ABSTRACT
The feeding ecology of five elasmobranch species was studied on the southern coast of Rio de Janeiro, southwestern Atlantic Ocean. The specimens were caught with a trawl or bottom longline between January 2006 and August 2007. The diets of Psammobatis rutrum and Psammobatis extenta appeared to be basically carcino-benthophagous, with a very small niche width. Rioraja agassizii was basically carcino/ichthyo-benthophagous, also with a narrow niche, including teleost fish and shrimp, but in different proportions according to age and sex. Rhizoprionodon lalandii was ichthyophagous, not influenced by age or sex. The analyses of these species and Atlantoraja cyclophora indicated two trophic groups: one composed of the smaller rays, P. extenta and P. rutrum, which basically fed on small crustaceans, polychaetes and nematodes; and the other of R. lalandii and the larger rays R. agassizii and A. cyclophora, which preyed on teleost fish and brachyurans.
Keywords:
Alimentary index; Carcharhinidae; Demersal fishes; Rajidae
RESUMO
Devido à lacuna de conhecimento sobre os elasmobrânquios estudamos a ecologia alimentar de cinco espécies desse grupo, no litoral sul do Rio de Janeiro. Os exemplares foram capturados por rede de arrasto e espinhel de fundo, entre janeiro de 2006 e agosto de 2007. As dietas de Psammobatis rutrum e Psammobatis extenta se mostraram basicamente carcino-bentofagas, com uma largura de nicho estreito. Rioraja agassizii foi considerada carcino/ictio-bentófaga, também com uma largura de nicho estreita. A espécie se alimenta com peixe e camarão, mas em proporções diferentes de acordo com a idade e sexo. A dieta de Rhizoprionodon lalandii foi considerada ictiófaga e não é influenciada pela idade ou sexo. As análises destas espécies e Atlantoraja cyclophora indicaram a formação de dois grupos tróficos: um composto pelas raias menores, P. extenta e P. rutrum, que basicamente se alimentam de pequenos crustáceos, poliquetos e nematodas; e o outro de R. lalandii e as raias maiores R. agassizii e A. cyclophora, que predam peixes teleósteos e brachyuras.
Palavras-chave:
Carcharhinidae; Índice alimentar; Peixes demersais; Rajidae
Introduction
Studies on the feeding of organisms in the natural environment are important mainly to extend knowledge of their trophic ecology, including resource sharing and competition, for the understanding, administration and management of ecosystems (Valls et al., 2011Valls M, Quetglas A, Ordines F, Moranta J. Feeding ecology of demersal elasmobranchs from the shelf and slope off the Balearic Sea (western Mediterranean). Sci Mar. 2011; 75(4):633-39.; Braga et al., 2012Braga RR, Bornatowski H, Vitule JRS. Feeding ecology of fishes: an overview of worldwide publications. Rev Fish Biol Fish. 2012; 22(4):915-29.; Estalles et al., 2015Estalles ML, Perier MR, Di Giácomo EE. Trophic ecology of the smallnose fanskate Sympterygia bonapartii in the San Matías Gulf, northern Patagonia, Argentina. Ichthyol Res. 2015; 63(2):207-17.). These studies contribute to explaining the variations in growth, reproduction, migration and predation behavior. Changes in food availability in the environment can lead to changes in the diet of a species, and species with broad geographic distributions may also show differences in the proportion of items in their diets over their area of occurrence (Vianna et al., 2000Vianna M, Arfelli CA, Amorim AF. Feeding of Mustelus canis (Elasmobranchii, Triakidae) caught off south-southeast coast of Brazil. Bol Inst Pesca. 2000; 26(1):79-84.; Bornatowski et al., 2014aBornatowski H, Braga RR, Abilhoa V, Corrêa MFM. Feeding ecology and trophic comparisons of six shark species in a coastal ecosystem off southern Brazil. J Fish Biol . 2014a; 85(2):246-63.; Carmo et al., 2015Carmo WPD, Bornatowski H, Oliveira EC, Fávaro LL. Diet of the chola guitarfish, Rhinobatos percellens (Rhinobatidae), in the Paranaguá estuarine complex. An Acad Bras Cienc. 2015; 87(2):721-31.; Viana, Vianna, 2014Viana AF, Vianna M. The feeding habits of the eyespot skate Atlantoraja cyclophora (Elasmobranchii: Rajiformes) in southeastern Brazil. Zoologia (Curitiba). 2014; 31(2):119-25.; Estalles et al., 2015Estalles ML, Perier MR, Di Giácomo EE. Trophic ecology of the smallnose fanskate Sympterygia bonapartii in the San Matías Gulf, northern Patagonia, Argentina. Ichthyol Res. 2015; 63(2):207-17.). In particular, knowledge of the feeding ecology of elasmobranchs is essential, as they play an important role in marine ecosystems, where they occupy the upper trophic levels (Vianna et al., 2000Vianna M, Arfelli CA, Amorim AF. Feeding of Mustelus canis (Elasmobranchii, Triakidae) caught off south-southeast coast of Brazil. Bol Inst Pesca. 2000; 26(1):79-84.; Ebert, Bizzarro, 2007Ebert DA, Bizzarro JJ. Standardized diet compositions and trophic levels of skates (Chondrichthyes: Rajiformes: Rajoidei). Environ Biol Fishes. 2007; 80(2); 80:221-37.; Valls et al., 2011Valls M, Quetglas A, Ordines F, Moranta J. Feeding ecology of demersal elasmobranchs from the shelf and slope off the Balearic Sea (western Mediterranean). Sci Mar. 2011; 75(4):633-39.; Bornatowski et al., 2014cBornatowski H, Navia AF, Braga RR, Abilhoa V, Corrêa MFM. Ecological importance of sharks and rays in a structural foodweb analysis in southern Brazil. ICES J Mar Sci. 2014c; 71(7):1586-92.; Estalles et al., 2015Estalles ML, Perier MR, Di Giácomo EE. Trophic ecology of the smallnose fanskate Sympterygia bonapartii in the San Matías Gulf, northern Patagonia, Argentina. Ichthyol Res. 2015; 63(2):207-17.).
Recently, Brazil was identified by Food and Agriculture Organization of the United Nations (FAO) as the largest shark sub-products consumer market (Barreto et al., 2016Barreto R, Ferretti F, Flemming JM, Amorim A, Andrade H, Worm B, Lessa R. Trends in the exploitation of South Atlantic shark populations. Conserv Biol. 2016; 30(4):792-804.). Since 1980, because of the high value of their fins and other byproducts, and also because of the depletion of traditional fishery resources such as teleosts, fishing for sharks and rays has intensified (Vooren, Klippel, 2005Vooren CM , Klippel S. Diretrizes para a conservação de espécies ameaçadas de elasmobrânquios. In: Vooren CM, Klippel S. Ações para a conservação de tubarões e raias do Brasil. Porto Alegre: Igaré; 2005. p.213-28.; Andrade et al., 2008Andrade AC, Silva Junior LC, Vianna M. Reproductive biology and population variables of the Brazilian sharpnose shark Rhizoprionodon lalandii (Müller & Henle, 1839) captured in coastal waters of south-eastern Brazil. J Fish Biol . 2008; 72(3):473-84.; Bornatowski et al., 2014bBornatowski H, Braga RR, Vitule JRS. Threats to sharks in a developing country: The need for effective and simple conservation measures. Nat Conservacao. 2014b; 12(1):11-18.; Dulvy et al., 2014Dulvy KN, Fowler SL, Musick JA, Cavanagh RD, Kyne PM, Harrison LR et al. Extinction risk and conservation of the world’s sharks and rays. eLife [serial on internet]. 2014; 3:e00590. DOI:10.7554/eLife.00590.
https://doi.org/10.7554/eLife.00590...
