Abstracts
Hemangioperycytoma is a rare perivascular tumor that seldom involves the urogenital system. This tumor often appears with an unspecific clinical picture, and sometimes is associated with hematuria or hypertension. Diagnosis is based on a combination of histological and immunohistological findings. We report a case of a 52-year-old patient with renal hemangiopericytoma who underwent surgical treatment at our service. This report also includes a literature review on the subject.
Hemangiopericytoma/diagnosis; Urologic neoplasms; Vascular neoplasms; Muscle, smooth; Case reports
Hemangiopericitoma é um raro tumor perivascular que raramente envolve o sistema urogenital. Esses tumores geralmente se manifestam com quadro clínico inespecífico, por vezes associado a hematúria ou hipertensão. O diagnóstico baseia-se numa combinação de alterações histológicas e imuno-histológica. Este artigo relatou o caso de uma paciente de 52 anos de idade com um hemangiopericitoma renal submetida a tratamento cirúrgico em nosso serviço e incluiu uma revisão de literatura sobre o assunto.
Hemangiopericitoma/diagnóstico; Neoplasias urológicas; Neoplasias vasculares; Músculo liso; Relatos de casos
INTRODUCTION
Hemangiopericytoma (HPC) was first described in 1942. This is a rare perivascular tumor due to uncontrolled proliferation capillary pericytes. This tumor affects mainly extremities of the body, pelvis, head and neck, and meningitis, but hemangiopericytoma is rarely found in urogenital system. Until 2009, only 42 cases of this neoplasia involving the kidney were reported in the world literature.(11. Bilici A, Ustaalioglu BB, Seker M, Salman T, Igdem AA, Celik E, et al. Metastatic renal hemangiopericytoma: A rare case report. Arch Oncol. 2009;17(1-2):32-5.)
Because of the rarity of this injury until to date, no therapeutic management and ideal diagnosis were established as well as progress and prognosis of the disease.(22. Argyropoulos A, Liakatas I, Lykourinas M. Renal haemangiopericytoma: the characteristics of a rare tumour. BJU Int. 2005;95(7):943-7. Review.) We report a case of renal HPC diagnosed and treated at Urological Service of the Irmandade Santa Casa de Misericórdia de São Paulo.
CASE REPORT
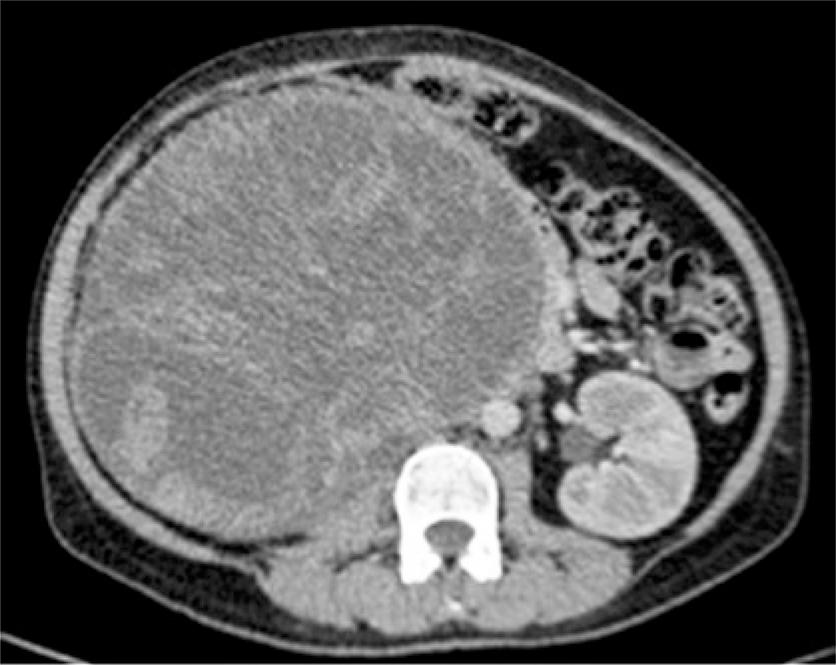
In November 2011, a 52-year-old, white and previously healthy woman was admitted at our because of an increase in abdominal volume. She denied any other complains, including abdominal pain or hematuria. The patient does not have a history of smoking or use of illicit substances. In March 2012, she began a follow-up at outpatient unit dedicated to urology care. She had blood hypertension and took medicine for this affection, and also mentioned to lose 10kg in that period. Her ultrasonography test of urinary tract showed evidence of tumors in topography of the right kidney. Laboratorial tests showed anemia with serum hemoglobin value of 8.9g/dL. The abdominal computed tomography (CT) scan revealed a cystic-solid expansive formation, multiloculated, heterogenous in the right kidney that replaced the parenchyma in the medial and inferior third. The formation measured 23.2x18.8x18.7cm with special emphasis of solid portions and superimposition of septal segments that dislocated superiorly the liver and gallbladder, crossing the medium line and opposing medially to the pancreas, portal and aortic vein with signs of vascular compromising (Figure 1).
