ABSTRACT.
The present study aimed to investigate the effects of pre-storage for 24h and 48h and different modified atmosphere packaging-MAP treatments (using low-density polyethylene - LDPE) on the antioxidant compounds and the postharvest quality of ‘Douradão’ peaches under cold storage. The peaches were submitted to the following treatments: a control (no packaging or pre-storage) and pre-storage for 24h or 48h at 25ºC combined with passive MAP (LDPE 60 µm or 80 µm). After the application of the treatments, the fruits were stored at 0ºC for 30 days plus 3 days at 25ºC to simulate marketing conditions. The results showed that on the simulated marketing period, all the treatments (except for the control) reduced woolliness and internal browning. Furthermore, both pre-storage and LDPE MAP enhanced the polygalacturonase (PG) activity, the levels of phenolic compounds and the antioxidant capacity. Thus, these results suggest that the pre-storage associated with LDPE packaging increases the shelf life of peaches by up to 30 days under cold storage.
Keywords:
Prunus persica (L.) Batsch; stone fruit; woolliness; internal browning; postharvest.
Introduction
The peach [Prunus persica (L.) Batsch] is the most important species of the genus Prunus and one of the most significant fruits in terms of production (Cantin et al., 2010Cantín, C. M., Crisosto, C. H., Ogundiwin, E. A., Gradziel, T., Torrents, J., Moreno, M. A., & Gogorcena, Y. (2010). Chilling injury susceptibility in an intra-specific peach [Prunus persica (L.) Batsch] progeny. Postharvest Biology and Technology, 58(2), 79-87. DOI: 10.1016/j.postharvbio.2010.06.002
https://doi.org/10.1016/j.postharvbio.20...
). It is a highly perishable climacteric fruit that deteriorates rapidly at room temperature (Lurie & Crisosto, 2005Lurie, S., & Crisosto, C. H. (2005). Chilling injury in peach and nectarine: review. Postharvest Biology and Technology, 37(3), 195-208. DOI: 10.1016/j.postharvbio.2005.04.012
https://doi.org/10.1016/j.postharvbio.20...
). Storage at temperatures of approximately 0ºC has been used to extend the shelf life of peaches by 3 - 4 weeks depending on the cultivar. However, long storage periods at low temperatures may cause chilling injuries such as woolliness and internal browning. These injuries are only perceived when the fruit is exposed to room temperature during marketing (Lurie & Crisosto, 2005Lurie, S., & Crisosto, C. H. (2005). Chilling injury in peach and nectarine: review. Postharvest Biology and Technology, 37(3), 195-208. DOI: 10.1016/j.postharvbio.2005.04.012
https://doi.org/10.1016/j.postharvbio.20...
). According to Zhou, Ben-Arie, and Lurie (2000Zhou, H.W., Ben-Arie, R., & Lurie, S. (2000). Pectin esterase, polygalacturonase and gel formation in peach pectin fractions. Phytochemistry, 55(3), 191-195. DOI: org/10.1016/S0031-9422(00)00271-5
https://doi.org/10.1016/S0031-9422(00)00...
), the woolliness in stone fruits is a chilling injury caused by an imbalance in the activity of pectinolytic enzymes during the maturation stage. This imbalance is caused by increased pectinmetylesterase (PME) activity and decreased polygalacturonase (PG) activity. It results in the accumulation of pectic substances with a low degree of esterification that can bind to the water present in the cells, forming a gel and leading to a lack of juiciness (Zhou, Dong, Ben-Arie, & Lurie, 2001Zhou, H. W., Dong, L., Ben-Arie, R., & Lurie, S. (2001). The role of ethylene in the prevention of chilling injury in nectarines. Journal of Plant Physiology, 158(1), 55-61. DOI: doi.org/10.1078/0176-1617-00126
https://doi.org/10.1078/0176-1617-00126...
). These partially degraded substances can also trespass into intercellular space and mesocarp cells and capture the water that is present (Bron, Jacomino, & Appezzato-da-Glória, 2002Bron, I. U., Jacomino, P. A., & Appezzato-da-Glória, B. (2002). Alterações anatômicas e físico-químicas associadas ao armazenamento refrigerado de pêssegos ‘Aurora-1’ e ‘Dourado-2’. Pesquisa Agropecuária Brasileira, 37(10), 1349-1358. DOI: 10.1590/S0100-204X2002001000001
https://doi.org/10.1590/S0100-204X200200...
).
To prolong the cold storage time and maintain fruit quality, it is necessary to use additional technologies such as pre-storage and a modified atmosphere. Pre-storage consists of exposing the harvested fruits to temperatures between 20ºC and 30ºC for 1 or 2 days before cold storage (Infante, Meneses, & Crisosto, 2009Infante, R., Meneses, C., & Crisosto, C. H. (2009). Preconditioning treatment maintains taste characteristic perception of ripe ‘September Sun’ peach following cold storage. Journal of Food Science and Technology, 44(5), 1011-1016. DOI: 10.1111/j.1365-2621.2008.01864.x
https://doi.org/10.1111/j.1365-2621.2008...
). Crisosto, Garner, Andris, and Day (2004) observed that the pre-storage of ‘Summer Lady’ and ‘Ryan Sun’ peaches for 48 hours at 20ºC reduced the woolliness and increased the storage period. The pre-storage treatment also enhanced PG activity in peaches (Von Mollendorff & De Villiers, 1988Von Mollendorff, L. J., & De Villiers, O. T. (1988). Role of pectolytic enzymes in the development of woolliness in peaches. Journal of Horticultural Science, 63(1), 53-58. DOI: org/10.1080/14620316.1988.11515827
https://doi.org/10.1080/14620316.1988.11...
) and nectarines (Zhou et al., 2000Zhou, H.W., Ben-Arie, R., & Lurie, S. (2000). Pectin esterase, polygalacturonase and gel formation in peach pectin fractions. Phytochemistry, 55(3), 191-195. DOI: org/10.1016/S0031-9422(00)00271-5
https://doi.org/10.1016/S0031-9422(00)00...
), which reduced the woolliness in the fruits when compared to those submitted to cold storage immediately after harvest. Peaches and nectarines that were submitted to pre-storage for 1 or 2 days presented a reduction in woolliness, which improved the sensory quality of these fruits and increased the consumer acceptance (Sasaki, Cerqueira, Sestari, Del Aguila, & Kluge, 2010Sasaki, F. F., Cerqueira, T. S., Sestari, I., Del Aguila, J. S., & Kluge, R. A. (2010). Woolliness control and pectin solubilization of ‘Douradao’ peach after heat shock treatment. Acta Horticulturae, 877, 539-542. DOI: 10.17660/AcataHortic.2010.877.6
https://doi.org/10.17660/AcataHortic.201...
; Neves, Campos, Prill, & Roberto, 2013Neves, L. C., Campos, J. C., Prill, M. A. S., & Roberto, S. R. (2013). Woolliness and leatheriness in late peach cultivars submitted to both pre-storage and to cold storage. Acta Scientiarum. Agronomy, 35(3), 363-369. DOI: 10.4025/actasciagron.v35i3.13347
https://doi.org/10.4025/actasciagron.v35...
; Infante et al., 2009Infante, R., Meneses, C., & Crisosto, C. H. (2009). Preconditioning treatment maintains taste characteristic perception of ripe ‘September Sun’ peach following cold storage. Journal of Food Science and Technology, 44(5), 1011-1016. DOI: 10.1111/j.1365-2621.2008.01864.x
https://doi.org/10.1111/j.1365-2621.2008...
). Nevertheless, one of the problems reported by these authors is the reduction in firmness and the increase in weight loss during the storage period.
Modified atmosphere is a widely used technique for limiting water loss, delaying ripening and suppressing postharvest diseases in fruits, and it contributes to prolonging shelf life during cold storage (Malakou & Nanos, 2005Malakou, A., & Nanos, G. D. (2005). A combination of hot water treatment and modified atmosphere packaging maintains quality of advanced maturity ‘Caldesi 2000’ nectarines and ‘Royal Glory’ peaches. Postharvest Biology and Technology, 38(2), 106-114. DOI: org/10.1016/j.postharvbio.2005.06.003
https://doi.org/10.1016/j.postharvbio.20...
). It reduces the levels of O2, while CO2 levels increase inside the packaging due to the combination of the product’s mass and the respiration rate, the gas transmission rate through the packaging and the permeation area (Kartal, Aday, & Caner, 2012Kartal, S., Aday, M. S., & Caner, C. (2012). Use of microperforated films and oxygen scavengers to maintain storage stability of fresh strawberries. Postharvest Biology and Technology, 71, 32-40. DOI: org/10.1016/j.postharvbio.2012.04.009
https://doi.org/10.1016/j.postharvbio.20...
). However, changes in the levels of O2 and CO2 should not exceed the critical limit to prevent the occurrence of physiological disorders such as fermentation (Beaudry, 1999Beaudry, R. M. (1999). Effect of O2 and CO2 partial pressure on selected phenomena affecting fruit and vegetable quality. Postharvest Biology and Technology, 15(3), 293-303. DOI: 10.1016/S0925-5214(98)00092-1
https://doi.org/10.1016/S0925-5214(98)00...
