Abstracts
A phylogeny of the species of the loricariid genus Acestridium and relevant outgroups is presented based on a parsimony analysis of 52 unweighted and unordered morphological characters. Acestridium is diagnosed as monophyletic based on the possession of the 17 exclusive synapomorphies. Two primary trees were found, and the strict consensus among those alternative trees resulted in the following relationships: ((A. dichromum + A. triplax)(A. gymnogaster + A. scutatum + (A. discus (A. colombiensis + A. martini)))). Acestridium was found to be sister to Niobichthys with this clade subsequently the sister-group to Oxyropsis + Hypoptopoma.
Uma filogenia das espécies do gênero de Loricariidae Acestridium e grupos externos relevantes é apresentada com base na análise de parcimônia de 52 caracteres morfológicos não pesados e desordenados. Acestridium é diagnosticado como monofilético com base na posse de 17 sinapomorfias exclusivas. Duas árvores primárias foram encontradas e o consenso estrito entre essas árvores alternativas resultou nas seguintes relações: ((A. dichromum + A. triplax)(A. gymnogaster + A. scutatum + (A. discus (A. colombiensis + A. martini)))). Acestridium foi encontrado como grupo-irmão de Niobichthys e este clado como grupo-irmão sucessivo de Oxyropsis + Hypoptopoma.
Amazon; Biodiversity; Evolution; Neotropical; Phylogeny
Introduction
Loricariidae is the largest catfish family with almost 900 species recognized as valid and more described each year. Loricariids are distributed throughout most of the Neotropics, extending from Costa Rica to northern Argentina. The vast majority of the species occur on the Andes, but with several species restricted to the western slopes of that mountain range (Reis et al., 2003Reis, R. E. 1998. Anatomy and phylogenetic analysis of the neotropical callichthyd catfishes (Ostariophysi, Siluriformes). Zoological Journal of the Linnean Society, 124: 105-168.). Six subfamilies are currently recognized: Delturinae, Hypoptopomatinae, Hypostominae, Lithogeninae, Loricariinae, and Neoplecostominae (Reis et al., 2006).
Hypoptopomatinae is a monophyletic subfamily comprised of 20 genera (Reis et al., 2012). Most species are relatively small as adults, ranging from 20 to 45 mm in SL. Like other members of the Loricariidae, hypoptopomatines are encased in dermal plates which bear numerous odontodes, or dermal teeth, and the mouth is surrounded by expanded fleshy lips forming a suctorial device. Hypoptopomatines are distinguished from all other loricariids, among other features, by the totally or partially ventrally exposed pectoral girdle that bears laminar extensions on the ventral surface of both the cleithrum and the coracoid that largely or entirely cover the fossae for the arrector muscles.
The species level taxonomic history of Acestridium is largely recent. Except for A. discusHaseman, 1911Haseman, J. D. 1911. Descriptions of some new species of fishes and miscellaneous notes on others obtained during the expedition of the Carnegie Museum to central South America. Annals of Carnegie Museum, 7: 315-328., all other species have been described in the last 15 years. Currently, seven species are recognized: Acestridium discus, A. dichromum Retzer, Nico & Provenzano, 1999Retzer, M. E., L. G. Nico & F. Provenzano R. 1999. Two new species of Acestridium (Siluformes: Loricariidae) from southern Venezuela, with observations on camouflage and color change. Ichthyological Exploration of Freshwaters, 10: 313-326., A. martini Retzer, Nico & Provenzano, 1999, A. colombiensis Retzer, 2005, A. triplaxRodriguez & Reis, 2007Rodriguez, M. S. & R. E. Reis. 2007. A new species of Acestridium Haseman, 1911 (Loricariidae: Hypoptopomatinae) from the Eastern Amazon basin, Brazil. Neotropical Ichthyology, 5: 429-434., A. gymnogaster Reis & Lehmann, 2009Lehmann, P. 2006. Otocinclus batmani, a new species of hypoptopomatine catfish (Siluriformes: Loricariidae) from Colombia and Peru. Neotropical Ichthyology, 4: 379-383. and A. scutatus Reis & Lehmann, 2009. The position of the genus has been inconsistent in different revisions of the Loricariidae. Gosline (1945)Gosline, W. A. 1945. Catálogo dos Nematognatos de água-doce da América do Sul e Central. Boletim do Museu Nacional, 33: 1-138. placed Acestridium in the subfamily Loricariinae. Subsequently, Isbrücker & Nijssen (1974)Nijssen, H. & I. J. H. Isbrücker. 1987. Spectracanthicus murinus, nouveaux genre et espèce de Poisson-Chat cuirassé du Rio Tapajós, Est. Pará, Brésil, avec des remarques su d'autres genres de Loricariidés (Pisces, Siluriformes, Loricariidae). Revue française Aquariologie et Herpetologie, 13: 93-98. relocated Acestridium to the subfamily Acestridiinae stating that it did not belong in Loricariinae, but providing no evidence to justify their action. Most recently, Isbrücker (1980) placed the genus in the subfamily Loricariinae, tribe Acestridiini. Finally, Nijssen & Isbrücker (1987) diagnosed and placed the genus in the Hypoptopomatinae.
In the first phylogenetic analysis of the hypoptopomatines, Schaefer (1991)Schaefer, S. A. 1987. Osteology of Hypostomus plecostomus (Linnaeus), with a phylogenetic analysis of the loricariid subfamilies (Pisces: Siluroidei). Contributions in Science, Natural History Museum of Los Angeles County, 394: 1-31. united Hypoptopoma Gunther, 1868, OxyropsisEigenmann & Eigenmann, 1889Eigenmann, C. H. 1914. On new species of fishes from the Rio Meta Basin of eastern Colombia and on albino or blind fishes from near Bogotá. Indiana University Studies, 23: 229-230., Acestridium, Niobichthys Schaefer & Provenzano, 1998 (then as new genus), Otocinclus Cope, 1871, and Microlepidogaster Eigenmann & Eigenmann, 1889 as the tribe Hypoptopomatini. The tribe was diagnosed by the absence of a levator arcus palatini crest on the hyomandibula and reduced levator muscle, the presence of few, relatively large plates at the anterior margin of the snout, and a ventrolateral reflection of the cheek plates. A number of synapomorphic traits were used to diagnose Acestridium: ten principal caudal-fin rays; the dermal plates anterior to first dorsal-fin proximal radial expanded, bearing a ventromedial keel contacting the dorsal margin of the neural arches of vertebrae six through twelve; and the presence of a medially reflected ridge of bone on the ventral surface of the lateral ethmoid contacting the metapterygoid channel. Schaefer (1991) also added as diagnosing features the plates at the anterior margin of the snout forming a conspicuous spatulate projection; the very slender, depressed head and body; the dorsal surface of the head and trunk with longitudinal ridges; a posteriorly placed dorsal fin, well beyond pelvics just anterior to anal-fin origin; and the absence of an adipose fin.
Schaefer (1997), in a revision of Otocinclus and reanalysis of the position of the genus in the subfamily using additional discovered character evidence, reaffirmed the earlier phylogeny of Schaefer (1991), with the exception that Microlepidogaster was transferred to the tribe Otothyrini. Finally, Schaefer (1998) diagnosed Hypoptopomatinae by the following six synapomorphies: the presence of a ventrally opened nasal capsule (reversed in Acestridium); the pterotic bone pierced by numerous fenestrae and enlarged in all hypoptopomatines; the presence of a metapterygoid channel; the closed arrector fossae of the ventral pectoral skeleton; the presence of a median rostral snout plate (reversed in Niobichthys Schaefer & Provenzano, 1998); and the possession of enlarged odontodes on both the dorsal and ventral margins of the snout (reversed in Niobichthys).
In the same year, Schaefer & Provenzano (1998) described Niobichthys and rediagnosed the tribe Hypoptopomatini based on the following synapomorphies: the elongate lateral ethmoid-antorbital lateral process, with the contact between the lateral ethmoid and the third infraorbital located well lateral to the lateral margin of the nasal capsule (reversed in Acestridium); a reduced pterotic lateral fenestrae (reversed from derived state for the Hypoptopomatinae); the number of bifid haemal spines reduced to 2; the reduced intervertebral interdigitations between successive neural and haemal spines; a loss of the first dorsal-fin spinelet (convergent in SchizolecisBritski & Garavello, 1984Britski, H. A. & J. C. Garavello. 1984. Two new southeastern brazilian genera of Hypoptopomatinae and a redescription of Pseudotocinclus Nichols, 1919 (Ostariophysi, Loricariidae). Papéis Avulsos de Zoologia, 35: 225-241. and Pseudotothyris Britski & Garavello, 1984 of the Otothyrini); the presence of median pectoral-fin spine serrae (reversed in Nannoptopoma Schaefer, 1996); a truncated mid-dorsal lateral plate series and the plates of the canal-bearing median series dorsally expanded; the presence of a conspicuous enlarged median pre-anal shield plate.
Although Acestridium has been demonstrated to be a member of the Hypoptomatinae, its position in the family is uncertain and the phylogenetic relationships of its species have never been investigated. In the present study, a phylogenetic analysis of the species of Acestridium is provided based on osteology and comparison with other members of the Hypoptopomatinae, and an expanded phylogenetic diagnosis is advanced for the genus and species groups.
Material and Methods
Unless otherwise indicated, the descriptions of characters and character states pertain only to the specimens listed in Material Examined. Skeletal terminology follows Schaefer (1987), except that parieto-supraoccipital replaces supraoccipital (Arratia & Gayet, 1995Arratia, G. & M. Gayet. 1995. Sensory canals and related bones of Tertiary Siluriform crania from Bolívia and North America and comparison with Recent forms. Journal of Vertebrate Paleontology, 15: 482-505.), and compound pterotic replaces pterotic-supracleithrum (Aquino & Schaefer, 2002Aquino, A. E. & S. A. Schaefer. 2002. The temporal region of the cranium of loricarioid catfishes (Teleostei: Siluriformes): morphological diversity and phylogenetic significance. Zoologischer Anzeiger, 241: 223-244.). Fishes were cleared and double-stained for cartilage and for bone according to procedures derived from Taylor & van Dyke (1985)Taylor, W. R. & G. C. van Dyke. 1985. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium, 9: 107-119..
The data matrix for the phylogenetic analysis provided in Table 1 was constructed using Mesquite, version 2.74 (Maddison & Maddison, 2006Maddison, W. P. & D. R. Maddison. 2006. Mesquite: a modular system for evolutionary analysis. Version 1.12. Available from http://mesquiteproject.org.
http://mesquiteproject.org...
