ABSTRACT
A new species of Moenkhausia is described from tributaries of the upper rio Sepotuba, Paraguay basin, Brazil. The new species is distinguished from its congeners by a combination of characters, including an inconspicuous oval-shaped vertically elongated humeral blotch, extending horizontally from third through five lateral-line scales, and vertically from third row above lateral line to first row below it, followed by a diffuse field of dark chromatophores in the flank, combined with a well-defined dark line at the base of the anal fin. Furthermore, the phylogenetic position of the new species is presented based on molecular data, showing a close relationship among species of Moenkhausia and Hemigrammus that have a well-defined dark line at the base of the anal fin. Until this moment, this species is only known from in the upper rio Sepotuba basin.
Keywords:
Hemigrammus; Molecular phylogeny; Neotropical tetras; Taxonomy
RESUMO
Uma nova espécie de Moenkhausia é descrita nos afluentes do rio Sepotuba, bacia do Paraguai, no Brasil. A nova espécie se distingue dos seus congêneres por uma combinação de caracteres, incluindo uma mancha umeral discreta de forma oval, alongada verticalmente, que se estende horizontalmente da terceira a quinta escamas da linha lateral e, verticalmente, da terceira fila de escamas acima da linha lateral até a primeira fila abaixo da linha lateral; seguida por escassos cromatóforos no flanco, combinado com uma linha escura bem definida na base da nadadeira anal. Além disso, a posição filogenética da nova espécie é apresentada com base em dados moleculares, mostrando um relacionamento próximo entre as espécies de Moenkhausia e Hemigrammus que possuem uma linha escura bem definida na base da nadadeira anal. Até o momento, essa espécie é conhecida apenas da bacia do Alto Sepotuba.
Palavras-chave:
Filogenia Molecular; Hemigrammus; Taxonomia; Tetras Neotropical
Introduction
The genus Moenkhausia Eigenmann basically differs from Hemigrammus Gill by possessing a completely pored lateral line (vs. an incompletely pored lateral line in the latter) (Eigenmann, Myers, 1917Eigenmann CH, Myers GS.The American Characidae. Part 1. Mem Mus Comp Zool . 1917; 43:1-102.; Géry, 1977Géry J. Characoids of the world. Neptune City: T. F. H. Publications; 1977.). However, several species have been described on Moenkhausia despite the fact of possessing incomplete or interrupted lateral lines (e.g. M. pyrophthalmaCosta, 1994Costa WJEM. Description of two new species of the genus Moenkhausia (Characiformes: Characidae) from the central Brazil. Zool Anz. 1994; 232(1-2):21-29.; M. diktyotaLima & Toledo-Piza, 2001Lima FCT, Toledo-Piza M. New species of Moenkhausia (Characiformes: Characidae) from the Rio Negro of Brazil. Copeia [serial on the Internet]. 2001; 2001(4):1058-63. Available from: http://dx.doi.org/10.1590/1982-0224-20150052
http://dx.doi.org/10.1590/1982-0224-2015...
; M. forestiiBenine, Mariguela & Oliveira, 2009Benine RC, Mariguela TC, Oliveira C. New species of Moenkhausia Eigenmann, 1903 (Characiformes: Characidae) with comments on the Moenkhausia oligolepis species complex. Neotrop Ichthyol [serial on the Internet]. 2009; 7(2):161-8. Available from: http://dx.doi.org/10.1590/S1679-62252009000200005
http://dx.doi.org/10.1590/S1679-62252009...
).
The non-monophyletic condition of these genera has long been discussed by several authors since their proposition (e.g.Géry, 1977Géry J. Characoids of the world. Neptune City: T. F. H. Publications; 1977.; Fink, 1979Fink WL. A new species of Moenkhausia from the Mato Grosso region of Brazil (Pisces: Characidae). Breviora. 1979; 450:12p.; Costa, 1994Costa WJEM. Description of two new species of the genus Moenkhausia (Characiformes: Characidae) from the central Brazil. Zool Anz. 1994; 232(1-2):21-29.; Weitzman, Palmer, 1997Weitzman SH, Palmer L. A new species of Hyphessobrycon (Teleostei: Characidae) from the Neblina region of Venezuela and Brazil, with comments on the putative ‘rosy tetra clade’. Ichthyol Explor Freshw . 1997; 7(3):209-42.; Lucena, Lucena, 1999Lucena ZMS, Lucena CAS. Moenkhausia tergimacula, a new species from the upper rio Tocantins, Brazil (Osteichthyes: Characidae). Ichthyol Explor Freshw . 1999; 10(3):231-36.; Lima, Toledo-Piza, 2001Lima FCT, Toledo-Piza M. New species of Moenkhausia (Characiformes: Characidae) from the Rio Negro of Brazil. Copeia [serial on the Internet]. 2001; 2001(4):1058-63. Available from: http://dx.doi.org/10.1590/1982-0224-20150052
http://dx.doi.org/10.1590/1982-0224-2015...
; Benine, 2002Benine RC. Moenkhausia levidorsa a new species from Rio Aripuanã, Amazon Basin, Brazil (Characiformes: Characidae). Ichthyol Explor Freshw. 2002; 13(4):289-94.; 2004Benine RC. Análise filogenética do gênero Moenkhausia (Characiformes: Characidae) com uma revisão dos táxons do alto Rio Paraná [PhD thesis].São Paulo: Universidade Estadual Paulista; 2004.; Benine et al., 2009Benine RC, Mariguela TC, Oliveira C. New species of Moenkhausia Eigenmann, 1903 (Characiformes: Characidae) with comments on the Moenkhausia oligolepis species complex. Neotrop Ichthyol [serial on the Internet]. 2009; 7(2):161-8. Available from: http://dx.doi.org/10.1590/S1679-62252009000200005
http://dx.doi.org/10.1590/S1679-62252009...
; Lima, Sousa, 2009Lima FCT, Sousa LM. A new species of Hemigrammus from the upper rio Negro basin, Brazil, with comments on the presence and arrangement of anal-fin hooks in Hemigrammus and related genera (Ostariophysi: Characiformes: Characidae). Aqua . 2009; 15(3):153-69.; Lima et al., 2007Lima FCT, Britski HA, Machado FA. A new Moenkhausia (Characiformes: Characidae) from central Brazil, with comments on the area relationship between the upper rio Tapajós and upper rio Paraguai systems. Aqua. 2007; 13(2):45-54.; Ota et al., 2014Ota RP, Lima FCT, Pavanelli CS. A new species of Hemigrammus Gill, 1858 (Characiformes: Characidae) from the rio Madeira and rio Paraguai basins, with a redescription of H. lunatus. Neotrop Ichthyol [serial on the Internet]. 2014; 12(2):265-79. Available from: http://dx.doi.org/10.1590/1982-0224-20130176
http://dx.doi.org/10.1590/1982-0224-2013...
) even though not based on tested hypothesis of phylogenetic relationships. In an attempt to test the monophyly of the Characidae, Mirande (2010Mirande JM. Phylogeny of the family Characidae (Teleostei: Characiformes) from characters to taxonomy. Neotrop Ichthyol [serial on the Internet]. 2010; 8(3):385-568. Available from: http://dx.doi.org/10.1590/S1679-62252010000300001
http://dx.doi.org/10.1590/S1679-62252010...
) and Oliveira et al. (2011Oliveira C, Avelino GS, Abe KT, Mariguela TC, Benine RC, Ortí G, Vari RP, Castro RMC. Phylogenetic relationships within the speciose family Characidae (Teleostei: Ostariophysi: Characiformes) based on multilocus analysis and extensive ingroup sampling. BMC Evol Biol [serial on the Internet]. 2011; 11(1):275. Available from: https://doi.org/10.1186/1471-2148-11-275
https://doi.org/10.1186/1471-2148-11-275...
