Abstract
OBJECTIVE:
To utilize low-cost and simple methods to assess airway and lung inflammation biomarkers related to air pollution.
METHODS:
A total of 87 male, non-smoking, healthy subjects working as street traffic-controllers or office-workers were examined to determine carbon monoxide in exhaled breath and to measure the pH in nasal lavage fluid and exhaled breath condensate. Air pollution exposure was measured by particulate matter concentration, and data were obtained from fixed monitoring stations (8-h work intervals per day, during the 5 consecutive days prior to the study).
RESULTS:
Exhaled carbon monoxide was two-fold greater in traffic-controllers than in office-workers. The mean pH values were 8.12 in exhaled breath condensate and 7.99 in nasal lavage fluid in office-workers; these values were lower in traffic-controllers (7.80 and 7.30, respectively). Both groups presented similar cytokines concentrations in both substrates, however, IL-1β and IL-8 were elevated in nasal lavage fluid compared with exhaled breath condensate. The particulate matter concentration was greater at the workplace of traffic-controllers compared with that of office-workers.
CONCLUSION:
The pH values of nasal lavage fluid and exhaled breath condensate are important, robust, easy to measure and reproducible biomarkers that can be used to monitor occupational exposure to air pollution. Additionally, traffic-controllers are at an increased risk of airway and lung inflammation during their occupational activities compared with office-workers.
Exhaled Breath Condensate; Nasal Lavage Fluid; Air Pollution; Airway Inflammation
INTRODUCTION
There is epidemiological evidence that air pollution is directly associated with increases in
respiratory symptoms, pulmonary inflammation, infections, emergency room visits and hospital
admissions (11. Johns DO, Stanek LW, Walker K, Benromdhane S, Hubbell B, Ross M, et al. Practical
advancement of multipollutant scientific and risk assessment approaches for ambient air pollution.
Environ Health Perspect. 2012;120(9):1238-42,
http://dx.doi.org/10.1289/ehp.1204939.
http://dx.doi.org/10.1289/ehp.1204939...
2. Ristovski ZD, Miljevic B, Surawski NC, Morawska L, Fong K, Goh F, et al.
Respiratory health effects of diesel particulate matter. Respirology. 2012;17(2):201-2,
http://dx.doi.org/10.1111/j.1440-1843.2011.02109.x.
http://dx.doi.org/10.1111/j.1440-1843.20...
3. Dockery DW, Brunekreef B. Longitudinal studies of air pollution effects on lung
function. Am J Respir Crit Care Med. 1996;154(6):S250-6,
http://dx.doi.org/10.1164/ajrccm/154.6_Pt_2.S250.
http://dx.doi.org/10.1164/ajrccm/154.6_P...
4. Pope CA 3rd, Kanner RE. Acute effects of PM10 pollution on pulmonary function of
smokers with mild to moderate chronic obstructive pulmonary disease. Am Rev Respir Dis.
1993;147(6):1336-40.-55. Olmo NRS, Saldiva PHN, Braga ALF, Lin CA, Santos UP, Pereira LAA. A review of
low-level air pollution and adverse effects on human health: implications for epidemiological
studies and public policy. Clinics. 2011;66(4):681-90,
http://dx.doi.org/10.1590/S1807-59322011000400025.
http://dx.doi.org/10.1590/S1807-59322011...
).
Among the pathophysiological mechanisms of these events, in vitro and in
vivo studies have shown that air pollutants, particularly fine and ultra-fine particles,
induce the release of reactive oxygen species into airways and cause lung inflammation (66. Matsumoto G, Nakagawa NK, Vieira RP, Mauad T, da Silva LF, de André CD, et
al. The time course of vasoconstriction and endothelin receptor A expression in pulmonary arterioles
of mice continuously exposed to ambient urban levels of air pollution. Environ Res.
2010;110(3):237-43, http://dx.doi.org/10.1016/j.envres.2010.01.003.
http://dx.doi.org/10.1016/j.envres.2010....
7. Shukla A, Timblin C, BeruBe K, Gordon T, McKinney W, Driscoll K, et al. Inhaled
particulate matter causes expression of nuclear factor (NF)-kappa B-related genes and
oxidant-dependent NF-kappa B activation in vitro. Am J Respir Cell Mol Biol.
2000;23(2):182-7.-88. Mukae H, English D, Anderson G, Terashima T, Hogg JC, van Eeden SF. Phagocytosis
of PM10 by human alveolar macrophages stimulates the release of PMN from the bone marrow.
Am J Respir Crit Care Med. 1999;159:A317.).
In São Paulo, which is one of the largest cities in the world, seven million vehicles circulate daily, resulting in traffic jam chaos in several areas of the city. Because of the high levels of vehicle-released pollutants in the atmosphere, outdoor activities in these areas may pose an increased health hazard, particularly to the respiratory system. Inflammation in the airways and lungs has an important role in the development and progression of several respiratory diseases.
We examined the air pollution-related airway and lung inflammation in non-smoking, healthy street
traffic-controllers and office-workers by measuring the pH and cytokines concentrations in exhaled
breath condensate (EBC) and nasal lavage fluid (NLF) using low-cost and simple methods (99. Kostikas K, Papatheodorou G, Ganas K, Psathakis K, Panagou P, Loukides S. Ph in
expired breath condensate of patients with inflammatory airway disease. Am J Respir Crit
Care Med. 2002; 165:1364-70, http://dx.doi.org/10.1164/rccm.200111-068OC.
http://dx.doi.org/10.1164/rccm.200111-06...
10. Koczulla R, Dragonieri S, Schot R, Bals R, Gauw SA, Vogelmeier C, et al.
Comparison of exhaled breath condensate pH using two commercially available devices in healthy
controls, asthma and COPD patients. Respir Res. 2009;10:78,
http://dx.doi.org/10.1186/1465-9921-10-78.
http://dx.doi.org/10.1186/1465-9921-10-7...
11. Borrill Z, Starkey C, Vestbo J, Singh D. Reproducibility of exhaled breath
condensate pH in chronic obstructive pulmonary disease. Eur Respir J. 2005;25(2):269-74,
http://dx.doi.org/10.1183/09031936.05.00085804.
http://dx.doi.org/10.1183/09031936.05.00...
12. Maestrelli P, Canova C, Scapellato ML, Visentin A, Tessari R, Bartolucci GB, et
al. Personal exposure to particulate matter is associated with worse health perception in adult
asthma. J Investig Allergol Clin Immunol. 2011;21(2):120-8.
13. Quirce S, Lemière C, de Blay F, del Pozo V, Gerth Van Wijk R, Maestrelli P,
et al. Noninvasive methods for assessment of airway inflammation in occupational settings. Allergy.
2010;65(4):445-58, http://dx.doi.org/10.1111/j.1398-9995.2009.02274.x.
http://dx.doi.org/10.1111/j.1398-9995.20...
14. Barraza-Villarreal A, Sunyer J, Hernandez-Cadena L, Escamilla-Nuñez MC,
Sienra-Monge JJ, Ramírez-Aguilar M, et al. Air pollution, airway inflammation, and lung
function in a cohort study of Mexico City schoolchildren. Environ Health Perspect.
