Abstracts
Lutjanidae, commonly known as snappers, includes 105 species, grouped in four subfamilies. In spite of the high number of species and of its worldwide distribution, the family has been little investigated and the phylogenetic relationships among some of its genera and species are still cause for debate. Only a small number of the species has been cytogenetically analysed. This study reports the first description of the karyotype of Rhomboplites aurorubens as well as data concerning the distribution of the constitutive heterochromatin and the location of the 18S rRNA and the 5S rRNA genes. Specimens of Ocyurus chrysurus from Venezuela were also investigated for the same cytogenetic features. Both species have a 48 uniarmed karyotype, but R. aurorubens has a single subtelocentric chromosome pair, the smallest of the chromosome complement, among the other acrocentric chromosomes. The C-positive heterochromatin is limited to the pericentromeric regions of all chromosomes. Both species show a single chromosome pair bearing the Nucleolus Organizer Regions, but NORs are differently located, in a terminal position on the short arms of the smallest chromosomes in R. aurorubens and in a paracentromeric position in a chromosome pair of large size in O. chrysurus. In O. chrysurus, the 5S rDNA gene cluster is located on a medium-sized chromosome pair, whereas in R. aurorubens it is syntenic with the 18S rDNA gene cluster on chromosome pair number 24. The obtained cytogenetic data, along with previous cytogenetic, morphological and molecular data for the family, reinforce the proposal to synonymize genus Ocyurus with Lutjanus. A review of Lutjanidae cytogenetics is also included.
C-banding; FISH, Karyotype; NORs; Ribosomal genes
Lutjanidae, comumente conhecidos como snappers, inclui 105 espécies, reunidas em quatro subfamílias. A despeito do grande número de espécies e de sua distribuição mundial, a família tem sido pouco estudada e as relações filogenéticas entre alguns de seus gêneros e espécies ainda é motivo de debates. Apenas um pequeno número de espécies foi citogeneticamente analisada. Esse estudo apresenta a primeira descrição do cariótipo de Rhomboplites aurorubens assim como dados relativos à distribuição de heterocromatina constitutiva e localização dos genes 18S rRNA e 5S rRNA. Espécimes de Ocyurus chrysurus da Venezuela foram também analisados quanto às mesmas características citogenéticas. Ambas as espécies têm cariótipos compostos de 48 cromossomos com um único braço, entretanto R. aurorubens tem um único par de cromossomos subtelocêntrico, o menor do complemento cromossômico, entre os outros cromossomos acrocêntricos. A heterocromatina C-positiva é limitada à região pericentromérica de todos os cromossomos. Ambas as species apresentam um único par com Regiões Organizadoras de Nucléolo, mas as RONs são localizadas em posições diferentes, em posição terminal no braço curto dos menores cromossomos de R. aurorubens e em posição paracentromérica no braço longo de um par de cromossomos grandes de O. chrysurus. Em O. chrysurus, os genes 5S rDNA estão localizados em um par de cromossomos de tamanho médio, enquanto em R. aurorubens eles são sintenicamente localizados com os genes 18S rDNA no par de cromossomos número 24. Os dados citogenéticos obtidos, junto com os dados morfológicos e moleculares disponíveis para a família reforçam a proposta de sinonimizar o gênero Ocyurus com Lutjanus. Uma revisão da citogenética dos Lutjanidae é também apresentada
Cytogenetic characterization of Rhomboplites aurorubens and Ocyurus chrysurus, two monotypic genera of Lutjaninae from Cubagua Island, Venezuela, with a review of the cytogenetics of Lutjanidae (Teleostei: Perciformes)
Mauro NirchioI; Claudio OliveiraII; Daniela C. FerreiraII; Rodolfo RondónI; Julio E. PérezIII; Anne Kathrin HettIV; Anna Rita RossiIV; Luciana SolaIV