) found Moenkhausia xinguensis (Steindachner, 1882) and Hemigrammus unilineatus (Gill, 1858), the type species of both genera, to be more closely related to species of Hasemania, Hyphessobrycon, Paracheirodon, Pristella, and Thayeria, evidencing their non-monophyletic condition.
Mariguela et al. (2013Mariguela TC, Benine RC, Abe KT, Avelino GS, Oliveira C. Molecular phylogeny of Moenkhausia (Characidae) inferred from mitochondrial and nuclear DNA evidence. J Zoolog Syst Evol Res [serial on the Internet]. 2013; 51(4):327-32. Available from: https://doi.org/10.1111/jzs.12025
https://doi.org/10.1111/jzs.12025...
) presented a phylogenetic analysis including 29 species out of the 91 current valid species of Moenkhausia (Eschmeyer et al., 2017Eschmeyer WN, Fricke R, van der Laan R, editors. Catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2017 [Updated 2017 June 30, cited 2017 July 01]. Avaliable from: Avaliable from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
http://researcharchive.calacademy.org/re...
). Their results showed that representatives of this genus appeared distributed in five different clades related to genera such as Bario, Hemigrammus, Hasemania, Nematocharax, Aphyodite, Parecbasis, and also with members of the subfamily Stethaprioninae.
Despite of these recent efforts in trying to identify the sister species of Moenkhausia xinguensis and Hemigrammus unilineatus, and propose a phylogenetic definition for both genera, the low number of taxa included in those analyses, along with the lack of understanding of closely related, species-rich genera such as Astyanax and Hyphessobrycon, has precluded a more consistent and useful definition for these and other characid genera.
Collecting efforts in headwater streams of the upper rio Sepotuba, rio Paraguay basin, revealed an undescribed species of small characin that, despite of its similarity to species of Hemigrammus, such as H. lunatus, would better fit to the artificial and traditional definition of Moenkhausia, since it has a completely pored lateral line. Herein, we present a phylogenetic analysis based on molecular data to infer and discuss its phylogenetic relationships within Characidae and formally describe this new species.
Material and Methods
Morphological data. Counts and measurements followed Fink, Weitzman (1974Fink WL, Weitzman SH. The so-called Cheirodontin fishes of Central America with descriptions of two new species (Pisces: Characidae). Washington (DC): Smithsonian Institution Press; 1974. (Smithsonian contributions to Zoology; No. 172).), except for counts of scale rows, which follow Lima et al. (2007Lima FCT, Britski HA, Machado FA. A new Moenkhausia (Characiformes: Characidae) from central Brazil, with comments on the area relationship between the upper rio Tapajós and upper rio Paraguai systems. Aqua. 2007; 13(2):45-54.) and with the addition of pelvic-fin origin to anal-fin origin measured at origin of pelvic-fin through the anal-fin origin.
Measurements were taken point to point with a digital caliper on the left side of specimens whenever possible (precision of 0.1 mm). All measurements are expressed as percentage of standard length (SL) or head length (HL). Values in the parentheses indicate the number of specimens with a particular count and an asterisk indicates values of the holotype. Vertebrae of the Weberian apparatus were counted as four elements and the fused PU1+U1 as a single element. Vertebrae, supraneural counts and gill-rakers of first arch were taken from seven cleared and stained (cs) specimens prepared following the method of Taylor, Van Dyke (1985Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage. Cybium. 1985; 9(2):107-20.).
Institutional abbreviations used are as follows: Laboratório de Biologia e Genética de Peixes (LBP), Universidade Estadual Paulista, Botucatu, Brazil; Museo de Ciencias Naturales de Guanare (MCNG), Guanare, Venezuela; Museum of Comparative Zoology (MCZ), Harvard, United States, Museu de Zoologia da Universidade de São Paulo (MZUSP), São Paulo, Brazil, and Museu de Zoologia da Universidade Estadual de Londrina (MZUEL), Londrina, Brazil; Naturhistorisches Museum Vienna (NMW), Vienna, Austria.
Molecular data. The present study included 30 species currently allocated in Moenkhausia, including the new species. As a framework for the selection of species to be included in our analysis we follow the results presented in Oliveira et al. (2011Oliveira C, Avelino GS, Abe KT, Mariguela TC, Benine RC, Ortí G, Vari RP, Castro RMC. Phylogenetic relationships within the speciose family Characidae (Teleostei: Ostariophysi: Characiformes) based on multilocus analysis and extensive ingroup sampling. BMC Evol Biol [serial on the Internet]. 2011; 11(1):275. Available from: https://doi.org/10.1186/1471-2148-11-275
https://doi.org/10.1186/1471-2148-11-275...
), selecting representatives of their node 31, which includes the Acestrorhynchidae, Bryconidae, Chalceidae, Characidae, Gasteropelecidae, Iguanodectidae, and Triportheidae.
Molecular methods. Total DNA was extracted from ethanol preserved muscle samples using DNeasy Tissue Extraction Kit (Qiagen), following manufacturer’s instructions. Partial sequences of the genes 16SrRNA, Cytochrome b (Cytb), recombination activating gene 1 (Rag1), recombination activating gene 2 (Rag2), and Myosin, heavy chain 6, cardiac muscle, alpha (Myh6) were amplified by polymerase chain reaction (PCR) with the same primers utilized by Oliveira et al. (2011Oliveira C, Avelino GS, Abe KT, Mariguela TC, Benine RC, Ortí G, Vari RP, Castro RMC. Phylogenetic relationships within the speciose family Characidae (Teleostei: Ostariophysi: Characiformes) based on multilocus analysis and extensive ingroup sampling. BMC Evol Biol [serial on the Internet]. 2011; 11(1):275. Available from: https://doi.org/10.1186/1471-2148-11-275
https://doi.org/10.1186/1471-2148-11-275...
). Amplifications were performed in a total volume of 25 ml with 2.5 ml of 10X buffer (10mM Tris-HCL + 15mM MgCl2 buffer), 0.5 ml MgCl2, 0.5 ml each primer (5 mM); 0.4 ml dNTPs (200 nM of each), 0.2 ml Taq Platinum polymerase (Invitrogen), 1 ml template DNA (10-50ng) and 19.4 ml ddH2O. The thermo-cycler profile used for the fragments 16SrRNA and Cyt b was with 35 cycles and annealing temperature of 50-55oC. Nested-PCR was used to amplify the nuclear genes rag1, rag2, and Myh6. Conditions for amplification of these genes for both rounds of PCR used 15 cycles with annealing temperature at 56oC followed by 15 cycles with annealing temperature at 54oC. PCR products were purified using ExoSap-IT® (USB Corporation), sequenced using the “Big DyeTM Terminator v 3.1 Cycle Sequencing Ready Reaction Kit” (Applied Biosystems), purified again by ethanol precipitation and loaded on an automatic sequencer 3130-Genetic Analyzer (Applied Biosystems) at Instituto de Biociências, Universidade Estadual Paulista, Botucatu, São Paulo, Brazil.
Alignment and phylogenetic analyses. Contigs were assembled and edited in Geneious v 5.4 software (Kearse et al., 2012Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S et al. Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics [serial on the Internet]. 2012; 28(12):1647-49. Available from: https://doi.org/10.1093/bioinformatics/bts199
https://doi.org/10.1093/bioinformatics/b...
). Sequences were independently aligned using the muscle algorithm under default parameters (Edgar, 2004Edgar RC. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res [serial on the Internet]. 2004; 32(5):1792-97. Available from: https://doi.org/10.1093/nar/gkh340
https://doi.org/10.1093/nar/gkh340...
), and alignments were inspected for any problems. The first alignments were initially analyzed by neighbour-joining (NJ) using MEGA 7.0 software (Kumar et al., 2016Kumar S, Stecher G, Tamura K. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol Biol Evol [serial on the Internet]. 2016; 33(7):1870-74. Available from: https://doi.org/10.1093/molbev/msw054
https://doi.org/10.1093/molbev/msw054...
