LETTER TO THE EDITOR
Fibromatosis of the male breast: a case report with immunohistochemistry study and review of the literature
Alexandre Henrique Macchetti; Heitor Ricardo Cosiski Marana; Alfredo Ribeiro-Silva; Jurandyr Moreira de Andrade; Carlos Francisco Erbolato Melo
Outpatient Breast Oncology Service, Department of Health of São Carlos and Department of Gynecology and Obstetrics (Breast Service) of Ribeirão Preto, SP/Brazil. Email: ahmacchetti@yahoo.com.br
Fibromatosis is an infiltrative proliferation of fibroblastic and myofibroblastic cells with significant risk for local recurrence, but no metastatic potential.1 It originates mainly from the fascia or aponeuroses of the abdominal wall muscles or from the muscles of the shoulders and pelvic girdles.1 Fibromatosis is uncommon in the mammary gland and accounts for less than 0.2% of all primary breast lesions.2,3 Although several series of this condition have been reported in women, mammary fibromatosis in men is extremely rare.47 Despite its rarity, this condition may mimic primary breast malignancy. In cases where suspicious breast findings do not correlate with the usual diagnostic measures, such as those from fine-needle aspiration or core needle biopsy, the diagnosis of a fibromatosis mimicking breast cancer should be considered in the differential diagnosis.89
We report the case of a 47-year-old man with mammary fibromatosis. To the best of our knowledge, this is the sixth case of fibromatosis reported in the male breast.47 The optimal management of breast fibromatosis in men is unknown because of the rarity of the disease. A review of articles published on breast fibromatosis was performed using the PUBMED-MEDLINE database with emphasis on articles published during the last 10 years.
CASE REPORT
We present the case of a 47-year-old man who was admitted to the Outpatient Breast Oncology Service, Department of Health of São Carlos, Brazil, with a 3-month history of a painless subcutaneous mass with rapid growth. Clinical examination revealed an ill-defined mass located in the upper inner quadrant of his left breast tissue measuring of 3.5 cm in diameter. The mass was firm and was fixed to the pectoral fascia and to skin. There was no axillary or cervical lymphadenopathy or nipple discharge.
At ultrasound, the lesion manifested as an irregular, hypoechoic mass with posterior acoustic shadowing, suggestive of malignancy. Fine-needle aspiration cytology was performed, but the result was inconclusive because the aspirate did not yield sufficient epithelial cells for diagnosis. The tumor was removed by en bloc resection with wide excision, the greater pectoral muscle being spared. During surgery the lesion was found to be firmly adherent to the underlying pectoralis major. The appearance was that of an infiltrative tumor, and the diagnosis of epithelial neoplasm was considered in the differential diagnosis. There was no history of trauma to the chest wall, so that a fibrous reaction to trauma was not considered in the differential diagnosis.
The surgical specimen was fixed in 4% formalin. Sequential 3-mm sections were obtained throughout the specimen. Representative samples of the tumor were embedded in paraffin, and histological sections of 4 µm obtained from the paraffin-embedded blocks were stained with hematoxylin-eosin. The histological sections stained with hematoxylin-eosin showed a proliferation of spindle cells without atypia forming sweeping or interlacing fascicles (Figure 1). The cellularity varied from mild to moderate. There was a variable amount of collagen predominantly located in the center of the lesion. Mitotic figures were absent. At the periphery of the lesions, there were focally irregular and infiltrating fingerlike projections extending into the adjacent breast tissue. There was no inflammatory infiltration at the periphery of the lesion. Dilated vessels and extravasated red cells were noted throughout the lesions.
For immunohistochemical staining, the paraffin-embedded blocks were cut into 3-µm sections, deparaffinized, and rehydrated. A standard avidin-biotin-peroxidase method was used (Novostain Super ABC kit, Novocastra, Newcastle upon Tyne, UK). The following primary antibodies were used: alpha smooth muscle actin (clone 1A4, Dako, Carpinteria, CA, 1:100), bcl-2 (clone Bcl-2/100/D5, Novocastra, 1:50), CD10 (clone 56C6, Novocastra, 1:200), CD117 (c-kit oncoprotein) (clone T595, Novocastra, 1:40), c-erbB-2 (clone CB11, Novocastra, 1:300), cytokeratin AE1/AE3 (clone AE1 & AE3, Dako, 1:1000), estrogen receptor (ER; clone 6F11, Novocastra, 1:100), Ki67 (clone MM1, Novocastra, 1:150), p53 (clone DO-7, Novocastra, 1:100), p63 (clone 4A4, Santa Cruz, Palo Alto, CA, 1:100), progesterone receptor (PR; clone 16, Novocastra, 1:150), S100 (polyclonal, Dako, 1:4000), and vimentin (clone V9, Dako, 1:100). After the reactions, the sections were developed for 5 minutes with 3.3-diaminobenzidine tetrahydrochloride (DAB) in phosphate-buffered saline, pH 7.5, containing 0.036% hydrogen peroxide. Light Mayer's hematoxylin was applied as a counterstain. The slides were then dehydrated in a series of ethanols and mounted with Permount (Fischer, Fairlawn, NJ).
