ABSTRACT
A new species of Characidium is described from the Cerrado biome, in Brasília, Distrito Federal, Central Brazil. The new species can be readily diagnosed by the presence of two or three longitudinal rows of dots along the body sides, absence of bars, by the short pectoral fin, which does not reach the vertical through the dorsal fin origin. Additional useful diagnostic characters are the scaled isthmus, absence of the adipose fin, and the terminal mouth. The new species is only known from the córrego Taquara and its tributaries, a tributary of ribeirão do Gama, upstream from lago Paranoá, in the upper rio Paraná basin. In accordance to the IUCN Red List Categories and Criteria, the new species is categorized as Critically Endangered.
Keywords:
Endangered species; IUCN; Lago Paranoá; Reserva Ecológica do Instituto Brasileiro de Geografia e Estatística; Rio Paraná basin
RESUMO
Uma nova espécie de Characidium é descrita do bioma Cerrado em Brasília, Distrito Federal, Brasil Central. A nova espécie pode ser diagnosticada pela presença de duas ou três fileiras longitudinais de pontos no corpo, ausência de barras e nadadeira peitoral curta, que não alcança a vertical que passa pela origem da nadadeira dorsal. Caracteres adicionais úteis na diagnose são o istmo escamado, a ausência da nadadeira adiposa e a boca terminal. A nova espécie é conhecida apenas do córrego Taquara e seus tributários, tributário do ribeirão do Gama a montante do Lago Paranoá, na bacia do alto rio Paraná. De acordo com os Critérios e Categorias para Listas Vermelhas da IUCN, a nova espécie é categorizada como uma Criticamente Ameaçada.
Palavras-chave:
Bacia do rio Paraná; Espécies Ameaçadas; IUCN; Lago Paranoá; Reserva Ecológica do Instituto Brasileiro de Geografia e Estatística
INTRODUCTION
Characidium Reinhardt, 1867 is a genus of Neotropical fishes currently including 79 valid species, distributed between Panamá and northern Argentina (Agudelo-Zamora et al., 2020aAgudelo-Zamora HD, Ortega-Lara A, Taphorn DC. Characidium chancoense, a new species of South American darter from the Río Cauca drainage, Colombia (Characiformes: Crenuchidae). Zootaxa. 2020a; 4768(2):249-63. https://doi.org/10.11646/zootaxa.4768.2.6
https://doi.org/10.11646/zootaxa.4768.2....
,bAgudelo-Zamora HD, Tavera J, Murillo YD, Ortega-Lara A. The unknown diversity of the genus Characidium (Characiformes: Crenuchidae) in the Chocó biogeographic region, Colombian Andes: two new species supported by morphological and molecular data. J Fish Biol. 2020b; 97(6):1662-75. https://doi.org/10.1111/jfb.14527
https://doi.org/10.1111/jfb.14527...
; Flausino Junior et al., 2020Flausino Junior N, Lima FCT, Machado FA, Melo MRS. A new species of Characidium Reinhardt (Characiformes: Crenuchidae) with a unique mid-water behavior from the upper rio Madeira basin, Brazil. Zootaxa. 2020; 4816(3):350-60. https://doi.org/10.11646/zootaxa.4816.3.5
https://doi.org/10.11646/zootaxa.4816.3....
; Fricke et al., 2020Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2020. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
http://researcharchive.calacademy.org/re...
; Zanata et al., 2020aZanata AM, Ribeiro TC, Araújo-Porto FA, Pessali TC, Oliveira-Silva L. A new species of Characidium (Characiformes: Crenuchidae) from the upper rio São Francisco basin, Minas Gerais, Brazil. Zootaxa. 2020a; 4790(3):505-14. https://doi.org/10.11646/zootaxa.4790.3.5
https://doi.org/10.11646/zootaxa.4790.3....
; Zanata, Ohara, 2020Zanata AM, Ohara WM. A new species of Characidium (Characiformes: Crenuchidae) from the rio Madeira basin, Brazil. J Fish Biol . 2020; 97(6):1681-89. https://doi.org/10.1111/jfb.14531
https://doi.org/10.1111/jfb.14531...
; Teixeira, Melo, 2021Teixeira TF, Melo MRS. A new species of Characidium Reinhardt (Characiformes: Crenuchidae) from the Río Huallaga, central Peruvian Amazon, with a review on secondary sexual characters in the genus. J Fish Biol 2021; 98(1): 178-188. https://doi.org/10.1111/jfb.14568
https://doi.org/10.1111/jfb.14568...
). The genus is considered to be monophyletic based on a single synapomorphy, the presence of a basicaudal spot, which is not present in all species (Buckup, 1993aBuckup PA. Phylogenetic interrelationships and reductive evolution in the neotropical characidiin fishes. Cladistics. 1993a; 9(3):305-41. https://doi.org/10.1111/j.1096-0031.1993.tb00227.x
https://doi.org/10.1111/j.1096-0031.1993...
).
The Brazilian Cerrado has high endemism of freshwater fishes because this biome contains the watersheds of some of the major South-American hydrological basins (Nogueira et al., 2010Nogueira C, Buckup PA, Menezes NA, Oyakawa OT, Kasecker TP, Ramos Neto MB, da Silva JMC. Restricted-Range Fishes and the Conservation of Brazilian Freshwaters. PLoS ONE. 2010; 5(6):e11390. http://dx.doi.org/10.1371/journal.pone.0011390
http://dx.doi.org/10.1371/journal.pone.0...
; Reis et al., 2016Reis RE, Albert JS, Di Dario F, Mincarone MM, Petry P, Rocha LA. Fish biodiversity and conservation in South America. J Fish Biol . 2016; 89(1):12-47. http://dx.doi.org/10.1111/jfb.13016
http://dx.doi.org/10.1111/jfb.13016...
). In the past 20 years, the taxonomic knowledge regarding the diversity of Characidium in the Brazilian Cerrado has received increased attention, resulting in descriptions of eight species: C. barbosaiFlausino Junior, Lima, Machado & Melo, 2020Flausino Junior N, Lima FCT, Machado FA, Melo MRS. A new species of Characidium Reinhardt (Characiformes: Crenuchidae) with a unique mid-water behavior from the upper rio Madeira basin, Brazil. Zootaxa. 2020; 4816(3):350-60. https://doi.org/10.11646/zootaxa.4816.3.5
https://doi.org/10.11646/zootaxa.4816.3....
, endemic to the tributaries of the upper rio Guaporé, Serra da Borda; C. cacahZanata, Ribeiro, Araújo-Porto, Pessali & Oliveira-Silva, 2020Zanata AM, Ohara WM. A new species of Characidium (Characiformes: Crenuchidae) from the rio Madeira basin, Brazil. J Fish Biol . 2020; 97(6):1681-89. https://doi.org/10.1111/jfb.14531
https://doi.org/10.1111/jfb.14531...
, from the rio das Velhas, rio São Francisco basin; C. mirimNetto-Ferreira, Birindelli & Buckup, 2013Netto-Ferreira AL, Birindelli JLO, Buckup PA. A new miniature species of Characidium Reinhardt (Ostariophysi: Characiformes: Crenuchidae) from the headwaters of the rio Araguaia, Brazil. Zootaxa. 2013; 3664(3):361-68. http://dx.doi.org/10.11646/zootaxa.3664.3.6
http://dx.doi.org/10.11646/zootaxa.3664....
, endemic to tributaries of the upper rio das Mortes, rio Tocantins basin, Chapada dos Guimarães; C. nupelia da Graça, Pavanelli & Buckup, 2008, from the rio Cuiabá upstream from the Pantanal Matogrossense, rio Paraguay basin; C. satoi Melo & Oyakawa, 2015, from rio Curral das Éguas, rio São Francisco basin; C. stigmosum Melo & Buckup, 2002, from rio Paranã, rio Tocantins basin Chapada dos Veadeiros; C. xanthopterum Silveira, Langeani, da Graça, Pavanelli & Buckup, 2008, widespread in the upper rio Paraná and upper rio Tocantins basins; and C. xavante da Graça, Pavanelli & Buckup, 2008, from rio Culuene, upper rio Xingu, Amazon basin (Melo, Buckup, 2002Melo MRS, Buckup PA. Characidium stigmosum (Characiformes: Crenuchidae): a new species of characidiin fish from Central Brazil. Copeia. 2002; 2002(4):988-93. http://dx.doi.org/10.1643/0045-8511(2002)002[0988:CSCCAN]2.0.CO;2; Da Graça et al., 2008Da Graça WJ, Pavanelli CS, Buckup PA. Two new species of Characidium (Characiformes: Crenuchidae) from Paraguay and Xingu basins, state of Mato Grosso, Brazil. Copeia. 2008; 2008(2):326-32. https://doi.org/10.1643/CI-06-167
https://doi.org/10.1643/CI-06-167...
; Netto-Ferreira et al., 2013Netto-Ferreira AL, Birindelli JLO, Buckup PA. A new miniature species of Characidium Reinhardt (Ostariophysi: Characiformes: Crenuchidae) from the headwaters of the rio Araguaia, Brazil. Zootaxa. 2013; 3664(3):361-68. http://dx.doi.org/10.11646/zootaxa.3664.3.6
http://dx.doi.org/10.11646/zootaxa.3664....
; Melo, Oyakawa, 2015Melo MRS, Oyakawa OT. A new species of Characidium Reinhardt (Characiformes, Crenuchidae) with a distinctively dimorphic male. Copeia. 2015; 103(2):281-89. http://dx.doi.org/10.1643/CI-14-073
http://dx.doi.org/10.1643/CI-14-073...