IEscuela de Ciencias Aplicadas del Mar, Universidad de Oriente, Porlamar, Venezuela. Apartado Postal 147-Porlamar. mnirchio@cantv.net
IIDepartamento de Morfologia, Instituto de Biociências Universidade Estadual Paulista, 18618-000 Botucatu, SP, Brazil
IIIInstituto Oceanografico de Venezuela, Universidad de Oriente, Cumaná, Venezuela
IVDepartment of Human and Animal Biology, University of Rome "La Sapienza", via Borelli 50, 00161 Rome, Italy
ABSTRACT
Lutjanidae, commonly known as snappers, includes 105 species, grouped in four subfamilies. In spite of the high number of species and of its worldwide distribution, the family has been little investigated and the phylogenetic relationships among some of its genera and species are still cause for debate. Only a small number of the species has been cytogenetically analysed. This study reports the first description of the karyotype of Rhomboplites aurorubens as well as data concerning the distribution of the constitutive heterochromatin and the location of the 18S rRNA and the 5S rRNA genes. Specimens of Ocyurus chrysurus from Venezuela were also investigated for the same cytogenetic features. Both species have a 48 uniarmed karyotype, but R. aurorubens has a single subtelocentric chromosome pair, the smallest of the chromosome complement, among the other acrocentric chromosomes. The C-positive heterochromatin is limited to the pericentromeric regions of all chromosomes. Both species show a single chromosome pair bearing the Nucleolus Organizer Regions, but NORs are differently located, in a terminal position on the short arms of the smallest chromosomes in R. aurorubens and in a paracentromeric position in a chromosome pair of large size in O. chrysurus. In O. chrysurus, the 5S rDNA gene cluster is located on a medium-sized chromosome pair, whereas in R. aurorubens it is syntenic with the 18S rDNA gene cluster on chromosome pair number 24. The obtained cytogenetic data, along with previous cytogenetic, morphological and molecular data for the family, reinforce the proposal to synonymize genus Ocyurus with Lutjanus. A review of Lutjanidae cytogenetics is also included.
Key words: C-banding, FISH, Karyotype, NORs, Ribosomal genes.
RESUMO
Lutjanidae, comumente conhecidos como snappers, inclui 105 espécies, reunidas em quatro subfamílias. A despeito do grande número de espécies e de sua distribuição mundial, a família tem sido pouco estudada e as relações filogenéticas entre alguns de seus gêneros e espécies ainda é motivo de debates. Apenas um pequeno número de espécies foi citogeneticamente analisada. Esse estudo apresenta a primeira descrição do cariótipo de Rhomboplites aurorubens assim como dados relativos à distribuição de heterocromatina constitutiva e localização dos genes 18S rRNA e 5S rRNA. Espécimes de Ocyurus chrysurus da Venezuela foram também analisados quanto às mesmas características citogenéticas. Ambas as espécies têm cariótipos compostos de 48 cromossomos com um único braço, entretanto R. aurorubens tem um único par de cromossomos subtelocêntrico, o menor do complemento cromossômico, entre os outros cromossomos acrocêntricos. A heterocromatina C-positiva é limitada à região pericentromérica de todos os cromossomos. Ambas as species apresentam um único par com Regiões Organizadoras de Nucléolo, mas as RONs são localizadas em posições diferentes, em posição terminal no braço curto dos menores cromossomos de R. aurorubens e em posição paracentromérica no braço longo de um par de cromossomos grandes de O. chrysurus. Em O. chrysurus, os genes 5S rDNA estão localizados em um par de cromossomos de tamanho médio, enquanto em R. aurorubens eles são sintenicamente localizados com os genes 18S rDNA no par de cromossomos número 24. Os dados citogenéticos obtidos, junto com os dados morfológicos e moleculares disponíveis para a família reforçam a proposta de sinonimizar o gênero Ocyurus com Lutjanus. Uma revisão da citogenética dos Lutjanidae é também apresentada.
