Abstracts
Recently discovered male specimens of Phallotorynus pankalos are described and diagnosed from males of congener species on the basis of gonopodial morphology, color pattern and number of dorsal and anal-fin rays. The phylogenetic hypothesis of relationships among Phallotorynus species is reappraised with the inclusion of P. pankalos. Phallotorynus pankalos is hypothesized as sister to P. jucundus, a clade sister to P. victoriae or to a clade composed of P. dispilos and P. psittakos. Biogeography of Phallotorynus species is discussed, and it is suggested: (1) an early basal split between the rio Paraíba do Sul drainage and the Paraná-Paraguay system, separating P. fasciolatus from the ancestor of the remaining Phallotorynus species followed by a dispersal to rio Paraíba do Sul; (2) that the formation of the Sete Quedas falls may have isolated the ancestors of P. psittakos and P. dispilos. However, the sequence of events associated with the evolutionary history of Phallotorynus remains unclear and unsatisfactory.
Areagram; Biogeography; Rio Iguatemi Basin; Systematics; Upper Rio Paraná Basin
Recentemente descobertos espécimes machos de Phallotorynus pankalos são descritos e diagnosticados dos machos de espécies congêneres com base na morfologia gonopodial, padrão de colorido e número de raios das nadadeiras dorsal e anal. A hipótese de relações filogenéticas entre as espécies de Phallotorynus foi reavaliada com a inclusão de P. pankalos. Phallotorynus pankalos é hipotetizado como irmão de P. jucundus, um clado irmão de P. victoriae ou a um clado composto por P. dispilos e P. psittakos. Biogeografia das espécies de Phallotorynus é discutida, e é sugerido: (1) uma separação basal entre as drenagens do rio Paraíba do Sul e o sistema Paraná-Paraguai, separando P. fasciolatus do ancestral dos demais Phallotorynus, seguida de uma dispersão para o rio Paraíba do Sul; (2) que a formação dos saltos das Sete Quedas podem ter isolado os ancestrais de P. psittakos e P. dispilos. No entanto, a sequência de eventos associados a história evolutiva de Phallotorynus continua obscura e insatisfatória.
Introduction
Phallotorynus Eigenmann, a genus of the cyprinodontiform Poeciliidae, is endemic to southern South American drainages of rio Paraíba do Sul, rio Paraná, and rio Paraguay. Lucinda et al. (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60. proposed Phallotorynus as monophyletic based on seven uniquely derived and unreversed synapomorphies of the cephalic sensory pore system, gonactinostal and gonopodium complexes. These authors recognized six valid Phallotorynus species describing three new species. The description of one of these species, Phallotorynus pankalos Lucinda, Rosa & Reis was based only on females because males were unavailable for study. For this reason, Lucinda & Reis (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60. did not include the species in their phylogenetic analysis of the subfamily Poeciliinae, which was based principally on male morphology.
Recent examination of specimens housed at Coleção Ictiológica do Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (NUP) yielded the discovery of male specimens of Phallotorynus pankalos. The aims of this paper are to formally describe male specimens of P. pankalos and to reappraise the phylogenetic hypothesis of relationships among Phallotorynus species with the inclusion of P. pankalos in the phylogenetic framework of Lucinda & Reis (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60..
Material and Methods
Museum acronyms are: MZUSP, Museu de Zoologia, Universidade de São Paulo, São Paulo; NUP, Coleção Ictiológica do Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura, Universidade Estadual de Maringá, Maringá; and, UNT, Coleção de Ictiologia Sistemática, Universidade Federal do Tocantins, Porto Nacional. Measurements and counts follow Lucinda et al. (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60. and are presented in Table 1. Specimens were cleared and stained following Taylor & van Dyke (1985)Taylor, W. R. & G. C. van Dyke. 1985. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium, 9: 107-119. and are indicated by "cs". Nomenclature of the gonopodium follows Rosen & Gordon (1953)Rosen, D. E. & M. Gordon. 1953. Functional anatomy and evolution of male genitalia in poeciliid fishes. Zoologica, 38: 1-47. and Lucinda & Reis (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60.. Numbers in parentheses following the counts indicate number of specimens for each count. Studied specimens of Phallotorynus pankalos are the ones listed under "Material Examined". Comparative material is as listed by Lucinda & Reis (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60. and Lucinda et al. (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60..
