Abstracts
We evaluate the simultaneous use of Sr: Ca and Zn: Ca ratios of the sagitta otolith as a potential indicator of the habitat of Percophis brasiliensis along a latitudinal gradient in the southwestern Atlantic Ocean (34-42ºS and 51-67ºW), in order to reliably identify fish stocks. Fish were collected in three sampling sites: Argentine-Uruguayan Common Fishing Zone (AUCFZ), El Rincón (ER) and San Matías Gulf (SMG). The otolith Sr:Ca and Zn:Ca ratios were determined by ICP-OES and EDTA volumetric method. The otolith Sr:Ca ratio was similar in the three sampling sites, while the Zn:Ca ratio was significantly higher in AUCFZ than in ER and SMG for all age groups. The discriminant analysis showed an association between the otolith Sr:Ca and Zn:Ca ratios from ER and SMG. Present results suggest the potential occurrence of two fish stocks of P. brasiliensis in the study area.
Argentina; Brazilian flathead; Fish stock; Microchemistry; Percophidae
Evaluamos el uso simultáneo de las relaciones Sr:Ca y Zn:Ca de los otolitos sagittae como un potencial indicador de hábitat de Percophis brasiliensis a lo largo de un gradiente longitudinal el Atlántico sudoccidental (34-42ºS - 51-67ºW) con el fin de contribuir a la identificación de los stocks pesqueros. Los peces fueron capturados en tres sitios de muestreo: Zona Común de Pesca Argentina-Uruguaya (ZCPAU), El Rincón (ER) y el Golfo San Matías (GSM). Las relaciones Sr:Ca y Zn:Ca se determinaron por ICP-OES y por titulación con EDTA. La relación Sr:Ca fue similar en los tres sitios de muestreo. La relación Zn:Ca fue mayor en la ZCPAU que en las demás areas (ER y GSM) para todos los rangos de edad. El análisis discriminante mostró una asociación entre las relaciones Sr:Ca y Zn:Ca de ER y GSM. Los resultados de este trabajo sugieren la presencia de al menos dos stocks de P. brasiliensis en el aérea de estudio.
Introduction
Fish otoliths are complex polycrystalline structures, composed of trace elements and calcium carbonate crystallized as aragonite (96%) in a protein matrix and small quantities of other minerals (Campana, 1999Campana, S. E. 1999. Chemistry and composition of fish otoliths: pathways, mechanisms and applications. Marine Ecology Progress Series, 188: 263-297.). These structures are placed in the inner ear of fish and are involved in hearing and maintenance of equilibrium (Popper & Zhongmin, 2000Popper, A. N. & L. Zhongmin. 2000. Structure-function relationship in fish otolith organs. Fisheries Research, 46: 15-25.). The concentration of the elements deposited in the otoliths, including strontium (Sr) and zinc (Zn), may be largely affected by the chemical composition of water, and secondarily by temperature (Campana, 1999Campana, S. E. 1999. Chemistry and composition of fish otoliths: pathways, mechanisms and applications. Marine Ecology Progress Series, 188: 263-297.; Wells et al., 2003Wells, B. K., B. E. Rieman, J. L. Clayton, D. L. Horan & C. M. Jones. 2003. Relationships between water, otoliths, and scale chemistries of Westslope cutthroat trout from the Coeur d'Alene River, Idaho: the potential application of hard-part chemistry to describe movements in freshwater. Transactions of the American Fisheries Society, 132: 409-424.; Ranaldi & Gagnon, 2008Ranaldi, M. M. & M. M. Gagnon. 2008. Zinc incorporation in the otoliths of juvenile pink snapper (Pagrus auratus Forster): The influence of dietary versus waterborne sources. Journal of Experimental Marine Biology and Ecology, 360: 56-62.). In particular, the concentration of Sr in the otoliths of anadromous and freshwater species was found to be positively correlated with water salinity (e.g., Kraus & Secor, 2004Kraus, R. T. & E. R. Secor. 2004. Incorporation of strontium into otoliths of an estuarine fish. Journal of Experimental Marine Biology and Ecology, 302: 85-106.; Sturrock et al., 2012Sturrock, A. M., C. N. Trueman, A. M. Darnaude & E. Hunter. 2012. Can otolith elemental chemistry retrospectively track migrations in fully marine fishes. Journal of Fish Biology, 81: 766-795.; Avigliano & Volpedo, 2013Avigliano, E., & A. V. Volpedo. 2013. Use of otolith strontium: calcium ratio as indicator of seasonal displacements of the silverside (Odontesthes bonariensis) in a freshwater-marine environment. Marine and Freshwater Research, 64: 746-751. ). However, the relationship between the otolith Sr and salinity tends to be weak and remains unclear for marine species (Brown & Severin, 2009Brown, R. J. & K. P. Severin. 2009. Otolith chemistry analyses indicate that water Sr:Ca is the primary factor influencing otolith Sr:Ca for freshwater and diadromous fish but not for marine fish. Canadian Journal of Fisheries and Aquatic Sciences, 66: 1790-1808.; Sturrock et al., 2012Sturrock, A. M., C. N. Trueman, A. M. Darnaude & E. Hunter. 2012. Can otolith elemental chemistry retrospectively track migrations in fully marine fishes. Journal of Fish Biology, 81: 766-795.). The fact that the incorporation of Sr into the otolith is affected by the environment makes this element a useful tool to study fish displacements and identify fish stocks (Secor et al., 1995Secor, D. H., A. Henderson-Arzapalob & P. M. Piccoli. 