Abstracts
A new genus and species of glandulocaudine, Lophiobrycon weitzmani, is described based on specimens collected in headwater tributary streams of the rio Grande, upper rio Paraná system, State of Minas Gerais, southeastern Brazil. The inclusion of the new species in the phylogeny of the subfamily Glandulocaudinae proposed by Weitzman & Menezes (1998), reveals a sister group relationship between the new genus and the monophyletic group composed of Glandulocauda and Mimagoniates that currently form the tribe Glandulocaudini. The new species can be readily distinguished from all other species of the tribe by the autapomorphic presence in adult male individuals (with more than 23.9 mm standard length) of an adipose-fin whose base extends for almost the entire distance between the posterior terminus of the base of the dorsal fin and the base of the upper lobe of the caudal fin and averages approximately 25% standard length, along with the presence of globular expansions formed by the lepidotrichia and hypertrophied soft tissue in the middle portions of the first and second pectoral-fin rays. The diagnosis of the tribe Glandulocaudini is modified to accommodate the new genus.
Glandulocaudinae; phylogeny; Mimagoniates; Glandulocauda
Um novo gênero e espécie de glandulocaudine, Lophiobrycon weitzmani, é descrito com base em exemplares coletados em riachos de cabeceira da drenagem do rio Grande, sistema do alto rio Paraná, Estado de Minas Gerais, sudeste do Brasil. A inclusão desta nova espécie na filogenia da subfamília Glandulocaudinae, proposta por Weitzman & Menezes (1998), revela uma relação de grupos irmãos entre o novo gênero e o grupo monofilético formado por Glandulocauda e Mimagoniates, atualmente compondo a tribo Glandulocaudini. A nova espécie pode ser facilmente distinguida das demais da tribo pela posse autapomórfica, nos machos adultos (com mais de 23,9 mm de comprimento padrão), de uma nadadeira adiposa com a base estendendo-se por praticamente a totalidade da distância entre a terminação posterior da base da nadadeira dorsal e base do lobo caudal superior, equivalendo em média a aproximadamente 25% do comprimento padrão, juntamente com a presença de expansões globulares, formadas por lepidotríquios e tecido mole hipertrofiado, na porção média do primeiro e segundo raios da nadadeira peitoral. A diagnose da tribo Glandulocaudini é modificada para acomodar o novo gênero.
Lophiobrycon weitzmani, a new genus and species of glandulocaudine fish (Characiformes: Characidae) from the rio Grande drainage, upper rio Paraná system, southeastern Brazil
Ricardo M. C. Castro; Alexandre C. Ribeiro; Ricardo C. Benine; Alex L. A. Melo
Laboratório de Ictiologia de Ribeirão Preto, Departamento de Biologia, FFCLRP-USP, Av. Bandeirantes 3900, 14040-901 Ribeirão Preto, SP, Brazil. e-mail: rmcastro@ffclrp.usp.br
ABSTRACT
A new genus and species of glandulocaudine, Lophiobrycon weitzmani, is described based on specimens collected in headwater tributary streams of the rio Grande, upper rio Paraná system, State of Minas Gerais, southeastern Brazil. The inclusion of the new species in the phylogeny of the subfamily Glandulocaudinae proposed by Weitzman & Menezes (1998), reveals a sister group relationship between the new genus and the monophyletic group composed of Glandulocauda and Mimagoniates that currently form the tribe Glandulocaudini. The new species can be readily distinguished from all other species of the tribe by the autapomorphic presence in adult male individuals (with more than 23.9 mm standard length) of an adipose-fin whose base extends for almost the entire distance between the posterior terminus of the base of the dorsal fin and the base of the upper lobe of the caudal fin and averages approximately 25% standard length, along with the presence of globular expansions formed by the lepidotrichia and hypertrophied soft tissue in the middle portions of the first and second pectoral-fin rays. The diagnosis of the tribe Glandulocaudini is modified to accommodate the new genus.
Key words: Glandulocaudinae, phylogeny, Mimagoniates, Glandulocauda.
RESUMO
Um novo gênero e espécie de glandulocaudine, Lophiobrycon weitzmani, é descrito com base em exemplares coletados em riachos de cabeceira da drenagem do rio Grande, sistema do alto rio Paraná, Estado de Minas Gerais, sudeste do Brasil. A inclusão desta nova espécie na filogenia da subfamília Glandulocaudinae, proposta por Weitzman & Menezes (1998), revela uma relação de grupos irmãos entre o novo gênero e o grupo monofilético formado por Glandulocauda e Mimagoniates, atualmente compondo a tribo Glandulocaudini. A nova espécie pode ser facilmente distinguida das demais da tribo pela posse autapomórfica, nos machos adultos (com mais de 23,9 mm de comprimento padrão), de uma nadadeira adiposa com a base estendendo-se por praticamente a totalidade da distância entre a terminação posterior da base da nadadeira dorsal e base do lobo caudal superior, equivalendo em média a aproximadamente 25% do comprimento padrão, juntamente com a presença de expansões globulares, formadas por lepidotríquios e tecido mole hipertrofiado, na porção média do primeiro e segundo raios da nadadeira peitoral. A diagnose da tribo Glandulocaudini é modificada para acomodar o novo gênero.
