Abstracts
Hemiancistrus cerrado is described from the tributaries of rio Araguaia, rio Tocantins basin. Hemiancistrus cerrado has external similarities with H. megalopteryx and H. punctulatus from coastal streams of southern Brazil, and can be distinguished by having a larger internarial width, 15.9-21.1% of head length (vs. 11.2-14.0% in H. megalopteryx and 11.2-13.9% in H. punctulatus) and, with little overlap, by the larger adipose-fin spine length, 9.4-13.6% of standard length (vs. 7.1-8.7% in H. megalopteryx and 7.4-10.0% in H. punctulatus). Hemiancistrus cerrado further differs from H. megalopteryx by having the pectoral-fin spine reaching maximally to the middle of the pelvic-fin spine when adpressed in adult males (vs. reaching tip). Hemiancistrus cerrado differs from other members of Hemiancistrus by color and numerous morphometric and meristic data.
Neotropics; Catfish; Ancistrini; Cerrado; Taxonomy
Hemiancistrus cerrado é descrito de tributários da margem esquerda do rio Araguaia, bacia do rio Tocantins. Hemiancistrus cerrado possui similaridades externas com H. megalopteryx e H. punctulatus de drenagens costeiras do sul do Brasil, e pode ser separado das duas espécies pela maior distância entre as narinas, 15.9-21.1% do comprimento da cabeça (vs. 11.2-14.0% em H. megalopteryx e 11.2-13.9% em H. punctulatus), e, com alguma sobreposição, pela maior nadadeira adiposa, 9.4-13.6% do comprimento padrão (vs. 7.1-8.7% em H. megalopteryx e 7.4-10.0% em H. punctulatus); de H. megalopteryx ainda difere por espinho da nadadeira peitoral de machos adultos se estendendo até o meio do espinho da nadadeira pélvica quando adpressa (vs. se estendendo até a ponta do espinho). Hemiancistrus cerrado difere de outros congêneres pela coloração e diversos dados merísticos e morfométricos.
A new species of Hemiancistrus from the rio Araguaia basin, Goiás state, Brazil (Siluriformes: Loricariidae)
Lesley S. de SouzaI; Marcelo R. S. MeloI; Carine C. ChamonII; Jonathan W. ArmbrusterI
IDepartment of Biological Sciences, Auburn University, 331 Funchess, Auburn, AL 36849, USA. desouls@auburn.edu, melomar@auburn.edu, armbrjw@auburn.edu
IIMuseu de Zoologia da Universidade de São Paulo, Seção de Ictiologia, Av. Nazaré, 481, Ipiranga, Caixa Postal 42494, 04299-970 São Paulo, SP, Brazil. chamon@ib.usp.br
ABSTRACT
Hemiancistrus cerrado is described from the tributaries of rio Araguaia, rio Tocantins basin. Hemiancistrus cerrado has external similarities with H. megalopteryx and H. punctulatus from coastal streams of southern Brazil, and can be distinguished by having a larger internarial width, 15.9-21.1% of head length (vs. 11.2-14.0% in H. megalopteryx and 11.2-13.9% in H. punctulatus) and, with little overlap, by the larger adipose-fin spine length, 9.4-13.6% of standard length (vs. 7.1-8.7% in H. megalopteryx and 7.4-10.0% in H. punctulatus). Hemiancistrus cerrado further differs from H. megalopteryx by having the pectoral-fin spine reaching maximally to the middle of the pelvic-fin spine when adpressed in adult males (vs. reaching tip). Hemiancistrus cerrado differs from other members of Hemiancistrus by color and numerous morphometric and meristic data.
Key words: Neotropics, Catfish, Ancistrini, Cerrado, Taxonomy.