) and exported and submitted to heuristic parsimony analyses in NONA (Goloboff, 1999Goloboff, P. A. 1999. NONA. Version 2.9. Tucuman, Argentina: Computer software distributed by the author.) under Winclada 1.00.08 (Nixon, 2002Nixon, K. C. 2002. WinClada ver. 1.00.08. Published by the author, Ithaca, NY, USA.) Windows shell. The heuristic search was performed with 1000 replications of Random Addition Sequence (RAS) and Tree Bisection Reconnection (TBR) branch swapping, followed by a final round of global TBR. All character transformations were unweighted and unordered. Branch support was calculated as Bremer decay index (Bremer, 1994Bremer, K. 1994. Branch support and tree stability. Cladistics, 10: 295-304.) in NONA. Character state transformations are given in Appendix 1
Appendix 1.
Character state transformations by clade, with only unambiguous changes listed, except for the Hypoptopomatini, in which transformations are presented under ACCTRAN optimization. Transformations are listed with the character number first and then the plesiomorphic > apomorphic state. Transformations listed in bold are exclusive synapomorphies.
.
Character state matrix used for the parsimony analysis. A = 0 and 1. Hyphen = missing data or inapplicable states.
Taxa were selected based on previous phylogenetic hypothesis of the Hypoptopomatini (Schaefer, 1997, 1998). Outgroups in the Hypoptopomatini were Hypoptopoma spectabile (Eigenmann, 1914), H. inexspectatum (Holmberg, 1893Holmberg, E. L. 1893. Dos peces argentinos: Aristommata inexpectata y Liposarcus ambrosetti. Revista Jardin Zoologico Buenos Aires, 1: 353-354.), Niobichthys ferrarisi Schaefer & Provenzano, 1998, Oxyropsis wrightiana Eigenmann & Eigenmann, 1889, Otocinclus batmani Lehmann, 2006 and O. arnoldiRegan, 1909Regan, C. T. 1909. Descriptions of three new freshwater fishes from South America, present to British Museum by Herr J. Paul Arnold. Annals and Magazine of Natural History, 3: 234-235. and in the Otothyrini Hisonotus maculipinnis Regan, 1912, which was also used to root the trees.
In Comparative Material Examined lots are grouped by country and within each country, by state or department, followed by institutional abbreviation, catalog number, number of specimens in the lot, number of cleared and stained (c&s) specimens when present, their range of standard length, and specific locality data. Institutional abbreviations follow Fricke & Eschmeyer (2014)Fricke, R & W. Eschmeyer. 2014. Guide to Fish Collections. Available from: http://research.calacademy.org/research/ichthyology/catalog/collections.asp. (Date of access 15 April 2014).
http://research.calacademy.org/research/...
.
Results
Character descriptions. The character descriptions below arranged by anatomical groups refer only the specimens examined in the present analysis.
Neurocranium. 1. Anterolateral expansions of the mesethmoid [Schaefer, 1991 char. 3; Schaefer, 1998 char. 2, modified from Martins et al., 2014Martins, F. O., H. A. Britski & F. Langeani. 2014. Systematics of Pseudotothyris (Loricariidae: Hypoptopomatinae). Zoological Journal of the Linnean Society, 170: 822-874. char. 3].
In the primitive condition among advanced loricarioids, the mesethmoid lacks the typical lateral cornua, or anterolateral expansions and the anterior portion of the mesethmoid is narrower than the posterior portion (state 0). Among examined taxa, this condition was found in Hisonotus, Niobichthys, and Acestridium. In Otocinclus, Oxyropsis, and Hypoptopoma the mesethmoid has conspicuous anterolateral expansions, with the anterior portion as wide as the posterior portion (state 1).
2. Ventral condyle of the mesethmoid [Schaefer, 2003 char. 2; Armbruster, 2004Armbruster, J. W. 1998. Modifications of the digestive tract for holding air in Loricariid and scoloplacid catfishes. Copeia, 1998: 663-675. char. 100].
There is a wide variation in shape and size of the mesethmoid ventral condyle or disk among loricarids. In the examined outgroups the mesethmoid condyle is a bi-concave, disk-like structure, more compressed anteriorly and with the posterior margin rounded (state 0), in all species of Acestridium the condyle is a uniformly oval disk (state 1).
3. Position of the mesethmoid condyle [modified from Schaefer, 1991 char 2 and Paixão & Toledo-Piza, 2009 char. 4].
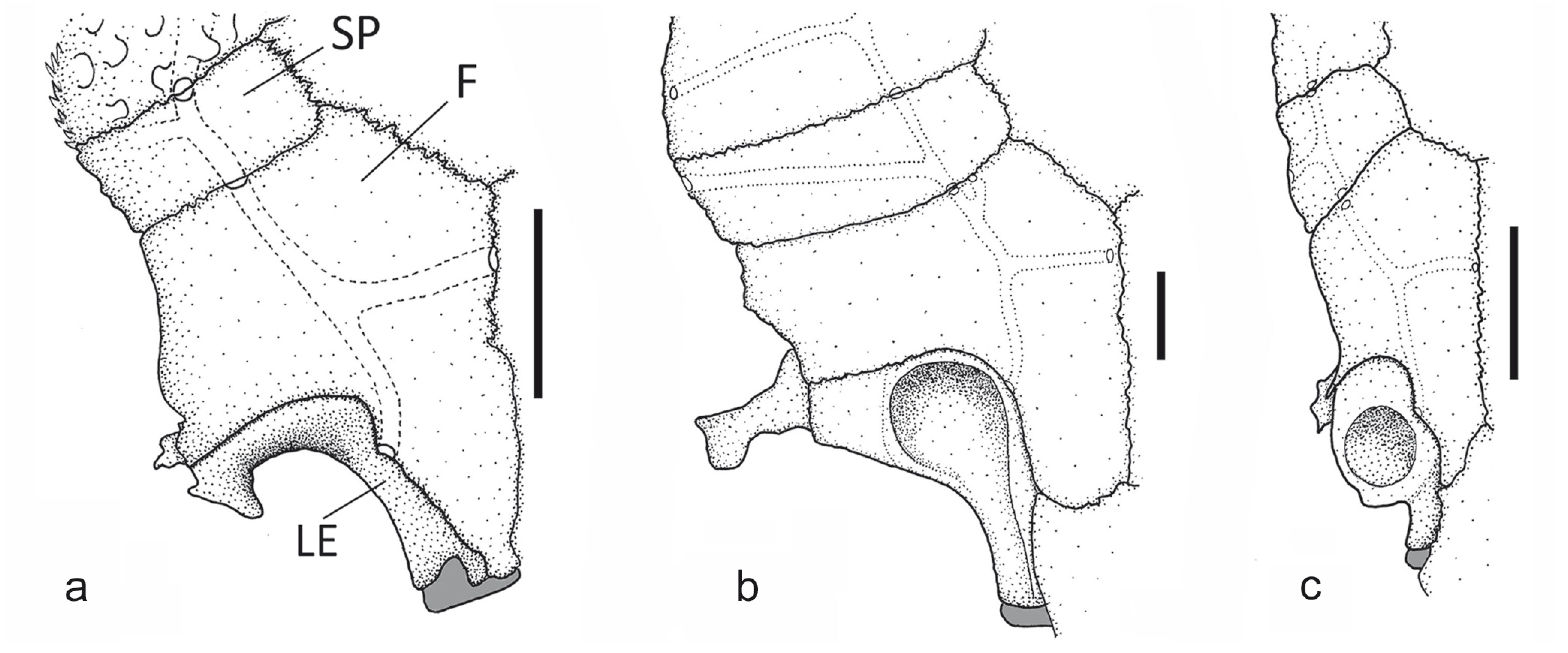
The position of the ventral condyle of the mesethmoid varies relative to the anterior margin of the bone. In Otocinclus, Oxyropsis wrightiana and Hisonotus maculipinnis the mesethmoid disk has an anterior position, being aligned with the anterior margin of the mesethmoid (state 0). Conversely, in Hypoptopoma spectabile, H. inexspectatum, and Niobichthys ferrarisi, the mesethmoid disk is shifted posteriorly, but still is located on the anterior one-half of the length of the bone (state 1). Alternatively, the species of Acestridium have an elongate mesethmoid with the mesethmoid disk on the posterior half of the bone (Fig. 1c; state 2).
Anterior portion of neurocranium of: a) Otocinclus arnoldi, MCP 25234; b) Oxyropsis wrightiana, MCP 34503; and c) Acestridium discus, MZUSP 85321. CPT = compound pterotic; F = frontal; LE = lateral ethmoid; ME = mesethmoid; SOC = parieto-supraoccipital; PN = prenasal plate; SP = sphenotic. Lateral view, left side, anterior to left. Scale bar = 2 mm.
4. Dorsal surface of the mesethmoid [modified from Gauger & Buckup, 2005Gauger, M. F. W. & P. A. Buckup. 2005. Two new species of Hypoptopomatinae from the rio Paraíba do Sul basin, with comments on the monophyly of Parotocinclus and the Otothyrini (Siluriformes: Loricariidae). Neotropical Ichthyology, 3: 509-518. char. 47, modified from Martins et al., 2014 char. 6].
The dorsal surface of the mesethmoid usually covered by dermal plates occurs in most loricariids, including Hisonotus and Otocinclus among the examined taxa (Fig. 1a; state 0). Conversely, the dorsal surface of the mesethmoid is exposed on the snout surface as a triangular patch that bears odontodes in most of the Hypoptopomatini (Figs. 1b-c; state 1).
5. Extent of exposure of the mesethmoid on the anterior portion of the snout.
The extent of exposure of the mesethmoid on the dorsum of the snout is variable. It can be short, not reaching or just reaching to a vertical passing by posterior margin of the mesethmoid disc, in Hypoptopoma, Niobichthys, Oxyropsis, and Acestridium dichromum, A. gymnogaster, A. scutatum, and A. triplax (Fig. 1b; state 0), or it can be long, extending past the mesethmoid disc towards the snout tip in A. discus, A. martini, and A. colombiensis (Fig. 1c; state 1). In Hisonotus and the species of Otocinclus the mesethmoid is not exposed, and this character is, thus, coded as inapplicable.
6. Degree of encapsulation of the nasal capsule [modified from Schaefer, 1991 char. 5; modified from Martins et al., 2014 char. 9].
The olfactory organ of catfishes is encapsulated in a cavity variably formed by the frontal, mesethmoid, lateral ethmoid, and palatine (Reis, 1998). Callichthys Scopoli, 1777 and many basal loricariids share an olfactory organ totally encapsulated by the lateral ethmoid. This represents the primitive condition in this family, but is present in reversed, derived state in some of the Hypoptopomatini. Among examined taxa, Hisonotus and Otocinclus share the condition in which the olfactory organ is only partially encapsulated, being widely open ventrally (Fig. 2a; state 0). Conversely, in most examined hypoptopomatines examined the nasal capsule is almost completely closed ventrally by the expanded lateral ethmoid (Fig. 2b; state 1), or is completely closed in the species of Acestridium (Fig. 2c; state 2).