1995. Can otolith microchemistry chart patterns of migration and habitat utilization in anadromous fishes? Journal of Experimental Marine Biology and Ecology, 192: 15-33.; Zlokovitz et al., 2003Zlokovitz, E. R., D. H. Secor & P. M. Piccoli. 2003. Patterns of migration in Hudson River striped bass as determined by otolith microchemistry. Fisheries Research, 63: 245-259.; Kraus & Secor, 2004Kraus, R. T. & E. R. Secor. 2004. Incorporation of strontium into otoliths of an estuarine fish. Journal of Experimental Marine Biology and Ecology, 302: 85-106.; Schuchert et al., 2010Schuchert, P. C., I. Alexander, R. Arkhipkin & A. E. Koenig. 2010. Traveling around Cape Horn: Otolith chemistry reveals a mixed stock of Patagonian hoki with separate Atlantic and Pacific spawning grounds. Fisheries Research, 102: 80-86.; Tabouret et al., 2010Tabouret, H., G. Bareille, F. Clverie, C. Pecheyran, P. Prouzet & O. F. Donard. 2010. Simultaneous use of strontium:calcium and barium:calcium ratios in otoliths as markers of habitat: application to the European eel (Anguilla anguilla) in Adour Basin, South West France. Marine Environmental Research, 70: 35-45.; Avigliano & Volpedo, 2013Avigliano, E., & A. V. Volpedo. 2013. Use of otolith strontium: calcium ratio as indicator of seasonal displacements of the silverside (Odontesthes bonariensis) in a freshwater-marine environment. Marine and Freshwater Research, 64: 746-751.; Avigliano et al., 2013Avigliano, E., & A. V. Volpedo. 2013. Use of otolith strontium: calcium ratio as indicator of seasonal displacements of the silverside (Odontesthes bonariensis) in a freshwater-marine environment. Marine and Freshwater Research, 64: 746-751.). On the other hand, the concentration of Zn in the otoliths may be closely associated with fish diet. For this reason, Zn is a potential habitat indicator (Ranaldi & Gagnon, 2008Ranaldi, M. M. & M. M. Gagnon. 2008. Zinc incorporation in the otoliths of juvenile pink snapper (Pagrus auratus Forster): The influence of dietary versus waterborne sources. Journal of Experimental Marine Biology and Ecology, 360: 56-62.), especially when the otolith Sr:Ca ratio proves to be inadequate, as for some coastal marine species (Brown & Severin, 2009Brown, R. J. & K. P. Severin. 2009. Otolith chemistry analyses indicate that water Sr:Ca is the primary factor influencing otolith Sr:Ca for freshwater and diadromous fish but not for marine fish. Canadian Journal of Fisheries and Aquatic Sciences, 66: 1790-1808.; Sturrock et al. 2012Sturrock, A. M., C. N. Trueman, A. M. Darnaude & E. Hunter. 2012. Can otolith elemental chemistry retrospectively track migrations in fully marine fishes. Journal of Fish Biology, 81: 766-795.). Unlike the otolith Sr:Ca ratio, the Zn:Ca ratio decreases with water salinity (McCulloch et al., 2005McCulloch, M., M. Cappo, J. Aumend & W. Müller. 2005. Tracing the life history of individual barramundi using laser ablation MC-ICP-MS Sr-isotopic and Sr/Ba ratios in otoliths. Marine and Freshwater Research, 56: 637-644.).
In the last years, the chemical composition of the otolith has been used to differentiate fish stocks of diverse commercially important species from the southwestern Atlantic Ocean, such as the whitemouth croaker (Micropogonias furnieri) (Volpedo et al., 2007Volpedo, A. V., P. Miretzky & A. Fernández Cirelli. 2007. Stocks pesqueros de Cynoscion guatucupa y Micropogonias furnieri de la costa atlántica de Sudamérica: comparación entre métodos de identificación. Memoria de la Fundación La Salle de Ciencias Naturales, 165: 115-130.) and longtail hake (Macruronus magellanicus, presentlya junior synonym of Macruronus novaezelandiae) (Schuchert et al., 2010Schuchert, P. C., I. Alexander, R. Arkhipkin & A. E. Koenig. 2010. Traveling around Cape Horn: Otolith chemistry reveals a mixed stock of Patagonian hoki with separate Atlantic and Pacific spawning grounds. Fisheries Research, 102: 80-86.).
The Brazilian flathead, Percophis brasiliensis Quoy & Gaimard, 1825, is a demersal species that inhabits the southwest Atlantic coastal waters, from 23ºS (Rio de Janeiro, Brazil) to 47ºS (Santa Cruz, Argentina) (Barreto et al., 2011Barreto, A. C. 2007. Influencia ambiental en la distribución espacial de las clases de edad de pez palo (Percophis brasiliensis) en el Sistema Costero del Atlántico Sudoccidental (34º- 41º S). Unpublished Ph.D. Dissertation, Universidad de Buenos Aires, Argentina, 43p .). Its distribution is mainly associated with water salinity, with 5- to 8-year-old specimens being more frequently found in high salinity waters (> 33.7SPU) and younger specimens (< 4-7 years old) in lower salinity waters (approx. 20-30 PSU) resulting from the discharge of different rivers (Barretto, 2007Barretto, A. C., M. B. Sáez, M. R. Rico & A. J. Jaureguizar. 2011. Age determination, validation, and growth of Brazilian flathead (Percophis brasiliensis) from the southwest Atlantic coastal waters (34º-41ºS). Latin American Journal of Aquatic Research, 39: 297-305.).
