Abstracts
The discus fishes of the genus Symphysodon are popular ornamental cichlids that occur in floodplain lakes and flooded forests of the lowland Amazon Basin. These habitats are characterized by extreme seasonal fluctuations in the availability of food, shelter and dissolved oxygen, and also the densities of predators and parasites. Most aspects of discus biology are influenced by these fluctuating conditions. This paper reports an autoecological study of the western Amazonian discus S. haraldi (until recently classified as S. aequifasciatus). This species feeds predominantly on algal periphyton, fine organic detritus, plant matter, and small aquatic invertebrates. At high water it forages alone or in small groups in flooded forests. At low water it forms large aggregations in fallen tree crowns along lake margins. Breeding occurs at the beginning of the flood season, ensuring that the progeny are well grown before the next low water period. Symphysodon haraldi is an iteroparous partial spawner, reaches reproductive maturity within a year, and undertakes parental care of its eggs and larvae. The timing of spawning events, and/or the rate of brood survival, may be influenced by fluctuations in the flood level, resulting in a non-unimodal distribution of size classes for the subsequent 1+ cohort.
Flooded forest; Ornamental fishes; Parasite; Reproduction
Os acarás-disco do gênero Symphysodon são peixes ornamentais comumente encontrados em lagos e florestas alagadas das planícies inundadas da Amazônia. Estes habitats são caracterizados por uma variação sazonal extrema na disponibilidade de alimento, abrigo e oxigênio dissolvido, e também pela densidade de predadores e parasitas. A maioria dos aspectos da biologia do acará-disco são influenciados por esta variabilidade de condições sazonais. Este artigo apresenta um estudo autoecológico de S. haraldi (até recentemente classificado como S. aequifasciatus) da Amazônia Ocidental. Os acarás-disco alimentam-se predominantemente de perifiton, detritos orgânico, material vegetal, e invertebratos aquáticos pequenos. Durante a estação da 'cheia' eles forrageiam sozinhos ou em pequenos grupos dentro das florestas alagadas. Porém, na estação da 'seca' eles formam grandes cardumes nas coroas de árvores caídas ao longo das margens de lagos. A reprodução ocorre no início do período das enchentes, assegurando que a prole esteja bem crescida antes da próxima seca. Os acarás-disco são iteróparos, executam desova parcial, alcançam a maturidade reprodutiva em um ano, e executam cuidado parental dos ovos e alevinos. O início da desova, e a taxa de sobrevivência da prole podem ser influenciados pelas flutuações do nível da água, resultando em distribuições não-unimodais das classes de tamanho do subsequente '1+ cohort'.
Ecology and life history of an Amazon floodplain cichlid: the discus fish Symphysodon (Perciformes: Cichlidae)
William G. R. Crampton
Department of Biology, University of Central Florida, P.O. Box 162368. Orlando, FL, 32816, U.S.A
ABSTRACT
The discus fishes of the genus Symphysodon are popular ornamental cichlids that occur in floodplain lakes and flooded forests of the lowland Amazon Basin. These habitats are characterized by extreme seasonal fluctuations in the availability of food, shelter and dissolved oxygen, and also the densities of predators and parasites. Most aspects of discus biology are influenced by these fluctuating conditions. This paper reports an autoecological study of the western Amazonian discus S. haraldi (until recently classified as S. aequifasciatus). This species feeds predominantly on algal periphyton, fine organic detritus, plant matter, and small aquatic invertebrates. At high water it forages alone or in small groups in flooded forests. At low water it forms large aggregations in fallen tree crowns along lake margins. Breeding occurs at the beginning of the flood season, ensuring that the progeny are well grown before the next low water period. Symphysodon haraldi is an iteroparous partial spawner, reaches reproductive maturity within a year, and undertakes parental care of its eggs and larvae. The timing of spawning events, and/or the rate of brood survival, may be influenced by fluctuations in the flood level, resulting in a non-unimodal distribution of size classes for the subsequent 1+ cohort.
Key words: Flooded forest, Ornamental fishes, Parasite, Reproduction.
RESUMO
Os acarás-disco do gênero Symphysodon são peixes ornamentais comumente encontrados em lagos e florestas alagadas das planícies inundadas da Amazônia. Estes habitats são caracterizados por uma variação sazonal extrema na disponibilidade de alimento, abrigo e oxigênio dissolvido, e também pela densidade de predadores e parasitas. A maioria dos aspectos da biologia do acará-disco são influenciados por esta variabilidade de condições sazonais. Este artigo apresenta um estudo autoecológico de S. haraldi (até recentemente classificado como S. aequifasciatus) da Amazônia Ocidental. Os acarás-disco alimentam-se predominantemente de perifiton, detritos orgânico, material vegetal, e invertebratos aquáticos pequenos. Durante a estação da 'cheia' eles forrageiam sozinhos ou em pequenos grupos dentro das florestas alagadas. Porém, na estação da 'seca' eles formam grandes cardumes nas coroas de árvores caídas ao longo das margens de lagos. A reprodução ocorre no início do período das enchentes, assegurando que a prole esteja bem crescida antes da próxima seca. Os acarás-disco são iteróparos, executam desova parcial, alcançam a maturidade reprodutiva em um ano, e executam cuidado parental dos ovos e alevinos. O início da desova, e a taxa de sobrevivência da prole podem ser influenciados pelas flutuações do nível da água, resultando em distribuições não-unimodais das classes de tamanho do subsequente '1+ cohort'.
Introduction
The discus fish genus Symphysodon (Perciformes: Cichlidae) comprises two or three species restricted to floodplain habitats of the lowland Amazon basin (Bleher, 2006; Bleher et al., 2007; Ready et al., 2006). Symphysodon spp. are popular ornamental fishes valued for their bright colors, unusual disk-like shape, and complex behavior in captivity. A lively international trade revolves around the export of wild discus from the Brazilian Amazon (Crampton, 1999 a,b). Little quantitative data has been published about the ecology and life history of wild discus, despite the importance of Symphysodon in the aquarium trade and an extensive aquarium literature (including some publications describing natural history and distribution, e.g. Mayland, 1994, Degen, 1995; Bleher, 2006), and despite recent taxonomic and cytogenetic interest in the genus (Kullander, 1986; 1996; Ready et al., 2006, Bleher et al., 2007; Mesquita et al., 2008).
The seasonally inundated floodplain systems inhabited by discus fishes are among the most productive and species-rich ecosystems on earth (Goulding et al., 2003). The annual rise and fall of the Amazon River has a typical amplitude of some 6-12 m, exposing floodplain organisms to extreme fluctuations in the availability of food and shelter, the density of predators and parasites, and physico-chemical properties of the water such as dissolved oxygen (Junk, 1997). Understanding how fishes are adapted to these constantly changing conditions is central to an understanding of fish diversification in the lowland Amazon basin.
The last two decades have seen tremendous advances in our understanding of how seasonal flooding influences the diversity, ecology, and physiology of Amazonian floodplain fishes. Physiological specializations for hypoxia are among the best understood adaptations to floodplain life (Val & Almeida-Val, 1995). The dietary specializations and energy sources of floodplain fishes are also relatively well known (e.g. Araujo-Lima et al., 1986; Goulding et al., 1988; Benedito-Cecilio et al., 2000). Likewise, the distributions and movements of fishes in response to changes in oxygen concentrations and food availability are well documented from the community perspective (e.g. Goulding, 1980; Cox-Fernandes, 1997; Crampton, 1998; Henderson et al., 1998; Petry et al., 2003; Granado-Lorencio et al., 2005; Correa et al., 2008). Despite these advances, most aspects of Amazonian floodplain fish ecology remain generalized to a broad ecological scale, with little emphasis placed on the life history of single species. Detailed autoecological studies are available for only a few species, notably Colossoma macropomum (Cuvier) (Araujo-Lima & Goulding, 1997), and Cichla spp. (Jepsen et al., 1997, 1999; Winemiller et al., 1997).
There is a pressing need for additional autecological studies of Amazonian floodplain species, especially those with commercial value such as discus fishes. The aim of this paper is to demonstrate how multiple specializations of life history, behavior, physiology, and anatomy adapt populations of Symphydodon to the constantly changing conditions of the Amazon floodplain. Emphasis is placed on three themes: diet, reproductive ecology, and social structure.