Central portion of neurocranium of: a) Otocinclus batmani, MCP 34087; b) Hypoptopoma inexspectatum, MCP 15744; and c) Acestridium martini, MZUSP 74275. F = frontal; LE = lateral ethmoid; SP = sphenotic. Dorsal view, anterior towards bottom. Scale bar = 1 mm.
7. Shape of the lateral ethmoid.
Retzer et al. (1999) noted that Acestridium dichromum has an elongate, rectangular lateral ethmoid, whereas A. martini has a squarish bone. According to Schaefer (1991) the lateral ethmoid of hypoptopomatines is almost triangular due to the broadening of the posterolateral process of that bone: an observation we corroborate herein (Fig. 3a; state 0). Alternatively, most species of Acestridium have an elongate, rectangular lateral ethmoid (Fig. 3b; state 1), while A. martini and A. colombiensis have a shorter, squarish lateral ethmoid (Fig. 3c; state 2).
Central portion of neurocranium of: a) Hypoptopoma spectabile, MCP 41467; b) Acestridium dichromum, ANSP 161494; and c) A. martini, MZUSP 74275. LE = lateral ethmoid; ME = mesethmoid; OS = orbitosphenoid; PS = parasphenoid; V = vomer. Ventral view, anterior towards top. Scale bar = 1 mm.
8. Length of the posterolateral process of the lateral ethmoid.
The posterolateral process of the lateral ethmoid demonstrates three different states in the examined specimens (Fig. 3). Among species of Acestridium the process is very short in A. martini and A. colombiensis, shorter than half the diameter of the nasal capsule (Fig. 3c; state 2), whereas in all other species of the genus, its length is approximately equal to one-half the diameter of the nasal capsule (Fig. 3b; state 1). Conversely, the posterolateral process of the lateral ethmoid is longer the diameter of the nasal capsule in all remaining taxa studied (Fig. 3a; state 0).
9. Anterior extension of the nasal.
The nasal forms the medial rim of the nasal opening, and varies in the relative positions of its anterior margin relative to the anterior margin of the lateral ethmoid. In Hisonotus, Otocinclus and most species of Acestridium, the anterior margin of the nasal surpasses the anterior margin of the lateral ethmoid (state 0). Conversely, in remaining outgroups and A. dichromum and A. gymnogaster, the nasal only reaches, or falls short of, the anterior margin of the lateral ethmoid (state 1).
Suspensorium and jaws. 10. Articulation between the hyomandibula and the neurocranium [modified from Schaefer, 1998 char. 7].
The hyomandibular dorsal condyle of the hiomandibula of loricariids usually articulates with both the prootic and the compound pterotic, a condition present in Hisonotus maculipinnis among the examined taxa (state 0). In the remaining outgroup taxa, however, the dorsal articular condyle of the hyomandibula contacts only the compound pterotic in Otocinclus and Niobichthys (state 1); or the junction between the compound pterotic and the sphenotic in Hypoptopoma and Oxyropsis (state 2). Finally, in all species of Acestridium, the hyomandibular condyle articulates with the prootic alone (state 3).
11.Levator arcus palatini crest [modified from Armbruster, 2004 char. 44; Paixão & Toledo-Piza, 2009 char. 31; modified from Martins et al., 2014 char. 24].
In most loricarids there is a ridge on the medial side of the hyomandibula for attachment of the levator arcus palatini muscle (LAP). Among the taxa examined the LAP crest is present in Hisonotus maculipinnis and all species of Acestridium (state 0), but the crest is either absent or inconspicuous in Otocinclus, Oxyropsis, Niobichthys, and Hypoptopoma (state 1).
12. Development of the transverse crest of the hyomandibula.
Retzer et al. (1999) described a conspicuous transverse crest on the hyomandibula of Acestridium, running transversely from the anterior of the hyomandibula dorsal articular condyle to the anterior of the condyle for articulation with the opercle. Such a crest provides an attachment area for the levator operculis muscle. Among the examined taxa, such a crest is present but not exposed in the surface of the cheek in all outgroups and most species of Acestridium (Fig. 4a, state 0). Conversely the crest is highly developed and exposed in the surface of the cheek between infraorbital 6 and the opercle in A. martini and A. colombiensis (Fig. 4b; state 1).
Head skeleton and snout plates of: a) Acestridium discus, MZUSP 85321; and b) A. martini, MZUSP 74275. CP = canal plate; HY = hyomandibula; IO = infraorbital; OP = opercle; PR = postrostral plates; R = rostral plate. Lateral view, left side, anterior to left. Scale bar = 2 mm.
13. Large foramen in the hyomandibula [Aquino & Schaefer, 2010 char. 19].
Retzer et al. (1999) described a large, dorsomedially located, circular foramen in the hyomandibula of Acestridium. Among the examined taxa this foramen is present in all species of Acestridium and Oxyropsis wrightiana, which has two foramina (Fig. 5b; state 1), but absent in all outgroups (Fig. 5a; state 0).
Suspensorium of: a) Hisonotus maculipinnis, MCP 48067; and b) Acestridium dichromum, ANSP 161494. AA = anguloarticular; D = dentary; HY = hyomandibula; MPT = metapterygoid; OP = opercle; POP = preopercle Q = quadrate. Lateral view, left side, anterior to left. Scale bar = 1 mm.
14. Subopercular plate [Schaefer, 1991 char. 19; Schaefer, 1998 char. 17].
Some neoplecostomines and hypoptomatines have a small, extra plate termed subopercular plate between the opercle and the cheek canal plate. Among the examined taxa the subopercular plate was found in Hisonotus maculipinnis, Hypoptopoma inexspectatum, Otocinclus arnoldi and Niobichthys ferrarisi (Figs. 6b-c; state 0). Conversely, the subopercular plate is absent in Acestridium, Oxyropsis wrightiana and Hypoptopoma spectabile (Figs. 4, 6a; state 1).
Head skeleton and snout plates of: a) Otocinclus batmani, MCP 34087; b) Niobichthys ferrarisi, MCP 34810; and c) Hypoptopoma inexspectatum, MCP 15744. CP = canal plate; IO = infraorbital; OP = opercle; PR= postrostral plate; R = rostral plate; SOP = subopercular plate. Lateral view, left side, anterior to left. Scale bar = 2 mm.
15. Size and position of the preopercle [Schaefer, 1991 char. 21; Schaefer, 1998 char. 19].
In most loricariids and hypoptopomines the preopercle is well developed and its posterior ramus contacts the adductor crest of the hyomandibula. This condition is found only in Hisonotus maculipinnis among the examined taxa (Fig. 5a; state 0). In all remaining taxa, the preopercle is reduced in size and displaced from the posterior margin of the hyomandibula, being rather articulated to the ventral margins of the quadrate and hyomandibula (Fig. 5b; state 1).
16. Preopercular latero sensory canal [Schaefer, 1991 char. 22; Schaefer, 1998 char. 20; Martins et al., 2014 char. 30].
The preopercle-mandibular latero sensory canal typically passes through the preopercle in most fishes, a condition shared among the examined taxa by Hisonotus maculipinnis, Hypoptopoma spectabile, and H. inexspectatum (Fig. 5a; state 0). Alternatively, the preopercle lacks a sensory canal in the remaining outgroups and all species of Acestridium (Fig. 5b; state 1).
17. Shape of the preopercular latero sensory canal [Schaefer, 1998 char. 21].
The latero sensory canal segment in the preopercle of loricariids is generally straight, a condition present on Hisonotus among the taxa examined (state 0). Conversely, the preopercle canal is semi-circular in the species of Hypoptopoma (state 1). All remaining examined taxa lack a canal in the preopercle and this character was thus coded as inapplicable for these species.
18. Path of the mandibular branch of the laterosensory canal within the fifth infraorbital [Schaefer, 1998 char. 38].
In the primitive condition in loricariids the preoperculo-mandibular sensory canal exits the compound pterotic and then enters either a small suprapreopercular plate or the preopercle directly, whereas the infraorbital sensory canal passes from the sphenotic to the last, usually sixth, infraorbital bone. In most loricariids the preoperculo-mandibular canal terminates in a small plate, the cheek plate or canal plate situated ventral to the preopercle. Among the examined taxa this condition is found in Hisonotus maculipinnis (state 0). Alternatively, in Niobichthys ferrarisi both the infraorbital and the preoperculo-mandibular sensory canals enter the sixth infraorbital bone and remain separate (Fig. 6b), with the canal reaching the cheek canal plate from the fifth infraorbital without passing through the preopercle (state 1). In Hypoptopoma inexspectatum and H. spectabile, however, both canals enter the infraorbital series and remain separate (Fig. 6c), but the mandibular canal passes through the ventral portion of the preopercle before entering into the canal plate (state 2). Finally, in Oxyropsis, Otocinclus, and Acestridium the branch from the compound pterotic disappears and then reappears on the cheek canal plate, without passing through the infraorbital series or the preopercle (state 3).
19. Presence versus absence of palatal splint [modified from Schaefer, 2003 char. 10; Paixão & Toledo-Piza, 2009 char. 20].
The palatal splint (Schaefer, 1987) is an elongate, thin bony shaft extending lateral to the palatine from the palatine cartilage, to the ventral face of the lateral ethmoid in the nasal capsule. Among the examined taxa, the palatal splint is present in the outgroups (state 0). Conversely, the palatal splint is absent in Niobichthys ferrarisi and in all species of Acestridium (state 1).
Hyoid and branchial skeleton. 20. Shape of the anterolateral margin of the anterohyal.
The anterolateral margin of the anterohyal has a conspicuous convex curvature, in the outgroups (Fig. 7a; state 0). Conversely, the anterolateral margin of the anterohyal is concave in all species of Acestridium (Fig. 7b; state 1).
Lower gill arch and hyoid elements of: a) Oxyropsis wrightiana, MCP 34503; and b) Acestridium martini, MZUSP 74275. AF = accessory flange of first ceratobranchial; AH = anterohyal; CB = ceratobranchial; HH = hypohyal; PH = posterohyal; UH = urohyal. Dorsal view, anterior towards top. Scale bar = 1 mm.
21. Presence of an interhyal [modified from Armbruster, 2004 char. 27].
The interhyal of most loricariids is reduced in size or is absent. Among the examined species, the interhyal is absent in Hisonotus maculipinnis and in all species of Acestridium (state 0). Conversely, a small interhyal closely associated with the inner surface of the hyomandibula is present in Otocinclus, Hypoptopoma, Oxyropsis wrightiana, and Niobichthys ferrarisi (state 1).
