ABSTRACT
The nutritional status of patients submitted to hematopoietic stem cell transplant is considered an independent risk factor, which may influence on quality of life and tolerance to the proposed treatment. The impairment of nutritional status during hematopoietic stem cell transplant occurs mainly due to the adverse effects resulting from conditioning to which the patient is subjected. Therefore, adequate nutritional evaluation and follow-up during hematopoietic stem cell transplant are essential. To emphasize the importance of nutritional status and body composition during treatment, as well as the main characteristics related to the nutritional assessment of the patient, the Brazilian Consensus on Nutrition in Hematopoietic Stem Cell Transplant: Adults was prepared, aiming to standardize and update Nutritional Therapy in this area. Dietitians, nutrition physicians and hematologists from 15 Brazilian centers thar are references in hematopoietic stem cell transplant took part.
Hematopoietic stem cell transplantation; Nutritional status; Nutritional therapy
RESUMO
O estado nutricional do paciente submetido ao transplante de células-tronco hematopoéticas é considerado fator de risco independente, podendo influenciar na qualidade de vida e na tolerância ao tratamento proposto. O comprometimento do estado nutricional durante o transplante de células-tronco hematopoéticas ocorre principalmente devido aos efeitos adversos decorrentes do condicionamento ao qual o paciente é submetido. Desta forma, a adequada avaliação nutricional e o acompanhamento durante o transplante de células-tronco hematopoéticas tornam-se imprescindíveis. Com o objetivo de salientar a importância do estado nutricional e da composição corporal durante o tratamento, bem como as principais características relacionadas à avaliação nutricional do paciente, o Consenso Brasileiro de Nutrição em Transplante de Células-Tronco Hematopoiéticas: Adulto foi elaborado visando uniformizar e atualizar a Terapia Nutricional nesta área. Com a participação de nutricionistas, nutrólogos e hematologistas de 15 centros brasileiros referências em transplante de células-tronco hematopoéticas
Transplante de células-tronco hematopoiéticas; Estado nutricional; Terapia nutricional
INTRODUCTION TO THE TYPES OF HEMATOPOIETIC STEM CELL TRANSPLANTATION
Hematopoietic stem cell transplantation (HSCT) is a well-established treatment modality for a wide range of benign and malignant hematological diseases.11. Copelan EA. Hematopoietic stem-cell transplantation. N Engl J Med. 2006; 354(17):1813-26.
Every year, about 50 thousand individuals undergo HSCT worldwide.22. Majhail NS, Rizzo JD, Lee SJ, Aljurf M, Atsuta Y, Bonfim C, Burns LJ, Chaudhri N, Davies S, Okamoto S, Seber A, Socie G, Szer J, Van Lint MT, Wingard JR, Tichelli A; Center for International Blood and Marrow Transplant Research (CIBMTR); American Society for Blood and Marrow Transplantation (ASBMT); European Group for Blood and Marrow Transplantation (EBMT); Asia-Pacific Blood and Marrow Transplantation Group (APBMT); Bone Marrow Transplant Society of Australia and New Zealand (BMTSANZ); East Mediterranean Blood and Marrow Transplantation Group (EMBMT),; Sociedade Brasileira de Transplante de Medula Ossea (SBTMO). Recommended screening and preventive practices for long-term survivors after hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2012;18(3):348–71. It is estimated that 3,091 HSCT were performed in Brazil, in 2018, based on data from the Brazilian Transplantation Registry, in line with the global trend towards an increase in the number of procedures carried out.33. Associação Brasileira de Transplante de Órgãos (ABTO). Registro Brasileiro de Transplantes (RBT). Dimensionamento dos transplantes no Brasil e em cada estado 2011-2018 [Internet]. São Paulo (SP): ABTO; 2018 [citado 2019 Jun 25]. Disponível em: http://www.abto.org.br/abtov03/Upload/file/RBT/2018/Lv_RBT-2018.pdf
http://www.abto.org.br/abtov03/Upload/fi...
In addition to the progressive increase in the number of transplant centers, this growth is due to the large number of donors registered with the Brazilian Registry of Volunteer Blood Marrow Donors (REDOME - Registro Brasileiro de Doadores Voluntários de Medula Óssea) which is now the third largest registry in the world, with more than 4 million people, which provides better chances of finding a suitable donor.44. Instituto Nacional do Câncer (INCA). Medula Net. Redome [Internet]. Rio de Janeiro: INCA: 2013 [citado 2019 Jun 27]. Disponível em: http://redome.inca.gov.br/o-redome/dados/
http://redome.inca.gov.br/o-redome/dados...
Based on the donor, HSCT can be referred to as autologous, when the hematopoietic stem cells (HSC) used are the patient’s own; allogeneic, when they come from a different donor; and syngeneic, when the donor is an identical twin. In allogeneic transplantation, the donor can be related (HLA matched or unmatched - haploidentical) or unrelated (preferably HLA matched), from the donor registry or the umbilical cord and placental blood bank (UCPB) - HLA matched or partly unmatched. Table 1 describes HSCT modalities. Table 2 lists the main diseases leading to HSCT indication.55. Passweg JR, Halter J, Bucher C, Gerull S, Heim D, Rovó A, et al. Hematopoietic stem cell transplantation: a review and recommendations for follow-up care for the general practitioner. Swiss Med Wkly. 2012;142:w13696. Review.
AUTOLOGOUS HEMATOPOIETIC STEM CELL TRANSPLANTATION
Autologous HSCT is based on the principle of using high-dose chemotherapy drugs followed by salvage HSC, because, otherwise, these doses would not be tolerated by patients due to intense myelotoxicity with very prolonged aplasia. The purpose is to overcome tumor cell resistance to conventional doses of chemotherapy, improving treatment response.44. Instituto Nacional do Câncer (INCA). Medula Net. Redome [Internet]. Rio de Janeiro: INCA: 2013 [citado 2019 Jun 27]. Disponível em: http://redome.inca.gov.br/o-redome/dados/
http://redome.inca.gov.br/o-redome/dados...
The most frequent indications for autologous HSCT (Table 2) include patients with myeloma, as the current treatment backbone for patients with no major comorbidities; and patients with lymphoma in first remission (such as mantle cell lymphoma) or those with non-Hodgkin or Hodgkin lymphomas relapsing after chemotherapy.
In this treatment modality, the HSC (CD34+) are the patient’s own cells, collected by apheresis and cryopreserved to be infused back after high-dose chemotherapy/radiotherapy. Cells are collected after 5 to 6 days of stimulation with granulocyte colony-stimulating factor (GCSF) at 10ug/kg/day infused into a peripheral vein.55. Passweg JR, Halter J, Bucher C, Gerull S, Heim D, Rovó A, et al. Hematopoietic stem cell transplantation: a review and recommendations for follow-up care for the general practitioner. Swiss Med Wkly. 2012;142:w13696. Review.
The complications of this therapy are related with toxicity of the conditioning regimen, such as mucositis and abnormalities in the liver, kidneys, heart and other organs, in addition to infection-related complications during aplasia.55. Passweg JR, Halter J, Bucher C, Gerull S, Heim D, Rovó A, et al. Hematopoietic stem cell transplantation: a review and recommendations for follow-up care for the general practitioner. Swiss Med Wkly. 2012;142:w13696. Review. Although autologous HSCT is a curative treatment modality for several diseases, relapse of the underlying disease is still the major post-transplant complication.
ALLOGENEIC HEMATOPOIETIC STEM CELL TRANSPLANTATION
Allogeneic HSCT consists in reconstituting the bone marrow by intravenously infusing HSC from a related or unrelated donor after the use of a conditioning regimen. The latter aims not just to eradicate neoplastic cells by direct cytotoxicity, but also to create an immunological space by immunoablation and immunosuppression, allowing for engraftment of allogeneic HSC.66. Buchholz S, Ganser A. [Hematopoietic stem cell transplantation. Indications, foundations and perspective]. Internist (Berl). 2009;50(5):572-80. Review. German.
Major histocompatibility complex (MHC) mediated genetic mismatches between donor and recipient trigger the main aloimmune reactions affecting the post-transplant period: rejection, graft versus host disease (GVHD) and graft versus tumor (GVT) effect. The donor’s T-lymphocytes have been considered the main effectors of these two effects.77. Sociedade Brasileira de Transplante de Medula Óssea. Transplantante de células-tronco hematopoéticas. São Paulo: Atheneu; 2010. 1280 pp.
The GVT effect has a central role in allogeneic HSCT, since the donor’s alloreactive T-cells are responsible for eliminating residual malignant cells, consequently contributing to a lower risk of relapse of the underlying disease.77. Sociedade Brasileira de Transplante de Medula Óssea. Transplantante de células-tronco hematopoéticas. São Paulo: Atheneu; 2010. 1280 pp. Thus, allogeneic HSCT is currently considered a cell therapy with antitumor activity.
SOURCE OF HEMATOPOIETIC STEM CELLS
For many years, HSC were collected exclusively by multiple bone marrow aspirations through the posterior iliac crest. However, an important milestone in the dissemination of the HSCT technique was the discovery of other HSC sources, such as peripheral cells and UCPB.55. Passweg JR, Halter J, Bucher C, Gerull S, Heim D, Rovó A, et al. Hematopoietic stem cell transplantation: a review and recommendations for follow-up care for the general practitioner. Swiss Med Wkly. 2012;142:w13696. Review.
DONOR SELECTION
In allogeneic HSCT, the selection of a donor with the appropriate degree of compatibility is essential for a successful HSCT. The genetic factors with the greatest impact on transplant results include HLA genes.77. Sociedade Brasileira de Transplante de Medula Óssea. Transplantante de células-tronco hematopoéticas. São Paulo: Atheneu; 2010. 1280 pp.
Of all the possibilities of potential donors for allogeneic HSCT, fully HLA-matched related donors still account for the best overall and progression-free survival rates.88. Symons HJ, Fuchs EJ. Hematopoietic SCT from partially HLA-mismatched (HLA-haploidentical) related donors. Bone Marrow Transplant. 2008; 42(6):365-77. Unfortunately, only 25% to 30% of allogeneic HSCT candidates have a HLA-matched related donor. Therefore, approximately 70% of patients do not have any related donors with no HLA matching. There are three possible HSC sources in this situation: cells from unrelated donors enrolled in bone marrow registries; cells from umbilical cord and placental blood, or those from partially HLA-matched related donors (haploidentical).
TYPES OF PREPARATION (CONDITIONING) REGIMENS
The preparation or conditioning regimen is intended to reduce and even eradicate an underlying neoplasm, in addition to inducing immunosuppression in the recipient to allow for HSC engraftment. Table 3 lists the conditioning regimens most commonly used in allogeneic HSCT.55. Passweg JR, Halter J, Bucher C, Gerull S, Heim D, Rovó A, et al. Hematopoietic stem cell transplantation: a review and recommendations for follow-up care for the general practitioner. Swiss Med Wkly. 2012;142:w13696. Review.
IMMEDIATE POST-HSCT COMPLICATIONS: TOXICITY, APLASIA AND ACUTE GRAFT-VERSUS-HOST DISEASE
After myeloablative conditioning (including all autologous HSCT) and some reduced-intensity therapies, we observe, after HSC infusion, an immediate toxicity phase due to the chemotherapy/radiotherapy administered, which can last for 10 to 15 days. In this phase, patients present nausea, vomiting and diarrhea, which can be associated with mild to severe mucositis, which occurs in 47% to 100% of patients.99. Chaudhry HM, Bruce AJ, Wolf RC, Litzow MR, Hogan WJ, Patnaik MS, et al. The Incidence and Severity of Oral Mucositis among Allogeneic Hematopoietic Stem Cell Transplantation Patients: A Systematic Review. Biol Blood Marrow Transplant. 2016;22(4):605-16.
The presence of mucositis depends on the conditioning received - the presence of total body irradiation, for example - the age of the patient, the use of methotrexate for GVHD prophylaxis, prior oral hygiene, and some genetic polymorphisms that affect chemotherapy metabolism.99. Chaudhry HM, Bruce AJ, Wolf RC, Litzow MR, Hogan WJ, Patnaik MS, et al. The Incidence and Severity of Oral Mucositis among Allogeneic Hematopoietic Stem Cell Transplantation Patients: A Systematic Review. Biol Blood Marrow Transplant. 2016;22(4):605-16.,1010. Rocha V, Porcher R, Fernandes JF, Filion A, Bittencourt H, Silva W Jr, et al. Association of drug metabolism gene polymorphisms with toxicities, graft-versus-host disease and survival after HLA-identical sibling hematopoietic stem cell transplantation for patients with leukemia. Leukemia. 2009;23(3):545-56. In this phase, patients also experience bone marrow toxicity with aplasia and, as a consequence, greater need for transfusion, and higher infection rates, particularly bacterial infections. Due to the high metabolic and inflammatory activity (known as cytokine release syndrome) in this phase, most patients do not eat and require opioids for the pain caused by mucositis, presenting with major weight loss, unless appropriate supplementation is administered. This phase continues until engraftment or neutrophil recovery. However, in this period, there is a risk for acute GVHD, which can manifest in the skin, liver or gastrointestinal tract. In the latter, patients can have nausea, vomiting and diarrhea, sometimes quite voluminous and, in more severe cases, intestinal bleeding and melena. Here, once again, patients have major metabolic consumption, requiring long-term corticosteroids, which is part of GVHD management, and fasting or dietary restriction.
IMPORTANCE OF NUTRITIONAL STATUS IN HEMATOPOIETIC STEM CELL TRANSPLANTATION
Patients undergoing HSCT are a heteregenous population from the nutritional standpoint, with differences regarding the type of intervention needed, which is why they require distinct strategies. We must also consider the intensity of the conditioning regimen and the presence of GVHD, which have a direct effect on the nutritional status of the patient. Decreased oral intake, post-HSCT complications, and increased nutritional requirements lead to the need for individualized nutritional interventions.1111. Macris PC, McMillen KK. Nutrition Support of the Hematopoietic Cell Transplant Recipient. In: S J Forman, Negrin RS, Antin JH, Appelbaum FR, editor. Thomas’ Hematopoietic Cell Transplantation: Stem Cell Transplantation. 5th edi. Wiley-Blackwell; 2016. p.1177-85
The early identification of patients at nutritional risk minimizes the deleterious effects of malnutrition and/or overweight/obesity. Both are risk factors for complications and increased mortality, either or not associated with relapse in transplanted patients.1212. Rzepecki P, Barzal J, Oborska S. Blood and marrow transplantation and nutritional support. Support Care Cancer. 2010;18(Suppl 2):S57-65. However, one of the difficulties which limits the impact of body weight (BW) on post-transplant prognosis is that most data available are limited to the first year after HSCT, in addition to the difference in methods applied and cutoff points used for nutritional status classification.1313. Urbain P, Ihorst G, Finke J, Bertz H. The impact of pre-transplant body weight on short- and long-term outcomes after allogeneic hematopoietic cell transplantation in adults using different weight classi fi cation tools. Bone Marrow Transplant. 2016;51(1):144-7. Initial and serial assessment of nutritional status is important, since it anticipates potential nutritional impacts of conditioning and other treatment-related toxicities, which affect nutrient intake, absorption and utilization.1111. Macris PC, McMillen KK. Nutrition Support of the Hematopoietic Cell Transplant Recipient. In: S J Forman, Negrin RS, Antin JH, Appelbaum FR, editor. Thomas’ Hematopoietic Cell Transplantation: Stem Cell Transplantation. 5th edi. Wiley-Blackwell; 2016. p.1177-85
OBESITY
Excess weight (overweight and obesity) is an important risk factor for cardiovascular diseases, diabetes mellitus, among others. The prevalence of obesity increases every year. According to the World Health Organization (WHO), in 2016, 39% of adult population was overweight and 13% of the world population was obese.1414. World Health Organization (WHO). Obesity and overweight [Internet]. Geneva: WHO; 2018 [cited 2019 Oct 1]. Available from: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight
https://www.who.int/news-room/fact-sheet...
It is not different in Brazil, where the agency Telephone-based Survey of Risk and Protective Factors for Chronic Diseases (VIGITEL - Vigilância de Fatores de Risco e Proteção para Doenças Crônicas por Inquérito Telefônico)1515. Brasil. Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância de Doenças e Agravos não Transmissíveis e Promoção da Saúde. Vigitel Brasil 2016: Vigilância de Fatores de Risco e Proteção Para Doenças Crônicas Por Inquérito Telefônico: estimativas sobre frequência e distribuição sociodemográfica de fatores de risco e proteção para doenças crônicas nas capitais dos 26 estados brasileiros e no Distrito Federal em 2016 [Internet]. Brasília (DF): Ministério da Saúde; 2017 [citado 2019 Out 1]. Disponível em: http://portalarquivos.saude.gov.br/images/pdf/2017/junho/07/vigitel_2016_jun17.pdf
http://portalarquivos.saude.gov.br/image...
showed, in 2016, that more than half of the population was overweight and 18.9% of Brazilians were obese. Thus, it is increasingly common to have overweight or bariatric patients requiring bone marrow transplantation.
Several publications have shown obesity as an independent prognostic factor for adverse events, such as increased mortality, risk of infection, incidence of acute grade II-IV GVHD, and toxicity. Tarella et al.,1616. Tarella C, Caracciolo D, Gavarotti P, Argentino C, Zallio F, Corradini P, et al. Overweight as an adverse prognostic factor for non-Hodgkin’s lymphoma patients receiving high-dose chemotherapy and autograft. Bone Marrow Transplant. 2000;26(11):1185-91. in a retrospective study with patients with non-Hodgkin lymphoma undergoing autologous bone marrow transplantation, showed worse results, with a 2.9-fold higher risk of death compared to the group of non-obese patients. Meloni et al.,1717. Meloni G, Proia A, Capria S, Romano A, Trapé G, Trisolini SM, et al. Obesity and autologous stem cell transplantation in acute myeloid leukemia. Bone Marrow Transplant. 2001;28(4):365-7. also showed that obesity is a positive predictive factor for increased treatment-related toxicity and mortality in patients with acute myeloid leukemia (AML) undergoing autologous bone marrow transplantation. The number of nucleated cells used in transplantation, which is smaller in obese patients, may influence transplant outcomes.1818. Fuji S, Kim SW, Yoshimura K, Akiyama H, Okamoto S, Sao H, Takita J, Kobayashi N, Mori S; Japan Marrow Donor Program. Possible association between obesity and posttransplantation complications including infectious diseases and acute graft-versus-host disease. Biol Blood Marrow Transplant. 2009;15(1):73-82. Vogl et al.,1919. Vogl DT, Wang T, Pérez WS, Stadtmauer EA, Heitjan DF, Lazarus HM, et al. Effect of obesity on outcomes after autologous hematopoietic stem cell transplantation for multiple myeloma. Biol Blood Marrow Transplant. 2011;17(12):1765-74. did not show any effect of overweight or obesity on progression-free survival, overall survival, progression and mortality related to relapse in patients undergoing autologous transplantation following multiple myeloma. In the same study, obese patients subjected to conditioning with melphalan and total body irradiation had a lower relapse rate; however, the rationale is uncertain.
Nevertheless, in a 2014 meta-analysis published in Bone Marrow Transplantation, the negative impact of obesity on bone marrow transplant outcomes was statistically significant for allogeneic and non-autologous transplants.2020. Nakao M, Chihara D, Niimi A, Ueda R, Tanaka H, Morishima Y, et al. Impact of being overweight on outcomes of hematopoietic SCT: a meta-analysis. Bone Marrow Transplant. 2014;49(1):66-72.
As for GVHD, there are studies that support increased incidence of acute grade II-IV GVHD as the body mass index (BMI) increases.1818. Fuji S, Kim SW, Yoshimura K, Akiyama H, Okamoto S, Sao H, Takita J, Kobayashi N, Mori S; Japan Marrow Donor Program. Possible association between obesity and posttransplantation complications including infectious diseases and acute graft-versus-host disease. Biol Blood Marrow Transplant. 2009;15(1):73-82.
Obesity is also associated with a higher incidence of infectious diseases (bacterial, fungal and viral) due to increased hyperglycemia when compared to patients with a BMI under 30kg/m22. Majhail NS, Rizzo JD, Lee SJ, Aljurf M, Atsuta Y, Bonfim C, Burns LJ, Chaudhri N, Davies S, Okamoto S, Seber A, Socie G, Szer J, Van Lint MT, Wingard JR, Tichelli A; Center for International Blood and Marrow Transplant Research (CIBMTR); American Society for Blood and Marrow Transplantation (ASBMT); European Group for Blood and Marrow Transplantation (EBMT); Asia-Pacific Blood and Marrow Transplantation Group (APBMT); Bone Marrow Transplant Society of Australia and New Zealand (BMTSANZ); East Mediterranean Blood and Marrow Transplantation Group (EMBMT),; Sociedade Brasileira de Transplante de Medula Ossea (SBTMO). Recommended screening and preventive practices for long-term survivors after hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2012;18(3):348–71..1818. Fuji S, Kim SW, Yoshimura K, Akiyama H, Okamoto S, Sao H, Takita J, Kobayashi N, Mori S; Japan Marrow Donor Program. Possible association between obesity and posttransplantation complications including infectious diseases and acute graft-versus-host disease. Biol Blood Marrow Transplant. 2009;15(1):73-82.
Obese patients may experience changes in drug pharmacokinetics. Some drugs are not fat soluble and do not disseminate well in fat tissue.1717. Meloni G, Proia A, Capria S, Romano A, Trapé G, Trisolini SM, et al. Obesity and autologous stem cell transplantation in acute myeloid leukemia. Bone Marrow Transplant. 2001;28(4):365-7. The clearance of a drug is the most relevant pharmacokinetic parameter due to its correlation with clinical outcomes. The liver is the main organ responsible for drug metabolism, and hepatic steatosis, very common in obese individuals, may have an impact on drug clearance.2121. Griggs JJ, Mangu PB, Anderson H, Balaban EP, Dignam JJ, Hryniuk WM, Morrison VA, Pini TM, Runowicz CD, Rosner GL, Shayne M, Sparreboom A, Sucheston LE, Lyman GH; American Society of Clinical Oncology. Appropriate chemotherapy dosing for obese adult patients with cancer: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2012;30(13):1553-61. Review. No positive relation has been shown between obesity, veno-occlusive disease of the liver, and liver dysfunction in these patients.1818. Fuji S, Kim SW, Yoshimura K, Akiyama H, Okamoto S, Sao H, Takita J, Kobayashi N, Mori S; Japan Marrow Donor Program. Possible association between obesity and posttransplantation complications including infectious diseases and acute graft-versus-host disease. Biol Blood Marrow Transplant. 2009;15(1):73-82. The effect of obesity on the kidneys is not very clear yet.2121. Griggs JJ, Mangu PB, Anderson H, Balaban EP, Dignam JJ, Hryniuk WM, Morrison VA, Pini TM, Runowicz CD, Rosner GL, Shayne M, Sparreboom A, Sucheston LE, Lyman GH; American Society of Clinical Oncology. Appropriate chemotherapy dosing for obese adult patients with cancer: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2012;30(13):1553-61. Review.
The American Society for Blood and Marrow Transplantation (ASBMT), in a 2014 review, did not find any significantly strong evidence that could specifically guide the prescription of conditioning chemotherapy to obese patients. The drugs are suggested empirically or by extrapolation of data from non-transplanted patients.2222. Bubalo J, Carpenter PA, Majhail N, Perales MA, Marks DI, Shaughnessy P, Pidala J, Leather HL, Wingard J, Savani BN; American Society for Blood and Marrow Transplantation practice guideline committee. Conditioning chemotherapy dose adjustment in obese patients: a review and position statement by the American Society for Blood and Marrow Transplantation practice guideline committee. Biol Blood Marrow Transplant. 2014;20(5):600-16. The American Society of Clinical Oncology (ASCO) reports that the poorer outcomes of chemotherapy in obese patients have multifactorial causes – however, the unexplained reduction in drug doses partly explains the higher mortality among overweight and obese patients. ASCO recommends that doses should be calculated as per the real weight of patients.2121. Griggs JJ, Mangu PB, Anderson H, Balaban EP, Dignam JJ, Hryniuk WM, Morrison VA, Pini TM, Runowicz CD, Rosner GL, Shayne M, Sparreboom A, Sucheston LE, Lyman GH; American Society of Clinical Oncology. Appropriate chemotherapy dosing for obese adult patients with cancer: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2012;30(13):1553-61. Review.
Hyperglycemia, when occurring during neutropenia, is a risk factor for acute GVHD, and associated with higher non-relapse mortality. Obesity is related with hyperglycemia and insulin resistance. Hyperglycemia also increases the incidence of infectious diseases.1818. Fuji S, Kim SW, Yoshimura K, Akiyama H, Okamoto S, Sao H, Takita J, Kobayashi N, Mori S; Japan Marrow Donor Program. Possible association between obesity and posttransplantation complications including infectious diseases and acute graft-versus-host disease. Biol Blood Marrow Transplant. 2009;15(1):73-82.
Metabolic syndrome is becoming more prevalent in post-transplant patients. Higher levels of C-reactive protein (CRP) and leptin, and lower levels of adiponectins may be indicative of association with inflammation. Leptin, which is proportional to the percentage of fat in the body, affects the function and proliferation of regulatory T-cells (Treg), suppressing their activity and increasing the risk of acute GVHD.1818. Fuji S, Kim SW, Yoshimura K, Akiyama H, Okamoto S, Sao H, Takita J, Kobayashi N, Mori S; Japan Marrow Donor Program. Possible association between obesity and posttransplantation complications including infectious diseases and acute graft-versus-host disease. Biol Blood Marrow Transplant. 2009;15(1):73-82. A higher level of visceral fat is found in survivors.
PROTEIN-CALORIE MALNUTRITION
Even with the current epidemiological transition, there is still a considerable percentage of patients with sarcopenia2323. Morishita S, Kaida K, Tanaka T, Itani Y, Ikegame K, Okada M, et al. Prevalence of sarcopenia and relevance of body composition, physiological function, fatigue, and health-related quality of life in patients before allogeneic hematopoietic stem cell transplantation. Support Care Cancer. 2012;20(12):3161-8. and protein-calorie malnutrition, and patients at nutritional risk.2424. Baumgartner A, Bargetzi M, Bargetzi A, Zueger N, Medinger M, Passweg J, et al. Nutritional support practices in hematopoietic stem cell transplantation centers: A nationwide comparison. Nutrition. 2017;35:43-50.
We have known for decades that malnutrition negatively affects the outcomes of HSCT. A retrospective study with 2,238 patients undergoing autologous and allogeneic transplantation found a significant association between weight loss within 150 days of HSCT and a worse prognosis.2828. Deeg HJ, Seidel K, Bruemmer B, Pepea M, Appelbaum F. Impact of patient weight on non-relapse mortality after marrow transplantation. Bone Marrow Transplant. 1995;15(3):461-8.
A study looking at 544 adult patients undergoing allogeneic transplantation found higher mortality from infection and disease relapse in patients with BMI under 20kg/m22. Majhail NS, Rizzo JD, Lee SJ, Aljurf M, Atsuta Y, Bonfim C, Burns LJ, Chaudhri N, Davies S, Okamoto S, Seber A, Socie G, Szer J, Van Lint MT, Wingard JR, Tichelli A; Center for International Blood and Marrow Transplant Research (CIBMTR); American Society for Blood and Marrow Transplantation (ASBMT); European Group for Blood and Marrow Transplantation (EBMT); Asia-Pacific Blood and Marrow Transplantation Group (APBMT); Bone Marrow Transplant Society of Australia and New Zealand (BMTSANZ); East Mediterranean Blood and Marrow Transplantation Group (EMBMT),; Sociedade Brasileira de Transplante de Medula Ossea (SBTMO). Recommended screening and preventive practices for long-term survivors after hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2012;18(3):348–71..2929. Le Blanc K, Ringdén O, Remberger M. A low body mass index is correlated with poor survival after allogeneic stem cell transplantation. Haematologica. 2003;88(9):1044-52. Weight loss over 10% is also associated with higher post-allogeneic-transplant mortality.2626. Urbain P, Birlinger J, Ihorst G, Biesalski HK, Finke J, Bertz H. Body mass index and bioelectrical impedance phase angle as potentially modifiable nutritional markers are independent risk factors for outcome in allogeneic hematopoietic cell transplantation. Ann Hematol. 2013;92(1):111-9.
In a stage of protein-calorie malnutrition, involuntary weight loss and decreased levels of plasma proteins, such as albumin, there is a high risk of toxicity from chemotherapy and the other drug classes administered.3030. Bay JO, Dendoncker C, Angeli M, Biot T, Chikhi M, Combal C, et al. Prise en charge nutritionnelle des patients hospitalisés pour allogreffe de CSH: recommandations de la Société francophone de greffe de moelle et de thérapie cellulaire (SFGM-TC). Bull Cancer. 2016;103(11S):S201-6. Albumin is the most abundant protein in human plasma, and it has multiple roles in body homeostasis, and an essential role as a drug-transport protein. This may have pharmacokinetic implications on clinical therapy, affecting plasma concentrations of chemotherapeutic agents.3131. Gatta A, Verardo A, Bolognesi M. Hypoalbuminemia. Intern Emerg Med. 2012;7(Suppl 3):S193-9. Review.
Pre-HSCT hypoalbuminemia has a significant impact on these patients’ survival. Patients with albumin levels under 3.2g/dL have significantly lower disease-free survival than those with levels over 3.2g/dL.3232. Sivgin S, Baldane S, Ozenmis T, Keklik M, Kaynar L, Kurnaz F, et al. The impact of pretransplant hypoalbuminemia on survival in patients with leukemia who underwent allogeneic hematopoietic stem cell transplantation (alloHSCT): a nutritional problem? Transplant Proc. 2013;45(9):3371-4. Low BW also impairs tolerability to treatment, contributing to higher chemotherapy-mediated toxicity due to related changes in drug pharmacokinetics.3333. Ladas EJ, Sacks N, Meacham L, Henry D, Enriquez L, Lowry G, et al. A Multidisciplinary Review of Nutrition Considerations in the Pediatric Oncology Population: A Perspective From Children’s Oncology Group. Nutr Clin Pract. 2005;20(4):377-93. Review.
Another major downside of an impaired nutritional status in patients undergoing HSCT is the longer period of aplasia during the transplant, in which low BMI, hypoalbuminemia, and increased urinary nitrogen excretion are associated with a longer time to neutrophil engraftment, leading to increased susceptibility to infections,3434. Hadjibabaie M, Tabeefar H, Alimoghaddam K, Iravani M, Eslami K, Honarmand H, et al. The relationship between body mass index and outcomes in leukemic patients undergoing allogeneic hematopoietic stem cell transplantation. Clin Transplant. 2012;26(1):149-55. in addition to financial losses, since these patients usually require prolonged hospitalization.3030. Bay JO, Dendoncker C, Angeli M, Biot T, Chikhi M, Combal C, et al. Prise en charge nutritionnelle des patients hospitalisés pour allogreffe de CSH: recommandations de la Société francophone de greffe de moelle et de thérapie cellulaire (SFGM-TC). Bull Cancer. 2016;103(11S):S201-6.
In addition to the anticipated harm, protein-calorie malnutrition and low oral intake are associated with increased GVHD rates3535. Mattsson J, Westin S, Edlund S, Remberger M. Poor oral nutrition after allogeneic stem cell transplantation correlates significantly with severe graft-versus-host disease. Bone Marrow Transplant. 2006;38(9):629-33. − particularly of the lungs, gastrointestinal tract and oral cavity. Vitamins A and D deficiency is also associated with higher incidence and severity of GVHD.3636. von Bahr L, Blennow O, Alm J, Björklund A, Malmberg KJ, Mougiakakos D, et al. Increased incidence of chronic GvHD and CMV disease in patients with vitamin D deficiency before allogeneic stem cell transplantation. Bone Marrow Transplant. 2015;50(9):1217-23.
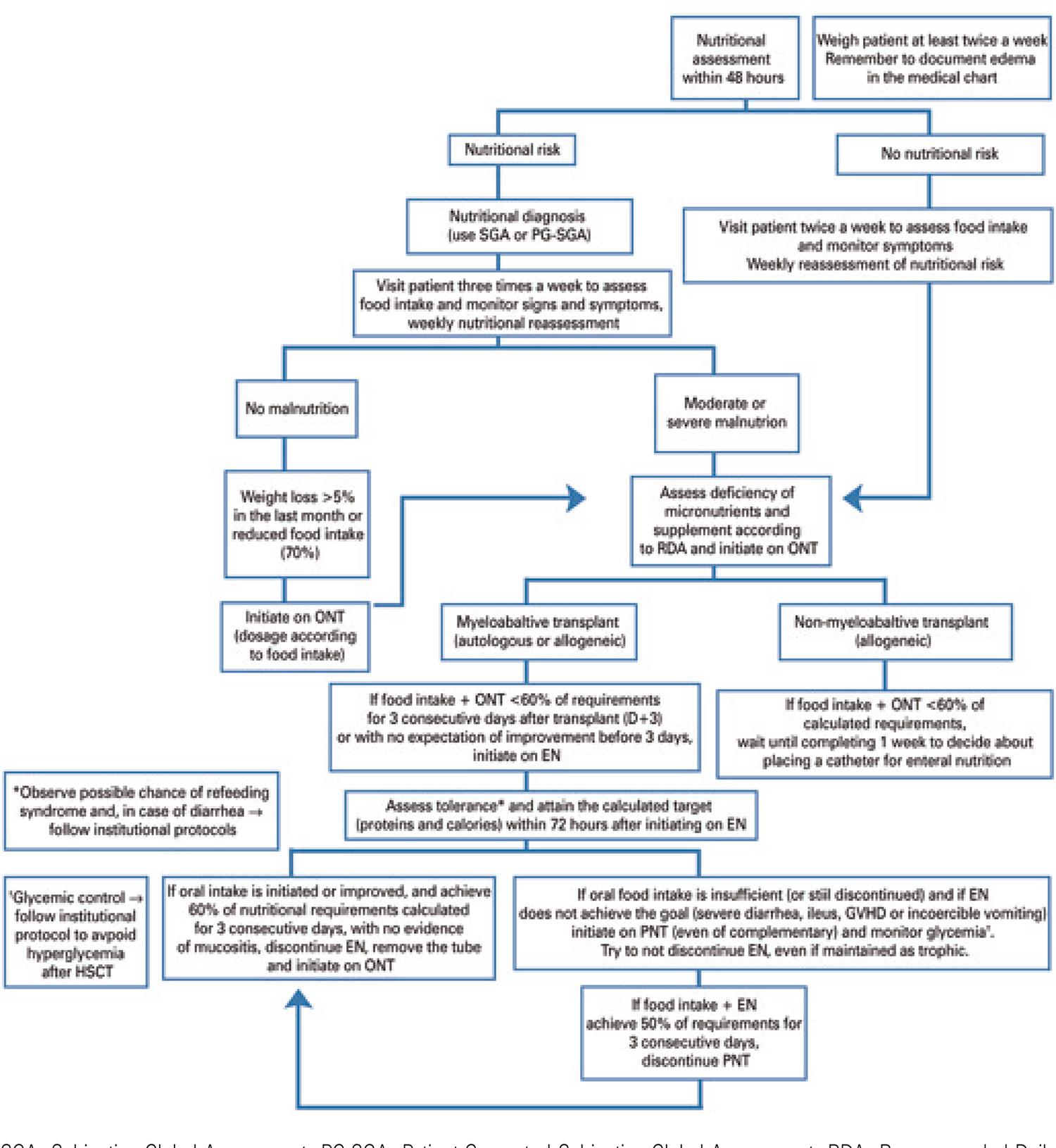
In this sense, it is recommended that the pre-HSCT nutritional assessment be conducted within 30 days before admission for HSCT, aiming to restore or maintain the pre-HSCT nutritional status and correct any nutritional deficiencies, with the view to minimize the deleterious effects of HSCT on nutritional status.
By evaluating the nutritional status, changes in food intake and nutritional semiology, it is possible to define groups of patients at a higher risk, allowing for rapid and effective nutritional intervention to curb protein catabolism.3737. Brasil. Ministério da Saúde. Secretaria de Atenção à Saúde. Departamento de Atenção Especializada e Temática. Manual de terapia nutricional na atenção especializada hospitalar no âmbito do Sistema Único de Saúde – SUS [Internet]. Brasília (DF): Ministério da Saúde; 2016.
NUTRITIONAL ASSESSMENT
Patients undergoing HSCT must be screened immediately upon admission for the procedure, or for any other hospital admission related with complications from the treatment or conditioning regimens administered. They are usually considered at nutritional risk or already malnourished, due to their underlying disease,3838. Gómez-Candela C, Luengo LM, Cos AI, Martínez-Roque V, Iglesias C, Zamora P, et al. [Subjective global assessment in neoplastic patients]. Nutr Hosp. 2003;18(6):353-7. Spanish. the chemotherapy received, and treatment toxicity. The different cytotoxic agents, radiation therapy, and other new drugs used in oncology-hematology treatment affect not only tumor cells, but also healthy cells, particularly those with high replication rates, such as lymphocytes and gastrointestinal tract cells (enterocytes and colonocytes). The effects on these cells lead to major functional changes in the digestive tract and the immune system, resulting in malabsorption, and seriously impairing the patient’s nutritional status.3939. Gómez-Candela C, Canales Albendea MA, Palma Milla S, De Paz Arias R, Díaz Gómez J, Rodríguez-Durán D, et al. Intervención nutricional en el paciente oncohematológico. Nutr Hosp. 2012;27(3):669-80.,4040. August DA, Huhmann MB; American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. A.S.P.E.N. clinical guidelines: nutrition support therapy during adult anticancer treatment and in hematopoietic cell transplantation. JPEN J Parenter Enteral Nutr. 2009;33(5):472-500. Reduced-intensity conditioning (RIC) regimens are mostly indicated for older patients, or individuals with multiple comorbidities, which would be a contraindication for myeloablative HSCT. Reduced-intensity conditioning reduces side effects, such as mucositis, and the duration of neutropenia.4141. Ljungman P, Urbano-Ispizua A, Cavazzana-Calvo M, Demirer T, Dini G, Einsele H, Gratwohl A, Madrigal A, Niederwieser D, Passweg J, Rocha V, Saccardi R, Schouten H, Schmitz N, Socie G, Sureda A, Apperley J; European Group for Blood and Marrow. Allogeneic and autologous transplantation for haematological diseases, solid tumours and immune disorders: definitions and current practice in Europe. Bone Marrow Transplant. 2006;37(5):439-49.,4242. Passweg JR, Baldomero H, Bader P, Bonini C, Cesaro S, Dreger P, et al. Hematopoietic stem cell transplantation in Europe 2014: more than 40 000 transplants annually. Bone Marrow Transplant. 2016;51(6):786-92.Allogeneic HSCT has considerable toxicity and leads to major inflammation, in addition to metabolic changes (such as cachexia), gastrointestinal symptoms and generalized effects, which can result in reduced diet intake and deterioration of the nutritional status. This behavior clearly puts patients at nutritional risk, which, additionally to the treatment, will negatively affect clinical outcomes.4343. Seguy D, Duhamel A, Rejeb MB, Gomez E, Buhl ND, Bruno B, et al. Better outcome of patients undergoing enteral tube feeding after myeloablative conditioning for allogeneic stem cell transplantation. Transplantation. 2012;94(3):287-94.
Considering a chronic disease, like cancer, and factors detrimental to the patient’s condition or clinical response to the treatment (such as age and psychosocial factors), there is a great chance of an impaired nutritional status at important stages of the treatment − and particularly during HSCT. Some diagnoses consider weight loss, muscle wasting, and inflammation, and characterize the presence of cachexia into different phases (pre-cachexia or refractory cachexia).
Of the 503 patients assessed in Germany4444. Schwarz S, Prokopchuk O, Esefeld K, Gröschel S, Bachmann J, Lorenzen S, et al. The clinical picture of cachexia: a mosaic of different parameters (experience of 503 patients). BMC Cancer. 2017;17(1):130. for the presence of cachexia as per the criteria by Fearon et al.,4545. Fearon K, Strasser F, Anker SD, Bosaeus I, Bruera E, Fainsinger RL, et al. Definition and classification of cancer cachexia: an international consensus. Lancet Oncol. 2011;12(5):489-95. (weight loss <5% for the past 6 months, or weight loss of 2 to 5%, combined with BMI <20kg/m2, or weight loss of 2 to 5% combined with the presence of sarcopenia), 131 were diagnosed with cachexia, of which 15.2% were hematology patients. The authors showed that patients with cachexia had multiple symptoms, such as anemia and impaired kidney and liver function (cholestasis).
However, in order to standardize the discussion, we will address only the diagnosis of malnutrition or low weight, based on anthropometric measurements.
Nutritional status assessment with a validated tool before, during and after HSCT is still little discussed and documented, but it is certainly needed. There is no consensus in the literature about different assessment methods to be used in each phase, but it is a fact that this assessment must take place according to each institution’s protocol and comprise the main phases of HSCT: admission; onset of preparation chemotherapy; day of the HSC infusion; onset of immunosuppression; hospital discharge; 1 month and 3 months after HSCT; 6 months and 1 year after allogeneic HSCT (in an outpatient setting).
The American Society Parenteral and Enteral Nutrition (ASPEN)4040. August DA, Huhmann MB; American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. A.S.P.E.N. clinical guidelines: nutrition support therapy during adult anticancer treatment and in hematopoietic cell transplantation. JPEN J Parenter Enteral Nutr. 2009;33(5):472-500.and the European Society Parenteral and Enteral Nutrition (ESPEN),4646. Bozzetti F, Arends J, Lundholm K, Micklewright A, Zurcher G, Muscaritoli M; ESPEN. ESPEN Guidelines on Parenteral Nutrition: non-surgical oncology. Clin Nutr. 2009;28(4):445-54. in their consensus statements, recommend nutritional screening and intervention if the patient is incapable of maintaining their nutritional status. The latest consensus for oncology patients, from 2017,4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48. recommends that, for early detection of nutritional disorders, food intake, weight changes and BMI be regularly assessed, starting on cancer diagnosis, and reviewed according to the patient’s clinical stability. It states that, for this purpose, parameters such as BMI, weight loss and food records must be used, or a validated screening tool, like the Nutritional Risk Screening (NRS-2002), Malnutrition Universal Screening Tool (MUST), Malnutrition Screening Tool (MST) or Mini Nutritional Assessment Short Form (MNA-SF). It is worth noting that an abnormal screening result (presence of risk), alone, does not provide enough information for developing a patient’s plan of care. Patients at risk must be followed and evaluated by more specific tools, and only then can an intervention be designed. Therefore, ESPEN strongly recommends that, after identification of the risk, the patient undergo an objective and quantitative evaluation of food intake, impact of symptoms, muscle mass, physical performance and grade of systemic inflammation. It also advises that this evaluation be repeated at frequent intervals (e.g. fortnightly, monthly, every 6 months, as appropriate). The tools listed for this step are: Subjective Global Assement (SGA), Patient-Generated Subjective Global Assessment (PG-SGA) or Mini Nutritional Assessment (MNA).
Most concerning to patient care teams and HSCT center managers is that nutritional risk is associated with increased mortality and higher hospital costs.4848. Khalatbari-Soltani S, Marques-Vidal P. Impact of nutritional risk screening in hospitalized patients on management, outcome and costs: A retrospective study. Clin Nutr. 2016;35(6):1340-6.
