Abstract
Kefir, a consortium of acetic and lactic bacteria and yeasts, is versatile for biotransformation of vegetable substrates and may improve their bioactive properties. Lentil is a legume rich in vitamins, minerals, complex carbohydrates and proteins, making it an excellent substrate for fermentation and obtaining bioactive molecules. We report the effect of different process variables on antioxidant properties of lentil extracts obtained using kefir as a biotransformation agent and experimental design. The results showed that the factors agitation (rpm) and incubation temperature exerted significant effects. Incubation temperature of 28 °C and agitation of 75 rpm were the most suitable conditions for kefir fermentation and maximizing the antioxidant properties of the fermented lentils. The highest antioxidant activities for fermented products were 4128.39, 35.87 and 23.20 μmol of Trolox equivalents per gram of sample (μmoL TE g-1) measured by Total Antioxidant Capacity, FRAP and DPPH-radical scavenging methods, respectively. Additionally, we detected that the initial fermentation times (in the first six hours) were enough to positively affect the antioxidant properties of the lentil extracts, reaching a percentage increase of up to 270% in the antioxidant activity of the fermented products in relation to those non-fermented samples.
Keywords:
Lentils; Kefir; Fermentation; Bioactive compounds; Experimental design; Antioxidant properties
Resumo
O kefir, um consórcio de bactérias acéticas e láticas, além de leveduras, mostra-se bastante versátil para adaptação a diversos meios de cultivo. A lentilha, por sua vez, é um grão rico em vitaminas, minerais, carboidratos complexos e proteínas, podendo ser um excelente substrato para fermentação e obtenção de moléculas bioativas. O objetivo deste estudo foi avaliar o efeito de diferentes variáveis de processo sobre as propriedades antioxidantes de extratos de lentilhas obtidos utilizando kefir como agente de transformação. Os resultados mostraram que as variáveis de processo exerceram efeitos significativos sobre a obtenção de extratos de lentilhas biotransformados com alto teor de compostos antioxidantes. As atividades antioxidantes mais elevadas para os produtos fermentados foram 4128,39, 35,87 e 23,20 μmol de equivalentes de Trolox por grama de amostra (μmoL TE g-1), detectadas para os métodos Capacidade Antioxidante Total, FRAP e DPPH, respectivamente. Adicionalmente, também foi observado que os tempos iniciais de fermentação (nas primeiras seis horas) foram suficientes para afetar positivamente as propriedades antioxidantes dos extratos de lentilhas, atingindo um aumento percentual de até 270% na atividade antioxidante dos produtos fermentados em relação às amostras não fermentadas.
Palavras-chave:
Lentilhas; Kefir; Fermentação; Compostos bioativos; Delineamento experimental; Propriedades antioxidantes
1 Introduction
Kefir grains comprise the association of different groups of microorganisms including yeasts, acidic lactic and acetic bacteria in a matrix composed of polysaccharides and proteins (Gulitz et al., 2011Gulitz, A., Stadie, J., Wenning, M., Ehrmann, M. A., & Vogel, R. F. (2011). The microbial diversity of water kefir. International Journal of Food Microbiology, 151(3), 284-288. PMid:22000549. http://dx.doi.org/10.1016/j.ijfoodmicro.2011.09.016
http://dx.doi.org/10.1016/j.ijfoodmicro....
; Amorim et al., 2019Amorim, F. G., Coitinho, L. B., Dias, A. T., Friques, A. G. F., Monteiro, B. L., Rezende, L. C. D., Pereira, T. M. C., Campagnaro, B. P., De Pauw, E., Vasquez, E. C., & Quinton, L. (2019). Identification of new bioactive peptides from Kefir milk through proteopeptidomics: Bioprospection of antihypertensive molecules. Food Chemistry, 282, 109-119. PMid:30711094. http://dx.doi.org/10.1016/j.foodchem.2019.01.010
http://dx.doi.org/10.1016/j.foodchem.201...
). The symbiotic activity of these microorganisms gives the kefir grains great versatility in the fermentation of different types of substrates, in addition to being an excellent source of probiotics with potential health benefits for the consumer (Prado et al., 2015Prado, M. R., Blandón, L. M., Vandenberghe, L. P. S., Rodrigues, C., Castro, G. R., Thomaz-Soccol, V., & Soccol, C. R. (2015). Milk kefir: Composition, microbial cultures, biological activities, and related products. Frontiers in Microbiology, 6, 1177. PMid:26579086. http://dx.doi.org/10.3389/fmicb.2015.01177
http://dx.doi.org/10.3389/fmicb.2015.011...
; Reis et al., 2019Reis, S. A., Conceição, L. L., Dias, M. M., Siqueira, N. P., Rosa, D. D., Oliveira, L. L., Matta, S. L. P., & Peluzio, M. C. G. (2019). Kefir reduces the incidence of pre-neoplastic lesions in an animal model for colorectal cancer. Journal of Functional Foods, 53, 1-6. http://dx.doi.org/10.1016/j.jff.2018.11.050
http://dx.doi.org/10.1016/j.jff.2018.11....
). Although most scientific studies aim at obtaining fermented products of milk origin, kefir grains have shown great potential for application in the production of different functional beverages based on fruits and vegetables. This alternative route presents a very interesting appeal to offer new products to a growing market that includes vegan consumers, lactose intolerant and those allergic to dairy products (Fiorda et al., 2017Fiorda, F. A., Pereira, G. V. M., Thomaz-Soccol, V., Rakshit, S. K., Pagnoncelli, M. G. B., Vandenberghe, L. P. S., & Soccol, C. R. (2017). Microbiological, biochemical, and functional aspects of sugary kefir fermentation: A review. Food Microbiology, 66, 86-95. PMid:28576377. http://dx.doi.org/10.1016/j.fm.2017.04.004
http://dx.doi.org/10.1016/j.fm.2017.04.0...
).
Lentils (Lens culinaris L.) are one of the most produced and consumed legumes worldwide. These grains are rich in vitamins, minerals, complex carbohydrates as well as being excellent sources of proteins and containing low lipid contents (Wang & Toews, 2011Wang, N., & Toews, R. (2011). Certain physicochemical and functional properties of fibre fractions from pulses. Food Research International, 44(8), 2515-2523. http://dx.doi.org/10.1016/j.foodres.2011.03.012
http://dx.doi.org/10.1016/j.foodres.2011...
