Abstract
This study analyzed the effect of different repetition durations on electromyographic and blood lactate responses of the bench press exercise. Fifteen recreationally trained male volunteers completed two training protocols, matched for intensity (% one-repetition maximum; 1RM), number of sets, number of repetitions, and rest intervals. One of the protocols was performed with a repetition duration of 4 s (2 s concentric: 2 s eccentric; 2:2 protocol), whereas the second protocol had a repetition duration of 6 s (2 s concentric: 4 s eccentric; 2:4 protocol). The results showed higher normalized integrated electromyography (pectoralis major and triceps brachii) for the 2:4 protocol. Blood lactate concentration was also higher in the 2:4 protocol across all sets. These results show that adding 2 s to the eccentric action in matched training protocols increases muscle activation and blood lactate response, which reinforces the notion that increasing repetition duration is an alternative load progression in resistance training.
Keywords:
resistance training; electromyography; lactate
Introduction
Repetition duration, defined as the time spent performing the concentric and eccentric muscle actions during a repetition, should be considered when prescribing a resistance training program (American College of Sports Medicine [ACSM], 2009American College of Sports Medicine. (2009). Position stand: Progression models in resistance training for healthy adults. Medicine and Science in Sports and Exercise, 41, 687-708. doi:10.1249/MSS.0b013e3181915670.
https://doi.org/10.1249/MSS.0b013e318191...
; Bird, Tarpenning, & Marino, 2005Bird, S., Tarpenning, M., & Marino F. (2005). Designing resistance training programmes to enhance muscular fitness: a review of the acute programme variables. Sports Medicine, 35, 841-851. doi: 10.2165/00007256-200535100-00002
https://doi.org/10.2165/00007256-2005351...
). Repetition durations between 4 s and 6 s are recommended for resistance training emphasizing muscle hypertrophy (ACSM, 2009American College of Sports Medicine. (2009). Position stand: Progression models in resistance training for healthy adults. Medicine and Science in Sports and Exercise, 41, 687-708. doi:10.1249/MSS.0b013e3181915670.
https://doi.org/10.1249/MSS.0b013e318191...
; Wernbom, Augustsson, & Thomeé, 2007Wernbom, M., Augustsson, J., & Thomeé, R. (2007). The influence of frequency, intensity, volume and mode of strength training on whole muscle cross-sectional area in humans. Sports Medicine, 37, 225-264. doi: 10.2165/00007256-200737030-00004
https://doi.org/10.2165/00007256-2007370...
) and are commonly used by weight trainers (Headley et al. , 2011Headley, S. A., Henry, K., Nindl, B. C., Thompson, B. A., Kraemer, W. J., & Jones, M. (2011). Effects of lifting tempo on one repetition maximum and hormonal responses to a bench press protocol. The Journal of Strength and Conditioning Research, 25, 406-413. doi: 10.1519/JSC.0b013e3181bf053b
https://doi.org/10.1519/JSC.0b013e3181bf...
). However, comparative physiological data about the effect of these durations on resis tance training are scarce.
The manipulation of the repetition duration affects electro myographic responses (Burd et al. , 2012Burd, N. A., Andrews, R. J., West, D. W., Little, J. P., Cochran, A. J., Hector, A. J., ... & Phillips, S. M. (2012). Muscle time under tension during resistance exercise stimulates differ ential muscle protein sub-fractional synthetic responses in men. The Journal of Physiology, 590, 351-362. doi: 10.1113/jphysiol.2011.221200
https://doi.org/10.1113/jphysiol.2011.22...
; Sakamoto & Sinclair, 2012Sakamoto, A., & Sinclair, P. J. (2012). Muscle activations under vary ing lifting speeds and intensities during bench press. European Journal of Applied Physiology, 112:1015-1025. doi: 10.1007/S00421-011-2059-0
https://doi.org/10.1007/S00421-011-2059-...
). The amplitude of the electromyographic signal has often been measured in resistance training protocols (Burd et al. , 2012Burd, N. A., Andrews, R. J., West, D. W., Little, J. P., Cochran, A. J., Hector, A. J., ... & Phillips, S. M. (2012). Muscle time under tension during resistance exercise stimulates differ ential muscle protein sub-fractional synthetic responses in men. The Journal of Physiology, 590, 351-362. doi: 10.1113/jphysiol.2011.221200
https://doi.org/10.1113/jphysiol.2011.22...
; Eckhardt, Wollny, Müller, Bärtsch, & Friedmann-Bette, 2011Eckhardt, H., Wollny, R., Müller, H., Bärtsch, P., & Friedmann-Bette, B. (2011). Enhanced myofiber recruitment during exhaustive squatting performed as whole-body vibration exercise. Journal of Strength and Conditioning Research, 25, 1120-1125. doi: 10.1519/JSC.0b013e3181d09e0e.
https://doi.org/10.1519/JSC.0b013e3181d0...
; Lacerda et al. , 2016Lacerda, L., Martins Costa, H., Diniz, R., Lima, F., Pereira Andrade, A., Tourino, F., ... Chagas, M. (2016). Variations in repetition duration and repetition numbers influences muscular activation and blood lactate response in protocols equalized by time under tension. The Journal of Strength and Conditioning Research, 30, 251-218. doi: 10.1519/JSC.0000000000001044
https://doi.org/10.1519/JSC.000000000000...
; Sakamoto & Sinclair, 2012Sakamoto, A., & Sinclair, P. J. (2012). Muscle activations under vary ing lifting speeds and intensities during bench press. European Journal of Applied Physiology, 112:1015-1025. doi: 10.1007/S00421-011-2059-0
https://doi.org/10.1007/S00421-011-2059-...
; Sampson, Donohoe, & Groeller, 2014Sampson, J. A., Donohoe, A., & Groeller, H. (2014). Effect of concen tric and eccentric velocity during heavy-load non-ballistic elbow flexion resistance exercise. Journal of Science and Medicine in Sport. 17, 306-311. doi: 10.1016/j.jsams.2013.04.012
https://doi.org/10.1016/j.jsams.2013.04....
; Smilios, Häkkinen, & Tokmakidis, 2010Smilios, I., Häkkinen, K., & Tokmakidis, S. P. (2010). Power output and electromyographic activity during and after a moderate load mus cular endurance session. The Journal of Strength and Conditioning Research, 24, 2122-2131. doi: 10.1519/JSC.0b013e3181a5bc44.
https://doi.org/10.1519/JSC.0b013e3181a5...
; Takarada, Takazawa, Sato, Takebayashi, Tanaka, & Ishii, 2000Takarada, Y., Takazawa, H., Sato, Y., Takebayashi, S., Tanaka, Y., & Ishii, N. (2000). Effects of resistance exercise combined with moderate vascular occlusion on muscular function in humans. Journal of Applied Physiology, 88, 2097-2106.; Walker, Taipale, Nyman, Kraemer, & Häkkinen, 2011Walker, S., Taipale, R.S., Nyman, K., Kraemer, W. J., & Häkkinen, K. (2011). Neuromuscular and hormonal responses to constant and variable resistance loadings. Medicine and Science in Sports and Exercise. 43, 26-33. doi: 10.1249/MSS.0b013e3181e71bcb.
https://doi.org/10.1249/MSS.0b013e3181e7...
), since it represents the degree of muscle activation and, at least in part, the recruitment of motor units during the training session (Hunter, Duchateau, & Enoka, 2004Hunter, S. K., Duchateau, J., & Enoka, R. M. (2004). Muscle fatigue and the mechanisms of task failure. Exercise and Sport Science Review, 32, 44-49. doi: 10.1097/00003677-200404000-00002
https://doi.org/10.1097/00003677-2004040...
; Suzuki, Conwit, Stashuk, Santarsiero, & Metter, 2002Suzuki, H., Conwit, R., Stashuk. D., Santarsiero, L., & Metter. E. (2002). Relationships between surface-detected EMG signals and motor unit activation. Medicine and Science in Sports and Exercise, 34, 1509-1517. doi:10.1249/01.MSS.0000027711.31651.AF
https://doi.org/10.1249/01.MSS.000002771...
). Some of these studies have also measured the concentration of blood lactate (Eckhardt et al. , 2011Eckhardt, H., Wollny, R., Müller, H., Bärtsch, P., & Friedmann-Bette, B. (2011). Enhanced myofiber recruitment during exhaustive squatting performed as whole-body vibration exercise. Journal of Strength and Conditioning Research, 25, 1120-1125. doi: 10.1519/JSC.0b013e3181d09e0e.
https://doi.org/10.1519/JSC.0b013e3181d0...