22. Size of the first ceratobranchial accessory flange [modified from Armbruster, 2004 char. 7; modified from Martins et al., 2014 char. 36].
Most loricariids posses an accessory flange on the first ceratobranchial which projects posterolaterally from the anterior margin of that bone. This accessory flange is unique to loricariids among Siluriformes, and supports the first row of branchial filaments along the anterior of the first ceratobranchial. In outgroups the accessory flange is wide and robust (Fig. 7a; state 0), whereas in Acestridium the accessory flange is small, pointed and fragile (Fig. 7b; state 1).
23. Flange of the first epibranchial [Schaefer, 1997 char. 9; modified from Martins et al., 2014 char. 40].
The epibranchial flange is a laminar extension of that bone extending from its proximal portion and supporting the accessory row of modified gill filaments anterior to the first gill arch (Schaefer, 1997). Such a flange is present in all species of Otocinclus among examined taxa (state 1), but is absent in all remaining species (state 0).
Post-cranial skeleton. 24. Dorsal-fin spinelet [modified from Schaefer, 1998 char. 27; Armbruster, 2004 char. 148; Paixão & Toledo-Piza, 2009 char. 52; modified from Martins et al. 2014 char. 55].
The first dorsal-fin element of loricariids, or spinelet, if present is reduced and articulated to the nuchal plate, sometimes acting as part of the dorsal-fin locking mechanism. The dorsal-fin spinelet is variably present among the examined taxa, being present in Otocinclus arnoldi, O. batmani, and Hisonotus maculipinnis (state 0), but absent in the remaining taxa (state 1).
25. Number of prenuchal plates.
The nuchal plate is a dorsally exposed area of the ossification formed by the sutured or fused first and second dorsal-fin pterygiophores, and perhaps the supraneural (Schaefer, 1987). This is located immediately anterior to the dorsal fin and is present in all loricariids. In addition to the nuchal plate, the species of Acestridium and Niobichthys possess additional unpaired middorsal plates with ventral keels that contact the neural spines of the anterior vertebral centra. Rodriguez & Reis (2007) termed these elements prenuchal plates. These are absent in almost all loricariids, including most outgroups in the present study (state 0). Conversely, one prenuchal plate is present in Acestridium dichromum, A. martini, A. scutatum, A. colombiensis, A. triplax and Niobichthys (state 1), and two or three prenuchal plates are found in A. discus and A. gymnogaster (state 2).
26. Possession of the connecting bone [modified from Schaefer, 2003 char. 30; Paixão & Toledo-Piza, 2009 char. 59; Martins et al., 2014 char. 52].
Most loricariids possess a rod-like connecting bone (Bailey & Baskin, 1976Bailey, R. M. & J. N. Baskin. 1976. Scoloplax dicra, a new armored catfish from the Bolivian Amazon. Occasional Papers of the Museum of Zoology of the University of Michigan, 674: 1-14.) or lateral bone (Schaefer, 1991, 2003), bridging the transverse process of the second dorsal-fin pterygiophore or the nuchal plate anteroventral to the distal portion of the rib of the sixth vertebral centrum. The connecting bone is present among the examined taxa (state 0) except Acestridium colombiensis and A. martini (state 1).
27. Dorsal attachment of the connecting bone.
Most loricariids have the connecting bone attached to the lateral wings of the second dorsal-fin pterygiophore, to the nuchal plate, or to both, a condition present in all outgroups examined (state 0). Conversely, in all species of Acestridium with a connecting bone, the ossification is dorsally attached to the most anterior prenuchal plate (state 1). This character is coded as inapplicable for A. colombiensis and A. martini which lack a connecting bone.
28. Articulation of the neural spine of the sixth vertebral centrum to the parieto-supraoccipital.
Most loricariids have the neural spine of the sixth vertebral centrum articulated with the posterior portion of the parieto-supraoccipital bone, thereby reinforcing the articulation between the vertebral column and cranium. This condition was found in all outgroups examined in this study (state 0). Alternatively, the neural spine of the sixth centrum in Acestridium is small and does not articulate with the parieto-supraoccipital (state 1).
29. Origin of the dorsal fin [modified from Schaefer, 1998 char. 26; modified from Martins et al., 2014 char. 54].
The first pterygiophore of the dorsal fin in most loricariids is attached to the neural spine of the vertebral centrum 7 with the dorsal fin being approximately above the pelvic fins, a condition observed in all outgroups (state 0). Alternatively, in all species of Acestridium the dorsal fin is displaced posteriorly, being above the anal fin, and with its first pterygiophore articulating with the neural spine of the centrum 12 or 13 (state 1).
30. Terminus of the sixth-centrum rib.
The rib associated with the sixth vertebral centrum is large and has a double articulation to the centrum. Its expanded distal tip is articulated to the lateral plates of the body. The terminus of the rib of the sixth centrum can be expanded both anteriorly and posteriorly, as in Acestridium colombiensis, A. discus, A. gymnogaster, A. martini, A. scutatum, Hypoptopoma spectabile, Niobichthys ferrarisi, Otocinclus arnoldi, O. batmani and Oxyropsis wrightiana (state 1), or expanded only posteriorly, as in A. dichromum, A. triplax, H. inexspectatum, and Hisonotus maculipinnis (state 0).
31. Number of bifid hemal spines [Schaefer, 1991 char. 28; Schaefer, 1998 char. 24].
Astroblepids and several loricariids including hypoptopomatines variably possess bifid hemal spines on the vertebral centra posterior to the first anal-fin pterygiophore. The number of bifid hemal spines, however, varies among the examined species. Three or more bifid hemal spines are present in Otocinclus arnoldi, O. batmani, and Hisonotus maculipinnis (state 0); two occur in all species of Acestridium except A. dichromum (state 1), only one bifid hemal spine was found in Hypoptopoma inexspectatum, H. spectabile, Niobichthys ferrarisi, and Oxyropsis wrightiana (state 2). Acestridium dichromum lacks bifid hemal spines (state 3).
32. Degree of development of the pleural ribs.
Pleural ribs posterior to that in the sixth centrum are delicate and fragile in the examined outgroup and most species of Acestridium (state 0). Alternatively, the ribs are well developed and robust in Acestridium dichromum and A. triplax (state 1). Hypoptopoma inexspectatum lacks ribs and this character was coded as inapplicable for that species.
33. Thickness of the pectoral-fin spine.
Among the examined species, Acestridium colombiensis, A. gymnogaster, A. martini and A. scutatum have remarkably weak pectoral-fin spines, which are barely thicker than the first branched fin ray in that fin (state 1). The species in the outgroup and A. discus, A. dichromum, and A. triplax, alternatively have more robust pectoral-fin spines, that are clearly thicker than the first branched fin ray (state 0).
34. Serrations on the inner margin of the pectoral-fin spine [modified from Schaefer, 1991 char. 39; Schaefer, 1998 char. 29; modified from Martins et al., 2014 char. 63].
Serrations are variably present on the inner margin of the pectoral-fin spine among the examined specimens. Some of the outgroups, including Hisonotus maculipinnis, and most species of Acestridium lacks no serrations on the pectoral-fin spine (state 0). Conversely, all other outgroups and A. dichromum and A. triplax have serrations along the inner margin of the pectoral spine (state 1).
35. Length of the ossified portion of the pectoral-fin spine.
In all outgroups and Acestridium martini and A. colombiensis the ossified portion of the pectoral-fin spine is as long as, or longer than, the first branched fin ray (state 0). Conversely, in the remaining species Acestridium the ossified portion of the pectoral-fin spine is shorter, just reaching beyond one-half the length of the first branched fin ray (state 1).
36. Width of the pelvic-fin spine [modified from Armbruster, 2004 char. 177].
In most examined taxa the pelvic-fin spine is thick and well developed (state 0). Alternatively, the pectoral-fin spines are thin and fragile in Acestridium martini and A. colombiensis (state 1).
37. Possession of a lateropterygium [Schaefer, 1987 char. 30; Schaefer, 2003 char. 36; Armbruster, 2004 char. 175; Paixão & Toledo-Piza, 2009 char. 66].
Astroblepids and loricariids share the derived presence of a lateropterygium, a small anterodorsally oriented bone shaft on the lateral region of the basipterygium of the pelvic (Schaefer, 1987). Among the examined taxa the lateropterygium is present in all outgroups (state 0), but absent in all species of Acestridium (state 1).
38. Number of branched rays in the caudal fin [modified from Schaefer, 1991 char. 35; Paixão & Toledo-Piza, 2009 char. 70; Martins et al., 2014 char. 62].
The number of branched caudal-fin rays in very stable among hypoptopomatines, which typically possess 14 branched rays in addition to the two outer, unbranched principal rays. Among the examined species, all outgroup taxa have 14 branched caudal-fin rays (state 0), while most species of Acestridium have 12 rays (state 1), and A. colombiensis and A. martini have 10 rays (state 2).
Dermal plates. 39. Degree of ventral contact of the paired rostral plates.
The snout tip of hypoptopomatines is usually covered by a single large medial plate, often divided at the midline in some groups. In both cases, the ventral margin of the rostral plate - or the ventral margins of each half-rostral plate - diverge laterally and are never in contact along the midventral line (state 0). Alternatively, in Acestridium dichromum, A. gymnogaster, A. scutatum, and A. triplax, the ventral margins of the paired rostral plates are partially united along the ventral midline anteriorly but their posterior portions still diverge laterally (Fig. 8a; state 1). In A. discus, A. martini, A. colombiensis, the ventral margins of the paired rostral plates are completely united at the midline, with their posterior portions not diverging laterally (Figs. 8b-c; state 2).
Spatulate projection of snout of: a) Acestridium triplax, MCP 41746; b) A. discus, MZUSP 85322; and c) A. martini, MZUSP 74275. IO = infraorbital; PR = postrostral plate; R = rostral plate. Ventral view, anterior towards top. Scale bar = 1 mm. Modified from Reis & Lehmann (2009).
40. Infraorbital 1 plate reaching to the ventral border of the snout.
A series of postrostral plates usually form the lateral margin of the snout posterior of the rostral plate in most hypoptopomatines; a condition shared by Hisonotus maculipinnis, Otocinclus, Niobichthys, and most species of Acestridium (Figs. 6a-b, 4a state 0). Alternatively, in Oxyropsis, Hypoptopoma, Acestridium martini, and A. colombiensis the infraorbital 1 is extended ventrolaterally and reaches to and forms part of the ventral border of the snout (Figs. 4b, 6c; state 1).