Phylogenetic procedures followed Lucinda & Reis (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60.. A phylogenetic analysis of the Poeciliinae was performed in order to elucidate the phylogenetic position of P. pankalos. A phylogenetic analysis was run with Hennig86 software vers. 1.5 (Farris, 1988Farris, J. S. 1988. Hennig86 reference and documentation for version 1.5. Published by the author, New York, Port Jefferson Station.), using the "ie*" command. This command generates trees by an exact searching algorithm (implicit enumeration) examining all trees, and retaining all best trees (most equally parsimonious trees). The implicit enumeration command "ie*" is guaranteed to find all the most parsimonious trees (Platnick, 1989Platnick, N. I. 1989. An empirical comparison of microcomputer parsimony programs, II. Cladistics, 5: 145-161.). Character optimization followed accelerated transformation model (ACCTRAN) for it is more consistent with the concepts of homology and synapomorphy as discussed by de Pinna (1991)de Pinna, M. C. C. 1991. Concepts and tests of homology in the cladistic paradigm. Cladistics, 7: 367-394..
Outgroups were chosen to be consistent with Lucinda & Reis (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60.. Outgroups are representatives of taxa most closely related to Poeciliinae according to previous phylogenetic hypotheses of relationships among cyprinodontiform fishes (Parenti, 1981Parenti, L. R. 1981. A phylogenetic and biogeographical analysis of Cyprinodontiform fishes (Teleostei, Atherinomorpha). Bulletin of the American Museum of Natural History, 168: 341-557.; Costa, 1998Costa, W. J. E. M. 1998. Phylogeny and classification of the Cyprinodontiformes (Euteleostei: Atherinomorpha): a reappraisal. Pp. 537-560. In: Malabarba, L. R., R. E. Reis, R. P. Vari, Z. M. S. Lucena & C. A. S. Lucena (Eds.). Phylogeny and classification of Neotropical fishes. Porto Alegre, Edipucrs, 603p.; Ghedotti, 2000Ghedotti, M. J. 2000. Phylogenetic analysis and taxonomy of the poeciloid fishes (Teleostei, Cyprinodontiformes). Zoological Journal of the Linnean Society, 130: 1-53.). Outgroups are the following cyprinodontiform species: Fundulus heteroclitus (Linnaeus), Cyprinodon macularius Baird & Girard, Jenynsia unitaenia Ghedotti & Weitzman, Aplocheilichthys spilauchen (Duméril), Fluviphylax pygmaeus (Myers & Carvalho), and Procatopus gracilis Clausen. Outgroup topology was unconstrained as recommended by Nixon & Carpenter (1993)Nixon, K. C. & J. M. Carpenter. 1993. On outgroups. Cladistics, 9: 413-426. and the unrooted networks generated by the phylogeny reconstruction program were rooted on Profundulus labialis (Günther). All transformation series were unordered. Character state assignments in transformation series are based on those presented in the phylogenetic analysis of the Poeciliinae by Lucinda & Reis (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60.. Data gathered from the new taxon were added to the phylogenetic data presented in Lucinda & Reis (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60.. These data are shown in Table 2. All character states were evaluated for the new species and analyzed in the phylogenetic framework of Lucinda & Reis (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60..
States assignments of 144 characters for Phallotorynus pankalos modified the Lucinda & Reis (2005) phylogenetic matrix.
Results
Phallotorynus pankalos Lucinda, Rosa & Reis, 2005
Phallotorynus pankalosLucinda, Rosa & Reis, 2005Lucinda, P. H. F., R. S. Rosa & R. E. Reis. 2005. Systematics and biogeography of the genus Phallotorynus Henn, 1916 (Cyprinodontiformes: Poeciliidae: Poeciliinae), with description of three new species. Copeia, 2005: 609-631.: 624, Fig. 11.
