Abstract
The aim of the study was to evaluate the effect of the incorporation of different fillers on an experimental vinyl polysiloxane (VPS) at two different concentrations, 20% and 40%. Different fillers were added to an experimental VPS. The study was developed in two stages: (i) incorporation of fillers in different concentrations: (a) 20 wt% fillers, and (b) 40 wt%. The fillers were added to experimental VPS and mixed with a speed mixer; (ii) characterization of experimental VPS; after the base paste and catalyst paste were mixed, the experimental VPS was used to make specimens specifically for each test, which were stored at 23°C for 24 hours. The tests were designed according to the specific standardization for the analysis of tensile strength, detail reproduction, Shore A hardness, and elastic recovery. For analysis of filler size pattern, scanning electron microscopy at 1500× magnification was used. The aerosil OX-50 40% (AE), and pure aluminum hydroxide 40% (PAH) groups presented the highest tensile strength and Shore A hardness values. However, those were the only groups that did not present continuous detail reproduction of an intersection of 20 μm line. The elastic recovery was not statistically significant. The undesirable characteristics of VPS (lowest Shore A hardness and tensile strength) were observed when it was added to the composition of acrylic polymer (AP) and fiberglass (FG) in both concentrations, 20% and 40%. In groups AE and PAH, agglomerates of nanofillers were shown in SEM micrography, while the other groups presented different shapes and fillers sizes.
Dental Impression Materials; Microscopy; Elementary Particles; Silicone Elastomers; Tensile Strength
Introduction
Addition silicones or vinyl polysiloxanes are elastomeric impression materials that have several advantages: resistance to rupture, moderate working times, fast elastic recovery, no smell or taste and usability even one week after preparation of the stone plaster cast model in dentistry.11. Al-Zarea BK, Sughaireen MG. Comparative analysis of dimensional precision of different silicone impression materials. J Contemp Dent Pract. 2011;12(3):208-15. doi:10.5005/jp-journals-10024-1036 The material is divided into two pastes, base and catalyst. Crosslinking occurs when the vinyl groups react with the hydride groups, which are catalyzed by platinum without generating by-products.22. Greco GD, Góes IMG, Ferreira JC, Greco ADL. Analysis of dimensional changes of plaster models obtained with the addition silicones in multiple leaks. Arq Bras Odontol. 2009;5(2):53-7. Portuguese. The material has different viscosities and can be used in different techniques. The light composition is fluid enough to flow and copy gingival crevices, while the putty version is viscous enough to promote removal of soft tissue and retain the impression.
Generally, vinyl polysiloxanes are used when a good copy is necessary for the production of dental prostheses and indirect restorations without distortions.33. Kumar S, Gandhi UV, Banerjee S. An in vitro study of the bond strength of five adhesives used for vinyl polysiloxane impression materials and tray materials. J Indian Prosthodont Soc. 2014;14(1):61-6. doi:10.1007/s13191-012-0243-5 Despite technical improvements, such as CAD/CAM and 3-D systems, the conventional impression process continues to play an important role in the transfer of patient information to the dental laboratory.44. Stober T, Johnson GH, Schmitter M. Accuracy of the newly formulated vinyl siloxanether elastomeric impression material. J Prosthet Dent. 2010;103(4):228-39. doi:10.1016/S0022-3913(10)60035-2 The impression is the main step in obtaining a well-adjusted restoration, therefore, the ability reproduce details, elastic recovery, and resistance to tensile strength are of primary importance for producing dental prostheses without the need for adjustments.