) to control potential sequencing errors. The sequences were translated using Geneious v 5.4 (Kearse et al., 2012Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S et al. Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics [serial on the Internet]. 2012; 28(12):1647-49. Available from: https://doi.org/10.1093/bioinformatics/bts199
https://doi.org/10.1093/bioinformatics/b...
) to check for the unexpected occurrence of stop codons. PartitionFinder v1.1.1 (Lanfear et al., 2012Lanfear R, Calcott B, Ho SYW, Guindon S. PartitionFinder: combined selection of partitioning schemes and substitution models for phylogenetic analyses. Mol Biol Evol [serial on the Internet]. 2012; 29(6):1695-701. Available from: https://doi.org/10.1093/molbev/mss020
https://doi.org/10.1093/molbev/mss020...
) was used to determine codon-specific models of molecular evolution for each gene under the Bayesian information criterion (BIC). A generalized time reversible model with rate heterogeneity of the remainder being model by gamma distribution with a proportion of invariable sites (GTR+Gamma+I) was identified as the best model of molecular evolution for the first, second and third codon position. Phylogenetic hypotheses were inferred from the data set with maximum-likelihood (ML), maximum parsimony (MP) and Bayesian inference (B). Maximum-likelihood (ML) analysis was performed using RAxML Web-Servers Black-Box (Stamatakis et al., 2008Stamatakis A, Hoover P, Rougemont J. A rapid bootstrap algorithm for the RAxML web servers. Syst Biol [serial on the Internet]. 2008; 57(5):758-71. Available from: https://doi.org/10.1080/10635150802429642
https://doi.org/10.1080/1063515080242964...
) with a mixed partition approach, with GTR + G + I as the model. Partitioned analysis was performed, with the data set divided into three sections corresponding to the first, second and third positions of the gene. Random starting trees were used for each independent ML tree search, and all other parameters were set at default values. Topological robustness was investigated using 1000 nonparametric bootstrap pseudoreplicates (Felsenstein, 1985Felsenstein J. Confidence limits on phylogenies: an approach using the bootstrap. Evolution. 1985; 39(4):783-91.). Maximum parsimony analyses were conducted with PAUP* 4.0b10 (Swofford, 2002Swofford DL. PAUP*. Phylogenetic Analysis Using Parsimony (*and other Methods) Version 4b10. Sunderland (MA): Sinauer Associates; 2002.). Heuristic searches were performed with random addition replicates (minimally 1000) and TBR branch swapping. Character transformations were equally weighted and branches with a maximum length of zero were collapsed. Gaps were treated as missing data. Clade robustness was assessed using 1000 bootstrap pseudoreplicates with the same parameters as above (Felsenstein, 1985Felsenstein J. Confidence limits on phylogenies: an approach using the bootstrap. Evolution. 1985; 39(4):783-91.). Phylogenetic analysis using a partitioned Bayesian approach was conducted in MrBayes 3.1.2 (Ronquist, Huelsenbeck, 2003Ronquist F, Huelsenbeck JP. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics. 2003; 19(12):1572-74.) with the models and partitions determined as the best scheme according PartitionFinder (Lanfear et al., 2012Lanfear R, Calcott B, Ho SYW, Guindon S. PartitionFinder: combined selection of partitioning schemes and substitution models for phylogenetic analyses. Mol Biol Evol [serial on the Internet]. 2012; 29(6):1695-701. Available from: https://doi.org/10.1093/molbev/mss020
https://doi.org/10.1093/molbev/mss020...
). MrBayes was configured to run for 10 million generations using eight chains (nchain = 8, being two parallel runs with one cold and 7 hot chains each; temperature parameter set to default). Results were analyzed in the Tracer v. 1.6 (Rambaut et al., 2014Rambaut A, Suchard MA, Xie D, Drummond AJ. Tracer version 1.6. [Computer software-Internet]. Edinburgh: University of Edinburgh ; 2014. Available from: http://beast.bio.ed.ac.uk/Tracer.
http://beast.bio.ed.ac.uk/Tracer...
), TreeAnnotator v. 1.8.0 (available as part of the BEAST package, Drummond et al., 2012Drummond AJ, Suchard MA, Xie D, Rambaut A. Bayesian phylogenetics with BEAUti and the BEAST 1.7. Mol Biol Evol [serial on the Internet]. 2012; 29(8):1969-73. Available from: http://dx.doi.org/10.1093/molbev/mss075
http://dx.doi.org/10.1093/molbev/mss075...
) and FigTree v. 1.4.2 (Rambaut, Drummond, 2012Rambaut A, Drummond AJ. Treeannotator version 1. 4.2. [Computer software-Internet]. Edinburgh: University of Edinburgh; 2012. Available from: http://beast.bio.ed.ac.uk/TreeAnnotator.
http://beast.bio.ed.ac.uk/TreeAnnotator...
) software programs, after deleting burn-in trees, which were approximately 30%.
Results
Moenkhausia flava, new species
urn:lsid:zoobank.org:act:FC435ADB-E0BC-4C44-B707-5B5F4852FC79
Holotype. MZUSP 123719, 34.0 mm SL, Brazil: Mato Grosso State, Tangará da Serra, Córrego São Jorge, tributary of rio Sepotuba, rio Paraguay basin, 14°27’24.9” S 57°34’32.7” W, 12 Nov 2009, R. Britzke, T. S. Zanini & W. P. Troy.
Paratypes. LBP 9031, 17 (7 cs), 20.9-36.8 mm SL, collected with holotype. LBP 18414, 15, 29-38.3 mm SL, same locality as holotype, 2 Apr 2013, W. P. Troy. MZUSP 123720, 10, 24.9-35.2 mm SL, same locality as holotype, 2 Apr 2013, W. P. Troy. MZUEL 8139, 3, 19.9-23.5 mm SL, Brazil: Mato Grosso State, Tangará da Serra, Ribeirão do Sapo, tributary of rio Sepotuba, rio Paraguay basin, 14°33’24.6” S 57°48’45.8” W, 29 Aug 2013, J. L. O. Birindelli, A. Claro-García, F. Assega & E. Santana.
Non types. Brazil: Mato Grosso State, Tangará da Serra: LBP 8418, 2, 34.9-39.4 mm SL, Córrego São Jorge, tributary of rio Sepotuba, rio Paraguay basin, 14°27’26.3” S 57°34’34” W. LBP 8406, 1, 23.4 mm SL, Riacho Águas Claras, tributary of rio Sepotuba, rio Paraguay basin, 14°21’03.2” S 57°33’07.2” W. Non-types also include dissected, alcohol-fixed, and poorly preserved specimens.
Moenkhausia flava , holotype, MZUSP 123719, 34.0 mm SL, Brazil, Mato Grosso, small stream tributary of rio Sepotuba.
Moenkhausia flava , paratype, LBP 18414, 34.1 mm SL. Overall coloration darker, yellowish tan, where most of dark pigments are preserved.
Map showing the localities of Moenkhausia flava. Red star represents the type locality. Black square represents the Salto das Nuvens fall and the white square represents Salto Maciel fall.
Moenkhausia flava . LBP 9031, 31.9 mm SL, paratype: right premaxilla (top), maxilla (middle), and dentary (bottom). Scale bar = 0.5 mm.