). Studies indicate that the use of passive modified atmosphere in peaches under cold storage reduces the loss of weight and firmness and may decrease chilling injuries in some cultivars (Santos, Castro, Picoli, & Rolim, 2008Santos, C. A. A., Castro, J. V., Picoli, A. A., & Rolim, G. S. (2008). Uso de quitosana e embalagem plástica na conservação pós-colheita de pêssegos ‘Douradão’. Revista Brasileira de Fruticultura, 30(1), 88-93. DOI: org/10.1590/S0100-29452008000100017
https://doi.org/10.1590/S0100-2945200800...
; Santana, Benedetti, Sigrist, & Sarantopóulos, 2010Santana, L. R. R., Benedetti, B. C., Sigrist, J. M. M., & Sarantopóulos, C. I. G. L. (2010). Modified atmosphere packaging extending the storage life of ‘Douradão’ peach. Revista Brasileira de Fruticultura, 32(4), 1009-1017. DOI: doi.org/10.1590/S0100-29452011005000006
https://doi.org/10.1590/S0100-2945201100...
). After evaluating the use of polypropylene and polyethylene, Akbudak and Eris (2004Akbudak, B., & Eris, A. (2004). Physical and chemical changes in peaches and nectarines during the modified atmosphere storage. Food Control, 15(4), 307-313. DOI: 10.1016/S0956-7135(03)00082-3
https://doi.org/10.1016/S0956-7135(03)00...
) reported that these packages were efficient in maintaining the quality of 'Flavorcrest' peaches and 'Fantasia' nectarines for 30 and 45 days of storage, respectively.
Although there are several studies regarding chilling injuries, they are still the major problem associated with the conservation of stone fruits around the world (Fruk, Cmelik, Jemric, Hribar, & Vidrih, 2014Fruk, G., Cmelik, Z., Jemric, T., Hribar, J., & Vidrih, R. (2014). Pectin role in woolliness development in peaches and nectarines: a review. Scientia Horticulturae, 180, 1-5. DOI: doi.org/10.1016/j.scienta.2014.09.042
https://doi.org/10.1016/j.scienta.2014.0...
; Brummell, Dal Cin, Lurie, Crisosto, & Labavitch, 2004Crisosto, C. H., Garner, D., Andris, H. L., & Day, K. R. (2004). Controlled delayed cooling extends peach market life. HortTechnoIogy, 14(1), 99-104.). Through the use of pre-storage and controlled atmosphere, Murray, Lucangeli, Polenta, and Budde (2007Murray, R., Lucangeli, C., Polenta, G., & Budde, C. (2007). Combined pre-torage heat treatment and controlled atmosphere storage reduce internal breakdown of ‘Flavorcred’ peach. Postharvest Biology and Technology, 44(2), 116-121. DOI: org/10.1016/j.postharvbio.2006.11.013
https://doi.org/10.1016/j.postharvbio.20...
) observed that combined techniques for the conservation of peaches and nectarines under refrigeration are more significant in the reduction of chilling injuries than the same treatments when separately applied. The same was observed by Jin, Zheng, Tang, Rui, and Wang (2009Jin, P., Zheng, Y., Tang, S., Rui, H., & Wang, C. Y. (2009). A combination of hot air and methyl jasmonate vapor treatment alleviates chilling injury of peach fruit. Postharvest Biology and Technology, 52(1), 24-29. DOI: org/10.1016/j.postharvbio.2008.09.011
https://doi.org/10.1016/j.postharvbio.20...
) with the use of pre-storage and methyl jasmonate, Malakou and Nanos (2005Malakou, A., & Nanos, G. D. (2005). A combination of hot water treatment and modified atmosphere packaging maintains quality of advanced maturity ‘Caldesi 2000’ nectarines and ‘Royal Glory’ peaches. Postharvest Biology and Technology, 38(2), 106-114. DOI: org/10.1016/j.postharvbio.2005.06.003
https://doi.org/10.1016/j.postharvbio.20...
) with pre-storage and modified atmosphere, and Cao, Hu, Zheng, and Lu (2010Cao, S., Hu, Z., Zheng, Y., & Lu, B. (2010). Synergistic effect of heat treatment and salicylic acid on alleviating internal browning in cold-stored peach fruit. Postharvest Biology and Technology, 58(2), 93-97. DOI: 10.1016/j.postharvbio.2010.05.010
https://doi.org/10.1016/j.postharvbio.20...
) with pre-storage and salicylic acid. ‘Douradão’ peaches are widely cultivated in Brazil with great consumer acceptance. As different varieties of peach present diverse responses to postharvest treatments (Ju, Duan, Ju, & Guo, 2001Ju, Z., Duan, Y., Ju, Z., & Guo, A. (2001). Different responses of ‘Snow Giant’ and ‘Elegant Lady’ peaches to fruit maturity and storage temperature. Journal of Horticultural Science and Biotechnology, 76(5), 575-580.), the study of new postharvest technologies that prolong the storage of ‘Douradão’ peaches is extremely relevant. This work aimed to evaluate the effects of the combination of pre-storage and passive modified atmosphere in reducing the woolliness and prolonging the shelf life of ‘Douradão’ peaches under cold storage.
Material and methods
‘Douradão’ peaches were harvested in the region of Itupeva, São Paulo State, at physiological maturity (breaking of the green background color) and were transported to the Laboratory of Postharvest Physiology and Biochemistry at the Biological Sciences Department of the “Luiz de Queiroz” College of Agriculture in Piracicaba, São Paulo State, Brazil. The fruits were sorted by size and selected according to the absence of mechanical injuries or visible signs of pathogenic agents.
The following treatments were applied: control (no packaging or pre-storage used before cold storage); pre-storage for 24h at 25ºC + LDPE 60 µm (CO2 transmission of 9,577 mL m-2 day-1 and O2 transmission of 2,872 mL m-2 day-1 with a moisture transmission that was 5.9 g water m-2 day-1); pre-storage for 48h at 25ºC + LDPE 60 µm; pre-storage for 24h at 25ºC + LDPE 80 µm (CO2 transmission of 7,425 mL m-2 day-1, O2 transmission of 1,705 mL m-2 day-1, and the moisture transmission was 4.1 g water m-2 day-1); and pre-storage for 48h at 25ºC + LDPE 80 µm. The fruits submitted to pre-storage were placed in hermetic boxes with a 186 L capacity and stored at 25ºC. The control fruits were placed in open plastic boxes. After pre-storage, the fruits were placed in LDPE packages to promote passive modified atmosphere during cold storage. All the treatments were stored at 0ºC and 90 ± 5% RH for 30 days. After this period, the packages were opened, and the fruits were kept at 25ºC and 60 ± 5% RH for 3 days to simulate marketing conditions.
The following variables were determined:
Package Internal Gas Composition: A gas analyzer (Illinois Instruments, model 6600 Headspace Oxygen/Carbon dioxide analyzer) was used to determine the gas composition within the package during cold storage. Samples of the inner atmosphere were collected using silicon septa previously fixed to the packages. The results were expressed in percentages of O2 and CO2.
Woolliness Index: Woolliness was determined according to Nava and Brackmann (2002Nava, G. A., & Brackmann, A. (2002). Armazenamento de pêssegos (Prunus persica (L.) Batsch), cv. Chiripá, em atmosfera controlada. Revista Brasileira de Fruticultura, 24(2), 328-332. DOI: 10.1590/S0100-29452002000200011
https://doi.org/10.1590/S0100-2945200200...
), with adaptations. The evaluation was subjective, considering the appearance and quality of the manually extracted juice. The fruits were divided into categories: 1 = fruits with high juice content (without woolliness), 2 = fruits with moderate juice content (moderate wooliness) and 3 = fruits without juice (totally woolly).
Internal browning index: This index was evaluated according to an adapted methodology of Ben-Arie and Sonego (1980Ben-Arie, R., & Sonego, L. (1980). Pectolytic enzyme activity involved in woolly breakdown of stored peaches. Phytochemistry, 19(2), 2553-2555. DOI: doi.org/10.1016/S0031-9422(00)83917-5
https://doi.org/10.1016/S0031-9422(00)83...
). The index of internal browning was calculated through a visual scale as follows: fruits without browning (0% of the fruit surface), slightly brown flesh (< 25% of the fruit surface), moderately brown flesh (25-50%), and severely brown flesh (> 50% of the fruit). The index of internal browning (IB) was calculated as the following quotient. IB (0 × % of fruits without browning) + (1 × % of slightly brown fruits) + (2 × % of moderately brown fruits) + (3 × % severely brown fruits)/4. The higher the value that was obtained, the more intense the internal browning.
PME activity (EC 3.1.1.11): The activity was determined by spectrophotometry and according to the adapted methodology of Ratner, Goren, and Monseline (1969Ratner, A., Goren, R., & Monseline, S. P. (1969). Activity of pectinesterase and cellulase in the abscission zone of citrus leaf explants. Plant Physiology, 44(12), 1717-1723. DOI: https://doi.org/10.1104/pp.44.12.1717
https://doi.org/10.1104/pp.44.12.1717...
