ABSTRACT
A new species of Ituglanis associated to the grasslands of the Pampa biome is described from the rio Uruguai basin, southern Brazil. The new species is distinguished from its congeners by the low number of ribs and by a unique color pattern composed of an outer layer with scattered round black blotches equivalent in size to the eye circumference over a reddish brown background on the lateral surface of the body. We provide the genetic sequences of the mitochondrial gene Cytochrome c Oxydase subunit I (COI) for three of the paratypes and discuss aspects about the recent discovery of the new species.
Keywords:
Genetype; Pampa Biome; Rio Ibicuí; South Brazilian Grasslands; Taxonomy
RESUMO
Uma nova espécie de Ituglanis, associada aos campos do bioma Pampa, é descrita para a bacia do rio Uruguai no sul do Brasil. A nova espécie distingue-se de todos seus congêneres pelo pequeno número de costelas e por um padrão de coloração único que consiste de manchas pretas arredondadas de tamanho equivalente à circunferência do olho sobre um fundo marrom avermelhado na superfície lateral do corpo. Sequências genéticas do gene mitocondrial Citocromo Oxidase subunidade I (COI) para três dos parátipos são fornecidas, e aspectos sobre a recente descoberta da nova espécie são discutidos.
Palavras-chave:
Bioma Pampa; Campos Sulinos; Gene-tipo; Rio Ibicuí; Taxonomia
Introduction
Trichomycteridae is the second most species-rich family of Siluriformes comprising about 300 valid species (Eschmeyer, Fong, 2017Eschmeyer WN, Fricke R, van der Laan R, editors. Catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science ; 2016 [updated 2017 Mar 01; cited 2017 Mar 29]. Available from: Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
http://researcharchive.calacademy.org/re...
). The trichomycterids are widespread in the Neotropical region from Costa Rica to Argentina, on both sides of the Andes. The genus Ituglanis Costa & Bockmann was proposed as a monophyletic group comprising nine species previously included in the putatively paraphyletic genus Trichomycterus Valenciennes (Costa, Bockmann, 1993Costa WJEM, Bockmann FA. Un nouveau genre néotropical de la famille des Trichomycteridae (Siluriformes: Loricarioidei). RFAHRevue Française d’Aquariologie et Herpétologie . 1993; 20(2):43-46.). Since then, a substantial number of species have been described and the genus is currently composed of 27 species (Datovo et al., 2016Datovo A, Aquino PPU, Langeani F. A new species of Ituglanis (Siluriformes: Trichomycteridae) from the Tocantins and Paranaíba river basins, central Brazil, with remarks on the systematics of the genus. Zootaxa . 2016; 4171(3):439-58.; Eschmeyer et al., 2017Eschmeyer WN, Fong JD. Species by Family/Subfamily [Internet]. San Francisco: California Academy of Science; 2016 [updated 2017 Mar 01; cited 2017 Mar 29]. Available from: Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/SpeciesByFamily.asp
http://researcharchive.calacademy.org/re...
).
Among the freshwater ecoregions of the southern Brazil sensuAbell et al. (2008Abell R, Thieme ML, Revenga C, Bryer M, Kottelat M, Bogutskaya N, Coad B, Mandrak N, Contreras-Balderas SL, Bussing W, Stiassny MLJ, Skelton P, Allen GR, Unmack P, Naseka A, Ng R, Sindorf N, Robertson J, Armijo E, Higgins V, Heibel TJ, Wikramanayake E, Olson D, López HL, Reis RE, Lundberg JG, Sabaj-Pérez MHS, Petry R. Freshwater ecoregions of the world: a new map of biogeographic units for freshwater biodiversity conservation. Bioscience. 2008; 58(5):403-14.) - which includes the upper and lower rio Uruguai basin, Tramandaí-Mampituba, and Laguna dos Patos - the genus Ituglanis was first recorded by Becker et al. (2013Becker FG, De Fries LCC, Ferrer J, Bertaco VA, Luz-Agostinho KDG, Silva JFP, Cardoso AR, Lucena ZMS, Lucena CAS. Fishes of the Taquari-Antas river basin (Patos Lagoon basin), southern Brazil. Braz J Biol. 2013; 73(1):79-90.) only in the latter drainage. Since then, two species were described from the region: I. australis Datovo & de Pinna from the lower rio Uruguai and Laguna dos Patos, and I. boitata Ferrer, Donin & Malabarba from Tramandaí-Mampituba (Datovo, de Pinna, 2014Datovo A, de Pinna MCC. A new species of Ituglanis representing the southernmost record of the genus, with comments on phylogenetic relationships (Teleostei: Siluriformes: Trichomycteridae). J Fish Biol. 2014; 84(2):314-27.; Ferrer et al., 2015Ferrer J, Donin LM, Malabarba LR. A new species of Ituglanis Costa & Bockmann, 1993 (Siluriformes: Trichomycteridae) endemic to the Tramandaí-Mampituba ecoregion, southern Brazil. Zootaxa , 2015; 4020(2):375-89.). An updated list of freshwater fishes from Rio Grande do Sul including those ecoregions of Abell et al. (2008Abell R, Thieme ML, Revenga C, Bryer M, Kottelat M, Bogutskaya N, Coad B, Mandrak N, Contreras-Balderas SL, Bussing W, Stiassny MLJ, Skelton P, Allen GR, Unmack P, Naseka A, Ng R, Sindorf N, Robertson J, Armijo E, Higgins V, Heibel TJ, Wikramanayake E, Olson D, López HL, Reis RE, Lundberg JG, Sabaj-Pérez MHS, Petry R. Freshwater ecoregions of the world: a new map of biogeographic units for freshwater biodiversity conservation. Bioscience. 2008; 58(5):403-14.) recognized three additional species of Ituglanis awaiting taxonomic description (Bertaco et al., 2016Bertaco VA, Ferrer J, Carvalho FR, Malabarba LR. Inventory of the freshwater fishes from a densely collected area in South America - a case study of the current knowledge of Neotropical fish diversity. Zootaxa. 2016; 4138(3):401-40.). One of them is known from only a few specimens discovered after a large collection effort in streams across the South Brazilian grasslands (see Dala-Corte et al., 2016Dala-Corte RB, Giam X, Olden JD, Becker FG, Guimarães TDF, Melo AS. Revealing the pathways by which agricultural land-use affects stream fish communities in South Brazilian grasslands. Freshwater Biol. 2016; 61(11):1921-34.) and is described herein.
Material and Methods
Measurements were taken point-to-point using a digital caliper to the nearest 0.1 mm and following Tchernavin (1944Tchernavin VV. A revision of some Trichomycterinae based on material preserved in the British Museum (Natural History). Proc Zool Soc London. 1944; 114(1-2):234-75.) for barbel length, Wosiacki, de Pinna (2008Wosiacki WB, de Pinna MCC. Trichomycterus igobi, a new catfish species from the rio Iguaçu drainage: the largest head in Trichomycteridae (Siluriformes: Trichomycteridae). Neotrop. Ichthyol. 2008; 6(1):17-23.) for length and depth of the caudal peduncle and for supraorbital pore s6 distance, Ferrer, Malabarba (2011Ferrer J, Malabarba LR. A new Trichomycterus lacking pelvic fins and pelvic girdle with a very restricted range in southern Brazil (Siluriformes: Trichomycteridae). Zootaxa , 2011; 2912:59-67.) for scapular girdle width, Ferrer et al. (2015Ferrer J, Donin LM, Malabarba LR. A new species of Ituglanis Costa & Bockmann, 1993 (Siluriformes: Trichomycteridae) endemic to the Tramandaí-Mampituba ecoregion, southern Brazil. Zootaxa , 2015; 4020(2):375-89.) for interopercular length, and Costa (1992Costa WJEM. Description de huit nouvelles espèces du genre Trichomycterus (Siluriformes: Trichomycteridae), du Brésil oriental. RFAHRevue Française d’Aquariologie et Herpétologie. 1992; 18(4):101-10.) for the remaining measurements. Nomenclature of bones and cartilages follows Bockmann et al. (2004Bockmann FA, Casatti L, de Pinna MCC. A new species of trichomycterid catfish from the Rio Paranapanema basin, southeastern Brazil (Teleostei: Siluriformes), with comments on the phylogeny of the family. Ichthyol Explor Freshw. 2004; 15(3):225-42.). Nomenclature of laterosensory canal system and associated pores follows Rizzato, Bichuette (2016Rizzato PP, Bichuette ME. The laterosensory canal system in epigean and subterranean Ituglanis (Siluriformes: Trichomycteridae), with comments about troglomorphism and the phylogeny of the genus. J Morphol. 2016; 278(1):4-28.). Number of branchiostegal rays, odontodes, procurrent rays, basal radials, ribs, teeth, vertebrae and unsegmented rays were counted in one cleared and counter-stained specimen prepared according to the protocol of Taylor, Van Dyke (1985Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107-19.). Vertebral count excludes those in the Weberian complex and the compound caudal centrum is counted as a single element. All type specimens were used for the count of branched and unbranched fin rays and for the analysis of the pores of the laterosensory canal system. Number of specimens with each count is given in parentheses, an asterisk indicating the count for the holotype.