; Zare et al., 2012Zare, F., Champagne, C. P., Simpson, B. K., Orsat, V., & Boye, J. I. (2012). Effect of the addition of pulse ingredients to milk on acid production by probiotic and yoghurt starter cultures. Food Science and Technology, 45, 155-160. http://dx.doi.org/10.1016/j.lwt.2011.08.012
http://dx.doi.org/10.1016/j.lwt.2011.08....
; Agil et al., 2013Agil, R., Gaget, A., Gliwa, J., Avis, T. J., Willmore, W. G., & Hosseinian, F. (2013). Lentils enhance probiotic growth in yogurt and provide added benefit of antioxidant protection. Lebensmittel-Wissenschaft + Technologie, 50(1), 45-49. http://dx.doi.org/10.1016/j.lwt.2012.07.032
http://dx.doi.org/10.1016/j.lwt.2012.07....
; Bautista-Expósito et al., 2018Bautista-Expósito, S., Peñas, E., Dueñas, M., Silván, J. M., Frias, J., & Martínez-Villaluenga, C. (2018). Individual contributions of Savinase and Lactobacillus plantarum to lentil functionalization during alkaline pH-controlled fermentation. Food Chemistry, 257, 341-349. PMid:29622220. http://dx.doi.org/10.1016/j.foodchem.2018.03.044
http://dx.doi.org/10.1016/j.foodchem.201...
). The presence of phytochemicals, such as phenolic acids, flavonoids and other related phenolic compounds can also be pointed out as a notable characteristic of these grains, since these substances are strongly associated with some biological properties, such as antioxidant activity (Zhang et al., 2015Zhang, B., Deng, Z., Ramdath, D. D., Tang, Y., Chen, P. X., Liu, R., Liu, Q., & Tsao, R. (2015). Phenolic profiles of 20 Canadian lentil cultivars and their contribution to antioxidant activity and inhibitory effects on α-glucosidase and pancreatic lipase. Food Chemistry, 172, 862-872. PMid:25442631. http://dx.doi.org/10.1016/j.foodchem.2014.09.144
http://dx.doi.org/10.1016/j.foodchem.201...
; Fiorda et al., 2017Fiorda, F. A., Pereira, G. V. M., Thomaz-Soccol, V., Rakshit, S. K., Pagnoncelli, M. G. B., Vandenberghe, L. P. S., & Soccol, C. R. (2017). Microbiological, biochemical, and functional aspects of sugary kefir fermentation: A review. Food Microbiology, 66, 86-95. PMid:28576377. http://dx.doi.org/10.1016/j.fm.2017.04.004
http://dx.doi.org/10.1016/j.fm.2017.04.0...
).
Biotransformation of vegetables, such as their fermentation, is a recognized strategy to improve the nutritional and biological properties of these substrates. The main effects of fermentation on the biological properties of legumes are related to the bioconversion of conjugated forms of phenolic compounds to their soluble forms with consequent change in the profile of bioactive substances and increase of related biological activities (Agil et al., 2013Agil, R., Gaget, A., Gliwa, J., Avis, T. J., Willmore, W. G., & Hosseinian, F. (2013). Lentils enhance probiotic growth in yogurt and provide added benefit of antioxidant protection. Lebensmittel-Wissenschaft + Technologie, 50(1), 45-49. http://dx.doi.org/10.1016/j.lwt.2012.07.032
http://dx.doi.org/10.1016/j.lwt.2012.07....
; Torino et al., 2013Torino, M. I., Limón, R. I., Martínez-Villaluenga, C., Mäkinen, S., Pihlanto, A., Vidal-Valverde, C., & Frias, J. (2013). Antioxidant and antihypertensive properties of liquid and solid state fermented lentils. Food Chemistry, 136(2), 1030-1037. PMid:23122159. http://dx.doi.org/10.1016/j.foodchem.2012.09.015
http://dx.doi.org/10.1016/j.foodchem.201...
; Simsek et al., 2014Simsek, S., El, S. N., Kancabas Kilinc, A., & Karakaya, S. (2014). Vegetable and fermented vegetable juices containing germinated seeds and sprouts of lentil and cowpea. Food Chemistry, 156, 289-295. PMid:24629970. http://dx.doi.org/10.1016/j.foodchem.2014.01.095
http://dx.doi.org/10.1016/j.foodchem.201...
; Tu et al., 2019Tu, C., Azi, F., Huang, J., Xu, X., Xing, G., & Dong, M. (2019). Quality and metagenomic evaluation of a novel functional beverage produced from soy whey using water kefir grains. Lebensmittel-Wissenschaft + Technologie, 113, 108258. http://dx.doi.org/10.1016/j.lwt.2019.108258
http://dx.doi.org/10.1016/j.lwt.2019.108...
).
Thus, this work reported an optimization study on the cultivation of kefir grains by submerged fermentation using lentils as substrate. The extracts obtained from non-fermented and fermented lentils were comparatively analyzed for their antioxidant properties. The process developed in this work presents an alternative way to obtain a fermented beverage using kefir grains with improved antioxidant properties.
2 Material and methods
2.1 Reagents
The reagents Folin and Ciocalteau’s phenol reagent, sodium carbonate, Trolox (6-hydroxy-2,5,7,8-tetramethlchroman-2-carboxylic acid), DPPH (2,2-diphenyl-1-picrylhydrazyl), ABTS (2,2’-azinobis-3-ethyl-benzthiazoline-6-sulfonic acid), sodium nitrite, aluminum chloride, sodium hydroxide; vanillin, 2,4,6-Tri(2-pyridyl)-s-triazine (TPTZ) and hydrated iron chloride were purchased from Sigma Aldrich. All the other reagents used in the experiments were of analytical grade.
2.2 Preparation of the lentils flour as substrate for submerged fermentation
We used fresh lentil, purchased from a local market (Campinas, Sao Paulo, Brazil), to prepare the lentil flour and used for fermentation process. The lentil grains were crushed in a blender, frozen and freeze-dried. The powdered product obtained was stored under refrigeration to use as substrate for submerged fermentation.