RESUMO
Hemiancistrus cerrado é descrito de tributários da margem esquerda do rio Araguaia, bacia do rio Tocantins. Hemiancistrus cerrado possui similaridades externas com H. megalopteryx e H. punctulatus de drenagens costeiras do sul do Brasil, e pode ser separado das duas espécies pela maior distância entre as narinas, 15.9-21.1% do comprimento da cabeça (vs. 11.2-14.0% em H. megalopteryx e 11.2-13.9% em H. punctulatus), e, com alguma sobreposição, pela maior nadadeira adiposa, 9.4-13.6% do comprimento padrão (vs. 7.1-8.7% em H. megalopteryx e 7.4-10.0% em H. punctulatus); de H. megalopteryx ainda difere por espinho da nadadeira peitoral de machos adultos se estendendo até o meio do espinho da nadadeira pélvica quando adpressa (vs. se estendendo até a ponta do espinho). Hemiancistrus cerrado difere de outros congêneres pela coloração e diversos dados merísticos e morfométricos.
Introduction
The Cerrado region of central Brazil, is a biologically rich savanna and currently one of the most threatened biomes of South America (Oliveira & Marquis, 2002). With the increasing expansion of agricultural activities, there has been a rapid reduction of the biodiversity in this unique ecosystem (Klink & Moreira, 2002). The 1.2 million km2 area contain tributaries of three of the major river basins in South America: the Amazon, Paraná-Paraguay and São Francisco rivers. In 2005 an expedition was taken to the Cerrado region of Brazil to explore the freshwater fish diversity, as part of the All Catfish Species Inventory Project. The focus was in the tributaries of the rio Araguaia in Goiás State. A new species of loricariid catfish was discovered and is being described in this issue, Hemiancistrus cerrado.
The genus Hemiancistrus Kner includes 15 valid species and is found in most tropical cis-Andean regions of South America and in Pacific coast drainages in Ecuador. Armbruster (2008) completed a phylogeny with representatives of all species groups of Hemiancistrus and found the genus to be paraphyletic; and he moved Peckoltia sabaji to Hemiancistrus. Armbruster (2008) defines Hemiancistrus phenetically as those species in his Panaque clade lacking the synapomorphies of the other genera and by having the dentaries meet at an angle greater than 120º (vs. <90º in Peckoltia). In addition to the 15 species currently recognized in Hemiancistrus, Armbruster (2004, 2008) removes several species to what he calls the Hemiancistrus annectens group; these species of trans-Andean fishes are unrelated to true Hemiancistrus and may represent an undescribed genus of the Pterygoplichthyini. Reviews of Hemiancistrus sensu stricto and the H. annectens group are under way by JWA, and morphometric data is based on that study. Five other species are known from the Amazon basin, H. micromattos, H. sabaji, H. snethlageae, H. spilomma and H. spinosissimus. Hemiancistrus micromattos, H. spilomma, and H. spinosissimus are also found in the Tocantins basin. Hemiancistrus cerrado is described herein from the rio Vermelho and rio Crixás-Açu, tributaries of rio Araguaia in the rio Tocantins basin.
Materials and Methods
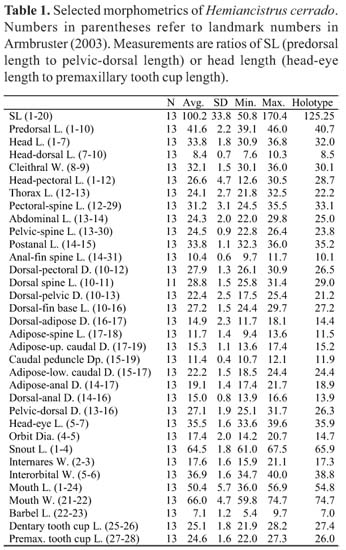
Counts and measurements follow Armbruster (2003). Institutional abbreviations are as listed at http://www.asih.org/codons.pdf. Specimens were cleared and stained for examination of bone and cartilage following the methods of Taylor & Van Dyke (1985). Names of plate rows follow Schaefer (1997). The following are abbreviations used in the text: CS = cleared and stained, D = distance, Dia = diameter, Dp = depth, dr = drainage, HL = Head length, L = length, W = width. All specimens were measured and examined for meristics.