Information about coloration of other species of Ituglanis was obtained in original descriptions - Steindachner (1882Steindachner F. Beiträge zur Kenntniss der Flussfische Südamerika’s (IV). Anz. Akad. Wiss. Wien, Math.-Naturwiss. Kl. 1882; 19(19):175-80.); Eigenmann (1912Eigenmann CH. The freshwater fishes of British Guiana, including a study of the ecological grouping of species, and the relation of the fauna of the plateau to that of the lowlands. Pittsburgh: Carnegie Institute; 1912. (Memoirs of the Carnegie Museum: vol 5)., 1917Eigenmann CH. Descriptions of sixteen new species of Pygidiidae. Proc Am Philos Soc. 1917; 56(7):690-703.); Miranda Ribeiro, A. (1912Miranda Ribeiro A. Loricariidae, Callichthyidae, Doradidae e Trichomycteridae. Commissão de Linhas Telegraphicas Estrategicas de Matto-Grosso ao Amazonas. 1912; Annexo 5:1-31.); Miranda Ribeiro, P. (1940Miranda Ribeiro P. Alguns peixes do sul de Mato Grosso. O Campo. 1940; 60:1., 1944Miranda Ribeiro P. Um Pigidídeo do Alto Amazonas (Pisces - Pygidiidae). Bol Mus Nac Rio de J (Zool) 1944; 19:1-3.); Dahl (1960Dahl G. Nematognathous fishes collected during the Macarena Expedition 1959. Part I. Novedades Colombianas. 1960; 1(5):302-17.); de Pinna, Keith (2003de Pinna M, Keith P. A new species of the catfish genus Ituglanis from French Guyana (Osteichthyes: Siluriformes: Trichomycteridae). Proc Biol Soc Wash. 2003; 116(4):873-82.); Datovo, Landim (2005Datovo A, Landim MI. Ituglanis
macunaima, a new catfish from rio Araguaia basin, Brazil (Siluriformes, Trichomycteridae). Neotrop Ichthyol. 2005; 3(4):455-64.); Wosiacki et al. (2012Wosiacki WB, Dutra GM, Mendonça MB. Description of a new species of Ituglanis (Siluriformes: Trichomycteridae) from Serra dos Carajás, rio Tocantins basin. Neotrop Ichthyol . 2012; 10(3):547-54.); Datovo (2014Datovo A. A new species of Ituglanis from the Rio Xingu basin, Brazil, and the evolution of pelvic fin loss in trichomycterid catfishes (Teleostei: Siluriformes: Trichomycteridae). Zootaxa . 2014; 3790(3):466-76.); Castro, Wosiacki (2017Castro IDS, Wosiacki WB. Ituglanis
compactus, a new species of catfish (Siluriformes: Trichomycteridae) from the rio Jari drainage, lower Amazon, Brazil. Zootaxa . 2017; 4244(2):207-18.) - with additional data provided by Canto (2009Canto ALC. Caracterização morfológica dos representantes do gênero Ituglanis (Siluriformes: Trichomycteridae) da bacia amazônica brasileira. [MSc Dissertation on the Internet]. Manaus, AM: Instituto Nacional de Pesquisas na Amazônia; 2009 [cited 2016 Dec 28]. Available from: Biblioteca Digital de Teses e Dissertações Instituto Nacional de Pesquisas da Amazônia. Available from: Biblioteca Digital de Teses e Dissertações Instituto Nacional de Pesquisas da Amazônia. http://bdtd.inpa.gov.br/handle/tede/2023
http://bdtd.inpa.gov.br/handle/tede/2023...
) and de Pinna (2013de Pinna M. Trichomycteridae. In: Queiroz LJ, Torrente-Vilara G, Ohara WM, Pires THS, Zuanon J, Doria CRC, organizers. Peixes do Rio Madeira Vol. 2. São Paulo: Dialeto Latin American Documentary; 2013. p.142-79.). Information about the number of pair of ribs in other species of Ituglanis was obtained from Rizzato, Bichuette (2016Rizzato PP, Bichuette ME. The laterosensory canal system in epigean and subterranean Ituglanis (Siluriformes: Trichomycteridae), with comments about troglomorphism and the phylogeny of the genus. J Morphol. 2016; 278(1):4-28.), plus Castro, Wosiacki (2017Castro IDS, Wosiacki WB. Ituglanis
compactus, a new species of catfish (Siluriformes: Trichomycteridae) from the rio Jari drainage, lower Amazon, Brazil. Zootaxa . 2017; 4244(2):207-18.) for I. compactus. The numbering of undescribed species in the discussion and in the comparative material (Ituglanis sp. 1 and Ituglanis sp. 3) follows the checklist of Bertaco et al. (2016Bertaco VA, Ferrer J, Carvalho FR, Malabarba LR. Inventory of the freshwater fishes from a densely collected area in South America - a case study of the current knowledge of Neotropical fish diversity. Zootaxa. 2016; 4138(3):401-40.). Osteological illustrations were prepared by the second author with a camera lucida attached to a stereomicroscope. The distribution map was generated with the help of Quantum GIS version 1.8.0 software (Scherman et al., 2016Scherman GE, Sutton T, Blazek R, Luthman L. QGIS 1.8 [Geographic Information Software System]. 2016. Available from: http://docs.qgis.org/1.8/pdf/
http://docs.qgis.org/1.8/pdf/...
).
Following the suggestion of Chakrabarty (2010Chakrabarty P. Genetypes: a concept to help integrate molecular phylogenetics and taxonomy. Zootaxa . 2010; 2632:67-68.), three paragenetypes are designated by sequencing of the mitochondrial gene Cytochrome c Oxydase subunit I (COI). Total DNA genomic was extracted from ethanol-preserved tissue following a slightly modified CTAB methodology of Doyle, Doyle (1990). PCRs had a total volume of 20 μL and included: 13.9 μL of ultra pure water; 2 μL of 10x PCR buffer; 0.2 μL of primers cocktail FishF2t1 (10 μM) and VF2t1 (10 μM) (Ivanova et al., 2007Ivanova NV, Zemlak TS, Hanner RH, Hebert PDN. Universal primer cocktails for fish DNA barcoding. Mol Ecol Resour. 2007; 7(4):544-48.); 0,2 μL of primers cocktail FishR2t1 (10 μM) and FishR1Dt1 (10 μM) (Ivanova et al., 2007Ivanova NV, Zemlak TS, Hanner RH, Hebert PDN. Universal primer cocktails for fish DNA barcoding. Mol Ecol Resour. 2007; 7(4):544-48.); 0.1 μL Platinum Taq DNA Polymerase (Invitrogen); 1.0 μL of DNA template (10-50 ng). The thermocycle profile consisted of 94°C for 5 min, 35 cycles of 94°C for 45 s, 52°C for 1,5 min, and 72°C for 2 min, with a final extension at 72°C for 10 min. PCR products were checked by electrophoresis on a 1% agarose gel, purified using ExoSAP (Exonuclease I and Shrimp Alkaline Phosphatase-GE Healthcare) and bi-directionally sequenced using BigDye kit (Applied Biosystems®) in capillary sequencer ABI-Prism 3500 Genetic Analyzer (Applied Biosystems®) at ACTGene Análises Moleculares Ltda. Each forward and reverse COI sequence was inspected by eye and obvious mistakes corrected. COI sequences were aligned using Clustal W (Thompson et al., 1994Thompson JD, Higgins DG, Gibson TJ. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994; 22(22):4673-80.) under default parameters implemented in MEGA 6.0 (Tamura et al., 2013Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: Molecular Evolutionary Genetics Analysis Version 6.0. Mol Biol Evol. 2013; 30(12):2725-29.). Sequences were uploaded to GenBank.
