Abstracts
No previous studies have investigated the use of graphite instead of activated charcoal in orchids. In this work, different concentrations of activated charcoal or graphite were added to KC medium to darken the culture medium and stimulate the in vitro propagation of Cattleya bicolor and of a double hybrid orchid ('BLC Pastoral Innocence'). The seedlings were inoculated on growth regulator-free KC medium; the effects of activated charcoal (0.0, 1.5, 3.0, 4.5, 6.0, and 7.5 g L-1) and graphite (0.0, 1.5, 3.0, 4.5, 6.0, and 7.5 g L-1) concentrations were tested separately. Each flask contained fifteen seedlings, and the experiments had a randomised, two-way factorial design. The two tested variables were culture medium and illumination levels; eleven culture levels (different concentrations of graphite or activate charcoal) and two illumination levels (14-hr photoperiod or continuous illumination) were evaluated. Three replicates of each combination were utilised. After six months of culture, the average numbers of induced buds and roots per seedling were recorded for each concentration of activated charcoal or graphite. Whereas the largest number of buds in C. bicolor seedlings occurred on media containing 6.0 or 7.5 g L-1 graphite, the largest number of roots occurred on media containing 6.0 g L-1 activated charcoal. In the hybrid 'BLC Pastoral Innocence', the largest number of buds and roots was reported in medium with 4.5 g L-1 activated charcoal. When using graphite in place of activated charcoal, we obtained disparate the results in root formation that suggest that graphite is not a recommended substitute for activated charcoal.
activated charcoal; graphite; orchids; micropropagation
Nenhum estudo prévio tem investigado o uso de grafite como substituto do carvão ativado em orquídeas. No presente trabalho, concentrações diferentes de carvão ativado ou de grafite foram adicionadas ao meio KC para escurecer o meio de cultura e estimular a propagação in vitro de Cattleya bicolor e de um duplo híbrido de orquídea ('BLC Pastoral Innocence'). As plântulas foram inoculadas em meio KC sem reguladores de crescimento; os efeitos de concentrações de carvão ativado (0,0; 1,5; 3,0; 4,5; 6,0 and 7,5 g L-1) e de grafite (0,0; 1,5; 3,0; 4,5; 6,0 and 7,5 g L-1) foram testados separadamente. Foram inoculadas quinze plântulas em cada frasco e os experimentos foram conduzidos sob esquema de planejamento fatorial com dois fatores: meios de cultura e luminosidade; o meio de cultura com onze níveis (diferentes concentrações de grafite ou de carvão ativado) e luminosidade com dois níveis (fotoperíodo de 14h e iluminação contínua). Para cada combinação do nível fatorial foram feitas três repetições. Após seis meses de cultivo, foram registrados os números médios de brotos e raízes induzidos em cada concentração de carvão ativado e de grafite. O maior número de brotos nas plântulas de C. bicolor foi observado no meio contendo 6,0 e 7,5 g L-1 de grafite, e o maior número de raízes foi verificado no meio contendo 6,0 g L-1 de carvão ativado. No duplo híbrido o maior número de brotos e raízes foi observado no meio contendo 4,5 g L-1 de carvão ativado. O grafite não pode ser recomendado para substituir o carvão ativado.