Introduction
The Glandulocaudinae are a group of 20 genera (including Lophiobrycon described herein) and approximately 60 species of small sized characiform fishes (less than 130 mm in standard length) that occur in practically all major South American drainages. All members of the Glandulocaudinae practice insemination among other derived characteristics. In the tribe Glandulocaudini, in which the new species is included, the adult males possess a unique kind of caudal-fin ray pheromone pump that consists of hypertrophied glandular tissue associated with modified caudal-fin scales and the principal fin rays. This structure is apparently used to signal females during courtship (Weitzman & Burns, 1995; Weitzman & Menezes, 1998).
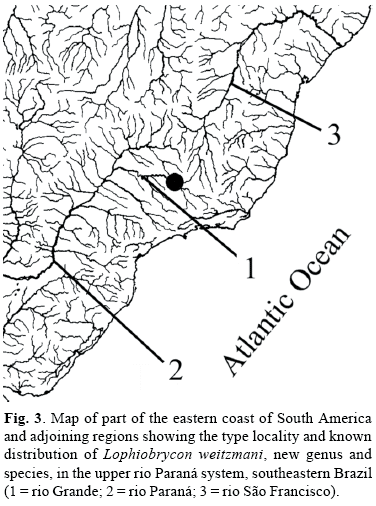
We herein describe Lophiobrycon weitzmani, a new genus and species of glandulocaudine fish (Figs. 1 and 2) and propose a hypothesis of its phylogenetic relationships, based on 84 specimens collected by us in a direct tributary stream of the rio Grande, upper Paraná River System, State of Minas Gerais, in southeastern Brazil (Fig. 3).
Materials and Methods
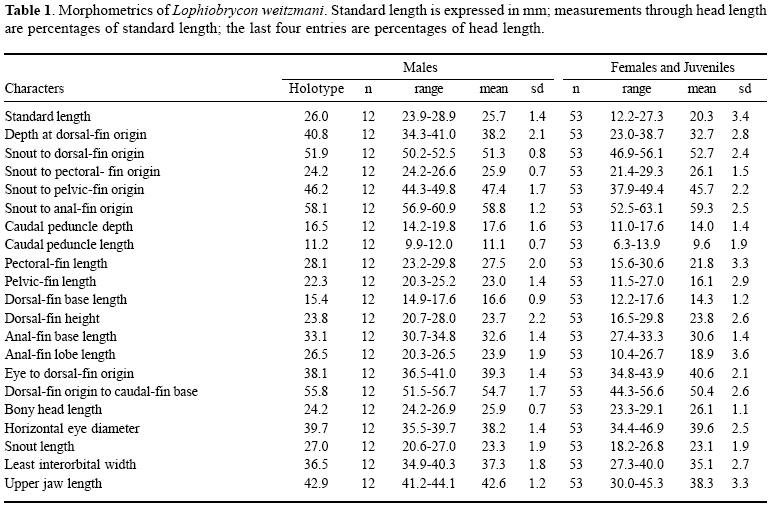
Counts and measurements follow Fink & Weitzman (1974: 1-2) and Menezes & Weitzman (1990: 382-383). For counts, given in the text, the holotype values are listed first, followed by the range, mode (the most frequent value) with the number of specimens having that count indicated in parentheses. Morphometrics are given in Table 1 with standard length (SL) in mm and all measurements other than proportions of the head expressed as a percentage of SL and subunits of the head recorded as percentages of head length (HL). Vertebral counts include the four centra associated with the Weberian apparatus and have the terminal half centrum and associated vertebral elements (PU1 + U1) counted as one element.
Cleared and stained specimens (C&S) were prepared according to Taylor & van Dyke (1985). For the illustrations of the pectoral fin, caudal fin, and associated structures, an adult male (LIRP 4338, 24.7 mm in standard length) was stained in KOH 5% Alizarin Red-S solution. Two adult specimens, a male and a female (LIRP 4338, 29.5 and 25.5 mm in standard length, respectively) had their gonads removed for histological preparation. The gonadal tissues were stained with Hematoxylin/Eosin and mounted in glass slides for examination under a microscope. The glass slides are stored in LIRP collection under the same numbers as used for their respective specimens.
