Abstract
An expedition crowdfunded by aquarists to a region of the Brazilian state of Pará drained by the Tapajós and Xingu river basins resulted in the capture of five species of Corydoras from the rio Jamanxim basin, a tributary to the rio Tapajós, of which three could be confirmed as new and described herein. The new species can be promptly diagnosed from their congeners by the combination of features related to color pattern and osteology. Considering the gathered information on color patterns and morphology plus field observations, the new species are considered to compose two distinct Müllerian mimicry rings, with two of them forming a mimetic pair, while the third one forms a pair with a fourth non-identified species captured in the same expedition. An identification key to the species of Corydoras from the rio Tapajós basin is also provided.
Keywords:
Amazon basin; Corydoradinae; Osteology; Rio Jamanxim; Taxonomy
Resumo
Uma expedição com financiamento coletivo por aquaristas a uma região do estado brasileiro do Pará, drenada pelas bacias dos rios Tapajós e Xingu, resultou na captura de cinco espécies de Corydoras da bacia do rio Jamanxim, um tributário do rio Tapajós, das quais três puderam ser confirmadas como novas e descritas aqui. As novas espécies podem ser prontamente diagnosticadas de suas congêneres por uma combinação de características relacionadas a padrão de colorido e osteologia. Considerando a informação reunida sobre padrões de coloração, morfologia e observações de campo, as espécies novas são consideradas como componentes de dois arcos de mimetismo Mülleriano distintos, com duas delas formando um par mimético, enquanto que a terceira forma par com uma quarta espécie, ainda não identificada, capturada na mesma expedição. Uma chave de identificação para as espécies de Corydoras da bacia do rio Tapajós também é fornecida.
Palavras-chave:
Bacia amazônica; Corydoradinae; Osteologia; rio Jamanxim; Taxonomia
INTRODUCTION
Callichthyidae is a family of Neotropical armored catfishes that can be promptly distinguished from other Siluriformes by having two longitudinal series of dermal plates on flanks (Reis, 1998Reis RE. Anatomy and phylogenetic analysis of the neotropical callichthyid catfishes (Ostariophysi, Siluriformes). Zool J Linn Soc. 1998; 124(2):105–68. https://doi.org/10.1111/j.1096-3642.1998.tb00571.x
https://doi.org/10.1111/j.1096-3642.1998...
, 2003Reis RE. Family Callichthyidae (Armored catfishes). In: Reis RE, Kullander SO, Ferraris Jr. CJ, organizers. Check list of the freshwater fishes of South and Central America. Porto Alegre: Edipucrs; 2003. p.291–309.). Currently, there are more than 220 valid species within Callichthyidae, and about 180 included in CorydorasLacépède, 1803Lacépède BGE. Histoire Naturelle des poissons. Paris: Chez Plassan; 1803. (Fricke et al., 2022Fricke R, Eschmeyer WN, Fong JD. Eschmeyer’s catalog of fishes: species by family/subfamily [Internet]. San Francisco: California; 2022. Available from: https://researcharchive.calacademy.org/research/ichthyology/catalog/SpeciesByFamily.asp.
https://researcharchive.calacademy.org/r...
), making it the most species-rich genus of the family and also of Siluriformes (Bentley et al., 2021Bentley RF, Grant S, Tencatt LFC. A new arc-striped species of Corydoras Lacépède, 1803 (Teleostei: Callichthyidae) from the Peruvian Amazon. Zootaxa. 2021; 4948(2):184–200. https://doi.org/10.11646/zootaxa.4948.2.2
https://doi.org/10.11646/zootaxa.4948.2....
; Tencatt et al., 2021Tencatt LFC, Santos SA, Evers HG, Britto MR. Corydoras fulleri (Siluriformes: Callichthyidae), a new catfish species from the rio Madeira basin, Peru. J Fish Biol. 2021; 99(2):614–28. https://doi.org/10.1111/jfb.14750
https://doi.org/10.1111/jfb.14750...
). Although a series of comprehensive studies aiming to elucidate the taxonomy (e.g., Eigenmann, Eigenmann, 1890Eigenmann CH, Eigenmann RS. A revision of the South American Nematognathi or cat-fishes. Occas Pap Calif Acad Sci. 1890; 1:1–508.; Ellis, 1913Ellis MD. The plated nematognaths. Anna Carnegie Mus. 1913; 8:384–413.; Gosline, 1940Gosline WA. A revision of the Neotropical catfishes of the family Callichthyidae. Stanford Ichthyol Bull. 1940; 2:1–36.; Nijssen, 1970Nijssen H. Revision of the Surinam catfishes of the genus Corydoras Lacépède, 1803 (Pisces, Siluriformes, Callichthyidae). Beaufortia. 1970; 18:1–75.; Nijssen, Isbrücker, 1967Nijssen H, Isbrücker IJH. Notes on the Guiana species of Corydoras Lacépède, 1803, with descriptions of seven new species and designation of a neotype for Corydoras punctatus (Bloch, 1794) (Pisces, Cypriniformes, Callichthyidae). Zool Meded. 1967; 42:21–50., 1980aNijssen H, Isbrücker IJH. A review of the genus Corydoras Lacépède, 1803 (Pisces, Siluriformes, Callichthyidae). Bijdr Dierkd. 1980a; 50:190–220., 1983Nijssen H, Isbrücker IJH. Sept espèces nouvelles de poissons-chats cuirassés du genre Corydoras Lacepède, 1803, de Guyane française, de Bolivie, d’Argentine, du Surinam et du Brésil (Pisces, Siluriformes, Callichthyidae). RFAH. 1983; 10:73–82., 1986Nijssen H, Isbrücker IJH. Review of the genus Corydoras from Peru and Ecuador (Pisces, Siluriformes, Callichthyidae). Stud Neotrop Fauna Environ. 1986; 21(1–2):1–68.) and phylogenetic relationships (e.g., Britto, 2003Britto MR. Phylogeny of the subfamily Corydoradinae Hoedeman, 1952 (Siluriformes: Callichthyidae), with a definition of its genera. Proc Acad Nat Sci Phila. 2003; 153(1):119–54. https://doi.org/10.1635/0097-3157(2003)153[0119:POTSCH]2.0.CO;2
https://doi.org/10.1635/0097-3157(2003)1...
; Alexandrou et al., 2011Alexandrou MA, Oliveira C, Maillard M, McGill RAR, Newton J, Creer S et al. Competition and phylogeny determine community structure in Müllerian co-mimics. Nature. 2011; 469:84–89. https://doi.org/10.1038/nature09660
https://doi.org/10.1038/nature09660...
) of the species within Corydoras have been made, the knowledge on these fields remains far from an elucidative scenario (Britto et al., 2007; Tencatt, Ohara, 2016aTencatt LFC, Ohara WM. A new long-snouted species of Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio Madre de Dios basin. Zootaxa. 2016a; 4144(3):430–42. https://doi.org/10.11646/zootaxa.4144.3.9
https://doi.org/10.11646/zootaxa.4144.3....
).
Corydoras is widely distributed within cis-Andean South America, having more than the half of its representatives occurring in the Amazon basin (Britto, 2003Britto MR. Phylogeny of the subfamily Corydoradinae Hoedeman, 1952 (Siluriformes: Callichthyidae), with a definition of its genera. Proc Acad Nat Sci Phila. 2003; 153(1):119–54. https://doi.org/10.1635/0097-3157(2003)153[0119:POTSCH]2.0.CO;2
https://doi.org/10.1635/0097-3157(2003)1...
; Tencatt, Ohara, 2016bTencatt LFC, Ohara WM. Two new species of Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio Madeira basin, Brazil. Neotrop Ichthyol. 2016b; 14(1):139–54. https://doi.org/10.1590/1982-0224-20150063
https://doi.org/10.1590/1982-0224-201500...
). Jézéquel et al., (2020)Jézéquel C, Tedesco PA, Darwall W, Dias MS, Frederico RG, Hidalgo M et al. Freshwater fish diversity hotspots for conservation priorities in the Amazon Basin. Conserv Biol. 2020; 34(4):956–65. https://doi.org/10.1111/cobi.13466
https://doi.org/10.1111/cobi.13466...
reported 982 fish species for the rio Tapajós basin, six of which belonging to Corydoras: C. apiaka Espíndola, Spencer, Rocha & Britto, 2014, C. benattiiEspíndola, Tencatt, Pupo, Villa‐Verde & Britto, 2018Espíndola VC, Tencatt LFC, Pupo FM, Villa-Verde L, Britto MR. From the inside out: a new species of armoured catfish Corydoras with the description of poorly-explored character sources (Teleostei, Siluriformes, Callichthyidae). J Fish Biol. 2018; 92(5):1463–86. https://doi.org/10.1111/jfb.13602
https://doi.org/10.1111/jfb.13602...
, C. bifasciatusNijssen, 1972Nijssen H. Records of the catfish genus Corydoras from Brazil and French Guiana with descriptions of eight new species (Pisces, Siluriformes, Callichthyidae). Neth J Zool. 1972; 21:412–33., C. hephaestus Ohara, Tencatt & Britto, 2016, C. ornatus Nijssen & Isbrücker, 1976, and C. rikbaktsa Lima & Britto, 2020 (Dagosta, de Pinna, 2019Dagosta FCP, de Pinna M. The Fishes of the Amazon: Distribution and biogeographical patterns, with a comprehensive list of species. Bull Am Mus Nat Hist. 2019; 2019(431):1–163. https://doi.org/10.1206/0003-0090.431.1.1
https://doi.org/10.1206/0003-0090.431.1....
; Lima, Britto, 2020Lima FCT, Britto MR. A new Corydoras (Ostariophysi: Siluriformes: Callichthyidae) with an unusual sexual dimorphism from the rio Juruena basin, Brazil. Zootaxa. 2020; 4742(3):518–30. https://doi.org/10.11646/zootaxa.4742.3.6
https://doi.org/10.11646/zootaxa.4742.3....
).
However, the number of putative undescribed species recognized by the fishkeeping hobby said to be from this basin is considerably higher, with 20 coded species apparently occurring in the rio Tapajós basin (C86, C133, C145, C146, CW4, CW66, CW101, CW127, CW135, CW146, CW155, CW156, CW162, CW167, CW168, CW170, CW171, CW172, CW173, and CW174; see Tencatt, Evers, (2016)Tencatt LFC, Evers H-G. A new species of Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the río Madre de Dios basin, Peru. Neotrop Ichthyol. 2016; 14(1):13–26. https://doi.org/10.1590/1982-0224-20150019
https://doi.org/10.1590/1982-0224-201500...
for further information on the coding system in Corydoradinae), showing that the diversity of this genus in the rio Tapajós drainage is certainly underestimated.
As evidenced above, the intake of new species introduced into the aquarium hobby tends to be much higher than the number of species described by science. Aquarists played an important role in the scientific advances on Corydoradinae systematics, providing grants for museum visits, photographs in life, preserved specimens and tissue samples (I. J. H. Isbrücker, 2017; I. A. M. Fuller, 2017; H. G. Evers, 2017, pers. comm.; LFCT pers. obs.). Considering the discrepancy between the high number of putative undescribed species in the aquarium trade and the ones gathered by scientific ichthyological surveys, eventually absent in Natural History collections, a crowdfunding initiative was created by Ian Fuller, founder of the “Corydoras World” website, the Research Assistance Program (RAP), in which hobbyists can make donations to scientific projects related to Corydoradinae (see https://www.corydorasworld.com/knowledgebase/research-assistance-programme).
Currently, one of the most notorious species in the hobby is the “Zebrina” or “Vulcan” Corydoras, which was coded as CW111 and was believed to be from the rio Curuá, a tributary to the rio Xingu basin at the Serra do Cachimbo region. Despite its uniqueness and consequently high demand on the aquarium trade, few specimens are available in scientific collections. Therefore, the first author submitted a project to RAP in order to visit the region of the Serra do Cachimbo attempting to collect specimens of Corydoras CW111 for examination and formal description, accordingly. With the financial support provided by the RAP and by the Museu Nacional, Universidade Federal do Rio de Janeiro (MNRJ), it was possible to perform an expedition to the region of the Serra do Cachimbo, starting from the surroundings of Sinop Municipality, Mato Grosso State, to the surroundings of Novo Progresso Municipality, Pará State, through the road BR-163, where some tributaries from both rio Xingu and rio Tapajós basins were sampled. In this expedition, five Corydoras species were found in tributaries draining to the rio Tapajós basin (rio Jamanxim and some of its tributaries): C. apiaka and four putative undescribed species, Corydoras spp. CW146, CW171, CW172, and CW173, all in the rio Tapajós basin, on the main channel of the rio Jamanxim and some of its tributaries. No specimen undoubtedly fitting CW111 color pattern was found neither in the rio Curuá or in any other tributary to the Xingu or to the Tapajós river basins.
After the analysis of the gathered material, it was possible to confirm the presence of three undescribed species, Corydoras sp. CW146, Corydoras sp. CW173, and Corydoras sp. CW172. Considering the available data, it was not possible to undoubtedly differ Corydoras sp. CW171 from C. guianensisNijssen, 1970Nijssen H. Revision of the Surinam catfishes of the genus Corydoras Lacépède, 1803 (Pisces, Siluriformes, Callichthyidae). Beaufortia. 1970; 18:1–75., which occurs in coastal rivers of French Guiana and Suriname (Reis, 2003Reis RE. Family Callichthyidae (Armored catfishes). In: Reis RE, Kullander SO, Ferraris Jr. CJ, organizers. Check list of the freshwater fishes of South and Central America. Porto Alegre: Edipucrs; 2003. p.291–309.). However, minor differences in snout shape and infraorbital morphology, as well as the considerable distance between their regions of occurrence, suggest that these populations may not be conspecific, and, for this reason, the population of the rio Tapajós basin will continue to be referred herein by its code until further analysis. Therefore, the aim of this study is to present the formal description of these three undescribed species of Corydoras from the rio Jamanxim basin, Pará State, Brazil. Additionally, an identification key to the valid species of Corydoras within the rio Tapajós drainage is provided.
MATERIAL AND METHODS
Measurements were obtained using digital calipers to the nearest tenth of millimeter. Morphometric and meristic data were taken following Reis, (1997)Reis RE. Revision of the Neotropical catfish genus Hoplosternum (Ostariophysi: Siluriformes: Callichthyidae), with the description of two new genera and three new species. Ichthyol Explor Freshw. 1997; 7:299–326. with the modifications of Tencatt et al., (2013)Tencatt LFC, Vera-Alcaraz HS, Britto MR, Pavanelli CS. A new Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio São Francisco basin, Brazil. Neotrop Ichthyol. 2013; 11(2):257–64. https://doi.org/10.1590/S1679-62252013000200003
https://doi.org/10.1590/S1679-6225201300...
. Morphometrics are reported as proportions of standard length (SL) or head length (HL). Terminology of barbels follows Britto, Lima, (2003)Britto MR, Lima FCT. Corydoras tukano, a new species of Corydoradine catfish from the rio Tiquié, upper rio Negro basin, Brazil (Ostariophysi: Siluriformes: Callichthyidae). Neotrop Ichthyol. 2003; 1(2):83–91. https://doi.org/10.1590/S1679-62252003000200002
https://doi.org/10.1590/S1679-6225200300...
. For the osteological analysis, some specimens were cleared and stained (cs) according to the protocol of Taylor, Van Dyke, (1985)Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107–19.. Osteological terminology was based on Reis, (1998)Reis RE. Anatomy and phylogenetic analysis of the neotropical callichthyid catfishes (Ostariophysi, Siluriformes). Zool J Linn Soc. 1998; 124(2):105–68. https://doi.org/10.1111/j.1096-3642.1998.tb00571.x
https://doi.org/10.1111/j.1096-3642.1998...
, except for the use of parieto-supraoccipital instead of supraoccipital (Arratia, Gayet, 1995Arratia G, Gayet M. Sensory canals and related bones of Tertiary siluriform crania from Bolivia and North America and comparison with recent forms. J Vertebr Paleontol. 1995; 15(3):482–505. https://www.jstor.org/stable/4523646
https://www.jstor.org/stable/4523646...