Fillers are used in dentistry in various materials such as composite resin, resin cements, glass ionomer and many others. There are different reasons for choosing fillers for dental materials, related to providing them with color, translucence,55. Naeimi AH, Moharamzadeh K, Wood DJ, Van Noort R. Relationship between color and translucency of multishaded dental composite resins. Int J Dent. 2012;2012:708032. doi:10.1155/2012/708032opalescence,66. Kelly JR, Benetti P. Ceramic materials in dentistry: historical evolution and current practice. Aust Dent J. 2011;56(Suppl 1):s84-96. doi:10.1111/j.1834-7819.2010.01299.x radiopacity,77. Fortin D, Vargas MA. The spectrum of composites: new techniques and materials. J Am Dent Assoc. 2000;131(Suppl 131):26S-30S. doi:10.14219/jada.archive.2000.0399,88. Sakaguchi RL, Power JM. Craig’s restorative dental materials. 13th ed. Philadelphia: Elsevier; 2012. rheology99. ramer NB, Stansbury JW, Bowman CN. Recent advances and developments in composite dental restorative materials. J Dent Res. 2011;90(4):402-16. doi:10.1177/0022034510381263 and appropriate resistance. In the formulation of vinyl polysiloxanes, many variations can usually be combined. The inherent characteristics of the filler determine the majority of the material properties. When size, surface area, and different shapes are properly combined, it improves the properties of vinyl polysiloxanes.1010. Mandikos MN. Polyvinyl siloxane impression materials: an update on clinical use. Aust Dent J. 1998;43(6):428-34. doi:10.1111/j.1834-7819.1998.tb00204.x
The addition of inorganic and organic fillers is very common to reinforce the mechanical strength of polymers1111. Suzuki N, Kiba S, Kamachi Y, Miyamoto N, Yamauchi Y. Unusual reinforcement of silicone rubber compounds containing mesoporous silica particles as inorganic fillers. Phys Chem Chem Phys. 2012;14:3400-7. doi:10.1039/c2cp23864kand to increase their mechanical properties, because the rigidity of inorganic agents is more relevant with regard to the organic portion of the polymer.1212. Shao-Yun F, Xi-Qiao F, Lauke B, Yiu-Wing M. Effects of particle size, particle/matrix interface adhesion and particle loading on mechanical properties of particulate-polymer composites. Compos Part B Eng. 2008;39(6):933-61. doi:10.1016/j.compositesb.2008.01.002 In most applications, sílica has been used as the main type of reinforcing filler used to increase the properties1313. Rattanasom N, Saowapark T, Deeprasertkul C. Reinforcement of natural rubber with silica/carbon black hybrid filler. Polym Test. 2007; 26(3):369-77. doi:10.1016/j.polymertesting.2006.12.003 of vinyl polysiloxanes, however, other types of non-black fillers are also used such as calcium carbonate, diatomite and fiberglass.
A filler must make intimate contact with the elastomer chains if it is going to contribute to the reinforcement of the rubber-filler composite. Fillers with a large surface area have more contact area available, and therefore, have a higher potential for reinforcement. This makes it very important to have a shape with the desired properties1414. Kingender RC, editor. Handbook of specialty elastomers. 1st ed: CRC Press; 2008. to influence the viscosity, and consequently, the material properties.1515. Tjan AH, Li T. Effects of reheating on the accuracy of addition silicone putty-wash impressions. J Prosthet Dent. 1991;65(6):743-8. doi:10.1016/S0022-3913(05)80005-8,1616. Craig RG, Sun Z. Trends in elastomeric impression materials. Oper Dent. 1994;19(4):138-45.
The aim of this study was to evaluate different properties of a vinyl polysiloxanes using ten different fillers in two different concentrations, 20% and 40%, added to a polymer matrix of an experimental vinyl polysiloxanes. The null hypothesis was that there would be no significant difference in the properties with regard to these types of fillers and their concentration in the material.
Methodology
The experimental vinyl polysiloxane without fillers (VPS) was produced by the company Yller and used to determine all the characterizations. The basic composition is described in Table 1.
Ten different types of fillers in two different concentrations, 20 wt% and 40 wt%, were incorporated into the experimental VPS for further evaluation. The fillers and the manufactures are described in Table 2.
The fillers were added to a composition of 30 mL of base paste and 30 mL of catalyst paste. The materials were inserted in individual vials and mixed in a rapid mixing machine (SpeedMixer DAC150.1FVZ; Synergy Devices; High Wycombe, United Kingdom) at 3500 rpm for 60 seconds. For all the groups and all the tests, the two pastes of VPS were mixed with self-mixing tips.
Tensile strength analysis was performed using a rectangular mold (50 mm x 16 mm x 1.5 mm) with a central constriction (8 mm), as an hourglass. The mold was filled with experimental VPS, and a polyester strip and a glass slide were placed onto the filled molds. After five minutes, the specimens (n = 10) were removed and stored at room temperature (23°C) for 24 hours. The universal testing machine (DL500; EMIC; São José dos Pinhais, Brazil) with two claws moving in opposite directions at a speed of 0.5 mm/min, was used to pull the specimen to the point of failure.
A detail reproduction test was performed in accordance with the ISO 4823:2000 standard, by using a mold with three horizontal lines measuring 20 μm, 50 μm and 75 μm and two vertical lines that cut their ends. The lines were molded, and the intersection of the horizontal and vertical lines created after taking the impression, was evaluated. After five minutes, the specimens (n = 3) were removed and stored at room temperature (23°C) for 24 hours. The samples were observed using a stereomicroscope (100×). The detail reproduction of an intersection of 20 μm, reproduced continuously, was considered satisfactory.