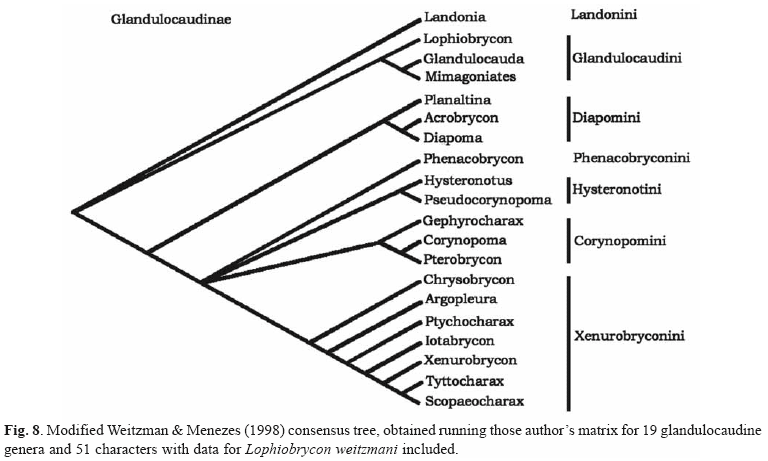
For the analysis of the phylogenetic relationships of the new taxon within the Glandulocaudinae a new line corresponding to L. weitzmani was introduced in the Weitzman & Menezes (1998: 188, Table 3) character state matrix for the 19 described glandulocaudine genera and 51 characters. The modified matrix was rerun, following Weitzman & Menezes (1998), through Hennig86 version 1.5, Farris (1988) and PAUP version 3.1.1, Swofford (1993), with multistate characters running unordered, until obtaining a tree with the same topology as the strict consensus tree of Weitzman & Menezes (1998: 174, Fig. 1). All available Hennig86 and PAUP commands produced the same cladogram topology for the tribe Glandulocaudini.
Stomach contents were analyzed using five specimens (including two C&S individuals) (LIRP 4337, 23.8 to 28.9 mm SL; all stomachs with contents) and utilizing the methods of frequency of occurrence and percent composition proposed by Bowen (1992) and Hynes (1950), respectively. The food items were grouped into broad taxonomic or ecological categories according to their origins. Aquatic insects, microcrustaceans, thecamoebas and algae were considered autochthonous, while terrestrial insects and arachnids were considered allochthonous.
Institutional abbreviations follow Leviton et al. (1985) with the addition of LIRP - Laboratório de Ictiologia de Ribeirão Preto, Faculdade de Filosofia, Ciências e Letras de Ribeirão Preto, Universidade de São Paulo, Ribeirão Preto, Brazil.
Lophiobrycon, new genus
Type species: Lophiobrycon weitzmani by monotypy and original designation.
Diagnosis. The tribe Glandulocaudini was diagnosed by Weitzman & Menezes (1998: 183) on the basis of three characters: 1) the scales of the dorsal caudal-fin lobe with a derived morphology and extending posteriorly to cover all or part of the caudal organ; 2) the presence of a caudal fin-ray pump of a kind unique to the tribe; and 3) the pored lateral line abbreviated and consisting of 3-7 scales with the terminal lateral-line tube absent. The inclusion of Lophiobrycon somewhat modifies this diagnosis because that genus lacks the modified dorsal caudal scales (a plesiomorphic feature) characteristic of members of the other glandulocaudine tribes (Fig. 4).
Lophiobrycon can be further distinguished from all other species of the Glandulocaudini in possessing the following autapomorphies: 1) anterior tip of pelvic bone located between or slightly anterior to ventral tips of anterior two pleural ribs in lateral view, versus ventral to ventral tips of second to third pleural ribs, anterior to ventral tip of first pleural rib, or laying near cleithrum; 2) posteromedial (parietal) branch of supraorbital (frontal) sensory canal reduced in length and never extending into parietal bone, versus sensory canal extending posteriorly into parietal bone; and 3) latero-sensory canal of posttemporal bone present, versus absent.
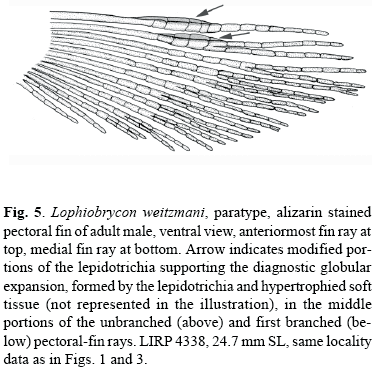
Additional autapomorphic diagnostic characters for the new genus not included in the analysis are: 1) the length of adult male adipose-fin base extends for almost the entire distance between the posterior termination of the base of the dorsal-fin and the base of the upper lobe of the caudal fin, its length averaging approximately 25% of SL (Figs. 1A and 2); and 2) middle portions of the unbranched and first branched pectoral-fin rays with globular expansions formed by the lepidotrichia and hypertrophied soft tissue (Fig. 5).
Etymology. The name Lophiobrycon is from the Greek lophia (= crest, used in reference to the uniquely elongate and crest-shaped adipose-fin of mature males) and brycho (= to gnash the teeth, in reference to the characid genus Brycon, hence a characid fish).