Diagnosis of male specimens. Male specimens of Phallotorynus pankalos can be distinguished from males of its congeners, except P. jucundus Ihering: (1) by the color pattern (six to nine dark brown round spots along lower half of flanks vs. one or two dark brown round to elliptical spots, respectively); (2) by the size of the lateral processes on the trowel-like appendix (large vs. small, respectively); (3) and by left and right halves of the trowel-like appendix united along its whole extension (vs. halves not entirely united and separate by a gap along its extension). A densely pigmented dorsal fin also distinguishes male P. pankalos from the males of remaining species in the genus, except P. jucundus. Males of P. pankalos may be distinguished from males of P. jucundus by the number of dorsal-fin rays (8 vs. 9 or 10, respectively) and by the number of anal-fin rays (eight vs. nine, respectively). Furthermore, males of P. pankalos and P. jucundus may be differentiated by the size of the membranous tip anterior to R4 and R5 (large vs. small, respectively).
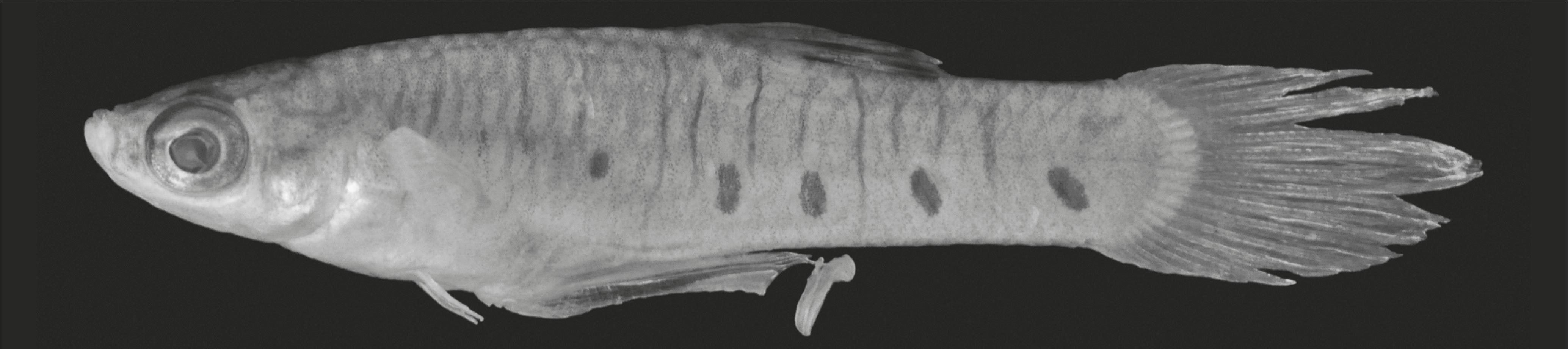
Description of male specimens. Morphometric data in Table 1. Maximum standard length: 19.2 mm SL. Body compressed. Body width in predorsal region uniform and about half body depth. Postdorsal region compressed near caudal peduncle. Dorsal profile of head slightly concave. Predorsal profile convex. Dorsal-fin base oblique. Postdorsal profile slightly concave. Preanal profile convex. Anal-fin base convex; postanal profile nearly straight. Dorsal fin with semi-elliptical border, located posterior to mid-body. Origin of dorsal fin posterior to vertical passing through origin of anal fin. Longest pectoral-fin ray slightly anterior to origin of pelvic fin. Pelvic fin small, almost reaching origin of gonopodium in adult males. Origin of anal fin closer to snout tip than to caudal peduncle. Mouth superior, almost aligned dorsally with base of dorsalmost pectoral-fin ray. Premaxillary and dentary teeth flattened and incisiform with long and narrow pedicel progressively wider towards distal end. Distal end of teeth curved towards oral cavity.
Premaxillary teeth varying from pointed to truncate, superior border varying from acute to oblique.