carvão ativado; grafite; orquídeas; micropropagação
CROP PRODUCTION
Activated charcoal and graphite for the micropropagation of Cattleya bicolor Lindl. and a orchid double-hybrid 'BLC Pastoral Innocence'
Carvão ativado e grafite para a micropropagação de Cattleya bicolor Lindl. e um duplo híbrido de orquídea 'BLC Pastoral Innocence'
Eliane Cristina PrizãoI; Letícia de Menezes GonçalvesI; Maria Auxiliadora Milaneze GutierreII; Claudete Aparecida MangolinI; Maria de Fátima Pires da Silva MachadoI, * * Author for correspondence. E-mail: mfpsmachado@uem.br
IDepartamento de Biologia Celular e Genética, Universidade Estadual de Maringá, Av. Colombo 5790, 87020-900, Maringá, Paraná, Brazil
IIDepartamento de Biologia, Universidade Estadual de Maringá, Maringá, Paraná, Brazil
ABSTRACT
No previous studies have investigated the use of graphite instead of activated charcoal in orchids. In this work, different concentrations of activated charcoal or graphite were added to KC medium to darken the culture medium and stimulate the in vitro propagation of Cattleya bicolor and of a double hybrid orchid ('BLC Pastoral Innocence'). The seedlings were inoculated on growth regulator-free KC medium; the effects of activated charcoal (0.0, 1.5, 3.0, 4.5, 6.0, and 7.5 g L-1) and graphite (0.0, 1.5, 3.0, 4.5, 6.0, and 7.5 g L-1) concentrations were tested separately. Each flask contained fifteen seedlings, and the experiments had a randomised, two-way factorial design. The two tested variables were culture medium and illumination levels; eleven culture levels (different concentrations of graphite or activate charcoal) and two illumination levels (14-hr photoperiod or continuous illumination) were evaluated. Three replicates of each combination were utilised. After six months of culture, the average numbers of induced buds and roots per seedling were recorded for each concentration of activated charcoal or graphite. Whereas the largest number of buds in C. bicolor seedlings occurred on media containing 6.0 or 7.5 g L-1 graphite, the largest number of roots occurred on media containing 6.0 g L-1 activated charcoal. In the hybrid 'BLC Pastoral Innocence', the largest number of buds and roots was reported in medium with 4.5 g L-1 activated charcoal. When using graphite in place of activated charcoal, we obtained disparate the results in root formation that suggest that graphite is not a recommended substitute for activated charcoal.
Keywords: activated charcoal, graphite, orchids, micropropagation.
RESUMO
Nenhum estudo prévio tem investigado o uso de grafite como substituto do carvão ativado em orquídeas. No presente trabalho, concentrações diferentes de carvão ativado ou de grafite foram adicionadas ao meio KC para escurecer o meio de cultura e estimular a propagação in vitro de Cattleya bicolor e de um duplo híbrido de orquídea ('BLC Pastoral Innocence'). As plântulas foram inoculadas em meio KC sem reguladores de crescimento; os efeitos de concentrações de carvão ativado (0,0; 1,5; 3,0; 4,5; 6,0 and 7,5 g L-1) e de grafite (0,0; 1,5; 3,0; 4,5; 6,0 and 7,5 g L-1) foram testados separadamente. Foram inoculadas quinze plântulas em cada frasco e os experimentos foram conduzidos sob esquema de planejamento fatorial com dois fatores: meios de cultura e luminosidade; o meio de cultura com onze níveis (diferentes concentrações de grafite ou de carvão ativado) e luminosidade com dois níveis (fotoperíodo de 14h e iluminação contínua). Para cada combinação do nível fatorial foram feitas três repetições. Após seis meses de cultivo, foram registrados os números médios de brotos e raízes induzidos em cada concentração de carvão ativado e de grafite. O maior número de brotos nas plântulas de C. bicolor foi observado no meio contendo 6,0 e 7,5 g L-1 de grafite, e o maior número de raízes foi verificado no meio contendo 6,0 g L-1 de carvão ativado. No duplo híbrido o maior número de brotos e raízes foi observado no meio contendo 4,5 g L-1 de carvão ativado. O grafite não pode ser recomendado para substituir o carvão ativado.
Palavras-chave: carvão ativado, grafite, orquídeas, micropropagação.
Introduction
In vitro tissue culture is a powerful alternative tool for germination and initial growth of many orchid species. Knudson 'C' medium (KC medium), described by Knudson (1946), has been the primary medium utilised for in vitro germination of most orchid species (GRIESBACH, 2002). Orchid in vitro propagation or micropropagation is particularly important when dealing with highly valued ornamental plants. These valuable plants are commercially priced and selected for certain phenotypic characteristics, such as their colour, size and shape of flowers, or their potential to allow a species' reintroduction into nature. Species of the genus Cattleya and their hybrids are currently the most commercialised orchids in Brazil due to their large, colourful flowers. Because large numbers of plants have been harvested by orchid collectors and producers, mass propagation of these species will be necessary to replace these orchids in nature.