2.3 Kefir grains preparation, pre-inoculum and inoculum production
Kefir grains were obtained from a private household located in Campinas (Sao Paulo, Brazil). The cultivation of the kefir grains was performed using 250 mL Erlenmeyer flasks containing 100 mL of 10% (w/v) sucrose solution. The flasks containing the culture medium were sterilized and the kefir grains were inoculated at an initial concentration of 10% w/v and incubated at 30 °C with renewal of culture medium every 24h. This procedure was carried out for 14 days to reach the amount of biomass required for the fermentation processes. The biomass produced over the days was frozen, lyophilized and stored for use as a pre-inoculum for fermentation. For each test, the lyophilized kefir grains were cultured as described previously for 96 h and then used as inoculum for submerged fermentation.
2.4 Submerged fermentation of lentil flour
For the fermentation process, 50 mL of a 10% (w/v) sucrose solution containing lentil flour at 5% (w/v) was distributed in 125 mL Erlenmeyer flasks and sterilized at 121 °C for 15 min. For each assay, the previously activated kefir grains were inoculated at an initial concentration of 5% (w/v) in the culture media and incubated for 24 and 48h. The control tests were carried out under the same fermentation conditions without addition of the inoculum and evaluated comparatively with their respective fermented samples.
Finally, the fermented media and controls were subjected to centrifugation and filtration through a filter membrane (Whatman® qualitative filter paper nº 1) to obtain an extract free of any solid material. The supernatants were frozen and used as extracts to evaluate the antioxidant properties.
2.5 Effects of the cultivation parameters on antioxidant properties of lentil extracts
We evaluated the effects of kefir grains culture parameters, including agitation (rpm) and incubation temperature using a Central Composite Rotatable Design (CCRD) at different levels (Table 1). The submerged fermentation of lentil flour was performed as previously described.
Matrix of the CCRD used to study the effects of the independent variables agitation and incubation temperature on antioxidant properties of the non-fermented and fermented extracts from lentils using kefir grains.
We used second order equations to define models for the experimental design as describe below (Equation 1):
where Y represents the estimated response by the model, i and j corresponds to the variation of 1 for the variables number (n); β0 is the mean value; βi and βij represent the linear and quadratic coefficients, respectively; xi and xj are the coded independent variables.
The multiple correlation coefficient (R2) and Fisher’s test (variance analysis – ANOVA) were used to verify the statistical adequacy of the proposed coded models to the real points. The software Statistica 13.3 from TIBCO Software Inc. Company (Palo Alto, California, USA) was used on the experimental design, to analyze the data and generate the models.
2.6 Effects of the fermentation time on antioxidant properties of lentil extracts
We evaluated the effect of the fermentation time on antioxidant properties of lentil extracts upon selecting the culture conditions that allowed the production of fermented products with high antioxidant activities from the analysis of CCRD. The fermentation process was carried out in the same way as previously described with fermentation times adjusted to 0, 2, 4, 6, 8, 10 and 12 h. Control assays (non-fermented samples) were also performed without the addition of kefir grains. We performed antioxidant activity assays for all samples, in addition to quantification of total phenolic compounds and pH determination.
2.7 Determination of antioxidant properties
2.7.1 Total antioxidant capacity
Total antioxidant capacity of the extracts from non-fermented and fermented lentils was performed according to the method described by Prieto et al. (1999)Prieto, P., Pineda, M., & Aguilar, M. (1999). Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: Specific application to the determination of vitamin E. Analytical Biochemistry, 269(2), 337-341. PMid:10222007. http://dx.doi.org/10.1006/abio.1999.4019
http://dx.doi.org/10.1006/abio.1999.4019...
. An aliquot of 0.1 mL of the lentil extracts at 0.30 mg mL-1 (non-fermented samples) and 0.50 mg mL-1 (fermented samples), was mixed with 1.0 mL of the reagent solution containing 0.6 M sulphuric acid, 28 mM sodium phosphate and 4 mM ammonium molybdate. The reaction mixtures were then incubated at 90 °C and kept in the dark for 90 min. The samples were cooled to room temperature and the absorbance was measured at 695 nm. We used an appropriate control prepared with 1.0 mL of the reagent solution and 0.1 mL distilled water. A standard curve was prepared with different concentrations of Trolox (0 to 2000 µM) and the results were expressed as µmol of Trolox Equivalents per gram of lentil flour in culture medium (µmol TE g-1).
2.7.2 Determination of DPPH-radical scavenging activity
The DPPH-radical (2,2-diphenyl-1-picrylhydrazyl) scavenging activity of the lentil extracts was determined as described by Bougatef et al. (2009)Bougatef, A., Hajji, M., Balti, R., Lassoued, I., Triki-Ellouz, Y., & Nasri, M. (2009). Antioxidant and free radical-scavenging activities of smooth hound (Mustelus mustelus) muscle protein hydrolysates obtained by gastrointestinal proteases. Food Chemistry, 114(4), 1198-1205. http://dx.doi.org/10.1016/j.foodchem.2008.10.075
http://dx.doi.org/10.1016/j.foodchem.200...
. A 500 µL aliquot of each sample at 2 mg mL-1 was mixed with 500 µL 99.5% ethanol and 125 µL 0.02% DPPH in 99.5% ethanol. The mixture was then kept at room temperature, in the dark for 60 min, and the reduction of the DPPH radical was measured at 517 nm using a UV-visible spectrophotometer (Beckman DU 70 spectrophotometer, Beckman-Coulter, Inc., Fullerton, CA, USA). The control reaction was performed in the same manner, except that distilled water was used instead of sample. A standard curve was prepared with different concentrations of Trolox (0-50 µM) and the results were expressed as µmol TE g-1.
2.7.3 Measurement of Ferric Reducing/Antioxidant Power (FRAP) assay
A FRAP assay was performed according to Benzie & Strain (1996)Benzie, I. F. F., & Strain, J. J. (1996). The ferric reducing ability of plasma (FRAP) as a measure of “Antioxidant Power”: The FRAP assay. Analytical Biochemistry, 239(1), 70-76. PMid:8660627. http://dx.doi.org/10.1006/abio.1996.0292
http://dx.doi.org/10.1006/abio.1996.0292...
with modifications proposed by Wiriyaphan et al. (2012)Wiriyaphan, C., Chitsomboon, B., & Yongsawadigul, J. (2012). Antioxidant activity of protein hydrolysates derived from threadfin bream surimi byproducts. Food Chemistry, 132(1), 104-111. PMid:26434269. http://dx.doi.org/10.1016/j.foodchem.2011.10.040
http://dx.doi.org/10.1016/j.foodchem.201...