). For the extraction, 5 g of frozen fruit flesh was triturated with 20 mL of NaCl (0.2 N) at 4ºC (enzymatic extract). The activity was quantified using 30 mL of a pectin solution (1% - diluted in NaCl 0.2 N) with the pH adjusted to 7.0 with NaOH (0.1 N). A total of 5 mL of enzymatic extract was added, and the pH was again adjusted to 7.0 with NaOH (0.1 N). Then, the sample was titrated with NaOH (0.01 N) for 10 minutes to maintain the pH at 7.0. The PME activity was considered the content of the enzyme able to catalyze pectin demethylation, which corresponds to 1 nmol of NaOH per gram of fresh weight per minute (nmol g-1 min.-1).
PG activity (EC 3.2.1.15): The enzymatic extract was obtained according to Denès, Baron, and Drilleau (2000Denès, J. M., Baron, A., & Drilleau, J. F. (2000). Purification, properties and heat inactivation of pectin methylesterase from apple (cv. Golden Delicious). Journal of the Science of Food and Agriculture, 80(10), 1503-1509. DOI: 10.1002/1097-0010(200008)80:10<1503:AID-JSFA676>3.0.CO;2-U
https://doi.org/10.1002/1097-0010(200008...
) with small modifications. A total of 0.5 g of lyophilized flesh was weighed and homogenized with 15 mL of cold acetone using a high-speed blender (Ultraturrax, UT) for 1 minute. Subsequently, the homogenate was kept in an expanded polystyrene box containing ice for 30 minutes. Then, the supernatant was discarded. The pellet was resuspended in 10 mL of cold acetone and kept in an expanded polystyrene box containing ice for 15 minutes. Next, the supernatant was discarded, and the pellet was used for enzyme determination. The pellet was resuspended in 10 mL of 0.2 M Tris (hydroxymethyl)-aminomethane buffer (Tris buffer), pH 7, containing 500 mg L-1 of sodium metabisulphite, 1% polyvinylpolypyrrolidone (PVPP) and 1 M of NaCl. The extraction was conducted for 2 hours using an orbital shaker at 200 × g in the dark and inside an expanded polystyrene box containing ice. The extract was transferred to Eppendorf tubes and centrifuged at 20,000 × g for 15 minutes at 4ºC. The supernatant was used as an extract for the enzymatic assay. The PG activity was determined according to the method proposed by Gross (1982Gross, K. C. (1982). A rapid and sensitive spectrophotometric method for assaying poligalacturonase using 2-cyanoacetamide. HortScience, 17(6), 933-934.), which measures the reduction of the formed groups using polygalacturonic acid as the substrate. The substrate contained 0.6 mL of a solution prepared with 0.4% (w/v) polygalacturonic acid dissolved in 0.05 M of sodium acetate buffer with a pH of 4.5. The reaction occurred by the addition of 0.15 mL of enzymatic extract followed by incubation at 37°C for 10 minutes under agitation at 30 × g. The reaction was stopped by adding 2 mL of 10 mM borate buffer at pH 9 and 0.4 mL of 1% (w/v) cyanoacetamide. The mixture was placed in a water bath at 100ºC for 10 minutes followed by a cooling period at room temperature. A total of 200 µL of the sample was pipetted to an ultraviolet-flat-bottom microplate and read in a multiple microplate reader at 22ºC. The absorbance was read at 276 nm. The calibration curve was conducted with D-galacturonic acid, and the PG activity was expressed as mmoL of galacturonic acid per gram of dry weight per minute (mmoL g-1 min.-1).
Flesh firmness: This parameter was determined using a manual penetrometer with an 8-mm diameter flat tip. After removing the peel, two measures were taken from each fruit from opposite sides of the equatorial section. The results were expressed in Newtons (N).
Total phenolic compounds: Samples of 0.2 g of lyophilized flesh were weighed and homogenized with 6 mL of methanol using a high-speed blender (Ultraturrax, UT) for 1 minute. The extraction was conducted for 1 hour in an orbital shaker at 200 × g in the dark and inside an expanded polystyrene box containing ice. Then, 2 mL of extract was transferred to Eppendorf tubes and centrifuged at 15,000 × g for 10 minutes at 4ºC. The content of the phenolic compounds solubilized in the supernatant was determined according to Swain and Hillis (1959Swain, T., & Hillis, W. E. (1959). The phenolic constituents of Prunus domestica. The quantitative analysis of phenolic constituents. Journal of the Science of Food and Agriculture, 10(1), 63-68. DOI: 10.1002/jsfa.2740100110
https://doi.org/10.1002/jsfa.2740100110...
) with modifications. A total of 19.2 µL of extract was placed in a flat-bottom microplate, and subsequently, 29 µL of Folin-Ciocalteu reagent (1N) was added. After 3 minutes of incubation in the dark and at room temperature, 192 µL of a solution containing Na2CO3 (0.4%) and NaOH (2%) was incorporated and then the samples were kept in the dark for 1 hour at room temperature. The absorbance was measured at 750 nm in a multiple microplate reader (Tecan Infinite M200, Männedorf, Switzerland) at 22ºC. The results are expressed in mg of gallic acid in 100 g of dry weight.
Antioxidant capacity (FRAP): Samples of 0.2 g of lyophilized flesh were weighed and homogenized with 6 mL of methanol using a high-speed blender (Ultraturrax, UT) for 1 minute. The extraction was conducted for 1 hour in an orbital shaker at 200 × g in the dark and inside an expanded polystyrene box containing ice. After that, 2 mL of extract was transferred to Eppendorf tubes and centrifuged at 15,000 × g for 10 minutes at 4ºC. The total antioxidant capacity was determined based on the methodology of Benzie and Strain (1996Benzie, I. F. F., & Strain, J. J. (1996). Ferric reducing ability of plasma (FRAP) as a measure of antioxidant power: The FRAP assay. Analytical Biochemitry, 239(1), 70-76. DOI: 0.1006/ABIO.1996.0292
https://doi.org/0.1006/ABIO.1996.0292...
). A buffer solution composed of 300 mmoL L-1 of acetate, pH 3.6 (3.1 g C2H3NaO2•3H2O) and 16 mL of C2H4O2 per liter of buffer solution was prepared. Two other solutions were made: one using 10 mmoL L-1 of 2, 4, 6-tripyridyl-s-triazina (TPTZ) in 40 mmoL L-1 of HCl and the other using 20 mmoL L-1 of FeCl3•6H2O. The FRAP reagent was prepared by the addition of 10 mL of acetate buffer, 1 mL of TPTZ and 1 mL FeCl3•6H2O followed by incubation for 2 hours at 37ºC. A 6 µL aliquot of the extract was placed in a flat-bottom microplate, and subsequently, a total of 198 µL of FRAP solution was added. The microplate was then kept in the dark for 50 minutes at room temperature, and the absorbance was measured at 593 nm in a multiple microplate reader (Tecan Infinite M200, Männedorf, Switzerland). The results were expressed in mg of trolox in 100 g of dry weight.
For each treatment, all these analyses were conducted on day 0, after 30 days of cold storage and after 30 days of cold storage plus 3 days at room temperature. Each treatment was comprised of 4 repetitions of eight fruits except for the gas composition analysis, which occurred on days 0, 5, 10, 15, 20, 25, and 30 of cold storage. In this case, each treatment comprised 5 repetitions of 4 fruits.
To determine the effect of each factor on each individual parameter, a one-way analysis of variance (ANOVA, p ≤ 0.05) was conducted (statistics software SAS, version 9.2, SAS Institute, Cary, NC, USA). The mean values were compared by Tukey’s test when significant differences among treatments and interactions between factors were found.
Results
Gas composition
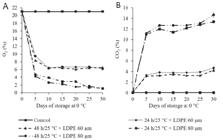
CO2 production increased, while O2 was reduced for all the pre-storage treatments in association with LDPE packaging when compared to the control. The LCPE with 60 µm packaging showed contents of CO2 and O2 approximately 6% and 5%, respectively. The LDPE with 80 µm packaging showed contents approximately 16% for CO2 and 1% for O2 (Figure 1A and B).
The O2 (A) and CO2 (B) composition inside the modified atmosphere packages (LDPE 60 or 80 µm) of ‘Douradão’ peaches after pre-storage (24 or 48h at 25°C) and during storage for 30 days at 0°C and 90 ± 5% RH. The bars represent a standard deviation of the mean (n = 5).
Woolliness index, Internal browning index, PME, and PG activity
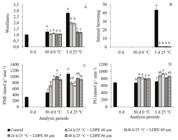
No significant difference was found between the treatments for the index of woolliness when the fruits were removed from the cold storage. However, all the combined treatments of pre-storage with modified atmosphere using LDPE 60 and 80 µm showed a lower index of woolliness, 2 and 1.20, respectively, when compared to the control (2.8) in the simulated marketing period (Figure 2A). The MAP using LDPE of 80 µm provided the lowest index of woolliness.
The index of woolliness (A), Internal browning index (B), PME activity (C) and PG activity (D) in ‘Douradão’ peaches submitted to pre-storage (24 or 48h at 25ºC), packaged under modified atmosphere (LDPE 60 or 80 µm) and stored for 30 days at 0°C and ± 5% RH followed by a simulated marketing period of 3 days at 25°C and 60 ± 5% RH. Means followed by the same letter do not differ between treatments by the Tukey’s test (p ≥ 0.05). The index of woolliness: 1 = fruits with a high content of juice (without woolliness); 2 = fruits with a moderate content of juice (moderate woolliness) and 3 = fruits without juice (totally woolly).