41. Ventral contact between the contralateral infraorbital 1 plates.
In most of the examined taxa, the infraorbital 1 is usually separated from its lateral counterpart ventrally (Figs. 8a-b; state 0). In Acestridium colombiensis and A.martini, however, the infraorbital 1 is enlarged and displaced ventromedially, such that the contralateral infraorbitals 1 are more ventrally positioned and articulate with each other along midline anterior to the lips (Fig. 8c; state 1). Reis & Lehmann (2009) described this condition in A. martini and A. colombiensis, but termed the ventrally united infraorbitals 1 as a postrostral plate, as illustrated in their fig. 7C.
42. Notch on the dorsal margin of the canal cheek plate (Schaefer, 1998 char. 22).
Most examined taxa have the dorsal margin of the canal plate rounded or straight, without any notch (state 0). Conversely, both examined species of Hypoptopoma have a notch along the dorsal margin of the canal plate, where the sensory canal enters from the preopercle (state 1).
43. Number of postrostral plates (not including canal cheek plate).
Among examined loricariids four or more postrostral plates are present in Hisonotus maculipinnis and Otocinclus species (Fig. 6a; state 0). Conversely, Hypoptopoma, Niobichthys and Oxyropsis possess three postrostral plates (Figs. 6b, c; state 1), whereas all species of Acestridium have lost the most anterior postrostral plates and have only two postrostral plates (Figs. 4b, c; state 2).
44. Presence versus absence of an enlarged, single preanal plate (modified from Schaefer, 1998 char. 36).
The abdominal plates immediately anterior to the anal opening are usually small and similar to the remaining plates covering the abdomen in shape and size, a condition observed in Hisonotus maculipinnis, Otocinclus arnoldi and O. batmani (state 0). In the alternative condition an enlarged, single preanal plate is found in all species of Acestridium and the remaining outgroups (state 1).
45. Number of middle abdominal plate series [modified from Armbruster, 2004 char. 203].
Basal loricariids have no plates covering the abdomen (Reis et al., 2006; Schaefer & Provenzano et al., 2008). Lateral abdominal plates (the thoracic plates of previous authors) and middle abdominal plates, however, are variably present in many loricariid groups. Among the examined specimens, more than one longitudinal series of middle abdominal plates are present between the lateral abdominal plates in Hisonotus maculipinnis and Otocinclus (state 0). Acestridium scutatum, A. triplax, Hypoptopoma inexspectatum, H. spectabile and Niobichthys ferrarisi, have one, sometimes irregular, series of middle abdominal plates (state 1), while A. discus, A. dichromum, A. martini, A. gymnogaster, and A. colombiensis lack middle abdominal plates (state 2).
46. Separation versus contact of lateral abdominal plates along the ventral midline of the abdomen.
In all outgroups and some species of Acestridium the lateral abdominal plates are separated from their counterparts along the ventral midline, by either one or more rows of middle abdominal plates or by a region of naked skin (state 0). Conversely, A. colombiensis, A. dichromum, A. discus and A. martini have the lateral abdominal plates close together and covering the entire ventral surface of the belly, without an intervening skin covered space (state 1).
Miscellaneous. 47. Shape of the caudal peduncle [Paixão & Toledo-Piza, 2009 char. 49].
The caudal peduncle of loricariids is typically round to ovoid in cross section, except in the loricariines, in which it is dorsoventrally flattened. Among hypoptopomatines the caudal peduncle is also ovoid in most species (state 0), but a dorsoventrally flattened condition similar to that of the loricariines is found in Niobichthys, Oxyropsis and Acestridium (state 1). As demonstrated by Retzer et al. (1999), because of the flattening, the vertebrae of species of Acestridium have a marked reduction in the size of both the neural and the hemal spines such that the shape of the vertebrae becomes rectangular and elongate, a condition common to the loricariines. This condition is also present on at least the posterior five or six vertebra of Niobichthys and Oxyropsis wrightiana. Morphological evidence indicates that the depressed trunk in Acestridium and Niobichthys might, however, not be homologous, in that the lateral line canal of Acestridium shifts ventrally to pass posteriorly through the ventral plate series to the caudal-fin base, whereas the canal path is shifted dorsally in Niobichthys (Schaefer & Provenzano, 2008). Optimization of this character herein, however, indicates that this condition is homologous in Acestridium and Niobichthys but not in Oxyropsis.
48. Truncation of the mid-dorsal series of lateral plates [modified from Schaefer, 1998 char. 32; modified from Martins et al., 2014 char. 69].
A complete or almost complete mid-dorsal series of lateral plates is the primitive condition among most loricariids. Loricariines and hypoptopomatines have independently reduced this series several times. Among the examined taxa Hisonotus maculipinnis and the species of Otocinclus share a mid-dorsal lateral series extending to posterior of the dorsal-fin base (state 0). Alternatively, Oxyropsis, Niobichthys, Hypoptopoma, and most species of Acestridium have a short mid-dorsal lateral series consisting of one to five plates and not reaching the dorsal-fin base (state 1). Acestridium martini and A. colombiensis completely lack the mid-dorsal series of lateral plates (state 2).
49. Truncation of the middle series of lateral plates [Schaefer, 1998 char. 33].
The middle series of lateral plates is primitively complete in loricariids, extending from the compound pterotic to the caudal fin, and carrying the lateral line canal (Schaefer, 1998). Among hypoptopomatines, however, the middle series of lateral plates is slightly truncated a few plates in advance of the caudal fin, such that the dorsal and ventral series of plates contact each other along the midlateral line for one to three plates. This is the basal condition for the subfamily, and is present in Hisonotus maculipinnis among the examined species (state 0). Alternatively, in Otocinclus arnoldi, O. batmani, Oxyropsis wrightiana, Hypoptopoma spectabile, and Hypoptopoma inexspectatum the middle series of plates is complete and extends to the caudal fin (state 1). In Acestridium and Niobichthys ferrarisi, however, the middle series in truncated more anteriorly, with the last plate in that series situated between the dorsal and anal fins, such that several plates of the dorsal and ventral series contact each other along the midlateral line (state 2).
50. Interruption of the lateral line [modified from Schaefer, 1991 char. 45; Armbruster, 2004 char. 92].
Most loricariids have a complete lateral line canal that ends near the caudal fin. In several hypoptopomatinae, alternatively, the lateral line is interrupted and divided in two segments which are separated by a gap of five or more plates without a canal below the dorsal fin. This condition occurs in Hisonotus maculipinnis, Otocinclus arnoldi, and O. batmani (state 0). Conversely, the lateral line is complete and uninterrupted in all remaining outgroups and in all species of Acestridium (state 1).
51. Presence versus absence of a spatulate projection of the snout with hypertrophied odontodes.
Species of Acestridium have a characteristic spatulate projection of the snout tip, which bears of a cluster of hypertrophied, retrorse odontodes (Fig. 8; state 1). A spatulate projection laden with enlarged odontodes is absent in all outgroups (state 0).
52. Presence versus absence of an accessory gas bladder formed via an esophageal diverticulum [Schaefer, 1997 char. 26].
Among loricariids only Otocinclus and some members of the Rhinelepini (Armbruster, 1998; Quevedo & Reis, 2002Quevedo, R. & R. E. Reis. 2002. Pogonopoma obscurum: a new species of loricariid catfish (Siluriformes: Loricariidae) from southern Brazil, with comments on the genus Pogonopoma. Copeia, 2002: 402-410.) possess an accessory gas bladder formed by a diverticulum of the esophagus (state 1), which is most parsimoniously interpreted as a convergence between these two groups. Alternatively, the esophageal diverticulum is absent in all other outgroups and all species of Acestridium (state 0).
Phylogenetic analysis. The phylogenetic analysis of the unweighted and unordered data matrix resulted in two maximum parsimonious trees with a length of 92 steps, a consistency index of 0.75 and a retention index of 0.87. These two primary cladograms were summarized in a strict consensus tree shown in Fig. 9. In the following, we provide phylogenetic diagnoses for Acestridium and all groups of species within Acestridium. Characters and character states are listed in the same sequence as presented in the character description section.
Strict consensus tree depicting the phylogenetic relationships of the species of Acestridium, based on a maximum parsimony analysis of the unordered and unweighted matrix. Primary trees = 2; Length = 92 steps; Consistency Index = 0.75; Retention Index = 0.87. Numbers above each branch are Bremer decay indices.
Acestridium Haseman, 1911
Diagnosis.Acestridium is diagnosed as monophyletic based on the possession of the following exclusive synapomorphies: ventral condyle of mesethmoid in the form of uniformly oval disk (char. 2.1); nasal capsule completely closed (char. 6.2); lateral ethmoid elongate and rectangular (char. 7.1); posterolateral process of lateral ethmoid short, approximately equal to on-half diameter of nasal capsule (char. 8.1); articulation between hyomandibula and neurocranium solely through prootic (char. 10.3); anterolateral margin of anterohyal concave (char. 20.1); accessory flange of first ceratobranchial small, pointed and fragile (char. 22.1); connecting bone dorsally attached to most anterior prenuchal plate (char. 27.1); neural spine of sixth vertebral centrum not articulated to posterior portion of parieto-supraoccipital (char. 28.1); dorsal fin displaced posteriorly, situated above anal fin (char. 29.1); two bifid hemal spines present on vertebral centra posterior to first anal-fin pterygiophore (char. 31.1); ossified portion of pectoral-fin spine short, just reaching beyond one-half length of first branched ray (char. 35.1); lateropterygium absent (char. 37.1); caudal fin with 12 branched rays (char. 38.1); ventral margins of paired rostral plates partially united at ventral midline anteriorly but their posterior portions diverging laterally (char. 39.1); only two postrostral plates (char. 43.2); and snout with spatulate projection (char. 51.1). In addition, there are three non-exclusive synapomorphies: presence of levator arcus palatine crest (char. 11.0); presence of large foramen in hyomandibula (char. 13.1); and interhyal absent (char. 21.0).
Acestridium dichromum + A. triplax. The clade is diagnosed as monophyletic based on the possession of one exclusive synapomorphy: the well developed and robust ribs (char. 32.1). In addition, there is also one non-exclusive synapomorphy: the terminal expansion of the tip of the rib on the sixth centrum only posteriorly (char. 30.0).
Acestridium scutatum + A. gymnogaster + A. discus + A. colombiensis + A. martini. The clade is diagnosed as monophyletic based on the possession of one exclusive synapomorphy: the weak pectoral-fin spines, hardly thicker than the first branched fin ray (char. 33.1). In addition, there is also one non-exclusive synapomorphy: the lack of serrations on the pectoral-fin spine (char. 34.0).