2008;116(6):832-8, http://dx.doi.org/10.1289/ehp.10926.
http://dx.doi.org/10.1289/ehp.10926...
15. Naclerio RM, Meier HL, Kagey-Sobotka A, Adkinson NF Jr, Meyers DA, Norman PS, et
al. Mediator release after nasal airway challenge with allergen. Am Rev Respir Dis.
1983;128:597-602.-1616. Nakagawa NK, Nakao M, Goto DM, Saraiva-Romanholo BM. Air pollution and
non-invasive respiratory assessments. In: Esquinas AM, ed. Applied Technologies in Pulmonary
Medicine. Basel: Karger AG. 2011:223-30.). Both EBC and NLF
contain particles from the upper and the lower airway lining fluid and may be potential sources of
air pollution-related inflammatory biomarkers.
MATERIALS AND METHODS
Study population
This cross-sectional study was approved by the local Ethics Committee of Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo (CAPPesq 0565/07). Non-smoking healthy male subjects aged 18 to 60 years were recruited from a list provided by the Engineering Traffic Company of São Paulo City (street traffic-controllers) and from Faculdade de Medicina da Universidade de São Paulo (office-workers). The subjects were enrolled in the study after providing informed consent. The exclusion criteria were as follows: the use of any chronic medications or the presence of any diagnosed acute or chronic disease. A healthy status was defined after a medical examination of each subject.
Study design
The subjects were assessed only once after one work week (five days of 8-h shifts) at the University Laboratory of Pulmonary Defense on a Saturday morning (from 8 A.M. until noon). The subjects were asked to sit in a chair in a quiet room. Clinical and job history, arterial blood pressure (mmHg), heart rate (bpm), pulse oximetry (%) and respiratory rate (rpm) were registered for each subject after 10 min of rest. The other variables were determined as described below.
Exhaled carbon monoxide measurements
The concentrations of exhaled carbon monoxide (CO) were determined (in ppm) with the aid of a Micro CO analyzer (Cardinal Health U.K., 232 Ltd., Chatham, UK). The subjects were asked to exhale slowly from their total lung capacity with a constant expiratory flow of 5-6 l min−1 over 10 to 15 sec. The mean of two reproducible measurements with a variation of less than 5% was considered.
Exhaled breath condensate
The EBC was obtained as previously described (1010. Koczulla R, Dragonieri S, Schot R, Bals R, Gauw SA, Vogelmeier C, et al.
Comparison of exhaled breath condensate pH using two commercially available devices in healthy
controls, asthma and COPD patients. Respir Res. 2009;10:78,
http://dx.doi.org/10.1186/1465-9921-10-78.
http://dx.doi.org/10.1186/1465-9921-10-7...
). At the
start of EBC collection, all subjects rinsed their mouths with distilled water and were instructed
to swallow saliva as necessary and to hold a slight head extension (approximately 15°). The EBC
sample was collected over 15 min of quiet and normal breathing (regular tidal volumes and
respiratory rate) through a mouthpiece that was connected to a collector device with dry ice
(−20°C). The total EBC (2.0-2.5 ml) was immediately divided and transferred to sterile
500 μl polypropylene tubes. One aliquot was immediately used for pH measurements. The
remaining EBC sample aliquots were coded (for blinding purposes) and stored for a maximum of 4 weeks
at −80°C for the determination of cytokine levels.
Nasal lavage collection
Subjects were asked to tilt their head back at a 45° angle and close the nasopharynx with the soft palate. Room temperature isotonic sodium chloride solution (0.9% NaCl, 5 ml) was instilled into each nostril. After 10 sec, the subject blew their nose forcefully into a sterile plastic container. The average recovery of fluid from NLF was approximately 70%. The lavage fluid was centrifuged (10 min, 300 g, 5°C), and the supernatant was separated from the pellet and divided into five aliquots of 500 μl. One of these supernatant aliquots was immediately used for pH measurements. The remaining aliquots were coded (for blinding purposes) and stored at −80°C for up to 4 weeks to determine the cytokine levels. The cell pellet was used for total and differential cell counts as previously described (1515. Naclerio RM, Meier HL, Kagey-Sobotka A, Adkinson NF Jr, Meyers DA, Norman PS, et al. Mediator release after nasal airway challenge with allergen. Am Rev Respir Dis. 1983;128:597-602.,1616. Nakagawa NK, Nakao M, Goto DM, Saraiva-Romanholo BM. Air pollution and non-invasive respiratory assessments. In: Esquinas AM, ed. Applied Technologies in Pulmonary Medicine. Basel: Karger AG. 2011:223-30.).
Total and differential cell counts in the NLF
The cell pellet was resuspended in 1 ml of phosphate buffer saline solution (PBS). Thereafter, 20 μl of the mixed solution was added to a Neubauer chamber, and the cells were counted using a 400x light microscope (Olympus CH2, Olympus America Inc., Palo Alto, USA). For differential cell proportions (%), 100 μl of the mixed solution was centrifuged (96 g, 25°C, 6 min) to obtain a slide with two areas of cells that were stained with hematoxylin and eosin. Differential cell counts were performed by two different observers with the aid of a 1000x light microscope (Olympus CH2, Olympus America Inc., Palo Alto, USA) (1515. Naclerio RM, Meier HL, Kagey-Sobotka A, Adkinson NF Jr, Meyers DA, Norman PS, et al. Mediator release after nasal airway challenge with allergen. Am Rev Respir Dis. 1983;128:597-602.,1616. Nakagawa NK, Nakao M, Goto DM, Saraiva-Romanholo BM. Air pollution and non-invasive respiratory assessments. In: Esquinas AM, ed. Applied Technologies in Pulmonary Medicine. Basel: Karger AG. 2011:223-30.).
pH measurements in EBC and NLF
In a room maintained at a constant ambient temperature (23°C) and relative humidity (65%),
500 μl of fluid (EBC or NLF) was de-aerated with a gentle 350 ml/min flow of ultrapure (99.9%)
argon gas (Gama Gases Ltd., Sao Paulo, Brazil) for 15 min. The pH was determined with the aid of a
microelectrode and a pH meter (827 pH Lab, Metrohm Ltd., Herisau, Switzerland). The pH meter was
calibrated before each measurement using solutions with pH values of 4, 7 and 9. After pH
determination, the EBC and NLF aliquots were discarded (99. Kostikas K, Papatheodorou G, Ganas K, Psathakis K, Panagou P, Loukides S. Ph in
expired breath condensate of patients with inflammatory airway disease. Am J Respir Crit
Care Med. 2002; 165:1364-70, http://dx.doi.org/10.1164/rccm.200111-068OC.
http://dx.doi.org/10.1164/rccm.200111-06...
10. Koczulla R, Dragonieri S, Schot R, Bals R, Gauw SA, Vogelmeier C, et al.
Comparison of exhaled breath condensate pH using two commercially available devices in healthy
controls, asthma and COPD patients. Respir Res. 2009;10:78,
http://dx.doi.org/10.1186/1465-9921-10-78.
http://dx.doi.org/10.1186/1465-9921-10-7...
-1111. Borrill Z, Starkey C, Vestbo J, Singh D. Reproducibility of exhaled breath
condensate pH in chronic obstructive pulmonary disease. Eur Respir J. 2005;25(2):269-74,
http://dx.doi.org/10.1183/09031936.05.00085804.
http://dx.doi.org/10.1183/09031936.05.00...