In Argentinean waters, P. brasiliensis is exploited by multi-species
multi-fleet fishery, with commercial capture operations being more frequent in spring
and summer, between 34º and 42ºS (Rico & Sáez,
2010Rico, M. R. & M. B. Sáez. 2010. Análisis de la estructura de tallas;
determinación de la edad, validación y crecimiento de pez palo (Percophis
brasiliensis) a partir de muestras del desembarque comercial en el puerto de Mar del
Plata. Informe Técnico INIDEP, 85: 1-20.). The catch of this species has progressively increased since 1997 (Rico & Perrotta, 2009Rico, M. R. & M. B. Sáez. 2010. Análisis de la estructura de tallas;
determinación de la edad, validación y crecimiento de pez palo (Percophis
brasiliensis) a partir de muestras del desembarque comercial en el puerto de Mar del
Plata. Informe Técnico INIDEP, 85: 1-20.), with approximately 8200
tons landed on Argentinean and Uruguayan coasts during 2012 (Minagri, 2012Minagri, 2012. Ministerio de Agricultura y Pesca de la Nación Argentina.
Available from:
http://www.minagri.gob.ar/site/pesca/pesca_maritima/02-desembarques/anio.php?anio=2012
(27 December 2012).
http://www.minagri.gob.ar/site/pesca/pes...
). However, sustainable fishery management practices
have never been implemented, due to the scarce information available for the
identification of P. brasiliensis stocks. On this regard, Braicovich & Timi (2008)Braicovich, P. E. & J. T. Timi. 2008. Parasites as biological tags
for stock discrimination of the Brazilian flathead in the south-west Atlantic.
Journal of Fish Biology, 73: 557-571. have suggested the
occurrence of three fish stocks in the Argentine-Uruguayan Common Fishing Zone (AUCFZ),
El Rincón (ER) and San Matías Gulf (SMG) (Fig. 1),
based on the study of parasites. However, this has not been properly validated with
other methodologies.
Sampling sites of the Brazilian flathead, Percophis brasiliensis, in the southwestern Atlantic Ocean.
The objective of the present study was to evaluate the simultaneous use of the Sr:Ca and Zn:Ca ratios of the sagitta otolith as a potential indicator of the habitat of P. brasiliensis along a latitudinal gradient in the southwestern Atlantic Ocean (34-42ºS and 51-67ºW), to reliably identify fish stocks. For this purpose, Sr:Ca and Zn:Ca ratios of the otolith were compared between age groups and three different areas from southwestern Atlantic Ocean (Argentine-Uruguayan Common Fishing Zone, El Rincón and San Matías Gulf).
This information is important for the sustainable exploitation of the species and the development of evaluation models.
Material and Methods
A sampling campaign was carried out from November to December 2005 (spring) in the Buenos Aires Coastal Ecosystem (ECB), between Argentine-Uruguayan Common Fishing Zone (AUCFZ; 34º-38.5ºS) and El Rincón (ER; 38.5-42ºS) (Fig. 1), over a bathymetric range of 5-54 m (Fig. 1). Another sampling campaign was carried out in November 2009 (spring) between latitudes 40.5º-41.5ºS, San Matías Gulf (SMG) (Fig. 1). Brazilian flatheads were caught in 225 bottom trawls that used an Engel trawl (mesh size of 200 mm in the wings and 120 mm in the codend, vertical height of 4 m and horizontal opening of 15 m), similar to that used by commercial fishing vessels operating in the San Matías Gulf (SMG). The total number, sex, total length (TL, in centimeters) and total weight (TW, in grams) of the Brazilian flatheads caught in each fishing set were recorded. The sagittae otoliths were removed from each specimen of Percophis brasiliensis (voucher specimen PPGSMB 684, at Instituto Nacional de Investigación y Desarrollo Pesquero (INIDEP) collection, Mar del Plata, Buenos Aires, Argentina); one otolith of the pair was used for age determination and the other for the analysis of Sr:Ca and Zn:Ca ratios.
Age determination.Sagittae otoliths were embedded in opaque epoxy resin and sectioned transversely through the core to a thickness of 0.5 mm using a micro-cutter (Maruto MC-201). The opaque bands of these sections were counted by two independent readers under incident and transmitted light using a stereomicroscope (Nikon SMZ 10A) at 40X magnification. The age of each specimen was determined to the nearest year unknowing its length and sex. If the two independent readings differed, then the otolith was discarded. The validation of the periodical occurrence of opaque bands was established from a previous study performed by Barretto et al. (2011); hence, age determination of the specimens was precise and acceptable.
Determination of otolith element:Ca ratios. A total of 143 sagittae otoliths were selected, cleaned with distilled water, dried, weighed to the nearest 0.1 mg in an analytical balance, and then digested with 50% nitric acid on a sand bath at 400-450ºC. Otolith Sr (line: 474.771) and Zn (line: 213.857) concentrations were determined by ICP-OES using a Perkin Elmer Optima 2000 DV optical emission spectrometer (Überlingen, Germany; method EPA, 1994EPA. 1994. Methods for the Determination of Metals in Environmental Samples. Supplement 1. Ohio.) equipped with a cross-flow nebulizer and a quartz ICP torch. A Perkin-Elmer AS-90 Plus autosampler was used for automated sample handling. All measurements were performed in triplicate (RSD < 4%). External calibration was carried out using the atomic spectroscopy standard QCS 21 (Quality Control Standard, Perkin Elmer(r) Pure, USA). The equipment was cleaned at regular intervals with MilliQ water (Millipore, Saõ Paulo, Brazil) and 10% nitric acid matrix to prevent sample memory effects. The operating conditions of the ICP-OES equipment are given in Table 1.