The patient had undergone radical nephrectomy, which is a procedure with technical difficult because of tumor proportion, without intercurrence or necessity of blood transfusion, and even intensive care in immediate postoperative. Patient progressed without complications, her drain presented low debt and good diuresis. She was discharged on the 3rd day after the surgery.
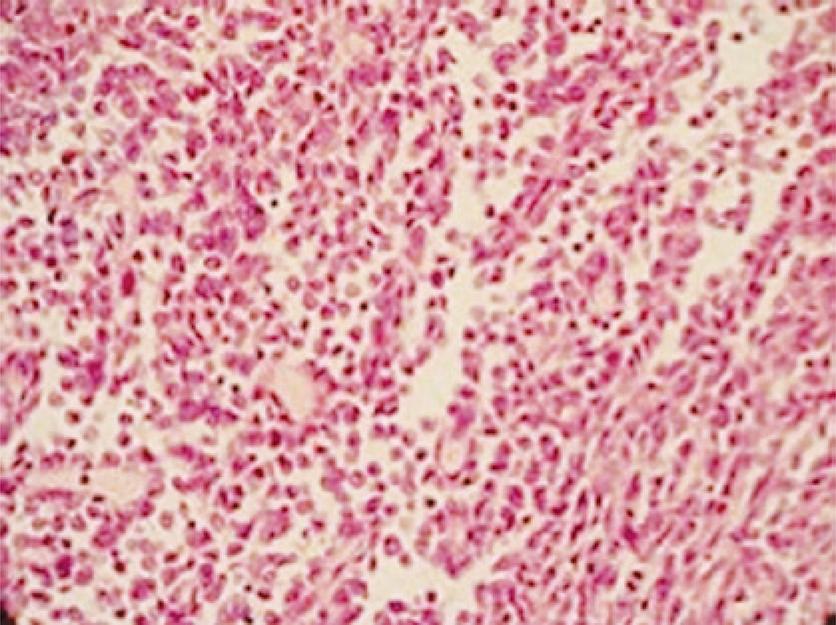
The anatomopathological study (Figure 2) confirmed the diagnosis of HPC, and immunohistochemical analysis showed tumoral cells positive for BCL-2 (Figure 3), CD99 (Figure 4), vimentin (Figure 5), CD34 (Figure 6), and negative for S-100. Mitotic index score was 2 (13 mitoses per 10 high-power fields).
In return visit at outpatient unit, the patient remained asymptomatic and without local or systemic complications. We removed points of surgical incision and requested abdominal CT and control chest radiography for 4 months. During reassessments on this period the patient was in good general status, normotensive and did not need anti-hypertensive medicine. In addition, no other complaints were reported. Radiography exam showed changes and CT revealed only sings of previous manipulations in topography of right kidney and mild increased paracaval lymph nodes, <1.0cm. Currently, 7th months after surgery, the patient is clinically well and without intercurrence.
DISCUSSION
HPC is classified as vascular tumor of soft tissues from the pericytes, modified cells of smooth muscle tissue that were described by Zimmerman in 1923. Such cells are organized around capillaries and post-capillary veins, and they are responsible to modulate blood flow and permeability of these vessels.(33. Stout AP, Murray MR. Hemangiopericytoma a vascular tumor featuring Zimmermann’s pericytes. Ann Surg. 1942;116(1):26-33.)
Our patient was at average age of diagnosis of this neoplasia, about 40.3 years old (16 to 68 years old), which suggest that such tumor affect more frequently in young patients than other types of renal cell cancer.(22. Argyropoulos A, Liakatas I, Lykourinas M. Renal haemangiopericytoma: the characteristics of a rare tumour. BJU Int. 2005;95(7):943-7. Review.,44. Brescia A, Pinto F, Gardi M, Maria Vecchio F, Bassi PF. Renal Hemangiopericytoma: case report and review of the literature. Urology. 2008;71(4):755.e9-12.) No significant difference is found concerning the incidence of this neoplasia between men and women, and no age variation between sexes in the diagnosis.