Lophiobrycon weitzmani, new species Figs. 1 and 2
Holotype: LIRP 4366, male, 26.0 mm SL, Brazil, Minas Gerais, rio Grande basin, Município de Delfinópolis, Estância Carmem Sílvia, córrego Bom Jesus (20º 12'10'' S 46º 55'22" W), 18 22 July 1998; A. C. Ribeiro, A. L. A. Melo & R. C. Benine.
Paratypes: LIRP 4337, 8 spms, 20.4-27.3 mm SL (1 male, C&S, 25.8mm SL, 1 female, C&S, 27.0 mm SL, 6 females, 20.4-27.3 mm SL), collected with holotype. LIRP 4338, 31 spms, 9.8-28.9 mm SL (3 males, 23.9-28.9 mm SL, 3 females, 23.8-25.5 mm SL, 24 juveniles, 9.8-25.5 mm SL), collected at type locality, 21 22 August 1998; A. C. Ribeiro & A. L. A. Melo. LIRP 4339, 6 spms C&S, 16.8-25.3 mm SL, collected at type locality, 20 21 July 1999; A. C. Ribeiro, A. L. A. Melo & R. C. Benine. MZUSP 83353, 30 spms, 10.9-25.3 mm SL (2 males, both 25.3 mm SL, 8 females, 18.6-21.4 mm SL, 20 juveniles 10.9-22.6 mm SL), collected at type locality, 20 21July 1999; A. C. Ribeiro, A. L. A. Melo & R. C. Benine. MCP 34194, 8 spms, 22.6-26.6 mm SL (4 males, 25.5-26.6 mm SL, 4 females, 22.3-23.4 mm SL), collected at type locality, 20 21July 1999; A. C. Ribeiro, A. L. A. Melo & R. C. Benine.
Diagnosis. As for the genus.
Description. Table 1 presents morphometrics of holotype and paratypes.
Body compressed, moderately elongate; body deepest approximately in region between verticals through pelvic-fin origin and close to dorsal-fin origin. Dorsal profile of head and predorsal profile of body convex. Body profile slightly elevated at dorsal-fin origin, straight along dorsal-fin base and nearly straight to origin of dorsal procurrent caudal-fin rays. Dorsal-fin origin located nearer snout tip than to caudal-fin base. Ventral profile of body convex from tip of lower jaw approximately to origin of pelvic fin. Ventral profile slightly concave to straight between pelvic-fin origin and caudal-fin base.
Head and snout of moderate size in proportion to body length. Lower jaw protruding and extending anterior to tip of upper jaw. Mouth angled posteroventrally with maxilla extending posteriorly to point slightly anterior of vertical through anterior border of pupil.
Dorsal-fin rays ii,8 (i-ii, mode = ii; 6-9, mode = 8; n = 62). Adipose-fin with shape and size unique to the genus, the length of its base in adult males extending for almost the entire distance between the posterior termination of the base of the dorsal-fin and the base of the upper lobe of the caudal fin, its length averaging approximately 25% of SL. Anal-fin rays iv, 22 (iii-iv, mode = iv; 20-24, mode = 22; n = 62). Anal fin with moderately developed anterior lobe including four unbranched rays and first six branched rays. Anal fin of sexually mature males with hooks on last unbranched ray and anterior five branched rays. Anteriormost rays bearing small hooks and other rays each with single or sometimes a few more highly developed hooks.
Pectoral-fin rays i,12 (i,11-i,14; mode = i,12; n = 62) (very young individuals not included); posterior tips of longest pectoral-fin ray extends posteriorly to origin of pelvic fin and of approximately equal lengths in both sexes. Pectoral-fin rays without hooks; middle portions of the unbranched and first branched pectoral-fin rays of sexually mature males with globular expansions, formed by the lepidotrichia and hypertrophied soft tissue, a condition autapomorphic for genus (Fig. 5). Pelvic-fin rays i,6 (in all specimens, n = 61). Pelvic-fin rays without hooks. Principal caudal-fin ray count 10/9 in all specimens (n = 62); sexually mature males with an anteriorly round soft tissue keel running below ventral procurrent caudal-fin rays (Figs. 1A and 2), feature apparently also present in sexually mature Glandulocauda melanogenys (Weitzman et al., 1988: 398-399, Figs. 13-14).
Scales cycloid; lateral line incomplete; perforated scales 4 (2-6; mode = 4; n = 58). Lateral series scales 35 (29-36; mode = 32; n = 59). Predorsal scales 10 (10-15; mode = 11; n = 60). Scale rows between dorsal-fin and anal-fin origin 12 in all specimens (n = 59). Scale rows around caudal peduncle 15 (11-15; mode = 14; n = 59).