Institutional acronyms: AMNH, American Museum of Natural History; MCN, Museu de Ciências Naturais, Fundação Zoobotânica do Rio Grande do Sul; MCP, Museu de Ciências e Tecnologia da Pontifícia Universidade Católica do Rio Grande do Sul; FMNH, Field Museum of Natural History; MNRJ, Museu Nacional, Universidade Federal do Rio de Janeiro; MZUEL, Museu de Zoologia, Universidade Estadual de Londrina; MZUSP, Museu de Zoologia, Universidade de São Paulo; UFRGS, Departamento de Zoologia, Universidade Federal do Rio Grande do Sul; USNM, Smithsonian Institution National Museum of Natural History. Other abbreviations in the text: c&s (cleared and stained specimens); SL (standard length); tec (specimens with tissue sample available).
Results
Ituglanisinusitatus, new species
urn:lsid:zoobank.org:act:4172BB61-BEE6-4060-B5CC-529A9A41C1B8
(Figs. 1-4, Table 1)
Ituglanis sp. 2: Bertaco et al. (2016Bertaco VA, Ferrer J, Carvalho FR, Malabarba LR. Inventory of the freshwater fishes from a densely collected area in South America - a case study of the current knowledge of Neotropical fish diversity. Zootaxa. 2016; 4138(3):401-40.): p. 421 (table 1; list of species from the Rio Grande do Sul State).
Holotype. UFRGS 21829, 62.2 mm SL, Brazil, Rio Grande do Sul, Município de Alegrete, arroio São João tributary of rio Ibicuí, rio Uruguai basin, 29°46’52”S 55°24’56”W, ca. 92 m, 28 Oct 2013, C. Hartmann, T. Guimarães, R. Dala-Corte, M. Dalmolin & L. Poldgaiski.
Paratypes. All from Brazil, Rio Grande do Sul, rio Uruguay basin: MCN 19975, 1, 54.9 mm SL, collected with holotype. UFRGS 19310, 1 (tec), 70.6 mm SL, Município de Alegrete, unnamed stream tributary of arroio São João, rio Ibicuí basin, 29°46’16”S 55°24’00”W, 28 Oct 2013, C. Hartmann, T. Guimarães, R. Dala-Corte, M. Dalmolin & L. Poldgaiski. UFRGS 19311, 4 (3 tec), 56.6-64.9 mm SL, collected with holotype. UFRGS 20201, 8 (6 tec, 1 c&s), 43.3-70.2 mm SL, boundary between Município de Itaquí and Município de Maçambará, unnamed stream tributary of rio Butuí at road RS-472, 29°00’06”S 56°10’38”W, 15 May 2015, J. Chuctaya, K. Bonato, L. R. Malabarba, M. C. Malabarba & R. Angrizani. UFRGS 21242, 1, 32.8 mm SL, Município de São Francisco de Assis, rio Ibicuí at Jacaquá beach, 29°41’24” S 55°11’40”W, 22 Mar 2007, B. Klotzel, L. E. Lanés & M. Volcán.
Paragenetype COI. GenBank accession numbers MF975518 (voucher tec 5502A): UFRGS 20201, MF975519 (voucher tec 5502B): UFRGS 20201, MF975520 (voucher tec 4966): UFRGS 19311.
Diagnosis.Ituglanisinusitatus is distinguished from most congeners by the presence of two to three pairs of ribs (vs. 4-10). The following species exhibit one to three pairs of ribs: I. amazonicus (Steindachner), I. apteryx Datovo, I. eichhorniarum (Miranda Ribeiro), I. gracilior (Eigenmann), I. herberti (Miranda Ribeiro), I. ina Wosiacki, Dutra & Mendonça, I. macunaima Datovo & Landim, I. metae (Eigenmann), I. nebulosus de Pinna & Keith, I. parkoi (Miranda Ribeiro); and I. compactus Castro & Wosiacki. Ituglanis inusitatus is distinguished from these species by the color pattern composed of an outer layer with scattered round black blotches equivalent in size to the eye circumference over a reddish brown background on the lateral surface of the body (Fig. 1; vs. color pattern uniform, i.e. lacking a conspicuous outer layer in I. compactus, I. herberti, I. ina and I. parkoi; color pattern composed of an outer layer with reticulated brown blotches over a pale yellow or whitish background on the lateral surface of the body in I. amazonicus, I. eichhorniarum, I. guayaberensis (Dahl), I. gracilior, I. macunaima and I. metae; color pattern composed of an outer layer with longitudinal stripes over a pale yellow background on the lateral surface of the body in I. apteryx; or color pattern composed of an outer layer with blotches larger than twice the eye diameter in I. nebulosus). Additionally, the presence of i+6 pectoral-fin rays distinguishes Ituglanis inusitatus from I. amazonicus (i+5), I. macunaima (i+3-5), I. metae (i+5) and I. nebulosus (i+5). Ituglanis inusitatus is further distinguished from I. apteryx by the presence of pelvic fins (vs. absence). Ituglanis inusitatus is further distinguished from I. compactus by the absence of the antorbital segment of infraorbital laterosensory canal (vs. presence). Ituglanis inusitatus is further distinguished from I. gracilior by the maxillary barbel not extending beyond the pectoral-fin insertion (vs. maxillary barbel reaching tip of pectoral fin). Ituglanis inusitatus is further distinguished from I. ina by the absence of neuromasts extending along the midlateral line of the body (vs. presence). Ituglanis inusitatus is distinguished from its sympatric congener in the rio Uruguay basin, I. australis, by the presence of the nasal canal of laterosensory system (vs. absence) and by the number of pectoral-fin rays (i+6 vs. i+4-5).
Ituglanisinusitatus, new species, holotype, UFRGS 21829, 62.2 mm SL; arroio São João, Alegrete, rio Ibicuí basin, Rio Grande do Sul State, Brazil. Cut over the dorsal margin of the caudal peduncle is absent in remaining type specimens.
Ituglanisinusitatus, new species, paratype, UFRGS 20201, 70.2 mm SL, c&s. Dorsal view of gill arches: basibranchials 2 to 4 (BB2-4), ceratobranchials 1 to 5 (CB1-5), epibranchials 1 to 4 (EB1-4), hypobranchials 1 to 3 (HB1-3), pharyngobranchial 3 (PB3), tooth plate attached to pharyngobranchial 4 (TP). Arrows indicate the process at distal portion of anterior margin of hypobranchial 1.
Description. Morphometric data in Tab. 1. Body elongate, trunk roughly cylindrical, gradually compressed towards caudal fin. Dorsal profile of body straight up to dorsal-fin insertion and slightly concave from this point to caudal-fin insertion. Ventral profile slightly convex up to pelvic-fin insertion and straight to slightly concave from this point to caudal-fin insertion.
Head depressed, trapezoidal from dorsal view, wider posteriorly. Dorsal profile straight and ventral profile straight to slightly convex. Snout straight to slightly round from dorsal view. Eyes dorsally oriented but also visible from lateral view; located on anterior half of head, near to posterior nostrils; orbital rim not free, eyes covered by thin and transparent skin. Nostrils smaller than eye diameter. Anterior nostril surrounded by fleshy flap of integument posterolaterally continuous with nasal barbel. Posterior nostril surrounded anterolaterally by thin flap of integument. Gill openings not constricted united with isthmus anteriorly forming free fold. Mouth subterminal and slightly convex from ventral view. Lower lip with lateral fleshy fold in corners continuous with rictal barbel. Lips with small papillae.
Barbels with large bases and tapering gradually towards tips. Nasal barbel emerging from posterolateral edge of anterior nostril and reaching posterior limit of skull. Maxillary barbel always extending beyond interopercle, usually reaching pectoral-fin insertion. Rictal barbel sligthly smaller than maxillary barbel.
Morphometric data of Ituglanis inusitatus, new species. Range refers to eight specimens in total including holotype (UFRGS 21829) and seven paratypes (MCN 19975: n = 1; UFRGS 19311: n = 1; UFRGS 20201: n = 4; UFRGS 21242: n = 1); SD: standard deviation.
Mesethmoid large at middle portion with anterior margin slightly concave and cornuas tapering at ends (Fig. 3). Anterior cranial fontanel absent; posterior cranial fontanel small and round located in posterior portion of parieto-supraoccipital. Antorbital short and extending into autopalatine. Tendon-bone supraorbital elongate, approximately three times larger than antorbital, anterior third over the posterior portion of autopalatine (Fig. 3). Sphenotic with anterior margin pointed surpassing adjacent tip of frontal. Sphenotic, prootic, and pterosphenoid totally fused. Vomer arrow-shaped with long posterior process over parasphenoid. Parasphenoid with pointed posterior process extending over basioccipital. Weberian capsule with small lateral opening and with anterior margin fused to basioccipital.