Thus, when caring for HSCT recipients, it is vital that nutritional screening and assessment be performed upon admission, as well as during outpatient care before admission, while preparing for HSCT. The frequency must be based on the presence or absence of nutritional risk, must not exceed 15 days for outpatients already at nutritional risk, and 30 days for those not yet at risk. Inpatients must be screened up to 48 hours after admission and reassessed after 7 days. This weekly reassessment must be carried out until patient is discharged home.4040. August DA, Huhmann MB; American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. A.S.P.E.N. clinical guidelines: nutrition support therapy during adult anticancer treatment and in hematopoietic cell transplantation. JPEN J Parenter Enteral Nutr. 2009;33(5):472-500.,4949. Brasil. Ministério da Saúde. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Consenso Nacional de Nutrição Oncológica. 2ª ed [Internet]. Rio de Janeiro (RJ): INCA; 2015. 186 p [citado 2019 Maio 10]. Disponível em: http://www1.inca.gov.br/inca/Arquivos/Consenso_Nutricao_vol_II_2_ed_2016.pdf
http://www1.inca.gov.br/inca/Arquivos/Co...
The screening period can change, depending on quality criteria of the nutritional care. Nutritional risk screening carried out within 24 hours of admission is one of the requirements of quality audit processes such as that of the Joint Commission International (JCI).5050. Consórcio Brasileiro de Acreditação de Sistemas e Serviços de Saúde. Padrões de Acreditação da Joint Commission International para Hospitais. Rio de Janeiro: 2011.
There is no definition regarding the best tool to be used in reassessing cancer patients. Many are not valid for reevaluations. However, in fact, hematological cancer patients undergoing HSCT must be followed and reevaluated on a regular basis. Nutritional visits allow for early and important interventions at each treatment stage, and changes in the nutritional or functional status must be identified. The tools that allow for calculation of final diagnostic scores can be an alternative to evaluate patients at different time points. Functional or quality of life assessments, in this sense, can be important tools to guide adjustments over the course of treatment (good examples included the PG-SGA and dynamometry for function assessment of muscle strength).
TOOLS FOR NUTRITIONAL ASSESSMENT
NRS-2002
The tool most suitable for assessing the nutritional risk of inpatients is the NRS-2002, which takes into account the patient’s age (added to the final screening score), includes all clinical and surgical patients,5151. Kondrup J, Allison SP, Elia M, Vellas B, Plauth M; Educational and Clinical Practice Committee, European Society of Parenteral and Enteral Nutrition (ESPEN). ESPEN guidelines for nutrition screening 2002. Clin Nutr. 2003;22(4):415-21. and is recommended by the ESPEN for in-hospital screening. It has been validated in a review of 128 randomized controlled nutritional support trials, to determine whether it could distinguish patients with positive clinical outcomes due to nutritional intervention from those that did not benefit from Nutritional Therapy (NT).5252. Polednak AP. Indicators of nutritional screening in hospital records of newly diagnosed Hispanic and Asian-American adult cancer patients in Connecticut. Nutrition. 2008;24(10):1053-6. NRS-2002 is the first nutritional screening tool developed on the ground of evidence-based medicine.5151. Kondrup J, Allison SP, Elia M, Vellas B, Plauth M; Educational and Clinical Practice Committee, European Society of Parenteral and Enteral Nutrition (ESPEN). ESPEN guidelines for nutrition screening 2002. Clin Nutr. 2003;22(4):415-21.,5353. Kondrup J, Rasmussen HH, Hamberg O, Stanga Z, Ad Hoc ESPEN Working Group. Nutritional risk screening (NRS 2002): a new method based on an analysis of controlled clinical trials. Clin Nutr. 2003;22(3):321-36.
Also, it has shown a high predictive value and low interobserver variation (k=0.67).5353. Kondrup J, Rasmussen HH, Hamberg O, Stanga Z, Ad Hoc ESPEN Working Group. Nutritional risk screening (NRS 2002): a new method based on an analysis of controlled clinical trials. Clin Nutr. 2003;22(3):321-36. Nutritional risk is assessed by the combination of current nutritional status and disease severity, with the first including as variables the BMI, recent weight loss and diet intake in the week before hospital admission.
In a national study of HSCT centers in Switzerland2424. Baumgartner A, Bargetzi M, Bargetzi A, Zueger N, Medinger M, Passweg J, et al. Nutritional support practices in hematopoietic stem cell transplantation centers: A nationwide comparison. Nutrition. 2017;35:43-50. with a total of 621 transplants carried out in 2014 (226 allogeneic and 395 autologous), all centers involved in allogeneic HSCT (n=3) were conducting nutritional screening mostly based on the NRS-2002. Only one center was using a screening method developed in-house, incorporating indirect calorimetry (IC) parameters and bioelectrical impedance. As for centers conducting autologous HSCT (n=7), only half had nutritional screening protocols. Three centers would call on a dietitian only if malnutrition was suspected during patient hospitalization.
Liu et al.,5454. Liu P, Zhang ZF, Cai JJ, Wang BS, Yan X. NRS2002 assesses nutritional status of leukemia patients undergoing hematopoietic stem cell transplantation. Chin J Cancer Res. 2012;24(4):299-303. investigated in 99 Chinese patients with leukemia, whether NRS-2002 was capable of properly assessing risk before and after HSCT, and if there were any age- or sex-related differences. The mean age was 32.4 years and the mean time in a laminar flow room was 30 days. Two dietitians were in charge of applying the tool upon admission (before HSCT) and the day the HSCT was completed (after). Out of patients screened, 22.2% were at nutritional risk. At the same time, 15.4% of patients with BMI ≥18.5kg/m2 were at nutritional risk, showing that the BMI alone would not have accurately identified the presence or absence of nutritional risk. Patients were grouped by sex, age and other conditions, and the presence of risk was compared. The authors showed there is no significant difference in the presence of risk by sex; however, they found a difference for age, matching degree (patients under 30 years and not fully-matched have higher nutritional risk), weight loss and decreased food intake or BMI (p<0.01). All patients were identified as at nutritional risk after HSCT, since the transplant alone, according to the authors, already confers score 3 in disease severity (i.e. nutritional risk), showing that the NRS-2002 is not an appropriate tool at this stage. However, the authors pointed out that 63,6% of patients had a score of 6 (the maximum score, considering there were no patients over 70 years in the sample), which means that, although the NRS-2002 cannot distinguish risk after HSCT, patients had higher scores than before the procedure. In addition, 77.8% of patients lost weight after HSCT, of which 63.6% lost >5% at one month.5454. Liu P, Zhang ZF, Cai JJ, Wang BS, Yan X. NRS2002 assesses nutritional status of leukemia patients undergoing hematopoietic stem cell transplantation. Chin J Cancer Res. 2012;24(4):299-303.
Subjective Global Assement and Patient-Generated Subjective Global Assessment
The SGA is an instrument commonly used to assess the nutritional status of inpatients. It is a clinical method developed by Detsky et al.,5555. Detsky a S, McLaughlin JR, Baker JP, Johnston N, Whittaker S, Mendelson RA, et al. What is subjective global assessment of nutritional status? JPEN J Parenter Enteral Nutr. 1987;11(1):8-13. capable of assessing nutritional risk and, most importantly, identify patients in need for more specific NT.5555. Detsky a S, McLaughlin JR, Baker JP, Johnston N, Whittaker S, Mendelson RA, et al. What is subjective global assessment of nutritional status? JPEN J Parenter Enteral Nutr. 1987;11(1):8-13.
The SGA, when applied sequentially after the NRS-2002, increases the sensitivity and specificity of both tests in predicting adverse clinical outcomes. Thus, NT can be targeted for patients in true nutritional debt.5656. Raslan M, Gonzalez MC, Torrinhas RS, Ravacci GR, Pereira JC, Waitzberg DL. Complementarity of Subjective Global Assessment (SGA) and Nutritional Risk Screening 2002 (NRS 2002) for predicting poor clinical outcomes in hospitalized patients. Clin Nutr. 2011;30(1):49-53.
The SGA showed an association with scores Acute Physiology and Chronic Health Evaluation II (APACHE II) and Simplified Acute Physiology Score II (SAPS II), as well as with mortality in a descriptive cohort of 124 clinical and surgical critically-ill patients.5757. Amaral TF, Antunes A, Cabral S, Alves P, Kent-Smith L. An evaluation of three nutritional screening tools in a Portuguese oncology centre. J Hum Nutr Diet. 200821(6):575-83.
The limitation of the SGA is that, as the name says, it is subjective, and depends on the experience of the observer.
In 1995, an SGA adapted for cancer patients was validated: the PG-SGA. It is a self-applicable questionnaire with two parts. In the first part, patients themselves answer the questions, describing any changes in weight, food intake, cancer-related symptoms, and changes in functional capacity. In the second part, answered by the professional applying the questionnaire, questions are based on diagnosis-related factors that may increase metabolic demand, such as stress, fever, depression, fatigue, tumor staging or cancer treatment, and physical examination.5858. Ottery FD. Definition of standardized nutritional assessment and interventional pathways in oncology. Nutrition. 1996;12(1 Suppl):S15-9.A disadvantage of this method is the fact that some patients may have difficulties answering questions related to weight loss in previous months, as well as to describe food intake in the previous month, which may, in some instances, result in a different nutritional status classification.5959. Raslan M, Gonzalez MC, Dias MC, Paes-Barbosa FC, Cecconello I, Waitzberg DL. Aplicabilidade dos m??todos de triagem nutricional no paciente hospitalizado. Rev Nutr. 2008;21(5):553-61.
Patient-Generated Subjective Global Assessment has the advantage of letting patients have an active role, and it optimizes the time spent by the professional to complete the evaluation. It is appropriate to identify cancer patients who would benefit from preventive nutritional intervention during cancer treatment.6060. Peres GB, Valim GS, Silva VL, El-kik RM. Comparação entre métodos de Avaliação Subjetiva Global em oncologia Comparison between Subjective Global Assessment methods in oncology. Rev Cienc Salud. 2009;2(1):37-42.,6161. Barbosa-Silva MC, de Barros AJ. Subjective global assessment: Part 2. Review of its adaptations and utilization in different clinical specialties. Arq Gastroenterol. 2002;39(4):248-52. Review. The limitations are related with patients’ lack of understanding of the questions relative to weight loss, and how to describe their food intake.6060. Peres GB, Valim GS, Silva VL, El-kik RM. Comparação entre métodos de Avaliação Subjetiva Global em oncologia Comparison between Subjective Global Assessment methods in oncology. Rev Cienc Salud. 2009;2(1):37-42.The PG-SGA has high sensitivity and specificity, at 80% and 89%, respectively.6262. Ravasco P, Monteiro-Grillo I, Vidal PM, Camilo ME. Nutritional deterioration in cancer: the role of disease and diet. Clin Oncol (R Coll Radiol). 2003; 15(8):443-50.
Barritta de Defranchi et al.,6363. Barritta de Defranchi RL, Bordalejo A, Cañueto I, Villar A, Navarro E. Evolution of nutritional status in patients with autologous and allogeneic hematopoietic stem cell transplant. Support Care Cancer. 2015;23(5):1341-7. in a prospective, longitudinal cohort study, described and compared the nutritional status of 123 patients undergoing HSCT, admitted to a teaching hospital in Buenos Aires, at three different time points: within 24 hours from admission for transplant (usually 3 to 7 days before transplant for chemotherapy), upon discharge, and at the follow-up visit (10 days after discharge). The nutritional status was assessed by the BMI and the PG-SGA. Of the total patients, 36 had missing data in some of the study evaluation phases for different reasons, such as readmission, transfer to the, and even death. The mean age was 50.5 years. Multiple myeloma was the most frequent diagnosis (44.8%). Most subjects (80.5%) had been subjected to autologous HSCT. The authors found a significant difference between nutritional status on admission, discharge and follow-up: the average PG-SGA scores were 3.39 on admission, 12.81 on discharge, and 6.71 on outpatient follow-up (p<0.001). Most inpatients (94.3%) were well nourished upon admission. Considering the BMI, there were no low-weight patients in the sample, and most were overweight (34.1%) or obese (33.3%). Upon discharge, 59.7% were malnourished (PG-SGA B and C). Patients with a length of stay longer than 21 days scored 2.9 points higher in the PG-SGA than patients who stayed for less than 21 days (p=0.034). Patients under 60 years had worse PG-SGA scores (p=0.0007). Patients undergoing allogeneic HSCT had a higher score on discharge (i.e., poorer outcome) than those undergoing autologous HSCT (p=0.0152). On outpatient follow-up, the PG-SGA score was even higher than the score on admission (3.66 points higher). A large part of the patients (74.7%) has a score ≥4, requiring immediate nutritional intervention. Body weight increased from discharge to the follow-up visit (mean of 74.5kg to 75.4kg) but was still lower than on admission (mean weight 78kg).
Malnutrition Universal Screening Tool
The malnutrition universal screening tool is mostly used in protein-calorie malnutrition, which includes three clinical parameters (BMI, involuntary weight loss in the last 3 to 6 months, acute disease or fasting exceeding 5 days) and assigns to each item a score of zero, 1 or 2, as per the following description:
-
BMI >20kg/m2, score 0; 18.5-20kg/m2, score 1; under 18.5kg/m2, score 2;
-
Weight loss under 5%, score 0; 5% to 10%, score 1; over 10%, score 2;
-
Acute disease or fasting exceeding 5 days, if absent, score 0; if present, score 2.
After the first three steps, all scores are added up to calculate a general risk for malnutrition. A score over 2 rates the patient as at high risk for malnutrition; a score of 1, medium risk for malnutrition; and score 0, low risk for malnutrition. For each nutritional risk category, guidelines are offered to help healthcare professionals act based on the results proposed.6464. Nutricional T, Must D, Oncológicos P. Triagem Nutricional por meio do MUST no Paciente Oncológico em Radioterapia. Rev Bras Cancerol. 2016;62(1):27-34.,6565. British Association for Parenteral and Enteral Nutrition (BAPEN). Nutritional screening and care planning with the “MUST”. The “MUST” Explanatory Booklet: a guide to the Malnutrition universal Screening Tool (“MUST”) for Adults [Internet]. Redditch: BAPEN; 2003 [cited 2017 Mar 3]. Available from: https://www.health.gov.il/download/ng/N500-19.pdf
https://www.health.gov.il/download/ng/N5...
ANTHROPOMETRY
Cancer-associated malnutrition involves a prior and ongoing history of nutritional deficits, and leads to changes in body composition, including loss of body fat and lean mass, resulting in weight loss and changes in other anthropometric parameters.6666. Gómez Candela C, Olivar Roldán J, García M, Marín M, Madero R, Pérez-Portabella C, et al. Utilidad de un método de cribado de malnutrición en pacientes con cáncer. Nutr Hosp. 2010;25(3):400-5.
Anthropometric measures are of great importance for establishing determinants of malnutrition and overweight, among others, as an instrument of nutritional surveillance.6767. World Health Organization (WHO). Physical status: the use and interpretation of anthropometry. Report of a WHO Expert Committee. Geneva: WHO; 1955. [World technical report series; 854].
The anthropometric measures most commonly used in clinical practice to assess the nutritional status of cancer patients include BW, height, BMI, triceps skinfold, arm circumference (AC) and arm muscle circumference (AMC).6868. Monteiro RS, Cunha TR, Santos ME, Mendonça SS. Estimativa de peso, altura e índice de massa corporal em adultos e idosos americanos: revisão. Rev Cienc Salud. 2009;20(4):341-50. However, in clinical practice, due to some mobility restrictions, presence of accesses, edema or immobility, the use of said parameters is not always possible. The inclusion into protocols should be evaluated with caution, even for comparing the diagnosis found to other tools applied (such as subjective assessments). An interesting option is to use an anthropometric indicator for comparing changes over time.
Anthropometry is the most commonly used method because it is non-invasive, easy to execute, fast, low-cost, feasible at the bedside, and yields reliable results if performed by trained professionals. The downside is that it is not capable of detecting recent disturbances to the nutritional status and identifying specific nutritional deficiencies. Of 226 adult outpatients with cancer followed-up in a Spanish study, 64% were malnourished (as assessed by the PG-SGA), and the BMI was not useful for NT-related decision-making, since half of the population (56.5%) had appropriate BMI; however, they observed that, as the BMI decreased, nutritional difficulties increased.6969. Marín Caro MM, Gómez Candela C, Castillo Rabaneda R, Lourenço Nogueira T, García Huerta M, Loria Kohen V, et al. Evaluación del riesgo nutricional e instauración de soporte nutricional en pacientes oncológicos, según el protocolo del grupo Español de Nutrición y Cáncer. Nutr Hosp. 2008;23(5):458-68.
WEIGHT AND BODY MASS INDEX
The weight is the sum of body compartments and reflects the protein-calorie balance of an individual. To measure weight, a weighing scale (platform or electronic) with 100g precision is needed. It must be installed on a level floor; the individual should be standing on the center of the scale, wearing as little as possible, barefoot, standing straight, with their feet together and arms extended alongside their body.7070. Brasil. Ministério da Saúde. Vigilância Alimentar e Nutricional. Orientações básicas para a coleta, processamento, análise de dados e informação em serviços de saúde [Internet]. Brasília (DF): Ministério da Saúde; 2004 [citado 2017 Jun 20]. Disponível em: http://189.28.128.100/nutricao/docs/geral/orientacoes_basicas_sisvan.pdf
http://189.28.128.100/nutricao/docs/gera...
The weight must be corrected for excess fluids (e.g. pleural edema, ascites and/or edema of any nature).4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48.
Japanese researchers evaluated 48 patients with lymphoma or myeloma undergoing autologous HSCT at a teaching hospital, in respect to the presence of dysgeusia as assessed via interview (divided into patients who developed dysgeusia during the treatment – 42% – and those who did not – 58%). The use of cryotherapy with ice chips before melphalan (120 minutes in total for each regimen) was significantly lower in the group that developed dysgeusia. Univariate and multivariate logistic regression analyses showed that the chemotherapy regimen used, and the presence of oral mucositis were independent risk factors for dysgeusia, while cryotherapy was an independent suppressor. There were no differences in weight loss between the groups; however, both lost approximately 4kg after autologous HSCT.7171. Okada N, Hanafusa T, Abe S, Sato C, Nakamura T, Teraoka K, et al. Evaluation of the risk factors associated with high-dose chemotherapy-induced dysgeusia in patients undergoing autologous hematopoietic stem cell transplantation: possible usefulness of cryotherapy in dysgeusia prevention. Support Care Cancer. 2016;24(9):3979-85.
Aoyama et al.,7272. Aoyama T, Imataki O, Mori K, Yoshitsugu K, Fukaya M, Okamura I, et al. Nutritional risk in allogeneic stem cell transplantation: rationale for a tailored nutritional pathway. Ann Hematol. 2017;96(4):617-25. studied 51 patients (mean age 51 years) undergoing allogeneic HSCT in a stem cell transplant department in Japan, in respect to weight loss, with some controlled variables in the NT indication flow chart at the study site. The items were assessed 1 to 2 days before the onset of pre-treatment and at the end of parenteral nutrition therapy (PNT; which can be indicated as a supplement), up to Day 100. Patients were divided into two groups: those who lost ≥7.5% of their weight in the past 3 months, and those who lost <7.5% in the same period. The authors found a strong, significant correlation (r=0.89; p<0.0001) between weight variations and changes in body composition (assessed by bioelectrical impedance). However, a weak correlation was found between weight variations and changes in fat mass (r=0.42; p=0.002). The mean BMI before the onset of pre-treatment was 22.1kg/m2, and weight loss was 4.5%. Acute GVHD was more frequent and severe in patients with weight loss ≥7.5%, suggesting that the nutritional status likely affects post-HSCT clinical outcomes. This group also showed higher scores for nutrition-related adverse events, and for a longer duration. The authors pointed out in the study that nutrition-related adverse events (such as nausea, mucositis, stomatitis, abnormal taste, anorexia and vomiting) were associated with increases in performance status, and it would be interesting if nutritional interventions would consider the severity of said adverse events.
Yang et al.,7373. Yang J, Xue SL, Zhang X, Zhou YN, Qin LQ, Shen YP, et al. Effect of body mass index on overall survival of patients with allogeneic hematopoietic stem cell transplantation. Eur J Clin Nutr. 2017;71(6):750-4. in a retrospective cohort study, investigated 267 Chinese patients (66.7% aged ≤40 years) diagnosed with AML or acute lymphoid leukemia (ALL), receiving total body irradiation or busulfan + cyclophosphamide as conditioning regimens. Patients were periodically monitored after HSCT until death or the last follow-up – 13 years of follow-up (mean group follow-up 19.7 months). Body mass index was sorted into patients with a lower BMI (low and normal weight as per the Chinese evaluation criteria, i.e. Body mass index <23kg/m2) and higher BMI (overweight and obesity). Of the total, 9.7% had low weight; 51.3%, normal weight; 16.9%, overweight; and 22.1%, obesity. There were no differences between BMI groups except for age (> and ≤40 years). A total of 93 patients (34.8%) died during the study follow-up. The overall survival was lower in the lower-BMI group, compared with the higher-BMI group (BMI ≥23kg/m2), with p=0.041. Within 3 years of HSCT, the overall survival was 55.7% for the lower-BMI group and 72.3% for the higher-BMI group. Compared with low-weight patients, normal-weight, overweight and obese patients had a lower odds ratio (OR), with a significant trend towards a higher overall survival as the BMI increased (p=0.019), after adjusting for age, sex, diagnosis, disease staging, type of graft, conditioning regimen, GVHD prophylaxis regimen, and time from diagnosis to transplant. Also, patients with a higher BMI survived longer, with a significant decrease of about 40% in the OR, compared with the low-BMI group. Authors believe that the difference in survival found between BMI groups (lower and higher) must be attributed to differences in the metabolism of chemotherapeutic drugs used for conditioning. Overweight and obese patients possibly have higher cumulative doses of the drugs or longer exposure times, which may lead to better outcomes.
Jaime-Pérez et al.,7474. Jaime-Pérez JC, Colunga-Pedraza PR, Gutiérrez-Gurrola B, Brito-Ramírez AS, Gutiérrez-Aguirre H, Cantú-Rodríguez OG, et al. Obesity is associated with higher overall survival in patients undergoing an outpatient reduced-intensity conditioning hematopoietic stem cell transplant. Blood Cells Mol Dis. 2013;51(1):61-5. looked at 77 patients with malignant or benign hematological diseases undergoing 6/6 HLA-compatible HSCT (64 allogeneic and 13 autologous), in an outpatient management model, treated at a teaching hospital in Mexico. The conditioning program consisted of a RIC regimen with ideal-body-weight-based dosing. There was no difference in mortality between patients with BMI <18 and ≥18kg/m2). Body mass index changes were recorded at each visit or at least every month. The data were reviewed in the first 6 months after HSCT and, then, every 3 months, until 1 year. Based on the BMI, 18.2% had low weight; 24.7% had normal weight; 33.7% were overweight and 23.4%, obese. There was a significant difference in patient weight before (mean weight 68.3kg) and after HSCT (mean weight 56.3kg), with p=0.014, but not in fat percentage or BMI (p=0.458 and p=0.067, respectively). There was no correlation between BMI > or <30 and the time to bone marrow engraftment (p=0.404) or duration of neutropenia – neutrophils <500x109/L (p=0.014). In the Kaplan-Meier curve for overall survival, there was no difference between the BMI and survival subgroups (p=0.059), however, when survival was compared between obese patients (n=18) and the rest of the group, a longer survival was seen for obese patients (p=0.017), for both autologous and allogeneic HSCT. There was no correlation between BMI and onset of GVHD in the allogeneic HSCT group investigated.
Espinoza et al.,7575. Espinoza M, Perelli J, Olmos R, Bertin P, Jara V, Ramírez P. Nutritional assessment as predictor of complications after hematopoietic stem cell transplantation. Rev Bras Hematol Hemoter. 2016;38(1):7-14. investigated 18 and 32 patients in Chile undergoing autologous and allogeneic HSCT, respectively, with a mean age of 41 years, of which 84% received myeloablative conditioning and 16% RIC. Nutritional assessment was conducted at the time of the HSCT, 10 days after, and upon discharge. The mean length of stay was 32 days and the mean follow-up was 41 months. On admission, there was no significant difference in nutritional parameters between the autologous and the allogeneic groups, and a significant difference in BMI after HSCT (27.3kg/m2 to 26,3kg/m2) was found only in the autologous group. Considering both types, the BMI decreased post-HSCT, compared to the pre-HSCT BMI (26.9kg/m2 to 26,1kg/m2), with p<0.01.
DYNAMOMETRY
The muscle capacity of malnourished individuals is significantly reduced, since protein-calorie malnutrition directly affects the loss of all skeletal muscle fibers and, as a consequence, decreases muscle strength.7676. Humphreys J, de la Maza P, Hirsch S, Barrera G, Gattas V, Bunout D. Muscle strength as a predictor of loss of functional status in hospitalized patients. Nutrition. 2002;18(7-8):616-20. Hand dynamometry, or hand grip strength, is a method used for nutritional assessment of patients, as a prognostic marker, and also in cancer patients. It is easy to apply, simple, fast and low-cost, and it predicts muscle function status.7777. Norman K, Stobäus N, Gonzalez MC, Schulzke JD, Pirlich M. Hand grip strength: outcome predictor and marker of nutritional status. Clin Nutr. 2011;30(2):135-42. Dynamometry is indicated as an auxiliary tool in nutritional screening.4949. Brasil. Ministério da Saúde. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Consenso Nacional de Nutrição Oncológica. 2ª ed [Internet]. Rio de Janeiro (RJ): INCA; 2015. 186 p [citado 2019 Maio 10]. Disponível em: http://www1.inca.gov.br/inca/Arquivos/Consenso_Nutricao_vol_II_2_ed_2016.pdf
http://www1.inca.gov.br/inca/Arquivos/Co...
The devices used for this grip strength measurement can be classified into four categories, hydraulic, pneumatic, mechanical and strain gauges (or load cells).7878. Schlüssel MM, Dos Anjos LA, Kac G. A dinamometria manual e seu uso na avaliação nutricional. Rev Nutr. 2008;21(2):223-35.
Pastore et al.,7979. Pastore CA, Oehlschlaeger MH, Gonzalez MC. Impacto do estado nutricional e da força muscular sobre o estado de saude geral e qualidade de vida em pacientes com cancer de trato gastrointestinal e de pulmão. Rev Bras Cancerol. 2013;59(1):43-9. assessed the impact of nutritional status and muscle strength on the quality of life of 77 patients with gastrointestinal and lung cancer, referred for chemotherapy for the first time, in southern Brazil. Patients has their nutritional status assessed by BMI and SGA, and muscle function by dynamometry (Jamar mechanical dynamometer). Three measures were obtained for each hand (dominant and non-dominant), and the highest was used. The general health status and quality of life domain was assessed with the European Organization for Research and Treatment of Cancer – Quality of Life Questionnaire Core-30 (EORTC QLQ-C30). Based on the BMI, 60% of sample had normal weight, however, the SGA found only 13% of well nourished patients. The mean quality of life score was 68±21.3. The mean grip strength in the non-dominant hand was 25.7±10.1Kgf, with no association with the quality of life (p=0.3). A poorer nutritional status was associated with lower quality of life scores, by both BMI (p=0.04) and SGA (p=0.007). There was a high prevalence of malnutrition, as per the SGA, and the nutritional status significantly affected the general health status and quality of life, however there was no correlation between nutritional status and muscle strength.
A study conducted in Switzerland with 37 patients with unresectable lung cancer found that hand dynamometry can be useful to assess functional and nutritional status.8080. Barata T, Santos C, Cravo M, Vinhas MD, Morais C, Carolino E, et al. MON-LB013: Handgrip Dynamometry and Patient-Generated Subjective Global Assessment in Patients With Non-Resectable Lung Cancer. Clin Nutr. 2015;34(Suppl 1):S255. It can be included in the evaluation of cancer patients along with other nutritional assessment tools. Nutritional status was obtained by the PG-SGA, and muscle function was assessed with a Jamar hand dynamometer in the non-dominant hand. Three measurements were obtained, and the highest measure was used. In the PG-SGA, 73% (n=27) of patients were moderately malnourished, and 8% (n=3) were severely malnourished. In total, 81% (n=30) were malnourished. The hand grip strength was below the 50th percentile in 57% of patients (n=21). There was a significant association between nutritional status and hand grip strength (p=0.026).
Tanaka et al.,8181. Tanaka S, Imataki O, Kitaoka A, Fujioka S, Hanabusa E, Ohbayashi Y, et al. Clinical impact of sarcopenia and relevance of nutritional intake in patients before and after allogeneic hematopoietic stem cell transplantation. J Cancer Res Clin Oncol. 2017;143(6):1083-92. investigated 34 patients with hematological disease undergoing allogeneic HSCT at a hospital, in Japan. The chemotherapy regimen was myeloablative for 14 patients and non-myeloablative for 20. A rehabilitation program was carried out five times a week, 7 days before the transplant, consisting of strength training and aerobic exercises, 20 minutes per day. The patient was followed-up until continuous infusion of immunosuppressants was discontinued (approximately 30 days after the transplant). In addition to tests assessing physical function (including hand grip), nutritional status was assessed at five different timepoints: upon admission, at the start of the preparation chemotherapy regimen, on the day of the transplant, when immunosuppression was started, and on discharge. Nutritional status was assessed based on calorie intake, weight changes, albumin, CRP and circumferences (for muscle measures). Sarcopenia was defined as per the criteria of the European Working Group on Sarcopenia in Older People (EWGSOP).8282. Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, Martin FC, Michel JP, Rolland Y, Schneider SM, Topinková E, Vandewoude M, Zamboni M; European Working Group on Sarcopenia in Older People. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010;39(4):412-23. The weight was significantly lower after HSCT than before (58.6±15.6kg and 56.0±14.5kg), p=0.001. Hand grip strength and upper-and lower-limb circumference decreased, bilaterally and significantly, after HSCT. Muscle strength significantly decreased by 6%, one month after HSCT, but recovered 3 months later. However, for patients with acute GVHD requiring corticosteroids, muscle loss was about 16% within 3 months after HSCT. The univariate analysis showed that oral energy intake after HSCT was a significant factor associated with weight loss. The authors concluded weight was a sensitive sarcopenia indicator; however, circumference was not the best method to assess muscle strength. In that study, the circumference varied significantly after HSCT, but the reduction was small. Nevertheless, patients with sarcopenia before HSCT are more susceptible to adverse events than patients without sarcopenia − and this explains the importance of this assessment.
COMPARISONS
Liu et al.,2525. Liu P, Wang B, Yan X, Cai J, Wang Y. Comprehensive evaluation of nutritional status before and after hematopoietic stem cell transplantation in 170 patients with hematological diseases. Chin J Cancer Res. 2016;28(6):626-33. assessed the nutritional status of 170 patients with hematological disease in Beijing, subjected to allogeneic HSCT, using anthropometric parameters and four tools: NRS-2002, MNA, SGA and MUST. The group included 116 men and 54 women, mean age 30 years (12±56), 65 cases of AML, 63 cases of ALL, 14 cases of chronic myeloid leukemia (CML), 3 lymphomas, 3 aplastic anemias and 22 myelodysplastic syndromes. Patients were assessed before undergoing HSCT and entering laminar flow rooms, and within 48 hours after leaving the room (mean length of stay was 30 days). Only 109 patients were assessed on the same day they left the room. Assessments were carried out by two dietitians. The treatment duration was 30 (±2.1) days. After HSCT, patients had a significant decrease in weight, hip circumference, waist-hip ratio, calf circumference, and AC, when compared to pre-HSCT measures. Before HSCT, the NRS-2002 identified that 21.2% of patients were at nutritional risk, compared to 100% after HSCT. The MUST indicated that before HSCT, 11.8% of patients had high nutritional risk, compared to 59.6% after HSCT. The MNA assessed that 0.06% of patients were malnourished before HSCT, compared with 19.3% after HSCT. The SGA identified that, before HSCT, 1.76% of patients had mild to severe malnutrition, a rate that increased to 83.3% after HSCT. The authors concluded that nutritional risk and malnutrition significantly increase after HSCT. A comparative analysis of nutritional screening after HSCT using the MUST yielded significantly different results than those obtained with the NRS-2002. The SGA indicated that 83.3% of patients had moderate to severe malnutrition, while the MNA identified 19.3% of patients as malnourished; this was probably due to the subjectivity of the SGA, which increased the likelihood of malnutrition when used for assessment. Irrespective of the assessment method, the evidence showed patients with hematological diseases suffer from a significantly deteriorated nutritional status after transplantation, and are at a relatively higher risk.
In a prospective study with 108 patients with leukemia after HSCT, Wang et al.,8383. Wang B, Yan X, Cai J, Wang Y, Liu P. Nutritional assessment with different tools in leukemia patients after hematopoietic stem cell transplantation. Chin J Cancer Res. 2013;25(6):762–9. assessed the nutritional status using different assessment methods (NRS-2002, MNA, MUST and SGA). The study included 77 men and 31 women, with a mean age of 8 (±56) years, 61 cases of acute lymphoid leukemia (ALL), 15 cases of AML, 14 cases of CML, and 19 cases of myelodysplastic syndrome (MDS). A total of 108 patients completed the SGA and 99 patients completed the NRS-2002, MNA and MUST. During the treatment, 85.2% of patients lost weight, with 50% losing more than 5%, and 42.6% significantly reducing their food intake. To assess the nutritional risk, the positive risk rates using the NRS-2002, the MNA and the MUST were 100%, 74.7% and 63.6%, respectively. There was a significant difference (p<0.05) between the NRS-2002, MNA and MUST. In the malnutrition assessment, the positive rate of the SGA (83.3%) was significantly higher than that of the MNA (17.2%), with p<0.05, and the rate of nutritional risk among leukemia patients aged ≤30 years was higher than that of patients >30 years (p<0.05). The authors suggested the NRS-2002 may be used to assess nutritional deficiencies, but its specificity is not high. The MNA is applicable for nutritional risk screening in the elderly, however it is not suitable for nutritional assessment. Thus, the combination of different screening tools and clinical laboratory indicators would be most recommendable for a precise and comprehensive assessment of nutritional status, providing better diagnostic accuracy.
SUMMARY TABLE
Table 4 presents suggested approached for nutritional screening and assessment of adult hematological cancer patients scheduled for HSCT at different treatment stages.
HSCT is a highly complex therapy, with major nutritional risks. Thus, it is of utmost importance that nutritional assessment and nutritional interventions be carried out properly, contributing to better clinical outcomes.
BODY COMPOSITION ASSESSMENT
The body composition assessment, including assessment of muscle mass and peripheral and visceral fat, has shown considerable association with the morbidity and mortality of different diseases, such as cancer.8484. Thibault R, Genton L, Pichard C. Body composition: why, when and for who? Clin Nutr. 2012;31(4):435-47.
85. Prado CM, Baracos VE, McCargar LJ, Reiman T, Mourtzakis M, Tonkin K, et al. Sarcopenia as a determinant of chemotherapy toxicity and time to tumor progression in metastatic breast cancer patients receiving capecitabine treatment. Clin Cancer Res. 2009;15(8):2920-6.
86. Thoresen L, Frykholm G, Lydersen S, Ulveland H, Baracos V, Prado CM, et al. Nutritional status, cachexia and survival in patients with advanced colorectal carcinoma. Different assessment criteria for nutritional status provide unequal results. Clin Nutr. 2013;32(1):65-72.-8787. Pichard C, Kyle UG, Morabia A, Perrier A, Vermeulen B, Unger P. Nutritional assessment: lean body mass depletion at hospital admission is associated with an increased length of stay. Am J Clin Nutr. 2004;79(4):613-8.
In HSCT, body composition has been studied, and has important correlations with complications and survival.8888. Kyle UG, Chalandon Y, Miralbell R, Karsegard VL, Hans D, Trombetti A, et al. Longitudinal follow-up of body composition in hematopoietic stem cell transplant patients. Bone Marrow Transplant. 2005;35(12):1171-7.
89. Gleimer M, Li Y, Chang L, Paczesny S, Hanauer DA, Frame DG, et al. Baseline body mass index among children and adults undergoing allogeneic hematopoietic cell transplantation: clinical characteristics and outcomes. Bone Marrow Transplant. 2015;50(3):402-10.-9090. Chughtai K, Song Y, Zhang P, Derstine B, Gatza E, Friedman J, et al. Analytic morphomics: a novel CT imaging approach to quantify adipose tissue and muscle composition in allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2016;51(3):446-50. Changes in nutritional status during HSCT serve as a prognostic indicator for these patients.9191. Hadjibabaie M, Iravani M, Taghizadeh M, Ataie-Jafari A, Shamshiri AR, Mousavi SA, et al. Evaluation of nutritional status in patients undergoing hematopoietic SCT. Bone Marrow Transplant. 2008;42(7):469-73.
92. Horsley P, Bauer J, Gallagher B. Poor nutritional status prior to peripheral blood stem cell transplantation is associated with increased length of hospital stay. Bone Marrow Transplant. 2005;35(11):1113-6.-9393. Ferreira ÉE, Guerra DC, Baluz K, de Resende Furtado W, da Silva Bouzas LF. Nutritional status of patients submitted to transplantation of allogeneic hematopoietic stem cells: a retrospective study. Rev Bras Hematol Hemoter. 2014;36(6):414-9.
Although most patients are not malnourished at the start of HSCT, low-weight and obese patients have a great risk of early death after HSCT; also, the deterioration in nutritional status during HSCT is also a negative prognostic indicator for these patients.9191. Hadjibabaie M, Iravani M, Taghizadeh M, Ataie-Jafari A, Shamshiri AR, Mousavi SA, et al. Evaluation of nutritional status in patients undergoing hematopoietic SCT. Bone Marrow Transplant. 2008;42(7):469-73.
Both protein-calorie malnutrition and obesity increase the morbidity and mortality risk, days of hospitalization, duration of immunosuppression, and the chance of developing GVHD.1818. Fuji S, Kim SW, Yoshimura K, Akiyama H, Okamoto S, Sao H, Takita J, Kobayashi N, Mori S; Japan Marrow Donor Program. Possible association between obesity and posttransplantation complications including infectious diseases and acute graft-versus-host disease. Biol Blood Marrow Transplant. 2009;15(1):73-82.,9191. Hadjibabaie M, Iravani M, Taghizadeh M, Ataie-Jafari A, Shamshiri AR, Mousavi SA, et al. Evaluation of nutritional status in patients undergoing hematopoietic SCT. Bone Marrow Transplant. 2008;42(7):469-73.,9292. Horsley P, Bauer J, Gallagher B. Poor nutritional status prior to peripheral blood stem cell transplantation is associated with increased length of hospital stay. Bone Marrow Transplant. 2005;35(11):1113-6.,9494. van der Meij BS, de Graaf P, Wierdsma NJ, Langius JA, Janssen JJ, van Leeuwen PA, et al. Nutritional support in patients with GVHD of the digestive tract: state of the art. Bone Marrow Transplant. 2013;48(4):474-82.,9595. Iestra JA, Fibbe WE, Zwinderman AH, van Staveren WA, Kromhout D. Body weight recovery, eating difficulties and compliance with dietary advice in the first year after stem cell transplantation: a prospective study. Bone Marrow Transplant. 2002;29(5):417-24.
The prevalence of obesity in HSCT varies between 10 and 34%,8888. Kyle UG, Chalandon Y, Miralbell R, Karsegard VL, Hans D, Trombetti A, et al. Longitudinal follow-up of body composition in hematopoietic stem cell transplant patients. Bone Marrow Transplant. 2005;35(12):1171-7.,8989. Gleimer M, Li Y, Chang L, Paczesny S, Hanauer DA, Frame DG, et al. Baseline body mass index among children and adults undergoing allogeneic hematopoietic cell transplantation: clinical characteristics and outcomes. Bone Marrow Transplant. 2015;50(3):402-10.,9393. Ferreira ÉE, Guerra DC, Baluz K, de Resende Furtado W, da Silva Bouzas LF. Nutritional status of patients submitted to transplantation of allogeneic hematopoietic stem cells: a retrospective study. Rev Bras Hematol Hemoter. 2014;36(6):414-9.,9696. Pereira AZ, Victor ES, Vidal Campregher P, Piovacari SM, Bernardo Barban JS, Pedreira WL Jr, et al. High body mass index among patients undergoing hematopoietic stem cell transplantation: results of a cross-sectional evaluation of nutritional status in a private hospital. Nutr Hosp. 2015;32(6):2874-9. and it is associated with a higher incidence of GVHD, infections and mortality.8989. Gleimer M, Li Y, Chang L, Paczesny S, Hanauer DA, Frame DG, et al. Baseline body mass index among children and adults undergoing allogeneic hematopoietic cell transplantation: clinical characteristics and outcomes. Bone Marrow Transplant. 2015;50(3):402-10.,9696. Pereira AZ, Victor ES, Vidal Campregher P, Piovacari SM, Bernardo Barban JS, Pedreira WL Jr, et al. High body mass index among patients undergoing hematopoietic stem cell transplantation: results of a cross-sectional evaluation of nutritional status in a private hospital. Nutr Hosp. 2015;32(6):2874-9. A recent study with patients undergoing allogeneic HSCT showed an inverse association between areas of visceral and peripheral fat and disease-free survival.9090. Chughtai K, Song Y, Zhang P, Derstine B, Gatza E, Friedman J, et al. Analytic morphomics: a novel CT imaging approach to quantify adipose tissue and muscle composition in allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2016;51(3):446-50.
Also the decreased muscle mass associated, among other factors, to the use of corticosteroids, is correlated with a poorer prognosis in the different types of HSCT.8888. Kyle UG, Chalandon Y, Miralbell R, Karsegard VL, Hans D, Trombetti A, et al. Longitudinal follow-up of body composition in hematopoietic stem cell transplant patients. Bone Marrow Transplant. 2005;35(12):1171-7. In allogeneic HSCT, this decrease is associated with a higher prevalence of chronic GVHD and low performance.8888. Kyle UG, Chalandon Y, Miralbell R, Karsegard VL, Hans D, Trombetti A, et al. Longitudinal follow-up of body composition in hematopoietic stem cell transplant patients. Bone Marrow Transplant. 2005;35(12):1171-7.,9494. van der Meij BS, de Graaf P, Wierdsma NJ, Langius JA, Janssen JJ, van Leeuwen PA, et al. Nutritional support in patients with GVHD of the digestive tract: state of the art. Bone Marrow Transplant. 2013;48(4):474-82.,9595. Iestra JA, Fibbe WE, Zwinderman AH, van Staveren WA, Kromhout D. Body weight recovery, eating difficulties and compliance with dietary advice in the first year after stem cell transplantation: a prospective study. Bone Marrow Transplant. 2002;29(5):417-24.
In most studies, body composition assessment in HSCT has been carried out by computed tomography (CT) scan, bone densitometry by whole-body dual-energy X-ray absorptiometry (DXA), and bioelectric impedance (BIA).8888. Kyle UG, Chalandon Y, Miralbell R, Karsegard VL, Hans D, Trombetti A, et al. Longitudinal follow-up of body composition in hematopoietic stem cell transplant patients. Bone Marrow Transplant. 2005;35(12):1171-7.,9090. Chughtai K, Song Y, Zhang P, Derstine B, Gatza E, Friedman J, et al. Analytic morphomics: a novel CT imaging approach to quantify adipose tissue and muscle composition in allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2016;51(3):446-50.,9797. Sommacal HM, Jochims AM, Schuch I, Silla LM. Comparação de métodos de avaliação nutricional empregados no acompanhamento de pacientes submetidos a transplante de células-tronco hematopoéticas alogênico. Rev Bras Hematol Hemoter. 2010;32(51):50-5.,9898. Inaba H, Yang J, Kaste SC, Hartford CM, Motosue MS, Chemaitilly W, et al. Longitudinal changes in body mass and composition in survivors of childhood hematologic malignancies after allogeneic hematopoietic stem-cell transplantation. J Clin Oncol. 2012;30(32):3991-7.However, the use of both CT scan and DXA, in this assessment, is associated with ionizing radiation and high cost, not allowing for appropriate patient follow-up,9090. Chughtai K, Song Y, Zhang P, Derstine B, Gatza E, Friedman J, et al. Analytic morphomics: a novel CT imaging approach to quantify adipose tissue and muscle composition in allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2016;51(3):446-50.,9999. Pereira AZ, Marchini JS, Carneiro G, Arasaki CH, Zanella MT. Lean and fat mass loss in obese patients before and after Roux-en-Y gastric bypass: a new application for ultrasound technique. Obes Surg. 2012;22(4):597-601.and presenting limitations for its performance in obese patients.9999. Pereira AZ, Marchini JS, Carneiro G, Arasaki CH, Zanella MT. Lean and fat mass loss in obese patients before and after Roux-en-Y gastric bypass: a new application for ultrasound technique. Obes Surg. 2012;22(4):597-601.