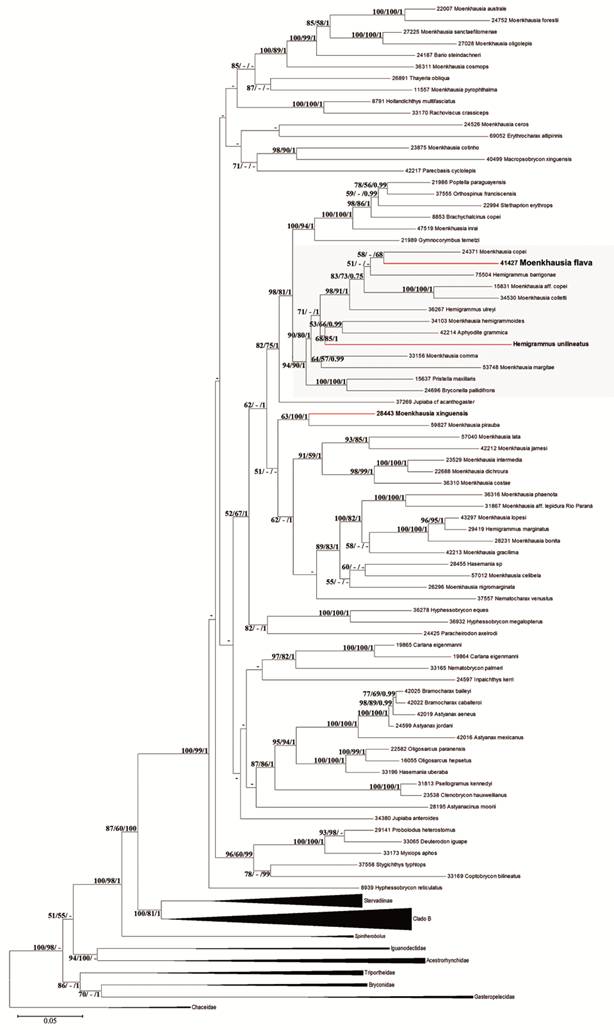
Maximum likelihood phylogenetic tree based on Partial sequences of two mitochondrial genes and three nuclear genes. The series of three numbers (e.g. 100/94/1) at each of the main nodes represents the percentage of bootstrap support obtained by Maximum Likelihood (ML), the percentage of bootstrap support obtained by the Maximum Parsimony (MP) analysis and the posterior probability for that split obtained in Bayesian analysis (B),respectively (1000 bootstrap replicates). Dashes represent values < 50% (ML, MP) or <0.5 (B).
Diagnosis. Moenkhausia flava is distinguished from all congeners and members of closely related genera, except Hemigrammus barrigonae Eigenmann & Henn, 1914; Hemigrammus lunatus Durbin, 1918; Hemigrammus machadoiOta, Lima & Pavanelli, 2014Ota RP, Lima FCT, Pavanelli CS. A new species of Hemigrammus Gill, 1858 (Characiformes: Characidae) from the rio Madeira and rio Paraguai basins, with a redescription of H. lunatus. Neotrop Ichthyol [serial on the Internet]. 2014; 12(2):265-79. Available from: http://dx.doi.org/10.1590/1982-0224-20130176
http://dx.doi.org/10.1590/1982-0224-2013...
; Hemigrammus ulreyi (Boulenger, 1895); Moenkhausia collettii (Steindachner, 1882); Moenkhausia conspicuaSoares & Bührnheim, 2016Soares IM, Bührnheim CM. A new species of Moenkhausia Eigenmann, 1903 (Characiformes: Characidae) from Amazon basin, Brazil. Zootaxa [serial on the Internet]. 2016; 4208(4):392-400. DOI: 10.11646/zootaxa.4208.4.6.
https://doi.org/10.11646/zootaxa.4208.4....
; Moenkhausia copei (Steindachner, 1882) and Moenkhausia venerei Petrolli, Azevedo-Santos & Benine, 2016, by the presence of a well-defined dark line at the base of the anal fin. Moenkhausia flava can be easily distinguished from H. barrigonae, H. ulreyi, M. conspicua and M. venerei by the presence of a midlateral dark thin stripe little evident on posterior half of the body (vs. conspicuous dark and well-defined longitudinal midlateral dark stripe from humeral region until caudal peduncle). Moenkhausia flava is quite similar with H. barrigonae and M. conspicua by sharing overall body shape and similar color pattern. In addition, some specimens of H. barrigonae also present complete lateral line (see Géry, 1977Géry J. Characoids of the world. Neptune City: T. F. H. Publications; 1977.:503; Soares, Bührnheim, 2016Soares IM, Bührnheim CM. A new species of Moenkhausia Eigenmann, 1903 (Characiformes: Characidae) from Amazon basin, Brazil. Zootaxa [serial on the Internet]. 2016; 4208(4):392-400. DOI: 10.11646/zootaxa.4208.4.6.
https://doi.org/10.11646/zootaxa.4208.4....
: 398), similar to the new species and M. conspicua. Moenkhausia flava differs from H. barrigonae and M. conspicua by presenting inner having premaxillary and dentary teeth pentacuspid (vs. inner premaxillary and dentary teeth heptacuspid in both species). Additionally, differs from H. barrigonae by larger body depth (31.0-42.6% SL, mean = 35.8% SL vs. 30.4-34.8% SL, mean = 32% SL); and differs of M. conspicua by the dorsal-fin length (24.3-31.3% vs. 31.2-36.8% SL, respectively), anal-fin length (14.8-22.1% vs. 22.6-27.1% SL, respectively) and orbital diameter (34.8-43.4% vs. 44.9-56.0% HL, respectively). Moenkhausia flava can be easily distinguished from H. machadoi and H. lunatus by an inconspicuous oval-shaped vertically elongated humeral blotch, extending horizontally from third through five lateral-line scales, and vertically from third row above lateral line to first row below it (vs. a conspicuous vertically elongated dark humeral blotch, extending horizontally from second through sixth lateral-line scales, and vertically from third row above lateral line to first row below it in H. machadoi; and a small roundish humeral blotch in H. lunatus). The new species differs from M. collettii and M. copei by having six longitudinal rows of scales above lateral line (vs. five longitudinal rows of scales). Additionally, it is distinguished from M. copei by a greater number of branched anal-fin rays (20-23 vs. 15-17 in M. copei) and distinguished from H. machadoi, H. lunatus and M. collettii by a three-scale deep band of sparse, scattered dark chromatophores extending along midlateral body (vs. a line of concentrated chromatophores along midlateral body).
Description. Morphometric data for Moenkhausia flava summarized in Tab. 1. Body relatively compressed and elongated, moderately high. Greatest body depth just before the origin of dorsal fin. Snout profile convex. Dorsal profile of head straight or slightly convex. Dorsal profile of body convex from posterior tip of supraoccipital spine to dorsal-fin origin, slightly convex and posteroventrally slanted along dorsal-fin base, slightly convex from posterior terminus of dorsal-fin base to end of adipose-fin origin, and concave along caudal peduncle. Ventral body profile convex from tip of lower jaw to caudal-peduncle origin, slightly concave along caudal peduncle. Prepelvic region transversally flattened, more so proximal to pelvic-fin insertion. Postpelvic region transversally flattened proximal to pelvic-fin insertion, becoming somewhat obtuse toward anal-fin origin. Mouth terminal. Maxilla slightly beyond vertical through anterior margin of orbit. Premaxillary teeth in two rows; outer teeth row with 3(19), 4*(21) tricuspid teeth, midcentral cusps longer than remaining cusps; inner teeth row with 4(2), 5*(38) tetra- to pentacuspid teeth, midcentral cusps longer than remaining cusps. Maxilla with 2(21), 3*(19) tricuspid teeth. Dentary with 4(5), 5*(27), 6(8) pentacuspid teeth, followed by a series of small teeth, with 1-3 cusps (Fig. 6). Dorsal profile of head convex from upper lip to vertical through anterior nostril. Suparaoccipital process short, its tip not reaching vertical through posterior margin of opercle. Dorsal-fin rays ii,9(30). Pectoral-fin rays i,10(30), i,11*(10). Tip of pectoral fin not reaching vertical through pelvic-fin insertion. Adipose fin well developed. Pelvic-fin rays i,7(30), when adpressed, its tip reaching first anal-fin ray in a few specimens. Anal-fin rays iv, 20(3), 21*(10), 22(12), 23(3). Caudal fin forked. Principal caudal-fin rays i,17,i. Scales cycloid, with few radii along posterior border. Lateral-line completely pored. Lateral line scales 33(4), 34(17), 35*(11), 36(1). Scale rows between dorsal-fin origin and lateral-line 6; scale rows between lateral-line and pelvic-fin origin 4. Circumpeduncular scale rows 14*(32). Predorsal scales 10*(30). Scale sheath along anal-fin base in a single series of 4 scales, extending from the first to fifth branched anal-fin ray. First gill arch with 9*(18) gill rakers on ventral limb and 5*(18) on dorsal limb.