The Shore A hardness test was performed in accordance with the ISO 868 standard, by using the matrix (thickness 0.6 cm x diameter 1.5 cm) to make specimens for test. The mould was filled with experimental VPS, and a polyester strip and a glass slide were placed onto the filled molds. After five minutes, the specimens (n = 3) were removed and stored at room temperature (23°C) for 24 hours. A digital durometer (Shore A Durometer; Mitutoyo; Kawasaki, Japan) was used to determine the hardness in three different points, and the mean values were calculated.
The elastic recovery test was performed in accordance with the ISO 4823:2000 standard, by using a cylindrical mold (thickness 1.25 cm x diameter 2.0 cm) filled with experimental VPS. After 5 minutes, the specimens (n = 3) were removed and stored at room temperature (23°C) for 24 hours and evaluated according to the following the equation:
where k is the constant of elastic recovery,h0 the height of the model,h1 the initial reading of the indicator andh2 the final reading of the indicator (immediately after the deforming force has been removed from the sample).
The morphology of inorganic fillers was examined by scanning electron microscopy (SEM) (JSM-6610/LV; JEOL; Waltham, USA). The fillers were covered with gold and analyzed at 1500× magnification.
Data were analyzed using ANOVA One Ranks, Dunn test with p < 0.001 for tensile strength tests, and one-way ANOVA and Tukey test with p < 0.05 for Shore A hardness and elastic recovery tests.
Results
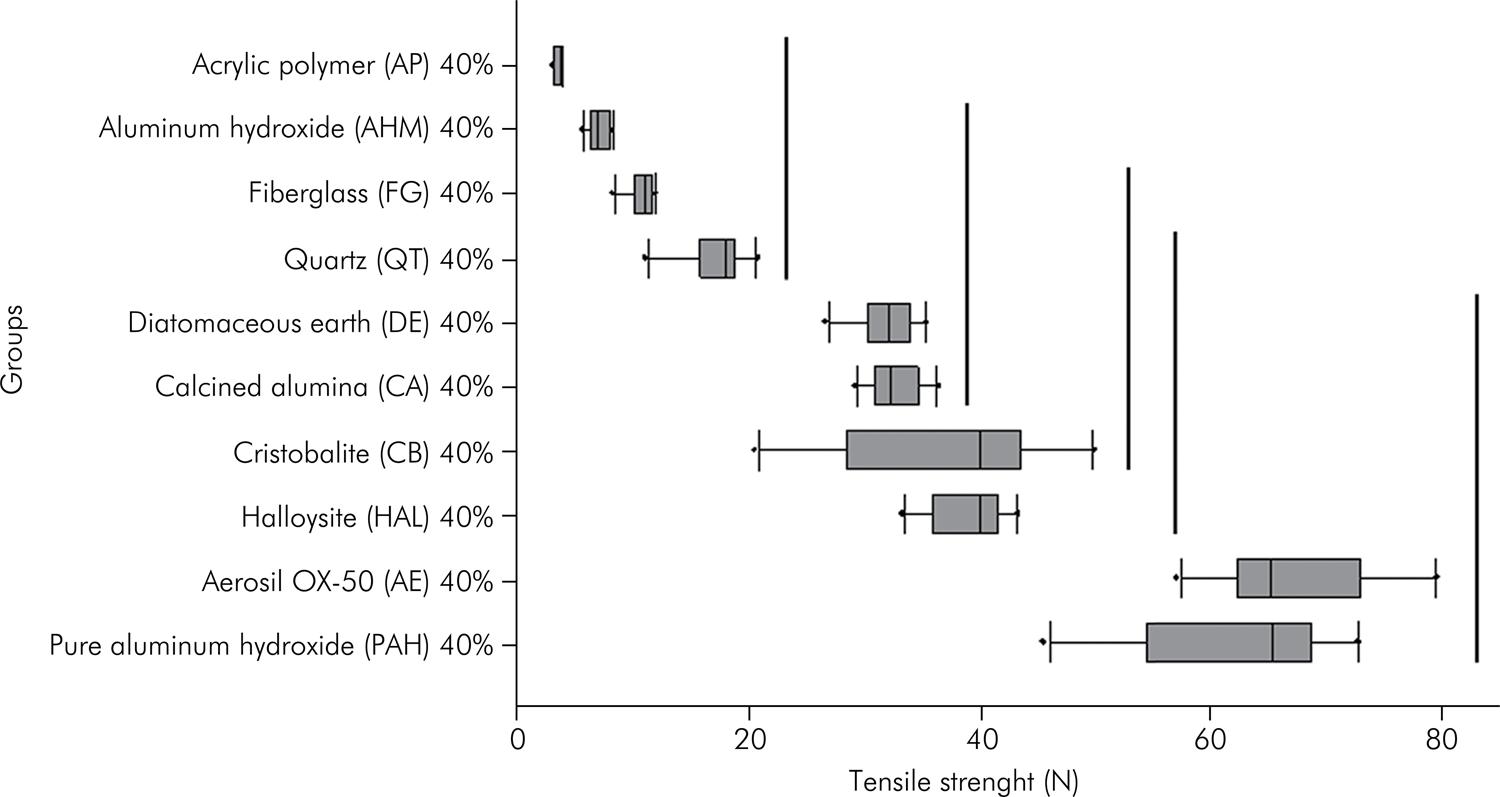
Figure 1 and the Figure 2 show the results of the mechanical tensile strength test. The groups with concentrations of 20 wt% fillers presented half the force values (N) compared with those with the concentration of 40 wt% fillers. However, for acrylic polymer (AP), aluminum hydroxide (AHM), fiberglass (FG) and quartz (QT) fillers, despite the increase in filler concentration, no significant increase in mechanical tensile strength was observed. The aerosil OX-5O (AE) and pure aluminum hydroxide (PAH) fillers showed increased resistance with increase in filler loading. The lowest results shown were for acrylic polymer (AP) aluminum hydroxide (AHM), fiberglass (FG) and quartz (QT) fillers for both concentrations.
Mechanical tensile strength found in groups with concentrations of 20 wt% fillers (n = 10) (ANOVA One Ranks, Dunn Test p < 0.001).
Mechanical tensile strength found in groups with concentrations of 40 wt% fillers (n = 10) (ANOVA One Ranks, Dunn Test p < 0.001).
Detail reproduction tests of groups aerosil OX-5O (AE) and pure aluminum hydroxide (PAH) fillers in both concentration showed they did not create a satisfactory reproduction, while for the other groups, a continuous detail reproduction of the 20 µm line was shown (Figure 3).
Detail reproduction of lines measuring 20 μm, 50 μm and 75 μm. (A) aerosil OX-50 (AE) 40 wt%, (B) pure aluminum hydroxide (PAH) 40 wt%, (C) calcined alumina (CA) 40 wt%.