; Davidson et al., 2015Davidson LNK, Krawchuk MA, Dulvy NK. Why have global shark and ray landings declined: improved management or overfishing?. Fish Fisheries. 2015; 17(2):438-58.), in addition to the already large numbers taken in bycatch (Costa, Chaves, 2006Costa L, Chaves PTC. Elasmobrânquios capturados pela pesca artesanal na costa sul do Paraná e norte de Santa Catarina, Brasil. Biota Neotrop [serial on internet]. 2006; 6(3). doi: 10.1590/S1676-06032006000300007.
https://doi.org/10.1590/S1676-0603200600...
; Bornatowski et al., 2014bBornatowski H, Braga RR, Vitule JRS. Threats to sharks in a developing country: The need for effective and simple conservation measures. Nat Conservacao. 2014b; 12(1):11-18.). Currently, the price per kilogram of shark fins is about $1,697 and the price per individual for marine species ranges from $11,121 for dwarf sawfish to $341,139 for the whale shark (McClenachan et al., 2016McClenachan L, Cooper AB, Dulvy NK. Rethinking trade-driven extinction risk in marine and terrestrial megafauna. Curr Biol. 2016; 26(12):1640-46.).
Intensive fisheries targeting elasmobranchs have an initial phase of large catches, followed by a rapid decline until collapse, since these fish have low rates of population increase and therefore low productivity of the stock. For example, in the South Atlantic, three phases of exploitation of sharks were identified. From 1979 to 1997, the shark catch rates increased, during a period with low fishing effort; then from 1998 to 2008 the catch rates decreased and the fishing effort increased; and from 2008 to 2011 shark catches remained stable or increased and the fishing effort was again low (Barreto et al., 2016Barreto R, Ferretti F, Flemming JM, Amorim A, Andrade H, Worm B, Lessa R. Trends in the exploitation of South Atlantic shark populations. Conserv Biol. 2016; 30(4):792-804.). Consequently, devising strategies for fishery management and biodiversity conservation is a complex matter (Vooren, Klippel, 2005Vooren CM , Klippel S. Diretrizes para a conservação de espécies ameaçadas de elasmobrânquios. In: Vooren CM, Klippel S. Ações para a conservação de tubarões e raias do Brasil. Porto Alegre: Igaré; 2005. p.213-28.; Andrade et al., 2008Andrade AC, Silva Junior LC, Vianna M. Reproductive biology and population variables of the Brazilian sharpnose shark Rhizoprionodon lalandii (Müller & Henle, 1839) captured in coastal waters of south-eastern Brazil. J Fish Biol . 2008; 72(3):473-84.; Dulvy et al., 2014Dulvy KN, Fowler SL, Musick JA, Cavanagh RD, Kyne PM, Harrison LR et al. Extinction risk and conservation of the world’s sharks and rays. eLife [serial on internet]. 2014; 3:e00590. DOI:10.7554/eLife.00590.
https://doi.org/10.7554/eLife.00590...
; Davidson et al., 2015Davidson LNK, Krawchuk MA, Dulvy NK. Why have global shark and ray landings declined: improved management or overfishing?. Fish Fisheries. 2015; 17(2):438-58.), principally in developing countries such as Brazil (Bornatowski et al., 2014bBornatowski H, Braga RR, Vitule JRS. Threats to sharks in a developing country: The need for effective and simple conservation measures. Nat Conservacao. 2014b; 12(1):11-18.), where the fauna is highly endemic in the southeastern region, with high species richness (Lucifora et al., 2011Lucifora LO, García VB, Worm B. Global diversity hotspots and conservation priorities for sharks. PLoS ONE. 2011; 6(5):e19356. doi:10.1371/journal.pone.0019356.
https://doi.org/10.1371/journal.pone.001...
; Davidson, Dulvy, 2017Davidson LNK, Dulvy NK. Global marine protected areas to prevent extinctions. Nat Ecol Evol. 2017; 40. DOI:10.1038/s41559-016-0040.
https://doi.org/10.1038/s41559-016-0040...
).
The elasmobranchs studied here are small-sized species that are often caught in coastal demersal fisheries. They include three species of Rajidae and one member of Carcharhinidae. Psammobatis rutrum Jordan, 1891, the Spade sandskate, is found in the South Atlantic from Rio de Janeiro to northern Argentina, at depths of 37 to 100 m. Psammobatis extenta (Garman, 1913), the Zipper sandskate, occurs in the South Atlantic from Rio de Janeiro to Argentina, from 20 m to 115 m depths, and is endemic to the Argentine Marine Zoogeographic Province. Rioraja agassizii (Müller & Henle, 1841), the Rio skate, is endemic to the South Atlantic and occurs from Espírito Santo to northern Argentina, at depths up to 130 m. Rhizoprionodon lalandii (Valenciennes, 1839), the Brazilian sharpnose shark, is found from Panama to Santa Catarina, at depths from 3 to 70 m (Gomes et al., 2010Gomes UL, Signori CN, Gadig OBF, Santos HRS. Guia para identificação de tubarões e raias do Rio de Janeiro. Rio de Janeiro: Technical Books; 2010.).
According to the Red List of the International Union for Conservation of Nature (IUCN), data deficient are available to evaluate the conservation status of P. rutrum and R. lalandii; in Brazil, the shark is considered vulnerable (Kyne, 2007Kyne PM, San Martín J, Stehmann MFW. Rioraja agassizii. The IUCN Red List of Threatened Species . 2007 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
http://www.iucnredlist.org/...
;Rosa et al., 2004Rosa RS, Gadig OBF, Santos Motta F, Namora RC. Rhizoprionodon lalandii. The IUCN Red List of Threatened Species . 2004 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
http://www.iucnredlist.org/...
). Psammobatis extenta is classified as of least concern, although in Brazil more information about the species is required (Chiaramonte, 2004Chiaramonte GE. Psammobatis extenta. The IUCN Red List of Threatened Species. 2004 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
http://www.iucnredlist.org/...
). Rioraja agassizii is classed as vulnerable because of heavy fishing pressure (Kyne et al., 2007Kyne PM, San Martín J, Stehmann MFW. Rioraja agassizii. The IUCN Red List of Threatened Species . 2007 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
http://www.iucnredlist.org/...
).
Since knowledge of feeding ecology is important and only incomplete information is available for all these species, the present study evaluated their feeding habits and the food overlap among them. The goal of this study was to generate information about the diets of Psammobatis rutrum (voucher UERJ 2142; UERJ = Universidade do Estado do Rio de Janeiro), Psammobatis extenta (UERJ 2125), Rioraja agassizi (UERJ 2147) and Rhizoprionodon lalandii (UERJ 2168) in southeastern Brazil, by identifying the main food items of the species; determine differences in their diets according to the sex and stage of maturity; and investigate the diet overlap of these four species, as well as of Atlantoraja cyclophora (UERJ 1256) (Viana, Vianna, 2014Viana AF, Vianna M. The feeding habits of the eyespot skate Atlantoraja cyclophora (Elasmobranchii: Rajiformes) in southeastern Brazil. Zoologia (Curitiba). 2014; 31(2):119-25.).