; Lacerda et al. , 2016Lacerda, L., Martins Costa, H., Diniz, R., Lima, F., Pereira Andrade, A., Tourino, F., ... Chagas, M. (2016). Variations in repetition duration and repetition numbers influences muscular activation and blood lactate response in protocols equalized by time under tension. The Journal of Strength and Conditioning Research, 30, 251-218. doi: 10.1519/JSC.0000000000001044
https://doi.org/10.1519/JSC.000000000000...
; Smilios et al. , 2010Smilios, I., Häkkinen, K., & Tokmakidis, S. P. (2010). Power output and electromyographic activity during and after a moderate load mus cular endurance session. The Journal of Strength and Conditioning Research, 24, 2122-2131. doi: 10.1519/JSC.0b013e3181a5bc44.
https://doi.org/10.1519/JSC.0b013e3181a5...
; Takarada et al. , 2000Takarada, Y., Takazawa, H., Sato, Y., Takebayashi, S., Tanaka, Y., & Ishii, N. (2000). Effects of resistance exercise combined with moderate vascular occlusion on muscular function in humans. Journal of Applied Physiology, 88, 2097-2106.; Walker et al. , 2011Walker, S., Taipale, R.S., Nyman, K., Kraemer, W. J., & Häkkinen, K. (2011). Neuromuscular and hormonal responses to constant and variable resistance loadings. Medicine and Science in Sports and Exercise. 43, 26-33. doi: 10.1249/MSS.0b013e3181e71bcb.
https://doi.org/10.1249/MSS.0b013e3181e7...
) because the augmented lactate response has been associated with an increased recruitment of fast-twitch motor units (Gladden, 2004Gladden, L. (2004). Lactate metabolism: a new paradigm for the third millennium. The Journal of Physiology, 558, 5-30.; Moritani, Tanaka, Yoshida, Ishii, Yoshida, & Shindo, 1984Moritani, T., Tanaka, H., Yoshida, T., Ishii, C., Yoshida, T., & Shindo, M. (1984). Relationship between myoelectric signals and blood lactate during incremental forearm exercise. American Journal of Physical Medicine, 63, 122-132.). Given the expectation that the activated muscle fibers are those that adapt to training (Spiering et al. , 2008Spiering, B. A., Kraemer, W. J., Anderson, J . M., Armstrong, L. E., Nindl, B. C., Volek, J. S., & Maresh, C. M. (2008). Resistance exercise biology: manipulation of resistance exercise programme variables determines the responses of cellular and molecu lar signalling pathways. Sports Medicine. 38, 527-540. doi: 10.2165/00007256-200838070-00001
https://doi.org/10.2165/00007256-2008380...
; Wakahara, Fukutani, Kawakami, & Yanai, 2013Wakahara, T., Fukutani, A., Kawakami, Y., & Yanai, T. (2013). Nonuniform muscle hypertrophy: its relation to muscle activation in training session. Medicine and Science in Sports and Exercise, 45, 2158-2165. doi: 10.1249/MSS.0b013e3182995349.
https://doi.org/10.1249/MSS.0b013e318299...
), protocols which show higher electromyographic amplitude would present greater potential to promote increases in strength and muscle hypertrophy (Takarada et al. ; 2000Takarada, Y., Takazawa, H., Sato, Y., Takebayashi, S., Tanaka, Y., & Ishii, N. (2000). Effects of resistance exercise combined with moderate vascular occlusion on muscular function in humans. Journal of Applied Physiology, 88, 2097-2106.; Watanabe, Madarame, Ogasawara, Nakazato, & Ishii, 2014Watanabe, Y., Madarame, H., Ogasawara, R., Nakazato, K., & Ishii, N. (2014). Effect of very low-intensity resistance training with slow movement on muscle size and strength in healthy older adults. Clinical Physiology and Functional Imaging, 34, 463-470. doi: 10.1111/cpf.12117.
https://doi.org/10.1111/cpf.12117...
).
Although it is expected that the increase in exercise time could result in a rise in electromyographic amplitude (Hunter et al. , 2004Hunter, S. K., Duchateau, J., & Enoka, R. M. (2004). Muscle fatigue and the mechanisms of task failure. Exercise and Sport Science Review, 32, 44-49. doi: 10.1097/00003677-200404000-00002
https://doi.org/10.1097/00003677-2004040...
), studies that examined the effect of repetition du ration in muscle activation have shown different results. While Burd et al. (2012)Burd, N. A., Andrews, R. J., West, D. W., Little, J. P., Cochran, A. J., Hector, A. J., ... & Phillips, S. M. (2012). Muscle time under tension during resistance exercise stimulates differ ential muscle protein sub-fractional synthetic responses in men. The Journal of Physiology, 590, 351-362. doi: 10.1113/jphysiol.2011.221200
https://doi.org/10.1113/jphysiol.2011.22...
verified higher concentric electromyographic amplitude when performing longer duration exercise (12 s vs. 2 s), Sampson et al. (2014)Sampson, J. A., Donohoe, A., & Groeller, H. (2014). Effect of concen tric and eccentric velocity during heavy-load non-ballistic elbow flexion resistance exercise. Journal of Science and Medicine in Sport. 17, 306-311. doi: 10.1016/j.jsams.2013.04.012
https://doi.org/10.1016/j.jsams.2013.04....
found no differences on the tested situations (maximum speed, ~3 s, and 4 s) for this same type of muscle action. Unlike these studies, Sakamoto and Sinclair (2012)Sakamoto, A., & Sinclair, P. J. (2012). Muscle activations under vary ing lifting speeds and intensities during bench press. European Journal of Applied Physiology, 112:1015-1025. doi: 10.1007/S00421-011-2059-0
https://doi.org/10.1007/S00421-011-2059-...
verified an increase in concentric muscle activation when using shorter repetition durations (maximum speed, 1.9 s, 2.8 s, and 5.6 s). Furthermore, in the studies of Sampson et al. (2014)Sampson, J. A., Donohoe, A., & Groeller, H. (2014). Effect of concen tric and eccentric velocity during heavy-load non-ballistic elbow flexion resistance exercise. Journal of Science and Medicine in Sport. 17, 306-311. doi: 10.1016/j.jsams.2013.04.012
https://doi.org/10.1016/j.jsams.2013.04....
and Sakamoto & Sinclair (2012)Sakamoto, A., & Sinclair, P. J. (2012). Muscle activations under vary ing lifting speeds and intensities during bench press. European Journal of Applied Physiology, 112:1015-1025. doi: 10.1007/S00421-011-2059-0
https://doi.org/10.1007/S00421-011-2059-...
the analysis of multiple sets was not carried out, neither the protocols were equalized by train ing volume (number of repetitions), training load component that affect deeply the degree of muscle activation (Lacerda et al. , 2016Lacerda, L., Martins Costa, H., Diniz, R., Lima, F., Pereira Andrade, A., Tourino, F., ... Chagas, M. (2016). Variations in repetition duration and repetition numbers influences muscular activation and blood lactate response in protocols equalized by time under tension. The Journal of Strength and Conditioning Research, 30, 251-218. doi: 10.1519/JSC.0000000000001044
https://doi.org/10.1519/JSC.000000000000...
; Yue, Alexander, Laidlaw, Gmitro, Unger, & Enoka, 1994Yue, G., Alexander, A., Laidlaw, D., Gmitro, A., Unger, E., & Enoka, R. (1994). Sensitivity of muscle proton spin-spin relaxation time as an index of muscle activation. Journal of Applied Physiology. 77, 84-92.). The lack of control of the repetition duration and of the equalization of all variables of the training protocol, except the independent variable, has been a methodological problem in many studies involving the manipulation of the repetition duration on resistance training (González-Badillo, Rodríguez- Rosell, Sánchez-Medina, Gorostiaga, & Pareja-Blanco, 2014González-Badillo, J.J., Rodríguez-Rosell, D., Sánchez-Medina, L., Gorostiaga, E. M, & Pareja-Blanco, F. (2014). Maximal intended velocity training induces greater gains in bench press performance than deliberately slower half-velocity training. European Journal of Sport Science, 14, 772-781. doi: 10.1080/17461391.2014.905987
https://doi.org/10.1080/17461391.2014.90...
).