Acestridium discus + A. colombiensis + A. martini. The clade is diagnosed as monophyletic based on the possession of two exclusive synapomorphies: the long exposure of the mesethmoid on the dorsum of the snout, extending anteriorly beyond the mesethmoid disc (char. 5.1); and the ventral margins of the paired rostral plates completely united along the ventral midline, with their posterior portions not diverging laterally (char. 39.2). In addition, there is also one non-exclusive synapomorphy: the lateral abdominal plates close together and covering the entire ventral surface of the abdomen (char. 46.1).
Acestridium colombiensis + A. martini. The clade is diagnosed as monophyletic based on the possession of eight exclusive synapomorphies: the short and squarish lateral ethmoid (char. 7.2); the very short posterolateral process of lateral ethmoid, with its length shorter than one-half the diameter of the nasal capsule (char. 8.2); the transverse crest of hyomadibula exposed on the external surface of the cheek between infraorbital 6 and the opercle (char. 12.1); the lack of a connecting bone (char. 26.1); the pelvic-fin spines thin and fragile (char. 36.1); a caudal fin with 10 branched fin rays (char. 38.2); left and right infraorbital 1 plates more ventrally positioned and articulated with each other along the midline ventrally in front of lips (char. 41.1); and the lack of mid-dorsal series of lateral plates (char. 48.2). In addition, there is also two non-exclusive synapomorphies: the ossified portion of pectoral-fin spine as long as, or longer than, the first branched fin ray (char. 35.0); and infraorbital 1 displaced ventrolaterally and reaching and forming part of the ventral border of the snout (char. 40.1).
Discussion
Acestridium is a highly specialized group of loricariids with a very distinctive morphology, making the assessment of their monophyly straightforward. Acestridium was recovered as monophyletic based on the possession of 17 synapomorphies, 14 of which being uniquely derived among the Hypoptopomatini. The position of Acestridium among the hypoptopomatins, however, differed from that suggested by Schaefer (1991, 1998), in which Acestridium discus was sister to the clade Hypoptopoma + Oxyropsis, and Niobichthys was basal to the these three genera. In the present phylogeny (Fig. 9) Acestridium was found to be sister to Niobichthys and this clade is subsequently the sister-group to Oxyropsis + Hypoptopoma. The clade Acestridium + Niobichthys is supported by three characters: the loss of the palatal splint (char. 19.1), the possession of one prenuchal plate (char. 25.1), and the middle series of lateral plates truncated anteriorly, with the last plate in that series situated between the dorsal and anal fins (char. 49.2). There is, however, evidence suggesting that the depressed caudal peduncle and the truncation of the median plate series might have evolved separately in Niobichthys and Acestridium. While Acestridium has the lateral line canal shifted ventrally after the truncation to pass posteriorly through the ventral plate series to the caudal-fin base, the canal path is shifted dorsally in Niobichthys (Schaefer & Provenzano, 1998).
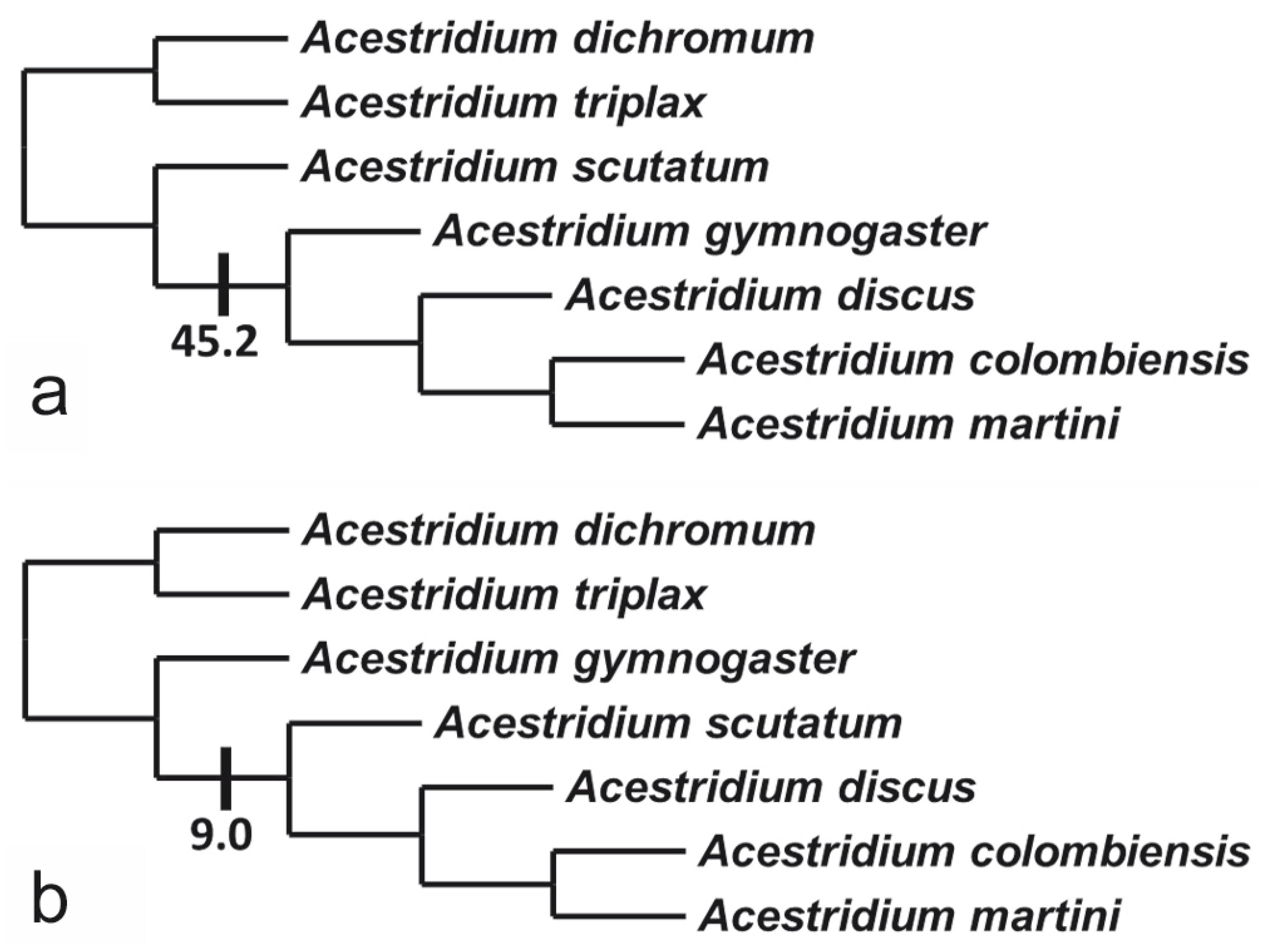
Interrelationships among the species of Acestridium have never been previously investigated, and the present analysis was unable to fully resolve their phylogenetic relationships. Two primary trees were found, and the strict consensus among those alternatives (Fig. 10) resulted in the polytomy involving A. scutatum, A. gymnogaster, and the clade composed of A. discus (A. colombiensis + A. martini). Under that hypothesis, the branch support for the entire genus and for the clade composed of A. colombiensis + A. martini are very high (Fig. 9), indicating that these groups are indeed monophyletic. The remaining branches within Acestridium, however, have low Bremer support and the relationships are not as well supported. Each of the two primary trees has a single character transformation in a branch that collapses under strict consensus. In one of the trees (Fig. 10a), Acestridium gymnogaster appears as the sister-group to the clade formed by A. discus (A. colombiensis + A. martini). These four species share the loss of the middle abdominal plates between the lateral abdominal plates (char. 45.2), which separately evolved in A. dichromum. Conversely, in the other primary tree (Fig. 10b), A. scutatum is recovered as sister to the clade formed by A. discus (A. colombiensis + A. martini), sharing the possession of a long nasal, surpassing the anterior margin of the lateral ethmoid (char. 9.0), a reversion to the condition in most of the rest of the Hypoptopomatini. Resolving this polytomy requires additional evidence (nucleotide sequences).
Two alternative primary trees depicting the phylogenetic relationships of the species of Acestridium.
The known distribution of Acestridium is somewhat patchy, since some species are only known from the type-localities or from very limited regions around their type-localities. Nonetheless, species of Acestridium appear to be endemic to two broad areas of the continent: the Orinoco and Negro basins (A. dichromum, A. discus, A. colombiensis, and A. martini), and the lower Madeira and lower Tapajós region (A. gymnogaster, A. scutatum, and A. triplax). The first divergence within the phylogeny of Acestridium includes components of both areas (Fig. 11), suggesting that the ancestral species of the genus was present in both the Orinoco, Negro and the lower Amazon basins. Subsequent biogeographic events presumably caused speciation between A. dichromum and A. triplax, and between a putative ancestor of A. gymnogaster and A. scutatum and the ancestor of the clade A. discus (A. colombiensis + A. martini). A more complete geographic distribution and a fully resolved phylogeny, is necessary to propose a conclusive biogeographic hypothesis for Acestridium.