Description of dentition based on eight C&S specimens. Premaxillary teeth in two distinct rows. Outer row teeth 2-3 (mode = 2, n = 8), tricuspid. Inner row teeth 3-6 (mode 5, n = 8), tricuspid. Maxillary teeth 4-6 (mode = 5, n = 8), all conic or anterior 1-3 teeth tricuspid and all 2-5 posterior teeth conic. Dentary teeth 10-13 (mode 12, n = 8) in single row with anterior 3-4 teeth largest and tricuspid and followed posteriorly by 7-9 tricuspid to conic teeth gradually diminishing in size posteriorly. Central cusp of premaxillary, maxillary, and dentary teeth larger than lateral cusps (Fig. 6).
Vertebra 33-35 (mode 34, n = 8 C&S) and branchiostegal rays 4 (n = 8 C&S). Three branchiostegal rays originating from anterior ceratohyal and one from posterior ceratohyal.
Color in alcohol. Ground coloration of preserved specimens tan. Slightly widened longitudinal stripe begins just posterior to opercle and extends to tip of medial caudal-fin rays. Several irregular dark vertical bars on body from just posterior of opercle to vertical line passing through posterior terminus of base of dorsal fin. First two bars more evident and posterior bars becoming thinner posteriorly. Dorsal portion of head dark brown. Dark mid-dorsal stripe extends from posterior tip of occipital bone to anterior region of caudal fin and passes through base of the dorsal fin. Dorsolateral portion of body darker than nearly unpigmented ventrolateral region. Dispersed dark pigment present on all fins. Base of anterior anal-fin rays darkly pigmented.
Color in life. Ground color greenish and dark pigmentation as in specimens in alcohol. Anterior two bar more obvious than others. Dorsal portion of body darker than yellowish-green ventral region. All fins yellowish, especially sexually dimorphic crest-shaped adipose-fin of mature males, with small black dots (Fig. 2).
Sexual dimorphism. Overall body shape of the males differs from that of the females and juveniles (Fig. 1). Mature, dominant and sexually active males (identified during observations of live wild specimens in aquaria) have deeper greater bodies and caudal peduncles and longer pectoral fins relative to mature and unequivocally identifiable females. Overlapping morphometric values in Table 1 between males, females, and juveniles possibly result of difficulties in distinguishing females and immature males.
Mature males with adipose fin much longer and more developed than females and/or juveniles and with middle portions of the unbranched and first branched pectoral-fin rays with globular expansions formed by the lepidotrichia and hypertrophied soft tissue (Fig. 5) that may be used in courtship behavior. Mature males bear numerous hooks on the anal-fin rays that are absent in the females. Sexually mature males with anteriorly rounded soft tissue keel that runs below ventral procurrent caudal-fin rays (Figs. 1A and 2).
Distribution. Known only from type locality (Fig. 3).
Etymology. The specific epithet is named for Stanley H. Weitzman, in recognition of his seminal work on the systematics of Neotropical characiforms, particularly of the characid subfamily Glandulocaudinae.
Discussion
Phylogenetic considerations. An analysis of the anatomical characters of Lophiobrycon within the phylogenetic framework for the Glandulocaudinae proposed by Weitzman & Menezes (1998) reveals that the new genus is undoubtedly a member of the subfamily Glandulocaudinae. As shown in Fig. 7 there occurs an aggregation of spermatozoa (the length of their elongated nuclei between 4.5 and 7.0 µm) that can be seen inside an adult female ovary, undoubtedly indicating the occurrence of insemination in the new species. According to Burns et al. (1995) and Weitzman & Menezes (1998), the possession of insemination and spermatozoa with elongated nuclei are, together with two other reproductive system characters, spermatozoa with elongate cytoplasmic collar and a sperm storage area in the testes, not observed by us, unequivocal synapomorphies for subfamily Glandulocaudinae. Although recent findings (see Burns et al., 2000) have shown that inseminating characids other than the Glandulocaudinae also have elongated sperm nuclei, in absence of published hypothesis on the distribution and homology of these characters, we keep the diagnosis of the Glandulocaudinae as proposed by Weitzman & Menezes (1998).
According to Weitzman & Menezes (1998) the monophyly of the tribe Glandulocaudini is supported by three synapomorphies: 1) the scales of the dorsal caudal-fin lobe are of a derived form and extend posteriorly to cover part or all the caudal organ; 2) the presence of a caudal fin-ray pump of a kind unique to the tribe and; 3) a pored lateral line that is abbreviated and consists of 3 to 7 scales, with the terminal lateral-line tube absent. The genus Glandulocauda although not supported by any synapomorphies (see Menezes & Weitzman, 1990: 381, Fig. 1) was kept as a separate genus, that was defined by the absence, in the adult males, of a caudal-fin ray pump like the derived one found in Mimagoniates, formed by highly modified caudal-fin rays 10-12, associated to the presence of hypertrophied glandular tissue confined to the area immediately around and on the caudal pump region of the gland (see Menezes & Weitzman, 1990: 381, Figs. 2, 5). Glandulocauda on the other hand possesses a plesiomorphic caudal fin-ray pump with only slightly ventrally decurved proximal portions of caudal-fin rays 11 and 12 associated with small bead like glandular tissue structures along their border (see Menezes & Weitzman, 1990: 390, Fig. 6). Furthermore Mimagoniates has no more than one hook on each anterior anal-fin ray that bears hooks, while Glandulocauda has more than one lateral hook on any anal-fin that bears hooks.