Hemiancistrus cerrado, new species
Holotype. MZUSP 89074, 125.3 mm SL, Brazil, Goiás, município Goiás Velho, rio Bugre, tributary of rio Vermelho, underneath bridge on road GO-164, 25 km NW of Goiás, 15º47'13"S, 50º07'53"W, 24 Jul 2005, M. R. S. Melo, L. S. de Souza, C. C. Chamon & L. M. de Sousa.
Paratypes. Brazil, Goiás, rio Araguaia drainage: AUM 45427, 4, 68.1-123.6 mm SL and MZUSP 98668, 6 (1 cs), 50.8-136.9 mm SL, same data as holotype. MZUSP 26549, 1, 170.4 mm SL, rio Vermelho at the mouth of rio Bugre, 15º49'S, 50º19'W, 7 Dec 1981, J. C. Garavello, A. Copriva, L. L. Ferreira. MZUSP 89389, 1, 112.2 mm SL, between município Crixás/Santa Terezinha de Goiás, rio Crixás-Açu, underneath bridge on road GO-465, 14º26'26"S, 49º42'07"W, 28 Jul 2005, M. R. S. Melo, L. S. de Souza, C. C. Chamon & L. M. de Sousa.
Non-type specimens (juvenile, tentative identification). MZUSP 89048, 1, 24.3 mm SL, município Faina, rio do Peixe 2, tributary of rio do Peixe at road GO-164 km 84, 15º20'51"S, 50º24'30"W, 4 Jul 2005, C. C. Chamon, M. R. S. Melo, L. M. de Sousa & L. S. de Souza.
Diagnosis. Hemiancistrus cerrado can be distinguished from H. chlorosticus, H. fuliginosus, H. guahiborum, H. meizospilos, H. subviridis, and H. votouro by having dark spots (vs. light spots); from H. landoni and H. medians by lacking spots on the abdomen (vs. spots present in adults); from H. micromattos, H. sabaji, H. snethlageae, H. spilomma, H. spinosissimus by having an emarginate caudal fin (vs. forked); from H. snethlageae by lacking faint zig-zag lines along the sides outlining dorsolateral plates (vs. faint zig-zag lines present); from H. landoni by lacking papillae on exposed surfaces (vs. papillae present, especially well developed on skin around orbit and eye); and from H. sabaji by having the spots on the sides all about the same size (vs. getting larger posteriorly) and the spots separate on the caudal fin (vs. combining to form bands on the lower lobe in adults).
Hemiancistrus cerrado is very similar to H. megalopteryx and H. punctulatus but can be distinguished by having a larger internares W/HL (15.9-21.1% vs. 11.2-14.0% in H. megalopteryx and 11.2-13.9% in H. punctulatus), can usually be distinguished by having a larger adipose-fin spine L/SL ratio (9.4-13.6% vs. 7.1-8.7% in H. megalopteryx and 7.4-10.0% in H. punctulatus); and from H. megalopteryx by having the pelvic-fin spine reaching maximally to the middle of the pelvic fin spine when adpressed in adult males (vs. reaching the tip).
Description. Morphometrics in Table 1. Largest specimen examined 170.3 mm SL. Counts and measurements based on 13 specimens. Head and nape forming arc from tip of snout to insertion of dorsal fin. Dorsal slope decreasing in straight line to insertion of dorsal procurrent caudal rays then ascending to caudal fin. Ventral profile flat to caudal fin. Caudal peduncle triangular in cross section with dorsal surface flattened. Body widest at insertion of pectoral fins, narrowest insertion of caudal fin. Snout rounded.
Eyes moderately sized, dorsal rim of orbit forming tall crest that continues forward to area just anterior of nares as low, rounded ridge. Interorbital space largely flat, but with slight, rounded, median hump contiguous with rounded ridge on snout formed from mesethmoid. Slight ridge formed between anterodorsal margin of orbit and nares. Supraoccipital pointed posteriorly with posterior point raised above nuchal region in small crest. Infraorbitals, frontal, nasal, pterotic, and supraoccipital supporting odontodes. Preopercle and opercle not supporting odontodes. Opercle sickle shaped.