Material and Methods
In southeastern Brazil, southwestern Atlantic Ocean (22°50-23°20 S; 44°00-44°45 W) (Fig. 1), 14 sampling campaigns in a fishing boat were carried out between January 2006 and August 2007, sampling at depths between 30 and 60 m, with the objective of capturing the demersal ichthyofauna. The vessel operated with double trawl nets with a body and sleeves, with a 20 mm mesh and 18 mm distance between adjacent knots, and trawls lasting one hour. In parallel, a bottom longline was set, with 100 hooks of different sizes (0.2, 0.3, 0.4 and 0.5), spaced 5m apart and attached to the main cable with a 30 cm snood. This gear was paid out before dawn and retrieved during the morning.
Location of samples taken between January 2006 and August 2007 on the southern coast of Rio de Janeiro, southeastern Brazil, E = longline; A= trawl.
In the laboratory, the specimens were identified with appropriate references (Gomes et al., 2010Gomes UL, Signori CN, Gadig OBF, Santos HRS. Guia para identificação de tubarões e raias do Rio de Janeiro. Rio de Janeiro: Technical Books; 2010.); measured, in centimeters (disc width for the rays - DW, and total length - TL, for the shark); weighed, in grams (total weight - TW); and the sex was recorded. Next, they were dissected and the stage of maturity (juveniles or adults) was determined; and the stomachs were removed, weighed (g), fixed in 10% formaldehyde, and preserved in 70% ethanol. The contents of each stomach were identified under a stereoscopic microscope and the items found were weighed and identified to the lowest possible taxonomic level. The smallest weight detectable with the balance employed was 0.01 g.
The cumulative prey curve was used to evaluate the reliability of the sample size used to analyze the diet of each species, a strategy recommended by Cortés (1997Cortés E. A critical review of methods of studying fish feeding based on analysis of stomach contents: Application to elasmobranch fishes. Can J Fish Aquat Sci. 1997; 54(3):726-38.) and Ferry, Cailliet (1996Ferry LA, Cailliet GM. Sample size and data analysis: are we characterizing and comparing diet properly? In: MacKinlay D, Shearer K, eds. Feeding Ecology and Nutrition in Fish. San Francisco: American Fisheries Society; 1996. p.71-80.), among others. This curve was constructed with the use of the program EstimateS Win820, with 50 randomizations.
The food items found were analyzed for Frequency of Occurrence (%FO) (Hynes, 1950Hynes HBN. The food of fresh-water sticklebacks (Gasterosteus aculeatus and Pygosteus pungitius), with a review of methods used in studies of the food of fishes. J Anim Ecol. 1950; 19(1):36-58. doi: 10.2307/1570.
https://doi.org/10.2307/1570...
), calculated as the percentage of stomachs in which a certain item occurs in relation to the total number of stomachs; and the Percentage Weight (%W), which is the percentage of the weight of a certain item in relation to the total weight of all items (Jones, 1973Jones A. The ecology of young turbot, Scophthalmus maximus (L.), at Borth, Cardiganshire, WaIes. J Fish Biol .1973; 5(3):367-83.). To determine the relative importance of each item in the diet, the Alimentary Index (%IAi) was calculated according to Kawakami and Vazzoler (1980Kawakami E, Vazzoler G. Método gráfico e estimativa de índice alimentar aplicado no estudo de alimentação de peixes. Bol Inst Oceanogr. 1980; 29(2):205-07.), modified by using the Percentage Weight (%W) instead of the Percentage Volume (%V). This is described by the equation: %IAi
1 = ((%FO
1 × %W
1) / ∑ (%FO
T × %W
T)) × 100; where: %FO
1 = percentage of stomachs in which a certain item occurs %W
1 = percentage of weight of a certain item.
The Frequency of Occurrence, Percentage Weight and Alimentary Index were calculated separately for the items and for the food groups (Crustaceans, Teleosts, Polychaetes, Nematodes and Elasmobranchs). The method of Costello (1990Costello MJ. Predator feeding strategy and prey importance: a new graphical analysis. J Fish Biol . 1990; 36(2):261-63.) as adapted by Amundsen et al. (1996Amundsen PA, Gabler HM, Staldvik FJ. A new approach to graphical analysis of feeding strategy from stomach contents data - modification of the Costello (1990) method. J Fish Biol. 1996; 48(4):607-14.) was used to evaluate the feeding strategy and the trophic-niche width.
Because of the difficulty of taxonomic identification of bony fishes in an advanced state of digestion, the fish were grouped into a single item. In contrast, crustaceans, which are easily identified by their exoskeleton, could be differentiated at lower taxonomic levels, in some cases to genus.
To evaluate the degree of dietary specialization, the trophic-niche width was calculated according to the standardized index of Levins (Hurlbert, 1978Hurlbert SH. The measurement of niche overlap and some relatives. Ecology. 1978; 59(1):67-77.). The values of the index also range from 0 to 1; values close to 0 correspond to a more specialized diet, and values close to 1 indicate a generalist diet. The following equation was used: Bi = [(∑jPij2)-1-1] (n-1)-1; where: Pij = The proportion of prey j in the diet of predator i n = number of prey categories.
The SIMPER (analyses of percentage of similarity) was used to determine the percentage of similarity between the sexes and the ontogenetic stages, and to estimate the contribution of each prey category. Comparative analyses of the diet between the sexes and the stages of maturity were not carried out for P. rutrum because of the small number of specimens obtained. For P. extenta, only the sex ratio was determined, because of the small number of juveniles caught.
To evaluate the similarity between the diets of the species and to investigate the formation of trophic groups, a Correspondence Analysis was carried out (Zar, 1999Zar JH. Biostatistical Analysis. 4th ed. New Jersey: Prentice-Hall; 1999.) in the Statistica program, applied to the data for percentage of items in the diet, considering males, females, juveniles and adults separately. This analysis included the data for Atlantoraja cyclophora (Tab. 1), collected in the same sampling campaigns and previously reported by Viana, Vianna (2014Viana AF, Vianna M. The feeding habits of the eyespot skate Atlantoraja cyclophora (Elasmobranchii: Rajiformes) in southeastern Brazil. Zoologia (Curitiba). 2014; 31(2):119-25.).
Frequency of occurrence (%FO), percentage weight (%W) and alimentary index (%IAi) of prey items in the total diet of Atlantoraja cyclophor a in southeastern Brazil, between January 2006 and August 2007. Published by Viana, Vianna (2014Viana AF, Vianna M. The feeding habits of the eyespot skate Atlantoraja cyclophora (Elasmobranchii: Rajiformes) in southeastern Brazil. Zoologia (Curitiba). 2014; 31(2):119-25.).
Results
Psammobatis rutrum. Only nine stomachs were analyzed, all of them with contents. The individuals measured between 13.5 and 16.0 cm disc width; all were adults, including four females (15.2-16.0 cm) and five males (13.5-14.0 cm).
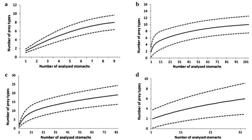
The cumulative prey curve (Fig. 2a) rose steadily with no tendency to stabilize, indicating that analyzing a larger number of stomachs would be necessary for a more complete dietary study for this species in the region. Three groups of food items were found: Crustaceans, Teleosts and Polychaetes (Tab. 2). Crustaceans were the most important component of the diet, and Teleosts and Polychaetes were also important.
Cumulative prey curve for the diet of (a) Psammobatis rutrum; (b) Psammobatis extenta; (c) Rioraja agassizii; (d) Rhizoprionodon lalandii ± Confidence Intervals (p=0.05) in the southeastern Brazil, between January 2006 and August 2007.
Frequency of occurrence (%FO), percentage weight (%W) and alimentary index (%IAi) of prey items in the total diet of Psammobatis rutru m in the southeastern Brazil, between January 2006 and August 2007.