), pterotic-extrascapular instead of pterotic-supracleithrum (Slobodian, Pastana, 2018Slobodian V, Pastana MNL. Description of a new Pimelodella (Siluriformes: Heptapteridae) species with a discussion on the upper pectoral girdle homology of Siluriformes. J Fish Biol. 2018; 93(5):901–16. https://doi.org/10.1111/jfb.13795
https://doi.org/10.1111/jfb.13795...
), and scapulocoracoid instead of coracoid (Lundberg, 1970Lundberg JG. The evolutionary history of North American catfishes, family Ictaluridae. [PhD Thesis]. Michigan: The University of Michigan; 1970.). Nomenclature of the latero-sensory canals and preopercular pores are according to Schaefer, Aquino, (2000)Schaefer SA, Aquino AE. Postotic laterosensory canal and pterotic branch homology in catfishes. J Morphol. 2000; 246(3):212–27. https://doi.org/10.1002/1097-4687(200012)246:3%3C212::aid-jmor5%3E3.0.co;2-s
https://doi.org/10.1002/1097-4687(200012...
and Schaefer, (1988)Schaefer AS. Homology and evolution of the opercular series in the loricarioid catfishes (Pisces: Siluroidei). J Zool. 1988; 214(1):81–93. https://doi.org/10.1111/j.1469-7998.1988.tb04988.x
https://doi.org/10.1111/j.1469-7998.1988...
, respectively. The supra-preopercle sensuHuysentruyt, Adriaens, (2005)Huysentruyt F, Adriaens D. Descriptive osteology of Corydoras aeneus (Siluriformes: Callichthyidae). Cybium. 2005; 29(3):261–73. https://doi.org/10.26028/cybium/2005-293-004
https://doi.org/10.26028/cybium/2005-293...
was treated here as a part of the hyomandibula according to Vera-Alcaraz, (2013)Vera-Alcaraz HS. Relações filogenéticas das espécies da família Callichthyidae (Ostariophysi, Siluriformes). [PhD Thesis]. Pontifícia Universidade Católica do Rio Grande do Sul; 2013.. To determine the development degree of the anterior laminar expansion of infraorbital 1 in relation to the nasal capsule, the specimen was positioned to maintain the largest diameter of the nasal capsule horizontally. The width of frontal bone was obtained at the same point as the least interorbital width. Vertebral counts include only free centra, with the compound caudal centrum (preural 1+ ural 1) counted as a single element. The last two dorsal-fin rays were counted as distinct elements. Pharyngeal teeth were counted in both sides of the branchial arches. In the description, numbers in parentheses represent the total number of specimens with those counts. Numbers with an asterisk refer to the counts of the holotype.
Institutional abbreviations follow Sabaj, (2020)Sabaj MH. Codes for Natural History Collections in Ichthyology and Herpetology. Copeia. 2020; 108(3):593–669. https://doi.org/10.1643/ASIHCODONS2020
https://doi.org/10.1643/ASIHCODONS2020...
, except for CITL, Coleção Ictiológica de Três Lagoas, Universidade Federal de Mato Grosso do Sul, Três Lagoas. The comparative material examined is the same as listed in Tencatt et al., (2021)Tencatt LFC, Santos SA, Evers HG, Britto MR. Corydoras fulleri (Siluriformes: Callichthyidae), a new catfish species from the rio Madeira basin, Peru. J Fish Biol. 2021; 99(2):614–28. https://doi.org/10.1111/jfb.14750
https://doi.org/10.1111/jfb.14750...
.
RESULTS
Corydoras thanatos, new species
urn:lsid:zoobank.org:act:3DD6AB86-416E-4C6C-B16C-015042A2A013
Corydoras CW146. —Lucanus, 2021:29 [AMAZONAS magazine, guide about Corydoras from Serra do Cachimbo].
Holotype. MNRJ 53287, 33.2 mm SL, Brazil, Pará State, Novo Progresso Municipality, stream with unknown name tributary to the rio Jamanxim, rio Tapajós basin, 08°23’06”S 55°19’43”W, 7 Jul 2019, M. R. Britto, W. M. Ohara and L. F. C. Tencatt.
Paratypes. All from Brazil, Pará State, Novo Progresso Municipality, rio Jamanxim basin, rio Tapajós drainage, collected by M. R. Britto, W. M. Ohara and L. F. C. Tencatt. CITL 382, 32, 12.6–18.5 mm SL, rio Jamanxim, 08°23’01”S 55°19’08”W, 7–8 Jul 2019. INPA 59776, 2, 12.9–14.2 mm SL; NUP 23540, 1, 33.2 mm SL, stream with unknown name, 08°23’33”S 55°22’36”W, 7–8 Jul 2019. CITL 383, 7 of 9, 15.0–37.0 mm SL, 2 cs of 9, 30.0–33.8 mm SL; INPA 59777, 3, 29.0–33.3 mm SL; INPA 59778, 1, 21.9 mm SL; MZUSP 126860, 7, 21.5–32.3 mm SL, collected with the holotype.
Corydoras thanatos, holotype, MNRJ 53287, 33.2 mm SL, Novo Progresso Municipality, Pará State, Brazil, stream with unknown name tributary to the rio Jamanxim, rio Tapajós basin.
Diagnosis.Corydoras thanatos can be distinguished from its congeners, except for the species from the lineages 6, 7 and 9 sensuAlexandrou et al., (2011)Alexandrou MA, Oliveira C, Maillard M, McGill RAR, Newton J, Creer S et al. Competition and phylogeny determine community structure in Müllerian co-mimics. Nature. 2011; 469:84–89. https://doi.org/10.1038/nature09660
https://doi.org/10.1038/nature09660...
plus C. difluviatilis Britto & Castro, 2002, C. hastatus Eigenmann & Eigenmann, 1888, and C. pygmaeus Knaack, 1966, by having posterior margin of pectoral-fin spine with most serrations directed towards the tip of the spine (vs. most serrations directed towards origin of spine); from C. difluviatilis and C. pygmaeus it differs by the presence of contact between nuchal plate and posterior process of the parieto-supraoccipital (vs. absence of contact between nuchal plate and the posterior process of the parieto-supraoccipital); from C. hastatus by the absence of a large-sized dark blotch on caudal-fin base (vs. caudal-fin base region with a dark brown or black large blotch, roughly diamond-shaped or rhomboid, or arrow-shaped, typically bordered anteriorly and posteriorly by conspicuously light areas); it can be distinguished from the species within lineage 7 by having dark brown or black markings on fins, except for the pelvic fin (vs. all fins devoid of dark markings); it differs from the species within lineages 6 and 9, except for C. coppenamesisNijssen, 1970Nijssen H. Revision of the Surinam catfishes of the genus Corydoras Lacépède, 1803 (Pisces, Siluriformes, Callichthyidae). Beaufortia. 1970; 18:1–75., C. lymnades Tencatt, Vera-Alcaraz, Britto & Pavanelli, 2013, C. garbei Ihering, 1911, and C. gosseiNijssen, 1972Nijssen H. Records of the catfish genus Corydoras from Brazil and French Guiana with descriptions of eight new species (Pisces, Siluriformes, Callichthyidae). Neth J Zool. 1972; 21:412–33., by having anterior laminar expansion of infraorbital 1 strongly well developed, conspicuously expanded towards the anteroventral portion of snout, significantly covering its lateral surface (vs. anterior laminar expansion of infraorbital 1 ranging from poorly to well developed, not conspicuously expanded towards the anteroventral portion of snout, leaving most of its lateral surface exposed); the new species differs from C. gossei by presenting dark brown or black markings on head (vs. presence of pale yellow to white blotches on head); from C. lymnades by having a robust body (vs. slender); from C. coppenamesis, C. lymnades, and C. garbei by having flank midline covered by small-sized dark brown or black blotches; flank midline variably with distinct longitudinal series of blotches, generally more evident on posterior half of flank (vs. flank midline with longitudinal dark brown or black stripe in C. coppenamesis; with a distinct series of longitudinally aligned moderate- to large-sized dark brown or black blotches in C. lymnades and C. garbei).
Description. Morphometric data in Tab. 1. Head laterally compressed with convex dorsal profile, roughly triangular in dorsal view. Snout short, rounded. Head profile convex from tip of snout to anterior nares, ascending nearly straight or slightly convex from this point to dorsal-fin origin; interorbital region slightly concave in some specimens. Profile slightly convex along dorsal-fin base. Postdorsal-fin body profile slightly concave to adipose-fin spine, concave from this point to caudal-fin base. Ventral profile of body nearly straight or slightly convex from isthmus to pectoral girdle, and slightly convex from this point until pelvic girdle. Profile nearly straight or slightly convex from pelvic girdle to base of first anal-fin ray, ascending abruptly concave until caudal-fin base. Body roughly elliptical in cross section at pectoral girdle, gradually becoming more compressed toward caudal fin.
Eye rounded, located dorsolaterally on head. Orbit delimited anteriorly by lateral ethmoid, anterodorsally by frontal, posterodorsally by sphenotic, posteroventrally by infraorbital 2, and anteroventrally by infraorbital 1. Anterior and posterior nares close to each other, only separated by flap of skin. Anterior naris tubular. Posterior naris close to anterodorsal margin of orbit, separated from it by distance similar to naris diameter. Mouth small, subterminal, width similar to bony orbit diameter. Maxillary barbel moderate in size, not reaching anteroventral limit of gill opening. Outer mental barbel slightly longer than maxillary barbel. Inner mental barbel fleshy, base of each counterpart slightly separated from each other. Small rounded papillae covering entire surface of all barbels, upper and lower lips, snout and isthmus.
Mesethmoid moderate in size, with anterior tip poorly developed, smaller than 50% of bone length (see Britto, 2003Britto MR. Phylogeny of the subfamily Corydoradinae Hoedeman, 1952 (Siluriformes: Callichthyidae), with a definition of its genera. Proc Acad Nat Sci Phila. 2003; 153(1):119–54. https://doi.org/10.1635/0097-3157(2003)153[0119:POTSCH]2.0.CO;2
https://doi.org/10.1635/0097-3157(2003)1...
:123, character 1, state 1; fig. 1B); posterior portion wide, partially exposed and bearing small odontodes. Nasal capsule delimited posteriorly and dorsally by frontal, anteriorly by mesethmoid, and ventrally and posteriorly by lateral ethmoid. Nasal relatively wide, laterally curved, inner margin with relatively well-developed laminar expansion contacting frontal and mesethmoid; outer margin with poorly-developed laminar expansion typically contacting lateral ethmoid. Lateral ethmoid moderately expanded anteriorly, with anterodorsal expansion contacting nasal, and anterior margin contacting posterior portion of mesethmoid. Frontal elongated, narrow, width less than half of entire length; anterior projection short, size clearly smaller than nasal length. Frontal fontanel large, slender, and somewhat ellipsoid; posterior tip extension slightly surpassing anterior margin of parieto-supraoccipital. Sphenotic somewhat trapezoid, contacting parieto-supraoccipital dorsally, pterotic-extrascapular posteriorly, second infraorbital posteroventrally and frontal anteriorly (Fig. 2A). Pterotic-extrascapular roughly pipe-shaped, with posteriormost portion contacting first lateral-line ossicle, posteroventral margin contacting cleithrum, and anteroventral margin contacting opercle and variably infraorbital 2; posterior expansion almost entirely covering lateral opening of swimbladder capsule, leaving slender area on its dorsal margin covered only by thick layer of skin. Parieto-supraoccipital wide, posterior process long and contacting nuchal plate; region of contact between posterior process and nuchal plate covered by thick layer of skin.
Two laminar infraorbitals with minute odontodes. Infraorbital 1 conspicuously large, ventral laminar expansion generally strongly well developed; some specimens with well-developed expansion; anterior portion with strongly well-developed laminar expansion, surpassing anterior margin of nasal capsule; inner laminar expansion moderately developed (Fig. 2A). Infraorbital 2 small, relatively slender, with posterior laminar expansion ranging from moderately to well developed; posteroventral margin contacting posterodorsal ridge of hyomandibula, posterodorsal edge contacting sphenotic and generally pterotic-extrascapular; posterodorsal edge not in contact with pterotic-extrascapular in some specimens; inner laminar expansion ranging from poorly- to moderately developed (Fig. 2A). Posterodorsal ridge of hyomandibula close to its articulation with opercle relatively slender, exposed, and bearing small odontodes. Dorsal ridge of hyomandibula between pterotic-extrascapular and opercle exposed and bearing odontodes. Interopercle partially covered by thick layer of skin, with posterior portion exposed and bearing odontodes; subtriangular, anterior projection ranging from moderately to well developed. Preopercle elongated, relatively slender; minute odontodes on external surface. Opercle dorsoventrally elongated; relatively compact in shape, with width equal to or slightly larger than half of its entire length; free margin slightly convex, without serrations and covered by small odontodes.
Four branchiostegal rays decreasing in size posteriorly. Hypobranchial 1 deep; hypobranchial 2 somewhat triangular, tip ossified and directed towards anterior portion, posterior margin cartilaginous; ossified portion well developed, its size about twice cartilaginous portion. Five ceratobranchials with expansions increasing posteriorly; ceratobranchial 1 with small process on anterior margin of mesial portion; ceratobranchial 3 with continuous laminar expansion on postero-lateral margin; ceratobranchial 5 toothed on posterodorsal surface, with 42 to 45(2) teeth aligned in one row. Four epibranchials with similar size; epibranchial 2 slightly larger than others, with small pointed process on laminar expansion of posterior margin; epibranchial 3 with mesially-curved uncinate process on laminar expansion of posterior margin. Two wide pharyngobranchials (3 and 4); pharyngobranchial 3 with small roughly triangular laminar expansion on posterior margin; rounded expansion in some specimens. Upper tooth plate roughly oval, 48 to 58(2) teeth aligned in two rows on posteroventral surface; rows closely aligned.
Lateral-line canal reaching cephalic laterosensory system through pterotic-extrascapular, branching twice before reaching sphenotic: pterotic branch, with single pore, preoperculomandibular branch conspicuously reduced, with single pore opening at postotic main canal; postotic main canal widens just posterior to pterotic branch. Sensory canal continuing through pterotic-extrascapula, reaching sphenotic as temporal canal, which splits into two branches: one branch giving rise to infraorbital canal, other branch connecting to frontal through supraorbital canal, both with single pore. Supraorbital canal branched, running through nasal bone. Epiphyseal branch conspicuously reduced; pore opening close to supraorbital main canal, directed towards frontal fontanel. Nasal canal with three openings, first on posterior edge, second on posterolateral portion and generally fused with first pore, and third on anterior edge. Infraorbital canal running through entire infraorbital 2, extending to infraorbital 1 and opening into two or three pores. Preoperculomandibular branch giving rise to preoperculo-mandibular canal, which runs through entire preopercle with three openings, leading to pores 3, 4, and 5, respectively.
