Abstract
Sugarcane bagasse from agro-industrial residues was autoclaved at 121 °C for 15 min, treated with 2% NaOH and activated with 2.5% glutaraldehyde for the immobilization of soybean β-glucosidase. Scanning electron microscopy, energy dispersive X-ray spectroscopy and Fourier transform infrared spectroscopy characterized and confirmed the immobilization of the β-glucosidase on the sugarcane bagasse. The immobilization efficiency was influenced by the type of bagasse modification and was 99% at maximum. The optimum immobilization conditions were 1 mg mL-1 protein, pH 7.0, 2.5% glutaraldehyde, 110 rpm and 8 h of incubation at 4 °C. The immobilized system could be reused for 15 cycles without the complete loss of activity. The thermal stability indicated a residual activity of 15% after 180 min incubation at 70 °C. The conversion efficiency of glucosides to aglycones in commercial soymilk by β-glucosidase immobilized on sugarcane bagasse was evaluated and the total aglycone content increased by 23.8% after incubation at 50 °C for 120 min.
Keywords:
Soybean; β-glucosidase; Sugarcane bagasse; Enzyme immobilization; Isoflavones; Commercial soymilk
Resumo
O bagaço de cana-de-açúcar, de resíduos agroindustriais, foi autoclavado a 121 °C durante 15 min, tratado com NaOH a 2% e ativado com 2,5% de glutaraldeído, para imobilização de β-glicosidase de soja. Análises de microscopia eletrônica de varredura, espectroscopia de raios X por dispersão de energia e espectroscopia de infravermelho por transformada de Fourier caracterizaram e confirmaram a imobilização de β-glicosidase em bagaço de cana. A eficiência da imobilização foi influenciada pelo tipo de modificação do bagaço com a máxima de 99%. As condições de imobilização ótimas foram de 1 mg de proteína mL-1, pH 7,0, 2,5% de glutaraldeído, 110 rpm e 8 h de incubação a 4 °C. O sistema imobilizado pode ser reutilizado por 15 ciclos sem perda completa de atividade. A estabilidade térmica indicou uma atividade residual de 15% após uma incubação de 180 min a 70 °C. A conversão de eficiência de isoflavonas glicosídicas em agliconas em um leite de soja comercial por β-glicosidase imobilizada em bagaço de cana foi avaliada e o teor de agliconas totais aumentou em 23,8% após incubação a 50 °C durante 120 minutos.
Palavras-chave:
Soja; β-glicosidase; Bagaço de cana; Imobilização de enzima; Isoflavonas; Extrato de soja comercial
1 Introduction
Brazil is the largest sugarcane producer in the world. The estimated average productivity of sugarcane for the 2017-2018 harvest was 73,728 kg/ha with an estimated 646.34 million tons of sugarcane crushed (Companhia Nacional de Abastecimento, 2017Companhia Nacional de Abastecimento – CONAB. (2017). Levantamento cana-de açúcar safra 2017/2018. Brasília. Retrieved in 2017, November 9, from http://www.conab.gov.br/OlalaCMS/uploads/arquivos/17_08_24_08_59_54_boletim_cana_portugues_-_2o_lev_-_17-18.pdf
http://www.conab.gov.br/OlalaCMS/uploads...
). Sugarcane bagasse (SCB) is one of the by-products of this process with an average production of 260 kilograms per ton of sugarcane crushed (União das Indústrias de Cana-de-Açúcar, 2011União das Indústrias de Cana-de-Açúcar – UNICA. (2011). Um fantástico potencial. São Paulo. Retrieved in 2014, May 4, from http://www.unica.com.br/convidados/5337876920338419546/um-fantastico-potencial
http://www.unica.com.br/convidados/53378...
). This agro-industrial residue is a fibrous material composed approximately of 55% cellulose, 17% hemicellulose, 25% lignin and 1% ash. These components present in sugarcane have varied concentrations and depends on many factors, such as climatic conditions and soil properties during cultivation (Hoareau et al., 2004Hoareau, W., Trindade, W. G., Siegmund, B., Castellan, A., & Frollini, E. (2004). Sugarcane bagasse and curaua lignins oxidized by chlorine dioxide and reacted with furfuryl alcohol: Characterization and stability. Polymer Degradation & Stability, 86(3), 567-576. http://dx.doi.org/10.1016/j.polymdegradstab.2004.07.005
http://dx.doi.org/10.1016/j.polymdegrads...
). A possible use of SCB is as a support for the immobilization of enzymes (Varavinit et al., 2002Varavinit, S., Chaokasem, N., & Shobsngob, S. (2002). Immobilization of a thermostable alpha-amylase. Scienceasia, 28(3), 247-251. http://dx.doi.org/10.2306/scienceasia1513-1874.2002.28.247
http://dx.doi.org/10.2306/scienceasia151...
). The lignocellulosic fibers of SCB contain hydroxyl groups which can react with other polar functional group, such as amine grouping, phosphate, carbonyl and carboxyl (Chen et al., 2012Chen, K. I., Lo, Y. C., Su, N. W., Chou, C. C., & Cheng, K. C. (2012). Enrichment of two isoflavone aglycones in black soymilk by immobilized β-glucosidase on solid carriers. Journal of Agricultural and Food Chemistry, 60(51), 12540-12546. PMid:23190054. http://dx.doi.org/10.1021/jf304405t
http://dx.doi.org/10.1021/jf304405t...
). The structural complexity of SCB can limit its use. However, the fiber can be modified by thermal, acid and basic treatments (Mosier et al., 2005Mosier, N., Wyman, C., Dale, B., Elander, R., Lee, Y. Y., Holtzapple, M., & Ladisch, M. (2005). Features of promising technologies for pretreatment of lignocellulosic biomass. Bioresource Technology, 96(6), 673-686. PMid:15588770. http://dx.doi.org/10.1016/j.biortech.2004.06.025
http://dx.doi.org/10.1016/j.biortech.200...
). Alkaline treatments remove lignin, hemicellulose and uronic acids, which facilitate the accessibility of cellulose (Khuong et al., 2014Khuong, L. D., Kondo, R., De Leon, R., Kim Anh, T., Shimizu, K., & Kamei, I. (2014). Bioethanol production from alkaline-pretreated sugarcane bagasse by consolidated bioprocessing using Phlebia sp. MG-60. International Biodeterioration & Biodegradation, 88, 62-68. http://dx.doi.org/10.1016/j.ibiod.2013.12.008
http://dx.doi.org/10.1016/j.ibiod.2013.1...
). The hydroxyl groups of anhydroglucose units can be modified with various chemicals (Potthast et al., 2006Potthast, A., Rosenau, T., & Kosma, P. (2006). Analysis of oxidized functionalities in cellulose. In D. Klemm (Ed.), Advancesin polymer science (Vol. 205, pp. 1-48). Switzerland: Springer. http://dx.doi.org/10.1007/12_099.
http://dx.doi.org/10.1007/12_099...
). Glutaraldehyde is one of these reagents and is used in the design of biocatalysts (Barbosa et al., 2014Barbosa, O., Ortiz, C., Berenguer-Murcia, A., Torres, R., Rodrigues, R. C., & Fernandez-Lafuente, R. (2014). Glutaraldehyde in bio-catalysts design: A useful crosslinker and a versatile tool in enzyme immobilization. RSC Advances, 4(4), 1583-1600. http://dx.doi.org/10.1039/C3RA45991H
http://dx.doi.org/10.1039/C3RA45991H...
).
β-Glucosidase (β-d-glucoside glucohydrolase, EC. 3.2.1.21) catalyzes the hydrolysis of the β-glycosidic bonds from the non-reducing ends of substrates. In the food industry, these enzymes play an important role in saccharification, fermentation, and the production of flavorings, teas, wines, fruit juices and cellulose (Krisch et al., 2010Krisch, J., Takó, M., Papp, T., & Vágvölgyi, C. (2010). Characteristics and potential use of β-glucosidases from zygomycetes. In A. Mendez-Vilas (Ed.), Current research, technology and education topics in applied microbiology and microbial biotechnology (pp. 891-896). Badajoz: Formatex Research Centre.). Purified soybean cotyledons β-glucosidase (BGL) can be applied to glucosides hydrolysis to aglycones (Santos et al., 2013Santos, R. F., Oliveira, C. F., Varéa, G. S., Orradi Da Silva, M. L. C., Ida, E. I., Mandarino, J. M. G., Carrão-Panizzi, M. C., & Ribeiro, M. L. L. (2013). Purification and characterization of soy cotyledon β-glucosidase. Journal of Food Biochemistry, 37(3), 302-312. http://dx.doi.org/10.1111/j.1745-4514.2011.00632.x
http://dx.doi.org/10.1111/j.1745-4514.20...
). However, the use of this soluble enzyme has been limited by economic factors, as purification processes and by the loss of the molecule in the solution, since the enzyme solubilizes in the medium, difficult to recover. The immobilization process in low-cost supports such as sugarcane bagasse, as well as its treatment, would enable the use in industrial processes, due to the added benefit of enzyme reuse, increased thermal stability, ease of separation from reaction mixtures and range of pH optimal (Singh et al., 2014Singh, N., Srivastava, G., Talat, M., Raghubanshi, H., Srivastava, O. N., & Kayastha, A. M. (2014). Cicer α-galactosidase immobilization onto functionalized graphene nanosheets using response surface method and its applications. Food Chemistry, 142, 430-438. PMid:24001862. http://dx.doi.org/10.1016/j.foodchem.2013.07.079
http://dx.doi.org/10.1016/j.foodchem.201...
).
Isoflavones are phenolic compounds of the flavonoid group, mainly accumulated in soybeans as products of secondary metabolism. Their chemical structures resemble that of an estrogenic compound estradiolThere are twelve different forms of isoflavones, including aglycones (daidzein, genistein and glycitein), glucosides (daidzin, genistin and glycitin), acetylglucosides (6-O-acetyldaidzin, 6-O-acetylgenistin and 6-O-acetylglycitin) and malonylglucosides (6-O-malonyldaidzin, 6-O-malonylgenistin and 6-O-malonylglycitin) (Kao et al., 2008Kao, T. H., Chien, J. T., & Chen, B. H. (2008). Extraction yield of isoflavones from soybean cake as affected by solvent and supercritical carbon dioxide. Food Chemistry, 107(4), 1728-1736. http://dx.doi.org/10.1016/j.foodchem.2007.10.015
http://dx.doi.org/10.1016/j.foodchem.200...
). Isoflavones have been widely investigated because of their potential health benefits, including anticarcinogenic activity (Hillman et al., 2011Hillman, G. G., Singh-Gupta, V., Al-Bashir, A. K., Yunker, C. K., Joiner, M. C., Sarkar, F. H., Abrams, J., & Haacke, E. M. (2011). Monitoring sunitinib-induced vascular effects to optimize radiotherapy combined with soy isoflavones in murine xenograft tumor1. Translational Oncology, 4(2), 110-121. PMid:21461174. http://dx.doi.org/10.1593/tlo.10274
http://dx.doi.org/10.1593/tlo.10274...
