ABSTRACT.
The relationship between soil and health is important for populations that depend on the local environment to meet their nutritional needs. This study aimed to evaluate the concentrations of major and trace elements in the soils, edible parts of crops and urine of farmers in agroecological communities. We collected samples of soil, edible parts of crops and urine of farmers and family members in 23 crop fields in agroecological communities from northeast Brazil. These samples were analyzed to concentrations of Al, As, Ca, Cr, Cu, Fe, Hg, Mg, Zn, and Pb in urine and discriminant analysis and principal component analysis were used to assess the data. Concentrations of potentially toxic elements in soils and crops of agroecosystems were below regulatory levels. Farmers living in agroecological communities present most of the essential and toxic elements in urine within the reference ranges. In general, results showed that urinary concentration of toxic elements among farmers and their families were below allowable limits, which may be due to the agroecological practices.
Keywords:
heavy metals; agroecossystems; biomonitoring
Introduction
Several major (Ca, Cl, Mg, P, K, Na, S, N, C, O, and H) and trace (Co, Cr, Cu, Fe, Mn, Mo, Se, Zn, F, and I) elements are essential for human nutrition, although excessive concentrations of some of these elements may cause toxicity problems. On the other hand, elements such as As, Cd, Pb, Hg, and Al have no biological functions and are only toxic (Selinus, 2004Selinus, O. (2004). Medical geology: An emerging specialty. Terræ, 1(1), 8-15. ). The concentrations of major and trace elements in crops are primarily dependent on the geological parent material of the soil, although anthropogenic sources may contribute to increases in these values.
Agroecology is the study of ecological processes that operate in agricultural production systems. Production systems based on agroecology are characterized by the use of environmentally friendly technologies that promote natural processes and biological interactions that enhance synergy so that diversified lands can support their own soil fertility, crop protection and productivity (Altieri, 2002Altieri, M. A. (2002). Agroecology: the science of natural resource management for poor farmers in marginal environments. Agriculture, Ecosystems and Environment, 93(1-3), 1-24. DOI: 10.1016/S0167-8809(02)00085-3
https://doi.org/10.1016/S0167-8809(02)00...
).
The exposure routes of humans to essential and toxic elements are the ingestion of food, water and soil, inhalation and dermal absorption. The ingestion of food and soil, the latter especially by children, have been identified as the main exposure routes for essential and potentially toxic elements in humans (Khillare et al., 2012Khillare, P. S., Jyethi, D. S., & Sarkar, S. (2012). Health risk assessment of polycyclic aromatic hydrocarbons and heavy metals via dietary intake of vegetables grown in the vicinity of thermal power plants. Food and Chemical Toxicology, 50(5), 1642-1652. DOI: 10.1016/j.fct.2012.01.032
https://doi.org/10.1016/j.fct.2012.01.03...
). The assessment of human exposure to chemicals can be performed by measuring the chemicals or their metabolites in human specimens, such as blood or urine. Urine has been regarded as a good biological indicator to evaluate the nutritional status due to its advantages over other biological matrices, such as its ease of sampling and effective indication of exposure to trace elements and various diseases (Horng et al., 2002Horng, C. J., Tsai, J. L., Horng, P. H., Lin, S. C., Lin, S. R., & Tzeng, C. C. (2002). Determination of urinary lead, cadmium and nickel in steel production workers. Talanta, 56(6), 1109-1115. DOI: 10.1016/S0039-9140(01)00645-2
https://doi.org/10.1016/S0039-9140(01)00...
; Parsons & Barbosa, 2007Parsons, P. J., & Barbosa, F. (2007). Atomic spectrometry and trends in clinical laboratory medicine. Spectrochimica Acta Part B: Atomic Spectroscopy, 62(9), 992-1003. DOI: 10.1016/j.sab.2007.03.007
https://doi.org/10.1016/j.sab.2007.03.00...
). Several studies have suggested that urine, as a non-invasive matrix, is preferred for the biomonitoring of potentially toxic elements (Moon et al., 1999Moon, C. S., Zhang, Z. W., Shimbo, S., Watanabe, T., Lee, C. -U., Lee, B. K., … Ikeda, M. (1999). Evaluation of urinary cadmium and lead as markers of background exposure of middle-aged women in Korea: dietary intake as an influential factor. Toxicology Letters, 108(2-3), 173-178. DOI: 10.1016/S0378-4274(99)00086-7
https://doi.org/10.1016/S0378-4274(99)00...
; Barbosa, Tanus-Santos, Gerlach, & Parsons, 2005Barbosa, F., Tanus-Santos, J. E., Gerlach, R. F., & Parsons, P. (2005). A critical review of biomarkers used for monitoring human exposure to lead: advantages, limitations and future needs. Environmental Health Perspectives, 113(12), 1669-1674. DOI: 10.1289/ehp.7917
https://doi.org/10.1289/ehp.7917...
; Berglund et al., 2005Berglund, M., Lind, B., Björnberg, K. A., Palm, B., Einarsson, Ö., & Vahter, M. (2005). Inter-individual variations of human mercury exposure biomarkers: a cross-sectional assessment. Environmental Health, 4(1), 20. DOI: 10.1186/1476-069X-4-20
https://doi.org/10.1186/1476-069X-4-20...
). In the present work, we hypothesize that the concentration of elements in the urine of people living in communities that produce their own food are closely related not only to the food itself but also to the soils and agricultural practices.
Although agroecological practices are widely regarded to protect human health and the environment, there have been very few studies assessing the validity of this assumption. This study aimed to evaluate the urinary concentration of major and trace elements among farmers and family members in order to correlate it with the concentration of these elements in the soils and food produced by agroecological communities. Both essential (Cu, Zn, Cr, Fe, Ca, and Mg) and toxic (Pb, As, Hg, and Al) elements were assessed.
Material and methods
Study sites
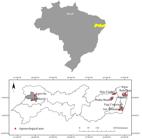
We selected farmers using agroecological practices from six communities from seven municipalities of Pernambuco State, northeast Brazil (Figure 1). In each community, three or four crop fields were selected, with a total of 23 production areas with diversified agroecological approaches, namely, the use of agroforestry, organic farming and vegetable gardens.
Sampling of soil, edible parts of crops and urine
Three composite soil samples (0 - 20 cm) were collected in each studied area; each composite sample consisted of fifteen randomly collected simple samples. Samples of the agricultural products produced and consumed by the farmers were also collected. Several samples of the edible parts of crops were randomly collected (Khillare, Jyethi, & Sarkar, 2012Khillare, P. S., Jyethi, D. S., & Sarkar, S. (2012). Health risk assessment of polycyclic aromatic hydrocarbons and heavy metals via dietary intake of vegetables grown in the vicinity of thermal power plants. Food and Chemical Toxicology, 50(5), 1642-1652. DOI: 10.1016/j.fct.2012.01.032
https://doi.org/10.1016/j.fct.2012.01.03...
). According to the seasonal availability, we collected samples from different food groups, including the following vegetables, tubers and fruits: green onion (Allium schoenoprasum), coriander (Coriandrum sativum); kale (Brassica oleracea L.); gherkin (Cucumis anguria L.); bell pepper (Capsicum annuum); okra (Abelmoschus esculentus L.); sweet potato (Ipomoea batatas); purple yam (Dioscorea alata L.); avocado (Persea americana); acerola (Malpighia punicifolia L.); banana (Musa sapientum); cocoa (Theobroma cacao); caja (Spondias mombin L.); star fruit (Averrhoa carambola); red mombin fruit (Spondias purpurea); guava (Psidium guajava); soursop (Annona muricata); orange (Citrus sinensis); lemon (Citrus limon); passion fruit (Passiflora edulis); and sugar apple (Annona squamosa).
For the urine sampling, farmers were divided into three groups: women (≥ 18 years old), men (≥ 18 years old), and children and teenagers (5 - 17 years old). For the children and teenagers, sampling was carried out with the consent of a legal guardian. Urine samples were collected in 80 mL fully transparent plastic bottles (universal collector) that had been previously sterilized by ionizing radiation. People were instructed to collect samples from the first morning urine, after disregarding the first jet, according to the procedures approved by the Research Ethics Committee of the University of Pernambuco (UPE), Brazil. All tubes were identified and transported in a cooler to the laboratory, where they were immediately frozen at - 4°C in a freezer.
Chemical analyses and quality control
Soil samples were dried at room temperature and subsequently ground and passed through a 2 mm mesh sieve. In order to determine the concentrations of trace and major elements, soil samples were further macerated in agate mortar and passed through a 0.3 mm mesh stainless steel sieve in order to avoid trace element contamination. The edible part of the crops was washed three times in distilled water and dried at 65°C. The material was then macerated in a mortar; leafy vegetables were milled in grinding mill.
