Abstracts
Amazonspinther dalmata, a new miniature characid from the streams of rio Purus and rio Madeira, right bank tributaries of the rio Amazonas, is described as a new genus and species of the subfamily Cheirodontinae. The unique characters of the new genus and species are the three remarkable black blotches on the base of the dorsal, anal, and caudal fins, the anteriormost proximal radial of the anal fin with an anteriorly extended lamina entering the abdominal cavity, and the extremely elongate caudal peduncle. The reduced number of ventral procurrent caudal-fin rays (7-9), and the hemal spines of only posterior one, two, or sometimes three caudal vertebrae directly articulating with the ventral procurrent caudal-fin rays further diagnoses the new taxon from remaining genera of the tribe Cheirodontini. The results of a phylogenetic analysis strongly support a close relationship between A. dalmata and Spintherobolus in the tribe Cheirodontini on the basis of fifteen unambiguous synapomorphies. Ten of these characters, previously hypothesized as exclusive synapomorphies for Spintherobolus, were now also identified in Amazonspinther, such as the pattern of exposed neuromasts on the head, the presence of a second pseudotympanum anterior to the first pleural rib, and the nearly discoidal coracoid bone. A discussion about the phylogenetic position of the closely related fossil †Megacheirodon to Spintherobolus and Amazonspinther is also provided. Convergent characters shared between Amazonspinther and the characid Priocharax are discussed in relation to miniaturization. Biogeographical implications of the diversification of Amazonspinther, Spintherobolus, and Megacheirodon lineages are discussed.
Phylogeny; Biogeography; Miniaturization; Spintherobolus; Priocharax
Amazonspinther dalmata, um novo caracídeo miniatura de igarapés dos rios Purus e Madeira, afluentes da margem direita do rio Amazonas, é descrito como um novo gênero e nova espécie da subfamília Cheirodontinae. Os caracteres únicos ao novo gênero são as manchas pretas conspícuas na base das nadadeiras dorsal, anal e caudal, o radial proximal mais anterior da nadadeira anal com uma lâmina estendida anteriormente entrando na cavidade abdominal e o pedúnculo caudal extremamente alongado. Caracteres adicionais que diagnosticam o novo táxon dos demais gêneros da tribo Cheirodontini incluem o número reduzido de raios procorrentes caudais (7-9) e os espinhos hemais de somente uma, duas, ou às vezes três das vértebras caudais, diretamente articulados com os raios procorrentes caudais ventrais. Os resultados de uma análise filogenética suportam fortemente uma relação de proximidade entre A. dalmata e Spintherobolus na tribo Cheirodontini com base em quinze sinapomorfias não ambíguas. Dez destes caracteres foram previamente propostos como sinapomorfias exclusivas de Spintherobolus, mas agora são reconhecidos também em Amazonspinther, e.g. o padrão de distribuição de neuromastos expostos na cabeça, o segundo pseudotímpano anterior à primeira costela pleural e o coracóide aproximadamente discóide. Uma discussão a respeito da posição filogenética do fóssil †Megacheirodon, proximamente relacionado a Spintherobolus e Amazonspinther, também é fornecida. Caracteres convergentes entre Amazonspinther e o caracídeo Priocharax são discutidos com relação à miniaturização. Implicações biogeográficas da diversificação das linhagens Amazonspinther, Spintherobolus e Megacheirodon são discutidas.
A new genus and species of characid fish from the Amazon basin - the recognition of a relictual lineage of characid fishes (Ostariophysi: Cheirodontinae: Cheirodontini)
Cristina M. BührnheimI,II; Tiago P. CarvalhoIII; Luiz R. MalabarbaIV; Stanley H. WeitzmanV
IUniversidade Federal do Amazonas, ICB, Lab. Zoologia, Av. Gen. Rodrigo Otávio Jordão Ramos, 3000, 69077-000 Manaus, AM, Brazil. cmbuhrn@yahoo.com.br
IIUniversidade do Estado do Amazonas, ENS, Av. Djalma Batista, 2470, 69050-010 Manaus, AM, Brazil
IIIUniversity of Louisiana at Lafayette, Department of Biology, P.O. Box 42451, Lafayette, LA 70504, USA. tiagobio2002@yahoo.com.br
IVUniversidade Federal do Rio Grande do Sul, Departamento de Zoologia, Av. Bento Gonçalves, 9500, 91501-970 Porto Alegre, RS, Brazil. malabarb@ufrgs.br
VNational Museum of Natural History, Division of Fishes, Department of Zoology, MRC 0159, PO Box 37012, Smithsonian Institution, Washington, D. C., 200013-7013, USA. weitzmas@si.edu
ABSTRACT
Amazonspinther dalmata, a new miniature characid from the streams of rio Purus and rio Madeira, right bank tributaries of the rio Amazonas, is described as a new genus and species of the subfamily Cheirodontinae. The unique characters of the new genus and species are the three remarkable black blotches on the base of the dorsal, anal, and caudal fins, the anteriormost proximal radial of the anal fin with an anteriorly extended lamina entering the abdominal cavity, and the extremely elongate caudal peduncle. The reduced number of ventral procurrent caudal-fin rays (7-9), and the hemal spines of only posterior one, two, or sometimes three caudal vertebrae directly articulating with the ventral procurrent caudal-fin rays further diagnoses the new taxon from remaining genera of the tribe Cheirodontini. The results of a phylogenetic analysis strongly support a close relationship between A. dalmata and Spintherobolus in the tribe Cheirodontini on the basis of fifteen unambiguous synapomorphies. Ten of these characters, previously hypothesized as exclusive synapomorphies for Spintherobolus, were now also identified in Amazonspinther, such as the pattern of exposed neuromasts on the head, the presence of a second pseudotympanum anterior to the first pleural rib, and the nearly discoidal coracoid bone. A discussion about the phylogenetic position of the closely related fossil Megacheirodon to Spintherobolus and Amazonspinther is also provided. Convergent characters shared between Amazonspinther and the characid Priocharax are discussed in relation to miniaturization. Biogeographical implications of the diversification of Amazonspinther, Spintherobolus, and Megacheirodon lineages are discussed.
Key words: Phylogeny, Biogeography, Miniaturization, Spintherobolus, Priocharax.
RESUMO
Amazonspinther dalmata, um novo caracídeo miniatura de igarapés dos rios Purus e Madeira, afluentes da margem direita do rio Amazonas, é descrito como um novo gênero e nova espécie da subfamília Cheirodontinae. Os caracteres únicos ao novo gênero são as manchas pretas conspícuas na base das nadadeiras dorsal, anal e caudal, o radial proximal mais anterior da nadadeira anal com uma lâmina estendida anteriormente entrando na cavidade abdominal e o pedúnculo caudal extremamente alongado. Caracteres adicionais que diagnosticam o novo táxon dos demais gêneros da tribo Cheirodontini incluem o número reduzido de raios procorrentes caudais (7-9) e os espinhos hemais de somente uma, duas, ou às vezes três das vértebras caudais, diretamente articulados com os raios procorrentes caudais ventrais. Os resultados de uma análise filogenética suportam fortemente uma relação de proximidade entre A. dalmata e Spintherobolus na tribo Cheirodontini com base em quinze sinapomorfias não ambíguas. Dez destes caracteres foram previamente propostos como sinapomorfias exclusivas de Spintherobolus, mas agora são reconhecidos também em Amazonspinther, e.g. o padrão de distribuição de neuromastos expostos na cabeça, o segundo pseudotímpano anterior à primeira costela pleural e o coracóide aproximadamente discóide. Uma discussão a respeito da posição filogenética do fóssil Megacheirodon, proximamente relacionado a Spintherobolus e Amazonspinther, também é fornecida. Caracteres convergentes entre Amazonspinther e o caracídeo Priocharax são discutidos com relação à miniaturização. Implicações biogeográficas da diversificação das linhagens Amazonspinther, Spintherobolus e Megacheirodon são discutidas.
Introduction
During an expedition made in 2004 along several southern streams of the rio Amazonas, a new characid species was caught in small streams of the rio Madeira and rio Purus. The fish was promptly recognized as a new species due to three black blotches on the body, unknown in other characid species. Investigation of the phylogenetic relationships of this species allows us to reconstruct sister taxa relationships to Spintherobolus Eigenmann, 1911, of the subfamily Cheirodontinae, a genus phylogenetically diagnosed by Weitzman & Malabarba (1999).
The new genus and species are herein described along with a presentation of a new phylogenetic analysis for the genus Spintherobolus that includes new synapomorphies for the taxon consisting of Spintherobolus and the new genus. Spintherobolus currently contains four species: S. papilliferus Eigenmann, 1911 from the headwaters of rio Tietê, upper rio Paraná basin, and three species distributed in southeastern coastal drainages of Brazil, S. broccae Myers, 1925, S. ankoseion Weitzman & Malabarba, 1999, and S. leptoura Weitzman & Malabarba, 1999. We demonstrate based on a global parsimony analysis of character distribution, that characters shared by the new species and Spintherobolus are homologous and not convergences. We additionally provide a discussion about the implication of the discovery of a closely related Amazonian species concerning evolutionary history of Spintherobolus, in relation to a hypothesis of biogeographical evolution among involved drainages.