Morphometrics data of holotype and paratypes of Moenkhausia flava. Standard length (SL) is expressed in mm, all other measurements are expressed as percentages of SL, except for subunits of head that are expressed as percentages of head length (HL). N=46.
Sexual dimorphism. Adult males with small hooks on the last unbranched and anterior four branched anal-fin rays. Also, found on the first and second pelvic-fin rays branched. In both fins, there are four to seven hooks per fin ray, located on the distal segments.
Color in alcohol. Humeral region with an inconspicuous oval-shaped vertically elongated humeral blotch located on second to fourth lateral line scales, extending from 3 horizontal series of scales above lateral to the series of scales immediately below the lateral line. Dark chromatophores scattered on infraorbitals and opercle, longitudinal dark lighter stripe along on the eye (Fig. 2). A dark thin stripe extending along horizontal septum, from humeral region to caudal peduncle, more evident on posterior half of the body. A three-scale deep band of sparse, scattered dark chromatophores extending along midlateral body.
Dorsal fin rays with few dispersed chromatophores, more concentrated on anterior half. Anal-fin rays with few dispersed chromatophores, more concentrated along its proximal and distal extension, resulting in a lighter medial area. Tip of anterior anal-fin rays densely pigmented by dark chromatophores resulting in a dark dash in this area. Paired fins hyaline with scattered dark pigmentation, more concentrated on unbranched rays. Caudal fin with a narrow field of dark chromatophores on its distal margin. Conspicuous dark line at anal-fin base. All fins hyaline with few dispersed chromatophores. Anterior rays of the anal fin and base of caudal-fin lobe present orange pigmentation in freshly preserved specimens.
Color in life. Body general color pattern pale yellowish. Dorsal region olive. Abdomen whitish to light yellow. Pelvic fin and adipose fin with yellow-orangish coloration. Dorsal fin and caudal fin with orange-reddish coloration. Anal fin with first rays with orange-reddish coloration and the remaining rays with hyaline coloration. A conspicuous dark line at the base of the anal fin. Pectoral fin yellowish (Fig. 4).
Ecological notes. All specimens of Moenkhausia flava were collected along the margin in semi-lentic stretches of a shallow river with clear water, with sand and scattered small stones on the bottom, aquatic vegetation (Echinodorus sp., Eleocharis sp., Hygrophila sp. and Mayaca fluviatilis), and riparian vegetation (Fig. 3). Species collected syntopically were Hyphessobrycon hebertaxelrodi Géry, 1961, Hyphessobrycon vilmae, Pyrrhulina australisEigenmann & Kennedy, 1903Eigenmann CH. New genera of South American fresh-water fishes, and new names for old genera. Smithson Misc Collect. 1903; 45(12):144-48., Characidium aff. zebra Eigenmann, 1909, Corydoras aeneus (Gill, 1858), Hypostomus sp., Brachyhypopomus sp., Gymnotus inaequilabiatus (Valenciennes, 1839), and Aequidens rondoni (Miranda Ribeiro, 1918).
Geographic distribution. Only known from tributaries of the upper rio Sepotuba, above the waterfalls Salto das Nuvens and Salto Maciel, rio Paraguay basin, Mato Grosso State, Brazil (Fig. 5).
Etymology. The name of species is derived from Latin flavus, meaning yellow. The name refers to the yellowish color pattern of the body in live specimens. An adjective.
Phylogenetic analysis. Partial sequences of two mitochondrial (16SrRNA and Cytb) and three nuclear genes (Myh6, Rag1 and Rag2) were obtained for 2 species of Hemigrammus and 29 species of Moenkhausia from the final matrix by Mariguela et al. (2013Mariguela TC, Benine RC, Abe KT, Avelino GS, Oliveira C. Molecular phylogeny of Moenkhausia (Characidae) inferred from mitochondrial and nuclear DNA evidence. J Zoolog Syst Evol Res [serial on the Internet]. 2013; 51(4):327-32. Available from: https://doi.org/10.1111/jzs.12025
https://doi.org/10.1111/jzs.12025...
) deposited in TreeBase (treebase.org) under access number 13922, plus the species Hemigrammus barrigonae, H. unilineatus and Moenkhausia flava. The combined sequence data resulted in a matrix with 4,640 base pairs (bp), out of which 1,911 were conserved, 2,726 were variable and 2,196 were information parsimony. The estimated index of substitution saturation (Iss) performed in DAMBE 5.2.31 (Xia, Xie, 2001Xia X, Xie Z. DAMBE: Data analysis in molecular biology and evolution. J Hered. 2001; 92(4):371-73.) showed that the data were not saturated (i.e., Iss.c value greater than Iss). In our analysis, Moenkhausia flava is more closely related to Moenkhausia collettii, M. copei, Hemigrammus barrigonae and H. ulreyi (Fig. 7). The new species was recovered within the clade 2 (sensuMariguela et al., 2013Mariguela TC, Benine RC, Abe KT, Avelino GS, Oliveira C. Molecular phylogeny of Moenkhausia (Characidae) inferred from mitochondrial and nuclear DNA evidence. J Zoolog Syst Evol Res [serial on the Internet]. 2013; 51(4):327-32. Available from: https://doi.org/10.1111/jzs.12025
https://doi.org/10.1111/jzs.12025...
), also composed of Aphyodite grammica Eigenmann, 1912; Bryconella pallidifrons (Fowler, 1946); Hemigrammus barrigonae, H. ulreyi, H. unilineatus (Gill, 1858); Moenkhausia comma Eigenmann, 1908; M. copei, M. aff. copei, M. collettii, M. hemigrammoides Géry, 1965; M. margitae Zarske & Géry, 2001; Pristella maxilaris (Ulrey, 1894), plus Moenkhausia flava. In this analysis, the groups proposed by Géry (1977Géry J. Characoids of the world. Neptune City: T. F. H. Publications; 1977.), reinforce that overall similarities are not sufficient to distinguish groups in Moenkhausia. However, few features seem to be still useful to designate natural groups within Moenkhausia and Hemigrammus (see discussion).
Conservation status. Moenkhausia flava is known exclusively from tributaries of the upper rio Sepotuba, above the waterfalls Salto das Nuvens and Salto Maciel, rio Paraguay basin. Because no specific threats have been detected, the species can be categorized as Least Concern (LC) according to IUCN criteria (IUCN, 2016International Union for Conservation of Nature (IUCN). Standards and Petitions Subcommittee. Guidelines for using the IUCN Red List Categories and Criteria. 2016; Version 12. Prepared by the Standards and Petitions Subcommittee. Available from: Available from: http://cmsdocs.s3.amazonaws.com/RedListGuidelines.pdf
http://cmsdocs.s3.amazonaws.com/RedListG...
), however, we emphasize the apparently narrowly distribution of the species, and strongly encourage the preservation of those environments.
Discussion
The taxonomic distinction between Moenkhausia and Hemigrammus is currently based solely on a single character state: completely pored lateral line vs. incompletely pored lateral line respectively, as proposed by Eigenmann (1903Eigenmann CH. New genera of South American fresh-water fishes, and new names for old genera. Smithson Misc Collect. 1903; 45(12):144-48.), Eigenmann, Myers (1917Eigenmann CH, Myers GS.The American Characidae. Part 1. Mem Mus Comp Zool . 1917; 43:1-102.) and Durbin (in Eigenmann, 1918Eigenmann CH. The American Characidae. Part 2. Mem Mus Comp Zool. 1918; 43:103-208.). Moenkhausia flava possesses a completely pored lateral line, but a body pattern similar to species of the genus Hemigrammus.