Soil and crop samples were digested in Teflon vessels with 9 mL of HNO3 and 3 mL of HCl in a microwave oven (USEPA, 2007United States Environmental Protection Agency [USEPA]. (2007). Method 3051a - Microwave assisted acid digestion of sediments, sludges, soils, and oils. Revision 1 Fev 2007. Retrieved on May. 13, 2012 from 13, 2012 from http://www.epa.gov/epawaste/hazard/testmethods/sw846/pdfs/3051a.pdf
http://www.epa.gov/epawaste/hazard/testm...
). All of the extracts were transferred to 50-mL certified flasks (NBR ISO/IEC) filled with ultrapure water and filtered in a slow filter paper. High-purity acids were used in the analysis (Merck PA).
Urine samples were defrosted to room temperature and homogenized. We pipetted 2.4 mL of the sample and transferred to 15 mL sterile Falcon tube, in which we added 9.6 mL of 5% HNO3 solution and ultrapure water for maintaining a 5-fold urine dilution (Goullé et al., 2005Goullé, J. P. M., Mahieu, L., Castermant, J., Neveu, N., Bonneau, L., Bouige, D., & Lacroix, C. (2005). Metal and metalloid multi-elementary ICP-MS validation in whole blood, plasma, urine and hair: Reference values. Forensic Science International, 153(1), 39-44. DOI: 10.1016/j.forsciint.2005.04.020
https://doi.org/10.1016/j.forsciint.2005...
).
We determined the concentrations of Al, Ca, Cr, Cu, Fe, Mg, Pb, and Zn by optical emission spectrometry (ICP-OES/Optima 7000, Perkin Elmer). As and Hg were determined by an atomic absorption spectrophotometer (Aanalyst 800 Perkin Elmer) coupled with a hydride generator (FIAS 100/Flow Injection System/Perkin Elmer).
Quality control of the analysis was carried out using samples of multielement reference solutions (spikes), which were prepared from 1000 mg L-1 standard (TITRISOL®, Merck). In general, good recoveries of elements were obtained in the spike for almost all the elements, ranging between 87 and 106%.
Statistical analysis
Results were evaluated and discussed using univariate and multivariate statistical procedures. For the univariate procedure, we used descriptive statistics. To evaluate the relationship between the concentrations of the chemical elements in the urine and soil or urine and crops, the Spearman correlation analysis was used (non-parametric correlation test). The nonparametric statistical method was chosen after verifying that the variables, even when transformed, did not follow a normal distribution. For the multivariate procedures, discriminant analysis (DA) and principal component analysis (PCA) were used.
Results and discussion
Trace and major elements in soils and edible parts of crops
The concentration of trace and major elements in the soil varied widely (Table 1). This result is likely because the studied areas had different soil management processes and were located in different geological contexts influencing the natural content of elements in the soil. The mean concentration of elements in the soils followed the order Al > Fe > Ca > Mg > Cr > Zn > Pb > Cu > As > Hg, which reflects the abundance of elements in the Earth's crust. The potentially toxic trace elements presented concentrations within the quality reference values established for the state of Pernambuco (CPRH, 2014Agência Estadual de Meio Ambiente [CPRH]. (2014). Instrução normativa CPRH n° 007/2014. Retrieved on Jan. 20, 2015 from 20, 2015 from https://www.legisweb.com.br/legislacao/?id=279789
https://www.legisweb.com.br/legislacao/?...
), except for Cu and As. However, the highest Cu and As concentrations found do not pose a risk to human health, according to the Brazilian guidelines for trace elements in soil (CONAMA, 2009Conselho Nacional do Meio Ambiente [CONAMA]. (2009). Resolution Nº 420, of December 28. Retrieved on Jan. 20, 2015 from 20, 2015 from http://www.mma.gov.br/port/conama/res/res09/res42009.pdf
http://www.mma.gov.br/port/conama/res/re...
).
The concentrations of trace and major elements in the group formed by vegetables and tubers presented the following order: Ca > Mg > Fe > Al > Zn > Cu > Cr > Pb > Hg >As (Table 2). For the fruits, the order was the same, except Al and Fe switched places.
The high concentrations of Fe, Ca, and Mg in the first group of agricultural products (Table 2) are due to the high contributions of these elements in leafy vegetables. For instance, Ali and Al-Qahtani (2012Ali, M. H. H., & Al-Qahtani, K. M. (2012). Assessment of some heavy metals in vegetables, cereals and fruits in Saudi Arabian markets. Egyptian Journal of Aquatic Research, 38(1), 31-37. DOI: 10.1016/j.ejar.2012.08.002
https://doi.org/10.1016/j.ejar.2012.08.0...
) found high Fe concentrations in leafy vegetables grown in Saudi Arabia and attributed these high concentrations to the fact that leaves absorb and accumulate more Fe than fruits. Only the maximum Cr concentration in the first group was higher than that established by the National Health Surveillance Agency of Brazil - ANVISA (1965Agência Nacional de Vigilância Sanitária [ANVISA]. (1965). Decree nº 55871, of March 26.) (0.1 mg kg-1). All the other potentially toxic elements were below the maximum allowable concentrations in food established by national and international legislation.
Trace and major elements in urine
The mean concentration of Cu in the urine of farmers (Table 3) was within the reference values for the populations of Brazil, France and the UK, which range from 2.2 to 18.4 µg L-1, (Batista et al., 2009Batista, B. L., Rodrigues, J. L., Tormen, L., Curtius, A. J., & Barbosa Jr. F. (2009). Reference concentrations for trace elements in urine for the Brazilian population based on q-ICP-MS with a simple dilute-and-shoot procedure. Journal of the Brazilian Chemical Society, 20(8), 1406-1413. DOI: 10.1590/S0103-50532009000800004
https://doi.org/10.1590/S0103-5053200900...
), 4.3 to 12.1 µg L-1 (Goullé et al., 2005Goullé, J. P. M., Mahieu, L., Castermant, J., Neveu, N., Bonneau, L., Bouige, D., & Lacroix, C. (2005). Metal and metalloid multi-elementary ICP-MS validation in whole blood, plasma, urine and hair: Reference values. Forensic Science International, 153(1), 39-44. DOI: 10.1016/j.forsciint.2005.04.020
https://doi.org/10.1016/j.forsciint.2005...
), and 4.6 to 40.4 µg L-1 (White & Sabbioni, 1998White, M. A., & Sabbioni, E. (1998). Trace element reference values in tissues from inhabitants of the European Union. X. A study of 13 elements in blood and urine of a United Kingdom population. Science of The Total Environment, 216(3), 253-270. DOI: 10.1016/S0048-9697(98)00156-9
https://doi.org/10.1016/S0048-9697(98)00...
), respectively. These reference values were obtained from a large number of samples distributed across several parts of the countries, where differences occur due to the environment, eating habits, and physiological parameters; therefore, reference ranges exhibit considerable variations (Batista, Rodrigues, Tormen, Curtius, & Barbosa, 2009). The mean Cu concentration in the urine of farmers demonstrated that their exposure to this element was not problematic. The mean concentrations of Cu were lower than those reported by Inoue et al. (2014Inoue, Y., Umezaki, M., Jiang, H., Li, D., Du, J., Jin, Y., … Watanabe, C. (2014). Urinary concentrations of toxic and essential trace elements among rural residents in Hainan Island, China. International Journal of Environmental Research and Public Health, 11(12), 13047-13064. DOI: 10.3390/ijerph111213047
https://doi.org/10.3390/ijerph111213047...
) for rural residents of Hainan Island (15.1 µg L-1), China.
We found that the mean Cu concentration was higher in men than in women (Table 3). On the other hand, Aguilera et al. (2008Aguilera, I., Daponte, A., Gil, F., Hernández, A. F., Godoy, P., Pla, A., & Ramos, J. L. (2008). Biomonitoring of urinary metals in a population living in the vicinity of industrial sources: A comparison with the general population of Andalusia. Science of the Total Environment, 407(1), 669-678. DOI: 10.1016/j.scitotenv.2008.08.041
https://doi.org/10.1016/j.scitotenv.2008...
) found higher Cu concentrations in women. These gender differences in urine Cu concentrations are linked to eating habits and hormonal influences, which may affect the absorption of this element (Olsson et al., 2002Olsson, I. M., Bensryd, I., Lundh, T., Ottosson, H., Skerfving, S., & Oskarsson, A. (2002). Cadmium in blood and urine - impact of sex, age, dietary intake, iron status, and former smoking-association of renal effects. Environmental Health Perspectives, 110(12), 1185-1190. DOI: 10.1289/ehp.021101185
https://doi.org/10.1289/ehp.021101185...
; Kazi et al., 2008Kazi, T. G., Afridi, H. I., Kazi, N., Jamali, M. K., Arain, M. B., Jalbani, N., & Kandhro, G.A. (2008). Copper, chromium, manganese, iron, nickel, and zinc levels in biological samples of diabetes mellitus patients. Biological Trace Element Research, 122(1), 1-18. DOI: 10.1007/s12011-007-8062-y
https://doi.org/10.1007/s12011-007-8062-...