Introduction
Snappers (Perciformes, Lutjanidae) are reef-associated marine fish which are distributed worldwide in the tropical and sub-tropical regions. The family includes 105 species grouped in four subfamilies (Paradichthyinae, Etelinae, Lutjaninae and Apsilinae) and 17 genera (Nelson, 2006). Lutjaninae is the largest subfamily and includes approximately 70 species, grouped in six genera: Lutjanus, with 64 species, Macolor and Pinjalo, with two species each, and three monotypic genera, Hoplopagrus, Ocyurus and Rhomboplites (Nelson, 2006). In spite of the high number of species and its worldwide distribution, the family has been little investigated, and contradictory results have been obtained concerning the phylogenetic relationships and the taxonomic status of some of its genera and species. For example, the validity of the genus Ocyurus has been extensively discussed (Anderson, 1967; Davis & Birdsong, 1973; Domeier & Clarke, 1992; Chow & Walsh, 1992; Loftus, 1992), leading some authors to propose the synonymization of Ocyurus with the genus Lutjanus (Loftus, 1992; Clarke et al., 1997).
Until quite recently, the number of snapper species whose karyotype had been described was limited to four (Rishi, 1973; Choudhury et al., 1979; Raghunath & Prasad, 1980; Ueno & Ojima, 1992) and for three of them only the Giemsa features had been reported. However, in 2008, three different studies added new data, mainly for species of the genus Lutjanus (Nirchio et al., 2008; Rocha & Molina, 2008; Ueno & Takai, 2008), reporting karyotypes as well as several finer cytogenetic features. All these studies have shown that a general chromosome homogeneity occurs within the family, but that derived karyotypes can also be found, whose phylogenetic interpretation remains unclear. Therefore, further data on other snapper species are needed to obtain a more general picture of the karyoevolutive trends in the family.
This study continues a cytogenetic survey of Venezuelan snappers (Nirchio et al., 2008, Table 1) by extending the investigation to the two monotypic genera, Ocyurus and Rhomboplites, which, along with Lutjanus, represent the three genera of Lutjaninae, with a total of 12 species, existing in Venezuela (Cervigón, 1993). The two monotypic species, Ocyurus chrysurus, the yellowtail snapper, and Rhomboplites aurorubens, the vermilion snapper, have an almost overlapping western Atlantic distribution, extending southwards from Massachusetts, USA, and Bermuda to southeastern Brazil, including the Gulf of Mexico and Caribbean Sea (Allen, 1985; Froese & Pauly, 2008). This study reports the first description of the karyotype of R. aurorubens and reports, in both species, data on the distribution of the constitutive heterochromatin and the locations of the 18S rRNA and the 5S rRNA genes obtained by conventional (Giemsa staining, C-banding, silver staining) and molecular techniques (Fluorescent In Situ Hybridization - FISH). A review of the available karyological literature of Lutjanidae is also presented.
Material and Methods
Cytogenetic analyses were performed on nine specimens (five males and four females) of Rhomboplites aurorubens and on four unsexed specimens of Ocyrus chrysurus captured in Cubagua Island, Venezuela. Voucher specimens were deposited at the Ichthyology collection of the Escuela de Ciencias Aplicadas del Mar (ECAM), Universidad de Oriente.
Twenty four hours prior to chromosome preparations, the fish were intramuscularly injected with a yeast glucose solution (Lee & Elder, 1980) for mitosis stimulation. Chromosomes were obtained from kidney cells according to Foresti et al. (1993). C-bands were obtained according to the method described by Sumner (1972). For the detection of the active Nucleolus Organizer Regions (NORs), the chromosome sites where major ribosomal genes -18S, 5.8S and 28S- are clustered, slides were stained with silver nitrate using the method of Howell & Black (1980).
The 18S and 5S rDNA sites were also identified by Fluorescent In Situ Hybridization (FISH), according to the method of Pinkel et al. (1986). A sequence of 1,800 base pairs of the 18S rRNA gene of Oreochromis niloticus (Nile tilapia), cloned in pGEM-T plasmid, was used as a probe to localize sites of 45S rDNA. Polymerase Chain Reaction (PCR) products containing 5S rDNA repeats from O. chrysurus were used as probes for the chromosome mapping of 5S rDNA. DNA was extracted from muscle (Sambrook & Russel, 2001) and the 5S rDNA repeats were generated by PCR with the primers 5SA (5'TAC GCC CGATCT CGT CCG ATC3') and 5SB (5'CAG GCT GGT ATG GCC GTA AGC3') according to Martins & Galetti Jr. (1999).