Ca concentration was estimated by the EDTA volumetric method (APHA, 1993APHA. 1993. Standard Methods for the Examination of Water and Wastes. Washington, D.C., American Public Health Association. ). All measurements were performed in triplicate. The digestion and analytical procedures were checked by analysis of Otolith Certified Reference Material for trace elements (FEBS-1, National Research Council, Canada). Replicate analysis of this material showed good accuracy, with the following metal recovery rates: 93% for Sr and 110% for Ca. The element:Ca ratios were expressed in mmol mol-1 to standardize the concentrations of the elements in relation to Ca.
Statistical Analysis. To avoid possible age effects on the otolith element:Ca ratios, four different age groups were defined (Table 2). An analysis of covariance (ANCOVA) was then used to determine the effect of fish length on the study variables within each age group. Variables that were significantly correlated to fish length were corrected using the common within-group slope (b) for the variable on fish length (e.g., Reist, 1985Reist, J. D. 1985. An empirical evaluation of several univariate methods that adjust for size variation in morphometric data. Canadian Journal of Zoology, 63: 1429-1439.; Begg et al., 2001Begg, G. A., W. J. Overholtz & N. J. Munroe. 2001. The use of internal otolith morphometrics for identification of haddock (Melanogrammus aeglefinus) stocks on Georges Bank. Fishery Bulletin, 99: 1-14.; Cardinale et al., 2004Cardinale, M., P. Doering-Arjes, M. Kastowsky & H. Mosegaard. 2004. Effects of sex, stock, and environment on the shape of known-age Atlantic cod (Gadus morhua) otoliths. Canadian Journal of Fisheries and Aquatic Sciences, 61: 158-167.; Galley et al., 2006Galley, E. A., P. J. Wright & F. M. Gibb. 2006. Combined methods of otolith shape analysis improve identification of spawning areas of Atlantic cod. ICES Journal of Marine Science, 63: 1710-1717.; Burke et al., 2008Burke N., D. Brophy & P. A. King. 2008. Otolith shape analysis: its application for discriminating between stocks of Irish Sea and Celtic Sea herring (Clupea harengus) in the Irish Sea. ICES Journal of Marine Science, 65: 1670-1675.). Subsequently, the otolith Sr:Ca and Zn:Ca ratios were compared among the three sampling sites and age groups using the Kruskal-Wallis non-parametric analysis of variance (Sokal & Rholf, 1995Sokal, R. R. & F. J. Rohlf. 1995. Biometry: The Principles and Practice of Statistics in Biological Research. 3rd ed. Local, W. H. Freeman.). In addition, the relationship between otolith Sr:Ca and Zn:Ca ratios was evaluated for each sampling site and age group using discriminant analysis. The analyses were performed with the statistical software InfoStat(r).
Number of specimens (N) of Percophis brasiliensis per age group and sampling site. Argentine-Uruguayan Common Fishing Zone (AUCFZ), El Rincón (ER), San Matías Gulf (SMG).
Results
Otolith Sr:Ca and Zn:Ca ratios. For all age groups, the otolith Sr:Ca ratio was similar (p>0.05) while the otolith Zn:Ca ratio was significantly different between fish from AUCFZ and fish from ER and SMG (p<0.05; Table 3).
Otolith Sr:Ca and Zn:Ca ratios (mean±standard deviation) for Percophis brasiliensis from the Argentine-Uruguayan Common Fishing Zone (AUCFZ), El Rincón (ER) and San Matías Gulf (SMG), within each age group. Different letters indicate statistical significance among sampling sites for each age group (p<0.05).
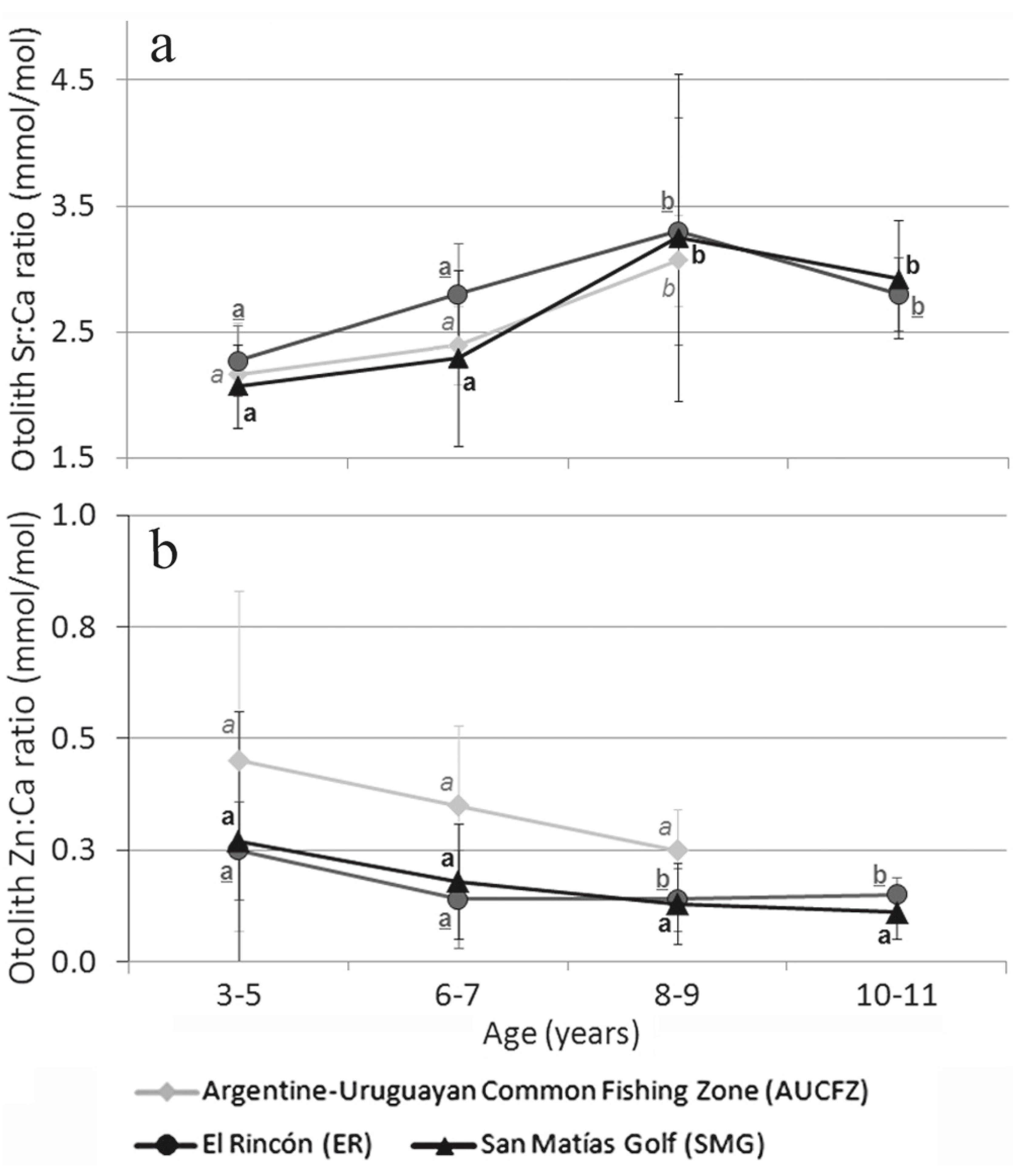
The relationship between the otolith Sr:Ca and Zn:Ca ratio and fish age, together with the significance levels from the Kruskal-Wallis test, are shown in Fig. 2a,b. The otolith Sr:Ca ratio significantly increased with age in the three sampling sites (Fig. 2). The otolith Zn:Ca ratio decreased with age in all the study sites, but this was only statistical significant for ER (Fig. 2).