Ituglanisinusitatus, new species, paratype, UFRGS 20201, 70.2 mm SL, c&s. Dorsal view of mesethmoid (ME), premaxilla (PM), maxilla (MX), tendon-bone supraorbital (SO), frontal (FR), autopalatine (AP), and antorbital (AN). Arrows indicate the enlargement at middle portion of mesethmoid. Gray area represents cartilage. Scale bar = 2 mm.
Premaxilla rectangular with 21 pointed teeth variable in size distributed in two well-defined rows and curved inward. Maxilla boomerang-shaped, shorter than premaxilla (Fig. 3). Lower jaw with 24-27 pointed teeth variable in size distributed in two or three irregular rows and curved inward. Autopalatine with anterior margin straight; medial margin with deep concavity and lateral margin concave (Fig. 2); posterior process of autopalatine extending over metapterygoid.
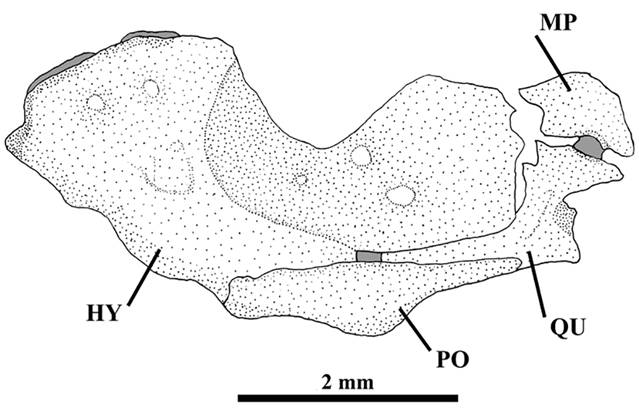
Metapterygoid large and laminar, connected with quadrate through cartilage (Fig. 4). Quadrate L-shaped with two small process in anterior margin. Hyomandibula well-developed with deep notch in dorsal margin. Preopercle long, narrow, in contact with ventral margins of quadrate and hyomandibula. Opercle elongate with small process directed upward and 17-19 pointed odontodes inserted in posterior margin. Interopercle elongate with dorsal process near anterior margin and 18-20 pointed odontodes along inferior margin extending from dorsal process up to posterior margin. Odontodes of opercle and interopercle curved at tips and gradually increasing in size posteriorly.
Ituglanisinusitatus, new species, paratype, UFRGS 20201, 70.2 mm SL, c&s. Lateral view of right suspensorium: metapterygoid (MP), quadrate (QU), preopercle (PO), and hyomandibula (HY). Gray area represents cartilage. Scale bar = 2 mm.
Ventral hypohyal trapezoid-shaped. Anterior ceratohyal elongated and with small concavity at middle length. Proximal and distal ceratohyals of approximately same depth. Posterior ceratohyal short with distal portion rounded. Eight branchiostegal rays: two or three in contact with anterior ceratohyal, four or five in contact with posterior ceratohyal and one laterally free of contact. Last four branchiostegal rays widest distally. Urohyal with expanded anterior head bearing two lateral elongated processes with wider base and pointed tips and smaller posterior process pointed, elongated and laminar; posterior process smaller than lateral processes.
Basibranchials 2 and 3 connected to each other by cartilage, basibranchial 2 slightly wider than basibranchial 3 (Fig. 2). Ossified portion of basibranchial 2 distinctly wider than basibranchial 3. Basibranchial 4 hexagonal and entirely cartilaginous. Hypobranchial 1 elongated with cartilage at tips and with small process at lateral portion of anterior margin. Hypobranchial 2 and 3 with narrow anterolateral ossified processes with large area of cartilage distally. Hypobranchial 2 more elongated than hypobranchial 3. Five elongate ceratobranchials with cartilage at tips. Ceratobranchial 3 with distinct concavity in posterior margin. Ceratobranchial 5 with approximately 12 teeth. Four epibranchials; first three elongate and narrow with cartilage at tips. Epibranchial 1 with elongate process in anterior margin; epibranchial 2 with small process in anterior margin. Epibranchial 3 with distinct curved process in posterior margin. Epibranchial 4 rectangular. Pharyngobranchial 3 narrow and curved at middle length with cartilage at tips, Pharyngobranchial 4 ossified and connected to curved plate with approximately 16 teeth arranged in up to two rows; teeth increasing in length posteriorly.
Vertebrae 42; abdominal vertebrae 5. Ribs 2 to 3 pairs, first rib thickest. Most anterior free vertebrae with parapophyses directed laterally. First complete haemal arch on sixth free vertebrae and first complete haemal spine on 15th free vertebrae.
Pectoral fin with distal margin rounded; one unbranched ray prolonged as short filament and six branched rays (16*). Pelvic fin with distal margin rounded, reaching anterior margin of urogenital papilla; one unbranched ray and four branched rays (16*). Dorsal fin with distal margin rounded, semicircular when fin expanded and with three unsegmented rays, one (8*) or two (8) unbranched rays and six (1) or seven (15*) branched rays. Origin of dorsal fin located at vertical through tip of pelvic fin. Dorsal fin with eight pterygiophores; first inserted anteriorly to neural spine of 26th vertebrae. Anal fin with distal margin slightly rounded and with three unsegmented rays (1), one (15*) or two (1) unbranched rays and five (2) or six (14*) branched rays. Origin of anal fin located slightly posterior to vertical through dorsal-fin origin. Anal fin with six pterygiophores; first inserted anterior to haemal spine of 28th vertebrae.
Caudal fin with distal margin rounded. Procurrent caudal-fin rays 15 dorsally and 13 ventrally. Upper plate with one unbranched ray and 5 branched rays. Lower caudal plate with one unbranched ray and 6 branched rays (16*). Lower caudal plate with parhypural and hypurals 1 and 2 co-ossified and fused to compound caudal centrum. Upper caudal plate with separate uroneural; hypural 3 autogenous; hypurals 4 and 5 fused.
Laterosensory canals with simple (non-dendritic) tubes ending in single pores. Nasal and frontal canals of supraorbital line fused to each other, with three pores (“s1”, “s3” and “s6”) present in all types (16*); three of them with extra pore on left side near to “s3” (1) or near to “s6” (2). Posterior segment of frontal, sphenotic and otic canals fused to each other. Antorbital segment of infraorbital line absent (16*). Sphenotic canal present (16*) with two pores (“i10” and “i11”); one of them with pore “i10” absent on left side and one of them with extra pore near to “i11” on left side. Otic, postotic and scapular canals present (16*) with preoperculo-mandibular and pterotic branches short and with one associated pore each. Trunk canal short with two pores (16*).
Coloration in alcohol. Dorsal and lateral surfaces of body with scattered black round blotches (equivalent in size to eye circumference) over reddish brown background (Fig. 1). Blotches more conspicuous in dorsolateral surface. Ventral surface of body white with few brown spots along pelvic fin insertion to caudal peduncle. Dorsal and lateral surfaces of head with black blotches over reddish brown background; larger black blotch in region of parieto-supraoccipital bone. Maxillary and nasal barbels with reddish brown blotches over light-yellow background; blotches more concentrated near base. Rictal barbel light yellow. Anal, dorsal, pelvic and pectoral fins with scattered round black blotches over light brackground. Caudal fin with black blotches forming vertical stripes over light brackground; distal stripe larger.
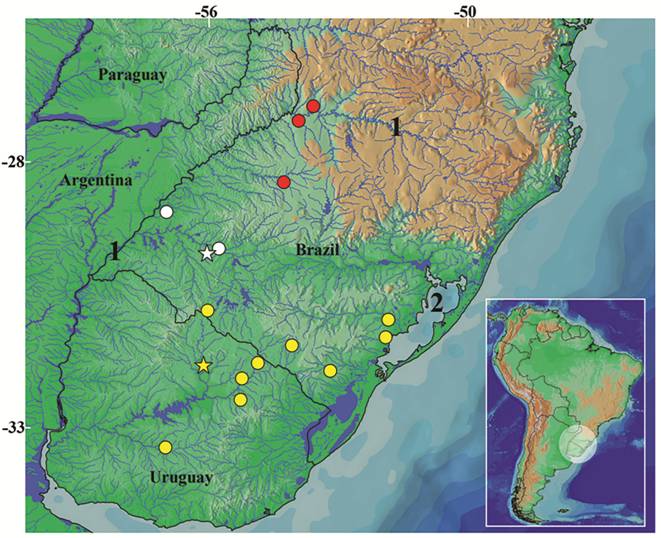
Geographical distribution.Ituglanis inusitatus is so far known only from four localities of the rio Ibicuí and rio Butuí basins, tributaries of the left bank of the rio Uruguai (Fig. 5), State of Rio Grande do Sul, southern Brazil. These localities are included in the Pampa grasslands, the southernmost Brazilian biome according to IBGE (2004Instituto Brasileiro de Geografia e Estatística (IBGE). Mapa de Biomas do Brasil [Internet]. Rio de Janeiro: IBGE; 2004 [updated 2013 Jan 10; cited 2016 Dec 28]. Available from: Available from: http://www.ibge.gov.br/home/presidencia/noticias/21052004biomashtml.shtm
http://www.ibge.gov.br/home/presidencia/...