), cardiovascular disease prevention (Rimbach et al., 2008Rimbach, G., Boesch-Saadatmandi, C., Frank, J., Fuchs, D., Wenzei, U., Daniel, H., Hall, W. L., & Weinberg, P. D. (2008). Dietary isoflavones in the prevention of cardiovascular disease: A molecular perspective. Food and Chemical Toxicology, 46(4), 1308-1319. PMid:17689850. http://dx.doi.org/10.1016/j.fct.2007.06.029
http://dx.doi.org/10.1016/j.fct.2007.06....
) improvement of renal function in menopausal women with hypertension and renal function reduction (Liu et al., 2014Liu, Z. M., Ho, S. C., Chen, Y. M., Tang, N., & Woo, J. (2014). Effect of whole soy and purified isoflavone daidzein on renal function: A 6 month randomized controlled trial in equol-producing postmenopausal women with prehypertension. Clinical Biochemistry, 47(13-14), 1250-1256. PMid:24877660. http://dx.doi.org/10.1016/j.clinbiochem.2014.05.054
http://dx.doi.org/10.1016/j.clinbiochem....
). The physiological effects of isoflavones may be associated with the biotransformation of the glucosides converted to forms aglycones absorbed in the intestine. However, the bioavailability of aglycones is influenced by the effects of habitual diet on gut microbiota (Tsuchihashi et al., 2008Tsuchihashi, R., Sakamoto, S., Kodera, M., Nohara, T., & Kinjo, J. (2008). Microbial metabolism of soy isoflavones by human intenstinal bacteria strains. Journal of Natural Medicines, 62(4), 456-460. PMid:18648905. http://dx.doi.org/10.1007/s11418-008-0271-y
http://dx.doi.org/10.1007/s11418-008-027...
). Thus, aglycone conversion technologies are applied to meet the increasing demands of soy products (Zaheer & Humayoun Akhtar, 2017Zaheer, K., & Humayoun Akhtar, M. (2017). An updated review of dietary isoflavones: Nutrition, processing, bioavailability and impacts on human health. Critical Reviews in Food Science and Nutrition, 57(6), 1280-1293. PMid:26565435. http://dx.doi.org/10.1080/10408398.2014.989958
http://dx.doi.org/10.1080/10408398.2014....
). The total isoflavone content of 18 soybean cultivars evaluated by Ribeiro et al. (2007)Ribeiro, M. L. L., Mandarino, J. M. G., Carrão-Panizzi, M. C., Oliveira, M. C. N., Campo, C. B. H., Nepomuceno, A. L., & Ida, E. I. (2007). Isoflavone content and β-glucosidase activity in soybean cultivars of different maturity groups. Journal of Food Composition and Analysis, 20(1), 19-24. http://dx.doi.org/10.1016/j.jfca.2006.07.004
http://dx.doi.org/10.1016/j.jfca.2006.07...
were from 174.30 to 61.83 mg 100 g-1 of which 67% was malonylglucosides, 31% glucosides and 2% aglycones. The application of exogenous BGL in unfermented soy products may be a viable alternative to increase the content of aglycones (Chen et al., 2013Chen, K. I., Lo, Y. C., Liu, C. W., Yu, R. C., Chou, C. C., & Cheng, K. C. (2013). Enrichment of two isoflavone aglycones in black soymilk by using spent coffee grounds as an immobiliser for β-glucosidase. Food Chemistry, 139(1-4), 79-85. PMid:23561081. http://dx.doi.org/10.1016/j.foodchem.2013.01.093
http://dx.doi.org/10.1016/j.foodchem.201...
; Grade et al., 2014Grade, L. C., Moreira, A. A., Varea, G. S., Mandarino, J. M. G., Silva, J. B., Ida, E. I., & Ribeiro, M. L. L. (2014). Soybean β-glucosidase immobilisated on chitosan beads and its application in soy drink increase the aglycones. Brazilian Archives of Biology and Technology, 57(5), 766-773. http://dx.doi.org/10.1590/S1516-8913201402331
http://dx.doi.org/10.1590/S1516-89132014...
). Therefore, in this work, enzyme immobilization conditions in SCB were optimized using response surface methodology (RMS) to development and application of an immobilized biocatalyst. SCB was characterized using scanning electron microscopy (SEM), energy dispersive X-ray spectroscopy (SEM-EDX) and Fourier transform infrared spectroscopy (FTIR). The properties of the SCB-immobilized enzyme were compared with those of soluble enzymes. The SCB-immobilized BGL was applied to commercial soymilk to evaluate the efficiency conversion of glucosides to aglycones.
2 Material and methods
2.1 Chemical and materials
Sugarcane bagasse (SCB) and soymilk were purchased from a local commercial market. Soybeans [Glycine max (L.) Merrill.; BRS 213], were obtained from the Empresa Brasileira de Pesquisa Agropecuária, Centro Nacional de Pesquisa de Soja (Embrapa-Soja; Londrina, PR, Brazil).
For the determination of BGL activity, p-nitrophenyl β-d-glucopyranoside (p-NPG) substrate, p-nitrophenol standard (p-NP) and bovine serum albumin (BSA) were purchased from Sigma-Aldrich Co. (St. Louis, MO, USA). For the determination of the isoflavones, the standards 6”-O-malonylglycosides and 6”-O-acetylglycosides (Wako Pure Chemical Industries, Ltd., Osaka, Japan), and β-glycosides and aglycones (Sigma-Aldrich Co., St. Louis, MO, USA) were used. All other reagents used were of analytical grade or chromatography grade from different sources.
2.2 Determination of β-glucosidase activity and protein content
BGL activity was assayed using p-NPG as substrate (Matsuura & Obata, 1993Matsuura, M., & Obata, A. (1993). β-glucosidases from soybeans hydrolyze daidzin and genistin. Journal of Food Science, 58(1), 144-147. http://dx.doi.org/10.1111/j.1365-2621.1993.tb03231.x
http://dx.doi.org/10.1111/j.1365-2621.19...
) with minor modification. Soluble enzyme (0.25 mL) was added to 1 mL phosphate-citrate buffer (100 mM, pH 5) containing p-NPG (1 mM) at 30 °C for 30 min. Reaction was stopped by addition of 1.25 mL of sodium carbonate (500 mM). Immobilized enzyme (100 mg) was added to 4 mL phosphate-citrate buffer (100 mM, pH 5) containing p-NPG (1 mM) at 30 °C for 30 min and was separated by filtration. To the filtrate (1 mL) was added 1.25 mL of sodium carbonate (500 mM) to stop and reaction. The activity of the soluble and immobilized enzyme was determined by absorbance measured at 420 nm. Control assay without enzyme or without substrate was included. The standard curve of p-NP (0.016-0.16 µmol) was prepared. One unit of β-glucosidase activity (U) was defined as the amount of enzyme required to liberate 1 µmol of p-nitrophenol min-1 under assay conditions.
Protein contents in the soluble enzyme solution and the residual protein contents in the immobilized enzymes washings solutions were quantified using BSA as standard (40-400 µg mL-1) (Lowry et al., 1951Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). Protein measurement with the folin phenol reagent. The Journal of Biological Chemistry, 193(1), 265-275. PMid:14907713.). The determination of the protein bound to the support was performed indirectly, by the difference of the initial amount of proteins (total content of the solution) and the protein content lost in the washings solutions. The specific activity of the soluble and immobilized BGL was determined as the relationship between the enzymatic activity and the protein contents, and this value was expressed as U mg-1 of protein.
2.3 Enzyme preparation
Soybean cotyledon flour and sodium phosphate buffer (100 mM; pH 6.6) were used in a 1:10 proportion (w/v) to obtain a crude extract (Matsuura & Obata, 1993Matsuura, M., & Obata, A. (1993). β-glucosidases from soybeans hydrolyze daidzin and genistin. Journal of Food Science, 58(1), 144-147. http://dx.doi.org/10.1111/j.1365-2621.1993.tb03231.x
http://dx.doi.org/10.1111/j.1365-2621.19...
). The crude extract was fractionated with ammonium sulfate at 40%, centrifuged at 4 °C 10000 x g for 20 min (ultracentrifuge Beckman Coulter Model optima XE-100) and obtained precipitate (P40) and supernatant (S40). At S40 ammonium sulfate was added until saturation of 85% and obtained precipitate (P85) and supernatant (S85). The precipitates were resuspended in the buffer and together with the supernatants were dialyzed with the same buffer for 14 h at 4 °C (Santos et al., 2013Santos, R. F., Oliveira, C. F., Varéa, G. S., Orradi Da Silva, M. L. C., Ida, E. I., Mandarino, J. M. G., Carrão-Panizzi, M. C., & Ribeiro, M. L. L. (2013). Purification and characterization of soy cotyledon β-glucosidase. Journal of Food Biochemistry, 37(3), 302-312. http://dx.doi.org/10.1111/j.1745-4514.2011.00632.x
http://dx.doi.org/10.1111/j.1745-4514.20...
). After, the precipitate (P85) was used as the enzyme source for immobilization on SCB, as it showed the highest enzymatic activity.
2.4 Sugarcane bagasse modifications
The sugarcane bagasse (SCB) fibers were recovered by hand processing, washed and dried at 60 °C (Marconi, MA 035), cut and passed through a 30 mesh (0.7 mm particle size). The modifications of the SCB proceeded in the following ways: SCB1: SCB was washed with distilled water at 60 °C to remove sugars until no sugars were detected (Dubois et al., 1956Dubois, M., Gilles, M., Hamilton, J. K., Rebers, P. A., & Smith, F. (1956). Colorimetric method for determination of sugars and related substances. Analytical Chemistry, 28(3), 350-356. http://dx.doi.org/10.1021/ac60111a017
http://dx.doi.org/10.1021/ac60111a017...
). SCB2: SCB1 was activated with 2.5% glutaraldehyde, pH 7, incubated at 25 °C for 30 min at 70 rpm.
SCB3: SCB1 was treated with 2% NaOH at 30 °C for 24 h and autoclaved at 121 °C for 15 min.
SCB4: SCB3 was activated with 2.5% glutaraldehyde, pH 7, incubated at 25 °C for 30 min at 70 rpm.
SCB5: was treated with 70% ethanol and autoclaved at 121 °C for 15 min.
SCB6: SCB5 was activated with 2.5% glutaraldehyde, pH 7, incubated at 25 °C for 30 min at 70 rpm.