Material and Methods
Counts and measurements follow Fink & Weitzman (1974), and were made primarily on the left side of each specimen. Measurements were taken under stereomicroscope with precision of 0.01 mm. Total vertebral number includes the four vertebrae of the Weberian apparatus, and the terminal "half centrum" as outlined by Malabarba & Weitzman (2003). The gill raker at the junction of the ceratobranchial and epibranchial is referred as the posteriormost gill raker of the lower branch of the gill arch, and was included in the count of gill rakers of the lower branch following Bührnheim & Malabarba (2006). Cleared and stained specimens (c&s) were prepared according to Taylor & Van Dyke (1985). Scanning electron micrographs were obtained from first branchial arches of alcohol preserved specimens. Values of meristic traits of the holotype are marked in bold. Values in parentheses along with counts in the description represent number of specimens with the given count. The following institutions provided material for this study: ANSP - Academy of National Sciences of Philadelphia, Philadelphia, USA; INPA - Instituto Nacional de Pesquisas da Amazônia, Manaus, Brazil; MCP - Museu de Ciências e Tecnologia, Porto Alegre, Brazil; MNRJ - Museu Nacional, Rio de Janeiro, Brazil; MZUSP - Museu de Zoologia da Universidade de São Paulo, São Paulo, Brazil.
In our discussions, comments about relationships of taxa are based on the concepts of phylogenetic systematics of Hennig (1966). The phylogenetic analysis was performed using TNT 1.0, 2005 by Goloboff, Farris & Nixon (1999), and Winclada 1.00.08, 1999-2002 by Nixon (2002). We used implicit enumeration, the branch and bound algorithm. All characters are given the same weight and are unordered. The matrix of characters includes data from all Spintherobolus species, putatively related cheirodontines, and outgroup characids. Characters 1-35 are numbered following Weitzman & Malabarba (1999), plus nine new characters numbered sequentially (36-44).
Results
Amazonspinther, new genus
Diagnosis.Amazonspinther is diagnosed among all characid species by the autapomorphic presence of three conspicuous black blotches on the base of the dorsal, anal, and caudal fins (ch. 43; Fig. 1).
Amazonspinther is diagnosed among all genera of the Cheirodontinae by two uniquely derived characters, the anteriormost proximal radial of the anal fin with an anteriorly extended lamina entering the abdominal cavity, between the distal portions of the 12th to 14th pleural ribs (ch. 44; Fig. 2) (vs. short anteriorly extended lamina, not entering the abdominal cavity and not between pleural ribs), and by the extremely elongate caudal peduncle, corresponding to 27.3-30.2% of SL. Caudal peduncle length is comparatively short in cheirodontines, ranging from 11.0 to 19.6% of SL. Spintherobolus papilliferus has an elongate caudal peduncle (21.3-27.0% of SL), but shorter than that observed for Amazonspinther.
Among all genera of the tribe Cheirodontini, Amazonspinther is diagnosed by two features: a small number of ventral procurrent caudal-fin rays (7-9 vs. 11-28; Malabarba, 1998:205-207, 209, ch. 42; Weitzman & Malabarba, 1999:8-9, ch. 5; 11-16 in Spintherobolus species) (Fig. 3); and hemal spines of one, two, or sometimes three posterior caudal vertebrae directly articulating with the ventral procurrent caudal-fin rays (Fig. 3; vs. hemal spines of at least the four posterior caudal vertebrae directly articulating with the ventral procurrent caudal-fin rays).
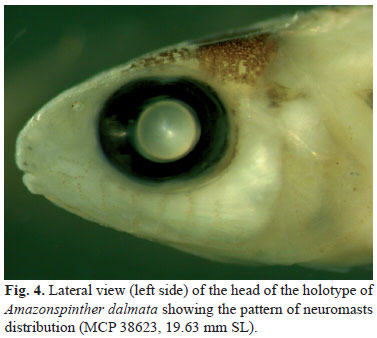
Etymology. Amazon, in reference to the Amazon basin, and spinther from the Greek spinther, masculine, meaning sparks, fire, in reference to both the closely related genus Spintherobolus and to the appearance of the yellow neuromasts of the head, also observed in Spintherobolus (Fig. 4).
Amazonspinther dalmata, new species
Holotype. MCP 38623 (1 unsexed 19.63 mm SL), Brazil, Amazonas, Humaitá-Canutama, stream crossing road Transamazônica about 12 km west of Humaitá to Lábrea, rio Madeira basin, 07º34'25"S 63º06'39"W, 27 July 2004, P. Lehman, F. T. Lima, P. A. Buckup, V. A. Bertaco & J. Pezzi da Silva.
Paratypes. All from Brazil, Amazonas: ANSP 187154 (1 unsexed 13.68 mm SL), INPA 28199 (1 unsexed 13.93 mm SL), MCP 37571 (1 female 14.05 mm SL c&s), MNRJ 31096 (2 unsexed 12.79-13.56 mm SL), collected with holotype. INPA 28659 (2 unsexed 14.49 mm SL & 14.74 mm SL), Autazes, stream of rio Tupana, rio Madeira basin, around 48 km southeast to the municipality of Careiro, 04º09'24"S 60º08'40"W, 3 July 2007, H. M. V. Espírito Santo, A. V. Galuch & D. Barros. INPA 28660 (2 unsexed 16.32 mm SL & 16.73 mm SL), same locality and collectors as INPA 28659, 5 July 2007. INPA 28661 (3 unsexed 14.72-15.41 mm SL), same locality and collectors as INPA 28659, 7 July 2007. INPA 28662 (1 female 19.68 mm SL, 2 unsexed 11.02 mm SL & 12.14 mm SL), Canutama, stream of upper rio Mucuim, about 50 km west of Porto Velho, 08º39'16"S 64º22'02"W, 25 April 2007, F. P. Mendonça & D. Barros. INPA 28663 (1 unsexed 13.86 mm SL), same locality and collectors as INPA 28662, 2 May 2007. INPA 28664 (7 unsexed 11.28-19.79 mm SL), same locality and collectors as INPA 28662, 3 May 2007. INPA 28665 (1 female 15.39 mm, 37 unsexed 12.18-15.77 mm SL), MCP 42017 (1 female 15.73 mm SL, 9 unsexed 12.24-15.82 mm SL), same locality and collectors as INPA 28662, 29 April 2007. MCP 37572, 2 (1 unsexed 14.98 mm SL c&s, 1 unsexed 14.85 mm SL), Canutama, stream of rio Açuá, rio Mucuim drainage, about 136 km southwest of Humaitá on road BR-319, rio Purus basin, 08º12'13"S 63º53'01"W, 28 July 2004, R. Reis, E. Pereira, F. Langeani & A. Cardoso.
Diagnosis. The same as for the genus.
Description. Morphometric and meristic data given in Table 1. Largest specimen 19.79 mm SL, possibly a female. General body shape relatively elongate. Greatest body depth at dorsal-fin origin corresponding to most elevated point in dorsal profile; greatest body depth slightly ahead of dorsal fin in females. Dorsal body profile slightly convex from snout to dorsal-fin origin, slightly concave to caudal peduncle. Ventral profile almost straight from isthmus to anal-fin origin. Anal-fin base slightly concave. Caudal peduncle very long, not deep. Two pseudotympanums, one anterior to first pleural rib and another between first and second pleural ribs (Fig. 1).
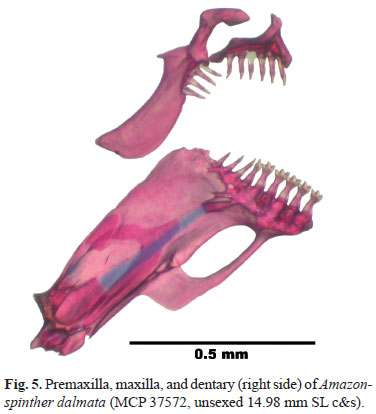
Head short, snout blunt, mouth subterminal. Posterior margin of opercle sinusoidal, with upper portion concave and lower portion convex. Obliquely positioned maxilla; posterior tip at vertical through anterior eye border and below projected longitudinal line through ventral eye border. Dentition examined in two c&s specimens (Fig. 5). Premaxillary teeth 6 or 7, conical. Maxillary teeth 3 or 4, conical. Dentary teeth 7 or 9, 4 or 5 largest tricuspid, 1 bicuspid and 2 or 3 smallest conical.
