Abstracts
It has been well established that, in addition to the pulmonary involvement, COPD has systemic consequences that can lead to peripheral muscle dysfunction, with greater muscle fatigue, lower exercise tolerance and lower survival in these patients. In view of the negative repercussions of early muscle fatigue in COPD, the objective of this review was to discuss the principal findings in the literature on the metabolic and bioenergy determinants of muscle fatigue, its functional repercussions, as well as the methods for its identification and quantification. The anatomical and functional substrate of higher muscle fatigue in COPD appears to include lower levels of high-energy phosphates, lower mitochondrial density, early lactacidemia, higher serum ammonia and reduced muscle perfusion. These alterations can be revealed by contraction failure, decreased firing rates of motor units and increased recruitment of motor units in a given activity, which can be functionally detected by a reduction in muscle strength, power and endurance. This review article also shows that various types of muscle contraction regimens and protocols have been used in order to detect muscle fatigue in this population. With this understanding, rehabilitation strategies can be developed in order to improve the resistance to muscle fatigue in this population.
Pulmonary disease, chronic obstructive; Neuromuscular manifestations; Muscle fatigue; Exercise tolerance; Energy metabolism; Evaluation
Está bem estabelecido que, além do acometimento pulmonar, a DPOC apresenta consequências sistêmicas que podem convergir para a disfunção muscular periférica, com maior fadigabilidade muscular, menor tolerância ao exercício e menor sobrevida nesses pacientes. Tendo em vista as repercussões negativas da fadiga muscular precoce na DPOC, esta revisão teve como objetivo discutir os principais achados da literatura relacionados aos seus determinantes metabólicos e bioenergéticos e suas repercussões funcionais, bem como seus métodos de identificação e de quantificação. O substrato anatomofuncional da maior fadigabilidade muscular na DPOC parece incluir a redução dos níveis de fosfatos de alta energia, a redução da densidade mitocondrial, a lactacidemia precoce, o aumento da amônia sérica e a perfusão muscular reduzida. Essas alterações podem ser evidenciadas por falência de contração, redução da taxa de disparo das unidades motoras e maior recrutamento de unidades motoras numa dada atividade, exteriorizando-se funcionalmente por redução da força, potência e resistência musculares. Esta revisão mostra também que diversos tipos de regimes contráteis e protocolos têm sido utilizados com o intuito de detectar fadiga nessa população. A partir de tais conhecimentos, estratégias reabilitadoras podem ser traçadas visando o aumento da resistência à fadiga muscular nessa população.
Doença pulmonar obstrutiva crônica; Manifestações neuromusculares; Fadiga muscular; Tolerância ao exercício; Metabolismo energético; Avaliação
REVIEW ARTICLE
*
Rafaella Rezende RondelliI; Simone Dal CorsoII; Alexandre SimõesIII; Carla MalagutiII
Methods for the assessment of peripheral muscle fatigue and its energy and metabolic determinants in COPD
ICollaborating Researcher. Masters Program in Rehabilitation Sciences, Nove de Julho University, São Paulo, Brazil
IIProfessor. Masters Program in Rehabilitation Sciences, Nove de Julho University, São Paulo, Brazil
IIIMasters Student. Masters Program in Rehabilitation Sciences, Nove de Julho University, São Paulo, Brazil
Correspondence to
ABSTRACT
It has been well established that, in addition to the pulmonary involvement, COPD has systemic consequences that can lead to peripheral muscle dysfunction, with greater muscle fatigue, lower exercise tolerance and lower survival in these patients. In view of the negative repercussions of early muscle fatigue in COPD, the objective of this review was to discuss the principal findings in the literature on the metabolic and bioenergy determinants of muscle fatigue, its functional repercussions, as well as the methods for its identification and quantification. The anatomical and functional substrate of higher muscle fatigue in COPD appears to include lower levels of high-energy phosphates, lower mitochondrial density, early lactacidemia, higher serum ammonia and reduced muscle perfusion. These alterations can be revealed by contraction failure, decreased firing rates of motor units and increased recruitment of motor units in a given activity, which can be functionally detected by a reduction in muscle strength, power and endurance. This review article also shows that various types of muscle contraction regimens and protocols have been used in order to detect muscle fatigue in this population. With this understanding, rehabilitation strategies can be developed in order to improve the resistance to muscle fatigue in this population.
Keywords: Pulmonary disease, chronic obstructive; Neuromuscular manifestations; Muscle fatigue; Exercise tolerance; Energy metabolism; Evaluation.