2.5 Immobilization of enzyme
The BGL was diluted with sodium phosphate buffer (200 mM; pH 7) to obtain a specific activity of 8.17 10-3 U mg-1 of protein, for each mL of soluble enzyme. For immobilization, 1 g of SCB native (SCB1) or modified (SCB2; SCB3; SCB4; SCB5; SCB6) was added to 30 mL of soluble enzyme. The systems were shaken at 70 rpm for 6 h at 4 °C. The SCB were collected by filtration, washed with phosphate-citrate buffer (100 mM, pH 5) and stored at 4 °C. The supernatants containing the soluble enzyme and washing solutions were analyzed for BGL activity (U), protein content and specific activity. The immobilization efficiency (IE) was calculated as follows: IE (%) = (specific activity of immobilized enzyme/specific activity of soluble enzyme) × 100. The assays were performed in triplicate and the results were submitted to an analysis of variance (ANOVA) and Tukey’s test (p < 0.05) to determine the modification of SCB most suitably to a higher immobilization efficiency of BGL.
2.6 Characterization of sugarcane bagasse
2.6.1 Determination of cellulose, lignin, hemicelluloses and ash
Cellulose, lignin and hemicellulose content were determined (Van Soest et al., 1991Van Soest, P. J., Robertson, J. B., & Lewis, B. A. (1991). Methods for dietary fiber, neutral detergent fiber and nonstarch polysaccharides in relation to animal nutrition. Journal of Dairy Science, 74(10), 3583-3597. PMid:1660498. http://dx.doi.org/10.3168/jds.S0022-0302(91)78551-2
http://dx.doi.org/10.3168/jds.S0022-0302...
) and ash content of was determined using muffle furnace at 550 °C (Association of Official Analytical Chemists, 2006Association of Official Analytical Chemists – AOAC. (2006). Official methods of analysis of AOAC International (18th ed.). Arlington: AOAC.) to evaluate the structure of SCB native and modified.
2.6.2 Scanning Electron Microscopy (SEM) and Energy Dispersive X-ray spectroscopy (SEM-EDX)
The morphology, chemical elements and functional groups on the surface of SCB native, modified and immobilized enzyme were examined by using scanning electron microscopy (SEM) coupled with energy dispersive X-ray spectroscopy (SEM-EDX) at an accelerating voltage of 10 keV (FEI Quanta 200, Hillsboro, OR, USA). For SEM analysis, sample were dried, placed on an aluminum stub and coated with a layer (25 nm) of gold using a sputter-coater (Bal-Tec Sputter Coater SDC 050, Scotia, NY, USA). For SEM-EDX studies, dried samples were coated with graphite.
2.6.3 Fourier Transform Infrared Spectroscopy (FTIR)
The FTIR spectra of the samples were analyzed using a IR Prestige-21 Shimadzu spectrometer (Columbia, MD, USA). The analyses were performed after, preparation of thesamples by mixing the material with KBr power followed by vacuum drying. The spectra were recorded in the range of 1000-4000 cm-1 and readings were performed with 100 scans for each sample at 2 cm-1 resolution using IRSolution Software 1.60SU2.
2.7 Experimental design and statistical analysis
Preliminary assays were performed to determine the initial values of factors [protein (0.5-1.5 mg mL-1), glutaraldehyde (0.5%-2.5%), pH (4.5-7.5), rpm (70-110) and incubation time (2-6 h)] involved in enzyme immobilization using 25-1 fractional factorial design (25-1 FFD). Based on these results, 22 rotatable central composite design (22 RCCD) was used for optimization of enzyme immobilization on SCB modified with 13 random assays (4 axial points and 5 central points). The independent variables and their levels were: incubation time (2, 4, 8, 12 and 14 h) and glutaraldehyde content (1.0%, 1.5%, 2.5%, 3.5% and 4.0%). The immobilization efficiency (IE) obtained was taken as the dependent variable (YIE, %). The mathematical model for the response function (IE) was expressed as: YIE = β0 + ΣβiXi + ΣβiiXi2 + ΣβiiXiXj + ε, where XiXj are the input variables, β are the estimated coefficients on the response surface and ε is the error. The model was obtained by analyses of variance (ANOVA, p = 0.05) and regression analysis using Statistic 10.0 (Statsoft, Tulsa, OK, USA). Response surface and desirability were generated for response function evaluated.
2.8 Characterization of immobilized β-glucosidase
2.8.1 Effect of pH and temperature
In all cases, control assays using soluble enzyme were carried out. The assays were performed in triplicate and analysis BGL activity. To evaluate the effect of pH and temperature on SCB-immobilized BGL, a 22RCCD was used, with 13 random assays (4 axial points and 5 central points). The independent variables and their levels were: pH (3.0, 4.0, 5.5, 7.0 and 8.0) and temperature (25, 35, 50, 65 and 75 °C). The SCB-immobilized BGL was assayed at an increasing content of p-NPG (0.1-20 mM) under optimal conditions (pH 5.5 at 50 °C) for 30 min. Km and Vmax were determined using Lineweaver-Burk plot method.
2.8.2 Thermal stability
Thermal stability was determined by incubating the enzyme at 30, 50 and 70 °C in phosphate-citrate buffer (100 mM, pH 5.5) for different time intervals (0-240 min), after this previous incubation, without p-NPG, the residual activity at 50 °C for 30 min using p-NPG as a substrate was determined.
2.8.3 Reusability
For reusability studies, SCB-immobilized BGL was determined under optimal conditions (pH 5.5 at 50 °C) for 30 min. After each assay, these were washed with phosphate-citrate buffer (100 mM, pH 5.5). The results were expressed as residual activity (%) of immobilized enzyme after each cycle.
2.9 Enzymatic treatment of soymilk for hydrolysis of isoflavones
The SCB-immobilized BGL (0.4 g) was incubated with 10 mL commercial soymilk and maintained at 25 or 50 °C for 30 to 120 min. At the same time, a control assay without enzyme was performed in the same conditions. After use, the enzyme was removed. The soymilk was lyophilized and defatted with hexane at a 1:10 (g mL-1) proportion and was continually stirred for 1 h at 25 °C followed by vacuum filtration. After, the isoflavones contents were determined.
2.10 Determination of isoflavones
The extraction of isoflavones was performed in triplicates with 0.3 g lyophilized and defatted samples in 6 mL of extraction solution containing ultra-pure water, acetone and ethanol (1:1:1; v:v:v) (Yoshiara et al., 2012Yoshiara, L. Y., Madera, T. B., Delaroza, F., Silva, J. B., & Ida, E. I. (2012). Optimization of soy isoflavone extraction with different solvents using the simplex-centroid mixture design. International Journal of Food Sciences and Nutrition, 63(8), 978-986. PMid:22621769. http://dx.doi.org/10.3109/09637486.2012.690026
http://dx.doi.org/10.3109/09637486.2012....
). Separation and quantification of isoflavones were performed using ultra-high-pressure liquid chromatography (UHPLC; Acquity UPLC® System, Waters, Milford, MA, USA) equipped with a reversed-phase BEH C18 column (50 mm × 2.1 mm, 1.7 mm; Waters) (Handa et al., 2014Handa, C. L., Couto, U. R., Vicensoti, A. H., Georgetti, S. R., & Ida, E. I. (2014). Optimisation of soy flour fermentation parameters to produce β-glucosidase for bioconversion into aglycones. Food Chemistry, 152, 56-65. PMid:24444906. http://dx.doi.org/10.1016/j.foodchem.2013.11.101
http://dx.doi.org/10.1016/j.foodchem.201...
). The results are expressed in µmol of isoflavone per mL of sample (µmol mL-1). The results were submitted to an analysis of variance (ANOVA) and Tukeys’s test (p < 0.05).
3 Results and discussion
3.1 Sugarcane bagasse modifications
The immobilization efficiency (IE) of BGL on SCB modified with alkali and temperature was 62% to 98% (Table 1) and protein content (0.7 mg mL-1 or 1.4 mg mL-1) did not affect (p > 0.05) the IE in all SCB used. The SCB modified with 2% NaOH, autoclaved (15 min at 121 °C) and activated with 2.5% glutaraldehyde (SCB4) presents greater IE.
Immobilization efficiency (IE) of soybean β-glucosidase in modified sugarcane bagasse (SCB) with chemical and thermal treatments.
Glutaraldehyde has been used widely in the construction of catalytic systems for application in food (Albuquerque et al., 2018Albuquerque, T. L., Gomes, S. D. L., D’Almeida, A. P., Fernandez-Lafuente, R., Gonçalves, L. R. B., & Rocha, M. V. P. (2018). Immobilization of β-galactosidase in glutaraldehyde-chitosan and its application to the synthesis of lactulose using cheese whey as feedstock. Process Biochemistry, 73, 65-73. http://dx.doi.org/10.1016/j.procbio.2018.08.010
http://dx.doi.org/10.1016/j.procbio.2018...
; Singh et al., 2014Singh, N., Srivastava, G., Talat, M., Raghubanshi, H., Srivastava, O. N., & Kayastha, A. M. (2014). Cicer α-galactosidase immobilization onto functionalized graphene nanosheets using response surface method and its applications. Food Chemistry, 142, 430-438. PMid:24001862. http://dx.doi.org/10.1016/j.foodchem.2013.07.079
http://dx.doi.org/10.1016/j.foodchem.201...
; Chen et al., 2013Chen, K. I., Lo, Y. C., Liu, C. W., Yu, R. C., Chou, C. C., & Cheng, K. C. (2013). Enrichment of two isoflavone aglycones in black soymilk by using spent coffee grounds as an immobiliser for β-glucosidase. Food Chemistry, 139(1-4), 79-85. PMid:23561081. http://dx.doi.org/10.1016/j.foodchem.2013.01.093
http://dx.doi.org/10.1016/j.foodchem.201...
). Thus, the immobilized α-galactosidase in functionalized graphene nanosheets activated with 2.8% glutaraldehyde was applied in soy drink and hydrolyzed the oligosaccharides (Singh et al., 2014Singh, N., Srivastava, G., Talat, M., Raghubanshi, H., Srivastava, O. N., & Kayastha, A. M. (2014). Cicer α-galactosidase immobilization onto functionalized graphene nanosheets using response surface method and its applications. Food Chemistry, 142, 430-438. PMid:24001862. http://dx.doi.org/10.1016/j.foodchem.2013.07.079
http://dx.doi.org/10.1016/j.foodchem.201...
). The activated spent coffee grounds with 2.5% glutaraldehyde were used for immobilization of β-glucosidase and applied in black soy drink for hydrolysis of aglycones (Chen et al., 2013Chen, K. I., Lo, Y. C., Liu, C. W., Yu, R. C., Chou, C. C., & Cheng, K. C. (2013). Enrichment of two isoflavone aglycones in black soymilk by using spent coffee grounds as an immobiliser for β-glucosidase. Food Chemistry, 139(1-4), 79-85. PMid:23561081. http://dx.doi.org/10.1016/j.foodchem.2013.01.093
http://dx.doi.org/10.1016/j.foodchem.201...