As in other plant species, orchid in vitro propagation and development may be induced by the inclusion of different supplements (vitamins, amino acids, sugar source and concentration, types and concentrations of growth regulators) in the culture medium and/or by the use of different light and temperature conditions during culture (KRAPIEC et al., 2003; TORRES et al., 2001). Although the addition of growth-regulating substances (auxins and/or cytokinins) is particularly effective and indicated for in vitro orchid propagation (HAMADA et al., 2010; KATSUMI, 2007), the use of these substances is expensive for producers. Producers endeavour to improve micropropagation techniques and reduce costs.
Alternative and inexpensive substances, such as activated charcoal, have been commonly used in tissue culture media to improve growth and development. The beneficial effects of activated charcoal on the in vitro tissue response may result from the darkening of the culture medium for multiple root induction (PASQUAL, 2001) or the adsorption of plant growth regulators and other organic compounds (CHERUVATHUR et al., 2010). The darkening of the culture medium may also be achieved through the addition of graphite. In general, 2.0 g L-1 is the recommended concentration of graphite (ARDITTI et al., 1982). However, no previous study has utilised graphite instead of activated charcoal. Graphite and activated charcoal, known for their carbon structures, are odourless, tasteless and nontoxic substances (JASZCZAK, 1997; MORAES et al., 2005). They may be used to darken the culture medium, to improve root and/or leaf growth and to speed development in in vitro orchid propagation.
The objective of the current study is to analyse different concentrations of activated charcoal and graphite that were separately added to KC medium to stimulate the in vitro propagation of Cattleya bicolor and of a double hybrid orchid ('BLC Pastoral Innocence').
Material and methods
Seeds of Cattleya bicolor Lindl., obtained from a producer in the state of Minas Gerais, Brazil, and seeds of a double hybrid from a cross of Brassavola, Cattleya and Laelia species ('BCL Pastoral Innocence'), obtained from a producer in the region of Mandaguaçu, in the northwestern region of the State of Paraná, Brazil, were germinated in vitro on Kundson-C medium (KNUDSON, 1946) containing 15% coconut water. After eight months, seedlings with two leaves and without roots (10.0 mm height) were placed in culture flasks (250 mL) containing 50 mL KC medium solidified with 0.7% agar and supplemented with 0.0, 1.5, 3.0, 4.5, 6.0, or 7.5 g L-1 activated charcoal or 0.0, 1.5, 3.0, 4.5, 6.0, or 7.5 g L-1 graphite. The pH of the medium was adjusted to 5.3 prior to autoclaving at 121°C for 20 min. The seedlings were inoculated on growth regulator-free KC medium, and the effects of activated charcoal and graphite were tested separately. Each flask contained fifteen seedlings, and the experiments had a completely randomised two-way factorial design with the following two factors: culture medium and illumination. Eleven culture medium conditions (different concentrations of graphite or activated charcoal) and two illumination conditions (14-h photoperiod or continuous illumination) were evaluated. Three replicates of each combination were tested.
The cultures were incubated in a growth chamber at 25 ± 2°C under 14.9 μmol m-2 s1, provided by light radiation with cold-white fluorescent lights (PPF) for a 14-h photoperiod or with continuous illumination (24h).
After six months of culture, the average numbers of induced buds and roots per seedling were recorded for each concentration of activated charcoal and graphite. The resulting differences in treatment conditions were tested by one-way analysis of variance (Anova) and by Tukey-Kramer Multiple Comparison Test (SAS, 1999-2000).