). Cu is an essential trace element for humans; however, it can become toxic when it accumulates in the body at high concentrations. According to Wapnir (1998Wapnir, R. A. (1998). Copper absorption and bioavailability. American Journal of Clinical Nutrition, 67(5), 1054S-1060S. DOI: 10.1093/ajcn/67.5.1054S
https://doi.org/10.1093/ajcn/67.5.1054S...
), exposure to Cu is influenced by the ingestion of contaminated food and water. Cu deficiency can lead to anemia and osteoporosis in children (Kanumakala, Boneh, & Zacharin, 2002Kanumakala, S., Boneh, A., & Zacharin, M. (2002). Pamidronate treatment improves bone mineral density in children with Menkes disease. Journal of Inherited Metabolic Disease, 25(5), 391-398. DOI: 10.1023/A:1020103901969
https://doi.org/10.1023/A:1020103901969...
).
The mean concentration of Pb (Table 3) in the urine of all farmers was within the reference concentration range (< 0.03-2.96 µg L-1) for the Brazilian population (Batista et al., 2009Batista, B. L., Rodrigues, J. L., Tormen, L., Curtius, A. J., & Barbosa Jr. F. (2009). Reference concentrations for trace elements in urine for the Brazilian population based on q-ICP-MS with a simple dilute-and-shoot procedure. Journal of the Brazilian Chemical Society, 20(8), 1406-1413. DOI: 10.1590/S0103-50532009000800004
https://doi.org/10.1590/S0103-5053200900...
). The mean Pb concentration was lower than those found by Inoue et al. (2014Inoue, Y., Umezaki, M., Jiang, H., Li, D., Du, J., Jin, Y., … Watanabe, C. (2014). Urinary concentrations of toxic and essential trace elements among rural residents in Hainan Island, China. International Journal of Environmental Research and Public Health, 11(12), 13047-13064. DOI: 10.3390/ijerph111213047
https://doi.org/10.3390/ijerph111213047...
) in China (4.3 µg L-1) and White and Sabbioni (1998White, M. A., & Sabbioni, E. (1998). Trace element reference values in tissues from inhabitants of the European Union. X. A study of 13 elements in blood and urine of a United Kingdom population. Science of The Total Environment, 216(3), 253-270. DOI: 10.1016/S0048-9697(98)00156-9
https://doi.org/10.1016/S0048-9697(98)00...
) in the United Kingdom (11.9 µg L-1) and higher than those reported in other countries, such as France (0,55 µg L-1) (Goullé et al., 2005Goullé, J. P. M., Mahieu, L., Castermant, J., Neveu, N., Bonneau, L., Bouige, D., & Lacroix, C. (2005). Metal and metalloid multi-elementary ICP-MS validation in whole blood, plasma, urine and hair: Reference values. Forensic Science International, 153(1), 39-44. DOI: 10.1016/j.forsciint.2005.04.020
https://doi.org/10.1016/j.forsciint.2005...
), the United States (1.3 µg L-1), (Komaromy-Hiller, Ash, Costa, & Howerton, 2000Komaromy-Hiller, G., Ash, K. O., Costa, R., & Howerton, K. (2000). Comparison of representative ranges based on U.S. patient population and literature reference intervals for urinary trace elements. Clinica Chimica Acta, 296(1-2), 71-90. DOI: 10.1016/S0009-8981(00)00205-9
https://doi.org/10.1016/S0009-8981(00)00...
), and Spain (1.05 µg L-1) (Castaño et al., 2012Castaño, A., Sánchez-Rodríguez, J. E., Cañas, A., Esteban, M., Navarro, C., Rodríguez-García, A. C., ... Jiménez-Guerrero, J. A. (2012). Mercury, lead and cadmium levels in the urine of 170 Spanish adults: A pilot human biomonitoring study. International Journal of Hygiene and Environmental Health, 215(2), 191-195. DOI: 10.1016/j.ijheh.2011.09.001
https://doi.org/10.1016/j.ijheh.2011.09....
). According to Kosnett (2003Kosnett, M. J. (2003). Intoxicação por metais pesados e quelantes. In B. G. Katzung (Ed.), Farmacologia básica e clínica (p. 867-870). Rio de Janeiro, RJ: Guanabara Koogan.), Pb contamination occurs due to the ingestion of contaminated food and beverages. According to Schifer, Bogusz, and Montano (2005Schifer, T. S., Bogusz, S., & Montano, M. A. E. (2005). Aspectos toxicológicos do chumbo. Infarma, 17(5-6), 67-71.), Pb concentrations in foods are higher in industrialized regions, where the metal and its compounds are widely used. Thus, it is expected that the Pb concentrations in the foods produced in agroecological communities are lower than those in the foods produced in industrialized regions, which is reflected in the farmers’ health.
In a study carried out by Moreira and Neves (2008Moreira, M. F. R., & Neves, E. B. (2008). Uso do chumbo em urina como indicador de exposição e sua relação com chumbo no sangue. Cadernos de Saúde Pública, 24(9), 2151-2159. DOI: 10.1590/S0102-311X2008000900021
https://doi.org/10.1590/S0102-311X200800...
), a significant correlation between the concentrations of lead in blood and urine was observed. Thus, the authors suggested that urine can be used to replace blood in the assessment of occupational lead exposure. By applying their exposure ranges to our data, we found that only one participant among the adults had a Pb concentration within the moderate occupational exposure range (Pb concentration in the blood between 100 to 275 µg L-1). Moreira and Neves (2008)Moreira, M. F. R., & Neves, E. B. (2008). Uso do chumbo em urina como indicador de exposição e sua relação com chumbo no sangue. Cadernos de Saúde Pública, 24(9), 2151-2159. DOI: 10.1590/S0102-311X2008000900021
https://doi.org/10.1590/S0102-311X200800...
failed to establish the occupational exposure range for children. Even if the adult ranges were applied to the group of children and teenagers in this work, they would all be classified as environmentally exposed (Pb concentration in the blood of < 100 µg L-1).
The Pb concentrations in the urine of children and teenagers were higher than those in the urine of men and women (Table 3). This is probably due to the greater contact of children with dust and topsoil since the streets of rural communities are not paved. Several studies have shown that the ingestion of soil or inhalation of soil particles (dust) are the most significant routes of Pb exposure in children (Maisonet, Bove, & Kaye, 1997Maisonet, M., Bove, F. J., & Kaye, W. E. (1997). A case-control for elevated blood lead levels in children, Idaho. Toxicology and Industrial Health, 13(1), 67-72. DOI: 10.1177/074823379701300106
https://doi.org/10.1177/0748233797013001...
; Meyer, Heinrich, & Lippold, 1999Meyer, I., Heinrich, J., & Lippold, U. (1999). Factors affecting lead, cadmium, and arsenic levels in house dust in a smelter town in eastern Germany. Environmental Research, 81(1), 32-44. DOI: 10.1006/enrs.1998.3950
https://doi.org/10.1006/enrs.1998.3950...
; Paoliello et al., 2002Paoliello, M. M. B., Capitani, E. M., Cunha, F. G., Matsuo, T., Carvalho, M. F., Sakuma, A., & Figueiredo, B. F. (2002). Exposure of children to lead and cadmium from a mining area of Brazil. Environmental Research, 88(2), 120-128. DOI: 10.1006/enrs.2001.4311
https://doi.org/10.1006/enrs.2001.4311...
). According to Cunha, Figueiredo, Paoliello, and De Capitani (2006Cunha, F. G., Figueiredo, B. R., Paoliello, M. M. B., & De Capitani, E. M. (2006). Diagnóstico ambiental e de saúde humana: contaminação por chumbo em Adrianópolis no estado do Paraná, Brasil. In C. R. Silva, B. R. Figueiredo, E. M. De Capitani , & F. G. Cunha (Eds.), Geologia médica no Brasil (p. 97-103). Rio de Janeiro, RJ: CPRM/Serviço Geológico do Brasil.), children are considered the group demonstrating the highest risk, since, compared to adults, they absorb and retain more ingested Pb. The Disease Control Center (DCC) of the United States reported that the current concerning concentration of Pb in children's blood is 100 µg L-1. However, adverse effects may occur at lower concentrations.
Due to the lack of Zn reference values in urine in Brazil, the concentrations were compared with values reported in France. The mean Zn concentrations of the three groups (Table 3) were within the range established by Goullé et al. (2005Goullé, J. P. M., Mahieu, L., Castermant, J., Neveu, N., Bonneau, L., Bouige, D., & Lacroix, C. (2005). Metal and metalloid multi-elementary ICP-MS validation in whole blood, plasma, urine and hair: Reference values. Forensic Science International, 153(1), 39-44. DOI: 10.1016/j.forsciint.2005.04.020
https://doi.org/10.1016/j.forsciint.2005...