The 18S rDNA and 5S rDNA probes were labeled by PCR with biotin-14-dATP for the single-FISH experiments. In the case of R. aurorubens, a double-FISH with both probes was carried out by labeling the 18S rDNA with 11-dUTP digoxigenin and 5S rDNA with biotin-14-dATP. Biotin signals were detected and amplified by a two-round application of Avidin-FITC/biotinilated Anti-avidin. Digoxigenin signals were not amplified and were detected by Anti-digoxigenin/ rhodamine. Chromosomes were counter-stained with Propidium Iodide (50 µg/ml) or DAPI diluted in Antifade.
The mitotic figures were photographed using a Motic B400 microscope equipped with a Moticam 5000C digital camera. FISH metaphases were photographed with a photomicroscope Olympus BX61 equipped with a DP70 digital camera.
Results
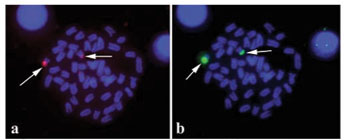
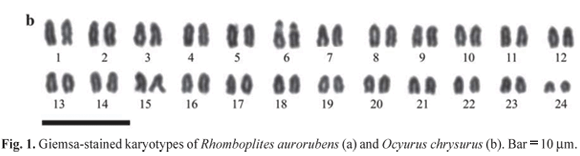
The counts of diploid metaphasic cells revealed a modal diploid number of 2n = 48 uniarmed chromosomes (NF = 48) in both species. However, in R. aurorubens (Fig. 1a) a single subtelocentric chromosome pair, the smallest of the chromosome complement, is present among the acrocentrics, while in O. chrysurus (Fig. 1b) all chromosomes are acrocentric. The chromosomes exhibit very small differences in size, thus, the precise classification of chromosomes in homologous pairs is not possible, with the exception of a large chromosome pair (numbered 6) in O. chrysurus, which shows a conspicuous secondary constriction, and chromosome pair number 24 in both species, clearly the smallest of the chromosome complement.
In both species, C-banding showed C-positive blocks of heterochromatin at the pericentromeric regions of all chromosomes (Fig. 2a-b), which were quite conspicuous in one of the largest chromosome pairs of R. aurorubens (Fig. 2a). In this latter species, the short arms of the subtelocentric chromosome pair 24 are also C-positive (Fig. 2a).
The analysis of the nucleolus organizer regions with the Ag-NOR technique sequential to Giemsa staining (Fig. 3), detected a maximum of two Ag-positive signals in both species. In R. aurorubens, the Ag-positive signals are located along the short arms of chromosome pair 24 (Fig. 3b), which are often heteromorphic and may appear heteropycnotic in Giemsa (Fig. 3a). This heteromorphism is unrelated to sex. In O. chrysurus, the Ag-positive signals are located proximally to centromeres (Fig. 3d), on the secondary constriction evident on chromosomes 6 in Giemsa-stained metaphases (Fig. 3c).
FISH with the 18S rDNA probe confirmed the unique location of NORs on the short arms of chromosome pair 24 in R. aurorubens (Fig. 4a) and in paracentromeric positions of chromosome pair number 6 in O. chrysurus (Fig. 4c). FISH with 5S rDNA produced one hybridization signal close to the centromere of the smallest subtelocentric chromosome pair in R. aurorubens (Fig. 4b) and the centromere of a mediumsized acrocentric chromosome pair in O. chrysurus (Fig. 4d). Thus, the double FISH (Fig. 4a-b) shows that both ribosomal gene clusters are located on the same chromosome pair in R. aurorubens.
Discussion
By adding the chromosome complement of R. aurorubens, reported in this study, to the Lutjanidae database, the number of the species of the family so far cytogenetically analysed rises to 13 (Table 1), out of the approximately 105 recognized species (Nelson, 2006).