Lips covered with short, wide papillae. Size of papillae decreasing towards posterior margin of lower lip, bucal central papillae present and well developed. Lower lip wide, upper lip narrow. Edge of lower lip with small crenulae. Maxillary barbel reaching about one-third of distance to gill opening.
Median plates 24-27 (mode 25). Plates unkeeled, but first four or five plates of mid-ventral series bent to form slight ridge. Five caudal peduncle plate rows. Plates on all dorsolateral surfaces of body except for extreme edge of snout that only has narrow column of platelets on either side of snout tip. Throat mostly covered in platelets except for area right below lower lip. Abdomen naked or sparsely covered in platelets over and slightly posterior to pectoral bridge, laterally between pectoral and pelvic fins, and small region around anus. Cheek plates supporting hypertrophied odontodes evertible perpendicular to head. Cheek odontodes nine to 29 (mode 15). Longest evertible cheek odontode reaching middle of lateral process of cleithrum. Hypertrophied cheek odontodes relatively weak. Slightly longer odontodes present along dorsal-, adipose-, pelvic-, caudal-, and pectoral-fin spines; larger individuals with hypertrophied odontodes at tip of pectoral spine.
Dorsal fin II,7; dorsal-fin spinelet V-shaped, dorsal-fin locking mechanism present, last ray of dorsal fin almost reaching adipose-fin base when adpressed. Adipose fin with single preadipose plate and moderately long spine. Caudal fin I,14,I; caudal fin slightly emarginate, ventral lobe longer than dorsal lobe; dorsal and ventral procurrent caudal rays four to five (mode four). Pectoral fin I,6; pectoral-fin spine reaching just posterior to insertion of pelvic fin when adpressed. Pelvic fin I,5; pelvic-fin spine extending to base of anal fin when adpressed. Anal fin I,4; anal-fin spine slightly shorter than first ray.
Teeth bicuspid with lateral lobe three-quarters length of medial lobe and distal tip of lateral cusp one-half width of tip of medial cusp. Left dentary teeth 58-95 (mode 72). Left premaxillary teeth 55-91 (mode 71).
Color. Base color brown or gray in preserved specimens. Head and nape almost completely dark brown with large dark spots sometimes becoming mottled distally. Pectoral fin brown with dark brown large oblong spots on fin membranes and rays. Pectoral, pelvic, dorsal, anal and caudal fins slightly lighter than body, with dark, large oblong spots on fin membranes and rays. Spots in caudal fin arranged in three to five regular (contiguous along height of fin) or irregular (ventral and dorsal parts offset) bands; lighter interspaces light gray, usually slightly narrower than spots. Largest individual examined with light interspaces much narrower than spots, spots very irregular. Body with four saddles slightly darker than intervening areas, first below anterior end of dorsal fin, second with anterior half below posterior end of dorsal fin and posterior half behind dorsal fin, third beginning one to two plates anterior of preadipose plate to about posterior third of adipose-fin membrane, and fourth beginning just posterior to adipose fin to end of caudal peduncle. Ventral surface uniformly light except for blotches from anterior insertion of anal fin to caudal fin. Color in life similar to preserved coloration except base light brown and spots on body more intense.
Distribution and habitat. Known from rio do Bugre, rio Vermelho and probably rio do Peixe, tributaries of rio Araguaia in Goiás State, Tocantins Basin (Fig. 3). Collected in second order streams in swift rocky riffles (Fig. 4).
Etymology. Named after the Brazilian Cerrado, where the species is found. A noun in apposition.