).
Measurement of cytokines in EBC and NLF
The concentrations of tumor necrosis factor-alpha (TNF-α), interleukin (IL)-1β, IL-8 and IL-10 in EBC and NLF samples were determined using high sensitivity enzyme-immunoassays (Quantikine HS, R&D Systems Inc., Minneapolis, USA). The limits of detection of the assays were as follows: (a) TNF-α: 0.106 pg/ml, with the standard curve fitted between 0 and 32 pg/ml; (b) IL-1β: 0.057 pg/ml, with the standard curve fitted between 0 and 8 pg/ml; (c) IL-8: 3.50 pg/ml, with the standard curve fitted between 0 and 2,000 pg/ml; and (d) IL-10: 0.50 pg/ml, with the standard curve fitted between 0 and 50 pg/ml.
Air pollutants
The estimation of the exposure of each volunteer (traffic-controller or office-worker) to 10 μm diameter particulate matter (PM10) was obtained during five consecutive days of an 8-h work shift from one of the seven fixed monitoring stations of the São Paulo State Environmental Agency in São Paulo City (Ibirapuera, Pinheiros, Cerqueira Cesar, Centro, Congonhas, Mooca and Parque Dom Pedro). The fixed monitoring stations that were geographically closest to the workplaces of the volunteers were chosen. None of the workplaces was >5 km distant from a fixed monitoring station.
Statistical analysis
Normality distribution was assessed using normal probability plots. The data are expressed as the mean and standard deviation (SD) when normally distributed or the median and interquartile range (IQR) when otherwise appropriate. Comparisons between groups (traffic-controllers and office-workers) were performed using the t-test or Mann-Whitney test. Pearson's correlation or Spearman's rank correlation coefficients were utilized to quantify the degree of association between variables. The estimated effect of traffic-controllers on the pH and the comparison between substrates (EBC and NLF) was determined using linear multiple regression adjusted for age and body mass index (BMI). Interactions between substrate and group and substrate and age were included in the model. The amount of variability in the response variable explained by the model was evaluated by the coefficient of determination, R2. To compare cytokines distributions between groups and between substrates, we created a factor with 4 levels (the combination between groups and substrates), and we used the Kruskal-Wallis test followed by Bonferroni's method (when necessary) to localize the differences. Statistical analyses were carried out using the SPSS statistical package, version 15 (SPSS Inc., Chicago, IL, USA). No correction for the multiplicity of tests was performed; however, p-values are given explicitly wherever reasonable. Statistical significance was set at 5%.
RESULTS
The demographic and clinical characteristics of the 87 adult male subjects who entered into the study (Table 1) showed differences in age and BMI between the street traffic-controllers and office-workers. However, the two occupational groups had similar vital signs (arterial blood pressure, heart rate, pulse oximetry and respiratory rate). Despite being within the normal range, the median levels of exhaled CO were two-fold higher in the street traffic-controllers compared with the office-workers.
pH measurements in EBC and NLF
The correlation between pH in the EBC and in the NLF was r = 0.12 and p = 0.38 in the traffic-controllers and r = −0.31 and p = 0.277 in the office-workers. The EBC pH was not correlated with age (r = 0.07 and p = 0.496); however, it was correlated with BMI (r = 0.22 and p = 0.039). The NLF pH was correlated with both age (r = −0.60 and p<0.001) and BMI (r = −0.27 and p = 0.011).
The pH measurements indicated that the EBC and NLF of traffic-controllers were more acidic than the EBC and NLF of office-workers (Figure 1). In the multiple linear regression model, there was a mean reduction in pH of −0.42 (SE = 0.13, p = 0.001), which was independent of the substrate (EBC or NLF, p = 0.608).
The pH of the exhaled breath condensate and nasal lavage fluid of the traffic-controllers and office-workers.
Cytokine expression in EBC and NLF
The concentrations of cytokines between EBC and NLF were not correlated. No significant differences in IL-10 were observed between the street traffic-controllers and the office-workers and between the substrates (EBC and NLF) (p = 0.455) (Table 2). However, significantly greater concentrations of IL-1 and IL-8 were observed in the NLF of traffic-controllers and office-workers compared with the EBC.
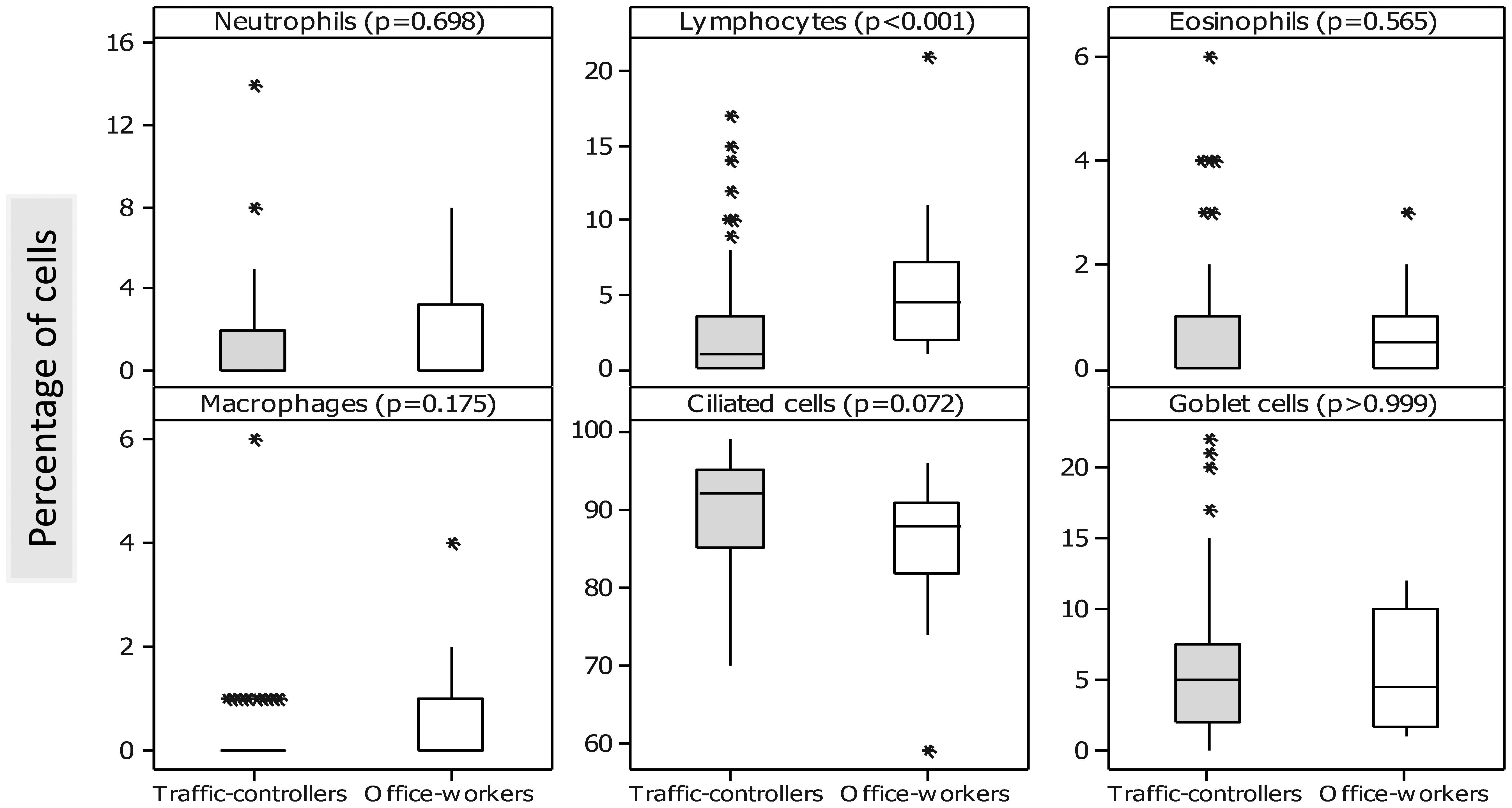
Cellularity of NLF
The number of total cells (median and IQR) in the NLF of the office-workers (126 and 170 cells/mm3) was greater than that of the traffic-controllers (42 and 77 cells/mm3), with an increase in lymphocytes (Figure 2). However, no other significant differences were observed in the NLF of office-workers and traffic-controllers with respect to the percentages of neutrophils, eosinophils, macrophages and epithelial cells (ciliated and goblet cells).