Evaluation of the feeding strategy and niche width (Fig. 3) did not indicate the presence of a single dominant item in the diet, but of items that were important for some individuals (Fig. 3a). With respect to the groups (Fig. 3b), Crustaceans were dominant, Teleosts were important for some individuals, and Polychaetes were rare. The diet of P. rutrum appeared to be basically carcino-benthophagous. The value of the trophic-niche width was 0.08, indicating a very narrow niche with a high importance of a few items and no single item dominating.
Diet of species analyzed by Costello’s method (1990Costello MJ. Predator feeding strategy and prey importance: a new graphical analysis. J Fish Biol . 1990; 36(2):261-63.) as adapted by Amundsen et al. (1996Amundsen PA, Gabler HM, Staldvik FJ. A new approach to graphical analysis of feeding strategy from stomach contents data - modification of the Costello (1990) method. J Fish Biol. 1996; 48(4):607-14.), in the southeastern Brazil, between January 2006 and August 2007, by food items and by groups. (a/b) Psammobatis rutrum: a. Food items: 1) Shrimp (not identified), 2) Teleost fragments, 3) Crustacean fragments, 4) Amphipoda, 5) Brachyura 6) Polychaeta. b. Groups: Crustacea (CRU), Teleosts (TEL) and Polychaeta (POL). (c/d) Psammobatis extenta: c. Food items: 1) Shrimp (not identified), 2) Teleost fragments, 3) Isopoda, 4) Stomatopoda, 5) Nematoda, 6) Polychaeta, 7) Brachyura, 8) Crustacean fragments, 9) Amphipoda. d. Groups: Crustacea (CRU), Teleosts (TEL), Polychaeta (POL), Nematoda (NEM). (e/f) Rioraja agassizii: e. Food items: 1) Teleost fragments, 2) Shrimp (not identified), 3) Brachyura, 4) Crustacean Fragments, 5) Thalassinidae, 6) Nematoda, 7) Stomatopoda, 8) Amphipoda, 9) Lophogastrida. f. Groups: Crustacea (CRU), Teleosts (TEL), Nematoda (NEM). (g/h) Rhizoprionodon lalandii: g. Food items: 1) Teleost fragments, 2) Dactylopterus volitans, 3) Triglidae, Merluccius hubbsi and elasmobranchs, 4) Loligo plei. h. Groups: Teleosts (TEL), Elasmobranchs (ELA), Cephalopoda (CEF).
Psammobatis extenta. The stomach contents of 83 individuals were analyzed; only one lacked contents and was omitted from the analyses. The specimens measured between 9.5 cm and 16.3 cm disc width. Of the 82 stomachs analyzed, 40 were from females (9.5-16.3 cm DW) and 42 from males (11.1-15.8 cm DW); 8 were from juveniles (9.5-12.4 cm) and 74 from adults (13.0-16.3 cm). The adult male with the smallest DW measured 13.0 cm, and the smallest adult females measured 13.4 cm.
The cumulative prey curve (Fig. 2b) stabilized with about 50 stomachs analyzed, which indicates that the number of stomachs examined was adequate for a dietary study of this species in the sampled area. The items found were Crustaceans, Teleosts, Polychaetes and Nematodes (Tab. 3). Crustaceans were the most important, mainly because of the importance of the caridean shrimp Leptochela sp., which showed the highest value of %IAi, followed by unidentified Shrimp.
Frequency of occurrence (%FO), percentage weight (%W) and alimentary index (%IAi) of prey items in the total diet of Psammobatis extent a in the southeastern Brazil, between January 2006 and August 2007, total and according to sex.
The evaluation of the feeding strategy and niche width (Fig. 3) indicated that unidentified Shrimp was the dominant item in the diet, with the presence of diverse rare items such as Stomatopoda, unidentified Nematodes, and Amphipoda, among others (Fig. 3c). With respect to the groups (Fig. 3d), Crustaceans were dominant, Teleosts were important in the diet of only some individuals, and Polychaetes and Nematodes were rare. The species showed a carcino-benthophagous diet. The index of niche width was 0.15, indicating that P. extenta has a narrow niche, with high importance of a single item and low importance of the others, in agreement with the result obtained using the method of Costello (1990Costello MJ. Predator feeding strategy and prey importance: a new graphical analysis. J Fish Biol . 1990; 36(2):261-63.).
The SIMPER analyses showed a similarity of 43.82% between the sexes, and Leptochela sp. contributed 31.35% of this similarity. Crustaceans showed high importance for both sexes, followed by Teleosts. Shrimp was the most important item; for females, Leptochela sp. was the main item, and for males, unidentified Shrimp was the most important (Tab. 3).
Rioraja agassizii. The stomach contents of 112 individuals were analyzed, of which eight were empty and were omitted from the analysis. The specimens analyzed measured between 14.8 and 43.3 cm disc width. Of the total of 104 stomachs, 55 were from females (15.1-43.3 cm DW) and 49 from males (14.8-31.6 cm DW); with 22 from juveniles (14.8-35.5 cm DW) and 82 from adults (25.1-43.3 cm DW). The adult male with the smallest DW measured 25.1 cm, and the smallest adult female measured 33.4 cm.
The cumulative prey curve (Fig. 2c) tended to stabilize with about 95 stomachs analyzed, indicating that the number of stomachs was sufficient to study the species’ diet in the sampling area. The food items were Crustaceans, Teleosts and Nematodes (Tab. 4). Crustaceans were the most important group with respect to %FO, %W and %IAi, followed by Teleosts. Of these items, Teleost fragments were most important, together with Portunidae.
Frequency of occurrence (%FO), percentage weight (%W) and alimentary index (%IAi) of prey items in the total diet of Rioraja agassizi i in the southeastern Brazil, between January 2006 and August 2007, total, according to sex and stage of maturity.
The evaluation of the feeding strategy and niche amplitude (Fig. 3) indicated that Teleost fragments were the dominant item, and unidentified Shrimp was also important (Fig. 3e). Of the groups (Fig. 3f), Crustaceans and Teleosts were dominant, and Nematodes were rare. The species’ diet was basically carcino/ichthyo-benthophagous. The value for niche width was 0.03, i.e. very narrow, with a few important items in the diet.
The SIMPER analysis showed a similarity of 52.4% between males and females, Teleost fragments contributed 57.6% of this similarity. Crustaceans showed the highest values of %IAi for both sexes (Tab. 4), but were more important for males. In males, Teleost fragments were an important item, followed by Penaeoidea (Dendrobranchiata) and Leptochela sp. For females, Teleost fragments were most important, followed by Portunidae and P. spinicarpus.
In the ontogenetic analysis, the similarity in SIMPER was 46% and Teleost fragments contributed 55%. For juveniles (Tab. 4), unidentified shrimp were quite important. For the adults, Brachyura, Portunidae and P. spinicarpus had higher importance. Teleosts were more important for adults than for juveniles, and Nematodes were more important for juveniles.
Rhizoprionodon lalandi. Of the 43 stomachs analyzed, 5 were omitted because of lack of contents. These sharks measured between 49.4 and 74.0 cm TL. Of the 37 remaining stomachs, 16 were from females (52.0-74.0 cm TL) and 21 from males (49.4-67.2 cm TL); with 14 from juveniles (49.4-57.7 cm TL) and 23 from adults (54.0-74.0 cm TL). The smallest adult male measured 54.0 cm TL, and the smallest adult female 57.0 cm.