Dorsal-fin rays: 8(4). Pectoral-fin rays: 10(2), 11(2). Pelvic-fin rays: 5(4). Anal-fin rays: 10(4). Branched caudal-fin rays: 9(2) or 10(2). Predorsal scales: 14(4). Longitudinal series scales: 28(3), 29(1). Scales around caudal peduncle: 16(4). Scales in transverse row: 8(4). Pleural ribs: 14(2). Epipleural ribs: 11(2). Vertebrae: 30(1) or 31(1). Expanded neural processes: 4(2). Premaxillary teeth: 12(3). Dentary teeth: 13(3). Branchiostegal rays: 5(2). Caudal-fin rays attached to hypural plate: 8(2). Upper accessory cartilages between distal neural spines of preural: 2(2). Lower accessory cartilages between distal hemal spines of preural: 2(2).
Gonopodial complex composed of three functional gonapophyses and 11 gonactinosts. Gonactinosts 2, 3, 4, and 5 fused. Gonactinost 5 with winglike expansions. Ligastyle present. Eight gonopodial rays. R1 and R2 unbranched and short. R3 with 31 segments. Segments 19 to 31 abruptly narrower than preceding segments. Segments 19 to 31 progressively narrower and shorter towards tip. Ventral V-shaped projection located at level of segment 17 and 18, and covered by a large membrane forming two conspicuous undulations. First (more basal) undulation approximately twice smaller the second (more distal) one. Last segment of R3 attached to bony style that supports trowel-like appendix. Bony style slightly bent dorsally. Terminal appendix long and narrow, with large lateral processes. Both halves of terminal appendix with straight lateral border and united along its whole extension. R4a with 31 segments. Last distal segment followed by cub-shaped membrane. Thirty segments on R4p; five paired serrae located near tip. Serrae anteriorly directed and gradually shorter towards tip of R4p. Distal half of R6 expanded. Tip of R6 and R7 ankylosed. R7 and R8 small and branched.
Color in alcohol (of male specimens). Eye pupil black. Ground color pale brown, darker on upper half. Border of scales and subjacent skin replete with brown chromatophores, conferring reticulate pattern to body sides, mainly on upper half. Brown chromatophores scattered over whole body, more concentrated on dorsal portion, mainly on head, snout, and ventral surface of lower jaw. Pale brown postorbital band. Dark brown mandibular stripe inconspicuous in some specimens. Dark brown line along predorsal surface. Dorsal-fin membrane black contrasting with hyaline rays. Black band on dorsal fin near its base. Pectoral-fin rays with brown chromatophores. Dark brown line along R3. Caudal-fin rays grayish brown. Six to nine (usually six) dark brown round to irregular spots along lower half of flanks, alternating with narrow dark brown bars that occasionally reach dorsal and ventral profiles.
Phylogenetic relationships. Analysis of the data set resulted in 288 most equally parsimonious trees (MEPT's) (length = 762, CI = 0.35, RI = 0.75) illustrating variation in topology of the ingroup (the Poeciliinae). Concerning the relationships within Phallotorynus, topologies obtained are congruent with those discussed in Lucinda & Reis (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60. and Lucinda et al. (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60. (q.v. fig. 3 and Lucinda et al., 2005Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60.: fig 2). All most parsimonious cladograms indicate P. fasciolatus Henn is sister to all other Phallotorynus species as well as sister relationships between P. jucundus + P. pankalos and P. dispilos Lucinda, Rosa & Reis + P. psittakos Lucinda, Rosa & Reis. However, a strict consensus of the MEPT's results in unresolved relationships among P. victoriae Oliveros, clade [P. jucundus + P. pankalos], and clade [P. dispilos + P. psittakos] (Fig. 3).
Consensus cladogram depicting hypothesis of phylogenetic relationships among Phallotorynus species. Consensus tree resultant from 288 equally most parsimonious cladograms for the ingroup. Length = 762, CI = 0.35, RI = 0.75. Character state assignments, transformation series, optimization and clade numbers follow Lucinda & Reis (2005). Uniquely derived and unreversed features indicated by two asterisks. Support for nodes within Phallotorynus are as follow: Clade 102: 7-0; 8-2**; 70-1; 76-1**; 78-2; 85-3; 89-1**; 100-1**; 102-1**; 104-1**; 110-2**; 136-1; 140-1. Clade 97: 66-1; 135-1**. Clade 90: 136-2**. Clade 84: 20-0; 71-2**; 74-1; 111-2**; 130-1**.