). Thus, the SCB4 was used for further immobilization assays, characterization and application.
3.2 Immobilization of β-glucosidase on modified sugarcane bagasse
From the exploratory model of the 25-1 FFD, the ANOVA and regression analysis, the effects of input variables X1 (protein, mg mL-1), X2 (pH), X3 (rpm), X4 (glutaraldehyde, %) and X5 (incubation, h) on the response function of IE (%) were observed (data not showed). The variable pH (X2), glutaraldehyde (X4) and incubation (X5) showed significant positive linear effects (p < 0.05). The variables protein (X1), rpm (X3) and interactions (X1. X 3, X 1. X 5, X 2. X 5, X 3. X 4 and X 3. X 5) were not significant (p > 0.05) and the coefficient of determination (R2) was 0.76. Preliminary experiments assays showed that the stability of the SCB4- immobilized BGL was greater at pH 7. Thus, the immobilization conditions were fixed 1 mg mL-1 of protein, 110 rpm and pH 7 at 4 °C. Therefore, to optimize SCB-immobilization BGL conditions, assays were performed (22 RCCD) with input variables X1” (incubation time: 2, 4, 8, 12 and 14 h) and X2” (glutaraldehyde: 1.0, 1.5, 2.5, 3.5 and 4.0%).
According to the ANOVA and the regression analysis for IE, X1” and X2” showed significant linear and quadratic effect. In addition, the interaction (X1”.X”) showed significant negative effect (p < 0.05) (data not showed). The quadratic polynomial equation with significant terms is: IE (%) = 17.85 + 12.05 X1” + 29.12 X2” - 0.62 X1”2 - 6.06 X2”2 - 0.63 X1”.X2”, where X1” denotes incubation time (h) and X2” is glutaraldehyde (%). The model showed an R2 of 0.92, indicating that 92% of the experimental results adequately adjusted with the proposed model, so the model can be used for predictive purposes.
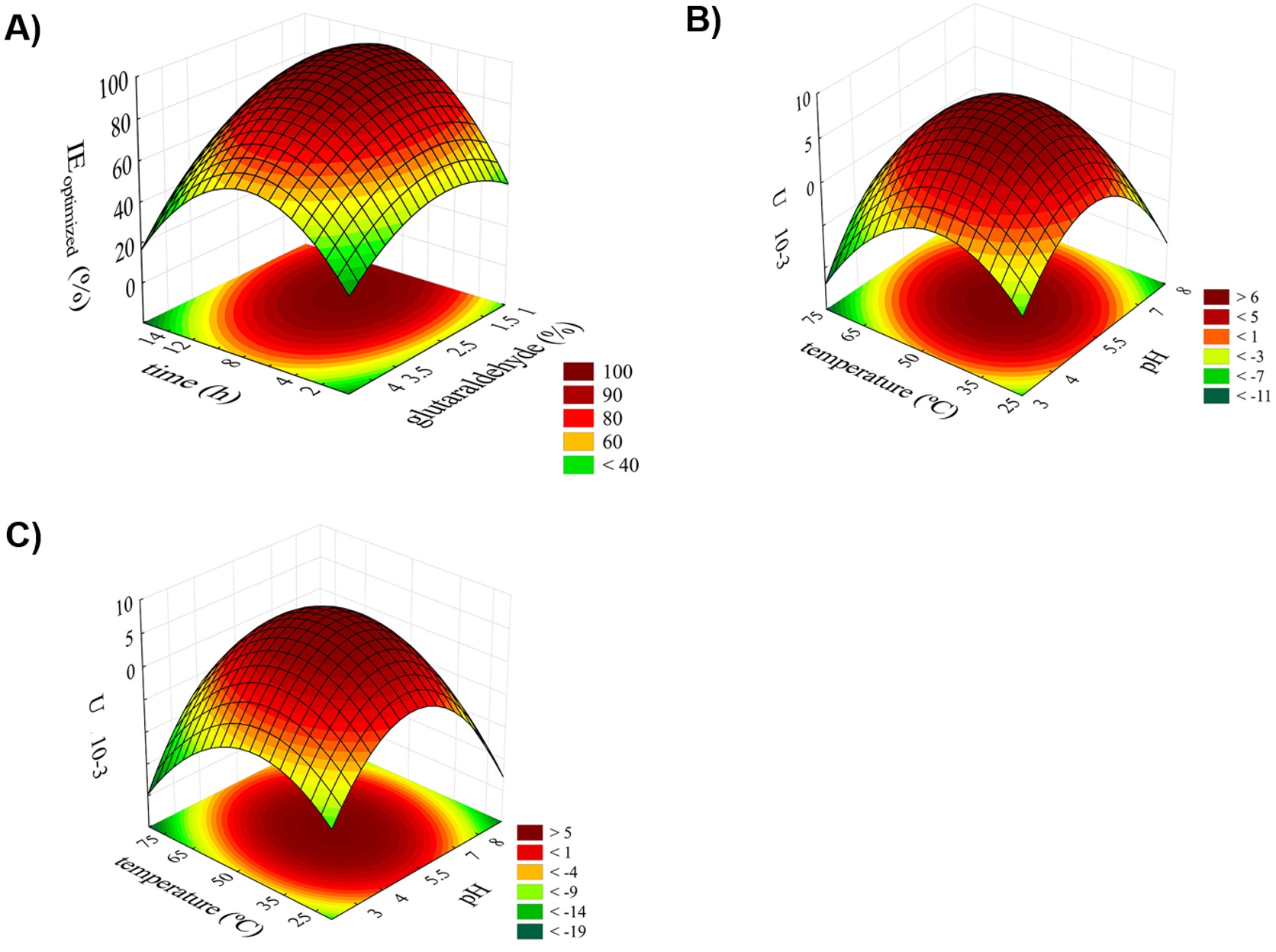
Analyzing the response surface (Figure 1A) the IE range from 70% to 99% and negative interaction between X1” and X2”. There is a region in which of maximum of IE, i.e., X1” was between 6.0 and 12 h and X2” was between 1.0% and 3.0% of glutaraldehyde. The desirability parameters obtained by the model indicated that the maximum IE would be 97%, which occurred when X1” = 8 h of incubation and X2” = 2.5% of glutaraldehyde (Figure 1A) which coincided with central point. Thus, the results demonstrated that to obtain maximum IE SCB4-immobilized BGL conditions should be used 1 mg mL-1 of protein, 110 rpm, pH 7.0, 8 h of incubation and 2.5% glutaraldehyde at 4 °C.
Response surfaces plots for soybean β-glucosidase immobilization in sugarcane bagasse as a function of time (h) and glutaraldehyde (%) for IE optimized (A) and as a function of effect of temperature (°C) and pH on the activity of soluble (B) and immobilized β-glucosidase (C).
Response surface methodology (RSM) was used to be a useful statistical tool for modeling multivariate that influences the response function. This statistical method was able of optimize the process with save time and reagents by reductions in the number of experiments of overall analysis, being advantageous to the industry. RSM has been used for optimization of biocatalysis processes (Singh et al., 2014Singh, N., Srivastava, G., Talat, M., Raghubanshi, H., Srivastava, O. N., & Kayastha, A. M. (2014). Cicer α-galactosidase immobilization onto functionalized graphene nanosheets using response surface method and its applications. Food Chemistry, 142, 430-438. PMid:24001862. http://dx.doi.org/10.1016/j.foodchem.2013.07.079
http://dx.doi.org/10.1016/j.foodchem.201...
) and enzyme production (Handa et al., 2014Handa, C. L., Couto, U. R., Vicensoti, A. H., Georgetti, S. R., & Ida, E. I. (2014). Optimisation of soy flour fermentation parameters to produce β-glucosidase for bioconversion into aglycones. Food Chemistry, 152, 56-65. PMid:24444906. http://dx.doi.org/10.1016/j.foodchem.2013.11.101
http://dx.doi.org/10.1016/j.foodchem.201...
).
The results of IE of BGL (97%), after optimization, were higher than those observed for BGL immobilized in chitosan beads activated with 2.5% glutaraldehyde with IE of 73.8% (Grade et al., 2014Grade, L. C., Moreira, A. A., Varea, G. S., Mandarino, J. M. G., Silva, J. B., Ida, E. I., & Ribeiro, M. L. L. (2014). Soybean β-glucosidase immobilisated on chitosan beads and its application in soy drink increase the aglycones. Brazilian Archives of Biology and Technology, 57(5), 766-773. http://dx.doi.org/10.1590/S1516-8913201402331
http://dx.doi.org/10.1590/S1516-89132014...
). Immobilization of BGL in supports cellulose-rich materials was reporting at immobilization of Aspergillus niger BGL on spent coffee grounds obtain IE of 82% (Chen et al., 2013Chen, K. I., Lo, Y. C., Liu, C. W., Yu, R. C., Chou, C. C., & Cheng, K. C. (2013). Enrichment of two isoflavone aglycones in black soymilk by using spent coffee grounds as an immobiliser for β-glucosidase. Food Chemistry, 139(1-4), 79-85. PMid:23561081. http://dx.doi.org/10.1016/j.foodchem.2013.01.093
http://dx.doi.org/10.1016/j.foodchem.201...
). Others enzymes were immobilized in agro-industrial residues such as Trametes versicolor laccase on green coconut and Bacillus licheneformis α-amylase on sugarcane bagasse, with immobilization efficiency of 98% and 44% (Bezerra et al., 2015Bezerra, T. M. S., Bassan, J. C., Santos, V. T. O., Ferraz, A., & Monti, R. (2015). Covalent immobilization of laccase in green coconut fiber and use in clarification of apple juice. Process Biochemistry, 50(3), 417-423. http://dx.doi.org/10.1016/j.procbio.2014.12.009
http://dx.doi.org/10.1016/j.procbio.2014...
; Potthast et al., 2006Potthast, A., Rosenau, T., & Kosma, P. (2006). Analysis of oxidized functionalities in cellulose. In D. Klemm (Ed.), Advancesin polymer science (Vol. 205, pp. 1-48). Switzerland: Springer. http://dx.doi.org/10.1007/12_099.
http://dx.doi.org/10.1007/12_099...
).
3.3 Characterization of sugarcane bagasse
The SCB washed contained cellulose (45.6%), lignin (5.6%), hemicellulose (42.6%) and ash (0.79%). After the alkaline and thermal treatments of SCB were significantly, (p < 0.05) removed lignin (71.4%) and hemicellulose (49.3%) of the SCB unmodified. No significant differences (p > 0.05) in the ash content between the treatments of SCB were observed in Table 2.
Chemical composition of sugarcane bagasse treated with 2% NaOH and autoclaved at 121 °C for 15 min.