Variation of Sr:Ca (a) and Zn:Ca (b) ratios of Percophis brasiliensis separated by age for the three sampling sites. Different letters indicate statistical significant differences among age groups (years) for each sampling site (p<0.05).
Dispersion analysis between the otolith Sr:Ca and Zn:Ca ratios. In the dispersion analysis, all age groups data for ER and SMG tended to cluster at low values of the otolith Zn:Ca ratio (Fig. 3a-d), while data for AUCFZ tended to scatter at high values of this ratio (Fig. 3a-c).
Relationship between otolith Sr:Ca and Zn:Ca ratios (mmol mol-1) for Percophis brasiliensis from three areas. Data for ER and SMG tended to cluster, while data for AUCFZ tended to disperse. Separation of data of AUCFZ and ER-SMG is observed. Triangles: Argentine-Uruguayan Common Fishing Zone (AUCFZ), stars: San Matías Gulf (SMG) and black circles: El Rincón (ER).
Discriminant analysis. The discriminant analysis showed an association between otolith Sr:Ca and Zn:Ca ratios in ER-SMG for the age groups 3-5, 6-7 and 8-9 years (Fig. 4a-c). For all cases, the first axis explained most of the variability in the data, being the most important variable in this axis, Zn:Ca ratio (Table 4). No fish from the age group 10-11 years was captured in AUCFZ.
Discriminant analysis of the otolith Sr:Ca and Zn:Ca ratios for Percophis brasiliensis. Plot of the first two discriminant functions for each age group (a-d). An association was observed between data for ER and SMG, which were separated from data for AUCFZ. Triangles: Argentine-Uruguayan Common Fishing Zone (AUCFZ), stars: San Matías Gulf (SMG) and black circles: El Rincón (ER).
Discussion
The Brazilian flathead is a marine species that inhabits areas with different salinity
levels throughout its life cycle. Juveniles and young adults (<4-7 years old) are
distributed in relatively low salinity waters (20-30 SPU), while adults (>5-8 years
old) are found in higher salinity waters (Barretto, 2007). In AUCFZ, this parameter
ranged between 22 and 33 SPU due to the in-flow of less salty waters from the freshwater
plume of the Río de la Plata (Marrari et
al., 2004Marrari, M., M. D. Viñas, P. Martos & D. Hernández. 2004. Spatial
patterns of mesozooplankton distribution in the Southwestern Atlantic Ocean
(34º-41ºS) during austral spring: relationship with the hydrographic conditions.
Journal of Marine Science, 61: 667-679.), which extends from 70 to 200 km into the open sea,
depending on the river flow (Guerrero et
al., 1997Guerrero, R.A., E. M. Acha, M. B. Framiñan & C. A. Lasta. 1997.
Physical oceanography of the Rio de la Plata estuary, Argentina. Continental Shelf
Research, 17: 727-742.). Waters from of ER are influenced by the discharge of
the Negro River and Colorado River (Guerrero et
al., 1997Guerrero, R.A., E. M. Acha, M. B. Framiñan & C. A. Lasta. 1997.
Physical oceanography of the Rio de la Plata estuary, Argentina. Continental Shelf
Research, 17: 727-742.). On this basis, the otolith Sr:Ca ratio was expected to
vary along the study region. However, this ratio was similar among sampling sites for
all age groups (Table 3). Despite the memory
effect that would have older samples due to the total digestion of otoliths, a positive
relationship was observed between the Sr:Ca ratio and fish age (Fig. 2). This is consistent with the movement of 4- to 7-year-old
fish toward areas of higher salinity. However, it may also be related to changes in the
rate of Sr incorporation into the otoliths due to physiological and ontogenetic factors.