Some aspects of the reproductive biology of Symphysodon spp. are known from aquarium observations. Like many other cichlids, discus form socially monogamous pairs and undertake biparental care of their broods (Matthaeus, 1992). Symphysodon and Uaru are unique among fishes in producing nourishing mucus that the newly hatched fry graze on. This mucus, known as 'discus milk', is exuded from the hypertrophied skin of both parents and is apparently essential for larval growth (Hildemann, 1959). The reproductive biology of wild discus has not previously been documented, and is explored here with emphasis on the influence of the flood cycle on spawning and life history.
Discus fishes are unique among Neotropical cichlids in forming static aggregations of up to hundreds of individuals during the low-water period (Mayland, 1994; Crampton, 1999a, Bleher, 2006). Local fishermen call these aggregations 'colonies'. Discus colonies shelter in coarse woody debris (known locally as 'galhadas'), the tangled branches and roots of fallen trees along lake margins, but disperse into seasonally flooded forests during the high-water period (Crampton, 1999a). The size, population structure, and site-fidelity of low-water aggregations of Symphysodon are described here, and their possible function discussed.
Notes on taxonomy and systematics of Symphysodon
Symphysodon belongs to the cichlasomatine tribe Heroini, which includes several other high-bodied cichlids that occur in floodplain systems of the lowland Amazon basin, e.g. Heros, Uaru, Pterophyllum and Mesonauta (Kullander, 1986). The phylogenetic position of Symphysodon within the Heroini is uncertain. Alternative hypotheses are given by Kullander (1998) on the basis of morphological data; by Farias et al. (1998, 1999, 2000, 2001) on the basis of molecular or combined molecular/morphological data, and by Ready et al. (2006) and Bleher et al. (2007) on the basis of molecular data.
The number of discus species is currently the subject of disagreement. Schultz (1960) published the first revision of the genus, and recognized two species on the basis of pigmentation: S. aequifasciatus Pellegrin from along the entire axis of the Amazon east of the lower rio Içá, and S. discus Heckel from the rio Negro, Abacaxis and Trombetas drainages. He also divided S. aequifasciatus into three subspecies based on pigmentation: S. aequifasciatus aequifasciatus ('green' discus - mainly from southern tributaries of the upper Amazon), S. a. haraldi ('blue' discus - mainly from northern tributaries of the upper Amazon) and S. a. axelrodi ('brown' discus - mainly from the lower Amazon). Regional variation in the color of discus species has also been the subject of many publications in the aquarium literature (reviews in Mayland, 1994 and Bleher, 2006). On the basis of pigmentation and meristic data, Kullander (1996) supported Schultz's (1960) division of the genus into two species but rejected the division of S. aequifasciatus into subspecies.
Based on studies of mitochondrial DNA sequence data, Ready et al. (2006) concluded that the genus Symphysodon comprises two distinct lineages and proposed that these correspond to two species: S. aequifasciatus, and a new species, S. tarzoo. All populations of 'green' and 'blue' discus previously assigned to S. aequifasciatus and occurring west of the Purus Arch were assigned to S. tarzoo. Symphysodon tarzoo was originally "described" by Lyons (1959) as "Symphysodon discus tarzoo" from near Leticia at the Colombia/Brazil border (although Lyons included a disclaimer stating that his notes on this form of discus were not intended to represent an official description). All populations of discus occurring to the east of the Purus Arch (a geological feature believed to have isolated eastern and western Amazonian drainages until some 6 MA, see Lundberg et al., 1998) were assigned by Ready et al. (2006) to S. aequifasciatus. These comprised some populations previously assigned to S. aequifasciatussensu Kullander (1996), and all populations that were previously assigned to S. discus.
Bleher et al. (2007) also used mitochondrial DNA sequence data to study interrelationships among Symphysodon populations, but reached different conclusions to Ready et al. (2006). They argued that there are three distinct species: S. discus ('Heckel discus'), S. aequifasciatus ('green discus'), and S. haraldi ('blue and brown discus'). In this scheme, S. aequifasciatus is the only one of these species to occur only to the west of the Purus Arch. Symphysodon haraldi is distributed widely from the mouth of the Amazon to the lower rio Içá, near the westernmost limit of the range of the genus. They limit the distribution of S. discus to the rio Negro, some north bank tributaries of the Central Amazon, and the rio Abacaxis (lower Madeira), and report extensive hybridization between S. discus and S. haraldi.
There is therefore disagreement over the number of species in the upper Amazon (including the Tefé region, where the study reported here was conducted). Ready et al. (2006) argue that only one species occurs west of the Purus Arch (S. tarzoo), while Bleher et al. (2007) argue that there are two: S. aequifasciatus (mostly from south bank tributaries), and S. haraldi (mostly from north bank tributaries). At this stage there may be insufficient genetic evidence to resolve the disagreement. Bleher et al. (2007) did not collect any north bank 'blue' discus tissue samples from west of the Purus Arch. Ready et al. (2006) sampled just one north bank population, from the lower rio Içá, and were unable to distinguish this from 'green' discus populations in south-bank tributaries. These uncertainties will likely be resolved by ongoing systematic investigations (T. Hrbek, pers. com.).
Material and Methods
Study area. Field work was undertaken near the town of Tefé, Brazil (Fig. 1). The climate of this region is typical of the Central Amazon, with a rainy season from February to April (300-450 mm/month) and a drier season from August to December (50-150 mm/month) (Ayres, 1993). The annual flood amplitude varies between 8 and 16 meters, reaching its peak between April and June, and its ebb in October (Fig. 2). The aquatic habitats of the Tefé region, also typical of the Central Amazon, are broadly divisible into two systems by nutrient content: the whitewater Amazon River and its floodplain (conductivity 80-200 µS.cm-1), and blackwater systems draining the terra firme forests above the maximum flood level (5-30 mS.cm-1) (Henderson et al., 1998). Both whitewater and blackwater systems of the Tefé region form expansive floodplains of seasonally inundated forests, lakes and channels. Large mouthbay lakes such as lago Tefé (Fig. 1) form where sediment-poor blackwater rivers (in this case the rio Tefé) drain into sediment-rich whitewater rivers (in this case the Amazon River). In the Tefé region, discus inhabit both whitewater and blackwater floodplain systems but are absent from large river channels and terra firme streams (Crampton, 1999a,b).
Study sites. This study focused on two sites in blackwater mouthbay lakes: Uxi Bay, a small embayment of lago Amanã; and the northern margin of nearby lago Urini (Fig. 1). These sites were selected because they had no previous history of commercial discus fishing and because, unlike in whitewater floodplain lakes, densities of piranhas (Pygocentrus nattereri (Kner) and Serrasalmus spp.) were low. Parallel studies of discus populations in the confluence area of the Amazon and Japurá Rivers had to be abandoned due to severe net damage by piranhas.
Uxi Bay was preferred for analyses of population structure because its small discus population permitted the efficient processing of all specimens within the limited time frame of rapidly changing water levels. Uxi Bay was also isolated from other discus populations by at least 6 km of shallow, wave-exposed shoreline (a habitat avoided by discus). The populations in lago Urini were much larger and were sampled with professional ornamental fish-catchers reportedly visiting for the first time. This allowed much larger numbers of discus to be captured, but storage limitations made it impossible to take measurements from individual fish.
Identification of discus. Discus population in lago Amanã and lago Urini exhibit the distinctive color phenotype of the blue/brown discus Symphysodon haraldi (sensu Bleher, 2006 and Bleher et al., 2007 - each of these publications includes detailed identification guides). This paper adopts the most recent classification scheme proposed by Bleher et al. (2007) - using the name S. haraldi (despite the absence of molecular data for the presence of S. haraldi west of Manacapuru) rather than the name S. tarzoo proposed by Ready et al. (2006) for discus occurring west of the Purus Arch.
Electrical conductivity. Conductivity was measured with a Hanna Hl 9142 meter at approximately two week intervals near the Uxi bay, between September 1997 and April 1998.
Sampling. To maximize the proportion of discus in breeding condition, sampling was undertaken at the beginning of the rising water season (December 1997 and November 1998 for Uxi Bay, December 1999 for lago Urini), but before the galhadas were deeply inundated (at which point they were not possible to sample effectively). Additional galhada samples were taken at the lowest flood ebb in October 1997 from lago Urini. With the assistance of a team of 8 - 10 fishermen, each galhada was surrounded by a 50 x 8 m seine net (5 mm mesh) and the branches removed with machetes and saws before the net was closed (Fig. 3). Captures, reported as the number of fish per netted galhada are relative estimates because some fish escaped around or over the net. Captures of discus per galhada are reported in the following format: 1 (4) = single specimens were captured in each of four galhadas; 19 (1) = 19 discus were captured together in one galhada.