The removal of lignin and hemicellulose increased the IE of 62% to 81% (Table 1). The complex association of cellulose, lignin and hemicellulose in SCB decrease the contact area and reduce their use as support of enzyme immobilization (Khuong et al., 2014Khuong, L. D., Kondo, R., De Leon, R., Kim Anh, T., Shimizu, K., & Kamei, I. (2014). Bioethanol production from alkaline-pretreated sugarcane bagasse by consolidated bioprocessing using Phlebia sp. MG-60. International Biodeterioration & Biodegradation, 88, 62-68. http://dx.doi.org/10.1016/j.ibiod.2013.12.008
http://dx.doi.org/10.1016/j.ibiod.2013.1...
). The alkaline treatment may partially remove lignin, hemicellulose and other residues from cellulose fiber surface, revealing chemical reactive functional cellulose groups like − OH. This treatment in sugarcane bagasse is advantageous for the industry, since it allows lower sugar degradation and formation of furan derivatives when compared to the thermal and acid pre-treatments, besides the improvement of the reactivity of the remaining polysaccharides and the elimination of acetyl and other hemicellulose substituent (Chen et al., 2011Chen, W. H., Ye, S. C., & Sheen, H. K. (2011). Hydrolysis characteristics of sugarcane bagasse pretreated by dilute acid solution in a microwave irradiation environment. Applied Energy, 93, 237-244. http://dx.doi.org/10.1016/j.apenergy.2011.12.014
http://dx.doi.org/10.1016/j.apenergy.201...
).
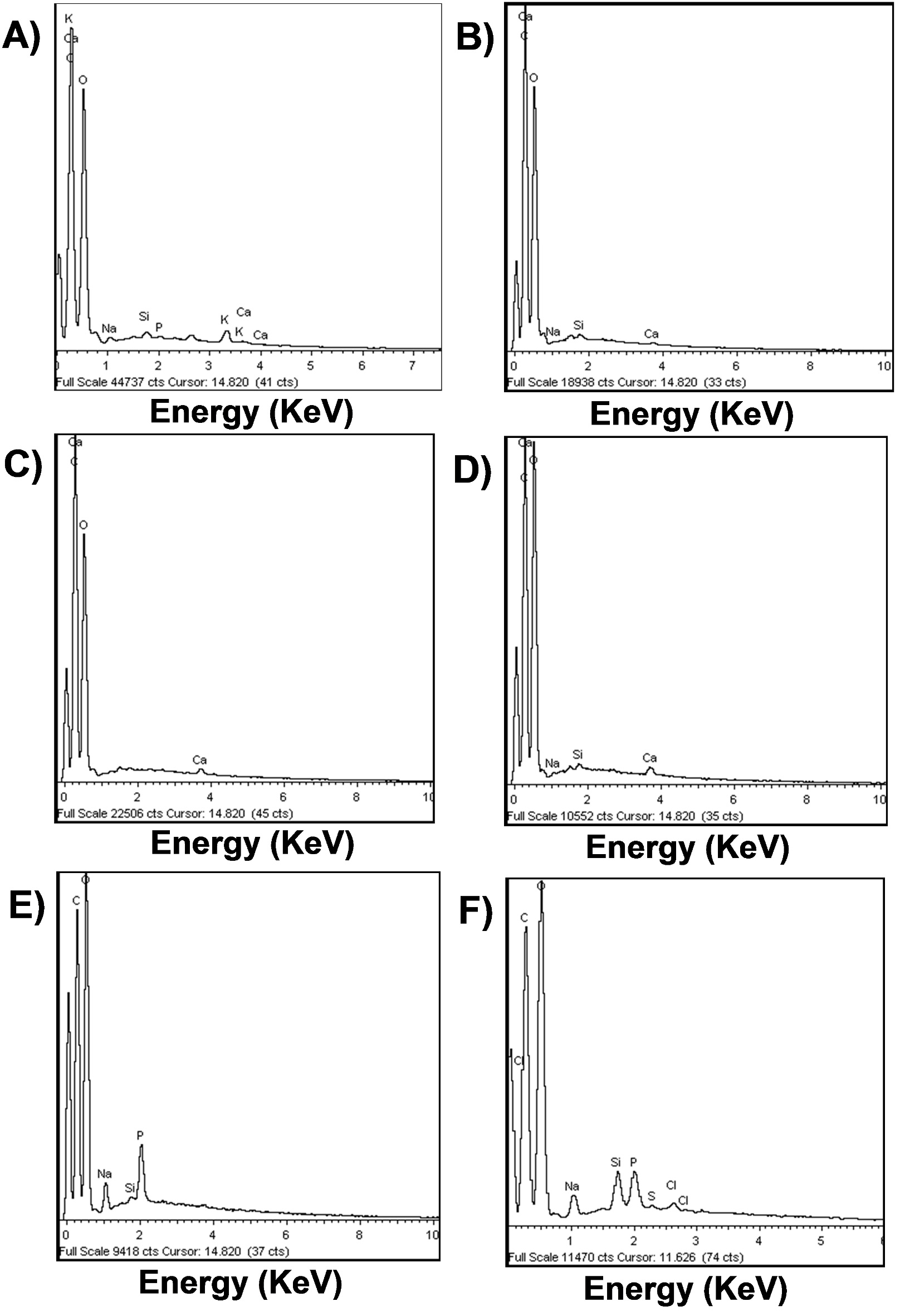
The SEM images of SCB unmodified, modified and with immobilized enzyme are presented in Figure 2. The morphology of SCB1 and SCB5 (Figure 22C) were similar a SCB unmodified (Figure 2A). SCB3 and SCB4 (Figure 22E) showed morphology different a SCB unmodified. SEM images suggest that the alkaline and thermal treatments were efficient in the removal of the external surface layers.
SEM images of sugarcane bagasse unmodified (A) and modified with: water at 60 °C (B), 70% ethanol and autoclaved (C), autoclaved and treated with 2% NaOH (D), autoclaved, treated with 2% NaOH and activated with 2.5% glutaraldehyde (E) and with immobilized β-glucosidase (F).
The similar morphological changes in SCB and coconut fiber were observed (Bezerra et al., 2015Bezerra, T. M. S., Bassan, J. C., Santos, V. T. O., Ferraz, A., & Monti, R. (2015). Covalent immobilization of laccase in green coconut fiber and use in clarification of apple juice. Process Biochemistry, 50(3), 417-423. http://dx.doi.org/10.1016/j.procbio.2014.12.009
http://dx.doi.org/10.1016/j.procbio.2014...
; Carvalho Neto et al., 2014Carvalho Neto, A. G. V., Ganzerli Ta, T. A., Cardoso, A. L., Fávaro, S. L., Pereira, A. G. B., Girotto, E. M., & Radovanovic, E. (2014). Development of composites based on recycled polyethylene/sugarcane bagasse fibers. Polymer Composites, 35(4), 768-774. http://dx.doi.org/10.1002/pc.22720
http://dx.doi.org/10.1002/pc.22720...
). The Figure 2F showed the SEM micrographs of enzyme immobilization on the surface of SCB4. The granulated areas marked by black arrows indicate that enzyme was immobilized on SCB modified. The presence of granulated areas on spent coffee grounds and glass microspheres was reported in the immobilization of Aspergillus niger BGL (Chen et al., 2012Chen, K. I., Lo, Y. C., Su, N. W., Chou, C. C., & Cheng, K. C. (2012). Enrichment of two isoflavone aglycones in black soymilk by immobilized β-glucosidase on solid carriers. Journal of Agricultural and Food Chemistry, 60(51), 12540-12546. PMid:23190054. http://dx.doi.org/10.1021/jf304405t
http://dx.doi.org/10.1021/jf304405t...
, 2013Chen, K. I., Lo, Y. C., Liu, C. W., Yu, R. C., Chou, C. C., & Cheng, K. C. (2013). Enrichment of two isoflavone aglycones in black soymilk by using spent coffee grounds as an immobiliser for β-glucosidase. Food Chemistry, 139(1-4), 79-85. PMid:23561081. http://dx.doi.org/10.1016/j.foodchem.2013.01.093
http://dx.doi.org/10.1016/j.foodchem.201...
).
Chemical elements (C, O, S, P, K, Ca, Na, Cl and Si) in SCB unmodified and modified were determined by SEM-EDX and were different between treatments (Figure 3AF). The presence of P in SCB4 can be attributed to sodium phosphate buffer used in the preparation of glutaraldehyde and was absent in SCB3. The Cl detected in SCB-immobilized BGL (Figure 3F) can be assign to HCl use to obtain the enzyme and S may be indicative of sulphydryl groups of amino acids present in the enzyme, indicating a possible immobilization de BGL in SCB4.
SEM/EDX images of sugarcane bagasse unmodified (A) and modified with: water at 60 °C (B), 70% ethanol and autoclaved (C), autoclaved and treated with 2% NaOH (D), autoclaved, treated with 2% NaOH and activated with 2.5% glutaraldehyde (E) and with immobilized β-glucosidase (F).