; Mendonça, Netto-Ferreira, 2015Mendonça MB, Ferreira-Netto AL. New species of Characidium (Characiformes: Crenuchidae) from the rio Tapajós and rio Xingu drainages, Pará, Brazil. Zootaxa. 2015; 4021(1):187-94. http://dx.doi.org/10.11646/zootaxa.4021.1.9
http://dx.doi.org/10.11646/zootaxa.4021....
; Flausino Junior et al., 2020Flausino Junior N, Lima FCT, Machado FA, Melo MRS. A new species of Characidium Reinhardt (Characiformes: Crenuchidae) with a unique mid-water behavior from the upper rio Madeira basin, Brazil. Zootaxa. 2020; 4816(3):350-60. https://doi.org/10.11646/zootaxa.4816.3.5
https://doi.org/10.11646/zootaxa.4816.3....
; Zanata et al., 2020bZanata AM, Ohara WM, Oyakawa OT, Dagosta FCP. A new rheophilic South American darter (Crenuchidae: Characidium) from the rio Juruena basin, Brazil, with comments on morphological adaptations to life in fast-flowing waters. J Fish Biol . 2020b; 97(5):1343-53. https://doi.org/10.1111/jfb.14485
https://doi.org/10.1111/jfb.14485 ...
).
A new Characidium species is herein described from Brasília, Distrito Federal, based on specimens collected in the córrego Taquara and its tributaries, in the headwaters of the ribeirão do Gama, upstream from the lago Paranoá, rio São Bartolomeu drainage, in the upper rio Paraná basin. Most specimens used for the present description were collected at the Reserva Ecológica do Instituto Brasileiro de Geografia e Estatística (RECOR). In addition, the conservation status of the species is also discussed.
MATERIAL AND METHODS
Morphometric and meristic data were obtained from the left side of specimens, under a binocular stereomicroscope, according to Buckup (1993bBuckup PA. Review of the characidiin fishes (Teleostei: Characiformes), with description of four new genera and ten new species. Ichthyol Explor Freshw. 1993b; 4(2):97-154.), with modifications proposed by Melo, Oyakawa (2015Melo MRS, Oyakawa OT. A new species of Characidium Reinhardt (Characiformes, Crenuchidae) with a distinctively dimorphic male. Copeia. 2015; 103(2):281-89. http://dx.doi.org/10.1643/CI-14-073
http://dx.doi.org/10.1643/CI-14-073...
). Measurements were taken using a digital caliper to 0.1 mm. Meristic data are given along the description, followed by the frequencies of each count in parentheses, and an asterisk (*) indicating the values for the holotype. Terminology for pigmentation of body follows Leitão, Buckup (2014Leitão RP, Buckup PA. A new species of Characidium (Characiformes: Crenuchidae) from coastal basins of Serra do Mar, southeastern Brazil. Copeia. 2014; 2014(1):14-22. http://dx.doi.org/10.1643/CI-12-137
http://dx.doi.org/10.1643/CI-12-137...
) and Flausino Junior et al. (2020Flausino Junior N, Lima FCT, Machado FA, Melo MRS. A new species of Characidium Reinhardt (Characiformes: Crenuchidae) with a unique mid-water behavior from the upper rio Madeira basin, Brazil. Zootaxa. 2020; 4816(3):350-60. https://doi.org/10.11646/zootaxa.4816.3.5
https://doi.org/10.11646/zootaxa.4816.3....
). Osteological features were observed in cleared-and-stained specimens (cs), prepared according to Taylor, Van Dyke (1985Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107-19.). Osteological nomenclature follows Weitzman (1962Weitzman SH. The osteology of Brycon meeki, a generalized characid fish, with an osteological definition of the family. Stanf Ichthyol Bull. 1962; 8:1-77.) and Vari, Harold (2001Vari RP, Harold AS. Phylogenetic study of the neotropical fish genera Creagrutus Günther and Piabina Reinhardt (Teleostei:Ostariophysi:Characiformes), with a revision of the cis-Andean species. Smithson Contr Zool. 2001; 613:1-239. https://doi.org/10.5479/si.00810282.613
https://doi.org/10.5479/si.00810282.613...
), with the addition of posterior cleithral process. The vertebral counts included the four anterior-most elements modified into the Weberian Apparatus counted individually and the compound element of caudal fin was counted as a single element. Supernumerary elements in dorsal and anal fins were counted only from cleared and stained individuals. Osteological illustrations were made on a Zeiss Discovery V12 stereomicroscope connected to a camera with auto-montage mechanism. The gonads examined macroscopically through a small incision made in the flank, and sex and stage of gonadal development were evaluated according to Vazzoler (1996Vazzoler AEAM. Biologia da Reprodução de Peixes Teleósteos: Teoria e Prática. Maringá: Editora da Universidade Estadual de Maringá; 1996.).
General abbreviations are as follows: SL, Standard Length; HL, Head Length; cs, cleared and stained. Acronyms for fish collections are as follows: MNRJ, Museu Nacional, Rio de Janeiro; MZUSP, Museu de Zoologia da Universidade de São Paulo; ZUEC, Museu de Zoologia da Universidade de Campinas; IBGE, Reserva Ecológica do Instituto Brasileiro de Geografia e Estatística fish collection. Abbreviations of main areas of conservation are as follows: EEJBB - Estação Ecológica Jardim Botânico de Brasília; FAL-UNB, Estação Experimental Fazenda Águas Limpas da Universidade de Brasília; EEC-UNB, Estação Ecológica da Universidade de Brasília; RECOR - Reserva Ecológica do Instituto Brasileiro de Geografia e Estatística. Only specimens in good conditions were selected as types. Maps presented in figure 4 were produced using Quantum GIS (QGIS.org, 2020QGIS.org. QGIS Geographic Information System. Version 3.14.16 [Internet]. Open Source Geospatial Foundation Project; 2020. Available from: http://qgis.org
http://qgis.org...
), with the QuickMapServices plugin. Maps presented in Figure 5 were produced using Google Earth Pro (Google, 2020Google. Google Earth Pro Version. 7.3.3.7699 (64-bit) free software. 2020. Available from: https://www.google.com/earth/
https://www.google.com/earth/...
), with the aid of the timeline function. The extent of occurrence was calculated with the aid of Google Earth Pro (Google, 2020Google. Google Earth Pro Version. 7.3.3.7699 (64-bit) free software. 2020. Available from: https://www.google.com/earth/
https://www.google.com/earth/...
), drawing the smallest polygon in which no internal angle exceeds 180 degrees and which contains all the sites of occurrence (IUCN Standards and Petitions Subcommittee, 2019International Union for Conservation of Nature (IUCN). Standards and petitions subcommittee. Guidelines for Using the IUCN Red List Categories and Criteria. Version 14 [Internet]. Gland; 2019. Available from: http://www.iucnredlist.org/documents/RedListGuidelines.pdf
http://www.iucnredlist.org/documents/Red...
). A regression analysis was performed using the computer program Microsoft Excel (MICROSOFT, 2020MICROSOFT. Microsoft Excel for Microsoft 365 MSO Version 16.0.13231; 2020.), comparing the standard length with the ratio between standard length and body depth at dorsal-fin origin.
RESULTS
Characidium onca, new species
urn:lsid:zoobank.org:act:A4755AF5-77AF-46FA-B034-49DD4A46D2B4
Holotype. MZUSP 125807, 40.1 mm SL, Brazil, Distrito Federal, Brasília, córrego Taquara at Reserva Ecológica do IBGE, tributary of ribeirão Gama, tributary of Lago Paranoá, tributary of the left bank of rio São Bartolomeu, upper rio Paraná basin, 15°54’55.04”S 47°54’23.87”W, 14 Nov 2016, M. R. S. Melo & M. C. L. B. Ribeiro.
Paratypes. All from Brazil, Brasília, Distrito Federal, rio Taquara, tributary of ribeirão do Gama, tributary of Lago Paranoá, tributary of right bank of rio São Bartolomeu. MNRJ 52124, 2, 27.1-28.4 mm SL, córrego Taquara, 15°56’29”S 47°53’52”W, 19 Oct 2005, M. C. L. B. Ribeiro; MZUSP 125795 (ex-IBGE 1175), 4, 28.6-30.6 mm SL, córrego Roncador, 15°56’08”S 47°53’18”W, 26 Mar 1990, M. C. L. B. Ribeiro; MZUSP 125796 (ex-IBGE1180), 3, 28.3-34.6 mm SL, córrego Taquara, 27 Mar 1990, M. C. L. B. Ribeiro; MZUSP 125797, 2, 39.0-44.1 mm SL, córrego Roncador, Reserva Ecológica do IBGE, 15°56’23”S 47°53’27”W, Oct 2005, M. C. L. B. Ribeiro; MZUSP 125798, 7, 22.1-39.1 mm SL, córrego Taquara, 15°55’09”S 47°54’13”W, 10 Oct 2005, M. C. L. B. Ribeiro; MZUSP 125799, 1, 30.4 mm SL, córrego da Onça, 15°56’25”S 47°54’05”W, 18 Oct 2005, M. C. L. B. Ribeiro; MZUSP 125800, 4, 23.2-25.7 mm SL, córrego Taquara, 15°55’09”S 47°54’13”W, 19 Oct 2005, M. C. L. B. Ribeiro; MZUSP 125801, 3 (1 cs), 28.8-32.8 mm SL, córrego Taquara, 15°54’36”S 47°54’30”W, 20 Oct 2005, M. C. L. B. Ribeiro; MZUSP 125802, 1, 30.6 mm SL, córrego Taquara, 15°54’32”S 47°54’45”W, 20 Oct 2005, M. C. L. B. Ribeiro; MZUSP 125803, 8 (2 cs), 28.4-41.3 mm SL, córrego Roncador, 24 Oct 2009, M. C. L. B. Ribeiro; MZUSP 125804, 1, 36.7 mm SL, córrego Roncador at Ponte do Corujão, 15°56’07”S 47°53’29”W, 23 Sep 2009, M. C. L. B. Ribeiro; MZUSP 125808, 1, 35.4 mm SL, collected with holotype; ZUEC 17241, 4, 27.0-34.7 mm SL, collected with MZUSP 125803; ZUEC 17242, 2, 38.7-40.1 mm SL, collected with MZUSP 125797; ZUEC 17243, 3, 23.5-26.8 mm SL, collected with MZUSP 125800; ZUEC 17244, 1, 31.2 mm SL, collected with MZUSP 125801.