In the case of BIA, although free from ionizing radiation, low-cost and allowing for appropriate follow-up, the test has limitations in its use for patients with edema and obese patients, affecting the quality of results.9999. Pereira AZ, Marchini JS, Carneiro G, Arasaki CH, Zanella MT. Lean and fat mass loss in obese patients before and after Roux-en-Y gastric bypass: a new application for ultrasound technique. Obes Surg. 2012;22(4):597-601. In HSCT, this method is used to assess body composition, due to its practicality; however, there are limitations, such as hyper-hydration in these patients, which can lead to overestimation of the lean mass.8888. Kyle UG, Chalandon Y, Miralbell R, Karsegard VL, Hans D, Trombetti A, et al. Longitudinal follow-up of body composition in hematopoietic stem cell transplant patients. Bone Marrow Transplant. 2005;35(12):1171-7.,9797. Sommacal HM, Jochims AM, Schuch I, Silla LM. Comparação de métodos de avaliação nutricional empregados no acompanhamento de pacientes submetidos a transplante de células-tronco hematopoéticas alogênico. Rev Bras Hematol Hemoter. 2010;32(51):50-5.
Dualenergy X-ray absorptiometry is not offered in most Brazilian services, due to its high cost and/or unavailability. However, in pediatric patients undergoing allogeneic HSCT, most studies use DXA.100100. Ruble K, Hayat M, Stewart KJ, Chen A. Body composition after bone marrow transplantation in childhood. Oncol Nurs Forum. 2012;39(2):186-92.,101101. Mostoufi-Moab S, Ginsberg JP, Bunin N, Zemel BS, Shults J, Thayu M, et al. Body composition abnormalities in long-term survivors of pediatric hematopoietic stem cell transplantation. J Pediatr. 2012;160(1):122-8. We did not find any studies in adults.
Ultrasound (US) is a simple, low-cost, practical and portable method, has emerged as a possible tool for assessment of body composition in hospitalized and obese patients.9999. Pereira AZ, Marchini JS, Carneiro G, Arasaki CH, Zanella MT. Lean and fat mass loss in obese patients before and after Roux-en-Y gastric bypass: a new application for ultrasound technique. Obes Surg. 2012;22(4):597-601.,102102. Abe T, Bemben MG, Kondo M, Kawakami Y, Fukunaga T. Comparison of skeletal muscle mass to fat-free mass ratios among different ethnic groups. J Nutr Health Aging. 2012;16(6):534-8.,103103. Tillquist M, Kutsogiannis DJ, Wischmeyer PE, Kummerlen C, Leung R, Stollery D, et al. Bedside ultrasound is a practical and reliable measurement tool for assessing quadriceps muscle layer thickness. JPEN J Parenter Enteral Nutr. 2014;38(7):886-90. This method has been applied in clinical studies and allows for measurement of muscle thickness, with good correlation with gold-standard methods, such as CT scan or magnetic resonance (MRI).103103. Tillquist M, Kutsogiannis DJ, Wischmeyer PE, Kummerlen C, Leung R, Stollery D, et al. Bedside ultrasound is a practical and reliable measurement tool for assessing quadriceps muscle layer thickness. JPEN J Parenter Enteral Nutr. 2014;38(7):886-90.
104. Sanada K, Kearns CF, Midorikawa T, Abe T. Prediction and validation of total and regional skeletal muscle mass by ultrasound in Japanese adults. Eur J Appl Physiol. 2006;96(1):24-31.-105105. Maurits NM, Bollen AE, Windhausen A, De Jager AE, Van Der Hoeven JH. Muscle ultrasound analysis: normal values and differentiation between myopathies and neuropathies. Ultrasound Med Biol. 2003;29(2):215-25. Ultrasound in the elderly allows for assessment of sarcopenia, with results comparable to those of DXA.106106. Berger J, Bunout D, Barrera G, de la Maza MP, Henriquez S, Leiva L, et al. Rectus femoris (RF) ultrasound for the assessment of muscle mass in older people. Arch Gerontol Geriatr. 2015;61(1):33-8. Also, it is possible to study the association between quality of muscle mass and strength, assessing the echogeneity, which is based on pixels on the images.107107. Watanabe Y, Yamada Y, Fukumoto Y, Ishihara T, Yokoyama K, Yoshida T, et al. Echo intensity obtained from ultrasonography images reflecting muscle strength in elderly men. Clin Interv Aging. 2013;8:993-8.
108. Fukumoto Y, Ikezoe T, Yamada Y, Tsukagoshi R, Nakamura M, Mori N, et al. Skeletal muscle quality assessed from echo intensity is associated with muscle strength of middle-aged and elderly persons. Eur J Appl Physiol. 2012;112(4):1519-25.
109. Pillen S, Arts IM, Zwarts MJ. Muscle ultrasound in neuromuscular disorders. Muscle Nerve. 2008;37(6):679-93.-110110. McLean RR, Kiel DP. Developing consensus criteria for sarcopenia: an update. J Bone Miner Res. 2015;30(4):588-92. There are no studies using this method in HSCT.
One of the routine tests conducted in cancer patients, particularly those eligible for HSCT, is chest computed tomography (CT scan), which has the advantage of providing a precise quantification of lean mass and an exact differentiation between muscle and fat. The review of images, using specific tomographic views, has a good correlation with fat mass and lean mass throughout the body. Between these points, we have the third lumbar vertebra and the fourth thoracic vertebra.111111. Shen W, Punyanitya M, Wang Z, Gallagher D, St-Onge MP, Albu J, et al. Total body skeletal muscle and adipose tissue volumes: estimation from a single abdominal cross-sectional image. J Appl Physiol (1985). 2004;97(6):2333-8.
112. Kinsey CM, San José Estépar R, van der Velden J, Cole BF, Christiani DC, Washko GR. Lower Pectoralis Muscle Area Is Associated with a Worse Overall Survival in Non-Small Cell Lung Cancer. Cancer Epidemiol Biomarkers Prev. 2017;26(1):38-43.
113. Camus V, Lanic H, Kraut J, Modzelewski R, Clatot F, Picquenot JM, et al. Prognostic impact of fat tissue loss and cachexia assessed by computed tomography scan in elderly patients with diffuse large B-cell lymphoma treated with immunochemotherapy. Eur J Haematol. 2014;93(1):9-18.-114114. Go SI, Park MJ, Song HN, Kim HG, Kang MH, Lee HR, et al. Prognostic impact of sarcopenia in patients with diffuse large B-cell lymphoma treated with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone. J Cachexia Sarcopenia Muscle. 2016;7(5):567-76.
In addition to allowing for visualization of fat and lean mass, CT scan also assesses radiodensity, the average radiation attenuation in Hounsfield units which, when low, in some studies, seems to lead to a better prognosis than sarcopenia in hematological tumors.115115. Antoun S, Lanoy E, Iacovelli R, Albiges-Sauvin L, Loriot Y, Merad-Taoufik M, et al. Skeletal muscle density predicts prognosis in patients with metastatic renal cell carcinoma treated with targeted therapies. Cancer. 2013;119(18):3377-84.,116116. Chu MP, Lieffers J, Ghosh S, Belch AR, Chua NS, Fontaine A, et al. Skeletal muscle radio-density is an independent predictor of response and outcomes in follicular lymphoma treated with chemoimmunotherapy. PLoS One. 2015;10(6):e0127589.
There are few studies in HSCT; however, one study found a larger amount of visceral and subcutaneous fat associated with a shorter disease-free survival.9090. Chughtai K, Song Y, Zhang P, Derstine B, Gatza E, Friedman J, et al. Analytic morphomics: a novel CT imaging approach to quantify adipose tissue and muscle composition in allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2016;51(3):446-50. Since it is included in most pre-HSCT assessment protocols, this could be a promising test for assessment of body composition.
Assessment of body composition should be part of the routine nutritional assessment of patients undergoing HSCT. Each service should select the most appropriate and cost-effective method for their patients.
BIOCHEMISTRY
The nutritional status diagnosis is essentially clinical. Laboratory; tests are used as an ancillary tool and should not replace clinical evaluation.117117. Viana L, Gazal C, Loss S, Azevedo M. Avaliação Nutricional. In: Xavier RM, Dora JM, Barros E, editors. Laboratório na Prática Clínica - Consulta Rápida. 3a edição. Porto Alegre: Artmed; 2016. p. 240-50.In patients undergoing HSCT, classic markers of nutritional status, such as lymphocyte count, anergy skin testing, and the presence of nutritional deficiency anemia, are impaired. Also, the interpretation of serum protein levels is also difficult due to the degree of inflammation and potential associated infections, particularly in inpatients.118118. Seres DS, Van Way CW, editors. Nutrition Support for the Critically III. EUA: Humana Press; 2016. 283 p. It is worth noting the importance of sampling nutritional markers before the transplant, in an attempt to reduce the interference of these factors in the previous nutritional assessment.
PROTEINS USED FOR NUTRITIONAL STATUS ASSESSMENT
In the absence of inflammation (CRP <10mg/L), albumin levels <3.0g/dL indicate a potentially malnourished patient with a poorer prognosis. Although albumin does not adequately correlate with malnutrition in the presence of inflammation, it is still a strong indicator of risk for morbidity and mortality in these cases. Albumin has a half-life of approximately 20 days, which limits its use as an immediate marker of improved nutritional status. Transthyretin, also called prealbumin, has a short half-life (2 days), and is sensitive to any changes in protein synthesis and catabolism. It can be used for PNT monitoring. Retinol-binding protein has the shortest half-life (12 hours) and is a fine marker of protein depletion.117117. Viana L, Gazal C, Loss S, Azevedo M. Avaliação Nutricional. In: Xavier RM, Dora JM, Barros E, editors. Laboratório na Prática Clínica - Consulta Rápida. 3a edição. Porto Alegre: Artmed; 2016. p. 240-50.
It is noteworthy that screening of said proteins must be carried out concomitantly with sampling of acute-phase proteins. The CRP tests aims to quantify inflammation, helping in the interpretation of the tests at issue.117117. Viana L, Gazal C, Loss S, Azevedo M. Avaliação Nutricional. In: Xavier RM, Dora JM, Barros E, editors. Laboratório na Prática Clínica - Consulta Rápida. 3a edição. Porto Alegre: Artmed; 2016. p. 240-50.
INDIRECT ASSESSMENT OF CATABOLISM AND MUSCLE STATUS
Creatinine is formed from creatine, a compound found almost exclusively in muscle tissue. In patients with marked cachexia, creatinine levels may be a marker of muscle loss. In these patients, increased serum creatinine may indicate loss of kidney function, even with creatinine levels within normal ranges. The creatinine height index (CHI), based on creatinine levels in urine, may be used to estimate muscle mass, with an index >60% suggesting severe muscle depletion:117117. Viana L, Gazal C, Loss S, Azevedo M. Avaliação Nutricional. In: Xavier RM, Dora JM, Barros E, editors. Laboratório na Prática Clínica - Consulta Rápida. 3a edição. Porto Alegre: Artmed; 2016. p. 240-50.
Reference values for estimated urine creatinine are117117. Viana L, Gazal C, Loss S, Azevedo M. Avaliação Nutricional. In: Xavier RM, Dora JM, Barros E, editors. Laboratório na Prática Clínica - Consulta Rápida. 3a edição. Porto Alegre: Artmed; 2016. p. 240-50.23mg/kg ideal weight for men and 18mg/kg ideal weight for women.
The 24-hour urinary urea levels are used to calculate the nitrogen balance, which guides protein replacement therapy, and represents the difference between the nitrogen administered (protein supply) and excreted (skin, feces, urine). Values ≥-5g in 24 hours are considered adequate. Although an interesting tool to assess NT, it has limitations in patients with loss of kidney function and large extrarenal losses, such as diarrhea in patients with intestinal GVHD.117117. Viana L, Gazal C, Loss S, Azevedo M. Avaliação Nutricional. In: Xavier RM, Dora JM, Barros E, editors. Laboratório na Prática Clínica - Consulta Rápida. 3a edição. Porto Alegre: Artmed; 2016. p. 240-50.
Where:
AN: administered nitrogen
EN: excreted nitrogen
UU: 24-hour urinary urea in g.
OTHER TESTS
Micronutrient and trace element testing is not a routine in nutritional status assessment. However, severely malnourished patients may have complications associated with their nutritional deficiency. A high level of suspicion for these deficiencies is needed, since clinical signs are discreet, which makes diagnosis difficult, and testing must be guided by clinical suspicion.117117. Viana L, Gazal C, Loss S, Azevedo M. Avaliação Nutricional. In: Xavier RM, Dora JM, Barros E, editors. Laboratório na Prática Clínica - Consulta Rápida. 3a edição. Porto Alegre: Artmed; 2016. p. 240-50.
Although not traditionally a part of nutritional status assessment, electrolyte testing has a major role in patients undergoing HSCT receiving PNT or at risk for refeeding syndrome. Sodium, potassium, calcium, phosphorus, chloride and magnesium are greatly affected by chemotherapeutic drugs given to these patients, which influences PNT prescription. Marked weight loss or prolonged fasting (>10 days) are risk factors for refeeding syndrome, the hallmark of which is hypophosphatemia. Electrolyte replacement in these patients must be given before feeding is resumed.117117. Viana L, Gazal C, Loss S, Azevedo M. Avaliação Nutricional. In: Xavier RM, Dora JM, Barros E, editors. Laboratório na Prática Clínica - Consulta Rápida. 3a edição. Porto Alegre: Artmed; 2016. p. 240-50.
Assessment of liver function and glucose metabolism119119. Fuji S, Einsele H, Savani BN, Kapp M. Systematic Nutritional Support in Allogeneic Hematopoietic Stem Cell Transplant Recipients. Biol Blood Marrow Transplant. 2015;21(10):1707-13. (preferably via glycated hemoglobin - HbA1c), though not directly related with nutritional status, is essential in these patients. Patients undergoing HSCT have higher risk for metabolic syndrome and diabetes, and the presence of hyperglycemia has direct implications on nutrition prescription and the risk of post-transplant infection. The hepatotoxicity of the drugs used, the use of PNT, and weight gain post-treatment, in addition to prior changes in liver function, are relevant during patient follow-up.
PHYSICAL EXAMINATION WITH SPECIAL FOCUS ON NUTRITIONAL STATUS
Hematological diseases are frequently associated with changes in the nutritional status. Particularly in patients undergoing HSCT, a physical examination with special focus on nutritional status is essential during the entire process (pre-transplant stage, immediate post-transplant, and late post-transplant periods). Although patient history and physical examination are considered cornerstones for appropriate clinical diagnosis, this aspect is sometimes neglected in daily practice.120120. Patient-Generated Subjective Global. Assessment (PG-SGA). Physical examination from a Nutritional Standpoint [Internet]. USA: (PG-SGA); 2001 [cited 2017 Jan 1]. Available from: http://pt-global.org/wp-content/uploads/2014/12/Physical_Examination_from_a_Nutritional_Standpoint_Ottery-FD-2001.pdf
http://pt-global.org/wp-content/uploads/...
Proper conduction of the physical examination by objective assessment is capable of detecting malnutrition or nutritional risk, and assisting121121. Fischer M, JeVenn A, Hipskind P. Evaluation of muscle and fat loss as diagnostic criteria for malnutrition. Nutr Clin Pract. 2015;30(2):239-48. in defining the best type of nutritional support for each patient. When speaking about malnutrition, the term includes both obesity and protein-calorie malnutrition − which are both associated with increased risk in patients undergoing HSCT.119119. Fuji S, Einsele H, Savani BN, Kapp M. Systematic Nutritional Support in Allogeneic Hematopoietic Stem Cell Transplant Recipients. Biol Blood Marrow Transplant. 2015;21(10):1707-13.
Physical examination is an efficient, low-cost and relatively simple method.122122. Esper DH. Utilization of nutrition-focused physical assessment in identifying micronutrient deficiencies. Nutr Clin Pract. 2015;30(2):194-202. It must be done from head to toe. For a complete physical examination, one must use inspection, palpation, percussion and auscultation techniques. On inspection, the patient must be fully examined, checking for skeletal muscle symmetry,120120. Patient-Generated Subjective Global. Assessment (PG-SGA). Physical examination from a Nutritional Standpoint [Internet]. USA: (PG-SGA); 2001 [cited 2017 Jan 1]. Available from: http://pt-global.org/wp-content/uploads/2014/12/Physical_Examination_from_a_Nutritional_Standpoint_Ottery-FD-2001.pdf
http://pt-global.org/wp-content/uploads/...
,121121. Fischer M, JeVenn A, Hipskind P. Evaluation of muscle and fat loss as diagnostic criteria for malnutrition. Nutr Clin Pract. 2015;30(2):239-48. and making sure to ask patients or relatives about any apparent changes in physical conditions and functional alterations observed in recent months, or after the onset of treatment.121121. Fischer M, JeVenn A, Hipskind P. Evaluation of muscle and fat loss as diagnostic criteria for malnutrition. Nutr Clin Pract. 2015;30(2):239-48.In patients with mucositis, for instance, examination of the oral cavity may be a determinant of the type of nutritional support. Its clinical manifestations range from soft tissue erythema to the presence of pseudomembranes and erosive lesions, which may cause secondary infection and sepsis. Poor oral hygiene, periodontal disease and tooth decay are risk factors for chemotherapy-mediated oral toxicity.123123. Negrin RS, Toljanic JA. Oral toxicity associated with chemotherapy [Internet]. UpToDate; 2017 [cited 2017 Feb 5]. Available from: http://www.uptodate.com/contents/oral-toxicity-associated-with-chemotherapy#H35
http://www.uptodate.com/contents/oral-to...
The appropriate techniques for the physical examination of areas allowing for classification of patients as well-nourished, moderate and severe loss of lean mass and subcutaneous fat, are described in tables 5 and 6. Evaluators must look out for signs of recent weight loss, fluid retention, loss of lean and fat mass, and specific signs of macro and micronutrient deficiencies.124124. White JV, Guenter P, Jensen G, Malone A, Schofield M; Academy Malnutrition Work Group; A.S.P.E.N. Malnutrition Task Force; A.S.P.E.N. Board of Directors. Consensus statement: Academy of Nutrition and Dietetics and American Society for Parenteral and Enteral Nutrition: characteristics recommended for the identification and documentation of adult malnutrition (undernutrition). JPEN J Parenter Enteral Nutr. 2012;36(3):275-83.
Subcutaneous fat assessment and nutritional status classification as per the physical examination
The loss of muscle and subcutaneous fat during treatment are often associated with worse food acceptance, and seen as a natural response to the stress caused by the disease.121121. Fischer M, JeVenn A, Hipskind P. Evaluation of muscle and fat loss as diagnostic criteria for malnutrition. Nutr Clin Pract. 2015;30(2):239-48. However, in obese patients or patients with marked edema, fat and muscle loss may be not so simple to assess, which corroborates the importance of a weight loss history.120120. Patient-Generated Subjective Global. Assessment (PG-SGA). Physical examination from a Nutritional Standpoint [Internet]. USA: (PG-SGA); 2001 [cited 2017 Jan 1]. Available from: http://pt-global.org/wp-content/uploads/2014/12/Physical_Examination_from_a_Nutritional_Standpoint_Ottery-FD-2001.pdf
http://pt-global.org/wp-content/uploads/...
Loss of subcutaneous fat is observed in areas where fat tissue is normally present. Patients with inflammation, in particular, such as patients subjected to HSCT, loss of subcutaneous fat may be more indolent, even with major catabolism, and become disproportional to the loss of muscle mass.118118. Seres DS, Van Way CW, editors. Nutrition Support for the Critically III. EUA: Humana Press; 2016. 283 p.
Muscle assessment must include volume, tonus and functional capacity. In general, superior muscles are more prone to nutrition-related wasting; muscle wasting from inactivity, however, is more associated with inferior muscles.120120. Patient-Generated Subjective Global. Assessment (PG-SGA). Physical examination from a Nutritional Standpoint [Internet]. USA: (PG-SGA); 2001 [cited 2017 Jan 1]. Available from: http://pt-global.org/wp-content/uploads/2014/12/Physical_Examination_from_a_Nutritional_Standpoint_Ottery-FD-2001.pdf
http://pt-global.org/wp-content/uploads/...
Muscle mass assessment can be performed through grasping and pinching of the areas observed: starting at the superior body, examining the temporal and orbital regions; the frontal clavicular bone and pectoris major, advancing to the deltoid and frontal/dorsal trapezius, shoulders, acromion process and scapulae, the upper arm and triceps, followed by the thoracic and lumbar regions, the ribs and iliac crest, as well as dorsal hands and interosseous muscles; in the inferior body, the quadriceps, patella and posterior calf should be examined.121121. Fischer M, JeVenn A, Hipskind P. Evaluation of muscle and fat loss as diagnostic criteria for malnutrition. Nutr Clin Pract. 2015;30(2):239-48. Calf circumference is a simple alternative to assess muscle mass, especially in elderly patients. Values under 31cm3 are considered abnormal.
Combined evaluation with muscle function is important, due to the high prevalence of sarcopenia in this population.125125. Morishita S, Kaida K, Tanaka T, Itani Y, Ikegame K, Okada M, et al. Prevalence of sarcopenia and relevance of body composition, physiological function, fatigue, and health-related quality of life in patients before allogeneic hematopoietic stem cell transplantation. Support Care Cancer. 2012;20(12):3161-8. Special highlight should be given to hand grip strength assessment using a dynamometer, which can be carried out at the bedside and correlated with prognosis in cancer patients.126126. Kilgour RD, Vigano A, Trutschnigg B, Lucar E, Borod M, Morais JA. Handgrip strength predicts survival and is associated with markers of clinical and functional outcomes in advanced cancer patients. Support Care Cancer. 2013;21(12):3261-70. There are specific tables with cutoff points for sex and age; however, values under 30kg for men and 20kg for women are classified as decreased muscle strength.127127. Ali S, Garcia JM. Sarcopenia, cachexia and aging: diagnosis, mechanisms and therapeutic options - a mini-review. Gerontology. 2014;60(4):294-305.
Weight gain in these patients, particularly in the immediate post-transplant phase, may be related with fluid build-up, which can range from subcutaneous edema to anasarca. To assess the presence of subcutaneous edema, the sacral region must be examined in bedridden patients and the ankle region in outpatients.120120. Patient-Generated Subjective Global. Assessment (PG-SGA). Physical examination from a Nutritional Standpoint [Internet]. USA: (PG-SGA); 2001 [cited 2017 Jan 1]. Available from: http://pt-global.org/wp-content/uploads/2014/12/Physical_Examination_from_a_Nutritional_Standpoint_Ottery-FD-2001.pdf
http://pt-global.org/wp-content/uploads/...
The presence of ascites, pleural or pericardial effusion and anasarca must be recorded in patient progression notes.
FOOD ACCEPTANCE DURING HEMATOPOIETIC STEM CELL TRANSPLANTATION
Hematopoietic stem cell transplantation-associated clinical changes lead to changes in food intake and eating experience. Taste is a sensory modality involving cognitive and emotional aspects deeply rooted in identity, references associated to pleasure and to a wide range of experiences throughout life.129129. Epstein JB, Barasch A. Taste disorders in cancer patients: pathogenesis, and approach to assessment and management. Oral Oncol. 2010;46(2):77-81. Conditioning chemotherapy for HSCT is a procedure that can modify the eating experience.130130. Meirelles CS, Diez-Garcia RW. Experiências alimentares e alterações no paladar em pacientes submetidos a transplante de células tronco [Tese]. São Paulo: Universidade de São Paulo; 2016.
Dietary care must be started pre-transplant and continue during all HSCT phases and modalities, not only to ensure the right supply of nutrients and calories, but also to improve coping with the disease. Dietary restrictions, cultural and religious particularities and diet types (vegetarian, omnivorous, vegan, kosher, among others) must be probed to be considered during hospitalization.
When eating, patients express the difficulties experienced and emotional oscillations occurring since the diagnosis. The conditioning period is permeated by a series of challenges, and the day-to-day is full of symptoms, such as pain, nausea and vomiting, associated with suffering and anguish, which impact different spheres of life and relation with food.130130. Meirelles CS, Diez-Garcia RW. Experiências alimentares e alterações no paladar em pacientes submetidos a transplante de células tronco [Tese]. São Paulo: Universidade de São Paulo; 2016. Hospital diets usually have little flexibility to overcome difficulties, i.e. strict meal service times and monotonous choices.131131. Calleja Fernández A, Pintor de la Maza B, Vidal Casariego A, Villar Taibo R, López Gómez JJ, Cano Rodríguez I, et al. Food intake and nutritional status influence outcomes in hospitalized hematology-oncology patients. Nutr Hosp. 2015;31(6):2598-605. Dietitians must understand these issues in an integrated manner and manage nutritional care in a way to overcome these difficulties.
Nutrition management must be clarified and negotiated with patient and caregivers with direct influence on dietary care.132132. Lindman A, Rasmussen HB, Andersen NF. Food caregivers influence on nutritional intake among admitted haematological cancer patients - a prospective study. Eur J Oncol Nurs. 2013;17(6):827-34. Patient compliance improves as caregivers get more involved and commited to their nutrition. The toxicity inherent to conditioning limits the intake of calories and nutrients, which deserves attention of the team, since it is directly related with treatment outcomes.133133. Sommacal HM, Gazal CH, Jochims AM, Beghetto M, Paz A, Silla LM, et al. Clinical impact of systematic nutritional care in adults submitted to allogeneic hematopoietic stem cell transplantation. Rev Bras Hematol Hemoter. 2012;34(5):334-8.
DIETARY MANAGEMENT IN HEMATOPOIETIC STEM CELL TRANSPLANTATION
Dietary management aims to preserve or restore nutritional status, contributing to a better prognosis and reducing toxic effects related to the treatment; attenuate and overcome digestive symptoms; and improve patient experience during hospitalization and treatment. Estimating nutritional requirements at the start of treatment, monitoring food intake, detecting difficulties and promptly managing them are vital roles of nutritional care. The literature shows that food intake during conditioning corresponds to approximately half of the prescribed diet.131131. Calleja Fernández A, Pintor de la Maza B, Vidal Casariego A, Villar Taibo R, López Gómez JJ, Cano Rodríguez I, et al. Food intake and nutritional status influence outcomes in hospitalized hematology-oncology patients. Nutr Hosp. 2015;31(6):2598-605. Difficulties adapting to the hospital environment, presentation and attractiveness of dishes, and meal times are some of the factors related with low intake.131131. Calleja Fernández A, Pintor de la Maza B, Vidal Casariego A, Villar Taibo R, López Gómez JJ, Cano Rodríguez I, et al. Food intake and nutritional status influence outcomes in hospitalized hematology-oncology patients. Nutr Hosp. 2015;31(6):2598-605.,133133. Sommacal HM, Gazal CH, Jochims AM, Beghetto M, Paz A, Silla LM, et al. Clinical impact of systematic nutritional care in adults submitted to allogeneic hematopoietic stem cell transplantation. Rev Bras Hematol Hemoter. 2012;34(5):334-8.
Several complications that occur during hospitalization, especially those deriving from drug toxicity, require specific dietary procedures. Symptoms like nausea, vomiting, hyporexia, diarrhea and mucositis can be circumvented with changes in food offer, diet temperature and consistency management, introduction of enzymes (lactase, alpha galactosidase, among others) into foods during episodes of diarrhea due to enzyme deficiency, use of special supplemental foods and nutrients, among others.9494. van der Meij BS, de Graaf P, Wierdsma NJ, Langius JA, Janssen JJ, van Leeuwen PA, et al. Nutritional support in patients with GVHD of the digestive tract: state of the art. Bone Marrow Transplant. 2013;48(4):474-82.
TASTE
Taste is a complex process of multisensory integration,134134. Spence C. Multisensory flavour perception. Curr Biol. 2013;23(9):R365-9. that allows for identification of nutrients that should be consumed, and toxins and non-digestable materials that should be avoided.129129. Epstein JB, Barasch A. Taste disorders in cancer patients: pathogenesis, and approach to assessment and management. Oral Oncol. 2010;46(2):77-81.,135135. Elman I, Soares NS, Pinto ME. Análise da Sensibilidade do Gosto Umami em Crianças com Câncer. Rev Bras Cancerol. 2010;56(2):237-42. Taste allows us to recognize and distinguish sweetness, saltiness, sourness, bitterness and umami, which are the basic tastes or flavors.
In the oral cavity, taste is created by chemical substances diluted in saliva, which are identified by specific receptors on taste buds.129129. Epstein JB, Barasch A. Taste disorders in cancer patients: pathogenesis, and approach to assessment and management. Oral Oncol. 2010;46(2):77-81. Recognizing flavors in our mouth also depends on retronasal odor perception. When we exhale, we blow small air streams from the food to the upper back of the mouth, through the nose passage, as we chew or swallow.135135. Elman I, Soares NS, Pinto ME. Análise da Sensibilidade do Gosto Umami em Crianças com Câncer. Rev Bras Cancerol. 2010;56(2):237-42.
Sensory changes are frequent with HSCT and orchestrated by interconnected, internal and external factors, which hinder understanding of the mechanisms involved.
Dysosmia, or impaired odor perception, affects the consumption and preparation of foods.136136. Gamper EM, Zabernigg A, Wintner LM, Giesinger JM, Oberguggenberger A, Kemmler G, et al. Coming to your senses: detecting taste and smell alterations in chemotherapy patients. A systematic review. J Pain Symptom Manage. 2012;44(6):880-95. These conditions may develop and persist even months after conditioning, due to toxicity of the drugs used in treatment.4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48.Changes in flavor and odor perception in patients undergoing chemotherapy are present in approximately 70% of cases4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48.,136136. Gamper EM, Zabernigg A, Wintner LM, Giesinger JM, Oberguggenberger A, Kemmler G, et al. Coming to your senses: detecting taste and smell alterations in chemotherapy patients. A systematic review. J Pain Symptom Manage. 2012;44(6):880-95. and may result in significantly decreased food intake and, consequently, decreased calorie consumption. A study has shown that patients with abnormal taste perception eat approximately 430kcal/day less than patients with normal taste perception.3535. Mattsson J, Westin S, Edlund S, Remberger M. Poor oral nutrition after allogeneic stem cell transplantation correlates significantly with severe graft-versus-host disease. Bone Marrow Transplant. 2006;38(9):629-33.,136136. Gamper EM, Zabernigg A, Wintner LM, Giesinger JM, Oberguggenberger A, Kemmler G, et al. Coming to your senses: detecting taste and smell alterations in chemotherapy patients. A systematic review. J Pain Symptom Manage. 2012;44(6):880-95.
In HSCT inpatient units, patients often complain about the smell of foods and how they make them feel nauseated. Unattractive meals can worsen hyporexia. Table 7 shows examples of situations that impact food intake.
Umami taste
Yet to be better explored, the umami taste, which derives from amino acid monosodium glutamate, is an alternative to stimulate taste perception.135135. Elman I, Soares NS, Pinto ME. Análise da Sensibilidade do Gosto Umami em Crianças com Câncer. Rev Bras Cancerol. 2010;56(2):237-42. It has gained attention because it is one of the last tastes or flavors to be discovered, and is therefore known as the fifth taste, in addition to the other four: sweet, sour, salty and bitter.138138. Ackroff K, Sclafani A. Flavor Preferences Conditioned by Dietary Glutamate. Adv Nutr. 2016;7(4):845S-52S. Because it activates specific receptors for glutamate and its byproducts, taste-m-GLUR4 (a metabotropic receptor of glutamate) seems to be one of the most preserved tastes during gustatory changes inherent to prolonged exposures to chemotherapy toxicity.135135. Elman I, Soares NS, Pinto ME. Análise da Sensibilidade do Gosto Umami em Crianças com Câncer. Rev Bras Cancerol. 2010;56(2):237-42.
Umami can be composed of, in addition to glutamate, other salts such as disodium guanilate and inosinate, naturally present in foods like cheese, tomato and fish.138138. Ackroff K, Sclafani A. Flavor Preferences Conditioned by Dietary Glutamate. Adv Nutr. 2016;7(4):845S-52S. It affects the salivary flow, promoting better dilution of the chemical compounds from foods in the oral cavity. In Brazil, a study showed that it is identified, even at low thresholds, by children diagnosed with leukemia and lymphomas undergoing conditioning chemotherapy,135135. Elman I, Soares NS, Pinto ME. Análise da Sensibilidade do Gosto Umami em Crianças com Câncer. Rev Bras Cancerol. 2010;56(2):237-42. which makes it a possible alternative that should get more attention from those in the field of dietary management. The dose compliant with the NOAEL (Non Observed Adversed Effect Level) was established at 1,600mg/kg of body weight for oral administration − which is quite high, even for big consumers.139139. Jinap S, Hajeb P. Glutamate. Its applications in food and contribution to health. Appetite. 2010;55(1):1-10. The use of monosodium glutamate to season food can be an alternative to be tested in patients with impaired taste perception.
Sweet, salty, sour and bitter tastes
Altered perception of sweet, salty, bitter or sour tastes is described in a review with 446 subjects on different chemotherapy regimens.140140. Boltong A, Keast R. The influence of chemotherapy on taste perception and food hedonics: a systematic review. Cancer Treat Rev. 2012;38(2):152-63.However, the diversity of methods, outcomes and chemotherapy protocols does not allow for identification of a taste perception change profile. Studies in patients undergoing allogeneic transplants showed that they have difficulties perceiving the intensity of high and low concentrations of sweetness, and the changes in taste persist for up to 3 years after HSCT, which could be considered a permanent change and a late complication of HSCT.141141. Boer CC, Correa ME, Miranda EC, de Souza CA. Taste disorders and oral evaluation in patients undergoing allogeneic hematopoietic SCT. Bone Marrow Transplant. 2010;45(4):705-11. This has a major impact on dietary management and requires professionals to monitor these changes and find food alternatives to attenuate patients’ disappointment when they cannot recognize the taste of food. There are two possible dietary approaches: resorting to known and appreciated foods and preparations, to encourage consumption of foods that bring positive past references to the patient; and investing in new preparations and foods to avoid the disappointment that comes with an impaired taste perception, which helps improve the eating experience.
NUTRITIONAL REQUIREMENTS IN HEMATOPOIETIC STEM CELL TRANSPLANTATION
After the nutritional diagnosis, the next step is to calculate nutritional requirements.4949. Brasil. Ministério da Saúde. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Consenso Nacional de Nutrição Oncológica. 2ª ed [Internet]. Rio de Janeiro (RJ): INCA; 2015. 186 p [citado 2019 Maio 10]. Disponível em: http://www1.inca.gov.br/inca/Arquivos/Consenso_Nutricao_vol_II_2_ed_2016.pdf
http://www1.inca.gov.br/inca/Arquivos/Co...
,142142. Pinho NB, Tartari RF. Terapia Nutricional Convencional versus Terapia Nutricional precoce no perioperatório de cirurgia do cancer colorretal. Rev Bras Cancerol. 2011;57(2):237-50.
The energy requirement of patients can be calculated with predictive equations or IC. According to the ASPEN, IC is the recommended method to determine energy requirements in critically-ill cancer patients. When it is not available, predictive equations, such as those of Harris Benedict, Scholfield, and others, can be used. Another fast and widely applicable method to calculate energy requirements is a simple formula that uses calories perkg/current weight or adjusted weight.4040. August DA, Huhmann MB; American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. A.S.P.E.N. clinical guidelines: nutrition support therapy during adult anticancer treatment and in hematopoietic cell transplantation. JPEN J Parenter Enteral Nutr. 2009;33(5):472-500.,143143. Brasil. Ministério da Saúde. Instituto Nacional do Câncer (INCA). Consenso Nacional de Nutrição Oncológica [Internet]. Rio de janeiro: INCA; 2011. Vol. II. [citado 2017 Abr 7]. Disponível em: http://www1.inca.gov.br/inca/Arquivos/comunicacao/consenso_nutricao_2011.pdf
http://www1.inca.gov.br/inca/Arquivos/co...
The nutritional status of HSCT candidates is on its own considered a risk factor, since these patients are already at nutritional risk, due to drug toxicity or to their underlying disease.4949. Brasil. Ministério da Saúde. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Consenso Nacional de Nutrição Oncológica. 2ª ed [Internet]. Rio de Janeiro (RJ): INCA; 2015. 186 p [citado 2019 Maio 10]. Disponível em: http://www1.inca.gov.br/inca/Arquivos/Consenso_Nutricao_vol_II_2_ed_2016.pdf
http://www1.inca.gov.br/inca/Arquivos/Co...
,144144. Albertini SM. O papel da glutamina na terapia nutricional do transplante de medula óssea. Rev Bras Hematolia e Hematoterapia. 2001;23(1):41-7.
During HSCT, two distinct situations occur simultaneously: a decreased oral intake of foods and increased metabolic requirements, which affect the patient’s nutritional status.145145. Anders JC, Soller VM, Brandão EM, Vendramini EC, Bertagnolli CL, Giovani PG, et al. Aspectos de enfermagem, nutrição, fisioterapia e serviço social no transplante de medula óssea. Medicina (Ribeirão Preto). 2000;33(4):463-85.,146146. Brasil. Ministério da Saúde. Instituto Nacional do Câncer. Consenso nacional de nutrição oncológica [Internet]. Rio de Janeiro: INCA; 2009. Vol. 53 [citado 2017 Mar 1]. Disponível em: https://www.inca.gov.br/sites/ufu.sti.inca.local/files//media/document//consenso-nacional-de-nutricao-oncologica-2009.pdf
https://www.inca.gov.br/sites/ufu.sti.in...
The maintenance of a good nutritional status is of utmost importance throughout the entire HSCT process, and depends on an appropriate supply of nutrients.145145. Anders JC, Soller VM, Brandão EM, Vendramini EC, Bertagnolli CL, Giovani PG, et al. Aspectos de enfermagem, nutrição, fisioterapia e serviço social no transplante de medula óssea. Medicina (Ribeirão Preto). 2000;33(4):463-85.,146146. Brasil. Ministério da Saúde. Instituto Nacional do Câncer. Consenso nacional de nutrição oncológica [Internet]. Rio de Janeiro: INCA; 2009. Vol. 53 [citado 2017 Mar 1]. Disponível em: https://www.inca.gov.br/sites/ufu.sti.inca.local/files//media/document//consenso-nacional-de-nutricao-oncologica-2009.pdf
https://www.inca.gov.br/sites/ufu.sti.in...
A patient with a good nutritional status has a high prognostic factor. Thus, nutritional support is indicated before the onset of digestive complications. The changes experienced by these patients affect energy, protein and micronutrient metabolism. The conditioning regimen and transplant complications (sepsis, GVHD, mucositis, diarrhea, among others) lead to a negative nitrogen balance and loss of skeletal muscle mass.147147. Martin-Salces M, de Paz R, Canales MA, Mesejo A, Hernandez-Navarro F. Nutritional recommendations in hematopoietic stem cell transplantation. Nutrition. 2008;24(7-8):769-75.
Hematopoietic stem cell transplantation is a procedure that requires high-dose chemotherapy due to its therapeutic aggressiveness, and therefore demands special nutrition care, since patients are at a higher risk of malnutrition before and after transplant. The conditioning regimen has effects on the gastrointestinal tract and immune system, leading to metabolic and nutritional changes. Altered nutritional status before transplant is a negative prognostic factor for patient progression.147147. Martin-Salces M, de Paz R, Canales MA, Mesejo A, Hernandez-Navarro F. Nutritional recommendations in hematopoietic stem cell transplantation. Nutrition. 2008;24(7-8):769-75.
Allogeneic HSCT tends to be more aggressive when compared to autologous HSCT; however, virtually all patients had complications, irrespective of the type of transplant, and they all benefited from nutritional support.147147. Martin-Salces M, de Paz R, Canales MA, Mesejo A, Hernandez-Navarro F. Nutritional recommendations in hematopoietic stem cell transplantation. Nutrition. 2008;24(7-8):769-75.
In the late post-HSCT, patients may present with several nutritional problems, such as malnutrition, due to insufficient oral intake, or metabolic disorders, such as diabetes, obesity, dyslipidemias and hypertension.119119. Fuji S, Einsele H, Savani BN, Kapp M. Systematic Nutritional Support in Allogeneic Hematopoietic Stem Cell Transplant Recipients. Biol Blood Marrow Transplant. 2015;21(10):1707-13.
Nutritional counseling must be appropriate to cover patient’s nutritional requirements, respecting their eating habits and food preferences.4949. Brasil. Ministério da Saúde. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Consenso Nacional de Nutrição Oncológica. 2ª ed [Internet]. Rio de Janeiro (RJ): INCA; 2015. 186 p [citado 2019 Maio 10]. Disponível em: http://www1.inca.gov.br/inca/Arquivos/Consenso_Nutricao_vol_II_2_ed_2016.pdf
http://www1.inca.gov.br/inca/Arquivos/Co...
Fluid requirements for HSCT patients are based on the recommendations for healthy individuals, i.e., 1m/kcal or 35ml/kg/body weight; however, due to dynamic losses and frequent fluid retention, some adjustments may be needed in these calculations.4949. Brasil. Ministério da Saúde. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Consenso Nacional de Nutrição Oncológica. 2ª ed [Internet]. Rio de Janeiro (RJ): INCA; 2015. 186 p [citado 2019 Maio 10]. Disponível em: http://www1.inca.gov.br/inca/Arquivos/Consenso_Nutricao_vol_II_2_ed_2016.pdf
http://www1.inca.gov.br/inca/Arquivos/Co...
,148148. Franceschini SC, Priore SE, Euclydes MP. Necessidades e recomendações de nutrientes. In: Cuppari L. Guias de Medicina Ambulatorial e Hospitalar - Nutrição - Nutrição Clínica no Adulto. Barueri (SP): Manole; 2005. p. 3-32.
ENERGY REQUIREMENTS
Hematopoietic stem cell transplantation is a very stressful condition with a high energy requirement due to hypermetabolism and increased catabolism, cytoreductive therapy, infections, multiple organ failure, and tissue repair.9191. Hadjibabaie M, Iravani M, Taghizadeh M, Ataie-Jafari A, Shamshiri AR, Mousavi SA, et al. Evaluation of nutritional status in patients undergoing hematopoietic SCT. Bone Marrow Transplant. 2008;42(7):469-73.
Hematopoietic stem cell transplantation predisposes patients to digestive complications. Aggressive immunosuppression can cause nausea and vomiting, mucositis, impaired taste perception and esophagitis, as well as diarrhea, a common complication that can persist for weeks after HSCT.147147. Martin-Salces M, de Paz R, Canales MA, Mesejo A, Hernandez-Navarro F. Nutritional recommendations in hematopoietic stem cell transplantation. Nutrition. 2008;24(7-8):769-75.
These undesirable digestive manifestations experienced by these patients have negative implications on the protein-calorie supply and nutrient absorption, in addition to the increase in energy requirements brought about by the treatment and prolonged hospitalization. The consequence of these factors combined is progressive deterioration of the nutritional status.149149. Sociedade Brasileira de Nutrição Parenteral e Enteral. Associação Brasileira de Nutrologia. Projeto Diretrizes - Associação Médica Brasileira e Conselho Federal de Medicina [Internet]. São Paulo: 2011 [citado 2019 Abr 26]. Disponível em: http://www.saudedireta.com.br/docsupload/1331171426terapia_nutricional_no_transplante_de_celula_hematopoietica.pdf
http://www.saudedireta.com.br/docsupload...