After sampling, each galhada was reconstructed and fresh-cut branches (approximately 2-4 m long) were added. One week before the 1998 samples in Uxi Bay, 25 'artificial' galhadas were built from piles of branches at locations interspersed between the existing natural ones. These were created to examine the propensity of discus and other cichlids to enter new substrate.
Beaches were sampled with a 50 x 8 m seine net (5 mm mesh). In Uxi Bay, floating patches of Cyperus were sampled with a 30 x 4 m seine net (5 mm mesh). Discus were also captured (incidentally) in 70 x 3 m gill-nets (60 mm mesh) set at 1700-0500 in flooded shore scrub or open water. Flashlights and hand nets were used to observe and capture discus at night. During the high water period some 100 hours of observation from canoes were made in flooded forests of lago Amanã.
Measurements and sex determination. Discus were held in aerated tanks on a boat. Soon after capture, standard length (SL) (in mm), and weight (in g) were measured for live specimens. Also, the number of a common isopod gill-chamber parasite, Braga cichlae Schiödte & Meinert (Cymothoidae), was scored for the right and left gills. A random sub-sample of 218 discus from Uxi Bay was sacrificed humanely and dissected to determine sex and maturity (which cannot always be reliably determined from external features, see Silva & Kotlar, 1980). The ovary of one pre-spawning female was fixed with Bouin's Solution and the eggs hand-counted and measured with a Wild M5 stereomicroscope fitted with an ocular micrometer. A modified version of Nikolsky's (1963) scale of reproductive maturity was devised to score discus maturity (Fig. 4). Male specimens with stage 1 testes could not be reliably discriminated from males/females with stage 0 gonads. Consequently, fishes with 0 and 1-stage gonads were classified as unsexed juveniles.
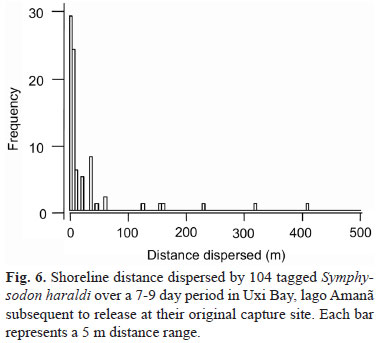
Site fidelity and movement. In December 1997, 91 discus were tagged and released at their capture sites in Uxi Bay. The expectation was that recaptures the following year would reveal patterns of growth and site fidelity. 219 discus were tagged and released (within 12 hr) at their capture sites in Uxi Bay during the period 16-18 November 1998. 7-9 days later all galhadas in Uxi Bay were resampled to document short-term dispersal. Because the tagged fishes were released at their exact capture sites, the proportion of tagged fish in the recaptures could not be used to estimate an actual population size in the Uxi bay from the sampled population size. To do so would have required 'mixing' the marked and unmarked portions of the population by releasing the tagged fishes in randomly chosen galhadas around the bay (see Southwood & Henderson, 2000). Numbered Floy 13 x 2 mm FD-68B T-bar tags were injected with a Dennison applicator at the base of the dorsal fin. Tag retention was tested by leaving 15 tagged fish (100-150 mm SL) in a crowded commercial holding tank for three weeks. Retention was 100% and no fish became infected.
Stomach content analysis. To emphasize seasonal effects, discus were collected at the ebb of the low-water season from galhadas, and at the peak of the high-water season from flooded forest. Fish in the 90-140 mm SL range were collected between 1600 and 2000 and size related dietary variation was not investigated. Stomachs were preserved in 10% formalin before analysis. The 'points method' of dietary analysis (Swynnerton & Worthington, 1940; Hyslop, 1980) was modified by assigning a percentage content (to nearest 5%), for each food type. This involved a subjective estimate by visual counts. The percentages were used to calculate the mean contribution of each food item in N stomachs. Frost's (1943) modification of the points system to take account of stomach fullness was not employed. Instead, each stomach was given a fullness index as a percentage of a fully distended stomach (to nearest 5%).
Results
Flood level and electrical conductivity. The electrical conductivity of the water near the Uxi bay exhibited an inverse relationship with water level, fluctuating from 8 µScm-1 at high water to 29 µScm-1at low water (Fig. 2). During the two years of the study, and also in the three preceding years during which the water level was monitored, events known locally as 'repiquetes' occurred during each rising water period. Repiquetes are periods of declining or static water level that interrupt the otherwise upward trend of the flood waters. For instance, at the ends of 1996, 1997 and 1998, a single large repiquete occurred between the 5 and 9 m datum (Fig. 2). Repiquetes were associated with fluctuations in electrical conductivity.
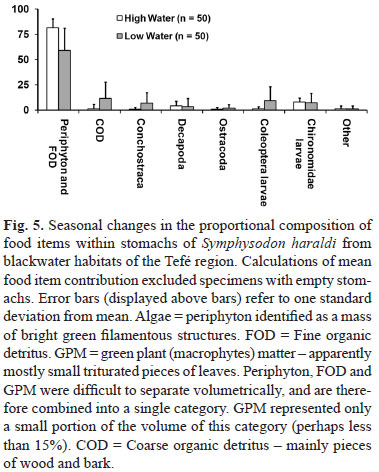
Diet. The proportional composition of food items in stomachs of S. haraldi are summarized for low and high water periods in Fig. 5. The mean stomach fullness at low water (including specimens with empty stomachs) was 28.4% (SD 30.1, n=75), with 33 % of specimens exhibiting completely empty stomachs, and 68% exhibiting a stomach fullness of 30% or less. At high water the mean stomach fullness was 89.2% (SD 15.2, n=50), with no specimens exhibiting a stomach fullness of less than 50%.
Microhabitat preferences and distribution of discus among galhadas. During the low water period, discus in Uxi Bay and lago Urini were only captured in galhadas or flooded shore scrub. None were found on muddy or sandy beaches (48 seine nets), on beaches vegetated with wild rice (Oryza) (20 seine nets), in patches of Cyperus (55 m2 with seine net), or in open lake water (210 m2 gill net x 10 nights). Gill nets set in shore scrub habitats of Uxi Bay and Urini captured 24 discus (210 m2 x 15 nights).
In Uxi Bay, a total of 19 galhadas (all natural) were sampled in 1997, and 58 (33 natural, 25 artificial) in 1998. The sampled population exhibited the following distribution of discus numbers per galhada (e.g. 1 (4) = single discus in each of 4 galhadas): 1997 (19 sites, 178 discus): 0 (14), 1 (4), 174 (1); 1998 (58 sites, 259 discus): 0 (50) 1 (3), 2 (1), 3 (2), 4 (1), 19 (1), 225 (1). In both years most of the discus in the Uxi bay were concentrated into one large (ca. 70 m2) galhada comprising several overlapping fallen tree crowns (i.e. 1997: 174/178 (97.8% of sampled discus); 1998: 225/259 (86.8% of sampled discus)). This large galhada was located in the deepest area of the bay (2-3 m), had negligible current (< 0.01 ms-1), and was well-sheltered from waves. Outside the large galhada, the remaining discus in the Uxi bay were only found at galhadas larger than 4 m diameter located in non-flowing water of 1.5 - 3.0 m depth, and sheltered from waves. 24 sheltered galhadas in static, deep water (2-3m) did not contain discus. Researchers working in the Uxi Bay after this study was completed also noted aggregations of discus comprising several dozen specimens in deep (1-2.5 m) pools of the lower reaches of a rainforest stream flowing into Uxi Bay (J. Alves de Oliveira, pers. comm.).