Dorsal fin subtriangular, located just posterior to second or third dorsolateral body plate. Dorsal-fin rays II,8*(20), posterior margin of dorsal-fin spine with 20 to 24 ranging from strongly reduced to poorly-developed serrations; most serrations directed towards tip of spine; some serrations variably perpendicularly directed; serrations absent close to origin of spine; small odontodes on anterior and lateral surfaces of spine (Fig. 2B). Nuchal plate moderately developed, almost entirely exposed, with minute odontodes. Spinelet short; spine typically well developed, with adpressed distal tip surpassing posterior origin of dorsal-fin base. Pectoral fin roughly triangular, its origin just posterior to gill opening. Pectoral-fin rays I,6,i(1), I,7*(3), I,7,i(12), I,8(3) or I,9(1), posterior margin of pectoral spine with 25 to 28 poorly- to moderately-developed serrations along almost its entire length, absent close to origin of spine; most serrations directed towards tip of spine; some serrations perpendicularly directed; small odontodes on anterior, dorsal and ventral surfaces of spine (Fig. 2C). Anteroventral portion of cleithrum exposed; posterolateral portion of scapulocoracoid moderately developed, exposed, with anterior portion slightly expanded anteriorly, not in contact with anteroventral portion of cleithrum; exposed areas bearing small odontodes. Opening of axillary gland sensuKiehl et al., (2006)Kiehl E, Rieger C, Greven H. Axillary gland secretions contribute to the stress-induced discharge of a bactericidal substance in Corydoras sterbai (Callichthyidae, Siluriformes). Verh Ges Ichthyol. 2006; 5:111–15. located just posterior to pectoral-fin spine base. Pelvic fin oblong, located just below first or second ventrolateral body plate, and at vertical through dorsal-fin spine or first branched dorsal-fin ray. Pelvic-fin rays i,5*(20). Adipose fin roughly triangular, separated from base of last dorsal-fin ray by six or seven dorsolateral body plates. Anal fin subtriangular, located just posterior to 12th or 13th ventrolateral body plates, and at vertical through adipose-fin spine base or region of preadipose platelets. Anal-fin rays i,4,ii(1), ii,5(16), i,6*(2) or ii,5,i(1). Caudal fin bilobed, with dorsal and ventral lobes similar in size or dorsal lobe slightly larger than ventral lobe. Caudal-fin rays i,11,i(1) or i,12,i*(19), generally four or five dorsal and ventral procurrent rays.
Two laterosensory canals on trunk; first ossicle tubular, second ossicle laminar, both bearing small odontodes. Body plates with minute odontodes scattered over exposed area, with conspicuous line of odontodes confined to posterior margins. Dorsolateral body plates 22(1) or 23*(19). Ventrolateral body plates 20*(17) or 21(3). Dorsolateral body plates along dorsal-fin base 6*(20). Dorsolateral body plates between adipose- and caudal-fin 6(1), 7*(16) or 8(3). Preadipose platelets 2(9) or 3*(11). Ventral surface of trunk between posteroventral margin of cleithrum and pelvic-fin origin laterally delimited only by first ventrolateral body plate; ventral portion of first ventrolateral body plate ranging from slightly to moderately expanded anteriorly. Small platelets covering base of caudal-fin rays. Small platelets disposed dorsally and ventrally between junctions of lateral plates on posterior portion of caudal peduncle. Anterior margin of orbit, above region of junction between frontal and lateral ethmoid, region around nasal capsule, on region above lateral ethmoid, and dorsal, lateral and variably ventrolateral portions of snout with small- to relatively large-sized platelets bearing odontodes; platelets on snout conspicuously more concentrated above mesethmoid. Ventral surface of trunk with numerous small- to relatively large-sized irregular platelets bearing odontodes; region around pectoral-fin origin typically with larger platelets.
Vertebral count 21(2); ribs 5(2); first pair conspicuously large, its middle portion closely connected to first ventrolateral body plate; its tip connected to anterior external process of basipterygium. Parapophysis of complex vertebra well developed.
Morphometric data of the holotype and 19 paratypes of Corydoras thanatos. SD = Standard deviation.
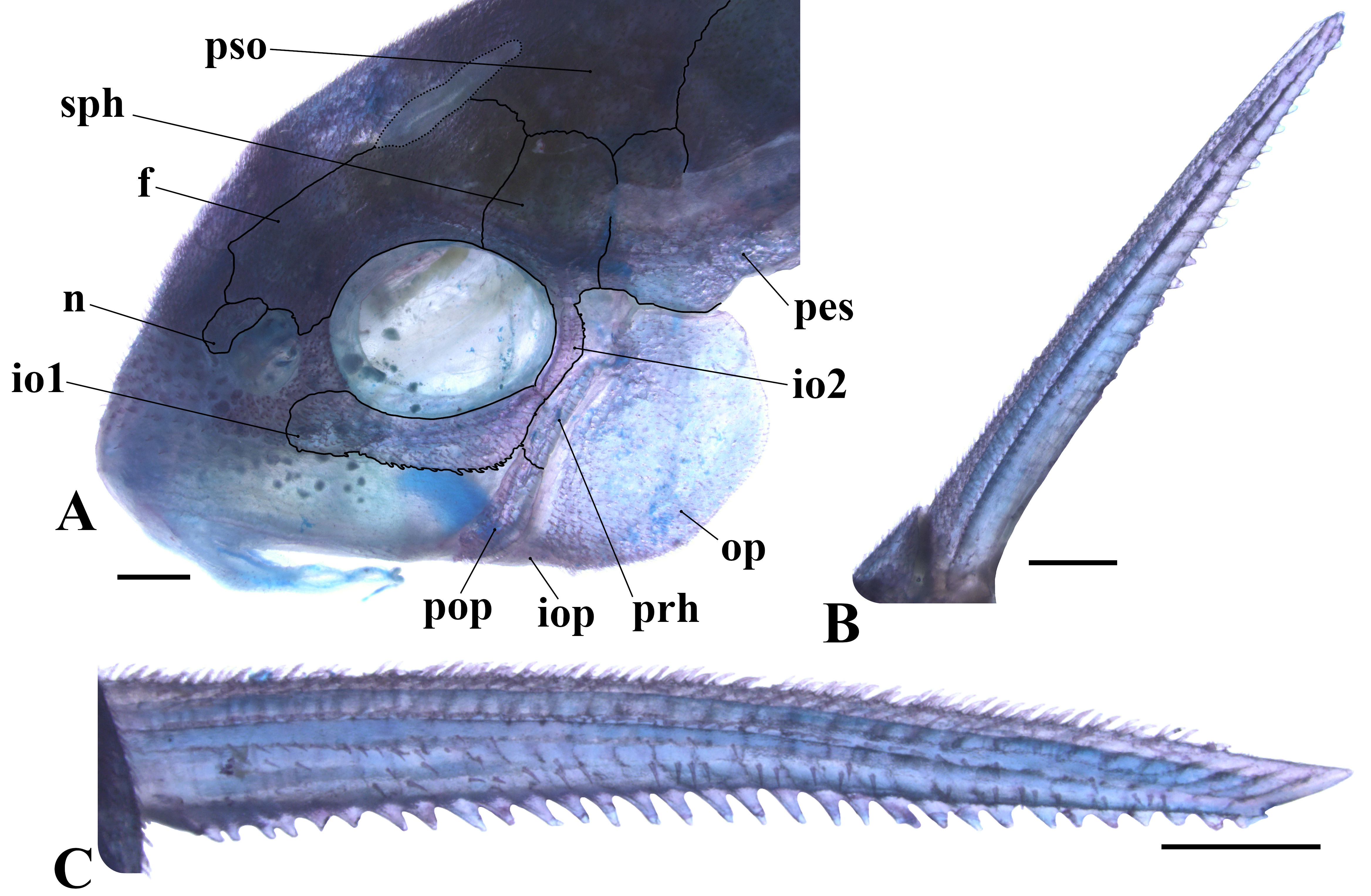
Lateral view of the head (A), lateral view of the dorsal-fin spine (B) and dorsal view of the left pectoral-fin spine (C) in cs paratype of Corydoras thanatos, CITL 383, 33.8 mm SL. Abbreviations: f: frontal, io1–2: infraorbital 1 and 2, iop: interopercle, n: nasal, op: opercle, pes: pterotic-extrascapular, pop: preopercle, prh: posterodorsal ridge of hyomandibula, pso: parieto-supraoccipital, sph: sphenotic. Scale bars = 1 mm.
Color in alcohol. Overall color of body in Fig. 1. Ground color of body pale- to brownish yellow or beige. Top of head dark brown; dark area typically extending mesially towards posterior tip of parieto-supraoccipital as thin longitudinal line. Dorsal and lateral surface of head with conspicuous concentrations of dark brown or black chromatophores, forming rounded, irregular or striated, diffuse blotches in some specimens. Lateral surface of cleithrum with conspicuous concentrations of dark brown or black chromatophores, forming rounded, irregular or vermiculated, blotches in some specimens; blotches, when present, diffuse or slightly more marked than blotches on head. Dorsolateral body plates with conspicuous concentration of dark brown or black chromatophores, typically forming rounded, irregular or vertically elongated conspicuous dark blotches; diffuse in some specimens. Ventrolateral body plates, except for region around pelvic-fin origin, with conspicuous concentration of dark brown or black chromatophores, typically forming rounded, irregular or vertically elongated conspicuous dark blotches; diffuse in some specimens; region around pelvic-fin origin with dark brown or black chromatophores but typically not forming any conspicuous pattern; ventral portion of ventrolateral body plates between pelvic-fin origin to area just anterior to anal-fin origin variably with sparse dark brown or black chromatophores. Flank midline variably with distinct longitudinal series of dark brown or black blotches, generally more evident on posterior half of flank. Blotches on flanks ranging from small to moderate in size. Posterior margin of body plates typically with dark brown or black chromatophores, variably forming thin dark lines along border of plates. Dorsal-fin with conspicuous concentrations of dark brown or black chromatophores, forming small dark blotches; blotches roughly longitudinally or obliquely aligned in some specimens, variably fused with each other and forming stripes; some specimens with less evident blotches on posterior portion of fin. Pectoral fin with conspicuous concentrations of dark brown or black chromatophores, forming small, irregular dark spots; spots roughly transversally aligned in some specimens; variably diffuse. Pelvic fin with sparse dark brown or black chromatophores, not forming spots. Adipose fin with dark brown or black chromatophores, generally more numerous and concentrated on spine and on region of membrane close to spine, typically forming single dark patch. Anal fin with conspicuous concentrations of dark brown or black chromatophores, generally more evident along its base, middle portion, and/or posterior portion, forming dark blotches roughly aligned transversally; blotches diffuse and/or not aligned in some specimens. Caudal fin with conspicuous concentrations of dark brown or black chromatophores, mainly on rays, forming dark blotches roughly aligned transversally in three to six slender bars; some specimens with less evident blotches on posterior portion of fin.
Color in life. Similar to color pattern of preserved specimens, but with light ground color of body, and with greenish yellow iridescent coloration (Figs. 3–4).
Paratypes of Corydoras thanatos photographed alive, showing general color pattern and morphology in lateral view in (A) INPA 59776, 14.2 mm SL, (B) INPA 59778, 21.9 mm SL, (C) INPA 59777, 29.0 mm SL, (D) INPA 59777, 30.0 mm SL, and (E) INPA 59777, 33.3 mm SL, all from tributaries to the rio Jamanxim, rio Tapajós basin.
Uncatalogued aquarium specimens of Corydoras thanatos photographed alive, showing general color pattern and morphology of (A) a female specimen, and (B) a dimorphic male specimen. Photos by Hans Evers.
Sexual dimorphism. As well-documented in Corydoradinae (see Nijssen, Isbrücker, 1980bNijssen H, Isbrücker IJH. Aspidorasvirgulatus n. sp., a plated catfish from Espírito Santo, Brazil (Pisces, Siluriformes, Callichthyidae). Bull Zoöl Mus Univ Amsterdam. 1980b; 7:133-39.; Britto, 2003Britto MR. Phylogeny of the subfamily Corydoradinae Hoedeman, 1952 (Siluriformes: Callichthyidae), with a definition of its genera. Proc Acad Nat Sci Phila. 2003; 153(1):119–54. https://doi.org/10.1635/0097-3157(2003)153[0119:POTSCH]2.0.CO;2
https://doi.org/10.1635/0097-3157(2003)1...
; Spadella et al., 2017Spadella MA, Desan SP, Henriques TCBPO, Oliveira C. Variation in male reproductive system characters in Corydoradinae (Loricarioidei: Callichthyidae) reflects the occurrence of different lineages in this subfamily. Neotrop Ichthyol. 2017; 15(1):e160039. https://doi.org/10.1590/1982-0224-20160039
https://doi.org/10.1590/1982-0224-201600...
), male specimens of C. thanatos present a genital papilla, which is somewhat tubular in shape. Aquarium male specimens eventually display a conspicuous elongation of the first and second dorsal-fin branched rays (Fig. 4B).
Geographical distribution.Corydoras thanatos is currently known from the main channel of the rio Jamanxim and two of its tributaries, both with unknown names, rio Tapajós basin, Novo Progresso Municipality, Pará State, Brazil (Fig. 5).
Map of the rio Tapajós and adjoining areas, showing the geographical distribution of Corydoras thanatos (black circle), C. hypnos (white star), and C. psamathos (red squares). Each symbol may represent more than one locality.
Ecological notes. The new species was observed to inhabit from main channel of the rio Jamanxim to its small tributary streams (Fig. 6). In the rio Jamanxim, it was mostly associated with the margins of the river, where juvenile specimens were observed shoaling together with juvenile and adult specimens of C. hypnos in the sandy shores, both species in relatively high abundance. In the small streams, adult and juvenile specimens were found shoaling together with juvenile and adult specimens of C. hypnos, generally in sites with submerged branches, leaf litter, and sandy and/or fine gravel substrate. In these streams, C. thanatos was found in higher numbers than C. hypnos. Although not shoaling together, the new species was found in syntopy with Corydoras sp. CW171 in these streams. In all sampled sites, the new species was mostly observed and captured at night.
Natural habitat of Corydoras thanatos and C. hypnos, showing (A) the rio Jamanxim, type locality of C. hypnos, and one of its tributaries (B), type locality of C. thanatos, rio Tapajós basin in Pará State, Brazil.
Etymology. The specific epithet “thanatos” refers to Thanatos (from the Greek θᾰ́νᾰτος, thánatos, which means “death”), the Greek god or personification of death, and the twin brother of Hypnos, the god/personification of sleep. The name makes an allusion to the fact that even though both C. thanatos and C. hypnos may present some similarities (in color pattern), they are completely different in other aspects (general morphological pattern). A noun in apposition.
Conservation status.Corydoras thanatos is currently known from its type locality and two additional records in its surroundings. However, these records are in the border of a conservation unit, the Floresta Nacional do Jamanxim, and no threat to the species as a whole is currently suspected. According to the International Union for Conservation of Nature (IUCN) categories and criteria (IUCN Standards and Petitions Subcommittee, 2019International Union for Conservation of Nature (IUCN). Standards and Petitions Subcommittee. Guidelines for using the IUCN red list categories and criteria. Version 14 [Internet]. Prepared by the Standards and Petitions Subcommittee; 2019. Available from: http://www.iucnredlist.org/documents/RedListGuidelines.pdf
http://www.iucnredlist.org/documents/Red...
), Corydoras thanatos can be classified as Least Concern (LC).
Corydoras hypnos, new species
urn:lsid:zoobank.org:act:841242F8-3AE8-4166-A0EA-D76D63340BCE
Holotype. MNRJ 53288, 31.7 mm SL, Brazil, Pará State, Novo Progresso Municipality, rio Jamanxim, a tributary to the rio Tapajós, 08°23’00”S 55°19’08”W, 8 Jul 2019, M. R. Britto, W. M. Ohara and L. F. C. Tencatt.
Paratypes. All from Brazil, Pará State, Novo Progresso Municipality, rio Jamanxim basin, rio Tapajós drainage, collected by M. R. Britto, W. M. Ohara and L. F. C. Tencatt. CITL 384, 2, 20.9–26.3 mm SL, stream with unknown name, 08°23’06”S 55°19’43”W, 7 Jul 2019. INPA 59775, 3, 22.2–27.2 mm SL, same locality as holotype, 7 Jul 2019. INPA 59774, 2, 22.6–28.2 mm SL, same locality as holotype, 11 Jul 2019. CITL 385, 11 of 13, 17.6–25.7 mm SL, 2 cs of 13, 26.6–32.0 mm SL; MZUSP 126861, 12, 13.2–24.7 mm SL; NUP 23541, 11, 18.9–26.1 mm SL, collected with the holotype.
Corydoras hypnos, holotype, MNRJ 53288, 31.7 mm SL, Brazil, Pará State, Novo Progresso Municipality, rio Jamanxim, a tributary to the rio Tapajós basin.