The 48 all-acrocentrics karyotype here reported for the examined specimens of O. chrysurus is consistent with data reported by Rocha & Molina (2008) for specimens from the Brazilian north-eastern coast. This karyotype is shared by 10 out of the 13 species so far analyzed (Table 1). The second species reported in this paper, R. aurorubens shows instead a karyotype characterized by the presence of a subtelocentric chromosome pair, which is a novelty among the so far studied species of the family. The remaining two species, Lutjanus quinquelineatus (Ueno & Takai, 2008) and L. synagris (Nirchio et al., 2008), show Robertsonian rearrangements (Table 1), so that a reduction of the diploid number to 47 and a karyotype composed by 46 acrocentric chromosomes and one metacentric chromosome has been observed in males of L. quinquelineatus (Ueno & Takai, 2008), and in two out of the 21 examined specimens of L. synagris (Nirchio et al., 2008). Thus, in the former species, the Robertsonian rearrangement is related to the presence of a chromosomal sex determination mechanism, while in the latter it is apparently unrelated to sex and reflects an intra-specific chromosome polymorphism. It is worth noting that specimens of L. synagris from Brazil (Rocha & Molina, 2008) do not show any biarmed chromosome.
The pattern of the heterochromatin distribution observed in O. chrysurus and R. aurorubens confirms that a limited presence of heterochromatic blocks at the centromeres of all chromosomes is a general characteristic of Lutjanidae, as this pattern is shared by all the 11 species investigated in this sense (Table 1), including those species for which more than one population has been studied, such as L. analis, L. synagris (Nirchio et al., 2008; Rocha & Molina, 2008) and O. chrysurus (Rocha & Molina, 2008; this paper). This evidence suggests that heterochromatinization processes have not played an important role in the karyotypic evolution of Lutjanidae.
Regarding the number and location of the major ribosomal genes, in O. chrysurus and R. aurorubens, the silver staining, which generally detects those NORs which are active in the preceding interphase (Hubbel, 1985; Jimènez et al., 1988; Sánchez-Pina et al., 1984), produced results which overlap with those obtained by FISH with 18S rDNA, i. e., two NOR sites were detected in both species, which show, however, different locations, on different chromosome pairs. It is useful to consider the obtained results together with the whole data set on nucleolar organizer regions in the family, mostly obtained by silver staining, in 11 species (Table 1). Most (nine) of the species show a single chromosome pair coding for major ribosomal genes. The remaining two species, L. griseus and L. jocu, show other variable NOR sites in addition to the main species-specific NOR-bearing chromosome pair.
The precise classification of chromosome pairs is difficult due to the small differences in chromosome size and to the absence of banding. However, in spite of this, at least three different main karyomorphs can be identified with certainty, which are summarized in Fig. 5. A group of seven species shows a large NOR-bearing chromosome pair and the NORs are located in a paracentromeric position. Different authors, in different studies (Table 1) have classified this chromosome pair as number 2, 5 or 6. However, in all species, in the Giemsa-stained metaphases this chromosome pair shows a large secondary constriction, corresponding to the NOR site, which may affect chromosome size and therefore its classification. Thus, pursuing a parsimonious criterion, this pair might be considered homeologous in this group of species (Fig. 5a). Two species, L. synagris and R. aurorubens, show NORs on the smallest chromosome pair, but their location is clearly different, representing therefore the two remaining karyomorphs. In L. synagris (Fig. 5b), NORs are located in a paracentromeric position of chromosome pair 24 (Nirchio et al., 2008; numbered 23 by Rocha & Molina, 2008), whereas in R. aurorubens (Fig. 5c) NORs are located on the short arms of this same chromosome pair, which however is, differently from the other Lutjanidae species, the only one made by subtelocentrics in the chromosome complement.
Considering that the karyomorph of Fig. 5a is the most common in Lutjanidae and widespread in Perciformes, it could be tentatively assumed that this might reflect the plesiomorphic condition in the family from which karyotypes with additional NORs (L. griseus, Nirchio et al., 2008; L. jocu, Rocha & Molina, 2008) or with different locations of NORs (L. synagris, Fig. 5b, Nirchio et al., 2008; R. aurorubens, Fig. 5c, present paper) derived.