Results and discussion
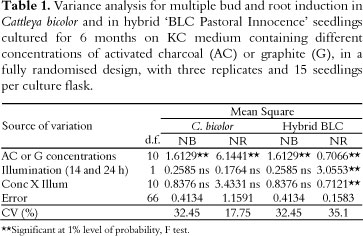
The largest number of buds in Cattleya bicolor seedlings occurred on medium containing 6.0 or 7.5 g L-1 graphite (Tables 1 and 2). The effect of the addition of graphite on the KC medium for in vitro culture of C. bicolor seedlings mimics the effect of auxin (indole-3-butyric acid; IBA) addition to culture medium in in vitro multiplication of Cattleya walkeriana, another species of the same genus (KRAPIEC et al., 2003). In the case of in vitro multiplication of C. walkeriana using seedlings as explants, the greatest frequency of bud induction occurred at concentrations of 1.0 or 1.5 mg L-1 IBA. This effect was observed in both the total absence of cytokinins and in the presence of different cytokinin concentrations.
Our results show that C. bicolor seedlings may be multiplied on KC medium without growth regulators. The addition of more than 2.0 g L-1 graphite (6.0 and 7.5 g L-1) during the culture of C. bicolor seedlings produces an endogenous auxin/cytokinin level that is adequate for the largest bud production on growth regulator-free media (Figure 1). Multiple bud induction is highly important when culturing a valuable plant selected for its colour and the shape of its flowers. Bud clusters may be divided into single buds and transferred to fresh medium for further multiplication. Micropropagation on growth regulator-free media is important for producers because of their focus on improvements in micropropagation techniques and cost reduction.
However, different concentrations of graphite in culture media resulted in no significant differences in root formation in C. bicolor seedlings (Table 1). The largest number of roots occurred on medium containing 6.0 g L-1 activated charcoal (Figure 2). Furthermore, the effect of activated charcoal on multiple root induction was revealed to be significantly different by Tukey's Student Range Test (Tables 1 and 2).
The varied effects of graphite and activated charcoal in root formation indicated that the use of graphite in place of activated charcoal is not recommended. The darkening of culture medium was not the main variable in determining the development of roots in C. bicolor seedlings in KC culture medium; we observed that the addition of activated charcoal led to other effects besides the darkening of culture medium.
Activated charcoal is pure carbon that has been specially processed to adsorb a wide variety of chemical compounds and gases (SÁENZ et al., 2010). It has been used since ancient times to cure a variety of ailments, including poisoning. According to Sáenz et al. (2010), the use of activated charcoal has been well documented by Egyptians from as early as 1550 B.C. When added to plant medium, activated charcoal adsorbs all toxic compounds released by explants. In addition to the adsorption of toxins, activated charcoal may also determine whether an endogenous auxin/cytokinin level that is adequate for root production in C. bicolor may be reached.
Another beneficial effect of activated charcoal is the induction of a high number of buds and roots in seedlings of the hybrid 'BLC Pastoral Innocence'. The largest numbers of buds and roots were observed in the medium containing 4.5 g L-1 activated charcoal (Table 2). The addition of activated charcoal to the KC culture medium may alter the interaction between endogenous auxin and cytokinin, which controls bud and root formation in seedling explants of the hybrid 'BLC Pastoral Innocence'. The result is an approximately 3-fold increase in the number of buds per seedlings (Table 2). The addition of 4.5 g L-1 activated charcoal to KC medium also led to a 13-fold increase in root length (results not shown).
The level of auxin to cytokinin is important in determining the formation of roots and buds during in vitro tissue culture (TAJI et al., 2002). Whereas roots are formed under conditions of high auxin and low cytokinin levels, buds are formed under conditions of low auxin and high cytokinin. If both are low, no growth or development occurs. The addition of 4.5 g L-1 activated charcoal to in vitro culture medium of the hybrid 'BLC Pastoral Innocence' seedlings may successfully induce the seedling to increase the number of buds and roots. Appropriate levels of auxin to cytokinin override the apical dominance effect and induce bud formation (TRIGIANO; GRAY, 2005); it is also sufficient to induce cell division activity in the root pericycle tissue, which ultimately result in root initiation (GALUSZKA et al., 2008; HOSSAIN et al., 2009, 2010; RESKI, 2006). The adsorptive capacity of activated charcoal has been shown to be related to a decrease in endogenous auxin levels and therefore leads to a larger number of expanded leaves in Catassetum fimbriatum, another species of orchids (MORALES et al., 2006). However, root induction from seedlings of C. fimbriatum showed no significant difference when seedlings were grown on medium with different concentrations of activated charcoal.