The proportions of cells (%) in the nasal lavage fluid of the traffic-controllers and office-workers. The data were analyzed by the Kruskal-Wallis test.
Environmental exposure data
The outdoor mean PM10 concentrations (SD) in the workplaces of the street traffic-controllers and office-workers were 26.4 (9.45) μg/m3 and 19.7 (1.71) μg/m3, respectively, and were significantly different (p = 0.006). No significant correlations were found between the PM10 concentrations and the EBC pH (r = −0.19 and p = 0.125), the NLF pH (r = −0.21 and p = 0.07), as either with exhaled CO (r = 0.21 and p = 0.876). However, among the office-workers, the correlation coefficient between the NLF pH and PM10 was r = −0.46 (p = 0.154) as a result of the lower statistical power due to the reduced PM10 sample size (n = 11).
DISCUSSION
We conducted a cross-sectional study to assess the effects of two levels of air pollution exposure on human airways: a busy street with considerable traffic and the inside of an office. Because street traffic-controllers perform their occupational activities under direct exposure to outdoor vehicle-related air pollution, they are at increased risk of airway inflammation. Street traffic-controllers presented lower NLF and EBC pH values accompanied by increased amounts of exhaled CO compared with office-workers. However, the concentrations of IL-1β and IL-8 in NLF and EBC were similar in traffic-controllers and office-workers. The workplace of the street traffic-controllers was 25% more polluted than the workplace of the office-workers, although the PM10 concentrations were within the air quality limits advocated by international environmental agencies.
Inflammation is a physiological response to a variety of stimuli comprising a complex series of
events that involve macrophages, monocytes and neutrophils among others and their molecular products
(i.e., cytokines and chemokines). Exposure to air pollution may pose a significant health risk to
some outdoor professionals. The nasal mucosa is the first barrier of the respiratory system that
protects against inhaled pollutants and other agents and plays an important role in the innate
immune response to environmental stimuli (1717. Auger F, Gendron MC, Chamot C, Marano F, Dazy AC. Responses of
well-differentiated nasal epithelial cells exposed to particles: role of the epithelium in airway
inflammation. Toxicol Appl Pharmacol. 2006;215(3):285-94,
http://dx.doi.org/10.1016/j.taap.2006.03.002.
http://dx.doi.org/10.1016/j.taap.2006.03...
). This response
is often studied using nasal biopsies, which themselves induce inflammatory responses. We have used
low-cost and simple methods to study the response of the nasal mucosa and lower airways; these
methods involve the collection of NLF and EBC. In the present study, we obtained sufficient amounts
of sampling fluid, and both were effective sources of inflammatory biomarkers that provided
complementary information. The acidification of the EBC suggests endogenous airway inflammation,
which is implicated in the pathophysiology of several respiratory disorders. All the volunteers in
this study were healthy non-smoking adults with no history of acute or chronic respiratory disease.
The office-workers had a mean EBC pH of 8.12, which is within the normal values reported by others
(1010. Koczulla R, Dragonieri S, Schot R, Bals R, Gauw SA, Vogelmeier C, et al.
Comparison of exhaled breath condensate pH using two commercially available devices in healthy
controls, asthma and COPD patients. Respir Res. 2009;10:78,
http://dx.doi.org/10.1186/1465-9921-10-78.
http://dx.doi.org/10.1186/1465-9921-10-7...
,1818. Vaughan J, Ngamtrakulpanit L, Pajewski TN, Turner R, Nguyen TA, Smith A, et al.
Exhaled breath condensate pH is a robust and reproducible assay of airway acidity. Eur
Respir J. 2003;22(6):889-94, http://dx.doi.org/10.1183/09031936.03.00038803.
http://dx.doi.org/10.1183/09031936.03.00...
19. Wells K, Vaughan J, Pajewski TN, Hom S, Ngamtrakulpanit L, Smith A, et al.
Exhaled breath condensate pH assays are not influenced by oral ammonia. Thorax. 2005;60(1):27-31,
http://dx.doi.org/10.1136/thx.2003.020602.
http://dx.doi.org/10.1136/thx.2003.02060...
20. Cruz MJ, Sanchez-Vidaurre S, Romero PV, Morell F, Munoz X. Impact of age on pH,
8-Isoprostane, and nitrogen oxides in exhaled breath condensate. Chest. 2009;135(2):462-7,
http://dx.doi.org/10.1378/chest.08-1007.
http://dx.doi.org/10.1378/chest.08-1007...
-2121. Koczulla AR, Noeske S, Herr C, Jörres RA, Römmelt H, Vogelmeier C, et
al. Acute and chronic effects of smoking on inflammation markers in exhaled breath condensate in
current smokers. Respiration. 2010;79(1):61-7, http://dx.doi.org/10.1159/000245325.
http://dx.doi.org/10.1159/000245325...
). However, the street traffic-controllers showed significant
reductions in the EBC pH values. The lower pH level was similar to that observed in mechanically
ventilated patients experiencing respiratory failure in the intensive care unit (1818. Vaughan J, Ngamtrakulpanit L, Pajewski TN, Turner R, Nguyen TA, Smith A, et al.
Exhaled breath condensate pH is a robust and reproducible assay of airway acidity. Eur
Respir J. 2003;22(6):889-94, http://dx.doi.org/10.1183/09031936.03.00038803.
http://dx.doi.org/10.1183/09031936.03.00...
).
Experimental and human studies have shown that exposure to air pollutants may generate reactive
oxygen species due to the presence of free radicals and oxidants on the particle surface (2222. Risom L, Moller P, Loft S. Oxidative stress-induced DNA damage by particulate
air pollution. Mutat Res. 2005;592(1-2):119-37,
http://dx.doi.org/10.1016/j.mrfmmm.2005.06.012.
http://dx.doi.org/10.1016/j.mrfmmm.2005....