Recent phylogenetic hypothesis including species of both Moenkhausia and Hemigrammus (Mirande, 2009Mirande JM. Weighted parsimony phylogeny of the family Characidae (Teleostei: Characiformes). Cladistics [serial on the Internet]. 2009; 25(6):574-613. Available from: https://doi.org/10.1111/j.1096-0031.2009.00262.x
https://doi.org/10.1111/j.1096-0031.2009...
, 2010Mirande JM. Phylogeny of the family Characidae (Teleostei: Characiformes) from characters to taxonomy. Neotrop Ichthyol [serial on the Internet]. 2010; 8(3):385-568. Available from: http://dx.doi.org/10.1590/S1679-62252010000300001
http://dx.doi.org/10.1590/S1679-62252010...
; Oliveira et al., 2011Oliveira C, Avelino GS, Abe KT, Mariguela TC, Benine RC, Ortí G, Vari RP, Castro RMC. Phylogenetic relationships within the speciose family Characidae (Teleostei: Ostariophysi: Characiformes) based on multilocus analysis and extensive ingroup sampling. BMC Evol Biol [serial on the Internet]. 2011; 11(1):275. Available from: https://doi.org/10.1186/1471-2148-11-275
https://doi.org/10.1186/1471-2148-11-275...
; Mariguela et al., 2013Mariguela TC, Benine RC, Abe KT, Avelino GS, Oliveira C. Molecular phylogeny of Moenkhausia (Characidae) inferred from mitochondrial and nuclear DNA evidence. J Zoolog Syst Evol Res [serial on the Internet]. 2013; 51(4):327-32. Available from: https://doi.org/10.1111/jzs.12025
https://doi.org/10.1111/jzs.12025...
) showed the non-monophyletic condition of these genera. In Mariguela et al. (2013Mariguela TC, Benine RC, Abe KT, Avelino GS, Oliveira C. Molecular phylogeny of Moenkhausia (Characidae) inferred from mitochondrial and nuclear DNA evidence. J Zoolog Syst Evol Res [serial on the Internet]. 2013; 51(4):327-32. Available from: https://doi.org/10.1111/jzs.12025
https://doi.org/10.1111/jzs.12025...
), 50% (14 species) of the analyzed species of Moenkhausia (29 in total) grouped with its type species, Moenkhausia xinguensis. It is noteworthy that this group also included the incomplete lateral-lined Hemigramus marginatus Ellis, 1911; Hasemania sp., and Nematocharax venustus Weitzman, Menezes & Britski, 1986.
According to our results, Moenkhausia flava is more close related to H. unilineatus (the type species of Hemigrammus) than to M. xinguensis (type species of Moenkhausia), reinforcing the weakness of the diagnostic characters between Moenkhausia (completely pored lateral line) and Hemigrammus (incompletely pored lateral line). Our new species is also close related to Moenkhausia copei, Hemigrammus barrigonae, M. collettii, M. aff. copei, and H. ulreyi. Such a close relationship may be phenotypically supported by the general color pattern of these species. However, until more comprehensive analyses encompassing more representatives of both genera are available, we traditionally and conservatively opted to describe this new species in Moenkhausia.
Moenkhausia flava has an inconspicuous oval-shaped vertically elongated humeral blotch, somewhat diffuse or slightly pigmented, and a diffuse longitudinal stripe along the flank and a dark line at the anal-fin base, quite similar to the pattern described for M. copei (sensuGéry, 1977Géry J. Characoids of the world. Neptune City: T. F. H. Publications; 1977.). This author affirmed that M. copei has a humeral spot absent or slightly pigmented, and that M. collettii has a conspicuous humeral spot. We observed that M. copei has an inconspicuous oval-shaped humeral mark, and a slightly dark longitudinal stripe along the body. Moenkhausia collettii presents a conspicuous humeral spot varying from round to square-shaped crossed by a longitudinal dark stripe extending along the flank, and both species present a dark line at the base of the anal fin (Eigenmann, Myers, 1917Eigenmann CH, Myers GS.The American Characidae. Part 1. Mem Mus Comp Zool . 1917; 43:1-102.; Géry, 1977Géry J. Characoids of the world. Neptune City: T. F. H. Publications; 1977.). Hemigrammus ulreyi also has a black line on the anal fin base, as found in M. copei, M. collettii, M. flava and, Hemigrammus barrigonae (Géry, 1977Géry J. Characoids of the world. Neptune City: T. F. H. Publications; 1977.). Therefore, these species present a characteristic color pattern, which basically consists of a well-marked dark line at the base of the anal fin and a longitudinal dark stripe from posterior margin of the operculum to the caudal peduncle; and a humeral spot more or less conspicuous, but always present, varying from horizontally elongated to square-shaped. Hemigrammus machadoi, H. lunatus, Moenkhausia conspicua, and M. venerei, not included in our analysis, also share this color pattern, and probably belong to the same clade. Ota et al. (2014Ota RP, Lima FCT, Pavanelli CS. A new species of Hemigrammus Gill, 1858 (Characiformes: Characidae) from the rio Madeira and rio Paraguai basins, with a redescription of H. lunatus. Neotrop Ichthyol [serial on the Internet]. 2014; 12(2):265-79. Available from: http://dx.doi.org/10.1590/1982-0224-20130176
http://dx.doi.org/10.1590/1982-0224-2013...
) had already suggested that the shared color pattern might support a monophyletic group, which they called “Hemigrammus lunatus species-group”.
Although the color based characters used by Mirande (2009Mirande JM. Weighted parsimony phylogeny of the family Characidae (Teleostei: Characiformes). Cladistics [serial on the Internet]. 2009; 25(6):574-613. Available from: https://doi.org/10.1111/j.1096-0031.2009.00262.x
https://doi.org/10.1111/j.1096-0031.2009...
; 2010Mirande JM. Phylogeny of the family Characidae (Teleostei: Characiformes) from characters to taxonomy. Neotrop Ichthyol [serial on the Internet]. 2010; 8(3):385-568. Available from: http://dx.doi.org/10.1590/S1679-62252010000300001
http://dx.doi.org/10.1590/S1679-62252010...
) showed mostly homoplastic, our results are evidence that, for some groups of species, the color pattern is useful for detecting potential natural groups and should be tested in congruence analysis. It is noteworthy that Costa (1994Costa WJEM. Description of two new species of the genus Moenkhausia (Characiformes: Characidae) from the central Brazil. Zool Anz. 1994; 232(1-2):21-29.), based on color pattern, discussed the putative monophyly of M. oligolepis, M. sanctaefilomenae, and M. pyrophthalma, species with complete, interrupted and incomplete lateral lines, respectively, which was posteriorly corroborated by Mariguela et al. (2013Mariguela TC, Benine RC, Abe KT, Avelino GS, Oliveira C. Molecular phylogeny of Moenkhausia (Characidae) inferred from mitochondrial and nuclear DNA evidence. J Zoolog Syst Evol Res [serial on the Internet]. 2013; 51(4):327-32. Available from: https://doi.org/10.1111/jzs.12025
https://doi.org/10.1111/jzs.12025...
).
In our phylogeny, we recover the same relationships of the clade 2 of Mariguela et al. (2013Mariguela TC, Benine RC, Abe KT, Avelino GS, Oliveira C. Molecular phylogeny of Moenkhausia (Characidae) inferred from mitochondrial and nuclear DNA evidence. J Zoolog Syst Evol Res [serial on the Internet]. 2013; 51(4):327-32. Available from: https://doi.org/10.1111/jzs.12025
https://doi.org/10.1111/jzs.12025...