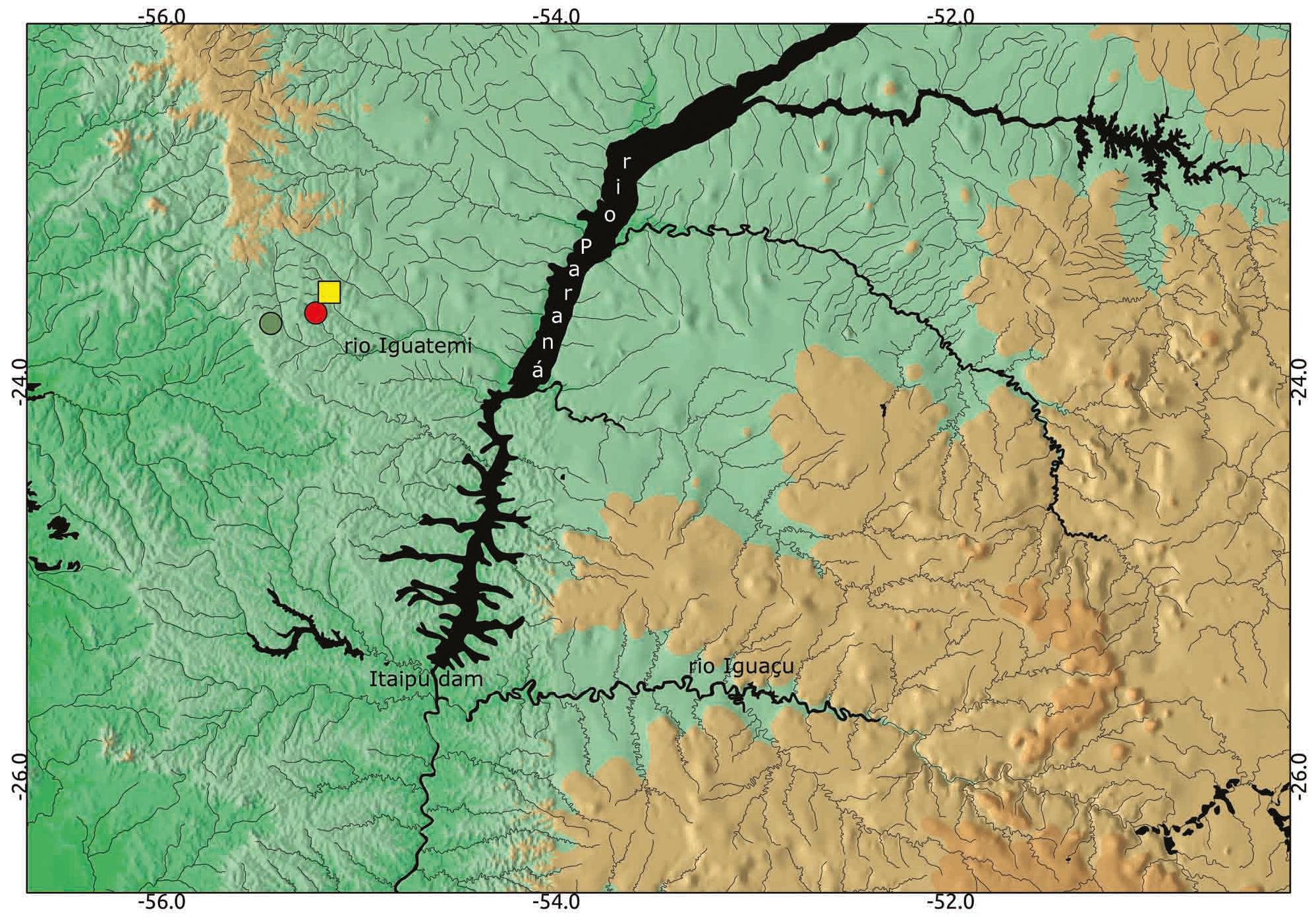
Distribution. Phallotorynus pankalos is known only from its type locality (córrego Sossego, rio Iguatemi drainage, upper portions of rio Paraná system) (Fig. 4). Phallotorynus pankalos has been reported as inhabiting the córrego Mirim and córrego Piraí by Súarez et al. (2009)Súarez, Y. R., J. P. Silva, L. P. Vasconcelos & W. F. Antonialli, Jr. 2009. Ecology of Phallotorynus pankalos (Cyprinodontiformes, Poeciliidae) in a first-order stream of the upper Paraná Basin. Neotropical Ichthyology, 7: 49-54.. We could not confirm this distribution.