The cumulative prey curve (Fig. 2d) rose continuously, indicating the need to sample more stomachs for a more complete study of feeding in the region. The food items were Teleosts, Elasmobranchs and Cephalopods (Tab. 5). In the total sample, Teleosts were the most important, with 99.9% IAi; Teleost fragments showed the highest FO and IAi; and Dactylopterus volitans was very important. Elasmobranchs and Cephalopods were represented by an elasmobranch egg and the beak of a squid (Doryteuthis plei).
Frequency of occurrence (%FO), percentage weight (%W) and alimentary index (%IAi) of prey items in the total diet of Rhizoprionodon lalandi i in the southeastern Brazil, between January 2006 and August 2007, total, according to sex and stage of maturity.
The trophic niche width was 0.01, i.e. very narrow, with high importance of a few food items, in agreement with the method of Costello (1990Costello MJ. Predator feeding strategy and prey importance: a new graphical analysis. J Fish Biol . 1990; 36(2):261-63.) (Figs. 3g-h). A single item, Teleost fragments, dominated and the other items were rare, indicating an ichthyophagous species. The food of males and females (Tab. 5) showed about 90.4% similarity in the SIMPER analysis. For both males and females, Teleosts were dominant, with a high %IAi, and fish fragments showed the highest importance. Elasmobranchs and Cephalopods were found only in females and with low importance. The food of juveniles and adults (Tab. 5) showed about 91.70% similarity in the SIMPER analysis. In this case, Teleosts dominated in the prey of both juveniles and adults, and Teleost fragments were the most important item.
Interspecies feeding overlap. The analysis of correspondence (Fig. 4) separated the species by their food items, in agreement with the analyses of feeding overlap. All individuals of R. lalandii were grouped with a diet mainly of Teleost fish and squid. The diet of R. agassizi was dominated by Teleosts and Crustaceans, with differences between males/females and juveniles/adults. Females and adults fed mainly on Teleost fish (23-63%), whereas Shrimp (70-77%) were more important for males and juveniles. The ray A. cyclophora was grouped separately, with a diet mainly of Brachyura (68-97%). Adult males and females of P. extenta had a diet dominated by Shrimp (69-83%) with a small participation of Nematodes (0.1-0.7%); whereas juveniles showed a more diversified diet, with Brachyurans (38.7%), Polychaetes (29%), Crustacean fragments (11.3%), Shrimp (10.8%), Amphipods (8.6%) and Isopods (1.6%). The diet of P. rutrum consisted of small Crustaceans.
Correspondence analysis. Projection on factorial planes 1-2 by food items, of species (Atl = Atlantoraja cyclophora, Rio = Rioraja agassizii, Psex = Psammobatis extenta, Psru = Psammobatis rutrum and Rhi = Rhizoprionodon lalandii) according to sex and stages of maturity (G= general, M = males, F = females, J = juveniles and A = adults), in the southeastern Brazil.
Discussion
Elasmobranchs are currently one of the most threatened vertebrate groups, not only by their inherent biological vulnerability which makes them extremely sensitive to fishing pressure, but also by habitat loss and environmental degradation, resulting in a discouraging prognosis (Bornatowski et al., 2014bBornatowski H, Braga RR, Vitule JRS. Threats to sharks in a developing country: The need for effective and simple conservation measures. Nat Conservacao. 2014b; 12(1):11-18.; Dulvy et al., 2014Dulvy KN, Fowler SL, Musick JA, Cavanagh RD, Kyne PM, Harrison LR et al. Extinction risk and conservation of the world’s sharks and rays. eLife [serial on internet]. 2014; 3:e00590. DOI:10.7554/eLife.00590.
https://doi.org/10.7554/eLife.00590...
; Davidson et al., 2015Davidson LNK, Krawchuk MA, Dulvy NK. Why have global shark and ray landings declined: improved management or overfishing?. Fish Fisheries. 2015; 17(2):438-58.). Alongside this critical situation, basic information about the biology and ecology of elasmobranch species is still limited or lacking. With this background, we seek to contribute with this study on the feeding ecology of four species of rays and one shark.
In the diet of Psammobatis rutrum the importance of teleost fragments may be overestimated, as they were treated as a group. In addition, evaluation of the relative importance of items showed that teleost fragments were not dominant, with a lower value than that of caridean shrimp. Consequently we believe that shrimp are actually the main item in the diet of this species. This supposition is supported by the low value obtained in the calculation of the trophic niche, suggesting a trend toward a specialist diet, despite the absence of a dominant item. However, the small number of specimens of P. rutrum studied may have compromised this analysis. The absence of scientific literature on the feeding of this species makes it difficult to discuss the results. However, studies with congeneric species have found similar diets. The feeding of Psammobatis normani and P. rudis were analyzed by Mabragaña, Gilberto (2007Mabragaña E, Giberto DA. Feeding ecology and abundance of two sympatric skates, the shortfin sand skate Psammobatis normani McEachran, and the smallthorn sand skate P. rudis Günther (Chondrichthyes, Rajidae), in the southwest Atlantic. ICES J Mar Sci . 2007; 64(5):1017-27.) in Argentina, and indicated a carcinophagous diet for both species; for P. normani, polychaetes also were an important group, and for both species, teleosts were only occasional. The study also indicated that both trended toward specializing in crustaceans.
The diet of Psammobatis extenta, based on benthic crustaceans, mainly Leptochela sp., is similar to the diet of the ray Pseudobatos percellens on the central coast of Paraná, southern Brazil (Carmo et al., 2015Carmo WPD, Bornatowski H, Oliveira EC, Fávaro LL. Diet of the chola guitarfish, Rhinobatos percellens (Rhinobatidae), in the Paranaguá estuarine complex. An Acad Bras Cienc. 2015; 87(2):721-31.). Polychaetes were also important in the diet, with predation focused not only on carnivorous polychaetes of the family Sigalionidae, but also on subsurface detritivores of the family Maldanidae. This suggests that P. extenta feeds by digging in the sediment. A carcinophagous diet was also observed in other studies, including those of Soares et al. (1992Soares LSH, Wongtschowski CLBR, Alvares LMC, Muto EY, Gasalla MA. Grupos tróficos de peixes demersais da plataforma continental interna de Ubatuba, Brasil. I. Chondrichthyes. Bol Inst Oceanogr . 1992; 40(1/2):79-85.) and Muto et al. (2001Muto EY, Soares LSH, Goitein R. Food resource utilization of the skates Rioraja agassizi (Müller & Henle, 1841) and Psammobatis extenta (Garman, 1913) on the continental shelf off Ubatuba, south-eastern Brazil. Rev Bras Biol. 2001; 61(2):217-38.) on the Brazilian southeast coast, and Braccini, Perez (2005Braccini JM, Perez JE. Feeding habits of the sandskate Psammobatis extenta (Garman, 1913): Sources of variation in dietary composition. Mar Freshw Res. 2005; 56:395-403.) in Argentina, where crustaceans also comprised the main part of the diet.