Non-types. All from Brazil, Brasília, Distrito Federal, upper rio Paraná basin, rio Taquara drainage. IBGE 1022, 2, 31.9-36.6 mm SL, córrego Roncador; IBGE 1023, 1, 20.4 mm SL, collected with IBGE 1022; IBGE 1070, 1, 24.2 mm SL, córrego Taquara; IBGE 1089, 1, 25.4 mm SL, córrego Onça; IBGE 12, 1, 39.8 mm SL, córrego Roncador; IBGE 152, 1, 23.5 mm SL, córrego Taquara; IBGE 170, 1, 31.1 mm SL, córrego Taquara; IBGE 899, 1, 28.3 mm SL, córrego Taquara; IBGE 934, 1, 35.9 mm SL, córrego da Onça; IBGE 935, 1, 17.8 mm SL, collected with IBGE 934; IBGE 936, 1, 21.9 mm SL, collected with IBGE 934; IBGE 988, 1, 22.5 mm SL, córrego Tapera; IBGE 1729, 1, 35.4 mm SL, córrego Taquara.
| Characidium onca, holotype, MZUSP 125807, 40.1 mm SL, male. A. Specimen in lateral view soon after collection; B-D. Preserved specimen in lateral dorsal, ventral views.
| Paratypes of Characidium onca. A-B. MZUSP 125798, 23.8-27.0 mm SL respectively, córrego Taquara; C. MZUSP 125795, 28.6 mm SL, córrego Roncador; D. ZUEC 17242, 38.7 mm SL, córrego Roncador; E-F. MZUSP 125797, 39.0-44.1 mm SL, respectively, córrego Roncador. Scale bar = 5 mm.
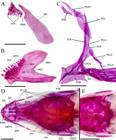
| Osteological characteristics in Characidium onca, paratypes: A. Right upper jaw in medial view (MZUSP 125803); B. Right dentary in medial view (MZUSP 125803); C. Left pectoral gridle in lateral view (MZUSP 125801); D. Skull in dorsal view (MZUSP 125803); E. Posterior portion of skull in dorsal view (MZUSP 125801). Abbreviations: ANT, antorbital; CLE, cleithrum; COR, coracoid; DEN, dentary; dentl, lateral row of dentary teeth; dentm; middle row of dentary teeth; dentr, replacement lateral row of dentary teeth; DPSO, dorsal process of the supraoccipital; ESC, extrascapular; FR, frontal; fbsc, frontal branch of the supraorbital canal; fo, fontanel; IO 1-6, infraorbitals 1 to 6; MCO, mesocoracoid; MEC, Meckel’s cartilage; METH, mesethmoid; MX, maxilla; NA, nasal; PAR, parietal; pbsc, parietal branch of the supraorbital canal; PCP, posterior cleithral process; PCL 1-3, postcleithrum 1 to 3; PMX, premaxilla; pmxt, premaxilarry teeth; pmxtr, replacement premaxillary teeth; POST, posttemporal; PTE, pterotic; SC, scapula; SCL, supracleithum; SUO, supraorbital. Scale bar = 1 mm.
| Pseudotympanum in Characidium onca (MZUSP 125801, paratype). Abbreviations: 5th pl, pleural rib of fifth vertebra; a, anterior window of pseudotympanum; lln, lateral line nerve; ls, lateralis superficialis; oi, obliquus inferioris; os, obliquus superioris; p, posterior window of pseudotympanum. Scale bar = 1 mm.
| The proportion of body depth at dorsal-fin origin (% SL) versus the standard length (mm) in females (red triangles) and males (blue dots) in Characidium onca. Symbols indicating the mature females are highlighted in the dashed area.
Diagnosis. Characidium onca can be distinguished from its congeners except C. chancoense Agudelo-Zamora, Ortega-Lara & Taphorn, 2020, C. japuhybense Travassos, 1949, C. lauroi Travassos, 1949, C. oiticicai Travassos, 1967, C. pellucidumEigenmann, 1909Eigenmann CH. Reports on the expedition to British Guiana of the Indiana University and the Carnegie Museum, 1908. Report no. 1. Some new genera and species of fishes from British Guiana. Ann Carnegie Mus. (1909); 6(1): 4-54., C. phoxocephalum Eigenmann, 1912, C. pteroides Eigenmann, 1909, C. schubarti Travassos, 1955, C. stigmosum, and C. travassosiMelo, Buckup & Oyakawa, 2016Melo MRS, Buckup PA, Oyakawa OT. A new species of Characidium Reinhardt, 1867 (Characiformes: Crenuchidae) endemic to the Atlantic Forest in Paraná State, southern Brazil. Neotrop Ichthyol. 2016; 14(3):e160014. http://dx.doi.org/10.1590/1982-0224-20160014
http://dx.doi.org/10.1590/1982-0224-2016...
by small dots on the side of body present (vs. spots absent), and from these exceptions by the dots arranged in two or three well-marked, longitudinal lines (vs. dots irregularly distributed); and by the pectoral fin short, not reaching the vertical that passes through the origin of the dorsal fin (vs. tip of pectoral fin reaching or extending posterior to the vertical that passes through the origin of dorsal fin). It further differs from C. chancoense, C. japuhybense, C. lauroi, C. helmeri, C. pellucidum, C. phoxocephalum, C. pteroides, and C. schubarti by the adipose fin absent (vs. adipose fin present); from C. stigmosum, C. japuhybense, C. longum, and C. pteroides by the bars absent (vs. bars present); from C. japuhybense, C. lauroi, C. helmeri, C. oiticicai, C. schubarti, and C. travassosi by the isthmus scaled (vs. isthmus scaleless); and from C. pellucidum and C. pteroides by the blotches on dorsum absent (vs. sickle-shaped, reddish-brown blotches present, formed by the fragmentation of vertical bars on sides of body present), and longitudinal stripe present (vs. absent).
Description. Morphometric data summarized in Tab. 1. Largest examined specimen 40.1 mm SL. Body elongate. Dorsal profile moderately convex between snout tip and dorsal-fin base, gently convex between dorsal and caudal-fin. Ventral profile gently convex between dentary tip and anal-fin origin, slightly convex at anal-fin base; almost straight between anal and caudal-fin bases. Belly strongly accentuated arched in females with well-developed ovarium. Greatest depth of body at dorsal-fin origin.
| Morphometric data for Characidium onca. Range values are for holotype and paratypes. N = total number of specimens examined; SD = Standard Deviation.
Snout in lateral view triangular and short, its tip at level of center of eye. Mouth small, terminal. Snout-maxillary tip about equal to diameter of orbit; tip of maxilla reaching level of anterior margin of orbit. Orbit nearly rounded, margin of orbit free. Cheek narrow, its depth less than one third of eye diameter. Nares distinctly separated, distance between nares shorter than distance between posterior naris and eye. Dermal flap along posterior border of anterior naris, and to anterior margin of posterior naris.
Premaxillary teeth arranged in single row, with 5(2), 6*(14), 7(7), or 8(1) unicuspid teeth, increasing in size from lateral to medial (Fig. 3A). Maxillary teeth absent (Fig. 3A). Dentary teeth arranged in two rows; outer row with 7(1), 6(3), 9*(16), 10(3), or 11(1) teeth, increasing in size from lateral to medial, medial teeth tricuspid with lateral cusps reduced, lateral teeth unicuspid, medial teeth larger and tricuspid; inner row with 9(1 cs), or 14(2 cs) teeth, minute and conical (Fig. 3B). Ectopterygoid teeth arranged in single row, with 6(1 cs), or 7(2 cs) teeth, minute and conical. Mesopterygoid teeth absent. Branchiostegal rays 5(3 cs); four attached to anterior ceratohyal (3 cs), one attached to posterior ceratohyal (3 cs). Total gill rakers on first arch 12(2); gill rakers attached to epibranchial 4(1), or 5(2); gill rakers attached to ceratobranchial 6(1), or 7(2); gill rakers attached to basibranchial 2(3).
Scales cycloid; parallel radii present on posterior field of scale, circuli on exposed field absent. Lateral line complete, with 33(4), 34*(10), 35(8), 36(1) perforated scales. Scales above lateral line 4.5*(24). Scales below lateral line 4*(24). Circumpeduncular scales 14*(24). Pre-dorsal scales series regularly arranged; scales on pre-dorsal series 11(4), 12(14), or 13*(8). Scales between anus and anal fin 2(9) or 3(15). Isthmus scaled.
Pectoral fin short, not reaching vertical that pass through dorsal-fin origin; origin of dorsal fin at level slight anterior to pelvic-fin origin; pectoral-fin rays iii,4,iii(1), iii,5,ii*(7), iii,6,i(7), iii,6,ii(8), or iii,6,iii(1). Pelvic fin short, not reaching anus; pelvic-fin rays ii,4,i(1), i,6,i*(21), ii,6,ii(1), or i,7,i(1). Dorsal-fin rays iii,8(3), ii,9*(18), iii,9(1), or i,10(2); supranumerary element on first pterygiophore of dorsal fin 1(3 cs); last dorsal-fin ray not adnate (3 cs). Anal fin not reaching ventralmost caudal-fin ray; anal-fin rays ii,6*(24); supranumerary element on first pterygiophore of anal fin 1(3 cs); last anal-fin ray not adnate (3 cs). Principal caudal-fin rays i,8,9,i*(24); lower procurrent rays 7(2 cs), or 9(1 cs); upper procurrent rays 7(1 cs) or 8(2 cs). Adipose fin absent (24).
