxins. Toxicon. 2011 Nov;58(6-7):455-63.
- 46. Lebois EP, Thorn C, Edgerton JR, Popiolek M, Xi S. Muscarinic receptor subtype distribution in the central nervous system and relevance to aging and Alzheimer's disease. Neuropharmacology. 2018 Jul 1;136(Pt C):362-73.
- 47. Caulfield MP. Muscarinic receptors--characterization, coupling and function. Pharmacol Ther. 1993 Jun;58(3):319-79.
- 48. Caulfield MP, Birdsall NJ. International Union of Pharmacology. XVII. Classification of muscarinic acetylcholine receptors. Pharmacol Rev. 1998 Jun;50(2):279-90.
- 49. Wess J. Molecular biology of muscarinic acetylcholine receptors. Crit Rev Neurobiol. 1996;10(1):69-99.
- 50. Ballinger EC, Ananth M, Talmage DA, Role LW. Basal forebrain cholinergic circuits and signaling in cognition and cognitive decline. Neuron. 2016 Sep 21;91(6):1199-218.
- 51. Freedman JE, Snyder SH. Vipoxin. A protein from Russell's viper venom with high affinity for biogenic amine receptors. J Biol Chem. 1981 Dec 25;256(24):13172-9.
- 52. Lambeau G, Barhanin J, Schweitz H, Qar J, Lazdunski M. Identification and properties of very high affinity brain membrane-binding sites for a neurotoxic phospholipase from the taipan venom. J Biol Chem. 1989 Jul 5;264(19):11503-10.
- 53. Vulfius CA, Kasheverov IE, Starkov VG, Osipov AV, Andreeva TV, Filkin SY, et al. Inhibition of nicotinic acetylcholine receptors, a novel facet in the pleiotropic activities of snake venom phospholipases A2 PLoS One. 2014 Dec 18;9(12): e115428.
- 54. Vulfius CA, Kasheverov IE, Kryukova EV, Spirova EN, Shelukhina IV, Starkov VG, et al. Pancreatic and snake venom presynaptically active phospholipases A2 inhibit nicotinic acetylcholine receptors. PLoS One. 2017 Oct 12;12(10):e0186206.
- 55. Cohen N, Weber G, Banner BL, Welton AF, Hope WC, Crowley H, et al. Analogs of arachidonic acid methylated at C-7 and C-10 as inhibitors of leukotriene biosynthesis. Prostaglandins. 1984 Apr;27(4):553-62.
-
Availability of data and materials
All data generated or analyzed during this study are included in this published article. -
Funding
This study was supported by São Paulo Research Foundation (FAPESP, grant n. 11/51600-2) and Instituto Nacional de Ciência e Tecnologia em Toxinas (INCTTOX-CNPq/FAPESP), Brazil. RTFS and MFPS were recipients of a Coordination for the Improvement of Higher Education Personnel (CAPES) fellowship of the Brazilian Ministry of Education. -
Ethics approval
All animal procedures involving experiments were approved by the Butantan Institute Committee for Ethics in Animal Experiments (license number CEUAIB 1100/13) in compliance with the recommendations of the National Council for the Control of Animal Experimentation of Brazil (CONCEA). -
Consent for publication
Not applicable.
Publication Dates
-
Publication in this collection
27 Jan 2020 -
Date of issue
2020
History
-
Received
05 July 2019 -
Accepted
04 Dec 2019