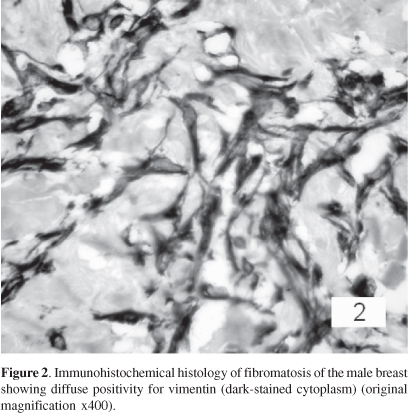
The immunohistochemistry study showed diffuse positivity for vimentin (Figure 2), and focal positivity for smooth muscle actin. The other markers were negative.
DISCUSSION
In spite of the abundance of reported cases and series of extramammary fibromatosis in the literature, this is only the sixth case reported in the male breast, and like previous descriptions, this patient presented with a history of a palpable nontender breast mass with breast carcinoma-like symptoms, such as fast growth and muscular infiltration, skin infiltration and dimpling, nipple retraction, mammographically indistinguishable from breast carcinoma.10,11 Although tumor location was unusual for breast carcinomas in menthe inner superior quadrantand breast malignancy is uncommon at this age, the presence of breast cancer was the principal suspected clinical diagnosis.10 McKinnon et al12 noted that the correct diagnosis of desmoid tumors was made preoperatively in only 50% of cases and that, even after biopsy, the diagnosis was often confused with low-grade fibrosarcoma, confirming the difficulty of diagnosis for this pathology.
Mammary fibromatosis presents macroscopically as a dense, poorly vascularized, hard, rubbery, grayish-white mass, and histologically, it is described as dense collagenous material with intertwining bundles of spindle cells and lacking epithelial components. These tumors do not metastasize, have no capsule, infiltrate into local structures (unlike fibrosarcomas), have normal mitotic characteristics, and frequently recur after attempted surgical excision.1,3,13
The extra-abdominal sites most commonly involved are the chest wall, shoulder girdle, inguinal region, and neck.1,3 Of this group, the chest wall is the most common site, usually involving the anterior muscles. Whether breast involvement is an extension from a primary site within the fibro-aponeurotic fascia or the pectoral muscle or whether it results from fibroblasts originating from within the breast parenchyma is undetermined. However, fibromatosis arising within the breast parenchyma appears to represent a separate entity from extramammary fibromatosis, although both lesions may display a similar morphology. The extramammary lesions display a higher propensity for local recurrence compared to mammary fibromatosis.13,14 Mammary fibromatosis accounts for 4% of extra-abdominal fibromatosis cases.15
The etiology of this lesion is still the subject of debate. Trauma, particularly surgical trauma, has been emphasized as an etiological factor. The occurrence of extramammary fibromatosis in younger women, often during pregnancy, the rapid tumor growth in women of childbearing age compared to juvenile and postmenopausal women and men, and spontaneous regression at menopause have suggested the influence of sex steroid hormones in the growth of this tumor.1,48
Since fibromatosis is an infiltrative proliferation of fibroblastic and myofibroblastic cells, the positivity for vimentin and smooth muscle actin was not surprising. Curiously, fibromatosis in the breast differs from fibromatosis arising in other parts of the body due to its hormone receptor profile. Although 30% of extramammary fibromatosis are positive for estrogen receptors, only one of the previously reported cases of mammary fibromatosis expressed hormonal receptors.16,17
Because of the consistent absence of immunoreactivity for estrogen and progesterone receptors in mammary fibromatosis, a positive reaction for these receptors in spindle cell neoplasms of the breast might be helpful in excluding fibromatosis from its differential diagnoses1719. Another major concern about mammary fibromatosis is the exclusion of the diagnosis of metaplastic carcinoma, because spindle cell tumors with a myoepithelial immunophenotype may be diagnosed as metaplastic carcinoma even with weak or absent cytokeratin expression. In the present report, both epithelial (AE1/AE3) and myoepithelial markers (CD14 and p63) were negative.21,22
Historically, wide excision has been the primary surgical modality for fibromatosis and the initial treatment of choice despite high local recurrence. However, due to the rarity of fibromatosis, there are no randomized, controlled surgical trials, and surgical treatment is most often dependent upon the tumor location and size, which influences the potential for a complete surgical resection.15 Other modalities of treatment, such as radiotherapy, hormonal agents, anti-inflammatory agents, and cytotoxic agents may be used in patients with unresectable tumors or tumors that would require extensive resection, including chest wall resection.1423
The reported case demonstrates that fibromatosis in the male breast may have features and a presentation that can be confused with breast cancer. In situations where obvious breast changes and history do not correlate with routine diagnostic measures, the presence of an unusual breast or chest wall tumor such as a fibromatosis should be considered, including this as part of the differential diagnosis.