. FRAP reagent was prepared by mixing 2.5 mL of 10 mM TPTZ solution in 40 mM HCl; 25 mL of 0.3 M acetate buffer (pH 3.6), and 2.5 mL of 20 mM FeCl3·6H2O solution. Aliquots of 100 µL of lentil extracts at 2 mg mL-1 (non-fermented samples) and 1 mg mL-1 (fermented samples), blank or Trolox (5-250 μM) were mixed with 1 mL of fresh FRAP reagent. The reaction mixture was then incubated at 37 °C for 15 min in a water bath. Absorbance of each reaction was monitored at 593 nm. Results were expressed as µmol TE g-1.
2.8 Total phenolic compounds
The total phenolic content was determined by the reaction with the Folin-Ciocalteau reagent, according to the method described by Roesler et al. (2007)Roesler, R., Catharino, R. R., Malta, L. G., Eberlin, M. N., & Pastore, G. (2007). Antioxidant activity of Annona crassiflora: Characterization of major components by electrospray ionization mass spectrometry. Food Chemistry, 104(3), 1048-1054. http://dx.doi.org/10.1016/j.foodchem.2007.01.017
http://dx.doi.org/10.1016/j.foodchem.200...
. Aliquots of 0.5 mL of each sample were mixed with 2.5 mL of Folin-Ciocalteau aqueous solution (10% v/v) and 2.0 mL of Na2CO3 solution (7.5% w/v). The reaction mixtures were incubated at 50 °C for 5 min and the absorbance was determined at 760 nm. Quantification of the phenolic compounds was performed using a calibration curve of gallic acid (0-0.10 mg mL-1) and the results were expressed as mg of gallic acid equivalent per gram of lentil flour in culture medium (mg GAE g-1).
2.9 Calculations and statistics
The results were statistically analyzed according to the Tukey test and Pearson correlation coefficient, using the software Minitab® 18 from Minitab Inc. (USA). The values were expressed as the arithmetic mean (n = 3) and were considered statistically different or correlated when the p ≤ 0.05.
3 Results and discussion
3.1 Effects of the process parameters on the antioxidant properties of lentil extracts using experimental design (CCRD)
The results obtained from CCRD for the different runs showed that the independent variables (agitation and incubation temperature) had interesting effects on the fermentation process, with great variation of the antioxidant activities according to each culture condition (Table 1). For example, the lentil extracts obtained at 48h fermentation presented antioxidant activity values from 297.81 to 4143.40 μmol TE g-1, from 9.88 to 17.54 μmol TE g-1 and from 26.22 to 56.82 μmol TE g-1 when measured by Total Antioxidant Activity, DPPH-radical scavenging and FRAP methods, respectively (Table 1).
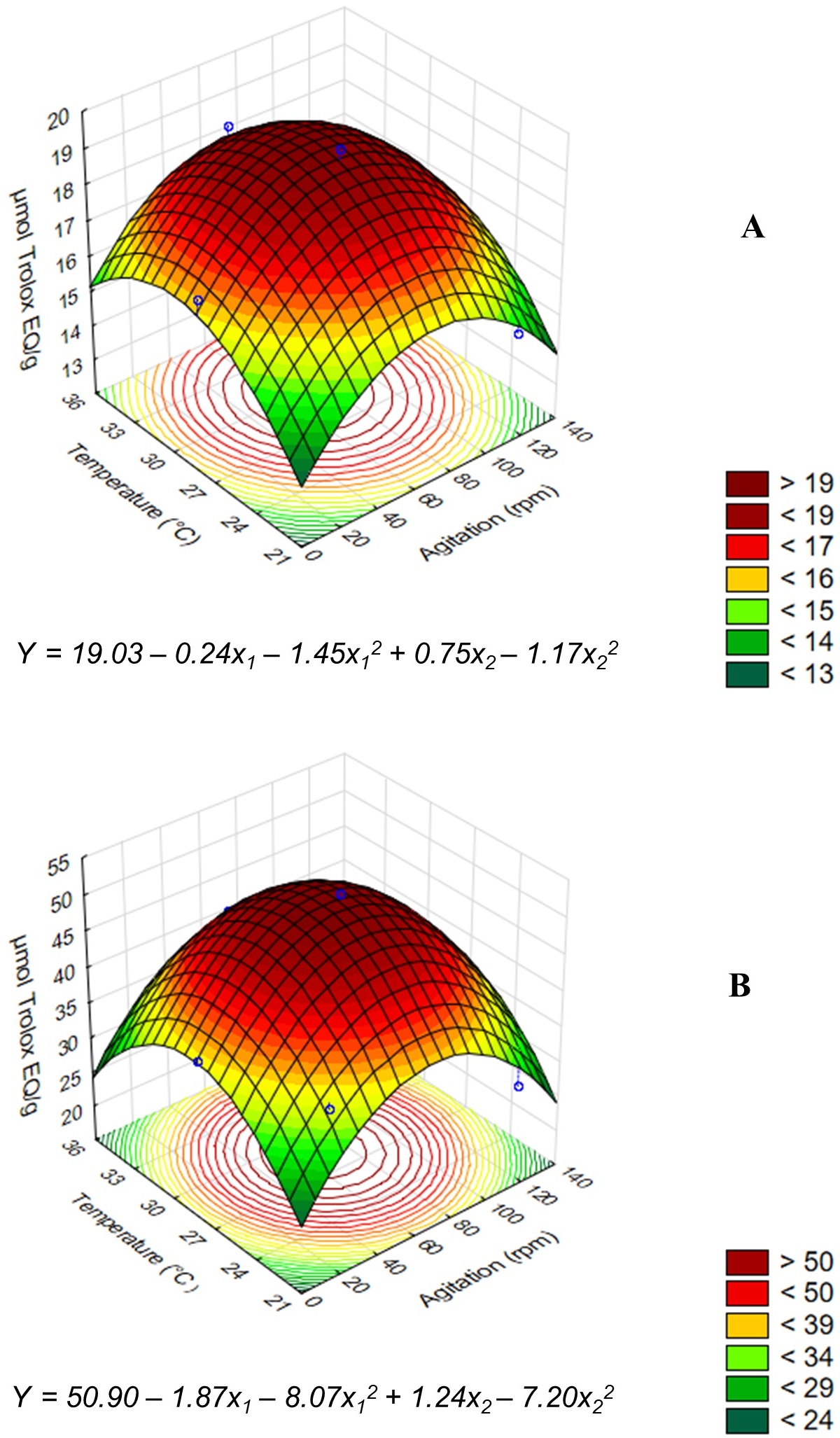
The analysis of effects of the independent variables allows to determine whether the factors studied had a significant or non-significant effect, which may be positive or negative. Regression coefficients that indicate the magnitude and significance of these factors are used to construct equations and predict experimental responses in different growing conditions. In addition to the effects assessment, analysis of variance (ANOVA) should be performed to verify the statistical adequacy of the experimental results. The data presented in Table 2 show the values of F calculated and F tabulated, R2 and p-value for each response. The models with R2 below 0.75, F calculated lower than F tabulated and p-values greater than 0.10 were considered unsatisfactory. As such, four models were generated, allowing the construction of four response surfaces for Total Antioxidant Capacity (Figure 1) and for FRAP (Figure 2). The results for DPPH-radical scavenging did not generate significant models.