In addition to those controversial results, it should be highlighted that the protocols used by Burd et al. (2012)Burd, N. A., Andrews, R. J., West, D. W., Little, J. P., Cochran, A. J., Hector, A. J., ... & Phillips, S. M. (2012). Muscle time under tension during resistance exercise stimulates differ ential muscle protein sub-fractional synthetic responses in men. The Journal of Physiology, 590, 351-362. doi: 10.1113/jphysiol.2011.221200
https://doi.org/10.1113/jphysiol.2011.22...
, even though equalized by training volume, had quite different repe tition duration values (Δ = 10 s), and are considered outside of the range suggested for muscle hypertrophy (ACSM, 2009American College of Sports Medicine. (2009). Position stand: Progression models in resistance training for healthy adults. Medicine and Science in Sports and Exercise, 41, 687-708. doi:10.1249/MSS.0b013e3181915670.
https://doi.org/10.1249/MSS.0b013e318191...
; Bird et al. , 2005Bird, S., Tarpenning, M., & Marino F. (2005). Designing resistance training programmes to enhance muscular fitness: a review of the acute programme variables. Sports Medicine, 35, 841-851. doi: 10.2165/00007256-200535100-00002
https://doi.org/10.2165/00007256-2005351...
; Schoenfeld, Ogborn, & Krieger, 2015Schoenfeld, B.J., Ogborn, D., & Krieger, J. (2015). Effect of repetition duration during resistance training on muscle hypertrophy: a sys tematic review and meta-analysis. Sports Medicine, 45, 577-585. doi: 10.1007/s40279-015-0304-0.
https://doi.org/10.1007/s40279-015-0304-...
; Wernbom et al. , 2007Wernbom, M., Augustsson, J., & Thomeé, R. (2007). The influence of frequency, intensity, volume and mode of strength training on whole muscle cross-sectional area in humans. Sports Medicine, 37, 225-264. doi: 10.2165/00007256-200737030-00004
https://doi.org/10.2165/00007256-2007370...
). It can also be noted that the training protocols analyzed in the aforementioned study (Burd et al. , 2012Burd, N. A., Andrews, R. J., West, D. W., Little, J. P., Cochran, A. J., Hector, A. J., ... & Phillips, S. M. (2012). Muscle time under tension during resistance exercise stimulates differ ential muscle protein sub-fractional synthetic responses in men. The Journal of Physiology, 590, 351-362. doi: 10.1113/jphysiol.2011.221200
https://doi.org/10.1113/jphysiol.2011.22...
) had low intensity (30% of one repetition maximum - 1RM). Considering that muscle activation response is affected by the intensity of training (%1RM) (Cook, Murphy, & Labarbera, 2013Cook, S., Murphy, B., & Labarbera K. (2013). Neuromuscular function after a bout of low-load blood flow-restricted exercise. Medicine and Science in Sports and Exercise, 45,67-74. doi: 10.1249/MSS.0b013e31826c6fa8.
https://doi.org/10.1249/MSS.0b013e31826c...
), and the fact that it has been identified in the literature the interaction between the intensity and repetition duration in the electromyographic amplitude (Sakamoto & Sinclair, 2012Sakamoto, A., & Sinclair, P. J. (2012). Muscle activations under vary ing lifting speeds and intensities during bench press. European Journal of Applied Physiology, 112:1015-1025. doi: 10.1007/S00421-011-2059-0
https://doi.org/10.1007/S00421-011-2059-...
), further research is necessary to establish the effect of repetition duration on electromyographic responses during the execution of protocols with higher intensity. Therefore, to our knowledge, no studies have compared the effect of repetition duration on electromyographic response in matched protocols with multiple sets and repetition duration configuration that it has been recommended for muscle hypertrophy. Research data involving this issue may allow insights into other physi ological responses considering matched protocols. Therefore, the purpose of the present study was to compare the effect of 4 s (2 s concentric:2 s eccentric; 2:2) and 6 s (2 s concentric:4 s eccentric; 2:4) repetition duration on muscle activation and blood lactate response using matched resistance training pro tocols. We hypothesized that the increase of the exercise time by manipulating the repetition duration would increase the amplitude of electromyographic signal. We also hypothesized that longer repetition duration would augment blood lactate concentration due to increased fast-twitch motor unit recruit ment and consequently higher anaerobic glycolysis.
Methods
Participants
Fifteen male weight trainers (mean age 24.4 ± 4.4 years; mean height 1.77 ± 0.07 m; mean body mass 78.3 ± 9.4 kg; mean 1RM 93.4 ± 10.4 kg) participated in this study. The inclusion criteria for participation were (a) currently performing resistance training continuously for at least 6 months before the start of the study; (b) no functional limitations with regard to performing the 1RM test or the training protocols; and (c) the ability to lift a weight corresponding to their own body mass on the 1RM bench press exercise. The volunteers were informed about the study objectives, procedures and risks and freely signed an informed consent form. The local ethics committee of the university ap proved this study, which complies with international standards. The volunteers' training routines were modified during data collection, in order to avoid performing exercises that use the pectoralis major, anterior deltoid, or triceps brachii muscles 48 hours prior to sessions. Participants were also instructed to maintain the same dietary practices before each session.
Experimental design
This study used a crossover design to examine the effect of resistance training protocols differentiated by repetition duration (2:2 and 2:4) on electromyographic responses and blood lactate concentration. Each volunteer attended the laboratory on 4 dif ferent days (Experimental Sessions 1 through 4) separated by at least 48 hours. The registration of electromyographic activity occurred during the performance of each set of the training proto cols, while blood samples for the estimation of the blood lactate were obtained before and after the sets. The same data collection schedule was maintained for each volunteer across all sessions.
Experimental session 1
After assessing the anthropometric measurements, the range of motion and the positions of the hands and head on the equipment were standardized. This standardization was carried out after each volunteer has been positioned as close as possible to his training routine in the bench press exercise and performed some repetitions without additional weight on the bar. Subsequently, the volunteers performed the 1RM test. The 1RM test was performed during the first session to famil iarize the volunteers with the procedures. The test began with an eccentric muscle action by lowering the bar to the sternum, followed by a concentric muscle action, determined by the extension of the elbows. It was performed a maximum of 6 attempts, with 5 min rests and a minimum increase of 2 kg on each attempt. An average of 4.2 ± 1.0 attempts was necessary to determine the 1RM performance for experimental session 1. As the last procedure of experimental session 1, participants were also familiarized with the use of the metronome (60 beats/min) performing one of the training protocols to be implemented in experimental sessions 3 and 4.
Experimental session 2
The first procedure the experimental session 2 was the execution of the 1RM test, as previously described. The 1RM test was performed to determine the weight for the following sessions. An average of 3.9 ± 0.8 attempts was necessary to determine the 1RM performance for experimental session 2. After completion of the 1RM test, as well as in experimental session 1, participants were also familiarized with the use of the metronome performing the other training protocol to be implemented in experimental sessions 3 or 4.
Experimental sessions 3 and 4
Training protocols
In experimental sessions 3 and 4 the subjects were randomly submitted to the training protocols 2:2 and 2:4. The training pro tocols consisted of 3 sets, 6 repetitions and 3 min rests between sets at an intensity of 60% 1RM, with different repetition dura tions (2:2 or 2:4). These training protocols were determined in a previous study (Diniz, Martins-Costa, Machado, Lima, & Chagas, 2014Diniz, R. C., Martins-Costa, H. C., Machado, S. C., Lima, F. V., & Chagas, M. H. (2014). Repetition duration influences ratings of perceived exertion. Perceptual and Motor Skills, 118, 261-273. doi: 10.2466/03.06.PMS.118k11w6
https://doi.org/10.2466/03.06.PMS.118k11...
), which verified that the volunteers could not complete the entire protocol for the greater repetition duration (2:4) using a greater number of sets, repetitions or intensity. Furthermore, we aimed to maintain the protocol configuration according to the recommendations for strength training for muscle hypertrophy (ACSM, 2009American College of Sports Medicine. (2009). Position stand: Progression models in resistance training for healthy adults. Medicine and Science in Sports and Exercise, 41, 687-708. doi:10.1249/MSS.0b013e3181915670.
https://doi.org/10.1249/MSS.0b013e318191...
; Bird et al. , 2005Bird, S., Tarpenning, M., & Marino F. (2005). Designing resistance training programmes to enhance muscular fitness: a review of the acute programme variables. Sports Medicine, 35, 841-851. doi: 10.2165/00007256-200535100-00002
https://doi.org/10.2165/00007256-2005351...