In the cladogram topology obtained herein (Fig. 8) (77 steps, CI = 0.85, RI = 0.91) the new genus appears as a member of the tribe Glandulocaudini based on two synapomorphies: 1) the presence of an abbreviated lateral line, with only 3 to 6 scales and without the terminal lateral-line tube; and 2) caudal fin-ray pump utilizing at least caudal-fin rays 10 and 11. In Lophiobrycon the modified scales of the caudal-fin lobe are absent (Fig. 4) but there is an apparent concentration of bead like hypertrophied glandular tissue along the borders of the proximal portions of caudal-fin rays 11 and 12, that are very slightly ventrally decurved in their distal half. This most plesiomorphic state of the glandulocaudin caudal organ present in Lophiobrycon is responsible for its position as the sister group of all other members of the tribe Glandulocaudini.
The inclusion of Lophiobrycon in the tribe Glandulocaudini, however, demands a reformulation of the tribal diagnosis. Among the three characters considered by Weitzman & Menezes, (1998) as putative synapomorphies of the tribe, the state 1 of character number four: "scales of the dorsal caudal-fin lobe derived and extending posteriorly to cover organized caudal glandular tissue" should be reconsidered. In the reformulation the phylogenetic diagnosis of the tribe Glandulocaudini to include Lophiobrycon, only the characters 9 (divided in a two state character): "caudal-fin ray pump utilizing at least principal caudal-fin rays 10 and 11: 1, plesiomorphic with only decurved presence of caudal-fin rays 11 and 12; 2, caudal-fin rays 11-13 and sometimes 14 expanded to form a pump", and 16: "pored lateral line abbreviated and consisting of 3 to 7 scales with the terminal lateral-line tube absent" are now applicable.
Biogeography. Faunistic evidence for area interrelationships between the upper rio Tietê, rio Paraíba do Sul and coastal drainages of eastern Brazil was previously reported by
Langeani (1989), Armbruster (1998) and Malabarba (1998). Weitzman et al. (1988), based on the phylogenetic patterns of the tribe Glandulocaudini, suggested that the diversification of Mimagoniates and other freshwater fishes in coastal eastern Brazil was under the influence of complex vicariant and dispersion events related to sea level changes during glacial and interglacial periods of Pleistocene. However, little has been discussed about the initial diversification of the tribe in the Paraná-Paraguay drainages in which the basal Glandulocaudinae taxa Glandulocauda and Lophiobrycon occur.
An area cladogram based on the phylogeny of the Glandulocaudini locates the area corresponding to the middle and upper portions of the rio Grande (where Lophiobrycon occurs) as the sister area of the upper rio Tietê, headwaters of the rio Iguaçú, (where the two known species of Glandulocauda, G. melanogenys and G. melanopleura occur, respectively), plus the coastal drainages of eastern Brazil (where most species of Mimagoniates occur).
According to Ab'Sáber (1998), the area corresponding to the southeastern portion of the Brazilian Crystalline Shield (where the headwaters of rio São Francisco, Serra da Mantiqueira eastern portion drainage, upper rio Paranaíba, upper rio Tietê, upper rio Grande, rio Paraíba do Sul drainage in the State of Minas Gerais and the upper rio Doce in the State of Espírito Santo are located) is a complex area due to tectonic activity since the beginning of the Tertiary of a rocky megadome. That process, in association with a complex fault system, is the main cause of several headwater stream captures, such as that between the rio Tietê and rio Paraíba do Sul basins. The consequences of such complex events for the biogeography of the aquatic biota remains obscure, but its effect on the distribution patterns is undoubtedly pronounced and is clearly reflected at least in the fish fauna of that region (see also Langeani, 1993).
Our results are consistent with the fact noted above, suggesting a biogeographic scenario with the southeastern portion of the Brazilian Crystalline Shield, including the northeastern portion of the upper rio Paraná and adjacent regions, sharing a common evolutionary history. This area, undoubtedly has an important role as a key for understanding the cladogenectic events responsible for the initial diversification of clades occurring in the drainages of upper rio Paraná, upper rio São Francisco, upper rio Paraíba do Sul and other coastal drainages of eastern Brazil. All the above clearly points to the possibly hybrid nature of the upper rio Paraná system's biogeographic history.