In lago Urini a total of 70 galhadas were sampled. All were greater than 4 m in diameter, located in areas sheltered from strong wave action, and exposed to negligible water flow (< 0.01 ms-1). Discus were absent from 48 of these galhadas, and present in 22. These 70 galhadas were divided into three arbitrary categories. The first comprised those in water of less than 1 m depth (at the lowest water level) (n = 14), the second in water of 1-2 m depth (n = 15), and the third in water of 2-4 m depth (n = 41). Discus were absent from the first depth category. The second depth category contained 24 discus (1.9 % of individuals from lago Urini galhadas) with the following distribution of individual fish per galhada: 1 [discus] (2 [galhadas]), 2 (1), 3 (1), 5 (1), 12 (1). The third depth category contained 1,246 discus (98.1% of individuals) with the following distribution of individual fish per galhada (or group of contiguous galhadas): 1 (3), 2 (3), 3 (1), 12 (1), 18 (1), 25 (1), 45 (1), 62 (1), 79 (1), 180 (1), 251 (1), 562 (1). Locals reported galhadas of similar depth in other parts of the lake hosting more than a thousand discus. Only 16 of the 41 galhadas in the 2-4 m depth category (39%) were occupied by discus. The remaining 'empty' galhadas did not differ obviously from those that contained discus in terms of their branching structure, sediment, shading, or by the extent to which they were protected from wind and wave action.
At high water, discus colonies dispersed into flooded forests and dense shore scrub. Samples of the galhadas containing the single large discus aggregation in Uxi Bay were conducted on 1 December 1998, 2 weeks (and a 2.5 m water level rise) after the colony was last sampled. No discus were encountered, even though the net was still able to reach the bottom and efficiently sample the galhada. During nightly observations in flooded forests during the high water period, discus were often seen alone or in groups of up to six, but never in larger groups. During the peak of the flood season 82% (237/289) of discus were seen in dense shore scrub or in forests with trees of moderate height (< 15 m). The remaining 18% were found in more shaded igapó forests dominated by high trees (15 - 25 m) (see Fig. 1).
Site fidelity and movement. Of 91 discus tagged in Uxi Bay in December 1997, only one (male) specimen was retrieved in November 1998. This was first captured at the site of the single large colony (Fig. 1), and then recaptured at the exact same site, 11 months later. During this period this specimen grew from 108 to 137 mm SL and 71 to 151 g in weight. This specimen provided a valuable measure of annual growth.
We recaptured 7-9 days later 104 of 219 discus tagged and released in November 1998 when all 58 natural and artificial galhadas in the bay were sampled. The shoreline distances covered by these fish during this period (Fig. 6) indicate little short-term dispersal, despite the fact that the stress of capture and tagging often causes animals to move (Southwood & Henderson, 2000). The 25 artificial galhadas made one week prior to the first round of 1998 samples (during which all discus were tagged) were colonized by 111 cichlid fishes, but only seven (6.3 %) of these were discus.
Predation. In lago Urini, the biomass (total live weight) of discus from galhadas in water of 2-4 m depth was positively correlated to the biomass of all other fishes (Spearman's R = 0.34, n = 41, p < 0.05) and to the biomass of other fishes excluding piscivorous fishes larger than 200 g (Spearman's R = 0.40, n = 41, p < 0.05), but negatively correlated to the biomass of piscivorous fishes larger than 200 g (Spearman's R = -0.29, n = 41, p < 0.05). All galhadas with 25 or more discus contained no predators larger than 200 g. Galhadas with 1-18 discus contained an average of 1.1 kg of predators larger than 200 g (range 0-6.3 kg, n = 9). Galhadas with no discus contained an average of 0.93 kg of predators larger than 200 g (range 0-5.4 kg, n = 25).
During this study, the remains of one small (75 mm SL), damaged discus was found in the stomach of a 1.1 kg specimen of Serrasalmus rhombeus (L.) (n = 38 examined), but not in any of the following syntopic species that were reported by local fishermen to prey on discus: Serrasalmus spp. (n = 72), Pygocentrus nattereri (n = 18), Hoplias malabaricus (Bloch) (n = 48), Hydrolycus spp. (n = 29), Electrophorus electricus (L.) (8), Pseudoplatystoma fasciatum (L.) (n = 8), P. tigrinum (Valenciennes) (n = 2), Plagioscion squamosissimus (Heckel) (n = 5), and Cichla monoculus Spix (n = 49).
Parasitization. 69 of 492 specimens of S. haraldi (14%) were infested with B. cichlae. Seven fish carried two B. cichlae (one in each opercular chamber), and the remaining 62 carried just one in either opercular chamber. None of the discus parasitized by B. cichlae had progressed in sexual maturity beyond stage 2, including those from the putative 2+ cohort (see discussion). Also, as summarized in Fig. 7, discus parasitized by B. cichlae (one or two parasites) weighed significantly less (at a given SL) than unparasitized individuals (General Linear Model [GLM] of the form Ln (Weight) = Ln (SL) + parasite + SL * Parasite, where weight and SL are continuous variables and the presence/absence of parasites is a discrete variable, F 1, 488, p < 0.001). Despite this stunting effect, there was no significant difference between the rate at which infested versus unparasitized fish accumulate weight with increasing length (GLM, F 1, 488, NS).
Sex and reproductive condition. 218 discus from Uxi Bay were dissected and sexed. 71 were male and 68 female. The remaining 79 specimens were juveniles of indeterminate sex (stages 0 and 1). The relationship between SL and weight for all male and female discus was not significantly different between the sexes (GLM, Ln (Weight) = Ln (SL) + sex + SL * sex: F 1, 135, NS) (Fig. 8). There was also no significant sex-related difference in weight (GLM, F 1, 135, NS). Nonetheless, the sampled males attained a larger size than the females (152 mm SL, 194g vs. 139 mm, 159g).
The size distributions of S. haraldi from the single large colony in Uxi Bay are illustrated in Fig. 9. In both the 1997 and 1998 samples there was an overlapping multi-modal distribution of size classes. Age groups were separated by the Petersen method of graphical distribution of modes (Bagenal & Tesch, 1978). The size distribution of seven discus captured with gill nets in newly-flooded shore scrub in Uxi Bay is superimposed onto the 1998 size distribution plot. The reproductive status of discus within the colony is compared to those in adjacent shore scrub in Fig. 10. The reproductive status of specimens from the putative 1+ cohort and 2+ cohorts (see discussion) within the colony are compared in Fig. 11.
Many of the discus in Uxi Bay reached pre-spawning condition by the early rising water phase (Fig. 9a,b). However, none of 45 discus captured from galhadas in lago Urini two month earlier (October 1997) had progressed beyond stage 2. At the peak of the flood period (in April 1999), only one of 28 discus captured from flooded forests was mature (female, stage 4), while all others were immature or resting (stage 1).
Observations of spawning. All of the branches removed from galhadas during samples were inspected for discus eggs, but none were found. One pair of large, brightly-colored discus was observed attending a brood of eggs on submerged aerial roots of shore shrub in Uxi Bay. The site was 0.3 - 0.5 m deep and about 10 m from the lake edge. On three consecutive nights in December 1997 the pair was visible by lamplight at the same position. During the day they were not visible. After three nights they disappeared, leaving some fertilized eggs (c. 1.3-1.5 mm diameter) on the root surface. The brood was in this case apparently abandoned. Intensive efforts to locate other brooding discus were unsuccessful.
Gonadosomatic index and egg size distribution. The gonads of six females with pre-spawning (gonad stage 4) ovaries weighed from 0.40 to 0.75 g. This corresponded to a gonadosomatic index of 0.340-0.652 % (mean 0.465, SD 0.104). All of the ripe ovaries exhibited more than one size class of eggs. The size distribution of eggs from the ovary of a single pre-spawning female illustrated a tri-modal size distribution (Fig. 12), which is typical of a partial-spawning fish.
Color. Digital photographs of accurately sexed discus from Uxi Bay revealed no obvious sex-related differences in colors. Juveniles are a mahogany or olive hue with relatively small areas of color restricted to the anal-fin base and nape. Individuals above 90 mm SL display wavy blue-green and red lines or spots in the nape and cheek region, blue and red marking on the pelvic fins and anal-fin base. These colors typically cover more of the body surface area in fish from the 2+ cohort than those in the 1+ cohort (see also Mayland, 1994; Degen, 1995). In the aquarium trade Symphysodon spp. are arbitrarily labeled 'royal discus' when the wavy blue-green and red lines extend uninterrupted from the nape to the base of the caudal fin. This gives them a spectacular appearance and much higher commercial value (Degen, 1995; Crampton, 1999a).