According to the Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA), determination of the energy value must take into consideration the baseline nutritional status of the patient, metabolic stress, age and weight.4949. Brasil. Ministério da Saúde. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Consenso Nacional de Nutrição Oncológica. 2ª ed [Internet]. Rio de Janeiro (RJ): INCA; 2015. 186 p [citado 2019 Maio 10]. Disponível em: http://www1.inca.gov.br/inca/Arquivos/Consenso_Nutricao_vol_II_2_ed_2016.pdf
http://www1.inca.gov.br/inca/Arquivos/Co...
Although energy expenditure varies between autologous and allogeneic transplants, due to intense catabolism, it is generally agreed that energy requirements will range between 130% and 150% of the baseline energy expenditure.146146. Brasil. Ministério da Saúde. Instituto Nacional do Câncer. Consenso nacional de nutrição oncológica [Internet]. Rio de Janeiro: INCA; 2009. Vol. 53 [citado 2017 Mar 1]. Disponível em: https://www.inca.gov.br/sites/ufu.sti.inca.local/files//media/document//consenso-nacional-de-nutricao-oncologica-2009.pdf
https://www.inca.gov.br/sites/ufu.sti.in...
,147147. Martin-Salces M, de Paz R, Canales MA, Mesejo A, Hernandez-Navarro F. Nutritional recommendations in hematopoietic stem cell transplantation. Nutrition. 2008;24(7-8):769-75.,150150. Muscaritoli M, Grieco G, Capria S, Iori AP, Rossi Fanelli F. Nutritional and metabolic support in patients undergoing bone marrow transplantation. Am J Clin Nutr. 2002;75(2):183-90.
In most HSCT centers, energy recommendation protocols are similar. Szeluga et al.,151151. Szeluga DJ, Stuart RK, Brookmeyer R, Utermohlen V, Santos GW. Energy requirements of parenterally fed bone marrow transplant recipients. JPEN J Parenter Enteral Nutr. 1985;9(2):139-43. demonstrated that maintaining nitrogen balance at zero required 30 to 50kcal/kg body weight/day in adolescents and adults.146146. Brasil. Ministério da Saúde. Instituto Nacional do Câncer. Consenso nacional de nutrição oncológica [Internet]. Rio de Janeiro: INCA; 2009. Vol. 53 [citado 2017 Mar 1]. Disponível em: https://www.inca.gov.br/sites/ufu.sti.inca.local/files//media/document//consenso-nacional-de-nutricao-oncologica-2009.pdf
https://www.inca.gov.br/sites/ufu.sti.in...
,151151. Szeluga DJ, Stuart RK, Brookmeyer R, Utermohlen V, Santos GW. Energy requirements of parenterally fed bone marrow transplant recipients. JPEN J Parenter Enteral Nutr. 1985;9(2):139-43.
According to the Fred Hutchinson Cancer Research Center criteria, in the post-HSCT period (days 30 to 50), energy requirements are higher due to conditioning, fever, infections, GVHD and other metabolic complications.146146. Brasil. Ministério da Saúde. Instituto Nacional do Câncer. Consenso nacional de nutrição oncológica [Internet]. Rio de Janeiro: INCA; 2009. Vol. 53 [citado 2017 Mar 1]. Disponível em: https://www.inca.gov.br/sites/ufu.sti.inca.local/files//media/document//consenso-nacional-de-nutricao-oncologica-2009.pdf
https://www.inca.gov.br/sites/ufu.sti.in...
,152152. Lenssen P. Aker SN. A Resource Manual Fred Hutchinson Cancer Research Center. BMT/PBSCT; 1985.
Critically-ill patients are usually in a hypermetabolic state and, therefore, have higher nutritional requirements.143143. Brasil. Ministério da Saúde. Instituto Nacional do Câncer (INCA). Consenso Nacional de Nutrição Oncológica [Internet]. Rio de janeiro: INCA; 2011. Vol. II. [citado 2017 Abr 7]. Disponível em: http://www1.inca.gov.br/inca/Arquivos/comunicacao/consenso_nutricao_2011.pdf
http://www1.inca.gov.br/inca/Arquivos/co...
,153153. El-Khalili N, Brodlie K, Kessel D. WebSTer: A web-based surgical training system. Stud Health Technol Inform. 2000;70:69-75.
Patients subjected to HSCT are at risk of malnutrition due to prior chemotherapy and/or radiation therapy, which lead to undesirable digestive changes, increased energy requirements, and prolonged hospitalization.149149. Sociedade Brasileira de Nutrição Parenteral e Enteral. Associação Brasileira de Nutrologia. Projeto Diretrizes - Associação Médica Brasileira e Conselho Federal de Medicina [Internet]. São Paulo: 2011 [citado 2019 Abr 26]. Disponível em: http://www.saudedireta.com.br/docsupload/1331171426terapia_nutricional_no_transplante_de_celula_hematopoietica.pdf
http://www.saudedireta.com.br/docsupload...
Regarding carbohydrate metabolism, glucose intolerance has been reported, caused by administration of cyclosporine and steroids, or due to sepsis. Lipid metabolism abnormalities are less frequent in the first phase of the transplant, however high levels of cholesterol and triglycerides are seen in the late post-transplant period. The lipids offered in the diet must contain long- and mid-chain triglycerides.147147. Martin-Salces M, de Paz R, Canales MA, Mesejo A, Hernandez-Navarro F. Nutritional recommendations in hematopoietic stem cell transplantation. Nutrition. 2008;24(7-8):769-75.
In the post-HSCT period, hyperglycemia and abnormal lipids are the most common, and they increase the risk of comorbidities. Glucose must be administered at no more than 5g/kg of body weight/day, and lipids must be used to complete the calorie requirements (20% to 30%). That is why it is important to supply appropriate doses of lipids, to reduce the amount of glucose in patients with hyperglycemia.119119. Fuji S, Einsele H, Savani BN, Kapp M. Systematic Nutritional Support in Allogeneic Hematopoietic Stem Cell Transplant Recipients. Biol Blood Marrow Transplant. 2015;21(10):1707-13.
Glucose control is important during the post-HSCT period, since hyperglycemia raises the risk of infectious diseases caused by neutropenia, immune cell dysfunctions, cytokine increase and proliferation, muscle and lipid catabolism, in addition to an increased risk of GVHD in allogeneic HSCT, as well as morbidity and mortality.119119. Fuji S, Einsele H, Savani BN, Kapp M. Systematic Nutritional Support in Allogeneic Hematopoietic Stem Cell Transplant Recipients. Biol Blood Marrow Transplant. 2015;21(10):1707-13.
PROTEIN REQUIREMENTS
Increased metabolism results from some factors, such as fever, infections, chemotherapy or radiation therapy, leading to tissue destruction.149149. Sociedade Brasileira de Nutrição Parenteral e Enteral. Associação Brasileira de Nutrologia. Projeto Diretrizes - Associação Médica Brasileira e Conselho Federal de Medicina [Internet]. São Paulo: 2011 [citado 2019 Abr 26]. Disponível em: http://www.saudedireta.com.br/docsupload/1331171426terapia_nutricional_no_transplante_de_celula_hematopoietica.pdf
http://www.saudedireta.com.br/docsupload...
Protein requirements are estimated to provide the right substrate for tissue repair after cytoreductive therapy, and minimize the loss of lean mass.146146. Brasil. Ministério da Saúde. Instituto Nacional do Câncer. Consenso nacional de nutrição oncológica [Internet]. Rio de Janeiro: INCA; 2009. Vol. 53 [citado 2017 Mar 1]. Disponível em: https://www.inca.gov.br/sites/ufu.sti.inca.local/files//media/document//consenso-nacional-de-nutricao-oncologica-2009.pdf
https://www.inca.gov.br/sites/ufu.sti.in...
,152152. Lenssen P. Aker SN. A Resource Manual Fred Hutchinson Cancer Research Center. BMT/PBSCT; 1985.
Most critically-ill patients have proportionally higher protein requirements than energy requirements, due to the fact that protein is the most important macronutrient in tissue repair, immunological support, and maintenance of lean mass.4040. August DA, Huhmann MB; American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. A.S.P.E.N. clinical guidelines: nutrition support therapy during adult anticancer treatment and in hematopoietic cell transplantation. JPEN J Parenter Enteral Nutr. 2009;33(5):472-500.,143143. Brasil. Ministério da Saúde. Instituto Nacional do Câncer (INCA). Consenso Nacional de Nutrição Oncológica [Internet]. Rio de janeiro: INCA; 2011. Vol. II. [citado 2017 Abr 7]. Disponível em: http://www1.inca.gov.br/inca/Arquivos/comunicacao/consenso_nutricao_2011.pdf
http://www1.inca.gov.br/inca/Arquivos/co...
There is a consensus regarding protein requirements in both types of transplant (autologous and allogeneic), which ranges between 1.4 and 1.5g/kg of body weight/day, and can reach up to 2.0g/kg/day) to meet nutritional demands related with the transplant.149149. Sociedade Brasileira de Nutrição Parenteral e Enteral. Associação Brasileira de Nutrologia. Projeto Diretrizes - Associação Médica Brasileira e Conselho Federal de Medicina [Internet]. São Paulo: 2011 [citado 2019 Abr 26]. Disponível em: http://www.saudedireta.com.br/docsupload/1331171426terapia_nutricional_no_transplante_de_celula_hematopoietica.pdf
http://www.saudedireta.com.br/docsupload...
,154154. Tvedt TH, Reikvam H, Bruserud Ø. Nutrition in Allogeneic Stem Cell Transplantion--Clinical Guidelines and Immunobiological Aspects. Curr Pharm Biotechnol. 2016;17(1):92-104. Review.
Table 8 lists the nutritional recommendations for adult patients undergoing HSCT.
MICRONUTRIENTS IN HEMATOPOIETIC STEM CELL TRANSPLANTATION
There are few studies about the use of micronutrients in HSCT, and still the level of recommendation for supplementation is low.9494. van der Meij BS, de Graaf P, Wierdsma NJ, Langius JA, Janssen JJ, van Leeuwen PA, et al. Nutritional support in patients with GVHD of the digestive tract: state of the art. Bone Marrow Transplant. 2013;48(4):474-82. It would not be possible to address all the micronutrients involved in this process and, therefore, here we discuss the most frequently mentioned in the literature: zync, magnesium, and vitamins D and B12.
Zync
Zync is a microelement which is very important for the body. It is a cofactor of >300 enzymes, responsible for the synthesis of nucleic acids, a structural component of different proteins, and it prevents formation of free radicals and maintains the immune system, among other roles.156156. Prasad AS. Zinc in human health: effect of zinc on immune cells. Mol Med. 2008;14(5-6):353-7.
Deficiency or low serum levels of zync have been seen in patients with leukemia. This situation has been thoroughly studied and characterized in children around the world, and also in Brazil.159159. Sgarbieri UR, Fisberg M, Tone LG, Latorre MR. Nutritional assessment and serum zinc and copper concentration among children with acute lymphocytic leukemia: a longitudinal study. Sao Paulo Med J. 2006;124(6):316-20.,160160. Zalina AZ Jr, Suzana S, A Rahman AJ, Noor Aini MY. Assessing the nutritional status of children with leukemia from hospitals in kuala lumpur. Malays J Nutr. 2009;15(1):45-51. Zync deficiency in children with leukemia is so relevant that some studies have proposed its supplementation as an adjuvant treatment.161161. Eby GA. Treatment of acute lymphocytic leukemia using zinc adjuvant with chemotherapy and radiation – a case history and hypothesis. Med Hypotheses. 2005;64(6):1124-6.
In older patients undergoing HSCT, the decrease in serum zync, already verified in studies, seems to be related with a greater susceptibility to infections, impaired taste perception, changes in the gastrointestinal system, among other reasons.156156. Prasad AS. Zinc in human health: effect of zinc on immune cells. Mol Med. 2008;14(5-6):353-7.,162162. Haase H, Rink L. The immune system and the impact of zinc during aging. Immun Ageing. 2009;6(1):9.
Clinical manifestations of zync deficiency may be caused by genetic changes (acrodermatitis entheropatica), use of PNT without zync supplementation, alcohol abuse, drugs (penicilamin, thiazides and glucagon), intake of high-protein and phytate-rich grains, malabsorption syndrome, sickle cell anemia, blood loss and excessive sweating (in tropical countries).156156. Prasad AS. Zinc in human health: effect of zinc on immune cells. Mol Med. 2008;14(5-6):353-7.,157157. Gibson RS, Hess SY, Hotz C, Brown KH. Indicators of zinc status at the population level: a review of the evidence. Br J Nutr. 2008;99(S3 Suppl 3):S14-23. Specifically in HSCT, zync deficiency is associated with low food intake, use of total parenteral nutrition (TPN), malabsorption and increased requirements for restructuring of the bone marrow.163163. Hadjibabaie M, Iravani M, Shamshiri AR, Zaker Z, Mousavi A, Alimoghaddam K, et al. Ghavamzadeh. Serum Zinc Values in Adult Patients Undergoing Bone Marrow Transplantation. IJHOSCR. 2009;3(3):12-8.
The symptoms of zync deficiency, such as alopecia, diarrhea, skin rash, failure to thrive, pustular and bullous dermatitis, weight loss, recurring infections, delayed wound healing, decreased lean mass, hypogeusia, dysgeusia, impaired nighttime visual acuity, among others,156156. Prasad AS. Zinc in human health: effect of zinc on immune cells. Mol Med. 2008;14(5-6):353-7.,157157. Gibson RS, Hess SY, Hotz C, Brown KH. Indicators of zinc status at the population level: a review of the evidence. Br J Nutr. 2008;99(S3 Suppl 3):S14-23.,164164. Raynard B. Nutritional support of the cancer patient: issues and dilemmas. Crit Rev Oncol Hematol. 2000;34(3):137-68. Review.can be mistaken for post-HSCT symptoms.165165. Papadopoulou A, Nathavitharana K, Williams MD, Darbyshire PJ, Booth IW. Diagnosis and clinical associations of zinc depletion following bone marrow transplantation. Arch Dis Child. 1996;74(4):328-31.
In HSCT, zync deficiency has been correlated with late mortality. During the new hematopoiesis and under inflammatory conditions, zync requirements are even higher. Therefore, keeping serum levels within normal ranges during the HSCT process seems to have an effect on the disease prognosis.163163. Hadjibabaie M, Iravani M, Shamshiri AR, Zaker Z, Mousavi A, Alimoghaddam K, et al. Ghavamzadeh. Serum Zinc Values in Adult Patients Undergoing Bone Marrow Transplantation. IJHOSCR. 2009;3(3):12-8.
During HSCT, high serum iron can be a negative prognostic factor, due to its build-up in the liver. To correct abnormal levels, iron chelating agents are used, which are not free from side effects. Zync is a potent iron chelator, and its supplementation and the consequent maintenance of higher serum levels, associated with chelating agents given to these patients, seem to improve the results, reducing side effects and, furthermore, reducing liver iron overload.166166. Beebe K, Olsen J, Sproat L. Impact Of Vitamin D Level Pre and Post Allogeneic Hematopoietic Stem Cell Transplant. Blood. 2013;122(21):4616.,167167. Bandeira F, Griz L, Dreyer P, Eufrazino C, Bandeira C, Freese E. Vitamin D deficiency: A global perspective. Arq Bras Endocrinol Metabol. 2006;50(4):640-6.
Also, HSCT-associated complications are more frequent in patients with lower serum zync, such as acute gastrointestinal GVHD, fever of longer duration, and higher susceptibility to infections. Therefore, it is suggested that low serum zync can be a risk factor for adverse events during and after HSCT.163163. Hadjibabaie M, Iravani M, Shamshiri AR, Zaker Z, Mousavi A, Alimoghaddam K, et al. Ghavamzadeh. Serum Zinc Values in Adult Patients Undergoing Bone Marrow Transplantation. IJHOSCR. 2009;3(3):12-8.,165165. Papadopoulou A, Nathavitharana K, Williams MD, Darbyshire PJ, Booth IW. Diagnosis and clinical associations of zinc depletion following bone marrow transplantation. Arch Dis Child. 1996;74(4):328-31.
Although this deficiency is underestimated in clinical practice, some studies have shown a 67% deficiency rate in children, particularly young children, after HSCT. In adults, in turn, this deficiency seems to be more prevalent in the pre-HSCT period.165165. Papadopoulou A, Nathavitharana K, Williams MD, Darbyshire PJ, Booth IW. Diagnosis and clinical associations of zinc depletion following bone marrow transplantation. Arch Dis Child. 1996;74(4):328-31.
In the post-HSCT period, zync supplementation has been suggested as a potential therapy, aiding immunosuppresants, but with no side effects.168168. Prietl B, Treiber G, Pieber TR, Amrein K. Vitamin D and immune function. Nutrients. 2013;5(7):2502-21. However, it is not a routine in HSCT centers.
Magnesium
The use of immunosuppressants may cause hypomagnesemia in patients undergoing HSCT.9494. van der Meij BS, de Graaf P, Wierdsma NJ, Langius JA, Janssen JJ, van Leeuwen PA, et al. Nutritional support in patients with GVHD of the digestive tract: state of the art. Bone Marrow Transplant. 2013;48(4):474-82.Nonetheless, there are reports of hypermagnesemia in these patients, which can be associated with the use of laxatives, kidney dysfunction or abnormal intestinal motility.169169. Leong DP, Kleinig TJ, Kimber TE, Bardy PG. Severe hypermagnesaemia related to laxative use in acute gastrointestinal graft-versus-host disease. Bone Marrow Transplant. 2006;38(1):71-2.,170170. Jaing TH, Hung IJ, Chung HT, Lai CH, Liu WM, Chang KW. Acute hypermagnesemia: a rare complication of antacid administration after bone marrow transplantation. Clin Chim Acta. 2002;326(1-2):201-3.
Magnesium supplementation must be given only when a laboratory diagnosis is established, since this micronutrient could be increased or decreased.
Vitamin D
Vitamin D can reduce the incidence of chronic GVHD and mortality in HSCT. Morbidity and mortality of HSCT are attributed to infection, organ system toxicity, GVHD and recurring diseases.166166. Beebe K, Olsen J, Sproat L. Impact Of Vitamin D Level Pre and Post Allogeneic Hematopoietic Stem Cell Transplant. Blood. 2013;122(21):4616.
There are two ways in which the human body produces vitamin D: by endogenous skin synthesis and food sources. Endogenous production depends on ultraviolet B (UVB) rays.167167. Bandeira F, Griz L, Dreyer P, Eufrazino C, Bandeira C, Freese E. Vitamin D deficiency: A global perspective. Arq Bras Endocrinol Metabol. 2006;50(4):640-6.,168168. Prietl B, Treiber G, Pieber TR, Amrein K. Vitamin D and immune function. Nutrients. 2013;5(7):2502-21. In diets, vitamin D is found in non-fortified natural products, such as fatty fish (salmon, sardines, cold liver and oil), or some types of mushrooms (shitake), which have relevant amounts of the two main forms, cholecalciferol and ergocalciferol.168168. Prietl B, Treiber G, Pieber TR, Amrein K. Vitamin D and immune function. Nutrients. 2013;5(7):2502-21.
In human skin, cholecalciferol is synthesized from 7-dihydrocholesterol when exposed to sunlight (UVB 290-315). Cholecalciferol is biologically inactive and immediately binds to vitamin-D-binding proteins or albumin. Thus, vitamin D3 is metabolized in the liver, into 25-hydroxyvitamin D3, catalyzed by CYP2R1 and CYP27A1 enzymes. In the kidney, 25-hydroxyvitamin D3 is metabolized into 1,25-dihydroxyvitamin D3 by enzyme 1-a-hydrolase (CYP27B1), which is strictly controlled by parathyroid hormone and fibroblast growth factor 23 (FGF-23).167167. Bandeira F, Griz L, Dreyer P, Eufrazino C, Bandeira C, Freese E. Vitamin D deficiency: A global perspective. Arq Bras Endocrinol Metabol. 2006;50(4):640-6.,168168. Prietl B, Treiber G, Pieber TR, Amrein K. Vitamin D and immune function. Nutrients. 2013;5(7):2502-21.
Vitamin D deficiency can lead to osteomalacia, bone pain, muscle weakness, fatigue, increased risk of fracture, and precipitate or exacerbate osteopenia and osteoporosis. It can be caused by reduced skin synthesis, reduced gastrointestinal absorption, and hereditary or acquired metabolic diseases. Also, liver failure and chronic kidney disease can lead to decreased vitamin D synthesis; the use of anticonvulsants, glucocorticoids and antirejection drugs can increase vitamin D catabolism.171171. Sproat L, Bolwell B, Rybicki L, Dean R, Sobecks R, Pohlman B, et al. Vitamin D level after allogeneic hematopoietic stem cell transplant. Biol Blood Marrow Transplant. 2011;17(7):1079-83.
The main cause of vitamin D deficiency is inadequate exposure to sunlight. People with darker skin are naturally protected from sun rays and need a 3-to-5-fold longer exposure time to produce the same amount of vitamin D than people with light skin. Also, obesity, malabsorption syndromes, bariatric and other gastrointestinal surgeries, medications, chronic granulomatous diseases, lymphomas and primary hyperparathyroidism are related with a higher risk for this deficiency.172172. Holick MF, Chen TC. Vitamin D deficiency: a worldwide problem with health consequences. Am J Clin Nutr. 2008;87(4):1080S-6S.
Vitamin D has different roles in the body. One of them is the important part it plays in mineral metabolism and bone health. Vitamin D increases intestinal calcium and phosphate absorption, estimulates osteoclast differentiation, as well as calcium resorption, and promotes bone matrix mineralization.
Vitamin D may also relate with the onset of certain diseases, such as cardiovascular diseases, cancer, autoimmune diseases, type 1 diabetes mellitus, multiple sclerosis and inflammatory bowel disease.168168. Prietl B, Treiber G, Pieber TR, Amrein K. Vitamin D and immune function. Nutrients. 2013;5(7):2502-21.,173173. Glotzbecker B, Ho VT, Aldridge J, Kim HT, Horowitz G, Ritz J, et al. Low levels of 25-hydroxyvitamin D before allogeneic hematopoietic SCT correlate with the development of chronic GVHD. Bone Marrow Transplant. 2013; 48(4):593-7.
174. Hall AC, Juckett MB. The role of vitamin D in hematologic disease and stem cell transplantation. Nutrients. 2013;5(6):2206-21.
175. Bunce CM, Brown G, Hewison M. Vitamin D and hematopoiesis. Trends Endocrinol Metab. 1997;8(6):245-51.-176176. Mielcarek M, Martin PJ, Leisenring W, Flowers ME, Maloney DG, Sandmaier BM, et al. Graft-versus-host disease after nonmyeloablative versus conventional hematopoietic stem cell transplantation. Blood. 2003; 102(2):756-62.
Vitamin D also regulates host immune response and prevents autoimmunity. Vitamin D inhibits maturation of dendritic cells, polarizes T-cell populations to express Th2 rather than Th1 cytokines, and blocks proliferation of allogeneic T-cells. The vitamin D receptor is expressed in multiple hematopoietic precursors, as well as monocytes, some thymocytes and active B and T lymphocytes. Its action is boosted by the binding between retinoid acid/retinoid X receptor.168168. Prietl B, Treiber G, Pieber TR, Amrein K. Vitamin D and immune function. Nutrients. 2013;5(7):2502-21.,173173. Glotzbecker B, Ho VT, Aldridge J, Kim HT, Horowitz G, Ritz J, et al. Low levels of 25-hydroxyvitamin D before allogeneic hematopoietic SCT correlate with the development of chronic GVHD. Bone Marrow Transplant. 2013; 48(4):593-7.
174. Hall AC, Juckett MB. The role of vitamin D in hematologic disease and stem cell transplantation. Nutrients. 2013;5(6):2206-21.
175. Bunce CM, Brown G, Hewison M. Vitamin D and hematopoiesis. Trends Endocrinol Metab. 1997;8(6):245-51.-176176. Mielcarek M, Martin PJ, Leisenring W, Flowers ME, Maloney DG, Sandmaier BM, et al. Graft-versus-host disease after nonmyeloablative versus conventional hematopoietic stem cell transplantation. Blood. 2003; 102(2):756-62.
In hematological disease, vitamin D also has an important role in anticancer therapy. Vitamina D analogs can take part in the maturation and differentiation of different cell lines into mature blood cells, particularly in MDS and AML. Another important role of vitamin D may be associated with modulated immune response in HSCT, where vitamin D receptors may impact immune reconstitution after HSCT and reduce the risk of infection, GVHD, and side effects.168168. Prietl B, Treiber G, Pieber TR, Amrein K. Vitamin D and immune function. Nutrients. 2013;5(7):2502-21.,173173. Glotzbecker B, Ho VT, Aldridge J, Kim HT, Horowitz G, Ritz J, et al. Low levels of 25-hydroxyvitamin D before allogeneic hematopoietic SCT correlate with the development of chronic GVHD. Bone Marrow Transplant. 2013; 48(4):593-7.
174. Hall AC, Juckett MB. The role of vitamin D in hematologic disease and stem cell transplantation. Nutrients. 2013;5(6):2206-21.
175. Bunce CM, Brown G, Hewison M. Vitamin D and hematopoiesis. Trends Endocrinol Metab. 1997;8(6):245-51.-176176. Mielcarek M, Martin PJ, Leisenring W, Flowers ME, Maloney DG, Sandmaier BM, et al. Graft-versus-host disease after nonmyeloablative versus conventional hematopoietic stem cell transplantation. Blood. 2003; 102(2):756-62.
Patients undergoing HSCT may have low levels of 25-hydroxyvitamin D due to less exposure to sunlight, the main cause of vitamin D deficiency.172172. Holick MF, Chen TC. Vitamin D deficiency: a worldwide problem with health consequences. Am J Clin Nutr. 2008;87(4):1080S-6S. This deficiency results from long hospital stays, low dietary vitamin D, use of steroids, limited outdoor activities, use of sunscreen and decreased oral intake due to gastrointestinal toxicity of the treatment.171171. Sproat L, Bolwell B, Rybicki L, Dean R, Sobecks R, Pohlman B, et al. Vitamin D level after allogeneic hematopoietic stem cell transplant. Biol Blood Marrow Transplant. 2011;17(7):1079-83.,177177. Philibert D, Desmeules S, Filion A, Poirier M, Agharazii M. Incidence and severity of early electrolyte abnormalities following autologous haematopoietic stem cell transplantation. Nephrol Dial Transplant. 2008;23(1):359-63.
Also, gastrointestinal GVHD limits vitamin D absorption, and some drugs administered during GVHD can increase this vitamin’s catabolism, affecting liver and kidney function.
Vitamin B12
Vitamin B12 is a cofactor of only two enzymes: methionine synthase and L-methylmalonyl-coenzyme A mutase. It is required for the development and initiation of central nervous system myelination.178178. Stabler SP. Clinical practice. Vitamin B12 deficiency. N Engl J Med. 2013; 368(2):149-60.
Its deficiency can cause megaloblastic anemia, neurological dysfunction, glossitis, malabsorption, infertility and thrombosis.178178. Stabler SP. Clinical practice. Vitamin B12 deficiency. N Engl J Med. 2013; 368(2):149-60.
In patients undergoing HSCT, pancreatic changes may occur due to atrophy or insufficiency, which may lead to greater loss of fat in stools, in addition to bacterial hyperproliferation.179179. Maringhini A, Gertz MA, DiMagno EP. Exocrine pancreatic insufficiency after allogeneic bone marrow transplantation. Int J Pancreatol. 1995;17(3):243-7. Also, the presence of intestinal GVHD may hinder absorption of vitamin B12.180180. Milligan DW, Quick A, Barnard DL. Vitamin B12 absorption after allogeneic bone marrow transplantation. J Clin Pathol. 1987;40(12):1472-4.
In case of vitamin B12 deficiency due to reduced absorption, oral supplementation may not be sufficient, and parenteral supplementation must be initiated.
Nutritional complications
Cancer patients are frequently submitted to antineoplastic therapy. During conditioning for HSCT, these drugs are used at high doses. Some of the side effects of the conditioning regimen are nutritional complications due to toxicity of chemotherapeutic agents. They include: mucositis, xerostomia, dysgeusia, dysphagia, odynophagia, nausea, vomiting, constipation, diarrhea, anorexia, anemia, leading to deterioration of the patient’s nutritional status and impacting on treatment response. Oral epithelial cells are the first affected by antineoplastic agents due to their high proliferation rate. The complexity of the oral cavity in these patients will depend on the drugs used, dose, general health status, patient tolerability. Oral complications directly impact on nutritional status, because they contribute to a decreased food intake. Patients receiving individualized nutritional counseling in the pre-transplant stage had a lower rate of side effects and better tolerance to treatment.181181. Paiva MD, Biase RC, Moraes JJ, Angelo AR, Honorato MC. Complicações orais decorrentes da terapia antineoplásica. Arq Odontol. 2010;46(1):48-55.
Most common chemotherapeutic agents
Patients undergoing HSCT are potential candidates for malnutrition. One of the reasons is related with toxicity of chemotherapeutic agents used during conditioning, until bone marrow engraftment or recovery, leading to decreased oral intake.182182. Ikemori EH, Oliveira T, Serralheiro IF, Shibuya E. Nutrição em Oncologia. São Paulo: Lemar; 2003.
Table 9 presents the main nutrition-related toxic effects of chemotherapeutic agents used in HSCT.
Nutrition-related toxic effects of the chemotherapeutic agents most commonly used in hematopoietic stem cell transplantation
Xerostomia
Xerostomia is defined as a subjective complaint of dry mouth, which can result from decreased saliva production. The main components of saliva are water, protein and electrolytes. These components improve taste, speech and swallowing, facilitating lubrication, irrigation and protection of mucous membranes of the upper digestive tract. The physiological functions of saliva include antimicrobial and buffer properties that protect the teeth. After the onset of symptoms, patients experience discomfort, such as pain, burning sensation and difficulty swallowing.182182. Ikemori EH, Oliveira T, Serralheiro IF, Shibuya E. Nutrição em Oncologia. São Paulo: Lemar; 2003.
Nutritional recommendations to relieve xerostomia are frequent intake of liquids in small quantities; encouraging consumption of mints and lemon drops (assess condition of the oral cavity; acid foods promote salivation); and offering moist diets (preparations with sauces, soups and broths).
Odynophagia
Odynophagia is caused by esophageal ulcers or erosions, resulting from inflammation of infectious (herpes, candidiasis and HIV) or chemical (drugs, acids and caustics) origin, causing pain when swallowing foods. Strong pain and/or burning sensation in the chest are typical symptoms of odynophagia.181181. Paiva MD, Biase RC, Moraes JJ, Angelo AR, Honorato MC. Complicações orais decorrentes da terapia antineoplásica. Arq Odontol. 2010;46(1):48-55.,183183. Garófolo A. Nutrição Clinica, Funcional e Preventiva Aplicada à Oncologia. Rio de Janeiro: Editora Rubio; 2012. p. 97-153.
Nutritional recommendations to alleviate symptom comprise to provide pain relief before meals; avoid consumption of foods that irritate the oral mucosa (carbonated drinks, acid foods, and dry/hard foods); prefer foods at room temperature, and avoid extreme heat or cold; modify diet consistency according to the level of impairment of the oral mucosa (bland, soft, liquefied and liquid diets); association of oral NT, EN, PNT, according to the patient’s severity and clinical condition.
Dysgeusia
It is characterized by loss or impairment of the sense of taste, where taste bud function is abnormal, and bitter taste is the first to be affected, followed by sweetness and saltiness.184184. Guimaraes J. Manual de Oncologia. 3a edição. v.2. São Paulo: BBS; 2008.
Nutritional recommendations in this situation include enhancing flavors with the use of condiments and spices; consume citric foods; serve food warm and cold, rather than very hot; assess the need to reduce offer of red meat, as well as iron-rich supplements, since these can worsen the sense of a metallic taste in foods, due to the use of platinum-based chemotherapeutic agents (cisplatin, carboplatin and oxaliplatin).
Dysphagia
Dysphagia is a change in the swallowing mechanism due to multiple causes, such as neurological and/or structural causes, head and neck trauma, stroke, degenerative neuromuscular diseases, head and neck cancer, toxicity of certain antineoplastic drugs, dementias and encephalopathies. Dysphagia most frequently presents as problems in the oral cavity, pharynx, esophagus or esophagogastric transition. Dysphagia or difficulty swallowing can result in food entering the airways, causing cough, choking/asphyxia, lung problems and aspiration. It may also lead to nutritional deficits and dehydration.182182. Ikemori EH, Oliveira T, Serralheiro IF, Shibuya E. Nutrição em Oncologia. São Paulo: Lemar; 2003.,185185. Padovani AR, Moraes DP, Mangili LD, De Andrade CR. Protocolo fonoaudiológico de avaliação do risco para disfagia (PARD). Rev Soc Bras Fonoaudiol. 2007;12(3):199-205.,186186. Waitzberg D, Cardenas T. Manual de Terapia Nutricional em Oncologia do ICESP. São Paulo: Atheneu; 2012. p. 253-261.
Nutritional recommendations in cases of dysphagia are referring patients for evaluation by a speech and language therapist (SLT), adapting diet consistency and using thickeners for liquids, at the SLT’s discretion.
Diarrhea
Diarrhea occurs when there is a change in the number of evacuation episodes and consistency of stools, which become liquid. Diarrhea leads to abnormal transport of water and electrolytes, which may cause dehydration. Most often, patients undergoing HSCT have diarrhea due to chemotherapy or radiation therapy toxicity of the intestinal mucosa.183183. Garófolo A. Nutrição Clinica, Funcional e Preventiva Aplicada à Oncologia. Rio de Janeiro: Editora Rubio; 2012. p. 97-153.
Diarrhea is a common complication in patients on high-dose chemotherapy, and even more common in those undergoing HSCT. Clostridium difficile has been frequently identified as the cause of infectious diarrhea at hospitals. Prolonged use of antibiotics may potentiate the risk of diarrhea in patients undergoing HSCT.187187. Spadão F, Gerhardt J, Guimarães T, Dulley F, Almeida Junior JN, Batista MV, et al. Incidence of diarrhea by Clostridium difficile in hematologic patients and hematopoietic stem cell transplantation patients: risk factors for severe forms and death. Rev Inst Med Trop São Paulo. 2014;56(4):325-31.
The presence of diarrhea can be frequent in any phase of HSCT, warranting investigation, since it could have different causes, ranging from shedding of cells due to mucositis to acute GVHD, and infections caused by bacterial or viral enteropathogens.
Nutritional recommendations in cases of diarrhea comprise controlling the intake of lactose, sucrose, as well as high-fat and naturally laxative foods; limit intake of fiber-rich or laxative foods; increase consumption of liquids for rehydration; and use drinks that promote electrolyte replacement, such as coconut water or isotonics. Also, due to the marked, prolonged immunosuppression expected in these cases, it is advisable that all hygiene-related recommendations be observed during handling and preparation of food intended for these patients, minimizing as much as possible the risk of infectious complications in the gastrointestinal tract.
Constipation
Constipation does not cause major nutritional losses like diarrhea, but it is a symptom that causes considerable discomfort to patients.183183. Garófolo A. Nutrição Clinica, Funcional e Preventiva Aplicada à Oncologia. Rio de Janeiro: Editora Rubio; 2012. p. 97-153.
Opioids are opium-derived substances indicated to treat pain and dyspnea, a common picture in patients undergoing HSCT. They are classified according to their chemical and pharmacological nature, as well as receptor-affinity. Opioid receptors are found throughout the gastrointestinal system, with higher concentrations in the gastric antrum and proximal duodenum. Their effects are linked to central and peripheral nervous system receptors. In the genesis of constipation, the action of opioids is related to different mechanisms: increased fluid absorption, inhibition of gastric, biliary and pancreatic secretions; participation in gastrointestinal neural activity, decreased propulsion; and increased segmental contractility. These mechanisms result in reduced intestinal mobility, prolonging contact between the intestinal content and the mucosa, favoring greater water and electrolyte absorption and forming, therefore, dry stools.188188. Henrique C, Silva D, Pereira K. Dietoterapia como estratégia de tratamento da constipação intestinal em (de la constipación intestinal en) cuidados paliativos. Saludi Ciencia. 2015;21(5):505-10.
As for treating opioid-induced constipation, there is still no clinical evidence on the efficacy of exclusively nutritional interventions, including fiber-rich diets and increased fluid intake (except in cases of dehydration) in regulating intestinal function. Thus, patients and caregivers must be informed, through nutritional education, on the adverse effects of these drugs, as well as on the prophylactic purpose of a laxative diet and adequate fluid intake.188188. Henrique C, Silva D, Pereira K. Dietoterapia como estratégia de tratamento da constipação intestinal em (de la constipación intestinal en) cuidados paliativos. Saludi Ciencia. 2015;21(5):505-10.
Nutritional recommendations in cases of constipation are to increase fluid intake; include soluble and insoluble fiber modules; include whole-grain foods such as grains, brown rice, whole-grain pasta, wheat bran, oat bran, flaxseed meal, and quinoa flakes; consume legumes (beans, lentils, peas, chickpeas and soybeans); fresh fruit is preferred (ensure proper washing); increase intake of leafy vegetables; and drink juices with laxative properties (papaya, orange and prunes).
Nausea and vomiting
These are common effects, present in different clinical conditions, particularly in cases of drug toxicity, which are frequent with antineoplastic drugs. These disorders may lead to nutritional deficits due to poor food acceptance, as well as dehydration, electrolytic changes, and prolonged hospitalization, which poses imminent risks to patients on prolonged neutropenia. The presence of these symptoms has a negative and quite significant impact on the patient’s nutritional status. Their occurrence and severity vary according to the drug, dose and combinations used.189189. Andrade V, Sawada NO, Barichello E. Qualidade de vida de pacientes com câncer hematológico em tratamento quimioterápico. Rev Esc Enferm USP. 2013;47(2):355-61.,190190. Gozzo TO, Moysés AM, da Silva PR, de Almeida AM. Náuseas, vômitos e qualidade de vida de mulheres com câncer de mama em tratamento quimioterápico. Rev Gaúcha Enferm. 2013;34(3):110-6.
The nutritional recommendations for relief of nausea and vomiting are to adjust diet according to patient tolerance (bland preparations); replace foods that cause discomfort; avoid contact of the patient with the food during preparation, because the smell can exacerbate symptoms; during meals, patients must be in a semirecumbent position (sitting or semi-sitting, with elevation of the head of bed), and chew thoroughly; avoid very sweet and/or fatty foods; prefer cold foods; avoid hot liquids when with nausea; hydrate by increasing fluid intake; maintain adequate oral hygiene; meals should be preferably eaten in a place with good air circulation, avoiding hot, poorly ventilated areas.
Ginger (Zingiber officinale)
Ginger is considered a medicinal plant/spice that, among other therapeutic benefits, has a major role in preventing nausea (antiemetic properties) in different situations, such as in patients undergoing chemotherapy.
The direct relation between consumption of ginger extract and control/reduction of side effects in patients undergoing cancer treatment is under investigation. Studies have shown that 1.5g of ginger powder, divided into three daily doses, is effective in women undergoing chemotherapy for advanced breast cancer.191191. Panahi Y, Saadat A, Sahebkar A, Hashemian F, Taghikhani M, Abolhasani E. Effect of ginger on acute and delayed chemotherapy-induced nausea and vomiting: a pilot, randomized, open-label clinical trial. Integr Cancer Ther. 2012;11(3):204-11.
Studies and reviews have found beneficial results when ginger is used in cancer patients undergoing antineoplastic therapy, reducing side effects, such as nausea and vomiting. However, further studies are warranted in humans to elucidate the real efficacy of this medicinal plant. Thus, it is still not possible to adopt the use of ginger in clinical practice.192192. Brasil. Ministério da Saúde. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Consenso Nacional de Nutrição Oncológica. 2a edição. Vol. II [Internet]. Rio de Janeiro: INCA; 2016. p.87-92 [citado 2019 Maio 10]. Disponível em: https://www.inca.gov.br/sites/ufu.sti.inca.local/files//media/document//consenso-nutricao-oncologica-vol-ii-2-ed-2016.pdf
https://www.inca.gov.br/sites/ufu.sti.in...
Mucositis
Mucositis is a very common adverse event in patients undergoing high-dose chemotherapy and HSCT.
The lesions are very painful, with difficult clinical management, and their occurrence can limit the duration and/or power of the treatment proposed, and also greatly impact on the quality of life of patients, increasing treatment-related costs.193193. McCann S, Schwenkglenks M, Bacon P, Einsele H, D’Addio A, Maertens J, Niederwieser D, Rabitsch W, Roosaar A, Ruutu T, Schouten H, Stone R, Vorkurka S, Quinn B, Blijlevens N; EBMT Mucositis Advisory Group. The Prospective Oral Mucositis Audit: relationship of severe oral mucositis with clinical and medical resource use outcomes in patients receiving high-dose melphalan or BEAM-conditioning chemotherapy and autologous SCT. Bone Marrow Transplant. 2009;43(2):141-7.
The injury of mucous barrier, or mucositis, is the tissue damage caused by cancer therapy, particularly high-dose chemotherapy and radiation therapy used in HSCT. From the clinical standpoint, two clinical syndromes associated with mucous barrier injury are recognized: oral mucositis, defined by the presence of erythematous, ulcerative lesions in the oral cavity; and gastrointestinal mucositis, which affects the entire digestive tract and can manifest as anorexia, nausea, vomiting and diarrhea.194194. Niscola P. Mucositis in malignant hematology. Expert Rev Hematol. 2010;3(1):57-65.
In 2003, a study with 599 patients demonstrated that 303 (51%) of those who received chemotherapy for solid tumors or lymphoma developed oral and/or gastrointestinal mucositis. Oral mucositis was found in 22% of 1,236 chemotherapy cycles, while the gastrointestinal form, in 7% of cycles. The two forms concomitantly presented in 8% of cycles. An even greater percentage (approximately 75% to 80%) of patients receiving high-dose chemotherapy before HSCT developed severe mucositis. This was considered the most disabling characteristic of the transplant.195195. Elting LS, Cooksley C, Chambers M, Cantor SB, Manzullo E, Rubenstein EB. The burdens of cancer therapy. Clinical and economic outcomes of chemotherapy-induced mucositis. Cancer. 2003;98(7):1531-9.
Several American reviews have demonstrated severe mucositis increases the use of hospital resources, such as opioids, antimicrobials and PNT, prolonging hospitalization.194194. Niscola P. Mucositis in malignant hematology. Expert Rev Hematol. 2010;3(1):57-65.,196196. Lalla RV, Sonis ST, Peterson DE. Management of oral mucositis in patients who have cancer. Dent Clin North Am. 2008;52(1):61-77.
The European Group for Blood and Marrow Transplantation (EBMT) carried out a study with 25 centers, in 13 European countries, with the primary purpose of verifying the presence of mucositis in patients undergoing autologous HSCT, who received conditioning regimens with high-dose melphalan or BEAM (carmustine, etoposide, cytarabine and melphalan). The study enrolled 197 patients and concluded that 44% of patients evolved with severe oral mucositis, with a median time to onset of ulcerative oral mucositis on D+11. The highest level of discomfort was reached on D+12, which coincided with the onset of neutrophil engraftment. The duration of the use of opioids was 6.5 days in patients with grade 4 oral mucositis, according to the World Health Organization classification. The use of PNT ranged between 19% in patients without oral mucositis to 59% in patients with grade 4 oral mucositis, and 35% for all patients.193193. McCann S, Schwenkglenks M, Bacon P, Einsele H, D’Addio A, Maertens J, Niederwieser D, Rabitsch W, Roosaar A, Ruutu T, Schouten H, Stone R, Vorkurka S, Quinn B, Blijlevens N; EBMT Mucositis Advisory Group. The Prospective Oral Mucositis Audit: relationship of severe oral mucositis with clinical and medical resource use outcomes in patients receiving high-dose melphalan or BEAM-conditioning chemotherapy and autologous SCT. Bone Marrow Transplant. 2009;43(2):141-7.