Clinical picture is unspecific. In 66% of cases reported in the literature, initial symptom is painless abdominal tumor, such as reported in our case. Other symptoms already described, in order of frequency, are: hematuria, hypoglycemia and arterial hypertension.(22. Argyropoulos A, Liakatas I, Lykourinas M. Renal haemangiopericytoma: the characteristics of a rare tumour. BJU Int. 2005;95(7):943-7. Review.) A study reported that hematuria was found in six patients in which an invasion of pyelocaliceal system by tumor occured.(11. Bilici A, Ustaalioglu BB, Seker M, Salman T, Igdem AA, Celik E, et al. Metastatic renal hemangiopericytoma: A rare case report. Arch Oncol. 2009;17(1-2):32-5.) Hypoglycemia is also associated to excess of glucose metabolism by the tumor. Blood hypertension, however, is believed to occur because of the production of renin by the neoplasia and, as observed in our patient, it often disappear after surgical treatment.(11. Bilici A, Ustaalioglu BB, Seker M, Salman T, Igdem AA, Celik E, et al. Metastatic renal hemangiopericytoma: A rare case report. Arch Oncol. 2009;17(1-2):32-5.,44. Brescia A, Pinto F, Gardi M, Maria Vecchio F, Bassi PF. Renal Hemangiopericytoma: case report and review of the literature. Urology. 2008;71(4):755.e9-12.) The most common site of metastasis of renal HPC is the lung, which was described in two case and patients had a poor prognosis.
Imaging exams, such as ultrasonography, CT or magnetic resonance did not show specific finding of the tumor that were able to collaborate in the differential diagnosis from other renal tumors.(11. Bilici A, Ustaalioglu BB, Seker M, Salman T, Igdem AA, Celik E, et al. Metastatic renal hemangiopericytoma: A rare case report. Arch Oncol. 2009;17(1-2):32-5.,44. Brescia A, Pinto F, Gardi M, Maria Vecchio F, Bassi PF. Renal Hemangiopericytoma: case report and review of the literature. Urology. 2008;71(4):755.e9-12.) However, Yagmani reported that these tumors can have standard characteristic in early phase of blood angiography – of principal arteries, presence of vessels surrounding the tumor and well-marked stain, delimiting the tumor.(22. Argyropoulos A, Liakatas I, Lykourinas M. Renal haemangiopericytoma: the characteristics of a rare tumour. BJU Int. 2005;95(7):943-7. Review.,55. Yaghmai I. Angiographic manifestations of soft-tissue and osseous hemangiopericytomas. Radiology. 1978;126(3):653-9.)
Macroscopically, most tumors are well circumscribed lesions or a thin capsule, associated to complex network of vessels and, sometimes, with small satellite nodules around the main mass. Only the minority is adhered to neighbouring tissues. Tumor size reported in the literature range between 2 and 25cm.(44. Brescia A, Pinto F, Gardi M, Maria Vecchio F, Bassi PF. Renal Hemangiopericytoma: case report and review of the literature. Urology. 2008;71(4):755.e9-12.)
In general, there is a cellular proliferation with small variability in cellularity, a “staghorn” vascular pattern with packed pericytes around vascular endothelium, and minimal collagenization that can vary both in size and form. The technique used is based on Hematoxylin and eosin stain (Figure 2) and silver associated with immunohistochemistry, using antibodies against CD31, CD34, CD99, S-100, vimentin, citokeratin and epithelia antigen membrane (renal). Only the combination of these two is can provide the diagnosis,(22. Argyropoulos A, Liakatas I, Lykourinas M. Renal haemangiopericytoma: the characteristics of a rare tumour. BJU Int. 2005;95(7):943-7. Review.,44. Brescia A, Pinto F, Gardi M, Maria Vecchio F, Bassi PF. Renal Hemangiopericytoma: case report and review of the literature. Urology. 2008;71(4):755.e9-12.) which in our case enabled the precise diagnosis of tumoral mass.