Dorsal-fin rays ii,9(8). Dorsal-fin origin at middle body length, slightly posterior to vertical through pelvic-fin origin. Posterior margin of dorsal-fin almost straight or slightly concave. Anal-fin rays iii,8(7), 9(1). Anal-fin border concave with 3rd unbranched and anterior 1st-3rd branched rays longer, decreasing moderately in length from 4th-8th branched ray. Pectoral-fin rays i,10(9). Pectoral fin pointed, 1st-3rd branched fin ray longer, lateral margin straight, posterior margin oblique and straight. Pectoral fin reaching to or slightly beyond pelvic-fin origin. Pelvic-fin rays i,5,i(9). Pelvic fin slightly rounded at tip, reaching anal-fin origin. Principal caudal-fin rays 17(1), 18(4), 19(3). Procurrent caudal-fin rays: dorsal 7(2), 8(5), 9(1), ventral 7(4), 8(3), 9(1). Adipose fin at vertical through middle of caudal peduncle. Caudal-fin lobes equal, somewhat pointed. No hooks on fins.
Scales cycloid. Counts estimated on scale pockets: lateral line incomplete with 4(1), 5(5) scales, row of longitudinal scales 32, 34(1); predorsal row 9(6), 10(1); scales between lateral line and dorsal-fin origin 4(8); scales between lateral line and pelvic-fin origin 3(1), 4(2).
Cleared and stained specimens (2): branchiostegal rays 4; supraneurals 4(1); precaudal vertebrae, including Weberian apparatus, 15; caudal vertebrae 18-19, including posterior half centrum. Anteriormost proximal radial of anal fin with an anteriorly extended lamina slightly entering abdominal cavity unique to A. dalmata, among all known Cheirodontinae (Fig. 2). Upper gill rakers 3, short; lower gill rakers absent (Fig. 6a).
Color in alcohol. General ground body color pale beige. Dorsal midline scales with few black chromatophores on scale borders reaching to caudal peduncle. Scarce or no chromatophores on pseudotympanum area; no humeral blotch. Black chromatophores on mid ventral line between anal-fin base termination and caudal peduncle, forming dotted line.
Three conspicuous black blotches on base of dorsal, anal, and caudal fins (Fig. 1). Dorsal blotch formed by black chromatophores on 1st and 2nd unbranched and 1st to 4th branched dorsal-fin rays, positioned on base and middle of fin rays, and laterally on body surface close to dorsal-fin base, forming nearly triangular blotch. Anal blotch formed by black chromatophores on 3rd unbranched and 1st to 7th branched anal-fin rays, on middle of anteriormost fin rays, decreasing in size to base of posterior pigmented branched rays, and laterally on body surface close to anal-fin base forming elongated blotch. Caudal black blotch rounded centered at posterior end of caudal peduncle and base of median caudal-fin rays, not reaching upper and lower border of caudal peduncle. Few scattered black chromatophores sometimes present along 2nd unbranched and 1st branched dorsal-fin rays. Scarce black chromatophores on base of 1st branched pectoral fins and 1st unbranched pelvic fin ray in a few specimens. Adipose fin hyaline. Caudal fin with few scattered black chromatophores on upper and lower lobes, clearer area just behind caudal blotch.
Snout with scarce black chromatophores; few chromatophores on upper lip. Epidermis covering fontanels well-pigmented; epidermis covering frontals and parietals, with deep-lying black chromatophores over brain underneath to frontals and parietals.
Color in life. Body translucent, allowing view of glass bladder and vertebral column (Fig. 7). A greenish metallic yellow iridescent line along vertebral column more evident depending on angle of light incidence. Proximal portion of pleural ribs and opercle iridescent (Fig. 7).
Sexual dimorphism. No external sexual dimorphism was observed. Four sexed specimens are females, two with immature gonads (MCP 37571, 1 c&s, 14.05 mm SL; INPA 28665, 1, 15.39 mm SL), two with mature gonads (MCP 42017, 1, 15.73 mm SL; INPA 28662, 1, 19.68 mm SL). A small incision was made on left side of abdominal region of holotype (19.63 mm SL), but no discernible gonads were found.
Distribution. Amazonspinther dalmata is known from small streams of middle rio Purus (rio Açuá) and middle and lower rio Madeira basins, State of Amazonas, Brazil (Fig. 8).
Ecological notes. The holotype of Amazonspinther dalmata was collected syntopically with several characid species, such as Axelrodia lindeae, Iguanodectes spp., Microschemobrycon geisleri, Tyttocharax madeirae, Gnathocharax steindachneri, Phenacogaster beni, Hemigrammus sp., Knodus sp., and with the crenuchids Odontocharacidium aphanes, Elachocharax pulcher, Ammocryptocharax elegans, and Microcharacidium sp. The type locality was characterized by possessing slow current, muddy substratum, and silty, turbid water. There was abundant riparian vegetation, and the maximum depth was 1.3 m (Fig. 9). The site of collection in the tributary of rio Purus (MCP 37572) had perceptible current, some submerged vegetation, transparent water, sandy and muddy substratum, riparian vegetation, and a maximum depth of 1 m.
Etymology. The epithet refers to the Portuguese word "dalmata", in allusion to the color pattern of the skin of the dogs of the Dalmatian breed which resembles the color of A. dalmata. The origin of this word is linked to the Dalmatia region currently in Croatia, where the dog breed was possibly developed. A noun in apposition.
Phylogeny of Amazonspinther
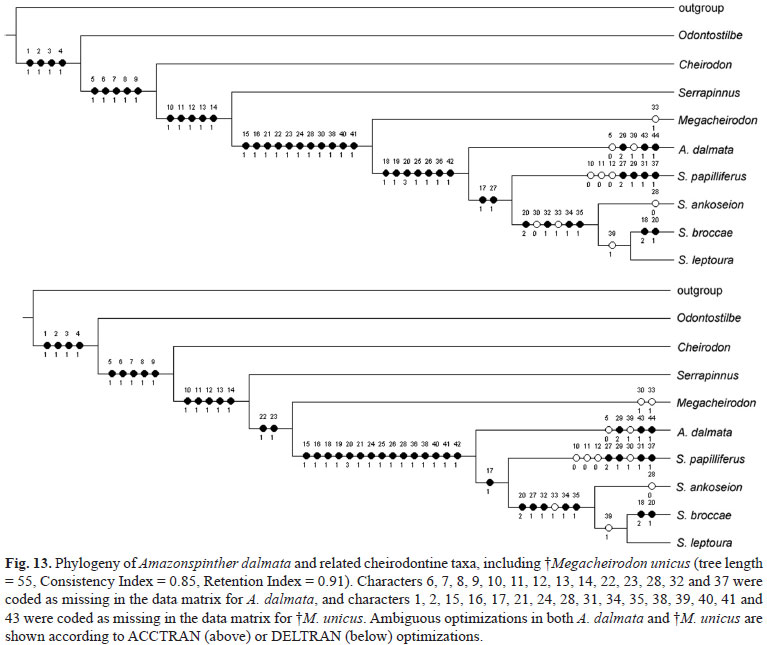
A single most parsimonious cladogram (tree length = 53, Consistency Index = 0.88, Retention Index = 0.92) was obtained from the analysis of 44 characters and 8 taxa (Table 2, excluding the fossil Megacheirodon, discussed below). Amazonspinther was found to be closely related to Spintherobolus, and both genera as sister group to Serrapinnus (Fig. 10). Amazonspinther dalmata is a miniature characid according to Weitzman & Vari's (1988) definition. Those authors stated that miniature characid fish species mature sexually at less than 20 mm SL, and are not reported to exceed 25 to 26 mm SL in the wild. Females of A. dalmata are fully mature at a very small size (15.73 mm SL), and the largest known specimen reached less than 20 mm SL. Mature males, however, were not found among the specimens examined.
The current phylogenetic hypothesis available (Malabarba, 1998) for the small sized cheirodontines is strongly based on secondary sexual characters of males, and the lack of mature males of A. dalmata does not allow the examination of thirteen characters potentially informative to access the relationships of A. dalmata with cheirodontines. Nevertheless the new species is found to share several uniquely derived characters with the species of Spintherobolus as defined by Weitzman & Malabarba (1999), supporting a hypothesis of phylogenetic relationship between A. dalmata and the Neotropical Cheirodontinae.
Coding of characters 6, 7, 8, 9, 10, 11, 12, 13, 14, 22, 23, 32 and 37 below depended on the examination of fully mature males. We performed two distinct analyses, one in which all these characters were coded as missing for A. dalmata, and the second with all these characters coded as the outgroup ("0") for A. dalmata. Character 28, the relatively short pectoral-fin was also coded as missing in the latter analysis, because the average percentuals of SL would fit in state 1, but minimum and maximum percentiles of SL would not (Appendix 1 Appendix 1. Character list. Characters 1-35 are numbered according to Weitzman & Malabarba (1999). Characters 36-44 are new. ). In both analyses, A. dalmata resulted as sister taxon to Spintherobolus (Fig. 10, characters coded as missing). Characters 1 through 35 were extensively described and discussed by Malabarba (1998) and Weitzman & Malabarba (1999), and are presented here in summarized format reporting the respective numeration in those papers (Appendix 1 Appendix 1. Character list. Characters 1-35 are numbered according to Weitzman & Malabarba (1999). Characters 36-44 are new. ).