) and may cause alterations in the transcription of inflammatory
cytokines (77. Shukla A, Timblin C, BeruBe K, Gordon T, McKinney W, Driscoll K, et al. Inhaled
particulate matter causes expression of nuclear factor (NF)-kappa B-related genes and
oxidant-dependent NF-kappa B activation in vitro. Am J Respir Cell Mol Biol.
2000;23(2):182-7.). Because cytokines may play important roles in
the pathophysiology of airway and lung parenchyma diseases, interest has been focused on cytokines
determinations (2323. Barnes PJ. The cytokine network in asthma and chronic obstructive pulmonary
disease. J Clin Invest. 2008;118(11):3546-56,
http://dx.doi.org/10.1172/JCI36130.
http://dx.doi.org/10.1172/JCI36130...
). IL-1β plays an important
protective role against pathogen invasion because it is a potent inflammatory mediator that
stimulates chemokine production and leukocyte recruitment to the site of injury. Additionally,
IL-1β is involved in a variety of cellular activities including cell proliferation,
differentiation and apoptosis (2424. Dinarello CA. Immunological and inflammatory functions of the interleukin-1
family. Annu Rev Immunol. 2009;27:519-50,
http://dx.doi.org/10.1146/annurev.immunol.021908.132612.
http://dx.doi.org/10.1146/annurev.immuno...
). IL-8 is responsible for
the recruitment of inflammatory cells from the circulation to the airways in many respiratory
diseases (2525. Riechelmann H, Deutschle T, Friemel E, Gross H-J, Bachem M. Biological markers
in nasal secretions. Eur Respir J. 2003;21(4):600-605,
http://dx.doi.org/10.1183/09031936.03.00072003.
http://dx.doi.org/10.1183/09031936.03.00...
,2626. Cyktor JC, Turner J. IL-10 and immunity against prokaryotic and eukaryotic
intracellular pathogens. Infect Immun. 2011;79(8):2964-73,
http://dx.doi.org/10.1128/IAI.00047-11.
http://dx.doi.org/10.1128/IAI.00047-11...
). Few
studies on health effects related to air pollution have explored cytokines levels in EBC, primarily
because low concentrations of cytokines have been reported (2727. Garey KW, Neuhauser MM, Robbins RA, Danziger LH, Rubinstein I. Markers of
inflammation in exhaled breath condensate of young healthy smokers. Chest. 2004;125(1):22-26,
http://dx.doi.org/10.1378/chest.125.1.22.
http://dx.doi.org/10.1378/chest.125.1.22...
,2828. Jungraithmayr W, Frings C, Zissel G, Prasse A, Passlick B, Stoelben E.
Inflammatory markers in exhaled breath condensate following lung resection for bronchial carcinoma.
Respirology. 2008;13(7):1022-7.). Indeed, in the present study, the IL-1β
concentrations in EBC were between 0 and 1 pg/ml, similar to the findings of other reports in
healthy subjects (2727. Garey KW, Neuhauser MM, Robbins RA, Danziger LH, Rubinstein I. Markers of
inflammation in exhaled breath condensate of young healthy smokers. Chest. 2004;125(1):22-26,
http://dx.doi.org/10.1378/chest.125.1.22.
http://dx.doi.org/10.1378/chest.125.1.22...
,2929. Gessner C, Scheibe R, Wötzel M, Hammerschmidt S, Kuhn H, Engelmann L, et
al. Exhaled breath condensate cytokine patterns in chronic obstructive pulmonary disease. Respir
Med. 2005;99(10):1229-40, http://dx.doi.org/10.1016/j.rmed.2005.02.041.
http://dx.doi.org/10.1016/j.rmed.2005.02...
). In this study, the IL-8 concentrations in EBC were similar in the office-workers and
the street traffic-controllers (∼10 pg/ml); however, these concentrations were four- to
nine-fold higher than previous findings in healthy individuals (1515. Naclerio RM, Meier HL, Kagey-Sobotka A, Adkinson NF Jr, Meyers DA, Norman PS, et
al. Mediator release after nasal airway challenge with allergen. Am Rev Respir Dis.
1983;128:597-602.,3030. Gessner C, Kuhn H, Toepfer K, Hammerschmidt S, Schauer J, Wirtz H. Detection of
p53 gene mutations in exhaled breath condensate of non-small cell lung cancer patients. Lung Cancer.
2004;43(2):215-22, http://dx.doi.org/10.1016/j.lungcan.2003.08.034.
http://dx.doi.org/10.1016/j.lungcan.2003...
,3131. Carpagnano GE, Resta O, De Pergola G, Sabato R, Foschino-Barbaro MP. The role of
obstructive sleep apnea syndrome and obesity in determining leptin in the exhaled breath condensate.
J Breath Res. 2010;4(3):036003,
http://dx.doi.org/10.1088/1752-7155/4/3/036003.
http://dx.doi.org/10.1088/1752-7155/4/3/...
) and
similar to those observed young smokers (3030. Gessner C, Kuhn H, Toepfer K, Hammerschmidt S, Schauer J, Wirtz H. Detection of
p53 gene mutations in exhaled breath condensate of non-small cell lung cancer patients. Lung Cancer.
2004;43(2):215-22, http://dx.doi.org/10.1016/j.lungcan.2003.08.034.
http://dx.doi.org/10.1016/j.lungcan.2003...
,3232. Carpagnano GE, Kharitonov SA, Resta O, Foschino-Barbaro MP, Gramiccioni E,
Barnes PJ. Increased 8-isoprostane and interleukin-6 in breath condensate of obstructive sleep apnea
patients. Chest. 2002;122(4):1162-7, http://dx.doi.org/10.1378/chest.122.4.1162.
http://dx.doi.org/10.1378/chest.122.4.11...
). We can not exclude the possibility that these differences can
be explained by the use of different standards in the assays.
In NLF, we found an increased number of total cells in office-workers, which was primarily due to
an increased number of lymphocytes. In contrast, traffic-workers presented a similar trend in the
number of ciliated cells, although statistical significance was not achieved. We also found higher
concentrations of IL-1β and IL-8 in NLF compared with EBC (eight-fold and three-fold,
respectively). Data related to IL-1β levels in NLF are very scarce in the literature.
Riechelmann et al. (2525. Riechelmann H, Deutschle T, Friemel E, Gross H-J, Bachem M. Biological markers
in nasal secretions. Eur Respir J. 2003;21(4):600-605,
http://dx.doi.org/10.1183/09031936.03.00072003.
http://dx.doi.org/10.1183/09031936.03.00...
) reported IL-1β concentrations
of 15±13 pg/ml in healthy non-smoking volunteers (aged 18-60 years); in the current study, we
observed IL-1β concentrations that were 50% lower. However, the IL-8 concentrations in the NLF
samples in the present study were ten-fold greater than those in volunteers with occupational
rhinitis (mean IL-8 concentrations of ∼27 pg/ml) (3333. Castano R, Maghni K, Castellanos L, Trudeau C, Malo J-L, Gaudrin D.
Proinflammatory mediators in nasal lavage of subjects with occupational rhinitis. Otolaryngol Head
Neck Surg. 2010;143(2):301-3, http://dx.doi.org/10.1016/j.otohns.2010.04.272.
http://dx.doi.org/10.1016/j.otohns.2010....