Brown & Severin (2009)Brown, R. J. & K. P. Severin. 2009. Otolith chemistry analyses
indicate that water Sr:Ca is the primary factor influencing otolith Sr:Ca for
freshwater and diadromous fish but not for marine fish. Canadian Journal of Fisheries
and Aquatic Sciences, 66: 1790-1808. indicated that the
water Sr:Ca ratio was more homogeneous in marine environments than in freshwater or
brackish environments. Therefore, the variation in the otolith Sr:Ca ratio in marine
species is more likely due to physiological factors, rather than the result of the
exposure to heterogeneous environments (Campana,
1999Campana, S. E. 1999. Chemistry and composition of fish otoliths:
pathways, mechanisms and applications. Marine Ecology Progress Series, 188:
263-297.; Brown & Severin, 2009Minagri, 2012. Ministerio de Agricultura y Pesca de la Nación Argentina.
Available from:
http://www.minagri.gob.ar/site/pesca/pesca_maritima/02-desembarques/anio.php?anio=2012
(27 December 2012).
http://www.minagri.gob.ar/site/pesca/pes...
).
Specific studies on Sr incorporation into the otoliths of P. brasiliensis
are required to explain present results.
Coastal areas show great chemical heterogeneity due to upwelling, fluvial, and anthropogenic inputs, but often the pollutants that would contribute to such geographic variation in the concentration of Zn (Sturrock et al., 2012Sturrock, A. M., C. N. Trueman, A. M. Darnaude & E. Hunter. 2012. Can otolith elemental chemistry retrospectively track migrations in fully marine fishes. Journal of Fish Biology, 81: 766-795.). Zn was proposed as an indicator of habitat because its incorporation into the otoliths is influenced by the diet of fish (Ranaldi & Gagnon, 2008Ranaldi, M. M. & M. M. Gagnon. 2008. Zinc incorporation in the otoliths of juvenile pink snapper (Pagrus auratus Forster): The influence of dietary versus waterborne sources. Journal of Experimental Marine Biology and Ecology, 360: 56-62.). The significantly different otolith Zn:Ca ratio between Brazilian flatheads from AUCFZ and ER-SMG, both of which correspond to the latitudinal extremes of the study region (Table 3), may suggest that they belong to different fish stocks. The otolith Zn:Ca ratio of fish from ER, located in the middle area of the study region, was significantly different from that of fish collected in AUCFZ but similar to that of fish from SMG (Table 3). In addition, a negative relationship was observed between this ratio and fish age. Considering that the otolith Zn:Ca ratio tend to decrease with water salinity (McCulloch et al., 2005McCulloch, M., M. Cappo, J. Aumend & W. Müller. 2005. Tracing the life history of individual barramundi using laser ablation MC-ICP-MS Sr-isotopic and Sr/Ba ratios in otoliths. Marine and Freshwater Research, 56: 637-644.), present results are consistent with the movement of older specimens to areas of high salinity, as previously reported by Barretto (2007).
On the other hand, an association was found between the otolith Sr:Ca and Zn:Ca ratios for fish groups from ER and SMG, with these groups being separated from the one located in AUCFZ (Fig. 3). This may be the result of interactions between Brazilian flatheads from ER and SMG, with both groups possibly constituting a stock. The discriminant analysis showed a separation between AUCFZ and ER-SMG for the age groups 3-5, 6-7 and 8-9 years, which was mainly explained by the otolith Zn:Ca ratio (Fig. 4).
The results of the present study may suggest the potential occurrence of at least two fish stocks of P. brasiliensis in the southwestern Atlantic Ocean, between the latitudes 34-42ºS and 51-67ºW. One of them may be found in the north of the study region (AUCFZ, coast of Uruguay), while the other may be located in the south of this region (ER: 39º-41ºS and 62º-64ºW and SMG: 41º S and 63ºW).
The occurrence of three different stocks of P. brasiliensis in the southwestern Atlantic Ocean has already been suggested by Braicovich & Timi (2008)Braicovich, P. E. & J. T. Timi. 2008. Parasites as biological tags for stock discrimination of the Brazilian flathead in the south-west Atlantic. Journal of Fish Biology, 73: 557-571., based on the analysis of parasite composition. According to these authors the stocks were located in AUCFZ, ER and SMG, respectively. The simultaneous use of the otolith Sr:Ca and Zn:Ca ratios did not allow us to differentiate populations from ER and SMG, but it allowed us to discriminate them from the population located in AUCFZ. Even though this result does not completely agree with those reported by Braicovich & Timi (2008)Braicovich, P. E. & J. T. Timi. 2008. Parasites as biological tags for stock discrimination of the Brazilian flathead in the south-west Atlantic. Journal of Fish Biology, 73: 557-571., it further supports the occurrence of different fish stocks. These populations may be divided, at least in part, by marine fronts. A high salinity water tongue from the SMG is located to the east of the relatively diluted waters of ER, and forms a north-south front (39ºS) that induces seawater recirculation (Piola & Rivas, 1997Piola, A. R. & A. L. Rivas. 1997. Masas de agua en la plataforma continental. Pp. 119-132. In: Boschi, E. (Ed.). El Mar Argentino y sus Recursos Pesqueros. Antecedentes Históricos de las Exploraciones en el Mar y las Características Ambientales. Mar del Plata, Argentina, El Mar Argentino y sus Recursos Pesqueros. v.1.). Previous studies on adult fishes and ichthyoplankton have shown that this is an important breeding area for P. brasiliensis (Acha et al., 2012Acha, E. M., M. Orduna, K. Rodrigues, M. I. Militelli & M. Braverman. 2012. Caracterización de la zona de "El Rincón" (Provincia de Buenos Aires) como área de reproducción de peces costeros. Revista de Investigación y Desarrollo Pesquero, 21: 31-43.), where seawater recirculation facilitates the retention of eggs and larvae and the life cycle of the species is completed (Rodrigues et al., 2010Rodrigues, K. A., M. R. Rico, M. I. Militelli, P. Osovnikar & M. Maggioni. 2010. Parámetros reproductivos y distribución de las hembras de pez palo (Percophis brasiliensis) y lenguado (Paralichthys patagonicus) en el Golfo San Matías. Informe Técnico INIDEP, 65: 1-20.; Acha et al., 2012Acha, E. M., M. Orduna, K. Rodrigues, M. I. Militelli & M. Braverman. 2012. Caracterización de la zona de "El Rincón" (Provincia de Buenos Aires) como área de reproducción de peces costeros. Revista de Investigación y Desarrollo Pesquero, 21: 31-43.). Hence, the low interaction between fish groups from AUCFZ and ER-SMG may be explained by the presence of a marine front.