A number of 33 of 258 (12.8 %) discus from the 1998 galhada samples in Uxi Bay, and 13/24 (54.2%) of discus from shore scrub habitats (Uxi Bay and lago Urini combined) exhibited royal coloration. Combing the samples from both these habitats, 42 of 46 royal discus (91%) were from the 2+ cohort and the remaining four were from the 1+ cohort. Considering only fish whose sex and maturity was confirmed, none of 36 immature specimens (stages 0-2) exhibited royal coloration. In contrast, 25 of 89 (28%) mature fishes (stages 3-5) exhibited royal coloration. Royal discus of both sexes were found, exhibiting a male: female ratio of 18:7. Of these 25, four (16%) had stage-3 gonads, 20 (80%) had stage-4 gonads, and one (4%) had stage-5 gonads. Among non-royal discus, mature specimens were also generally more colorful than immature ones.
Without exception, and regardless of size, all discus infested with B. cichlae were a drab coloration - presenting only small, lackluster areas of blue-green or red color. The vertical flank bars, which become intensified in stressed or socially subordinate fish, were often permanently highlighted in these individuals. Similar condition factors were observed in four discus with severe infestations of the intestinal nematode Capillaria.
Discussion
Feeding adaptations to flooded forests. In volumetric terms, Symphysodon haraldi appeared to feed predominantly on a mixture of algal periphyton, fine organic detritus and green plant matter. These three categories were combined into a single category because of practical difficulties in separating them. Nonetheless, green plant matter in the form of triturated leaves probably represented no more than 15% of the total volume (a crude estimate). Of the remaining material, it was hard to distinguish between partially digested periphyton and fine organic detritus - which comprises mainly decomposing plant residues.
Periphyton comprises multi-species filamentous algal communities covering the branches and leaves of flooded forests. Periphyton represents a major form of primary production, and an important source of energy for floodplain fish populations (Araujo-Lima et al., 1986; Forsberg et al., 1993). Fine organic detritus, which becomes trapped by periphyton and accumulates on submerged structures, is in itself also an important source of nutrition for many species of Amazonian fishes, and a dominant source of energy for fish communities as a whole (Araujo-Lima et al., 1986).
At low water, far less submerged substrate is available for periphyton growth. Also, lake waters tend to become clouded by the action of waves on clay beaches at low water - reducing the photic zone for algal growth. These factors may explain why a large proportion of discus exhibited only partially filled stomachs at low water, and why invertebrates comprised a greater fraction of their diet. During the high water period discus were usually found in open, well lit flooded forests where periphyton grows most profusely. At this time of the year, invertebrates constituted a surprisingly small volumetric component of discus diet, although they may represent a vital source of protein. Further study is required to investigate the relative energetic and nutritional contribution (versus volumetric representation) of detritus, algae, plant matter and animal matter in the diet of Symphysodon.
The alimentary canal of Symphysodon is characterized by a poorly defined stomach and an elongate intestine, some 300 mm long and 3 mm wide (in a 180 mm SL specimen). This intestinal morphology is typical of a cichlid with a dominantly vegetarian, detritivorous, or omnivorous diet. Predominantly piscivorous cichlids such as Cichla and Crenicichla exhibit shorter alimentary canals with well developed stomachs (Zihler, 1982).
Bleher (2006, p. 510-595) reports detailed observational notes on the diet of discus, taken over many years of field visits to the Amazon basin. He undertook stomach content analyses on over 8,500 discus specimens and also made direct observations of feeding in the wild. Although most of his findings are reported qualitatively, Bleher (2006) presents some quantitative data for the volumetric dietary intake of S. haraldi (although numbers of specimens are not given, p. 593). During the high-water period he reports average stomach contents of: 12% algae and microalgae, 44% plant matter (flowers, fruits, seeds, leaves), 6% detritus, 16% aquatic invertebrates, and 22% terrestrial and arboreal arthropods. During the low water period he reports 25% algae and microalgae, 39% detritus, 9% plant matter, 22% aquatic invertebrates, and 5% terrestrial and arboreal arthropods. Data for S. aequifasciatus and S. discus indicate a larger proportion of algae, plant matter and detritus both for during the low and high water periods. The data presented here for S. haraldi from the Amanã region indicate a pattern of lower dietary variability, and a much larger proportion of periphyton/detritus than reported by Bleher (2006). These discrepancies might reflect the small sample sizes reported here; perhaps much larger samples are required to show the true breadth of discus diet. Alternatively these discrepancies might reflect natural variation in diet among populations and species of discus.
Non trophic adaptations to flooded forests. The exaggerated lateral compression and body height of discus are shared, to a lesser degree, by Mesonauta and Pterophyllum. This morphology is presumably well suited to entering the narrow interstices of submerged branches. The dark vertical stripes and drab green-brown background colors of discus presumably provide camouflage, and the stripes are rapidly intensified by chromatophore expansion in alarmed fish.
Flooded forests waters are typically poorly oxygenated or anoxic due to the decomposition of leaf litter and other organic debris (Crampton, 1998). Like other Amazonian cichlids, S. haraldi exhibits a suite of biochemical adaptations that permit efficient anaerobic respiration during periods of hypoxia. These are documented by Chippari-Gomes et al. (2005).
Group living behavior in discus. Discus form multi-cohort 'colonies' in galhadas, which appear to function as low-water refuges. The short dispersal distances indicated by mark-release data, coupled with the relatively small number of discus captured from newly constructed ('artificial') galhadas suggest that discus colonies are relatively static during the low water period. Discus were not evenly distributed among galhadas at the study sites. Instead, many apparently suitable galhadas (sheltered sites in 2 - 4 m depth), were devoid of discus or contained only a few specimens, while other, similar galhadas contained hundreds of specimens. These patterns suggest that discus actively form social aggregations, rather than simply fill available space.
During the rising water period, colonies in the galhadas of lago Amanã disintegrated. Discus dispersed into adjacent flooded forests where they were thereafter observed alone or in small groups. The persistence of a sizable colony at one site in the Uxi bay throughout this study, and the return of a single tagged fish to this site over an 11 month period suggest that individual discus may routinely return to the same low water refuge from one year to the next. Crampton (1999b) also reports the return of individual discus to galhadas in a white water floodplain between successive low-water periods.
Group living and predation. The formation of large, multi-cohort aggregations is unique to Symphysodon among Neotropical cichlids. Most South American cichlids live alone or in small groups but form breeding pairs and undertake biparental care of the eggs and young (Breder & Rosen, 1966; Lowe-McConnell, 1969; Goodwin et al., 1999). Aggregations of more than a few dozen individuals are rarely if ever seen in other South American cichlids (H. Bleher, H. López-Fernandez, R. Lowe-McConnell, pers. comms.). In the Tefé region, the only cichlid species other than discus ever seen in adult aggregations exceeding six individuals is the angelfish Pterophyllum scalare (Lichtenstein). This species forms groups of up to around 30 individuals during the low water period (Crampton, 1999c). Some other cichlids form groups of no more than six specimens, including: C. monoculus, Geophagus proximus, Heros efasciatus, Mesonauta mirificus, Uaru amphiacanthoides, and Satanoperca jurupari (pers. obs.).
Predatory fishes reach spectacular densities in Amazonian floodplains during the low-water period (Goulding, 1980), and large cohesive groups of discus may mitigate predation. Pitcher (1986) and Magurran et al. (1985) review mechanisms by which fish shoals may reduce predation. Queiroz & Magurran (2005) proposed that shoaling in an Amazonian floodplain fish, the piranha Pygocentrus nattereri, may have evolved in response to predation. Magurran & Queiroz (2003) noted that shoals of this species were size-structured with larger, mature specimens occupying central positions. Gomez-Laplaza (2002) suggested that predators play a role in the formation of small shoals of the floodplain cichlid P. scalare, and demonstrated that predator inspection behavior varied among group-members by social dominance.
Two observations suggest that colonies of discus might actively discourage the presence of fish predators. First, piscivorous fishes larger than 200 g were absent from galhadas holding groups of 25 or more discus. Second, there was a (weak) negative correlation between the biomass of discus and piscivorous fishes larger than 200 g. Bleher (2006, p. 503) documents brightly colored 'alpha individuals' swimming outside galhadas containing groups of discus. Here they reportedly "protect the group from predatory fishes by acting as a decoy". Nonetheless, it is not yet clear which fish species are important discus predators. Piranhas (Serrasalmus spp., P. nattereri) are voracious piscivores, but because they typically do not swallow their prey, stomach content analyses may drastically overlook the extent to which they eat discus. The importance of caiman, river dolphins, otters, snakes and birds as predators is also unknown.