; Wernbom et al. , 2007Wernbom, M., Augustsson, J., & Thomeé, R. (2007). The influence of frequency, intensity, volume and mode of strength training on whole muscle cross-sectional area in humans. Sports Medicine, 37, 225-264. doi: 10.2165/00007256-200737030-00004
https://doi.org/10.2165/00007256-2007370...
).
Procedures overview
An electrogoniometer was positioned on the volunteer's elbow, and electrodes were fixed to the pectoralis major and triceps brachii muscles as part of the first procedure in experimental sessions 3 and 4. The skin was marked using a semi-permanent pen to reposition the electrogoniometer and electrodes and the same researcher determined landmarks and carried out the positioning of equipment in both exper imental sessions. The electrodes and the electrogoniometer were initially fixed, and the volunteers remained seated for 10 min; at this point, the first blood sample was collected to analyze the blood lactate concentration at rest. The remaining samples were collected 1 minute after each set of the training protocols. Electromyographic activity was recorded while performing each set of the training protocols.
Electrogoniometer
A calibrated electrogoniometer (Biovision, Wehrheim, Germany) was fixed on the right elbow of participants using double-sided adhesive tape and elastic bands. Once stored, the electrogoniometer raw data were converted into angular displacement data and filtered through a 4th-order Butterworth low-pass filter with a cut-off frequency of 10 Hz. The dura tion of each repetition was calculated by the time between the points of lowest angular displacement (elbow extension) recorded by electrogoniometer during the execution of the training protocols. Additionally, the electrogoniometer was also used to determine the angular velocity and elbow range of motion.
Electromyography
The surface electromyography procedure (Biovision, Wehrheim, Germany) followed the recommendations of Hermens, Freriks, Disselhorst-Klug, and Rau (2000)Hermens, H. J., Freriks, B., Disselhorst-Klug, C., & Rau, G.J. (2000). Development of recommendations for SEMG sensors and sen sor placement procedures. Journal of Electromyography and Kinesiology, 10, 361-374. doi: 0.1016/S1050-6411(00)00027-4
https://doi.org/0.1016/S1050-6411(00)000...
. Bipolar surface electrodes (Ag/AgCl) were placed parallel to the muscle fibers of the volunteers' right pectoralis major (sternal portion) and triceps brachii (long head portion) muscles. The skin area was shaved and cleaned with alcohol and cotton prior to placing the electrodes. The electrodes were placed in pairs, 2 cm apart from their centers at the point of greatest muscle belly. The ground electrode was fixed at the olecranon. The electromyo graphic data acquisition was amplified 500 times. After stored, these data were filtered (2nd-order Butterworth band-pass filter of 20-500 Hz) and rectified to calculate the signal amplitude through the integrated electromyography (iEMG). The iEMG was cal culated for the concentric muscle actions in each set and then divided by the respective time to analyze the electromyographic response during the training protocols (Ahtiainen & Hakkinen, 2009Ahtiainen J., & Hakkinen K. (2009). Strength athletes are capable to produce greater muscle activation and neural fatigue during high-intensity resistance exercise than nonathletes. Journal of Strength and Conditioning Research, 23, 1129-1134. doi:10.1519/JSC.0b013e3181aa1b72.
https://doi.org/10.1519/JSC.0b013e3181aa...
; Pincivero, Gandhi, Timmons, & Coelho, 2006Pincivero, D. M., Gandhi, V., Timmons, M. K, & Coelho, A J. (2006). Quadriceps femoris electromyogram during concentric, isometric and eccentric phases of fatiguing dynamic knee ex tensions. Journal of Biomechanics, 39, 246-254. doi: 10.1016/j.jbiomech.2004.11.023
https://doi.org/10.1016/j.jbiomech.2004....
). The mean concentric iEMG of the set was divided by the iEMG of the first repetition of each protocol performed (Ahtiainen & Hakkinen, 2009Ahtiainen J., & Hakkinen K. (2009). Strength athletes are capable to produce greater muscle activation and neural fatigue during high-intensity resistance exercise than nonathletes. Journal of Strength and Conditioning Research, 23, 1129-1134. doi:10.1519/JSC.0b013e3181aa1b72.
https://doi.org/10.1519/JSC.0b013e3181aa...
). Therefore, the mean set percentage values normalized by de first muscle action of the day were generated (mean nor malized iEMG). The electrogoniometer was used to separate the muscle actions. The electromyographic and electrogoniometer signals were synchronized and converted using an A/D board (Biovision, Wehrheim, Germany) and sampled at a frequency of 2,000 Hz. Appropriate software (DasyLab 10.0, Measurement Computing, MA, USA) was used to record and treat the data.
Blood lactate concentration
Blood samples were collected from a puncture to volunteers' left earlobes using sterile, disposable lancets. The earlobe was cleaned with neutral soap and water and then sterilized with alcohol 70% before puncturing. A 30 μl sample of blood was collected into heparinized capillary tubes, which were trans ferred into other tubes containing 60 μl of 1% sodium fluoride and then stored in a refrigerator maintained at a temperature of -20º C. Subsequently, the samples were defrosted and analyzed in duplicates on the Yellow Springs Sport 1500 Lactate Analyzer device (Yellow Springs, OH, USA).
Data analyses
Distribution normality and homogeneity of variances were initially assessed using the Shapiro-Wilk and Levene's tests, respectively. The data were expressed as means and standard deviations, and the level of significance adopted for all analyses was P < 0.05. Logarithmic transformation of the normalized iEMG data was necessary to obtain a normal distribution or homogeneity of variances. The repeated-measures two-way ANOVA (training protocol x sets) was used for each muscle separately in the electromyographic analyses, as well as to compare lactate concentration. When necessary, a post hoc Tukey's test was used to identify the differences reported in the ANOVAs. Related-sample t -tests were used to compare repetition durations, ranges of motion, and angular velocities between protocols. Post hoc power analysis was performed for both ANOVA and t -test. The Intraclass Correlation Coefficient (ICC[3, 1]) of the concentric electromyographic activity found in the first repetition of Experimental Sessions 3 and 4 was calculated; these inter-session values were 0.85 for the triceps brachii and 0.93 for the pectoralis major.
Results
The main effects of protocol (F1,14 = 5.69; P = 0.032; power = 0.60) and set (F1,14 = 17.01; P < 0.001; power = 0.99) were significant for pectoralis major muscle activation. Our data show that the normalized iEMG in 2:4 protocol was higher than the 2:2 protocol, as well as the protocols increased the normalized iEMG throughout the sets. Post hoc comparisons indicated a progressive increase in muscle activation over the sets (Figure 1). No significant interaction was observed between protocol and set (F2,28 = 1.37; P = 0.271; power = 0.17).
Mean normalized iEMG of the pectoralis major muscle for each training protocol (values normalized using the 1st repetition of the day).
The main effects of protocol (F1,14 = 5.48; P = 0.034; power = 0.59) and set (F1,14 = 10.66; P = 0.004; power = 0.94) were also significant for the triceps brachii muscle. Similarly, 2:4 protocol showed higher normalized iEMG than the 2:2 protocol, and the protocols increased the normalized iEMG throughout the sets. However, Tukey post hoc test found differences only when the 3rd set is compared with the 1st and 2nd sets (Figure 2). No significant interaction was observed between protocol and set (F2,28 = 1.53; P = 0.234; power = 0.19).
Mean normalized iEMG of the triceps brachii muscle for each training protocol (values normalized using the 1st repetition of the day).
The main effects of protocol and set were significant with regard to blood lactate concentration. In addition, a significant interaction effect was observed between protocol and set (F3,42 = 46.59; P < 0.001; power = 1.00). Figure 3 shows the blood lactate concentrations for the different training protocols. These results indicate that blood lactate concentrations increased for both protocols throughout the sets, and blood lactate concen tration was higher in the 2:4 protocol than the 2:2 protocol for all sets, except during rest measurement.
Blood lactate concentration at rest and after each set with regard to the 2:2 and 2:4 protocols.