) plus Moenkhausia flava, evidencing a close relationship among species with very similar color pattern (e.g. Moenkhausia flava, M. copei, H. barrigonae, M. aff. copei, M. collettii and H. ulreyi sister to H. unilineatus, M. hemigrammoides and Aphyodite grammica).
Comparative material examined. Hemigrammus barrigonae. LBP 19637, 3, 34.6-35.5 mm SL, Colombia, Depto. Meta, Vista Hermosa, rio Guapaya, rio Guaviare basin; LBP 18725, 22, 27.3-37.2 mm SL, Colombia, Meta, San Martin, río Meta, río Orinoco basin. Hemigrammus lunatus. MZUSP 90274, 10, 28.1-29.4 mm SL, Brazil, Mato Grosso State, Cáceres, rio Sepotuba. Hemigrammus machadoi. Paratypes. MZUSP 37611, 20, 24.6-35.8 mm SL, Brazil, Mato Grosso State, Nova Lacerda, stream tributary of rio Guaporé, road BR-174. Hemigrammus ulreyi. LBP 7604, 2, 25.0-26.0 mm SL, Brazil, Mato Grosso State, Barão de Melgaço, lagoa marginal, rio Cuiabá basin. Hemigrammus unilineatus. MZUSP 63759, 5, 28.2-33.2 mm SL, Brazil, Pernambuco State, Igarassu, Igarapé Jacoca, Refúgio Ecológico Charles Darwin, rio Botafogo basin. Moenkhausia collettii. MCZ 52041, 5, 39.6-45.6 mm SL, Suriname, Brokopondo, Makambi Creek. LBP 4411, 67, 27.3-43.6 mm SL, Brazil, Amazonas State, Barcelos, rio Negro. Moenkhausia aff. collettii. LBP 9128, 8, 33.67-40.3 mm SL, Brazil, Pará State, Capitão Poço, Igarapé Açu, rio Guamá basin. Moenkhausia comma. LBP 14187, 8, 50.5-68.0 mm SL, Brazil, Pará State, Itaituba, Igarapé sem nome, rio Tapajós basin. LBP 17745, 2, 29.0-30.0 mm SL, Perú, Depto. Ucayali, Pucallpa, Quebrada sin nombre, rio Ucayali basin. Moenkhausia conspicua. Paratype. MZUSP 120821, 10, 22.1-37.6 mm SL, Brazil, Pará, Mojuí dos Campos, rio Curuá-Una, igarapé near Tabocal community. Moenkhausia copei. Syntypes. NMW 57383, 5, 20.4-34.1 mm SL, Brazil, Pará, Santarém. MCZ 89960, 1 of 30, 32.7 mm SL. Brazil, Pará, Santarém. Non types. MCZ 30016, 5, 25.9-34.2 mm SL, Guyana, Demerara, rio Essequibo. LBP14068, 2, 35.5-38.6 mm SL, Igarapé Montanha, rio Tapajós basin, Itaituba, Pará State. Moenkhausia forestii. Paratype. LBP 3793, 1, 33.4 mm SL, Brazil, Mato Grosso do Sul State, Aquidauana, rio Negro, rio Paraguay basin. Moenkhausia heikoi. MZUSP 97184, 1, 58.6 mm SL, Brazil, Pará State, Altamira, rio Curuá, na vila Castelo dos Sonhos, rio Xingu basin. Moenkhausia hemigrammoides. LBP 7029, 24.4 - 30.25 mm, SL, Brazil, Amazonas State, São Gabriel da Cachoeira, Igarapé Ya Mirim, rio Negro basin; MZUSP 92501, 10, 28.1-33.9 mm SL, Brazil, Amazonas State, Crixas, Igarapé Castanha, rio Negro basin. Moenkhausia margitae. LBP 12439, 5, 48.0-79.0 mm SL, Perú, Depto. Pevas, Quebrada sin nombre, rio Ampiyacu basin. Moenkhausia petymbuaba. MZUSP 96867, 7, 39.5-46.5 mm SL, Brazil, Pará State, Altamira, rio Curuá, rio Xingu basin. Moenkhausia pittieri. MCNG 14301, 1, 42.2 mm SL, Venezuela, Miranda, Pte. cerca de Araguita al Sur, Drainage Caribe. Moenkhausia simulata. MZUSP 26074, 1, 45.3 mm SL, Perú, Depto. Ucayali, Estrada Pucallpa-Huánuco, rio Huacamayo, rio Ucayali basin. Moenkhausia xinguensis. MZUSP 36806, 1, 35.2 mm SL, Brazil, Pará State, Cachoeira do Espelho, rio Xingu basin. LBP 16745, 10, 38.5-53.4 mm SL, Brazil, Pará State, rio Xingu, rio Amazonas drainage. Moenkhausia venerei. Paratypes. LBP 9028, 3, 26.7-28.2 mm SL, Brazil, Mato Grosso State, Barra do Garças, Córrego Taquaral, rio Araguaia basin; LBP 1533, 5, 25.6-32.6 mm SL, Brazil, Mato Grosso State, Barra do Garças, Ribeirão Ínsula, rio das Mortes/Araguaia. LBP 2425, 17, 18.8-36.1 mm SL, Brazil, Mato Grosso State, Barra do Garças, Ribeirão Ínsula, rio das Mortes/Araguaia.
Acknowledgments
Flávio C. T. Lima (ZUEC), Fernando C. P. Dagosta (UFGD) and anonymous reviewer for comments and suggestions on the manuscript. Talitha Zanini for help in the field. Osvaldo T. Oyakawa and Michel Gianeti (MZUSP), José Luis Birindelli (MZUEL) for the loan part of the material used in this study and curatorial assistance. Added thanks to José Luis Birindelli for live photograph of MZUEL paratype. Thanks Gleisy Avelino for help with Hemigrammus unilineatus sequence. Thanks Caroline Silva Oliveira for helping on the preparation of Fig. 6. RB is financially supported by CNPq (132968/2009-6) and FAPESP (2011/00269-4), WPT is financially supported by the UNEMAT, CO is financially supported by CNPq (303854/2009-0) and FAPESP (2014/26508-3), RCB is financially supported by CNPq (308784/2016-2). This study benefited in part from the FAPESP Thematic Project ‘‘South American Characiformes Inventory” (FAPESP grant 2011/50282-7).
References
- Benine RC. Moenkhausia levidorsa a new species from Rio Aripuanã, Amazon Basin, Brazil (Characiformes: Characidae). Ichthyol Explor Freshw. 2002; 13(4):289-94.
- Benine RC. Análise filogenética do gênero Moenkhausia (Characiformes: Characidae) com uma revisão dos táxons do alto Rio Paraná [PhD thesis].São Paulo: Universidade Estadual Paulista; 2004.
- Benine RC, Mariguela TC, Oliveira C. New species of Moenkhausia Eigenmann, 1903 (Characiformes: Characidae) with comments on the Moenkhausia oligolepis species complex. Neotrop Ichthyol [serial on the Internet]. 2009; 7(2):161-8. Available from: http://dx.doi.org/10.1590/S1679-62252009000200005
» http://dx.doi.org/10.1590/S1679-62252009000200005 - Costa WJEM. Description of two new species of the genus Moenkhausia (Characiformes: Characidae) from the central Brazil. Zool Anz. 1994; 232(1-2):21-29.
- Drummond AJ, Suchard MA, Xie D, Rambaut A. Bayesian phylogenetics with BEAUti and the BEAST 1.7. Mol Biol Evol [serial on the Internet]. 2012; 29(8):1969-73. Available from: http://dx.doi.org/10.1093/molbev/mss075
» http://dx.doi.org/10.1093/molbev/mss075 - Edgar RC. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res [serial on the Internet]. 2004; 32(5):1792-97. Available from: https://doi.org/10.1093/nar/gkh340
» https://doi.org/10.1093/nar/gkh340 - Eigenmann CH. New genera of South American fresh-water fishes, and new names for old genera. Smithson Misc Collect. 1903; 45(12):144-48.