Diagnosis.Corydoras hypnos can be distinguished from its congeners, except for the species from the lineages 6, 7 and 9 sensuAlexandrou et al., (2011)Alexandrou MA, Oliveira C, Maillard M, McGill RAR, Newton J, Creer S et al. Competition and phylogeny determine community structure in Müllerian co-mimics. Nature. 2011; 469:84–89. https://doi.org/10.1038/nature09660
https://doi.org/10.1038/nature09660...
plus C. difluviatilis, C. hastatus, and C. pygmaeus, by having posterior margin of pectoral-fin spine with most serrations directed towards the tip of the spine (vs. most serrations directed towards origin of spine); from C. difluviatilis and C. pygmaeus it differs by the presence of contact between nuchal plate and posterior process of the parieto-supraoccipital (vs. absence of contact between nuchal plate and the posterior process of the parieto-supraoccipital); from C. hastatus by the absence of a large-sized dark blotch on caudal-fin base (vs. caudal-fin base region with a dark brown or black large blotch, roughly diamond-shaped or rhomboid, or arrow-shaped, typically bordered anteriorly and posteriorly by conspicuously light areas); it can be distinguished from the species within lineage 7 by having dark brown or black markings on fins, except for the pectoral and pelvic fins (vs. fins devoid of conspicuous dark markings); it differs from the species within lineages 6 and 9, except for C. benattii, C. eversi Tencatt & Britto, 2016, C. froehlichi Tencatt, Britto & Pavanelli, 2016, C. granti Tencatt, Lima & Britto, 2019, C. gryphus Tencatt, Britto & Pavanelli, 2014, C. psamathos, and C. thanatos, by having dorsal surface of snout with numerous platelets bearing odontodes (vs. dorsal surface of snout lacking platelets); from C. benattii and C. froehlichi by having moderately- to relatively well-developed and smoothly rounded snout (vs. conspicuously short and rounded snout); from C. eversi and C. granti, it can be distinguished by having ventral surface of trunk only with small-sized, non-coalescent platelets (vs. ventral surface of trunk entirely or partially covered by moderate- to relatively large-sized, coalescent platelets); from C. gryphus by having midline of flank with longitudinal series of dark brown or black blotches, when present, diffuse (vs. midline of flank with a longitudinal series of four to six conspicuous dark brown or black blotches), and mesethmoid partially exposed (vs. entirely covered by thick layer of skin); from C. psamathos by the presence of conspicuous dark brown or black markings at least in dorsal and caudal fins (vs. absence of conspicuous dark brown or black markings on body; dark markings, when present, diffuse); from C. thanatos by having anterior laminar expansion moderately developed, poorly expanded towards the anteroventral portion of snout, leaving most of its lateral surface exposed (vs. anterior laminar expansion of infraorbital 1 strongly well developed, conspicuously expanded towards the anteroventral portion of snout, significantly covering its lateral surface).
Description. Morphometric data in Tab. 2. Head laterally compressed with convex dorsal profile, roughly triangular in dorsal view. Snout ranging from moderately to relatively well developed and smoothly rounded. Head profile convex from tip of snout to anterior nares; ascending nearly straight or slightly convex from this point to dorsal-fin origin; region of frontal or parieto-supraoccipital slightly concave in some specimens. Profile slightly convex along dorsal-fin base. Postdorsal-fin body profile slightly concave to adipose-fin spine, slightly concave from this point to caudal-fin base. Ventral profile of body nearly straight or slightly convex from isthmus to pectoral girdle, and slightly convex from this point until pelvic girdle. Profile nearly straight or slightly convex from pelvic girdle to base of first anal-fin ray, ascending slightly concave until caudal-fin base. Body roughly elliptical in cross section at pectoral girdle, gradually becoming more compressed toward caudal fin.
Eye rounded, located dorsolaterally on head. Orbit delimited anteriorly by lateral ethmoid, anterodorsally by frontal, posterodorsally by sphenotic, posteroventrally by infraorbital 2, and anteroventrally by infraorbital 1. Anterior and posterior nares close to each other, only separated by flap of skin. Anterior naris tubular. Posterior naris close to anterodorsal margin of orbit, separated from it by distance similar to naris diameter. Mouth small, subterminal, width similar to bony orbit diameter. Maxillary barbel ranging from short to moderate in size, not reaching to anteroventral limit of gill opening. Outer mental barbel slightly longer than maxillary barbel. Inner mental barbel fleshy, base of each counterpart slightly separated from each other. Small rounded papillae covering entire surface of all barbels, upper and lower lips, snout and isthmus.
Mesethmoid moderate in size, with anterior tip poorly developed, slightly smaller than 50% of bone length (see Britto, 2003Britto MR. Phylogeny of the subfamily Corydoradinae Hoedeman, 1952 (Siluriformes: Callichthyidae), with a definition of its genera. Proc Acad Nat Sci Phila. 2003; 153(1):119–54. https://doi.org/10.1635/0097-3157(2003)153[0119:POTSCH]2.0.CO;2
https://doi.org/10.1635/0097-3157(2003)1...
:123, character 1, state 1; fig. 1B); posterior portion wide, partially exposed and bearing small odontodes. Nasal capsule delimited posteriorly and dorsally by frontal, anteriorly by mesethmoid, and ventrally and posteriorly by lateral ethmoid. Nasal slender, laterally curved, inner margin with poorly- to moderately-developed laminar expansion generally contacting frontal and mesethmoid; variably close but not in direct contact with mesethmoid; outer margin with poorly-developed to strongly reduced laminar expansion. Lateral ethmoid slightly expanded anteriorly, with anterodorsal expansion relatively distant from nasal, and anterior margin contacting posterior portion of mesethmoid. Frontal elongated, narrow, width less than half of entire length; anterior projection short, size smaller than nasal length. Frontal fontanel large, slender, and somewhat ellipsoid; posterior tip extension slightly surpassing anterior margin of parieto-supraoccipital. Sphenotic somewhat trapezoid, contacting parieto-supraoccipital dorsally, pterotic-extrascapular posteriorly, second infraorbital posteroventrally and frontal anteriorly (Fig. 8A). Pterotic-extrascapular roughly pipe-shaped, with posteriormost portion contacting first lateral-line ossicle, posteroventral margin contacting cleithrum, and anteroventral margin contacting opercle; posterior expansion almost entirely covering lateral opening of swimbladder capsule, leaving slender area on its dorsal margin covered only by thick layer of skin. Parieto-supraoccipital wide, posterior process long and contacting nuchal plate; region of contact between posterior process and nuchal plate covered by thick layer of skin.
Two laminar infraorbitals with minute odontodes. Infraorbital 1 large, ventral laminar expansion ranging from poorly to moderately developed; anterior portion with laminar expansion moderately developed, reaching to middle portion of nasal capsule; inner laminar expansion poorly developed (Fig. 8A). Infraorbital 2 small, slender, with posterior laminar expansion ranging from strongly reduced to poorly developed; posteroventral margin contacting posterodorsal ridge of hyomandibula, posterodorsal edge contacting only sphenotic; inner laminar expansion ranging from strongly reduced to poorly developed (Fig. 8A). Posterodorsal ridge of hyomandibula close to its articulation with opercle relatively slender, exposed, and bearing small odontodes. Dorsal ridge of hyomandibula between pterotic-extrascapular and opercle typically covered by thin skin layer. Interopercle entirely covered by thick layer of skin or almost entirely covered by thick layer of skin, with posterior portion exposed, and typically bearing small odontodes; subtriangular, anterior projection moderately developed. Preopercle elongated, relatively slender; minute odontodes on external surface. Opercle dorsoventrally elongated, with width slightly smaller or similar to half of its entire length; free margin slightly convex, without serrations and covered by small odontodes.
Four branchiostegal rays decreasing in size posteriorly. Hypobranchial 1 deep; hypobranchial 2 somewhat triangular, tip ossified and directed towards anterior portion, posterior margin cartilaginous; ossified portion well developed, its size about twice or triple of cartilaginous portion. Five ceratobranchials with expansions increasing posteriorly; ceratobranchial 1 with small process on anterior margin of mesial portion; ceratobranchial 3 notched on postero-lateral margin; variably with continuous laminar expansion on postero-lateral margin; ceratobranchial 5 toothed on posterodorsal surface, with 34 to 37(3) teeth aligned in one row. Four epibranchials with similar size; epibranchial 2 slightly larger than others, with small pointed process on laminar expansion of posterior margin; epibranchial 3 with mesially-curved uncinate process on laminar expansion of posterior margin. Two wide pharyngobranchials (3 and 4); pharyngobranchial 3 with smoothly rounded to nearly straight laminar expansion on posterior margin. Upper tooth plate roughly oval, 40 to 53(3) teeth aligned in two rows on posteroventral surface; rows closely aligned.
Lateral-line canal reaching cephalic laterosensory system through pterotic-extrascapular, branching twice before reaching sphenotic: pterotic branch, with single pore, preoperculomandibular branch conspicuously reduced, with single pore opening at postotic main canal; postotic main canal widens just posterior to pterotic branch. Sensory canal continuing through pterotic-extrascapular, reaching sphenotic as temporal canal, which splits into two branches: one branch giving rise to infraorbital canal, other branch connecting to frontal through supraorbital canal, both with single pore. Supraorbital canal branched, running through nasal bone. Epiphyseal branch conspicuously reduced; pore opening close to supraorbital main canal, directed towards frontal fontanel. Nasal canal with three openings, first on posterior edge, second on posterolateral portion, generally fused with first pore, and third on anterior edge. Infraorbital canal running through entire infraorbital 2, extending to infraorbital 1 and opening into two or three pores. Preoperculomandibular branch giving rise to preoperculo-mandibular canal, which runs through entire preopercle with three openings, leading to pores 3, 4, and 5, respectively; pore 3 variably opening at posterodorsal ridge of hyomandibula.
Dorsal fin subtriangular, located just posterior to second or third dorsolateral body plate. Dorsal-fin rays II,8*(19), II,9(1), posterior margin of dorsal-fin spine with 16 to 17 strongly reduced to poorly-developed serrations; most serrations directed towards tip of spine; some serrations variably perpendicularly directed; serrations absent close to origin of spine; small odontodes on anterior and lateral surfaces of spine (Fig. 8B). Nuchal plate moderately developed, almost entirely exposed, with minute odontodes. Spinelet short; spine moderately developed, with adpressed distal tip slightly surpassing posterior origin of dorsal-fin base. Pectoral fin roughly triangular, its origin just posterior to gill opening. Pectoral-fin rays I,7(3), I,7,i(1) or I,8*(16), posterior margin of pectoral spine with 20 to 32 strongly reduced to moderately-developed serrations along its entire length; most serrations directed towards tip of spine; some serrations perpendicularly directed or directed towards origin of spine; serrations variably bifid; small odontodes on anterior, dorsal and ventral surfaces of spine (Fig. 8C). Anteroventral portion of cleithrum exposed; posterolateral portion of scapulocoracoid moderately developed, exposed, with anterior portion slightly expanded anteriorly, not in contact with anteroventral portion of cleithrum; exposed areas bearing small odontodes. Opening of axillary gland sensuKiehl et al., (2006)Kiehl E, Rieger C, Greven H. Axillary gland secretions contribute to the stress-induced discharge of a bactericidal substance in Corydoras sterbai (Callichthyidae, Siluriformes). Verh Ges Ichthyol. 2006; 5:111–15. located just posterior to pectoral-fin spine base. Pelvic fin oblong, located just below first or second ventrolateral body plate, and at vertical through first branched dorsal-fin ray. Pelvic-fin rays i,5*(20). Adipose fin roughly triangular, separated from base of last dorsal-fin ray by generally six dorsolateral body plates. Anal fin subtriangular, typically located just posterior to 12th ventrolateral body plates, and at vertical through adipose-fin spine base or region of preadipose platelets. Anal-fin rays ii,5(19) or ii,6(1). Caudal fin bilobed, with dorsal and ventral lobes similar in size or dorsal lobe slightly larger than ventral lobe. Caudal-fin rays i,12,i*(20), generally four or five dorsal and ventral procurrent rays.
Typically, three laterosensory canals on trunk; first ossicle tubular, second ossicle laminar, both bearing small odontodes; third, encased in third dorsolateral body plates. Body plates with minute odontodes scattered over exposed area, with conspicuous line of odontodes confined to posterior margins. Dorsolateral body plates 23(17) or 24*(3). Ventrolateral body plates 20(13) or 21*(7). Dorsolateral body plates along dorsal-fin base 6*(18) or 7(2). Dorsolateral body plates between adipose- and caudal-fin 7(18) or 8*(2). Preadipose platelets 3*(6) or 4(14). Ventral surface of trunk between posteroventral margin of cleithrum and pelvic-fin origin laterally delimited only by first ventrolateral body plate; ventral portion of first ventrolateral body plate slightly expanded anteriorly. Small platelets covering base of caudal-fin rays. Small platelets disposed dorsally and ventrally between junctions of lateral plates on posterior portion of caudal peduncle. Anterior margin of orbit, above region of junction between frontal and lateral ethmoid, ventral margin of nasal capsule, above lateral ethmoid, and dorsal surface of snout with numerous small- to relatively large-sized platelets bearing odontodes; platelets on anterior margin of orbit and ventral margin of nasal capsule typically larger and coalescent. Ventral surface of trunk with scarce to relatively numerous small-sized irregular platelets bearing odontodes; platelets more concentrated anteriorly and/or around pectoral-fin base.
Vertebral count 21(3); ribs 5(3); first pair conspicuously large, its middle portion closely connected to first ventrolateral body plate; its tip connected to anterior external process of basipterygium. Parapophysis of complex vertebra well developed.
Morphometric data of the holotype and 19 paratypes of Corydoras hypnos. SD = Standard deviation.
Lateral view of the head (A), lateral view of the dorsal-fin spine (B) and dorsal view of the left pectoral-fin spine (C) in cs paratype of Corydoras hypnos, CITL 385, 32.0 mm SL. Abbreviations: f: frontal, io1–2: infraorbital 1 and 2, iop: interopercle, n: nasal, op: opercle, pes: pterotic-extrascapular, pop: preopercle, prh: posterodorsal ridge of hyomandibula, pso: parieto-supraoccipital, sph: sphenotic. Scale bars = 1 mm.
Color in alcohol. Overall color of body in Fig. 7. Ground color of body pale- to brownish yellow or beige. Top of head dark brown; dark area typically extending mesially towards posterior tip of parieto-supraoccipital as thin longitudinal line. Dorsal and lateral surface of head, and lateral surface of cleithrum covered by dark brown or black chromatophores, typically not forming blotches; diffuse stripe from anteroventral portion of eye to upper lip lateral area in some specimens. Dorsolateral body plates with conspicuous concentration of dark brown or black chromatophores, typically forming rounded, irregular or vertically elongated diffuse dark blotches. Ventrolateral body plates, except for ventral portion of plates between pelvic-fin origin to area just anterior to anal-fin origin, with conspicuous concentration of dark brown or black chromatophores, typically forming rounded, irregular or vertically elongated diffuse dark blotches; ventral portion of plates between pelvic-fin origin to area just anterior to anal-fin origin, especially around pelvic-fin origin, typically with sparse dark brown or black chromatophores, not forming any conspicuous pattern. Midline of flank generally with distinct longitudinal series of typically diffuse dark brown or black blotches, generally more evident on posterior half of flank. Blotches on flanks ranging from small to moderate in size; strongly faded in some specimens. Dorsal portion of body with conspicuous concentration of dark brown or black chromatophores at anterior and posterior origins of dorsal fin, adipose-fin base, and caudal-fin base, variably forming longitudinal series of four typically diffuse dark brown or black blotches. Posterior margin of body plates typically with dark brown or black chromatophores, variably forming thin dark lines along border of plates. Dorsal-fin with conspicuous concentrations of dark brown or black chromatophores, forming small dark blotches; blotches roughly longitudinally or obliquely aligned in some specimens, variably fused with each other and forming stripes; blotches diffuse in some specimens. Pectoral and pelvic fins with sparse dark brown or black chromatophores, generally more concentrated on rays and not forming dark spots. Adipose fin with dark brown or black chromatophores, generally more numerous and concentrated on spine and on posterior portion of membrane, typically forming single dark patch; diffuse patch in some specimens. Anal fin with conspicuous concentrations of dark brown or black chromatophores, generally more evident on its anterior half, typically forming diffuse or conspicuous dark blotches roughly aligned transversally; blotches fused with each other, forming dark stripe in some specimens. Caudal fin with conspicuous concentrations of dark brown or black chromatophores, mainly on rays, forming dark blotches roughly aligned transversally in two to five slender bars; some specimens with diffuse blotches/bars.