Group living and parasites. Group living, even if temporary, has disadvantages - notably the increased transmission of diseases and parasites (Trivers, 1985). A large proportion (14%) of discus from the Uxi Bay was infested with the isopod B. cichlae. This parasite not only appears to retard discus growth, but appears also to limit the development of bright colors (see 'color' in Results). Braga cichlae devours gill lamellae, causing reduced oxygen uptake and presumably therefore hindering normal growth and development. In a separate paper, data will be presented to show that discus are infested far more frequently by B. cichlae than other syntopic cichlids, despite the generalist nature of this parasite (Thatcher, 1991); this lack of host specificity can be demonstrated by providing free-swimming B. cichlae with a choice of Symphysodon versus other cichlid hosts. Because discus develop bright color as a secondary sexual ornament (see 'color' in Results), and because parasites both diminish color and stunt growth, a case will be made for bright color serving as an index of parasite load in mate choice. There is a strong theoretical basis for color serving as an index of mate quality, including general health and parasite load (e.g. Maynard-Smith & Harper, 2003). As such, there may be a parasite-mediated link between the formation of extremely large aggregations, and the possession of bright color extending over much of the body surface - both features unique to Symphysodon among South American cichlids.
Population structure. Although male discus grow larger than females, both sexes have the same length to weight relationship (Fig. 8), and the sampled sex ratio is close to 1:1. This simplifies the interpretation of age groups from the distribution of modes in a histogram of size distribution (sensu Everhart et al., 1975; Jearld, 1983). The speculative schematic model of age structure in Fig. 9 is based on the following assumption: modal classes 1b and 2 in 1998 and, likewise 2b and 3 in 1997, are separated by approximately one year. This assumption derives from the known growth of the single 1998 recapture of a fish marked 11 months earlier (X1,2 in Fig. 9). In contrast, groups 1a and 1b in 1998 (and likewise groups 2a and 2b in 1997) are too close together to represent a full year of growth (the modes are approximately 15.5 mm apart compared to approximately 32 mm between 1b and 2 or 2b and 3). There is no significant disparity in the number of each sex between 1a and 1b, or 2a and 2b, ruling out a sex basis for the bimodal size distribution. Likewise, there is no disparity in the number of parasitized specimens between 1a and 1b, or 2a and 2b. Instead, the modes of groups 1a and 2a must correspond to progeny from breeding events in the same season as 1b and 2b respectively. This is compatible with the period over which female discus were observed in pre-spawning condition.
The 1998 and 1997 populations are therefore hypothesized to comprise two main age groups: first, a younger 1+ cohort comprising fish 12 (+/- 6) months old (mode 1a,b, 1998; mode 2a,b, 1997); and second, an older 2+ cohort comprising fish 24 (+/- 6) months old (mode 2, 1998; mode 3, 1997). Fig. 9 is organized so that mode 2 for 1997 (1+ cohort) and 1998 (2+ cohort) represents fish from the same spawning event. Survivors of a 3+ cohort, comprising fish 36 (+/- 6) months old, if they exist, do not form a readily distinguishable modal class. The questionable mode 4 in 1997 (absent in 1998) comprises only male specimens (maximum female SL is 139 mm), and therefore possibly represents the male-dominated upper size range of the 2+ cohort. Captive discus typically live for 3-5 years, or exceptionally longer (T. Silva, pers. comm.), but wild specimens are expected to have shorter lives. The single small specimen to the left of the 1a modal class in 1998 is probably a member of a 0+ cohort (<6 months old) from an unusually late spawning event in the previous breeding season.
Some aspects of these data are difficult to interpret. The sampled ratio of females to males was 41:39 for cohorts 1+ and 2+ combined. However the male: female ratio differed between the 1+ cohort (30:20) and 2+ cohort (9:21). The approximately equal ratio of female to male specimens in adjacent shore scrub (Fig. 10) suggests that this sex ratio discrepancy cannot be explained by sex-biased dispersal out of the galhadas, nor are there grounds to suspect different mortality rates between males and females. This discrepancy may instead be an artifact of the relatively small sample sizes. Another curious aspect of the data is that the ratio of 1+ to 2+ fishes changed from 2.9 : 1(138:48) in 1997 to 1.5 : 1 (127:82) in 1998. One explanation for this is that disturbance during the 1997 capture/tagging may have suppressed breeding and resulted in a proportionally smaller 1+cohort in the following year.
The existence of two distinct size modes within the 1+ group (Fig. 9) implies either that discus either do not generate progeny continuously, and/or that the survival of progeny is not constant through the rising water period. Three hypotheses are elaborated here to explain these size modes. The first is that the two modes derive from a single group of females breeding on two separate occasions. Female discus exhibit a multi-modal distribution of egg sizes typical of partial spawners, and in aquaria are known to breed at least twice each year in aquaria (Silva & Kotlar, 1980). After spawning, discus are occupied with the care and defense of their broods for approximately three to four weeks (Matthaeus, 1992). Following the raising of a first brood, there may also be a refractory period in which the parents recuperate. The data in Fig. 11 indicate that reproductive maturity (gonad stages 3-4) of discus within galhadas is attained by fish from both 1+ and 2+ cohorts, but proportionally much more so in the 2+ cohort. Hence, two waves of spawning by 2+ females, separated by this latency period, could, in principle, contribute to a bimodal size distribution of the subsequent 1+ cohort. However, this scenario is unlikely because the observation of multiple states of reproductive maturity in the 2+ cohort (Fig. 11) at the onset of flooding does not predict a synchronized first episode of spawning.
A second hypothesis for the two modes in the size distribution of the 1+ cohort is that they derive from separate episodes of spawning for different age classes of females. For example, 2+ females might spawn earlier in the season, while 1+ females might spawn later in the season as they reach full maturity. However, the observation of multiple states of reproductive maturity in both the 1+ cohort and 2+ cohorts (Fig. 11) suggest that neither cohort would be expected to begin breeding at a given time. Instead, 2+ females are expected to breed repeatedly through the season and 1+ females mostly towards the end.
A third hypothesis for the two size modes in the 1+ progeny assumes that females come into breeding condition at a more or less regular rate through the preceding rising-water period (with 2+ females dominating, especially earlier). Instead, external events in the flood cycle are proposed to either trigger synchronized breeding of females, and/or provoke periods of brood failure or larval mortality. This third hypothesis is expanded below (timing of spawning).
Location of spawning. Pairs of captive discus avoid tank-mates and seek sheltered locations when they spawn - expedients believed to reduce brood cannibalism (Matthaeus, 1992). Two observations suggest that discus do not spawn in crowded galhadas. First, the low proportion of pre-spawning (gonad stage 4) discus inside galhadas relative to those in shore scrub (Fig. 10) and, second, the complete absence of discus eggs on the submerged branches of galhadas hosting discus. In the Tefé region, many cichlids use floating meadows as nurseries (e.g. A. nassa, M. mirificus, and P. scalare, pers. obs.), but adult or juvenile discus were never captured in this habitat. The observation of an egg-guarding pair of discus in shallow shore scrub, along with the capture of several pre-spawning (gonad stage 4) discus (Fig. 10), suggests that this habitat may be the first available spawning substrate.
Timing of spawning. The bulk of discus spawning occurred at the beginning of the rising water period - and not substantially earlier or later. This phenomenon is common in Amazonian fishes (Lowe-McConnell, 1979; Crampton, 1999b). It ensures that the resulting progeny are able to benefit from the abundant food resources and low predator densities of flooded forests, and that they are also well grown at the onset of the subsequent low-water season, when predator densities are at their highest. The cues that provoke spawning and cycles of gonadal maturation and recrudescence in Amazonian fishes have been debated (Kirschbaum, 1975; Schwassmann, 1978; Lowe-McConnell, 1979). Experiments by Kirschbaum (1992) implicated a combination of declining conductivity and the physical action of rainfall as key cues. Kirschbaum & Schugardt (2002) later demonstrated that declining conductivity alone can provoke spawning in gymnotiform and mormyriform electric fishes. In the Tefé region, the peak of discus breeding (November-January) corresponds to the period of rising water when conductivity declines rapidly (Fig. 2). This period does not coincide with the period of maximum rainfall in the Tefé region (February-April). The initial rise of the Amazon and Japurá rivers in the Tefé region is driven largely by rainfall in the upper Amazon basin and Andean piedmont region, and not by local rainfall (Ayres, 1993; Goulding et al., 2003).