Ecological notes. Specimens were collected during winter months in 18-22 July 1998, 21-22 August 1998, and 20-21 July 1999, in two spatially close points in the drainage basin of the córrego Bom Jesus, a first order tributary of the rio Grande drainage basin, in the upper Paraná River system, State of Minas Gerais, southeastern Brazil (Figs. 3 and 9). Five specimens, including the holotype, were captured in the main channel of the córrego Bom Jesus, close to the margins, in areas of slower current under riparian vegetation over a bottom composed of rocks, pebbles and sand, with submerged litter only on the bottom of the larger and deeper pools. At the time of collection (approximately at 09:00 am) the temperature and pH of the water were 19ºC and 7.0, respectively. The remaining specimens were collected (Fig. 9) in a flooded/marshy area draining directly into the córrego Bom Jesus, with little to very little current over a muddy bottom with a large amount of decomposing litter. At the time of collection (approximately 10:30 am) the temperature and pH of the water were 21ºC and 6.9, respectively.
Geomorphologically the collection locality belongs to the "Patamares da Canastra" unit, that includes the whole area south of the Canastra Range (just outside the Parque Nacional da Serra da Canastra), with altitudes usually between 750 and 1000 m. In the collection area the altitude is approximately 900 m and the general climate belongs to the tropical type, with an annual mean temperature varying between 18 and 20ºC and with a mean annual rainfall precipitation larger than 1,630 mm. December and January are the rainiest months, with mean rainfall precipitation over 100 mm and May, June, July and August are the driest months, with mean rainfall precipitation below 40 mm. The general vegetation of the area belongs to the so-called "Província Central", with savanna-like "cerrado" prevalent inserted with gallery forests and open high altitude fields on the range tops (cf. Radan Brasil, 1983; Nimer, 1989; Rizzini, 1992).
The new species seems to feed mostly on aquatic and terrestrial arthropods. The analysis of the stomach contents of five C&S specimens revealed a modest prevalence of autochthonous items (52.5 %) over the allochthonous items (47.5 %). The most frequent autochthonous items were Chironomidae larvae (100%), Ephemeroptera nymphs (60%), Odonata nymphs (60%) and thecamoebas (60%), followed by Trichoptera larvae (40%), copepods (40%), Cladocera (40%), algae (40%), and aquatic Coleoptera (20%). The most frequent allochthonous items were adult Diptera (100%), Formicidae (60%), Hymenoptera (40%), Thysanoptera (40%), Coleoptera (40%), followed by Hemiptera, adult Trichoptera, Coleoptera larvae, and Aranae (all with 20% frequency of occurrence).
Acknowledgments
Some of the specimens of Lophiobrycon weitzmani that served as the basis for this description were collected during one of the LIRP initial exploratory expeditions of the upper rio Paraná stream and headwaters fish diversity BIOTA/FAPESP Thematic Project (FAPESP Grant number: 98/05072-8) (see www.biota.org.br). The stomach extractions and stomachs contents identification for L. weitzmani diet analysis were performed by Lilian Casatti (IBILCE-UNESP) and Humberto F. Mendes (FFCLRP-USP). Antônio J. Colusso and Wagner F. dos Santos (both FFCLRP-USP) helped with the histological preparations. Elza T. S. Hojo (FFCLRP-USP) permitted the use of her optical equipment to produce the ovary photomicrograph. This paper was greatly improved by the suggestions and criticisms of Richard P. Vari (NMNH, Smithsonian Institution) and Flávio A. Bockmann (LIRP, FFCLRP-USP). We thank all of the above for their assistance. Partial funding for field and laboratory studies was provided by FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo) within the "BIOTA/FAPESP The Virtual Biodiversity Institute Program" (www.biota.org.br/) through the Thematic Project "Fish diversity of the headwaters and streams of the upper Paraná River system in the State of São Paulo, Brazil" (FAPESP grant No. 98/05072-8) and by PRONEX Project "Conhecimento, conservação e utilização racional da diversidade da fauna de peixes do Brasil" (FINEP/CNPq grant No. 661058/1997-2). The first author is a Conselho Nacional de Desenvolvimento Científico e Tecnológico do Brasil researcher (grant No. 301309/91-4).
Literature Cited
Received February 26, 2003
Accepted July 8, 2003
- Ab'Sáber, N. A. 1998. Megageomorfologia do território brasileiro. Pp. 71-106. In: Cunha, S. B. da & Guerra, A. J. T. (Eds.). Geomorfologia do Brasil. Rio de Janeiro, Bertand Brasil, 388p.
- Armbruster, J. W. 1998. Phylogenetic relationships of the suckermouth armored catfishes of the Rhinelepis group (Loricariidae: Hypostominae). Copeia, 1998 (3): 620-636.