Color in life. Similar to color pattern of preserved specimens, but with light ground color of body and slightly more evident stripe from anteroventral portion of eye to upper lip lateral area. Body covered by greenish yellow iridescent coloration (Fig. 9). Aquarium specimens were observed to present dorsal and lateral surface of head plus lateral surface of cleithrum with irregular, rounded or vermiculated, conspicuous dark brown or black blotches, and conspicuous blotches on body. Some specimens with ground color of body greyish yellow (Fig. 10).
Paratypes of Corydoras hypnos photographed alive, showing general color pattern and morphology in lateral view in (A) INPA 59775, 26.2 mm SL, (B) INPA 59775, 27.2 mm SL, (C) INPA 59775, 22.2 mm SL, (D) INPA 59774, 28.2 mm SL, and (E) INPA 59774, 22.6 mm SL, all from the rio Jamanxim, rio Tapajós basin, Pará State, Brazil.
Uncatalogued aquarium specimens of Corydoras hypnos photographed alive, showing general morphology and color pattern of (A) a specimen with brownish yellow ground color of body, and (B) and two specimens with greyish yellow ground color of body. Photos by Robert McLure (A) and Oliver Lucanus (B).
Sexual dimorphism. Similar to the described for Corydoras thanatos, except by the absence of elongated dorsal fin in males, which was not observed so far in both wild caught or aquarium specimens.
Geographical distribution.Corydoras hypnos is currently known from the rio Jamanxim and one of its tributaries, with unknown name, rio Tapajós basin, Novo Progresso Municipality, Pará State, Brazil (Fig. 5).
Ecological notes. Specimens of Corydoras hypnos were always observed by the authors in syntopy and shoaling together with C. thanatos, information on its natural history and habitat notes can be found in the “Ecological notes” section of C. thanatos.
Etymology. The specific epithet “hypnos” refers to Hypnos (from the Greek Ύπνος, Ýpnos, which means “sleep”), the Greek god or personification of sleep, and the twin brother of Thanatos, the god/personification of death. The name makes an allusion to the fact that even though both C. hypnos and C. thanatos may present some similarities (in color pattern), they are completely different in other aspects (general morphological pattern). A noun in apposition.
Conservation status.Corydoras hypnos is currently known from its type locality and an additional record in its surroundings. However, both records are in the border of a conservation unit, the Floresta Nacional do Jamanxim, and no threat to the species as a whole is currently suspected. According to the International Union for Conservation of Nature (IUCN) categories and criteria (IUCN Standards and Petitions Subcommittee, 2019International Union for Conservation of Nature (IUCN). Standards and Petitions Subcommittee. Guidelines for using the IUCN red list categories and criteria. Version 14 [Internet]. Prepared by the Standards and Petitions Subcommittee; 2019. Available from: http://www.iucnredlist.org/documents/RedListGuidelines.pdf
http://www.iucnredlist.org/documents/Red...
), Corydoras hypnos can be classified as Least Concern (LC).
Corydoras psamathos, new species
urn:lsid:zoobank.org:act:76871FE9-BC24-4B3D-99DA-0249A1E672C7
Holotype. MNRJ 53289, 29.4 mm SL, Brazil, Pará State, Novo Progresso Municipality, igarapé Santa Júlia, a tributary to the rio Jamanxim, rio Tapajós basin, 06°45’38”S 55°28’44”W, 10 Jul 2019, M. R. Britto, W. M. Ohara and L. F. C. Tencatt.
Paratypes. All from Brazil, Pará State, Novo Progresso Municipality, rio Jamanxim basin, rio Tapajós drainage, collected by M. R. Britto, W. M. Ohara and L. F. C. Tencatt. CITL 386, 3, 18.1–21.8 mm SL; INPA 59772, 1, 23.7 mm SL, rio Jamanxim, 07°03’52”S 55°26’28”W, 9 Jul 2019. CITL 387, 9 of 12, 19.3–29.6 mm SL, 3 cs of 12, 22.0–28.6 mm SL; INPA 59773, 2, 29.3–30.2 mm SL; MZUSP 126862, 10, 19.6–27.8 mm SL; NUP 23542, 10, 18.7–26.9 mm SL, collected with holotype.
Corydoras psamathos, holotype, MNRJ 53289, 29.4 mm SL, Novo Progresso Municipality, Pará State, Brazil, igarapé Santa Júlia, a tributary to the rio Jamanxim, rio Tapajós basin.
Diagnosis.Corydoras psamathos can be distinguished from its congeners, except for the species from the lineages 6, 7 and 9 sensuAlexandrou et al., (2011)Alexandrou MA, Oliveira C, Maillard M, McGill RAR, Newton J, Creer S et al. Competition and phylogeny determine community structure in Müllerian co-mimics. Nature. 2011; 469:84–89. https://doi.org/10.1038/nature09660
https://doi.org/10.1038/nature09660...
plus C. difluviatilis, C. hastatus and C. pygmaeus, by having posterior margin of pectoral-fin spine with most serrations directed towards the tip of the spine (vs. most serrations directed towards origin of spine); from C. difluviatilis and C. pygmaeus it differs by the presence of contact between nuchal plate and posterior process of the parieto-supraoccipital (vs. absence of contact between nuchal plate and the posterior process of the parieto-supraoccipital); from C. hastatus by the absence of a large-sized dark blotch on caudal-fin base (vs. caudal-fin base region with a dark brown or black large blotch, roughly diamond-shaped or rhomboid, or arrow-shaped, typically bordered anteriorly and posteriorly by conspicuously light areas); from the species within lineage 7, it differs by having posterodorsal edge of infraorbital 2 only in contact with sphenotic (vs. in contact with sphenotic and pterotic-extrascapular); from the species of lineages 6 and 9, except for C. concolor Weitzman, 1961, C. esperanzae Castro, 1987, C. guianensis, C. polystictusRegan, 1912Regan CT. A revision of the South-American siluroid fishes of the genus Corydoras, with a list of the specimens in the British Museum (Natural History). Ann Mag Nat Hist. 1912; 10(56):209–20., and C. sanchesi Nijssen & Isbrücker, 1967, by the absence of conspicuous dark brown or black markings on body; dark markings, when present, diffuse (vs. presence of conspicuous small- to large- sized dark brown or black markings in at least some region of the body); from C. concolor, C. esperanzae, C. guianensis, C. polystictus, and C. sanchesi by having posterior laminar expansion of infraorbital 2 strongly reduced, nearly imperceptible in some specimens (vs. posterior laminar expansion at least poorly developed, but clearly more developed and perceptible), by the slender body (vs. more robust), and by the narrower frontal bone (vs. wider). The new species can be further distinguished from C. concolor and C. esperanzae by the presence of ventral surface of trunk only with small-sized, irregular platelets (vs. ventral surface of trunk entirely or almost entirely covered by larger coalescent platelets); from C. sanchesi by having anterior portion of dorsal fin with scattered dark brown or black chromatophores, not forming any conspicuous pattern (vs. anterior portion of dorsal fin typically with a conspicuous concentration of dark brown or black chromatophores, forming a dark patch); from C. polystictus by presenting dorsal surface of snout with numerous platelets bearing odontodes (vs. dorsal surface of snout lacking platelets).
Description. Morphometric data in Tab. 3. Head laterally compressed with convex dorsal profile, roughly triangular in dorsal view. Snout ranging from relatively short to moderately developed and generally smoothly rounded; slightly more rounded in some specimens. Head profile convex from tip of snout to anterior nares; ascending nearly straight or slightly convex from this point to dorsal-fin origin; region of frontal fontanel slightly concave in some specimens. Profile slightly convex along dorsal-fin base. Postdorsal-fin body profile slightly concave to adipose-fin spine, slightly concave from this point to caudal-fin base. Ventral profile of body nearly straight or slightly convex from isthmus to pectoral girdle, and slightly convex from this point until pelvic girdle. Profile nearly straight or slightly convex from pelvic girdle to base of first anal-fin ray, ascending slightly concave until caudal-fin base. Body roughly elliptical in cross section at pectoral girdle, gradually becoming more compressed toward caudal fin.
Eye rounded, located dorsolaterally on head. Orbit delimited anteriorly by lateral ethmoid, anterodorsally by frontal, posterodorsally by sphenotic, posteroventrally by infraorbital 2, and anteroventrally by infraorbital 1. Anterior and posterior nares close to each other, only separated by flap of skin. Anterior naris tubular. Posterior naris close to anterodorsal margin of orbit, separated from it by distance similar to naris diameter. Mouth small, subterminal, width similar to bony orbit diameter. Maxillary barbel ranging from short to moderate in size, not reaching to anteroventral limit of gill opening. Outer mental barbel slightly longer than maxillary barbel. Inner mental barbel fleshy, base of each counterpart slightly separated from each other. Small rounded papillae covering entire surface of all barbels, upper and lower lips, snout and isthmus.
Mesethmoid moderate in size, with anterior tip poorly developed, slightly smaller than 50% of bone length (see Britto, 2003Britto MR. Phylogeny of the subfamily Corydoradinae Hoedeman, 1952 (Siluriformes: Callichthyidae), with a definition of its genera. Proc Acad Nat Sci Phila. 2003; 153(1):119–54. https://doi.org/10.1635/0097-3157(2003)153[0119:POTSCH]2.0.CO;2
https://doi.org/10.1635/0097-3157(2003)1...
:123, character 1, state 1; fig. 1B); posterior portion wide, partially exposed and bearing small odontodes. Nasal capsule delimited posteriorly and dorsally by frontal, anteriorly by mesethmoid, and ventrally and posteriorly by lateral ethmoid. Nasal slender, laterally curved, inner margin with poorly- to moderately-developed laminar expansion generally contacting frontal and mesethmoid; variably close but not in direct contact with mesethmoid; outer margin typically with strongly reduced laminar expansion. Lateral ethmoid slightly expanded anteriorly, with anterodorsal expansion relatively distant from nasal, and anterior margin contacting posterior portion of mesethmoid. Frontal elongated, narrow, width less than half of entire length; anterior projection short, size smaller than nasal length. Frontal fontanel large, slender, and somewhat ellipsoid; posterior tip extension slightly surpassing anterior margin of parieto-supraoccipital. Sphenotic somewhat trapezoid, contacting parieto-supraoccipital dorsally, pterotic-extrascapular posteriorly, second infraorbital posteroventrally and frontal anteriorly (Fig. 12A). Pterotic-extrascapular roughly pipe-shaped, with posteriormost portion contacting first lateral-line ossicle, posteroventral margin contacting cleithrum, and anteroventral margin contacting opercle; posterior expansion almost entirely covering lateral opening of swimbladder capsule, leaving slender area on its dorsal margin covered only by thick layer of skin. Parieto-supraoccipital wide, posterior process long and contacting nuchal plate; region of contact between posterior process and nuchal plate covered by thick layer of skin.
Two laminar infraorbitals with minute odontodes. Infraorbital 1 large, ventral laminar expansion ranging from poorly to moderately developed; anterior portion with laminar expansion moderately developed, reaching to middle portion of nasal capsule; inner laminar expansion poorly developed (Fig. 12A). Infraorbital 2 small, slender, with posterior laminar expansion strongly reduced, nearly imperceptible in some specimens; posteroventral margin contacting posterodorsal ridge of hyomandibula, posterodorsal edge contacting only sphenotic; inner laminar expansion ranging from strongly reduced to poorly developed (Fig. 12A). Posterodorsal ridge of hyomandibula close to its articulation with opercle relatively slender, exposed, and bearing small odontodes. Dorsal ridge of hyomandibula between pterotic-extrascapular and opercle typically covered by thick skin layer. Interopercle entirely covered by thick layer of skin; subtriangular, anterior projection ranging from moderately developed to relatively well developed. Preopercle elongated, relatively slender; minute odontodes on external surface. Opercle dorsoventrally elongated, with width similar to half of its entire length; free margin slightly convex, without serrations and covered by small odontodes.
Four branchiostegal rays decreasing in size posteriorly. Hypobranchial 1 deep; hypobranchial 2 somewhat triangular, tip ossified and directed towards anterior portion, posterior margin cartilaginous; ossified portion ranging from strongly reduced, with only region of tip ossified, to well developed, its size about twice of cartilaginous portion. Five ceratobranchials with expansions increasing posteriorly; ceratobranchial 1 with small process on anterior margin of mesial portion; ceratobranchial 3 typically notched on postero-lateral margin; ceratobranchial 5 toothed on posterodorsal surface, with 28 to 38(3) teeth aligned in one row. Four epibranchials with similar size; epibranchial 2 slightly larger than others, with small pointed process on laminar expansion of posterior margin; epibranchial 3 with mesially-curved uncinate process on laminar expansion of posterior margin. Two wide pharyngobranchials (3 and 4); pharyngobranchial 3 with slightly triangular to smoothly rounded laminar expansion on posterior margin. Upper tooth plate roughly oval, 34 to 43(3) teeth aligned in two rows on posteroventral surface; rows closely aligned.
Lateral-line canal reaching cephalic laterosensory system through pterotic-extrascapular, branching twice before reaching sphenotic: pterotic branch, with single pore, preoperculomandibular branch conspicuously reduced, with single pore opening at postotic main canal; postotic main canal widens just posterior to pterotic branch. Sensory canal continuing through pterotic-extrascapular, reaching sphenotic as temporal canal, which splits into two branches: one branch giving rise to infraorbital canal, other branch connecting to frontal through supraorbital canal, both with single pore. Supraorbital canal branched, running through nasal bone. Epiphyseal branch conspicuously reduced; pore opening close to supraorbital main canal, directed towards frontal fontanel. Nasal canal typically with three openings, first on posterior edge, second on posterolateral portion, generally fused with first pore, and third on anterior edge. Infraorbital canal running through entire infraorbital 2, extending to infraorbital 1 and generally opening into two pores. Preoperculomandibular branch giving rise to preoperculo-mandibular canal, which runs through entire preopercle with three openings, leading to pores 3, 4, and 5, respectively.
Dorsal fin subtriangular, generally located just posterior to second dorsolateral body plate. Dorsal-fin rays II,7*(1) or II,8(19), posterior margin of dorsal-fin spine with 10 to 16 strongly reduced to poorly-developed serrations; most serrations directed towards tip of spine; some serrations variably perpendicularly directed; serrations absent close to origin of spine; small odontodes on anterior and lateral surfaces of spine (Fig. 12B). Nuchal plate moderately developed, almost entirely exposed, with minute odontodes. Spinelet short; spine moderately developed, with adpressed distal tip slightly surpassing posterior origin of dorsal-fin base. Pectoral fin roughly triangular, its origin just posterior to gill opening. Pectoral-fin rays I,7*(5), I,7,I(3) or I,8(12), posterior margin of pectoral spine with 17 to 26 strongly reduced to moderately-developed serrations along its entire length; most serrations directed towards tip of spine; some serrations perpendicularly directed; small odontodes on anterior, dorsal and ventral surfaces of spine (Fig. 12C). Anteroventral portion of cleithrum exposed; posterolateral portion of scapulocoracoid moderately developed, exposed, with anterior portion slightly expanded anteriorly, not in contact with anteroventral portion of cleithrum; exposed areas bearing small odontodes. Opening of axillary gland sensuKiehl et al., (2006)Kiehl E, Rieger C, Greven H. Axillary gland secretions contribute to the stress-induced discharge of a bactericidal substance in Corydoras sterbai (Callichthyidae, Siluriformes). Verh Ges Ichthyol. 2006; 5:111–15. located just posterior to pectoral-fin spine base. Pelvic fin oblong, located just below first or second ventrolateral body plate, and at vertical through first branched dorsal-fin ray. Pelvic-fin rays I,5*(20). Adipose fin roughly triangular, separated from base of last dorsal-fin ray by generally six dorsolateral body plates. Anal fin subtriangular, typically located just posterior to 12th or 13th ventrolateral body plates, and at vertical through adipose-fin spine base. Anal-fin rays ii,5(18), ii,5,i*(1), ii,7(1). Caudal fin bilobed, with dorsal and ventral lobes similar in size or dorsal lobe slightly larger than ventral lobe. Caudal-fin rays I,12,i*(20), generally five dorsal and ventral procurrent rays.