Precaudal vertebrae 20(1 cs), or 21(2 cs); total vertebrae 35(1 cs), or 36(2 cs). Supraneurals 5(1 cs), or 7(2 cs). Postcleithrum 1 rudimentary (1) or absent (2); postcleithrum 2 and 3 present and well developed (3) (Fig. 3C). Epurals 3(3 cs); hypurals 4(1 cs) or 6(2 cs). Parietal branch of supraorbital laterosensory canal present (3 cs), elongate, reaching the parietal bone. Epiphysial branch of the supraorbital laterosensory canal present. Fontanel limited antero-laterally by parietals and posteriorly by supraoccipital (2 cs) (Fig. 3D), or extending anteriorly and contacting frontals (1 cs) (Fig. 3E). Posterior chamber of swim bladder larger than anterior chamber, 21.9-35.3% in SL (3 cs).
Pseudotympanum immediately posterior to supracleithrum, underneath anterior part of longitudinal stripe, antero-dorsally elongate, limited dorsally by lateralis superficialis, anteriorly and posteriorly, by obliquus inferioris, and ventrally by obliquus superioris; lined dorsally by lateral-line nerve. Humeral hiatus divided into anterior and posterior chambers by pleural rib of fifth. Fatty tissue filling humeral hiatus absent (Fig. 4).
Coloration in alcohol. Ground color of head and trunk light yellow, darker on dorsum, lighter on belly (Figs. 1-2). Cheek, distal portion of upper jaw, ventral part of head and opercle pale with widely spaced melanophores. Dorsal portion of snout and head light brown. Oblique preorbital stripe extending from snout tip to anterior portion of eye present and conspicuous. Postorbital stripe present, conspicuous. Bars on body absent. Three thin stripes present on dorsal part of body; dorsal stripe extending from head to dorsal-fin base; middle bar extending from head to caudal peduncle; and ventral bar extending from head to base do dorsa-caudal fin ray. Midlateral stripe, stout, extending from supracleithrum to caudal peduncle, not reaching base of middle caudal-fin rays. Humeral blotch absent. Basicaudal spot inconspicuous, vertically elongated. Dots arranged in two or three longitudinal rows; dorsal row of dots extending from level of cleithrum to caudal peduncle, slightly dorsal to, or overlapped by, midlateral stripe; middle and ventral rows of dots extending from posterior angle of opercle to caudal peduncle, ventrally to lateral line. Pectoral, pelvic, dorsal, anal and caudal fins mostly hyaline, or caudal and pectoral fins dusky; melanophores present only on lepidotrichia.
Coloration in life. Overall color pattern same as preserved. Ground color yellow to brownish with dorsum darker and belly whitish, with gold tint on opercle, eye and parts of head. A longitudinal, gold stripe present, extending along body immediately dorsal to longitudinal stripe.
Sexual dimorphism. A total of 22 specimens had the sex and gonadal development stage evaluated, including eight immature females (23.3-27.2 mm SL), seven mature females (31.1-44.1 mm SL), and seven males (26.7-40.1 mm SL). The body depth at origin of dorsal fin has a statistically significant, positive growth in both females and males, varying from 22.2 to 27.0% in SL (mean 24.3%) in immature females; from 26.5 to 29.6% in SL (mean 27.3%) in mature females; and from 21.8 to 25.5 % in SL (mean 23.4%) in mature males (Fig. 5). The mature females with fully developed eggs have a largely expanded belly, resulting on a considerable deeper body and high standard deviation for the body depth at origin of dorsal (Tab. 1). Further secondary sexual dimorphic characters, such as differences in the color pattern or the lepidotrichia bony process, are absent.
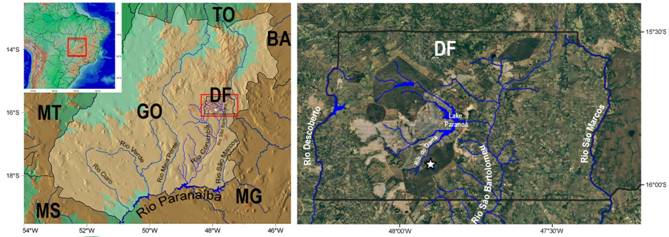
Geographical distribution.Characidium onca is only known from córrego Taquara and its tributaries, itself being a tributary of the ribeirão do Gama which nowadays drains into the lago Paranoá, a tributary of the right margin of the rio São Bartolomeu, tributary of the left margin of rio the Corumbá, rio Paranaíba drainage, in the upper rio Paraná basin, Distrito Federal, Brazil (Fig. 6).
| Maps of the tributaries of rio Paranaíba in Goiás State, Brazil in general view (left), with the rio São Bartolomeu basin detailed in the Distrito Federal (right). The type locality of Characidium onca is indicated by a star in the córrego Taquara. Abbreviations: BA, Bahia; DF, Distrito Federal; GO, Goiás; MG, Minas Gerais; MS, Mato Grosso do Sul; MT, Mato Grosso; and TO, Tocantins.
Etymology. The specific name onca refers to the Portuguese name onça, used for the jaguar Panthera onca (Linnaeus) (Mammalia: Felidae), in an allusion to the black spots in a bright gold-yellow body. It is derived from the Latin lyncea, meaning lynx (Italian “lonza”, old French “l’once”), and should be pronounced as ˈõ.sɐ. A noun in apposition.
Conservation status.Characidium onca is known only from córrego Taquara and its tributaries, which have about 6.5 km of extension. Since the 1980’s, the second author (MCLBR) has been sampling the drainages in the Distrito Federal and surroundings building a collection of over 1,000 lots of Characidium hosted mainly at the IBGE-RECOR, MZUSP and ZUEC fish collections, but C. onca was never found in any other drainages. Additional ichthyofaunistic inventories conducted in the Distrito Federal also did not report the presence of C. onca elsewhere, supporting the evidence that it truly has a narrow distribution (Ribeiro et al., 2001Ribeiro MCLB, Starling FLRM, Walter T, Farah EM. Fauna - Peixes - Evolução da comunidade de peixes do lago Paranoá. In: Fonseca FO, editor. Olhares sobre o lago Paranoá. Brasília: Secretaria de Meio Ambiente e Recursos Hídricos; 2001. p.121-28., 2008Ribeiro MCLB, Perdigão VSJ, Ramos HAC. Ictiofauna. In: Fonseca FO, editor. Águas Emendadas. Brasília: Secretaria de Desenvolvimento Urbano e Meio Ambiente - Seduma; 2008. p.253-72. Available from: http://www.recursoshidricos.df.gov.br/aguas_emendadas/documentos/Aguas_Emendadas.pdf
http://www.recursoshidricos.df.gov.br/ag...
; Aquino et al., 2009Aquino PPU, Schneider M, Silva MJM, Fonseca CP, Arakawa HB, Cavalcanti DR. Ictiofauna dos córregos do Parque Nacional de Brasília, bacia do Alto rio Paraná, Distrito Federal, Brasil Central. Biota Neotrop. 2009; 9(1):217-30. https://doi.org/10.1590/S1676-06032009000100021
https://doi.org/10.1590/S1676-0603200900...
; Ribeiro, 2012Ribeiro MCLB. Ictiofauna da bacia hidrográfica do Ribeirão do Torto: Condições atuais, impactos dos barramentos e recomendações. Brasília: CAESB; 2012.). The extent of occurrence of C. onca is calculated in 15 km2 and considered a single location. Additionally, the species is naturally rare and have extremely low abundance in nature (MRSM and MCLBR, pers. obs.).
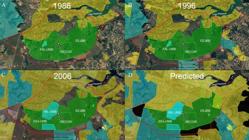
Since the construction of Brasilia in 1960’s, the impacts on the ribeirão do Gama drainage are gradually increasing (Figs. 7A-D). The lago Paranoá is an artificial lake formed by damming the rio Paranoá in 1959, to provide water supply, electric power and to increase the air humidity in Brasília. The Lago Paranoá extends to the lower part of the ribeirão do Gama, slightly downstream (>8 km) from the collection sites of C. onca, resulting on habitat loss by changing the original characteristics of a lotic into a lentic system, and by the destruction of the original riparian vegetation. The formation of the lake was followed by the introduction of several exotic species, such as the Congo tilapia Coptodon rendalli (Boulenger, 1897), the largemouth bass (known in Brazil as black bass) Micropterus salmoides (Lacepède, 1802), the bluegill Lepomis macrochirus Rafinesque, 1819 and the carp Cyprinus carpio Linnaeus, 1758 (Ribeiro et al., 2001Ribeiro MCLB, Starling FLRM, Walter T, Farah EM. Fauna - Peixes - Evolução da comunidade de peixes do lago Paranoá. In: Fonseca FO, editor. Olhares sobre o lago Paranoá. Brasília: Secretaria de Meio Ambiente e Recursos Hídricos; 2001. p.121-28.).
| Satellite images of the Area de Preservação Ambiental das Bacias do Gama e Cabeça de Veado (highlighted in red), showing the land use and cover changes along the past 34 years, and predicted: in A. year of 1986, with urban areas concentrated north of the FAL-UNB, RECOR and EEJBB; B. year of 1996, illustrating the beginning of expansion of urban and rural areas just east of the FAL-UNB, RECOR and EEJBB; C. year of 2006, after the construction of the Juscelino Kubitschek bridge in 2002, illustrating a rapid expansion of urban areas in the eastern area; and D., predicted situation according to the Distrito Federal Territorial Planning Master Plan (2009, 2019). Green polygons indicate protected areas; yellow, urban areas; blue, rural areas; black, public land; and, red arrow, the Juscelino Kubitschek bridge. Abbreviations: FAL-UNB, Estação Experimental Fazenda Águas Limpas of the University of Brasília; RECOR, Reserva Ecológica do Instituo Brasileiro de Geografia e Estatística; and EEJBB, Estação Ecológica do Jardim Botânico de Brasília. Source of maps: Google Earth.