The protocols 2:2 and 2:4 showed, respectively, repetition durations of 4.03 ± 0.05 s (1.84 ± 0.08 s concentric; 2.19 ± 0.08 s eccentric), and 6.03 ± 0.06 s (2.06 ± 0.08 s concentric; 3.97 ± 0.08 s eccentric), and average angular velocities of 0.80 ± 0.07 rad∙s-1 (0.73 ± 0.08 rad∙s-1 concentric; 0.88 ± 0.11 rad∙s-1 eccentric), and 0.59 ± 0.09 rad∙s-1 (0.40 ± 0.05 rad∙s-1 concentric; 0.78 ± 0.11 rad∙s-1 eccentric). As expected, protocol 2:2 showed shorter mean repetition duration than protocol 2:4 (P < 0.001; power = 1.00). The average angular velocity was lower in protocol 2:4 than in protocol 2:2 (P < 0.001; power = 1.00). Finally, no differences were found on the average range of motion between the protocols 2:2 and 2:4 (91.6 ± 9.9° vs. 90.4 ± 10.3°, respectively; P = 0.24; power = 0.20).
Discussion
This study examined whether longer repetition duration (2:2 vs. 2:4) would result in greater electromyographic responses and higher blood lactate concentrations in matched training protocols. The results showed that normalized iEMG response for concentric actions was greater in the 2:4 protocol than in the 2:2 protocol for the pectoralis major and triceps brachii muscles. Furthermore, normalized iEMG response increased throughout the sets for both protocols. Blood lactate concen tration was higher with longer repetition duration and increased throughout all sets.
In the present study, a higher normalized iEMG response in concentric muscle actions indicated that an increase in muscle activation was necessary to maintain the level of force necessary to complete the 2:4 protocol. Considering that the training protocols were matched, this result may be attributed to a longer duration of eccentric muscle action in the 2:4 pro tocol when compared to the 2:2 protocol. This longer duration denotes a decreased movement velocity during eccentric action, which has been linked to a decreased use of elastic energy in the stretch-shortening cycle (SSC) (Bosco, Komi, & Ito, 1981Bosco, C., Komi, P., & Ito, A. (1981). Prestretch potentiation of human skeletal muscle during ballistic movement. Acta Physiologica Scandinavica, 111, 135-140. doi: 10.1111/j.1748-1716.1981.tb06716.x
https://doi.org/10.1111/j.1748-1716.1981...
; Cronin, Mcnair, & Marshall, 2002Cronin, J. B., Mcnair, P. J., & Marshall, R. N. (2002). Power absorption and production during slow, large-amplitude stretch-shorten cycle motions. European Journal of Applied Physiology, 87, 59-65. doi: 10.1007/s00421-002-0585-5
https://doi.org/10.1007/s00421-002-0585-...
). In addition, previous stud ies have shown that training protocols performed with longer duration of eccentric action caused an increase in eccentric-con centric transition time (Martins-Costa, Diniz, Machado, Lima, & Chagas, 2012Martins-Costa, H. C., Diniz, R. R., Machado, S. C., Lima, F. V., & Chagas, M. H. (2012). Impacto de diferentes velocidades de mov imento no tempo de transição entre ações musculares excêntricas e concêntricas no exercício supino [Impact of different movement velocities on coupling time between eccentric and concentric mus cle actions in the bench press exercise]. Motricidade, 8(Suppl.2), 365-372. doi: 10.6063/motricidade.8(0).649
https://doi.org/10.6063/motricidade.8(0)...
; Sampson et al. , 2014Sampson, J. A., Donohoe, A., & Groeller, H. (2014). Effect of concen tric and eccentric velocity during heavy-load non-ballistic elbow flexion resistance exercise. Journal of Science and Medicine in Sport. 17, 306-311. doi: 10.1016/j.jsams.2013.04.012
https://doi.org/10.1016/j.jsams.2013.04....
). Wilson, Elliott, and Wood (1991)Wilson, G. J., Elliott, B. C., & Wood, G. A. (1991). The effect on performance of imposing a delay during a stretch-shorten cycle movement. Medicine and Science in Sports and Exercise, 23, 364-370. doi: 10.1249/00005768-199103000-00016
https://doi.org/10.1249/00005768-1991030...
showed that an increased transition time between eccen tric and concentric actions (from 0.6 s to 1.27 s) resulted in lower impulse values early in the concentric muscle action following a decreased use of elastic energy, which reduced muscle force production. Thus, differences in transition time associated with the increase in the duration of the eccentric action in the 2:4 protocol might also have negatively affected the use of elastic energy. Given that the external resistance to overcome the con centric actions was the same in all repetitions for both protocols, it is possible that the participation of additional motor units was required in the 2:4 protocol to maintain strength performance during the concentric muscle action. Similar results were found by Burd et al. (2012)Burd, N. A., Andrews, R. J., West, D. W., Little, J. P., Cochran, A. J., Hector, A. J., ... & Phillips, S. M. (2012). Muscle time under tension during resistance exercise stimulates differ ential muscle protein sub-fractional synthetic responses in men. The Journal of Physiology, 590, 351-362. doi: 10.1113/jphysiol.2011.221200
https://doi.org/10.1113/jphysiol.2011.22...
. Although these authors analyzed different repetition durations (12 s vs. 2 s) of those compared in this study (4 s vs. 6 s), the longer duration used by Burd et al. (2012)Burd, N. A., Andrews, R. J., West, D. W., Little, J. P., Cochran, A. J., Hector, A. J., ... & Phillips, S. M. (2012). Muscle time under tension during resistance exercise stimulates differ ential muscle protein sub-fractional synthetic responses in men. The Journal of Physiology, 590, 351-362. doi: 10.1113/jphysiol.2011.221200
https://doi.org/10.1113/jphysiol.2011.22...
also caused an increase in electromyographic amplitude, especially at the end of each set. Therefore, the data from the present study reinforce the expectation that an increase in the duration of muscle contraction at submaximal intensities can promote higher levels of muscle activation, a neuromuscular behavior also commonly seen in the electromyographic amplitude anal ysis of isometric sustained submaximal contractions (Hunter et al. , 2004Hunter, S. K., Duchateau, J., & Enoka, R. M. (2004). Muscle fatigue and the mechanisms of task failure. Exercise and Sport Science Review, 32, 44-49. doi: 10.1097/00003677-200404000-00002
https://doi.org/10.1097/00003677-2004040...
; Suzuki et al. , 2002Suzuki, H., Conwit, R., Stashuk. D., Santarsiero, L., & Metter. E. (2002). Relationships between surface-detected EMG signals and motor unit activation. Medicine and Science in Sports and Exercise, 34, 1509-1517. doi:10.1249/01.MSS.0000027711.31651.AF
https://doi.org/10.1249/01.MSS.000002771...
). As mentioned previously, this rise in the amplitude of electromyographic signal is at least in part an indicative of the occurrence of increased recruitment of motor units in an attempt to preserve the desired strength levels (Hunter et al. , 2004Hunter, S. K., Duchateau, J., & Enoka, R. M. (2004). Muscle fatigue and the mechanisms of task failure. Exercise and Sport Science Review, 32, 44-49. doi: 10.1097/00003677-200404000-00002
https://doi.org/10.1097/00003677-2004040...
), which in turn has been pointed out as an important neuromuscular response related to increased muscle strength and hypertrophy adaptation (Schoenfeld, 2013Schoenfeld, B. J. (2013). Is there a minimum intensity threshold for resistance training-induced hypertrophic adaptations? Sports Medicine, 43,1279-1288. doi: 10.1007/s40279-013-0088-z.
https://doi.org/10.1007/s40279-013-0088-...
; Spiering et al. , 2008Spiering, B. A., Kraemer, W. J., Anderson, J . M., Armstrong, L. E., Nindl, B. C., Volek, J. S., & Maresh, C. M. (2008). Resistance exercise biology: manipulation of resistance exercise programme variables determines the responses of cellular and molecu lar signalling pathways. Sports Medicine. 38, 527-540. doi: 10.2165/00007256-200838070-00001
https://doi.org/10.2165/00007256-2008380...
). However, other factors, such as the increased firing frequency and synchronization of motor units, may also influence the electromyographic amplitude (Hunter et al. , 2004Hunter, S. K., Duchateau, J., & Enoka, R. M. (2004). Muscle fatigue and the mechanisms of task failure. Exercise and Sport Science Review, 32, 44-49. doi: 10.1097/00003677-200404000-00002
https://doi.org/10.1097/00003677-2004040...
; Suzuki et al. , 2002Suzuki, H., Conwit, R., Stashuk. D., Santarsiero, L., & Metter. E. (2002). Relationships between surface-detected EMG signals and motor unit activation. Medicine and Science in Sports and Exercise, 34, 1509-1517. doi:10.1249/01.MSS.0000027711.31651.AF
https://doi.org/10.1249/01.MSS.000002771...
).