). Ituglanis australis and Ituglanis sp. 3 are the two congeners found in the rio Uruguai basin. Despite this fact, the three species have never been collected sintopically: I. australis is distributed over a larger area in the rio Uruguai basin and occurs in the south tributaries of the laguna dos Patos system, restricted to the Pampa biome; and Ituglanis sp. 3 is so far known from only a few localities in the rio Uruguai basin (Fig. 5), all inside the Atlantic forest biome.
Geographic distribution of Ituglanis inusitatus, new species (white symbols), I. australis (yellow symbols) and Ituglanis sp. 3 (red symbols). Some symbols represent more than one collection locality. Stars represent type localities. Numbers 1 and 2 indicate rio Uruguay and laguna dos Patos, respectively.
Ecological notes. The arroio São João (Fig. 6; type locality) and its unnamed tributary are surrounded by preserved riparian vegetation. The bottom is mostly sandy (granulation of 0.6-2.0 mm) with few fallen leaves and branches. Water velocity was moderate at type locality and its physicochemical parameters between 2.40 and 4.00 pm were as follows: temperature (20.4°C), dissolved oxygen (6.85 mg/L), pH (5.76) and specific conductive (22.7 µS/cm). According to R. Dala-Corte, PhD (oral communication, November 2016Dala-Corte RB, Giam X, Olden JD, Becker FG, Guimarães TDF, Melo AS. Revealing the pathways by which agricultural land-use affects stream fish communities in South Brazilian grasslands. Freshwater Biol. 2016; 61(11):1921-34.), all three specimens collected at type locality were not visible on sand bottom until the use of the electroshock. The following species were collected with Ituglanis inusitatus at the type locality, not necessarily in the same microhabitat: Bryconamericus iheringii (Boulenger), Bunocephalus doriae Boulenger, Crenicichla lepidota Heckel, Eigenmannia trilineata López & Castello, Hisonotus charrua Almirón, Azpelicueta, Casciotta & Litz, Phalloceros caudimaculatus (Hensel), Pseudohemiodon laticeps (Regan), Rineloricaria stellata Ghazzi and Scleronema operculatum Eigenmann.
Type locality of Ituglanis inusitatus, new species: arroio São João, Alegrete, rio Ibicuí basin, Rio Grande do Sul State, Brazil.
Etymology. From Latin inusitatus (unusual, rare) in reference to the recent discovery of the new species (see discussion). Treated as an adjective.
Conservation status.Ituglanis inusitatus is known from only four localities corresponding to an Extent of Occurrence (EOO) of approximately 1,400 km2. Although the species seems to be rare and known only from habitats with preserved riparian vegetation, no specific treats were detected and the species can be classified as LC according to IUCN criteria (IUCN, 2016International Union for Conservation of Nature (IUCN). Standard and Petitions Subcommittee. Guidelines for using the IUCN Red List Categories and Criteria. Version 12 [Internet]. 2016 [updated 2016 Feb]. Available from: http://cmsdocs.s3.amazonaws.com/RedListGuidelines.pdf
http://cmsdocs.s3.amazonaws.com/RedListG...
).
Discussion
The genus Ituglanis was proposed by Costa, Bockmann (1993Costa WJEM, Bockmann FA. Un nouveau genre néotropical de la famille des Trichomycteridae (Siluriformes: Loricarioidei). RFAHRevue Française d’Aquariologie et Herpétologie . 1993; 20(2):43-46.) as a monophyletic group based on three putative autapomorphies: the posterior cranial fontanel present as an orifice situated posteriorly in the parieto-supraoccipital; the anterior portion of the sphenotic-prootic-pterosphenoid directed anteriorly; and the presence of a deep concavity in the medial margin of the autopalatine. These three characters are present in Ituglanis inusitatus (see Fig. 3 for the autopalatine shape).
Wosiacki (2002Wosiacki WB. Estudo das relações filogenéticas de Trichomycterinae (Teleostei, Siluriformes, Trichomycteridae) com uma proposta de classificação. [PhD Thesis]. São Paulo: Universidade de São Paulo; 2002.) based on a phylogenetic analysis including four species of Ituglanis (I. amazonicus, I. eichhorniarum, I. proops and an undescribed species) cited four characters shared by them, posteriorly inferred as putative synapomorphies (Wosiacki et al., 2012Wosiacki WB, Dutra GM, Mendonça MB. Description of a new species of Ituglanis (Siluriformes: Trichomycteridae) from Serra dos Carajás, rio Tocantins basin. Neotrop Ichthyol . 2012; 10(3):547-54.). Datovo, de Pinna (2014Datovo A, de Pinna MCC. A new species of Ituglanis representing the southernmost record of the genus, with comments on phylogenetic relationships (Teleostei: Siluriformes: Trichomycteridae). J Fish Biol. 2014; 84(2):314-27.) confirmed the presence of two of those characters in a broader sample of species of Ituglanis examined by them: five or more abdominal vertebrae and two or fewer vertebrae between the first dorsal-fin pterygiophore and the first anal-fin pterygiophore. Indeed, these two conditions are present in I. inusitatus; which has five abdominal vertebrae and one vertebra between the first dorsal-fin pterygiophore and the first anal-fin pterygiophore.
Recently, Rizzato, Bichuette (2016Rizzato PP, Bichuette ME. The laterosensory canal system in epigean and subterranean Ituglanis (Siluriformes: Trichomycteridae), with comments about troglomorphism and the phylogeny of the genus. J Morphol. 2016; 278(1):4-28.) conducted a complete analysis of the laterosensory canal system within Ituglanis and discussed the previous hypotheses of groupings inside the genus (i.e.de Pinna, Keith, 2003de Pinna M, Keith P. A new species of the catfish genus Ituglanis from French Guyana (Osteichthyes: Siluriformes: Trichomycteridae). Proc Biol Soc Wash. 2003; 116(4):873-82.; Rizzato, Bichuette, 2014Rizzato PP, Bichuette ME. Ituglanis boticario, a new troglomorphic catfish (Teleostei: Siluriformes: Trichomycteridae) from Mambaí karst area, central Brazil. Zoologia (Curitiba). 2014; 31(6):577-98.). The authors inferred some putative phylogenetic relationships relevant to discussion with the discovery of I. inusitatus.
De Pinna, Keith (2003de Pinna M, Keith P. A new species of the catfish genus Ituglanis from French Guyana (Osteichthyes: Siluriformes: Trichomycteridae). Proc Biol Soc Wash. 2003; 116(4):873-82.) suggested that a reduced number of ribs (three or less) could be interpreted as a derived character shared by a group of exclusively Amazonian species, composed by the time of I. amazonicus, I. gracilior, I.nebulosus and an undescribed species. According to an updated list presented by Rizzato, Bichuette (2016Rizzato PP, Bichuette ME. The laterosensory canal system in epigean and subterranean Ituglanis (Siluriformes: Trichomycteridae), with comments about troglomorphism and the phylogeny of the genus. J Morphol. 2016; 278(1):4-28.), this group should include all species occurring in the drainages inside the Amazon forest plus two species from Paraná-Paraguai basin (I. eichhorniarum and I. herberti). Rizzato, Bichuette (2016Rizzato PP, Bichuette ME. The laterosensory canal system in epigean and subterranean Ituglanis (Siluriformes: Trichomycteridae), with comments about troglomorphism and the phylogeny of the genus. J Morphol. 2016; 278(1):4-28.) named the latter the “northwestern South America group”, in contrast to the “southeastern group”, which should comprise the remaining species of the genus. Ituglanis inusitatus has less than three ribs and inhabits tributaries of the left bank of the rio Uruguai (southern Brazil), a drainage encompassed by the rio Paraná-Paraguai basin (Reis et al., 2016Reis RE, Albert JS, Di Dario F, Mincarone MM, Petry P, Rocha LA. Fish biodiversity and conservation in South America. J Fish Biol . 2016; 89(1):12-47.), thus contrasting with the biogeographical distribution of the groups proposed by de Pinna, Keith (2003de Pinna M, Keith P. A new species of the catfish genus Ituglanis from French Guyana (Osteichthyes: Siluriformes: Trichomycteridae). Proc Biol Soc Wash. 2003; 116(4):873-82.) and Rizzato, Bichuette (2016Rizzato PP, Bichuette ME. The laterosensory canal system in epigean and subterranean Ituglanis (Siluriformes: Trichomycteridae), with comments about troglomorphism and the phylogeny of the genus. J Morphol. 2016; 278(1):4-28.).