HPC diagnosis is done mainly by exclusion. The histological pattern and immunophenotyping provide information to differential diagnosis with other vascular neoplasias and other mesenchymal tumors. Such tumors include juxtaglomerular origin, which often is presented as blood hypertension, especially in young adults. These tumors are considered of different origin of HPC.(22. Argyropoulos A, Liakatas I, Lykourinas M. Renal haemangiopericytoma: the characteristics of a rare tumour. BJU Int. 2005;95(7):943-7. Review.) Other vascular neoplasias, histiocytosis, neural and of smooth muscle must be excluded before the definitive diagnosis of HPC.(44. Brescia A, Pinto F, Gardi M, Maria Vecchio F, Bassi PF. Renal Hemangiopericytoma: case report and review of the literature. Urology. 2008;71(4):755.e9-12.)
The main treatment for this disease is surgery, and exeresis must be as radical as possible in order to avoid incomplete resection and recurrence. Therefore, an appropriate surgical plan associated with a previous study of tumor vascular architecture (CT angiography or arteriography) must be done.(22. Argyropoulos A, Liakatas I, Lykourinas M. Renal haemangiopericytoma: the characteristics of a rare tumour. BJU Int. 2005;95(7):943-7. Review.) Radiotherapy, chemotherapy or adjuvant or neoadjuvant immunotherapy seem to be inefficient to treat HPC. Although, the majority of renal HPCs are benign, outcomes cannot be precisely foreseen by histological appearance, however, rigorous follow-up must be conducted to detect long-term recurrence.(66. Chaudhary A, Seenu V, Sedain G, Ray R, Sharma S, Agarwal S, et al. Hemangiopericytoma of renal pelvis – an unusual tumor in an adolescent. Urology. 2007;70(4):811.e13-4.)
Main factor associated to prognosis seems to be the histological pattern. Enzinger and Smith observed survival of 77% in 10 years in patients with 0 to 3 mitoses /10versus 29% in those patients with 4 or more by field.(77. Enzinger FM, Smith BH. Hemangiopericytoma. An analysis of 106 cases. Hum Pathol. 1976;7(1):61-82.) Similarly, survival was 81% in patients with tumoral necrosisversus 29% in those who had necrotic areas in the tumor. They also observed the importance of tumor size concerning prognosis. In tumors <6.5cm, survival in 10 years reached 95% and 63% for large tumors.(44. Brescia A, Pinto F, Gardi M, Maria Vecchio F, Bassi PF. Renal Hemangiopericytoma: case report and review of the literature. Urology. 2008;71(4):755.e9-12.)
CONCLUSION
Because HPC is rare and present a vague clinical picture associated with imaging exams that do not indicate a specific disease, the precisely diagnosis of hemangiopericytoma is made only after surgery and with the immunohistochemical result.
Best treatment consists in complete surgical excision because complementary treatment such as chemotherapy and/or radiotherapy is not efficient.
REFERENCES
-
1Bilici A, Ustaalioglu BB, Seker M, Salman T, Igdem AA, Celik E, et al. Metastatic renal hemangiopericytoma: A rare case report. Arch Oncol. 2009;17(1-2):32-5.
-
2Argyropoulos A, Liakatas I, Lykourinas M. Renal haemangiopericytoma: the characteristics of a rare tumour. BJU Int. 2005;95(7):943-7. Review.
-
3Stout AP, Murray MR. Hemangiopericytoma a vascular tumor featuring Zimmermann’s pericytes. Ann Surg. 1942;116(1):26-33.
-
4Brescia A, Pinto F, Gardi M, Maria Vecchio F, Bassi PF. Renal Hemangiopericytoma: case report and review of the literature. Urology. 2008;71(4):755.e9-12.
-
5Yaghmai I. Angiographic manifestations of soft-tissue and osseous hemangiopericytomas. Radiology. 1978;126(3):653-9.
-
6Chaudhary A, Seenu V, Sedain G, Ray R, Sharma S, Agarwal S, et al. Hemangiopericytoma of renal pelvis – an unusual tumor in an adolescent. Urology. 2007;70(4):811.e13-4.
-
7Enzinger FM, Smith BH. Hemangiopericytoma. An analysis of 106 cases. Hum Pathol. 1976;7(1):61-82.
Publication Dates
-
Publication in this collection
01 May 2015 -
Date of issue
Apr-Jun 2015
History
-
Received
25 Mar 2013 -
Accepted
1 Dec 2013