As far as the 5S rDNA sites are concerned, by adding the results here reported for O. chrysurus and R. aurorubens, data are available for only five species (Table 1). In O. chrysurus, the minor ribosomal genes show a number and location corresponding to those observed in the other three species that have been investigated so far, Lutjanus analis, L. synagris and L. griseus (Nirchio et al., 2008), i. e., a single site in a paracentromeric position of a medium-sized acrocentric chromosome pair, which might be homeologous in all of them, numbered as 9 in Table 1 and Fig. 5 (a, b). Rhomboplites aurorubens similarly shows a single pair of 5S rDNA bearing chromosomes. However, this pair is certainly different, being the smallest of the chromosome complement, where major ribosomal genes are co-located (Fig. 5c). The syntenic organization of 45S and 5S rDNA loci is quite uncommon in vertebrates and in fish in particular (Martins, 2007).
By framing these cytogenetic data within a systematic and phylogenetic context, some considerations concerning the validity of genus Ocyurus and the relationships among the Lutjaninae species can be made. There is a long term debate regarding the validity of the genus Ocyurus. According to Domeier & Clarke (1992), the morphological characters in Ocyurus, which allows the separation of this genus from Lutjanus, are merely adaptations to a pelagic lifestyle. On the other hand, the numbers of morphological and meristic similarities among these genera are far greater. In addition, Domeier & Clarke (1992) and Loftus (1992), based on the evidence of natural and laboratory hybrids between L. synagris (and likely L. griseus) and O. chrysurus, claimed that Ocyurus probably does not represent a distinct evolutionary lineage from Lutjanus. Finally, according to Clarke et al. (1997), the resemblance of the larval forms also provides further evidence for the synonymization of the two genera.
Similar evidence, suggesting the synonymization of Ocyurus with Lutjanus, was obtained using molecular markers. In fact, in a phylogenetic study based on mitochondrial 12S rRNA and cytochrome b genes sequences of 14 species of snappers occurring in western Atlantic, Sarver et al. (1996) emphasized that the single most-parsimonious tree obtained from analysis of weighted characters placed Ocyurus in a clade with the red snapper group (Lutjanus campechanus and L. vivanus); whereas the strict consensus of the three most-parsimonious trees from the analysis of unweighted characters placed it in a polytomy with several species of Lutjanus and the monotypic genus Rhomboplites. In addition to these, recent mitochondrial data (16S rDNA and cytochrome b) obtained by Miller & Cribb (2007), who investigated phylogenetic relationships of Indo-Pacific snappers (27 species), with the inclusion of three western Atlantic snappers, showed that the genus Lutjanus is paraphyletic.
In this context, O. chrysurus shares the cytogenetic features shown by most of the species of Lutjanus from both the Indo-Pacific and western Atlantic Oceans, while R. aurorubens shows the most derived features. Therefore, the cytogenetic data here reported, while supporting the classification of Rhomboplites as a monotypic genus, do not rule out the inclusion of Ocyurus chrysurus into the genus Lutjanus.
Considering the more general picture of karyotype evolution in Perciformes, the presence of 48 chromosomes has been regarded as a primitive condition for this fish group (Accioly & Molina, 2008; Galetti Jr. et al., 2000, 2006). It can be stated, however, that there can be found both extreme karyotype conservativeness, as for example in the Haemulidae, as well as tendencies for karyotype differentiation, such as in Gobiidae (Molina, 2007). The marine family of Lutjanidae, including the species studied in this work, show a higher level of chromosomal stability, compared to closely related taxa such as Sparidae (Johnson, 1980; Orrell & Carpenter, 2004; Miller & Cribb, 2007), which shows higher levels of chromosomal diversification. Nevertheless, in spite of their morphologically conservative karyotype, when investigated for the finer cytogenetic features, Lutjaninae species were found to have undergone a certain degree of chromosome divergence.