The internal browning did not affect the fruits when they were removed from the cold storage. However, in the simulated marketing period, the control treatment presented the highest index of internal browning (Figure 2B). PME showed low activity in the control treatment when the fruits were removed from cold storage. Nonetheless, in the simulated marketing period, the control treatment presented the highest activity of this enzyme when compared to the other treatments except for the pre-storage for 24h + LDPE 80 µm (Figure 2C). PG presented lower activity for the control treatment when the fruits were removed from the cold storage and during the simulated marketing period when compared to all the pre-storage treatments (Figure 2D).
Flesh firmness, total phenolic compounds, and antioxidant capacity
The firmness decreased with the time of storage in all treatments. In addition, in the simulated marketing period, lower values of firmness were found in all the treatments with combined pre-storage and modified atmosphere when compared to the control (Figure 3A).
The treatments of pre-storage for 48h in combination with LDPE packaging of 60 or 80 µm presented the highest contents of total phenolic compounds on the day the fruits were removed from cold storage. After the simulated marketing period, the contents of the total phenolic compounds of all the treatments submitted to pre-storage and LPDE packaging were higher than the control treatment. In addition, the treatments submitted to pre-storage of 48h presented higher contents of total phenolic compounds in relation to the treatments with only 24h, regardless of the thickness of the film used in the package (Figure 3B).
The antioxidant capacity measured by FRAP was higher for the treatment of pre-storage for 48h + LDPE 80 µm when the fruits were removed from cold storage. In the simulated marketing period, all the treatments of pre-storage in association with LDPE packaging showed a higher antioxidant capacity compared to the control treatment (Figure 3C).
Firmness (A), total phenolics compounds (B) and antioxidant capacity (FRAP) (C) in ‘Douradão’ peaches submitted to pre-storage (24 or 48h at 25ºC), packaged under modified atmosphere (LDPE 60 or 80 µm) and stored for 30 days at 0°C and ± 5% RH, followed by a simulated marketing period of 3 days at 25°C and 60 ± 5% RH. The means followed by the same letter do not differ between treatments by the Tukey’s test (p ≥ 0.05).
Discussion
Gas composition
Reductions in the concentrations of CO2 and O2 were observed for all the treatments of pre-storage and packaging. The pre-storage for 24 and 48h associated with LDPE packaging of 80 µm presented the largest differences in the concentrations of CO2 and O2 when compared to the control. As expected, a thicker LDPE packaging presented a lower permeability to gasses: 80 µm versus 60 µm. According to Kartal, Aday, and Caner (2012Kartal, S., Aday, M. S., & Caner, C. (2012). Use of microperforated films and oxygen scavengers to maintain storage stability of fresh strawberries. Postharvest Biology and Technology, 71, 32-40. DOI: org/10.1016/j.postharvbio.2012.04.009
https://doi.org/10.1016/j.postharvbio.20...
), the use of passive MAP promotes a reduction in O2 levels and an increase in CO2 levels inside the packaging. This occurs due to the mass and respiration rate of the fruits, the surface area and the permeability rate of the packaging material. In this study, the use of MAP did not cause fermentation of the fruits. The levels of O2 and CO2 achieved inside the package were under the limits of tolerance to prevent anaerobic fermentation in peaches, which is less than 2% for O2 and up to 10 to 25% for CO2 depending on each peach cultivar (Zagory & Kader, 1988Zagory, D., & Kader, A. A. (1988). Modified atmosphere packaging of fresh produce. Food Technology, 42(9), 76-77. DOI: 10.1080/10408398909527506
https://doi.org/10.1080/1040839890952750...
; Fernandez-Trujillo, Salmeron, & Artés, 1997Fernández-Trujillo, J. P., Salmerón, M. C., & Artés, F. (1997). Effect of intermittent warming and modified atmosphere packaging on fungal growth in peaches. Plant Disease Journal, 81(8), 880-884. DOI: 10.1094/PDIS.1997.81.8.880
https://doi.org/10.1094/PDIS.1997.81.8.8...
). Retamales, Cooper, Streif, and Kania (1992Retamales, J., Cooper, T., Streif, J., & Kania, J. C. (1992). Preventing cold storage disorders in nectarines. Journal of Horticultural Science, 67(5), 619-626. DOI: abs/10.1080/00221589.1992.11516291
https://doi.org/10.1080/00221589.1992.11...
) achieved great results in maintaining the quality of refrigerated nectarine using 20% CO2 without the presence of any deleterious effects in the fruit. Zoffoli, Rodriguez, Aldunce, and Crisosto (1997Zoffoli, J. P., Rodriguez, J., Aldunce, P., & Crisosto, C. H. (1997). Development of high concentration carbon dioxide modified atmosphere packaging systems to maintain peach quality. In A. A. Kader (Ed.), 7th
International Controlled Atmosphere Research Conference (p. 132-138). Davis, US: University of California .), after studying different packages for cold stored peaches, observed that the levels of CO2 varied from 10 to 25% and the levels of O2 varied from 1.5 to 10%. They also noted that greater reductions in chilling injuries occurred in packages with high levels of CO2. Lee (2014Lee, E. J. (2014). Chilling Injury and Phytochemical Composition of Peach Fruits as Affected by High Carbon Dioxide Treatment before Cold Storage. Horticulture, Environment, and Biotechnology, 55(3), 190-195. DOI: 10.1007/s13580-014-0187-2
https://doi.org/10.1007/s13580-014-0187-...
) also observed the quality maintenance and reduction of chilling injuries in ‘Mibaekdo’ peaches submitted to a pretreatment with 30% CO2.
Woolliness index, Internal browning index, PME, and PG activity
The effect of pre-storage combined with LDPE packaging on the cold storage of ‘Douradão’ peaches was shown to be effective in reducing woolliness when the fruits were cold stored for 30 days at 0ºC followed by 3 days at room temperature. The pre-storage also reduced the woolliness in different varieties of peaches and nectarines when the fruits were exposed to room temperature before the cold storage (Zhou et al., 2000Zhou, H. W., Lurie, S., Lers, A., Khatchitski, A., Sonego, L., & Ben-Arie, R. (2000). Pre-storage and controlled atmosphere storage of nectarines: two strategies to prevent woolliness. Postharvest Biology and Technology, 18(2), 133-141. DOI: doi.org/10.1016/S0925-5214(99)00072-1
https://doi.org/10.1016/S0925-5214(99)00...
; Sasaki at al., 2010Sasaki, F. F., Cerqueira, T. S., Sestari, I., Del Aguila, J. S., & Kluge, R. A. (2010). Woolliness control and pectin solubilization of ‘Douradao’ peach after heat shock treatment. Acta Horticulturae, 877, 539-542. DOI: 10.17660/AcataHortic.2010.877.6
https://doi.org/10.17660/AcataHortic.201...
; Neves, Campos, Prill, & Roberto, 2013Neves, L. C., Campos, J. C., Prill, M. A. S., & Roberto, S. R. (2013). Woolliness and leatheriness in late peach cultivars submitted to both pre-storage and to cold storage. Acta Scientiarum. Agronomy, 35(3), 363-369. DOI: 10.4025/actasciagron.v35i3.13347
https://doi.org/10.4025/actasciagron.v35...
).
Choi and Lee (1997Choi, J. H., & Lee, S. K. (1997). Effect of MA storage on wooliness of ‘Yumyeong’ peaches. In A. A. Kader (Ed.), 7th
International Controlled Atmosphere Research Conference (p. 132-138). Davis, US: University of California. DOI: 10.1016/j.postharvbio.2010.05.010
https://doi.org/10.1016/j.postharvbio.20...
) observed a significant effect on the reduction of woolliness using a passive modified atmosphere in ‘Yumyeong’ peaches when the fruits were compared with unpackaged ones. Santana, Benedetti, Sigrist, and Sarantopóulos (2010Santana, L. R. R., Benedetti, B. C., Sigrist, J. M. M., & Sarantopóulos, C. I. G. L. (2010). Modified atmosphere packaging extending the storage life of ‘Douradão’ peach. Revista Brasileira de Fruticultura, 32(4), 1009-1017. DOI: doi.org/10.1590/S0100-29452011005000006
https://doi.org/10.1590/S0100-2945201100...
) and Santana, Benedetti, Sigrist, and Sato (2011Santana, L. R. R., Benedetti, B. C., Sigrist, J. M. M., & Sato, H. H. (2011). Effects of modified atmosphere packaging on ripening of ‘Douradão’. Revista Brasileira de Fruticultura, 33(4), 1084-1094. DOI: dx.doi.org/10.1590/S0100-29452011000400006
https://doi.org/10.1590/S0100-2945201100...
) observed a significant reduction in woolliness with the use of active modified atmosphere and LDPE packaging (with values of 1% O2 and 3% CO2).