Analysis of variance (ANOVA) including Fisher's test (F calculated and F tabulated), multiple correlation coefficient (R2) and probability values (p-value) for the antioxidant properties of the non-fermented and fermented extracts from lentils using kefir grains.
Response surfaces and models equations for antioxidant activity measured by the Total Antioxidant Capacity method for lentil extracts obtained from non-fermented samples (A) after 48 h incubation and for the fermented samples (B) after 48 h fermentation in function of the significant parameters (p ≤ 0.10). The coded values in the equations represent the independent variables and their interactions: x1 = Agitation (rpm) and x2 = Temperature (°C).
Response surfaces and models equations for antioxidant activity measured by FRAP method for lentil extracts obtained from non-fermented samples (A) after 24 h incubation and for the fermented samples (B) after 48 h fermentation in function of the significant parameters (p ≤ 0.10). The coded values in the equations represent the independent variables and their interactions: x1 = Agitation (rpm) and x2 = Temperature (°C).
When analyzing the responses with significant effects (p ≤ 0.10), we observe that incubation temperature and agitation, promoted considerable changes in the antioxidant activity of the lentil extracts. For the non-fermented samples incubated for 48 h and evaluated by the Total Antioxidant Capacity method, it was observed that the linear terms of agitation and temperature were significant (p ≤ 0.10) and positive in the response, whereas the quadratic terms of these same variables were significant (p ≤ 0.10) and negative (Figure 1A). This evaluation allowed to conclude that the increase in the level of the evaluated parameters promoted an increase in the recovery of antioxidant compounds to a certain point, from which there is reduction of the activity. For the lentil extract produced under 48h fermentation, all the significant effects observed were negative, indicating that the increase in the values of agitation and temperature promoted a decrease in the antioxidant properties of the products obtained (Figure 1B).
For the antioxidant activities of the lentil extracts produced after incubation for 24 h (non-fermented lentils) measured by the FRAP method, the linear and quadratic terms of the independent variables were significant (p ≤ 0.10), detecting negative effect for agitation and positive and negative effects for the linear and quadratic terms of temperature, respectively (Figure 2A). In this case, the evaluation indicated that any increase in the agitation level resulted in a decrease in the ferric ion reducing antioxidant power (FRAP), while the increase in temperature exerted an initial positive and then negative effect. For the fermented lentil extract obtained at 48h, the same behavior described above was observed (Figure 2B).
By evaluating the response surfaces, we infer that temperatures in the range from 28 to 30 °C resulted in lentil extracts with higher antioxidant properties (Figure 1 and Figure 2). For the fermented products, the results are in agreement with previous studies already conducted with kefir grains. Viana et al. (2017)Viana, R. O., Magalhães-Guedes, K. T., Braga Junior, R. A., Dias, D. R., & Schwan, R. F.. . (2017). Fermentation process for production of apple-based kefir vinegar: Microbiological, chemical and sensory analysis. Brazilian Journal of Microbiology, 48(3), 592-601. PMid:28283415. http://dx.doi.org/10.1016/j.bjm.2016.11.006
http://dx.doi.org/10.1016/j.bjm.2016.11....
used 28 °C as incubation temperature in a fermentative process using kefir in order to produce an apple-based kefir vinegar and obtained a yield of 79% acetic acid. Fiorda et al. (2016a)Fiorda, F. A., Pereira, G. V. M., Soccol, V. T., Rakshit, S. K., & Soccol, C. R. (2016a). Evaluation of a potentially probiotic non-dairy beverage developed with honey and kefir grains: Fermentation kinetics and storage study. Food Science & Technology International, 22(8), 732-742. PMid:27118768. http://dx.doi.org/10.1177/1082013216646491
http://dx.doi.org/10.1177/10820132166464...
showed that an incubation temperature of 30 °C was the most adequate for high growth of kefir grains. Thus, the temperature range observed as being the most suitable for obtaining compounds with antioxidant properties in our study, is also favorable for the growth and metabolism of kefir microorganisms which is also due to the high content and mesophilic character of lactic and acetic bacteria as well the yeasts present in the grain (Fiorda et al., 2016aFiorda, F. A., Pereira, G. V. M., Soccol, V. T., Rakshit, S. K., & Soccol, C. R. (2016a). Evaluation of a potentially probiotic non-dairy beverage developed with honey and kefir grains: Fermentation kinetics and storage study. Food Science & Technology International, 22(8), 732-742. PMid:27118768. http://dx.doi.org/10.1177/1082013216646491
http://dx.doi.org/10.1177/10820132166464...
).
The agitation effects on antioxidant properties of the lentil extracts were different for each method. When the lentil extracts were obtained under low agitation conditions for the 48h fermentation time, the Total Antioxidant Capacity was higher. Meanwhile, for the FRAP method, the best results were obtained for the lentil extracts produced at 75 rpm agitation for non-fermented and fermented samples (Figure 2). Based on these observations, the culture conditions of the central points from CCRD (75 rpm agitation and 28 °C as incubation temperature) were selected to study the effects of the incubation time on the antioxidant properties of the lentil extracts.
3.2 Effects of the incubation time on the antioxidant properties of lentil extracts
The variation of the antioxidant properties of the lentil extracts as a function of the incubation time was performed in order to verify that shorter fermentation times would exert positive effects on the biological activity of the fermented products, since the observed effects in most of the CCRD runs were negative after 24 and 48 h fermentation. In this case, the analysis of total phenolic compounds was also performed for each incubation time to study possible correlations among the results. Table 3 shows the results obtained for each test as well as the percentage variation calculated as a function of the values observed for the fermented samples in relation to their respective non-fermented samples. The correlation analyzes using the Pearson coefficient showed that there was no significant correlation (p ≥ 0.05) between the antioxidant activity and the phenolic compounds content.