De Pinna, Keith (2003de Pinna M, Keith P. A new species of the catfish genus Ituglanis from French Guyana (Osteichthyes: Siluriformes: Trichomycteridae). Proc Biol Soc Wash. 2003; 116(4):873-82.) also cited a less inclusive group inside their Amazon clade (I. amazonicus, I.nebulosus and the same undescribed species cited above) sharing a widening of the mesethmoid shaft. However, Rizzato, Bichuette (2016Rizzato PP, Bichuette ME. The laterosensory canal system in epigean and subterranean Ituglanis (Siluriformes: Trichomycteridae), with comments about troglomorphism and the phylogeny of the genus. J Morphol. 2016; 278(1):4-28.) highlighted the intraspecific variability of this character in the genus, thereby contesting its usability to group species of Ituglanis. In the unique c&s specimen of I. inusitatus analyzed, the mesethmoid shaft has a conspicuous enlargement at its middle portion (Fig. 3).
The absence of the antorbital segment of the infraorbital laterosensory canal was suggested as a putative apomorphic condition in Ituglanis shared by all the species of the so-called northwestern group of Rizzato, Bichuette (2014Rizzato PP, Bichuette ME. Ituglanis boticario, a new troglomorphic catfish (Teleostei: Siluriformes: Trichomycteridae) from Mambaí karst area, central Brazil. Zoologia (Curitiba). 2014; 31(6):577-98., 2016Rizzato PP, Bichuette ME. The laterosensory canal system in epigean and subterranean Ituglanis (Siluriformes: Trichomycteridae), with comments about troglomorphism and the phylogeny of the genus. J Morphol. 2016; 278(1):4-28.), plus three species from the so-called southeastern group: I. cayensis, I. parahybae and I. goyaDatovo, Aquino & Langeani (Rizzato, Bichuette, 2014Datovo A, de Pinna MCC. A new species of Ituglanis representing the southernmost record of the genus, with comments on phylogenetic relationships (Teleostei: Siluriformes: Trichomycteridae). J Fish Biol. 2014; 84(2):314-27., 2016Rizzato PP, Bichuette ME. The laterosensory canal system in epigean and subterranean Ituglanis (Siluriformes: Trichomycteridae), with comments about troglomorphism and the phylogeny of the genus. J Morphol. 2016; 278(1):4-28.). The antorbital segment of the infraorbital laterosensory canal is invariably absent in I. inusitatus, while it is variably present in its congener occurring in the rio Uruguai basin, I. australis; see Datovo, de Pinna (2014Datovo A, de Pinna MCC. A new species of Ituglanis representing the southernmost record of the genus, with comments on phylogenetic relationships (Teleostei: Siluriformes: Trichomycteridae). J Fish Biol. 2014; 84(2):314-27.).
In the single c&s specimen of Ituglanis inusitatus analyzed, we observed a process at the distal portion of the anterior margin of the first hypobranchial (Fig. 2). A similar process was observed in representatives of the basal trichomycterid subfamilies Copionodontinae and Trichogeninae, in two Trichomycterus species - T. pardus Cope (= T. rivulatus Valenciennes) and T. spelaeus DoNascimiento, Villarreal & Provenzano - and in the glanapterygine Listrura costai Villa-Verde, Lazzarotto & Lima (de Pinna, 1998de Pinna MCC. Phylogenetic relationships of Neotropical Siluriformes (Teleostei: Ostariophysi): historical overview and synthesis of hypotheses. In: Malabarba LR, Reis RE, Vari RP, Lucena ZMS, Lucena CAS, editors. Phylogeny and classification of Neotropical fishes. Porto Alegre: Edipucrs; 1998. p.279-330.; DoNascimiento et al., 2001DoNascimiento C, Villarreal O, Provenzano F. Descripción de una nueva especie de bagre anoftalmo del género Trichomycterus (Siluriformes, Trichomycteridae), de una cueva de la Sierra de Perijá, Venezuela. Bol Soc Venez Espeleol. 2001; 35:20-26.; Wosiacki, 2002Wosiacki WB. Estudo das relações filogenéticas de Trichomycterinae (Teleostei, Siluriformes, Trichomycteridae) com uma proposta de classificação. [PhD Thesis]. São Paulo: Universidade de São Paulo; 2002.; Villa-Verde et al., 2012Villa-Verde L, Lazzarotto H, Lima SMQ. A new glanapterygine catfish of the genus Listrura (Siluriformes: Trichomycteridae) from southeastern Brazil, corroborated by morphological and molecular data. Neotrop Ichthyol . 2012; 10(3):527-38.). In Ituglanis, a similar process was only observed at the right side of a single paratype of I. goya (F. Langeani, PhD: written communication, March 2017). Therefore, this character also may be interspecifically variable in I. inusitatus considering that only one c&s specimen was analyzed. Even though is not possible to confirm its absence in all the known species of Ituglanis based on the current knowledge about the genus, the distribution of these character should be further investigated in order to analyze its putative phylogenetic significance.
The new species was recognized after identifying seven specimens from material collected recently on an extensive survey using electrofishing gear in 54 streams located in the South Brazilian grasslands (cf.Dala-Corte et al., 2016Dala-Corte RB, Giam X, Olden JD, Becker FG, Guimarães TDF, Melo AS. Revealing the pathways by which agricultural land-use affects stream fish communities in South Brazilian grasslands. Freshwater Biol. 2016; 61(11):1921-34.). Thereafter, eight new specimens of I. inusitatus were captured employing the same methodology and only the smallest type-specimen (UFRGS 21242: 32.8 mm SL), identified after recognition of the new species, was collected without the help of electroshocking. The exploration of streams with preserved riparian vegetation located in less sampled areas in the Rio Grande do Sul State ((lower rio Ibicuí and rio Butuí; see fig. 2 of Bertaco et al. (2016Bertaco VA, Ferrer J, Carvalho FR, Malabarba LR. Inventory of the freshwater fishes from a densely collected area in South America - a case study of the current knowledge of Neotropical fish diversity. Zootaxa. 2016; 4138(3):401-40.)) with the aid of electrofishing, a method currently known for its high performance in capturing benthic species in hidden places, may explain such a recent discovery.
Dala-Corte et al. (2016Dala-Corte RB, Giam X, Olden JD, Becker FG, Guimarães TDF, Melo AS. Revealing the pathways by which agricultural land-use affects stream fish communities in South Brazilian grasslands. Freshwater Biol. 2016; 61(11):1921-34.) emphasized the importance of riparian zones in the maintenance of fish communities and recommended their protection and recovery as an attempt to mitigate the effects of agriculture in South Brazilian grasslands. The presence of I. inusitatus in only two of the 54 streams sampled by these authors, both with preserved riparian vegetation and without agricultural influence in the vicinities, provides additional support to this recommendation.