The internal browning presented the highest values for the control treatment in the simulated marketing period. This result is probably due to the deterioration and senescence of tissues after a long period under refrigeration, which leads to changes in cell membrane permeability. These changes in permeability modify both the cellular metabolism and the normal activity of membrane enzymes, especially the oxidative enzymes. The modification of flesh color is due to the damages caused in the cells by toxic intermediary products that accumulate during cold storage and to the oxidation of phenolic compounds by the polyphenoloxidase enzyme (Macheix, Fleuriet, & Billot 1990Macheix, J. J., Fleuriet, A., & Billot, J. (1990). Fruit phenolics. Boca Raton, FL: CRC Press.; Espin, Morales, Vàron, Tudela, & Garcia, 1997Espin, J. C., Morales, M., Vàron, R., Tudela, J., & Garcia, C. F. (1997). Monophenolase activity of poliphenol oxidase from Blanquila pear. Phytochemistry, 44(1), 17-22. DOI: 10.4025/actasciagron.v34i3.13346
https://doi.org/10.4025/actasciagron.v34...
).
The exposition of stone fruits to pre-storage before cold storage promotes the stability of cellular tissues and reduces the activity of oxidative enzymes such as polyphenoloxidase (Neves, Campos, Prill, & Roberto, 2013Neves, L. C., Campos, J. C., Prill, M. A. S., & Roberto, S. R. (2013). Woolliness and leatheriness in late peach cultivars submitted to both pre-storage and to cold storage. Acta Scientiarum. Agronomy, 35(3), 363-369. DOI: 10.4025/actasciagron.v35i3.13347
https://doi.org/10.4025/actasciagron.v35...
). This enables the peach to resist a longer period of exposition to low storage temperatures without presenting chilling injuries such as internal browning (Crisosto, Garner, Andris, & Day 2004Crisosto, C. H., Garner, D., Andris, H. L., & Day, K. R. (2004). Controlled delayed cooling extends peach market life. HortTechnoIogy, 14(1), 99-104.). The results found in this study corroborate the findings of Jin, Zheng, Tang, Rui, and Wang (2009Jin, P., Zheng, Y., Tang, S., Rui, H., & Wang, C. Y. (2009). A combination of hot air and methyl jasmonate vapor treatment alleviates chilling injury of peach fruit. Postharvest Biology and Technology, 52(1), 24-29. DOI: org/10.1016/j.postharvbio.2008.09.011
https://doi.org/10.1016/j.postharvbio.20...
), who observed a reduction in internal browning in ‘Baifeng’ peaches after heat treatment at 38°C for 12 hours. Neves, Tosin, Silva, Vasconcelos, and Roberto (2012Neves, L. C., Tosin, J. M., Silva, S., Vasconcelos, L. L., & Roberto, S. R. (2012). Inhibition of pulp browning and quality maintenance of late peach cultivars. Acta Scientiarum. Agronomy, 34(3), 299-308. DOI: 10.4025/actasciagron.v34i3.13346
https://doi.org/10.4025/actasciagron.v34...
) reported the efficiency of pre-storage for 24 or 48 hours in preventing internal browning in three peach cultivars that had been stored for 28 days under cold storage. Retamales, Cooper, Streif, and Kania (1992Retamales, J., Cooper, T., Streif, J., & Kania, J. C. (1992). Preventing cold storage disorders in nectarines. Journal of Horticultural Science, 67(5), 619-626. DOI: abs/10.1080/00221589.1992.11516291
https://doi.org/10.1080/00221589.1992.11...
) found that the application of pre-storage for 48 hours associated with a modified atmosphere with a high CO2 concentration (20%) significantly reduced internal browning.
The increase in PME activity and the decrease in PG activity observed in this study are related to the high index of woolliness in the control fruits in the simulated marketing period. The index of woolliness in stone fruits is considered to occur due to an imbalance between the activities of the pectinolytic enzymes PME and PG during cold storage (Ben-Arie & Sonego, 1980Ben-Arie, R., & Sonego, L. (1980). Pectolytic enzyme activity involved in woolly breakdown of stored peaches. Phytochemistry, 19(2), 2553-2555. DOI: doi.org/10.1016/S0031-9422(00)83917-5
https://doi.org/10.1016/S0031-9422(00)83...
). The low activity of PG combined with the de-esterification of pectin molecules from the action of PME results in the accumulation of pectins with high molecular weight and low degrees of esterification that bind calcium ions, which is what generates the pectates. These compounds remove the free water from the cells and form a pectic gel, thus promoting the loss of juiciness in fruit and a farinaceous aspect (Obenland, Crisosto, & Rose, 2003Obenland, D. M., Crisosto, C. H., & Rose, J. K. C. (2003). Expansin protein levels decline with the development of mealiness in peaches. Postharvest Biology and Technology, 29(1), 11-18. DOI: doi.org/10.1016/S0925-5214(02)00245-4
https://doi.org/10.1016/S0925-5214(02)00...
). Brummell et al. (2004Brummell, D. A., Dal Cin, V., Lurie, S., Crisosto, C. H., & Labavitch, J. M. (2004). Cell wall metabolism during the development of chilling injury in cold stored peach fruit: association of mealiness with arrested disassembly of cell wall pectins. Journal of Experimental Botany, 55(245), 2041-2052. DOI: 10.1093/jxb/erh228
https://doi.org/10.1093/jxb/erh228...
) observed that PG synthesis is regulated by ethylene. The low ethylene production during cooling reduces the synthesis and activity of the enzyme and causes chilling injuries such as woolliness in stone fruits. Other authors found similar results to this study and reported an increase in PME activity at the same time as the increase in woolliness (Artés, Cano, & Fernandéz-Trujillo, 1996Artés, F., Cano, A., & Fernandéz-Trujillo, J. P. (1996). Pectolytic enzyme activity during intermittent warning storage of peaches. Journal of Food Science, 61(2), 311-321. DOI: 10.1111/j.1365-2621.1996.tb14183.x
https://doi.org/10.1111/j.1365-2621.1996...
). Choi and Lee (1997Choi, J. H., & Lee, S. K. (1997). Effect of MA storage on wooliness of ‘Yumyeong’ peaches. In A. A. Kader (Ed.), 7th
International Controlled Atmosphere Research Conference (p. 132-138). Davis, US: University of California. DOI: 10.1016/j.postharvbio.2010.05.010
https://doi.org/10.1016/j.postharvbio.20...
) and Santana et al. (2011Santana, L. R. R., Benedetti, B. C., Sigrist, J. M. M., & Sato, H. H. (2011). Effects of modified atmosphere packaging on ripening of ‘Douradão’. Revista Brasileira de Fruticultura, 33(4), 1084-1094. DOI: dx.doi.org/10.1590/S0100-29452011000400006
https://doi.org/10.1590/S0100-2945201100...
) also noticed low PG activity and a high index of woolliness in peaches without packaging when compared to packaged fruits stored for 4 weeks at 0ºC. Zhou et al. (2000Zhou, H. W., Lurie, S., Lers, A., Khatchitski, A., Sonego, L., & Ben-Arie, R. (2000). Pre-storage and controlled atmosphere storage of nectarines: two strategies to prevent woolliness. Postharvest Biology and Technology, 18(2), 133-141. DOI: doi.org/10.1016/S0925-5214(99)00072-1
https://doi.org/10.1016/S0925-5214(99)00...
) and Brummell et al. (2004) also found a lower PG activity in woolly peaches and nectarines after cold storage.
Flesh firmness, total phenolic compounds, and antioxidant capacity.
The decrease in firmness found during the simulated marketing period in treatments that combined pre-storage and LDPE were expected since the exposure to higher temperatures before the cold storage accelerates the metabolism of stone fruits and affects the firmness and all the other aspects related to ripening (Vitti, Kluge, Jacomino, & Lima, 2007Vitti, D. C. C., Kluge, R. A., Jacomino, A. P., & Lima, G. P. P. (2007). Tratamento térmico para controle da lanosidade em pêssegos ‘Dourado-2’ refrigerados. Pesquisa Agropecuária Brasileira, 42(12), 1705-1713. DOI: org/10.1590/S0100-204X200700120000
https://doi.org/10.1590/S0100-204X200700...
). Camargo, Lima, Scalon, and Siqueira (2000Camargo, Y. R., Lima, L. C. O., Scalon, S. P. Q., & Siqueira, A. C. (2000). Efeito do cálcio sobre o amadurecimento de morangos (Fragaria ananassa Duch.) cv. campineiro. Ciência e Agrotecnologia, 24(4), 968-972.) reported that the loss of flesh firmness during ripening is due to the action of enzymes from the wall such as PG. The reduction in the firmness of the fruits submitted to the pre-storage and MAP may be related to the higher PG activity (Figure 2D). According to Evangelista, Chitarra, and Chitarra (2000Evangelista, R. M., Chitarra, A. B., & Chitarra, M. I. F. (2000). Influência da aplicação pré-colheita de cálcio na textura e na atividade das enzimas PG, PME e β-galactosidase de mangas ‘Tommy Atkins’ armazenadas sob refrigeração. Ciência e Agrotecnologia, 24(Edição Especial), 174-181.), PG catalyzes the hydrolysis of the β-1,4 bonds between the galacturonic acid residues in the pectin chain and consequently promotes the reduction of firmness in the fruits. The lower firmness found in the treated fruit during the simulated marketing period may also be related to the higher juiciness and lack of woolliness in these fruits (Figure 2A). Other authors observed similar results in peaches and nectarines submitted to pre-storage and concluded that the decline in firmness is related to an increase in juiciness and to a decrease in woolliness (Infante et al., 2009Infante, R., Meneses, C., & Crisosto, C. H. (2009). Preconditioning treatment maintains taste characteristic perception of ripe ‘September Sun’ peach following cold storage. Journal of Food Science and Technology, 44(5), 1011-1016. DOI: 10.1111/j.1365-2621.2008.01864.x
https://doi.org/10.1111/j.1365-2621.2008...