Antioxidant properties and total phenolic compounds for lentil extracts obtained from non-fermented samples and fermented with kefir at 28 °C and 75 rpm agitation during fermentation kinetics.
Results are expressed as mean (n=3) ± standard deviation and those with different letters in the same line for determination in each antioxidant method were considered statistically different at p ≤ 0.05. The variation (%) was calculated as a function of the values of µmol TE g-1 or mg GAE g-1 obtained for the fermented samples in relation to their respective non-fermented sample.
The highest values of antioxidant activity and total phenolic compounds for fermented products were detected in the first incubation times, especially in the first 6h of the process. This knowledge indicates fermentations with kefir for short periods as a good alternative, since time is an important parameter related to the process cost. In addition, high antioxidant activity percentage gains for the extracts obtained from fermented lentils in comparison to their respective non-fermented samples were detected in Total Antioxidant Capacity and FRAP methods, reaching maximum increases of 270% and 105%, respectively, after 4 h of fermentation (Table 3).
The increase in antioxidant properties of lentil extracts may be linked to the biotransformation of macromolecules that carry these possible antioxidants, but in an inactive form, which would be bound to the substrate and without activity. Gunenc et al. (2017)Gunenc, A., Yeung, M. H., Lavergne, C., Bertinato, J., & Hosseinian, F. (2017). Enhancements of antioxidant activity and mineral solubility of germinated wrinkled lentils during fermentation in kefir. Journal of Functional Foods, 3, 72-79. http://dx.doi.org/10.1016/j.jff.2017.02.016
http://dx.doi.org/10.1016/j.jff.2017.02....
suggested that fermentation with kefir is capable of hydrolyzing complex lentil polysaccharides and releasing phenolic compounds, increasing antioxidant activity. These phenolic compounds are commonly associated with fiber and proteins, naturally present in lentils (Gunenc et al., 2017Gunenc, A., Yeung, M. H., Lavergne, C., Bertinato, J., & Hosseinian, F. (2017). Enhancements of antioxidant activity and mineral solubility of germinated wrinkled lentils during fermentation in kefir. Journal of Functional Foods, 3, 72-79. http://dx.doi.org/10.1016/j.jff.2017.02.016
http://dx.doi.org/10.1016/j.jff.2017.02....
).
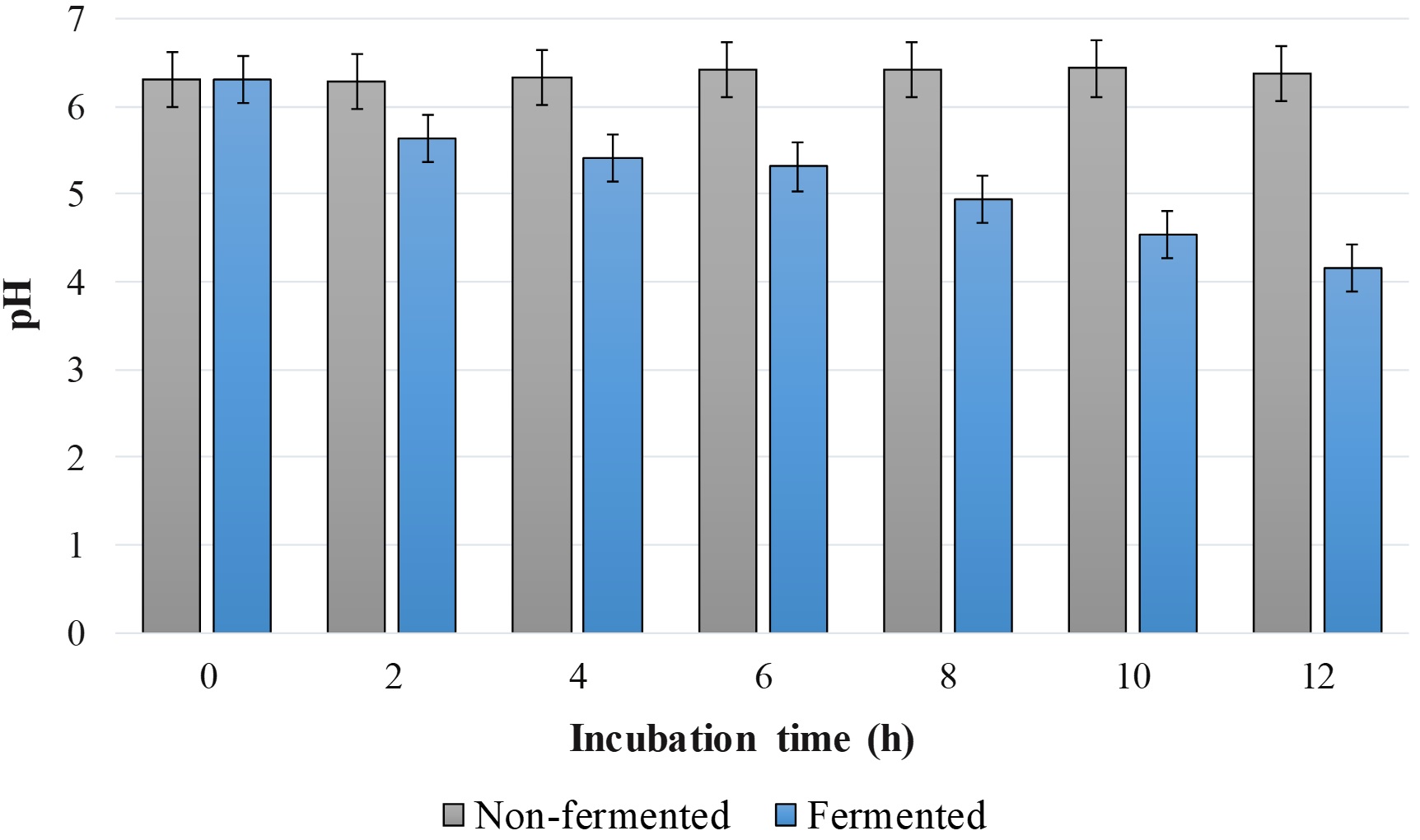
The variation of the pH values in the lentil extracts was monitored in function of the incubation time and the results expressed in Figure 3 confirmed the intense action of the microorganisms in the culture medium from the beginning of the fermentation process.