Typically, three laterosensory canals on trunk; first ossicle tubular, second ossicle laminar, both bearing small odontodes; third, encased in third dorsolateral body plates. Body plates with minute odontodes scattered over exposed area, with conspicuous line of odontodes confined to posterior margins. Dorsolateral body plates 23(2), 24*(15) or 25(3). Ventrolateral body plates 21(15) or 22*(5). Dorsolateral body plates along dorsal-fin base 6*(17) or 7(3). Dorsolateral body plates between adipose- and caudal-fin 7(2), 8*(17) or 9(1). Preadipose platelets 3*(14) or 4(6). Ventral surface of trunk between posteroventral margin of cleithrum and pelvic-fin origin laterally delimited only by first ventrolateral body plate; ventral portion of first ventrolateral body plate slightly expanded anteriorly. Small platelets covering base of caudal-fin rays. Small platelets disposed dorsally and ventrally between junctions of lateral plates on posterior portion of caudal peduncle. Anterior margin of orbit, above region of junction between frontal and lateral ethmoid, ventral margin of nasal capsule, above lateral ethmoid, and dorsal surface of snout with numerous small- to relatively large-sized platelets bearing odontodes; platelets on anterior margin of orbit and ventral margin of nasal capsule typically larger and coalescent. Ventral surface of trunk with scarce small-sized irregular platelets bearing odontodes; platelets more concentrated anteriorly and/or around pectoral-fin base.
Vertebral count 22(3); ribs 5(3); first pair conspicuously large, its middle portion closely connected to first ventrolateral body plate; its tip connected to anterior external process of basipterygium. Parapophysis of complex vertebra well developed.
Morphometric data of the holotype and 19 paratypes of Corydoras psamathos. SD = Standard deviation.
Lateral view of the head (A), lateral view of the dorsal-fin spine (B) and dorsal view of the left pectoral-fin spine (C) in cs paratype of Corydoras psamathos, CITL 387, 28.6 mm SL. Abbreviations: f: frontal, io1–2: infraorbital 1 and 2, iop: interopercle, n: nasal, op: opercle, pes: pterotic-extrascapular, pop: preopercle, prh: posterodorsal ridge of hyomandibula, pso: parieto-supraoccipital, sph: sphenotic. Scale bars = 1 mm.
Color in alcohol. Overall color of body in Fig. 11. Ground color of body pale- to brownish yellow or beige. Top of head dark brown; dark area typically extending mesially towards posterior tip of parieto-supraoccipital as thin longitudinal line. Dorsal and lateral surface of head and lateral surface of cleithrum with conspicuous concentrations of dark brown or black chromatophores, typically not forming blotches; top of head with diffuse irregular dark blotches in some specimens; diffuse stripe from anteroventral portion of eye to upper lip lateral area typically present. Dorsolateral body plates with conspicuous concentration of dark brown or black chromatophores, typically not forming dark blotches; diffuse rounded, irregular or vertically elongated dark blotches in some specimens; dorsal portion of plates with slightly more concentrated chromatophores on anterior and posterior origins of dorsal fin, adipose-fin base, and caudal-fin base in some specimens. Ventrolateral body plates, except for ventral portion of plates between pelvic-fin origin to area just anterior to anal-fin origin, with conspicuous concentration of dark brown or black chromatophores, typically not forming dark blotches; diffuse rounded, irregular or vertically elongated dark blotches in some specimens; ventral portion of plates between pelvic-fin origin to area just anterior to anal-fin origin, especially around pelvic-fin origin, typically with sparse dark brown or black chromatophores, not forming any distinctive pattern. Midline of flank with distinct longitudinal series of diffuse dark brown or black blotches, more evident on posterior half of flank in single juvenile specimen (18.4 mm SL). Blotches on flanks typically small. Posterior margin of body plates typically with dark brown or black chromatophores, variably forming thin dark lines along border of plates. Dorsal-fin with dark brown or black chromatophores, especially on rays, generally not forming evident blotches; forming strongly faded to diffuse small dark blotches in some specimens; chromatophores typically more numerous and concentrated on dorsal half of first branched-ray region, including membranes, and along dorsal-fin proximal portion. Pectoral, pelvic, and anal fins with sparse to relatively numerous dark brown or black chromatophores, especially on rays, typically not forming dark spots; chromatophores on anal fin more concentrated on its proximal half. Adipose fin with conspicuous concentrations of dark brown or black chromatophores on spine; dark brown or black chromatophores on membrane typically more numerous and concentrated on its posterior margin and/or along its proximal portion, not forming any conspicuous pattern. Caudal fin with dark brown or black chromatophores, mainly on rays, typically forming closely-spaced, strongly faded to diffuse dark blotches; some specimens with blotches roughly aligned transversally in up to nine slender and closely-spaced rows of blotches; some specimens with non-aligned blotches; dark blotches variably absent.
Color in life. Similar to color pattern of preserved specimens, but with light ground color of body and slightly more evident stripe from anteroventral portion of eye to upper lip lateral area. Body covered by greenish yellow iridescent coloration (Fig. 13). Some aquarium specimens with greyish-white ground color of body (Fig. 14).
Paratypes of Corydoras psamathos photographed alive, showing general color pattern and morphology in lateral view in (A) INPA 59772, 23.7 mm SL, (B) INPA 59773, 30.2 mm SL, and (C) INPA 59773, 29.3 mm SL. Specimen (A) is from the rio Jamanxim, and specimens (B–C) are from the igarapé Santa Júlia, rio Tapajós basin.
Uncatalogued aquarium specimen of Corydoras psamathos photographed alive. Photo by Takayuki Shimizu.
Sexual dimorphism. Same as described for Corydoras hypnos.
Geographical distribution.Corydoras psamathos is currently known from the main channel of the rio Jamanxim and one of its tributaries, the igarapé Santa Júlia, rio Tapajós basin Novo Progresso Municipality, Pará State, Brazil (Fig. 5).
Ecological notes. The new species was observed to inhabit the rio Jamanxim and at least one of its tributaries, the igarapé Santa Júlia. In the rio Jamanxim, it was mostly associated with the margins of the river, where juvenile and adult specimens were observed in the shallow (up to about 20 cm deep) sandy shores with slow water flow. In the igarapé Santa Júlia (Fig. 15), juvenile and adult specimens were also observed associated in marginal shallow (up to about 20 cm deep) portions, with more complex substrate, mainly composed by sand, fine gravel and larger rocks. In this site, both juvenile and adult specimens of C. psamathos were observed shoaling together with juvenile specimens of Corydoras sp. CW171, being the new species clearly more abundant. Additionally, C. apiaka was observed syntopically with the new species and Corydoras sp. CW171 in the igarapé Santa Júlia. In both sampled sites, the new species was mostly captured at night.
Type locality of Corydoras psamathos, the igarapé Santa Júlia, a tributary to the rio Jamanxim, rio Tapajós basin in Pará State, Brazil.
Etymology. The specific epithet “psamathos” refers to Psamathos Psamathides, the oldest wizard among the Psamathists, the sand sorcerers, a fictional character created by J. R. R. Tolkien in his book Roverandom. The name derives from the Greek psammos, which means sand. The name alludes to the sand-dwelling behavior of the new species. A noun in apposition.
Conservation status.Corydoras psamathos is currently known from its type locality and an additional record from the same region. Although the region is severely impacted by anthropic action, no threat to the species as a whole is currently suspected. According to the International Union for Conservation of Nature (IUCN) categories and criteria (IUCN Standards and Petitions Subcommittee, 2019International Union for Conservation of Nature (IUCN). Standards and Petitions Subcommittee. Guidelines for using the IUCN red list categories and criteria. Version 14 [Internet]. Prepared by the Standards and Petitions Subcommittee; 2019. Available from: http://www.iucnredlist.org/documents/RedListGuidelines.pdf
http://www.iucnredlist.org/documents/Red...
), Corydoras psamathos can be classified as Least Concern (LC).
Identification key to the valid species of Corydoras from the rio Tapajós basin
1a. Posterior margin of pectoral-fin spine with most serrations directed towards the origin of the spine 2
1b. Posterior margin of pectoral-fin spine with most serrations directed towards the tip of the spine 3
2a. Two relatively wide, dark brown or black, longitudinal stripes on flanks C. bifasciatus
2b. Three or four slender, dark brown or black, longitudinal stripes on flanks C. ornatus
3a. Body devoid of conspicuous dark brown or black blotches 4
3b. Body with conspicuous dark brown or black blotches 5
4a. Posterior process of the parieto-supraoccipital not in contact with the nuchal plate C. hephaestus
4b. Posterior process of the parieto-supraoccipital in contact with the nuchal plate C. psamathos
5a. Broad, dark vertical bar at the level of eye present, occupying top of head, opercle, posterior portion of infraorbital 1 and entire infraorbital 2, and preopercle C. rikbaktsa
5b. Dark vertical bar at the level of eye absent 6
6a. Anterior laminar expansion of infraorbital 1 strongly well developed, conspicuously expanded towards the anteroventral portion of snout, significantly covering its lateral surface C. thanatos
6b. Anterior laminar expansion of infraorbital 1 ranging from moderately to well developed, not conspicuously expanded towards the anteroventral portion of snout, leaving most of its lateral surface uncovered 7
7a. Body depth 36.9–42.6% of SL; least interorbital distance (13.8–17.3% of HL) C. apiaka
7b. Body depth 30.0–35.7% of SL; least interorbital distance (18.8–27.7% of HL) 8
8a. Body with conspicuous dark brown or black markings C. benattii
8b. Body lacking conspicuous dark brown or black markings; dark markings, when present, diffuse C. hypnos
DISCUSSION
The presence of syntopic species sharing similar color patterns but distinct morphology has been widely reported for Corydoras (e.g., Nijssen, Isbrücker, 1980aNijssen H, Isbrücker IJH. A review of the genus Corydoras Lacépède, 1803 (Pisces, Siluriformes, Callichthyidae). Bijdr Dierkd. 1980a; 50:190–220.,cNijssen H, Isbrücker IJH. Three new Corydoras species from French Guiana and Brazil (Pisces, Siluriformes, Callichthyidae). Neth J Zool. 1980c; 30(3):494-503.; Britto, 2003Britto MR. Phylogeny of the subfamily Corydoradinae Hoedeman, 1952 (Siluriformes: Callichthyidae), with a definition of its genera. Proc Acad Nat Sci Phila. 2003; 153(1):119–54. https://doi.org/10.1635/0097-3157(2003)153[0119:POTSCH]2.0.CO;2
https://doi.org/10.1635/0097-3157(2003)1...
; Britto et al., 2009; Alexandrou et al., 2011Alexandrou MA, Oliveira C, Maillard M, McGill RAR, Newton J, Creer S et al. Competition and phylogeny determine community structure in Müllerian co-mimics. Nature. 2011; 469:84–89. https://doi.org/10.1038/nature09660
https://doi.org/10.1038/nature09660...
; Tencatt et al., 2013Tencatt LFC, Vera-Alcaraz HS, Britto MR, Pavanelli CS. A new Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio São Francisco basin, Brazil. Neotrop Ichthyol. 2013; 11(2):257–64. https://doi.org/10.1590/S1679-62252013000200003
https://doi.org/10.1590/S1679-6225201300...
, 2019Tencatt LFC, Lima FCT, Britto MR. Deconstructing an octogenarian misconception reveals the true Corydoras arcuatus Elwin 1938 (Siluriformes: Callichthyidae) and a new Corydoras species from the Amazon basin. J Fish Biol. 2019; 95(2):453–71. https://doi.org/10.1111/jfb.13980
https://doi.org/10.1111/jfb.13980...
, 2021Tencatt LFC, Santos SA, Evers HG, Britto MR. Corydoras fulleri (Siluriformes: Callichthyidae), a new catfish species from the rio Madeira basin, Peru. J Fish Biol. 2021; 99(2):614–28. https://doi.org/10.1111/jfb.14750
https://doi.org/10.1111/jfb.14750...
; Tencatt, Pavanelli, 2015Tencatt LFC, Pavanelli CS. Redescription of Corydoras guapore Knaack, 1961 (Siluriformes: Callichthyidae), a midwater Corydoradinae species from the rio Guaporé basin. Neotrop Ichthyol. 2015; 13(2):287–96. https://doi.org/10.1590/1982-0224-20150018
https://doi.org/10.1590/1982-0224-201500...
; Tencatt, Britto, 2016Tencatt LFC, Britto MR. A new Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio Araguaia basin, Brazil, with comments about Corydoras araguaiaensis Sands, 1990. Neotrop Ichthyol. 2016; 14(1):53–64. https://doi.org/10.1590/1982-0224-20150062
https://doi.org/10.1590/1982-0224-201500...
; Tencatt, Ohara, 2016aTencatt LFC, Ohara WM. A new long-snouted species of Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio Madre de Dios basin. Zootaxa. 2016a; 4144(3):430–42. https://doi.org/10.11646/zootaxa.4144.3.9
https://doi.org/10.11646/zootaxa.4144.3....
,bTencatt LFC, Ohara WM. Two new species of Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio Madeira basin, Brazil. Neotrop Ichthyol. 2016b; 14(1):139–54. https://doi.org/10.1590/1982-0224-20150063
https://doi.org/10.1590/1982-0224-201500...
; Lima, Sazima, 2017Lima FCT, Sazima I. Corydoras desana, a new plated catfish from the upper rio Negro, Brazil, with comments on mimicry within Corydoradinae (Ostariophysi: Siluriformes: Callichthyidae). Aqua. 2017; 23(1):19–32.), having almost certainly played an important role in the evolution of the group (Tencatt et al., 2021Tencatt LFC, Santos SA, Evers HG, Britto MR. Corydoras fulleri (Siluriformes: Callichthyidae), a new catfish species from the rio Madeira basin, Peru. J Fish Biol. 2021; 99(2):614–28. https://doi.org/10.1111/jfb.14750
https://doi.org/10.1111/jfb.14750...
). These convergent color patterns were generally reported as cases of Müllerian mimicry (Alexandrou et al., 2011Alexandrou MA, Oliveira C, Maillard M, McGill RAR, Newton J, Creer S et al. Competition and phylogeny determine community structure in Müllerian co-mimics. Nature. 2011; 469:84–89. https://doi.org/10.1038/nature09660
https://doi.org/10.1038/nature09660...
), with a single mention to the Batesian mode of mimicry (Lima, Sazima, 2017Lima FCT, Sazima I. Corydoras desana, a new plated catfish from the upper rio Negro, Brazil, with comments on mimicry within Corydoradinae (Ostariophysi: Siluriformes: Callichthyidae). Aqua. 2017; 23(1):19–32.). During the fieldwork, it was possible to observe C. thanatos and C. hypnos, which share similar color patterns but clearly different general morphological patterns, shoaling together in some sites. The same was observed between C. psamathos and Corydoras sp. CW171. Considering that the four species present the defensive tripod formed by their pungent dorsal and pectoral spines (Fine, Ladich, 2003Fine ML, Ladich F. Sound production, spine locking and related adaptations. In: Arratia G, Kapoor BG, Chardon M, Diogo R, editors. Catfishes. Enfield: Science Publishers; 2003. p.249–90.), allied to the presence of axillary glands sensuKiehl et al., (2006)Kiehl E, Rieger C, Greven H. Axillary gland secretions contribute to the stress-induced discharge of a bactericidal substance in Corydoras sterbai (Callichthyidae, Siluriformes). Verh Ges Ichthyol. 2006; 5:111–15., all of them can be considered as unpalatable. Therefore, it seems reasonable to conclude that these pairs of species compose two Müllerian mimicry rings (see Alexandrou et al., 2011Alexandrou MA, Oliveira C, Maillard M, McGill RAR, Newton J, Creer S et al. Competition and phylogeny determine community structure in Müllerian co-mimics. Nature. 2011; 469:84–89. https://doi.org/10.1038/nature09660
https://doi.org/10.1038/nature09660...
).
The Serra do Cachimbo is one of the highest elevations of the Brazilian Shield, located on the southernmost border of the Amazon basin. The mountain chain can reach 500 meters above the sea level and forms an arc, running from Southeast to Northwest dividing the headwaters of the middle rio Xingu basin to the East (rio Curuá basin) and the middle rio Tapajós basin to the West (headwaters of the rio Jamanxim basin and tributaries of the rio Teles Pires) (Goulding et al., 2003Goulding M, Barthem R, Ferreira E. The Smithsonian Atlas of the Amazon. Washington: Smithsonian Books; 2003.). Goulding et al., (2003)Goulding M, Barthem R, Ferreira E. The Smithsonian Atlas of the Amazon. Washington: Smithsonian Books; 2003. identified the Serra do Cachimbo as an area with high endemism, a theory proven in following years by the description of a relatively high number of fish species reported as endemic to this region (e.g., Lima, Birindelli, 2006Lima FCT, Birindelli JLO. Moenkhausiapetymbuaba, a new species of characid from the Serra do Cachimbo, Rio Xingu basin, Brazil (Characiformes: Characidae). Ichthyol Explor Freshw. 2006; 17(1):53–58.; Birindelli, Britski, 2009Birindelli JLO, Britski HA. New species of the genus Leporinus Agassiz (Characiformes: Anostomidae) from the rio Curuá, rio Xingu basin, Serra do Cachimbo, Brazil, with comments on Leporinus reticulatus. Neotrop Ichthyol. 2009; 7(1):1–09. https://doi.org/10.1590/S1679-62252009000100001
https://doi.org/10.1590/S1679-6225200900...
; Birindelli et al., 2009Birindelli JLO, Zanata AM, Sousa LM, Netto-Ferreira AL. New species of Jupiaba Zanata (Characiformes: Characidae) from Serra do Cachimbo, with comments on the endemism of upper rio Curuá, rio Xingu basin, Brazil. Neotrop Ichthyol. 2009; 7(1):11–18. https://doi.org/10.1590/S1679-62252009000100002
https://doi.org/10.1590/S1679-6225200900...
; Sousa et al., 2010Sousa LM, Netto-Ferreira AL, Birindelli JL. Two new species of Moenkhausia Eigenmann (Characiformes: Characidae) from Serra do Cachimbo, Pará State, Northern Brazil. Neotrop Ichthyol. 2010; 8(2):255–64. https://doi.org/10.1590/S1679-62252010000200003
https://doi.org/10.1590/S1679-6225201000...
; Netto-Ferreira, 2012Netto-Ferreira AL. Three new species of Lebiasina (Characiformes: Lebiasinidae) from the Brazilian shield border at Serra do Cachimbo, Pará State, Brazil. Neotrop Ichthyol. 2012; 10(3):487–98. https://doi.org/10.1590/S1679-62252012000300002
https://doi.org/10.1590/S1679-6225201200...
; Netto-Ferreira et al., 2013Netto-Ferreira AL, Birindelli JLO, Sousa LM, Mariguela TC, Oliveira C. A new miniature characid (Ostariophysi: Characiformes: Characidae), with phylogenetic position inferred from morphological and molecular data. PLoS ONE. 2013; 8(1):e52098. https://doi.org/10.1371/journal.pone.0052098
https://doi.org/10.1371/journal.pone.005...
; Datovo, 2014Datovo A. A new species of Ituglanis from the Rio Xingu basin, Brazil, and the evolution of pelvic fin loss in trichomycterid catfishes (Teleostei: Siluriformes: Trichomycteridae). Zootaxa. 2014; 3790(3):466–76. https://doi.org/10.11646/zootaxa.3790.3.5
https://doi.org/10.11646/zootaxa.3790.3....
; Varella, Sabaj Pérez, 2014Varella HR, Sabaj Pérez MH. A titan among dwarfs: Apistogramma kullanderi, new species (Teleostei: Cichlidae). Ichthyol Explor Freshw. 2014; 25(3):243–58.; Varella, Britzke, 2016Varella HR, Britzke R. Apistogramma eleutheria and A. flavipedunculata, two new species of dwarf cichlids from the rio Curuá on Serra do Cachimbo, Brazil (Teleostei: Cichlidae). Ichthyol Explor Freshw. 2016; 27(1):81–95.; Garcia-Ayala et al., 2017Garcia-Ayala JR, Ohara WM, Pastana MNL, Benine RC. A new species of Brachychalcinus (Characiformes: Characidae) from the rio Xingu basin, Serra do Cachimbo, Brazil. Zootaxa. 2017; 4362(4):564–74. https://doi.org/10.11646/zootaxa.4362.4.5
https://doi.org/10.11646/zootaxa.4362.4....
; Oyakawa et al., 2018Oyakawa OT, Fichberg I, Rapp Py-Daniel L. Three new species of Harttia (Loricariidae: Loricariinae) from Serra do Cachimbo, Rio Xingu basin, Pará State, Northern Brazil. Zootaxa. 2018; 4387(1):75–90. https://doi.org/10.11646/zootaxa.4387.1.3
https://doi.org/10.11646/zootaxa.4387.1....
). Even though the specimens analyzed herein were not captured in the Serra do Cachimbo itself, they were recorded from an area in its surroundings (especially C. thanatos and C hypnos), in the upper portion of the rio Jamanxim, which drains part of the Serra do Cachimbo. This explains the reason why the three new species together with Corydoras sp. CW111 and CW171 are called the “Serra do Cachimbo Corydoras” in the aquarium trade (see Lucanus, 2021Lucanus O. Corydoras of the Serra do Cachimbo. AMAZONAS Magazine. 2021; 10(6):26–31. ).
Until recently, the area was relatively poorly sampled, but four major scientific expeditions were carried out in the early twenty-first century (for more details about those expeditions, see Birindelli et al., 2009Birindelli JLO, Zanata AM, Sousa LM, Netto-Ferreira AL. New species of Jupiaba Zanata (Characiformes: Characidae) from Serra do Cachimbo, with comments on the endemism of upper rio Curuá, rio Xingu basin, Brazil. Neotrop Ichthyol. 2009; 7(1):11–18. https://doi.org/10.1590/S1679-62252009000100002
https://doi.org/10.1590/S1679-6225200900...
). One of them was the PIPE Expedition, funded by the US National Science Foundation project “All Catfish Species Inventory” (DEB-0315963 PI José Birindelli), the first scientific endeavor to the Cachimbo that shared its results on the internet. In this way, photos of live specimens were published in a report-like website in 2008 (http://silurus.acnatsci.org/ACSI/field/Pipe_Expedition/Home.html), making the species of the region known around the world. Some species depicted on the website immediately got the attention of fish-keeping hobbyists around the world, such as Apistogramma sp. “gigas” (later described as Apistogramma kullanderi Varella & Sabaj Pérez, 2014) and Corydoras sp. “bonita” (= Corydoras sp. CW111), both reported from the rio Curuá.
The demand for these species by aquarists made exporters focus their efforts on catching these fishes in the Serra do Cachimbo region. Fishermen from several places in Brazil (as well as from Colombia and Peru) traveled to the area and extensively seined the tributaries of the rio Curuá in order to capture the new fishes. Apistogramma kullanderi and A. eleutheria Varella & Britzke, 2016 were exported in great numbers, mainly to Asia, but also to Europe and North America, and have been successfully bred in captivity (O. Lucanus, 2022, pers. comm.; LMS pers. obs.). However, the impact of so many individuals being removed from wild populations of species with restrict geographical distribution is yet to be fully understood. In a recent expedition to the rio Curuá tributaries at the Serra do Cachimbo, very few specimens of A. kullanderi were spotted during field observations (O. Lucanus, 2022, pers. comm.), and the habitat was considerably impacted by fishing activity (e.g., clearly overturned substrate, and disposed collection gear), in addition to the habitat degradation on the surroundings caused by the paving works of the BR-163 road, which runs close to these sites.
The absence of Corydoras CW111 in the most recent survey efforts, as well as reports from several fishermen of the scarcity of this species, brings a real concern. Is this species naturally rare, or was it overfished to supply the ornamental trade? Perhaps both factors combined. The capture of wild fishes to supply the ornamental fish trade is an activity that can be beneficial to both local human populations and ecosystems if made in a sustainable way (see Evers et al., 2019Evers H-G, Pinnegar JK, Taylor MI. Where are they all from? – sources and sustainability in the ornamental freshwater fish trade. J Fish Biol. 2019; 94(6):909–16. https://doi.org/10.1111/jfb.13930
https://doi.org/10.1111/jfb.13930...
; Sousa et al., 2021Sousa LM, Lucanus O, Arroyo-Mora JP, Kalacska M. Conservation and trade of the endangered Hypancistrus zebra (Siluriformes, Loricariidae), one of the most trafficked Brazilian fish. Glob Ecol Conserv. 2021; 27:e01570. https://doi.org/10.1016/j.gecco.2021.e01570
https://doi.org/10.1016/j.gecco.2021.e01...
). However, the over-exploitation of a species caused by the ornamental trade can lead to a series of negative impacts (Dey, 2016Dey VK. The global trade in ornamental fish. Infofish International. 2016; 4:52–55. Available from: www.infofish.org; Evers et al., 2019Evers H-G, Pinnegar JK, Taylor MI. Where are they all from? – sources and sustainability in the ornamental freshwater fish trade. J Fish Biol. 2019; 94(6):909–16. https://doi.org/10.1111/jfb.13930
https://doi.org/10.1111/jfb.13930...
; Sousa et al., 2021Sousa LM, Lucanus O, Arroyo-Mora JP, Kalacska M. Conservation and trade of the endangered Hypancistrus zebra (Siluriformes, Loricariidae), one of the most trafficked Brazilian fish. Glob Ecol Conserv. 2021; 27:e01570. https://doi.org/10.1016/j.gecco.2021.e01570
https://doi.org/10.1016/j.gecco.2021.e01...
), especially in the case of narrowly endemic species with naturally low abundance, which are often already affected by the local environmental degradation itself. This delicate balance between allowing local people to profit from native species found in their region, and affording an alternative income stream to the much more destructive practices (e.g., gold mining, logging, industrial agriculture) (for a broader discussion on this theme, see Evers et al., 2019Evers H-G, Pinnegar JK, Taylor MI. Where are they all from? – sources and sustainability in the ornamental freshwater fish trade. J Fish Biol. 2019; 94(6):909–16. https://doi.org/10.1111/jfb.13930
https://doi.org/10.1111/jfb.13930...
), and the potential damage to some species with small and/or geographically restricted populations, especially in the fragile headwater streams of the Xingu and Tapajós basins, is a difficult and elusive problem that requires further study and input from all stakeholders.
ACKNOWLEDGEMENTS
This work is dedicated to all donators of the Research Assistance Program (RAP), hosted by the “Corydoras World” website. The Universidade Estadual de Mato Grosso do Sul, the Laboratório de Zoologia of INBIO of Universidade Federal de Mato Grosso do Sul, and the Museu Nacional, Universidade Federal do Rio de Janeiro (MNRJ) provided logistical support. The authors are grateful to Carlos Lucena and Margarete Lucena (MCP), Claudio Oliveira (LBP), Mário de Pinna, Aléssio Datovo and Osvaldo Oyakawa (MZUSP), Kris Murphy, Sandra Raredon and Jeffrey Clayton (USNM), Mark Sabaj and Mariangeles Arce (ANSP), and Otávio Froehlich (in memoriam) (ZUFMS) for hosting museum visits and loaning of material. To Francisco Severo-Neto and Thomaz Sinani (ZUFMS-PIS), Carlos Lucena and Héctor Vera-Alcaraz (MCP), Cláudio Oliveira, Ricardo Britzke, Fábio Roxo, Bruno Melo and Gabriel Silva (LBP), Willian Ohara, Vinícius Espíndola and Túlio Teixeira (MZUSP) for generously welcoming LFCT during museum visits. To Lucélia Nobre de Carvalho and Andressa Costa (Laboratório de Ictiologia Tropical da Universidade Federal de Mato Grosso, campus Sinop) for logistical support to fieldwork. To Alêny Francisco and Alan Eriksson by permitting the use and by the assistance with the image capture equipment of the Laboratório de Ecologia of INBIO of Universidade Federal de Mato Grosso do Sul. To Hans Evers, Robert McLure, Oliver Lucanus, and Takayuki Shimizu for sending and allowing the use of photos in life of Corydoras thanatos, C. hypnos, and C. psamathos. To Steven Grant for kindly reviewing the English language of this manuscript. To the members of the fish keeping groups Corydoras World (United Kingdom), Catfish Study Group (United Kingdom), Internationale Gemeinschaft Barben Salmler Schmerlen Welse e.V. (Germany) and Potomac Valley Aquarium Society (United States of America) for the invaluable support to LFCT. The Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) provided financial support to LFCT (process #160674/2019–0), and MRB (process #309285/2018). The Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) also provided financial support to MRB (process #E–200.103/2019).
REFERENCES
- Alexandrou MA, Oliveira C, Maillard M, McGill RAR, Newton J, Creer S et al Competition and phylogeny determine community structure in Müllerian co-mimics. Nature. 2011; 469:84–89. https://doi.org/10.1038/nature09660
» https://doi.org/10.1038/nature09660 - Arratia G, Gayet M. Sensory canals and related bones of Tertiary siluriform crania from Bolivia and North America and comparison with recent forms. J Vertebr Paleontol. 1995; 15(3):482–505. https://www.jstor.org/stable/4523646
» https://www.jstor.org/stable/4523646 - Bentley RF, Grant S, Tencatt LFC. A new arc-striped species of Corydoras Lacépède, 1803 (Teleostei: Callichthyidae) from the Peruvian Amazon. Zootaxa. 2021; 4948(2):184–200. https://doi.org/10.11646/zootaxa.4948.2.2
» https://doi.org/10.11646/zootaxa.4948.2.2 - Birindelli JLO, Britski HA. New species of the genus Leporinus Agassiz (Characiformes: Anostomidae) from the rio Curuá, rio Xingu basin, Serra do Cachimbo, Brazil, with comments on Leporinus reticulatus Neotrop Ichthyol. 2009; 7(1):1–09. https://doi.org/10.1590/S1679-62252009000100001
» https://doi.org/10.1590/S1679-62252009000100001 - Birindelli JLO, Zanata AM, Sousa LM, Netto-Ferreira AL. New species of Jupiaba Zanata (Characiformes: Characidae) from Serra do Cachimbo, with comments on the endemism of upper rio Curuá, rio Xingu basin, Brazil. Neotrop Ichthyol. 2009; 7(1):11–18. https://doi.org/10.1590/S1679-62252009000100002
» https://doi.org/10.1590/S1679-62252009000100002 - Britto MR. Phylogeny of the subfamily Corydoradinae Hoedeman, 1952 (Siluriformes: Callichthyidae), with a definition of its genera. Proc Acad Nat Sci Phila. 2003; 153(1):119–54. https://doi.org/10.1635/0097-3157(2003)153[0119:POTSCH]2.0.CO;2
» https://doi.org/10.1635/0097-3157(2003)153[0119:POTSCH]2.0.CO;2 - Britto MR, Lima FCT Corydoras tukano, a new species of Corydoradine catfish from the rio Tiquié, upper rio Negro basin, Brazil (Ostariophysi: Siluriformes: Callichthyidae). Neotrop Ichthyol. 2003; 1(2):83–91. https://doi.org/10.1590/S1679-62252003000200002
» https://doi.org/10.1590/S1679-62252003000200002 - Dagosta FCP, de Pinna M. The Fishes of the Amazon: Distribution and biogeographical patterns, with a comprehensive list of species. Bull Am Mus Nat Hist. 2019; 2019(431):1–163. https://doi.org/10.1206/0003-0090.431.1.1
» https://doi.org/10.1206/0003-0090.431.1.1 - Datovo A. A new species of Ituglanis from the Rio Xingu basin, Brazil, and the evolution of pelvic fin loss in trichomycterid catfishes (Teleostei: Siluriformes: Trichomycteridae). Zootaxa. 2014; 3790(3):466–76. https://doi.org/10.11646/zootaxa.3790.3.5
» https://doi.org/10.11646/zootaxa.3790.3.5 - Dey VK. The global trade in ornamental fish. Infofish International. 2016; 4:52–55. Available from: www.infofish.org
- Eigenmann CH, Eigenmann RS. A revision of the South American Nematognathi or cat-fishes. Occas Pap Calif Acad Sci. 1890; 1:1–508.