The presence of activated charcoal has led to various effects in different species. The differences may be attributed to the specific type of activated charcoal used. Different results have been obtained utilising activated charcoal of various degrees of purity (MORAES et al., 2005). However, current results using the same type of activated charcoal indicated that the effect of activated charcoal in orchids is dependent upon the plant species or genotype.
Experiments in which plants were subjected to either a 14-h photoperiod or continuous illumination (24h) revealed no significant differences in multiple bud and root induction in C. bicolor seedlings (Table 1). However, a significant effect on root induction was observed in the hybrid 'BLC Pastoral Innocence' when continuous illumination was employed. On a commercial scale, the use of continuous illumination in micropropagation would be highly expensive for orchid producers. However, results showed that 24-h light may be important for multiple root induction in some plants with high commercial value, such as the hybrid 'BLC Pastoral Innocence'.
Another effect of activated charcoal and graphite is related to the final pH changes in the KC medium. Our results showed that the addition of activated charcoal or graphite led to an alkaline medium after autoclaving (initial pH). However, the final pH change was evident only in the KC medium containing activated charcoal and C. bicolor or 'BLC Pastoral Innocence' seedlings (results not shown). The final pH of the KC medium containing graphite remained unchanged during 6 months of in vitro seedling culture. The lower pH-buffering capacity of activated charcoal has also been reported in the in vitro culture of other orchid species and hybrids (MORAES et al., 2005; MORALES et al., 2006).
In our study, the single beneficial effect of graphite, compared to activated charcoal, was in bud induction in C. bicolor seedlings. However, the results of the current research are important because they indicate that graphite and activated charcoal may be used for bud and root induction, respectively, in C. bicolor seedlings. Furthermore, activated charcoal may be used for bud and root induction in the hybrid 'BLC Pastoral Innocence' in media lacking growth regulators. The continuous multiplication of buds in growth regulator-free media is important because new seedlings should be genetically uniform. In spite of the varied effects of activated charcoal and graphite in our study, the addition of graphite, in place of growth regulators, to KC medium could allow for a new labour-saving operation that minimises cost and operational requirements for orchid producers.
Conclusion
The varied effects of graphite and activated charcoal on root formation demonstrated that graphite is not recommended as a substitute for activated charcoal. Additionally, the darkening of culture medium was not the main factor in determining root development in seedlings of C. bicolor and the hybrid 'BLC Pastoral Innocence'. Our results show that graphite and activated charcoal may be important for bud and root induction, respectively, in C. bicolor seedlings. Furthermore, when using medium lacking growth regulators, activated charcoal may be important for bud and root induction in the hybrid 'BLC Pastoral Innocence'.
Acknowledgements
Financial support for this project was provided by CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior).
Received on January 16, 2011.
Accepted on May 30, 2011.
License information: This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
- ARDITTI, J.; CLEMENTS, M. A.; SAST, G.; HADLEY, G.; NICHIMURA, G.; ERNEST, R. Orchid seed germination and seedling culture - a manual. In: ARDITTI, J. (Ed.). Orchid biology: reviews and perspectives II. New York: Cornel University Press, 1982.
- CHERUVATHUR, M. K.; ABRAHAM, J.; MANI, B.; THOMAS, T. D. Adventitious shoot induction from cultured intermodal explants of Malaxis acuminate D. Don, a valuable terrestrial medicinal orchid. Plant Cell, Tissue and Organ Culture, v. 101, n. 2, p. 163-170, 2010.
- GALUSZKA, P.; SPICHAL, L.; KOPECNY, D.; TARKOWSKI, P.; FREBORTOVA, J.; SEBELA, M.; FREBORT, I. Metabolism of plant hormones cytokinins and their function in signaling, cell differentiation and plant development. Studies in Natural Products Chemistry, v. 34, part N, p. 203-264, 2008.
- GRIESBACH, R. J. Development of Phalaenopsis orchids for the mass-market. In: JANICK, J.; WHIPKEY, A. (Ed.). Trends in new crops and new uses Alexandria: ASHS Press, 2002. (West Lafayette). p. 458-465.