Comparative Material Examined.Acestridium colombiensis: Colombia: Guainía, río Orinoco drainage: FMNH 115255, holotype, 50.9 mm SL and FMNH 105169, 9 + 1 c&s paratypes, 38.0-45.6 mm SL, lagoon 1 km upriver from Puerto Inirida, río Orinoco drainage. INHS 99093, 2 paratypes, 37.9-43.2 mm SL, lagoon 1 km upriver from Puerto Inirida. Acestridium dichromum: Venezuela: Amazonas State: ANSP 160692, 1, 40.8 mm SL, río Sipapo, 500 m upstream of Pendare. ANSP 161494, 6+2 c&s, 34.6-50.7 mm SL, outflow stream from series of morichales ca 5 km from mouth of río Pamoni. ANSP 187061, 3, 32.2-38.8 mm SL, río Sipapo, ca. 6 km upstream of Pendare. MCP 35016, 3, 37.4-56.1 mm SL, caño Cucurito, ca. 1 km upstream mouth of río Autana. FMNH 103326, 6 paratypes, 27.2-57.4 mm SL caño Cucurito ca. 1 km above mouth in río Autana. FMNH 105326, 1, 59.8 mm SL, caño Cucurito ca. 1 km above mouth in río Autana. FMNH 105327, 3, 34.4-39.3 mm SL, río Autana at playa Cucurito in front of caño Cucurito. INHS 27642, 1 paratype, 52.9 mm SL, caño Topocho at bridge on highway between Puerto Paez and Puerto Ayacucho. INHS 40427, 1 allotype, 44.7 mm SL, Pozo de Lucas, 7 km from San Fernando de Atabapo. USNM 269949, 25, 27.0-52.0 mm SL, caño Chola, where crossed by road from San Carlos de Rio Negro to Solano. Acestridium discus: Brazil: Amazonas State: FMNH 54339, holotype, 69.1 mm SL and FMNH 54340, 1 paratype, 52.1 mm SL, igarapé Cachoeira Grande, near Manaus. CAS 52153, 1 paratype, 49.4 mm SL, igarapé do Maia, Iraue, Manaus. CAS-SU 64095, 8, 41.9-59.4 mm SL, igarapé da Mãe Joana into rio Negro near Manaus. MHNG 2557.055, 5, 42.2-44.7 mm SL, rio Aracá, rio Negro basin. MHNG 2575.61, 2, 31.2-39.7 mm SL, rio Cuiuni, rio Negro basin. MZUSP 85320, 7+2 c&s, 26.8-56.6 mm SL, igarapé Barroso, tributary to rio Preto da Eva on Francisco Mendes road. MZUSP 85321, 12+2 c&s, 22.4-62.4 mm SL, bathing spot at igarapé do Manu, rio Preto da Eva basin. MZUSP 85322, 9+1 c&s, 16.3-64.5 mm SL, igarapé nearby sítio Bom Jesus, km 13 of road to Francisca Mendes, rio Preto da Eva basin. MZUSP 85323, 6+1 c&s, 18.8-55.3 mm SL, rio Preto da Eva, 15 km N of the town of Rio Preto da Eva. MZUSP 88878, 6, 30.4-64.3 mm SL, bathing spot ca. 2 km downstream of Rio Preto da Eva. MZUSP 88944, 7, 32.8-65.1 mm SL, creek tributary to rio Preto da Eva, after the bathing spot of Encanto da Mata. MZUSP 88962, 15, 19.9-62.1 mm SL, rio Preto da Eva, ca. 4 km upstream of town. MZUSP 88966, 4, 35.4-66.9 mm SL, creek tributary to rio Preto da Eva, after the bathing spot of Encanto da Mata. MZUSP 88986, 10, 17.0-63.4 mm SL, creek on road from Rio Preto da Eva to Pousada do Paraíso. Venezuela: Amazonas State, Río Negro Department: USNM 269947, 2, 43.6 and 46.6 mm SL, shore of río Negro, ca half hour above San Carlos de Río Negro, Río Negro Department. ANSP 165830, 1, 44.0 mm SL, río Matiyure at Achaguas. Acestridium gymnogaster: Brazil: Amazonas State: MCP 37783, 12, 27.9-60.2 mm SL, creek ca 68 km E of rio Madeira on Trans-Amazon road, Humaitá. MCP 37784, 13 + 2 c&s, 25.2-62.0 mm SL, rio Traira, ca. 35 km E of rio Madeira Trans-Amazon road, Humaitá. Acestridium martini: Brazil: Amazonas State, rio Negro basin: MHNG 2576.32, 1, 47.6 mm SL, rio Aracá below confluence with rio Cueiras. MHNG 2577.55, 4, 33.7-44.6 mm SL, rio Aracá. MZUSP 26820, 3+1 c&s, 46.6-48.2 mm SL, rio Cuieiras. MZUSP 61945, 4, 38.6-54.6 mm SL, rio Aiuanã, near Santa Isabel do Rio Negro (=Tapurucuava). MZUSP 74275, 6+1 c&s, 41.6-60.5 mm SL, igarapé Jaradá, tributary to rio Cuieiras, ca. 40 km upstream from mouth. MZUSP 77945, 2, 54.4 and 54.9 mm SL, rio Aiuanã, tributary to rio Negro. USNM 268903, 3, 36.3-57.5 mm SL, rio Negro at Anavilhanas archipelago. CAS-SU 64202, 2, 46.1 and 46.8 mm SL, small creek into igarapé Castanha, tributary to rio Negro, ca. 2 h by boat upstream from Manaus. Venezuela: Amazonas Department: ANSP 160644, 1, 40.8 mm SL, río Sipapo, backwater channel behind sandbar 6-7 km above Pendare. ANSP 160701, 8, 31.9-42.9 mm SL, río Sipapo, ca. 6 km upstream of Pendare. ANSP 165831, 6, 43.3-62.8 mm SL, caño Horeda, at border of Bolívar-Amazonas Territories, ca. 68 km NE of Puerto Ayacucho. MCP 35015, 4 + 1 c&s, 46.5-69.7 mm SL, río Sipapo, above Pendare. FMNH 85827, 2, 42.4 and 59.6 mm SL, laguna Titi near Puerto Atabapo (same as San Fernando de Atabapo) on road past airstrip. FMNH 103323, 34, 33.9-64.4 mm SL, río Sipapo, ca. 1.5 h above Pendare. FMNH 103324, 8, 42.5-51.0 mm SL, río Autana at beach below mouth of caño Cabeza de Manteco. FMNH 103325, 25 paratypes, 39.1-66.8 mm SL, caño Cucurito, ca. 1 km above mouth in río Autana. FMNH 103555, 20 paratypes, 39.6-65.1 mm SL, río Autana at playa Cucurito in front of caño Cucurito. USNM 269945, 2, 42.7 and 49.5 mm SL, caño Manu, tributary of Casiquiare canal, approximately 250 m upstream of Solano, Río Negro Department. USNM 269946, 2, 40.0-51.6 mm SL, small creek of caño Urami, just upriver of Santa Lucia, Río Negro Department. USNM 389920, 3, 47.2-59.1 mm SL, caño Chola, where crossed by road from San Carlos de Río Negro to Solano, Río Negro Department. INHS 40425, 1 allotype, 62.0 mm SL, caño Pozo Azul, río Orinoco drainage. INHS 61564, 6 paratypes, 40.2-57.8 mm SL, caño Pozo Azul, río Orinoco drainage. Acestridium scutatum: Brazil: Amazonas State: MCP 37785, 15+2 c&s, 29.0-62.9 mm SL, rio Traíra, ca. 35 km E of rio Madeira on Trans-Amazon road, Humaitá. MCP 37786, 3, 39.4-43.8 mm SL, rio Maici-Mirim, ca. 45 km E of rio Madeira on Trans-Amazon road, Humaitá. Acestridium triplax: Brazil: Pará State: AMNH 240826, 10, 41.1-51.8 mm SL, paratypes, and MPEG 10122, 3, 48.9-49.1 mm SL, paratypes, igarapé Socó Barroso, Jurutí. AMNH 240827, 10, 32.4-50.2 mm SL, paratypes, MCP 41746, 8 + 2 c&s, 30.6-52.0 mm SL, paratypes, MPEG 12614, 8 + 2 c&s, 41.0-53.5 mm SL, paratypes, ANSP 187168, 10, 36.7-55.4 mm SL, paratypes, MPEG 12492, 4, 23.8-28.9 mm SL, paratypes, and MPEG 12493, 4, 27.4-52.3 mm SL, paratypes, Jurutí. ANSP 187167, 11, 29.2-51.0 mm SL and MPEG 12451, 10, 32.3-49.1 mm SL, Jurutí. MPEG 12467, 10, 35.4-48.0 mm SL, paratypes, Jurutí. MPEG 13355, 55.5 mm SL, holotype, MCP 41745, 9+1 c&s, 41.8-52.8 mm SL, paratypes, and MPEG 12461, 9, 28.9-50.4 mm SL, paratypes, MPEG 10123, 5, 27.8-42.7 mm SL, paratypes, and MPEG 10124, 10, 27.6-49.5 mm SL, paratypes, and MPEG 12476, 5, 37.4-46.8 mm SL, paratypes, igarapé Mutum, lower rio Tapajós basin, Jurutí. MPEG 12462, 5, 34.8-50.0 mm SL, paratypes, rio São Francisco, Jurutí. MPEG 12468, 10, 34.1-53.3 mm SL, paratypes, MPEG 12475, 7, 34.1-52.3 mm SL, paratypes, and MPEG 12453, 1, 53.4 mm SL, paratype, Jurutí. Hisonotus maculipinnis: Argentina: Santa Fé Province: MCP 48067, 2 c&s, 27.8-28.7 mm SL, laguna El Rey, río Salado basin. Hypoptopoma inexspectatum: Brazil: Mato Grosso State: MCP 15744, 24 + 2 c&s, 49.5-51.9 mm SL, rio Paraguay at Cáceres and neighborhood, Cáceres. Hypoptopoma spectabile: Peru: Loreto Department: MCNG 26579, 2 c&s, 21.5 and 25.5 mm SL, río Orinoco drainage; MCP 41467, 2 + 1 c&s, 28.7 mm SL, río Tapiche, sierra del Divisor, Requena. Niobichthys ferrarisi: Venezuela: Amazonas State: MCP 34810, 2 + 1 c&s, 40.6 mm SL, río Bária, 200 m upriver base camp, La Neblina, río Negro basin. Otocinclus arnoldi: Argentina: Santa Fé Province: MCP 48077, 1 c&s, 24.8 mm SL, laguna El Rey, rio Salado basin. Brazil: Rio Grande do Sul State: MCP 25234, 2 c&s, 34.3 and 43.0 mm SL, rio Inhacunda near São Francisco de Assis, rio Uruguai basin, São Francisco de Assis. Otocinclus batmani: Peru: Loreto Department, Provincia de Maynas: ANSP 178616, 1 c&s, 38.4 mm SL, paratype, small creek 25 miles south of Iquitos, tributary of río Amazonas. Oxyropsis wrightiana: Peru: Loreto Department: MCP 34503, 22 + 3 c&s, 42.5-55.3 mm SL, río Pacaya, lago Tamara, río Amazonas basin.
Acknowledgments
The following colleagues contributed specimens and/or hospitality during MSR's visits: William Fink, Sara Fink, and Douglas Nelson (UMMZ), John Lundberg, Mark Sabaj, and Kyle Luckenbill (ANSP), Scott Schaefer and Barbara Brown (AMNH), Richard Vari, Sandra Raredon, and Jeffrey Williams (USNM), David Catania (CAS), Mark Westneat, Mary Anne Rogers, Susan Mochel, Kevin Swagel, and Philip Willink (FMNH), Raphäel Covain and Sonia Fisch-Muller (MHNG). Special thanks to Michael Retzer (INHS), who kindly provided material of Acestridium under his care, during MSR's visit to the FMNH. Philip Willink and Mary Anne Rogers provided special technical support at FMNH. Osvaldo Oyakawa (MZUSP) sent material for this study. Marcelo Gonzaga (UFU) provided access to microphotography equipment. Rubens Pazza and Karine Kavalco (UFV-CRP) made available the resources of their laboratory. Richard Vari, Carl Ferraris Jr. and Pablo Lehmann reviewed the manuscript. RER thanks William Crampton for support at the University of Central Florida where this manuscript was partially completed during a sabbatical semester. MSR was financed by All Catfish Species Inventory (NSF-DEB #0315963) and postdoctoral fellowships from CNPq (process # 152041/2004-7) and FAPESP (process #2007/04701-2). RER is partially financed by CNPq (processes #303362/2007-3, #305180/2010-0 and #207038/2013-9), and Fundação de Amparo à Pesquisa do Estado Rio Grande do Sul - FAPERGS (process # 11/0936-5). MLSD is partially financed by a fellowship from the Coordenação de Aperfeiçoamento do Pessoal de Nível Superior - CAPES-PROSUP.