After analysis, it was concluded there was no statistically significant difference between the groups for elastic recovery, whereas Shore A hardness presented different results for the different fillers. The highest hardness values were shown in the aerosil OX-50 (AE 40%) and pure aluminum hydroxide (PAH 40%) groups, and the lowest for the cristobalite (CB 20%) and fiberglass (FG 20%) groups, which were statistically similar (Table 3).
The morphological characteristics of different types of fillers are shown in Figure 4. Some were shown to be stick-shaped, such as those of fiberglass (FG), others were spherical-shaped such as those of acrylic polymer (AP). Nevertheless, some had the appearance of single fillers, and in reality they were agglomerates of nanofillers, such as those of aerosil OX-50 (AE) and pure aluminum hydroxide (PAH); irregular shapes were also shown in the SEM micrograph, with more superficial areas, such as those of the diatomaceous earth (DE), halloysite (HAL), calcined alumina (CA) fillers, the cited Figure 4 are F, H, A, E, J, G and B, respectively.
SEM micrographs showing the morphology of the different fillers: (A) aerosil OX-50; (B) calcined alumina; (C) cristobalite; (D) aluminum hydroxide; (E) pure aluminum hydroxide; (F) fiberglass; (G) halloysite; (H) acrylic polymer; (I) quartz and (J) diatomaceous earth.
Discussion
Filler is the most-used term for referring to material that is intended to augment weight or fill space. Although the original purpose of fillers in commodity materials was to lower the cost of products, nowadays it is very important to select the filler characteristics desirable for promoting better mechanical or optical properties in the material. Unreinforced polymers often exhibit properties that do not meet the technical specifications demanded. The addition of mineral/inorganic fillers, adequately surface-treated, contribute to changes with respect to mechanical properties such as hardness and tensile strength.
The fillers shown in Figure 2. have different morphological characteristics. Most fillers are isometric; others are spherical; others are fiber-shaped or aggregate into clusters.1717. Carlo HL, Fonseca RB, Soares CJ, Correr AB, Correr-Sobrinho L, Sinhoreti MA. Inorganic particle analysis of dental impression elastomers. Braz Dent J. 2010;21(6):520-7. doi:10.1590/S0103-64402010000600007 In Figure 4A(aerosil OX-50) it is possible to observe clustered fillers. Silica has small primary fillers that are joined together with other fillers of a similar size to make up larger aggregate structures. This cluster structure is not permanent, because hydrogen bonding among fillers will form clusters or aggregates constantly.1414. Kingender RC, editor. Handbook of specialty elastomers. 1st ed: CRC Press; 2008. The small size of this filler favors the increased viscosity of the elastomer, reducing the flow and copy capacity.