In contrast, previous studies have reported different results (Sakamoto & Sinclair, 2012Sakamoto, A., & Sinclair, P. J. (2012). Muscle activations under vary ing lifting speeds and intensities during bench press. European Journal of Applied Physiology, 112:1015-1025. doi: 10.1007/S00421-011-2059-0
https://doi.org/10.1007/S00421-011-2059-...
; Sampson et al. 2014Sampson, J. A., Donohoe, A., & Groeller, H. (2014). Effect of concen tric and eccentric velocity during heavy-load non-ballistic elbow flexion resistance exercise. Journal of Science and Medicine in Sport. 17, 306-311. doi: 10.1016/j.jsams.2013.04.012
https://doi.org/10.1016/j.jsams.2013.04....
). However, the studies of Sakamoto and Sinclair (2012)Sakamoto, A., & Sinclair, P. J. (2012). Muscle activations under vary ing lifting speeds and intensities during bench press. European Journal of Applied Physiology, 112:1015-1025. doi: 10.1007/S00421-011-2059-0
https://doi.org/10.1007/S00421-011-2059-...
and Sampson et al. (2014)Sampson, J. A., Donohoe, A., & Groeller, H. (2014). Effect of concen tric and eccentric velocity during heavy-load non-ballistic elbow flexion resistance exercise. Journal of Science and Medicine in Sport. 17, 306-311. doi: 10.1016/j.jsams.2013.04.012
https://doi.org/10.1016/j.jsams.2013.04....
investigated one set of exercise to muscle failure, probably resulting in a non-equalized volume (number of repetitions) in each of the experimental conditions, so that the observed differences on electromyographic activity could not be mainly attributed to repetition duration.
The present investigation revealed also an increase in the electromyographic activity throughout the sets, findings ob served in other studies (Smilios et al. , 2010Smilios, I., Häkkinen, K., & Tokmakidis, S. P. (2010). Power output and electromyographic activity during and after a moderate load mus cular endurance session. The Journal of Strength and Conditioning Research, 24, 2122-2131. doi: 10.1519/JSC.0b013e3181a5bc44.
https://doi.org/10.1519/JSC.0b013e3181a5...
; Walker et al. , 2011Walker, S., Taipale, R.S., Nyman, K., Kraemer, W. J., & Häkkinen, K. (2011). Neuromuscular and hormonal responses to constant and variable resistance loadings. Medicine and Science in Sports and Exercise. 43, 26-33. doi: 10.1249/MSS.0b013e3181e71bcb.
https://doi.org/10.1249/MSS.0b013e3181e7...
). As previously highlighted, participants should overcome the same external resistance for all repetitions. Thus, the increased electromyographic activity may reflect the insufficient rest in terval for adequate recovery of some previously activated motor units, thereby demanding the participation of additional motor units in the next set. Mendez-Villanueva, Baudry, Riley, and Rudroff (2009)Mendez-Villanueva, A., Baudry, S., Riley, Z. A., & Rudroff, T. (2009). Influence of rest length on muscle activation during submaximal in termittent contractions with the elbow flexor muscles. The Journal of Sports Medicine and Physical Fitness, 49, 255-264. Retrieved from http://www.minervamedica.it/en/journals/sports-med-phys ical-fitness/article.php?cod=R40Y2009N03A0255
http://www.minervamedica.it/en/journals/...
also suggested this mechanism after finding an increase in electromyographic activity across repeated isometric actions with a reduced pause for recovery.
Similarly to the electromyographic responses, blood lac tate concentration increased along the sets in both protocols, result already seen in previous investigations (Lacerda et al. , 2016Lacerda, L., Martins Costa, H., Diniz, R., Lima, F., Pereira Andrade, A., Tourino, F., ... Chagas, M. (2016). Variations in repetition duration and repetition numbers influences muscular activation and blood lactate response in protocols equalized by time under tension. The Journal of Strength and Conditioning Research, 30, 251-218. doi: 10.1519/JSC.0000000000001044
https://doi.org/10.1519/JSC.000000000000...
; Smilios et al. , 2010Smilios, I., Häkkinen, K., & Tokmakidis, S. P. (2010). Power output and electromyographic activity during and after a moderate load mus cular endurance session. The Journal of Strength and Conditioning Research, 24, 2122-2131. doi: 10.1519/JSC.0b013e3181a5bc44.
https://doi.org/10.1519/JSC.0b013e3181a5...
; Wirtz , Wahl, Kleinöder, & Mester, 2014Wirtz, N., Wahl, P., Kleinöder, H., & Mester, J. (2014). Lactate Kinetics during Multiple Set Resistance Exercise. Journal of Sports Science and Medicine, 13, 73-77.). These data reinforce the expectation of high anaerobic demand required during the execution of each set of the resis tance training protocols (Wirtz et al. , 2014Wirtz, N., Wahl, P., Kleinöder, H., & Mester, J. (2014). Lactate Kinetics during Multiple Set Resistance Exercise. Journal of Sports Science and Medicine, 13, 73-77.). With regard to the comparison of protocols, higher blood lactate concentration values were recorded in the 2:4 protocol compared with the 2:2 protocol, confirming data from previous studies that also equalized the training protocols (Mazzetti, Douglass, Yocum, & Harber, 2007Mazzetti, S., Douglass, M. S., Yocum, A., & Harber, M. (2007). Effect of explosive versus slow contractions and exercise intensity on energy expenditure. Medicine and Science in Sports Exercise, 39, 1291-1301. doi: 10.1249/mss.0b013e318058a603
https://doi.org/10.1249/mss.0b013e318058...
). Given the higher normalized iEMG value found in the concentric muscle actions of the 2:4 protocol, additional motor units with higher glycolytic capacities were presumably recruited (Eckhardt et al. , 2011Eckhardt, H., Wollny, R., Müller, H., Bärtsch, P., & Friedmann-Bette, B. (2011). Enhanced myofiber recruitment during exhaustive squatting performed as whole-body vibration exercise. Journal of Strength and Conditioning Research, 25, 1120-1125. doi: 10.1519/JSC.0b013e3181d09e0e.
https://doi.org/10.1519/JSC.0b013e3181d0...
; Takarada et al. , 2000Takarada, Y., Takazawa, H., Sato, Y., Takebayashi, S., Tanaka, Y., & Ishii, N. (2000). Effects of resistance exercise combined with moderate vascular occlusion on muscular function in humans. Journal of Applied Physiology, 88, 2097-2106.; Walker et al. , 2011Walker, S., Taipale, R.S., Nyman, K., Kraemer, W. J., & Häkkinen, K. (2011). Neuromuscular and hormonal responses to constant and variable resistance loadings. Medicine and Science in Sports and Exercise. 43, 26-33. doi: 10.1249/MSS.0b013e3181e71bcb.
https://doi.org/10.1249/MSS.0b013e3181e7...
), which might promote an increase in blood lactate production compared with the 2:2 protocol. In contrast, Headley et al. (2011)Headley, S. A., Henry, K., Nindl, B. C., Thompson, B. A., Kraemer, W. J., & Jones, M. (2011). Effects of lifting tempo on one repetition maximum and hormonal responses to a bench press protocol. The Journal of Strength and Conditioning Research, 25, 406-413. doi: 10.1519/JSC.0b013e3181bf053b
https://doi.org/10.1519/JSC.0b013e3181bf...
found no difference in blood lactate concentration when comparing the protocols (4 s vs. 6 s), although the protocols performed were not matched by training volume, a training load component that also interfere with the blood lactate responses (Buitrago, Wirtz, Flenker, & Kleinöder, 2014Buitrago, S., Wirtz, N., Flenker, U., & Kleinöder H. (2014). Physiological and metabolic responses as function of the mechan ical load in resistance exercise. Applied Physiology, Nutrition, and Metabolism, 39, 345-350. doi: 10.1139/apnm-2013-0214.
https://doi.org/10.1139/apnm-2013-0214...
; Lacerda et al. , 2016Lacerda, L., Martins Costa, H., Diniz, R., Lima, F., Pereira Andrade, A., Tourino, F., ... Chagas, M. (2016). Variations in repetition duration and repetition numbers influences muscular activation and blood lactate response in protocols equalized by time under tension. The Journal of Strength and Conditioning Research, 30, 251-218. doi: 10.1519/JSC.0000000000001044
https://doi.org/10.1519/JSC.000000000000...