) for the French population, which is 44 - 499 µg L-1. The wide Zn concentration range observed in men, with a maximum concentration of 1,567.5 µg L-1, was probably due to the two farmers of that group who are smokers. Schuhmacher, Domingo, and Corbella (1994Schuhmacher, M., Domingo, J. L., & Corbella, J. (1994). Zinc and copper levels in serum and urine: Relationship to biological, habitual and environmental factors. The Science of the Total Environment, 148(1), 67-72. DOI: 10.1016/0048-9697(94)90376-X
https://doi.org/10.1016/0048-9697(94)903...
) compared the urinary Zn concentrations of smokers and nonsmokers and found that smokers presented the highest values; in blood serum, in contrast, nonsmokers had higher Zn concentrations. Therefore, as confirmed by the data of our study, the use of cigarettes results in higher Zn excretion in urine. The mean Zn concentrations (Table 3) were lower than those found in the populations of the following countries: China, 470 µg L-1 (Inoue et al., 2014Inoue, Y., Umezaki, M., Jiang, H., Li, D., Du, J., Jin, Y., … Watanabe, C. (2014). Urinary concentrations of toxic and essential trace elements among rural residents in Hainan Island, China. International Journal of Environmental Research and Public Health, 11(12), 13047-13064. DOI: 10.3390/ijerph111213047
https://doi.org/10.3390/ijerph111213047...
); the United States, 371.5 µg L-1 (Komaromy-Hiller et al., 2000Komaromy-Hiller, G., Ash, K. O., Costa, R., & Howerton, K. (2000). Comparison of representative ranges based on U.S. patient population and literature reference intervals for urinary trace elements. Clinica Chimica Acta, 296(1-2), 71-90. DOI: 10.1016/S0009-8981(00)00205-9
https://doi.org/10.1016/S0009-8981(00)00...
); and Spain, 698.7 µg L-1 (Schuhmacher et al., 1994). The Zn concentrations in the group comprising children and teenagers were lower than that reported for children in Poland, which is 660 µg L-1 (Błażewicz, Klatka, Astel, Partyka, & Kocjan, 2013Błażewicz, A., Klatka, M., Astel, A., Partyka, M., & Kocjan, R. (2013). Differences in trace metal concentrations (Co, Cu, Fe, Mn, Zn, Cd, and Ni) in whole blood, plasma, and urine of obese and monobese children. Biological Trace Element Research, 155(2), 190-200. DOI: 10.1007/s12011-013-9783-8
https://doi.org/10.1007/s12011-013-9783-...
).
The mean concentration of Cr (Table 3) in this study differed from the reference values of the United States and the United Kingdom. The mean Cr concentrations in the urine of women (0.56 µg L-1) was within the range established by Paschal et al. (1998Paschal, D. C., Ting, B. G., Morrow, J. C., Pirkle, J. L., Jackson, R. J., Sampson, E. J., … Caldwell, K. L. (1998). Trace metals in urine of United States residents: Reference range concentrations. Environmental Research, 76(1), 53-59. DOI: 10.1006/enrs.1997.3793
https://doi.org/10.1006/enrs.1997.3793...
) for the United States, ranging from < 0.1 to 0.7 µg L-1. In men, the mean Cr concentration (0.67 µg L-1) was close to the upper limit of the reference range. The group of children and teenagers showed a mean Cr concentration (0.77 µg L-1) slightly above the upper reference limit. When compared with the reference values for the UK (0.04 - 0.96 µg L-1) (White & Sabbioni, 1998White, M. A., & Sabbioni, E. (1998). Trace element reference values in tissues from inhabitants of the European Union. X. A study of 13 elements in blood and urine of a United Kingdom population. Science of The Total Environment, 216(3), 253-270. DOI: 10.1016/S0048-9697(98)00156-9
https://doi.org/10.1016/S0048-9697(98)00...
), all groups showed mean Cr concentrations within the range.
The mean Al concentration in urine (Table 3) was above the reference concentrations established for the Brazilian population, which are between 0.22 and 17.5 µg L-1 (Batista et al., 2009Batista, B. L., Rodrigues, J. L., Tormen, L., Curtius, A. J., & Barbosa Jr. F. (2009). Reference concentrations for trace elements in urine for the Brazilian population based on q-ICP-MS with a simple dilute-and-shoot procedure. Journal of the Brazilian Chemical Society, 20(8), 1406-1413. DOI: 10.1590/S0103-50532009000800004
https://doi.org/10.1590/S0103-5053200900...
). The mean concentration was also higher than the reference values for France, which are between 0.16 and 11.2 µg L-1 (Goullé et al., 2005Goullé, J. P. M., Mahieu, L., Castermant, J., Neveu, N., Bonneau, L., Bouige, D., & Lacroix, C. (2005). Metal and metalloid multi-elementary ICP-MS validation in whole blood, plasma, urine and hair: Reference values. Forensic Science International, 153(1), 39-44. DOI: 10.1016/j.forsciint.2005.04.020
https://doi.org/10.1016/j.forsciint.2005...
). When compared with the reference values of the United Kingdom, which are between 1.2 and 168 µg L-1 (White & Sabbioni, 1998White, M. A., & Sabbioni, E. (1998). Trace element reference values in tissues from inhabitants of the European Union. X. A study of 13 elements in blood and urine of a United Kingdom population. Science of The Total Environment, 216(3), 253-270. DOI: 10.1016/S0048-9697(98)00156-9
https://doi.org/10.1016/S0048-9697(98)00...
), the mean Al concentrations for all three groups were within the range. Al is toxic to the human body. The World Health Organization (WHO, 1996World Health Organization [WHO]. (1996). Trace elements in human nutrition and health. Revised ed. Geneva, SW: WHO.) reports that Al ingestion is increased when acidic foods are cooked with utensils made of Al.
The concentrations of As in the urine of farmers (Table 3) were within the reference range for the population of the United Kingdom (White & Sabbioni, 1998White, M. A., & Sabbioni, E. (1998). Trace element reference values in tissues from inhabitants of the European Union. X. A study of 13 elements in blood and urine of a United Kingdom population. Science of The Total Environment, 216(3), 253-270. DOI: 10.1016/S0048-9697(98)00156-9
https://doi.org/10.1016/S0048-9697(98)00...
) and France (Goullé et al., 2005Goullé, J. P. M., Mahieu, L., Castermant, J., Neveu, N., Bonneau, L., Bouige, D., & Lacroix, C. (2005). Metal and metalloid multi-elementary ICP-MS validation in whole blood, plasma, urine and hair: Reference values. Forensic Science International, 153(1), 39-44. DOI: 10.1016/j.forsciint.2005.04.020
https://doi.org/10.1016/j.forsciint.2005...
) (0.4 and 48.2 µg L-1 and 2.3 and 161 µg L-1, respectively). The values found in this study were higher than those found by Aguilera et al. (2008Aguilera, I., Daponte, A., Gil, F., Hernández, A. F., Godoy, P., Pla, A., & Ramos, J. L. (2008). Biomonitoring of urinary metals in a population living in the vicinity of industrial sources: A comparison with the general population of Andalusia. Science of the Total Environment, 407(1), 669-678. DOI: 10.1016/j.scitotenv.2008.08.041
https://doi.org/10.1016/j.scitotenv.2008...
) (2.11 µg L-1) in Spain and lower than those reported by Komaromy-Hiller et al. (2000Komaromy-Hiller, G., Ash, K. O., Costa, R., & Howerton, K. (2000). Comparison of representative ranges based on U.S. patient population and literature reference intervals for urinary trace elements. Clinica Chimica Acta, 296(1-2), 71-90. DOI: 10.1016/S0009-8981(00)00205-9
https://doi.org/10.1016/S0009-8981(00)00...
) (25.1 µg L-1) for the United States, Calderón et al. (2001Calderón, J., Navarro, M. E., Jimenez-Capdeville, M. E., Santos-Diaz, M. A., Golden, A., Rodriguez-Levya, I., ... Diaz-Barriga, F. (2001). Exposure to arsenic and lead and neuropsychological development in Mexican children. Environmental Research, 85(2), 69-76. DOI: 10.1006/enrs.2000.4106
https://doi.org/10.1006/enrs.2000.4106...
) (40.28 µg L-1) for Mexico, and Nordberg et al. (2005Nordberg, G. F., Jin, T., Hong, F., Zhang, A., Buchet, J. P., & Bernard, A. (2005). Biomarkers of cadmium and arsenic interactions. Toxicology and Applied Pharmacology, 206(2), 191-197. DOI: 10.1016/j.taap.2004.11.028
https://doi.org/10.1016/j.taap.2004.11.0...
) (56.23 µg L-1) for China. Children and teenagers showed a mean As concentration higher than that of adults (Table 4). According to Zhang, Deng, Lu, and Richardson (2002Zhang, Z., Deng, C., Lu, Q., & Richardson, B. (2002). Age-dependent DNA methylation changes in the ITGAL (CD11a) promoter. Mechanisms of Ageing and Development, 123(9), 1257-1268. DOI: 10.1016/S0047-6374(02)00014-3.
https://doi.org/10.1016/S0047-6374(02)00...