Acknowledgements
Financial support was provided by Consejo de Investigación, Universidad de Oriente, Venezuela, by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) Brazil and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) Brazil and by the Italian Ministry of Instruction, University and Research (MIUR).
Literature Cited
Accepted October 7, 2009
Published December 18, 2009
- Accioly, I. V. & W. F. Molina. 2008. Cytogenetic studies in Brazilian marine Sciaenidae and Sparidae fishes (Perciformes). Genetics and Molecular Research, 7(2): 358-370.
- Allen, G. R. 1985. FAO species catalogue. Volume 6. Snappers of the world. An annotated and illustrated catalogue of lutjanid species known to date. FAO Fisheries Synopsis, 6(125): 1-208.
- Anderson, W. D. Jr. 1967. Field guide to the snappers (Lutjanidae) of the western Atlantic. United States Fish and Wildlife Service Circular, 252: 1-14.
- Cervigón, F. 1993. Los Peces Marinos de Venezuela. Volumen II. Caracas, Fundación Científica Los Roques, 499p.
- Choudhury, R. C., R. Prasad & C. C. Das. 1979. Chromosomes of six species of marine fishes. Caryologia, 32(1): 15-21.
- Chow, S. & P. J. Walsh. 1992. Biochemical and morphometric analyses for phylogenetic relationships between seven snapper species (Subfamily: Lutjaninae) of the western Atlantic. Bulletin of Marine Science, 50: 508-519.
- Clarke, M. E., M. L. Domeier & W. A. Laroche. 1997. Development of larvae and juveniles of the mutton snapper (Lutjanus analis), lane snapper (Lutjanus synagris) and yellowtail snapper (Lutjanus chrysurus). Bulletin of Marine Science, 61(3): 511-537.
- Davis, W. P. & R. S. Birdsong. 1973. Coral reef fishes which forage in the water column. A review of their morphology, behaviour, ecology and evolutionary implications. Helgolaender Wissenschaftliche Meeresuntersuchungen, 24: 292-306.
- Domeier, M. L. & M. E. Clarke. 1992. A laboratory produced hybrid between Lutjanus synagris and Ocyurus chrysurus and a probable hybrid between L. griseus and O. chrysurus (Perciformes: Lutjanidae). Bulletin of Marine Science, 50(3): 501-507.
- Foresti, F., C. Oliveira & L. F. Almeida-Toledo. 1993. A method for chromosome preparations from large specimens of fishes using in vitro short treatment with colchicine. Experientia, 49: 810-813.
- Froese, R. & D. Pauly. 2008. FishBase. World Wide Web electronic publication. Available from: http://www.fishbase.org/ (Accessed in December, 2008).
- Galetti, P. M. Jr., C. T. Aguilar & W. F. Molina. 2000. An overview of marine fish cytogenetics. Hydrobiologia, 420: 55-62.
- Galetti, P. M. Jr., W. F. Molina, P. R. A. M. Affonso & C. T. Aguilar. 2006. Assessing genetic diversity of Brazilian reef fishes by chromosomal and DNA markers. Genetica, 126: 161-177.
- Howell, W. M. & D. A. Black. 1980. Controlled silver staining of nucleolus organizer regions with a protective colloidal developer: a 1-step method. Experientia, 3: 1014-1015.
- Hubbel, H. R. 1985. Silver staining as an indicator of active ribosomal genes. Stain Technology, 60: 285-294.
- Jiménez, R., M. Burgos & R. Diaz de La Guardia. 1988. A study of the silver staining significance in mitotic NORs. Heredity, 60: 125-127.
- Johnson, G. D. 1980. The limits and relationships of the Lutjanidae and associated families. Bulletin Scripps Institution of Oceanography, 24: 1-11
- Lee, M. R. & F. F. B. Elder. 1980. Yeast stimulation of bone marrow mitosis for cytogenetic investigations. Cytogenetics and Cell Genetics, 26: 36-40.
- Loftus, W. F. 1992. Lutjanus ambiguus (Poey), a natural intergeneric hybrid of Ocyurus chrysurus (Bloch) and Lutjanus synagris (Linnaeus). Bulletin of Marine Science, 50: 489-500.