). On other hand, in the study of González-Badillo et al. (2014)González-Badillo, J.J., Rodríguez-Rosell, D., Sánchez-Medina, L., Gorostiaga, E. M, & Pareja-Blanco, F. (2014). Maximal intended velocity training induces greater gains in bench press performance than deliberately slower half-velocity training. European Journal of Sport Science, 14, 772-781. doi: 10.1080/17461391.2014.905987
https://doi.org/10.1080/17461391.2014.90...
training protocols were equalized and higher lactate values were obtained at the end of the protocols of shorter repetition duration (higher repetition velocity). The authors explained this result by arguing that greater force production would be required to perform faster movements, which would also result in a greater recruitment of muscle fibers with higher glycolytic potential.
These controversial results may be related to the method ological differences, for example, the time under tension adopted in different studies. While in the research of González-Badillo et al. (2014)González-Badillo, J.J., Rodríguez-Rosell, D., Sánchez-Medina, L., Gorostiaga, E. M, & Pareja-Blanco, F. (2014). Maximal intended velocity training induces greater gains in bench press performance than deliberately slower half-velocity training. European Journal of Sport Science, 14, 772-781. doi: 10.1080/17461391.2014.905987
https://doi.org/10.1080/17461391.2014.90...
the approximate time under tension per set performed in the longer repetition duration protocol was 18 s, in the study of Mazzetti et al. (2007)Mazzetti, S., Douglass, M. S., Yocum, A., & Harber, M. (2007). Effect of explosive versus slow contractions and exercise intensity on energy expenditure. Medicine and Science in Sports Exercise, 39, 1291-1301. doi: 10.1249/mss.0b013e318058a603
https://doi.org/10.1249/mss.0b013e318058...
the protocol of longer repetition du ration resulted in 32 s. In the present study, time under tension was 36 s for the 2:4 protocol. Therefore, possibly the training loads imposed by longer repetition duration protocols adopted in current study, as well as in Mazzetti et al. (2007)Mazzetti, S., Douglass, M. S., Yocum, A., & Harber, M. (2007). Effect of explosive versus slow contractions and exercise intensity on energy expenditure. Medicine and Science in Sports Exercise, 39, 1291-1301. doi: 10.1249/mss.0b013e318058a603
https://doi.org/10.1249/mss.0b013e318058...
, have promoted greater degree of fatigue than the longer repetition duration pro tocol of the González-Badillo et al. (2014)González-Badillo, J.J., Rodríguez-Rosell, D., Sánchez-Medina, L., Gorostiaga, E. M, & Pareja-Blanco, F. (2014). Maximal intended velocity training induces greater gains in bench press performance than deliberately slower half-velocity training. European Journal of Sport Science, 14, 772-781. doi: 10.1080/17461391.2014.905987
https://doi.org/10.1080/17461391.2014.90...
study. Accordingly, greater recruitment of motor units may have occurred during exercise (Hunter et al. , 2004Hunter, S. K., Duchateau, J., & Enoka, R. M. (2004). Muscle fatigue and the mechanisms of task failure. Exercise and Sport Science Review, 32, 44-49. doi: 10.1097/00003677-200404000-00002
https://doi.org/10.1097/00003677-2004040...
), as well as increased blood lactate responses (Gladden, 2004Gladden, L. (2004). Lactate metabolism: a new paradigm for the third millennium. The Journal of Physiology, 558, 5-30.).
Conclusion
This study revealed that longer repetition duration with similar concentric duration, but higher eccentric action duration, increases the blood lactate response and muscle activation. These findings indicate higher physiological demands with longer repetition duration. The relevance of this data is that increasing repetition duration should be considered as a pro gression in resistance training.
References
- Ahtiainen J., & Hakkinen K. (2009). Strength athletes are capable to produce greater muscle activation and neural fatigue during high-intensity resistance exercise than nonathletes. Journal of Strength and Conditioning Research, 23, 1129-1134. doi:10.1519/JSC.0b013e3181aa1b72.
» https://doi.org/10.1519/JSC.0b013e3181aa1b72 - American College of Sports Medicine. (2009). Position stand: Progression models in resistance training for healthy adults. Medicine and Science in Sports and Exercise, 41, 687-708. doi:10.1249/MSS.0b013e3181915670.
» https://doi.org/10.1249/MSS.0b013e3181915670 - Bird, S., Tarpenning, M., & Marino F. (2005). Designing resistance training programmes to enhance muscular fitness: a review of the acute programme variables. Sports Medicine, 35, 841-851. doi: 10.2165/00007256-200535100-00002
» https://doi.org/10.2165/00007256-200535100-00002 - Bosco, C., Komi, P., & Ito, A. (1981). Prestretch potentiation of human skeletal muscle during ballistic movement. Acta Physiologica Scandinavica, 111, 135-140. doi: 10.1111/j.1748-1716.1981.tb06716.x
» https://doi.org/10.1111/j.1748-1716.1981.tb06716.x - Buitrago, S., Wirtz, N., Flenker, U., & Kleinöder H. (2014). Physiological and metabolic responses as function of the mechan ical load in resistance exercise. Applied Physiology, Nutrition, and Metabolism, 39, 345-350. doi: 10.1139/apnm-2013-0214.
» https://doi.org/10.1139/apnm-2013-0214 - Burd, N. A., Andrews, R. J., West, D. W., Little, J. P., Cochran, A. J., Hector, A. J., ... & Phillips, S. M. (2012). Muscle time under tension during resistance exercise stimulates differ ential muscle protein sub-fractional synthetic responses in men. The Journal of Physiology, 590, 351-362. doi: 10.1113/jphysiol.2011.221200
» https://doi.org/10.1113/jphysiol.2011.221200 - Cook, S., Murphy, B., & Labarbera K. (2013). Neuromuscular function after a bout of low-load blood flow-restricted exercise. Medicine and Science in Sports and Exercise, 45,67-74. doi: 10.1249/MSS.0b013e31826c6fa8.
» https://doi.org/10.1249/MSS.0b013e31826c6fa8 - Cronin, J. B., Mcnair, P. J., & Marshall, R. N. (2002). Power absorption and production during slow, large-amplitude stretch-shorten cycle motions. European Journal of Applied Physiology, 87, 59-65. doi: 10.1007/s00421-002-0585-5
» https://doi.org/10.1007/s00421-002-0585-5 - Diniz, R. C., Martins-Costa, H. C., Machado, S. C., Lima, F. V., & Chagas, M. H. (2014). Repetition duration influences ratings of perceived exertion. Perceptual and Motor Skills, 118, 261-273. doi: 10.2466/03.06.PMS.118k11w6
» https://doi.org/10.2466/03.06.PMS.118k11w6 - Eckhardt, H., Wollny, R., Müller, H., Bärtsch, P., & Friedmann-Bette, B. (2011). Enhanced myofiber recruitment during exhaustive squatting performed as whole-body vibration exercise. Journal of Strength and Conditioning Research, 25, 1120-1125. doi: 10.1519/JSC.0b013e3181d09e0e.
» https://doi.org/10.1519/JSC.0b013e3181d09e0e - Gladden, L. (2004). Lactate metabolism: a new paradigm for the third millennium. The Journal of Physiology, 558, 5-30.