For R. agassizi the diet was also based on benthic prey. However, as in P. rutrum the importance of teleosts may have been overestimated. Although crustaceans and teleosts formed the basis of the diet, the analysis indicated a fairly narrow niche and a diet trending toward specialization. A carcinophagous diet, with a secondary importance of teleosts, was found in other studies of R. agassizi. In the studies of Soares et al. (1992Soares LSH, Wongtschowski CLBR, Alvares LMC, Muto EY, Gasalla MA. Grupos tróficos de peixes demersais da plataforma continental interna de Ubatuba, Brasil. I. Chondrichthyes. Bol Inst Oceanogr . 1992; 40(1/2):79-85.; 1999Soares LSH, Vazzoler AEAM, Correa AR. Diel feeding chronology of the skate Raja agassizii (Müller & Henle) (Pisces, Elasmobranchii) on the continental shelf off Ubatuba, southeastern Brazil. Rev Bras Zool. 1999; 16(1):201-12.) and Muto et al. (2001Muto EY, Soares LSH, Goitein R. Food resource utilization of the skates Rioraja agassizi (Müller & Henle, 1841) and Psammobatis extenta (Garman, 1913) on the continental shelf off Ubatuba, south-eastern Brazil. Rev Bras Biol. 2001; 61(2):217-38.) in São Paulo, crustaceans were the main food item and teleosts were secondary, in addition to polychaetes. The same behavior was observed for the species in Uruguay and Argentina by Barbini, Lucifora (2012Barbini SA, Lucifora LO. Feeding habits of a large endangered skate from the south-west Atlantic: the spotback skate, Atlantoraja castelnaui. Mar. Freshw. Res. 2012; 63(2):180-88.). Bornatowski et al. (2014dBornatowski H, Wosnick N, Carmo WPD, Corrêa MFM, Abilhoa V. Feeding comparisons of four batoids (Elasmobranchii) in coastal waters of southern Brazil. J Mar Biol Assoc U.K. 2014d; 94(7):1491-99.) found that teleosts were the main item, followed by gammarideans, caridean shrimp and dendobranchiate shrimp.
The shark R. lalandii was basically ichthyophagous, feeding on demersal teleosts, but also on cephalopods. The low value of the trophic-niche width and the evaluation of the feeding strategy indicated specialized feeding, with only one item dominating. The difficulty of identifying the fish species contributed to the indication of a specialist diet, as these prey were mainly grouped in the item “Teleost fragments”. Published reports, such as those by Lima et al. (2000Lima GHL, Daros FA, Mazzoleni R, Silva MH. Aspectos da alimentação natural do cação-frango Rhizoprionodon lalandii (Valenciennes, 1841) (Elasmobranchii: Carcharhinidae) no município de Barra Velha, Santa Catarina. Notas Técnicas da Facimar. 2000; 4:91-96.) in Santa Catarina, and Bornatowski (2012Bornatowski H, Heithaus MR, Abilhoa V, Corrêa MFM. Feeding of the Brazilian sharpnose shark Rhizoprionodon lalandii (Müller & Henle, 1839) from southern Brazil. J Appl Ichthyol. 2012; 28(4):623-27.) and Bornatowski et al. (2014aBornatowski H, Braga RR, Abilhoa V, Corrêa MFM. Feeding ecology and trophic comparisons of six shark species in a coastal ecosystem off southern Brazil. J Fish Biol . 2014a; 85(2):246-63.) in Paraná also describe a fish-based diet for this species. However, differing from the present study, Bornatowski et al. (2014aBornatowski H, Braga RR, Abilhoa V, Corrêa MFM. Feeding ecology and trophic comparisons of six shark species in a coastal ecosystem off southern Brazil. J Fish Biol . 2014a; 85(2):246-63.) found ontogenetic differences: crustaceans, sciaenids and clupeids were more important for juveniles and sciaenids for adults.
Comparison of the diets of the rays of the genus Psammobatis, which are smaller-bodied species, showed that they were based on small crustaceans such as unidentified shrimp, polychaetes and nematodes. Rioraja agassizii and A. cyclophora, larger-bodied species, had a more varied diet, with a higher importance of larger crustaceans such as portunid crabs, in addition to teleosts. The shark R. lalandii, which feeds at a higher trophic level, showed a diet dominated by teleosts. This behavior can be explained by the greater capacity of larger individuals to capture larger prey, while smaller-sized predators feed on smaller, more abundant prey (Keast, 1977Keast A. Diet overlaps and feeding relationships between the year classes in the yellow perch (Perca flavescens). Environ Biol Fish. 1977; 2(1):53-70.; Vianna et al., 2000Vianna M, Arfelli CA, Amorim AF. Feeding of Mustelus canis (Elasmobranchii, Triakidae) caught off south-southeast coast of Brazil. Bol Inst Pesca. 2000; 26(1):79-84.; Viana, Vianna, 2014Viana AF, Vianna M. The feeding habits of the eyespot skate Atlantoraja cyclophora (Elasmobranchii: Rajiformes) in southeastern Brazil. Zoologia (Curitiba). 2014; 31(2):119-25.). This analysis may also explain the differences between sexes and ontogenetic stages for P. extenta and R. agassizii. In both cases males are smaller than females, and adult females capture more teleosts and crustaceans such as crabs. On the other hand, juveniles and males capture more small crustaceans such as shrimp, polychaetes, and fewer teleosts. Estalles et al. (2015Estalles ML, Perier MR, Di Giácomo EE. Trophic ecology of the smallnose fanskate Sympterygia bonapartii in the San Matías Gulf, northern Patagonia, Argentina. Ichthyol Res. 2015; 63(2):207-17.), however, believed that this relationship with size is not direct, and that the increase in caloric expenditure during reproduction obligates adult elasmobranchs to increase their consumption of more energy-rich prey such as fish and cephalopods.
The present results indicated that the five species showed differences in their feeding ecology, and formed two trophic groups: one composed of the smaller-sized rays P. extenta and P. rutrum; and the other of the larger rays R. agassizii and A. cyclophora and the shark R. lalandii. Soares et al. (1992Soares LSH, Wongtschowski CLBR, Alvares LMC, Muto EY, Gasalla MA. Grupos tróficos de peixes demersais da plataforma continental interna de Ubatuba, Brasil. I. Chondrichthyes. Bol Inst Oceanogr . 1992; 40(1/2):79-85.) found similar trophic guilds of elasmobranchs on the northern coast of São Paulo. Rioraja agassizi and A. cyclophora, which fed on crustaceans (principally brachyurans and shrimp) and fish, were grouped together; whereas P. extenta belonged to another trophic guild, consumers of benthic invertebrates (principally smaller crustaceans such as gammarideans, and polychaetes). The formation of the two guilds indicated that although these species occupy the same region, they show differences in their diets, enabling them to coexist with less competition for resources in the same locale. Similar small differences in diets between sympatric elasmobranchs with similar feeding habits were observed for demersal species in the Mediterranean (Valls et al., 2011Valls M, Quetglas A, Ordines F, Moranta J. Feeding ecology of demersal elasmobranchs from the shelf and slope off the Balearic Sea (western Mediterranean). Sci Mar. 2011; 75(4):633-39.). However, the formation of guilds does not indicate that these groups differ completely in their diets, but rather in the relative contribution of items to each group. Members of the two guilds have many food items in common, but the importance of these items differs.
The study of feeding ecology of elasmobranchs is essential because in general they occupy intermediate or high trophic levels (Vianna et al., 2000Vianna M, Arfelli CA, Amorim AF. Feeding of Mustelus canis (Elasmobranchii, Triakidae) caught off south-southeast coast of Brazil. Bol Inst Pesca. 2000; 26(1):79-84.; Ebert, Bizzarro, 2007Ebert DA, Bizzarro JJ. Standardized diet compositions and trophic levels of skates (Chondrichthyes: Rajiformes: Rajoidei). Environ Biol Fishes. 2007; 80(2); 80:221-37.); because of the intensifying fishery exploitation that is reducing populations of these organisms (Vooren, Klippel, 2005Vooren CM , Klippel S. Diretrizes para a conservação de espécies ameaçadas de elasmobrânquios. In: Vooren CM, Klippel S. Ações para a conservação de tubarões e raias do Brasil. Porto Alegre: Igaré; 2005. p.213-28.; Andrade et al., 2008Andrade AC, Silva Junior LC, Vianna M. Reproductive biology and population variables of the Brazilian sharpnose shark Rhizoprionodon lalandii (Müller & Henle, 1839) captured in coastal waters of south-eastern Brazil. J Fish Biol . 2008; 72(3):473-84.), the scarcity of biological data that hinders knowledge of the species (Vianna et al., 2000Vianna M, Arfelli CA, Amorim AF. Feeding of Mustelus canis (Elasmobranchii, Triakidae) caught off south-southeast coast of Brazil. Bol Inst Pesca. 2000; 26(1):79-84.; Chiaramonte et al., 2004Chiaramonte GE. Psammobatis extenta. The IUCN Red List of Threatened Species. 2004 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
http://www.iucnredlist.org/...