).
In agreement with this finding, other reports have shown direct associations between exposure to
PM2,5 and nasal inflammation (3434. Chen BY, Chan CC, Lee CT, Cheng TJ, Huang WC, Jhou JC, et al. The association of
ambient air pollution with airway inflammation in schoolchildren. Am J Epidemiol.
2012;175(8):764-74.) as well as
increases in IL-8 production in the airways of healthy individuals after acute exposure to diesel
exhaust (3535. Salvi SS, Nordenhall C, Blomberg A, Rudell B, Pourazar J, Kelly FJ, et al. Acute
exposure to diesel exhaust increases interleukin-8 and GRO-alpha production in healthy human
airways. Am J Respir Crit Care Med. 2000;161(2 Pt 1):550-7,
http://dx.doi.org/10.1164/ajrccm.161.2.9905052.
http://dx.doi.org/10.1164/ajrccm.161.2.9...
). In the present study, the levels of IL-1β
and IL-8 were increased in NLF compared with EBC in the traffic-controllers and office-workers, and
these increases were not accompanied by changes in the percentage of neutrophils. This finding
raised the possibility that the increase in these pro-inflammatory mediators (IL-1β and IL-8)
may have resulted from epithelial cell production, as reported in in vitro studies
(3636. Sakamoto N, Hayashi S, Gosselink J, Ishii H, Ishimatsu Y, Mukae H, et al.
Calcium dependent and independent cytokine synthesis by air pollution particle-exposed human
bronchial epithelial cells. Toxicol Appl Pharmacol. 2007;225(2):134-41,
http://dx.doi.org/10.1016/j.taap.2007.07.006.
http://dx.doi.org/10.1016/j.taap.2007.07...
) with human bronchial epithelial cells exposed to
PM10 and in studies of the nasal epithelial cells of asthmatic children (3737. Pringle EJ, Richardson HB, Miller D, Cornish DS, Devereux GS, Walsh GM, et al.
Nasal and bronchial airway epithelial cell mediator release in children. Ped Pulmonol.
2012;47(12):1215-25, http://dx.doi.org/10.1002/ppul.22672.
http://dx.doi.org/10.1002/ppul.22672...
).
Human activity patterns and microenvironmental exposure can significantly affect exhaled CO
levels; however, it has been suggested that regular monitoring of exhaled CO levels in healthy
subjects has the potential to be used as a functional index of air pollution (3838. Jones AY, Lam PK. End-expiratory carbon monoxide levels in healthy subjects
living in a densely populated urban environment. Sci Total Environ. 2006;354(2-3):150-6,
http://dx.doi.org/10.1016/j.scitotenv.2005.02.018.
http://dx.doi.org/10.1016/j.scitotenv.20...
). We observed higher exhaled CO levels in traffic-controllers (two-fold) than in
office-workers, although both levels were within the normal values for non-smokers (<10 ppm,
according to the ATS guidelines).
This study has limitations. There were some differences in demographic characteristics between
our study groups. For example, the street traffic-controllers were older than the office-workers.
However, previous studies have shown that aging has no effect on the pH of EBC (1818. Vaughan J, Ngamtrakulpanit L, Pajewski TN, Turner R, Nguyen TA, Smith A, et al.
Exhaled breath condensate pH is a robust and reproducible assay of airway acidity. Eur
Respir J. 2003;22(6):889-94, http://dx.doi.org/10.1183/09031936.03.00038803.
http://dx.doi.org/10.1183/09031936.03.00...
), with the exception of individuals between 60 and 80 years old
(2020. Cruz MJ, Sanchez-Vidaurre S, Romero PV, Morell F, Munoz X. Impact of age on pH,
8-Isoprostane, and nitrogen oxides in exhaled breath condensate. Chest. 2009;135(2):462-7,
http://dx.doi.org/10.1378/chest.08-1007.
http://dx.doi.org/10.1378/chest.08-1007...
), which was not the age range of our study population.
Additionally, the BMI of the traffic-controllers was higher than that of the office-workers. Obesity
has been reported to be associated with systemic inflammation, particularly if coupled with
sedentary behavior (3939. Handschin C, Spiegelman BM. The role of exercise and PGC1 in inflammation and
chronic disease. Nature. 2008;454(7200):24-5.). However, the difference in BMI
between the two groups was only approximately 10; thus, it may not have affected our results. In the
present study, the outdoor air pollution data must be interpreted with caution. We used the
PM10 concentration data provided by fixed monitoring stations that were located near the
workplace of each street traffic-controller and office-worker (mean values of 8-hr intervals over 5
consecutive work days). We detected a 25% difference in exposure between the traffic-controllers and
the office-workers. However, this difference may still underestimate the PM10 exposure
among traffic-controllers because considerably higher levels of pollutants are present at the
location where the traffic-controllers work compared with the city average, as we have recently
demonstrated in vehicle corridors with high traffic (4040. Carneiro MFH, Ribeiro FQ, Fernandes-Filho FN, et al. Pollen abortion rates,
nitrogen dioxide by passive diffusive tubes and bioaccumulation in tree barks are effective in the
characterization of air pollution. Environ Exp Botany. 2011;72(2):272-7,
http://dx.doi.org/10.1016/j.envexpbot.2011.04.001.
http://dx.doi.org/10.1016/j.envexpbot.20...
). In
addition, the PM10 results likely do not reflect the real difference in personal exposure
(4141. Baxter LK, Wright RJ, Paciorek C, Laden F, Suh HH, Levy JI. Effects of exposure
measurements error in the analysis of health effects from traffic-related air pollution. J Expo
Sci Environ Epidemiol. 2010;20(1):101-11, http://dx.doi.org/10.1038/jes.2009.5.
http://dx.doi.org/10.1038/jes.2009.5...
). However, unpublished data from our laboratory show that
the mean PM10 concentration measured by fixed monitoring stations is 50% lower than the
personal exposure. Another issue is that the personal exposure of office-workers may be lower than
the value reported by the fixed monitoring stations because these individuals work indoors. Several
reports indicate that exposure to urban traffic is markedly attenuated in the indoor environment
(4141. Baxter LK, Wright RJ, Paciorek C, Laden F, Suh HH, Levy JI. Effects of exposure
measurements error in the analysis of health effects from traffic-related air pollution. J Expo
Sci Environ Epidemiol. 2010;20(1):101-11, http://dx.doi.org/10.1038/jes.2009.5.
http://dx.doi.org/10.1038/jes.2009.5...
). Additionally, the present study was performed during a
period of school vacation when there were 30-40% fewer vehicles circulating in the city. However, we
found that the pH in NLF and EBC was an efficient biomarker that can be used to assess the
inflammatory effects of air pollution on the airways and lungs. Inflammatory disturbances were
markedly present in the nasal cavities of both groups. However, only traffic-controllers showed
extended inflammation in the airways based on acidification of their EBC.
In conclusion, the pH values of NLF and EBC are important, robust, easy-to-measure and reproducible biomarkers that can be used to monitor occupational exposure to air pollution. Additionally, traffic-controllers are at an increased risk of airway and lung inflammation during their occupational activities compared with office-workers.