Variation of pH during incubation of non-fermented and fermented by kefir culture media using lentil flour as substrate.
By evaluating the process kinetics performed, we verified the rapid and continuous reduction of the pH of the medium promoted by the fermentation with the kefir grains, while the samples that were not fermented presented stable pH values for all incubation times evaluated (Figure 3). This behavior corroborates with Fiorda et al. (2016b)Fiorda, F. A., Pereira, G. V. M., Soccol, V. T., Medeiros, A. P., Rakshit, S. K., & Soccol, C. R. (2016b). Development of kefir-based probiotic beverages with DNA protection and antioxidant activities using soybean hydrolyzed extract, colostrum and honey. Food Science and Technology, 68, 690-697. http://dx.doi.org/10.1016/j.lwt.2016.01.003
http://dx.doi.org/10.1016/j.lwt.2016.01....
, who evaluated the fermentation with kefir in soy drink, milk and bovine colostrum. In all cases, the highest pH variation occurred during the first 24 h fermentation. This decrease is due to the production of organic acids, ethanol, carbon dioxide and other volatile substances (Fiorda et al., 2016bFiorda, F. A., Pereira, G. V. M., Soccol, V. T., Medeiros, A. P., Rakshit, S. K., & Soccol, C. R. (2016b). Development of kefir-based probiotic beverages with DNA protection and antioxidant activities using soybean hydrolyzed extract, colostrum and honey. Food Science and Technology, 68, 690-697. http://dx.doi.org/10.1016/j.lwt.2016.01.003
http://dx.doi.org/10.1016/j.lwt.2016.01....
).
Studies involving other biological activities of products fermented with kefir have been reported in the literature. Rodrigues et al. (2016)Rodrigues, K. L., Araújo, T. H., Schneedorf, J. M., Ferreira, C. S., Moraes, G. O. I., Coimbra, R. S., & Rodrigues, M. R. (2016). A novel beer fermented by kefir enhances anti-inflammatory and anti-ulcerogenic activities found isolated in its constituents. Journal of Functional Foods, 21, 58-69. http://dx.doi.org/10.1016/j.jff.2015.11.035
http://dx.doi.org/10.1016/j.jff.2015.11....
evaluated the production of a beer using kefir as a single fermenting agent and verified the anti-inflammatory and anti-ulcerogenic activities of the product obtained. The authors investigated the polyphenol content of beers and in the tests with rats, the following parameters were determined: serum cholesterol, triacylglycerol, HDL cholesterol, as well as the enzymatic activities of alanine aminotransferase, aspartate aminotransferase, catalase and glutathione peroxidase. The results showed that the administration of beer in the animals markedly reduced the inflammatory and ulcerogenic processes. According to the authors, the results obtained suggest a special functionality of a beer made with kefir as a single fermenter. It is important to point out that the determinations carried out in the aforementioned study involve mechanisms directly correlated with the antioxidant compounds of the beer.
4 Conclusion
From the results obtained, we conclude that the independent variables agitation and incubation temperature exerted significant effects on the antioxidant properties of lentil extracts fermented by kefir grains. The fermentation time is also directly linked to the production or biotransformation of compounds with better antioxidant properties from lentils. The conditions defined as more suitable for kefir cultivation, namely: 28 °C as incubation temperature, 75 rpm agitation and 4h of fermentation, resulted in extracts with high antioxidant activity and percentage gains of up to 270% in comparison to the non-fermented samples. Therefore, these results are promising to help in future studies related to the fermentation of lentils by kefir, as well as in research with different substrates.
-
Cite as: Oliveira, I. M. A., & Castro, R. J. S. (2020). Kefir fermentation as a bioprocess to improve lentils antioxidant properties: is it worthwhile?. Brazilian Journal of Food Technology, 23, e2019120. https://doi.org/10.1590/1981-6723.12019
-
Funding: Fundação de Amparo à Pesquisa do Estado de São Paulo. Project nº 2017/02000-9.
References
- Agil, R., Gaget, A., Gliwa, J., Avis, T. J., Willmore, W. G., & Hosseinian, F. (2013). Lentils enhance probiotic growth in yogurt and provide added benefit of antioxidant protection. Lebensmittel-Wissenschaft + Technologie, 50(1), 45-49. http://dx.doi.org/10.1016/j.lwt.2012.07.032
» http://dx.doi.org/10.1016/j.lwt.2012.07.032 - Amorim, F. G., Coitinho, L. B., Dias, A. T., Friques, A. G. F., Monteiro, B. L., Rezende, L. C. D., Pereira, T. M. C., Campagnaro, B. P., De Pauw, E., Vasquez, E. C., & Quinton, L. (2019). Identification of new bioactive peptides from Kefir milk through proteopeptidomics: Bioprospection of antihypertensive molecules. Food Chemistry, 282, 109-119. PMid:30711094. http://dx.doi.org/10.1016/j.foodchem.2019.01.010
» http://dx.doi.org/10.1016/j.foodchem.2019.01.010 - Bautista-Expósito, S., Peñas, E., Dueñas, M., Silván, J. M., Frias, J., & Martínez-Villaluenga, C. (2018). Individual contributions of Savinase and Lactobacillus plantarum to lentil functionalization during alkaline pH-controlled fermentation. Food Chemistry, 257, 341-349. PMid:29622220. http://dx.doi.org/10.1016/j.foodchem.2018.03.044
» http://dx.doi.org/10.1016/j.foodchem.2018.03.044 - Benzie, I. F. F., & Strain, J. J. (1996). The ferric reducing ability of plasma (FRAP) as a measure of “Antioxidant Power”: The FRAP assay. Analytical Biochemistry, 239(1), 70-76. PMid:8660627. http://dx.doi.org/10.1006/abio.1996.0292