Phallotorynus pankalos distribution, córrego Sossego (square, type-locality), córrego Mirim (red dot) and córrego Piraí (green dot).
Ecological Notes. Despite the small number of specimens deposited in fish collections (only 28), Súarez et al. (2009)Súarez, Y. R., J. P. Silva, L. P. Vasconcelos & W. F. Antonialli, Jr. 2009. Ecology of Phallotorynus pankalos (Cyprinodontiformes, Poeciliidae) in a first-order stream of the upper Paraná Basin. Neotropical Ichthyology, 7: 49-54. recorded 2,680 specimens (948 males and 1,732 females) during one year of sampling in three first-order streams in the rio Iguatemi basin (córrego Sossego, córrego Mirim and córrego Piraí). Unfortunately, Súarez et al. (2009)Súarez, Y. R., J. P. Silva, L. P. Vasconcelos & W. F. Antonialli, Jr. 2009. Ecology of Phallotorynus pankalos (Cyprinodontiformes, Poeciliidae) in a first-order stream of the upper Paraná Basin. Neotropical Ichthyology, 7: 49-54.'s voucher specimens are restricted to only one holding with a few specimens (NUP 5839). As a result, most specimens studied by these authors were probably discarded and their identity cannot be confirmed. If the specimens studied by Súarez et al. (2009)Súarez, Y. R., J. P. Silva, L. P. Vasconcelos & W. F. Antonialli, Jr. 2009. Ecology of Phallotorynus pankalos (Cyprinodontiformes, Poeciliidae) in a first-order stream of the upper Paraná Basin. Neotropical Ichthyology, 7: 49-54. are Phallotorynus pankalos, this is an abundant yet restricted species. Therefore, Phallotorynus pankalos is likely a threatened species, because it is known to exist at no more than three locations imperiled by decline of habitat quality, caused mainly by agricultural activities. Furthermore, Súarez et al. (2009)Súarez, Y. R., J. P. Silva, L. P. Vasconcelos & W. F. Antonialli, Jr. 2009. Ecology of Phallotorynus pankalos (Cyprinodontiformes, Poeciliidae) in a first-order stream of the upper Paraná Basin. Neotropical Ichthyology, 7: 49-54. concluded that P. pankalos has small fecundity and higher mortality rate for females after first reproduction. Thus, conservational actions on sites of occurrence of P. pankalos should be considered.
Discussion
The character states exhibited by P. pankalos allow its classification as a member of the monophyletic Phallotorynus as diagnosed by Lucinda et al. (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60.. However, P. pankalos was not included in Lucinda & Reis (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60. phylogenetic analysis (which was primarily based on gonopodial morphology) due to the lack of males. Judging from its distribution, Lucinda et al. (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60. speculated that P. pankalos was more closely related to the clade composed of P. victoriae, P. jucundus, P. dispilos, and P. psittakos, and therefore also originated in upper Paraná. Since males have now become available for study this assumption may be confirmed. Global parsimony of character states in the consensus tree supports P. pankalos as sister to P. jucundus, a clade sister to P. victoriae or to a clade composed of P. dispilos and P. psittakos.