; Kyne, 2007Kyne PM, San Martín J, Stehmann MFW. Rioraja agassizii. The IUCN Red List of Threatened Species . 2007 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
http://www.iucnredlist.org/...
; Massa et al., 2006Massa A, Hozbor N, Vooren CM. Atlantoraja cyclophora. The IUCN Red List of Threatened Species . 2006 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
http://www.iucnredlist.org/...
; Rosa et al., 2004Rosa RS, Gadig OBF, Santos Motta F, Namora RC. Rhizoprionodon lalandii. The IUCN Red List of Threatened Species . 2004 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
http://www.iucnredlist.org/...
; Andrade et al., 2008Andrade AC, Silva Junior LC, Vianna M. Reproductive biology and population variables of the Brazilian sharpnose shark Rhizoprionodon lalandii (Müller & Henle, 1839) captured in coastal waters of south-eastern Brazil. J Fish Biol . 2008; 72(3):473-84.; Silva Junior et al., 2011Silva Junior LC, Andrade AC, Vianna M. Length-weight relationships for elasmobranchs from southeastern Brazil. J Appl Ichthyol . 2011; 27(6):1408-10.; Viana, Vianna, 2014Viana AF, Vianna M. The feeding habits of the eyespot skate Atlantoraja cyclophora (Elasmobranchii: Rajiformes) in southeastern Brazil. Zoologia (Curitiba). 2014; 31(2):119-25.; Bornatowski et al., 2014cBornatowski H, Navia AF, Braga RR, Abilhoa V, Corrêa MFM. Ecological importance of sharks and rays in a structural foodweb analysis in southern Brazil. ICES J Mar Sci. 2014c; 71(7):1586-92.) and the possibility that elasmobranchs may become contaminated by persistent organic pollutants, with consequences for the entire food web (Rosenfelder et al., 2012Rosenfelder N, Lehnert K, Kaffarnik S, Torres JPM, Vianna M, Vetter W. Thorough analysis of polyhalogenated compounds in ray liver samples off the coast of Rio de Janeiro, Brazil. Environ Sci Pollut Res. 2012; 19(2):379-89.).
Studies reveal that some sharks and rays may act as key elements in a food-web structure. Reductions in their populations not only lead to changes in their main prey (direct effect) but also to changes in other species through indirect effects on the food web (Bornatowski et al., 2014bBornatowski H, Braga RR, Vitule JRS. Threats to sharks in a developing country: The need for effective and simple conservation measures. Nat Conservacao. 2014b; 12(1):11-18.).
Further studies will be useful in monitoring the responses of the species to the conditions to which they are being subjected, as well as in improving understanding of the relationships among the species in the region, the food web structures and the ecosystem.
Acknowledgments
The authors are grateful to the Laboratório de Biologia e Tecnologia Pesqueira group for the help in sampling, measurements and dissection of the skates, to Paulo C. Paiva for helping with polychaete identification, and to Tereza C. G. da Silva and Karina A. Keunecke for helping with crustacean identification. Financial support was received from FAPERJ (Fundo de Amparo à Pesquisa do Estado do Rio de Janeiro), CAPES and CNPQ (Programa PELD). Janet W. Reid (JWR Associates) revised the English text.
References
- Amundsen PA, Gabler HM, Staldvik FJ. A new approach to graphical analysis of feeding strategy from stomach contents data - modification of the Costello (1990) method. J Fish Biol. 1996; 48(4):607-14.
- Andrade AC, Silva Junior LC, Vianna M. Reproductive biology and population variables of the Brazilian sharpnose shark Rhizoprionodon lalandii (Müller & Henle, 1839) captured in coastal waters of south-eastern Brazil. J Fish Biol . 2008; 72(3):473-84.
- Barbini SA, Lucifora LO. Feeding habits of a large endangered skate from the south-west Atlantic: the spotback skate, Atlantoraja castelnaui Mar. Freshw. Res. 2012; 63(2):180-88.
- Barreto R, Ferretti F, Flemming JM, Amorim A, Andrade H, Worm B, Lessa R. Trends in the exploitation of South Atlantic shark populations. Conserv Biol. 2016; 30(4):792-804.
- Bornatowski H, Braga RR, Abilhoa V, Corrêa MFM. Feeding ecology and trophic comparisons of six shark species in a coastal ecosystem off southern Brazil. J Fish Biol . 2014a; 85(2):246-63.
- Bornatowski H, Braga RR, Vitule JRS. Threats to sharks in a developing country: The need for effective and simple conservation measures. Nat Conservacao. 2014b; 12(1):11-18.
- Bornatowski H, Heithaus MR, Abilhoa V, Corrêa MFM. Feeding of the Brazilian sharpnose shark Rhizoprionodon lalandii (Müller & Henle, 1839) from southern Brazil. J Appl Ichthyol. 2012; 28(4):623-27.
- Bornatowski H, Navia AF, Braga RR, Abilhoa V, Corrêa MFM. Ecological importance of sharks and rays in a structural foodweb analysis in southern Brazil. ICES J Mar Sci. 2014c; 71(7):1586-92.
- Bornatowski H, Wosnick N, Carmo WPD, Corrêa MFM, Abilhoa V. Feeding comparisons of four batoids (Elasmobranchii) in coastal waters of southern Brazil. J Mar Biol Assoc U.K. 2014d; 94(7):1491-99.
- Braccini JM, Perez JE. Feeding habits of the sandskate Psammobatis extenta (Garman, 1913): Sources of variation in dietary composition. Mar Freshw Res. 2005; 56:395-403.
- Braga RR, Bornatowski H, Vitule JRS. Feeding ecology of fishes: an overview of worldwide publications. Rev Fish Biol Fish. 2012; 22(4):915-29.
- Carmo WPD, Bornatowski H, Oliveira EC, Fávaro LL. Diet of the chola guitarfish, Rhinobatos percellens (Rhinobatidae), in the Paranaguá estuarine complex. An Acad Bras Cienc. 2015; 87(2):721-31.
- Chiaramonte GE. Psammobatis extenta The IUCN Red List of Threatened Species. 2004 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
» http://www.iucnredlist.org/ - Cortés E. A critical review of methods of studying fish feeding based on analysis of stomach contents: Application to elasmobranch fishes. Can J Fish Aquat Sci. 1997; 54(3):726-38.
- Costa L, Chaves PTC. Elasmobrânquios capturados pela pesca artesanal na costa sul do Paraná e norte de Santa Catarina, Brasil. Biota Neotrop [serial on internet]. 2006; 6(3). doi: 10.1590/S1676-06032006000300007.
» https://doi.org/10.1590/S1676-06032006000300007 - Costello MJ. Predator feeding strategy and prey importance: a new graphical analysis. J Fish Biol . 1990; 36(2):261-63.