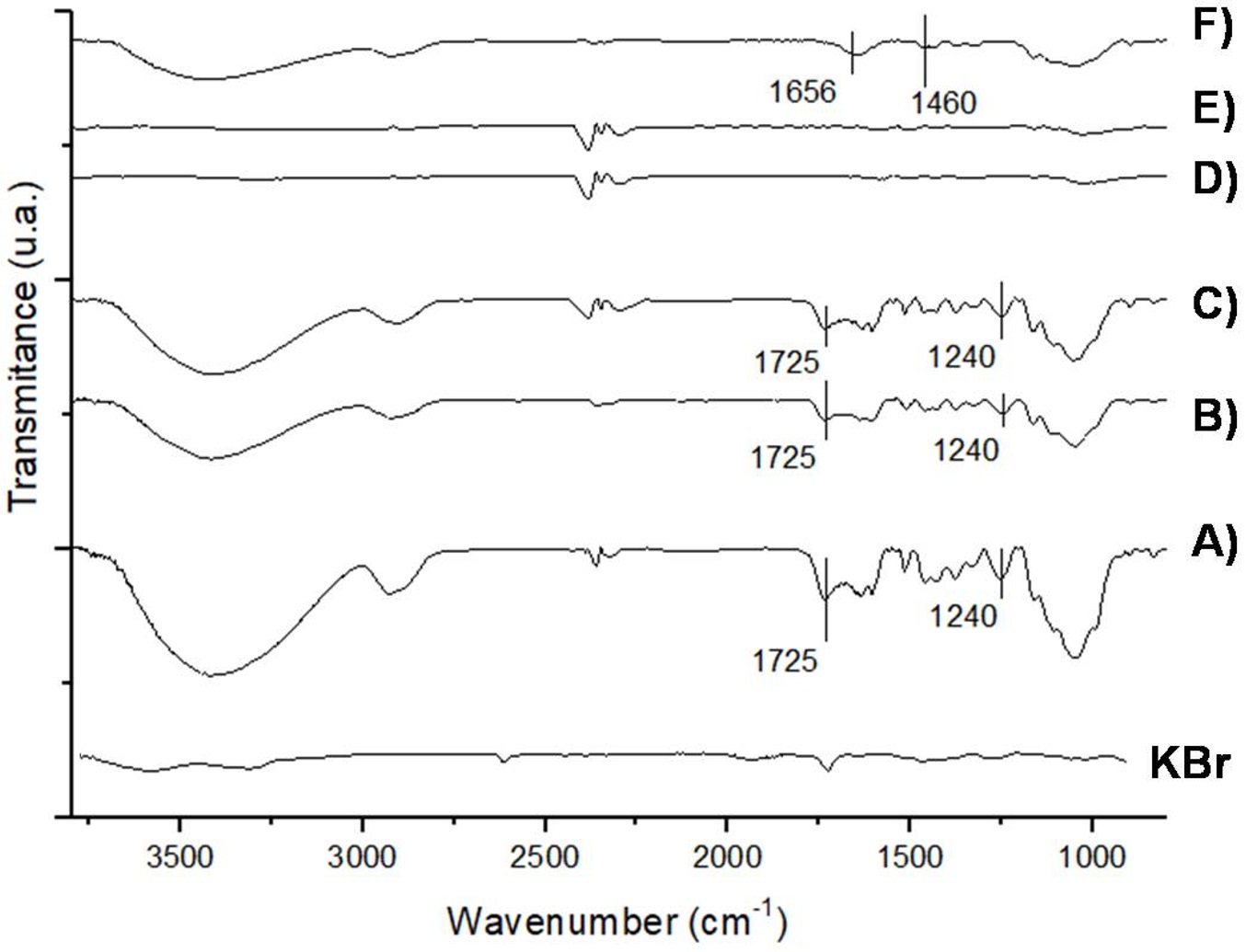
FTIR spectra were recorded for unmodified, modified SCB and SCB-immobilized BGL. The peaks at 1725 cm-1 and 1240 cm-1 of the unmodified SCB, SCB1 and SCB5 (Figure 444C) corresponds to C=O and C−O−C stretching vibrational, respectively, and are characteristics of lignin and hemicellulose. The disappearance of these peaks in spectra of the SCB3 and SCB4 can indicate that lignin and hemicellulose were partially removed during alkaline treatment with 2% NaOH at 121 °C for 15 min. These results also were observed in SCB treated with 10% NaOH at room temperature for 15 h (Carvalho Neto et al., 2014Carvalho Neto, A. G. V., Ganzerli Ta, T. A., Cardoso, A. L., Fávaro, S. L., Pereira, A. G. B., Girotto, E. M., & Radovanovic, E. (2014). Development of composites based on recycled polyethylene/sugarcane bagasse fibers. Polymer Composites, 35(4), 768-774. http://dx.doi.org/10.1002/pc.22720
http://dx.doi.org/10.1002/pc.22720...
).
FTIR spectra of sugarcane bagasse unmodified (A) and modified with: water at 60 °C (B), 70% ethanol and autoclaved (C), treated with 2% NaOH and autoclaved (D), autoclaved, treated with 2% NaOH and activated with 2.5% glutaraldehyde (E) and with immobilized β-glucosidase (F).
The spectra of the SCB4-immobilized BGL (Figure 4F) showed peaks at 1656 cm-1 and 1460 cm-1. Studies evaluated the immobilization of BGL (Sigma) on smectite nanoclays (Serefoglou et al., 2008Serefoglou, E., Litina, K., Gournis, D., Kalogeris, E., Tzialla, A. A., Pavlidis, I. V., Stamatis, H., Maccallini, E., Lubomska, M., & Rudolf, P. (2008). Smectite clays as solid supports for immobilization of β-glucosidase: Synthesis, characterization, and biochemical properties. Chemistry of Materials, 20(12), 4106-4115. http://dx.doi.org/10.1021/cm800486u
http://dx.doi.org/10.1021/cm800486u...
) and magnetic Fe3O4 nanoparticles (Zhou et al., 2013Zhou, Y., Pan, S., Wei, X., Wang, L., & Liu, Y. (2013). Immobilization of β-glucosidase onto magnetic nanoparticles and evaluation the enzymatic properties. BioResources, 8(2), 2605-2619. http://dx.doi.org/10.15376/biores.8.2.2605-2619
http://dx.doi.org/10.15376/biores.8.2.26...
), peaks at 1412 cm-1 and 1660 cm-1 were attributed to the stretching vibrational of amide groups of peptides and therefore confirmed the immobilization of enzyme.
3.4 Characterization of immobilized β-glucosidase
The optimum pH and temperature of soluble and SCB-immobilized BGL were determined by 22 RCCD and RMS (shown only figures). The RMS showed an optimal area of activity to soluble and SCB-immobilized BGL (i.e.), pH values of 4 to 6 and temperatures from 30 to 60 °C; Figure 11C). The greater activity was observed in at pH 5.5 at 50 °C for soluble and immobilized enzyme (3.69 10-3 U and 4.47 10-3 U). The optimum pH of 5.5 was attributed to the presence of two carboxylic acids involved in the catalytic activity of BGL (Nam et al., 2010Nam, K. H., Sung, M. W., & Hwang, K. Y. (2010). Structural insights into the substrate recognition properties of beta-glucosidase. Biochemical and Biophysical Research Communications, 391(1), 1131-1135. PMid:20005197. http://dx.doi.org/10.1016/j.bbrc.2009.12.038
http://dx.doi.org/10.1016/j.bbrc.2009.12...
).
Kinetic constants of soluble (Figure 5A) and SCB-immobilized BGL (Figure 5B) were determined with p-NPG at pH 5.5 and 50 °C (optimal condition). Km of the soluble and immobilized enzyme were 0.26 mM and 0.46 mM, respectively, and Vmax values of 7.2 µmol ρ-NP min-1 and 4.37 µmol p-NP min-1. These results indicated that the affinity of immobilized BGL by p-NPG was lower than when compared to the soluble enzyme. This difference in affinity can be associated to the microenvironment created by the enzyme-support interaction, which promoted conformational changes that affected the catalytic function (Simões et al., 2011Simões, A. S., Mori, R. Y., Faria, R., Mendes, A. A., & Castro, H. F. (2011). Performance of hybrid matrix silica-chitosan to immobilize microbial lipase from Candida rugosa. Química Nova, 34, 33-38.).
Effect of p-NPG concentration on the activity of soluble (A) and sugarcane bagasse-immobilized soybean β-glucosidase (B).
The stability of soluble and SCB-immobilized BGL’s was evaluated for different periods of time and temperatures at pH 5.5 (Figure 6A). Soluble and immobilized enzyme was thermally stable at 30 °C, with activity reductions of 16% and 5%, respectively, after 240 min of incubation. At 50 °C, the activities of the soluble and immobilized enzyme decreased by 78% and 15% after 60 min incubation. After 240 min, the immobilized enzymes retained 37% of the original activity, while the soluble enzyme retained only 6% of the initial activity. At 70 °C, the soluble enzyme showed no activity after 20 min, whereas the immobilized enzymes retained 15% activity after 180 min incubation. The reduction in the activity of the soluble enzyme was greater than the one of immobilized enzyme, indicating that the immobilization increased the thermal stability. The increase of thermal stability in immobilized systems can be due to the increased stiffness of the enzymes, which preserved the conformation of the tertiary structure in different environments (Wang et al., 2010Wang, F., Su, R., Qi, W., Zhang, M. J., & He, Z. M. (2010). Preparation and activity of bubbling-immobilized cellobiase within chitosan-alginate composite. Preparative Biochemistry & Biotechnology, 40(1), 57-64. PMid:20024795. http://dx.doi.org/10.1080/10826060903392939
http://dx.doi.org/10.1080/10826060903392...
).
Effect of thermal stability of soluble and immobilized β-glucosidase at pH 5.5 (A) and reusability of immobilized β-glucosidase with the substrate p-NPG at pH 5.5 at 50 °C for 30 min (B).
The reusability of SCB-immobilized BGL using p-NPG was analyzed over 15 cycles (Figure 6B). After 4 cycles, the immobilized enzyme retained 84% of its activity. After 15 cycles, the immobilized enzyme showed 13% of its initial activity. The activity loss could be associated to progressive weakening bonds in the immobilization matrix due to release and three-dimensional distortions that likely occurred from the recurrent exposure of the catalytic site to substrates (Dwevedi & Kayastha, 2009Dwevedi, A., & Kayastha, A. M. (2009). Optimal immobilization of β-galactosidase from Pea (PsBGAL) onto Sephadex and chitosan beads using response surface methodology and its applications. Bioresource Technology, 100(10), 2667-2675. PMid:19195879. http://dx.doi.org/10.1016/j.biortech.2008.12.048
http://dx.doi.org/10.1016/j.biortech.200...
).
3.5 Conversion of glucosides to aglycones in soymilk
The different forms of isoflavones from commercial soymilk were quantified by UHPLC (Table 3) and the contents were 0.311 µmol de glucosides mL-1, 0.046 µmol de malonilglucosides mL-1 e 0.021 µmol de aglycones mL-1.
Content of different forms of isoflavones in commercial soymilk treated with soybean β-glucosidase immobilized in modified sugarcane bagasse with 2% NaOH, autoclaved (15 min at 121 °C) and activation with 2.5% glutaraldehyde.
The forms acetilglucosides, malonilglicitin, glicitin, glicitein and daidzein were not detected. The addition of SCB-immobilized BGL (0.003 U) in commercial soymilk at 25 °C or 50 °C for 120 min increased in 14.3% and 23.8% of genistein contents, respectively, compared with the control. The daidzein content was not detected. Possibly, the difference of daidzin content between the control and treatments (0.011-0.012 µmol mL-1) was not enough to the detection of the conversion from daidzin to daidzein. But the difference of genistin content between the control and treatments (0.027-0.031 µmol mL-1) made possible the observation of the conversion to genistein.
Thus, the addition SCB-immobilized enzyme in commercial soymilk increased the aglycone content, even though it was not in optimal conditions (pH 5.5), since the application was carried out in soymilk without changing its characteristics, including its pH being 7.2. Quitosan-immobilized soybean BGL applied in soymilk at optimal conditions of enzyme (pH 5.5 at 50 °C) increased by 24% in the aglycones contents after 60 min of incubation (Grade et al., 2014Grade, L. C., Moreira, A. A., Varea, G. S., Mandarino, J. M. G., Silva, J. B., Ida, E. I., & Ribeiro, M. L. L. (2014). Soybean β-glucosidase immobilisated on chitosan beads and its application in soy drink increase the aglycones. Brazilian Archives of Biology and Technology, 57(5), 766-773. http://dx.doi.org/10.1590/S1516-8913201402331
http://dx.doi.org/10.1590/S1516-89132014...