Pure aluminum hydroxide and aluminum hydroxide Al(OH)3 fillers, shown inFigure 4E and 4D respectively, are also agglomerate fillers, but in aluminum hydroxide, the fillers are of a larger size than they are in the others. A filler must make intimate contact with the elastomer chains if it is going to contribute to reinforcement of the polymer. Fillers that have a large surface area have more contact area available, and therefore, have a higher potential of reinforcing the elastomer.1414. Kingender RC, editor. Handbook of specialty elastomers. 1st ed: CRC Press; 2008. Even when using the same chemical composition, a pure version of the material was able to promote better results. The higher the concentration of pure aluminum hydroxide added, the higher was the Shore A hardness, and the greater was the material resistance to traction. Purity seems to have been decisive for the material to perform better.
Halloysite nanotubes are formed from aluminosilicate minerals and have a hollow nanotube structure and a larger luminal diameter that is capable of accommodating different polymer molecules, which offers a larger variety of polymeric composites. Halloysite nanotubes are made up of siloxane and hydroxyl groups, which provide the potential for hydrogen bond formation, thereby improving dispersion. Their surface modification also enhances their wettability and bond formation with different polymers.1818. Rawtani D, Agrawal YK. Multifarious Applications of halloysite nanotubes: a review. Rev Adv Mater Sci. 2012;30(3):282-95. In Figure 1, it is possible to compare the tensile strength tests of the diatomaceous earth, calcined alumina, cristobalite, and quartz groups. The diatomaceous earth is composed of 80-90% silica; calcined alumina is composed of Al2O3; cristobalite presents a structure alternately pointing up and down; and the quartz has helical arrangements of SiO4 tetrahedra. Although they are similar, even those with the same chemical composition have different properties, because they have a different crystal structure. This difference did not create differences in the tests evaluated, however, these groups with additions of 20% and 40% were statistically similar in both the hardness and tensile strength tests.
The fiberglass filler is a synthetic material primarily made of silica, which does not have a melting point and has long been used for its hardness properties. Generally, fiberglass was added to the matrix of materials with different compositions, which are not vinyl polysiloxane, and was expected to increase the material strength. In Figure 4F, it is possible to observe the dispersed sticks showing a small surface area. In all the tests performed with groups to which glass fiber was added, the performance of the material was the worst. It is possible that the amount of fiber added was not sufficient to promote additional strengthening for this type of silicone formulation.
The only filler that was not a mineral and had a spherical shape was the acrylic polymer, mainly composed of methyl methacrylate, shown in Figure 4H. This organic filler generally offers less affinity and less surface activity in comparison with elastomers.1414. Kingender RC, editor. Handbook of specialty elastomers. 1st ed: CRC Press; 2008. It does not have the ability to bond to the rubber matrix, because the strongest bond it creates is via Van Der Waals force, which is a relatively weak force. The reduction in area of contact with the organic matrix is directly related to the results of the tensile strength test, because when all the spaces of the organic matrix are not filled, there is a freer polymeric matrix, providing the material with less strength and making it an inactive filler. Furthermore, the acrylic polymer has a very large filler size of 50 microns, and theoretically, the larger the size of filler added to promote the material strength, the lower the tensile strength.
As previously mentioned, fillers may be of the reinforcing or active type, increase the viscosity and hardness of the rubber compound and improve the properties of tensile strength and abrasion resistance up to a limit, as the amount of the load increases. Typically, while the hardness values increase, the values of other properties, such as resilience, decrease.
The fillers aggregation tendency increases as the filler size decreases, and extensive aggregation leads to a polymer with insufficient homogeneity, lower stiffness, and lower compressive strength. These filler aggregate tends to act as a break for occasional initiators when affected by some impact.
The filler size is not only related to viscosity but also to tensile strength and detail reproduction. Shao-Yun et al.1212. Shao-Yun F, Xi-Qiao F, Lauke B, Yiu-Wing M. Effects of particle size, particle/matrix interface adhesion and particle loading on mechanical properties of particulate-polymer composites. Compos Part B Eng. 2008;39(6):933-61. doi:10.1016/j.compositesb.2008.01.002 stated that fillers have sizes ranging from 10 nm to 80 nm to 1.3 micrometers to 58 micrometers, and for a given filler volume, the tensile strength increases as the filler size decreases.1212. Shao-Yun F, Xi-Qiao F, Lauke B, Yiu-Wing M. Effects of particle size, particle/matrix interface adhesion and particle loading on mechanical properties of particulate-polymer composites. Compos Part B Eng. 2008;39(6):933-61. doi:10.1016/j.compositesb.2008.01.002 This indicates that the resistance increases with the increase in filler surface area, favoring copy ability in the same way as smaller filler sizes are better able to copy small spaces.