REFERENCES
1. Brennan MF, Alektiar KM, Maki RG. Sarcomas of the soft tissue. In: DeVita VT, Hellman S, Rosenberg SA, editors. Cancer: Principles and Practice of Oncology. Philadelphia: Lippincott Williams & Wilkins, 2001:1841935.
2. Bombonati A, Parra JS, Schwartz GF, Palazzo JP. Solitary fibrous tumor of the breast. Breast J. 2003;9:251.
3. Shields CJ, Winter DC, Kirwan WO, Redmond HP. Desmoid tumours. Eur J Surg Oncol. 2001;27:7016.
4. Burrell HC, Sibbering DM, Wilson AR. Case report: fibromatosis of the breast in a male patient. Br J Radiol. 1995;68:1128-9.
5. Smarrito S, Salmon R. Desmoid tumor in a male breast in the context of Gardner's syndrome. Case report. Ann Chir. 2005;130:40-3.
6. Magro G, Gurrera A, Scavo N, Lanzafame S, Bisceglia M. Fibromatosis of the breast: a clinical, radiological and pathological study of 6 cases. Pathologica. 2002;94:238-46.
7. Rudan I, Rudan N, Skoric T, Sarcevic B. Fibromatosis of male breast. Acta Med Croatica. 1996;50:157-9.
8. Zanella M, Falconieri G, Della Libera D. The value of fine needle aspiration cytology in breast fibromatosis: study of two new cases and review of the literature. Breast J. 1999;5:264-8.
9. Kouriefs C, Leris ACA, Mokbel K, Wells C, Carpenter R. Infiltrating fibromatosis of the breast: a potential pitfall. Int J Clin Pract .2002;56:4012.
10. Giordano SH, Buzdar AU, Hortobagyi GN. Breast cancer in men. Ann Intern Med. 2002;137:678-87.
11 Caruso G, Ienzi R, Piovana G, Ricotta V, Cirino A, Salvaggio G, et al. High-frequency ultrasound in the study of male breast palpable masses. Radiol Med (Torino). 2004;108:185-93.
12 McKinnon JG, Neifeld JP, Kay S, Parker GA, Foster WC, Lawrence W Jr. Management of desmoid tumors. Surg Gynecol Obstet 1989;169:104-6.
13 Rosen PP, Ernsberger D. Mammary fibromatosis. A benign spindle-cell tumor with significant risk for local recurrence. Cancer. 1989;63:1363-9.
14 Greenberg D, McIntyre H, Ramsaroop R, Arthur J, Harman J. Aggressive fibromatosis of the breast: a case report and literature review. Breast J. 2002;8:55-57.
15 Gronchi A, Casali PG, Mariani L, Lo Vullo S, Colecchia M, Lozza L, et al. Quality of surgery and outcome in extra-abdominal aggressive fibromatosis: a series of patients surgically treated at a single institution. J Clin Oncol. 2003;21:1390-7.
16. Pettinato G, Manivel C, Gould E, Albores-Saavedra J. Inclusion body fibromatosis of the breast: two cases with immunohistochemical and ultrastructural findings. Am J Clin Pathol. 1994;101:714-8.
17 Rasbridge SA, Gillett CE, Millis RR. Oestrogen and progesterone receptor expression in mammary fibromatosis. J Clin Pathol. 1993;46:349-51.
18 Dunne B, Lee AH, Pinder SE, Bell JA, Ellis IO. An immunohistochemical study of metaplastic spindle cell carcinoma, phyllodes tumor and fibromatosis of the breast. Hum Pathol. 2003;34:1009-15.
19 Falconieri G, Lamovec J, Mirra M, Pizzolitto S. Solitary fibrous tumor of the mammary gland: a potential pitfall in breast pathology. Ann Diagn Pathol. 2004;8:121-5.
20 Devouassoux-Shisheboran M, Schammel MD, Man YG, Tavassoli FA. Fibromatosis of the breast: age-correlated morphofunctional features of 33 cases. Arch Pathol Lab Med. 2000;124:276-80.
21 Leibl S, Gogg-Kammerer M, Sommersacher A, Denk H, Moinfar F. Metaplastic breast carcinomas: are they of myoepithelial differentiation?: immunohistochemical profile of the sarcomatoid subtype using novel myoepithelial markers. Am J Surg Pathol. 2005;29:347-53.
22 Reis-Filho JS, Milanezi F, Paredes J, Silva P, Pereira EM, Maeda SA, et al. Novel and classic myoepithelial/stem cell markers in metaplastic carcinomas of the breast. Appl Immunohistochem Mol Morphol. 2003;11:1-8.
23. Nuyttens JJ, Rust PF, Thomas CR, Turrisi AT. Surgery versus radiation therapy for patients with aggressive fibromatosis or desmoid tumors. Cancer. 2000;88:151723.
Publication Dates
-
Publication in this collection
16 Aug 2006 -
Date of issue
Aug 2006