The simultaneous use of the Sr:Ca and Zn:Ca ratios of the sagitta otolith is a potential indicator of the habitat of P. brasiliensis along a latitudinal gradient in the southwestern Atlantic Ocean. The otolith Sr:Ca ratio was associated with fish age and could be used as an indicator of habitat. However, this ratio did not allow differentiating stocks of P. brasiliensis. The otolith Zn:Ca ratio tended to decrease with age and allowed the differentiation of two fish groups (AUCFZ and ER-SMG). For this reason, it could also be considered as a potential indicator of habitat. Based on these results, the incorporation of other otolith ratios (e.g., Ba:Ca, Mn:Ca, Mg:Ca, Li:Ca) to the analysis is suggested in order to complement the study of potential habitat indicators for the Brazilian flathead. This may validate the identification of fish stocks, thus contributing to the sustainable management of that resource in the southwestern Atlantic Ocean.
Acknowledgments
We are grateful to "Programa Especies Demersales Costeras" from INIDEP (Argentina) and IBMPAS, Universidad Nacional del Comahue and Dr. R. González for the samples collected during the cruise REDE 2009, Universidad de Buenos Aires (UBACYT 20620110100007), CONICET (PIP 112-20120100543CO) and ANPCyT (PICT2010-1372). This paper corresponds to INIDEP Contribution n.º 074.
References
- Acha, E. M., M. Orduna, K. Rodrigues, M. I. Militelli & M. Braverman. 2012. Caracterización de la zona de "El Rincón" (Provincia de Buenos Aires) como área de reproducción de peces costeros. Revista de Investigación y Desarrollo Pesquero, 21: 31-43.
- APHA. 1993. Standard Methods for the Examination of Water and Wastes. Washington, D.C., American Public Health Association.
- Avigliano, E., & A. V. Volpedo. 2013. Use of otolith strontium: calcium ratio as indicator of seasonal displacements of the silverside (Odontesthes bonariensis) in a freshwater-marine environment. Marine and Freshwater Research, 64: 746-751.
- Avigliano, E., C. Riaños Martinez & A. V. Volpedo. 2013. Combined use of otolith microchemistry and morphometry as indicators of the habitat of the silverside (Odontesthes bonariensis) in a freshwater-estuarine environment. Fisheries Reseach, 149: 55-60.
- Barreto, A. C. 2007. Influencia ambiental en la distribución espacial de las clases de edad de pez palo (Percophis brasiliensis) en el Sistema Costero del Atlántico Sudoccidental (34º- 41º S). Unpublished Ph.D. Dissertation, Universidad de Buenos Aires, Argentina, 43p .
- Barretto, A. C., M. B. Sáez, M. R. Rico & A. J. Jaureguizar. 2011. Age determination, validation, and growth of Brazilian flathead (Percophis brasiliensis) from the southwest Atlantic coastal waters (34º-41ºS). Latin American Journal of Aquatic Research, 39: 297-305.
- Begg, G. A., W. J. Overholtz & N. J. Munroe. 2001. The use of internal otolith morphometrics for identification of haddock (Melanogrammus aeglefinus) stocks on Georges Bank. Fishery Bulletin, 99: 1-14.
- Braicovich, P. E. & J. T. Timi. 2008. Parasites as biological tags for stock discrimination of the Brazilian flathead in the south-west Atlantic. Journal of Fish Biology, 73: 557-571.
- Brown, R. J. & K. P. Severin. 2009. Otolith chemistry analyses indicate that water Sr:Ca is the primary factor influencing otolith Sr:Ca for freshwater and diadromous fish but not for marine fish. Canadian Journal of Fisheries and Aquatic Sciences, 66: 1790-1808.
- Burke N., D. Brophy & P. A. King. 2008. Otolith shape analysis: its application for discriminating between stocks of Irish Sea and Celtic Sea herring (Clupea harengus) in the Irish Sea. ICES Journal of Marine Science, 65: 1670-1675.
- Campana, S. E. 1999. Chemistry and composition of fish otoliths: pathways, mechanisms and applications. Marine Ecology Progress Series, 188: 263-297.
- Cardinale, M., P. Doering-Arjes, M. Kastowsky & H. Mosegaard. 2004. Effects of sex, stock, and environment on the shape of known-age Atlantic cod (Gadus morhua) otoliths. Canadian Journal of Fisheries and Aquatic Sciences, 61: 158-167.
- EPA. 1994. Methods for the Determination of Metals in Environmental Samples. Supplement 1. Ohio.
- Galley, E. A., P. J. Wright & F. M. Gibb. 2006. Combined methods of otolith shape analysis improve identification of spawning areas of Atlantic cod. ICES Journal of Marine Science, 63: 1710-1717.