The existence of two distinct size modes in the 1+ cohort may be linked to events in the flood cycle (see third hypothesis in Population structure). The Tefé region usually experiences one large repiquete during each rising water period because of asynchrony between the peak flood pulses of the Amazon and Japurá rivers (Ayres, 1993) (see Fig. 2). These repiquetes influence the spawning of discus because, like many cichlids, discus spawn on anchored substrates near the water surface (Crampton, 1999b). Eggs placed near the surface are better supplied with oxygen and require parental vigilance of a smaller volume of water from which predators could attack (Lowe McConnell, 1969). Reports of local fishermen, and personal observations indicate that many cichlids, including discus, and also Astronotus ocellatus (Agassiz), C. monoculus, and H. efasciatus avoid spawning during periods of static or declining water levels. In contrast, they often spawn after a few days of consistently rising water levels following the end of a repiquete. Cichlids that spawn near the surface will also abandon eggs during rapid declines in water level, or lose broods as they become exposed (pers. obs.; Lowe-McConnell, 1969). These considerations suggest that the single large repiquetes at the ends of 1996 and 1997 could have provoked a period of reduced spawning, and/or higher brood failure or larval mortality. Either mechanism would account directly for the observed gap in the size distribution of the subsequent 1+ cohort. Repiquetes were associated with fluctuations in the electrical conductivity of the water during this study (Fig. 2), and changes in conductivity are known to cue spawning (Kirschbaum & Schugardt, 2002). Therefore, conductivity changes following repiquetes could trigger synchronized spawning in discus populations.
Acknowledgements
J. Albert, H. Bleher, T. Hrbek, E. Huusela, H. López Fernandez, R. Lowe-McConnell, J. Ready, T. Silva, and S. Willis shared ideas. For field assistance I thank J. Alves de Oliveira and S. Esashika. C. Paxton provided statistical advice. J. Celso-Malta identified B. cichlae. Funding and support were provided by the Mamirauá Institute, Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (grants 380602/96, 2/381597/97-0), and the UK Department for International Development. Funding during the writing stage was provided by National Science Foundation grant DEB-0614334. Collecting was authorized by IBAMA permits, including 0492/99-12. Animal care followed CNPq guidelines.
Literature Cited
Accepted July 2008
Published December 22, 2008
- Araujo-Lima, C. A. R. M., B. Forsberg, R .Victoria & L. Martinelli. 1986. Energy sources for detritivorous fishes in the Amazon. Science, 234:1256-1258.
- Araujo-Lima, C. A. R. M. & M. Goulding. 1997. So Fruitful a Fish: Ecology, Conservation and Aquaculture of the Amazon's Tambaqui. New York, Columbia University Press.
- Ayres, J. M. 1993. As Matas de Várzea do Mamirauá - Médio Rio Solimões. Brasília, Brazil, Sociedade Civil Mamirauá/CNPq.
- Bagenal, T. B. & F. W. Tesch. 1978. Age and growth. Pp. 101-136. In: Bagenal, T (Ed.). Methods for Assessment of Fish Production in Fresh Water. Oxford, Blackwell Scientific Publications.
- Benedito-Cecilio, E., C. A. R. M. Araujo-Lima, B. R. Forsberg, M. M. Bittencourt & L. A. Martinelli. 2000. Carbon sources of Amazonian fisheries. Fisheries Management and Ecology, 7:305-314.
- Bleher, H. 2006. Bleher's discus. Volume I. Pavia, Italy, Aquapress.
- Bleher, H., K.N. Stölting, W. Salzburger, & A. Meyer. 2007. Revision of the genus Symphysodon Heckel, 1840 (Teleostei: Perciformes: Cichlidae) based on molecular and morphological characters. Aqua, International Journal of Ichthyology, 12(4):133-174.
- Breder, C. M. & D. E. Rosen. 1966. Modes of Reproduction in Fishes. New York, The Natural History Press.
- Chippari-Gomes, A. R., L. C. Gomes, N. P. Lopes, A. L. Val & V. M. Almeida-Val, 2005. Metabolic adjustments in two Amazonian cichlids exposed to hypoxia and anoxia. Journal of Comparative Physiology, B., 141:347-355.
- Correa, S. B., W. G. R. Crampton, L. J. Chapman & J. S. Albert. 2008. Fish assemblages in an upper Amazon floodplain; a comparison of adjacent habitats during the flood season. Journal of Fish Biology, 72:629-644.
- Cox-Fernandes, C. 1997. Lateral migration of fishes in Amazon floodplains. Ecology of Freshwater Fish, 6:36-44.
- Crampton, W. G. R. 1998. Effects of anoxia on the distribution, respiratory strategies and electric signal diversity of gymnotiform fishes. Journal of Fish Biology, 53(Supplement A):307-330.
- Crampton, W. G. R. 1999a. The impact of the ornamental fish trade on the discus Symphysodon aequifasciatus: a case study from the flood plain forests of Estação Ecológica Mamirauá. Pp. 29-44. In: Padoch, C., J. M. Ayres, M. Pinedo-Vasquez & A. Henderson (Eds.). Várzea: Diversity, Conservation and Development of Amazonia's Whitewater Floodplains. New York, New York Botanical Garden Press.
- Crampton, W. G. R. 1999b. Plano de manejo para o uso sustentável de peixes ornamentais na Reserva Mamirauá. Pp. 159-176. In: Queiroz, H. L., & W. G. R Crampton (Eds.). Estratégias para Manejo de Recursos Pesqueiros em Mamirauá. Brasília, Brazil, Sociedade Civil Mamirauá/CNPq.
- Crampton, W. G. R. 1999c. Os peixes da Reserva Mamirauá: diversidade e história natural na planície alagável da Amazônia. Pp. 10-36. In: Queiroz, H. L. & W. G. R Crampton (Eds.). Estratégias para Manejo de Recursos Pesqueiros em Mamirauá. Brasília, Brazil, Sociedade Civil Mamirauá/CNPq.
- Degen, B. 1995. Wild-Caught Discus. Neptune City, N.J., T.F.H. Publications.
- Everhart, W. H., A. W. Eipper & W. D. Young. 1975. Principles of Fishery Science. Ithaca, N.Y., Cornell University Press.
- Farias, I. P., G. Ortí & A. Meyer. 2000. Total evidence: molecules, morphology, and the phylogenetics of cichlid fishes. Journal of Experimental Zoology, 288:76-92.
- Farias, I. P, G. Ortí, I. Sampaio, H. Schneider & A. Meyer. 2001. The cytochrome b gene as a phylogenetic marker: the limits of resolution for analyzing relationships among cichlid fishes. Journal of Molecular Evolution, 53:89-103.
- Farias, I. P., G. Ortí, I. Sampaio, H. Schneider & A. Meyer. 1999. Mitochondrial DNA phylogeny of the family Cichlidae: monophyly and fast molecular evolution of the Neotropical assemblage. Journal of Molecular Evolution, 48:703-711.
- Farias, I. P., H. Schneider & I. Sampaio. 1998. Molecular phylogeny of Neotropical cichlids: the relationships of cichlasomines and heroines. Pp. 499-508. In: Malabarba, L. R., R. R. Reis, R. P. Vari, Z. M. S. Lucena & C. A. S. Lucena (Eds.). Phylogeny and Classification of Neotropical Fishes. Porto Alegre, Brazil, Edipucrs.
- Forsberg, B. R., C. A. R. M. Araujo-Lima, L. A. Martinelli, R. L. Victoria & J. A. Bonassi. 1993. Autotrophic carbon sources for fish of the Central Amazon. Ecology, 74:643-652.
- Frost, W. E. 1943. The natural history of the minnow Phoxinus phoxinus Journal of Animal Ecology, 12:139-162.
- Gomez-Laplaza, L. M. 2002. Social status and investigatory behaviour in the angelfish (Pterophyllum scalare). Behaviour, 139:1469-1490.
- Goodwin, N. B., S. Balshine-Earn & J. Reynolds. 1999. Evolutionary transitions in parental care in cichlid fishes. Proceedings of the Royal Society of London, B., 265:2265-2272.