- Martins, C. 2007. Chromosomes and repetitive DNAs: a contribution to the knowledge of fish genome. Pp. 421-454. In: Pisano E., C. Ozouf-Costaz, F. Foresti & B. G. Kapoor (Eds.). Fish Cytogenetics. Enfield, Science Publishers, 502p.
- Martins, C. & P. M. Galetti Jr. 1999. Chromosomal localization of 5S rDNA genes in Leporinus fish (Anostomidae, Characiformes). Chromosome Research, 7(5): 363-367.
- Miller, T. L. & T. H. Cribb. 2007. Phylogenetic relationships of some common Indo-Pacific snappers (Perciformes: Lutjanidae) based on mitochondrial sequences, with comments on the taxonomic position of the Caesioninae. Molecular Phylogenetics and Evolution, 44: 450-460.
- Molina W. F. 2007. Chromosomal changes and stasis in marine fish groups. Pp. 69-110. In: Pisano, E., C. Ozouf-Costaz, F. Foresti & B. G. Kapoor (Eds.). Fish Cytogenetics. Enfield, Science Publishers, 502p.
- Nelson, J. S. 2006. Fishes of the World. New York, John Wiley and Sons Inc., 601p.
- Nirchio, M., R. Rondon, J. E. Pérez, C. Oliveira, I. A. Ferreira, C. Martins, L. Sola & A. R. Rossi. 2008. Cytogenetic studies in three species of Lutjanus (Perciformes: Lutjanidae, Lutjaninae) from the Isla Margarita, Venezuela. Neotropical Ichthyology, 6(1): 101-108.
- Orrell, T. M. & K. E. Carpenter. 2004. A phylogeny of the fish family Sparidae (porgies) inferred from mitochondrial sequence data. Molecular Phylogenetics and Evolution, 32: 425-434.
- Pinkel, D., T. Straume & J. W. Gray. 1986. Cytogenetic analysis using quantitative, high-sensitivity, fluorescence hybridization. Proceedings of the National Academy of Sciences, 83: 2934-2938.
- Raghunath, P. & R. Prasad. 1980. Chromosomes of six marine percoids from the Indian Sea. Indian Biologist, 11: 9-12.
- Rishi, K. K. 1973. A preliminary report on the karyotypes of eighteen marine fishes. Research Bulletin of the Panjab University, 24: 161-162.
- Rocha, E. C. & W. F. Molina. 2008. Cytogenetic analysis in western Atlantic snapper (Perciformes, Lutjanidae). Genetics and Molecular Biology, 31: 461-463.
- Sambrook, J. & D. W. Russell. 2001. Molecular Cloning: A Laboratory Manual. New York, Cold Spring Harbor Laboratory, 999p.
- Sánchez-Pina, M. A., F. J. Medina, M. M. Fernández-Gómez & M. C. Risueno. 1984. Ag-NOR proteins are present when transcription is impaired. Biology of the Cell, 5: 199-202.
- Sarver, S. K., D. W. Freshwater & P. J. Walsh. 1996. Phylogenetic relationships of Western Atlantic snappers (Family Lutjanidae) based on mitochondrial DNA sequences. Copeia, 1996(3): 715-721.
- Sumner, A. T. 1972. A simple technique for demonstrating centromeric heterochromatin. Experimental Cell Research, 75: 304-306.
- Ueno, K. & Y. Ojima. 1992. Notes on the chromosomes of Girella melanichthys and Lutjanus russelli (Pisces, Perciformes). Chromosome Information Service, 52: 3-5
- Ueno, K. & A. Takai. 2008. Multiple sex chromosome system of X1X1X2X2/X1X2Y type in lutjanid fish, Lutjanus quinquelineatus (Perciformes). Genetica, 132: 35-41.
Publication Dates
-
Publication in this collection
18 Jan 2010 -
Date of issue
2009
History
-
Accepted
18 Dec 2009 -
Received
07 Oct 2009