Monophyly of Amazonspinther dalmata + Spintherobolus
A close relationship between Amazonspinther dalmata and Spintherobolus is strongly supported by fifteen unambiguous synapomorphies, described below. The first ten characters herein analyzed were more extensively discussed by Malabarba (1998) and Weitzman & Malabarba (1999) as synapomorphies for Spintherobolus, and are herein expanded as synapomorphies of A. dalmata + Spintherobolus.
A complex, patterned series of exposed neuromasts is distributed on the head and body (ch. 69 in Malabarba, 1998: 216, see discussion under ch. 15 in Weitzman & Malabarba, 1999: 12-13, figs. 4, 10 and 11). This is a character shared by A. dalmata and Spintherobolus species. Patterns of exposed neuromasts found in A. dalmata are similar and homologous to those of Spintherobolus (compare Fig. 4 here with Weitzman & Malabarba, 1999: 124, 10).
The dentary has a large anterior fenestra (Fig. 5), associated with a large epidermal, papilla-like structure surrounded by a deep groove that has its deep internal portion lodged in the dentary fenestra. The external surface of this papilla bears several exposed neuromasts. The ventral face of the dentary bone, posterior to the fenestra, is concave (ch. 5 in Malabarba, 1998: 216, and ch. 16 in Weitzman & Malabarba, 1999: 128, fig. 12).
Infraorbital bones are reduced in number and possibly fused. Amazonspinther dalmata has the first and second infraorbitals fused and slightly bifurcated anteriorly, and the third infraorbital reduced (Fig. 11). Fourth to sixth infraorbitals are absent, as observed for Spintherobolus (see discussion under ch. 9 in Malabarba, 1998: 216, and ch. 18 in Weitzman & Malabarba, 1999: 128, fig. 13).
There is a small number of pelvic-fin rays, not exceeding six branched rays (see ch. 14 in Malabarba, 1998: 216, and discussion under ch. 19 in Weitzman & Malabarba, 1999: 129, fig. 14). Amazonspinther dalmata has i,5,i and Spintherobolus species i,4,i; i,5; i,5,i or i,6, while remaining cheirodontines show i,7-8 branched rays.
The anal fin has a small number of 9-16 branched rays. This number varies among the species of this clade, with the highest range observed in S. broccae (13-16 branched rays; state 1), an intermediate range in S. ankoseion and S. leptoura (11-14; state 2), and the smallest number found in A. dalmata and S. papilliferus (8-9 and 9-10, respectively; state 3). State 0 corresponds to 16 to 24 branched rays, following Weitzman & Malabarba (1999: character 20; figs. 9 and 15). Reduction in the number of branched anal-fin rays is a synapomorphy of A. dalmata + Spintherobolus. The remarkably reduced number of branched anal-fin rays in A. dalmata and S. papilliferus is the lowest observed in Cheirodontinae but it is most parsimoniously accepted as basal in the clade Amazonspinther + Spintherobolus.
An anterior pseudotympanum lies anterior to the first pleural rib (Fig. 1) (ch. 2 in Malabarba, 1998: 216, and ch. 21 in Weitzman & Malabarba, 1999: 130).
The symphyseal dentary joint surfaces are smooth oval articulations lacking the intercalated and folded bony surfaces found in outgroup characids. The articulation is supported by tough ligamentous tissue (ch. 4 in Malabarba, 1998: 216 and ch. 24 in Weitzman & Malabarba, 1999: 131).
Lateral line is reduced to 2-6 perforated scales (see ch. 60 in Malabarba, 1998: 216, and discussion under ch. 25 in Weitzman & Malabarba, 1999: 131). Amazonspinther dalmata has 4-5 scales, averaging 4.8, similar to that of S. papilliferus (4.6).
The coracoid bone of the pectoral girdle (Fig. 12) is reduced in length, and more or less discoid in shape (ch. 13 in Malabarba, 1998: 216, and ch. 26 in Weitzman & Malabarba, 1999: 131, fig. 18).
The pectoral-fin is relatively short. Weitzman & Malabarba (1999: 132; ch. 28) found this character to be ambiguous, supporting two equally parsimonious hypotheses: the acquisition in a common ancestor to Spintherobolus and a reversal in S. ankoseion, or the independent acquisition in S. papilliferus and in the clade S. broccae + S. leptoura. The presence of a short pectoral fin in A. dalmata (13.82-16.30 % of SL, mean 15.19) supports this character as a synapomorphy of Amazonspinther + Spintherobolus, and the longer pectoral fin of S. ankoseion as autapomorphic and a reversal.
New characters added herein are as follows:
The teeth are conical to tricuspid (ch. 36, Fig. 5). We treat the conical or tricuspid teeth separately and independent of teeth pedunculation (ch. 3). The conical to tricuspid teeth are proposed as a synapomorphy of A. dalmata + Spintherobolus (vs. multicuspid teeth of remaining cheirodontines - except the compsurin Macropsobrycon uruguayanae).
The antorbital of A. dalmata and Spintherobolus species is short and rounded to oval (ch. 38, Fig. 11), instead of elongate, slender and ventrally expanded, as observed in the remaining Cheirodontinae. It resembles that of Carnegiella, Gasteropelecidae (adnasal in Weitzman, 1954). Not checked in S. leptoura.
The gill rakers are short and conical, instead of elongate and lanceolate (ch. 40, Fig. 6).
The gill rakers on the lower branch of the first gill arch are absent or only the posteriormost gill raker at the junction of the ceratobranchial and the epibranchial is present (ch. 41, Fig. 6). Gill rakers on the lower branch of the first gill arch are always present on remaining cheirodontines. First branchial arches of the four known Spintherobolus species were checked for comparisons with A. dalmata. All the species lack gill rakers or have only one gill raker (the posteri rmost) on lower branchial branch, and the gill rakers are conical and the shortest among cheirodontines (Figs. 6a-d). Amazonspinther dalmata has 3 upper gill rakers and none on the lower branch (two c&s specimens) (Fig. 6a). Spintherobolus papilliferus has none (1), 5(4), 6(6), 7(2) upper gill rakers, and one (13) lower gill raker (the posteriormost). The anteriormost gill rakers on upper branch are very short in the specimen photographed (Fig. 6b). Spintherobolus broccae has 1(2), 2(4) on upper gill rakers, and none (2) or 1(4) lower gill rakers (Fig. 6c). Spintherobolus ankoseion has 1(4), 2(2), 3(2), 4(1), 5(2) upper gill rakers and one (11) lower gill raker (the posteriormost, in photographed specimen not visible, damaged) (Fig. 6d). In contrast, Spintherobolus leptoura has 2-3 upper gill rakers, and one lower gill raker (two alcohol specimens examined). For comparison, Cheirodon ibicuhiensis has 7 gill rakers on upper branch and 11 on lower branch, and Serrapinnus heterodon has 5 on upper branch and 13 on lower branch (Fig. 6e-f).
The maxillary shape is irregular, not bearing a smooth dorsal border. Instead, there are two concave sections in the dorsal border of the maxilla separated by a dorsal short projection in the bearing tooth region of the bone (ch. 42, Fig. 5). The anterior arm of the maxilla that articulates to the premaxilla is thicker than usual in the Characidae. Remaining cheirodontines have a smooth dorsal border in the maxilla.
Monophyly of Spintherobolus
Most characters previously used to diagnose Spintherobolus by either Malabarba (1998) or Weitzman & Malabarba (1999) are herein proposed as synapomorphies of Amazonspinther + Spintherobolus. Two characters remain synapomorphic for Spintherobolus: the lack of an adipose fin (Weitzman & Malabarba, 1999: 131, ch. 17), and the relatively small eye. The eyes of Spintherobolus species are remarkably small compared to that of remaining cheirodontines (see discussion in Weitzman & Malabarba, 1999: 131, ch. 27). Amazonspinther dalmata has large horizontal eye diameter 33.0-39.4% of head length compared to 21.3-28.7% observed in Spintherobolus.
The hypothesis of relationships among Spintherobolus species proposed herein (Fig. 10) agrees with that proposed by Weitzman & Malabarba (1999), in which S. papilliferus is the sister species of one clade formed by the other three species of the genus. Additionally, the polytomy formed by S. broccae, S. leptoura, and S. ankoseion (Weitzman & Malabarba, 1999) was resolved in the present study. The low number of gill rakers on the upper branch of the first gill arch (1-3, ch. 39) supports a hypothesis of sister group relationship between S. broccae and S. leptoura (Fig. 10), a character that independently appears in A. dalmata. The higher number of gill rakers observed in S. ankoseion and S. papilliferus is suggested as plesiomorphic.