- Goulding, M. 1980. The Fishes and the Forest. Berkeley, University of California Press.
- Goulding, M., R. Barthem & E. Ferreira. 2003. The Smithsonian Atlas of the Amazon. Washington D.C., The Smithsonian Institution.
- Goulding, M., M. L. Carvalho & E. G. Ferreira. 1988. Rio Negro, Rich Life in Poor Water. Amazonian Diversity and Foodchain Ecology as seen through Fish Communities. The Hague, Netherlands, SPB.
- Granado-Lorencio, C., C. A. R. M. Araujo-Lima & J. Lobón-Cerviá. 2005. Abundance - distribution relationships in fish assembly of the Amazonas floodplain lakes. Ecography, 28:515-520.
- Henderson, P. A., W. D. Hamilton & W. G. R. Crampton. 1998. Evolution and diversity in Amazonian floodplain communities. Pp. 385-419. In: Newbery, D. M., H. H. T. Prins & N. D. Brown (Eds.). Dynamics of Tropical Communities. Oxford, Blackwell Science.
- Hildemann, W. H. 1959. A cichlid fish Symphysodon discus with unique nurture habits. American Naturalist, 93:27-34.
- Hyslop, E. J. 1980. Stomach contents analysis - a review of methods and their application. Journal of Fish Biology, 17:411-429.
- Jearld, J. A. 1983. Age determination. Pp. 301-324. In: Nielsen, L.A., & D. L. Johnson (Eds.). Fisheries Techniques. Bethesda, MD, American Fisheries Society.
- Jepsen, D. B., K. O. Winemiller & D. C. Taphorn. 1997. Temporal patterns of resource partitioning among Cichla species in a Venezuelan blackwater river. Journal of Fish Biology, 51:1085-1108.
- Jepsen, D. B., K. O. Winemiller, D. C. Taphorn & D. R. Olarte. 1999. Age structure and growth of peacock cichlids from rivers and reservoirs of Venezuela. Journal of Fish Biology, 55:433-450.
- Junk, W. J. 1997. General aspects of floodplain ecology with special reference to Amazonian floodplains. Pp. 3-20. In: Junk, W. J. (Ed.). The Central Amazon floodplain: ecology of a pulsing system. Berlin, Springer-Verlag.
- Kirschbaum, F. 1975. Environmental factors control the periodical reproduction of tropical electric fish. Experientia, 31:1159-1160.
- Kirschbaum, F. 1992. Cyclic reproduction of tropical freshwater fishes: comparative experimental aspects. Pp. 115-123. In: Adamek, Z. & M. Flajshans (Eds.). Fish Reproduction 1992, Conference Proceedings. Vodnany, Czechoslovakia.
- Kirschbaum, F., & C. Schugardt. 2002. Reproductive strategies and developmental aspects in mormyrid and gymnotiform fishes. Journal of Physiology-Paris, 96:557-566.
- Kullander, S. O. 1986. The Cichlid Fishes of the Amazon River Drainage of Peru. Stockholm, Swedish Museum of Natural History.
- Kullander, S. O. 1996. Eine weitere Ubersicht der Diskusfische, Gattung Symphysodon Heckel. Pp. 10-16. In: R. Stawikowski (Ed.). DATZ Sonderheft Diskus. Verlag Eugen Ulmer, Stuttgart.
- Kullander, S.O. 1998. A phylogeny and classification of the South American Cichlidae (Teleostei: Perciformes). Pp. 461-498. In: L. R. Malabarba, R. R. Reis, R. P. Vari, Z. M. S. Lucena and C. A. S. Lucena (Eds.). Phylogeny and Classification of Neotropical Fishes. Porto Alegre, Brazil, Edipucrs.
- Lowe-McConnell, R. H. 1969. The cichlid fishes of Guyana, South America, with notes on their ecology and breeding behaviour. Zoological Journal of the Linnean Society, 48:255-302.
- Lowe-McConnell, R. H. 1979. Ecological aspects of seasonality in fishes of tropical waters. Symposia of the Zoological Society of London, 44:219-241.
- Lundberg, J. G., L. G. Marshall, J. Guerrero, B. Horton, M. C. S. L. Malabarba & F. Wesselingh. 1998. The Stage for Neotropical Fish Diversification: A History of Tropical South American Rivers. Pp. 13-48. In: L. R. Malabarba, R. R. Reis, R. P. Vari, Z. M. S. Lucena, and C. A. S. Lucena (Eds.). Phylogeny and Classification of Neotropical Fishes. Edipucrs, Porto Alegre.
- Lyons, E. 1959. Symphysodon discus tarzoo. New blue discus electrify aquarium world. Tropicals Magazine, 4:6-8,10.
- Magurran, A. E., W. Oulton & T. J. Pitcher. 1985. Vigilant behaviour and shoal size in minnows. Zeitschrift für Tierpyschologie, 67:167-178.
- Magurran, A. E. & H. L. Queiroz. 2003. Partner choice in piranha shoals. Behaviour, 140:289-299.
- Matthaeus, W. 1992. Observations on the behaviour of Symphysodon aequifasciatus Freshwater and Marine Aquarium, 15:12-16.
- Mayland, H. J. 1994. Adventures with Discus. Neptune City, N.J., T.F.H. Publications.
- Maynard Smith, J. & D. Harper. 2003. Animal Signals. Oxford, Oxford University Press.
- Mesquita, D. R., J. I. R. Porto & E. Feldberg. 2008. Chromosomal variability in the wild ornamental species of Symphysodon (Perciformes: Cichlidae) from Amazon. Neotropical Ichthyology, 6(2):181-190.
- Nikolsky, G. V. 1963. The Ecology of Fishes. Academic Press, London.
- Petry, P., P. B. Bayley & D. F. Markle. 2003. Relationships between fish assemblages, macrophytes and environmental gradients in the Amazon River floodplain. Journal of Fish Biology, 63:547-579.
- Pitcher, T. J. 1986. Functions of shoaling behaviour in teleosts. Pp. 294-338. In: Pitcher, T. J. (Ed.). The Behaviour of Teleost Fishes. London, Croom Helm.
- Queiroz, H. L. & A. Magurran. 2005. Safety in numbers? Shoaling behaviour of the Amazonian red-bellied piranha. Biology Letters, 1:155-157.
- Ready, J. S., E. J. G. Ferreira & S. O. Kullander. 2006. Discus fishes: mitochondrial DNA evidence for a phylogeographic barrier in the Amazon genus Symphysodon (Teleostei: Cichlidae). Journal of Fish Biology, 69(Supplement B):200-211.
- Schultz, L. P. 1960. A review of the pompadour or discus fishes, genus Symphysodon of South America. Tropical Fish Hobbyist, 8:5-17.
- Schwassmann, H. O. 1978. Times of annual spawning and reproductive strategies in Amazonian fishes. Pp. 187-200. In: Thorpe, J.E. (Ed.). Rhythmic Activity of Fishes. London, Academic Press.
- Silva, T. & B. Kotlar. 1980. Discus. Neptune City, N.J., T.F.H. Publications.
- Southwood, T. R. E., & Henderson, P. A. 2000. Ecological Methods. Third Edition. Oxford, Blackwell Science.
- Swynnerton, G. H. & E. B. Worthington. 1940. Notes on the food of fish in Haweswater (Westmorland). Journal of Animal Ecology, 9:183-187.
- Thatcher, V. E. 1991. Amazon fish parasites. Amazoniana, 11:236-572.
- Trivers, R. L. 1985. Social Evolution. Menlo Park, CA., Benjamin-Cummings.
- Val, A. L. & V. M. F. Almeida-Val. 1995. Fishes of the Amazon and their Environment. New York, Springer.
- Winemiller, K. O., D. C. Taphorn & A. Barbarino-Duque. 1997. Ecology of Cichla (Cichlidae) in two blackwater rivers of southern Venezuela. Copeia, 1997:690-696.
- Zihler, F. 1982. Gross morphology and configuration of digestive tracts of Cichlidae (Teleostei, Perciformes): phylogenetic and functional significance. Netherland Journal of Zoology, 32:544-571.
Publication Dates
-
Publication in this collection
22 Jan 2009 -
Date of issue
Dec 2008
History
-
Accepted
22 Dec 2008 -
Received
July 2008