Comparative material examined.Ituglanis apteryx: MZUSP 115048, 1, 62.3 mm SL, holotype of I. apteryxDatovo, 2014Datovo A, de Pinna MCC. A new species of Ituglanis representing the southernmost record of the genus, with comments on phylogenetic relationships (Teleostei: Siluriformes: Trichomycteridae). J Fish Biol. 2014; 84(2):314-27.. Ituglanis australis: UFRGS 12578, 1, 40.2 mm SL, paratype of I. australis Datovo & de Pinna, 2014; UFRGS 13600, 1, 75,2 mm SL, paratype of I. australis Datovo & de Pinna, 2014; UFRGS 19184, 2 (tec), 43.07-43.19 mm SL; UFRGS 19656, 1, 51.1 mm SL. Ituglanis bambui Bichuette & Trajano: MZUSP 79860, 1, 41.7 mm SL, holotype of I. bambui Bichuette & Trajano, 2004. Ituglanis boitata: UFRGS 17617, 5 (1 c&s), 43.0-66.2 mm SL, paratypes of I. boitataFerrer, Donin & Malabarba, 2015Ferrer J, Donin LM, Malabarba LR. A new species of Ituglanis Costa & Bockmann, 1993 (Siluriformes: Trichomycteridae) endemic to the Tramandaí-Mampituba ecoregion, southern Brazil. Zootaxa , 2015; 4020(2):375-89.. Ituglanis cahyensis Sarmento-Soares, Martins-Pinheiro, Aranda & Chamon: MNRJ 28404, 46.9 mm SL, holotype of I. cahyensis Sarmento-Soares, Martins-Pinheiro, Aranda & Chamon, 2006. Ituglanis eichhorniarum: AMNH 233244, 1, 64.9 mm SL; AMNH 233230, 2, 42.8-60.1 mm SL; MNRJ 780, 2, 34.2 mm SL, paralectotypes of T. eichorniarum (Miranda Ribeiro, 1912Miranda Ribeiro A. Loricariidae, Callichthyidae, Doradidae e Trichomycteridae. Commissão de Linhas Telegraphicas Estrategicas de Matto-Grosso ao Amazonas. 1912; Annexo 5:1-31.) designated by Miranda Ribeiro (1953: 405); MZUEL 09074, 5, 34.9-47.3 mm SL. MZUEL 12190, 6, 20.7-29.9 mm SL. Ituglanis epikarsticus Bichuette & Trajano: MZUSP 79869, 1, 26.0 mm SL, holotype of I. epikarsticus Bichuette & Trajano, 2004. Ituglanis gracilior: FMNH 53264, not measured, holotype of Pygidium gracilior. Ituglanis herberti: MNRJ 1429, 2, 52.7-54.9 mm SL, syntypes of T. herberti Miranda Ribeiro, 1940; MNRJ 1428, 1, 60.5 mm SL, syntype of T. herberti Miranda Ribeiro, 1940. Ituglanis ina: MZUSP 112508, 4, 47.4-57.8 mm SL. Ituglanis macunaima: MZUSP 88452, 30.7 mm SL, holotype of I. macunaima Datovo & Landim, 2005. Ituglanis mambai Bichuette & Trajano: MCP 42538, 53.8 mm SL, holotype of I. mambai Bichuette & Trajano, 2008; MCP 42537, 1, 68.9 mm SL, paratype of I. mambai Bichuette & Trajano, 2008. Ituglanis paraguassuensis Campos-Paiva & Costa: USNM 301016, 5, 20.9-37.7 mm SL paratypes of I. paraguassuensis Campos-Paiva & Costa, 2007. Ituglanis parahybae (Eigenmann): MNRJ 22980, 3, 39.8-45.0 mm SL; FMNH 58576, 1, 29.3 mm SL. I. passensis Fernández & Bichuette: MZUSP 79869, 26.2 mm SL, holotype I. passensis Fernández & Bichuette, 2002; MZUSP 80099, 2, 50.5-54.1 mm SL. Ituglanis proops (Eigenmann): MCP 20620, 8, 60.1-68.3 mm SL. MZUSP 60255, 10 (2 c&s), 57.5-60.4 mm SL. MNRJ 781, 3, 49.4-63.5 mm SL, paralectotypes of T. proops desiganted by Miranda Ribeiro (1953: 405). Ituglanis ramiroi Bichuette & Trajano: MZUSP 79865, 28.1 mm SL, holotype of I. ramiroi Bichuette & Trajano, 2004. Ituglanis sp. 1 (cited in Bertaco et al., 2016Bertaco VA, Ferrer J, Carvalho FR, Malabarba LR. Inventory of the freshwater fishes from a densely collected area in South America - a case study of the current knowledge of Neotropical fish diversity. Zootaxa. 2016; 4138(3):401-40.): UFRGS 20372, 5 (3 tec), 41.8-57.6 mm SL. UFRGS 20371, 25 (8 tec), 54.2-19,9 mm SL. Ituglanis sp. 3 (cited in Bertaco et al., 2016Bertaco VA, Ferrer J, Carvalho FR, Malabarba LR. Inventory of the freshwater fishes from a densely collected area in South America - a case study of the current knowledge of Neotropical fish diversity. Zootaxa. 2016; 4138(3):401-40.): MCP 20882, 1, 41.4 mm SL. MCP 40011, 3, 47.3-48.2 mm SL. UFRGS 20134, 1 (tec), 49.3 mm SL. UFRGS 21920, 1, 80.0 mm SL. UFRGS 21921, 1 (tec), 84.1-80.0 mm SL.
Acknowledgments
We are grateful to Renato Dala-Corte and Taís Guimarães (UFRGS) for collection data and photo of the type locality. Thanks to Aléssio Datovo (MZUSP), Carlos. A. S. Lucena (MCP), Oscar Shibatta (MZUEL) and Laísa W. Cavalheiro (UFRGS) who loaned or donated the comparative material analyzed. Rafael Angrizani (UFRGS) and Juliana Wingert (UFRGS) gently helped with molecular procedures and draws under camera lucida, respectively. We thank for comments and suggestions made by two anonymous referees and by Francisco Langeani (DZSJRP). Casey Dillman (USNM) generously reviewed the English language of the manuscript. The authors are supported by Capes and CNPq (Proc. 1681448 to LMD) to LMD and 152354/2016-6 to JF).
References
- Abell R, Thieme ML, Revenga C, Bryer M, Kottelat M, Bogutskaya N, Coad B, Mandrak N, Contreras-Balderas SL, Bussing W, Stiassny MLJ, Skelton P, Allen GR, Unmack P, Naseka A, Ng R, Sindorf N, Robertson J, Armijo E, Higgins V, Heibel TJ, Wikramanayake E, Olson D, López HL, Reis RE, Lundberg JG, Sabaj-Pérez MHS, Petry R. Freshwater ecoregions of the world: a new map of biogeographic units for freshwater biodiversity conservation. Bioscience. 2008; 58(5):403-14.
- Becker FG, De Fries LCC, Ferrer J, Bertaco VA, Luz-Agostinho KDG, Silva JFP, Cardoso AR, Lucena ZMS, Lucena CAS. Fishes of the Taquari-Antas river basin (Patos Lagoon basin), southern Brazil. Braz J Biol. 2013; 73(1):79-90.
- Bertaco VA, Ferrer J, Carvalho FR, Malabarba LR. Inventory of the freshwater fishes from a densely collected area in South America - a case study of the current knowledge of Neotropical fish diversity. Zootaxa. 2016; 4138(3):401-40.
- Bockmann FA, Casatti L, de Pinna MCC. A new species of trichomycterid catfish from the Rio Paranapanema basin, southeastern Brazil (Teleostei: Siluriformes), with comments on the phylogeny of the family. Ichthyol Explor Freshw. 2004; 15(3):225-42.
- Canto ALC. Caracterização morfológica dos representantes do gênero Ituglanis (Siluriformes: Trichomycteridae) da bacia amazônica brasileira. [MSc Dissertation on the Internet]. Manaus, AM: Instituto Nacional de Pesquisas na Amazônia; 2009 [cited 2016 Dec 28]. Available from: Biblioteca Digital de Teses e Dissertações Instituto Nacional de Pesquisas da Amazônia. Available from: Biblioteca Digital de Teses e Dissertações Instituto Nacional de Pesquisas da Amazônia. http://bdtd.inpa.gov.br/handle/tede/2023
» http://bdtd.inpa.gov.br/handle/tede/2023 - Castro IDS, Wosiacki WB. Ituglanis compactus, a new species of catfish (Siluriformes: Trichomycteridae) from the rio Jari drainage, lower Amazon, Brazil. Zootaxa . 2017; 4244(2):207-18.
- Chakrabarty P. Genetypes: a concept to help integrate molecular phylogenetics and taxonomy. Zootaxa . 2010; 2632:67-68.
- Costa WJEM. Description de huit nouvelles espèces du genre Trichomycterus (Siluriformes: Trichomycteridae), du Brésil oriental. RFAHRevue Française d’Aquariologie et Herpétologie. 1992; 18(4):101-10.
- Costa WJEM, Bockmann FA. Un nouveau genre néotropical de la famille des Trichomycteridae (Siluriformes: Loricarioidei). RFAHRevue Française d’Aquariologie et Herpétologie . 1993; 20(2):43-46.
- Dahl G. Nematognathous fishes collected during the Macarena Expedition 1959. Part I. Novedades Colombianas. 1960; 1(5):302-17.