). The black soymilk added of spent coffee grounds or glass microspheres-immobilized Aspergillus niger BGL increased by 56% in the aglycones content, approximately, after 60 min or 120 min at optimal conditions of enzyme (pH 6 at 50 °C) (Chen et al., 2012Chen, K. I., Lo, Y. C., Su, N. W., Chou, C. C., & Cheng, K. C. (2012). Enrichment of two isoflavone aglycones in black soymilk by immobilized β-glucosidase on solid carriers. Journal of Agricultural and Food Chemistry, 60(51), 12540-12546. PMid:23190054. http://dx.doi.org/10.1021/jf304405t
http://dx.doi.org/10.1021/jf304405t...
,2013Chen, K. I., Lo, Y. C., Liu, C. W., Yu, R. C., Chou, C. C., & Cheng, K. C. (2013). Enrichment of two isoflavone aglycones in black soymilk by using spent coffee grounds as an immobiliser for β-glucosidase. Food Chemistry, 139(1-4), 79-85. PMid:23561081. http://dx.doi.org/10.1016/j.foodchem.2013.01.093
http://dx.doi.org/10.1016/j.foodchem.201...
).
Several factors, such as optimal conditions of activity, its source and substrate availability can influence in the obtainment of the aglycones. The isoflavones content in soymilk varies accordingly raw material and genetic variability of cultivar (harvest, cultivated land) (Lee et al., 2007Lee, S. J., Ahn, J. K., Khanh, T. D., Chun, S. C., Kim, S. L., Ro, H. M., Song, H. K., & Chung, I. M. (2007). Comparison of isoflavone concentrations in soybean (Glycine Max (L.) Merrill) sprouts grown under two different light conditions. Journal of Agricultural and Food Chemistry, 55(23), 9415-9421. PMid:17941689. http://dx.doi.org/10.1021/jf071861v
http://dx.doi.org/10.1021/jf071861v...
) and processing condition (Genovese et al., 2007Genovese, M. I., Barbosa, A. C. L., Pinto, M. S., & Lajolo, F. M. (2007). Commercial soy protein ingredients as isoflavone sources for functional foods. Plant Foods for Human Nutrition, 62(2), 53-58. PMid:17333396. http://dx.doi.org/10.1007/s11130-007-0041-0
http://dx.doi.org/10.1007/s11130-007-004...
). Also, storage and shelf life of commercial soybean products promote significant reduction of isoflavones (Callou et al., 2010Callou, K. R. A., Sadigov, S., Lajolo, F. M., & Genovese, M. I. (2010). Isoflavones and antioxidant capacity of commercial soy-based beverages: Effect of storage. Journal of Agricultural and Food Chemistry, 58(7), 4284-4291. PMid:20199084. http://dx.doi.org/10.1021/jf904130z
http://dx.doi.org/10.1021/jf904130z...
). Consumption of soy beverages has increased in recent years because of its health benefits and absence of lactose and cholesterol. These drinks are the most consumed between soy products (United Soybean Board, 2014United Soybean Board. (2014). Food Industry insights: Consumer attitudes: About soyfood & health, 26th annual survey. Chesterfield. Retrieved in 2019, September 18, from https://www.soyconnection.com/foodindustry/consumer-insights
https://www.soyconnection.com/foodindust...
).
4 Conclusions
The optimal conditions for the immobilization soybean cotyledons BGL in SCB modified with 2% NaOH, autoclaved at 125 °C for 15 min and activated with 2.5% glutaraldehyde show maximum immobilization efficiency of 97%. The bagasse treatments modify the morphological structures of lignin-hemicellulose-cellulose complex, influence the compositions of the minerals and the chemical groups of the fibers and enable the enzyme immobilization. The thermal stability of the immobilized enzymes is greater than that of solubilized enzymes, and it is possible to reuse the same immobilized structure for 15 cycles without a complete loss of activity. The application of a catalyst system content SCB-immobilized soybean BGL, proposed here, can be a promising alternative due to the ease of use, high thermal stability and can be implemented in industrial processes.
Acknowledgements
This work was partially funded by the CNPq (14/2014). AAM would like to thank CAPES for a scholarship.
-
Cite as: Moreira, A. A., Ferreira, A. C. V., Silva, J. B., & Ribeiro, M. L. L. (2019). Treatment of sugarcane bagasse for the immobilization of soybean β-glucosidase and application in soymilk isoflavones. Brazilian Journal of Food Technology, 22, e2018243. https://doi.org/10.1590/1981-6723.24318
-
Funding: CNPq (14/2014) and CAPES for a scholarship.
References
- Albuquerque, T. L., Gomes, S. D. L., D’Almeida, A. P., Fernandez-Lafuente, R., Gonçalves, L. R. B., & Rocha, M. V. P. (2018). Immobilization of β-galactosidase in glutaraldehyde-chitosan and its application to the synthesis of lactulose using cheese whey as feedstock. Process Biochemistry, 73, 65-73. http://dx.doi.org/10.1016/j.procbio.2018.08.010
» http://dx.doi.org/10.1016/j.procbio.2018.08.010 - Association of Official Analytical Chemists – AOAC. (2006). Official methods of analysis of AOAC International (18th ed.). Arlington: AOAC.
- Barbosa, O., Ortiz, C., Berenguer-Murcia, A., Torres, R., Rodrigues, R. C., & Fernandez-Lafuente, R. (2014). Glutaraldehyde in bio-catalysts design: A useful crosslinker and a versatile tool in enzyme immobilization. RSC Advances, 4(4), 1583-1600. http://dx.doi.org/10.1039/C3RA45991H
» http://dx.doi.org/10.1039/C3RA45991H - Bezerra, T. M. S., Bassan, J. C., Santos, V. T. O., Ferraz, A., & Monti, R. (2015). Covalent immobilization of laccase in green coconut fiber and use in clarification of apple juice. Process Biochemistry, 50(3), 417-423. http://dx.doi.org/10.1016/j.procbio.2014.12.009
» http://dx.doi.org/10.1016/j.procbio.2014.12.009 - Callou, K. R. A., Sadigov, S., Lajolo, F. M., & Genovese, M. I. (2010). Isoflavones and antioxidant capacity of commercial soy-based beverages: Effect of storage. Journal of Agricultural and Food Chemistry, 58(7), 4284-4291. PMid:20199084. http://dx.doi.org/10.1021/jf904130z
» http://dx.doi.org/10.1021/jf904130z - Carvalho Neto, A. G. V., Ganzerli Ta, T. A., Cardoso, A. L., Fávaro, S. L., Pereira, A. G. B., Girotto, E. M., & Radovanovic, E. (2014). Development of composites based on recycled polyethylene/sugarcane bagasse fibers. Polymer Composites, 35(4), 768-774. http://dx.doi.org/10.1002/pc.22720
» http://dx.doi.org/10.1002/pc.22720 - Chen, K. I., Lo, Y. C., Liu, C. W., Yu, R. C., Chou, C. C., & Cheng, K. C. (2013). Enrichment of two isoflavone aglycones in black soymilk by using spent coffee grounds as an immobiliser for β-glucosidase. Food Chemistry, 139(1-4), 79-85. PMid:23561081. http://dx.doi.org/10.1016/j.foodchem.2013.01.093
» http://dx.doi.org/10.1016/j.foodchem.2013.01.093 - Chen, K. I., Lo, Y. C., Su, N. W., Chou, C. C., & Cheng, K. C. (2012). Enrichment of two isoflavone aglycones in black soymilk by immobilized β-glucosidase on solid carriers. Journal of Agricultural and Food Chemistry, 60(51), 12540-12546. PMid:23190054. http://dx.doi.org/10.1021/jf304405t
» http://dx.doi.org/10.1021/jf304405t - Chen, W. H., Ye, S. C., & Sheen, H. K. (2011). Hydrolysis characteristics of sugarcane bagasse pretreated by dilute acid solution in a microwave irradiation environment. Applied Energy, 93, 237-244. http://dx.doi.org/10.1016/j.apenergy.2011.12.014
» http://dx.doi.org/10.1016/j.apenergy.2011.12.014 - Companhia Nacional de Abastecimento – CONAB. (2017). Levantamento cana-de açúcar safra 2017/2018 Brasília. Retrieved in 2017, November 9, from http://www.conab.gov.br/OlalaCMS/uploads/arquivos/17_08_24_08_59_54_boletim_cana_portugues_-_2o_lev_-_17-18.pdf
» http://www.conab.gov.br/OlalaCMS/uploads/arquivos/17_08_24_08_59_54_boletim_cana_portugues_-_2o_lev_-_17-18.pdf - Dubois, M., Gilles, M., Hamilton, J. K., Rebers, P. A., & Smith, F. (1956). Colorimetric method for determination of sugars and related substances. Analytical Chemistry, 28(3), 350-356. http://dx.doi.org/10.1021/ac60111a017
» http://dx.doi.org/10.1021/ac60111a017 - Dwevedi, A., & Kayastha, A. M. (2009). Optimal immobilization of β-galactosidase from Pea (PsBGAL) onto Sephadex and chitosan beads using response surface methodology and its applications. Bioresource Technology, 100(10), 2667-2675. PMid:19195879. http://dx.doi.org/10.1016/j.biortech.2008.12.048
» http://dx.doi.org/10.1016/j.biortech.2008.12.048 - Genovese, M. I., Barbosa, A. C. L., Pinto, M. S., & Lajolo, F. M. (2007). Commercial soy protein ingredients as isoflavone sources for functional foods. Plant Foods for Human Nutrition, 62(2), 53-58. PMid:17333396. http://dx.doi.org/10.1007/s11130-007-0041-0
» http://dx.doi.org/10.1007/s11130-007-0041-0 - Grade, L. C., Moreira, A. A., Varea, G. S., Mandarino, J. M. G., Silva, J. B., Ida, E. I., & Ribeiro, M. L. L. (2014). Soybean β-glucosidase immobilisated on chitosan beads and its application in soy drink increase the aglycones. Brazilian Archives of Biology and Technology, 57(5), 766-773. http://dx.doi.org/10.1590/S1516-8913201402331
» http://dx.doi.org/10.1590/S1516-8913201402331 - Handa, C. L., Couto, U. R., Vicensoti, A. H., Georgetti, S. R., & Ida, E. I. (2014). Optimisation of soy flour fermentation parameters to produce β-glucosidase for bioconversion into aglycones. Food Chemistry, 152, 56-65. PMid:24444906. http://dx.doi.org/10.1016/j.foodchem.2013.11.101
» http://dx.doi.org/10.1016/j.foodchem.2013.11.101 - Hillman, G. G., Singh-Gupta, V., Al-Bashir, A. K., Yunker, C. K., Joiner, M. C., Sarkar, F. H., Abrams, J., & Haacke, E. M. (2011). Monitoring sunitinib-induced vascular effects to optimize radiotherapy combined with soy isoflavones in murine xenograft tumor1. Translational Oncology, 4(2), 110-121. PMid:21461174. http://dx.doi.org/10.1593/tlo.10274
» http://dx.doi.org/10.1593/tlo.10274 - Hoareau, W., Trindade, W. G., Siegmund, B., Castellan, A., & Frollini, E. (2004). Sugarcane bagasse and curaua lignins oxidized by chlorine dioxide and reacted with furfuryl alcohol: Characterization and stability. Polymer Degradation & Stability, 86(3), 567-576. http://dx.doi.org/10.1016/j.polymdegradstab.2004.07.005
» http://dx.doi.org/10.1016/j.polymdegradstab.2004.07.005 - Kao, T. H., Chien, J. T., & Chen, B. H. (2008). Extraction yield of isoflavones from soybean cake as affected by solvent and supercritical carbon dioxide. Food Chemistry, 107(4), 1728-1736. http://dx.doi.org/10.1016/j.foodchem.2007.10.015
» http://dx.doi.org/10.1016/j.foodchem.2007.10.015 - Khuong, L. D., Kondo, R., De Leon, R., Kim Anh, T., Shimizu, K., & Kamei, I. (2014). Bioethanol production from alkaline-pretreated sugarcane bagasse by consolidated bioprocessing using Phlebia sp. MG-60. International Biodeterioration & Biodegradation, 88, 62-68. http://dx.doi.org/10.1016/j.ibiod.2013.12.008
» http://dx.doi.org/10.1016/j.ibiod.2013.12.008 - Krisch, J., Takó, M., Papp, T., & Vágvölgyi, C. (2010). Characteristics and potential use of β-glucosidases from zygomycetes. In A. Mendez-Vilas (Ed.), Current research, technology and education topics in applied microbiology and microbial biotechnology (pp. 891-896). Badajoz: Formatex Research Centre.