References
- Aquino, A. E. & S. A. Schaefer. 2002. The temporal region of the cranium of loricarioid catfishes (Teleostei: Siluriformes): morphological diversity and phylogenetic significance. Zoologischer Anzeiger, 241: 223-244.
- Aquino, A. E. & S. A. Schaefer. 2010. Systematics of the genus Hypoptopoma Günther, 1868 (Siluriformes, Loricariidae). Bulletin of the American Museum of Natural History, 336: 1-110.
- Armbruster, J. W. 1998. Modifications of the digestive tract for holding air in Loricariid and scoloplacid catfishes. Copeia, 1998: 663-675.
- Armbruster, J. W. 2004. Phylogenetic relationships of the suckermouth armoured catfishes (Loricariidae) with emphasis on the Hypostominae and the Ancistrinae. Zoological Journal of the Linnean Society, 141: 1-80.
- Arratia, G. & M. Gayet. 1995. Sensory canals and related bones of Tertiary Siluriform crania from Bolívia and North America and comparison with Recent forms. Journal of Vertebrate Paleontology, 15: 482-505.
- Bailey, R. M. & J. N. Baskin. 1976. Scoloplax dicra, a new armored catfish from the Bolivian Amazon. Occasional Papers of the Museum of Zoology of the University of Michigan, 674: 1-14.
- Bremer, K. 1994. Branch support and tree stability. Cladistics, 10: 295-304.
- Britski, H. A. & J. C. Garavello. 1984. Two new southeastern brazilian genera of Hypoptopomatinae and a redescription of Pseudotocinclus Nichols, 1919 (Ostariophysi, Loricariidae). Papéis Avulsos de Zoologia, 35: 225-241.
- Eigenmann, C. H. 1914. On new species of fishes from the Rio Meta Basin of eastern Colombia and on albino or blind fishes from near Bogotá. Indiana University Studies, 23: 229-230.
- Eigenmann, C. H. & R. S. Eigenmann. 1889. Preliminary notes on South American Nematognathi. Proceedings of the California Academy of Sciences, 1: 119-172.
- Fricke, R & W. Eschmeyer. 2014. Guide to Fish Collections. Available from: http://research.calacademy.org/research/ichthyology/catalog/collections.asp (Date of access 15 April 2014).
» http://research.calacademy.org/research/ichthyology/catalog/collections.asp - Gauger, M. F. W. & P. A. Buckup. 2005. Two new species of Hypoptopomatinae from the rio Paraíba do Sul basin, with comments on the monophyly of Parotocinclus and the Otothyrini (Siluriformes: Loricariidae). Neotropical Ichthyology, 3: 509-518.
- Goloboff, P. A. 1999. NONA. Version 2.9. Tucuman, Argentina: Computer software distributed by the author.
- Gosline, W. A. 1945. Catálogo dos Nematognatos de água-doce da América do Sul e Central. Boletim do Museu Nacional, 33: 1-138.
- Haseman, J. D. 1911. Descriptions of some new species of fishes and miscellaneous notes on others obtained during the expedition of the Carnegie Museum to central South America. Annals of Carnegie Museum, 7: 315-328.
- Isbrücker, I. J. H. 1980. Classification and catalogue of the mailed Loricariidae (Pisces, Siluriformes). Verslagen en Technische Gegevens, 22: 1-181.
- Holmberg, E. L. 1893. Dos peces argentinos: Aristommata inexpectata y Liposarcus ambrosetti. Revista Jardin Zoologico Buenos Aires, 1: 353-354.
- Isbrücker, I. J. H. & H. Nijssen. 1974. Rhadinoloricaria gen. nov. and Planiloricaria, two genera of South American mailed catfishes (Pisces, Siluriformes, Loricariidae). Beaufortia, 22: 67-81.
- Lehmann, P. 2006. Otocinclus batmani, a new species of hypoptopomatine catfish (Siluriformes: Loricariidae) from Colombia and Peru. Neotropical Ichthyology, 4: 379-383.
- Maddison, W. P. & D. R. Maddison. 2006. Mesquite: a modular system for evolutionary analysis. Version 1.12. Available from http://mesquiteproject.org
» http://mesquiteproject.org - Martins, F. O., H. A. Britski & F. Langeani. 2014. Systematics of Pseudotothyris (Loricariidae: Hypoptopomatinae). Zoological Journal of the Linnean Society, 170: 822-874.
- Nijssen, H. & I. J. H. Isbrücker. 1987. Spectracanthicus murinus, nouveaux genre et espèce de Poisson-Chat cuirassé du Rio Tapajós, Est. Pará, Brésil, avec des remarques su d'autres genres de Loricariidés (Pisces, Siluriformes, Loricariidae). Revue française Aquariologie et Herpetologie, 13: 93-98.
- Nixon, K. C. 2002. WinClada ver. 1.00.08. Published by the author, Ithaca, NY, USA.
- Paixão, A. C. & M. Toledo-Piza. 2009. Systematics of Lamontichthys Miranda-Ribeiro (Siluriformes: Loricariidae), with the description of two new species. Neotropical Ichthyology, 7: 519-568.
- Quevedo, R. & R. E. Reis. 2002. Pogonopoma obscurum: a new species of loricariid catfish (Siluriformes: Loricariidae) from southern Brazil, with comments on the genus Pogonopoma. Copeia, 2002: 402-410.
- Regan, C. T. 1909. Descriptions of three new freshwater fishes from South America, present to British Museum by Herr J. Paul Arnold. Annals and Magazine of Natural History, 3: 234-235.
- Regan, C. T. 1912. Descriptions of new fishes of the family Loricariidae in the British Museum collection. Proceedings of the Zoological Society of London, 3: 666-670.
- Reis, R. E. 1998. Anatomy and phylogenetic analysis of the neotropical callichthyd catfishes (Ostariophysi, Siluriformes). Zoological Journal of the Linnean Society, 124: 105-168.
- Reis, R. E., E. H. L. Pereira & J. W. Armbruster. 2006. Delturinae, a new loricariid catfish subfamily (Teleostei: Siluriformes), with revisions of Delturus and Hemipsilichthys. Zoological Journal of the Linnean Society, 147: 277-299.
- Reis, R. E., E. H. L. Pereira & P. Lehmann A. 2012. A new genus and species of Hypoptopomatine catfish (Siluriformes: Loricariidae) from the upper Rio São Francisco basin, Brazil. Copeia, 2012: 6-11.
- Reis, R. E. & P. Lehmann A. 2009. Two new species of Acestridium Haseman, 1911 (Loricariidae: Hypoptopomatinae) from the Rio Madeira basin, Brazil. Copeia, 2009: 446-452.
- Reis, R. E., S. O. Kullander & C. J. Ferraris, Jr. 2003. Check list of the freshwater fishes of South and Central America. Porto Alegre, Edipucrs.
- Retzer, M. E., L. G. Nico & F. Provenzano R. 1999. Two new species of Acestridium (Siluformes: Loricariidae) from southern Venezuela, with observations on camouflage and color change. Ichthyological Exploration of Freshwaters, 10: 313-326.
- Retzer, M. E. 2005. Description of a new species of Acestridium (Siluriformes: Loricariidae) from Colombia. Zootaxa, 972: 1-6.
- Rodriguez, M. S. & R. E. Reis. 2007. A new species of Acestridium Haseman, 1911 (Loricariidae: Hypoptopomatinae) from the Eastern Amazon basin, Brazil. Neotropical Ichthyology, 5: 429-434.
- Schaefer, S. A. 1987. Osteology of Hypostomus plecostomus (Linnaeus), with a phylogenetic analysis of the loricariid subfamilies (Pisces: Siluroidei). Contributions in Science, Natural History Museum of Los Angeles County, 394: 1-31.
- Schaefer, S. A. 1991. Phylogenetic analysis of the loricariid subfamily Hypoptopomatinae (Pisces: Siluroidei: Loricariidae), with comments on generic diagnoses and geographic distribution. Zoological Journal of the Linnean Society, 102: 1-41.
- Schaefer, S. A. 1996. Nannoptopoma, a new genus of loricariid catfishes (Siluriformes: Loricariidae) from the Amazon and Orinoco river basins. Copeia, 1996: 913-926.
- Schaefer, S. A. 1997. The Neotropical cascudinhos: Systematics and biogeography of the Otocinclus catfishes (Siluriformes: Loricariidae). Proceedings of the Academy of Natural Sciences of Philadelphia, 148: 1-120.
- Schaefer, S. A. 1998. Conflict and resolution: Impact of new taxa on phylogenetic studies of the Neotropical cascudinhos (Siluroidei: Loricariidae). Pp. 375-400. In: Malabarba, L. R., R. E. Reis, R. P. Vari, Z. M. Lucena & C. A. S. Lucena (Eds.). Phylogeny and classification of Neotropical fishes. Porto Alegre, Edipucrs.
- Schaefer, S. A. 2003. Relationships of Lithogenes villosus Eigenmann, 1909 (Siluriformes, Loricariidae): evidence from high-resolution computed microtomography. American Museum Novitates, 3401: 1-55.
- Schaefer, S. A. & F. Provenzano R. 1998. Niobichthys ferrarisi, a new genus and species of armored catfish from southern Venezuela (Siluriformes: Loricariidae). Ichthyological Exploration of Freshwaters, 8: 221-230.
- Schaefer, S. A. & F. Provenzano R. 2008. The Lithogeninae (Siluriformes, Loricariidae): anatomy, interrelationships and description of a new species. American Museum Novitates, 3637: 1-49.
- Taylor, W. R. & G. C. van Dyke. 1985. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium, 9: 107-119.
Appendix 1.
Character state transformations by clade, with only unambiguous changes listed, except for the Hypoptopomatini, in which transformations are presented under ACCTRAN optimization. Transformations are listed with the character number first and then the plesiomorphic > apomorphic state. Transformations listed in bold are exclusive synapomorphies.

Publication Dates
-
Publication in this collection
02 June 2015 -
Date of issue
Apr-Jun 2015
History
-
Received
02 Sept 2014 -
Accepted
23 Oct 2014