), studies have shown that the expression of genes coding the methyltransferases involved in DNA methylation decreases significantly as humans age. Other authors have speculated that methylation increases over human growth and that the exposure to factors that may inhibit the methylation, such as cigarettes, alcoholic beverages, and environmental pollutants, increases with the age (Hsueh et al., 2003Hsueh, Y. M., Ko, Y. F., Huang, Y. K., Chen, H. W., Chiou, H. Y., Huang, Y. L., … Chen, C. J. (2003). Determinants of inorganic arsenic methylation capability among residents of the Lanyang Basin, Taiwan: arsenic and selenium exposure and alcohol consumption. Toxicology Letters, 137(1-2), 49-63. DOI: 10.1016/S0378-4274(02)003806
https://doi.org/10.1016/S0378-4274(02)00...
; Lindberg et al., 2008Lindberg, A-L., Ekström, E-C., Nermell, B., Rahman, M., Lönnerdal, B., Persson, L-A., & Vahter, M. (2008). Gender and age differences in the metabolism of inorganic arsenic in a highly exposed population in Bangladesh. Environmental Research, 106(1), 110-120. DOI: 10.1016/j.envres.2007.08.011
https://doi.org/10.1016/j.envres.2007.08...
). The methylation of inorganic As in the human body reduces the affinity of the compound to the tissue, and urine is the main form of As elimination (Vahter, 2002Vahter, M. (2002). Mechanisms of arsenic biotransformation. Toxicology, 181-182, 211-217. DOI: 10.1016/S0300-483X(02)00285-8
https://doi.org/10.1016/S0300-483X(02)00...
).
The mean Hg concentration in the urine of farmers (Table 3) was within the reference range for the United Kingdom (White & Sabbioni, 1998White, M. A., & Sabbioni, E. (1998). Trace element reference values in tissues from inhabitants of the European Union. X. A study of 13 elements in blood and urine of a United Kingdom population. Science of The Total Environment, 216(3), 253-270. DOI: 10.1016/S0048-9697(98)00156-9
https://doi.org/10.1016/S0048-9697(98)00...
), France (Goullé et al., 2005Goullé, J. P. M., Mahieu, L., Castermant, J., Neveu, N., Bonneau, L., Bouige, D., & Lacroix, C. (2005). Metal and metalloid multi-elementary ICP-MS validation in whole blood, plasma, urine and hair: Reference values. Forensic Science International, 153(1), 39-44. DOI: 10.1016/j.forsciint.2005.04.020
https://doi.org/10.1016/j.forsciint.2005...
), and the Czech Republic (Batáriová et al., 2006Batáriová, A., Speváckova, V., Benes, B., Cejchanova, M., Smíd, J., & Cerná, M. (2006). Blood and urine levels of Pb, Cd and Hg in the general population of the Czech Republic and proposed reference values. International Journal of Hygiene and Environmental Health, 209(4), 359-366. DOI: 10.1016/j.ijheh.2006.02.005
https://doi.org/10.1016/j.ijheh.2006.02....
), which established intervals of < 0.5 - 10.0 µg L-1; 0.14 - 2.21 µg L-1, and 0.55 - 3.45 µg L-1, respectively. These mean concentrations of Hg were lower than those found by Castaño et al. (2012Castaño, A., Sánchez-Rodríguez, J. E., Cañas, A., Esteban, M., Navarro, C., Rodríguez-García, A. C., ... Jiménez-Guerrero, J. A. (2012). Mercury, lead and cadmium levels in the urine of 170 Spanish adults: A pilot human biomonitoring study. International Journal of Hygiene and Environmental Health, 215(2), 191-195. DOI: 10.1016/j.ijheh.2011.09.001
https://doi.org/10.1016/j.ijheh.2011.09....
), with a mean concentration of 1.19 µg L-1, and Komaromy-Hiller et al. (2000Komaromy-Hiller, G., Ash, K. O., Costa, R., & Howerton, K. (2000). Comparison of representative ranges based on U.S. patient population and literature reference intervals for urinary trace elements. Clinica Chimica Acta, 296(1-2), 71-90. DOI: 10.1016/S0009-8981(00)00205-9
https://doi.org/10.1016/S0009-8981(00)00...
), with a mean concentration of 1.4 µg L-1. The maximum acceptable concentration of Hg in the urine of adults in the state of New York is 20 µg L-1. In contrast with the pattern for As, the mean values of Hg were lower in children and teenagers than in adults. This was also observed by Gil et al. (2006Gil, F., Capitán-Vallvey, L. F., De Santiago, E., Ballesta, J., Pla, A., Hernández, A. F., ... Villanueva, E. (2006). Heavy metal concentrations in the general population of Andalusia, South of Spain: a comparison with the population within the area of influence of Aznalcóllar mine spill (SW Spain). Science of The Total Environment, 372(1), 49-57. DOI: 10.1016/j.scitotenv.2006.08.004
https://doi.org/10.1016/j.scitotenv.2006...
), who reported higher Hg concentrations in the urine of adults than in the urine of children.
The mean concentration of Fe (Table 3) was below the value reported by Cui et al. (2005Cui, Y., Zhu, Y-G., Zhai, R., Huang, Y., Qiu, Y., & Liang, J. (2005). Exposure to metal mixtures and human health impacts in a contaminated area in Nanning, China. Environment International, 31(6), 784-790. DOI: 10.1016/j.envint.2005.05.025
https://doi.org/10.1016/j.envint.2005.05...
), who compared the Fe concentration in the urine of a group of people from an uncontaminated area (310 µg L-1) with those in the urine of two groups exposed to contaminated areas (190 and 180 µg L-1). It is also below the mean Fe concentration (72.3 µg L-1) found in a study carried out in Poland (Długaszek, Kaszczuk, & Mularczyk-Oliwa, 2011Długaszek, M., Kaszczuk, M., & Mularczyk-Oliwa, M. (2011). Magnesium, calcium, and trace elements excretion in 24-h urine. Biological Trace Element Research, 142(1), 1-10. DOI: 10.1007/s12011-010-8745-7
https://doi.org/10.1007/s12011-010-8745-...
). Długaszek et al. (2011) also found higher iron concentrations in men than in women. This gender difference is because women are more susceptible to iron deficiency caused by menstrual blood flow, especially those of childbearing age (Tefferi, 2003Tefferi, A. (2003). Anemia in adults: A contemporary in adults approach to diagnosis. Mayo Clinic Proceedings, 78(10), 1274-1280. DOI: 10.4065/78.10.1274
https://doi.org/10.4065/78.10.1274...
). According to the WHO (2001World Health Organization [WHO]. (2001). Iron deficiency anaemia: Assessment, prevention, and control. A guide for programme managers. Geneva, SW: WHO .), Fe deficiency anemia is the most prominent nutritional disease worldwide, affecting 20-30% of the world population; women are the highest risk group (Coad & Conlon, 2011Coad, J., & Conlon, C. (2011). Iron deficiency in women: assessment, causes and consequences. Current Opinion in Clinical Nutrition and Metabolic Care, 14(6), 625-634. DOI: 10.1097/MCO.0b013e32834be6fd
https://doi.org/10.1097/MCO.0b013e32834b...
).
Ca is an essential element and one of the most abundant nutrients in the human body (Azin, Raie, & Mahmoudi, 1998Azin, F., Raie, R. M., & Mahmoudi, M. M. (1998). Correlation between the levels of certain carcinogenic and anticarcinogenic trace elements and esophageal cancer in northern Iran. Ecotoxicology and Environmental Safety, 39(3), 179-184. DOI: 10.1006/eesa.1997.1601
https://doi.org/10.1006/eesa.1997.1601...
). The calcium concentrations in the urine of framers were 45.01, 94.49, and 77.86 mg L-1 for women, men and children and teenagers, respectively (Table 3). These concentrations were lower than those found in Nanning, China (Cui et al. 2005Cui, Y., Zhu, Y-G., Zhai, R., Huang, Y., Qiu, Y., & Liang, J. (2005). Exposure to metal mixtures and human health impacts in a contaminated area in Nanning, China. Environment International, 31(6), 784-790. DOI: 10.1016/j.envint.2005.05.025
https://doi.org/10.1016/j.envint.2005.05...
), and Warsaw, Poland (Długaszek et al., 2011Długaszek, M., Kaszczuk, M., & Mularczyk-Oliwa, M. (2011). Magnesium, calcium, and trace elements excretion in 24-h urine. Biological Trace Element Research, 142(1), 1-10. DOI: 10.1007/s12011-010-8745-7
https://doi.org/10.1007/s12011-010-8745-...