- Davidson LNK, Dulvy NK. Global marine protected areas to prevent extinctions. Nat Ecol Evol. 2017; 40. DOI:10.1038/s41559-016-0040.
» https://doi.org/10.1038/s41559-016-0040 - Davidson LNK, Krawchuk MA, Dulvy NK. Why have global shark and ray landings declined: improved management or overfishing?. Fish Fisheries. 2015; 17(2):438-58.
- Dulvy KN, Fowler SL, Musick JA, Cavanagh RD, Kyne PM, Harrison LR et al Extinction risk and conservation of the world’s sharks and rays. eLife [serial on internet]. 2014; 3:e00590. DOI:10.7554/eLife.00590.
» https://doi.org/10.7554/eLife.00590 - Ebert DA, Bizzarro JJ. Standardized diet compositions and trophic levels of skates (Chondrichthyes: Rajiformes: Rajoidei). Environ Biol Fishes. 2007; 80(2); 80:221-37.
- Estalles ML, Perier MR, Di Giácomo EE. Trophic ecology of the smallnose fanskate Sympterygia bonapartii in the San Matías Gulf, northern Patagonia, Argentina. Ichthyol Res. 2015; 63(2):207-17.
- Ferry LA, Cailliet GM. Sample size and data analysis: are we characterizing and comparing diet properly? In: MacKinlay D, Shearer K, eds. Feeding Ecology and Nutrition in Fish. San Francisco: American Fisheries Society; 1996. p.71-80.
- Gomes UL, Signori CN, Gadig OBF, Santos HRS. Guia para identificação de tubarões e raias do Rio de Janeiro. Rio de Janeiro: Technical Books; 2010.
- Hurlbert SH. The measurement of niche overlap and some relatives. Ecology. 1978; 59(1):67-77.
- Hynes HBN. The food of fresh-water sticklebacks (Gasterosteus aculeatus and Pygosteus pungitius), with a review of methods used in studies of the food of fishes. J Anim Ecol. 1950; 19(1):36-58. doi: 10.2307/1570.
» https://doi.org/10.2307/1570 - Jones A. The ecology of young turbot, Scophthalmus maximus (L.), at Borth, Cardiganshire, WaIes. J Fish Biol .1973; 5(3):367-83.
- Kawakami E, Vazzoler G. Método gráfico e estimativa de índice alimentar aplicado no estudo de alimentação de peixes. Bol Inst Oceanogr. 1980; 29(2):205-07.
- Keast A. Diet overlaps and feeding relationships between the year classes in the yellow perch (Perca flavescens). Environ Biol Fish. 1977; 2(1):53-70.
- Kyne, PM. Psammobatis rutrum The IUCN Red List of Threatened Species. 2007 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
» http://www.iucnredlist.org/ - Kyne PM, San Martín J, Stehmann MFW. Rioraja agassizii The IUCN Red List of Threatened Species . 2007 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
» http://www.iucnredlist.org/ - Lima GHL, Daros FA, Mazzoleni R, Silva MH. Aspectos da alimentação natural do cação-frango Rhizoprionodon lalandii (Valenciennes, 1841) (Elasmobranchii: Carcharhinidae) no município de Barra Velha, Santa Catarina. Notas Técnicas da Facimar. 2000; 4:91-96.
- Lucifora LO, García VB, Worm B. Global diversity hotspots and conservation priorities for sharks. PLoS ONE. 2011; 6(5):e19356. doi:10.1371/journal.pone.0019356.
» https://doi.org/10.1371/journal.pone.0019356 - Mabragaña E, Giberto DA. Feeding ecology and abundance of two sympatric skates, the shortfin sand skate Psammobatis normani McEachran, and the smallthorn sand skate P. rudis Günther (Chondrichthyes, Rajidae), in the southwest Atlantic. ICES J Mar Sci . 2007; 64(5):1017-27.
- Massa A, Hozbor N, Vooren CM. Atlantoraja cyclophora The IUCN Red List of Threatened Species . 2006 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
» http://www.iucnredlist.org/ - McClenachan L, Cooper AB, Dulvy NK. Rethinking trade-driven extinction risk in marine and terrestrial megafauna. Curr Biol. 2016; 26(12):1640-46.
- Muto EY, Soares LSH, Goitein R. Food resource utilization of the skates Rioraja agassizi (Müller & Henle, 1841) and Psammobatis extenta (Garman, 1913) on the continental shelf off Ubatuba, south-eastern Brazil. Rev Bras Biol. 2001; 61(2):217-38.
- Rosa RS, Gadig OBF, Santos Motta F, Namora RC. Rhizoprionodon lalandii The IUCN Red List of Threatened Species . 2004 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
» http://www.iucnredlist.org/ - Rosenfelder N, Lehnert K, Kaffarnik S, Torres JPM, Vianna M, Vetter W. Thorough analysis of polyhalogenated compounds in ray liver samples off the coast of Rio de Janeiro, Brazil. Environ Sci Pollut Res. 2012; 19(2):379-89.
- Silva Junior LC, Andrade AC, Vianna M. Length-weight relationships for elasmobranchs from southeastern Brazil. J Appl Ichthyol . 2011; 27(6):1408-10.
- Soares LSH, Wongtschowski CLBR, Alvares LMC, Muto EY, Gasalla MA. Grupos tróficos de peixes demersais da plataforma continental interna de Ubatuba, Brasil. I. Chondrichthyes. Bol Inst Oceanogr . 1992; 40(1/2):79-85.
- Soares LSH, Vazzoler AEAM, Correa AR. Diel feeding chronology of the skate Raja agassizii (Müller & Henle) (Pisces, Elasmobranchii) on the continental shelf off Ubatuba, southeastern Brazil. Rev Bras Zool. 1999; 16(1):201-12.
- Valls M, Quetglas A, Ordines F, Moranta J. Feeding ecology of demersal elasmobranchs from the shelf and slope off the Balearic Sea (western Mediterranean). Sci Mar. 2011; 75(4):633-39.
- Viana AF, Vianna M. The feeding habits of the eyespot skate Atlantoraja cyclophora (Elasmobranchii: Rajiformes) in southeastern Brazil. Zoologia (Curitiba). 2014; 31(2):119-25.
- Vianna M, Arfelli CA, Amorim AF. Feeding of Mustelus canis (Elasmobranchii, Triakidae) caught off south-southeast coast of Brazil. Bol Inst Pesca. 2000; 26(1):79-84.
- Vooren CM , Klippel S. Diretrizes para a conservação de espécies ameaçadas de elasmobrânquios. In: Vooren CM, Klippel S. Ações para a conservação de tubarões e raias do Brasil. Porto Alegre: Igaré; 2005. p.213-28.
- Zar JH. Biostatistical Analysis. 4th ed. New Jersey: Prentice-Hall; 1999.
Data availability
Data citations
Chiaramonte GE. Psammobatis extenta The IUCN Red List of Threatened Species. 2004 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
Kyne, PM. Psammobatis rutrum The IUCN Red List of Threatened Species. 2007 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
Kyne PM, San Martín J, Stehmann MFW. Rioraja agassizii The IUCN Red List of Threatened Species . 2007 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
Massa A, Hozbor N, Vooren CM. Atlantoraja cyclophora The IUCN Red List of Threatened Species . 2006 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
Rosa RS, Gadig OBF, Santos Motta F, Namora RC. Rhizoprionodon lalandii The IUCN Red List of Threatened Species . 2004 (updated 2016; cited 2017 Jan 7). Available from: Available from: http://www.iucnredlist.org/
Publication Dates
-
Publication in this collection
2017
History
-
Received
25 May 2016 -
Accepted
21 May 2017