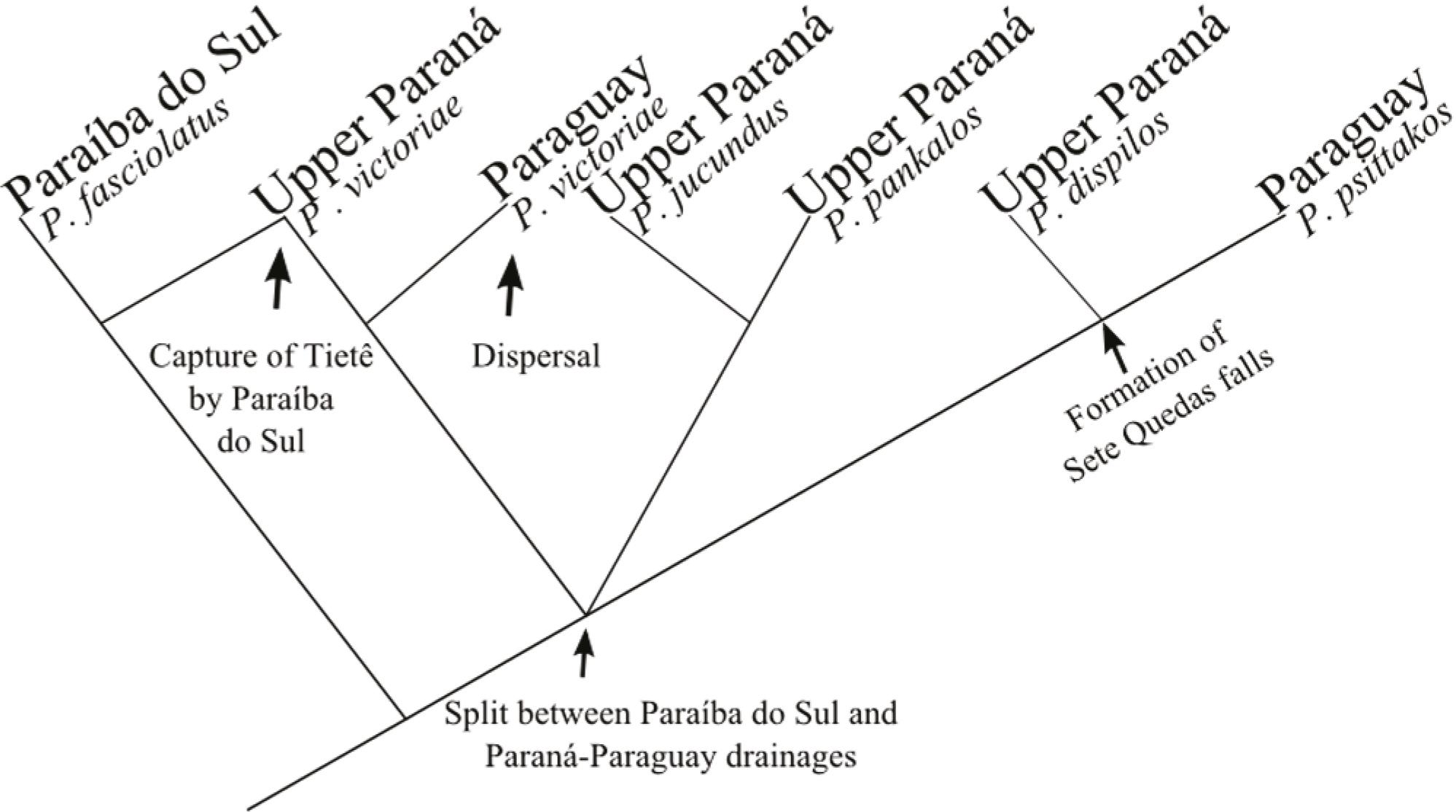
An area cladogram (Fig. 5) (derived from the consensus tree is congruent with previous biogeographic hypothesis (Lucinda et al., 2005Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60.: fig. 12). These authors suggested an early basal split between the rio Paraíba do Sul drainage + upper rio Tietê and the Paraná-Paraguay system, separating P. fasciolatus from the ancestor of the remaining species followed by a dispersal to rio Paraíba do Sul, an event likely associated with stream capture of rio Tietê headwaters. Lucinda et al. (2005)Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60. also raised a hypothesis that the formation of the Sete Quedas falls may have isolated the ancestors of P. psittakos and P. dispilos.