» http://dx.doi.org/10.1006/abio.1996.0292 - Bougatef, A., Hajji, M., Balti, R., Lassoued, I., Triki-Ellouz, Y., & Nasri, M. (2009). Antioxidant and free radical-scavenging activities of smooth hound (Mustelus mustelus) muscle protein hydrolysates obtained by gastrointestinal proteases. Food Chemistry, 114(4), 1198-1205. http://dx.doi.org/10.1016/j.foodchem.2008.10.075
» http://dx.doi.org/10.1016/j.foodchem.2008.10.075 - Fiorda, F. A., Pereira, G. V. M., Soccol, V. T., Rakshit, S. K., & Soccol, C. R. (2016a). Evaluation of a potentially probiotic non-dairy beverage developed with honey and kefir grains: Fermentation kinetics and storage study. Food Science & Technology International, 22(8), 732-742. PMid:27118768. http://dx.doi.org/10.1177/1082013216646491
» http://dx.doi.org/10.1177/1082013216646491 - Fiorda, F. A., Pereira, G. V. M., Soccol, V. T., Medeiros, A. P., Rakshit, S. K., & Soccol, C. R. (2016b). Development of kefir-based probiotic beverages with DNA protection and antioxidant activities using soybean hydrolyzed extract, colostrum and honey. Food Science and Technology, 68, 690-697. http://dx.doi.org/10.1016/j.lwt.2016.01.003
» http://dx.doi.org/10.1016/j.lwt.2016.01.003 - Fiorda, F. A., Pereira, G. V. M., Thomaz-Soccol, V., Rakshit, S. K., Pagnoncelli, M. G. B., Vandenberghe, L. P. S., & Soccol, C. R. (2017). Microbiological, biochemical, and functional aspects of sugary kefir fermentation: A review. Food Microbiology, 66, 86-95. PMid:28576377. http://dx.doi.org/10.1016/j.fm.2017.04.004
» http://dx.doi.org/10.1016/j.fm.2017.04.004 - Gulitz, A., Stadie, J., Wenning, M., Ehrmann, M. A., & Vogel, R. F. (2011). The microbial diversity of water kefir. International Journal of Food Microbiology, 151(3), 284-288. PMid:22000549. http://dx.doi.org/10.1016/j.ijfoodmicro.2011.09.016
» http://dx.doi.org/10.1016/j.ijfoodmicro.2011.09.016 - Gunenc, A., Yeung, M. H., Lavergne, C., Bertinato, J., & Hosseinian, F. (2017). Enhancements of antioxidant activity and mineral solubility of germinated wrinkled lentils during fermentation in kefir. Journal of Functional Foods, 3, 72-79. http://dx.doi.org/10.1016/j.jff.2017.02.016
» http://dx.doi.org/10.1016/j.jff.2017.02.016 - Prado, M. R., Blandón, L. M., Vandenberghe, L. P. S., Rodrigues, C., Castro, G. R., Thomaz-Soccol, V., & Soccol, C. R. (2015). Milk kefir: Composition, microbial cultures, biological activities, and related products. Frontiers in Microbiology, 6, 1177. PMid:26579086. http://dx.doi.org/10.3389/fmicb.2015.01177
» http://dx.doi.org/10.3389/fmicb.2015.01177 - Prieto, P., Pineda, M., & Aguilar, M. (1999). Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: Specific application to the determination of vitamin E. Analytical Biochemistry, 269(2), 337-341. PMid:10222007. http://dx.doi.org/10.1006/abio.1999.4019
» http://dx.doi.org/10.1006/abio.1999.4019 - Reis, S. A., Conceição, L. L., Dias, M. M., Siqueira, N. P., Rosa, D. D., Oliveira, L. L., Matta, S. L. P., & Peluzio, M. C. G. (2019). Kefir reduces the incidence of pre-neoplastic lesions in an animal model for colorectal cancer. Journal of Functional Foods, 53, 1-6. http://dx.doi.org/10.1016/j.jff.2018.11.050
» http://dx.doi.org/10.1016/j.jff.2018.11.050 - Rodrigues, K. L., Araújo, T. H., Schneedorf, J. M., Ferreira, C. S., Moraes, G. O. I., Coimbra, R. S., & Rodrigues, M. R. (2016). A novel beer fermented by kefir enhances anti-inflammatory and anti-ulcerogenic activities found isolated in its constituents. Journal of Functional Foods, 21, 58-69. http://dx.doi.org/10.1016/j.jff.2015.11.035
» http://dx.doi.org/10.1016/j.jff.2015.11.035 - Roesler, R., Catharino, R. R., Malta, L. G., Eberlin, M. N., & Pastore, G. (2007). Antioxidant activity of Annona crassiflora: Characterization of major components by electrospray ionization mass spectrometry. Food Chemistry, 104(3), 1048-1054. http://dx.doi.org/10.1016/j.foodchem.2007.01.017
» http://dx.doi.org/10.1016/j.foodchem.2007.01.017 - Simsek, S., El, S. N., Kancabas Kilinc, A., & Karakaya, S. (2014). Vegetable and fermented vegetable juices containing germinated seeds and sprouts of lentil and cowpea. Food Chemistry, 156, 289-295. PMid:24629970. http://dx.doi.org/10.1016/j.foodchem.2014.01.095
» http://dx.doi.org/10.1016/j.foodchem.2014.01.095 - Torino, M. I., Limón, R. I., Martínez-Villaluenga, C., Mäkinen, S., Pihlanto, A., Vidal-Valverde, C., & Frias, J. (2013). Antioxidant and antihypertensive properties of liquid and solid state fermented lentils. Food Chemistry, 136(2), 1030-1037. PMid:23122159. http://dx.doi.org/10.1016/j.foodchem.2012.09.015
» http://dx.doi.org/10.1016/j.foodchem.2012.09.015 - Tu, C., Azi, F., Huang, J., Xu, X., Xing, G., & Dong, M. (2019). Quality and metagenomic evaluation of a novel functional beverage produced from soy whey using water kefir grains. Lebensmittel-Wissenschaft + Technologie, 113, 108258. http://dx.doi.org/10.1016/j.lwt.2019.108258
» http://dx.doi.org/10.1016/j.lwt.2019.108258 - Viana, R. O., Magalhães-Guedes, K. T., Braga Junior, R. A., Dias, D. R., & Schwan, R. F.. . (2017). Fermentation process for production of apple-based kefir vinegar: Microbiological, chemical and sensory analysis. Brazilian Journal of Microbiology, 48(3), 592-601. PMid:28283415. http://dx.doi.org/10.1016/j.bjm.2016.11.006
» http://dx.doi.org/10.1016/j.bjm.2016.11.006 - Wang, N., & Toews, R. (2011). Certain physicochemical and functional properties of fibre fractions from pulses. