Abstract
Little is known about the effects of stress hormones on the etiologic agents of halitosis. Thus, the aim of this study was to evaluate in vitro the effects of adrenaline (ADR), noradrenaline (NA) and cortisol (CORT) on bacteria that produce volatile sulfur compounds (VSC), the major gases responsible for bad breath. Cultures of Fusobacterium nucleatum (Fn), Porphyromonas endodontalis (Pe), Prevotella intermedia (Pi) and Porphyromonas gingivalis (Pg) were exposed to 50 µM ADR, NA and CORT or equivalent volumes of sterile water as controls for 12 and 24 h. Growth was evaluated based on absorbance at 660 nm. Portable gas chromatography was used to measure VSC concentrations. Kruskal-Wallis and the Dunn post-hoc test were used to compare the groups. For Fn, ADR, NA and CORT significantly reduced bacterial growth after 12 h and 24 h (p < 0.05). All the substances tested increased hydrogen sulfide (H2S) production (p < 0.05). For Pe, all the substances tested reduced bacterial development after 24 h (p < 0.05), and NA significantly increased the H2S concentration after 12 h (p < 0.05). In the Pg and Pi cultures, no effects on bacterial growth were observed (p > 0.05). In the Pi cultures, ADR, NA and CORT increased H2S (p < 0.05). Catecholamines and cortisol can interfere with growth and H2S production of sub-gingival species in vitro. This process appears to be complex and supports the association between stress and the production of VSC.
Halitosis; Catecholamines; Hydrocortisone; Gram-Negative Anaerobic Bacteria
Introduction
Halitosis, also known as bad breath or malodor, is a term used to describe unpleasant, fetid odors present in air exhaled from the mouth.11 Aylikci BU, Colak H. Halitosis: From diagnosis to management. J Nat Sci Biol Med. 2013 Jan;4(1):14-23. Being unpleasant, bad breath is a problem that affects both the patient and the family.11 Aylikci BU, Colak H. Halitosis: From diagnosis to management. J Nat Sci Biol Med. 2013 Jan;4(1):14-23.
Studies have indicated that in the majority of cases, malodor is associated with the degradation of sulfur-containing amino acids (methionine, cysteine and cystine) by anaerobic gram-negative bacteria present in the oral cavity, including Porphyromonas gingivalis, Fusobacterium nucleatum, Prevotella intermedia, Tannerella forsythia and Porphyromonas endodontalis.22 Persson S, Claesson R, Carlsson J. The capacity of subgingival microbiotas to produce volatile sulfur compounds in human serum. Oral Microbiol Immunol. 1989 Sep;4(3):169-72.,33 Krespi YP, Shrime MG, Kacker A. The relationship between oral malodor and volatile sulfur compound-producing bacteria. Otolaryngol Head Neck Surg. 2006 Nov;135(5):671-6. Volatile sulfur compounds (VSC), such as hydrogen sulfide (H2S), methyl mercaptan (CH3SH) and dimethyl sulfide [(CH3)2S], are generated as a product of this metabolism.33 Krespi YP, Shrime MG, Kacker A. The relationship between oral malodor and volatile sulfur compound-producing bacteria. Otolaryngol Head Neck Surg. 2006 Nov;135(5):671-6.
Some patients with halitosis do not present clinical evidence of oral pathologies or systemic disorders. Stress, depression and anxiety have been identified as risk factors for a shift in oral homeostasis.44 Peruzzo DC, Benatti BB, Ambrosano GM, Nogueira-Filho GR, Sallum EA, Casati MZ, et al. A systematic review of stress and psychological factors as possible risk factors for periodontal disease. J Periodontol. 2007 Aug;78(8):1491-504.,55 Rosania AE, Low KG, McCormick CM, Rosania DA. Stress, depression, cortisol, and periodontal disease. J Periodontol. 2009 Feb;80(2):260-6. It has been proposed that emotional alterations may be a co-factor in halitosis development. Calil and Marcondes66 Calil CM, Marcondes FK. Influence of anxiety on the production of oral volatile sulfur compounds. Life Sci. 2006 Jul 10;79(7):660-4. reported increased VSC concentrations in healthy men faced with an anxiety-evoking situation compared with the basal situation, independent of salivary flow.
The relationship between stress and the progression of infectious diseases, including periodontitis, has been the focus of some studies over the last few decades.44 Peruzzo DC, Benatti BB, Ambrosano GM, Nogueira-Filho GR, Sallum EA, Casati MZ, et al. A systematic review of stress and psychological factors as possible risk factors for periodontal disease. J Periodontol. 2007 Aug;78(8):1491-504.,77 Ng SK, Keung Leung W. A community study on the relationship between stress, coping, affective dispositions and periodontal attachment loss. Community Dent Oral Epidemiol. 2006 Aug;34(4):252-66.,88 Ishisaka A, Ansai T, Soh I, Inenaga K, Yoshida A, Shigeyama C, et al. Association of salivary levels of cortisol and dehydroepiandrosterone with periodontitis in older Japanese adults. J Periodontol. 2007 Sep;78(9):1767-73.,99 Huang S, Lu F, Zhang Z, Yang X, Chen Y. The role of psychologic stress-induced hypoxia-inducible factor-1alpha in rat experimental periodontitis. J Periodontol. 2011 Jun;82(6):934-41.,1010 Semenoff TA, Rosa Junior A, Borges AH, Porto AN, Caporossi C, Semenoff Segundo A. Effect of chronic stress in newborn rats on the progression of ligature-induced-periodontitis in adulthood. Acta Cir Bras. 2013 Sep;28(9):652-6. However, the mechanisms involved in this process are still not clear. Potentially pathogenic microorganisms can recognize stress hormones, a fact that has led to the development of the concept of “microbial endocrinology.”1111 Freestone PP, Sandrini SM, Haigh RD, Lyte M. Microbial endocrinology: how stress influences susceptibility to infection. Trends Microbiol. 2008 Feb;16(2):55-64. These microorganisms could use the hormones produced by the host as ‘environmental trails’ to initiate their growth and pathogenic processes.1212 Belay T, Aviles H, Vance M, Fountain K, Sonnenfeld G. Catecholamines and in vitro growth of pathogenic bacteria: enhancement of growth varies greatly among bacterial species. Life Sci. 2003 Aug 8;73(12):1527-35.,1313 Roberts A, Matthews JB, Socransky SS, Freestone PP, Williams PH, Chapple IL. Stress and the periodontal diseases: effects of catecholamines on the growth of periodontal bacteria in vitro. Oral Microbiol Immunol. 2002 Oct;17(5):296-303.,1414 Roberts A, Matthews JB, Socransky SS, Freestone PP, Williams PH, Chapple IL. Stress and the periodontal diseases: growth responses of periodontal bacteria to Escherichia coli stress-associated autoinducer and exogenous Fe. Oral Microbiol Immunol. 2005 Jun;20(3):147-53.,1515 Saito T, Inagaki S, Sakurai K, Okuda K, Ishihara K. Exposure of P. gingivalis to noradrenaline reduces bacterial growth and elevates ArgX protease activity. Arch Oral Biol. 2011 Mar;56(3):244-50.,1616 Pullinger GD, Carnell SC, Sharaff FF, van Diemen PM, Dziva F, Morgan E, et al. Norepinephrine augments Salmonella enterica-induced enteritis in a manner associated with increased net replication but independent of the putative adrenergic sensor kinases QseC and QseE. Infect Immun. 2010 Jan;78(1):372-80.,1717 Jentsch HF, März D, Krüger M. The effects of stress hormones on growth of selected periodontitis related bacteria. Anaerobe. 2013 Dec;24:49-54 In one study, the growth rate of bacteria commonly found in the oral cavity, such as Actinomyces naeslundii, Eikenella corrodens, and Campylobacter gracilis, was stimulated by exposure to noradrenaline.1313 Roberts A, Matthews JB, Socransky SS, Freestone PP, Williams PH, Chapple IL. Stress and the periodontal diseases: effects of catecholamines on the growth of periodontal bacteria in vitro. Oral Microbiol Immunol. 2002 Oct;17(5):296-303. In addition, recent studies show conflicting results of noradrenaline and adrenaline on the growth of P. gingivalis, with studies showing either no effects of these hormones on growth1212 Belay T, Aviles H, Vance M, Fountain K, Sonnenfeld G. Catecholamines and in vitro growth of pathogenic bacteria: enhancement of growth varies greatly among bacterial species. Life Sci. 2003 Aug 8;73(12):1527-35.,1717 Jentsch HF, März D, Krüger M. The effects of stress hormones on growth of selected periodontitis related bacteria. Anaerobe. 2013 Dec;24:49-54 or demonstrating a reduction in viability.1313 Roberts A, Matthews JB, Socransky SS, Freestone PP, Williams PH, Chapple IL. Stress and the periodontal diseases: effects of catecholamines on the growth of periodontal bacteria in vitro. Oral Microbiol Immunol. 2002 Oct;17(5):296-303.,1515 Saito T, Inagaki S, Sakurai K, Okuda K, Ishihara K. Exposure of P. gingivalis to noradrenaline reduces bacterial growth and elevates ArgX protease activity. Arch Oral Biol. 2011 Mar;56(3):244-50. Recently, it was demonstrated that noradrenaline can stimulate the production and activity of arg-gingipain B, an important protease of P. gingivalis.1515 Saito T, Inagaki S, Sakurai K, Okuda K, Ishihara K. Exposure of P. gingivalis to noradrenaline reduces bacterial growth and elevates ArgX protease activity. Arch Oral Biol. 2011 Mar;56(3):244-50. Thus, the effects of stress could be mediated by two potentially synergistic pathways involving the down-regulation of the host’s defenses and the up-regulation of bacterial pathogenicity.
Because there is clinical evidence that stress is related to halitosis,66 Calil CM, Marcondes FK. Influence of anxiety on the production of oral volatile sulfur compounds. Life Sci. 2006 Jul 10;79(7):660-4. we hypothesized that periodontal bacteria would respond to stress substances by stimulating their growth and their production of VSC. Because there have been no studies on the effects of stress hormones on VSC production, the aim of the present study was to investigate the effects of catecholamines and cortisol on the growth and VSC production of periodontopathogens.
Methodology
Substances Used
The following substances were tested: adrenaline - ADR (epinephrine bitartrate, Sigma Chemical Co.®, St Louis, USA), noradrenaline – NA (arterenol, Sigma®) and cortisol - CORT (hydrocortisone, Sigma®). The 50 µm concentration of ADR, NA1313 Roberts A, Matthews JB, Socransky SS, Freestone PP, Williams PH, Chapple IL. Stress and the periodontal diseases: effects of catecholamines on the growth of periodontal bacteria in vitro. Oral Microbiol Immunol. 2002 Oct;17(5):296-303.,1818 Lyte M, Ernst S. Catecholamine induced growth of gram negative bacteria. Life Sci. 1992;50(3):203-12. and CORT1919 Plotkin BJ, Roose RJ, Erikson Q, Viselli SM. Effect of androgens and glucocorticoids on microbial growth and antimicrobial susceptibility. Curr Microbiol. 2003 Dec;47(6):514-20. was chosen based on previous studies.The solutions were prepared using distilled water as the solvent and were sterilized using Millex® (Millipore®, Billerica, USA) sterile filters with a pore size of 0.22 µm.
Bacterial Samples and Culture Conditions
The growth patterns and VSC production of four bacterial species (Porphyromonas endodontalis from clinical specimens - Pe, Porphyromonas gingivalis W83 - Pg, Fusobacterium nucleatum nucleatum ATCC 25586 - Fn and Prevotella intermedia ATCC 25611 - Pi) were examined. These species were selected based on their ability to produce VSC constitutively. The bacteria were grown statically in brain heart infusion broth (BHI; Difco Co., Detroit, USA). To cultivate Pg and Pe, 5 µg/mL hemin (Sigma®) and 1 µg/mL menadione (Sigma®) were added to the BHI. Bacterial cultures were grown at 37 °C under anaerobic conditions (10% CO2, 10% H2 and 80% N2; MiniMacs Anaerobic Workstation, Don Whitley Scientific®, Shipley, UK).
Study design and Experimental Groups
Cultures of Pg, Pe, Pi and Fn were exposed to 50 µM ADR, NOR and CORT for 12 and 24 h (early and mid-logarithmic growth phases). Tubes containing bacteria without the tested substances but containing equivalent volumes of distilled water (vehicle) were used as positive control for growth. Negative controls contained only the tested substances and the culture medium. After incubation, bacterial growth and VSC production were assessed as follows. At least two separate experiments were performed for each hormone tested. In each experiment, 8 replicates were tested for each group (ADR, NA, CORT and controls).2020 Tanabe S, Desjardins J, Bergeron C, Gafner S, Villinski JR, Grenier D. Reduction of bacterial volatile sulfur compound production by licoricidin and licorisoflavan A from licorice. J Breath Res. 2012 Mar;6(1):016006.
Susceptibility Tests (Macrodilution Broth Tests)
Susceptibility was assayed using a macrodilution broth test as previously described.2121 Winn Jr WC, Janda WM, Koneman EW, Schreckenberger PC, Procop GW, Woods GL. Koneman’s color atlas and textbook of diagnostic microbiology. 6th ed. Philadelphia: Lippincott Willians & Wilkins Company; 2006. 1531 p. Tubes containing 5 mL of BHI broth medium (plus 5 µg/mL hemin (Sigma®) and 1 µg/mL menadione (Sigma®) for the Pg and Pe cultures) were prepared for each experimental group. A standardized bacterial suspension was prepared in sterile saline solution (0.9% NaCl) and adjusted with a spectrophotometer (Unico 1100 RS;Unico®, Dayton, USA) to an optical density of 0.5 (8 x 106 CFU/mL). An inoculum of 500 µL was added to each tube to give a final concentration of 4 x 106 CFU/mL. The tubes were then incubated at 37°C in anaerobic conditions for 12 or 24 h. After these periods, bacterial growth was assessed spectrophotometrically at a wavelength of 660 nm.
Bacterial VSC Production Tests
This experiment was conducted as previously described, with some modifications.2020 Tanabe S, Desjardins J, Bergeron C, Gafner S, Villinski JR, Grenier D. Reduction of bacterial volatile sulfur compound production by licoricidin and licorisoflavan A from licorice. J Breath Res. 2012 Mar;6(1):016006. For bacterial VSC production analyses, the experimental groups and inocula were prepared as described above. After incubation at 37 °C in anaerobic conditions, 0.1 mL of air from the tubes was aspirated using a 1 mL syringe (BD®, Franklin Lakes, USA) coupled to a needle (BD®) (Figure 1) and injected into Oral Chroma® (Abimedical, Osaka, Japan). For a period of 8 minutes, the apparatus performed molecule counts in parts per billion (ppb) of hydrogen sulfite (H2S) and methylmercaptan (CH3SH). VSC analyses were performed at two different time intervals (12 and 24 h). These incubation lengths were chosen because at these periods, the bacteria are growing exponentially, and there are more viable cells than dead cells.
Gaseous standards of the VSC (H2S and CH3SH) were prepared as described previously2222 Winkel EG, Tangerman A. Appropriate sample bags and syringes for preserving breath samples in breath odor research: a technical note. J Breath Res. 2008 Mar;2(1):017011. by White Martins (Campinas, Brazil) (RBC-INMETRO n M-32433/10), and the device was calibrated every three months according to Tangerman and Winkel.2323 Tangerman A, Winkel EG. Extra-oral halitosis: an overview. J Breath Res. 2010 Mar;4(1):017003.
Statistical Analysis
Levene’s test was used to test the homogeneity of variance, and the normality of distribution was tested by the Shapiro-Wilk’s test. Kruskal-Wallis and the Dunn post-hoc test were used to compare the groups (ADR, NA and CORT compared to control for each bacteria and each time interval separately). Statistical analysis was performed using BioEstat 5.0 (Fundação Mamirauá, Belém, Brazil) at a significance level of 5%.
Results
Effects of Adrenaline, Noradrenaline and Cortisol on Bacterial Growth
Figure 2 summarizes the effects of these substances on bacterial growth (optical density, 660 nm). For the Fn cultures, ADR, NA and CORT significantly reduced bacterial growth after 12 and 24 h (p < 0.05, Kruskal Wallis test). For Pe, the tested substances reduced bacterial growth, but only after 24 h of exposure (p < 0.05). No statistically significant differences in optical density (p > 0.05) were observed for the Pg and Pi cultures in any of the time periods evaluated.
Mean and standard deviation of optical density (660 nm) representing the bacterial growth. Statistically significant p values are shown (*) on the upper side of the columns (p < 0.05, Kruskal Wallis, Dunn).
Effects of Noradrenaline, Adrenaline and Cortisol on Bacterial VSC Production
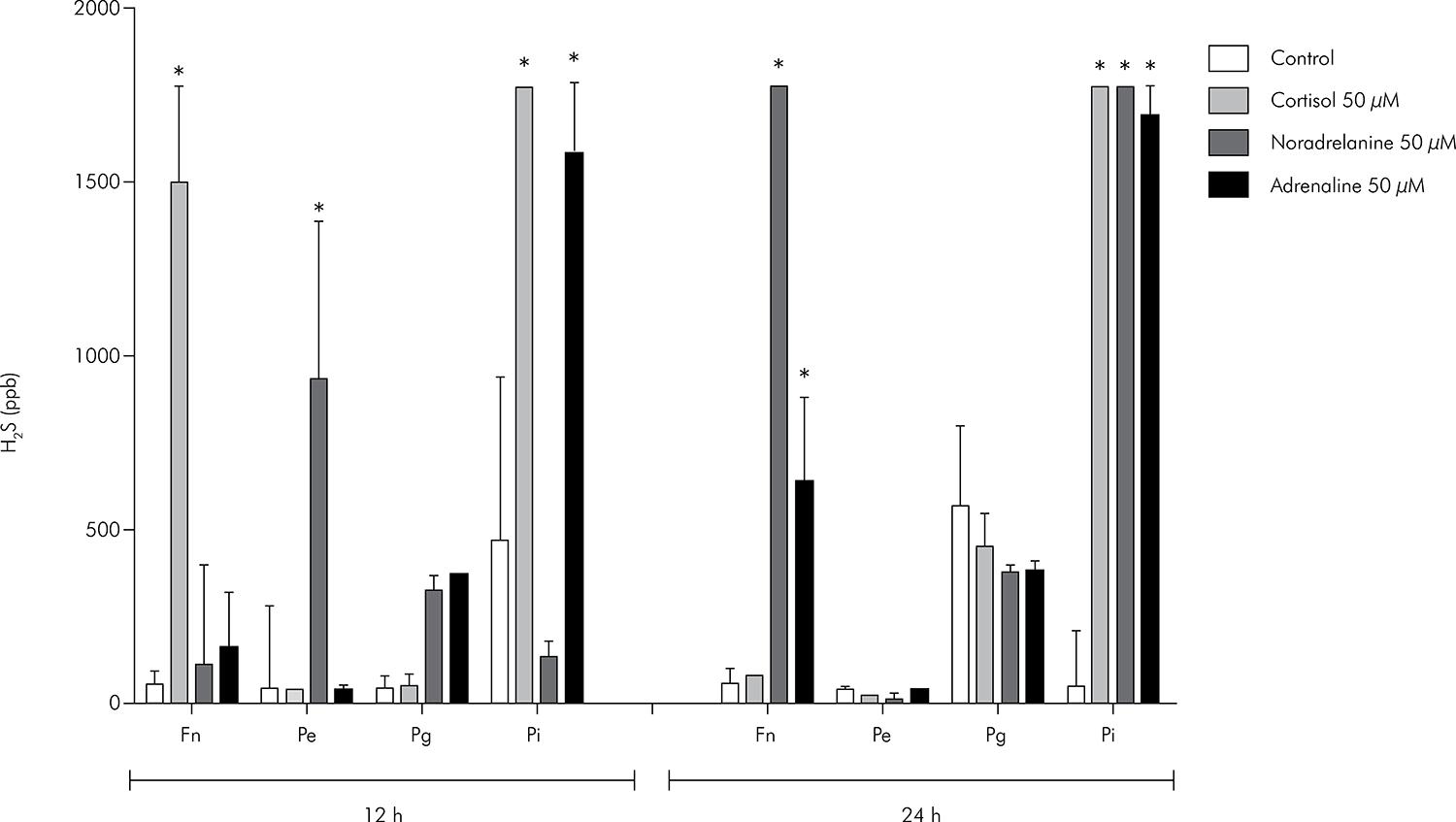
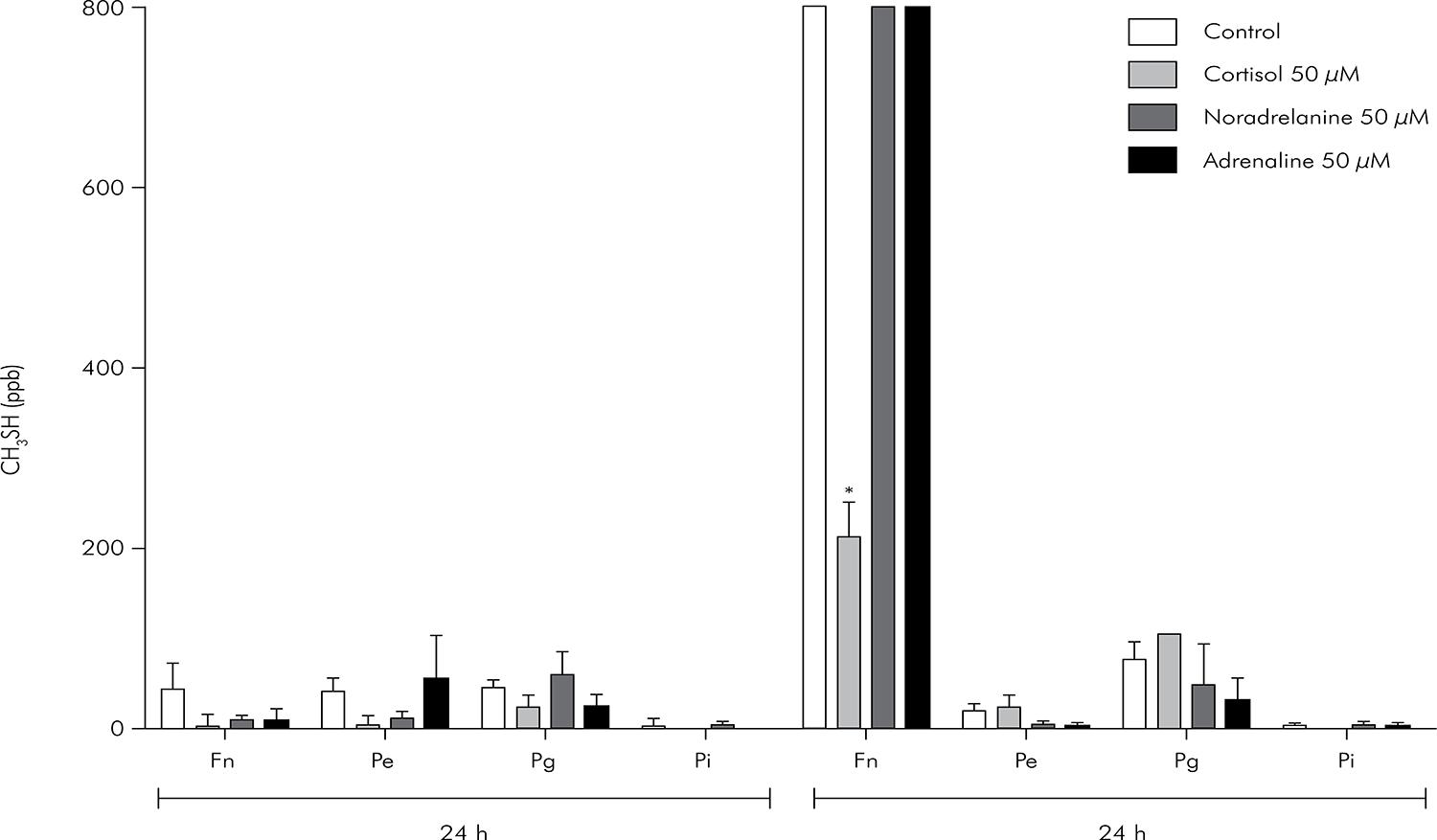
The median values and interquartile deviations of H2S and CH3SH concentrations (in ppb) for Fn, Pe, Pg and Pi cultures exposed for 12 and 24 h to ADR, NA and CORT are presented in Figures 3 and 4, respectively.
Median and interquartile deviation of H2S concentrations (ppb) representing the VSCs production. Statistically significant p values are shown (*) on the upper side of the columns (p < 0.05, Kruskal Wallis, Dunn).
Median and interquartile deviation of CH3SH concentrations (ppb) representing the VSCs production. Statistically significant p values are shown (*) on the upper side of the columns (p < 0.05, Kruskal Wallis, Dunn).
In Fn, only CORT caused a significant increase in H2S levels at 12 h (Kruskal-Wallis, p < 0.05), while no changes in H2S concentration were observed at 24 h (Kruskal-Wallis, p > 0.05). However, ADR and NA did increase the H2S concentration in Fn cultures after 24 h of exposure (p < 0.05). Only Fn showed a reduction in CH3SH production by CORT after 24 h (p < 0.05). No other conditions changed the CH3SH levels.
H2S concentrations were significantly increased only in Pe cultures exposed to NA for 12 h (p < 0.05). No difference in H2S production was observed for the groups exposed to ADR and CORT (p > 0.05). In addition, CH3SH levels were not altered by exposure of Pe to catecholamines and CORT (p > 0.05).
No statistically significant differences (p > 0.05) were found in H2S and CH3SH levels among groups and their respective controls in Pg cultured for 12 or 24 h.
Pi was more sensitive to the effects of catecholamines and CORT than the other bacterial cultures. In this case, H2S production was up-regulated in the groups exposed to ADR, CORT (12 and 24 h) and NA (24 h) (p < 0.05). None of the experimental groups showed differences in CH3SH production compared to their controls (p > 0.05).
Discussion
Clinical evidence has shown that stress can increase VSC concentrations, thus contributing to halitosis.66 Calil CM, Marcondes FK. Influence of anxiety on the production of oral volatile sulfur compounds. Life Sci. 2006 Jul 10;79(7):660-4. The present study provides additional support for the role of stress substances on halitosis, as it was shown that ADR, NOR and CORT can up-regulate the production of VSC by microorganisms commonly found in sub-gingival biofilms.
This study reveals a variation in the growth rates of four periodontopathogenic microorganisms in response to the addition of stress hormones to their culture media. The growth of Fn and Pe was reduced by stress hormones, primarily after 24 h of exposure. In contrast, no significant effects on growth were observed in Pg and Pi for any of the substances tested. Most of these results are in agreement with the literature regarding the effects of catecholamines on bacterial growth. Roberts et al.1313 Roberts A, Matthews JB, Socransky SS, Freestone PP, Williams PH, Chapple IL. Stress and the periodontal diseases: effects of catecholamines on the growth of periodontal bacteria in vitro. Oral Microbiol Immunol. 2002 Oct;17(5):296-303. demonstrated an inhibitory effect of catecholamines on Fn growth and no effect on the growth of Pi cultures, while Belay et al.1212 Belay T, Aviles H, Vance M, Fountain K, Sonnenfeld G. Catecholamines and in vitro growth of pathogenic bacteria: enhancement of growth varies greatly among bacterial species. Life Sci. 2003 Aug 8;73(12):1527-35. and Jentsch et al.1717 Jentsch HF, März D, Krüger M. The effects of stress hormones on growth of selected periodontitis related bacteria. Anaerobe. 2013 Dec;24:49-54 showed no change in Pg growth patterns. However, some of the present findings are contrary to previous reports. It was reported that the growth rate of Pg was reduced when exposed to catecholamines.1313 Roberts A, Matthews JB, Socransky SS, Freestone PP, Williams PH, Chapple IL. Stress and the periodontal diseases: effects of catecholamines on the growth of periodontal bacteria in vitro. Oral Microbiol Immunol. 2002 Oct;17(5):296-303.,1515 Saito T, Inagaki S, Sakurai K, Okuda K, Ishihara K. Exposure of P. gingivalis to noradrenaline reduces bacterial growth and elevates ArgX protease activity. Arch Oral Biol. 2011 Mar;56(3):244-50. Additionally, cortisol was demonstrated to stimulate the growth of Pg cultures.2424 Akcali A, Huck O, Buduneli N, Davideau JL, Kose T, Tenenbaum H. Exposure of Porphyromonas gingivalis to cortisol increases bacterial growth. Arch Oral Biol. 2014 Jan;59(1):30-4. Our results showed no effects of NA, ADR or CORT on the growth rate of this pathogen. There were several differences in the experimental factors used in these studies, including the period of hormone exposure, bacterial strains and culture media used, catecholamine concentration and the method of growth assessment, which may have contributed to the divergent findings.
There are some reports in the literature demonstrating VSC production by periodontopathogenic bacteria. Previous studies revealed that saliva and dental plaque from patients with periodontal disease produces large quantities of VSC, thus contributing to malodor.2525 Coli JM, Tonzetich J. Characterization of volatile sulphur compounds production at individual gingival crevicular sites in humans. J Clin Dent. 1992;3(4):97-103.,2626 Yaegaki K, Sanada K. Biochemical and clinical factors influencing oral malodor in periodontal patients. J Periodontol. 1992 Sep;63(9):783-9.,2727 Kishi M, Ohara-Nemoto Y, Takahashi M, Kishi K, Kimura S, Aizawa F, et al. Prediction of periodontopathic bacteria in dental plaque of periodontal healthy subjects by measurement of volatile sulfur compounds in mouth air. Arch Oral Biol. 2013 Mar;58(3):324-30. Our study shows that periodontal bacteria produce significant VSC levels in vitro and that this production is altered by stress hormones. We observed that the major effects of catecholamines and CORT were on H2S levels, mainly in the case of Fn, Pe and Pi. For these bacteria, exposure to these substances caused an increase in H2S concentration. Only CORT changed the production of CH3SH, and this effect was inhibitory in Fn cultures treated with this glucocorticoid. Given these results, it is possible to assume that the main roles of ADR, NA and CORT are in H2S production. To better understand the effects of stress hormones, it will be necessary to utilize enzymatic, genomic and proteomic approaches.
Interestingly, the growth and VSC production of Pg was not affected by any of the stress hormones tested. It is possible that Pg is less sensitive to hormonal influence in the favorable environment of our experiment, which offered a rich-nutrient culture media along with controlled atmosphere and temperature. Previous studies have observed that adrenaline and noradrenaline have strong effects on bacterial growth mainly when the bacteria are cultivated in poor nutrient media.1818 Lyte M, Ernst S. Catecholamine induced growth of gram negative bacteria. Life Sci. 1992;50(3):203-12.,2828 O’Donnell PM, Aviles H, Lyte M, Sonnenfeld G. Enhancement of in vitro growth of pathogenic bacteria by norepinephrine: importance of inoculum density and role of transferrin. Appl Environ Microbiol. 2006 Jul;72(7):5097-9. For this reason, those authors designed a special medium, serum-SAPI minimal medium, to mimic the challenging in vivo environment. However, this medium was not used in our study. This could be the reason for our observations of slight effects of hormones on growth and VSC production, especially in the case of Pg. It is possible, for example, that these organisms (and others showing negative growth responses) utilize catecholamines for up-regulation of virulence expression rather than for growth1313 Roberts A, Matthews JB, Socransky SS, Freestone PP, Williams PH, Chapple IL. Stress and the periodontal diseases: effects of catecholamines on the growth of periodontal bacteria in vitro. Oral Microbiol Immunol. 2002 Oct;17(5):296-303. and VSC production.
When taking into consideration the times of exposure (12 and 24 h) in our study, differing effects were found. In some experiments, significant effects of the hormones were noted at both times, while the hormone effects were observed at only one time period (12 or 24 h) in other experiments. We expected to observe that a substance that showed an effect at 12 h of exposure would have the same effect at 24 h. As demonstrated, however, this was not always the case. These stress-related substances have short half-lives and could be degraded during the time-period of our experiments. Kennedy et al.2929 Kennedy B, Dillon E, Mills PJ, Ziegler MG. Catecholamines in human saliva. Life Sci. 2001 May 25;69(1):87-99. demonstrated that catecholamines are stable in saliva for up to 2 h at 4 °C. In our study, however, catecholamine concentrations in the culture medium after 12 and 24 h were not determined, and one can imagine that degradation was faster due to the higher temperature. We hypothesized that the bacteria might be able to use this degradation by-product to stimulate VSC production; however, further studies are necessary to elucidate this hypothesis. With regard to the degradation of CORT, it is known that this hormone remains stable in saliva at ambient temperature for up to one week or more3030 Liening SH, Stanton SJ, Saini EK, Schultheiss OC. Salivary testosterone, cortisol, and progesterone: two-week stability, interhormone correlations, and effects of time of day, menstrual cycle, and oral contraceptive use on steroid hormone levels. Physiol Behav. 2010 Jan 12;99(1):8-16. and, therefore, would not be degraded during our experimental period.
By comparing the VSC production and growth patterns of bacteria exposed to NA, ADR and CORT, it was possible to observe differing results. These findings suggest a species-specific interaction of ADR, NA and CORT, which leads to differing effects in terms of growth and gas production. It is worth highlighting that, even in some situations where growth was reduced, VSC levels were stimulated. Because of this, it is possible that these stress-related substances have different cellular mechanisms whereby they affect growth and VSC production. Additionally, considering that fewer bacteria could produce higher levels of VSC in the presence of these hormones, it is important to evaluate the clinical importance of this observation. It is possible that these results could be the beginning of an explanation for why patients without advanced periodontal disease or other clinical signs of inflammation have increased complaints of halitosis in stressful situations and have higher VSC levels than non-stressed patients.66 Calil CM, Marcondes FK. Influence of anxiety on the production of oral volatile sulfur compounds. Life Sci. 2006 Jul 10;79(7):660-4. Investigations of the exposure of catecholamines and cortisol in multi-species biofilm models, animal models for halitosis and clinical studies should be carried out to better understand the relationship of stress and VSC production.
Conclusions
In conclusion, our results indicate that the hormones adrenaline, noradrenaline and cortisol can modulate the growth of periodontopathogenic microorganisms and the production of VSC, thus elucidating another possible mechanism of action for the relationship between stress and the development of halitosis.
Acknowledgements
We thank Fundação de Amparo à Pesquisa do Estado de São Paulo (Fapesp) for its support. GMO was the recipient of a Fapesp fellowship (no. 2008/00750-1).
References
-
1Aylikci BU, Colak H. Halitosis: From diagnosis to management. J Nat Sci Biol Med. 2013 Jan;4(1):14-23.
-
2Persson S, Claesson R, Carlsson J. The capacity of subgingival microbiotas to produce volatile sulfur compounds in human serum. Oral Microbiol Immunol. 1989 Sep;4(3):169-72.
-
3Krespi YP, Shrime MG, Kacker A. The relationship between oral malodor and volatile sulfur compound-producing bacteria. Otolaryngol Head Neck Surg. 2006 Nov;135(5):671-6.
-
4Peruzzo DC, Benatti BB, Ambrosano GM, Nogueira-Filho GR, Sallum EA, Casati MZ, et al. A systematic review of stress and psychological factors as possible risk factors for periodontal disease. J Periodontol. 2007 Aug;78(8):1491-504.
-
5Rosania AE, Low KG, McCormick CM, Rosania DA. Stress, depression, cortisol, and periodontal disease. J Periodontol. 2009 Feb;80(2):260-6.
-
6Calil CM, Marcondes FK. Influence of anxiety on the production of oral volatile sulfur compounds. Life Sci. 2006 Jul 10;79(7):660-4.
-
7Ng SK, Keung Leung W. A community study on the relationship between stress, coping, affective dispositions and periodontal attachment loss. Community Dent Oral Epidemiol. 2006 Aug;34(4):252-66.
-
8Ishisaka A, Ansai T, Soh I, Inenaga K, Yoshida A, Shigeyama C, et al. Association of salivary levels of cortisol and dehydroepiandrosterone with periodontitis in older Japanese adults. J Periodontol. 2007 Sep;78(9):1767-73.