). For men and children, we found higher concentrations of Ca than those reported in Hyderabad City, Pakistan (Afridi et al., 2008Afridi, H. I., Kazi, T. G., Kazi, N., Jamali, M. K., Arain, M. B., Jalbani, N., & Baig, J. A. (2008). Potassium, calcium, magnesium, and sodium levels in biological samples of hypertensive and nonhypertensive diabetes Mellitus patients. Biological Trace Element Research, 124(3), 206-224. DOI: 10.1007/s12011-008-8142-7
https://doi.org/10.1007/s12011-008-8142-...
). The Ca concentrations presented high variability, with a coefficient of variation of 122% for women. The concentration of Ca in urine is related to many factors, such as daily food consumption, eating habits, malnutrition, absorption and elimination processes, renal diseases, thyroid and parathyroid gland functions, bone metabolism, stress, excessive aldosterone, smoking, alcohol and medications that interfere with the absorption of this element and promote an increase in its excretion (Siener & Hesse, 2002Siener, R., & Hesse, A. (2002). The effect of different diets on urine composition and the risk of calcium oxalate crystalisation in healthy subjects. European Urology, 42(3), 289-296. DOI: 10.1016/S0302-2838(02)00316-0
https://doi.org/10.1016/S0302-2838(02)00...
; Długaszek et al., 2011Długaszek, M., Kaszczuk, M., & Mularczyk-Oliwa, M. (2011). Magnesium, calcium, and trace elements excretion in 24-h urine. Biological Trace Element Research, 142(1), 1-10. DOI: 10.1007/s12011-010-8745-7
https://doi.org/10.1007/s12011-010-8745-...
).
The mean concentrations of Mg in the urine of farmers (Table 3) were higher for the groups of men and children and teenagers than the concentrations found by Afridi et al. (2008Afridi, H. I., Kazi, T. G., Kazi, N., Jamali, M. K., Arain, M. B., Jalbani, N., & Baig, J. A. (2008). Potassium, calcium, magnesium, and sodium levels in biological samples of hypertensive and nonhypertensive diabetes Mellitus patients. Biological Trace Element Research, 124(3), 206-224. DOI: 10.1007/s12011-008-8142-7
https://doi.org/10.1007/s12011-008-8142-...
) and Długaszek et al. (2011Długaszek, M., Kaszczuk, M., & Mularczyk-Oliwa, M. (2011). Magnesium, calcium, and trace elements excretion in 24-h urine. Biological Trace Element Research, 142(1), 1-10. DOI: 10.1007/s12011-010-8745-7
https://doi.org/10.1007/s12011-010-8745-...
). Urinary excretion is the main form of eliminating absorbed Mg, with 1/3 of the Mg ingested daily excreted in the urine (Alpers, Clouse, & Stenson, 1988Alpers, D. H., Clouse, R. E., & Stenson, W. F. (1988). Manual of Nutritional Therapeutics. Boston, US/Toronto, CA: Little, Brown and Company.). Considering a Mg elimination rate of 90 mg day-1 per the ingestion of 270 mg day-1 and a mean production of 1 L day-1 urine, women were the most affected group in our study. A total of 80% of the women had Mg concentrations in their urine that were lower than the appropriate level, followed by men (52%) and children and teenagers (33%). Mg deficiency can be classified into primary and secondary forms, in which the insufficient consumption of foods rich in Mg, the excessive consumption of sugar and fat, protein-energy malnutrition, and Mg-deficient parenteral nutrition contribute to the primary deficiency. Secondary deficiency is affected by alcoholism, decreased Mg absorption, diarrhea, laxative abuse, malabsorption syndrome, vomiting, increased renal excretion, tubular disease, glomerulonephritis, metabolic and endocrine disorders, medications, pregnancy, and physical and mental stress (Barbagallo, Belvedere, & Dominguez, 2009Barbagallo, M., Belvedere, M., & Dominguez, L. J. (2009). Magnesium homeostasis and aging. Magnesium Research, 22(4), 235-246. DOI: 10.1684/mrh.2009.0187
https://doi.org/10.1684/mrh.2009.0187...
).
Discriminant analysis
To verify the differences in the concentrations of trace and major elements in the urine of different groups (men, women and children), discriminant analysis (DA) was applied. For this analysis, in addition to the concentrations of the elements in the urine, we added information about the body mass and height (to calculate the body mass index - BMI), age, and smoking status of each farmer. The three groups showed different behaviors (Figure 2), with an accuracy greater than 80% (Table 4).
Based on DA, we carried out principal component analysis (PCA) by groups of individuals (Figure 3). Data were standardized, and eigenvalues superior to unity were selected. Additional variables were added to the PCA, namely, BMI, age and smoking habit.
Based on the PCA for the group of men (Figure 3a and b), it was possible to reduce the variables to three components, which together explained 57.56% of the total variation in the data. The overlap of Zn and the complementary variable smoking (Figure 3b) in men confirmed the link between higher urinary Zn excretion and the smoking habit. The readily observed overlap between Ca and Mg (Figure 3a) was due to the proximity of the mean concentrations of the elements in the urine of men, with values of 94.49 and 99.66 mg L-1, respectively (Table 3).
For women, the PCA revealed little influence of complementary variables (BMI, age, and smoking habit) on the concentrations of trace and major elements in urine (Figure 3c and d). The three components together explained 65.52% of the total variation in the data.
Age and BMI influenced the Ca and Mg in the urine of children and teenagers (Figure 3e). This association is desirable, since children and teenagers require a greater amount of energy than adults, with an appropriate vitamin and mineral supply (Greer & Krebs, 2006Greer, F. R., & Krebs, N. F. (2006). Optimizing bone health and calcium intakes of infants, children, and adolescents. Pediatrics, 117(2), 578-585. DOI: 10.1542/peds.2005-2822
https://doi.org/10.1542/peds.2005-2822...
), due to the growth phase. The three PCA components explained 66.73% of the total variation in the data. For the first component (Figure 3f), there was greater expressiveness for the potentially toxic trace elements Cu and Pb. This result corroborates the increased exposure of children to these elements, possibly because their high contact with dust and surface soil makes them more vulnerable to contamination (Sapcanin, Cakal, Pehlic, & Jancan, 2016Sapcanin, A., Cakal, M., Pehlic, E., & Jancan, L. (2016). Soil pollution fingerprints of children playgrounds in Sarajevo city, Bosnia and Herzegovina. Environmental Science and Pollution Research, 24(12), 10949-10954. DOI: 10.1007/s11356-016-6301-5
https://doi.org/10.1007/s11356-016-6301-...
).
Concentrations of chemical elements in urine of farmers of agroecological systems and their relations with body mass index, age and smoking habits. a and b (men), c and d (women), e and f (children and teenagers).
Conclusion
The concentrations of essential and toxic elements in the urine of farmers living in agroecological communities were within the reference ranges of Brazil and other countries. Children and teenagers were more susceptible than adults to the influence of elements in soil due to their higher direct contact with the soil. In general, the results showed that the urinary concentrations of toxic elements among farmers and their families were below the allowable limits, which may be due to the agroecological practices in the studied communities. PCA analysis demonstrated an association between the smoking habit and the Zn concentration in urine.
Acknowledgements
The authors acknowledge the valuable assistance of the NGOs "Caatinga - Sowing life in the semi-arid" and "Sabiá - Agro-ecological Development Center", and of the Association of Agro-ecological Farmers of Bom Jardim (Agroflor) in the choices of the sites and in the samplings of soil, agricultural products and urine. The authors also thank the farmers for providing the necessary samples for the study.
References
- Afridi, H. I., Kazi, T. G., Kazi, N., Jamali, M. K., Arain, M. B., Jalbani, N., & Baig, J. A. (2008). Potassium, calcium, magnesium, and sodium levels in biological samples of hypertensive and nonhypertensive diabetes Mellitus patients. Biological Trace Element Research, 124(3), 206-224. DOI: 10.1007/s12011-008-8142-7
» https://doi.org/10.1007/s12011-008-8142-7 - Aguilera, I., Daponte, A., Gil, F., Hernández, A. F., Godoy, P., Pla, A., & Ramos, J. L. (2008). Biomonitoring of urinary metals in a population living in the vicinity of industrial sources: A comparison with the general population of Andalusia. Science of the Total Environment, 407(1), 669-678. DOI: 10.1016/j.scitotenv.2008.08.041
» https://doi.org/10.1016/j.scitotenv.2008.08.041 - Ali, M. H. H., & Al-Qahtani, K. M. (2012). Assessment of some heavy metals in vegetables, cereals and fruits in Saudi Arabian markets. Egyptian Journal of Aquatic Research, 38(1), 31-37. DOI: 10.1016/j.ejar.2012.08.002
» https://doi.org/10.1016/j.ejar.2012.08.002 - Alpers, D. H., Clouse, R. E., & Stenson, W. F. (1988). Manual of Nutritional Therapeutics Boston, US/Toronto, CA: Little, Brown and Company.