; Sasaki et al., 2010Sasaki, F. F., Cerqueira, T. S., Sestari, I., Del Aguila, J. S., & Kluge, R. A. (2010). Woolliness control and pectin solubilization of ‘Douradao’ peach after heat shock treatment. Acta Horticulturae, 877, 539-542. DOI: 10.17660/AcataHortic.2010.877.6
https://doi.org/10.17660/AcataHortic.201...
). Murray et al. (2007Murray, R., Lucangeli, C., Polenta, G., & Budde, C. (2007). Combined pre-torage heat treatment and controlled atmosphere storage reduce internal breakdown of ‘Flavorcred’ peach. Postharvest Biology and Technology, 44(2), 116-121. DOI: org/10.1016/j.postharvbio.2006.11.013
https://doi.org/10.1016/j.postharvbio.20...
) reported that the combination of pre-storage and controlled atmosphere reduced the fruit firmness to values of approximately 6 N, as well as the woolliness, thus maintaining the fruit quality. Cano-Salazar, López, Crisosto, and Echeverría (2013Cano-Salazar, J., López, M. L., Crisosto, C. H., & Echeverría, G. (2013). Volatile compound emissions and sensory attributes of ‘Big Top’ nectarine and ‘Early Rich’ peach fruit in response to a pre-storage treatment before cold storage and subsequent shelf-life. Postharvest Biology and Technology, 76, 152-162. DOI: org/10.1016/j.postharvbio.2012.10.001
https://doi.org/10.1016/j.postharvbio.20...
) also reported a reduction in firmness in ‘Early Rich’ peaches to 4.5 N after exposure to room temperature (20ºC) for 36 hours, whereas the untreated fruits presented values of 14.9 N for firmness.
The control treatment presented a lower content of phenolic compounds. Genetic and environmental factors are determining factors in the content of phenolic compounds (Robards, Prenzler, Tucker, Swatsitang, & Glover, 1999Robarts, K., Prenzler, P. D., Tucker, G., Swatsitang, P., Glover, W. (1999). Phenolic compounds and their role in oxidative processes in fruits. Food Chemistry, 66(4), 401-436. DOI: 10.1016/s0308-8146(99)00093-x
https://doi.org/10.1016/s0308-8146(99)00...
). However, oxidative reactions that occur during processing or storage can also modify these compounds because the increase in their content is related to responses to stress conditions. In general, oxidative regulation is a process that promotes dynamic balance in the system and produces and scavenges reactive oxygen species (ROS). According to Toivonen (2004Toivonen, P. M. A. (2004). Postharvest storage procedures and oxidative stress. HortScience, 39(5), 938-942.), the tissue initially responds to the stress, leading to the production of ROS and consequently activating the system responsible for the production of antioxidant compounds. Nevertheless, if there is a high level of stress, the tissue may not be able to maintain the necessary balance and causes damage to the cells. Phenolic compounds are one of the main mechanisms for the protection of cellular tissue (Mittler 2002Mittler, R. (2002). Oxidative stress, antioxidants and stress tolerance. Trends in Plant Science, 7(9), 405-410. DOI: org/10.1016/S1360-1385(02)02312-9
https://doi.org/10.1016/S1360-1385(02)02...
) and act directly as antioxidant agents or substrates for superoxide (Robards et al., 1999Robarts, K., Prenzler, P. D., Tucker, G., Swatsitang, P., Glover, W. (1999). Phenolic compounds and their role in oxidative processes in fruits. Food Chemistry, 66(4), 401-436. DOI: 10.1016/s0308-8146(99)00093-x
https://doi.org/10.1016/s0308-8146(99)00...
). The internal browning is generally related to the increase in the activities of polyphenoloxidade and peroxidase that can lead to the oxidation of phenolic compounds to quinones or to quinine compounds, resulting in brown polymerized pigments (Lill, O’Donaghue, & King, 1989Lill, R. E., O’Donaghue, E. M., & King, G. A. (1989). Postharvest physiology of peaches and nectarines. Horticultural Reviews, 11, 413-452. DOI: ). Jin et al. (2009Jin, P., Zheng, Y., Tang, S., Rui, H., & Wang, C. Y. (2009). A combination of hot air and methyl jasmonate vapor treatment alleviates chilling injury of peach fruit. Postharvest Biology and Technology, 52(1), 24-29. DOI: org/10.1016/j.postharvbio.2008.09.011
https://doi.org/10.1016/j.postharvbio.20...
) also related the high index of internal browning with the low content of phenolic compounds in peaches when they were exposed to room temperature after refrigeration.
According to Malakou and Nanos (2005Malakou, A., & Nanos, G. D. (2005). A combination of hot water treatment and modified atmosphere packaging maintains quality of advanced maturity ‘Caldesi 2000’ nectarines and ‘Royal Glory’ peaches. Postharvest Biology and Technology, 38(2), 106-114. DOI: org/10.1016/j.postharvbio.2005.06.003
https://doi.org/10.1016/j.postharvbio.20...
), the exposure of fruits to high temperatures before cold storage leads to an increase in the content of phenolic compounds and is a way to protect the vegetal tissue from the stress caused by the temperature. The same authors found an increase in the content of phenolic compounds in ‘Royal Glory’ peaches during the simulated marketing period with the application of heat treatment (immersion in hot water) before cold storage. However, Neves et al. (2013Neves, L. C., Campos, J. C., Prill, M. A. S., & Roberto, S. R. (2013). Woolliness and leatheriness in late peach cultivars submitted to both pre-storage and to cold storage. Acta Scientiarum. Agronomy, 35(3), 363-369. DOI: 10.4025/actasciagron.v35i3.13347
https://doi.org/10.4025/actasciagron.v35...
) did not find any difference between the treatments both with and without the application of pre-storage in peaches kept under refrigeration for 28 days.
The use of pre-storage associated with LDPE packaging promoted a higher antioxidant capacity in ‘Douradão’ peaches during the simulated marketing period. This finding can be related to the lower internal browning index and the higher content of total phenolic compounds observed in these treatments over the same period. Studies report that the antioxidant capacity of phenolic compounds is due to the reducing power of the aromatic hydroxyl groups that reduce reactive free radicals, such as “singlet” oxygen, or decomposed peroxides, which produce the radical phenoxyl that is less reactive (Campos, Martino, Sabarense, & Pinheiro-Sant’Ana, 2008Campos, F. M., Martino, H. S. D., Sabarense, C. M., & Pinheiro-Sant’Ana, H. M. (2008). Estabilidade de compostos antioxidantes em hortaliças processadas: uma revisão. Alimentos e Nutrição, 19(4), 481-490.; Silva, Costa, Santana, & Koblitz, 2010Silva, M. L. C., Costa, R. S., Santana, A. S., & Koblitz, M. G. B. (2010). Compostos fenólicos, carotenóides e atividade antioxidante em produtos vegetais. Semina: Ciências Agrárias, 31(3), 669-682.). These results are in accordance with those found by Gil, Tomas-Barberan, Hess-Pierce, and Kader (2002Gil, M. I., Tomas-Barberan, F. A., Hess-Pierce, B., & Kader, A. A. (2002). Antioxidant capacities, phenolic compounds, carotenoids, and vitamin C contents of nectarine, peach, and plum cultivars from California. Journal of Agricultural and Food Chemitry, 50(17), 4976-4982. DOI: 10.1021/jf020136b
https://doi.org/10.1021/jf020136b...
) and Mokrani et al. (2016Mokrani, A., Krisa, S., Cluzet, S., Da Costa, G., Temsamani, H., Renouf, E., ... Richard, T. (2016). Phenolic contents and bioactive potential of peach fruit extracts. Food Chemistry, 202, 212-220. DOI: 10.1016/j.foodchem.2015.12.026
https://doi.org/10.1016/j.foodchem.2015....
), who observed a high correlation between the antioxidant capacity and the content of total phenolic compounds. According to these authors, phenolic compounds are mainly responsible for the antioxidant capacity in peaches.
Conclusion
The combination of pre-storage for 24 and 48 hours and LDPE packaging of 60 and 80 µm is a strategy to prevent or reduce chilling injuries in ‘Douradão’ peaches while maintaining the quality of fruits during cold storage for 30 days. The LDPE packaging of 80 µm associated with pre-storage for both 24 and 48 hours showed the best results in the reduction in woolliness. All the treatments that used a combination of pre-storage and LDPE were efficient in inhibiting internal browning, highest content of phenolic compounds and antioxidant capacity during the simulated marketing period the fruit.