The accuracy of impression materials such as VPS is demonstrably better than other types of impression taking materials.1919. Chen SY, Liang WM, Chen FN. Factors affecting the accuracy of elastometric impression materials. J Dent. 2004;32(8):603-9. doi:10.1016/j.jdent.2004.04.002 One of the factors that enhances the accuracy of impressions is the copy ability, which is related to the filler size and viscosity of these materials. This is partly determined by the amount of filler, demonstrating that materials with high viscosity have a larger amount of filler material than those with low viscosity. Materials with a smaller amount of filler have lower viscosity and consequently a greater likelihood of impression distortion. This is probably due to the larger organic matrix, which does not have the ability to impart shape to materials, only to promote the agglutination of the filler.1010. Mandikos MN. Polyvinyl siloxane impression materials: an update on clinical use. Aust Dent J. 1998;43(6):428-34. doi:10.1111/j.1834-7819.1998.tb00204.x,1717. Carlo HL, Fonseca RB, Soares CJ, Correr AB, Correr-Sobrinho L, Sinhoreti MA. Inorganic particle analysis of dental impression elastomers. Braz Dent J. 2010;21(6):520-7. doi:10.1590/S0103-64402010000600007
However, when a larger amount of filler was added to the organic matrix in this study, the opposite was observed only with aerosil OX-50 and pure aluminum hydroxide fillers at concentrations of 40%, which showed increase in viscosity and worse copy ability. When comparing concentrations of 20%, the increase in the concentration of the fillers decreased the quality of detail reproduction, because the material was unable to drain from the mold grooves so that it could copy the really necessary details. Whereas the other materials, because they have hybrid filler sizes (10 to 80 nm), showed good copy ability, whether in concentrations of 20% or 40%, probably because the fillers could not enter into full contact with the organic matrix, thereby enhancing the flow of the matrix between the fillers.
However, is not sufficient to have good copy ability only. When an impression is removed from the oral cavity, the material needs to withstand the force that separates it from the teeth and surrounding tissues. If a small strain is applied to a solid material, deformation begins, and the material will continue to deform until the molecular strain establishes a balance with external strains. Most solids exhibit some degree of elastic response, meaning complete deformation recovery after deforming strains.2020. Guerra BB, Furtado CRGC, Coutinho FMB. Rheological evaluation of elastomers and their compounds. Polimeros. 2004;14(4):289-94. Portuguese. doi:10.1590/S0104-14282004000400017 Thus, elastic recovery becomes important in determining the precision of an impression material.2121. Lu H, Nguyen B, Powers JM. Mechanical properties of 3 hydrophilic addition silicone and polyether elastomeric impression materials. J Prosthet Dent. 2004;92(2):151-4. doi:10.1016/j.prosdent.2004.05.016 In this study, the ISO 4823 standard specification was used, which requires recovery of over 96.5%, yielding values between 98.91% and 99.91% for pure aluminum hydroxide and quartz, respectively, as shown in Table 2. These values can be explained by the interstitial filling of organic matrix. In contrast, when complete or almost total filling of the matrix is used, the displacement of the particulates becomes more difficult, and depending on the filler format, slippage occurs. Whereas, the resultant elastic recovery values are due to the impossibility of filler movement. In the case of VPS, this is very small, which explains the strain values of less than 2%.
The tensile strength of a material is defined as the maximum stress the material can withstand under uniaxial traction.1212. Shao-Yun F, Xi-Qiao F, Lauke B, Yiu-Wing M. Effects of particle size, particle/matrix interface adhesion and particle loading on mechanical properties of particulate-polymer composites. Compos Part B Eng. 2008;39(6):933-61. doi:10.1016/j.compositesb.2008.01.002 As can be seen in Figure 1, the tensile strength values were derived from the maximum stress that could be applied to the specimens through elongation before they ruptured. This property can be used to indicate the ability of an impression material to withstand interproximal tearing in thin areas. Indeed, clinically it has been observed when taking impressions of a gingival sulcus; where the flap should be copied, it often leads to having no continuity because it is necessary to use the minimum amount of impression material2222. Yi MH, Shim JS, Lee KW, Chung MK. Drying time of tray adhesive for adequate tensile bond strength between polyvinylsiloxane impression and tray resin material. J Adv Prosthodont. 2009;1(12):63-7. doi:10.4047/jap.2009.1.2.63 to achieve the required uniform thickness. Materials with less filler have lower tensile strength when compared with each other, as can be seen Figure 1 when comparing the 20% fillers with the 40% fillers, which show an increased resistance in terms of values. Thus, more viscous materials have higher tensile strength values due to the increased amount of filler and tend not to tear so easily. Less viscous materials, on the other hand, produce a smaller film thickness and tear more easily.1717. Carlo HL, Fonseca RB, Soares CJ, Correr AB, Correr-Sobrinho L, Sinhoreti MA. Inorganic particle analysis of dental impression elastomers. Braz Dent J. 2010;21(6):520-7. doi:10.1590/S0103-64402010000600007,2121. Lu H, Nguyen B, Powers JM. Mechanical properties of 3 hydrophilic addition silicone and polyether elastomeric impression materials. J Prosthet Dent. 2004;92(2):151-4. doi:10.1016/j.prosdent.2004.05.016,2222. Yi MH, Shim JS, Lee KW, Chung MK. Drying time of tray adhesive for adequate tensile bond strength between polyvinylsiloxane impression and tray resin material. J Adv Prosthodont. 2009;1(12):63-7. doi:10.4047/jap.2009.1.2.63
As regards Shore A hardness, we observed a large discrepancy between the values according to filler type. The VPS with 20% fiberglass revealed mean hardness values of 14.50, which differed statistically from all other fillers except for cristobalite 20%. Its morphology can be observed in Figure 2, in which the filler sizes are similar, and this is explained by the ease of dispersion of the organic matrix around the fillers. The similarity between 40% aerosil OX-50 and 40% pure aluminum hydroxide fillers can be explained by the nanometer size of the fillers, which promotes increased contact between the organic and inorganic matrix. This, in turn, reduces the interstices of the matrix and increases the hardness values of the material, shown by means of the tip of the tester that cannot penetrate into the VPS. However, the fact that the material is harder does not mean it will be more difficult to handle or remove from the oral cavity.2323. Lawson NC, Burgess JO, Litaker M. Tear strength of five elastomeric impression materials at two setting times and two tearing rates. J Esthet Restor Dent. 2008;20(3):186-93. doi:10.1111/j.1708-8240.2008.00176.x,2424. Walker MP, Alderman N, Petrie CS, Melander J, McGuire J. Correlation of impression removal force with elastomeric impression material rigidity and hardness. J Prosthodont. 2013;22(5):362-6. doi:10.1111/jopr.12011
Therefore, the null hypothesis of this study was rejected because there was no statistically significant difference between the mechanical tests, except for the elastic recovery test.
Conclusion
This study, teaches us about the properties and uses of vinyl polysiloxanes. The incorporation of different filler concentrations considerably modified the properties of most materials. The composition, shape and particle size influences the material properties. The correct selection and use of different particles in the same formulation can significantly improve the properties of materials. The type of fillers used in the composition have the potential to influence the clinical performance of an impression material. The use of different fillers may be an important factor for promoting synergy between materials.
Acknowledgements
F.A.O. is grateful for the support from Financiadora de Estudos e Projeto - FINEP/Brazil (grant 01.10.0709.00). The authors are grateful to Centro de Microscopia Eletrônica do Sul - CEME-SUL -Fundação Universidade Federal do Rio Grande do Sul - FURG/ Brazil for SEM analysis. The authors thank Yller for donating of experimental polyvinylsiloxane, without fillers used in the study.
References
-
1Al-Zarea BK, Sughaireen MG. Comparative analysis of dimensional precision of different silicone impression materials. J Contemp Dent Pract. 2011;12(3):208-15. doi:10.5005/jp-journals-10024-1036
-
2Greco GD, Góes IMG, Ferreira JC, Greco ADL. Analysis of dimensional changes of plaster models obtained with the addition silicones in multiple leaks. Arq Bras Odontol. 2009;5(2):53-7. Portuguese.