- Altieri, M. A. (2002). Agroecology: the science of natural resource management for poor farmers in marginal environments. Agriculture, Ecosystems and Environment, 93(1-3), 1-24. DOI: 10.1016/S0167-8809(02)00085-3
» https://doi.org/10.1016/S0167-8809(02)00085-3 - Agência Estadual de Meio Ambiente [CPRH]. (2014). Instrução normativa CPRH n° 007/2014 Retrieved on Jan. 20, 2015 from 20, 2015 from https://www.legisweb.com.br/legislacao/?id=279789
» https://www.legisweb.com.br/legislacao/?id=279789 - Agência Nacional de Vigilância Sanitária [ANVISA]. (1965). Decree nº 55871, of March 26
- Azin, F., Raie, R. M., & Mahmoudi, M. M. (1998). Correlation between the levels of certain carcinogenic and anticarcinogenic trace elements and esophageal cancer in northern Iran. Ecotoxicology and Environmental Safety, 39(3), 179-184. DOI: 10.1006/eesa.1997.1601
» https://doi.org/10.1006/eesa.1997.1601 - Barbagallo, M., Belvedere, M., & Dominguez, L. J. (2009). Magnesium homeostasis and aging. Magnesium Research, 22(4), 235-246. DOI: 10.1684/mrh.2009.0187
» https://doi.org/10.1684/mrh.2009.0187 - Barbosa, F., Tanus-Santos, J. E., Gerlach, R. F., & Parsons, P. (2005). A critical review of biomarkers used for monitoring human exposure to lead: advantages, limitations and future needs. Environmental Health Perspectives, 113(12), 1669-1674. DOI: 10.1289/ehp.7917
» https://doi.org/10.1289/ehp.7917 - Batáriová, A., Speváckova, V., Benes, B., Cejchanova, M., Smíd, J., & Cerná, M. (2006). Blood and urine levels of Pb, Cd and Hg in the general population of the Czech Republic and proposed reference values. International Journal of Hygiene and Environmental Health, 209(4), 359-366. DOI: 10.1016/j.ijheh.2006.02.005
» https://doi.org/10.1016/j.ijheh.2006.02.005 - Batista, B. L., Rodrigues, J. L., Tormen, L., Curtius, A. J., & Barbosa Jr. F. (2009). Reference concentrations for trace elements in urine for the Brazilian population based on q-ICP-MS with a simple dilute-and-shoot procedure. Journal of the Brazilian Chemical Society, 20(8), 1406-1413. DOI: 10.1590/S0103-50532009000800004
» https://doi.org/10.1590/S0103-50532009000800004 - Berglund, M., Lind, B., Björnberg, K. A., Palm, B., Einarsson, Ö., & Vahter, M. (2005). Inter-individual variations of human mercury exposure biomarkers: a cross-sectional assessment. Environmental Health, 4(1), 20. DOI: 10.1186/1476-069X-4-20
» https://doi.org/10.1186/1476-069X-4-20 - Błażewicz, A., Klatka, M., Astel, A., Partyka, M., & Kocjan, R. (2013). Differences in trace metal concentrations (Co, Cu, Fe, Mn, Zn, Cd, and Ni) in whole blood, plasma, and urine of obese and monobese children. Biological Trace Element Research, 155(2), 190-200. DOI: 10.1007/s12011-013-9783-8
» https://doi.org/10.1007/s12011-013-9783-8 - Calderón, J., Navarro, M. E., Jimenez-Capdeville, M. E., Santos-Diaz, M. A., Golden, A., Rodriguez-Levya, I., ... Diaz-Barriga, F. (2001). Exposure to arsenic and lead and neuropsychological development in Mexican children. Environmental Research, 85(2), 69-76. DOI: 10.1006/enrs.2000.4106
» https://doi.org/10.1006/enrs.2000.4106 - Castaño, A., Sánchez-Rodríguez, J. E., Cañas, A., Esteban, M., Navarro, C., Rodríguez-García, A. C., ... Jiménez-Guerrero, J. A. (2012). Mercury, lead and cadmium levels in the urine of 170 Spanish adults: A pilot human biomonitoring study. International Journal of Hygiene and Environmental Health, 215(2), 191-195. DOI: 10.1016/j.ijheh.2011.09.001
» https://doi.org/10.1016/j.ijheh.2011.09.001 - Coad, J., & Conlon, C. (2011). Iron deficiency in women: assessment, causes and consequences. Current Opinion in Clinical Nutrition and Metabolic Care, 14(6), 625-634. DOI: 10.1097/MCO.0b013e32834be6fd
» https://doi.org/10.1097/MCO.0b013e32834be6fd - Conselho Nacional do Meio Ambiente [CONAMA]. (2009). Resolution Nº 420, of December 28 Retrieved on Jan. 20, 2015 from 20, 2015 from http://www.mma.gov.br/port/conama/res/res09/res42009.pdf
» http://www.mma.gov.br/port/conama/res/res09/res42009.pdf - Cui, Y., Zhu, Y-G., Zhai, R., Huang, Y., Qiu, Y., & Liang, J. (2005). Exposure to metal mixtures and human health impacts in a contaminated area in Nanning, China. Environment International, 31(6), 784-790. DOI: 10.1016/j.envint.2005.05.025
» https://doi.org/10.1016/j.envint.2005.05.025 - Cunha, F. G., Figueiredo, B. R., Paoliello, M. M. B., & De Capitani, E. M. (2006). Diagnóstico ambiental e de saúde humana: contaminação por chumbo em Adrianópolis no estado do Paraná, Brasil. In C. R. Silva, B. R. Figueiredo, E. M. De Capitani , & F. G. Cunha (Eds.), Geologia médica no Brasil (p. 97-103). Rio de Janeiro, RJ: CPRM/Serviço Geológico do Brasil.
- Długaszek, M., Kaszczuk, M., & Mularczyk-Oliwa, M. (2011). Magnesium, calcium, and trace elements excretion in 24-h urine. Biological Trace Element Research, 142(1), 1-10. DOI: 10.1007/s12011-010-8745-7
» https://doi.org/10.1007/s12011-010-8745-7 - Gil, F., Capitán-Vallvey, L. F., De Santiago, E., Ballesta, J., Pla, A., Hernández, A. F., ... Villanueva, E. (2006). Heavy metal concentrations in the general population of Andalusia, South of Spain: a comparison with the population within the area of influence of Aznalcóllar mine spill (SW Spain). Science of The Total Environment, 372(1), 49-57. DOI: 10.1016/j.scitotenv.2006.08.004
» https://doi.org/10.1016/j.scitotenv.2006.08.004 - Goullé, J. P. M., Mahieu, L., Castermant, J., Neveu, N., Bonneau, L., Bouige, D., & Lacroix, C. (2005). Metal and metalloid multi-elementary ICP-MS validation in whole blood, plasma, urine and hair: Reference values. Forensic Science International, 153(1), 39-44. DOI: 10.1016/j.forsciint.2005.04.020
» https://doi.org/10.1016/j.forsciint.2005.04.020 - Greer, F. R., & Krebs, N. F. (2006). Optimizing bone health and calcium intakes of infants, children, and adolescents. Pediatrics, 117(2), 578-585. DOI: 10.1542/peds.2005-2822
» https://doi.org/10.1542/peds.2005-2822 - Horng, C. J., Tsai, J. L., Horng, P. H., Lin, S. C., Lin, S. R., & Tzeng, C. C. (2002). Determination of urinary lead, cadmium and nickel in steel production workers. Talanta, 56(6), 1109-1115. DOI: 10.1016/S0039-9140(01)00645-2
» https://doi.org/10.1016/S0039-9140(01)00645-2 - Hsueh, Y. M., Ko, Y. F., Huang, Y. K., Chen, H. W., Chiou, H. Y., Huang, Y. L., … Chen, C. J. (2003). Determinants of inorganic arsenic methylation capability among residents of the Lanyang Basin, Taiwan: arsenic and selenium exposure and alcohol consumption. Toxicology Letters, 137(1-2), 49-63. DOI: 10.1016/S0378-4274(02)003806
» https://doi.org/10.1016/S0378-4274(02)003806 - Inoue, Y., Umezaki, M., Jiang, H., Li, D., Du, J., Jin, Y., … Watanabe, C. (2014). Urinary concentrations of toxic and essential trace elements among rural residents in Hainan Island, China. International Journal of Environmental Research and Public Health, 11(12), 13047-13064. DOI: 10.3390/ijerph111213047
» https://doi.org/10.3390/ijerph111213047 - Kanumakala, S., Boneh, A., & Zacharin, M. (2002). Pamidronate treatment improves bone mineral density in children with Menkes disease. Journal of Inherited Metabolic Disease, 25(5), 391-398. DOI: 10.1023/A:1020103901969
» https://doi.org/10.1023/A:1020103901969 - Kazi, T. G., Afridi, H. I., Kazi, N., Jamali, M. K., Arain, M. B., Jalbani, N., & Kandhro, G.A. (2008). Copper, chromium, manganese, iron, nickel, and zinc levels in biological samples of diabetes mellitus patients. Biological Trace Element Research, 122(1), 1-18. DOI: 10.1007/s12011-007-8062-y
» https://doi.org/10.1007/s12011-007-8062-y - Khillare, P. S., Jyethi, D. S., & Sarkar, S. (2012). Health risk assessment of polycyclic aromatic hydrocarbons and heavy metals via dietary intake of vegetables grown in the vicinity of thermal power plants. Food and Chemical Toxicology, 50(5), 1642-1652. DOI: 10.1016/j.fct.2012.01.032
» https://doi.org/10.1016/j.fct.2012.01.032 - Komaromy-Hiller, G., Ash, K. O., Costa, R., & Howerton, K. (2000). Comparison of representative ranges based on U.S. patient population and literature reference intervals for urinary trace elements. Clinica Chimica Acta, 296(1-2), 71-90. DOI: 10.1016/S0009-8981(00)00205-9
» https://doi.org/10.1016/S0009-8981(00)00205-9 - Kosnett, M. J. (2003). Intoxicação por metais pesados e quelantes. In B. G. Katzung (Ed.), Farmacologia básica e clínica (p. 867-870). Rio de Janeiro, RJ: Guanabara Koogan.