Introduction
Exercise intolerance constitutes a central aspect in COPD, being associated with progressive incapacitation and lower survival. The fundamental pathophysiological determinants are classically related to a chronic imbalance between the increased ventilatory demand and the reduced capacity to meet this demand.(1) Studies have shown that, in addition to the pulmonary involvement, peripheral muscle dysfunction (PMD) contributes to the reduced exercise capacity(2) and lower survival in these patients.(3)
Although the mechanisms related to PMD in COPD have yet to be clarified, PMD has been characterized by the presence of atrophy, loss of strength and power, in addition to early muscle fatigue, which explains the exercise intolerance in these patients.(2)
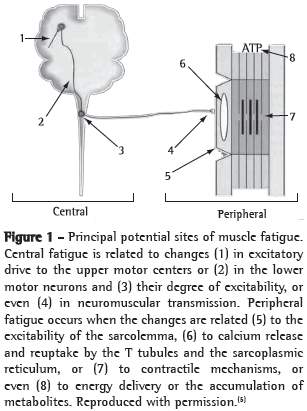
Classically, muscle fatigue is defined as the inability of the muscle to generate or maintain the levels of strength required for a given work rate.(4) Muscle fatigue can be divided into two components: central and peripheral. This terminology is based on the anatomical and functional sites involved in the fatigue process. In central fatigue, muscle activation by the central nervous system is impaired, whereas, in peripheral or contractile fatigue, the involvement occurs distally to the neuromuscular junction.(5) A pictorial representation of the structures involved in central and peripheral fatigue is presented in Figure 1.
The interest in studying peripheral muscle fatigue in COPD patients is very recent, since the scientific focus has always been on the contributions of respiratory muscle fatigue to and on its implications for exercise tolerance. A study conducted in the early 1990s can be considered the precursor of subsequent studies on peripheral muscle fatigue, having made a contribution in terms of exercise limitation, since lower limb fatigue was the predominant system when maximal exercise was interrupted in 43% of the sample of COPD patients of that study. At that time, the authors reported that it was an unexpected result.(6) In that same decade, another group of authors demonstrated that quadriceps muscle strength was an important determinant of physical capacity in this population.(7) These findings had a significant impact, furthering the study of the relationship between muscle performance and exercise capacity. More recently, one group of authors demonstrated that, in patients with moderate to severe COPD, quadriceps muscle strength is a better predictor of mortality than are age, body mass index and FEV1.(3)
In view of the negative repercussions of peripheral muscle fatigue in COPD, the objective of this review was to discuss the principal findings in the literature on the metabolic and bioenergy determinants of muscle fatigue, as well as the methods for its identification and quantification. The databases PubMed, Medline, SciELO and LILACS were searched using the following keywords: Pulmonary disease, chronic obstructive; Neuromuscular manifestations; Muscle fatigue; Exercise tolerance; Energy metabolism; and Evaluation.
Metabolic and bioenergy determinants of peripheral muscle fatigue in COPD
As mentioned previously, PMD contributes significantly to peripheral muscle fatigue. The impact of this dysfunction on muscle performance stems from abnormalities in muscle structure,(8) bioenergy(9) and function.(7) Some important clinical implications are related to changes in muscle structure (mass) and function (power and endurance), such as reduced exercise capacity, reduced quality of life and lower survival in these patients.(3,4,10)
The etiology of these abnormalities seems to be multifactorial, involving factors such as deconditioning, hypoxia or hypercapnia, oxidative stress, senescence, hormonal dysfunction, systemic inflammation, chronic or repetitive use of drugs (corticosteroids) and nutritional depletion.(2)
Local or systemic inflammatory factors have been identified in COPD. In stable patients, increased levels of C-reactive protein (CRP), fibrinogen, circulating leukocytes and proinflammatory cytokines, including TNF-α, IL-8 and IL-6, have been observed. Recently, increased serum IL-18 levels have also been identified in patients with moderate to severe COPD.(11-15)
The involvement of inflammatory mediators in PMD is suggested by the observation that systemic inflammatory markers are related to poor contractile muscle performance in COPD. For instance, quadriceps muscle strength was negatively associated with IL-8 levels during disease exacerbation(15) and with IL-6 and TNF-α levels in elderly individuals.(16) In critically ill patients, increased PCR levels were associated with a reduction in power, endurance and quality of life.(17)
Conversely, another study revealed that severe quadriceps muscle weakness was significantly related to the mean daily dose of steroids used by patients during acute disease exacerbation. The deleterious effects of steroids on skeletal muscle function have been attributed to the inhibition of protein synthesis and the increase in protein degradation. However, due to the fact that patients receive corticosteroids for the treatment of acute inflammation, it is difficult to differentiate the muscle effect of steroid administration from the direct deleterious effects of COPD exacerbation.(18)
Other evidence suggests the participation of oxidative stress in muscle dysfunction in COPD patients.(15) An imbalance between antioxidants and oxidants can be hypothesized. One group of authors established an inverse relationship between the magnitude of oxidative stress and quadriceps muscle endurance in COPD patients.(19) In addition, that same group showed that short-term therapy with high doses of N-acetylcysteine (an antioxidant) prevent oxidative stress and significantly improves quadriceps muscle endurance.(20)
Furthermore, it has been shown that systemic inflammation and hypoxemia are strongly associated with increased oxidative stress in COPD patients.(21) Chronic hypoxemia also seems to be involved in the reduction in muscle strength and endurance, increasing atrophy and attenuating mitochondrial enzymatic activity. It is also suggested that these effects are rapidly reversed by oxygen supplementation.(22)
Similarly, another group of authors showed that the levels of ATP, glycogen and phosphocreatine (PCr) are significantly reduced in the quadriceps muscle fibers in hypoxemic COPD patients.(23)
Senescence seems to be an important factor related to loss of muscle strength in healthy individuals. There is approximately a 30% decline in muscle strength and a 40% decline in muscle mass between the second and the seventh decade of life.(24) This decline in muscle strength is associated with the degree of type II fiber atrophy and, usually, is not limited to the lower limbs. Since COPD patients frequently have advanced age, the effects of this disease on skeletal muscle function should be considered.
Finally, the sedentary lifestyle adopted to prevent or minimize the sensation of dyspnea and muscle fatigue promotes muscle deconditioning, which results in weakness due to a reduction in motor neuron activity. This cascade culminates in a decrease in the proportion of type I fibers, an increase in the proportion of type IIx fibers and a reduction in the oxidation potential. Similarly, a significant correlation has been reported between physical inactivity and quadriceps muscle endurance in COPD patients.(25) It is worthy of note that these changes are more pronounced in the locomotor muscles in these patients.(2,10)
A recent systematic review has established that, based on reference values, there usually is a 27% reduction in the proportion of type I fibers and a 29% increase in the proportion of type IIx fibers in patients with severe COPD.(26)
The elevated baseline energy expenditure in activities of daily living suggests a high cost due to the increase in ventilatory demand.(2) In addition, the low protein-calorie intake, caused by symptoms of dyspnea, fatigue and early satiety, results in an imbalance between energy demand and energy consumption in these patients, contributing to an increase in their daily energy expenditure.(27) The reduction in body mass index in COPD patients correlates with a poor muscle contractile performance, suggesting that nutritional aspects play a key role in muscle function in these patients.(28)
One group of authors demonstrated that COPD patients with muscle mass depletion complained more of lower limb fatigue during the six-minute walk test than did COPD patients with preserved muscle mass.(29)
From a microstructural point of view, the histochemical and bioenergy changes observed in skeletal muscle can be summarized as follows: maintenance or increase in glycolytic enzyme activity; reduction in oxidative stress; decrease in capillary density; reduction in the cross-sectional area of slow and fast fibers; reduction in mitochondrial activity; and reduction in the capillary/fiber ratio. These changes increase the contribution of the anaerobic system to the generation of energy, resulting in early lactacidemia.(30)
In summary, the association of abnormalities in peripheral muscle structure, bioenergy and function results in significant peripheral muscle fatigue in these patients. This peripheral muscle fatigue can be intensified in cases of disease exacerbation.(7)Chart 1 presents a summary of the metabolic and bioenergy determinants of muscle fatigue in COPD patients.
Methods for the determination of peripheral muscle fatigue in COPD
Currently, there is a wide range of exercise models, tests and protocols being used in order to identify parameters indicative of muscle fatigue in COPD patients.(4)
Methods for the assessment of peripheral muscle fatigue involve three basic components:
i) muscle activation strategy: voluntary effort or exogenous stimulation
ii) exercise condition: isometric; isoinertial; or isokinetic
iii) exercise pattern: duration and intensity, either involving a single prolonged contraction or a series of repeated contractions
Most of the methods used in this assessment in COPD patients preferably focus on lower limb muscles. However, it is of note that upper limb function is maintained relatively preserved in these patients.
Maximal and submaximal voluntary contraction
The assessment of muscle fatigue through maximal voluntary contraction (MVC) is performed by instructing the patient to generate as much voluntary strength as possible, without changing muscle length. In this assessment, equipment such as tensiometers or dynamometers is used to quantify the isometric strength generated. Since this is a volitional technique, external complicating factors, such as the functional ability and motivation of the patient, can generate contractions using submaximal activation.(31,32) However, due to its user-friendliness, the use of verbal encouragement and the experience of the technician, little variability has been described in MVC. In COPD patients, the loss of the capacity to generate or maintain MVC during knee extension exercise has been representative of muscle fatigue.(7,31,32)
Studies have used series of submaximal voluntary contractions in the assessment of muscle fatigue in COPD patients.(25-28) Typically, it is required that relative values of approximately 20-60% of MVC be sustained until exhaustion. Endurance is measured by how long the subject can maintain the task. The test must be interrupted when there is a failure in maintaining the intensity of the contraction (a 5% decrease).(4,25,33)
Isokinetic dynamometry
Isokinetic movement consists of muscle contractions at a constant velocity throughout the movement. This type of contraction is only obtained by using isokinetic equipment, in which resistance is variable and accommodative, adapting itself to the strength applied. With this equipment, it is possible to determine the strength developed at each angle of movement and the duration of the contraction. Therefore, in addition to strength, it is possible to determine the variable work, which is the product of strength multiplied by velocity and power (i.e., work performed per unit of time).
When isokinetic assessment is performed, peak torque is the most appropriate term to express the maximal strength generated, since strength occurs due to the rotational movement of the joint in relation to an axis. In order to assess strength, lower angular velocities, typically 60-90º/s, are recommended.(34) In order to assess endurance, the most widely used angular velocities are 180º/s(30) and 300º/s.(7,34) Greater torque is generated when low angular velocities are applied. Therefore, in the assessment of strength, 3-5 repetitions are used, and the highest value obtained corresponds to peak torque. In the assessment of endurance, 15-30 successive repetitions are used, since lower peak torques are generated and a greater number of muscle contractions are possible.
Endurance test results can be interpreted in two ways: total work, which corresponds to the sum of the area under the muscle contraction curve, or a decrease in peak torque or work during 15, 20, 30 or 50 repetitions.(30) The decrease is calculated by dividing the peak torque or the work of the last five contractions by that of the first five, and this result is expressed in percentage. A greater decrease translates to greater fatigue.
In clinical populations, it is strongly recommended that total work be used, since the peak torque generated in each contraction when these patients start the test is already lower, and, therefore, its reduction is not pronounced.
The most obvious advantage of using isokinetic assessment, whose reliability and reproducibility are higher than those of conventional techniques, is the control of the velocity and angle of the movement.(35) Conversely, the disadvantages are related to the excessive cost, the need to train raters and the extremely limited external validity, since constant angular velocity does not constitute a physiological movement.(36)
Typically, in comparison with healthy controls, COPD patients were found to present lower isometric torque, lower isokinetic torque and lower total work.(30,37)
Surface electromyography
Surface electromyography (sEMG) is a widely used noninvasive method for the assessment of muscle function through the placement of electrodes on the skin. The study of muscle function through sEMG results from the analysis of the electrical activity of the muscle by determining the sum of the action potentials of all muscle fibers.(38,39) In addition to determining the electrical activity of the muscle, sEMG has been used to analyze fatigue, assess the training, determine the sEMG signal/strength ratio and identify pathological conditions.(40,41)
During muscle contraction, the electromyographic record basically provides two parameters: square root of the mean (SMR) and median frequency (MF). The SRM represents the electromyographic signal amplitude, that is, it quantifies the electrical activity during contraction. The MF is related to the firing rates of motor units. Therefore, muscle fatigue is present when, in the electromyographic analysis, there is(35):
higher SRM, which results from greater muscle activation due to a reduction in the capacity to sustain a contraction
lower MF due to a reduction in the action potential of the fibers during muscle contraction
The electromyographic signal resulting from this method is an action potential designated M-wave. The M-wave is proportional to the number of depolarized motor units and, consequently, is a reflex of the extent of the activation of muscle fibers produced during a muscle contraction. A reduction in contractile strength with unchanged M-wave is indicative of contractile fatigue, that is, excitation-contraction coupling failure.(42) However, when the reduction in contractile force is associated with a decrease in M-wave amplitude, transmission failure can be suspected. Therefore, the possible sites of muscle dysfunction during exercise can be determined.(4,35)
In pulmonary rehabilitation, sEMG has been widely used to assess muscle function, including during exercise.(43) In this context, one group of authors compared, through sEMG, the electrophysiological changes in the quadriceps femoris of COPD patients, who were slightly hypoxemic during exercise, with those of healthy individuals during cycle ergometer incremental exercise.(44) The COPD patients presented lower MF and higher SRM than did the control group patients. Subsequently, in a randomized study, another group of authors revealed changes in the electromyographic responses of the quadriceps femoris during exercise performed using oxygen. The same group suggested that those changes would be associated with improved aerobic metabolism, delayed fatigue, greater muscle excitability and greater muscle activation at a given level of exercise when using oxygen.(45)
In another study, COPD patients presented a lower capacity to sustain an isometric contraction of the quadriceps femoris at 60% of voluntary contraction until exhaustion than did healthy individuals (42 s and 80 s, respectively; p < 0.05). Despite the lower muscle resistance, the decrease in MF was similar in both groups.(33)
Twitch
Non-volitional techniques for the assessment of muscle fatigue have the advantage of ensuring maximal muscle activation, regardless of the cooperation of the individual. Twitch is an indirect method for the assessment of fatigue. In this method, the contraction is elicited by a single electrical or magnetic stimulus offered to the nerve.
A more sensitive assessment of fatigue by this method is twitch interpolation. In this technique, the individual is asked to perform a MVC, and, in addition, a stimulus is applied to the nerve. Concomitantly, electrical activity recording will show the additional recruitment of motor units by using twitch. A greater difference between MVC alone and MVC combined with twitch in terms of the muscle activity generated translates to greater muscle fatigue. A difference > 15% between MVC and interpolated twitch is characterized as contractile fatigue. In addition to allowing the differentiation between central fatigue and peripheral fatigue, this interpolated technique, in association with the M-wave, makes it possible to detect transmission failure or central activation failure.(4) In the scientific field, twitch is being currently used to further the understanding of the effects of dynamic exercise(32,46) and localized exercise on contractile fatigue of the quadriceps muscle in COPD patients.(31,43,47,48) However, despite being currently considered the gold standard in this assessment, this method is not yet available in clinical practice.(4)
Critical power
A physiological approach in the assessment of fatigue is the test of anaerobic capacity or power. This method attempts to assess the organic ability to provide energy regardless of the oxygen supply or of the use of oxygen, that is, the capacity to sustain predominantly anaerobic work. In this context, the concept of critical power (CP) involves the performance of high-intensity activities of short duration, and the limits of exercise tolerance are determined using a mathematical model. The clinical importance of this test consists in quantifying the workload at which fatigue occurs, that is, in determining the fatigue threshold.(37) Despite involving a series of dynamic and exhausting exercise tests, this protocol was feasible in COPD patients.(37,49) Therefore, one group of authors demonstrated that, in these patients, CP and anaerobic work capacity present different determinants from those observed in healthy controls, being dependent on airflow limitation and the consequent dyspnea sensation in COPD patients.(37)
Fatigue perception scales
The assessment of the subjective perception of fatigue by the patient during exercise tests or training tests has been commonly performed using scales. Although more than 30 scales can be found, the most widely used are the modified Borg scale(50) and the visual analog scale.(51) Although these scales are user-friendly, their outcomes can be influenced by various factors, such as motivation and understanding. Therefore, despite being widely used, these scales should be used in combination with other objective assessments.
Phosphorus-31 magnetic resonance spectroscopy
Phosphorus-31 magnetic resonance spectroscopy (31P-MRS) is a method for the direct and noninvasive measurement of muscle bioenergy (ATP, PCr, inorganic phosphate [IP] and intracellular pH) during repetitive localized exercise and during the subsequent recovery period. This method has been clinically used in the treatment of patients with severe COPD, with evidence of pronounced changes in their muscle metabolism. These changes are revealed by lower aerobic capacity ( PCr/PI and
PCr/PI and  PCr/[PCr +PI]) during exercise, as well as by lower intracellular pH and lower PCr resynthesis during the recovery period, when compared with those observed in controls.(52-54) It has also been shown that acute oxygen administration partially improves theses indices,(54) suggesting that factors other than hypoxemia affect muscle metabolism.
PCr/[PCr +PI]) during exercise, as well as by lower intracellular pH and lower PCr resynthesis during the recovery period, when compared with those observed in controls.(52-54) It has also been shown that acute oxygen administration partially improves theses indices,(54) suggesting that factors other than hypoxemia affect muscle metabolism.
Muscle biopsy
A study using techniques of vastus lateralis muscle biopsy revealed a higher strength/frequency ratio in patients with severe COPD, that is, these patients required a higher frequency of stimulation to produce the same relative vastus lateralis muscle strength than did normal controls.(55)
In addition, studies of muscle biopsy in this population have shown changes in enzymatic and metabolic activity, as well as changes in the proportion of muscle fiber types and in the relationship with exercise intolerance.(8,9)
Determination of lactate and serum ammonia levels
Blood lactate levels determined during dynamic exercise and during the recovery period indicate that the COPD patients who interrupted the exercise complaining of lower limb fatigue tended to present significantly higher lactate levels than did those who interrupted the exercise complaining of dyspnea. Therefore, one group of authors observed that, in patients with fatigue, there was a relationship between increased lactate levels and decreased quadriceps twitch after dynamic exercise.(56)
The early onset of lactic acidosis observed in these patients occurs mainly due to the reduced mitochondrial density and the reduced muscle oxidative capacity, leading to the decrease in pH and the accumulation of acid lactic resulting from the inability to convert oxygen into energy in acid situations.(43)
Another marker to monitor fatigue in these patients is the level of serum ammonia, which typically accompanies the response to lactate during exercise. In one study, it was observed that, in comparison with healthy individuals, 60% of the COPD patients showed an increase in serum ammonia levels during exercise. It is likely that this event is due to the degradation of the AMP deaminase enzyme, subsequently converted into inosine monophosphate and ammonia, with a consequent reduction in the production of ATP, making its level insufficient to meet the energy demand.(57)
Near-infrared spectroscopy
Physiological changes, such as reduced muscle perfusion and hypoxemia, are common findings in COPD patients.(2) In addition, reduced muscle perfusion and hypoxemia have been considered potential factors for the development of contractile fatigue. Near-infrared spectroscopy (NIRS) constitutes a state-of-the-art method for noninvasive measurement of indicators of peripheral perfusion, such as muscle oxygenation. In this technique, the oxygen-dependent differences in the absorption spectrum of iron (in the heme prosthetic group of hemoglobin) or copper (in cytochrome oxidase) make it possible to estimate the changes in the quantity of these metals, providing the local volumes of oxyhemoglobin and deoxyhemoglobin, as well as the sum of both volumes in the local muscle blood flow.(58,59)
Using this technique in COPD patients, one group of authors showed that increased ventilatory work during moderate to intense exercise promotes blood flow redistribution, preferably to the respiratory muscles, reducing skeletal muscle perfusion and consequently increasing lactacidemia and muscle fatigue.(58) Recently, using NIRS, another group of authors showed more eloquently that, in comparison with control group patients, COPD patients present higher peripheral muscle O2 extraction fraction accompanied by lower estimated muscle perfusion during intense exercise.(59) Therefore, the evident imbalance between oxygen delivery and oxygen uptake also plays a negative role in exercise tolerance in these individuals.(58)
In view of the evidence of peripheral muscle function impairment and in view of the different mechanisms determining susceptibility to fatigue in COPD patients, knowledge of the distinct functional assessment methods available (Chart 2) is absolutely necessary so that it is possible to discriminate the main component involved in the process of peripheral muscle fatigue in these patients. However, some aspects in the choice of the method of assessment, such as the availability and reliability of the equipment, its cost-benefit ratio and its user-friendliness, in addition to patient cooperation, should also be considered.
Final considerations
The anatomical and functional substrate of higher muscle fatigue in COPD appears to include lower levels of high-energy phosphates, lower mitochondrial density, early lactacidemia, higher serum ammonia and reduced muscle perfusion. These changes can be revealed by contraction failure, decreased firing rates of motor units and increased recruitment of motor units in a given activity, which can be functionally detected by a reduction in muscle strength, power and endurance. The present review also shows that various types of contractile regimens and protocols have been used in order to detect fatigue in COPD patients. With this understanding, rehabilitation strategies can be developed in order to improve the resistance to muscle fatigue in this population.
References
- 1. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. Bethesda: Global Initiative for Chronic Obstructive Lung Disease; 2007.
- 2. Skeletal muscle dysfunction in chronic obstructive pulmonary disease. A statement of the American Thoracic Society and European Respiratory Society. Am J Respir Crit Care Med. 1999;159(4 Pt 2):S1-40.
- 3. Swallow EB, Reyes D, Hopkinson NS, Man WD, Porcher R, Cetti EJ, et al. Quadriceps strength predicts mortality in patients with moderate to severe chronic obstructive pulmonary disease. Thorax. 2007;62(2):115-20.
- 4. Vøllestad NK. Measurement of human muscle fatigue. J Neurosci Methods. 1997;74(2):219-27.
- 5. Neder JA, Neder LE. Fisiologia Clínica do Exercício: Teoria e Prática. São Paulo: Artes Médicas; 2003.
- 6. Killian KJ, Leblanc P, Martin DH, Summers E, Jones NL, Campbell EJ. Exercise capacity and ventilatory, circulatory, and symptom limitation in patients with chronic airflow limitation. Am Rev Respir Dis. 1992;146(4):935-40.
- 7. Gosselink R, Troosters T, Decramer M. Peripheral muscle weakness contributes to exercise limitation in COPD. Am J Respir Crit Care Med. 1996;153(3):976-80.
- 8. Whittom F, Jobin J, Simard PM, Leblanc P, Simard C, Bernard S, et al. Histochemical and morphological characteristics of the vastus lateralis muscle in patients with chronic obstructive pulmonary disease. Med Sci Sports Exerc. 1998;30(10):1467-74.
- 9. Maltais F, Simard AA, Simard C, Jobin J, Desgagnés P, LeBlanc P. Oxidative capacity of the skeletal muscle and lactic acid kinetics during exercise in normal subjects and in patients with COPD. Am J Respir Crit Care Med. 1996;153(1):288-93.
- 10. Dourado VZ, Tanni SE, Vale SA, Faganello MM, Sanchez FF, Godoy I. Systemic manifestations in chronic obstructive pulmonary disease. J Bras Pneumol. 2006;32(2):161-71.
- 11. Schols AM, Buurman WA, Staal van den Brekel AJ, Dentener MA, Wouters EF. Evidence for a relation between metabolic derangements and increased levels of inflammatory mediators in a subgroup of patients with chronic obstructive pulmonary disease. Thorax. 1996;51(8):819-24.
- 12. Dentener MA, Creutzberg EC, Schols AM, Mantovani A, van't Veer C, Buurman WA, et al. Systemic anti-inflammatory mediators in COPD: increase in soluble interleukin 1 receptor II during treatment of exacerbations. Thorax. 2001;56(9):721-6.
- 13. Gan WQ, Man SF, Senthilselvan A, Sin DD. Association between chronic obstructive pulmonary disease and systemic inflammation: a systematic review and a meta-analysis. Thorax. 2004;59(7):574-80.
- 14. Elevated levels of IL-18 in plasma and skeletal muscle in chronic obstructive pulmonary disease. Lung. 2007;185(3):161-71.
- 15. Spruit MA, Gosselink R, Troosters T, Kasran A, Gayan-Ramirez G, Bogaerts P, et al. Muscle force during an acute exacerbation in hospitalised patients with COPD and its relationship with CXCL8 and IGF-I. Thorax. 2003;58(9):752-6.
- 16. Yende S, Waterer GW, Tolley EA, Newman AB, Bauer DC, Taaffe DR, et al. Inflammatory markers are associated with ventilatory limitation and muscle dysfunction in obstructive lung disease in well functioning elderly subjects. Thorax. 2006;61(1):10-6.
- 17. Broekhuizen R, Wouters EF, Creutzberg EC, Schols AM. Raised CRP levels mark metabolic and functional impairment in advanced COPD. Thorax. 2006;61(1):17-22.
- 18. Decramer M, Lacquet LM, Fagard R, Rogiers P. Corticosteroids contribute to muscle weakness in chronic airflow obstruction. Am J Respir Crit Care Med. 1994;150(1):11-6.
- 19. Richardson RS, Leek BT, Gavin TP, Haseler LJ, Mudaliar SR, Henry R, et al. Reduced mechanical efficiency in chronic obstructive pulmonary disease but normal peak VO2 with small muscle mass exercise. Am J Respir Crit Care Med. 2004;169(1):89-96.
- 20. Koechlin C, Couillard A, Cristol JP, Chanez P, Hayot M, Le Gallais D, et al. Does systemic inflammation trigger local exercise-induced oxidative stress in COPD? Eur Respir J. 2004;23(4):538-44.
- 21. Boots AW, Haenen GR, Bast A. Oxidant metabolism in chronic obstructive pulmonary disease. Eur Respir J Suppl. 2003;46:14s-27s.
- 22. Faucher M, Steinberg JG, Barbier D, Hug F, Jammes Y. Influence of chronic hypoxemia on peripheral muscle function and oxidative stress in humans. Clin Physiol Funct Imaging. 2004;24(2):75-84.
- 23. Jakobsson P, Jorfeldt L, Brundin A. Skeletal muscle metabolites and fibre types in patients with advanced chronic obstructive pulmonary disease (COPD), with and without chronic respiratory failure. Eur Respir J. 1990;3(2):192-6.
- 24. Lexell J. Human aging, muscle mass, and fiber type composition. J Gerontol A Biol Sci Med Sci. 1995;50 Spec No:11-6.
- 25. Serres I, Gautier V, Varray A, Préfaut C. Impaired skeletal muscle endurance related to physical inactivity and altered lung function in COPD patients. Chest. 1998;113(4):900-5.
- 26. Gosker HR, Zeegers MP, Wouters EF, Schols AM. Muscle fibre type shifting in the vastus lateralis of patients with COPD is associated with disease severity: a systematic review and meta-analysis. Thorax. 2007;62(11):944-9.
- 27. Schols AM, Soeters PB, Dingemans AM, Mostert R, Frantzen PJ, Wouters EF. Prevalence and characteristics of nutritional depletion in patients with stable COPD eligible for pulmonary rehabilitation. Am Rev Respir Dis. 1993;147(5):1151-6.
- 28. Kim HC, Mofarrahi M, Hussain SN. Skeletal muscle dysfunction in patients with chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis. 2008;3(4):637-58.
- 29. Pelegrino NR, Lucheta PA, Sanchez FF, Faganello MM, Ferrari R, Godoy I. Influence of lean body mass on cardiopulmonary repercussions during the six-minute walk test in patients with COPD. J Bras Pneumol. 2009;35(1):20-6.
- 30. Malaguti C, Nery LE, Dal Corso S, Nápolis L, De Fuccio MB, Castro M, et al. Scaling skeletal muscle function to mass in patients with moderate-to-severe COPD. Eur J Appl Physiol. 2006;98(5):482-8.
- 31. Allen GM, Gandevia SC, McKenzie DK. Reliability of measurements of muscle strength and voluntary activation using twitch interpolation. Muscle Nerve. 1995;18(6):593-600.
- 32. Man WD, Soliman MG, Gearing J, Radford SG, Rafferty GF, Gray BJ, et al. Symptoms and quadriceps fatigability after walking and cycling in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2003;168(5):562-7.
- 33. Allaire J, Maltais F, Doyon JF, Noël M, LeBlanc P, Carrier G, et al. Peripheral muscle endurance and the oxidative profile of the quadriceps in patients with COPD. Thorax. 2004;59(8):673-8.
- 34. Gleeson NP, Mercer TH. The utility of isokinetic knee extension and flexion strength characteristics of adult males: a comparative examination of gravity corrected and uncorrected data. J Sports Sci. 1996;14(5):415-6.
- 35. Man WD, Soliman MG, Nikoletou D, Harris ML, Rafferty GF, Mustfa N, et al. Non-volitional assessment of skeletal muscle strength in patients with chronic obstructive pulmonary disease. Thorax. 2003;58(8):665-9.
- 36. Mador MJ, Kufel TJ, Pineda LA, Steinwald A, Aggarwal A, Upadhyay AM, et al. Effect of pulmonary rehabilitation on quadriceps fatiguability during exercise. Am J Respir Crit Care Med. 2001;163(4):930-5.
- 37. Neder JA, Jones PW, Nery LE, Whipp BJ. Determinants of the exercise endurance capacity in patients with chronic obstructive pulmonary disease. The power-duration relationship. Am J Respir Crit Care Med. 2000;162(2 Pt 1):497-504.
- 38. De Luca CJ. The use of surface electromyography in biomechanics. J Appl Biomech. 1997;13(1):135-63.
- 39. Duchêne J, Goubel F. Surface electromyogram during voluntary contraction: processing tools and relation to physiological events. Crit Rev Biomed Eng. 1993;21(4):313-97.
- 40. Hausswirth C, Brisswalter J, Vallier JM, Smith D, Lepers R. Evolution of electromyographic signal, running economy, and perceived exertion during different prolonged exercises. Int J Sports Med. 2000;21(6):429-36.
- 41. Soderberg GL, Knutson LM. A guide for use and interpretation of kinesiologic electromyographic data. Phys Ther. 2000;80(5):485-98.
- 42. Farina D, Blanchietti A, Pozzo M, Merletti R. M-wave properties during progressive motor unit activation by transcutaneous stimulation. J Appl Physiol. 2004;97(2):545-55.
- 43. Saey D, Côté CH, Mador MJ, Laviolette L, LeBlanc P, Jobin J, et al. Assessment of muscle fatigue during exercise in chronic obstructive pulmonary disease. Muscle Nerve. 2006;34(1):62-71.
- 44. Gosselin N, Matecki S, Poulain M, Ramonatxo M, Ceugniet F, Préfaut C, et al. Electrophysiologic changes during exercise testing in patients with chronic obstructive pulmonary disease. Muscle Nerve. 2003;27(2):170-9.
- 45. Gosselin N, Durand F, Poulain M, Lambert K, Ceugniet F, Préfaut C, et al. Effect of acute hyperoxia during exercise on quadriceps electrical activity in active COPD patients. Acta Physiol Scand. 2004;181(3):333-43.
- 46. Man WD, Soliman MG, Gearing J, Radford SG, Rafferty GF, Gray BJ, et al. Symptoms and quadriceps fatigability after walking and cycling in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2003;168(5):562-7.
- 47. Verin E, Ross E, Demoule A, Hopkinson N, Nickol A, Fauroux B, et al. Effects of exhaustive incremental treadmill exercise on diaphragm and quadriceps motor potentials evoked by transcranial magnetic stimulation. J Appl Physiol. 2004;96(1):253-9.
- 48. Swallow EB, Gosker HR, Ward KA, Moore AJ, Dayer MJ, Hopkinson NS, et al. A novel technique for nonvolitional assessment of quadriceps muscle endurance in humans. J Appl Physiol. 2007;103(3):739-46.
- 49. Malaguti C, Nery LE, Dal Corso S, De Fuccio MB, Lerario MC, Cendon S, et al. Alternative strategies for exercise critical power estimation in patients with COPD. Eur J Appl Physiol. 2006;96(1):59-65.
- 50. Borg GA. Psychophysical bases of perceived exertion. Med Sci Sports Exerc. 1982;14(5):377-81.
- 51. Hayes M, Patterson D. Experiental development of the graphic rating method. Psychol Bull 1921;18:98-9.
- 52. Kutsuzawa T, Shioya S, Kurita D, Haida M, Ohta Y, Yamabayashi H. Muscle energy metabolism and nutritional status in patients with chronic obstructive pulmonary disease. A 31P magnetic resonance study. Am J Respir Crit Care Med. 1995;152(2):647-52.
- 53. Tada H, Kato H, Misawa T, Sasaki F, Hayashi S, Takahashi H, et al. 31P-nuclear magnetic resonance evidence of abnormal skeletal muscle metabolism in patients with chronic lung disease and congestive heart failure. Eur Respir J. 1992;5(2):163-9.
- 54. Lévy P, Wuyam B, Pépin JL, Reutenauer H, Payen JF. Skeletal muscle abnormalities in chronic obstructive lung disease with respiratory insufficiency. Value of P31 magnetic resonance spectroscopy [Article in French]. Rev Mal Respir. 1997;14(3):183-91. Erratum in: Rev Mal Respir. 1997;14(4):333.
- 55. Debigaré R, Côte CH, Hould FS, LeBlanc P, Maltais F. In vitro and in vivo contractile properties of the vastus lateralis muscle in males with COPD. Eur Respir J. 2003;21(2):273-8.
- 56. Saey D, Michaud A, Couillard A, Côté CH, Mador MJ, LeBlanc P, et al. Contractile fatigue, muscle morphometry, and blood lactate in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2005;171(10):1109-15.
- 57. Calvert LD, Singh SJ, Greenhaff PL, Morgan MD, Steiner MC. The plasma ammonia response to cycle exercise in COPD. Eur Respir J. 2008;31(4):751-8.
- 58. Borghi-Silva A, Oliveira CC, Carrascosa C, Maia J, Berton DC, Queiroga F Jr, et al. Respiratory muscle unloading improves leg muscle oxygenation during exercise in patients with COPD. Thorax. 2008;63(10):910-5.
- 59. Chiappa GR, Borghi-Silva A, Ferreira LF, Carrascosa C, Oliveira CC, Maia J, et al. Kinetics of muscle deoxygenation are accelerated at the onset of heavy-intensity exercise in patients with COPD: relationship to central cardiovascular dynamics. J Appl Physiol. 2008;104(5):1341-50.
Publication Dates
-
Publication in this collection
16 Dec 2009 -
Date of issue
Nov 2009
History
-
Received
17 Feb 2009 -
Accepted
07 July 2009