Remarks on Amazonspinther apomorphic characters
A high number of ventral procurrent caudal-fin rays (11-28) is a synapomorphy for the Cheirodontini, and the small number (7-9) observed in Amazonspinther (Fig. 3) is a reversal (Malabarba, 1998; Bührnheim, 2006). Similarly, the ventral procurrent caudal-fin rays articulating with the hemal spines of at least the four posterior caudal vertebrae is a synapomorphy of the tribe Cheirodontini, and the reduced articulation observed in Amazonspinther (Fig. 3) is a reversal (Weitzman & Malabarba, 1999: 9, ch. 9; fig. 8).
The phylogenetic position of Megacheirodon unicus
Megacheirodon unicus (Travassos & Santos, 1955) is an extinct cheirodontine, previously proposed as sister-group to Spintherobolus by M. C. Malabarba (1998a,b). The species is known only through two fossilized specimens, a female and a highly sexually dimorphic male. Support for its inclusion in the tribe Cheirodontini is given by the apomorphic traits found in both anal and caudal fins of the male specimen.
The inclusion of the fossil in the analysis and the most parsimonious resulting tree allowed the recognition of Megacheirodon as sister group to Spintherobolus + Amazonspinther (Fig. 13), but we have reservations about these results. Three apomorphic traits are shared by M. unicus and Spintherobolus species and are absent in Amazonspinther, and six synapomorphies are shared by Spintherobolus species and Amazonspinther, and are absent in M. unicus. Sixteen characters, however, are missing in the fossilized remains of this fish (1, 2, 15, 16, 17, 21, 24, 28, 31, 34, 35, 38, 39, 40, 41, and 43). Further complications are related to the lack of mature males of Amazonspinther dalmata that resulted in coding 13 characters as missing in this species (6, 7, 8, 9, 10, 11, 12, 13, 14, 22, 23, 32 and 37). Although monophyly of the Clade Megacheirodon + Spintherobolus + Amazonspinther, seems to be strongly supported, the internal relationships among the three genera may change with the addition of the missing information.
So far, characters supporting a close relationship between Spintherobolus and Amazonspinther and that are absent in Megacheirodon unicus (see M. C. Malabarba, 1998 for characters description in M. unicus) are, infraorbital bones reduced or fused (ch. 18); one unbranched and five or six branched pelvic-fin rays (ch. 19); anal fin with iii unbranched and 8-16 branched rays (ch. 20); lateral line with 2 to 6 perforated scales (ch. 25), the coracoid bone of the pectoral girdle discoid in shape (ch. 26), and the teeth conical or with three cusps (ch. 36).
Two synapomorphies are shared by all Spintherobolus species and Megacheirodon unicus, but these are observed only in mature males, and mature males of A. dalmata were unavailable for comparisons. Both characters, however, are related to the fusion and reduced proximal portion of the anteriormost ventral procurrent caudal-fin rays. Since A. dalmata has a small number of ventral procurrent caudal-fin rays, located posteriorly and in the usual position found in other characids (Fig. 3), we expect the following characters are unlikely to occur in A. dalmata: the anterior ventral procurrent caudal-fin rays in adult males, those that have their proximal ends inserted anterior to the hemal spine of the antepenultimate vertebrae, are proximally fused to one another (Weitzman & Malabarba, 1999: 130, ch. 22; fig. 8); and the anterior ventral procurrent caudal-fin rays of males have reduced proximal portions, not rising above the area of fusion between the rays, while the posterior dorsal portions of these rays are fused into a flat compressed plate that inserts between the hemal spine of the antepenultimate vertebra and the hemal spines of the anterior vertebrae (Weitzman & Malabarba, 1999: 131, ch. 23; fig. 8).
One additional apomorphic trait present in Megacheirodon unicus, clearly discernible in the male specimen (M. C. Malabarba, 1998: 195, fig. 3), is the pterygiophore of the sixth branched anal-fin ray, directed dorsally, away from the fifth which is directed anteriorly and in parallel with the pterygiophores anterior to it (Sarraf, 1997: Fig. 6; Weitzman & Malabarba, 1999: character 33; fig. 9). Such character is shared with S. ankoseion, S. leptoura and S. broccae, again suggesting a close relationship of Megacheirodon unicus to Spintherobolus.
Discussion
The proposal of the new genus Amazonspinther is based on the recognition of a new lineage in the Amazon basin, closely related to a group that succeeded in the coastal drainages of southeastern Brazil. The new species is easily identified by the presence of three conspicuous black blotches on each on the base of the dorsal, anal, and caudal fins, not observed in any other characid. It has two other uniquely derived features among all cheirodontines, the anteriormost proximal radial of the anal fin with an anteriorly extended lamina entering the abdominal cavity, and the extremely elongate caudal peduncle, corresponding to 27.3-30.2% of SL. Two additional characters distinguishes A. dalmata in the inclusive tribe Cheirodontini, the small number of ventral procurrent caudal-fin rays (7-9 vs. 11-28), and the hemal spines of only posterior one, two, or sometimes three caudal vertebrae directly articulating with the ventral procurrent caudal-fin rays. Although the close relationship between A. dalmata and Spintherobolus is supported by fifteen unambiguous synapomorphies, the five apomorphic features of A. dalmata distinguish a stem with an unexpected geographical distribution, the Amazon basin. The biogeographical implications of that are further discussed.
Priocharax and Amazonspinther, a case of convergence associated with miniaturization
The genus Priocharax contains two miniature characin species P. ariel and P. pygmaeus (maximum 17 mm SL), that appear morphologically similar to Amazonspinther dalmata. Priocharax species, however, do not have the synapomorphies of cheirodontines (Malabarba, 1998). According to Weitzman & Vari (1987), the apparent derived and non-paedomorphic characters of numerous conic teeth and elongate maxillae in the genus suggest that its relationships lie in the subfamily Characinae of Géry (1977). Priocharax has or appears to have plesiomorphic character states described in various characines and cynopotamines. Among his studies with characine taxa, Lucena (1998) found a clade that includes most of the suggested Characinae genera by Géry (1977). In this clade Priocharax was found to be a basal sister group of the broader clade composed by Gnathocharax, Hoplocharax, Heterocharax, Lonchogenys, Phenacogaster, Cynopotamus, Acestrocephalus, Galeocharax, Acanthocharax, Charax, and Roeboides.
We have compared the Priocharax and Amazonspinther species and found that the external similarity of these species is due to convergence related to miniaturization. Priocharax species do not share the following synapomorphies described above for Amazonspinther dalmata and Spintherobolus: a complex patterned series of exposed neuromasts on the head and body; the teeth elongate and conical or tricuspid; the dentary bearing a large anterior fenestra; the symphyseal dentary joint surfaces smooth oval articulations lacking the intercalated and folded bony surfaces; the coracoid bone reduced in length, and more or less discoid in shape; the relatively short pectoral-fin length; the anal fin with a reduced number of 9-16 branched rays; the gill rakers on upper branch short and conical; the gill rakers on lower branch of the first gill arch absent or with only the posteriormost gill raker at the junction of the ceratobranchial and the epibranchial; and maxilla shape irregular, not bearing a smooth dorsal border.
Similar to Amazonspinther dalmata and Spintherobolus species, however, Priocharax species show infraorbital bones reduced in number, the antorbital short and rounded to oval, an anterior pseudotympanum anterior to the first pleural rib, low number of pelvic-fin rays, not exceeding six branched rays, and lateral line reduced to 2-6 perforated scales (no pored lateral line scales observed in Priocharax). Similar to Spintherobolus species, Priocharax also lacks an adipose fin. By parsimony, and considering characters grouping Amazonspinther to Spintherobolus and Cheirodontinae and characters grouping Priocharax to Characinae, we consider these characters as convergences related to miniaturization. The reductive nature of the absence of the adipose fin, the body size reduced, absent and possibly fused infraorbitals, low number of branched pelvic-rays, and reduction of the lateral line related to the small size were discussed to Spintherobolus by Weitzman & Malabarba (1999). Other reductive characters related to small size not discussed by Weitzman & Malabarba (1999), but common to Priocharax and to the clade Amazonspinther + Spintherobolus are the absence of laterosensory canals on parietal, frontal, and preopercle, discussed in the small size species of Paracheirodon by Weitzman & Fink (1983).
Malabarba & Lucena (1995) and Malabarba (1998:201) have registered the presence of similar and potentially homologous pseudotympanuns in all species of the Cheirodontinae and in the characine genera Phenacogaster, Charax, Roeboides, and Cynopotamus. Malabarba (1998) also stated that "in the absence of additional synapomorphies supporting the Characinae + Cheirodontinae, or at least the Cheirodontinae plus the group formed by Charax, Roeboides, and Phenacogaster as monophyletic, the pseudotympanums of both groups are considered homoplastic." However, phylogenetic hypotheses based on molecular characters supports a close relationship between taxa of the Characinae and Cheirodontinae taxa such as Cynopotamus + Cheirodon (Ortí & Meyer, 1997), and (Exodon + Roeboides) + ((Aphyocheirodon + Cheirodon) + Cheirodontops + Prodontocharax)) (Calcagnotto et al., 2005), and in this case the pseudotympanum of characines and cheirodontines could be interpreted as homologous and a probable synapomorphy grouping both subfamilies. If so, the external resemblance of the miniature characid of the genera Priocharax and Amazonspinther may be related to their common and not long ancestry among characid fishes.