Recent studies have shown that the fundamental mechanism involving mucositis pathogenesis is more complex than just the direct injury to the epithelium. The mechanisms through which chemotherapy and radiation therapy can induce oral mucositis seem to be similar.
The following model, with five stages of pathogenesis, is based on current evidence:
-
Initiation: radiation therapy and/or cheotherapy induce cell damage, which results in death of epithelial cells. It is believed that the production and release of free radicals also play a role in initial injury in mucositis.
-
Signalling: additionally to direct tissue damage, free radicals activate secondary messengers, which transmit signals from cell surface receptors to the inside of cells, leading to release of inflammatory cytokines, and causing tissue damage and cell death.
-
Amplification: the release of tumor necrosis factor alpha (TNF-α) produced mainly by macrophages, causes damage to mucosa cells, and also activates molecular pathways that amplify this mucosal injury.
-
Ulceration: there is significant infiltrate of inflammatory cells associated with ulceration of the mucosa, based, in part, on metabolic byproducts of the microbiome colonizing the oropharynx. This secondary infection is also responsible for the production of inflammatory cytokines.
-
Healing: this phase is characterized by epithelial proliferation and cell and tissue differentiation, restoring the integrity of the epithelium.194194. Niscola P. Mucositis in malignant hematology. Expert Rev Hematol. 2010;3(1):57-65.
Clinical manifestations of mucositis include signs and symptoms of inflammation, ranging from discreet erythema, edema and discomfort, to disabling pain and ulcerations. These changes interfere with the patient’s activities of daily living, such as speaking and eating. They can hinder proper nutrition or, in some cases, eating can become impossible.195195. Elting LS, Cooksley C, Chambers M, Cantor SB, Manzullo E, Rubenstein EB. The burdens of cancer therapy. Clinical and economic outcomes of chemotherapy-induced mucositis. Cancer. 2003;98(7):1531-9. Dehydration, malnutrition and infections and, consequently, decreased overall survival, have been repeatedly associated with mucositis.197197. Fanning SR, Rybicki L, Kalaycio M, Andresen S, Kuczkowski E, Pohlman B, et al. Severe mucositis is associated with reduced survival after autologous stem cell transplantation for lymphoid malignancies. Br J Haematol. 2006;135(3):374-81.
There are three main classifications of mucositis severity, presented in table 10. Yet, the most commonly used oral mucositis classification was proposed by the WHO.
There is no consensus regarding prevention or even management of oral mucositis. Different measures have been used to prevent severe mucositis. Good oral hygiene, regular dental checks – including prevention and treatment of cavities and gingivitis – and comprehensive guidance to patients are important measures.
Cryotherapy seems to have an important role in preventing mucositis from high-dose melphalan.198198. Niscola P, Romani C, Cupelli L, Scaramucci L, Tendas A, Dentamaro T, et al. Mucositis in patients with hematologic malignancies: an overview. Haematologica. 2007;92(2):222-31. Review. Its purpose is to promote vasoconstriction in the oral cavity and, thus, reduce toxicity from the drug administered. Cryotherapy has been described in the literature due to its low cost, absence of side effects, and influence on the outcomes of the complication at issue.199199. Chen J, Seabrook J, Fulford A, Rajakumar I. Icing oral mucositis: oral cryotherapy in multiple myeloma patients undergoing autologous hematopoietic stem cell transplant. J Oncol Pharm Pract. 2015;23(2):116-20. It is recommended to offer ice 30 minutes before initiating the drug infusion, until administration is completed. Caution is recommended in respect to the quality of water for production of the ice cubes.199199. Chen J, Seabrook J, Fulford A, Rajakumar I. Icing oral mucositis: oral cryotherapy in multiple myeloma patients undergoing autologous hematopoietic stem cell transplant. J Oncol Pharm Pract. 2015;23(2):116-20. Since patients will receive the drug over the course of many hours, and have to simultaneously keep the ice in their mouth, ice prepared with nutritional supplements, fruit juice and infusions has been an alternative for cryotherapy, and could increase the energy and nutrients offered to patients.200200. Bressan V, Stevanin S, Bianchi M, Aleo G, Bagnasco A, Sasso L. The effects of swallowing disorders, dysgeusia, oral mucositis and xerostomia on nutritional status, oral intake and weight loss in head and neck cancer patients: A systematic review. Cancer Treat Rev. 2016;45:105-19. Review.
Antibacterial prophylaxis, although adopted in recipients of different types of transplant, has not been not proven effective in reducing microorganisms present on the surface of the oral mucosa, which have a major role in the onset of mucositis.201201. Lövenich H, Schütt-Gerowitt H, Keulertz C, Waldschmidt D, Bethe U, Söhngen D, et al. Failure of anti-infective mouth rinses and concomitant antibiotic prophylaxis to decrease oral mucosal colonization in autologous stem cell transplantation. Bone Marrow Transplant. 2005;35(10):997-1001.
Likewise, the use of chlorhexidine and stimulators of salivary secretion, such as pilocarpine, has not been proven effective in preventing severe mucositis.
TREATMENTS
Palliative treatment
Systemic analgesia with morphine in infusion pump is the treatment of choice for oral mucositis-related pain, in both adults and children. The use of transdermal fentanyl for this purpose has also been published, but its efficacy is yet to be confirmed.202202. Epstein JB, Schubert MM. Managing pain in mucositis. Semin Oncol Nurs. 2004;20(1):30-7.
Low-intensity laser therapy
The first studies of laser therapy for oral mucositis have shown reduction of inflammation and pain, when compared to groups not subjected to this therapy.203203. Cowen D, Tardieu C, Schubert M, Peterson D, Resbeut M, Faucher C, Franquin JC. Low energy Helium-Neon laser in the prevention of oral mucositis in patients undergoing bone marrow transplant: results of a double blind randomized trial. Int J Radiat Oncol Biol Phys. 1997;38(4):697-703.
A Brazilian, retrospective study with 43 patients undergoing allogeneic transplants between 2004 and 2008, comparing two groups – one receiving dental care and low-intensity laser therapy, and a second group with no interventions – found that these procedures can reduce the extent and severity of oral mucositis.
Another study showed that the use of laser therapy and dental care reduced the morbidity of oral mucositis, also reducing the costs of hospitalization.204204. Eduardo FP, Bezinelli LM, Orsi MC, Rodrigues M, Ribeiro MS, Hamerschlak N, et al. The influence of dental care associated with laser therapy on oral mucositis during allogeneic hematopoietic cell transplant: retrospective study. einstein (Sao Paulo). 2011;9(2):201-6.
Keratinocyte growth factor (palifermin)
Palifermin is a mytogenic agent for fibroblasts, keratinocytes and endothelial cells, which increases mucosal thickness and also attenuates the effects of TNF-α, reducing the typical inflammation found in mucositis. Clinical trials have demonstrated a decrease in the incidence and duration of severe oral mucositis (grades 3 and 4).205205. Nguyen DT, Shayani S, Palmer J, Dagis A, Forman SJ, Epstein J, et al. Palifermin for prevention of oral mucositis in allogeneic hematopoietic stem cell transplantation: a single-institution retrospective evaluation. Support Care Cancer. 2015;23(11):3141-7.
Nutritional therapy in mucositis
NT in cancer patients aims to minimize weight loss and prevent specific nutritional deficiencies. Patients with mucositis also have difficulty swallowing and, sometimes, keeping food in their mouth. Diarrhea may also be present. In these cases, the NT of choice is other than oral and must not exacerbate diarrhea.
In mild mucositis, while oral feeding is still possible, it is important to change the consistency of the diet. In addition, foods that do not irritate the mucosa must be used (reduced acids, spices and salt), as well as foods with a soft texture.
Gastrointestinal mucositis causes damage to crypt cells, decreases production of digestive enzymes, and transient intolerance to lactose can occur. Therefore, a dairy-light diet may be required.
For patients unable to meet their nutritional targets, enteral nutrition (EN) or PNT must be considered. It is possible to safely insert an enteral tube in patients with more severe mucositis. Parenteral nutrition therapy may be indicated for patients unable to be fed orally or enterally due to ileus, incoercible vomiting, or obstructive conditions. Patients with severe mucositis, although they are still candidates for EN, may have additional benefits with PNT, especially for prolonged periods.
Glutamine supplementation was, for a long time, used as part of the management of gastrointestinal mucositis, but recent studies have shown conflicting results regarding its benefits.206206. Keefe DM, Rassias G, O’Neil L, Gibson RJ. Severe mucositis: how can nutrition help? Curr Opin Clin Nutr Metab Care. 2007;10(5):627-31.
The typical lesions of oral mucositis are, by and large, very painful, and may compromise the nutrition and oral hygiene of patients, in addition to increasing the risk of local and systemic infection. Due to its complex and multifactorial pathogenesis, the treatment of mucositis consists of palliative care, such as pain management and oral hygiene, as well as NT, which must be a multidisciplinary decision.196196. Lalla RV, Sonis ST, Peterson DE. Management of oral mucositis in patients who have cancer. Dent Clin North Am. 2008;52(1):61-77.
ANOREXIA
Anorexia can occur due to the disease process, metabolic disorder, side effects of treatment, or even depression, and is common in cancer patients, particularly in more advanced disease states. It can also be related with the effects of nausea and vomiting.4949. Brasil. Ministério da Saúde. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Consenso Nacional de Nutrição Oncológica. 2ª ed [Internet]. Rio de Janeiro (RJ): INCA; 2015. 186 p [citado 2019 Maio 10]. Disponível em: http://www1.inca.gov.br/inca/Arquivos/Consenso_Nutricao_vol_II_2_ed_2016.pdf
http://www1.inca.gov.br/inca/Arquivos/Co...
,207207. Nunes MP. Síndrome da anorexia-caquexia em portadores de câncer. Rev Brasil Cancerol. 2006;52(1):59-77.
Anorexia is defined as early satiety and/or loss of appetite, and is classified as a biopsychosocial phenomenon, present in approximately 40% of cancer patients; its etiology is still under debate.207207. Nunes MP. Síndrome da anorexia-caquexia em portadores de câncer. Rev Brasil Cancerol. 2006;52(1):59-77.,208208. Molassiotis A. Anorexia and weight loss in long-term survivors of haematological malignancies. J Clin Nurs. 2003;12(6):925-7.
Both hormonal factors and cytokine activity are involved in appetite regulation. Neuropeptide Y and ghrelin are orexigenic hormones, and the first was found at lower levels in animals with tumors. Melanocortin is attenuated in the presence of cancer, and has anorexigenic action. Some cytokines, such as TNF-α, interleukins 1 and 6, and interferon gamma, are released by tumors and act to reduce food intake.207207. Nunes MP. Síndrome da anorexia-caquexia em portadores de câncer. Rev Brasil Cancerol. 2006;52(1):59-77.,209209. Porporato PE. Understanding cachexia as a cancer metabolism syndrome. Oncogenesis. 2016;5:e200. Review.
Anorexia is not the only symptom involved in the weight loss of cancer patients, although it is relevant in the context of cachexia.208208. Molassiotis A. Anorexia and weight loss in long-term survivors of haematological malignancies. J Clin Nurs. 2003;12(6):925-7.,210210. Associação Brasileira de Cuidados Paliativos. Consenso Brasileiro de Caquexia / anorexia. Rev Bras Cuidados Paliativos. 2011;3(3 Suppl 1):1-42.
It is the most frequent cause of insufficient intake after conditioning regimens, and early satiety and delayed gastric emptying are the main factors contributing to reduced hunger. The appropriate nutritional interventions advocate smaller meals, eating slowly and drinking liquids only between main meals. Other factors impacting appetite include impaired sense of taste and emotional problems, such as anxiety and depression − frequently present in transplant patients, mostly due to prolonged hospitalization.211211. Rust DM, Simpson JK, Lister J. Nutritional issues in patients with severe neutropenia. Semin Oncol Nurs. 2000;16(2):152-62.
Anorexia usually occurs between the second and the third weeks of the transplant, mostly caused by the conditioning regimen.212212. Paton E, Coutinho M, Voltarelli J. Diagnóstico e tratamento de Complicações agudas do Transplante de Células Progenitoras Hematopoéticas. Medicina (Ribeirão Preto). 2000;33(3):264-77. Oral intake is heavily affected during the transplant and remains low for up to 3 weeks after treatment.213213. Malone FR, Leisenring WM, Storer BE, Lawler R, Stern JM, Aker SN, et al. Prolonged anorexia and elevated plasma cytokine levels following myeloablative allogeneic hematopoietic cell transplant. Bone Marrow Transplant. 2007;40(8):765-72.
A study investigated 147 transplanted patients, and decreased oral intake was observed in 92%, with a mean calorie intake of 3% of baseline energy requirements of these patients. This marked decrease in oral intake took place predominantly between days 10 and 12 of the conditioning regimen.213213. Malone FR, Leisenring WM, Storer BE, Lawler R, Stern JM, Aker SN, et al. Prolonged anorexia and elevated plasma cytokine levels following myeloablative allogeneic hematopoietic cell transplant. Bone Marrow Transplant. 2007;40(8):765-72.
Another study observed that, in 69 patients followed-up in the post-transplant period, 66% had feeding difficulties on day 50 after the bone marrow infusion, and anorexia was one of the reasons for reduced oral intake. This loss of appetite regressed over time after treatment, but was still prevalent in 16% of patients on day 200 after infusion.9595. Iestra JA, Fibbe WE, Zwinderman AH, van Staveren WA, Kromhout D. Body weight recovery, eating difficulties and compliance with dietary advice in the first year after stem cell transplantation: a prospective study. Bone Marrow Transplant. 2002;29(5):417-24.
Some studies have investigated the prevalence of malnutrition and its correlation with the presence of side effects in cancer patients on adjuvant or neoadjuvant therapy. A recent multicenter study with 65 hospitals and 561 patients on adjuvant therapy showed that 90.7% had lost weight. The same study found that 96.4% had nutritional complications such as anorexia (70.9%), gastrointestinal disorders (32.6%), dysgeusia (40.5%) and dysphagia, among others.4949. Brasil. Ministério da Saúde. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Consenso Nacional de Nutrição Oncológica. 2ª ed [Internet]. Rio de Janeiro (RJ): INCA; 2015. 186 p [citado 2019 Maio 10]. Disponível em: http://www1.inca.gov.br/inca/Arquivos/Consenso_Nutricao_vol_II_2_ed_2016.pdf
http://www1.inca.gov.br/inca/Arquivos/Co...
Since toxicity effects lead to decreased food intake for extended periods, this makes it even more important to monitor the nutritional status of patients undergoing transplant.9595. Iestra JA, Fibbe WE, Zwinderman AH, van Staveren WA, Kromhout D. Body weight recovery, eating difficulties and compliance with dietary advice in the first year after stem cell transplantation: a prospective study. Bone Marrow Transplant. 2002;29(5):417-24.
In the nutritional approach to anorexia,4949. Brasil. Ministério da Saúde. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Consenso Nacional de Nutrição Oncológica. 2ª ed [Internet]. Rio de Janeiro (RJ): INCA; 2015. 186 p [citado 2019 Maio 10]. Disponível em: http://www1.inca.gov.br/inca/Arquivos/Consenso_Nutricao_vol_II_2_ed_2016.pdf
http://www1.inca.gov.br/inca/Arquivos/Co...
educating patients on the need to eat; eating smaller, more frequent meals; increasing calorie and protein content in meals; increasing consumption of high-calorie snacks; increasing consumption of better accepted foods; adding high-calorie, high-protein supplements; and choosing higher-calorie drinks are important points to be observed.
WEIGHT LOSS
Hematopoietic stem cell transplantation is now indicated in different hematological and high-morbidity, non-hematological diseases. High-dose cytotoxic chemotherapy, with or without total body irradiation, results in hypercatabolism, breakdown of the gastrointestinal mucosal barrier, and immunosuppression. Gastrointestinal and infectious complications from conditioning are frequent, impairing the nutritional status and contributing to high morbidity and mortality. In addition, for patients already in a catabolic state, the increased metabolic demand associated with treatment further aggravates the problem.214214. Albertini SM. O transplante de células-tronco hematopoéticas e o fator nutricional na evolução dos pacientes. Rev Bras Hematol Hemoter. 2010;32(1):8-9.
A study looked into the impact of patient weight on transplant outcomes. When compared with patients close to their ideal weight, patients with low weight had worse results after allogeneic HSCT, and this was probably related with the aggressive pre-transplant therapy. Also, worse results can reflect differences in drug distribution volumes and an effect of weight on pharmacokinetics.215215. Fleming DR, Rayens MK, Garrison J. Impact of obesity on allogeneic stem cell transplant patients: a matched case-controlled study. Am J Med. 1997;102(3):265-8.
Patients scheduled to undergo HSCT are at higher risk for malnutrition related with prior chemotherapy and/or radiation therapy, due to side effects − many of which involve the gastrointestinal tract, leading to increased energy requirements and prolonged hospitalization.
Hematopoietic stem cell transplantation has multiple side effects, such as anorexia, diarrhea, mucositis and dysgeusia, which affect food acceptance and, as a consequence, deteriorate patients’ nutritional status.9595. Iestra JA, Fibbe WE, Zwinderman AH, van Staveren WA, Kromhout D. Body weight recovery, eating difficulties and compliance with dietary advice in the first year after stem cell transplantation: a prospective study. Bone Marrow Transplant. 2002;29(5):417-24.,216216. Associação Médica Brasileira (AMB). Conselho Federal de Medicina. Projeto Diretrizes. Terapia. Terapia Nutricional no Transplante de Célula Hematopoiética [Internet]. São Paulo: ABM; 2011 [citado 2018 Jul 11]. Disponível em: https://diretrizes.amb.org.br/_BibliotecaAntiga/terapia_nutricional_no_transplante_de_celula_hematopoietica.pdf
https://diretrizes.amb.org.br/_Bibliotec...
A significant part of patients undergoing transplant develop mucositis, with considerable difficulty maintaining adequate calorie intake, which is even worse in patients with acute GVHD affecting the digestive tract.217217. Blijlevens NM, Donnelly JP, De Pauw BE. Mucosal barrier injury: biology, pathology, clinical counterparts and consequences of intensive treatment for haematological malignancy: an overview. Bone Marrow Transplant. 2000;25(12):1269-78. Review.,218218. Stiff P. Mucositis associated with stem cell transplantation: current status and innovative approaches to management. Bone Marrow Transplant. 2001;27 Suppl 2:S3-S11. Review.
Nutritional status is an important prognostic factor in patients undergoing HSCT. There are three important periods for patients undergoing HSCT: the initial period, which comprises the conditioning phase, and the cytoreductive phase, causing extensive tissue damage. The second period, right after the transplant, is characterized by intense pancytopenia, followed by repair of damaged tissues. The third period, when the implantation of transplanted cells takes place, is when infectious complications can occur.219219. Albertini S, Ruiz MA. Nutrição em transplante de medula óssea: a importância da terapia nutricional. Arq Ciênc Saúde 2004;11(3):182-8
In a retrospective study, morbidity and mortality were higher among patients with lymphoma undergoing autologous HSCT when these patients were malnourished at admission, in comparison to those with normal weight.220220. Navarro WH, Loberiza FR Jr, Bajorunaite R, van Besien K, Vose JM, Lazarus HM, et al. Effect of body mass index on mortality of patients with lymphoma undergoing autologous hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2006;12(5):541-51.
All patients undergoing HSCT are at nutritional risk, mainly due to adverse effects from prior treatment. Moreover, half of the patients undergoing HSCT have impaired nutritional status for up to one year after transplant, and weight loss and nutritional complications are more frequent in patients submitted to allogeneic HSCT.221221. Spexoto MC, Oliveira MR. Consumo alimentar orientado pode prevenir a queda ponderal no pós-transplante de células- tronco hematopoéticas imediato. Revi Bras Nutr Clin. 2013;28(2):91-7.
Another study also found weight loss after autologous and allogeneic transplant. The authors found that weight loss persisted for up to 75 days after transplant of 118 study subjects, even for those who did not complete the study.9595. Iestra JA, Fibbe WE, Zwinderman AH, van Staveren WA, Kromhout D. Body weight recovery, eating difficulties and compliance with dietary advice in the first year after stem cell transplantation: a prospective study. Bone Marrow Transplant. 2002;29(5):417-24.
A survey conducted in a cancer institution in the state of São Paulo, in 2016, studied 123 autologous and allogeneic transplants, and found that the mean weight at pre-conditioning was 75.6kg, and the mean weight at post-conditioning was 73.8kg. Of the total patients, 83 lost weight during the transplant, with an average weight loss of 3.2kg.
Some studies have assessed the impact of nutritional status on patients undergoing HSCT. In a study for the ASBMT, which investigated the impact of nutritional status in 1,087 patients treated with high-dose melphalan and undergoing autologous HSCT with a diagnosis of multiple myeloma, patients were sorted based on BMI as normal, overweight, obese or severely obese. There was no overall effect of BMI on progression-free survival, overall survival, disease progression, or relapse-free mortality.1919. Vogl DT, Wang T, Pérez WS, Stadtmauer EA, Heitjan DF, Lazarus HM, et al. Effect of obesity on outcomes after autologous hematopoietic stem cell transplantation for multiple myeloma. Biol Blood Marrow Transplant. 2011;17(12):1765-74.,222222. Costa LJ, Micallef IN, Inwards DJ, Johnston PB, Porrata LF, Litzow MR, et al. Effect of the dose per body weight of conditioning chemotherapy on severity of mucositis and risk of relapse after autologous haematopoietic stem cell transplantation in relapsed diffuse large B cell lymphoma. Br J Haematol. 2008;143(2):268-73.
The pre-HSCT nutritional status is related with the post-HSCT clinical progression, directly affecting the time to engraftment. Due to the intense toxicity to which these patients are subjected, the risk for malnutrition in the pre- and post-transplant phases must be monitored, as well as complications related to nutritional status, and the consequent decrease in food intake.214214. Albertini SM. O transplante de células-tronco hematopoéticas e o fator nutricional na evolução dos pacientes. Rev Bras Hematol Hemoter. 2010;32(1):8-9.
Patients with a nutritional diagnosis of malnutrition and obesity are at higher risk of death in the immediate post-HSCT period, and patients with major muscle mass depletion have more clinical complications and longer lengths of stay.8888. Kyle UG, Chalandon Y, Miralbell R, Karsegard VL, Hans D, Trombetti A, et al. Longitudinal follow-up of body composition in hematopoietic stem cell transplant patients. Bone Marrow Transplant. 2005;35(12):1171-7.
Malnutrition, very often characterized by weight loss, deteriorates patient prognosis and survival.9292. Horsley P, Bauer J, Gallagher B. Poor nutritional status prior to peripheral blood stem cell transplantation is associated with increased length of hospital stay. Bone Marrow Transplant. 2005;35(11):1113-6.,133133. Sommacal HM, Gazal CH, Jochims AM, Beghetto M, Paz A, Silla LM, et al. Clinical impact of systematic nutritional care in adults submitted to allogeneic hematopoietic stem cell transplantation. Rev Bras Hematol Hemoter. 2012;34(5):334-8. A study investigated the length of stay according to patients’ nutritional status. Patients classified as well-nourished, as per the PG-SGA, had a mean length of stay of 16.9±6.3 days, while patients classified as at risk for malnutrition, or patients with moderate and severe malnutrition, had a mean length of stay of 23.9±9.9 days, with a significant difference between the groups (p=0.002).9292. Horsley P, Bauer J, Gallagher B. Poor nutritional status prior to peripheral blood stem cell transplantation is associated with increased length of hospital stay. Bone Marrow Transplant. 2005;35(11):1113-6.
Irrespective of whether the current food intake and nutritional status are maintained, patients scheduled to undergo HSCT require individualized nutritional monitoring, aiming to offer and educate on the appropriate NT for each phase of the transplant, minimizing involution of the nutritional status and future nutritional complications. Transplanted patients present with unintentional weight loss, regardless of the type of HSCT to which they are submitted to.223223. Paiva M, Biase RC, Moraes JJ, Ângelo AR, Honorato MC. Complicações orais decorrentes da terapia antineoplásica. Arq Odontol. 2010;46(1): 48-55.224.
ORAL DIET
Nutritional supplements
Nutritional supplements are one of the alternatives when patients are not consuming the calories and nutrients required to cover their nutritional demands,224224. Fadoni MR, Sicchieri JM. Suplementos nutricionais e nutrição enteral. In: Navarro AM, Japur CC, Sicchieri JMF, Chiarello PG, Diez Garcia RW, editors. Atualiadades em Alimentação e Nutrição Hospitalar. 1st ed. Rio de Janeiro-RJ: editora Atheneu; 2017. p. 215-24. and to offer nutrients for therapeutic purposes.
Oral nutritional supplements are defined as mixtures containing nutrients for oral ingestion, and can be purchased as ready-to-consume formulations, prepared with fresh foods, and industrially processed; they can also be purchased based on a combination, in order to supply calories and nutrients in a concentrated form.4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48.,224224. Fadoni MR, Sicchieri JM. Suplementos nutricionais e nutrição enteral. In: Navarro AM, Japur CC, Sicchieri JMF, Chiarello PG, Diez Garcia RW, editors. Atualiadades em Alimentação e Nutrição Hospitalar. 1st ed. Rio de Janeiro-RJ: editora Atheneu; 2017. p. 215-24.Table 11 presents some of the formulations available in the market.
Prolonged, repetitive use of the same supplements may hinder adherence and reduce tolerance to this strategy. Special caution should be taken with high-osmolar-concentration formulations, when diarrhea is present, and with nutrients that can aggravate symptoms or irritate the mucosa, such as fibers, mono- and disaccharides.225225. Halmos EP. Role of FODMAP content in enteral nutrition-associated diarrhea. J Gastroenterol Hepatol. 2013;28 Suppl 4:25-8. Review. Options with higher energy density are interesting due to their greater calorie supply, however they usually have this limitation. Some strategies, such as using flavorless supplements, alternating options, or even mixing these formulations with foods may improve adherence,224224. Fadoni MR, Sicchieri JM. Suplementos nutricionais e nutrição enteral. In: Navarro AM, Japur CC, Sicchieri JMF, Chiarello PG, Diez Garcia RW, editors. Atualiadades em Alimentação e Nutrição Hospitalar. 1st ed. Rio de Janeiro-RJ: editora Atheneu; 2017. p. 215-24. as well as resorting to supplements compounded with foods, which offer greater diversity of flavors and textures.
Modular supplements
Modular supplements can be an interesting option to improve the consumption of a nutrient alone, such as to adjust protein intake.224224. Fadoni MR, Sicchieri JM. Suplementos nutricionais e nutrição enteral. In: Navarro AM, Japur CC, Sicchieri JMF, Chiarello PG, Diez Garcia RW, editors. Atualiadades em Alimentação e Nutrição Hospitalar. 1st ed. Rio de Janeiro-RJ: editora Atheneu; 2017. p. 215-24. They are interesting in dietary management, since they can be used in preparations with foods that are better accepted by or more familiar to patients.
Carbohydrate module
Usually based on maltodextrin, sucrose or sucrose polymer. Powdered, and used to increase energy densit,y without significantly altering the flavor of the final preparation.
Lipid module
Based on middle-chain triglycerides, lipid modules are used for their better absorption, their offer of essential fatty acids, or both. Mostly found in liquid formulations, they are also used to increase the energy density of preparations.
Protein module
Usually based on whey proteins, protein modules may contain essential amino acids, branched chain amino acids, protein hydrolysate, calcium caseinate etc. They are used to increase the protein density of preparations without changing texture or volume. Mostly found in powdered form.
Supplements with aminoacids glutamine and beta-hydroxy-beta-methylbutyrate
Beta-hydroxy-beta-methylbutyrate (HMB) is a leucine metabolite, which has been used in studies on sarcopenia, with more significant results in young patients than in the elderly, as an alternative for restoring protein synthesis.4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48. Patients under treatment for HSCT, using anthracyclines and corticosteroids, which contribute to accentuate protein catabolism, may have intense muscle fatigue.
The role of glutamine (conditionally essential amino acid) and its benefits in patients undergoing HSCT are not yet well established. Its oral administration has been indicated for symptoms such as mucositis and associated odynophagia; however, studies do not provide conclusive results due to different methodologies.4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48.,226226. Mochamat CH, Cuhls H, Marinova M, Kaasa S, Stieber C, Conrad R, et al. A systematic review on the role of vitamins, minerals, proteins, and other supplements for the treatment of cachexia in cancer: a European Palliative Care Research Centre cachexia project. J Cachexia Sarcopenia Muscle. 2017;8(1):25-39.
Probiotic supplements
According to the WHO, probiotics are living organisms that, at the right amount, can promote health and benefits to their hosts.227227. Wang W, Xu S, Ren Z, Jiang J, Zheng S. Gut microbiota and allogeneic transplantation. J Transl Med. 2015;13(1):275. In related publications, there is no evidence of any pharmacological action of probiotics in the host, however a symbiotic effect has been verified in experimental studies, by comparing parameters such as weight of organs, cardiac output and immune response of experimental germ-free animals.227227. Wang W, Xu S, Ren Z, Jiang J, Zheng S. Gut microbiota and allogeneic transplantation. J Transl Med. 2015;13(1):275.
It is known that patients undergoing HSCT have intense dysbiosis, frequently associated with severe diarrhea, GVHD and infection. The change in the percentage of beneficial bacteria is even more marked with the use of antibiotics, which are frequently given to transplant patients due to the intense immunosuppression to which they are subjected. However, regarding probiotic supplementation in patients undergoing HSCT, few but promising studies have shown that probiotic supplementation, in addition to reestablishing intestinal dysbiosis, is capable of preventing some post-HSCT complications, such as GVHD.228228. Ladas EJ, Bhatia M, Chen L, Sandler E, Petrovic A, Berman DM, et al. The safety and feasibility of probiotics in children and adolescents undergoing hematopoietic cell transplantation. Bone Marrow Transplant. 2016;51(2):262-6. Some findings argued that supplements containing Lactobacillus may prevent the proliferation of Enterococcus in the gut.228228. Ladas EJ, Bhatia M, Chen L, Sandler E, Petrovic A, Berman DM, et al. The safety and feasibility of probiotics in children and adolescents undergoing hematopoietic cell transplantation. Bone Marrow Transplant. 2016;51(2):262-6. Feared adverse events, such as bacteremia and bacterial translocation, have not been found in the study by Ladas et al.,228228. Ladas EJ, Bhatia M, Chen L, Sandler E, Petrovic A, Berman DM, et al. The safety and feasibility of probiotics in children and adolescents undergoing hematopoietic cell transplantation. Bone Marrow Transplant. 2016;51(2):262-6. who administered Lactobacillus plantarum to children undergoing HSCT.228228. Ladas EJ, Bhatia M, Chen L, Sandler E, Petrovic A, Berman DM, et al. The safety and feasibility of probiotics in children and adolescents undergoing hematopoietic cell transplantation. Bone Marrow Transplant. 2016;51(2):262-6.
The main microorganisms commercialy available in Brazil are Lactobacillus acidophilus, Lactobacillus casei shirota, Lactobacillus casei variety rhamnosus, Lactobacillus casei variety defensis, Lactobacillus delbrueckii subsp. bulgaricus, Lactobacillus paracasei, Lactococcus lactis, Bifidobacterium bifidum, Bifidobacterium lactis, Bifidobacterium longum, Bifidobacterium animalis, Enterococcus faecium and Streptococcus salivarius subsp. thermophillus. The minimum quantity to be considered a probiotic is 108 to 101010. Rocha V, Porcher R, Fernandes JF, Filion A, Bittencourt H, Silva W Jr, et al. Association of drug metabolism gene polymorphisms with toxicities, graft-versus-host disease and survival after HLA-identical sibling hematopoietic stem cell transplantation for patients with leukemia. Leukemia. 2009;23(3):545-56.colony forming units (CFU) per portion, and they can be sold in capsules, powdered, freeze-dried or in yoghurts.229229. De Paula I, Theophilo P, Cintra B. Tratamento com probióticos na síndrome do intestino irritável. Rev Bras Ciências da Saúde. 2008;19(3):271-81. There is no consensus in the literature about which dose to use and at which point during HSCT this supplemention should be initiated; although the idea seems promising, further clinical studies are needed to establish the safety and efficacy in patients undergoing HSCT.
Herbal supplements
The integrative approach, which combines knowledge, practices and natural resources with conventional treatments, has been well accepted by health professionals, allowing for investigation of new alternatives to relieve symptoms, stimulate the appetite and help alleviate anxiety.4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48.
It is always necessary to reinforce to patients and family members that they must inform the care team about any herbal supplements or other natural substances they may be on.230230. Sierpina V, Levine L, McKee J, Campbell C, Lian S, Frenkel M. Nutrition, metabolism, and integrative approaches in cancer survivors. Semin Oncol Nurs. 2015;31(1):42-52. Some infusions, herbs and compounds may contain substances that interfere with the metabolism of some drugs, affecting their action.230230. Sierpina V, Levine L, McKee J, Campbell C, Lian S, Frenkel M. Nutrition, metabolism, and integrative approaches in cancer survivors. Semin Oncol Nurs. 2015;31(1):42-52. For instance, Hypericum perforatum (Saint John’s wort), popularly recommended for depressive conditions, may interfere with imatinib mesylate (Glivec®).230230. Sierpina V, Levine L, McKee J, Campbell C, Lian S, Frenkel M. Nutrition, metabolism, and integrative approaches in cancer survivors. Semin Oncol Nurs. 2015;31(1):42-52.Camellia sinensis (green tea or matcha), known for its antioxidant and thermogenic properties, interferes with bortezomib (Velcade®).230230. Sierpina V, Levine L, McKee J, Campbell C, Lian S, Frenkel M. Nutrition, metabolism, and integrative approaches in cancer survivors. Semin Oncol Nurs. 2015;31(1):42-52.
Nutritional supplements added to foods
The use of supplements with foods, combined in such a way as to offer calories and specific nutrients, with different temperatures, consistencies and flavors, may be an option to meet nutritional demands during treatment. These alternatives involve food preparations with different presentations and can stimulate food intake and nutritional demands in periods of hyporexia. They can be formulated as needed.
Diet for patients with neutropenia
Neutropenia is defined as a neutrophil count under 500/µL or lower than 1.000/µL, expected to drop to 500/µL within two days. In these situations, some prophylactic measures are adopted to control the risk of infection.231231. Galati PC, Brandão EM. Abordagem nutricional no transplante de medula óssea. In: Navarro AM, Japur CC, Sicchieri JM, Chiarello PG, Diez Garcia RW, editors. Atualidades em Alimentação e Nutrição Hospitalar. 1st ed. editora Atheneu; 2017. p. 235-49. However, there is no consensus in the literature about the benefits of using a neutropenic diet. Its use was instituted in the 1980’s to prevent digestive tract infections, and its main characteristic was restricting foods that could act as vectors of pathogenic microorganisms.4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48.,221221. Spexoto MC, Oliveira MR. Consumo alimentar orientado pode prevenir a queda ponderal no pós-transplante de células- tronco hematopoéticas imediato. Revi Bras Nutr Clin. 2013;28(2):91-7. Until 2006, the literature had studies with diets restricting consumption of raw vegetables, fresh and sun-dried fruit, nuts, yoghurts, eggs and undercooked meats. Studies comparing diets with these restrictions with conventional diets, with properly washed foods, showed that said restrictions did not protect neutropenic patients and, in some cases, the number of complications was higher for the more restrictive diets.232232. Baumgartner A, Bargetzi A, Zueger N, Bargetzi M, Medinger M, Bounoure L, et al. Revisiting nutritional support for allogeneic hematologic stem cell transplantation-a systematic review. Bone Marrow Transplant. 2017; 52(4):506-13.
According to the ESPEN consensus, due to the difficulty comparing studies with different methodologies and divergent concepts, including in respect to the neutropenic condition, which warrants these approaches, the use of these diets is not supported in the scientific literature.4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48.The guidance has been pointing to an opposite direction: instead of restrictive diets, the focus has been on the quality of sanitation procedures, i.e., allowing more flexible diets, provided the right precautions are taken when purchasing, storing and preparing foods.4949. Brasil. Ministério da Saúde. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Consenso Nacional de Nutrição Oncológica. 2ª ed [Internet]. Rio de Janeiro (RJ): INCA; 2015. 186 p [citado 2019 Maio 10]. Disponível em: http://www1.inca.gov.br/inca/Arquivos/Consenso_Nutricao_vol_II_2_ed_2016.pdf
http://www1.inca.gov.br/inca/Arquivos/Co...
,232232. Baumgartner A, Bargetzi A, Zueger N, Bargetzi M, Medinger M, Bounoure L, et al. Revisiting nutritional support for allogeneic hematologic stem cell transplantation-a systematic review. Bone Marrow Transplant. 2017; 52(4):506-13.
For sanitization of fruit and vegetables, it is safe to use chlorine-based solutions, such as the hypochloride solutions intended for this purpose, with 200 ppm of active chlorine, which ensures great antimicrobial power with no corrosive action.233233. Silva Junior EA, editor. Manual de controle higiênico sanitário em serviços de alimentação. São Paulo: Varela; 2007. p. 239-66. As for residues, the amount ingested is much lower than tolerated, even considering the intake of washed, raw foods, several times a day. After washing, foods can be rinsed with treated water.233233. Silva Junior EA, editor. Manual de controle higiênico sanitário em serviços de alimentação. São Paulo: Varela; 2007. p. 239-66.
The recommendations about the precautions adopted in this sense are those used for the general population, as presented in table 12.
Water is another important item that is worth discussing, considering aspects related with water treatment, as well as sales and distribution channels. The definition of treated water is water from the water supply system, filtered or boiled for at least 2 minutes, or filtered and chlorinated.233233. Silva Junior EA, editor. Manual de controle higiênico sanitário em serviços de alimentação. São Paulo: Varela; 2007. p. 239-66. There are national programs under the Ministry of Health, such as the (SISÁGUA - Sistema de Informação de Vigilância da Qualidade da Água para Consumo Humano) which monitor the quality of water for human consumption, and must be periodically consulted by healthcare professionals for better understanding of recommendations.234234. Barbosa AM, Solano ML, Umbuzeiro GA. Pesticides in Drinking Water - The Brazilian Monitoring Program. Front Public Health. 2015;3:246. In cities with no watter supply network, it is recommended to add one drop of sodium hypochlorite for each liter of water consumed, or boil the water in a clean pan.235235. Pontara AV, de Oliveira CD, Barbosa AH, Dos Santos RA, Pires RH, Martins CH. Microbiological monitoring of mineral water commercialized in Brazil. Braz J Microbiol. 2011;42(2):554-9.,236236. Agência Nacional de Vigilância Sanitária (ANVISA). Guia de alimentos e vigilância sanitária [Internet]. Brasília (DF): ANVISA; 2004 [citado 2017 Mar 18]. Disponível em: http://portal.anvisa.gov.br/resultado-de-busca?p_p_id=101&p_p_lifecycle=0&p_p_state=maximized&p_p_mode=view&p_p_col_id=column-1&p_p_col_count=1&_101_struts_action=%2Fasset_publisher%2Fview_content&_101_assetEntryId=395967&_101_type=document
http://portal.anvisa.gov.br/resultado-de...
As for bottled spring water, a study has shown that fungal contamination may occur during the bottling and/or storage processes, therefore, this alternative is not necessarily safe.235235. Pontara AV, de Oliveira CD, Barbosa AH, Dos Santos RA, Pires RH, Martins CH. Microbiological monitoring of mineral water commercialized in Brazil. Braz J Microbiol. 2011;42(2):554-9.
Furthermore, one should pay attention to the cleaning of domestic water tanks every 6 months, as well as make sure to know the origin and the way water containers were stored and transported. These procedures must be explained to patients to guide them on the best options to use high chemical- and microbiological-quality water.234234. Barbosa AM, Solano ML, Umbuzeiro GA. Pesticides in Drinking Water - The Brazilian Monitoring Program. Front Public Health. 2015;3:246.
Table 13 presents the main recommendations for patients with neutropenia.
Parenteral nutrition
Nutritional Therapy in HSCT has the purpose of maintaining and recovering nutritional status, avoiding or reducing nutritional deficits resulting from chemotherapy and/or radiation therapy, minimizing the effects of the conditioning regimen, and providing appropriate substrate for recovery of the hematopoietic and immune systems.
Patients undergoing HSCT can be considered as at nutritional risk, due to reduced calorie supply, impaired nutrient absorption, and increased metabolic demands, which may compromise the nutritional status of these patients throughout the process.1212. Rzepecki P, Barzal J, Oborska S. Blood and marrow transplantation and nutritional support. Support Care Cancer. 2010;18(Suppl 2):S57-65.,216216. Associação Médica Brasileira (AMB). Conselho Federal de Medicina. Projeto Diretrizes. Terapia. Terapia Nutricional no Transplante de Célula Hematopoiética [Internet]. São Paulo: ABM; 2011 [citado 2018 Jul 11]. Disponível em: https://diretrizes.amb.org.br/_BibliotecaAntiga/terapia_nutricional_no_transplante_de_celula_hematopoietica.pdf
https://diretrizes.amb.org.br/_Bibliotec...
,238238. Akbulut G. Medical Nutritional Therapy in Hematopoietic Stem Cell Transplantation (HSCT). Int J Hematol Oncol. 2013;23(1):55-65.,239239. Zatarain L, Savani BN. The role of nutrition and effects on the cytokine milieu in allogeneic hematopoietic stem cell transplantation. Cell Immunol. 2012;276(1-2):6-9.Thus, these patients are prone to developing multiple metabolic disorders of variable severity − mostly during the immediate post-transplant period. The main causes include adverse effects from the conditioning regimen, immunosuppressants and PNT.240240. Lee JH, Choi SJ, Lee JH, Kim SE, Seol M, Lee YS, et al. Severe metabolic abnormalities after allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2005;35(1):63-9.
Adverse effects of chemotherapy and radiation therapy predominantly affect the gastrointestinal tract and the immune system.238238. Akbulut G. Medical Nutritional Therapy in Hematopoietic Stem Cell Transplantation (HSCT). Int J Hematol Oncol. 2013;23(1):55-65. Therefore, in addition to nausea and vomiting, severe mucositis frequently occurs, associated with intense odynophagia, abdominal pain, diarrhea, and infections.147147. Martin-Salces M, de Paz R, Canales MA, Mesejo A, Hernandez-Navarro F. Nutritional recommendations in hematopoietic stem cell transplantation. Nutrition. 2008;24(7-8):769-75. These factors further impair oral tolerability, contributing to aggravation of weight loss, which can persist for up to 40 days after admission for the transplant.241241. Urbain P, Birlinger J, Lambert C, Finke J, Bertz H, Biesalski HK. Longitudinal follow-up of nutritional status and its influencing factors in adults undergoing allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2013;48(3):446-51. As a consequence, malnutrition can rapidly occur while the patients is hospitalized for HSCT.
Regarding the type of NT to be used, data suggest a trend towards fewer complications during the use of EN when compared to PNT in HSCT, particularly for infectious complications and propensity for more severe forms of intestinal GVHD.242242. Guièze R, Lemal R, Cabrespine A, Hermet E, Tournilhac O, Combal C, et al. Enteral versus parenteral nutritional support in allogeneic haematopoietic stem-cell transplantation. Clin Nutr. 2014;33(3):533-8. However, a study with patients requiring admission to the intensive care unit (ICU) did not show any association of poorer outcomes in the group receiving PNT.243243. Azoulay E, Pène F, Darmon M, Lengliné E, Benoit D, Soares M, et al.; Groupe de Recherche Respiratoire en Réanimation Onco-Hématologique (Grrr-OH). Managing critically Ill hematology patients: time to think differently. Blood Rev. 2015;29(6):359-67.,244244. Bayraktar UD, Nates JL. Intensive care outcomes in adult hematopoietic stem cell transplantation patients. World J Clin Oncol. 2016;7(1):98-105.