The authors would like to thank Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP 07/51605-9 and 09/50056-7) and the National Council of Technological and Scientific Development (CNPq 555.223/06-0) for providing financial support for this study. We also thank Carolina Tieko Yoshida for revising tables, figures and references.
REFERENCES
-
1Johns DO, Stanek LW, Walker K, Benromdhane S, Hubbell B, Ross M, et al. Practical advancement of multipollutant scientific and risk assessment approaches for ambient air pollution. Environ Health Perspect. 2012;120(9):1238-42, http://dx.doi.org/10.1289/ehp.1204939.
» http://dx.doi.org/10.1289/ehp.1204939 -
2Ristovski ZD, Miljevic B, Surawski NC, Morawska L, Fong K, Goh F, et al. Respiratory health effects of diesel particulate matter. Respirology. 2012;17(2):201-2, http://dx.doi.org/10.1111/j.1440-1843.2011.02109.x.
» http://dx.doi.org/10.1111/j.1440-1843.2011.02109.x -
3Dockery DW, Brunekreef B. Longitudinal studies of air pollution effects on lung function. Am J Respir Crit Care Med. 1996;154(6):S250-6, http://dx.doi.org/10.1164/ajrccm/154.6_Pt_2.S250.
» http://dx.doi.org/10.1164/ajrccm/154.6_Pt_2.S250 -
4Pope CA 3rd, Kanner RE. Acute effects of PM10 pollution on pulmonary function of smokers with mild to moderate chronic obstructive pulmonary disease. Am Rev Respir Dis. 1993;147(6):1336-40.
-
5Olmo NRS, Saldiva PHN, Braga ALF, Lin CA, Santos UP, Pereira LAA. A review of low-level air pollution and adverse effects on human health: implications for epidemiological studies and public policy. Clinics. 2011;66(4):681-90, http://dx.doi.org/10.1590/S1807-59322011000400025.
» http://dx.doi.org/10.1590/S1807-59322011000400025 -
6Matsumoto G, Nakagawa NK, Vieira RP, Mauad T, da Silva LF, de André CD, et al. The time course of vasoconstriction and endothelin receptor A expression in pulmonary arterioles of mice continuously exposed to ambient urban levels of air pollution. Environ Res. 2010;110(3):237-43, http://dx.doi.org/10.1016/j.envres.2010.01.003.
» http://dx.doi.org/10.1016/j.envres.2010.01.003 -
7Shukla A, Timblin C, BeruBe K, Gordon T, McKinney W, Driscoll K, et al. Inhaled particulate matter causes expression of nuclear factor (NF)-kappa B-related genes and oxidant-dependent NF-kappa B activation in vitro. Am J Respir Cell Mol Biol. 2000;23(2):182-7.
-
8Mukae H, English D, Anderson G, Terashima T, Hogg JC, van Eeden SF. Phagocytosis of PM10 by human alveolar macrophages stimulates the release of PMN from the bone marrow. Am J Respir Crit Care Med. 1999;159:A317.
-
9Kostikas K, Papatheodorou G, Ganas K, Psathakis K, Panagou P, Loukides S. Ph in expired breath condensate of patients with inflammatory airway disease. Am J Respir Crit Care Med. 2002; 165:1364-70, http://dx.doi.org/10.1164/rccm.200111-068OC.
» http://dx.doi.org/10.1164/rccm.200111-068OC -
10Koczulla R, Dragonieri S, Schot R, Bals R, Gauw SA, Vogelmeier C, et al. Comparison of exhaled breath condensate pH using two commercially available devices in healthy controls, asthma and COPD patients. Respir Res. 2009;10:78, http://dx.doi.org/10.1186/1465-9921-10-78.
» http://dx.doi.org/10.1186/1465-9921-10-78 -
11Borrill Z, Starkey C, Vestbo J, Singh D. Reproducibility of exhaled breath condensate pH in chronic obstructive pulmonary disease. Eur Respir J. 2005;25(2):269-74, http://dx.doi.org/10.1183/09031936.05.00085804.
» http://dx.doi.org/10.1183/09031936.05.00085804 -
12Maestrelli P, Canova C, Scapellato ML, Visentin A, Tessari R, Bartolucci GB, et al. Personal exposure to particulate matter is associated with worse health perception in adult asthma. J Investig Allergol Clin Immunol. 2011;21(2):120-8.
-
13Quirce S, Lemière C, de Blay F, del Pozo V, Gerth Van Wijk R, Maestrelli P, et al. Noninvasive methods for assessment of airway inflammation in occupational settings. Allergy. 2010;65(4):445-58, http://dx.doi.org/10.1111/j.1398-9995.2009.02274.x.
» http://dx.doi.org/10.1111/j.1398-9995.2009.02274.x -
14Barraza-Villarreal A, Sunyer J, Hernandez-Cadena L, Escamilla-Nuñez MC, Sienra-Monge JJ, Ramírez-Aguilar M, et al. Air pollution, airway inflammation, and lung function in a cohort study of Mexico City schoolchildren. Environ Health Perspect. 2008;116(6):832-8, http://dx.doi.org/10.1289/ehp.10926.
» http://dx.doi.org/10.1289/ehp.10926 -
15Naclerio RM, Meier HL, Kagey-Sobotka A, Adkinson NF Jr, Meyers DA, Norman PS, et al. Mediator release after nasal airway challenge with allergen. Am Rev Respir Dis. 1983;128:597-602.
-
16Nakagawa NK, Nakao M, Goto DM, Saraiva-Romanholo BM. Air pollution and non-invasive respiratory assessments. In: Esquinas AM, ed. Applied Technologies in Pulmonary Medicine. Basel: Karger AG. 2011:223-30.
-
17Auger F, Gendron MC, Chamot C, Marano F, Dazy AC. Responses of well-differentiated nasal epithelial cells exposed to particles: role of the epithelium in airway inflammation. Toxicol Appl Pharmacol. 2006;215(3):285-94, http://dx.doi.org/10.1016/j.taap.2006.03.002.
» http://dx.doi.org/10.1016/j.taap.2006.03.002 -
18Vaughan J, Ngamtrakulpanit L, Pajewski TN, Turner R, Nguyen TA, Smith A, et al. Exhaled breath condensate pH is a robust and reproducible assay of airway acidity. Eur Respir J. 2003;22(6):889-94, http://dx.doi.org/10.1183/09031936.03.00038803.
» http://dx.doi.org/10.1183/09031936.03.00038803 -
19Wells K, Vaughan J, Pajewski TN, Hom S, Ngamtrakulpanit L, Smith A, et al. Exhaled breath condensate pH assays are not influenced by oral ammonia. Thorax. 2005;60(1):27-31, http://dx.doi.org/10.1136/thx.2003.020602.
» http://dx.doi.org/10.1136/thx.2003.020602 -
20Cruz MJ, Sanchez-Vidaurre S, Romero PV, Morell F, Munoz X. Impact of age on pH, 8-Isoprostane, and nitrogen oxides in exhaled breath condensate. Chest. 2009;135(2):462-7, http://dx.doi.org/10.1378/chest.08-1007.
» http://dx.doi.org/10.1378/chest.08-1007 -
21Koczulla AR, Noeske S, Herr C, Jörres RA, Römmelt H, Vogelmeier C, et al. Acute and chronic effects of smoking on inflammation markers in exhaled breath condensate in current smokers. Respiration. 2010;79(1):61-7, http://dx.doi.org/10.1159/000245325.