Biogeographic implications of the discovery of Amazon spinther dalmata
The age of the fossil Megacheirodon unicus given by M. C. Malabarba (1998) allow us to hypothesize a minimum age of 30-25 Myr (Late Oligocene-Early Miocene) to the diversification of Spintherobolus, Amazonspinther, and Megacheirodon lineages, according to the phylogenetic hypothesis presented herein.
Previous hypotheses have supported a sister-group relationship of Serrapinnus, a genus widespread in Amazonian, Paraná-Paraguay, São Francisco and other small drainages, to the clade Spintherobolus + Megacheirodon from coastal drainages of southeastern Brazil. Such pattern of sister group relationships usually at genus level between Atlantic coastal drainages and adjacent continental shields in which both sister-clades underwent subsequent radiation was described by Ribeiro (2006) as "Pattern B" and exemplified with Spintherobolus and a few different lineages of fishes. The discovery of Amazonspinther does not affect the classification of Ribeiro (2006) for Spintherobolus, but changes drastically a pattern of sister group with highly diversified and widespread genus (Serrapinnus) to a so far monotypic and apparently relictual species (Amazonspinther dalmata).
Distribution of Spintherobolus, Amazonspinther, and Megacheirodon lineages seems to demonstrate that the clade formed by them has suffered extensive local extirpation. The modern genus Spintherobolus is represented by four species with very limited and allopatric distributional ranges. Three species are found in small Atlantic coastal drainages from Santa Catarina to Rio de Janeiro States, in Brazil, and Spintherobolus papilliferus, is a rare species occurring in a very restricted area, in the headwaters of the rio Tietê. The last species of this Clade, Amazonspinther dalmata, is found considerably far away from eastern Brazil, into the Amazon basin area. There is, consequently, a large gap in the distribution of that clade, with a large extension of the central portion of South America lacking records of either Amazonspinther or Spintherobolus. It seems feasible to hypothesize a relictual pattern of distribution for the clade that probably underwent a process of extinction through much of its former range. Extinction seems still to be a current process in this Clade. All four Spintherobolus species are listed as critically endangered in Brazil due to their very limited, relictual distributional ranges, in areas subject to high level of anthropogenic disturbances.
Otherwise, the discovery of Amazonspinther dalmata may represent the first of new findings of this clade in the Amazon basin. A great part of Neotropical fish fauna has been recognized in the last decades in an accelerated rate of description of new species (Vari & Malabarba, 1998) and several species remain to be discovered. The limited knowledge of the Brazilian Amazonian ichthyofauna, with a few drainages relatively well surveyed, such as the rio Negro basin (Goulding et al., 1988; Chao, 2001), makes difficult the evaluation of its overall biodiversity, as well as the recognition of priority areas for conservation and sustainable use (Barthem, 2001).
Comparative material examined. Cheirodon ibicuhiensis, MCP 13663, 1 male 36.1 mm SL of 52, Brazil, Rio Grande do Sul, Capão da Canoa, pool near channel João Pedro. Serrapinnus heterodon, MZUSP 16740, 1 male 30.2 mm SL of 42, Brazil, São Paulo, Corumbataí, rio Corumbataí. Spintherobolus ankoseion, MCP 19253, paratypes, 4 (1 male 20.7 mm SL, 3 females 24.4-28.1 mm SL), Brazil, Santa Catarina, Barra do Saí, stream in the forest, between Barra do Saí and Itapema, northern Santa Catarina. MCP 19260, 7 (1 female 23.4 mm SL, 6 unsexed 17.1-20.0 mm SL), Brazil, Paraná, Paranaguá, rio Colônia Pereira under bridge on the road Alexandre Matinhos. MCP 38625, 6 (2 females 28.2-28.4 mm SL, 1 female 25.9 mm SL c&s, 2 males 25.4-26.6 mm SL, 1 male 24.4 mm SL c&s), Brazil, Santa Catarina, Ilha de São Francisco, Ribeirão Grande stream, 26º17'36''S 48º35'20''W. Spintherobolus broccae, MCP 19196, 5 (2 males 17.5-17.6 mm SL, 1 male 17.4 mm SL c&s, 3 females 17.9-18.1 mm SL), Brazil, Rio de Janeiro, Cachoeira de Macacu, rio Macacu near town of Cachoeira de Macacu, small tributary about 1-2 km from town, highway bridge over stream. Spintherobolus leptoura, MCP 19254, paratypes, 3 (1 male 19.0 mm SL, 1 female 25.6 mm SL, 1 unsexed 16.5 mm SL), Brazil, São Paulo, Registro, rio Quilombo, Fazenda Dalila. Spintherobolus papilliferus, MZUSP 49408, 4 (1 male 58.2 mm SL, 1 female 50.3 mm SL, 1 female 49.9 mm SL, 1 female not measured c&s), Brazil, São Paulo, Paranapiacaba, rio Tietê basin, last creek on road to Paranapiacaba. MZUSP 51021, 1 female 60.9 mm SL, Brazil, São Paulo, rio Ipiranga, rio Tietê basin. MZUSP 51022, 16 immature 20.6-24.0 mm SL, 1 immature 23.5 mm SL c&s, Brazil, São Paulo, Paranapiacaba and Campo Grande, rio Tietê basin.
Acknowledgements
We thank José L. de Figueiredo and Osvaldo Oyakawa (MZUSP) for loaning specimens. Specimens of the new species were caught during the Transcontinental Expedition and supported by the All Catfish Species Inventory (NSF DEB 0315963). Specimens were also acquired through GEOMA project/INPA collections carried out by Jansen Zuanon, Fernando Mendonça (live specimens photo), Helder Espírito Santo, André Galuch and Daniela Barros. We thank their collaboration to this study. SEM micrographs were taken at "Centro de Microscopia e Microanálises" - PUCRS and "Centro de Microscopia Eletrônica" - UFRGS.
Literature Cited
Accepted December 2008
Published December 22, 2008
(1) Humeral blotch absent (state 1). Humeral blotch present (state 0).
(2) A characteristic hiatus occurs in musculature of the body wall in the area of the swimbladder between the first and second pleural ribs, exposing the swimbladder (Weitzman & Malabarba, 1999: character 2; figs. 4, 5 & 6) (state 1) A hiatus if present involving different muscles and pleural rib limits (state 0).
(3) Teeth have slight proximal peduncle or pedicle and an expanded and compressed distal region bordered by a few to three cusps (state 1). This is different fromWeitzman & Malabarba (1999: character 3; fig. 7) that coded teeth with proximal peduncle or pedicle and an expanded and compressed distal region bordered by a few to many cusps as state 0. Herein teeth not pedunculated, tooth base enlarged (state 0), and teeth pedunculated, largely expanded, and compressed distally (state 2).
(4) A single series of teeth on the premaxilla (Weitzman & Malabarba, 1999: character 4) (state 1). Two tooth rows or different from state 1 (state 0).
(5) Twelve to twenty-eight ventral procurrent caudal-fin rays in both males and females (Weitzman & Malabarba, 1999: character 5) (state 1). Five-eleven ventral procurrent caudal-fin rays in both males and females (state 0).
(6) The ventral procurrent caudal-fin rays of males are elongate and the ray halves of each side are fused their entire length (Weitzman & Malabarba, 1999: character 6) (state 1). Ventral procurrent caudal-fin rays of males with the ray halves forms a V shape bone in frontal view with its two halves fused only distally (state 0). Tested with state 0 and ? in the matrix.
(7) The anteriormost ventral procurrent caudal-fin rays of the females have the proximal portions of their ray-halves fused to each other, but retain an opening near their distal tips, giving a needle-like shape to these rays (Weitzman & Malabarba, 1999: character 7) (state 1). Females have the entire lengths of their ray halves separate (state 0). Tested with state 0 and ? in the matrix.
(8) Several of the anteriormost ventral procurrent caudal-fin rays of males project through the muscles at which point they are covered only by extremely thin skin, thus they can be easily seen along the ventral margin of the caudal peduncle (Weitzman & Malabarba, 1999: character 8; fig. 8) (state 1). Anterior ventral procurrent caudal-fin rays buried in the muscles and skin and are not visible along the ventral surface of the caudal peduncle in males (state 0). Tested with state 0 and ? in the matrix.
(9) Males have the hemal spines of four or more posterior caudal vertebrae anterior to the terminal "half centrum" and its processes elongated and associated with the ventral procurrent caudal-fin rays support (Weitzman & Malabarba, 1999: character 9; fig. 8) (state 1). Only the posterior one, two, or sometimes three caudal vertebrae directly articulating with the ventral procurrent caudal-fin rays in males (state 0). Tested with state 0 and ? in the matrix.