Parenteral nutrition therapy has been shown to be safer and more effective in maintaining and recovering nutritional status when compared to patients on ENT, with gastrointestinal intolerance and severe mucositis. Cost-effectiveness assessments based on definitive outcome indicators have influenced the clinical practice of delaying PNT initiation for patients at normal weight undergoing autologous HSCT, in contrast to those undergoing allogeneic HSCT, since the duration of food intolerance in the former is shorter.4040. August DA, Huhmann MB; American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. A.S.P.E.N. clinical guidelines: nutrition support therapy during adult anticancer treatment and in hematopoietic cell transplantation. JPEN J Parenter Enteral Nutr. 2009;33(5):472-500.,245245. Bozzetti F. Nutritional support of the oncology patient. Crit Rev Oncol Hematol. 2013;87(2):172-200.,246246. Andersen S, Brown T, Kennedy G, Banks M. Implementation of an evidenced based nutrition support pathway for haematopoietic progenitor cell transplant patients. Clin Nutr. 2015;34(3):536-40.
Indications
Parenteral nutrition therapy must be initiated at the pre-transplant phase in all patients eligible for treatment of their underlying disease, presenting significant weight loss with clinical repercussion. Underweight patients (BMI under 18.5kg/m2) are candidates for early PNT when oral intake falls below 50% within the first 3 days after infusion. Most of the time, patients candidate for HSCT arrive with good enough nutritional status, and must initiate PNT when gastrointestinal toxicity associated with grade III or IV mucositis occurs, with inadequate food intake for the past 10 days with no expected improvements, and when enteral nutrition has been excluded.147147. Martin-Salces M, de Paz R, Canales MA, Mesejo A, Hernandez-Navarro F. Nutritional recommendations in hematopoietic stem cell transplantation. Nutrition. 2008;24(7-8):769-75.,232232. Baumgartner A, Bargetzi A, Zueger N, Bargetzi M, Medinger M, Bounoure L, et al. Revisiting nutritional support for allogeneic hematologic stem cell transplantation-a systematic review. Bone Marrow Transplant. 2017; 52(4):506-13.
Parenteral nutrition therapy with central venous access allow for adequate supply of fluids and electrolytes, in addition to macronutrients, vitamins and trace elements.247247. Muscaritoli M, Grieco G, Capria S, Iori AP, Rossi Fanelli F. Nutritional and metabolic support in patients undergoing bone marrow transplantation. Am J Clin Nutr. 2002;75(2):183-90. In order to meet nutritional requirements, malnourished patients need progressive PNT prescription to prevent the incidence of refeeding syndrome. Parenteral nutrition therapy must be gradually introduced after assessment of the following criteria: availability of central venous access, nutritional status or performance status, presence of organ dysfunctions, and metabolic disorders.
The transition from PNT to oral diet can be done as soon as gastrointestinal symptoms improve. Foods must be introduced based on patient tolerability, starting with liquid and soft foods, and then progressing to the usual diet. Parenteral nutrition therapy discontinuation must also be gradual, by cutting the calorie-protein supply in half, according to food acceptance. Parenteral nutrition therapy can be discontinued when oral intake exceeds 50% of nutritional recommendations.4646. Bozzetti F, Arends J, Lundholm K, Micklewright A, Zurcher G, Muscaritoli M; ESPEN. ESPEN Guidelines on Parenteral Nutrition: non-surgical oncology. Clin Nutr. 2009;28(4):445-54.
Although PNT is a valuable resource in the management of patients undergoing HSCT, some circumstances may limit its use, such as an infected venous access, severe lipid and glucose metabolism disorders, fluid overload, and progressive liver dysfunction. Transplant units that have developed protocols that prioritize EN have shown favorable results in the group of patients studied.246246. Andersen S, Brown T, Kennedy G, Banks M. Implementation of an evidenced based nutrition support pathway for haematopoietic progenitor cell transplant patients. Clin Nutr. 2015;34(3):536-40.
Vascular access
The advent of deep venous catheters, with their semi-implantable and fully-implantable variants, allowed for more safety and better local care for prolonged PNT in HSCT. In general terms, patients scheduled to undergo autologous transplantation receive a short-term, multilumen, deep venous catheter, with one of the lumens dedicated to PNT, when needed. Patients scheduled for allogeneic transplant, on the other hand, receive long-term, semi-implantable deep venous catheter, to facilitate care in the post-transplant period.
Determination of the vascular access used for administration of PNT must consider two fundamental factors: the duration of PNT and the osmolarity of the solution. Table 14 presents the criteria for determination of vascular access.
Formulation and prescription
The basis of protein supply in PNT are essential and non-essential amino acid formulations. The standard 10% formulation for adults contains approximately 4kcal/g. Protein supply to critically-ill patients is crucial for availability and optimization of the metabolic demand for protein synthesis and tissue repair, and the muscle compartment is the main endogenous source of protein turnover. Glucose solutions also provide 4 kcal/g and probably represent, on average, 70% of non-protein calorie sources. Lipid emulsions are the most variable component of the formulation, due to different sources, and provides 10% to 20% of medium-chain, long-chain, monounsaturated-rich, or omega-3 triglycerides. On average, the lipid source at 20% provides 2kcal/mL and accounts for 30% of calorie supply of the solution. Macronutrient recommendations are presented in table 15.
Patients with moderate mucositis and diarrhea must use the protein target defined for clinically stable patients, i.e. 1.2 to 1.5g/kg/day. In the presence of severe mucositis, or changes related with severe infections, a higher protein target must be aimed at. To make the most of the protein supply, the non-protein calorie content for each gram of nitrogen should be 100:1 to 130:1. Although the physiological properties of glutamine have incited great interest in its use for EN and PNT, there are no robust studies recommending its inclusion in the PNT solution.232232. Baumgartner A, Bargetzi A, Zueger N, Bargetzi M, Medinger M, Bounoure L, et al. Revisiting nutritional support for allogeneic hematologic stem cell transplantation-a systematic review. Bone Marrow Transplant. 2017; 52(4):506-13. In fact, there could be a negative effect on the risk of late disease relapse (lymphomas).250250. Crowther M, Avenell A, Culligan DJ. Systematic review and meta-analyses of studies of glutamine supplementation in haematopoietic stem cell transplantation. Bone Marrow Transplant. 2009;44(7):413-25.
Glucose control is of utmost importance during PNT, since it minimizes the risk of complications and favors restoration of the nutritional status.216216. Associação Médica Brasileira (AMB). Conselho Federal de Medicina. Projeto Diretrizes. Terapia. Terapia Nutricional no Transplante de Célula Hematopoiética [Internet]. São Paulo: ABM; 2011 [citado 2018 Jul 11]. Disponível em: https://diretrizes.amb.org.br/_BibliotecaAntiga/terapia_nutricional_no_transplante_de_celula_hematopoietica.pdf
https://diretrizes.amb.org.br/_Bibliotec...
Hyperglycemia is related to an increased risk of infections, metabolic disorders, organ dysfunctions and mortality,251251. Fuji S, Kim SW, Mori S, Kamiya S, Yoshimura K, Yokoyama H, et al. Intensive glucose control after allogeneic hematopoietic stem cell transplantation: a retrospective matched-cohort study. Bone Marrow Transplant. 2009; 44(2):105-11. and is a common effect of the use of calcineurin inhibitors and corticosteroids, requiring glucose monitoring and insulin use. Thus it is necessary to design a glucose control protocol that fits the type of NT used and allows for prevention of hypoglycemia, glycemic variability, and significant hyperglycemia.252252. Hammer MJ, D’Eramo Melkus G, Knobf MT, Casper C, Fletcher J, Cleland CM. Glycemic Status and Infection Risk in Nondiabetic Autologous Hematopoietic Cell Transplantation Recipients. Biol Res Nurs. 2016;18(3):344-50.,253253. Olausson JM, Hammer MJ, Brady V. The impact of hyperglycemia on hematopoietic cell transplantation outcomes: an integrative review. Oncol Nurs Forum. 2014;41(5):E302-12. There are no specific recommendations for the ideal glucose range, and the consensus for inpatients is between 110 and 180mg/dL.
In patients undergoing allogeneic HSCT for hematological tumors, lower rates of fatal GVHD were observed in those receiving parenteral formula with a high content of long-chain fatty acids (80% of non-protein calories versus carbohydrates); however this is a ketogenic nutrition strategy that should only be used for short periods.254254. Muscaritoli M, Conversano L, Torelli GF, Arcese W, Capria S, Cangiano C, et al. Clinical and metabolic effects of different parenteral nutrition regimens in patients undergoing allogeneic bone marrow transplantation. Transplantation. 1998;66(5):610-6. On the other hand, with transplanted patients exposed to the lipid-increasing effects of immunosuppressants, the use of long-chain triglycerides in the PNT composition must be restricted.255255. Baena-Gómez MA, de la Torre-Aguilar MJ, Aguilera-García CM, Olza J, Pérez-Navero JL, Gil-Campos M. Inflammatory Response Using Different Lipid Parenteral Nutrition Formulas in Children After Hematopoietic Stem Cell Transplantation. Nutr Cancer. 2016;68(5):804-10.
Still today, there are few studies on the use of omega-3 lipids during HSCT.255255. Baena-Gómez MA, de la Torre-Aguilar MJ, Aguilera-García CM, Olza J, Pérez-Navero JL, Gil-Campos M. Inflammatory Response Using Different Lipid Parenteral Nutrition Formulas in Children After Hematopoietic Stem Cell Transplantation. Nutr Cancer. 2016;68(5):804-10. The literature recognizes that its parenteral use attenuates inflammatory activity in a short time (days); however, the ideal dose required, the time of administration, and safety-related issues due to changes in platelet function during critical levels of thrombocytopenia, result in the lack of recommendations for its use in the initial phase of the transplant.
Electrolytes, minerals, vitamins and trace elements must be adjusted to individual needs. Some factors, such as GVHD, antibiotics, metabolic stress, immunosuppressants, diarrhea, and vomiting may alter micronutrient requirements. Therefore, serum electrolytes should be assessed daily for potential adjustments.216216. Associação Médica Brasileira (AMB). Conselho Federal de Medicina. Projeto Diretrizes. Terapia. Terapia Nutricional no Transplante de Célula Hematopoiética [Internet]. São Paulo: ABM; 2011 [citado 2018 Jul 11]. Disponível em: https://diretrizes.amb.org.br/_BibliotecaAntiga/terapia_nutricional_no_transplante_de_celula_hematopoietica.pdf
https://diretrizes.amb.org.br/_Bibliotec...
As for micronutrients, there are no definitive studies with recommendations targeted at patients undergoing HSCT, and the context in which the main changes in metabolic demands occur, such as oxidative stress, liver or kidney dysfunction, must be kept in mind.256256. Nannya Y, Shinohara A, Ichikawa M, Kurokawa M. Serial profile of vitamins and trace elements during the acute phase of allogeneic stem cell transplantation. Biol Blood Marrow Transplant. 2014;20(3):430-4. Unfortunately, there are no commercially available products containing trace elements in isolation, which leads to discontinuation of all trace element prescriptions when these organ dysfunctions occur.260260. Agarwal A, Khanna P, Baidya KD, Arora MK. Trace Elements in Critical Illness. J Endocrinol Metab. 2011;1(2):57-63.
261. Parrish CR. Trace Element Supplementation and Monitoring in the Adult Patient on Parenteral Nutrition. Pract Gastroenterol. 2014;4(129):27-38.-262262. Wong T. Parenteral trace elements in children: clinical aspects and dosage recommendations. Curr Opin Clin Nutr Metab Care. 2012;15(6):649-56. Biological deficiency of a micronutrient precedes its clinical manifestation.263263. Singer P, Berger MM, Van den Berghe G, Biolo G, Calder P, Forbes A, et al. ESPEN Guidelines on Parenteral Nutrition: intensive care. Clin Nutr. 2009;28(4):387-400. Cases of Wernicke encephalopathy by acute thiamine deficiency during PNT initiation have been reported after long fasting times, vitamin C depletion due to oxidative stress, and zync deficiency associated with voluminous diarrhea.256256. Nannya Y, Shinohara A, Ichikawa M, Kurokawa M. Serial profile of vitamins and trace elements during the acute phase of allogeneic stem cell transplantation. Biol Blood Marrow Transplant. 2014;20(3):430-4. The daily recommendations for electrolytes, vitamins and trace elements are presented in tables 16 to 18
, respectively.
Recommended daily amounts of electrolytes prescribed for adult patients on parenteral nutrition therapy (ideal weight)
In respect to adjustment of macro and micronutrients when prescribing PNT, we must consider the existence of two systems: the individualized PNT and industrialized PNT, previously described as ready to use (RTU). Although the individualized version gets closer to meeting the requirements of each patient, most centers use the industrialized version, without any harm to patients.264264. Mühlebach S, Franken C, Stanga Z; Working group for developing the guidelines for parenteral nutrition of The German Association for Nutritional Medicine. Practical handling of AIO admixtures - Guidelines on Parenteral Nutrition, Chapter 10. Ger Med Sci. 2009;7:18.
The addition of these PNT components upon installation favors their bioavailability, but compromises safety due to the generation of physical and chemical imbalances in the solution, or increased risk of infection during handling.264264. Mühlebach S, Franken C, Stanga Z; Working group for developing the guidelines for parenteral nutrition of The German Association for Nutritional Medicine. Practical handling of AIO admixtures - Guidelines on Parenteral Nutrition, Chapter 10. Ger Med Sci. 2009;7:18. As regards commercially available products, their composition, in general, also strays away from the recommendations in table 15.
Compounded PNT solutions follow pharmacological principles and are finished by the pharmacist, which favors a more adequate supply of nutrients to transplanted patients, however they must be produced in compliance with safety standards (therapeutically and pharmaceutically appropriate, free from pyrogenic substances, sterile, at the right dose and composition, identified, stored and administered within the defined shelf-life). Nonetheless, when the calculation of nutritional needs fits into a RTU formulation, this option becomes cost-effective, and facilitates the process involved.264264. Mühlebach S, Franken C, Stanga Z; Working group for developing the guidelines for parenteral nutrition of The German Association for Nutritional Medicine. Practical handling of AIO admixtures - Guidelines on Parenteral Nutrition, Chapter 10. Ger Med Sci. 2009;7:18.
The use of IC is greatly relevant for PNT prescription, promoting better understanding of the metabolic demand and individualization of calorie-protein requirements. The combination of specific recommendations for critically-ill patients and large-scale production of new RTU formulations seeks to promote their use even in children.265265. Meyer R, Timmermann M, Schulzke S, Kiss C, Sidler MA, Furlano RI. Developing and implementing all-in-one standard paediatric parenteral nutrition. Nutrients. 2013;5(6):2006-18.
Complications
Parenteral nutrition therapy is related with adverse metabolic effects due to inadequate estimates of nutritional requirements, extent of the catabolic state, and problems with central or peripheral venous access; in addition PNT might reduce response to chemotherapy There is a relative consensus that it is preferable to adopt conservative calorie-protein supply, of about 25kcal/kg/day and up to 1.5g/kg/day, in order not to induce metabolic or fluid overload, which could impair liver or kidney function. The onset of sinusoidal obstruction syndrome is the most serious non-infectious complication of the initial stage of HSCT, resulting from conditioning toxicity in both autologous and allogeneic HSCT, and it usually manifests in the first three weeks of treatment. The clinical context of weight gain, hyperbilirubinemia, followed by oliguria and hepatic encephalopathy, in its more severe form, will require discontinuarion of PNT. Other metabolic complications are related with hyperglycemia, prolonged use of diuretics, and slowed-down appetite recovery. There is a recommendation for addition of regular insulin to the PNT solution, considering a safe ratio of grams of glucose per unit of insulin (usually starting at 20:1 to 10:1). In peripherally administered PNT solutions, the addition of hydrocortisone and heparin prevents inflammatory complications and early withdrawal of the catheter. In case of increased liver enzymes outside the context of sinusoidal obstruction syndrome, it is recommended to change the PNT infusion frequency to 12- to 20-hour cycles, reduce the supply of non-protein calories (10% to 15% less than the previous), check for any micronutrient defiencies (coline, carnitine and taurine), stimulate oral intake, and adjust the ursodeoxycholate dose to 20 to 30mg/kg/day. Consider the use of metronidazole to reduce portal endotoxemia. In cholestatic conditions, the use of copper and manganese must be limited due to neurotoxicity.
The nursing team must supervise PNT administration, including management of the PNT infusion time, adjusting according to other drug infusions scheduled. It is recommendable to administer multivitamin supplements in the first half of the day, and trace elemnts in the second half of the day. Electrolytic deficits must be corrected with concentrated solutions, to minimize fluid overload (in case of central venous access), with observation of the maximum safe limits for infusion of each electrolyte.
Venous access is the most common source of PNT-related complications, particularly those of infectious origin. Other complications include local thrombosis, venous thromboembolism, displaced and fractured catheters, and leaks from mechanical damage.1212. Rzepecki P, Barzal J, Oborska S. Blood and marrow transplantation and nutritional support. Support Care Cancer. 2010;18(Suppl 2):S57-65.
Parenteral nutrition therapy is a procedure inherent to HSCT, and must observe the same quality and safety criteria applicable to the transplant. Changes associated with PNT administration and enteral nutrient deprivation are complex and only partially known, with multiple potential fields of study, which could help increase the safety of this therapy. In the future, further studies are needed to address the impact of sequential or combined use of EN and PNT on clinical outcomes, and complication rates.4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48. Specifically in the case of glutamine and omega-3 fatty acids, there is still a need for studies investigating their effects on mucositis, diarrhea, infections, GVHD, and relapse rates.
However, it is important that these studies separate patients by nutritional status, tumor type (solid versus hematological) and transplant type (autologous versus allogeneic), in order to reduce interpretation biases resulting from the combined analysis of these characteristics in the same study.
Enteral nutrition
Enteral nutrition consists of a set of therapeutic procedures for maintenance or recovery of nutritional status, through enteral nutrition, administered either orally or through a tube accessing the digestive system. American Society Parenteral and Enteral Nutrition and ESPEN have published guidelines on nutritional support for HSCT patients, recommending artificial NT to malnourished patients, or those with impaired food intake or intestinal absorption for a prolonged period of time.4040. August DA, Huhmann MB; American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. A.S.P.E.N. clinical guidelines: nutrition support therapy during adult anticancer treatment and in hematopoietic cell transplantation. JPEN J Parenter Enteral Nutr. 2009;33(5):472-500.,4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48.,266266. Brasil. Ministério da Saúde. Agência Nacional de Vigilância Sanitária. Resolução de Diretoria Colegiada - RDC Nº 63, de 25 de Novembro de 2011. Dispõe sobre os Requisitos de Boas Práticas de Funcionamento para os Serviços de Saúde [Internet]. Rio de Janeiro: ANVISA; 2011[citado 2017 Mar 1]. Disponível em: http://portal.anvisa.gov.br/documents/33880/2568070/rdc0063_25_11_2011.pdf/94c25b42-4a66-4162-ae9b-bf2b71337664
http://portal.anvisa.gov.br/documents/33...
In the context of HSCT, the best NT must be established to prevent weight loss and deterioration of the nutritional status, considering the increased nutritional requirements, gastrointestinal toxicities interfering with food tolerance, and the psychosocial conflicts involved in this procedure.
These factors favor the deterioration of nutritional status, quality of life, physical activity, and lean body mass, for up to 100 days post-HSCT.6363. Barritta de Defranchi RL, Bordalejo A, Cañueto I, Villar A, Navarro E. Evolution of nutritional status in patients with autologous and allogeneic hematopoietic stem cell transplant. Support Care Cancer. 2015;23(5):1341-7.,9393. Ferreira ÉE, Guerra DC, Baluz K, de Resende Furtado W, da Silva Bouzas LF. Nutritional status of patients submitted to transplantation of allogeneic hematopoietic stem cells: a retrospective study. Rev Bras Hematol Hemoter. 2014;36(6):414-9.,267267. Hung YC, Bauer J, Horsley P, Waterhouse M, Bashford J, Isenring E. Changes in nutritional status, body composition, quality of life, and physical activity levels of cancer patients undergoing autologous peripheral blood stem cell transplantation. Support Care Cancer. 2013;21(6):1579-86.,268268. Rieger CT, Wischumerski I, Rust C, Fiegl M. Weight Loss and Decrease of Body Mass Index during Allogeneic Stem Cell Transplantation Are Common Events with Limited Clinical Impact. PLoS One. 2015;10(12):e0145445. Also, patients presenting with weight loss for up to 3 months after allogeneic HSCT are at high risk of mortality not associated to relapse, and worse survival.269269. Fuji S, Mori T, Khattry N, Cheng J, Do YR, Yakushijin K, et al. Severe weight loss in 3 months after allogeneic hematopoietic SCT was associated with an increased risk of subsequent non-relapse mortality. Bone Marrow Transplant. 2015;50(1):100-5. It is clear that nutritional intervention should be part of HSCT therapy, since it contributes to treatment success.
In respect to the NT of choice, PNT is still the first choice in many HSCT centers, due to the easiness of administration through a central venous access, and due to it being considered by healthcare teams, family members and patients as less traumatic and uncomfortable.2424. Baumgartner A, Bargetzi M, Bargetzi A, Zueger N, Medinger M, Passweg J, et al. Nutritional support practices in hematopoietic stem cell transplantation centers: A nationwide comparison. Nutrition. 2017;35:43-50.,270270. Williams-Hooker R, Adams M, Havrilla DA, Leung W, Roach RR, Mosby TT. Caregiver and health care provider preferences of nutritional support in a hematopoietic stem cell transplant unit. Pediatr Blood Cancer. 2015; 62(8):1473-6. However, EN is more physiologic and maintains intestinal tropism, which is essential to avoid bacterial translocation and subsequent systemic infection.242242. Guièze R, Lemal R, Cabrespine A, Hermet E, Tournilhac O, Combal C, et al. Enteral versus parenteral nutritional support in allogeneic haematopoietic stem-cell transplantation. Clin Nutr. 2014;33(3):533-8.
Challenging the usual choice for PNT, studies have shown that it can promote intestinal atrophy and abnormal gastrointestinal immunity, due to decreased cellularity of the GALT (gut-associated lymphatic system) and vascular perfusion, significantly reducing the number of lymphocytes in the lamina propria. There are also important changes in intestinal IgA release and antimicrobial production by Paneth cells (ileal lymph node conglomerates), compromising the epithelial barrier and chemical cleaving, altering the microbiome (dysbiosis) and increasing susceptibility to infections.271271. Pierre JF. Gastrointestinal immune and microbiome changes during parenteral nutrition. Am J Physiol Gastrointest Liver Physiol. 2017;312(3):G246-56.,272272. Sigalet DL, Mackenzie SL, Hameed SM. Enteral nutrition and mucosal immunity: implications for feeding strategies in surgery and trauma. Can J Surg. 2004;47(2):109-16.
The loss of integrity of the intestinal mucosa decreases nutrient absorption, facilitates colonization by pathogenic bacteria, and promotes infections by translocation. In addition, exsudative enteropathy, proteolysis and nitrogen loss, as consequences of this process, contribute to some complications, such as sepsis and GVHD. These can be prevented with the use of EN, which makes it a good choice.7575. Espinoza M, Perelli J, Olmos R, Bertin P, Jara V, Ramírez P. Nutritional assessment as predictor of complications after hematopoietic stem cell transplantation. Rev Bras Hematol Hemoter. 2016;38(1):7-14.,271271. Pierre JF. Gastrointestinal immune and microbiome changes during parenteral nutrition. Am J Physiol Gastrointest Liver Physiol. 2017;312(3):G246-56.
A study by Seguy et al.,273273. Seguy D, Berthon C, Micol JB, Darré S, Dalle JH, Neuville S, et al. Enteral feeding and early outcomes of patients undergoing allogeneic stem cell transplantation following myeloablative conditioning. Transplantation. 2006; 82(6):835-9. demonstrated better 100-day survival in patients on EN, compared to patients on PNT. In another study by the same authors, EN resulted in lower GVHD incidences since it limited intestinal atrophy.4343. Seguy D, Duhamel A, Rejeb MB, Gomez E, Buhl ND, Bruno B, et al. Better outcome of patients undergoing enteral tube feeding after myeloablative conditioning for allogeneic stem cell transplantation. Transplantation. 2012;94(3):287-94.In another comparison between the two treatments, patients on PNT had higher risk of infection.242242. Guièze R, Lemal R, Cabrespine A, Hermet E, Tournilhac O, Combal C, et al. Enteral versus parenteral nutritional support in allogeneic haematopoietic stem-cell transplantation. Clin Nutr. 2014;33(3):533-8.
In line with these findings, several other clinical trials have considered EN just as effective as PNT, however with lower complication rates, in addition to better survival, lower incidence of acute GVHD, and faster neutrophil recovery. Parenteral nutrition therapy was only recommended in cases of severe mucositis or gastrointestinal insufficiency.4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48.,232232. Baumgartner A, Bargetzi A, Zueger N, Bargetzi M, Medinger M, Bounoure L, et al. Revisiting nutritional support for allogeneic hematologic stem cell transplantation-a systematic review. Bone Marrow Transplant. 2017; 52(4):506-13.
Thus, EN must be stimulated during HSCT, challenging preconceptions of patients, family members and the professionals involved. For this purpose, we must encourage the creation of nutritional care protocols246246. Andersen S, Brown T, Kennedy G, Banks M. Implementation of an evidenced based nutrition support pathway for haematopoietic progenitor cell transplant patients. Clin Nutr. 2015;34(3):536-40.that value EN as a feasible and viable option, even if it requires skill development for management of gastrointestinal toxicities arising from this therapeutic modality.
Indication for enteral nutrition therapy
Enteral nutrition is indicated for patients with oral acceptance <60% of nutritional requirements, before HSCT, or in the late post-transplant period, as well as malnourished patients with dietary acceptance (diet + oral NT) <60% of nutritional requirements prior to HSCT or in the late post-transplant period (without the presence of mucositis and thrombocytopenia).246246. Andersen S, Brown T, Kennedy G, Banks M. Implementation of an evidenced based nutrition support pathway for haematopoietic progenitor cell transplant patients. Clin Nutr. 2015;34(3):536-40. Consider the risks and benefits of EN if there is an indication for refusal of oral NT pre-HSCT.4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48.
Patients with a history of inadequate/erratic eating and poor adherence to nutritional guidance, based on observations of the pre-HSCT assessment, must be individually assessed for indication of EN, in an attempt to identify, together with professionals from each specific area, if there are any psychosocial issues interfering with treatment.
Indication may be considered in patients undergoing myeloablative HSCT, due to the potentially higher toxicity, and in elderly patients on high-dose melphalan for autologous HSCT, because of the higher risk of developing severe mucositis, although there is no evidence for formal EN indication in this population.242242. Guièze R, Lemal R, Cabrespine A, Hermet E, Tournilhac O, Combal C, et al. Enteral versus parenteral nutritional support in allogeneic haematopoietic stem-cell transplantation. Clin Nutr. 2014;33(3):533-8.,274274. Zalcberg J, Kerr D, Seymour L, Palmer M; Tomudex International Study Group. Haematological and non-haematological toxicity after 5-fluorouracil and leucovorin in patients with advanced colorectal cancer is significantly associated with gender, increasing age and cycle number. Eur J Cancer. 1998;34(12):1871-5.
The enteral feeding tube must be inserted early on, due to gastrointestinal toxicities.275275. Andersen S, Kennedy G, Banks M. A randomised controlled comparison of enteral versus parenteral nutritional support post allogeneic haematopoietic cell transplantation. Clin Nutr ESPEN. 2015;10(3):e102-6. It is suggested the insertion of the catheter of enteral nutrition until D+3, when gastrointestinal toxicities are not yet pronounced.
The healthcare team, the patient and family members must be approached about the possibility of EN indication at the pre-HSCT assessment, when questions and myths about NT should be clarified, and benefits explained.3030. Bay JO, Dendoncker C, Angeli M, Biot T, Chikhi M, Combal C, et al. Prise en charge nutritionnelle des patients hospitalisés pour allogreffe de CSH: recommandations de la Société francophone de greffe de moelle et de thérapie cellulaire (SFGM-TC). Bull Cancer. 2016;103(11S):S201-6.,242242. Guièze R, Lemal R, Cabrespine A, Hermet E, Tournilhac O, Combal C, et al. Enteral versus parenteral nutritional support in allogeneic haematopoietic stem-cell transplantation. Clin Nutr. 2014;33(3):533-8. The healthcare team must be encouraged to use EN as something beneficial to the treatment and the patient, and understand that NT in HSCT must be focused on preventing7272. Aoyama T, Imataki O, Mori K, Yoshitsugu K, Fukaya M, Okamura I, et al. Nutritional risk in allogeneic stem cell transplantation: rationale for a tailored nutritional pathway. Ann Hematol. 2017;96(4):617-25.,275275. Andersen S, Kennedy G, Banks M. A randomised controlled comparison of enteral versus parenteral nutritional support post allogeneic haematopoietic cell transplantation. Clin Nutr ESPEN. 2015;10(3):e102-6. deterioration of the nutritional status and nutrition-related risks to patients.
In patients able to receive oral nutrition, once EN is initiated, oral feeding may be continued and should be stimulated for faster and adequate weaning from EN. There is an indication for EN in patients weaning from PNT with food acceptance ≥60% of requirements, for 3 consecutive days.246246. Andersen S, Brown T, Kennedy G, Banks M. Implementation of an evidenced based nutrition support pathway for haematopoietic progenitor cell transplant patients. Clin Nutr. 2015;34(3):536-40.
Position of the nasoenteral catheter/tube
Gastric positioning is preferred due to better tolerability to enteral formulations, withstanding osmotic overload, and greater storage capacity when compared to the bowel. If the tube is displaced due to vomiting, it must be reinserted in an enteric position. If the nasoenteral tube has to be reinserted more than twice, PNT should be evaluated for its risks and benefits.
The use of gastrostomies must be reserved for patients with malnutrition and impaired oral intake due to the underlying disease,4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48. and the procedure must be done before the transplant, as part of disease management.
Enteral diet formulations
The use of industrialized EN is suggested due to the lower risk of contamination and the better supply of macro- and micronutrients for HSCT patients. The enteral formulation indicated is one that meets the nutritional requirements of patients with good tolerability. The use of polymeric, high-calorie, high-protein EN is advocated.3030. Bay JO, Dendoncker C, Angeli M, Biot T, Chikhi M, Combal C, et al. Prise en charge nutritionnelle des patients hospitalisés pour allogreffe de CSH: recommandations de la Société francophone de greffe de moelle et de thérapie cellulaire (SFGM-TC). Bull Cancer. 2016;103(11S):S201-6.,4040. August DA, Huhmann MB; American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. A.S.P.E.N. clinical guidelines: nutrition support therapy during adult anticancer treatment and in hematopoietic cell transplantation. JPEN J Parenter Enteral Nutr. 2009;33(5):472-500.,246246. Andersen S, Brown T, Kennedy G, Banks M. Implementation of an evidenced based nutrition support pathway for haematopoietic progenitor cell transplant patients. Clin Nutr. 2015;34(3):536-40. Continuous infusion is recommended, with the use of a pump for better control of the diet. It is fundamental that the team establish the criteria for fulfillment of patients’ nutritional needs in the shortest time possible.276276. Mcclave SA, Martindale RG, Vanek VW, Mccarthy M, Roberts P, Taylor B, Ochoa JB, Napolitano L, Cresci G; A.S.P.E.N. Board of Directors; American College of Critical Care Medicine; Society of Critical Care Medicine. Guidelines for the Provision and Assessment of Nutrition Support Therapy in the Adult Critically Ill Patient: Society of Critical Care Medicine (SCCM) and AmericanSociety for Parenteral and Enteral Nutrition (A.S.P.E.N.). J Parenter Enter Nutr. 2009;33:277-316.
To complement oral nutrition, Bay et al.,3030. Bay JO, Dendoncker C, Angeli M, Biot T, Chikhi M, Combal C, et al. Prise en charge nutritionnelle des patients hospitalisés pour allogreffe de CSH: recommandations de la Société francophone de greffe de moelle et de thérapie cellulaire (SFGM-TC). Bull Cancer. 2016;103(11S):S201-6. recommended nocturnal EN infusion, with a 10-hour infusion duration, starting with 20mL per hour and progressing to 10mL to 20mL per night, aiming to administer 1,000 to 1,500kcal per day.3030. Bay JO, Dendoncker C, Angeli M, Biot T, Chikhi M, Combal C, et al. Prise en charge nutritionnelle des patients hospitalisés pour allogreffe de CSH: recommandations de la Société francophone de greffe de moelle et de thérapie cellulaire (SFGM-TC). Bull Cancer. 2016;103(11S):S201-6.
Enteral nutrition monitoring must occur to ensure patient tolerability of the therapy initiated, metabolic balance, and toxicity detection.3030. Bay JO, Dendoncker C, Angeli M, Biot T, Chikhi M, Combal C, et al. Prise en charge nutritionnelle des patients hospitalisés pour allogreffe de CSH: recommandations de la Société francophone de greffe de moelle et de thérapie cellulaire (SFGM-TC). Bull Cancer. 2016;103(11S):S201-6.,277277. Stroud M, Duncan H, Nightingale J; British Society of Gastroenterology. Guidelines for enteral feeding in adult hospital patients. Gut. 2003;52(90007 Suppl 7):vii1-12. This monitoring, irrespective of the NT adopted (oral nutrition, EN or PNT), must be associated with surveillance of nutritional status, fluid balance, and kidney and liver function throughout the entire HSCT procedure.3030. Bay JO, Dendoncker C, Angeli M, Biot T, Chikhi M, Combal C, et al. Prise en charge nutritionnelle des patients hospitalisés pour allogreffe de CSH: recommandations de la Société francophone de greffe de moelle et de thérapie cellulaire (SFGM-TC). Bull Cancer. 2016;103(11S):S201-6.
Contraindications of enteral nutrition therapy
The main contraindications for maintaining or indicating Enteral nutrition are mucositis, digestive bleeding, neutropenic colitis, incoercible vomiting and diarrhea (grades III and IV) refractory to drug therapy, gastrointestinal tract obstruction, ileus, severe intestinal GVHD (grades III and IV) refractory to drug therapy, and sinus disease. The risk of mucositis, if not already installed, is not a contraindication for EN.4040. August DA, Huhmann MB; American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. A.S.P.E.N. clinical guidelines: nutrition support therapy during adult anticancer treatment and in hematopoietic cell transplantation. JPEN J Parenter Enteral Nutr. 2009;33(5):472-500.,4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48.,9494. van der Meij BS, de Graaf P, Wierdsma NJ, Langius JA, Janssen JJ, van Leeuwen PA, et al. Nutritional support in patients with GVHD of the digestive tract: state of the art. Bone Marrow Transplant. 2013;48(4):474-82.,246246. Andersen S, Brown T, Kennedy G, Banks M. Implementation of an evidenced based nutrition support pathway for haematopoietic progenitor cell transplant patients. Clin Nutr. 2015;34(3):536-40.,275275. Andersen S, Kennedy G, Banks M. A randomised controlled comparison of enteral versus parenteral nutritional support post allogeneic haematopoietic cell transplantation. Clin Nutr ESPEN. 2015;10(3):e102-6.
Table 19 presents the main EN-related complications.
Gastrointestinal toxicities
During HSCT, multiple gastrointestinal toxicities are observed as a result of the action of chemotherapeutic agents during conditioning, total body irradiation, and antimicrobials. There are many conditioning regimens, leading to variable compromise of the gastrointestinal tract, which can be graded using the Common Terminology Criteria for Adverse Events (CTAE), the monitoring of which is extremely important during EN use.281281. National Institute of Cancer (NIH). Common Terminology Criteria for Adverse Events (CTCAE). [Internet]. NIH, 2010 [cited 2018 Jun 12]. Available from: https://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm.
https://ctep.cancer.gov/protocolDevelopm...
In non-Hodgkin lymphoma, a comparison of toxicities from three conditioning regimens showed greater toxicities for the BEAM regimen, with mucosal impairment (greater than grade II), diarrhea and fever; with BEAC (carmustine, etoposide, cytarabine and cyclophosphamide), in turn, patients had more anorexia, vomiting and bleeding; and in CBV (cyclophosphamide, carmustine and etoposide), the incidence of toxicities was lower.282282. Shi Y, Liu P, Zhou S, Yang J, Han X, He X, et al. Comparison of CBV, BEAM and BEAC high-dose chemotherapy followed by autologous hematopoietic stem cell transplantation in non-Hodgkin lymphoma: efficacy and toxicity. Asia Pac J Clin Oncol. 2017;13(5):e423-9. In another investigation, neutropenic colitis was more frequent in patients with non-Hodgkin lymphoma treated with BEAM.283283. Gil L, Poplawski D, Mol A, Nowicki A, Schneider A, Komarnicki M. Neutropenic enterocolitis after high-dose chemotherapy and autologous stem cell transplantation: incidence, risk factors, and outcome. Transpl Infect Dis. 2013;15(1):1-7.
Nausea, vomiting, loss of appetite, diarrhea and mucositis do not contraindicate the use of EN, but these symptoms hinder appropriate nutrition of patients undergoing HSCT, and must be evaluated and closely monitored for better adjustment of NT.
The use of protocols for management of nausea and vomiting must be encouraged in these services. Vomiting causing tube displacement does not contraindicate EN, however it requires post-pyloric placement of the enteral nutrition tube.
Cases of diarrhea must be evaluated for the possibility of infections such as C. difficile, and frequency and volume must be checked daily. Management must be conducted by the medical and nutrition teams. In case of voluminous diarrhea, with no improvement for three days, after ruling out potential infectious causes and gastrointestinal GVHD, diet volume and formulation must be reassessed. It is essential that the patient be informed that the diarrhea is not caused by EN.
However, a study investigating gastrointestinal symptoms during autologous and allogeneic HSCT with different conditioning regimens found that most patients consuming <60% of calorie requirements until engraftment had less diarrhea. This fact may be due to the intake of nutrients that contribute for recovery of the intestinal epithelium, with a positive impact on oral intake.284284. Walrath M, Bacon C, Foley S, Fung HC. Gastrointestinal side effects and adequacy of enteral intake in hematopoietic stem cell transplant patients. Nutr Clin Pract. 2015;30(2):305-10.
One possible therapy for diarrhea is the use of probiotics, which contribute for prevention and treatment of antibiotic-associated diarrhea.285285. Hickson M. Probiotics in the prevention of antibiotic-associated diarrhoea and Clostridium difficile infection. Therap Adv Gastroenterol. 2011;4(3):185-97.,286286. Surendran Nair M, Amalaradjou MA, Venkitanarayanan K. Antivirulence Properties of Probiotics in Combating Microbial Pathogenesis. Adv Appl Microbiol. 2017;98:1-29. However, their safety and efficacy have not yet been established and, therefore, they are not yet indicated via EN in patients undergoing HSCT. Only one strain has been proven safe in children and adolescents during HSCT-associated neutropenia, but its use in adults and the elderly are not yet established, as well as the appropriate doses.228228. Ladas EJ, Bhatia M, Chen L, Sandler E, Petrovic A, Berman DM, et al. The safety and feasibility of probiotics in children and adolescents undergoing hematopoietic cell transplantation. Bone Marrow Transplant. 2016;51(2):262-6.
As for the choice of enteral formulations during episodes of diarrhea, the use of elemental or semielemental formulations is controversial and must be individually assessed, considering these formulations have higher osmolarity, and diarrhea taking place during the course of HSCT is usually osmotic.
Addressing gastrointestinal toxicities and the use of EN in patients submitted to myeloablative HSCT, a recent study with 28 patients in each group showed that 92% of those on EN and 95% of those in PNT has nausea and vomiting. Diarrhea was observed in 44% and 54% of patients on EN and PNT, respectively. Enteral nutrition was discontinued in only 28% of patients due to change in body image, incoercible vomiting, and displacement or obstruction of the enteral nutrition tube, when mucositis was already present and prevented the feeding tube from being reinserted.242242. Guièze R, Lemal R, Cabrespine A, Hermet E, Tournilhac O, Combal C, et al. Enteral versus parenteral nutritional support in allogeneic haematopoietic stem-cell transplantation. Clin Nutr. 2014;33(3):533-8. This observation demonstrated the feasibility of routine use of EN in patients undergoing HSCT.
The American, French and European societies agreed that the first-choice NT, when the gastrointestinal tract is functioning, should be enteral rather than parenteral, due to the potential infectious complications or metabolic implications. Management of gastrointestinal toxicities must be adopted as a daily practice by healthcare teams.3030. Bay JO, Dendoncker C, Angeli M, Biot T, Chikhi M, Combal C, et al. Prise en charge nutritionnelle des patients hospitalisés pour allogreffe de CSH: recommandations de la Société francophone de greffe de moelle et de thérapie cellulaire (SFGM-TC). Bull Cancer. 2016;103(11S):S201-6.,4040. August DA, Huhmann MB; American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. A.S.P.E.N. clinical guidelines: nutrition support therapy during adult anticancer treatment and in hematopoietic cell transplantation. JPEN J Parenter Enteral Nutr. 2009;33(5):472-500.,4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48. Gastrointestinal toxicities, such as mucositis, are not a limitation for the use of EN, provided the tube has been previously inserted.3030. Bay JO, Dendoncker C, Angeli M, Biot T, Chikhi M, Combal C, et al. Prise en charge nutritionnelle des patients hospitalisés pour allogreffe de CSH: recommandations de la Société francophone de greffe de moelle et de thérapie cellulaire (SFGM-TC). Bull Cancer. 2016;103(11S):S201-6.
Infection related to the use of enteral feeding tube and enteral nutrition therapy
Comparatively, EN has lower correlation with infection than PNT.242242. Guièze R, Lemal R, Cabrespine A, Hermet E, Tournilhac O, Combal C, et al. Enteral versus parenteral nutritional support in allogeneic haematopoietic stem-cell transplantation. Clin Nutr. 2014;33(3):533-8. The risk of EN-related infection results from insertion of the tube for enteral feeding, inadequate handling of enteral diets as well as of the enteral feeding tube, incorrect storage of the diet to be offered, and incorrect reuse of tubes and feeding containers for food.
Insertion of the enteral nutrition tube
The three must frequent accesses are nasogastric, nasojejunal, and percutaneous endoscopic gastrostomy. The risk of infection is related with insertion of the tube (nasogastric and nasojejunal), contact with the hand of the healthcare professional guiding the catheter and, in gastrostomy, placement of the tube and infection of the insertion site.
Compounding of enteral diet
The healthcare unit must follow the Good Manufacturing Practices for Enteral Nutrition, as per ANVISA’s Collegiate Board Resolution 63, from 2000.266266. Brasil. Ministério da Saúde. Agência Nacional de Vigilância Sanitária. Resolução de Diretoria Colegiada - RDC Nº 63, de 25 de Novembro de 2011. Dispõe sobre os Requisitos de Boas Práticas de Funcionamento para os Serviços de Saúde [Internet]. Rio de Janeiro: ANVISA; 2011[citado 2017 Mar 1]. Disponível em: http://portal.anvisa.gov.br/documents/33880/2568070/rdc0063_25_11_2011.pdf/94c25b42-4a66-4162-ae9b-bf2b71337664
http://portal.anvisa.gov.br/documents/33...