- Bowen, S. H. 1992. Quantitative description of the diet. Pp.325-336. In: Nielsen, L. A. & D. L. Johnson (Eds.). Fisheries Techniques. Blacksburg, American Fisheries Society, 468 p.
- Burns, J. R., S. H. Weitzman, H. J. Grier & N. A. Menezes. 1995. Internal fertilization, testis and sperm morphology in glandulocaudine fishes (Teleostei: Characidae: Glandulocaudinae). Journal of Morphology, 224: 131-145.
- Burns, J. R., S. H. Weitzman, L. R. Malabarba & D. Meisner. 2000. Sperm modifications in inseminating ostariophysan fishes, with new documentation of inseminating species. P. 255. In: Norberg, B, O. S. Kjesbu, G. L. Taranger, E. Andersson & S. O. Stefasson (Eds). Proceedings of the 6th International Symposium on the Reproductive Physiology of Fish, July 4-9, 1999. Norway, Institute of Marine Research and University of Bergen.
- Farris, J. S. 1998. "Henning86, version 1.5", program and documentation, New York, Farris, Port Jefferson Station.
- Fink, W. & S. H. Weitzman. 1974. The so-called cheirodontin fishes of Central America with descriptions of two new species (Pisces: Characidae). Smithsonian Contributions to Zoology, 172: 1-46.
- Hynes, H. B. N. 1950. The food of fresh-water sticklebacks (Gasterosteus aculeatus and Pygosteus pungitius), with a review of methods used in studies of food fishes. Journal of Animal Ecology, 19: 36-57.
- Langeani, F. 1989. Ictiofauna do alto curso do rio Tietê (SP): taxonomia. Unpublished M.SC. Dissertation, Universidade de São Paulo, São Paulo. 231p.
- Leviton, A. E., R. H. Gibbs, E. Heal & C. E. Dawson. 1985. Standards in herpetology and ichthyology: part. I. Standard symbolic codes for institutional resource collections in herpetology and ichthyology. Copeia, 1985: 802-832.
- Malabarba, M. C. 1998. Phylogeny of fossil Characiformes and paleobiogeography of the Tremembé Formation. Pp.69-84. In: Malabarba, L. R., R. E. Reis, R. P. Vari, Z. M. Lucena & C. A. Lucena (Eds.). Phylogeny and Classification of Neotropical Fishes. Porto Alegre, Edipucrs, 603 p.
- Menezes, N. A. & S. H. Weitzman. 1990. Two new Species of Mimagoniates (Teleostei: Characidae: Glandulocaudinae), their phylogeny and biogeography and a key to the glandulocaudin fishes of Brazil and Paraguay. Proceedings of the Biological Society of Washington, 103 (2): 380-446.
- Nimer, E. 1989. Climatologia do Brasil. Rio de Janeiro, Instituto Brasileiro de Geografia e Estatística, 422p.
- Radan Brasil. 1983. Folhas SF. 23/24: Rio de Janeiro/Vitória: geologia, pedologia, vegetação e uso potencial da terra. Rio de Janeiro, 780p.
- Rizzini, C. D. 1992. Tratado de Fitogeografia do Brasil: Aspectos Ecológicos, Sociológicos e Florísticos (segunda edição). Rio de Janeiro, Ambito Cultural Edições, 747p.
- Swofford, D. L. 1993. "PAUP": Phylogenetic analysis using parsimony, version 3.1.1. Washington, D.C., Smithsonian Institution.
- Taylor, W. R. & G. C. Van Dyke. 1985. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage. Cybium, 9(2): 107-119.
- Weitzman, S. H., N. A. Menezes & M. J. Weitzman. 1988. Phylogenetic biogeography of the Glandulocaudini (Teleostei: Characiformes, Characidae) with comments on the distribution of other freshwater fishes in eastern and southeastern Brazil. Pp. 379-427. In: Vanzolini, P. E. & W. R. Heyer (Eds). Proceedings of a Workshop on Neotropical Distribution Patterns. Rio de Janeiro, Academia Brasileira de Ciências, 488 p.
- Weitzman, S. H. & J. Burns. 1995. Glandulocaudine fishes, a brief history from the perspective of the biologist/aquarist. Tropical Fish Hobbyist, 44(2):102-104, 106, 108, 110-113.
- Weitzman, S. H. & N. A. Menezes. 1998. Relationships of the tribes and genera of Glandulocaudinae (Ostariophysi: Characiformes: Characidae) with a description of a new genus, Chrysobrycon Pp. 171-192. In: Malabarba, L. R., R. E. Reis, R. P. Vari, Z. M. Lucena & C. A. Lucena (Eds.). Phylogeny and Classification of Neotropical Fishes. Porto Alegre, Edipucrs, 603 p.
Publication Dates
-
Publication in this collection
02 Jan 2008 -
Date of issue
Sept 2003
History
-
Accepted
08 July 2003 -
Received
26 Feb 2003