- Ellis MD. The plated nematognaths. Anna Carnegie Mus. 1913; 8:384–413.
- Espíndola VC, Tencatt LFC, Pupo FM, Villa-Verde L, Britto MR. From the inside out: a new species of armoured catfish Corydoras with the description of poorly-explored character sources (Teleostei, Siluriformes, Callichthyidae). J Fish Biol. 2018; 92(5):1463–86. https://doi.org/10.1111/jfb.13602
» https://doi.org/10.1111/jfb.13602 - Evers H-G, Pinnegar JK, Taylor MI. Where are they all from? – sources and sustainability in the ornamental freshwater fish trade. J Fish Biol. 2019; 94(6):909–16. https://doi.org/10.1111/jfb.13930
» https://doi.org/10.1111/jfb.13930 - Fine ML, Ladich F. Sound production, spine locking and related adaptations. In: Arratia G, Kapoor BG, Chardon M, Diogo R, editors. Catfishes. Enfield: Science Publishers; 2003. p.249–90.
- Fricke R, Eschmeyer WN, Fong JD. Eschmeyer’s catalog of fishes: species by family/subfamily [Internet]. San Francisco: California; 2022. Available from: https://researcharchive.calacademy.org/research/ichthyology/catalog/SpeciesByFamily.asp.
» https://researcharchive.calacademy.org/research/ichthyology/catalog/SpeciesByFamily.asp. - Garcia-Ayala JR, Ohara WM, Pastana MNL, Benine RC. A new species of Brachychalcinus (Characiformes: Characidae) from the rio Xingu basin, Serra do Cachimbo, Brazil. Zootaxa. 2017; 4362(4):564–74. https://doi.org/10.11646/zootaxa.4362.4.5
» https://doi.org/10.11646/zootaxa.4362.4.5 - Gosline WA. A revision of the Neotropical catfishes of the family Callichthyidae. Stanford Ichthyol Bull. 1940; 2:1–36.
- Goulding M, Barthem R, Ferreira E. The Smithsonian Atlas of the Amazon. Washington: Smithsonian Books; 2003.
- Huysentruyt F, Adriaens D. Descriptive osteology of Corydoras aeneus (Siluriformes: Callichthyidae). Cybium. 2005; 29(3):261–73. https://doi.org/10.26028/cybium/2005-293-004
» https://doi.org/10.26028/cybium/2005-293-004 - International Union for Conservation of Nature (IUCN). Standards and Petitions Subcommittee. Guidelines for using the IUCN red list categories and criteria. Version 14 [Internet]. Prepared by the Standards and Petitions Subcommittee; 2019. Available from: http://www.iucnredlist.org/documents/RedListGuidelines.pdf
» http://www.iucnredlist.org/documents/RedListGuidelines.pdf - Jézéquel C, Tedesco PA, Darwall W, Dias MS, Frederico RG, Hidalgo M et al Freshwater fish diversity hotspots for conservation priorities in the Amazon Basin. Conserv Biol. 2020; 34(4):956–65. https://doi.org/10.1111/cobi.13466
» https://doi.org/10.1111/cobi.13466 - Kiehl E, Rieger C, Greven H. Axillary gland secretions contribute to the stress-induced discharge of a bactericidal substance in Corydoras sterbai (Callichthyidae, Siluriformes). Verh Ges Ichthyol. 2006; 5:111–15.
- Lacépède BGE. Histoire Naturelle des poissons. Paris: Chez Plassan; 1803.
- Lima FCT, Birindelli JLO Moenkhausiapetymbuaba, a new species of characid from the Serra do Cachimbo, Rio Xingu basin, Brazil (Characiformes: Characidae). Ichthyol Explor Freshw. 2006; 17(1):53–58.
- Lima FCT, Britto MR. A new Corydoras (Ostariophysi: Siluriformes: Callichthyidae) with an unusual sexual dimorphism from the rio Juruena basin, Brazil. Zootaxa. 2020; 4742(3):518–30. https://doi.org/10.11646/zootaxa.4742.3.6
» https://doi.org/10.11646/zootaxa.4742.3.6 - Lima FCT, Sazima I Corydoras desana, a new plated catfish from the upper rio Negro, Brazil, with comments on mimicry within Corydoradinae (Ostariophysi: Siluriformes: Callichthyidae). Aqua. 2017; 23(1):19–32.
- Lucanus O Corydoras of the Serra do Cachimbo. AMAZONAS Magazine. 2021; 10(6):26–31.
- Lundberg JG. The evolutionary history of North American catfishes, family Ictaluridae. [PhD Thesis]. Michigan: The University of Michigan; 1970.
- Netto-Ferreira AL. Three new species of Lebiasina (Characiformes: Lebiasinidae) from the Brazilian shield border at Serra do Cachimbo, Pará State, Brazil. Neotrop Ichthyol. 2012; 10(3):487–98. https://doi.org/10.1590/S1679-62252012000300002
» https://doi.org/10.1590/S1679-62252012000300002 - Netto-Ferreira AL, Birindelli JLO, Sousa LM, Mariguela TC, Oliveira C. A new miniature characid (Ostariophysi: Characiformes: Characidae), with phylogenetic position inferred from morphological and molecular data. PLoS ONE. 2013; 8(1):e52098. https://doi.org/10.1371/journal.pone.0052098
» https://doi.org/10.1371/journal.pone.0052098 - Nijssen H. Revision of the Surinam catfishes of the genus Corydoras Lacépède, 1803 (Pisces, Siluriformes, Callichthyidae). Beaufortia. 1970; 18:1–75.
- Nijssen H. Records of the catfish genus Corydoras from Brazil and French Guiana with descriptions of eight new species (Pisces, Siluriformes, Callichthyidae). Neth J Zool. 1972; 21:412–33.
- Nijssen H, Isbrücker IJH. Notes on the Guiana species of Corydoras Lacépède, 1803, with descriptions of seven new species and designation of a neotype for Corydoras punctatus (Bloch, 1794) (Pisces, Cypriniformes, Callichthyidae). Zool Meded. 1967; 42:21–50.
- Nijssen H, Isbrücker IJH. A review of the genus Corydoras Lacépède, 1803 (Pisces, Siluriformes, Callichthyidae). Bijdr Dierkd. 1980a; 50:190–220.
- Nijssen H, Isbrücker IJH Aspidorasvirgulatus n. sp., a plated catfish from Espírito Santo, Brazil (Pisces, Siluriformes, Callichthyidae). Bull Zoöl Mus Univ Amsterdam. 1980b; 7:133-39.
- Nijssen H, Isbrücker IJH. Three new Corydoras species from French Guiana and Brazil (Pisces, Siluriformes, Callichthyidae). Neth J Zool. 1980c; 30(3):494-503.
- Nijssen H, Isbrücker IJH. Sept espèces nouvelles de poissons-chats cuirassés du genre Corydoras Lacepède, 1803, de Guyane française, de Bolivie, d’Argentine, du Surinam et du Brésil (Pisces, Siluriformes, Callichthyidae). RFAH. 1983; 10:73–82.
- Nijssen H, Isbrücker IJH. Review of the genus Corydoras from Peru and Ecuador (Pisces, Siluriformes, Callichthyidae). Stud Neotrop Fauna Environ. 1986; 21(1–2):1–68.
- Oyakawa OT, Fichberg I, Rapp Py-Daniel L. Three new species of Harttia (Loricariidae: Loricariinae) from Serra do Cachimbo, Rio Xingu basin, Pará State, Northern Brazil. Zootaxa. 2018; 4387(1):75–90. https://doi.org/10.11646/zootaxa.4387.1.3
» https://doi.org/10.11646/zootaxa.4387.1.3 - Regan CT. A revision of the South-American siluroid fishes of the genus Corydoras, with a list of the specimens in the British Museum (Natural History). Ann Mag Nat Hist. 1912; 10(56):209–20.
- Reis RE. Revision of the Neotropical catfish genus Hoplosternum (Ostariophysi: Siluriformes: Callichthyidae), with the description of two new genera and three new species. Ichthyol Explor Freshw. 1997; 7:299–326.
- Reis RE. Anatomy and phylogenetic analysis of the neotropical callichthyid catfishes (Ostariophysi, Siluriformes). Zool J Linn Soc. 1998; 124(2):105–68. https://doi.org/10.1111/j.1096-3642.1998.tb00571.x
» https://doi.org/10.1111/j.1096-3642.1998.tb00571.x - Reis RE. Family Callichthyidae (Armored catfishes). In: Reis RE, Kullander SO, Ferraris Jr. CJ, organizers. Check list of the freshwater fishes of South and Central America. Porto Alegre: Edipucrs; 2003. p.291–309.
- Sabaj MH. Codes for Natural History Collections in Ichthyology and Herpetology. Copeia. 2020; 108(3):593–669. https://doi.org/10.1643/ASIHCODONS2020
» https://doi.org/10.1643/ASIHCODONS2020 - Schaefer AS. Homology and evolution of the opercular series in the loricarioid catfishes (Pisces: Siluroidei). J Zool. 1988; 214(1):81–93. https://doi.org/10.1111/j.1469-7998.1988.tb04988.x
» https://doi.org/10.1111/j.1469-7998.1988.tb04988.x - Schaefer SA, Aquino AE. Postotic laterosensory canal and pterotic branch homology in catfishes. J Morphol. 2000; 246(3):212–27. https://doi.org/10.1002/1097-4687(200012)246:3%3C212::aid-jmor5%3E3.0.co;2-s
» https://doi.org/10.1002/1097-4687(200012)246:3%3C212::aid-jmor5%3E3.0.co;2-s - Slobodian V, Pastana MNL. Description of a new Pimelodella (Siluriformes: Heptapteridae) species with a discussion on the upper pectoral girdle homology of Siluriformes. J Fish Biol. 2018; 93(5):901–16. https://doi.org/10.1111/jfb.13795
» https://doi.org/10.1111/jfb.13795 - Sousa LM, Netto-Ferreira AL, Birindelli JL. Two new species of Moenkhausia Eigenmann (Characiformes: Characidae) from Serra do Cachimbo, Pará State, Northern Brazil. Neotrop Ichthyol. 2010; 8(2):255–64. https://doi.org/10.1590/S1679-62252010000200003
» https://doi.org/10.1590/S1679-62252010000200003 - Sousa LM, Lucanus O, Arroyo-Mora JP, Kalacska M. Conservation and trade of the endangered Hypancistrus zebra (Siluriformes, Loricariidae), one of the most trafficked Brazilian fish. Glob Ecol Conserv. 2021; 27:e01570. https://doi.org/10.1016/j.gecco.2021.e01570
» https://doi.org/10.1016/j.gecco.2021.e01570 - Spadella MA, Desan SP, Henriques TCBPO, Oliveira C. Variation in male reproductive system characters in Corydoradinae (Loricarioidei: Callichthyidae) reflects the occurrence of different lineages in this subfamily. Neotrop Ichthyol. 2017; 15(1):e160039. https://doi.org/10.1590/1982-0224-20160039
» https://doi.org/10.1590/1982-0224-20160039 - Taylor WR, Van Dyke GC. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium. 1985; 9(2):107–19.
- Tencatt LFC, Vera-Alcaraz HS, Britto MR, Pavanelli CS. A new Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio São Francisco basin, Brazil. Neotrop Ichthyol. 2013; 11(2):257–64. https://doi.org/10.1590/S1679-62252013000200003
» https://doi.org/10.1590/S1679-62252013000200003 - Tencatt LFC, Pavanelli CS. Redescription of Corydoras guapore Knaack, 1961 (Siluriformes: Callichthyidae), a midwater Corydoradinae species from the rio Guaporé basin. Neotrop Ichthyol. 2015; 13(2):287–96. https://doi.org/10.1590/1982-0224-20150018
» https://doi.org/10.1590/1982-0224-20150018 - Tencatt LFC, Britto MR. A new Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio Araguaia basin, Brazil, with comments about Corydoras araguaiaensis Sands, 1990. Neotrop Ichthyol. 2016; 14(1):53–64. https://doi.org/10.1590/1982-0224-20150062
» https://doi.org/10.1590/1982-0224-20150062 - Tencatt LFC, Evers H-G. A new species of Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the río Madre de Dios basin, Peru. Neotrop Ichthyol. 2016; 14(1):13–26. https://doi.org/10.1590/1982-0224-20150019
» https://doi.org/10.1590/1982-0224-20150019 - Tencatt LFC, Ohara WM. A new long-snouted species of Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio Madre de Dios basin. Zootaxa. 2016a; 4144(3):430–42. https://doi.org/10.11646/zootaxa.4144.3.9
» https://doi.org/10.11646/zootaxa.4144.3.9 - Tencatt LFC, Ohara WM. Two new species of Corydoras Lacépède, 1803 (Siluriformes: Callichthyidae) from the rio Madeira basin, Brazil. Neotrop Ichthyol. 2016b; 14(1):139–54. https://doi.org/10.1590/1982-0224-20150063
» https://doi.org/10.1590/1982-0224-20150063 - Tencatt LFC, Lima FCT, Britto MR. Deconstructing an octogenarian misconception reveals the true Corydoras arcuatus Elwin 1938 (Siluriformes: Callichthyidae) and a new Corydoras species from the Amazon basin. J Fish Biol. 2019; 95(2):453–71. https://doi.org/10.1111/jfb.13980
» https://doi.org/10.1111/jfb.13980 - Tencatt LFC, Santos SA, Evers HG, Britto MR Corydoras fulleri (Siluriformes: Callichthyidae), a new catfish species from the rio Madeira basin, Peru. J Fish Biol. 2021; 99(2):614–28. https://doi.org/10.1111/jfb.14750
» https://doi.org/10.1111/jfb.14750 - Varella HR, Britzke R Apistogramma eleutheria and A. flavipedunculata, two new species of dwarf cichlids from the rio Curuá on Serra do Cachimbo, Brazil (Teleostei: Cichlidae). Ichthyol Explor Freshw. 2016; 27(1):81–95.
- Varella HR, Sabaj Pérez MH. A titan among dwarfs: Apistogramma kullanderi, new species (Teleostei: Cichlidae). Ichthyol Explor Freshw. 2014; 25(3):243–58.
- Vera-Alcaraz HS. Relações filogenéticas das espécies da família Callichthyidae (Ostariophysi, Siluriformes). [PhD Thesis]. Pontifícia Universidade Católica do Rio Grande do Sul; 2013.
ADDITIONAL NOTES
-
HOW TO CITE THIS ARTICLE
Tencatt LFC, Ohara WM, Sousa LM, Britto MR. Science and hobby joining forces for the discovery of three new Corydoras (Siluriformes: Callichthyidae) from the rio Tapajós basin, Brazil, with comments on Corydoras sp. CW111. Neotrop Ichthyol. 2022; 20(3):e220070. https://doi.org/10.1590/1982-0224-2022-0070
Edited-by
Publication Dates
-
Publication in this collection
14 Oct 2022 -
Date of issue
2022
History
-
Received
12 July 2022 -
Accepted
01 Sept 2022