The dietitian is responsible for supervising preparation of enteral nutrition. Enteral nutrition preparation involves reviewing the diet prescription, compounding, quality control, conservation and transport of enteral nutrition, under responsibility and direct supervision of the dietitian. Enteral nutrition compounding must be carried out using aseptic technique, and following written and validated procedures.
Enteral nutrition must be labeled with clear identification of the patient’s name, composition, and other legal and specific information, to ensure safe use and traceability. Dietitians are responsible for maintaining the quality of EN until its delivery to the professional responsible for administration, and they must guide and train the employees in charge of its transport.
For closed-system EN, the manufacturer’s recommendations regarding conservation and transport must be followed. The use of a closed system is generally preferred, since the product is sterilized, there is no local handling, and the tube is accessed fewer times, which reduces the chance of infection.287287. Fogg L. Home enteral feeding: part 2 current issues in community practice. Br J Community Nurs. 2007;12(7):296-300.
Enteral feeding produced the same organisms that cause food poisoning, with similar symptoms, such as abdominal discomfort and/or bloating, nausea, vomiting and diarrhea, which, in immunocompromised patients, can lead to bacteremia, sepsis and even death.277277. Stroud M, Duncan H, Nightingale J; British Society of Gastroenterology. Guidelines for enteral feeding in adult hospital patients. Gut. 2003;52(90007 Suppl 7):vii1-12.,288288. Ho SS, Tse MM, Boost MV. Effect of an infection control programme on bacterial contamination of enteral feed in nursing homes. J Hosp Infect. 2012;82(1):49-55.,289289. Anderton A. Bacterial contamination of enteral feeds and feeding systems. Clin Nutr. 1993;12:S16-32.
Storage and administration
After EN products are received, the nurse is responsible for their conservation and administration.266266. Brasil. Ministério da Saúde. Agência Nacional de Vigilância Sanitária. Resolução de Diretoria Colegiada - RDC Nº 63, de 25 de Novembro de 2011. Dispõe sobre os Requisitos de Boas Práticas de Funcionamento para os Serviços de Saúde [Internet]. Rio de Janeiro: ANVISA; 2011[citado 2017 Mar 1]. Disponível em: http://portal.anvisa.gov.br/documents/33880/2568070/rdc0063_25_11_2011.pdf/94c25b42-4a66-4162-ae9b-bf2b71337664
http://portal.anvisa.gov.br/documents/33...
To reduce biological risks, the nursing team must:
-
Visually inspect EN before its administration. If any abnormalities are detected in enteral nutrition, it must not be administered, the dietitian in charge must be contacted, and the EN product must be returned.
-
Confirm the position and patency of the tube before initiating enteral nutrition administration.
-
Properly wash hands before proceeding with arrangements for administration of enteral nutrition.
-
Administer enteral nutrition with strict observation of the duration established.
-
If the administration has to be paused, closed-system EN must comply with manufacturer’s instructions regarding its stability time once opened. Compounded EN (open system) must be administered within 4 hours once opened.287287. Fogg L. Home enteral feeding: part 2 current issues in community practice. Br J Community Nurs. 2007;12(7):296-300.
Enteral nutrition is inviolable until the end of administration, and cannot be transferred to any other container. Administration must ensure the safety of the patient and maximize effectiveness in respect to costs, using standardized materials and techniques.
There are multiple ways EN can be contaminated in a hospital, home or clinic, such as:290290. Malhi H. Enteral tube feeding: using good practice to prevent infection. Br J Nurs. 2017;26(1):8-14.
-
Poor hand washing: lack of hand washing can lead to contamination of enteral nutrition, tube, system, and EN container.290290. Malhi H. Enteral tube feeding: using good practice to prevent infection. Br J Nurs. 2017;26(1):8-14.
-
Inadequate cleaning of reusable devices during and after use: the system and the infusion pump must be kept clean while in use, and completely cleaned after feeding is completed.287287. Fogg L. Home enteral feeding: part 2 current issues in community practice. Br J Community Nurs. 2007;12(7):296-300.
-
Inadequate installation of enteral feeding: handling of the catheter and container without the appropriate technique.287287. Fogg L. Home enteral feeding: part 2 current issues in community practice. Br J Community Nurs. 2007;12(7):296-300.
-
Poor tube maintenance: the tube must be regularly flushed with filtered water to reduce the build-up of residues and drug inside, which could cause obstruction and become a source of contamination. Tubes for enteral feeding must be flushed with 20mL to 30mL of water, every 4 to 6 hours, during continuous feeding, during feeding pauses, and before and after drug infusions.
-
Indequate use of the system: all tubes used in enteral feeding are designed for this specific purpose. Ideally, an enteral feeding system should not connect to any other access. It is possible to administer drugs through the enteral feeding tube; however, the pharmacy staff at the hospital must educate on the correct handling and administration of these substances.
Education and training on infection prevention and control are essential.288288. Ho SS, Tse MM, Boost MV. Effect of an infection control programme on bacterial contamination of enteral feed in nursing homes. J Hosp Infect. 2012;82(1):49-55.,291291. Patchell CJ, Anderton A, MacDonald A, George RH, Booth IW. Bacterial contamination of enteral feeds. Arch Dis Child. 1994;70(4):327-30. The expertise of the nurse is vital to prevent bacterial contamination in enteral nutrition.290290. Malhi H. Enteral tube feeding: using good practice to prevent infection. Br J Nurs. 2017;26(1):8-14. Training of the nursing staff on proper handling procedures and enteral nutrition protocols can reduce the level and incidence of bacterial contamination in EN, as well as improve EN offer and impact clinical outcomes of HSCT patients.287287. Fogg L. Home enteral feeding: part 2 current issues in community practice. Br J Community Nurs. 2007;12(7):296-300.,288288. Ho SS, Tse MM, Boost MV. Effect of an infection control programme on bacterial contamination of enteral feed in nursing homes. J Hosp Infect. 2012;82(1):49-55.,291291. Patchell CJ, Anderton A, MacDonald A, George RH, Booth IW. Bacterial contamination of enteral feeds. Arch Dis Child. 1994;70(4):327-30.,292292. Padar M, Uusvel G, Starkopf L, Starkopf J, Reintam Blaser A. Implementation of enteral feeding protocol in an intensive care unit: before-and-after study. World J Crit Care Med. 2017;6(1):56-64.
Reuse of materials
Most hospitals and clinics recommend single use of devices, which must be discarded afterwards. Nurses are responsible to ensure the proper disposal of all devices used. A tamper-proof safety mechanism must be considered. These errors may not sound as serious as drug administration errors, for example, however the effects of such misconducts can be very serious to patients.289289. Anderton A. Bacterial contamination of enteral feeds and feeding systems. Clin Nutr. 1993;12:S16-32.,290290. Malhi H. Enteral tube feeding: using good practice to prevent infection. Br J Nurs. 2017;26(1):8-14. The reuse of devices may affect their integrity and increase the likelihood of bacterial contamination.290290. Malhi H. Enteral tube feeding: using good practice to prevent infection. Br J Nurs. 2017;26(1):8-14.
Another product that must be used only once is the syringe used to flush catheters. If syringes must be reused, they must be washed right after use with soap and water. The plunger should be removed to allow for thorough cleaning. Other cleaning methods approved include dishwasher, soaking in boiling water (3 minutes), cold sterilization solution, and microwave steam sterilizers. Once dry, store in a clean, dry container. Wait to reassemble the syringe right before use. It must be strictly used in only one patient. Syringes are validated for 30 uses. For most patients, the syringe must be changed once a week (equivalent to a maximum of four doses per day). However, if the syringe is used more than four times a day, it must be changed more frequently.
Weaning
Enteral nutrition discontinuation depends on acceptance of oral nutrition, and oral intake must reach at least 60% of nutritional requirements for three consecutive days.246246. Andersen S, Brown T, Kennedy G, Banks M. Implementation of an evidenced based nutrition support pathway for haematopoietic progenitor cell transplant patients. Clin Nutr. 2015;34(3):536-40. After resolution of all gastrointestinal toxicities, such as mucositis, and restablishment of oral feeding, oral intake must be stimulated and oral nutrition therapy must be initiated for adequate supply and consumption of nutrients. When the oral intake of foods and supplements combined is equal to or greater than 60% of individual nutritional requirements, for three consecutive days, EN must be suspended.4040. August DA, Huhmann MB; American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. A.S.P.E.N. clinical guidelines: nutrition support therapy during adult anticancer treatment and in hematopoietic cell transplantation. JPEN J Parenter Enteral Nutr. 2009;33(5):472-500.,4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48.,246246. Andersen S, Brown T, Kennedy G, Banks M. Implementation of an evidenced based nutrition support pathway for haematopoietic progenitor cell transplant patients. Clin Nutr. 2015;34(3):536-40.
The importance of using nutritional therapy indicators
Nutritional therapy indicators enable quality management of the nutritional care provided to patients. These indicators, after analysis of their results, allow for critical assessment of the actions performed, and implementation of corrective measures to improve results and the quality of patient care.293293. Waitzberg DL, editor. Indicadores de qualidade nutricional: aplicação e resultados. São Paulo: Atheneu; 2010.
When these indicators are included in the NT quality assurance program, there are some proven benefits, such as greater efficiency in daily routines, cost reduction, health care practice supported by scientific literature, greater capacity for process analysis, and better clinical and quality of life outcomes for patients.293293. Waitzberg DL, editor. Indicadores de qualidade nutricional: aplicação e resultados. São Paulo: Atheneu; 2010.,294294. Isosaki M, Gandolfo A, Jorge A, Evazian D, Castanheira F, Bittar O. Indicadores de Nutrição Hospitalar. São Paulo: Atheneu; 2015.
The selection of indicators is based on the needs and possibilities of each service.278278. Waitzberg D, Dias MC, Isosaki M. Manual de boas práticas em terapia nutricional enteral e parenteral do HCFMUSP (Hospital das Clínicas da Faculdade de Medicida da Universidade de São Paulo). 2nd ed. São Paulo: Atheneu; 2015. 431 pp. Three indicators presented in tables 20 to 22 , as technical charts, are related with the content of the EN topic.
Figures 1 and 2 present the flow charts for decision-making.3939. Gómez-Candela C, Canales Albendea MA, Palma Milla S, De Paz Arias R, Díaz Gómez J, Rodríguez-Durán D, et al. Intervención nutricional en el paciente oncohematológico. Nutr Hosp. 2012;27(3):669-80.,4747. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48.,119119. Fuji S, Einsele H, Savani BN, Kapp M. Systematic Nutritional Support in Allogeneic Hematopoietic Stem Cell Transplant Recipients. Biol Blood Marrow Transplant. 2015;21(10):1707-13.,242242. Guièze R, Lemal R, Cabrespine A, Hermet E, Tournilhac O, Combal C, et al. Enteral versus parenteral nutritional support in allogeneic haematopoietic stem-cell transplantation. Clin Nutr. 2014;33(3):533-8.,246246. Andersen S, Brown T, Kennedy G, Banks M. Implementation of an evidenced based nutrition support pathway for haematopoietic progenitor cell transplant patients. Clin Nutr. 2015;34(3):536-40.,295295. Raynard B, Nitenberg G, Gory-Delabaere G, Bourhis JH, Bachmann P, Bensadoun RJ, et al.; FNCLCC. Summary of the Standards, Options and Recommendations for nutritional support in patients undergoing bone marrow transplantation (2002). Br J Cancer. 2003;89(S1 Suppl 1):S101-6.
296. This S, Nurse TS, Must I, Advice TPN, Nurse W, Assistant N, et al. Nutritional Protocol for Blood and Bone Marrow Transplantation (BMT). 2015;(May):1-7.-297297. Lemal R, Cabrespine A, Pereira B, Combal C, Ravinet A, Hermet E, et al. Could enteral nutrition improve the outcome of patients with haematological malignancies undergoing allogeneic haematopoietic stem cell transplantation? A study protocol for a randomized controlled trial (the NEPHA study). Trials. 2015;16(1):136.
Flow chart for decision-making regarding admission for hematopoietic stem cell transplantation
SGA: Subjective Global Assessment; PG-SGA: Patient-Generated Subjective Global Assessment; RDA: Recommended Daily Allowance; ONT: oral nutrition therapy; EN: enteral nutrition; GVHD: graft-versus-host disease; PNT: parenteral nutrition therapy.
REFERENCES
-
1Copelan EA. Hematopoietic stem-cell transplantation. N Engl J Med. 2006; 354(17):1813-26.
-
2Majhail NS, Rizzo JD, Lee SJ, Aljurf M, Atsuta Y, Bonfim C, Burns LJ, Chaudhri N, Davies S, Okamoto S, Seber A, Socie G, Szer J, Van Lint MT, Wingard JR, Tichelli A; Center for International Blood and Marrow Transplant Research (CIBMTR); American Society for Blood and Marrow Transplantation (ASBMT); European Group for Blood and Marrow Transplantation (EBMT); Asia-Pacific Blood and Marrow Transplantation Group (APBMT); Bone Marrow Transplant Society of Australia and New Zealand (BMTSANZ); East Mediterranean Blood and Marrow Transplantation Group (EMBMT),; Sociedade Brasileira de Transplante de Medula Ossea (SBTMO). Recommended screening and preventive practices for long-term survivors after hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2012;18(3):348–71.
-
3Associação Brasileira de Transplante de Órgãos (ABTO). Registro Brasileiro de Transplantes (RBT). Dimensionamento dos transplantes no Brasil e em cada estado 2011-2018 [Internet]. São Paulo (SP): ABTO; 2018 [citado 2019 Jun 25]. Disponível em: http://www.abto.org.br/abtov03/Upload/file/RBT/2018/Lv_RBT-2018.pdf
» http://www.abto.org.br/abtov03/Upload/file/RBT/2018/Lv_RBT-2018.pdf -
4Instituto Nacional do Câncer (INCA). Medula Net. Redome [Internet]. Rio de Janeiro: INCA: 2013 [citado 2019 Jun 27]. Disponível em: http://redome.inca.gov.br/o-redome/dados/
» http://redome.inca.gov.br/o-redome/dados/ -
5Passweg JR, Halter J, Bucher C, Gerull S, Heim D, Rovó A, et al. Hematopoietic stem cell transplantation: a review and recommendations for follow-up care for the general practitioner. Swiss Med Wkly. 2012;142:w13696. Review.
-
6Buchholz S, Ganser A. [Hematopoietic stem cell transplantation. Indications, foundations and perspective]. Internist (Berl). 2009;50(5):572-80. Review. German.
-
7Sociedade Brasileira de Transplante de Medula Óssea. Transplantante de células-tronco hematopoéticas. São Paulo: Atheneu; 2010. 1280 pp.
-
8Symons HJ, Fuchs EJ. Hematopoietic SCT from partially HLA-mismatched (HLA-haploidentical) related donors. Bone Marrow Transplant. 2008; 42(6):365-77.
-
9Chaudhry HM, Bruce AJ, Wolf RC, Litzow MR, Hogan WJ, Patnaik MS, et al. The Incidence and Severity of Oral Mucositis among Allogeneic Hematopoietic Stem Cell Transplantation Patients: A Systematic Review. Biol Blood Marrow Transplant. 2016;22(4):605-16.
-
10Rocha V, Porcher R, Fernandes JF, Filion A, Bittencourt H, Silva W Jr, et al. Association of drug metabolism gene polymorphisms with toxicities, graft-versus-host disease and survival after HLA-identical sibling hematopoietic stem cell transplantation for patients with leukemia. Leukemia. 2009;23(3):545-56.
-
11Macris PC, McMillen KK. Nutrition Support of the Hematopoietic Cell Transplant Recipient. In: S J Forman, Negrin RS, Antin JH, Appelbaum FR, editor. Thomas’ Hematopoietic Cell Transplantation: Stem Cell Transplantation. 5th edi. Wiley-Blackwell; 2016. p.1177-85
-
12Rzepecki P, Barzal J, Oborska S. Blood and marrow transplantation and nutritional support. Support Care Cancer. 2010;18(Suppl 2):S57-65.
-
13Urbain P, Ihorst G, Finke J, Bertz H. The impact of pre-transplant body weight on short- and long-term outcomes after allogeneic hematopoietic cell transplantation in adults using different weight classi fi cation tools. Bone Marrow Transplant. 2016;51(1):144-7.
-
14World Health Organization (WHO). Obesity and overweight [Internet]. Geneva: WHO; 2018 [cited 2019 Oct 1]. Available from: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight
» https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight -
15Brasil. Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância de Doenças e Agravos não Transmissíveis e Promoção da Saúde. Vigitel Brasil 2016: Vigilância de Fatores de Risco e Proteção Para Doenças Crônicas Por Inquérito Telefônico: estimativas sobre frequência e distribuição sociodemográfica de fatores de risco e proteção para doenças crônicas nas capitais dos 26 estados brasileiros e no Distrito Federal em 2016 [Internet]. Brasília (DF): Ministério da Saúde; 2017 [citado 2019 Out 1]. Disponível em: http://portalarquivos.saude.gov.br/images/pdf/2017/junho/07/vigitel_2016_jun17.pdf
» http://portalarquivos.saude.gov.br/images/pdf/2017/junho/07/vigitel_2016_jun17.pdf -
16Tarella C, Caracciolo D, Gavarotti P, Argentino C, Zallio F, Corradini P, et al. Overweight as an adverse prognostic factor for non-Hodgkin’s lymphoma patients receiving high-dose chemotherapy and autograft. Bone Marrow Transplant. 2000;26(11):1185-91.
-
17Meloni G, Proia A, Capria S, Romano A, Trapé G, Trisolini SM, et al. Obesity and autologous stem cell transplantation in acute myeloid leukemia. Bone Marrow Transplant. 2001;28(4):365-7.
-
18Fuji S, Kim SW, Yoshimura K, Akiyama H, Okamoto S, Sao H, Takita J, Kobayashi N, Mori S; Japan Marrow Donor Program. Possible association between obesity and posttransplantation complications including infectious diseases and acute graft-versus-host disease. Biol Blood Marrow Transplant. 2009;15(1):73-82.
-
19Vogl DT, Wang T, Pérez WS, Stadtmauer EA, Heitjan DF, Lazarus HM, et al. Effect of obesity on outcomes after autologous hematopoietic stem cell transplantation for multiple myeloma. Biol Blood Marrow Transplant. 2011;17(12):1765-74.
-
20Nakao M, Chihara D, Niimi A, Ueda R, Tanaka H, Morishima Y, et al. Impact of being overweight on outcomes of hematopoietic SCT: a meta-analysis. Bone Marrow Transplant. 2014;49(1):66-72.
-
21Griggs JJ, Mangu PB, Anderson H, Balaban EP, Dignam JJ, Hryniuk WM, Morrison VA, Pini TM, Runowicz CD, Rosner GL, Shayne M, Sparreboom A, Sucheston LE, Lyman GH; American Society of Clinical Oncology. Appropriate chemotherapy dosing for obese adult patients with cancer: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2012;30(13):1553-61. Review.
-
22Bubalo J, Carpenter PA, Majhail N, Perales MA, Marks DI, Shaughnessy P, Pidala J, Leather HL, Wingard J, Savani BN; American Society for Blood and Marrow Transplantation practice guideline committee. Conditioning chemotherapy dose adjustment in obese patients: a review and position statement by the American Society for Blood and Marrow Transplantation practice guideline committee. Biol Blood Marrow Transplant. 2014;20(5):600-16.
-
23Morishita S, Kaida K, Tanaka T, Itani Y, Ikegame K, Okada M, et al. Prevalence of sarcopenia and relevance of body composition, physiological function, fatigue, and health-related quality of life in patients before allogeneic hematopoietic stem cell transplantation. Support Care Cancer. 2012;20(12):3161-8.
-
24Baumgartner A, Bargetzi M, Bargetzi A, Zueger N, Medinger M, Passweg J, et al. Nutritional support practices in hematopoietic stem cell transplantation centers: A nationwide comparison. Nutrition. 2017;35:43-50.
-
25Liu P, Wang B, Yan X, Cai J, Wang Y. Comprehensive evaluation of nutritional status before and after hematopoietic stem cell transplantation in 170 patients with hematological diseases. Chin J Cancer Res. 2016;28(6):626-33.
-
26Urbain P, Birlinger J, Ihorst G, Biesalski HK, Finke J, Bertz H. Body mass index and bioelectrical impedance phase angle as potentially modifiable nutritional markers are independent risk factors for outcome in allogeneic hematopoietic cell transplantation. Ann Hematol. 2013;92(1):111-9.
-
27Fuji S, Mori T, Khattry N, Cheng J, Do YR, Yakushijin K, et al. Severe weight loss in 3 months after allogeneic hematopoietic SCT was associated with an increased risk of subsequent non-relapse mortality. Bone Marrow Transplant. 2015;50(1):100-5.
-
28Deeg HJ, Seidel K, Bruemmer B, Pepea M, Appelbaum F. Impact of patient weight on non-relapse mortality after marrow transplantation. Bone Marrow Transplant. 1995;15(3):461-8.
-
29Le Blanc K, Ringdén O, Remberger M. A low body mass index is correlated with poor survival after allogeneic stem cell transplantation. Haematologica. 2003;88(9):1044-52.
-
30Bay JO, Dendoncker C, Angeli M, Biot T, Chikhi M, Combal C, et al. Prise en charge nutritionnelle des patients hospitalisés pour allogreffe de CSH: recommandations de la Société francophone de greffe de moelle et de thérapie cellulaire (SFGM-TC). Bull Cancer. 2016;103(11S):S201-6.
-
31Gatta A, Verardo A, Bolognesi M. Hypoalbuminemia. Intern Emerg Med. 2012;7(Suppl 3):S193-9. Review.
-
32Sivgin S, Baldane S, Ozenmis T, Keklik M, Kaynar L, Kurnaz F, et al. The impact of pretransplant hypoalbuminemia on survival in patients with leukemia who underwent allogeneic hematopoietic stem cell transplantation (alloHSCT): a nutritional problem? Transplant Proc. 2013;45(9):3371-4.
-
33Ladas EJ, Sacks N, Meacham L, Henry D, Enriquez L, Lowry G, et al. A Multidisciplinary Review of Nutrition Considerations in the Pediatric Oncology Population: A Perspective From Children’s Oncology Group. Nutr Clin Pract. 2005;20(4):377-93. Review.
-
34Hadjibabaie M, Tabeefar H, Alimoghaddam K, Iravani M, Eslami K, Honarmand H, et al. The relationship between body mass index and outcomes in leukemic patients undergoing allogeneic hematopoietic stem cell transplantation. Clin Transplant. 2012;26(1):149-55.
-
35Mattsson J, Westin S, Edlund S, Remberger M. Poor oral nutrition after allogeneic stem cell transplantation correlates significantly with severe graft-versus-host disease. Bone Marrow Transplant. 2006;38(9):629-33.
-
36von Bahr L, Blennow O, Alm J, Björklund A, Malmberg KJ, Mougiakakos D, et al. Increased incidence of chronic GvHD and CMV disease in patients with vitamin D deficiency before allogeneic stem cell transplantation. Bone Marrow Transplant. 2015;50(9):1217-23.
-
37Brasil. Ministério da Saúde. Secretaria de Atenção à Saúde. Departamento de Atenção Especializada e Temática. Manual de terapia nutricional na atenção especializada hospitalar no âmbito do Sistema Único de Saúde – SUS [Internet]. Brasília (DF): Ministério da Saúde; 2016.
-
38Gómez-Candela C, Luengo LM, Cos AI, Martínez-Roque V, Iglesias C, Zamora P, et al. [Subjective global assessment in neoplastic patients]. Nutr Hosp. 2003;18(6):353-7. Spanish.
-
39Gómez-Candela C, Canales Albendea MA, Palma Milla S, De Paz Arias R, Díaz Gómez J, Rodríguez-Durán D, et al. Intervención nutricional en el paciente oncohematológico. Nutr Hosp. 2012;27(3):669-80.
-
40August DA, Huhmann MB; American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. A.S.P.E.N. clinical guidelines: nutrition support therapy during adult anticancer treatment and in hematopoietic cell transplantation. JPEN J Parenter Enteral Nutr. 2009;33(5):472-500.
-
41Ljungman P, Urbano-Ispizua A, Cavazzana-Calvo M, Demirer T, Dini G, Einsele H, Gratwohl A, Madrigal A, Niederwieser D, Passweg J, Rocha V, Saccardi R, Schouten H, Schmitz N, Socie G, Sureda A, Apperley J; European Group for Blood and Marrow. Allogeneic and autologous transplantation for haematological diseases, solid tumours and immune disorders: definitions and current practice in Europe. Bone Marrow Transplant. 2006;37(5):439-49.
-
42Passweg JR, Baldomero H, Bader P, Bonini C, Cesaro S, Dreger P, et al. Hematopoietic stem cell transplantation in Europe 2014: more than 40 000 transplants annually. Bone Marrow Transplant. 2016;51(6):786-92.
-
43Seguy D, Duhamel A, Rejeb MB, Gomez E, Buhl ND, Bruno B, et al. Better outcome of patients undergoing enteral tube feeding after myeloablative conditioning for allogeneic stem cell transplantation. Transplantation. 2012;94(3):287-94.
-
44Schwarz S, Prokopchuk O, Esefeld K, Gröschel S, Bachmann J, Lorenzen S, et al. The clinical picture of cachexia: a mosaic of different parameters (experience of 503 patients). BMC Cancer. 2017;17(1):130.
-
45Fearon K, Strasser F, Anker SD, Bosaeus I, Bruera E, Fainsinger RL, et al. Definition and classification of cancer cachexia: an international consensus. Lancet Oncol. 2011;12(5):489-95.
-
46Bozzetti F, Arends J, Lundholm K, Micklewright A, Zurcher G, Muscaritoli M; ESPEN. ESPEN Guidelines on Parenteral Nutrition: non-surgical oncology. Clin Nutr. 2009;28(4):445-54.
-
47Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, Fearon K, Hütterer E, Isenring E, Kaasa S, Krznaric Z, Laird B, Larsson M, Laviano A, Mühlebach S, Muscaritoli M, Oldervoll L, Ravasco P, Solheim T, Strasser F, de van der Schueren M, Preiser JC. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36(1):11-48.
-
48Khalatbari-Soltani S, Marques-Vidal P. Impact of nutritional risk screening in hospitalized patients on management, outcome and costs: A retrospective study. Clin Nutr. 2016;35(6):1340-6.
-
49Brasil. Ministério da Saúde. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Consenso Nacional de Nutrição Oncológica. 2ª ed [Internet]. Rio de Janeiro (RJ): INCA; 2015. 186 p [citado 2019 Maio 10]. Disponível em: http://www1.inca.gov.br/inca/Arquivos/Consenso_Nutricao_vol_II_2_ed_2016.pdf
» http://www1.inca.gov.br/inca/Arquivos/Consenso_Nutricao_vol_II_2_ed_2016.pdf -
50Consórcio Brasileiro de Acreditação de Sistemas e Serviços de Saúde. Padrões de Acreditação da Joint Commission International para Hospitais. Rio de Janeiro: 2011.
-
51Kondrup J, Allison SP, Elia M, Vellas B, Plauth M; Educational and Clinical Practice Committee, European Society of Parenteral and Enteral Nutrition (ESPEN). ESPEN guidelines for nutrition screening 2002. Clin Nutr. 2003;22(4):415-21.
-
52Polednak AP. Indicators of nutritional screening in hospital records of newly diagnosed Hispanic and Asian-American adult cancer patients in Connecticut. Nutrition. 2008;24(10):1053-6.
-
53Kondrup J, Rasmussen HH, Hamberg O, Stanga Z, Ad Hoc ESPEN Working Group. Nutritional risk screening (NRS 2002): a new method based on an analysis of controlled clinical trials. Clin Nutr. 2003;22(3):321-36.
-
54Liu P, Zhang ZF, Cai JJ, Wang BS, Yan X. NRS2002 assesses nutritional status of leukemia patients undergoing hematopoietic stem cell transplantation. Chin J Cancer Res. 2012;24(4):299-303.
-
55Detsky a S, McLaughlin JR, Baker JP, Johnston N, Whittaker S, Mendelson RA, et al. What is subjective global assessment of nutritional status? JPEN J Parenter Enteral Nutr. 1987;11(1):8-13.
-
56Raslan M, Gonzalez MC, Torrinhas RS, Ravacci GR, Pereira JC, Waitzberg DL. Complementarity of Subjective Global Assessment (SGA) and Nutritional Risk Screening 2002 (NRS 2002) for predicting poor clinical outcomes in hospitalized patients. Clin Nutr. 2011;30(1):49-53.
-
57Amaral TF, Antunes A, Cabral S, Alves P, Kent-Smith L. An evaluation of three nutritional screening tools in a Portuguese oncology centre. J Hum Nutr Diet. 200821(6):575-83.
-
58Ottery FD. Definition of standardized nutritional assessment and interventional pathways in oncology. Nutrition. 1996;12(1 Suppl):S15-9.
-
59Raslan M, Gonzalez MC, Dias MC, Paes-Barbosa FC, Cecconello I, Waitzberg DL. Aplicabilidade dos m??todos de triagem nutricional no paciente hospitalizado. Rev Nutr. 2008;21(5):553-61.
-
60Peres GB, Valim GS, Silva VL, El-kik RM. Comparação entre métodos de Avaliação Subjetiva Global em oncologia Comparison between Subjective Global Assessment methods in oncology. Rev Cienc Salud. 2009;2(1):37-42.
-
61Barbosa-Silva MC, de Barros AJ. Subjective global assessment: Part 2. Review of its adaptations and utilization in different clinical specialties. Arq Gastroenterol. 2002;39(4):248-52. Review.
-
62Ravasco P, Monteiro-Grillo I, Vidal PM, Camilo ME. Nutritional deterioration in cancer: the role of disease and diet. Clin Oncol (R Coll Radiol). 2003; 15(8):443-50.
-
63Barritta de Defranchi RL, Bordalejo A, Cañueto I, Villar A, Navarro E. Evolution of nutritional status in patients with autologous and allogeneic hematopoietic stem cell transplant. Support Care Cancer. 2015;23(5):1341-7.
-
64Nutricional T, Must D, Oncológicos P. Triagem Nutricional por meio do MUST no Paciente Oncológico em Radioterapia. Rev Bras Cancerol. 2016;62(1):27-34.
-
65British Association for Parenteral and Enteral Nutrition (BAPEN). Nutritional screening and care planning with the “MUST”. The “MUST” Explanatory Booklet: a guide to the Malnutrition universal Screening Tool (“MUST”) for Adults [Internet]. Redditch: BAPEN; 2003 [cited 2017 Mar 3]. Available from: https://www.health.gov.il/download/ng/N500-19.pdf
» https://www.health.gov.il/download/ng/N500-19.pdf -
66Gómez Candela C, Olivar Roldán J, García M, Marín M, Madero R, Pérez-Portabella C, et al. Utilidad de un método de cribado de malnutrición en pacientes con cáncer. Nutr Hosp. 2010;25(3):400-5.
-
67World Health Organization (WHO). Physical status: the use and interpretation of anthropometry. Report of a WHO Expert Committee. Geneva: WHO; 1955. [World technical report series; 854].
-
68Monteiro RS, Cunha TR, Santos ME, Mendonça SS. Estimativa de peso, altura e índice de massa corporal em adultos e idosos americanos: revisão. Rev Cienc Salud. 2009;20(4):341-50.
-
69Marín Caro MM, Gómez Candela C, Castillo Rabaneda R, Lourenço Nogueira T, García Huerta M, Loria Kohen V, et al. Evaluación del riesgo nutricional e instauración de soporte nutricional en pacientes oncológicos, según el protocolo del grupo Español de Nutrición y Cáncer. Nutr Hosp. 2008;23(5):458-68.
-
70Brasil. Ministério da Saúde. Vigilância Alimentar e Nutricional. Orientações básicas para a coleta, processamento, análise de dados e informação em serviços de saúde [Internet]. Brasília (DF): Ministério da Saúde; 2004 [citado 2017 Jun 20]. Disponível em: http://189.28.128.100/nutricao/docs/geral/orientacoes_basicas_sisvan.pdf
» http://189.28.128.100/nutricao/docs/geral/orientacoes_basicas_sisvan.pdf -
71Okada N, Hanafusa T, Abe S, Sato C, Nakamura T, Teraoka K, et al. Evaluation of the risk factors associated with high-dose chemotherapy-induced dysgeusia in patients undergoing autologous hematopoietic stem cell transplantation: possible usefulness of cryotherapy in dysgeusia prevention. Support Care Cancer. 2016;24(9):3979-85.
-
72Aoyama T, Imataki O, Mori K, Yoshitsugu K, Fukaya M, Okamura I, et al. Nutritional risk in allogeneic stem cell transplantation: rationale for a tailored nutritional pathway. Ann Hematol. 2017;96(4):617-25.
-
73Yang J, Xue SL, Zhang X, Zhou YN, Qin LQ, Shen YP, et al. Effect of body mass index on overall survival of patients with allogeneic hematopoietic stem cell transplantation. Eur J Clin Nutr. 2017;71(6):750-4.
-
74Jaime-Pérez JC, Colunga-Pedraza PR, Gutiérrez-Gurrola B, Brito-Ramírez AS, Gutiérrez-Aguirre H, Cantú-Rodríguez OG, et al. Obesity is associated with higher overall survival in patients undergoing an outpatient reduced-intensity conditioning hematopoietic stem cell transplant. Blood Cells Mol Dis. 2013;51(1):61-5.
-
75Espinoza M, Perelli J, Olmos R, Bertin P, Jara V, Ramírez P. Nutritional assessment as predictor of complications after hematopoietic stem cell transplantation. Rev Bras Hematol Hemoter. 2016;38(1):7-14.
-
76Humphreys J, de la Maza P, Hirsch S, Barrera G, Gattas V, Bunout D. Muscle strength as a predictor of loss of functional status in hospitalized patients. Nutrition. 2002;18(7-8):616-20.
-
77Norman K, Stobäus N, Gonzalez MC, Schulzke JD, Pirlich M. Hand grip strength: outcome predictor and marker of nutritional status. Clin Nutr. 2011;30(2):135-42.
-
78Schlüssel MM, Dos Anjos LA, Kac G. A dinamometria manual e seu uso na avaliação nutricional. Rev Nutr. 2008;21(2):223-35.
-
79Pastore CA, Oehlschlaeger MH, Gonzalez MC. Impacto do estado nutricional e da força muscular sobre o estado de saude geral e qualidade de vida em pacientes com cancer de trato gastrointestinal e de pulmão. Rev Bras Cancerol. 2013;59(1):43-9.
-
80Barata T, Santos C, Cravo M, Vinhas MD, Morais C, Carolino E, et al. MON-LB013: Handgrip Dynamometry and Patient-Generated Subjective Global Assessment in Patients With Non-Resectable Lung Cancer. Clin Nutr. 2015;34(Suppl 1):S255.
-
81Tanaka S, Imataki O, Kitaoka A, Fujioka S, Hanabusa E, Ohbayashi Y, et al. Clinical impact of sarcopenia and relevance of nutritional intake in patients before and after allogeneic hematopoietic stem cell transplantation. J Cancer Res Clin Oncol. 2017;143(6):1083-92.
-
82Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, Martin FC, Michel JP, Rolland Y, Schneider SM, Topinková E, Vandewoude M, Zamboni M; European Working Group on Sarcopenia in Older People. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010;39(4):412-23.
-
83Wang B, Yan X, Cai J, Wang Y, Liu P. Nutritional assessment with different tools in leukemia patients after hematopoietic stem cell transplantation. Chin J Cancer Res. 2013;25(6):762–9.
-
84Thibault R, Genton L, Pichard C. Body composition: why, when and for who? Clin Nutr. 2012;31(4):435-47.
-
85Prado CM, Baracos VE, McCargar LJ, Reiman T, Mourtzakis M, Tonkin K, et al. Sarcopenia as a determinant of chemotherapy toxicity and time to tumor progression in metastatic breast cancer patients receiving capecitabine treatment. Clin Cancer Res. 2009;15(8):2920-6.
-
86Thoresen L, Frykholm G, Lydersen S, Ulveland H, Baracos V, Prado CM, et al. Nutritional status, cachexia and survival in patients with advanced colorectal carcinoma. Different assessment criteria for nutritional status provide unequal results. Clin Nutr. 2013;32(1):65-72.
-
87Pichard C, Kyle UG, Morabia A, Perrier A, Vermeulen B, Unger P. Nutritional assessment: lean body mass depletion at hospital admission is associated with an increased length of stay. Am J Clin Nutr. 2004;79(4):613-8.
-
88Kyle UG, Chalandon Y, Miralbell R, Karsegard VL, Hans D, Trombetti A, et al. Longitudinal follow-up of body composition in hematopoietic stem cell transplant patients. Bone Marrow Transplant. 2005;35(12):1171-7.
-
89Gleimer M, Li Y, Chang L, Paczesny S, Hanauer DA, Frame DG, et al. Baseline body mass index among children and adults undergoing allogeneic hematopoietic cell transplantation: clinical characteristics and outcomes. Bone Marrow Transplant. 2015;50(3):402-10.
-
90Chughtai K, Song Y, Zhang P, Derstine B, Gatza E, Friedman J, et al. Analytic morphomics: a novel CT imaging approach to quantify adipose tissue and muscle composition in allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2016;51(3):446-50.
-
91Hadjibabaie M, Iravani M, Taghizadeh M, Ataie-Jafari A, Shamshiri AR, Mousavi SA, et al. Evaluation of nutritional status in patients undergoing hematopoietic SCT. Bone Marrow Transplant. 2008;42(7):469-73.
-
92Horsley P, Bauer J, Gallagher B. Poor nutritional status prior to peripheral blood stem cell transplantation is associated with increased length of hospital stay. Bone Marrow Transplant. 2005;35(11):1113-6.
-
93Ferreira ÉE, Guerra DC, Baluz K, de Resende Furtado W, da Silva Bouzas LF. Nutritional status of patients submitted to transplantation of allogeneic hematopoietic stem cells: a retrospective study. Rev Bras Hematol Hemoter. 2014;36(6):414-9.
-
94van der Meij BS, de Graaf P, Wierdsma NJ, Langius JA, Janssen JJ, van Leeuwen PA, et al. Nutritional support in patients with GVHD of the digestive tract: state of the art. Bone Marrow Transplant. 2013;48(4):474-82.
-
95Iestra JA, Fibbe WE, Zwinderman AH, van Staveren WA, Kromhout D. Body weight recovery, eating difficulties and compliance with dietary advice in the first year after stem cell transplantation: a prospective study. Bone Marrow Transplant. 2002;29(5):417-24.
-
96Pereira AZ, Victor ES, Vidal Campregher P, Piovacari SM, Bernardo Barban JS, Pedreira WL Jr, et al. High body mass index among patients undergoing hematopoietic stem cell transplantation: results of a cross-sectional evaluation of nutritional status in a private hospital. Nutr Hosp. 2015;32(6):2874-9.
-
97Sommacal HM, Jochims AM, Schuch I, Silla LM. Comparação de métodos de avaliação nutricional empregados no acompanhamento de pacientes submetidos a transplante de células-tronco hematopoéticas alogênico. Rev Bras Hematol Hemoter. 2010;32(51):50-5.
-
98Inaba H, Yang J, Kaste SC, Hartford CM, Motosue MS, Chemaitilly W, et al. Longitudinal changes in body mass and composition in survivors of childhood hematologic malignancies after allogeneic hematopoietic stem-cell transplantation. J Clin Oncol. 2012;30(32):3991-7.
-
99Pereira AZ, Marchini JS, Carneiro G, Arasaki CH, Zanella MT. Lean and fat mass loss in obese patients before and after Roux-en-Y gastric bypass: a new application for ultrasound technique. Obes Surg. 2012;22(4):597-601.
-
100Ruble K, Hayat M, Stewart KJ, Chen A. Body composition after bone marrow transplantation in childhood. Oncol Nurs Forum. 2012;39(2):186-92.
-
101Mostoufi-Moab S, Ginsberg JP, Bunin N, Zemel BS, Shults J, Thayu M, et al. Body composition abnormalities in long-term survivors of pediatric hematopoietic stem cell transplantation. J Pediatr. 2012;160(1):122-8.
-
102Abe T, Bemben MG, Kondo M, Kawakami Y, Fukunaga T. Comparison of skeletal muscle mass to fat-free mass ratios among different ethnic groups. J Nutr Health Aging. 2012;16(6):534-8.
-
103Tillquist M, Kutsogiannis DJ, Wischmeyer PE, Kummerlen C, Leung R, Stollery D, et al. Bedside ultrasound is a practical and reliable measurement tool for assessing quadriceps muscle layer thickness. JPEN J Parenter Enteral Nutr. 2014;38(7):886-90.
-
104Sanada K, Kearns CF, Midorikawa T, Abe T. Prediction and validation of total and regional skeletal muscle mass by ultrasound in Japanese adults. Eur J Appl Physiol. 2006;96(1):24-31.
-
105Maurits NM, Bollen AE, Windhausen A, De Jager AE, Van Der Hoeven JH. Muscle ultrasound analysis: normal values and differentiation between myopathies and neuropathies. Ultrasound Med Biol. 2003;29(2):215-25.
-
106Berger J, Bunout D, Barrera G, de la Maza MP, Henriquez S, Leiva L, et al. Rectus femoris (RF) ultrasound for the assessment of muscle mass in older people. Arch Gerontol Geriatr. 2015;61(1):33-8.
-
107Watanabe Y, Yamada Y, Fukumoto Y, Ishihara T, Yokoyama K, Yoshida T, et al. Echo intensity obtained from ultrasonography images reflecting muscle strength in elderly men. Clin Interv Aging. 2013;8:993-8.
-
108Fukumoto Y, Ikezoe T, Yamada Y, Tsukagoshi R, Nakamura M, Mori N, et al. Skeletal muscle quality assessed from echo intensity is associated with muscle strength of middle-aged and elderly persons. Eur J Appl Physiol. 2012;112(4):1519-25.
-
109Pillen S, Arts IM, Zwarts MJ. Muscle ultrasound in neuromuscular disorders. Muscle Nerve. 2008;37(6):679-93.
-
110McLean RR, Kiel DP. Developing consensus criteria for sarcopenia: an update. J Bone Miner Res. 2015;30(4):588-92.
-
111Shen W, Punyanitya M, Wang Z, Gallagher D, St-Onge MP, Albu J, et al. Total body skeletal muscle and adipose tissue volumes: estimation from a single abdominal cross-sectional image. J Appl Physiol (1985). 2004;97(6):2333-8.
-
112Kinsey CM, San José Estépar R, van der Velden J, Cole BF, Christiani DC, Washko GR. Lower Pectoralis Muscle Area Is Associated with a Worse Overall Survival in Non-Small Cell Lung Cancer. Cancer Epidemiol Biomarkers Prev. 2017;26(1):38-43.
-
113Camus V, Lanic H, Kraut J, Modzelewski R, Clatot F, Picquenot JM, et al. Prognostic impact of fat tissue loss and cachexia assessed by computed tomography scan in elderly patients with diffuse large B-cell lymphoma treated with immunochemotherapy. Eur J Haematol. 2014;93(1):9-18.
-
114Go SI, Park MJ, Song HN, Kim HG, Kang MH, Lee HR, et al. Prognostic impact of sarcopenia in patients with diffuse large B-cell lymphoma treated with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone. J Cachexia Sarcopenia Muscle. 2016;7(5):567-76.
-
115Antoun S, Lanoy E, Iacovelli R, Albiges-Sauvin L, Loriot Y, Merad-Taoufik M, et al. Skeletal muscle density predicts prognosis in patients with metastatic renal cell carcinoma treated with targeted therapies. Cancer. 2013;119(18):3377-84.
-
116Chu MP, Lieffers J, Ghosh S, Belch AR, Chua NS, Fontaine A, et al. Skeletal muscle radio-density is an independent predictor of response and outcomes in follicular lymphoma treated with chemoimmunotherapy. PLoS One. 2015;10(6):e0127589.