» http://dx.doi.org/10.1159/000245325 -
22Risom L, Moller P, Loft S. Oxidative stress-induced DNA damage by particulate air pollution. Mutat Res. 2005;592(1-2):119-37, http://dx.doi.org/10.1016/j.mrfmmm.2005.06.012.
» http://dx.doi.org/10.1016/j.mrfmmm.2005.06.012 -
23Barnes PJ. The cytokine network in asthma and chronic obstructive pulmonary disease. J Clin Invest. 2008;118(11):3546-56, http://dx.doi.org/10.1172/JCI36130.
» http://dx.doi.org/10.1172/JCI36130 -
24Dinarello CA. Immunological and inflammatory functions of the interleukin-1 family. Annu Rev Immunol. 2009;27:519-50, http://dx.doi.org/10.1146/annurev.immunol.021908.132612.
» http://dx.doi.org/10.1146/annurev.immunol.021908.132612 -
25Riechelmann H, Deutschle T, Friemel E, Gross H-J, Bachem M. Biological markers in nasal secretions. Eur Respir J. 2003;21(4):600-605, http://dx.doi.org/10.1183/09031936.03.00072003.
» http://dx.doi.org/10.1183/09031936.03.00072003 -
26Cyktor JC, Turner J. IL-10 and immunity against prokaryotic and eukaryotic intracellular pathogens. Infect Immun. 2011;79(8):2964-73, http://dx.doi.org/10.1128/IAI.00047-11.
» http://dx.doi.org/10.1128/IAI.00047-11 -
27Garey KW, Neuhauser MM, Robbins RA, Danziger LH, Rubinstein I. Markers of inflammation in exhaled breath condensate of young healthy smokers. Chest. 2004;125(1):22-26, http://dx.doi.org/10.1378/chest.125.1.22.
» http://dx.doi.org/10.1378/chest.125.1.22 -
28Jungraithmayr W, Frings C, Zissel G, Prasse A, Passlick B, Stoelben E. Inflammatory markers in exhaled breath condensate following lung resection for bronchial carcinoma. Respirology. 2008;13(7):1022-7.
-
29Gessner C, Scheibe R, Wötzel M, Hammerschmidt S, Kuhn H, Engelmann L, et al. Exhaled breath condensate cytokine patterns in chronic obstructive pulmonary disease. Respir Med. 2005;99(10):1229-40, http://dx.doi.org/10.1016/j.rmed.2005.02.041.
» http://dx.doi.org/10.1016/j.rmed.2005.02.041 -
30Gessner C, Kuhn H, Toepfer K, Hammerschmidt S, Schauer J, Wirtz H. Detection of p53 gene mutations in exhaled breath condensate of non-small cell lung cancer patients. Lung Cancer. 2004;43(2):215-22, http://dx.doi.org/10.1016/j.lungcan.2003.08.034.
» http://dx.doi.org/10.1016/j.lungcan.2003.08.034 -
31Carpagnano GE, Resta O, De Pergola G, Sabato R, Foschino-Barbaro MP. The role of obstructive sleep apnea syndrome and obesity in determining leptin in the exhaled breath condensate. J Breath Res. 2010;4(3):036003, http://dx.doi.org/10.1088/1752-7155/4/3/036003.
» http://dx.doi.org/10.1088/1752-7155/4/3/036003 -
32Carpagnano GE, Kharitonov SA, Resta O, Foschino-Barbaro MP, Gramiccioni E, Barnes PJ. Increased 8-isoprostane and interleukin-6 in breath condensate of obstructive sleep apnea patients. Chest. 2002;122(4):1162-7, http://dx.doi.org/10.1378/chest.122.4.1162.
» http://dx.doi.org/10.1378/chest.122.4.1162 -
33Castano R, Maghni K, Castellanos L, Trudeau C, Malo J-L, Gaudrin D. Proinflammatory mediators in nasal lavage of subjects with occupational rhinitis. Otolaryngol Head Neck Surg. 2010;143(2):301-3, http://dx.doi.org/10.1016/j.otohns.2010.04.272.
» http://dx.doi.org/10.1016/j.otohns.2010.04.272 -
34Chen BY, Chan CC, Lee CT, Cheng TJ, Huang WC, Jhou JC, et al. The association of ambient air pollution with airway inflammation in schoolchildren. Am J Epidemiol. 2012;175(8):764-74.
-
35Salvi SS, Nordenhall C, Blomberg A, Rudell B, Pourazar J, Kelly FJ, et al. Acute exposure to diesel exhaust increases interleukin-8 and GRO-alpha production in healthy human airways. Am J Respir Crit Care Med. 2000;161(2 Pt 1):550-7, http://dx.doi.org/10.1164/ajrccm.161.2.9905052.
» http://dx.doi.org/10.1164/ajrccm.161.2.9905052 -
36Sakamoto N, Hayashi S, Gosselink J, Ishii H, Ishimatsu Y, Mukae H, et al. Calcium dependent and independent cytokine synthesis by air pollution particle-exposed human bronchial epithelial cells. Toxicol Appl Pharmacol. 2007;225(2):134-41, http://dx.doi.org/10.1016/j.taap.2007.07.006.
» http://dx.doi.org/10.1016/j.taap.2007.07.006 -
37Pringle EJ, Richardson HB, Miller D, Cornish DS, Devereux GS, Walsh GM, et al. Nasal and bronchial airway epithelial cell mediator release in children. Ped Pulmonol. 2012;47(12):1215-25, http://dx.doi.org/10.1002/ppul.22672.
» http://dx.doi.org/10.1002/ppul.22672 -
38Jones AY, Lam PK. End-expiratory carbon monoxide levels in healthy subjects living in a densely populated urban environment. Sci Total Environ. 2006;354(2-3):150-6, http://dx.doi.org/10.1016/j.scitotenv.2005.02.018.
» http://dx.doi.org/10.1016/j.scitotenv.2005.02.018 -
39Handschin C, Spiegelman BM. The role of exercise and PGC1 in inflammation and chronic disease. Nature. 2008;454(7200):24-5.
-
40Carneiro MFH, Ribeiro FQ, Fernandes-Filho FN, et al. Pollen abortion rates, nitrogen dioxide by passive diffusive tubes and bioaccumulation in tree barks are effective in the characterization of air pollution. Environ Exp Botany. 2011;72(2):272-7, http://dx.doi.org/10.1016/j.envexpbot.2011.04.001.
» http://dx.doi.org/10.1016/j.envexpbot.2011.04.001 -
41Baxter LK, Wright RJ, Paciorek C, Laden F, Suh HH, Levy JI. Effects of exposure measurements error in the analysis of health effects from traffic-related air pollution. J Expo Sci Environ Epidemiol. 2010;20(1):101-11, http://dx.doi.org/10.1038/jes.2009.5.
» http://dx.doi.org/10.1038/jes.2009.5
-
No potential conflict of interest was reported.
Publication Dates
-
Publication in this collection
Dec 2013
History
-
Received
28 Mar 2013 -
Reviewed
14 May 2013 -
Accepted
8 June 2013