- Lindberg, A-L., Ekström, E-C., Nermell, B., Rahman, M., Lönnerdal, B., Persson, L-A., & Vahter, M. (2008). Gender and age differences in the metabolism of inorganic arsenic in a highly exposed population in Bangladesh. Environmental Research, 106(1), 110-120. DOI: 10.1016/j.envres.2007.08.011
» https://doi.org/10.1016/j.envres.2007.08.011 - Maisonet, M., Bove, F. J., & Kaye, W. E. (1997). A case-control for elevated blood lead levels in children, Idaho. Toxicology and Industrial Health, 13(1), 67-72. DOI: 10.1177/074823379701300106
» https://doi.org/10.1177/074823379701300106 - Meyer, I., Heinrich, J., & Lippold, U. (1999). Factors affecting lead, cadmium, and arsenic levels in house dust in a smelter town in eastern Germany. Environmental Research, 81(1), 32-44. DOI: 10.1006/enrs.1998.3950
» https://doi.org/10.1006/enrs.1998.3950 - Moon, C. S., Zhang, Z. W., Shimbo, S., Watanabe, T., Lee, C. -U., Lee, B. K., … Ikeda, M. (1999). Evaluation of urinary cadmium and lead as markers of background exposure of middle-aged women in Korea: dietary intake as an influential factor. Toxicology Letters, 108(2-3), 173-178. DOI: 10.1016/S0378-4274(99)00086-7
» https://doi.org/10.1016/S0378-4274(99)00086-7 - Moreira, M. F. R., & Neves, E. B. (2008). Uso do chumbo em urina como indicador de exposição e sua relação com chumbo no sangue. Cadernos de Saúde Pública, 24(9), 2151-2159. DOI: 10.1590/S0102-311X2008000900021
» https://doi.org/10.1590/S0102-311X2008000900021 - Nordberg, G. F., Jin, T., Hong, F., Zhang, A., Buchet, J. P., & Bernard, A. (2005). Biomarkers of cadmium and arsenic interactions. Toxicology and Applied Pharmacology, 206(2), 191-197. DOI: 10.1016/j.taap.2004.11.028
» https://doi.org/10.1016/j.taap.2004.11.028 - Olsson, I. M., Bensryd, I., Lundh, T., Ottosson, H., Skerfving, S., & Oskarsson, A. (2002). Cadmium in blood and urine - impact of sex, age, dietary intake, iron status, and former smoking-association of renal effects. Environmental Health Perspectives, 110(12), 1185-1190. DOI: 10.1289/ehp.021101185
» https://doi.org/10.1289/ehp.021101185 - Paoliello, M. M. B., Capitani, E. M., Cunha, F. G., Matsuo, T., Carvalho, M. F., Sakuma, A., & Figueiredo, B. F. (2002). Exposure of children to lead and cadmium from a mining area of Brazil. Environmental Research, 88(2), 120-128. DOI: 10.1006/enrs.2001.4311
» https://doi.org/10.1006/enrs.2001.4311 - Parsons, P. J., & Barbosa, F. (2007). Atomic spectrometry and trends in clinical laboratory medicine. Spectrochimica Acta Part B: Atomic Spectroscopy, 62(9), 992-1003. DOI: 10.1016/j.sab.2007.03.007
» https://doi.org/10.1016/j.sab.2007.03.007 - Paschal, D. C., Ting, B. G., Morrow, J. C., Pirkle, J. L., Jackson, R. J., Sampson, E. J., … Caldwell, K. L. (1998). Trace metals in urine of United States residents: Reference range concentrations. Environmental Research, 76(1), 53-59. DOI: 10.1006/enrs.1997.3793
» https://doi.org/10.1006/enrs.1997.3793 - Sapcanin, A., Cakal, M., Pehlic, E., & Jancan, L. (2016). Soil pollution fingerprints of children playgrounds in Sarajevo city, Bosnia and Herzegovina. Environmental Science and Pollution Research, 24(12), 10949-10954. DOI: 10.1007/s11356-016-6301-5
» https://doi.org/10.1007/s11356-016-6301-5 - Schifer, T. S., Bogusz, S., & Montano, M. A. E. (2005). Aspectos toxicológicos do chumbo. Infarma, 17(5-6), 67-71.
- Schuhmacher, M., Domingo, J. L., & Corbella, J. (1994). Zinc and copper levels in serum and urine: Relationship to biological, habitual and environmental factors. The Science of the Total Environment, 148(1), 67-72. DOI: 10.1016/0048-9697(94)90376-X
» https://doi.org/10.1016/0048-9697(94)90376-X - Selinus, O. (2004). Medical geology: An emerging specialty. Terræ, 1(1), 8-15.
- Siener, R., & Hesse, A. (2002). The effect of different diets on urine composition and the risk of calcium oxalate crystalisation in healthy subjects. European Urology, 42(3), 289-296. DOI: 10.1016/S0302-2838(02)00316-0
» https://doi.org/10.1016/S0302-2838(02)00316-0 - Tefferi, A. (2003). Anemia in adults: A contemporary in adults approach to diagnosis. Mayo Clinic Proceedings, 78(10), 1274-1280. DOI: 10.4065/78.10.1274
» https://doi.org/10.4065/78.10.1274 - United States Environmental Protection Agency [USEPA]. (2007). Method 3051a - Microwave assisted acid digestion of sediments, sludges, soils, and oils Revision 1 Fev 2007. Retrieved on May. 13, 2012 from 13, 2012 from http://www.epa.gov/epawaste/hazard/testmethods/sw846/pdfs/3051a.pdf
» http://www.epa.gov/epawaste/hazard/testmethods/sw846/pdfs/3051a.pdf - Vahter, M. (2002). Mechanisms of arsenic biotransformation. Toxicology, 181-182, 211-217. DOI: 10.1016/S0300-483X(02)00285-8
» https://doi.org/10.1016/S0300-483X(02)00285-8 - Wapnir, R. A. (1998). Copper absorption and bioavailability. American Journal of Clinical Nutrition, 67(5), 1054S-1060S. DOI: 10.1093/ajcn/67.5.1054S
» https://doi.org/10.1093/ajcn/67.5.1054S - White, M. A., & Sabbioni, E. (1998). Trace element reference values in tissues from inhabitants of the European Union. X. A study of 13 elements in blood and urine of a United Kingdom population. Science of The Total Environment, 216(3), 253-270. DOI: 10.1016/S0048-9697(98)00156-9
» https://doi.org/10.1016/S0048-9697(98)00156-9 - World Health Organization [WHO]. (1996). Trace elements in human nutrition and health Revised ed. Geneva, SW: WHO.
- World Health Organization [WHO]. (2001). Iron deficiency anaemia: Assessment, prevention, and control A guide for programme managers. Geneva, SW: WHO .
- Zhang, Z., Deng, C., Lu, Q., & Richardson, B. (2002). Age-dependent DNA methylation changes in the ITGAL (CD11a) promoter. Mechanisms of Ageing and Development, 123(9), 1257-1268. DOI: 10.1016/S0047-6374(02)00014-3.
» https://doi.org/10.1016/S0047-6374(02)00014-3.
Data availability
Data citations
United States Environmental Protection Agency [USEPA]. (2007). Method 3051a - Microwave assisted acid digestion of sediments, sludges, soils, and oils Revision 1 Fev 2007. Retrieved on May. 13, 2012 from 13, 2012 from http://www.epa.gov/epawaste/hazard/testmethods/sw846/pdfs/3051a.pdf
Publication Dates
-
Publication in this collection
28 Mar 2019 -
Date of issue
2019
History
-
Received
28 Oct 2017 -
Accepted
19 Mar 2018