-
117Viana L, Gazal C, Loss S, Azevedo M. Avaliação Nutricional. In: Xavier RM, Dora JM, Barros E, editors. Laboratório na Prática Clínica - Consulta Rápida. 3a edição. Porto Alegre: Artmed; 2016. p. 240-50.
-
118Seres DS, Van Way CW, editors. Nutrition Support for the Critically III. EUA: Humana Press; 2016. 283 p.
-
119Fuji S, Einsele H, Savani BN, Kapp M. Systematic Nutritional Support in Allogeneic Hematopoietic Stem Cell Transplant Recipients. Biol Blood Marrow Transplant. 2015;21(10):1707-13.
-
120Patient-Generated Subjective Global. Assessment (PG-SGA). Physical examination from a Nutritional Standpoint [Internet]. USA: (PG-SGA); 2001 [cited 2017 Jan 1]. Available from: http://pt-global.org/wp-content/uploads/2014/12/Physical_Examination_from_a_Nutritional_Standpoint_Ottery-FD-2001.pdf
» http://pt-global.org/wp-content/uploads/2014/12/Physical_Examination_from_a_Nutritional_Standpoint_Ottery-FD-2001.pdf -
121Fischer M, JeVenn A, Hipskind P. Evaluation of muscle and fat loss as diagnostic criteria for malnutrition. Nutr Clin Pract. 2015;30(2):239-48.
-
122Esper DH. Utilization of nutrition-focused physical assessment in identifying micronutrient deficiencies. Nutr Clin Pract. 2015;30(2):194-202.
-
123Negrin RS, Toljanic JA. Oral toxicity associated with chemotherapy [Internet]. UpToDate; 2017 [cited 2017 Feb 5]. Available from: http://www.uptodate.com/contents/oral-toxicity-associated-with-chemotherapy#H35
» http://www.uptodate.com/contents/oral-toxicity-associated-with-chemotherapy#H35 -
124White JV, Guenter P, Jensen G, Malone A, Schofield M; Academy Malnutrition Work Group; A.S.P.E.N. Malnutrition Task Force; A.S.P.E.N. Board of Directors. Consensus statement: Academy of Nutrition and Dietetics and American Society for Parenteral and Enteral Nutrition: characteristics recommended for the identification and documentation of adult malnutrition (undernutrition). JPEN J Parenter Enteral Nutr. 2012;36(3):275-83.
-
125Morishita S, Kaida K, Tanaka T, Itani Y, Ikegame K, Okada M, et al. Prevalence of sarcopenia and relevance of body composition, physiological function, fatigue, and health-related quality of life in patients before allogeneic hematopoietic stem cell transplantation. Support Care Cancer. 2012;20(12):3161-8.
-
126Kilgour RD, Vigano A, Trutschnigg B, Lucar E, Borod M, Morais JA. Handgrip strength predicts survival and is associated with markers of clinical and functional outcomes in advanced cancer patients. Support Care Cancer. 2013;21(12):3261-70.
-
127Ali S, Garcia JM. Sarcopenia, cachexia and aging: diagnosis, mechanisms and therapeutic options - a mini-review. Gerontology. 2014;60(4):294-305.
-
128Academy of Nutrition and Dietetics. Physical Exam – Parameters Useful in the Assessment of Nutritional Status [Internet]. Meridian ID: Academy of Nutrition and Dietetics; 2013 [cited 2017 Jan 1]. Available from: http://www.idhca.org/wp-content/uploads/2018/07/SCOLLARD_NFPE-Idaho-Physical_Exam_MN.pdf
» http://www.idhca.org/wp-content/uploads/2018/07/SCOLLARD_NFPE-Idaho-Physical_Exam_MN.pdf -
129Epstein JB, Barasch A. Taste disorders in cancer patients: pathogenesis, and approach to assessment and management. Oral Oncol. 2010;46(2):77-81.
-
130Meirelles CS, Diez-Garcia RW. Experiências alimentares e alterações no paladar em pacientes submetidos a transplante de células tronco [Tese]. São Paulo: Universidade de São Paulo; 2016.
-
131Calleja Fernández A, Pintor de la Maza B, Vidal Casariego A, Villar Taibo R, López Gómez JJ, Cano Rodríguez I, et al. Food intake and nutritional status influence outcomes in hospitalized hematology-oncology patients. Nutr Hosp. 2015;31(6):2598-605.
-
132Lindman A, Rasmussen HB, Andersen NF. Food caregivers influence on nutritional intake among admitted haematological cancer patients - a prospective study. Eur J Oncol Nurs. 2013;17(6):827-34.
-
133Sommacal HM, Gazal CH, Jochims AM, Beghetto M, Paz A, Silla LM, et al. Clinical impact of systematic nutritional care in adults submitted to allogeneic hematopoietic stem cell transplantation. Rev Bras Hematol Hemoter. 2012;34(5):334-8.
-
134Spence C. Multisensory flavour perception. Curr Biol. 2013;23(9):R365-9.
-
135Elman I, Soares NS, Pinto ME. Análise da Sensibilidade do Gosto Umami em Crianças com Câncer. Rev Bras Cancerol. 2010;56(2):237-42.
-
136Gamper EM, Zabernigg A, Wintner LM, Giesinger JM, Oberguggenberger A, Kemmler G, et al. Coming to your senses: detecting taste and smell alterations in chemotherapy patients. A systematic review. J Pain Symptom Manage. 2012;44(6):880-95.
-
137Gustafsson IB, Öström Å, Johansson J, Mossberg L. The Five Aspects Meal Model: a tool for developing meal services in restaurants. J Foodserv. 2006;17(2):84-93.
-
138Ackroff K, Sclafani A. Flavor Preferences Conditioned by Dietary Glutamate. Adv Nutr. 2016;7(4):845S-52S.
-
139Jinap S, Hajeb P. Glutamate. Its applications in food and contribution to health. Appetite. 2010;55(1):1-10.
-
140Boltong A, Keast R. The influence of chemotherapy on taste perception and food hedonics: a systematic review. Cancer Treat Rev. 2012;38(2):152-63.
-
141Boer CC, Correa ME, Miranda EC, de Souza CA. Taste disorders and oral evaluation in patients undergoing allogeneic hematopoietic SCT. Bone Marrow Transplant. 2010;45(4):705-11.
-
142Pinho NB, Tartari RF. Terapia Nutricional Convencional versus Terapia Nutricional precoce no perioperatório de cirurgia do cancer colorretal. Rev Bras Cancerol. 2011;57(2):237-50.
-
143Brasil. Ministério da Saúde. Instituto Nacional do Câncer (INCA). Consenso Nacional de Nutrição Oncológica [Internet]. Rio de janeiro: INCA; 2011. Vol. II. [citado 2017 Abr 7]. Disponível em: http://www1.inca.gov.br/inca/Arquivos/comunicacao/consenso_nutricao_2011.pdf
» http://www1.inca.gov.br/inca/Arquivos/comunicacao/consenso_nutricao_2011.pdf -
144Albertini SM. O papel da glutamina na terapia nutricional do transplante de medula óssea. Rev Bras Hematolia e Hematoterapia. 2001;23(1):41-7.
-
145Anders JC, Soller VM, Brandão EM, Vendramini EC, Bertagnolli CL, Giovani PG, et al. Aspectos de enfermagem, nutrição, fisioterapia e serviço social no transplante de medula óssea. Medicina (Ribeirão Preto). 2000;33(4):463-85.
-
146Brasil. Ministério da Saúde. Instituto Nacional do Câncer. Consenso nacional de nutrição oncológica [Internet]. Rio de Janeiro: INCA; 2009. Vol. 53 [citado 2017 Mar 1]. Disponível em: https://www.inca.gov.br/sites/ufu.sti.inca.local/files//media/document//consenso-nacional-de-nutricao-oncologica-2009.pdf
» https://www.inca.gov.br/sites/ufu.sti.inca.local/files//media/document//consenso-nacional-de-nutricao-oncologica-2009.pdf -
147Martin-Salces M, de Paz R, Canales MA, Mesejo A, Hernandez-Navarro F. Nutritional recommendations in hematopoietic stem cell transplantation. Nutrition. 2008;24(7-8):769-75.
-
148Franceschini SC, Priore SE, Euclydes MP. Necessidades e recomendações de nutrientes. In: Cuppari L. Guias de Medicina Ambulatorial e Hospitalar - Nutrição - Nutrição Clínica no Adulto. Barueri (SP): Manole; 2005. p. 3-32.
-
149Sociedade Brasileira de Nutrição Parenteral e Enteral. Associação Brasileira de Nutrologia. Projeto Diretrizes - Associação Médica Brasileira e Conselho Federal de Medicina [Internet]. São Paulo: 2011 [citado 2019 Abr 26]. Disponível em: http://www.saudedireta.com.br/docsupload/1331171426terapia_nutricional_no_transplante_de_celula_hematopoietica.pdf
» http://www.saudedireta.com.br/docsupload/1331171426terapia_nutricional_no_transplante_de_celula_hematopoietica.pdf -
150Muscaritoli M, Grieco G, Capria S, Iori AP, Rossi Fanelli F. Nutritional and metabolic support in patients undergoing bone marrow transplantation. Am J Clin Nutr. 2002;75(2):183-90.
-
151Szeluga DJ, Stuart RK, Brookmeyer R, Utermohlen V, Santos GW. Energy requirements of parenterally fed bone marrow transplant recipients. JPEN J Parenter Enteral Nutr. 1985;9(2):139-43.
-
152Lenssen P. Aker SN. A Resource Manual Fred Hutchinson Cancer Research Center. BMT/PBSCT; 1985.
-
153El-Khalili N, Brodlie K, Kessel D. WebSTer: A web-based surgical training system. Stud Health Technol Inform. 2000;70:69-75.
-
154Tvedt TH, Reikvam H, Bruserud Ø. Nutrition in Allogeneic Stem Cell Transplantion--Clinical Guidelines and Immunobiological Aspects. Curr Pharm Biotechnol. 2016;17(1):92-104. Review.
-
155Atualização da diretriz brasileira de dislipidemias e prevenção da aterosclerose – 2017. Arq Bras Cardiol. 2017;109(2 Supl 1):1-76.
-
156Prasad AS. Zinc in human health: effect of zinc on immune cells. Mol Med. 2008;14(5-6):353-7.
-
157Gibson RS, Hess SY, Hotz C, Brown KH. Indicators of zinc status at the population level: a review of the evidence. Br J Nutr. 2008;99(S3 Suppl 3):S14-23.
-
158Ertekin MV, Koç M, Karslioğlu I, Sezen O. Zinc sulfate in the prevention of radiation-induced oropharyngeal mucositis: a prospective, placebo-controlled, randomized study. Int J Radiat Oncol Biol Phys. 2004;58(1):167-74.
-
159Sgarbieri UR, Fisberg M, Tone LG, Latorre MR. Nutritional assessment and serum zinc and copper concentration among children with acute lymphocytic leukemia: a longitudinal study. Sao Paulo Med J. 2006;124(6):316-20.
-
160Zalina AZ Jr, Suzana S, A Rahman AJ, Noor Aini MY. Assessing the nutritional status of children with leukemia from hospitals in kuala lumpur. Malays J Nutr. 2009;15(1):45-51.
-
161Eby GA. Treatment of acute lymphocytic leukemia using zinc adjuvant with chemotherapy and radiation – a case history and hypothesis. Med Hypotheses. 2005;64(6):1124-6.
-
162Haase H, Rink L. The immune system and the impact of zinc during aging. Immun Ageing. 2009;6(1):9.
-
163Hadjibabaie M, Iravani M, Shamshiri AR, Zaker Z, Mousavi A, Alimoghaddam K, et al. Ghavamzadeh. Serum Zinc Values in Adult Patients Undergoing Bone Marrow Transplantation. IJHOSCR. 2009;3(3):12-8.
-
164Raynard B. Nutritional support of the cancer patient: issues and dilemmas. Crit Rev Oncol Hematol. 2000;34(3):137-68. Review.
-
165Papadopoulou A, Nathavitharana K, Williams MD, Darbyshire PJ, Booth IW. Diagnosis and clinical associations of zinc depletion following bone marrow transplantation. Arch Dis Child. 1996;74(4):328-31.
-
166Beebe K, Olsen J, Sproat L. Impact Of Vitamin D Level Pre and Post Allogeneic Hematopoietic Stem Cell Transplant. Blood. 2013;122(21):4616.
-
167Bandeira F, Griz L, Dreyer P, Eufrazino C, Bandeira C, Freese E. Vitamin D deficiency: A global perspective. Arq Bras Endocrinol Metabol. 2006;50(4):640-6.
-
168Prietl B, Treiber G, Pieber TR, Amrein K. Vitamin D and immune function. Nutrients. 2013;5(7):2502-21.
-
169Leong DP, Kleinig TJ, Kimber TE, Bardy PG. Severe hypermagnesaemia related to laxative use in acute gastrointestinal graft-versus-host disease. Bone Marrow Transplant. 2006;38(1):71-2.
-
170Jaing TH, Hung IJ, Chung HT, Lai CH, Liu WM, Chang KW. Acute hypermagnesemia: a rare complication of antacid administration after bone marrow transplantation. Clin Chim Acta. 2002;326(1-2):201-3.
-
171Sproat L, Bolwell B, Rybicki L, Dean R, Sobecks R, Pohlman B, et al. Vitamin D level after allogeneic hematopoietic stem cell transplant. Biol Blood Marrow Transplant. 2011;17(7):1079-83.
-
172Holick MF, Chen TC. Vitamin D deficiency: a worldwide problem with health consequences. Am J Clin Nutr. 2008;87(4):1080S-6S.
-
173Glotzbecker B, Ho VT, Aldridge J, Kim HT, Horowitz G, Ritz J, et al. Low levels of 25-hydroxyvitamin D before allogeneic hematopoietic SCT correlate with the development of chronic GVHD. Bone Marrow Transplant. 2013; 48(4):593-7.
-
174Hall AC, Juckett MB. The role of vitamin D in hematologic disease and stem cell transplantation. Nutrients. 2013;5(6):2206-21.
-
175Bunce CM, Brown G, Hewison M. Vitamin D and hematopoiesis. Trends Endocrinol Metab. 1997;8(6):245-51.
-
176Mielcarek M, Martin PJ, Leisenring W, Flowers ME, Maloney DG, Sandmaier BM, et al. Graft-versus-host disease after nonmyeloablative versus conventional hematopoietic stem cell transplantation. Blood. 2003; 102(2):756-62.
-
177Philibert D, Desmeules S, Filion A, Poirier M, Agharazii M. Incidence and severity of early electrolyte abnormalities following autologous haematopoietic stem cell transplantation. Nephrol Dial Transplant. 2008;23(1):359-63.
-
178Stabler SP. Clinical practice. Vitamin B12 deficiency. N Engl J Med. 2013; 368(2):149-60.
-
179Maringhini A, Gertz MA, DiMagno EP. Exocrine pancreatic insufficiency after allogeneic bone marrow transplantation. Int J Pancreatol. 1995;17(3):243-7.
-
180Milligan DW, Quick A, Barnard DL. Vitamin B12 absorption after allogeneic bone marrow transplantation. J Clin Pathol. 1987;40(12):1472-4.
-
181Paiva MD, Biase RC, Moraes JJ, Angelo AR, Honorato MC. Complicações orais decorrentes da terapia antineoplásica. Arq Odontol. 2010;46(1):48-55.
-
182Ikemori EH, Oliveira T, Serralheiro IF, Shibuya E. Nutrição em Oncologia. São Paulo: Lemar; 2003.
-
183Garófolo A. Nutrição Clinica, Funcional e Preventiva Aplicada à Oncologia. Rio de Janeiro: Editora Rubio; 2012. p. 97-153.
-
184Guimaraes J. Manual de Oncologia. 3a edição. v.2. São Paulo: BBS; 2008.
-
185Padovani AR, Moraes DP, Mangili LD, De Andrade CR. Protocolo fonoaudiológico de avaliação do risco para disfagia (PARD). Rev Soc Bras Fonoaudiol. 2007;12(3):199-205.
-
186Waitzberg D, Cardenas T. Manual de Terapia Nutricional em Oncologia do ICESP. São Paulo: Atheneu; 2012. p. 253-261.
-
187Spadão F, Gerhardt J, Guimarães T, Dulley F, Almeida Junior JN, Batista MV, et al. Incidence of diarrhea by Clostridium difficile in hematologic patients and hematopoietic stem cell transplantation patients: risk factors for severe forms and death. Rev Inst Med Trop São Paulo. 2014;56(4):325-31.
-
188Henrique C, Silva D, Pereira K. Dietoterapia como estratégia de tratamento da constipação intestinal em (de la constipación intestinal en) cuidados paliativos. Saludi Ciencia. 2015;21(5):505-10.
-
189Andrade V, Sawada NO, Barichello E. Qualidade de vida de pacientes com câncer hematológico em tratamento quimioterápico. Rev Esc Enferm USP. 2013;47(2):355-61.
-
190Gozzo TO, Moysés AM, da Silva PR, de Almeida AM. Náuseas, vômitos e qualidade de vida de mulheres com câncer de mama em tratamento quimioterápico. Rev Gaúcha Enferm. 2013;34(3):110-6.
-
191Panahi Y, Saadat A, Sahebkar A, Hashemian F, Taghikhani M, Abolhasani E. Effect of ginger on acute and delayed chemotherapy-induced nausea and vomiting: a pilot, randomized, open-label clinical trial. Integr Cancer Ther. 2012;11(3):204-11.
-
192Brasil. Ministério da Saúde. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Consenso Nacional de Nutrição Oncológica. 2a edição. Vol. II [Internet]. Rio de Janeiro: INCA; 2016. p.87-92 [citado 2019 Maio 10]. Disponível em: https://www.inca.gov.br/sites/ufu.sti.inca.local/files//media/document//consenso-nutricao-oncologica-vol-ii-2-ed-2016.pdf
» https://www.inca.gov.br/sites/ufu.sti.inca.local/files//media/document//consenso-nutricao-oncologica-vol-ii-2-ed-2016.pdf -
193McCann S, Schwenkglenks M, Bacon P, Einsele H, D’Addio A, Maertens J, Niederwieser D, Rabitsch W, Roosaar A, Ruutu T, Schouten H, Stone R, Vorkurka S, Quinn B, Blijlevens N; EBMT Mucositis Advisory Group. The Prospective Oral Mucositis Audit: relationship of severe oral mucositis with clinical and medical resource use outcomes in patients receiving high-dose melphalan or BEAM-conditioning chemotherapy and autologous SCT. Bone Marrow Transplant. 2009;43(2):141-7.
-
194Niscola P. Mucositis in malignant hematology. Expert Rev Hematol. 2010;3(1):57-65.
-
195Elting LS, Cooksley C, Chambers M, Cantor SB, Manzullo E, Rubenstein EB. The burdens of cancer therapy. Clinical and economic outcomes of chemotherapy-induced mucositis. Cancer. 2003;98(7):1531-9.
-
196Lalla RV, Sonis ST, Peterson DE. Management of oral mucositis in patients who have cancer. Dent Clin North Am. 2008;52(1):61-77.
-
197Fanning SR, Rybicki L, Kalaycio M, Andresen S, Kuczkowski E, Pohlman B, et al. Severe mucositis is associated with reduced survival after autologous stem cell transplantation for lymphoid malignancies. Br J Haematol. 2006;135(3):374-81.
-
198Niscola P, Romani C, Cupelli L, Scaramucci L, Tendas A, Dentamaro T, et al. Mucositis in patients with hematologic malignancies: an overview. Haematologica. 2007;92(2):222-31. Review.
-
199Chen J, Seabrook J, Fulford A, Rajakumar I. Icing oral mucositis: oral cryotherapy in multiple myeloma patients undergoing autologous hematopoietic stem cell transplant. J Oncol Pharm Pract. 2015;23(2):116-20.
-
200Bressan V, Stevanin S, Bianchi M, Aleo G, Bagnasco A, Sasso L. The effects of swallowing disorders, dysgeusia, oral mucositis and xerostomia on nutritional status, oral intake and weight loss in head and neck cancer patients: A systematic review. Cancer Treat Rev. 2016;45:105-19. Review.
-
201Lövenich H, Schütt-Gerowitt H, Keulertz C, Waldschmidt D, Bethe U, Söhngen D, et al. Failure of anti-infective mouth rinses and concomitant antibiotic prophylaxis to decrease oral mucosal colonization in autologous stem cell transplantation. Bone Marrow Transplant. 2005;35(10):997-1001.
-
202Epstein JB, Schubert MM. Managing pain in mucositis. Semin Oncol Nurs. 2004;20(1):30-7.
-
203Cowen D, Tardieu C, Schubert M, Peterson D, Resbeut M, Faucher C, Franquin JC. Low energy Helium-Neon laser in the prevention of oral mucositis in patients undergoing bone marrow transplant: results of a double blind randomized trial. Int J Radiat Oncol Biol Phys. 1997;38(4):697-703.
-
204Eduardo FP, Bezinelli LM, Orsi MC, Rodrigues M, Ribeiro MS, Hamerschlak N, et al. The influence of dental care associated with laser therapy on oral mucositis during allogeneic hematopoietic cell transplant: retrospective study. einstein (Sao Paulo). 2011;9(2):201-6.
-
205Nguyen DT, Shayani S, Palmer J, Dagis A, Forman SJ, Epstein J, et al. Palifermin for prevention of oral mucositis in allogeneic hematopoietic stem cell transplantation: a single-institution retrospective evaluation. Support Care Cancer. 2015;23(11):3141-7.
-
206Keefe DM, Rassias G, O’Neil L, Gibson RJ. Severe mucositis: how can nutrition help? Curr Opin Clin Nutr Metab Care. 2007;10(5):627-31.
-
207Nunes MP. Síndrome da anorexia-caquexia em portadores de câncer. Rev Brasil Cancerol. 2006;52(1):59-77.
-
208Molassiotis A. Anorexia and weight loss in long-term survivors of haematological malignancies. J Clin Nurs. 2003;12(6):925-7.
-
209Porporato PE. Understanding cachexia as a cancer metabolism syndrome. Oncogenesis. 2016;5:e200. Review.
-
210Associação Brasileira de Cuidados Paliativos. Consenso Brasileiro de Caquexia / anorexia. Rev Bras Cuidados Paliativos. 2011;3(3 Suppl 1):1-42.
-
211Rust DM, Simpson JK, Lister J. Nutritional issues in patients with severe neutropenia. Semin Oncol Nurs. 2000;16(2):152-62.
-
212Paton E, Coutinho M, Voltarelli J. Diagnóstico e tratamento de Complicações agudas do Transplante de Células Progenitoras Hematopoéticas. Medicina (Ribeirão Preto). 2000;33(3):264-77.
-
213Malone FR, Leisenring WM, Storer BE, Lawler R, Stern JM, Aker SN, et al. Prolonged anorexia and elevated plasma cytokine levels following myeloablative allogeneic hematopoietic cell transplant. Bone Marrow Transplant. 2007;40(8):765-72.
-
214Albertini SM. O transplante de células-tronco hematopoéticas e o fator nutricional na evolução dos pacientes. Rev Bras Hematol Hemoter. 2010;32(1):8-9.
-
215Fleming DR, Rayens MK, Garrison J. Impact of obesity on allogeneic stem cell transplant patients: a matched case-controlled study. Am J Med. 1997;102(3):265-8.
-
216Associação Médica Brasileira (AMB). Conselho Federal de Medicina. Projeto Diretrizes. Terapia. Terapia Nutricional no Transplante de Célula Hematopoiética [Internet]. São Paulo: ABM; 2011 [citado 2018 Jul 11]. Disponível em: https://diretrizes.amb.org.br/_BibliotecaAntiga/terapia_nutricional_no_transplante_de_celula_hematopoietica.pdf
» https://diretrizes.amb.org.br/_BibliotecaAntiga/terapia_nutricional_no_transplante_de_celula_hematopoietica.pdf -
217Blijlevens NM, Donnelly JP, De Pauw BE. Mucosal barrier injury: biology, pathology, clinical counterparts and consequences of intensive treatment for haematological malignancy: an overview. Bone Marrow Transplant. 2000;25(12):1269-78. Review.
-
218Stiff P. Mucositis associated with stem cell transplantation: current status and innovative approaches to management. Bone Marrow Transplant. 2001;27 Suppl 2:S3-S11. Review.
-
219Albertini S, Ruiz MA. Nutrição em transplante de medula óssea: a importância da terapia nutricional. Arq Ciênc Saúde 2004;11(3):182-8
-
220Navarro WH, Loberiza FR Jr, Bajorunaite R, van Besien K, Vose JM, Lazarus HM, et al. Effect of body mass index on mortality of patients with lymphoma undergoing autologous hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2006;12(5):541-51.
-
221Spexoto MC, Oliveira MR. Consumo alimentar orientado pode prevenir a queda ponderal no pós-transplante de células- tronco hematopoéticas imediato. Revi Bras Nutr Clin. 2013;28(2):91-7.
-
222Costa LJ, Micallef IN, Inwards DJ, Johnston PB, Porrata LF, Litzow MR, et al. Effect of the dose per body weight of conditioning chemotherapy on severity of mucositis and risk of relapse after autologous haematopoietic stem cell transplantation in relapsed diffuse large B cell lymphoma. Br J Haematol. 2008;143(2):268-73.
-
223Paiva M, Biase RC, Moraes JJ, Ângelo AR, Honorato MC. Complicações orais decorrentes da terapia antineoplásica. Arq Odontol. 2010;46(1): 48-55.224.
-
224Fadoni MR, Sicchieri JM. Suplementos nutricionais e nutrição enteral. In: Navarro AM, Japur CC, Sicchieri JMF, Chiarello PG, Diez Garcia RW, editors. Atualiadades em Alimentação e Nutrição Hospitalar. 1st ed. Rio de Janeiro-RJ: editora Atheneu; 2017. p. 215-24.
-
225Halmos EP. Role of FODMAP content in enteral nutrition-associated diarrhea. J Gastroenterol Hepatol. 2013;28 Suppl 4:25-8. Review.
-
226Mochamat CH, Cuhls H, Marinova M, Kaasa S, Stieber C, Conrad R, et al. A systematic review on the role of vitamins, minerals, proteins, and other supplements for the treatment of cachexia in cancer: a European Palliative Care Research Centre cachexia project. J Cachexia Sarcopenia Muscle. 2017;8(1):25-39.
-
227Wang W, Xu S, Ren Z, Jiang J, Zheng S. Gut microbiota and allogeneic transplantation. J Transl Med. 2015;13(1):275.
-
228Ladas EJ, Bhatia M, Chen L, Sandler E, Petrovic A, Berman DM, et al. The safety and feasibility of probiotics in children and adolescents undergoing hematopoietic cell transplantation. Bone Marrow Transplant. 2016;51(2):262-6.
-
229De Paula I, Theophilo P, Cintra B. Tratamento com probióticos na síndrome do intestino irritável. Rev Bras Ciências da Saúde. 2008;19(3):271-81.
-
230Sierpina V, Levine L, McKee J, Campbell C, Lian S, Frenkel M. Nutrition, metabolism, and integrative approaches in cancer survivors. Semin Oncol Nurs. 2015;31(1):42-52.
-
231Galati PC, Brandão EM. Abordagem nutricional no transplante de medula óssea. In: Navarro AM, Japur CC, Sicchieri JM, Chiarello PG, Diez Garcia RW, editors. Atualidades em Alimentação e Nutrição Hospitalar. 1st ed. editora Atheneu; 2017. p. 235-49.
-
232Baumgartner A, Bargetzi A, Zueger N, Bargetzi M, Medinger M, Bounoure L, et al. Revisiting nutritional support for allogeneic hematologic stem cell transplantation-a systematic review. Bone Marrow Transplant. 2017; 52(4):506-13.
-
233Silva Junior EA, editor. Manual de controle higiênico sanitário em serviços de alimentação. São Paulo: Varela; 2007. p. 239-66.
-
234Barbosa AM, Solano ML, Umbuzeiro GA. Pesticides in Drinking Water - The Brazilian Monitoring Program. Front Public Health. 2015;3:246.
-
235Pontara AV, de Oliveira CD, Barbosa AH, Dos Santos RA, Pires RH, Martins CH. Microbiological monitoring of mineral water commercialized in Brazil. Braz J Microbiol. 2011;42(2):554-9.
-
236Agência Nacional de Vigilância Sanitária (ANVISA). Guia de alimentos e vigilância sanitária [Internet]. Brasília (DF): ANVISA; 2004 [citado 2017 Mar 18]. Disponível em: http://portal.anvisa.gov.br/resultado-de-busca?p_p_id=101&p_p_lifecycle=0&p_p_state=maximized&p_p_mode=view&p_p_col_id=column-1&p_p_col_count=1&_101_struts_action=%2Fasset_publisher%2Fview_content&_101_assetEntryId=395967&_101_type=document
» http://portal.anvisa.gov.br/resultado-de-busca?p_p_id=101&p_p_lifecycle=0&p_p_state=maximized&p_p_mode=view&p_p_col_id=column-1&p_p_col_count=1&_101_struts_action=%2Fasset_publisher%2Fview_content&_101_assetEntryId=395967&_101_type=document -
237Fox N, Freifeld AG. The neutropenic diet reviewed: moving toward a safe food handling approach. Oncology (Williston Park). 2012;26(6):572-5.
-
238Akbulut G. Medical Nutritional Therapy in Hematopoietic Stem Cell Transplantation (HSCT). Int J Hematol Oncol. 2013;23(1):55-65.
-
239Zatarain L, Savani BN. The role of nutrition and effects on the cytokine milieu in allogeneic hematopoietic stem cell transplantation. Cell Immunol. 2012;276(1-2):6-9.
-
240Lee JH, Choi SJ, Lee JH, Kim SE, Seol M, Lee YS, et al. Severe metabolic abnormalities after allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2005;35(1):63-9.
-
241Urbain P, Birlinger J, Lambert C, Finke J, Bertz H, Biesalski HK. Longitudinal follow-up of nutritional status and its influencing factors in adults undergoing allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2013;48(3):446-51.
-
242Guièze R, Lemal R, Cabrespine A, Hermet E, Tournilhac O, Combal C, et al. Enteral versus parenteral nutritional support in allogeneic haematopoietic stem-cell transplantation. Clin Nutr. 2014;33(3):533-8.
-
243Azoulay E, Pène F, Darmon M, Lengliné E, Benoit D, Soares M, et al.; Groupe de Recherche Respiratoire en Réanimation Onco-Hématologique (Grrr-OH). Managing critically Ill hematology patients: time to think differently. Blood Rev. 2015;29(6):359-67.
-
244Bayraktar UD, Nates JL. Intensive care outcomes in adult hematopoietic stem cell transplantation patients. World J Clin Oncol. 2016;7(1):98-105.
-
245Bozzetti F. Nutritional support of the oncology patient. Crit Rev Oncol Hematol. 2013;87(2):172-200.
-
246Andersen S, Brown T, Kennedy G, Banks M. Implementation of an evidenced based nutrition support pathway for haematopoietic progenitor cell transplant patients. Clin Nutr. 2015;34(3):536-40.
-
247Muscaritoli M, Grieco G, Capria S, Iori AP, Rossi Fanelli F. Nutritional and metabolic support in patients undergoing bone marrow transplantation. Am J Clin Nutr. 2002;75(2):183-90.
-
248Pittiruti M, Hamilton H, Biffi R, MacFie J, Pertkiewicz M; ESPEN. ESPEN Guidelines on Parenteral Nutrition: central venous catheters (access, care, diagnosis and therapy of complications). Clin Nutr. 2009;28(4):365-77.
-
249Peterson S, Chen Y. Systemic approach to parenteral nutrition in the ICU. Curr Drug Saf. 2010;5(1):33-40.
-
250Crowther M, Avenell A, Culligan DJ. Systematic review and meta-analyses of studies of glutamine supplementation in haematopoietic stem cell transplantation. Bone Marrow Transplant. 2009;44(7):413-25.
-
251Fuji S, Kim SW, Mori S, Kamiya S, Yoshimura K, Yokoyama H, et al. Intensive glucose control after allogeneic hematopoietic stem cell transplantation: a retrospective matched-cohort study. Bone Marrow Transplant. 2009; 44(2):105-11.
-
252Hammer MJ, D’Eramo Melkus G, Knobf MT, Casper C, Fletcher J, Cleland CM. Glycemic Status and Infection Risk in Nondiabetic Autologous Hematopoietic Cell Transplantation Recipients. Biol Res Nurs. 2016;18(3):344-50.
-
253Olausson JM, Hammer MJ, Brady V. The impact of hyperglycemia on hematopoietic cell transplantation outcomes: an integrative review. Oncol Nurs Forum. 2014;41(5):E302-12.
-
254Muscaritoli M, Conversano L, Torelli GF, Arcese W, Capria S, Cangiano C, et al. Clinical and metabolic effects of different parenteral nutrition regimens in patients undergoing allogeneic bone marrow transplantation. Transplantation. 1998;66(5):610-6.
-
255Baena-Gómez MA, de la Torre-Aguilar MJ, Aguilera-García CM, Olza J, Pérez-Navero JL, Gil-Campos M. Inflammatory Response Using Different Lipid Parenteral Nutrition Formulas in Children After Hematopoietic Stem Cell Transplantation. Nutr Cancer. 2016;68(5):804-10.
-
256Nannya Y, Shinohara A, Ichikawa M, Kurokawa M. Serial profile of vitamins and trace elements during the acute phase of allogeneic stem cell transplantation. Biol Blood Marrow Transplant. 2014;20(3):430-4.
-
257Osland EJ, Ali A, Nguyen T, Davis M, Gillanders L. Australasian society for parenteral and enteral nutrition (AuSPEN) adult vitamin guidelines for parenteral nutrition. Asia Pac J Clin Nutr. 2016;25(3):636-50.
-
258Osland EJ, Ali A, Isenring E, Ball P, Davis M, Gillanders L. Australasian Society for Parenteral and Enteral Nutrition guidelines for supplementation of trace elements during parenteral nutrition. Asia Pac J Clin Nutr. 2014;23(4):545-54.
-
259Berger MM, Oudemans-van Straaten HM. Vitamin C supplementation in the critically ill patient. Curr Opin Clin Nutr Metab Care. 2015;18(2):193-201.
-
260Agarwal A, Khanna P, Baidya KD, Arora MK. Trace Elements in Critical Illness. J Endocrinol Metab. 2011;1(2):57-63.
-
261Parrish CR. Trace Element Supplementation and Monitoring in the Adult Patient on Parenteral Nutrition. Pract Gastroenterol. 2014;4(129):27-38.
-
262Wong T. Parenteral trace elements in children: clinical aspects and dosage recommendations. Curr Opin Clin Nutr Metab Care. 2012;15(6):649-56.
-
263Singer P, Berger MM, Van den Berghe G, Biolo G, Calder P, Forbes A, et al. ESPEN Guidelines on Parenteral Nutrition: intensive care. Clin Nutr. 2009;28(4):387-400.
-
264Mühlebach S, Franken C, Stanga Z; Working group for developing the guidelines for parenteral nutrition of The German Association for Nutritional Medicine. Practical handling of AIO admixtures - Guidelines on Parenteral Nutrition, Chapter 10. Ger Med Sci. 2009;7:18.
-
265Meyer R, Timmermann M, Schulzke S, Kiss C, Sidler MA, Furlano RI. Developing and implementing all-in-one standard paediatric parenteral nutrition. Nutrients. 2013;5(6):2006-18.
-
266Brasil. Ministério da Saúde. Agência Nacional de Vigilância Sanitária. Resolução de Diretoria Colegiada - RDC Nº 63, de 25 de Novembro de 2011. Dispõe sobre os Requisitos de Boas Práticas de Funcionamento para os Serviços de Saúde [Internet]. Rio de Janeiro: ANVISA; 2011[citado 2017 Mar 1]. Disponível em: http://portal.anvisa.gov.br/documents/33880/2568070/rdc0063_25_11_2011.pdf/94c25b42-4a66-4162-ae9b-bf2b71337664
» http://portal.anvisa.gov.br/documents/33880/2568070/rdc0063_25_11_2011.pdf/94c25b42-4a66-4162-ae9b-bf2b71337664 -
267Hung YC, Bauer J, Horsley P, Waterhouse M, Bashford J, Isenring E. Changes in nutritional status, body composition, quality of life, and physical activity levels of cancer patients undergoing autologous peripheral blood stem cell transplantation. Support Care Cancer. 2013;21(6):1579-86.
-
268Rieger CT, Wischumerski I, Rust C, Fiegl M. Weight Loss and Decrease of Body Mass Index during Allogeneic Stem Cell Transplantation Are Common Events with Limited Clinical Impact. PLoS One. 2015;10(12):e0145445.
-
269Fuji S, Mori T, Khattry N, Cheng J, Do YR, Yakushijin K, et al. Severe weight loss in 3 months after allogeneic hematopoietic SCT was associated with an increased risk of subsequent non-relapse mortality. Bone Marrow Transplant. 2015;50(1):100-5.
-
270Williams-Hooker R, Adams M, Havrilla DA, Leung W, Roach RR, Mosby TT. Caregiver and health care provider preferences of nutritional support in a hematopoietic stem cell transplant unit. Pediatr Blood Cancer. 2015; 62(8):1473-6.
-
271Pierre JF. Gastrointestinal immune and microbiome changes during parenteral nutrition. Am J Physiol Gastrointest Liver Physiol. 2017;312(3):G246-56.
-
272Sigalet DL, Mackenzie SL, Hameed SM. Enteral nutrition and mucosal immunity: implications for feeding strategies in surgery and trauma. Can J Surg. 2004;47(2):109-16.
-
273Seguy D, Berthon C, Micol JB, Darré S, Dalle JH, Neuville S, et al. Enteral feeding and early outcomes of patients undergoing allogeneic stem cell transplantation following myeloablative conditioning. Transplantation. 2006; 82(6):835-9.
-
274Zalcberg J, Kerr D, Seymour L, Palmer M; Tomudex International Study Group. Haematological and non-haematological toxicity after 5-fluorouracil and leucovorin in patients with advanced colorectal cancer is significantly associated with gender, increasing age and cycle number. Eur J Cancer. 1998;34(12):1871-5.
-
275Andersen S, Kennedy G, Banks M. A randomised controlled comparison of enteral versus parenteral nutritional support post allogeneic haematopoietic cell transplantation. Clin Nutr ESPEN. 2015;10(3):e102-6.
-
276Mcclave SA, Martindale RG, Vanek VW, Mccarthy M, Roberts P, Taylor B, Ochoa JB, Napolitano L, Cresci G; A.S.P.E.N. Board of Directors; American College of Critical Care Medicine; Society of Critical Care Medicine. Guidelines for the Provision and Assessment of Nutrition Support Therapy in the Adult Critically Ill Patient: Society of Critical Care Medicine (SCCM) and AmericanSociety for Parenteral and Enteral Nutrition (A.S.P.E.N.). J Parenter Enter Nutr. 2009;33:277-316.
-
277Stroud M, Duncan H, Nightingale J; British Society of Gastroenterology. Guidelines for enteral feeding in adult hospital patients. Gut. 2003;52(90007 Suppl 7):vii1-12.
-
278Waitzberg D, Dias MC, Isosaki M. Manual de boas práticas em terapia nutricional enteral e parenteral do HCFMUSP (Hospital das Clínicas da Faculdade de Medicida da Universidade de São Paulo). 2nd ed. São Paulo: Atheneu; 2015. 431 pp.
-
279Bankhead R, Boullata J, Brantley S, Corkins M, Guenter P, Krenitsky J, et al. Special Report Enteral Nutrition Practice Recommendations. 2009; 33(2):122-67.
-
280Harris AE, Styczynski J, Bodge M, Mohty M, Savani BN, Ljungman P. Pretransplant vaccinations in allogeneic stem cell transplantation donors and recipients: an often-missed opportunity for immunoprotection? Bone Marrow Transplant. 2015;50(7):899-903. Review.
-
281National Institute of Cancer (NIH). Common Terminology Criteria for Adverse Events (CTCAE). [Internet]. NIH, 2010 [cited 2018 Jun 12]. Available from: https://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm
» https://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm -
282Shi Y, Liu P, Zhou S, Yang J, Han X, He X, et al. Comparison of CBV, BEAM and BEAC high-dose chemotherapy followed by autologous hematopoietic stem cell transplantation in non-Hodgkin lymphoma: efficacy and toxicity. Asia Pac J Clin Oncol. 2017;13(5):e423-9.
-
283Gil L, Poplawski D, Mol A, Nowicki A, Schneider A, Komarnicki M. Neutropenic enterocolitis after high-dose chemotherapy and autologous stem cell transplantation: incidence, risk factors, and outcome. Transpl Infect Dis. 2013;15(1):1-7.
-
284Walrath M, Bacon C, Foley S, Fung HC. Gastrointestinal side effects and adequacy of enteral intake in hematopoietic stem cell transplant patients. Nutr Clin Pract. 2015;30(2):305-10.
-
285Hickson M. Probiotics in the prevention of antibiotic-associated diarrhoea and Clostridium difficile infection. Therap Adv Gastroenterol. 2011;4(3):185-97.
-
286Surendran Nair M, Amalaradjou MA, Venkitanarayanan K. Antivirulence Properties of Probiotics in Combating Microbial Pathogenesis. Adv Appl Microbiol. 2017;98:1-29.
-
287Fogg L. Home enteral feeding: part 2 current issues in community practice. Br J Community Nurs. 2007;12(7):296-300.
-
288Ho SS, Tse MM, Boost MV. Effect of an infection control programme on bacterial contamination of enteral feed in nursing homes. J Hosp Infect. 2012;82(1):49-55.
-
289Anderton A. Bacterial contamination of enteral feeds and feeding systems. Clin Nutr. 1993;12:S16-32.
-
290Malhi H. Enteral tube feeding: using good practice to prevent infection. Br J Nurs. 2017;26(1):8-14.
-
291Patchell CJ, Anderton A, MacDonald A, George RH, Booth IW. Bacterial contamination of enteral feeds. Arch Dis Child. 1994;70(4):327-30.
-
292Padar M, Uusvel G, Starkopf L, Starkopf J, Reintam Blaser A. Implementation of enteral feeding protocol in an intensive care unit: before-and-after study. World J Crit Care Med. 2017;6(1):56-64.
-
293Waitzberg DL, editor. Indicadores de qualidade nutricional: aplicação e resultados. São Paulo: Atheneu; 2010.
-
294Isosaki M, Gandolfo A, Jorge A, Evazian D, Castanheira F, Bittar O. Indicadores de Nutrição Hospitalar. São Paulo: Atheneu; 2015.
-
295Raynard B, Nitenberg G, Gory-Delabaere G, Bourhis JH, Bachmann P, Bensadoun RJ, et al.; FNCLCC. Summary of the Standards, Options and Recommendations for nutritional support in patients undergoing bone marrow transplantation (2002). Br J Cancer. 2003;89(S1 Suppl 1):S101-6.
-
296This S, Nurse TS, Must I, Advice TPN, Nurse W, Assistant N, et al. Nutritional Protocol for Blood and Bone Marrow Transplantation (BMT). 2015;(May):1-7.
-
297Lemal R, Cabrespine A, Pereira B, Combal C, Ravinet A, Hermet E, et al. Could enteral nutrition improve the outcome of patients with haematological malignancies undergoing allogeneic haematopoietic stem cell transplantation? A study protocol for a randomized controlled trial (the NEPHA study). Trials. 2015;16(1):136.
-
Consensus approved by the Sociedade Brasileira de Transplante de Medula Óssea.
Publication Dates
-
Publication in this collection
07 Feb 2020 -
Date of issue
2020
History
-
Received
12 May 2018 -
Accepted
16 May 2019