- Eigenmann CH. The American Characidae. Part 2. Mem Mus Comp Zool. 1918; 43:103-208.
- Eigenmann CH, Myers GS.The American Characidae. Part 1. Mem Mus Comp Zool . 1917; 43:1-102.
- Eschmeyer WN, Fricke R, van der Laan R, editors. Catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2017 [Updated 2017 June 30, cited 2017 July 01]. Avaliable from: Avaliable from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
» http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp - Felsenstein J. Confidence limits on phylogenies: an approach using the bootstrap. Evolution. 1985; 39(4):783-91.
- Fink WL. A new species of Moenkhausia from the Mato Grosso region of Brazil (Pisces: Characidae). Breviora. 1979; 450:12p.
- Fink WL, Weitzman SH. The so-called Cheirodontin fishes of Central America with descriptions of two new species (Pisces: Characidae). Washington (DC): Smithsonian Institution Press; 1974. (Smithsonian contributions to Zoology; No. 172).
- Géry J. Characoids of the world. Neptune City: T. F. H. Publications; 1977.
- International Union for Conservation of Nature (IUCN). Standards and Petitions Subcommittee. Guidelines for using the IUCN Red List Categories and Criteria. 2016; Version 12. Prepared by the Standards and Petitions Subcommittee. Available from: Available from: http://cmsdocs.s3.amazonaws.com/RedListGuidelines.pdf
» http://cmsdocs.s3.amazonaws.com/RedListGuidelines.pdf - Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S et al Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics [serial on the Internet]. 2012; 28(12):1647-49. Available from: https://doi.org/10.1093/bioinformatics/bts199
» https://doi.org/10.1093/bioinformatics/bts199 - Kumar S, Stecher G, Tamura K. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol Biol Evol [serial on the Internet]. 2016; 33(7):1870-74. Available from: https://doi.org/10.1093/molbev/msw054
» https://doi.org/10.1093/molbev/msw054 - Lanfear R, Calcott B, Ho SYW, Guindon S. PartitionFinder: combined selection of partitioning schemes and substitution models for phylogenetic analyses. Mol Biol Evol [serial on the Internet]. 2012; 29(6):1695-701. Available from: https://doi.org/10.1093/molbev/mss020
» https://doi.org/10.1093/molbev/mss020 - Lima FCT, Britski HA, Machado FA. A new Moenkhausia (Characiformes: Characidae) from central Brazil, with comments on the area relationship between the upper rio Tapajós and upper rio Paraguai systems. Aqua. 2007; 13(2):45-54.
- Lima FCT, Sousa LM. A new species of Hemigrammus from the upper rio Negro basin, Brazil, with comments on the presence and arrangement of anal-fin hooks in Hemigrammus and related genera (Ostariophysi: Characiformes: Characidae). Aqua . 2009; 15(3):153-69.
- Lima FCT, Toledo-Piza M. New species of Moenkhausia (Characiformes: Characidae) from the Rio Negro of Brazil. Copeia [serial on the Internet]. 2001; 2001(4):1058-63. Available from: http://dx.doi.org/10.1590/1982-0224-20150052
» http://dx.doi.org/10.1590/1982-0224-20150052 - Lucena ZMS, Lucena CAS. Moenkhausia tergimacula, a new species from the upper rio Tocantins, Brazil (Osteichthyes: Characidae). Ichthyol Explor Freshw . 1999; 10(3):231-36.
- Mariguela TC, Benine RC, Abe KT, Avelino GS, Oliveira C. Molecular phylogeny of Moenkhausia (Characidae) inferred from mitochondrial and nuclear DNA evidence. J Zoolog Syst Evol Res [serial on the Internet]. 2013; 51(4):327-32. Available from: https://doi.org/10.1111/jzs.12025
» https://doi.org/10.1111/jzs.12025 - Mirande JM. Weighted parsimony phylogeny of the family Characidae (Teleostei: Characiformes). Cladistics [serial on the Internet]. 2009; 25(6):574-613. Available from: https://doi.org/10.1111/j.1096-0031.2009.00262.x
» https://doi.org/10.1111/j.1096-0031.2009.00262.x - Mirande JM. Phylogeny of the family Characidae (Teleostei: Characiformes) from characters to taxonomy. Neotrop Ichthyol [serial on the Internet]. 2010; 8(3):385-568. Available from: http://dx.doi.org/10.1590/S1679-62252010000300001
» http://dx.doi.org/10.1590/S1679-62252010000300001 - Oliveira C, Avelino GS, Abe KT, Mariguela TC, Benine RC, Ortí G, Vari RP, Castro RMC. Phylogenetic relationships within the speciose family Characidae (Teleostei: Ostariophysi: Characiformes) based on multilocus analysis and extensive ingroup sampling. BMC Evol Biol [serial on the Internet]. 2011; 11(1):275. Available from: https://doi.org/10.1186/1471-2148-11-275
» https://doi.org/10.1186/1471-2148-11-275 - Ota RP, Lima FCT, Pavanelli CS. A new species of Hemigrammus Gill, 1858 (Characiformes: Characidae) from the rio Madeira and rio Paraguai basins, with a redescription of H. lunatus Neotrop Ichthyol [serial on the Internet]. 2014; 12(2):265-79. Available from: http://dx.doi.org/10.1590/1982-0224-20130176
» http://dx.doi.org/10.1590/1982-0224-20130176 - Rambaut A, Drummond AJ. Treeannotator version 1. 4.2. [Computer software-Internet]. Edinburgh: University of Edinburgh; 2012. Available from: http://beast.bio.ed.ac.uk/TreeAnnotator
» http://beast.bio.ed.ac.uk/TreeAnnotator - Rambaut A, Suchard MA, Xie D, Drummond AJ. Tracer version 1.6. [Computer software-Internet]. Edinburgh: University of Edinburgh ; 2014. Available from: http://beast.bio.ed.ac.uk/Tracer
» http://beast.bio.ed.ac.uk/Tracer - Ronquist F, Huelsenbeck JP. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics. 2003; 19(12):1572-74.
- Soares IM, Bührnheim CM. A new species of Moenkhausia Eigenmann, 1903 (Characiformes: Characidae) from Amazon basin, Brazil. Zootaxa [serial on the Internet]. 2016; 4208(4):392-400. DOI: 10.11646/zootaxa.4208.4.6.
» https://doi.org/10.11646/zootaxa.4208.4.6 - Stamatakis A, Hoover P, Rougemont J. A rapid bootstrap algorithm for the RAxML web servers. Syst Biol [serial on the Internet]. 2008; 57(5):758-71. Available from: https://doi.org/10.1080/10635150802429642
» https://doi.org/10.1080/10635150802429642 - Swofford DL. PAUP*. Phylogenetic Analysis Using Parsimony (*and other Methods) Version 4b10. Sunderland (MA): Sinauer Associates; 2002.
- Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage. Cybium. 1985; 9(2):107-20.
- Weitzman SH, Palmer L. A new species of Hyphessobrycon (Teleostei: Characidae) from the Neblina region of Venezuela and Brazil, with comments on the putative ‘rosy tetra clade’. Ichthyol Explor Freshw . 1997; 7(3):209-42.
- Xia X, Xie Z. DAMBE: Data analysis in molecular biology and evolution. J Hered. 2001; 92(4):371-73.
Edited by
Data availability
Data citations
Eschmeyer WN, Fricke R, van der Laan R, editors. Catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2017 [Updated 2017 June 30, cited 2017 July 01]. Avaliable from: Avaliable from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Publication Dates
-
Publication in this collection
16 July 2018 -
Date of issue
2018
History
-
Received
17 July 2017 -
Accepted
11 June 2018