The construction of Brasilia also resulted in dynamic land use and vegetation coverage changes, with the replacement of natural areas of open fields (campo nativo) and Cerrado vegetation for agriculture and urban uses (Figs. 7A-D). Over the last two decades, the population in the Distrito Federal increased in more than 70%, from 1,737,813 in 1995 to 2,977,216, in 2016 (UNESCO, 2002UNESCO. Vegetação no Distrito Federal, Tempo e Espaço, uma Avaliação Multitemporal da Perda de Cobertura Vegetal no DF e da Diversidade Florística da Reserva da Biosfera do Cerrado. Fase I. 2nd Editon. Brasília: UNESCO; 2002. Available from: http://bibliotecadigital.puc-campinas.edu.br/services/e-books-MS/ue000208.pdf
http://bibliotecadigital.puc-campinas.ed...
). The rapid urbanization has replaced large areas of the natural Cerrado ecosystem with urban areas and agriculture land for cultivating soybean, corn, and bean. The intense use of surface and underground hydrological resources for such purposes caused water depletion and local climate changes of longer dry seasons and a predicted increase of temperature by up to 5o C (Campos, 2004Campos JEG. Hidrogeologia do Distrito Federal: bases para a gestão dos recursos hídricos subterrâneos. Rev Bras Geoc . 2004; 34(1):41-48.; Cadamuro, Campos, 2005Cadamuro ALM, Campos JEG. Recarga Artificial de Aqüíferos fraturados no Distrito Federal: uma ferramenta para a gestão dos recursos hídricos. Rev Bras Geoc. 2005; 35(1):89-98.; Lorz et al., 2012Lorz C, Abbt-Braun G, Bakker F, Borges P, Börnick H, Fortes L et al . Challenges of an integrated water resource management for the Distrito Federal, Western Central Brazil: climate, land-use and water resources. Environ Earth Sci. 2012; 65:1575-86. https://doi.org/10.1007/s12665-011-1219-1
https://doi.org/10.1007/s12665-011-1219-...
; IBGE, 2017IBGE. Instituto Brasileiro de Geografia e Estatística - Estimativa de População [internet]. Brasília, 2017 Available from: http://www.ibge.gov.br/home/estatistica/populacao/estimativa2016/default.shtm
http://www.ibge.gov.br/home/estatistica/...
). The most severe consequences of those impacts are loss of ecological integrity of both terrestrial and aquatic ecosystems, such as the reduction of water level in lakes, rivers and floodplains, the deterioration of water quality, changes in the fire regimes, loss of gallery forests, and the increase of river channel sedimentation that reduces habitats conditions for the species of Characidium (Alley et al., 1999Alley W, Reilly TE, Franke OL. Sustainability of Ground-Water Resources. U.S. Geological Survey. Circular 1186 Denver; 1999. Available from: https://pubs.usgs.gov/circ/circ1186/pdf/circ1186.pdf
https://pubs.usgs.gov/circ/circ1186/pdf/...
; Ribeiro et al., 2018Ribeiro MCLB, Rosa JWC, Veríssimo dos Santos M. Integridade ecológica e sustentabilidade: novos paradigmas para a proteção aos ecossistemas aquáticos e a segurança hídrica no Distrito Federal, Brasil. Brasília em Debate. 2018; 18:10-19. Available from: http://www.codeplan.df.gov.br/wp-content/uploads/2018/03/Brasília-em-Debate-18.pdf
http://www.codeplan.df.gov.br/wp-content...
).
The distribution of C. onca is within the Area de Proteção Ambiental (APA) das Bacias do Gama e Cabeça de Veado, created by the Decreto Distrital Nº 9.471, of April 21st, 1986 and protected by both district (Reserva da Biosfera do Cerrado, Lei Distrital Nº 742, of July 28th, 1994) and federal laws (ARIE dos córregos Capetinha e Taquara, Decreto Federal Nº 91.303, of June 3rd, 1985; APA do Planalto Central, Decreto s/nº da Presidência da República, of January 10th, 2002). The APA das Bacias do Gama e Cabeça de Veado is composed by a relatively large mosaic with more than 10,000 hectares of protected areas, including the Reserva Ecológica do IBGE (RECOR), Estação Experimental Fazenda Águas Limpas (FAL-UNB) of the University of Brasília and Estação Ecológica do Jardim Botânico (EEJBB) de Brasília, among others. The ARIE dos córregos Capetinha e Taquara was initially classified as a “Zona de Conservação Ambiental” and had a relatively low protection stability (UNESCO, 2003UNESCO. Subsídios ao zoneamento da APA Gama-Cabeça de Veado e Reserva da Biosfera do Cerrado: Caracterização e Conflitos Socioambientais. Brasília: UNESCO, MAB, Reserva da Biosfera do Cerrado; 2003. Available from: http://www.ibram.df.gov.br/wp-content/uploads/2019/05/Subs%C3%ADdios-ao-Zoneamento-da-APA-Gama-Cabe%C3%A7a-de-Veado-e-Reserva-da-Biosfera-do-Cerrado-compactado.pdf
http://www.ibram.df.gov.br/wp-content/up...
) (Fig. 7).
Because of its proximity to downtown Brasília, a considerable extension of the APA das Bacias do Gama e Cabeça de Veado is already occupied by urban areas, especially in the ribeirão do Gama left margin. The mosaic composed of the FAL-UNB, RECOR and EEJBB is also under strong agricultural and real state speculations and the plans to expand the city limits includes the construction of the Juscelino Kubitschek bridge in 2002 and several changes in delimitation and management categories of the protected areas occasioned by the revision of the Distrito Federal Master Plan (Plano Diretor de Ordenamento Territorial - PDOT, Lei Complementar Nº 17/1997, of January 23rd, 1997, revised by the Lei Complementar Nº 803/2009, of April 25th, 2009, and instituted by the Lei Distrital No 6.269, of January 29th, 2019). The FAL-UNB, was replaced with the Estação Experimenal da UNB and, within that area, the Estação Ecológica da UNB (EEc-UNB) was circumscribed as a “Macrozona de Proteção Integral”, the RECOR and the EEJBB were classified as a “Macrozona de Proteção Integral”, but the latter had a part of its territory changed to “Urban Zone”. The expansion of human occupation in the vicinity of the mosaic resulted on the habitat fragmentation and the loss of ecological integrity of both terrestrial and aquatic ecosystems, compromising the creation of buffer zones and ecological corridors (UNESCO, 2003UNESCO. Subsídios ao zoneamento da APA Gama-Cabeça de Veado e Reserva da Biosfera do Cerrado: Caracterização e Conflitos Socioambientais. Brasília: UNESCO, MAB, Reserva da Biosfera do Cerrado; 2003. Available from: http://www.ibram.df.gov.br/wp-content/uploads/2019/05/Subs%C3%ADdios-ao-Zoneamento-da-APA-Gama-Cabe%C3%A7a-de-Veado-e-Reserva-da-Biosfera-do-Cerrado-compactado.pdf
http://www.ibram.df.gov.br/wp-content/up...
; Ribeiro et al., 2018Ribeiro MCLB, Rosa JWC, Veríssimo dos Santos M. Integridade ecológica e sustentabilidade: novos paradigmas para a proteção aos ecossistemas aquáticos e a segurança hídrica no Distrito Federal, Brasil. Brasília em Debate. 2018; 18:10-19. Available from: http://www.codeplan.df.gov.br/wp-content/uploads/2018/03/Brasília-em-Debate-18.pdf
http://www.codeplan.df.gov.br/wp-content...
).
In summary, C. onca has a very narrow distribution in a single location that has observed impacts of human occupation and introduction of exotic species in the basin and nearby areas, and predicted impacts of reduction of the river flow and loss of water quality caused by the longer periods of the dry season and higher temperatures due to local climate changes allied to the intense use of surface and underground hydrological resources. Following the IUCN Red List Categories and Criteria (IUCN Standards and Petitions Subcommittee, 2019International Union for Conservation of Nature (IUCN). Standards and petitions subcommittee. Guidelines for Using the IUCN Red List Categories and Criteria. Version 14 [Internet]. Gland; 2019. Available from: http://www.iucnredlist.org/documents/RedListGuidelines.pdf
http://www.iucnredlist.org/documents/Red...
), C. onca should be categorized as Critically Endangered (CR) because of its restricted geographic distribution (B1: Extent of occurrence <100 km2) and the following two conditions: number of location equals one (a); and continuing decline observed, estimated, inferred or projected of area extent and/or quality of habitat (biii).
Comparative material examined. Same as listed in Melo et al. (2016Melo MRS, Buckup PA, Oyakawa OT. A new species of Characidium Reinhardt, 1867 (Characiformes: Crenuchidae) endemic to the Atlantic Forest in Paraná State, southern Brazil. Neotrop Ichthyol. 2016; 14(3):e160014. http://dx.doi.org/10.1590/1982-0224-20160014
http://dx.doi.org/10.1590/1982-0224-2016...
); Melo, Espíndola (2016Melo MRS, Espíndola VC. Description of a new species of Characidium Reinhardt, 1867 (Characiformes: Crenuchidae) from the Chapada Diamantina, Bahia, and redescription of Characidium bimaculatum Fowler, 1941. Zootaxa. 2016; 4196(4):552-68. http://dx.doi.org/10.11646/zootaxa.4196.4.5
http://dx.doi.org/10.11646/zootaxa.4196....