Acknowledgements
The authors would like to thank the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for granting the scholarships. They would also like to thank the Universidad Politécnica de Cartagena (UPCT) for the use of some equipment.
References
- Akbudak, B., & Eris, A. (2004). Physical and chemical changes in peaches and nectarines during the modified atmosphere storage. Food Control, 15(4), 307-313. DOI: 10.1016/S0956-7135(03)00082-3
» https://doi.org/10.1016/S0956-7135(03)00082-3 - Artés, F., Cano, A., & Fernandéz-Trujillo, J. P. (1996). Pectolytic enzyme activity during intermittent warning storage of peaches. Journal of Food Science, 61(2), 311-321. DOI: 10.1111/j.1365-2621.1996.tb14183.x
» https://doi.org/10.1111/j.1365-2621.1996.tb14183.x - Beaudry, R. M. (1999). Effect of O2 and CO2 partial pressure on selected phenomena affecting fruit and vegetable quality. Postharvest Biology and Technology, 15(3), 293-303. DOI: 10.1016/S0925-5214(98)00092-1
» https://doi.org/10.1016/S0925-5214(98)00092-1 - Ben-Arie, R., & Sonego, L. (1980). Pectolytic enzyme activity involved in woolly breakdown of stored peaches. Phytochemistry, 19(2), 2553-2555. DOI: doi.org/10.1016/S0031-9422(00)83917-5
» https://doi.org/10.1016/S0031-9422(00)83917-5 - Benzie, I. F. F., & Strain, J. J. (1996). Ferric reducing ability of plasma (FRAP) as a measure of antioxidant power: The FRAP assay. Analytical Biochemitry, 239(1), 70-76. DOI: 0.1006/ABIO.1996.0292
» https://doi.org/0.1006/ABIO.1996.0292 - Bron, I. U., Jacomino, P. A., & Appezzato-da-Glória, B. (2002). Alterações anatômicas e físico-químicas associadas ao armazenamento refrigerado de pêssegos ‘Aurora-1’ e ‘Dourado-2’. Pesquisa Agropecuária Brasileira, 37(10), 1349-1358. DOI: 10.1590/S0100-204X2002001000001
» https://doi.org/10.1590/S0100-204X2002001000001 - Brummell, D. A., Dal Cin, V., Lurie, S., Crisosto, C. H., & Labavitch, J. M. (2004). Cell wall metabolism during the development of chilling injury in cold stored peach fruit: association of mealiness with arrested disassembly of cell wall pectins. Journal of Experimental Botany, 55(245), 2041-2052. DOI: 10.1093/jxb/erh228
» https://doi.org/10.1093/jxb/erh228 - Camargo, Y. R., Lima, L. C. O., Scalon, S. P. Q., & Siqueira, A. C. (2000). Efeito do cálcio sobre o amadurecimento de morangos (Fragaria ananassa Duch.) cv. campineiro. Ciência e Agrotecnologia, 24(4), 968-972.
- Campos, F. M., Martino, H. S. D., Sabarense, C. M., & Pinheiro-Sant’Ana, H. M. (2008). Estabilidade de compostos antioxidantes em hortaliças processadas: uma revisão. Alimentos e Nutrição, 19(4), 481-490.
- Cano-Salazar, J., López, M. L., Crisosto, C. H., & Echeverría, G. (2013). Volatile compound emissions and sensory attributes of ‘Big Top’ nectarine and ‘Early Rich’ peach fruit in response to a pre-storage treatment before cold storage and subsequent shelf-life. Postharvest Biology and Technology, 76, 152-162. DOI: org/10.1016/j.postharvbio.2012.10.001
» https://doi.org/10.1016/j.postharvbio.2012.10.001 - Cantín, C. M., Crisosto, C. H., Ogundiwin, E. A., Gradziel, T., Torrents, J., Moreno, M. A., & Gogorcena, Y. (2010). Chilling injury susceptibility in an intra-specific peach [Prunus persica (L.) Batsch] progeny. Postharvest Biology and Technology, 58(2), 79-87. DOI: 10.1016/j.postharvbio.2010.06.002
» https://doi.org/10.1016/j.postharvbio.2010.06.002 - Cao, S., Hu, Z., Zheng, Y., & Lu, B. (2010). Synergistic effect of heat treatment and salicylic acid on alleviating internal browning in cold-stored peach fruit. Postharvest Biology and Technology, 58(2), 93-97. DOI: 10.1016/j.postharvbio.2010.05.010
» https://doi.org/10.1016/j.postharvbio.2010.05.010 - Choi, J. H., & Lee, S. K. (1997). Effect of MA storage on wooliness of ‘Yumyeong’ peaches. In A. A. Kader (Ed.), 7th International Controlled Atmosphere Research Conference (p. 132-138). Davis, US: University of California. DOI: 10.1016/j.postharvbio.2010.05.010
» https://doi.org/10.1016/j.postharvbio.2010.05.010 - Crisosto, C. H., Garner, D., Andris, H. L., & Day, K. R. (2004). Controlled delayed cooling extends peach market life. HortTechnoIogy, 14(1), 99-104.
- Denès, J. M., Baron, A., & Drilleau, J. F. (2000). Purification, properties and heat inactivation of pectin methylesterase from apple (cv. Golden Delicious). Journal of the Science of Food and Agriculture, 80(10), 1503-1509. DOI: 10.1002/1097-0010(200008)80:10<1503:AID-JSFA676>3.0.CO;2-U
» https://doi.org/10.1002/1097-0010(200008)80:10<1503:AID-JSFA676>3.0.CO;2-U - Fernández-Trujillo, J. P., Salmerón, M. C., & Artés, F. (1997). Effect of intermittent warming and modified atmosphere packaging on fungal growth in peaches. Plant Disease Journal, 81(8), 880-884. DOI: 10.1094/PDIS.1997.81.8.880
» https://doi.org/10.1094/PDIS.1997.81.8.880 - Espin, J. C., Morales, M., Vàron, R., Tudela, J., & Garcia, C. F. (1997). Monophenolase activity of poliphenol oxidase from Blanquila pear. Phytochemistry, 44(1), 17-22. DOI: 10.4025/actasciagron.v34i3.13346
» https://doi.org/10.4025/actasciagron.v34i3.13346 - Evangelista, R. M., Chitarra, A. B., & Chitarra, M. I. F. (2000). Influência da aplicação pré-colheita de cálcio na textura e na atividade das enzimas PG, PME e β-galactosidase de mangas ‘Tommy Atkins’ armazenadas sob refrigeração. Ciência e Agrotecnologia, 24(Edição Especial), 174-181.
- Fruk, G., Cmelik, Z., Jemric, T., Hribar, J., & Vidrih, R. (2014). Pectin role in woolliness development in peaches and nectarines: a review. Scientia Horticulturae, 180, 1-5. DOI: doi.org/10.1016/j.scienta.2014.09.042
» https://doi.org/10.1016/j.scienta.2014.09.042 - Gil, M. I., Tomas-Barberan, F. A., Hess-Pierce, B., & Kader, A. A. (2002). Antioxidant capacities, phenolic compounds, carotenoids, and vitamin C contents of nectarine, peach, and plum cultivars from California. Journal of Agricultural and Food Chemitry, 50(17), 4976-4982. DOI: 10.1021/jf020136b
» https://doi.org/10.1021/jf020136b - Gross, K. C. (1982). A rapid and sensitive spectrophotometric method for assaying poligalacturonase using 2-cyanoacetamide. HortScience, 17(6), 933-934.
- Infante, R., Meneses, C., & Crisosto, C. H. (2009). Preconditioning treatment maintains taste characteristic perception of ripe ‘September Sun’ peach following cold storage. Journal of Food Science and Technology, 44(5), 1011-1016. DOI: 10.1111/j.1365-2621.2008.01864.x
» https://doi.org/10.1111/j.1365-2621.2008.01864.x - Jin, P., Zheng, Y., Tang, S., Rui, H., & Wang, C. Y. (2009). A combination of hot air and methyl jasmonate vapor treatment alleviates chilling injury of peach fruit. Postharvest Biology and Technology, 52(1), 24-29. DOI: org/10.1016/j.postharvbio.2008.09.011
» https://doi.org/10.1016/j.postharvbio.2008.09.011 - Ju, Z., Duan, Y., Ju, Z., & Guo, A. (2001). Different responses of ‘Snow Giant’ and ‘Elegant Lady’ peaches to fruit maturity and storage temperature. Journal of Horticultural Science and Biotechnology, 76(5), 575-580.
- Kartal, S., Aday, M. S., & Caner, C. (2012). Use of microperforated films and oxygen scavengers to maintain storage stability of fresh strawberries. Postharvest Biology and Technology, 71, 32-40. DOI: org/10.1016/j.postharvbio.2012.04.009
» https://doi.org/10.1016/j.postharvbio.2012.04.009 - Lee, E. J. (2014). Chilling Injury and Phytochemical Composition of Peach Fruits as Affected by High Carbon Dioxide Treatment before Cold Storage. Horticulture, Environment, and Biotechnology, 55(3), 190-195. DOI: 10.1007/s13580-014-0187-2
» https://doi.org/10.1007/s13580-014-0187-2 - Lill, R. E., O’Donaghue, E. M., & King, G. A. (1989). Postharvest physiology of peaches and nectarines. Horticultural Reviews, 11, 413-452. DOI:
- Lurie, S., & Crisosto, C. H. (2005). Chilling injury in peach and nectarine: review. Postharvest Biology and Technology, 37(3), 195-208. DOI: 10.1016/j.postharvbio.2005.04.012
» https://doi.org/10.1016/j.postharvbio.2005.04.012 - Macheix, J. J., Fleuriet, A., & Billot, J. (1990). Fruit phenolics Boca Raton, FL: CRC Press.