(10) In males the anterior branched anal-fin rays 1 through 5-8 are slab shaped and five to eight times more expanded in the sagittal plane than comparable rays in the females (Weitzman & Malabarba, 1999: character 10; fig. 9) (state 1). Anal-fin rays in males usually circular in cross section and progressively reduced in diameter from the anteriormost to the most posterior one (state 0). Tested with state 0 and ? in the matrix.
(11) The ray segments of the expanded rays progressively fuse to one another as the male specimens become fully mature (Weitzman & Malabarba, 1999: character 11, fig. 9) (state 1). Ray segmentes not fused (state 0). Tested with state 0 and ? in the matrix.
(12) The proximal ends of the anal-fin rays of males have their lepidotrich bases extended anteriorly (Weitzman & Malabarba, 1999: character 12; fig. 9) (state 1). The proximal ends of the anal-fin rays of males lacking an anterior extension. Tested with state 0 and ? in the matrix.
(13) Adult males with two to four (or sometimes five) anal-fin ray hooks on the posterior border of the hook bearing segments (Weitzman & Malabarba, 1999: character 13) (state 1). Only one or two hooks per segment on the posterolateral border of the anal-fin rays I adult males (state 0). Tested with state 0 and ? in the matrix.
(14) Adult males with spatulate ventral procurrent caudal-fin rays that are rounded in profile (Weitzman & Malabarba, 1999: character 14; fig. 8) (state 1). Distal ends of these rays acutely pointed in adult males (state 0).
(15) A complex, patterned series of exposed neuromasts are distributed on the head and body (Weitzman & Malabarba, 1999: character 15, figs. 6, 10 & 11) (state 1). Neuromasts not as state 1 (state 0).
(16) The dentary has a large anterior fenestra, associated with a large epidermal, papilla-like structure surrounded by a deep groove that has its deep internal portion lodged in the dentary fenestra. The external surface of this papilla bears several exposed neuromasts. The ventral face of the dentary bone, posterior to the fenestra, is concave (Weitzman & Malabarba, 1999: character 16; fig. 12) (state 1). Dentary lacking an anterior fenestra or with a different fenestra as state 1 (state 0).
(17) The adipose fin is absent (Weitzman & Malabarba, 1999: character 17) (state 1). Adipose fin present (state 0).
(18) Infraorbital bones reduced and possibly fused (Weitzman & Malabarba, 1999: character 18; figs. 13 a & b) (state 1). Infraorbitals one to six present (state 0).
(19) There are one unbranched and five to six branched pelvic-fin rays (Weitzman & Malabarba, 1999: character 19) (state 1). One unbranched and seven branched pelvic-fin rays (state 0).
(20) The anal fin has iii-iv unbranched and nine to sixteen branched rays (Weitzman & Malabarba, 1999: character 20; figs. 9 & 15) (state 1). Branched anal-fin rays eleven to fourteen branched rays (state 2). Branched anal-fin rays 8--10 (state 3). Branched anal-fin rays at least fifteen branched rays (state 0).
(21) An anterior pseudotympanum lies anterior to the first pleural rib (Weitzman & Malabarba, 1999: character 21) (state 1). None anterior pseudotympanum anterior to the first pleural rib or present but involving different muscle and rib limits than those described in state 1 (state 0).
(22) In adult males the anterior ventral procurrent caudal-fin rays, those that have their proximal ends inserted anterior to the hemal spine of the antepenultimate vertebrae, are fused to one another proximally (Weitzman & Malabarba, 1999: character 22; fig. 8) (state 1). Fused elements absent in males (state 0).Tested with state 0 and ? in the matrix.
(23) The anterior ventral procurrent caudal-fin rays of males have reduced proximal portions, not rising above the area of fusion between the rays, while the posterior dorsal portions of these rays are fused into a flat compressed plate that inserts between the hemal spine of the antepenultimate vertebra and the hemal spines of the anterior vertebrae (Weitzman & Malabarba, 1999: character 23; fig. 8) (state 1). Anterior ventral procurrent caudal-fin rays of males not reduced in proximal portions (state 0). Tested with state 0 and ? in the matrix.
(24) The symphyseal dentary joint surfaces are smooth oval articulations lacking the intercalated and folded bony surfaces found in other characiforms. The articulation is supported by tough ligamentous tissue (Weitzman & Malabarba, 1999: character 24) (state 1). Symphyseal dentary joint with intercalated bony folds (state 0).
(25) Lateral line reduced to 2-6 perforated scales (Weitzman & Malabarba, 1999: character 25) (state 1). Lateral line complete or with higher number of scales than in state 1 (0).
(26) The coracoid bone of the pectoral girdle is reduced in length, and more or less discoid in shape (Weitzman & Malabarba, 1999: character 26; fig 18) (state 1). Elongate coracoids (state 0).
(27) The eyes of Spintherobolus papilliferus (18.4-26.0, in average 21.3 % HL) are the smallest of the four species of Spintherobolus (state 2) (Weitzman & Malabarba, 1999: character 27). Eyes small , S. ankoseion, S. broccae, and S. leptoura, in average 27.2-28.7% HL ( state 1). Relatively large eyes, 25-35 % HL (state 0).
(28) Relatively short pectoral-fin length, 15.2-18.7% SL in S. broccae, S. leptoura, and S. papilliferus (Weitzman & Malabarba, 1999: character 28) (state 1). By parcimonious analysis, A. dalmata, 13.82-16.30 % SL, mean 15.19 (coded as uninformed), fit the short pectoral fin. Relatively long pectoral-fin length, 17.0-20.9% SL (state 0). Tested with state ? in the matrix.
(29) The caudal peduncle length extremely elongate, corresponding to 27.3-30.2% SL (Weitzman & Malabarba, 1999: character 29) (state 2). Elongate caudal peduncle, 21.3-27.0% SL in Spintherobolus papilliferus (state 1). Caudal peduncle length comparatively short, around 11.0 to 19.6% SL, mean 14.3-15.2 (state 0).
(30) Large adult size, reaching at least 60.8 mm SL in Spintherobolus papilliferus, and 51.5-55.0 mm SL in Megacheirodon unicus (Weitzman & Malabarba, 1999: character 30; Malabarba, M. C. 1998b) (state 1). Small adult size, reaching 18 to 28 mm SL (state 0).
(31) High vertebral count of 35-36 (Weitzman & Malabarba, 1999: character 31) (state 1). Comparatively low vertebral count of 32-34 or different from state 1 (state 0).
(32) The ray segments of the posterior branch of the second through fifth anal-fin rays of fully adult males are thickened and have a characteristic somewhat asymmetrical chevron shape in lateral profile (Weitzman & Malabarba, 1999: character 32; figs. 9 & 15) (state 1). Ray segments relatively rectangular, either short or elongate, in males (state 0). Tested with state 0 and ? in the matrix.
(33) The pterygiophore of the sixth branched anal-fin ray in both males and females is directed dorsally, away from the fifth which is directed anteriorly and in parallel with the pterygiophores anterior to it (Weitzman & Malabarba, 1999: character 33; fig. 9) (state 1). Anteriorly directed pterygiophores of the branched anal-fin ray in both males and females (state 0).
(34) A black pigmented area lies ventral to the eyes (Weitzman & Malabarba, 1999: character 34; figs. 2, 4, 29-37) (state 1). None black pigmented area ventral to the eyes or different from state 1 (state 0).
(35) A longitudinal stripe occurs on the body dorsal to the anterior lobe of the anal-fin and extends onto the short posterior branched anal-fin rays (Weitzman & Malabarba, 1999: character 35; figs. 2, 29-37) (state 1). None longitudinal stripe as described in state 1 (state 0).
(36) Number of teeth cusps. Conical to tricuspid teeth (state 1). Teeth with four cusps or more (state 0). Amazonspinther dalmata has conical to tricuspid dentary teeth, intermediated by a bicuspid tooth, and unicuspid premaxillary teeth. In Spintherobolus species there are small lateral cusps near the distal apices of what are essentially teeth having an elongate pedicle and a slightly recurved apical cone; see Weitzman & Malabarba, 1999: Fig. 12. The anterior dentary teeth of Spintherobolus have three small cusps. Teeth bearing these small cusps are more numerous than the strictly conical teeth present in the large species, Spintherobolus papilliferus. The premaxillary teeth are usually conical in Spintherobolus species, but in S. papilliferus they bear some very small lateral cusps in their somewhat laterally expanded distal portions. The maxillary teeth in all species, A. dalmata and Spintherobolus species are conical.
(37) Anal-fin rays of males. The last unbranched and about four branched anal-fin rays slab-shaped, including all the branches expanded forming a typical fan (state 1). Different from the state 1 (state 0). This is an autopomorphy Spintherobolus papilliferus, not verified in A. dalmata. Tested with state 0 and ? in the matrix.