); Teixeira, Melo (2021Teixeira TF, Melo MRS. A new species of Characidium Reinhardt (Characiformes: Crenuchidae) from the Río Huallaga, central Peruvian Amazon, with a review on secondary sexual characters in the genus. J Fish Biol 2021; 98(1): 178-188. https://doi.org/10.1111/jfb.14568
https://doi.org/10.1111/jfb.14568...
).
DISCUSSION
Only a few species of Characidium have dots on sides of body: C. chancoense, C. japuhybense, C. lauroi, C. longum, C. oiticicai, C. pellucidum, C. phoxocephalum, C. pteroides, C. schubarti, C. stigmosum, and C. travassosi (Eigenmann, 1909Eigenmann CH. Reports on the expedition to British Guiana of the Indiana University and the Carnegie Museum, 1908. Report no. 1. Some new genera and species of fishes from British Guiana. Ann Carnegie Mus. (1909); 6(1): 4-54., 1912Eigenmann CH. Some results from an ichthyological reconnaissance of Colombia, South America. Part I. Indiana U Stud. (1912); 16(8): 1-27.; Travassos, 1949aTravassos H. Notas ictiológicas. I. Characidium lauroi n. sp. (Actinopterygii, Ostareophysi). Rev Bras Biol. 1949a; 9:87-92.,bTravassos H. Notas ictiológicas. II. Characidium japuhybensis n. sp. (Actinopterygii, Ostariophysi). Bras Biol. 1949b; 9:229-33., 1955Travassos H. Sobre um novo peixe do estado do Paraná (Actinopterygii, Cypriniformes, Characoidei). An Acad Bras Ciênc. 1955; 27:365-71., 1967Travassos H. Três novas espécies do gênero Characidium Reinhardt, 1866 (Actinopterigii, Characoidei). Pap Avulsos Zool. 1967; 20:45-53.; Melo, Buckup, 2002Melo MRS, Buckup PA. Characidium stigmosum (Characiformes: Crenuchidae): a new species of characidiin fish from Central Brazil. Copeia. 2002; 2002(4):988-93. http://dx.doi.org/10.1643/0045-8511(2002)002[0988:CSCCAN]2.0.CO;2; Melo et al., 2016Melo MRS, Buckup PA, Oyakawa OT. A new species of Characidium Reinhardt, 1867 (Characiformes: Crenuchidae) endemic to the Atlantic Forest in Paraná State, southern Brazil. Neotrop Ichthyol. 2016; 14(3):e160014. http://dx.doi.org/10.1590/1982-0224-20160014
http://dx.doi.org/10.1590/1982-0224-2016...
; Agudelo-Zamora et al., 2020aAgudelo-Zamora HD, Ortega-Lara A, Taphorn DC. Characidium chancoense, a new species of South American darter from the Río Cauca drainage, Colombia (Characiformes: Crenuchidae). Zootaxa. 2020a; 4768(2):249-63. https://doi.org/10.11646/zootaxa.4768.2.6
https://doi.org/10.11646/zootaxa.4768.2....
). Buckup (1993aBuckup PA. Phylogenetic interrelationships and reductive evolution in the neotropical characidiin fishes. Cladistics. 1993a; 9(3):305-41. https://doi.org/10.1111/j.1096-0031.1993.tb00227.x
https://doi.org/10.1111/j.1096-0031.1993...
) produced the most inclusive phylogenetic hypothesis for the species of Characidium, obtaining three major clades, namely Clades C1, C4 and C4, and a polytomy at the base among those clades and the species that did not fit in any of them, such as C. zebra Eigenmann, 1909 and C. hasemani Steindachner, 1915. In accordance to such hypothesis, the species of Characidium having spots do not consist in a monophyletic group, as discussed below, and therefore, despite being very distinctive from its congeners, C. onca cannot be promptly included in any of the clades proposed by Buckup (1993aBuckup PA. Phylogenetic interrelationships and reductive evolution in the neotropical characidiin fishes. Cladistics. 1993a; 9(3):305-41. https://doi.org/10.1111/j.1096-0031.1993.tb00227.x
https://doi.org/10.1111/j.1096-0031.1993...
).
The Clade C1, including Clades C2 and C3, is composed of species having unscaled isthmus, the postcleithrum 1 reduced or absent, and the fontanel reduced, limited antero-laterally by the parietals, such as C. amailaLujan, Agudelo-Zamora, Taphorn, Booth & López-Fernández, 2013Lujan NK, Agudelo-Zamora H, Taphorn DC, Booth PN, López-Fernández H. Description of a new, narrowly endemic South American Darter (Characiformes: Crenuchidae) from the Central Guiana Shield highlands of Guyana. Copeia. 2013; 2013 (3):454-63. http://dx.doi.org/10.1643/CI-12-079
http://dx.doi.org/10.1643/CI-12-079...
, C. fasciatum Reinhardt, 1867, and C. gomesi Travassos, 1956 (Buckup, 1993aBuckup PA. Phylogenetic interrelationships and reductive evolution in the neotropical characidiin fishes. Cladistics. 1993a; 9(3):305-41. https://doi.org/10.1111/j.1096-0031.1993.tb00227.x
https://doi.org/10.1111/j.1096-0031.1993...
; Lujan et al., 2013). According to Buckup (1993aBuckup PA. Phylogenetic interrelationships and reductive evolution in the neotropical characidiin fishes. Cladistics. 1993a; 9(3):305-41. https://doi.org/10.1111/j.1096-0031.1993.tb00227.x
https://doi.org/10.1111/j.1096-0031.1993...
), the size of the fontanel is variable among the members of Clade C1, but interspecific variation was never reported so far. Characidium japuhybense, C. lauroi, C. oiticicai, C. schubarti, and C. travassosi also are considered as members Clade C1 and compose the C. lauroi group, having as main diagnostic feature the presence of small dots on body (Travassos, 1949aTravassos H. Notas ictiológicas. I. Characidium lauroi n. sp. (Actinopterygii, Ostareophysi). Rev Bras Biol. 1949a; 9:87-92.,bTravassos H. Notas ictiológicas. II. Characidium japuhybensis n. sp. (Actinopterygii, Ostariophysi). Bras Biol. 1949b; 9:229-33., 1955Travassos H. Sobre um novo peixe do estado do Paraná (Actinopterygii, Cypriniformes, Characoidei). An Acad Bras Ciênc. 1955; 27:365-71., 1967Travassos H. Três novas espécies do gênero Characidium Reinhardt, 1866 (Actinopterigii, Characoidei). Pap Avulsos Zool. 1967; 20:45-53.; Melo et al., 2016Melo MRS, Buckup PA, Oyakawa OT. A new species of Characidium Reinhardt, 1867 (Characiformes: Crenuchidae) endemic to the Atlantic Forest in Paraná State, southern Brazil. Neotrop Ichthyol. 2016; 14(3):e160014. http://dx.doi.org/10.1590/1982-0224-20160014
http://dx.doi.org/10.1590/1982-0224-2016...
). Within the C. lauroi group, the postcleithrum 1 is well developed in C. japuhybense and C. travassosi and reduced in the remaining species. In C. onca, the postcleithrum 1 is either reduced or absent (Fig. 3C); the fontanel can either be reduced and limited antero-laterally by the parietals or elongated, reaching the frontals anteriorly (Figs. 3D-E); but the isthmus is completely scaled.
The Clade C4 is composed of the species with a single row of dentary teeth, the absence of the parietal branch of the laterosensory canal of head, and 12 or more vertical bars on body, such as C. cacah, C. bahiense Almeida, 1971, C. interruptum Pellegrin, 1909, and C. stigmosum (Buckup, 1993aBuckup PA. Phylogenetic interrelationships and reductive evolution in the neotropical characidiin fishes. Cladistics. 1993a; 9(3):305-41. https://doi.org/10.1111/j.1096-0031.1993.tb00227.x
https://doi.org/10.1111/j.1096-0031.1993...
; Netto-Ferreira et al., 2013Netto-Ferreira AL, Birindelli JLO, Buckup PA. A new miniature species of Characidium Reinhardt (Ostariophysi: Characiformes: Crenuchidae) from the headwaters of the rio Araguaia, Brazil. Zootaxa. 2013; 3664(3):361-68. http://dx.doi.org/10.11646/zootaxa.3664.3.6
http://dx.doi.org/10.11646/zootaxa.3664....
; Mendonça, Netto-Ferreira, 2015Mendonça MB, Ferreira-Netto AL. New species of Characidium (Characiformes: Crenuchidae) from the rio Tapajós and rio Xingu drainages, Pará, Brazil. Zootaxa. 2015; 4021(1):187-94. http://dx.doi.org/10.11646/zootaxa.4021.1.9
http://dx.doi.org/10.11646/zootaxa.4021....
; Melo, Espíndola, 2016Melo MRS, Espíndola VC. Description of a new species of Characidium Reinhardt, 1867 (Characiformes: Crenuchidae) from the Chapada Diamantina, Bahia, and redescription of Characidium bimaculatum Fowler, 1941. Zootaxa. 2016; 4196(4):552-68. http://dx.doi.org/10.11646/zootaxa.4196.4.5
http://dx.doi.org/10.11646/zootaxa.4196....