- Lee, S. J., Ahn, J. K., Khanh, T. D., Chun, S. C., Kim, S. L., Ro, H. M., Song, H. K., & Chung, I. M. (2007). Comparison of isoflavone concentrations in soybean (Glycine Max (L.) Merrill) sprouts grown under two different light conditions. Journal of Agricultural and Food Chemistry, 55(23), 9415-9421. PMid:17941689. http://dx.doi.org/10.1021/jf071861v
» http://dx.doi.org/10.1021/jf071861v - Liu, Z. M., Ho, S. C., Chen, Y. M., Tang, N., & Woo, J. (2014). Effect of whole soy and purified isoflavone daidzein on renal function: A 6 month randomized controlled trial in equol-producing postmenopausal women with prehypertension. Clinical Biochemistry, 47(13-14), 1250-1256. PMid:24877660. http://dx.doi.org/10.1016/j.clinbiochem.2014.05.054
» http://dx.doi.org/10.1016/j.clinbiochem.2014.05.054 - Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). Protein measurement with the folin phenol reagent. The Journal of Biological Chemistry, 193(1), 265-275. PMid:14907713.
- Matsuura, M., & Obata, A. (1993). β-glucosidases from soybeans hydrolyze daidzin and genistin. Journal of Food Science, 58(1), 144-147. http://dx.doi.org/10.1111/j.1365-2621.1993.tb03231.x
» http://dx.doi.org/10.1111/j.1365-2621.1993.tb03231.x - Mosier, N., Wyman, C., Dale, B., Elander, R., Lee, Y. Y., Holtzapple, M., & Ladisch, M. (2005). Features of promising technologies for pretreatment of lignocellulosic biomass. Bioresource Technology, 96(6), 673-686. PMid:15588770. http://dx.doi.org/10.1016/j.biortech.2004.06.025
» http://dx.doi.org/10.1016/j.biortech.2004.06.025 - Nam, K. H., Sung, M. W., & Hwang, K. Y. (2010). Structural insights into the substrate recognition properties of beta-glucosidase. Biochemical and Biophysical Research Communications, 391(1), 1131-1135. PMid:20005197. http://dx.doi.org/10.1016/j.bbrc.2009.12.038
» http://dx.doi.org/10.1016/j.bbrc.2009.12.038 - Potthast, A., Rosenau, T., & Kosma, P. (2006). Analysis of oxidized functionalities in cellulose. In D. Klemm (Ed.), Advancesin polymer science (Vol. 205, pp. 1-48). Switzerland: Springer. http://dx.doi.org/10.1007/12_099
» http://dx.doi.org/10.1007/12_099 - Ribeiro, M. L. L., Mandarino, J. M. G., Carrão-Panizzi, M. C., Oliveira, M. C. N., Campo, C. B. H., Nepomuceno, A. L., & Ida, E. I. (2007). Isoflavone content and β-glucosidase activity in soybean cultivars of different maturity groups. Journal of Food Composition and Analysis, 20(1), 19-24. http://dx.doi.org/10.1016/j.jfca.2006.07.004
» http://dx.doi.org/10.1016/j.jfca.2006.07.004 - Rimbach, G., Boesch-Saadatmandi, C., Frank, J., Fuchs, D., Wenzei, U., Daniel, H., Hall, W. L., & Weinberg, P. D. (2008). Dietary isoflavones in the prevention of cardiovascular disease: A molecular perspective. Food and Chemical Toxicology, 46(4), 1308-1319. PMid:17689850. http://dx.doi.org/10.1016/j.fct.2007.06.029
» http://dx.doi.org/10.1016/j.fct.2007.06.029 - Santos, R. F., Oliveira, C. F., Varéa, G. S., Orradi Da Silva, M. L. C., Ida, E. I., Mandarino, J. M. G., Carrão-Panizzi, M. C., & Ribeiro, M. L. L. (2013). Purification and characterization of soy cotyledon β-glucosidase. Journal of Food Biochemistry, 37(3), 302-312. http://dx.doi.org/10.1111/j.1745-4514.2011.00632.x
» http://dx.doi.org/10.1111/j.1745-4514.2011.00632.x - Serefoglou, E., Litina, K., Gournis, D., Kalogeris, E., Tzialla, A. A., Pavlidis, I. V., Stamatis, H., Maccallini, E., Lubomska, M., & Rudolf, P. (2008). Smectite clays as solid supports for immobilization of β-glucosidase: Synthesis, characterization, and biochemical properties. Chemistry of Materials, 20(12), 4106-4115. http://dx.doi.org/10.1021/cm800486u
» http://dx.doi.org/10.1021/cm800486u - Simões, A. S., Mori, R. Y., Faria, R., Mendes, A. A., & Castro, H. F. (2011). Performance of hybrid matrix silica-chitosan to immobilize microbial lipase from Candida rugosa Química Nova, 34, 33-38.
- Singh, N., Srivastava, G., Talat, M., Raghubanshi, H., Srivastava, O. N., & Kayastha, A. M. (2014). Cicer α-galactosidase immobilization onto functionalized graphene nanosheets using response surface method and its applications. Food Chemistry, 142, 430-438. PMid:24001862. http://dx.doi.org/10.1016/j.foodchem.2013.07.079
» http://dx.doi.org/10.1016/j.foodchem.2013.07.079 - Tsuchihashi, R., Sakamoto, S., Kodera, M., Nohara, T., & Kinjo, J. (2008). Microbial metabolism of soy isoflavones by human intenstinal bacteria strains. Journal of Natural Medicines, 62(4), 456-460. PMid:18648905. http://dx.doi.org/10.1007/s11418-008-0271-y
» http://dx.doi.org/10.1007/s11418-008-0271-y - União das Indústrias de Cana-de-Açúcar – UNICA. (2011). Um fantástico potencial São Paulo. Retrieved in 2014, May 4, from http://www.unica.com.br/convidados/5337876920338419546/um-fantastico-potencial
» http://www.unica.com.br/convidados/5337876920338419546/um-fantastico-potencial - United Soybean Board. (2014). Food Industry insights: Consumer attitudes: About soyfood & health, 26th annual survey Chesterfield. Retrieved in 2019, September 18, from https://www.soyconnection.com/foodindustry/consumer-insights
» https://www.soyconnection.com/foodindustry/consumer-insights - Van Soest, P. J., Robertson, J. B., & Lewis, B. A. (1991). Methods for dietary fiber, neutral detergent fiber and nonstarch polysaccharides in relation to animal nutrition. Journal of Dairy Science, 74(10), 3583-3597. PMid:1660498. http://dx.doi.org/10.3168/jds.S0022-0302(91)78551-2
» http://dx.doi.org/10.3168/jds.S0022-0302(91)78551-2 - Varavinit, S., Chaokasem, N., & Shobsngob, S. (2002). Immobilization of a thermostable alpha-amylase. Scienceasia, 28(3), 247-251. http://dx.doi.org/10.2306/scienceasia1513-1874.2002.28.247
» http://dx.doi.org/10.2306/scienceasia1513-1874.2002.28.247 - Wang, F., Su, R., Qi, W., Zhang, M. J., & He, Z. M. (2010). Preparation and activity of bubbling-immobilized cellobiase within chitosan-alginate composite. Preparative Biochemistry & Biotechnology, 40(1), 57-64. PMid:20024795. http://dx.doi.org/10.1080/10826060903392939
» http://dx.doi.org/10.1080/10826060903392939 - Yoshiara, L. Y., Madera, T. B., Delaroza, F., Silva, J. B., & Ida, E. I. (2012). Optimization of soy isoflavone extraction with different solvents using the simplex-centroid mixture design. International Journal of Food Sciences and Nutrition, 63(8), 978-986. PMid:22621769. http://dx.doi.org/10.3109/09637486.2012.690026
» http://dx.doi.org/10.3109/09637486.2012.690026 - Zaheer, K., & Humayoun Akhtar, M. (2017). An updated review of dietary isoflavones: Nutrition, processing, bioavailability and impacts on human health. Critical Reviews in Food Science and Nutrition, 57(6), 1280-1293. PMid:26565435. http://dx.doi.org/10.1080/10408398.2014.989958
» http://dx.doi.org/10.1080/10408398.2014.989958 - Zhou, Y., Pan, S., Wei, X., Wang, L., & Liu, Y. (2013). Immobilization of β-glucosidase onto magnetic nanoparticles and evaluation the enzymatic properties. BioResources, 8(2), 2605-2619. http://dx.doi.org/10.15376/biores.8.2.2605-2619
» http://dx.doi.org/10.15376/biores.8.2.2605-2619
Publication Dates
-
Publication in this collection
17 Oct 2019 -
Date of issue
2019
History
-
Received
19 Sept 2018 -
Accepted
05 Apr 2019