(38) Form of antorbital. Short, rounded to oval (state 1). Form different from state 1, elongate, slender, ventrally expanded, in form of a inverse comma in Cheirodontinae (state 0). The Spintherobolus antorbital is relatively similar to that of Carnegiella, Gasteropelecidae (adnasal in Weitzman, 1954). The Gasteropelecidae is a group of uncertain relationships in Characiformes not sharing the synapomorphies of Cheirodontinae (Malabarba, 1998). Not checked in S. leptoura.
(39) Number of gill rakers on upper branch of first gill arch. Low number of gill rakers, 1-3 (state 1). High number gill rakers, 4-7 (or higher in outgroup) (state 0). The higher number of gill rakers observed in S. ankoseion and S. papilliferus is suggested as plesiomorphic. However, this character is variable in S. ankoseion, reaching 4-5 gill rakers, or 1-2 gill rakers. We assume S. ankoseion is plesiomorphic (Character 39, state 0) because some specimens reach a higher number of gill rakers than A. dalmata, S. broccae and S. leptoura, with only none to 3 gill rakers.
(40) Form of gill rakers. Short in a conical form (state 1). Elongate, lanceolate (state 0).
(41) Gill rakers on lower branch of the first gill arch. Absent or with only the posteriormost gill raker at the junction of the ceratobranchial and the epibranchial (state 1). Present (state 0).
(42) Form of maxilla, dorsal profile. Irregular, with a concave protuberance on middle portion of maxilla, above the inferior border with teeth, slightly convex on posteriormost border (state 1). Almost straight, slightly convex on posteriormost border or not (state 0).
(43) Presence of three conspicuous rounded dark black blotches on the base of dorsal, anal, and caudal fins (ch. 1; Fig. 1) (state 1). None blotches as state 1 (state 0).
(44) The anteriormost proximal radial of anal fin with an anteriorly extended lamina slightly entering abdominal cavity, between distal portions of the 12th to 14th pleural ribs (ch. 2; Fig. 2) (state 1). Short anteriorly extended lamina, not entering the abdominal cavity and not between pleural ribs (state 0).
- Barthem, R. B. 2001. Componente Biota Aquática. Pp. 60-78. In: Biodiversidade na Amazônia brasileira: avaliação e ações prioritárias para a conservação, uso sustentável e repartição de benefícios. Capobianco, J. P. R., A. Veríssimo, A. Moreira, D. Sawyer, I. Santos & L. P. Pinto (Eds.). Estação Liberdade: Instituto Socioambiental, São Paulo, 540p.
- Bührnheim, C. M. 2006. Sistemática de Odontostilbe Cope. 1870 com a proposição de uma nova tribo Odontostilbini e redefinição dos gêneros incertae sedis de Cheirodontinae (Ostariophysi: Characiformes: Characidae). Unpublished Ph. D. dissertation. PUCRS, Porto Alegre, 315p.
- Bührnheim, C. M. & L. R. Malabarba. 2006. Redescription of the type species of Odontostilbe Cope, 1870 (Teleostei: Characidae: Cheirodontinae), and description of three new species from the Amazon basin. Neotropical Ichthyology, 4(2):167-196.
- Calcagnotto, D., S. A. Schaefer & R. DeSalle. 2005. Relationships among characiform fishes inferred from analysis of nuclear and mitochondrial gene sequences. Molecular Phylogenetics and Evolution, 36(1):135-153.
- Chao, N. L. 2001. The fishery, diversity, and conservation of ornamental fishes in the Rio Negro Basin, Brazil - a review of the Project Piaba (1989-99). Pp. 161-204. In: Chao N. L., P. Petry, G. Prang, L. Sonneschien & M. Tlusty (Eds.). Conservation and management of ornamental fish resources of the Rio Negro Basin, Amazonia, Brazil - Project Piaba. EDUA, Manaus, 310p.
- Fink, W. L. & S. H. Weitzman. 1974. The so-called Cheirodontin fishes of Central America with descriptions of two new species (Pisces: Characidae). Smithsonian Contributions to Zoology, 172:1-46.
- Goloboff, P., J. S. Farris & K. Nixon. 1999. TNT: Tree Analysis using new technology. Currently available at http://www.zmuc.dk/public/phylogeny/TNT/
- Goulding, M., M. L. Carvalho & E. G. Ferreira. 1988. Rio Negro, rich life in poor water: Amazonian diversity and foodchain ecology as seen through fish communities, SPB Academic Publishing, The Hague, 200p.
- Lucena, C. A. S. 1998. Relações filogenéticas e definição do gênero Roeboides (Ostariophysi; Characiformes; Characidae). Comunicações do Museu de Ciências e Tecnologia da PUCRS. Série Zoologia, 11:19-59.
- Malabarba, L. R. 1998. Monophyly of the Cheirodontinae, characters and majors clades (Ostariophysi: Characidae). Pp. 193-233. In: Malabarba, L. R., R. E. Reis, R. P. Vari, Z. M. S. Lucena & C. A. S. Lucena (Eds.). Phylogeny and Classification of Neotropical Fishes. Edipucrs, Porto Alegre, 603p.
- Malabarba, L. R. 2003. Subfamily Cheirodontinae (Characins, tetras). Pp. 215-221. In: Reis, R. E., S. O. Kullander & C. Ferraris Jr. (Eds.). Check list of the freshwater fishes of South and Central America. Edipucrs, Porto Alegre, 729p.
- Malabarba, L. R. & Z. M. S. Lucena. 1995. Phenacogaster jancupa, new species, with comments on the relationships and a new diagnosis of the genus (Ostariophysi: Characidae). Ichthyological Exploration of Freshwaters: 6(4):337-344.
- Malabarba, M. C. S. L. 1998a. Megacheirodon, a new fossil genus of characiform fish (Ostariophysi: Characidae) from Tremembé Formation, Tertiary of São Paulo, Brazil. Ichthyological Exploration of Freshwaters, 8(3):193-200.
- Malabarba, M. C. S. L. 1998b. Phylogeny of fossil characiformes and paleobiogeography of the Tremembé Formation, São Paulo, Brazil. Pp 68-84. In: Malabarba, L. R., R. E. Reis, R. P. Vari, Z. M. S. Lucena & C.A.S. Lucena (Eds.). Phylogeny and Classification of Neotropical Fishes. Edipucrs, Porto Alegre, 603p.
- Nixon, K. C. 2002. Winclada version 1.00.08. Published by the Author, Ithaca, New York. Available at http://www.cladistics.com
- Ortí, G. & Meyer, A. 1997. The radiation of Characiformes fishes and the limits of resolution of mitochondrial ribosomal DNA sequences. Systematic Biology, 46(1):75-100.
- Ribeiro, A. C. 2006. Tectonic history and the biogeography of the freshwater fishes from the coastal drainages of eastern Brazil: an example of faunal evolution associated with a divergent continental margin. Neotropical Ichthyology, 4(2):225-246.
- Sarraf, A. 1997. Redescription and distribution of Spintherobolus broccae Myers (Characiformes: Characidae). Revue française d' Aquariologie Herpétologie, 24:27-30.
- Taylor, W. R. & G. C. Van Dyke. 1985. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage. Cybium, 9(2):107-119.
- Vari, R. P. & L. R. Malabarba. 1998. Neotropical ichthyology: an overview. Pp. 1-11. In: Malabarba, L. R., R. E. Reis, R. P. Vari, Z. M. S. Lucena & C. A. S. Lucena (Eds). Edipucrs, Porto Alegre, 603p.
- Weitzman, S. H. 1954. The osteology and relationships of the South American characid fishes of the subfamily Gasteropelecinae. Stanford Ichthyological Bulletin, 4(4):213-263.
- Weitzman, S. H. & S. V. Fink. 1983. Relationships of the neon tetras, a group of South American freshwater fishes (Teleostei: Characidae), with comments on the phylogeny of the New World characiforms. Bulletin of the Museum of Comparative Zoology, 150(6):339-395.
- Weitzman, S. H. & L. R. Malabarba. 1999. Systematics of Spintherobolus (Teleostei: Characidae: Cheirodontinae) from eastern Brazil. Ichthyological Exploration of Freshwaters, 10(1):1-43.
- Weitzman, S. H. & R. P. Vari. 1987. Two new species and a new genus of miniature Characid fishes (Teleostei, Characiformes) from Northern South America. Proceedings of the Biological Society of Washington, 100(3):640-652.
- Weitzman, S. H. & R. P. Vari. 1988. Miniaturization in South American freshwater fishes; an overview and discussion. Proceedings of the Biological Society of Washington, 101(2):444-465.
Appendix 1. Character list. Characters 1-35 are numbered according to Weitzman & Malabarba (1999). Characters 36-44 are new.
Publication Dates
-
Publication in this collection
22 Jan 2009 -
Date of issue
Dec 2008
History
-
Received
Dec 2008 -
Accepted
22 Dec 2008