- Guerrero, R.A., E. M. Acha, M. B. Framiñan & C. A. Lasta. 1997. Physical oceanography of the Rio de la Plata estuary, Argentina. Continental Shelf Research, 17: 727-742.
- Kraus, R. T. & E. R. Secor. 2004. Incorporation of strontium into otoliths of an estuarine fish. Journal of Experimental Marine Biology and Ecology, 302: 85-106.
- Marrari, M., M. D. Viñas, P. Martos & D. Hernández. 2004. Spatial patterns of mesozooplankton distribution in the Southwestern Atlantic Ocean (34º-41ºS) during austral spring: relationship with the hydrographic conditions. Journal of Marine Science, 61: 667-679.
- McCulloch, M., M. Cappo, J. Aumend & W. Müller. 2005. Tracing the life history of individual barramundi using laser ablation MC-ICP-MS Sr-isotopic and Sr/Ba ratios in otoliths. Marine and Freshwater Research, 56: 637-644.
- Minagri, 2012. Ministerio de Agricultura y Pesca de la Nación Argentina. Available from: http://www.minagri.gob.ar/site/pesca/pesca_maritima/02-desembarques/anio.php?anio=2012 (27 December 2012).
» http://www.minagri.gob.ar/site/pesca/pesca_maritima/02-desembarques/anio.php?anio=2012 - Piola, A. R. & A. L. Rivas. 1997. Masas de agua en la plataforma continental. Pp. 119-132. In: Boschi, E. (Ed.). El Mar Argentino y sus Recursos Pesqueros. Antecedentes Históricos de las Exploraciones en el Mar y las Características Ambientales. Mar del Plata, Argentina, El Mar Argentino y sus Recursos Pesqueros. v.1.
- Popper, A. N. & L. Zhongmin. 2000. Structure-function relationship in fish otolith organs. Fisheries Research, 46: 15-25.
- Ranaldi, M. M. & M. M. Gagnon. 2008. Zinc incorporation in the otoliths of juvenile pink snapper (Pagrus auratus Forster): The influence of dietary versus waterborne sources. Journal of Experimental Marine Biology and Ecology, 360: 56-62.
- Reist, J. D. 1985. An empirical evaluation of several univariate methods that adjust for size variation in morphometric data. Canadian Journal of Zoology, 63: 1429-1439.
- Rico, M. R. & M. B. Sáez. 2010. Análisis de la estructura de tallas; determinación de la edad, validación y crecimiento de pez palo (Percophis brasiliensis) a partir de muestras del desembarque comercial en el puerto de Mar del Plata. Informe Técnico INIDEP, 85: 1-20.
- Rico, M. R. & R. G. Perrotta. 2009. Evolución reciente de la captura por unidad de esfuerzo (1993-2004) y de las tallas medias (1993-2005) del pez palo (Percophis brasiliensis) en el litoral marítimo entre 34ºS y 42ºS. Informe Técnico INIDEP, 73: 1-21.
- Rodrigues, K. A., M. R. Rico, M. I. Militelli, P. Osovnikar & M. Maggioni. 2010. Parámetros reproductivos y distribución de las hembras de pez palo (Percophis brasiliensis) y lenguado (Paralichthys patagonicus) en el Golfo San Matías. Informe Técnico INIDEP, 65: 1-20.
- Schuchert, P. C., I. Alexander, R. Arkhipkin & A. E. Koenig. 2010. Traveling around Cape Horn: Otolith chemistry reveals a mixed stock of Patagonian hoki with separate Atlantic and Pacific spawning grounds. Fisheries Research, 102: 80-86.
- Secor, D. H., A. Henderson-Arzapalob & P. M. Piccoli. 1995. Can otolith microchemistry chart patterns of migration and habitat utilization in anadromous fishes? Journal of Experimental Marine Biology and Ecology, 192: 15-33.
- Sokal, R. R. & F. J. Rohlf. 1995. Biometry: The Principles and Practice of Statistics in Biological Research. 3rd ed. Local, W. H. Freeman.
- Sturrock, A. M., C. N. Trueman, A. M. Darnaude & E. Hunter. 2012. Can otolith elemental chemistry retrospectively track migrations in fully marine fishes. Journal of Fish Biology, 81: 766-795.
- Tabouret, H., G. Bareille, F. Clverie, C. Pecheyran, P. Prouzet & O. F. Donard. 2010. Simultaneous use of strontium:calcium and barium:calcium ratios in otoliths as markers of habitat: application to the European eel (Anguilla anguilla) in Adour Basin, South West France. Marine Environmental Research, 70: 35-45.
- Volpedo, A. V., P. Miretzky & A. Fernández Cirelli. 2007. Stocks pesqueros de Cynoscion guatucupa y Micropogonias furnieri de la costa atlántica de Sudamérica: comparación entre métodos de identificación. Memoria de la Fundación La Salle de Ciencias Naturales, 165: 115-130.
- Wells, B. K., B. E. Rieman, J. L. Clayton, D. L. Horan & C. M. Jones. 2003. Relationships between water, otoliths, and scale chemistries of Westslope cutthroat trout from the Coeur d'Alene River, Idaho: the potential application of hard-part chemistry to describe movements in freshwater. Transactions of the American Fisheries Society, 132: 409-424.
- Zlokovitz, E. R., D. H. Secor & P. M. Piccoli. 2003. Patterns of migration in Hudson River striped bass as determined by otolith microchemistry. Fisheries Research, 63: 245-259.
Publication Dates
-
Publication in this collection
24 Mar 2015 -
Date of issue
Jan-Mar 2015
History
-
Received
30 Dec 2013 -
Accepted
06 Oct 2014