- HAMADA, K.; SHIMASAKI, K.; OGATA, T.; NISHIMURA, Y.; NAKAMURA, K.; OYAMA-EGAWA, H.; YOSHIDA, K. Effects of spectral composition conversion film and plant growth regulators on proliferation of Cymbidium protocorm like body (PLB) cultured in vitro. Environmental Control Biology, v. 48, n. 3, p. 127-132, 2010.
- HOSSAIN, M. M.; SHARMA, M.; PATHAK, P. Cost effective protocol for in vitro mass propagation of Cymbidium aloifolium (L.) Sw.- a medicinally important orchid. Engineering in Life Sciences, v. 9, n. 6, p. 444-453, 2009.
- HOSSAIN, M. M.; SHARMA, M.; SILVA, J. A. T.; PATHAK, P. Seed germination and tissue culture of Cymbidium giganteum Wall. ex Lindl. Scientia Horticulturae, v. 123, n. 4, p. 479-487, 2010.
- JASZCZAK, J. A. Unusual graphite crystals from the lime crest quarry, Sparta, New Jersey. Rocks and Minerals, v. 72, n. 4, p. 330-334, 1997.
- KATSUMI, M. Studies on plant growth substances in Japan before 1945. Plant Biotechnology, v. 24, n. 2, p. 155-163, 2007.
- KNUDSON L. A new nutrient solution for the germination of orchid seeds. American Orchid Society Bulletin, v. 15, n. 9, p. 214-217, 1946.
- KRAPIEC, P. V.; MILANEZE, M. A. G.; MACHADO, M. F. P. S. Effects of different combinations of growth regulators for bud induction from seedlings of Cattleya walkeriana Gardner (Orchidaceae). Acta Scientiarum. Biological Sciences, v. 25, n. 1, p. 179-182, 2003.
- MORAES, L.; FARIA, R. T.; CUQUEL, F. L. Activated charcoal for in vitro propagation of Brazilian orchids. Acta Hortculturae, v. 683, n. 3, p. 383-389, 2005.
- MORALES, S.; MILANEZE, M. A. G.; MACHADO, M. F. P. S. Effect of activated charcoal for seedlings development of Catassetum fimbriatum Lindl (Orchidaceae). Journal of Plant Science, v. 1, n. 4, p. 388-391, 2006.
- PASQUAL, M. Meios de cultura Lavras: UFL/FAEPE, 2001.
- RESKI, R. Small molecules on the move: homeostasis, crosstalk, and molecular action of phytohormones. Plant Biology, v. 8, n. 3, p. 277-280, 2006.
- SÁENZ, L.; HERRERA-HERRERA, G.; UICAB-BALLOTE, F.; CHAN, J. L.; OROPEZA, C. Influence of formo f activated charcoal on embryogenic callis formation in coconut (Cocos nufera). Plant Cell, Tissue and Organ Culture, v. 100, n. 3, p. 301-308, 2010.
- SAS-Statistical Analisys System. Statistical analisys system user's guide Cary: Statistical Analysis System Institute, 1999-2000.
- TAJI, A.; KUMAR, P.; LAKSHMANAN, P. Micropropagation. In: TAJI, A.; KUMAR, P.; LAKSHMANAN, P. (Ed.). In vitro plant breeding New York: The Haworth Press Inc., 2002. p. 15-32.
- TORRES, A. C.; BARBOSA, N. V. R.; WILLADIONO, L.; GUERRA, M. P.; FERRERIA, C. F.; PAIVA, S. A. V. Meio e condições de incubação para cultura de tecidos de plantas Brasília: Embrapa/CNPH, 2001. (Circular Técnica).
- TRIGIANO, R. N.; GRAY, D. J. Plant development and biotechnology Boca Raton: CRC Press LLC, 2005.
Publication Dates
-
Publication in this collection
05 June 2012 -
Date of issue
June 2012
History
-
Received
16 Jan 2011 -
Accepted
30 May 2011