Areagram showing drainage relationships based on phylogenetic analysis of Phallotorynus (modified from Lucinda et al., 2005). Arrows indicate major vicariant and dispersion events.
The generic phylogenetic relationships and the sequence of geological events associated with cladogeneses along the evolutionary history of Phallotorynus is still partially unclear and unsatisfactory given the polytomy (P. victoriae (P. dispilos, P. psittakos)(P. pankalos, P. jucundus)), yet the cladogram and areagram express the state of our knowledge of the relationships among Phallotorynus species. Discovery of new characters and/or species likely would help clarify and may shed light on our understanding of Phallotorynus systematics and biogeography.
Material Examined. Brazil: Mato Grosso do Sul, rio Iguatemi drainage (upper rio Paraná basin). MZU
Acknowledgements
For loan of specimens we thank Osvaldo Oyakawa (MZUSP). We are grateful to Everton Oliveira (UNT) for assistance on various tasks during this study. We thank Ciro Y. Joko (Centro de Ensino Unificado do Distrito Federal) for drawing the figure 2 and Celso Ikedo (UEM) for figure 1. PHFL is partially supported the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq proc. 308018/2011-7) and WJG is partially supported by a research grant from Fundação Araucária (Secretaria da Ciência, Tecnologia e Ensino Superior do Paraná, covenant 471/2013, protocol 36204).
References
- Costa, W. J. E. M. 1998. Phylogeny and classification of the Cyprinodontiformes (Euteleostei: Atherinomorpha): a reappraisal. Pp. 537-560. In: Malabarba, L. R., R. E. Reis, R. P. Vari, Z. M. S. Lucena & C. A. S. Lucena (Eds.). Phylogeny and classification of Neotropical fishes. Porto Alegre, Edipucrs, 603p.
- Farris, J. S. 1988. Hennig86 reference and documentation for version 1.5. Published by the author, New York, Port Jefferson Station.
- Ghedotti, M. J. 2000. Phylogenetic analysis and taxonomy of the poeciloid fishes (Teleostei, Cyprinodontiformes). Zoological Journal of the Linnean Society, 130: 1-53.
- Lucinda, P. H. F. & R. E. Reis. 2005. Systematics of the subfamily Poeciliinae Bonaparte (Cyprinodontiformes: Poeciliidae), with an emphasis on the tribe Cnesterodontini Hubbs. Neotropical Ichthyology, 3: 1-60.
- Lucinda, P. H. F., R. S. Rosa & R. E. Reis. 2005. Systematics and biogeography of the genus Phallotorynus Henn, 1916 (Cyprinodontiformes: Poeciliidae: Poeciliinae), with description of three new species. Copeia, 2005: 609-631.
- Nixon, K. C. & J. M. Carpenter. 1993. On outgroups. Cladistics, 9: 413-426.
- Parenti, L. R. 1981. A phylogenetic and biogeographical analysis of Cyprinodontiform fishes (Teleostei, Atherinomorpha). Bulletin of the American Museum of Natural History, 168: 341-557.
- de Pinna, M. C. C. 1991. Concepts and tests of homology in the cladistic paradigm. Cladistics, 7: 367-394.
- Platnick, N. I. 1989. An empirical comparison of microcomputer parsimony programs, II. Cladistics, 5: 145-161.
- Rosen, D. E. & M. Gordon. 1953. Functional anatomy and evolution of male genitalia in poeciliid fishes. Zoologica, 38: 1-47.
- Súarez, Y. R., J. P. Silva, L. P. Vasconcelos & W. F. Antonialli, Jr. 2009. Ecology of Phallotorynus pankalos (Cyprinodontiformes, Poeciliidae) in a first-order stream of the upper Paraná Basin. Neotropical Ichthyology, 7: 49-54.
- Taylor, W. R. & G. C. van Dyke. 1985. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium, 9: 107-119.
Publication Dates
-
Publication in this collection
Jan-Mar 2015
History
-
Received
09 May 2014 -
Reviewed
15 Dec 2014 -
Accepted
31 Mar 2015