-
9Huang S, Lu F, Zhang Z, Yang X, Chen Y. The role of psychologic stress-induced hypoxia-inducible factor-1alpha in rat experimental periodontitis. J Periodontol. 2011 Jun;82(6):934-41.
-
10Semenoff TA, Rosa Junior A, Borges AH, Porto AN, Caporossi C, Semenoff Segundo A. Effect of chronic stress in newborn rats on the progression of ligature-induced-periodontitis in adulthood. Acta Cir Bras. 2013 Sep;28(9):652-6.
-
11Freestone PP, Sandrini SM, Haigh RD, Lyte M. Microbial endocrinology: how stress influences susceptibility to infection. Trends Microbiol. 2008 Feb;16(2):55-64.
-
12Belay T, Aviles H, Vance M, Fountain K, Sonnenfeld G. Catecholamines and in vitro growth of pathogenic bacteria: enhancement of growth varies greatly among bacterial species. Life Sci. 2003 Aug 8;73(12):1527-35.
-
13Roberts A, Matthews JB, Socransky SS, Freestone PP, Williams PH, Chapple IL. Stress and the periodontal diseases: effects of catecholamines on the growth of periodontal bacteria in vitro. Oral Microbiol Immunol. 2002 Oct;17(5):296-303.
-
14Roberts A, Matthews JB, Socransky SS, Freestone PP, Williams PH, Chapple IL. Stress and the periodontal diseases: growth responses of periodontal bacteria to Escherichia coli stress-associated autoinducer and exogenous Fe. Oral Microbiol Immunol. 2005 Jun;20(3):147-53.
-
15Saito T, Inagaki S, Sakurai K, Okuda K, Ishihara K. Exposure of P. gingivalis to noradrenaline reduces bacterial growth and elevates ArgX protease activity. Arch Oral Biol. 2011 Mar;56(3):244-50.
-
16Pullinger GD, Carnell SC, Sharaff FF, van Diemen PM, Dziva F, Morgan E, et al. Norepinephrine augments Salmonella enterica-induced enteritis in a manner associated with increased net replication but independent of the putative adrenergic sensor kinases QseC and QseE. Infect Immun. 2010 Jan;78(1):372-80.
-
17Jentsch HF, März D, Krüger M. The effects of stress hormones on growth of selected periodontitis related bacteria. Anaerobe. 2013 Dec;24:49-54
-
18Lyte M, Ernst S. Catecholamine induced growth of gram negative bacteria. Life Sci. 1992;50(3):203-12.
-
19Plotkin BJ, Roose RJ, Erikson Q, Viselli SM. Effect of androgens and glucocorticoids on microbial growth and antimicrobial susceptibility. Curr Microbiol. 2003 Dec;47(6):514-20.
-
20Tanabe S, Desjardins J, Bergeron C, Gafner S, Villinski JR, Grenier D. Reduction of bacterial volatile sulfur compound production by licoricidin and licorisoflavan A from licorice. J Breath Res. 2012 Mar;6(1):016006.
-
21Winn Jr WC, Janda WM, Koneman EW, Schreckenberger PC, Procop GW, Woods GL. Koneman’s color atlas and textbook of diagnostic microbiology. 6th ed. Philadelphia: Lippincott Willians & Wilkins Company; 2006. 1531 p.
-
22Winkel EG, Tangerman A. Appropriate sample bags and syringes for preserving breath samples in breath odor research: a technical note. J Breath Res. 2008 Mar;2(1):017011.
-
23Tangerman A, Winkel EG. Extra-oral halitosis: an overview. J Breath Res. 2010 Mar;4(1):017003.
-
24Akcali A, Huck O, Buduneli N, Davideau JL, Kose T, Tenenbaum H. Exposure of Porphyromonas gingivalis to cortisol increases bacterial growth. Arch Oral Biol. 2014 Jan;59(1):30-4.
-
25Coli JM, Tonzetich J. Characterization of volatile sulphur compounds production at individual gingival crevicular sites in humans. J Clin Dent. 1992;3(4):97-103.
-
26Yaegaki K, Sanada K. Biochemical and clinical factors influencing oral malodor in periodontal patients. J Periodontol. 1992 Sep;63(9):783-9.
-
27Kishi M, Ohara-Nemoto Y, Takahashi M, Kishi K, Kimura S, Aizawa F, et al. Prediction of periodontopathic bacteria in dental plaque of periodontal healthy subjects by measurement of volatile sulfur compounds in mouth air. Arch Oral Biol. 2013 Mar;58(3):324-30.
-
28O’Donnell PM, Aviles H, Lyte M, Sonnenfeld G. Enhancement of in vitro growth of pathogenic bacteria by norepinephrine: importance of inoculum density and role of transferrin. Appl Environ Microbiol. 2006 Jul;72(7):5097-9.
-
29Kennedy B, Dillon E, Mills PJ, Ziegler MG. Catecholamines in human saliva. Life Sci. 2001 May 25;69(1):87-99.
-
30Liening SH, Stanton SJ, Saini EK, Schultheiss OC. Salivary testosterone, cortisol, and progesterone: two-week stability, interhormone correlations, and effects of time of day, menstrual cycle, and oral contraceptive use on steroid hormone levels. Physiol Behav. 2010 Jan 12;99(1):8-16.
Publication Dates
-
Publication in this collection
2014
History
-
Received
20 Aug 2013 -
Reviewed
13 Jan 2014 -
Accepted
06 Feb 2014