-
3Kumar S, Gandhi UV, Banerjee S. An in vitro study of the bond strength of five adhesives used for vinyl polysiloxane impression materials and tray materials. J Indian Prosthodont Soc. 2014;14(1):61-6. doi:10.1007/s13191-012-0243-5
-
4Stober T, Johnson GH, Schmitter M. Accuracy of the newly formulated vinyl siloxanether elastomeric impression material. J Prosthet Dent. 2010;103(4):228-39. doi:10.1016/S0022-3913(10)60035-2
-
5Naeimi AH, Moharamzadeh K, Wood DJ, Van Noort R. Relationship between color and translucency of multishaded dental composite resins. Int J Dent. 2012;2012:708032. doi:10.1155/2012/708032
-
6Kelly JR, Benetti P. Ceramic materials in dentistry: historical evolution and current practice. Aust Dent J. 2011;56(Suppl 1):s84-96. doi:10.1111/j.1834-7819.2010.01299.x
-
7Fortin D, Vargas MA. The spectrum of composites: new techniques and materials. J Am Dent Assoc. 2000;131(Suppl 131):26S-30S. doi:10.14219/jada.archive.2000.0399
-
8Sakaguchi RL, Power JM. Craig’s restorative dental materials. 13th ed. Philadelphia: Elsevier; 2012.
-
9ramer NB, Stansbury JW, Bowman CN. Recent advances and developments in composite dental restorative materials. J Dent Res. 2011;90(4):402-16. doi:10.1177/0022034510381263
-
10Mandikos MN. Polyvinyl siloxane impression materials: an update on clinical use. Aust Dent J. 1998;43(6):428-34. doi:10.1111/j.1834-7819.1998.tb00204.x
-
11Suzuki N, Kiba S, Kamachi Y, Miyamoto N, Yamauchi Y. Unusual reinforcement of silicone rubber compounds containing mesoporous silica particles as inorganic fillers. Phys Chem Chem Phys. 2012;14:3400-7. doi:10.1039/c2cp23864k
-
12Shao-Yun F, Xi-Qiao F, Lauke B, Yiu-Wing M. Effects of particle size, particle/matrix interface adhesion and particle loading on mechanical properties of particulate-polymer composites. Compos Part B Eng. 2008;39(6):933-61. doi:10.1016/j.compositesb.2008.01.002
-
13Rattanasom N, Saowapark T, Deeprasertkul C. Reinforcement of natural rubber with silica/carbon black hybrid filler. Polym Test. 2007; 26(3):369-77. doi:10.1016/j.polymertesting.2006.12.003
-
14Kingender RC, editor. Handbook of specialty elastomers. 1st ed: CRC Press; 2008.
-
15Tjan AH, Li T. Effects of reheating on the accuracy of addition silicone putty-wash impressions. J Prosthet Dent. 1991;65(6):743-8. doi:10.1016/S0022-3913(05)80005-8
-
16Craig RG, Sun Z. Trends in elastomeric impression materials. Oper Dent. 1994;19(4):138-45.
-
17Carlo HL, Fonseca RB, Soares CJ, Correr AB, Correr-Sobrinho L, Sinhoreti MA. Inorganic particle analysis of dental impression elastomers. Braz Dent J. 2010;21(6):520-7. doi:10.1590/S0103-64402010000600007
-
18Rawtani D, Agrawal YK. Multifarious Applications of halloysite nanotubes: a review. Rev Adv Mater Sci. 2012;30(3):282-95.
-
19Chen SY, Liang WM, Chen FN. Factors affecting the accuracy of elastometric impression materials. J Dent. 2004;32(8):603-9. doi:10.1016/j.jdent.2004.04.002
-
20Guerra BB, Furtado CRGC, Coutinho FMB. Rheological evaluation of elastomers and their compounds. Polimeros. 2004;14(4):289-94. Portuguese. doi:10.1590/S0104-14282004000400017
-
21Lu H, Nguyen B, Powers JM. Mechanical properties of 3 hydrophilic addition silicone and polyether elastomeric impression materials. J Prosthet Dent. 2004;92(2):151-4. doi:10.1016/j.prosdent.2004.05.016
-
22Yi MH, Shim JS, Lee KW, Chung MK. Drying time of tray adhesive for adequate tensile bond strength between polyvinylsiloxane impression and tray resin material. J Adv Prosthodont. 2009;1(12):63-7. doi:10.4047/jap.2009.1.2.63
-
23Lawson NC, Burgess JO, Litaker M. Tear strength of five elastomeric impression materials at two setting times and two tearing rates. J Esthet Restor Dent. 2008;20(3):186-93. doi:10.1111/j.1708-8240.2008.00176.x
-
24Walker MP, Alderman N, Petrie CS, Melander J, McGuire J. Correlation of impression removal force with elastomeric impression material rigidity and hardness. J Prosthodont. 2013;22(5):362-6. doi:10.1111/jopr.12011
Publication Dates
-
Publication in this collection
2016
History
-
Received
09 Aug 2015 -
Received
22 Sept 2015 -
Accepted
18 Nov 2015