- Dala-Corte RB, Giam X, Olden JD, Becker FG, Guimarães TDF, Melo AS. Revealing the pathways by which agricultural land-use affects stream fish communities in South Brazilian grasslands. Freshwater Biol. 2016; 61(11):1921-34.
- Datovo A. A new species of Ituglanis from the Rio Xingu basin, Brazil, and the evolution of pelvic fin loss in trichomycterid catfishes (Teleostei: Siluriformes: Trichomycteridae). Zootaxa . 2014; 3790(3):466-76.
- Datovo A, Landim MI. Ituglanis macunaima, a new catfish from rio Araguaia basin, Brazil (Siluriformes, Trichomycteridae). Neotrop Ichthyol. 2005; 3(4):455-64.
- Datovo A, de Pinna MCC. A new species of Ituglanis representing the southernmost record of the genus, with comments on phylogenetic relationships (Teleostei: Siluriformes: Trichomycteridae). J Fish Biol. 2014; 84(2):314-27.
- Datovo A, Aquino PPU, Langeani F. A new species of Ituglanis (Siluriformes: Trichomycteridae) from the Tocantins and Paranaíba river basins, central Brazil, with remarks on the systematics of the genus. Zootaxa . 2016; 4171(3):439-58.
- DoNascimiento C, Villarreal O, Provenzano F. Descripción de una nueva especie de bagre anoftalmo del género Trichomycterus (Siluriformes, Trichomycteridae), de una cueva de la Sierra de Perijá, Venezuela. Bol Soc Venez Espeleol. 2001; 35:20-26.
- Eigenmann CH. The freshwater fishes of British Guiana, including a study of the ecological grouping of species, and the relation of the fauna of the plateau to that of the lowlands. Pittsburgh: Carnegie Institute; 1912. (Memoirs of the Carnegie Museum: vol 5).
- Eigenmann CH. Descriptions of sixteen new species of Pygidiidae. Proc Am Philos Soc. 1917; 56(7):690-703.
- Eschmeyer WN, Fong JD. Species by Family/Subfamily [Internet]. San Francisco: California Academy of Science; 2016 [updated 2017 Mar 01; cited 2017 Mar 29]. Available from: Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/SpeciesByFamily.asp
» http://researcharchive.calacademy.org/research/ichthyology/catalog/SpeciesByFamily.asp - Eschmeyer WN, Fricke R, van der Laan R, editors. Catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science ; 2016 [updated 2017 Mar 01; cited 2017 Mar 29]. Available from: Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
» http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp - Ferrer J, Donin LM, Malabarba LR. A new species of Ituglanis Costa & Bockmann, 1993 (Siluriformes: Trichomycteridae) endemic to the Tramandaí-Mampituba ecoregion, southern Brazil. Zootaxa , 2015; 4020(2):375-89.
- Ferrer J, Malabarba LR. A new Trichomycterus lacking pelvic fins and pelvic girdle with a very restricted range in southern Brazil (Siluriformes: Trichomycteridae). Zootaxa , 2011; 2912:59-67.
- Instituto Brasileiro de Geografia e Estatística (IBGE). Mapa de Biomas do Brasil [Internet]. Rio de Janeiro: IBGE; 2004 [updated 2013 Jan 10; cited 2016 Dec 28]. Available from: Available from: http://www.ibge.gov.br/home/presidencia/noticias/21052004biomashtml.shtm
» http://www.ibge.gov.br/home/presidencia/noticias/21052004biomashtml.shtm - International Union for Conservation of Nature (IUCN). Standard and Petitions Subcommittee. Guidelines for using the IUCN Red List Categories and Criteria. Version 12 [Internet]. 2016 [updated 2016 Feb]. Available from: http://cmsdocs.s3.amazonaws.com/RedListGuidelines.pdf
» http://cmsdocs.s3.amazonaws.com/RedListGuidelines.pdf - Ivanova NV, Zemlak TS, Hanner RH, Hebert PDN. Universal primer cocktails for fish DNA barcoding. Mol Ecol Resour. 2007; 7(4):544-48.
- Miranda Ribeiro A. Loricariidae, Callichthyidae, Doradidae e Trichomycteridae. Commissão de Linhas Telegraphicas Estrategicas de Matto-Grosso ao Amazonas. 1912; Annexo 5:1-31.
- Miranda Ribeiro P. Alguns peixes do sul de Mato Grosso. O Campo. 1940; 60:1.
- Miranda Ribeiro P. Um Pigidídeo do Alto Amazonas (Pisces - Pygidiidae). Bol Mus Nac Rio de J (Zool) 1944; 19:1-3.
- de Pinna MCC. Phylogenetic relationships of Neotropical Siluriformes (Teleostei: Ostariophysi): historical overview and synthesis of hypotheses. In: Malabarba LR, Reis RE, Vari RP, Lucena ZMS, Lucena CAS, editors. Phylogeny and classification of Neotropical fishes. Porto Alegre: Edipucrs; 1998. p.279-330.
- de Pinna M. Trichomycteridae. In: Queiroz LJ, Torrente-Vilara G, Ohara WM, Pires THS, Zuanon J, Doria CRC, organizers. Peixes do Rio Madeira Vol. 2. São Paulo: Dialeto Latin American Documentary; 2013. p.142-79.
- de Pinna M, Keith P. A new species of the catfish genus Ituglanis from French Guyana (Osteichthyes: Siluriformes: Trichomycteridae). Proc Biol Soc Wash. 2003; 116(4):873-82.
- Reis RE, Albert JS, Di Dario F, Mincarone MM, Petry P, Rocha LA. Fish biodiversity and conservation in South America. J Fish Biol . 2016; 89(1):12-47.
- Rizzato PP, Bichuette ME. Ituglanis boticario, a new troglomorphic catfish (Teleostei: Siluriformes: Trichomycteridae) from Mambaí karst area, central Brazil. Zoologia (Curitiba). 2014; 31(6):577-98.
- Rizzato PP, Bichuette ME. The laterosensory canal system in epigean and subterranean Ituglanis (Siluriformes: Trichomycteridae), with comments about troglomorphism and the phylogeny of the genus. J Morphol. 2016; 278(1):4-28.
- Scherman GE, Sutton T, Blazek R, Luthman L. QGIS 1.8 [Geographic Information Software System]. 2016. Available from: http://docs.qgis.org/1.8/pdf/
» http://docs.qgis.org/1.8/pdf/ - Steindachner F. Beiträge zur Kenntniss der Flussfische Südamerika’s (IV). Anz. Akad. Wiss. Wien, Math.-Naturwiss. Kl. 1882; 19(19):175-80.
- Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: Molecular Evolutionary Genetics Analysis Version 6.0. Mol Biol Evol. 2013; 30(12):2725-29.
- Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107-19.
- Tchernavin VV. A revision of some Trichomycterinae based on material preserved in the British Museum (Natural History). Proc Zool Soc London. 1944; 114(1-2):234-75.
- Thompson JD, Higgins DG, Gibson TJ. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994; 22(22):4673-80.
- Villa-Verde L, Lazzarotto H, Lima SMQ. A new glanapterygine catfish of the genus Listrura (Siluriformes: Trichomycteridae) from southeastern Brazil, corroborated by morphological and molecular data. Neotrop Ichthyol . 2012; 10(3):527-38.
- Wosiacki WB. Estudo das relações filogenéticas de Trichomycterinae (Teleostei, Siluriformes, Trichomycteridae) com uma proposta de classificação. [PhD Thesis]. São Paulo: Universidade de São Paulo; 2002.
- Wosiacki WB, Dutra GM, Mendonça MB. Description of a new species of Ituglanis (Siluriformes: Trichomycteridae) from Serra dos Carajás, rio Tocantins basin. Neotrop Ichthyol . 2012; 10(3):547-54.
- Wosiacki WB, de Pinna MCC. Trichomycterus igobi, a new catfish species from the rio Iguaçu drainage: the largest head in Trichomycteridae (Siluriformes: Trichomycteridae). Neotrop. Ichthyol. 2008; 6(1):17-23.
Data availability
Data citations
Eschmeyer WN, Fong JD. Species by Family/Subfamily [Internet]. San Francisco: California Academy of Science; 2016 [updated 2017 Mar 01; cited 2017 Mar 29]. Available from: Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/SpeciesByFamily.asp
Eschmeyer WN, Fricke R, van der Laan R, editors. Catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science ; 2016 [updated 2017 Mar 01; cited 2017 Mar 29]. Available from: Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Publication Dates
-
Publication in this collection
2017
History
-
Received
03 Feb 2017 -
Accepted
01 Sept 2017