- González-Badillo, J.J., Rodríguez-Rosell, D., Sánchez-Medina, L., Gorostiaga, E. M, & Pareja-Blanco, F. (2014). Maximal intended velocity training induces greater gains in bench press performance than deliberately slower half-velocity training. European Journal of Sport Science, 14, 772-781. doi: 10.1080/17461391.2014.905987
» https://doi.org/10.1080/17461391.2014.905987 - Headley, S. A., Henry, K., Nindl, B. C., Thompson, B. A., Kraemer, W. J., & Jones, M. (2011). Effects of lifting tempo on one repetition maximum and hormonal responses to a bench press protocol. The Journal of Strength and Conditioning Research, 25, 406-413. doi: 10.1519/JSC.0b013e3181bf053b
» https://doi.org/10.1519/JSC.0b013e3181bf053b - Hermens, H. J., Freriks, B., Disselhorst-Klug, C., & Rau, G.J. (2000). Development of recommendations for SEMG sensors and sen sor placement procedures. Journal of Electromyography and Kinesiology, 10, 361-374. doi: 0.1016/S1050-6411(00)00027-4
- Hunter, S. K., Duchateau, J., & Enoka, R. M. (2004). Muscle fatigue and the mechanisms of task failure. Exercise and Sport Science Review, 32, 44-49. doi: 10.1097/00003677-200404000-00002
» https://doi.org/10.1097/00003677-200404000-00002 - Lacerda, L., Martins Costa, H., Diniz, R., Lima, F., Pereira Andrade, A., Tourino, F., ... Chagas, M. (2016). Variations in repetition duration and repetition numbers influences muscular activation and blood lactate response in protocols equalized by time under tension. The Journal of Strength and Conditioning Research, 30, 251-218. doi: 10.1519/JSC.0000000000001044
» https://doi.org/10.1519/JSC.0000000000001044 - Martins-Costa, H. C., Diniz, R. R., Machado, S. C., Lima, F. V., & Chagas, M. H. (2012). Impacto de diferentes velocidades de mov imento no tempo de transição entre ações musculares excêntricas e concêntricas no exercício supino [Impact of different movement velocities on coupling time between eccentric and concentric mus cle actions in the bench press exercise]. Motricidade, 8(Suppl.2), 365-372. doi: 10.6063/motricidade.8(0).649
» https://doi.org/10.6063/motricidade.8(0).649 - Mazzetti, S., Douglass, M. S., Yocum, A., & Harber, M. (2007). Effect of explosive versus slow contractions and exercise intensity on energy expenditure. Medicine and Science in Sports Exercise, 39, 1291-1301. doi: 10.1249/mss.0b013e318058a603
» https://doi.org/10.1249/mss.0b013e318058a603 - Mendez-Villanueva, A., Baudry, S., Riley, Z. A., & Rudroff, T. (2009). Influence of rest length on muscle activation during submaximal in termittent contractions with the elbow flexor muscles. The Journal of Sports Medicine and Physical Fitness, 49, 255-264. Retrieved from http://www.minervamedica.it/en/journals/sports-med-phys ical-fitness/article.php?cod=R40Y2009N03A0255
» http://www.minervamedica.it/en/journals/sports-med-phys ical-fitness/article.php?cod=R40Y2009N03A0255 - Moritani, T., Tanaka, H., Yoshida, T., Ishii, C., Yoshida, T., & Shindo, M. (1984). Relationship between myoelectric signals and blood lactate during incremental forearm exercise. American Journal of Physical Medicine, 63, 122-132.
- Pincivero, D. M., Gandhi, V., Timmons, M. K, & Coelho, A J. (2006). Quadriceps femoris electromyogram during concentric, isometric and eccentric phases of fatiguing dynamic knee ex tensions. Journal of Biomechanics, 39, 246-254. doi: 10.1016/j.jbiomech.2004.11.023
» https://doi.org/10.1016/j.jbiomech.2004.11.023 - Sampson, J. A., Donohoe, A., & Groeller, H. (2014). Effect of concen tric and eccentric velocity during heavy-load non-ballistic elbow flexion resistance exercise. Journal of Science and Medicine in Sport. 17, 306-311. doi: 10.1016/j.jsams.2013.04.012
» https://doi.org/10.1016/j.jsams.2013.04.012 - Sakamoto, A., & Sinclair, P. J. (2012). Muscle activations under vary ing lifting speeds and intensities during bench press. European Journal of Applied Physiology, 112:1015-1025. doi: 10.1007/S00421-011-2059-0
» https://doi.org/10.1007/S00421-011-2059-0 - Schoenfeld, B. J. (2013). Is there a minimum intensity threshold for resistance training-induced hypertrophic adaptations? Sports Medicine, 43,1279-1288. doi: 10.1007/s40279-013-0088-z.
» https://doi.org/10.1007/s40279-013-0088-z - Schoenfeld, B.J., Ogborn, D., & Krieger, J. (2015). Effect of repetition duration during resistance training on muscle hypertrophy: a sys tematic review and meta-analysis. Sports Medicine, 45, 577-585. doi: 10.1007/s40279-015-0304-0.
» https://doi.org/10.1007/s40279-015-0304-0 - Smilios, I., Häkkinen, K., & Tokmakidis, S. P. (2010). Power output and electromyographic activity during and after a moderate load mus cular endurance session. The Journal of Strength and Conditioning Research, 24, 2122-2131. doi: 10.1519/JSC.0b013e3181a5bc44.
» https://doi.org/10.1519/JSC.0b013e3181a5bc44 - Spiering, B. A., Kraemer, W. J., Anderson, J . M., Armstrong, L. E., Nindl, B. C., Volek, J. S., & Maresh, C. M. (2008). Resistance exercise biology: manipulation of resistance exercise programme variables determines the responses of cellular and molecu lar signalling pathways. Sports Medicine. 38, 527-540. doi: 10.2165/00007256-200838070-00001
» https://doi.org/10.2165/00007256-200838070-00001 - Suzuki, H., Conwit, R., Stashuk. D., Santarsiero, L., & Metter. E. (2002). Relationships between surface-detected EMG signals and motor unit activation. Medicine and Science in Sports and Exercise, 34, 1509-1517. doi:10.1249/01.MSS.0000027711.31651.AF
» https://doi.org/10.1249/01.MSS.0000027711.31651.AF - Takarada, Y., Takazawa, H., Sato, Y., Takebayashi, S., Tanaka, Y., & Ishii, N. (2000). Effects of resistance exercise combined with moderate vascular occlusion on muscular function in humans. Journal of Applied Physiology, 88, 2097-2106.
- Wakahara, T., Fukutani, A., Kawakami, Y., & Yanai, T. (2013). Nonuniform muscle hypertrophy: its relation to muscle activation in training session. Medicine and Science in Sports and Exercise, 45, 2158-2165. doi: 10.1249/MSS.0b013e3182995349.
» https://doi.org/10.1249/MSS.0b013e3182995349 - Walker, S., Taipale, R.S., Nyman, K., Kraemer, W. J., & Häkkinen, K. (2011). Neuromuscular and hormonal responses to constant and variable resistance loadings. Medicine and Science in Sports and Exercise. 43, 26-33. doi: 10.1249/MSS.0b013e3181e71bcb.
» https://doi.org/10.1249/MSS.0b013e3181e71bcb - Watanabe, Y., Madarame, H., Ogasawara, R., Nakazato, K., & Ishii, N. (2014). Effect of very low-intensity resistance training with slow movement on muscle size and strength in healthy older adults. Clinical Physiology and Functional Imaging, 34, 463-470. doi: 10.1111/cpf.12117.
» https://doi.org/10.1111/cpf.12117 - Wernbom, M., Augustsson, J., & Thomeé, R. (2007). The influence of frequency, intensity, volume and mode of strength training on whole muscle cross-sectional area in humans. Sports Medicine, 37, 225-264. doi: 10.2165/00007256-200737030-00004
» https://doi.org/10.2165/00007256-200737030-00004 - Wilson, G. J., Elliott, B. C., & Wood, G. A. (1991). The effect on performance of imposing a delay during a stretch-shorten cycle movement. Medicine and Science in Sports and Exercise, 23, 364-370. doi: 10.1249/00005768-199103000-00016
» https://doi.org/10.1249/00005768-199103000-00016 - Wirtz, N., Wahl, P., Kleinöder, H., & Mester, J. (2014). Lactate Kinetics during Multiple Set Resistance Exercise. Journal of Sports Science and Medicine, 13, 73-77.
- Yue, G., Alexander, A., Laidlaw, D., Gmitro, A., Unger, E., & Enoka, R. (1994). Sensitivity of muscle proton spin-spin relaxation time as an index of muscle activation. Journal of Applied Physiology. 77, 84-92.
Publication Dates
-
Publication in this collection
Mar 2016
History
-
Received
29 Oct 2014 -
Accepted
30 Nov 2015

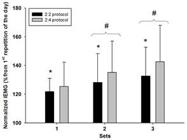



 * Different from the 2:4 protocol (main effect); # Different from previous set (main effect); iEMG: integrated electromyographic signal.
* Different from the 2:4 protocol (main effect); # Different from previous set (main effect); iEMG: integrated electromyographic signal.
 *Different from the 2:4 protocol (main effect); # Different from previous sets (main effect); iEMG: integrated electromyographic signal.
*Different from the 2:4 protocol (main effect); # Different from previous sets (main effect); iEMG: integrated electromyographic signal.
 * Different from the 2:4 protocol in the respective set; ‡ Different from the other sets in the respective protocols; $ Different from the 1st set in the respective protocols; # Different from the value at rest in the respective protocols.
* Different from the 2:4 protocol in the respective set; ‡ Different from the other sets in the respective protocols; $ Different from the 1st set in the respective protocols; # Different from the value at rest in the respective protocols.