- Malakou, A., & Nanos, G. D. (2005). A combination of hot water treatment and modified atmosphere packaging maintains quality of advanced maturity ‘Caldesi 2000’ nectarines and ‘Royal Glory’ peaches. Postharvest Biology and Technology, 38(2), 106-114. DOI: org/10.1016/j.postharvbio.2005.06.003
» https://doi.org/10.1016/j.postharvbio.2005.06.003 - Mittler, R. (2002). Oxidative stress, antioxidants and stress tolerance. Trends in Plant Science, 7(9), 405-410. DOI: org/10.1016/S1360-1385(02)02312-9
» https://doi.org/10.1016/S1360-1385(02)02312-9 - Mokrani, A., Krisa, S., Cluzet, S., Da Costa, G., Temsamani, H., Renouf, E., ... Richard, T. (2016). Phenolic contents and bioactive potential of peach fruit extracts. Food Chemistry, 202, 212-220. DOI: 10.1016/j.foodchem.2015.12.026
» https://doi.org/10.1016/j.foodchem.2015.12.026 - Murray, R., Lucangeli, C., Polenta, G., & Budde, C. (2007). Combined pre-torage heat treatment and controlled atmosphere storage reduce internal breakdown of ‘Flavorcred’ peach. Postharvest Biology and Technology, 44(2), 116-121. DOI: org/10.1016/j.postharvbio.2006.11.013
» https://doi.org/10.1016/j.postharvbio.2006.11.013 - Nava, G. A., & Brackmann, A. (2002). Armazenamento de pêssegos (Prunus persica (L.) Batsch), cv. Chiripá, em atmosfera controlada. Revista Brasileira de Fruticultura, 24(2), 328-332. DOI: 10.1590/S0100-29452002000200011
» https://doi.org/10.1590/S0100-29452002000200011 - Neves, L. C., Tosin, J. M., Silva, S., Vasconcelos, L. L., & Roberto, S. R. (2012). Inhibition of pulp browning and quality maintenance of late peach cultivars. Acta Scientiarum. Agronomy, 34(3), 299-308. DOI: 10.4025/actasciagron.v34i3.13346
» https://doi.org/10.4025/actasciagron.v34i3.13346 - Neves, L. C., Campos, J. C., Prill, M. A. S., & Roberto, S. R. (2013). Woolliness and leatheriness in late peach cultivars submitted to both pre-storage and to cold storage. Acta Scientiarum. Agronomy, 35(3), 363-369. DOI: 10.4025/actasciagron.v35i3.13347
» https://doi.org/10.4025/actasciagron.v35i3.13347 - Obenland, D. M., Crisosto, C. H., & Rose, J. K. C. (2003). Expansin protein levels decline with the development of mealiness in peaches. Postharvest Biology and Technology, 29(1), 11-18. DOI: doi.org/10.1016/S0925-5214(02)00245-4
» https://doi.org/10.1016/S0925-5214(02)00245-4 - Ratner, A., Goren, R., & Monseline, S. P. (1969). Activity of pectinesterase and cellulase in the abscission zone of citrus leaf explants. Plant Physiology, 44(12), 1717-1723. DOI: https://doi.org/10.1104/pp.44.12.1717
» https://doi.org/10.1104/pp.44.12.1717 - Retamales, J., Cooper, T., Streif, J., & Kania, J. C. (1992). Preventing cold storage disorders in nectarines. Journal of Horticultural Science, 67(5), 619-626. DOI: abs/10.1080/00221589.1992.11516291
» https://doi.org/10.1080/00221589.1992.11516291 - Robarts, K., Prenzler, P. D., Tucker, G., Swatsitang, P., Glover, W. (1999). Phenolic compounds and their role in oxidative processes in fruits. Food Chemistry, 66(4), 401-436. DOI: 10.1016/s0308-8146(99)00093-x
» https://doi.org/10.1016/s0308-8146(99)00093-x - Santana, L. R. R., Benedetti, B. C., Sigrist, J. M. M., & Sarantopóulos, C. I. G. L. (2010). Modified atmosphere packaging extending the storage life of ‘Douradão’ peach. Revista Brasileira de Fruticultura, 32(4), 1009-1017. DOI: doi.org/10.1590/S0100-29452011005000006
» https://doi.org/10.1590/S0100-29452011005000006 - Santana, L. R. R., Benedetti, B. C., Sigrist, J. M. M., & Sato, H. H. (2011). Effects of modified atmosphere packaging on ripening of ‘Douradão’. Revista Brasileira de Fruticultura, 33(4), 1084-1094. DOI: dx.doi.org/10.1590/S0100-29452011000400006
» https://doi.org/10.1590/S0100-29452011000400006 - Santos, C. A. A., Castro, J. V., Picoli, A. A., & Rolim, G. S. (2008). Uso de quitosana e embalagem plástica na conservação pós-colheita de pêssegos ‘Douradão’. Revista Brasileira de Fruticultura, 30(1), 88-93. DOI: org/10.1590/S0100-29452008000100017
» https://doi.org/10.1590/S0100-29452008000100017 - Sasaki, F. F., Cerqueira, T. S., Sestari, I., Del Aguila, J. S., & Kluge, R. A. (2010). Woolliness control and pectin solubilization of ‘Douradao’ peach after heat shock treatment. Acta Horticulturae, 877, 539-542. DOI: 10.17660/AcataHortic.2010.877.6
» https://doi.org/10.17660/AcataHortic.2010.877.6 - Silva, M. L. C., Costa, R. S., Santana, A. S., & Koblitz, M. G. B. (2010). Compostos fenólicos, carotenóides e atividade antioxidante em produtos vegetais. Semina: Ciências Agrárias, 31(3), 669-682.
- Swain, T., & Hillis, W. E. (1959). The phenolic constituents of Prunus domestica The quantitative analysis of phenolic constituents. Journal of the Science of Food and Agriculture, 10(1), 63-68. DOI: 10.1002/jsfa.2740100110
» https://doi.org/10.1002/jsfa.2740100110 - Toivonen, P. M. A. (2004). Postharvest storage procedures and oxidative stress. HortScience, 39(5), 938-942.
- Vitti, D. C. C., Kluge, R. A., Jacomino, A. P., & Lima, G. P. P. (2007). Tratamento térmico para controle da lanosidade em pêssegos ‘Dourado-2’ refrigerados. Pesquisa Agropecuária Brasileira, 42(12), 1705-1713. DOI: org/10.1590/S0100-204X200700120000
» https://doi.org/10.1590/S0100-204X200700120000 - Von Mollendorff, L. J., & De Villiers, O. T. (1988). Role of pectolytic enzymes in the development of woolliness in peaches. Journal of Horticultural Science, 63(1), 53-58. DOI: org/10.1080/14620316.1988.11515827
» https://doi.org/10.1080/14620316.1988.11515827 - Zagory, D., & Kader, A. A. (1988). Modified atmosphere packaging of fresh produce. Food Technology, 42(9), 76-77. DOI: 10.1080/10408398909527506
» https://doi.org/10.1080/10408398909527506 - Zhou, H.W., Ben-Arie, R., & Lurie, S. (2000). Pectin esterase, polygalacturonase and gel formation in peach pectin fractions. Phytochemistry, 55(3), 191-195. DOI: org/10.1016/S0031-9422(00)00271-5
» https://doi.org/10.1016/S0031-9422(00)00271-5 - Zhou, H. W., Dong, L., Ben-Arie, R., & Lurie, S. (2001). The role of ethylene in the prevention of chilling injury in nectarines. Journal of Plant Physiology, 158(1), 55-61. DOI: doi.org/10.1078/0176-1617-00126
» https://doi.org/10.1078/0176-1617-00126 - Zhou, H. W., Lurie, S., Lers, A., Khatchitski, A., Sonego, L., & Ben-Arie, R. (2000). Pre-storage and controlled atmosphere storage of nectarines: two strategies to prevent woolliness. Postharvest Biology and Technology, 18(2), 133-141. DOI: doi.org/10.1016/S0925-5214(99)00072-1
» https://doi.org/10.1016/S0925-5214(99)00072-1 - Zoffoli, J. P., Rodriguez, J., Aldunce, P., & Crisosto, C. H. (1997). Development of high concentration carbon dioxide modified atmosphere packaging systems to maintain peach quality. In A. A. Kader (Ed.), 7th International Controlled Atmosphere Research Conference (p. 132-138). Davis, US: University of California .
Publication Dates
-
Publication in this collection
17 Dec 2018 -
Date of issue
2019
History
-
Received
25 Sept 2017 -
Accepted
05 Jan 2018