Comparative material.Hemiancistrus punctulatus: MZUSP 37857, 13, Brazil, Rio Grande do Sul State, Município Jaguarão, confluence of rio Telho into rio Jaguarão, laguna Mirim drainage, approx. 32º30'S 53º27'W, MCP 17622, 5 of 23, Rio Grande do Sul State, rio dos Sinos, 2 km from Caraá, at Passo da Forquilha, 29º46'S 50º23'W. Further material listed by Armbruster (2008).
Discussion
Hemiancistrus cerrado, H. micromattos, H. spilomma, and H. spinosissimus are species known from the rio Araguaia-Tocantins basin. Hemiancistrus cerrado is distinguishable from H. micromattos, H. spilomma and H. spinosissimus by having an emarginate caudal fin (vs. forked). In addition, the dentaries of H. cerrado form almost a straight line, whereas the dentaries in H. micromattos, H. spilomma, and H. spinosissimus form a distinct angle approaching 90º in some specimens. Morphologically Hemiancistrus cerrado is most similar to H. punctulatus, which is found in laguna dos Patos drainage in Southern Brazil. Considering the great distance between the drainages systems of Southern Brazil and the Cerrado region, it is extremely unlikely that these two populations are conspecific; however, only one difference (internareal width) was found to distinguish them.
All specimens of Hemiancistrus cerrado were found in the Araguaia drainage in close proximity of each other. Their range is in the central region of the Cerrado, which is being heavily impacted by the rapid destruction of the Cerrado biome (Cavalcanti & Joly, 2002). Threats to the Araguaia drainage include agricultural erosion, pesticide runoff and mercury in the tributaries from gold mining. The uniqueness of the region as well as the fauna of the Araguaia is in urgent need of protection. Oliveira & Marquis (2002) recognized this region to be a priority for conservation and urged for a proactive conservation approach.
Acknowledgements
This project represents part of Planetary Biodiversity Inventory: All Catfish Species (Siluriformes) Phase I of an inventory of the Otophysi, a five year grant through the US National Science Foundation to describe all species of catfishes (NSF DEB-0315963). We would like to thank Mario de Pinna for his help in obtaining permits and logistical support. Additional thanks to Leandro de Sousa for aid in collecting specimens and taking pictures, Osvaldo Oyakawa for collections help and support, Neusa de Oliveira for support while in São Paulo, and Geoffrey Sorrell for comments on manuscript.
Literature Cited
Accepted August, 2008
Published September 30, 2008
- Armbruster, J. W. 2003. Peckoltia sabaji, a new species from the Guyana Shield (Siluriformes: Loricariidae). Zootaxa, 344:1-12.
- Armbruster, J. W. 2004. Phylogenetic relationships of the suckermouth armoured catfishes (Loricariidae) with emphasis on the Hypostominae and the Ancistrinae. Zoological Journal of the Linnean Society, 141:1-80.
- Cavalcanti, R. & C. Joly. 2002. Biodiversity and Conservation Priorities in the Cerrado Region. Pp. 351-367. In: Oliveira, P. S. & R. J. Marquis (Eds.). The cerrados of Brazil: ecology and natural history of a neotropical savanna. New York, Columbia University Press, 398p.
- Klink, C. & A. Moreira. 2002. Biodiversity and Conservation Priorities in the Cerrado Region. Pp. 69-88. Oliveira, P. S. & R. J. Marquis (Eds.). The cerrados of Brazil: ecology and natural history of a neotropical savanna. New York, Columbia University Press, 398p.
- Oliveira, P. & R. Marquis. 2002. The cerrados of Brazil: ecology and natural history of neotropical savanna. Columbia University Press, New York.
- Schaefer, S. A. 1997. The neotropical cascudinhos: systematics and biogeography of the Otocinclus catfishes (Siluriformes: Loricariidae). Proceedings of the Academy of Natural Sciences of Philadelphia, 148:1-120.
- Taylor, W. R. & G. C. Van Dyke. 1985. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium, 9:107-119.
Publication Dates
-
Publication in this collection
14 Oct 2008 -
Date of issue
2008
History
-
Accepted
30 Sept 2008 -
Received
Aug 2008