; Zanata et al., 2020Zanata AM, Ohara WM. A new species of Characidium (Characiformes: Crenuchidae) from the rio Madeira basin, Brazil. J Fish Biol . 2020; 97(6):1681-89. https://doi.org/10.1111/jfb.14531
https://doi.org/10.1111/jfb.14531...
a). In addition, the pigmentation pattern of C. stigmosum is very distinctive and unique among its congeners, as the spots are enlarged and vertically elongate (Melo, Buckup, 2002Melo MRS, Buckup PA. Characidium stigmosum (Characiformes: Crenuchidae): a new species of characidiin fish from Central Brazil. Copeia. 2002; 2002(4):988-93. http://dx.doi.org/10.1643/0045-8511(2002)002[0988:CSCCAN]2.0.CO;2). Characidium onca does not share any synapomorphies with the species of Clade C4 and, therefore, we refute a hypothesis of closer relationships between C. onca and C. stigmosum. Likewise, C. onca lacks the synapomorphies defining the Clade C5, which includes the species with narrow and fragmented bars, the reddish-brown blotches on dorsum, and a slender body, such as C. longum, C. pellucidum and C. pteroides (Buckup, 1993a; Taphorn et al., 2006Taphorn DC, Montaña CG, Buckup PA. Characidium longum (Characiformes: Crenuchidae), a new fish from Venezuela. Zootaxa. 2006; 1247(1):1-12. https://doi.org/10.11646/zootaxa.1247.1.1
https://doi.org/10.11646/zootaxa.1247.1....
).
Characidium chancoense and C. phoxocephalum are trans-Andean species with scaled isthmus and dots on the sides not assigned to any clade of Characidium (Agudelo-Zamora et al., 2020aAgudelo-Zamora HD, Ortega-Lara A, Taphorn DC. Characidium chancoense, a new species of South American darter from the Río Cauca drainage, Colombia (Characiformes: Crenuchidae). Zootaxa. 2020a; 4768(2):249-63. https://doi.org/10.11646/zootaxa.4768.2.6
https://doi.org/10.11646/zootaxa.4768.2....
). Characidium chancoense has considerable fewer dots, restricted to an irregular line below the longitudinal stripe, apparently associated to the transversal bars, being remarkably similar to C. japuhybense. Characidium phoxocephalum is the species that most closely resembles C. onca because of the numerous dots on body and the short pectoral and pelvic fins, and but can be externally distinguished by having an adipose fin. Nevertheless, osteological features suggest such external similarity would be the result of evolutionary convergence, with C. phoxocephalum having a well-developed postcleithrum 1 and an elongated and a finger-like posterior cleithral process (Agudelo-Zamora et al., 2020aAgudelo-Zamora HD, Ortega-Lara A, Taphorn DC. Characidium chancoense, a new species of South American darter from the Río Cauca drainage, Colombia (Characiformes: Crenuchidae). Zootaxa. 2020a; 4768(2):249-63. https://doi.org/10.11646/zootaxa.4768.2.6
https://doi.org/10.11646/zootaxa.4768.2....
) while, in C. onca, the postcleithrum 1 is reduced or absent, and the posterior cleithral process being short and triangular (Fig. 4).
ACKNOWLEDGMENTS
We would like to thank Caius Ferreira (RECOR-IBGE) for his-long time dedication to the RECOR fish collection and fieldwork assistance; for curatorial assistance, we thank Alessio Datovo, Michel D. Gianetti, Mario C. C. de Pinna and Osvaldo T. Oyakawa (MZUSP), Cristiano L. R. Moreira, Marcelo R. Britto and Paulo A. Buckup (MNRJ). We are indebted to Carla Polaz (Instituto Chico Mendes de Conservação da Biodiversidade - ICMBio) for the valuable discussion about the conservation status of the newly described species, and three anonymous reviewers for general comments along the manuscript. MRSM is supported by research grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq Universal process 433050/2016-0) and Fundação de Amparo à Pesquisa (FAPESP process 2017/12909-4), and FCTL by FAPESP (processes 2011/51532-7 and 2013/20936-0). Collecting permits were issued by the Instituto Brasileiro de Meio Ambiente e dos Recursos Naturais Renováveis (IBAMA).
REFERENCES
- Agudelo-Zamora HD, Ortega-Lara A, Taphorn DC. Characidium chancoense, a new species of South American darter from the Río Cauca drainage, Colombia (Characiformes: Crenuchidae). Zootaxa. 2020a; 4768(2):249-63. https://doi.org/10.11646/zootaxa.4768.2.6
» https://doi.org/10.11646/zootaxa.4768.2.6 - Agudelo-Zamora HD, Tavera J, Murillo YD, Ortega-Lara A. The unknown diversity of the genus Characidium (Characiformes: Crenuchidae) in the Chocó biogeographic region, Colombian Andes: two new species supported by morphological and molecular data. J Fish Biol. 2020b; 97(6):1662-75. https://doi.org/10.1111/jfb.14527
» https://doi.org/10.1111/jfb.14527 - Alley W, Reilly TE, Franke OL. Sustainability of Ground-Water Resources. U.S. Geological Survey. Circular 1186 Denver; 1999. Available from: https://pubs.usgs.gov/circ/circ1186/pdf/circ1186.pdf
» https://pubs.usgs.gov/circ/circ1186/pdf/circ1186.pdf - Aquino PPU, Schneider M, Silva MJM, Fonseca CP, Arakawa HB, Cavalcanti DR. Ictiofauna dos córregos do Parque Nacional de Brasília, bacia do Alto rio Paraná, Distrito Federal, Brasil Central. Biota Neotrop. 2009; 9(1):217-30. https://doi.org/10.1590/S1676-06032009000100021
» https://doi.org/10.1590/S1676-06032009000100021 - Buckup PA. Phylogenetic interrelationships and reductive evolution in the neotropical characidiin fishes. Cladistics. 1993a; 9(3):305-41. https://doi.org/10.1111/j.1096-0031.1993.tb00227.x
» https://doi.org/10.1111/j.1096-0031.1993.tb00227.x - Buckup PA. Review of the characidiin fishes (Teleostei: Characiformes), with description of four new genera and ten new species. Ichthyol Explor Freshw. 1993b; 4(2):97-154.
- Cadamuro ALM, Campos JEG. Recarga Artificial de Aqüíferos fraturados no Distrito Federal: uma ferramenta para a gestão dos recursos hídricos. Rev Bras Geoc. 2005; 35(1):89-98.
- Campos JEG. Hidrogeologia do Distrito Federal: bases para a gestão dos recursos hídricos subterrâneos. Rev Bras Geoc . 2004; 34(1):41-48.
- Eigenmann CH. Reports on the expedition to British Guiana of the Indiana University and the Carnegie Museum, 1908. Report no. 1. Some new genera and species of fishes from British Guiana. Ann Carnegie Mus. (1909); 6(1): 4-54.
- Eigenmann CH. Some results from an ichthyological reconnaissance of Colombia, South America. Part I. Indiana U Stud. (1912); 16(8): 1-27.
- Flausino Junior N, Lima FCT, Machado FA, Melo MRS. A new species of Characidium Reinhardt (Characiformes: Crenuchidae) with a unique mid-water behavior from the upper rio Madeira basin, Brazil. Zootaxa. 2020; 4816(3):350-60. https://doi.org/10.11646/zootaxa.4816.3.5
» https://doi.org/10.11646/zootaxa.4816.3.5 - Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2020. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
» http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp - Google. Google Earth Pro Version. 7.3.3.7699 (64-bit) free software. 2020. Available from: https://www.google.com/earth/
» https://www.google.com/earth/ - Da Graça WJ, Pavanelli CS, Buckup PA. Two new species of Characidium (Characiformes: Crenuchidae) from Paraguay and Xingu basins, state of Mato Grosso, Brazil. Copeia. 2008; 2008(2):326-32. https://doi.org/10.1643/CI-06-167
» https://doi.org/10.1643/CI-06-167 - IBGE. Instituto Brasileiro de Geografia e Estatística - Estimativa de População [internet]. Brasília, 2017 Available from: http://www.ibge.gov.br/home/estatistica/populacao/estimativa2016/default.shtm
» http://www.ibge.gov.br/home/estatistica/populacao/estimativa2016/default.shtm - International Union for Conservation of Nature (IUCN). Standards and petitions subcommittee. Guidelines for Using the IUCN Red List Categories and Criteria. Version 14 [Internet]. Gland; 2019. Available from: http://www.iucnredlist.org/documents/RedListGuidelines.pdf
» http://www.iucnredlist.org/documents/RedListGuidelines.pdf - Leitão RP, Buckup PA. A new species of Characidium (Characiformes: Crenuchidae) from coastal basins of Serra do Mar, southeastern Brazil. Copeia. 2014; 2014(1):14-22. http://dx.doi.org/10.1643/CI-12-137
» http://dx.doi.org/10.1643/CI-12-137 - Lorz C, Abbt-Braun G, Bakker F, Borges P, Börnick H, Fortes L et al . Challenges of an integrated water resource management for the Distrito Federal, Western Central Brazil: climate, land-use and water resources. Environ Earth Sci. 2012; 65:1575-86. https://doi.org/10.1007/s12665-011-1219-1
» https://doi.org/10.1007/s12665-011-1219-1 - Lujan NK, Agudelo-Zamora H, Taphorn DC, Booth PN, López-Fernández H. Description of a new, narrowly endemic South American Darter (Characiformes: Crenuchidae) from the Central Guiana Shield highlands of Guyana. Copeia. 2013; 2013 (3):454-63. http://dx.doi.org/10.1643/CI-12-079
» http://dx.doi.org/10.1643/CI-12-079 - Melo MRS, Buckup PA. Characidium stigmosum (Characiformes: Crenuchidae): a new species of characidiin fish from Central Brazil. Copeia. 2002; 2002(4):988-93. http://dx.doi.org/10.1643/0045-8511(2002)002[0988:CSCCAN]2.0.CO;2
- Melo MRS, Espíndola VC. Description of a new species of Characidium Reinhardt, 1867 (Characiformes: Crenuchidae) from the Chapada Diamantina, Bahia, and redescription of Characidium bimaculatum Fowler, 1941. Zootaxa. 2016; 4196(4):552-68. http://dx.doi.org/10.11646/zootaxa.4196.4.5
» http://dx.doi.org/10.11646/zootaxa.4196.4.5 - Melo MRS, Oyakawa OT. A new species of Characidium Reinhardt (Characiformes, Crenuchidae) with a distinctively dimorphic male. Copeia. 2015; 103(2):281-89. http://dx.doi.org/10.1643/CI-14-073
» http://dx.doi.org/10.1643/CI-14-073 - Melo MRS, Buckup PA, Oyakawa OT. A new species of Characidium Reinhardt, 1867 (Characiformes: Crenuchidae) endemic to the Atlantic Forest in Paraná State, southern Brazil. Neotrop Ichthyol. 2016; 14(3):e160014. http://dx.doi.org/10.1590/1982-0224-20160014
» http://dx.doi.org/10.1590/1982-0224-20160014 - Mendonça MB, Ferreira-Netto AL. New species of Characidium (Characiformes: Crenuchidae) from the rio Tapajós and rio Xingu drainages, Pará, Brazil. Zootaxa. 2015; 4021(1):187-94. http://dx.doi.org/10.11646/zootaxa.4021.1.9
» http://dx.doi.org/10.11646/zootaxa.4021.1.9 - MICROSOFT. Microsoft Excel for Microsoft 365 MSO Version 16.0.13231; 2020.
- Netto-Ferreira AL, Birindelli JLO, Buckup PA. A new miniature species of Characidium Reinhardt (Ostariophysi: Characiformes: Crenuchidae) from the headwaters of the rio Araguaia, Brazil. Zootaxa. 2013; 3664(3):361-68. http://dx.doi.org/10.11646/zootaxa.3664.3.6
» http://dx.doi.org/10.11646/zootaxa.3664.3.6 - Nogueira C, Buckup PA, Menezes NA, Oyakawa OT, Kasecker TP, Ramos Neto MB, da Silva JMC. Restricted-Range Fishes and the Conservation of Brazilian Freshwaters. PLoS ONE. 2010; 5(6):e11390. http://dx.doi.org/10.1371/journal.pone.0011390
» http://dx.doi.org/10.1371/journal.pone.0011390 - QGIS.org. QGIS Geographic Information System. Version 3.14.16 [Internet]. Open Source Geospatial Foundation Project; 2020. Available from: http://qgis.org
» http://qgis.org - Reis RE, Albert JS, Di Dario F, Mincarone MM, Petry P, Rocha LA. Fish biodiversity and conservation in South America. J Fish Biol . 2016; 89(1):12-47. http://dx.doi.org/10.1111/jfb.13016
» http://dx.doi.org/10.1111/jfb.13016 - Ribeiro MCLB. Ictiofauna da bacia hidrográfica do Ribeirão do Torto: Condições atuais, impactos dos barramentos e recomendações. Brasília: CAESB; 2012.
- Ribeiro MCLB, Perdigão VSJ, Ramos HAC. Ictiofauna. In: Fonseca FO, editor. Águas Emendadas. Brasília: Secretaria de Desenvolvimento Urbano e Meio Ambiente - Seduma; 2008. p.253-72. Available from: http://www.recursoshidricos.df.gov.br/aguas_emendadas/documentos/Aguas_Emendadas.pdf
» http://www.recursoshidricos.df.gov.br/aguas_emendadas/documentos/Aguas_Emendadas.pdf - Ribeiro MCLB, Rosa JWC, Veríssimo dos Santos M. Integridade ecológica e sustentabilidade: novos paradigmas para a proteção aos ecossistemas aquáticos e a segurança hídrica no Distrito Federal, Brasil. Brasília em Debate. 2018; 18:10-19. Available from: http://www.codeplan.df.gov.br/wp-content/uploads/2018/03/Brasília-em-Debate-18.pdf
» http://www.codeplan.df.gov.br/wp-content/uploads/2018/03/Brasília-em-Debate-18.pdf - Ribeiro MCLB, Starling FLRM, Walter T, Farah EM. Fauna - Peixes - Evolução da comunidade de peixes do lago Paranoá. In: Fonseca FO, editor. Olhares sobre o lago Paranoá. Brasília: Secretaria de Meio Ambiente e Recursos Hídricos; 2001. p.121-28.
- Taphorn DC, Montaña CG, Buckup PA. Characidium longum (Characiformes: Crenuchidae), a new fish from Venezuela. Zootaxa. 2006; 1247(1):1-12. https://doi.org/10.11646/zootaxa.1247.1.1
» https://doi.org/10.11646/zootaxa.1247.1.1 - Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107-19.
- Teixeira TF, Melo MRS. A new species of Characidium Reinhardt (Characiformes: Crenuchidae) from the Río Huallaga, central Peruvian Amazon, with a review on secondary sexual characters in the genus. J Fish Biol 2021; 98(1): 178-188. https://doi.org/10.1111/jfb.14568
» https://doi.org/10.1111/jfb.14568 - Travassos H. Notas ictiológicas. I. Characidium lauroi n. sp. (Actinopterygii, Ostareophysi). Rev Bras Biol. 1949a; 9:87-92.
- Travassos H. Notas ictiológicas. II. Characidium japuhybensis n. sp. (Actinopterygii, Ostariophysi). Bras Biol. 1949b; 9:229-33.
- Travassos H. Sobre um novo peixe do estado do Paraná (Actinopterygii, Cypriniformes, Characoidei). An Acad Bras Ciênc. 1955; 27:365-71.
- Travassos H. Três novas espécies do gênero Characidium Reinhardt, 1866 (Actinopterigii, Characoidei). Pap Avulsos Zool. 1967; 20:45-53.
- UNESCO. Vegetação no Distrito Federal, Tempo e Espaço, uma Avaliação Multitemporal da Perda de Cobertura Vegetal no DF e da Diversidade Florística da Reserva da Biosfera do Cerrado. Fase I. 2nd Editon. Brasília: UNESCO; 2002. Available from: http://bibliotecadigital.puc-campinas.edu.br/services/e-books-MS/ue000208.pdf
» http://bibliotecadigital.puc-campinas.edu.br/services/e-books-MS/ue000208.pdf - UNESCO. Subsídios ao zoneamento da APA Gama-Cabeça de Veado e Reserva da Biosfera do Cerrado: Caracterização e Conflitos Socioambientais. Brasília: UNESCO, MAB, Reserva da Biosfera do Cerrado; 2003. Available from: http://www.ibram.df.gov.br/wp-content/uploads/2019/05/Subs%C3%ADdios-ao-Zoneamento-da-APA-Gama-Cabe%C3%A7a-de-Veado-e-Reserva-da-Biosfera-do-Cerrado-compactado.pdf
» http://www.ibram.df.gov.br/wp-content/uploads/2019/05/Subs%C3%ADdios-ao-Zoneamento-da-APA-Gama-Cabe%C3%A7a-de-Veado-e-Reserva-da-Biosfera-do-Cerrado-compactado.pdf - Vari RP, Harold AS. Phylogenetic study of the neotropical fish genera Creagrutus Günther and Piabina Reinhardt (Teleostei:Ostariophysi:Characiformes), with a revision of the cis-Andean species. Smithson Contr Zool. 2001; 613:1-239. https://doi.org/10.5479/si.00810282.613
» https://doi.org/10.5479/si.00810282.613 - Vazzoler AEAM. Biologia da Reprodução de Peixes Teleósteos: Teoria e Prática. Maringá: Editora da Universidade Estadual de Maringá; 1996.
- Weitzman SH. The osteology of Brycon meeki, a generalized characid fish, with an osteological definition of the family. Stanf Ichthyol Bull. 1962; 8:1-77.
- Zanata AM, Ohara WM. A new species of Characidium (Characiformes: Crenuchidae) from the rio Madeira basin, Brazil. J Fish Biol . 2020; 97(6):1681-89. https://doi.org/10.1111/jfb.14531
» https://doi.org/10.1111/jfb.14531 - Zanata AM, Ribeiro TC, Araújo-Porto FA, Pessali TC, Oliveira-Silva L. A new species of Characidium (Characiformes: Crenuchidae) from the upper rio São Francisco basin, Minas Gerais, Brazil. Zootaxa. 2020a; 4790(3):505-14. https://doi.org/10.11646/zootaxa.4790.3.5
» https://doi.org/10.11646/zootaxa.4790.3.5 - Zanata AM, Ohara WM, Oyakawa OT, Dagosta FCP. A new rheophilic South American darter (Crenuchidae: Characidium) from the rio Juruena basin, Brazil, with comments on morphological adaptations to life in fast-flowing waters. J Fish Biol . 2020b; 97(5):1343-53. https://doi.org/10.1111/jfb.14485
» https://doi.org/10.1111/jfb.14485
ADDITIONAL NOTES
-
HOW TO CITE THIS ARTICLE
Melo MRS, Ribeiro MCLB, Lima FCT. A new, narrowly distributed, and critically endangered species of Characidium (Characiformes: Crenuchidae) from the Distrito Federal, Central Brazil. Neotrop Ichthyol. 2021; 19(1):e200061. https://doi.org/10.1590/1982-0224-2020-0061 -
ZOOBANK REGISTER
urn:lsid:zoobank.org:pub:7A74DAE7-2100-4D64-8A5A-B6D01F888CFD
Edited by
Data availability
Data citations
Fricke R, Eschmeyer WN, Van der Laan R. Eschmeyer’s catalog of fishes: genera, species, references [Internet]. San Francisco: California Academy of Science; 2020. Available from: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp
Publication Dates
-
Publication in this collection
29 Jan 2021 -
Date of issue
2021
History
-
Received
09 July 2020 -
Accepted
24 Dec 2020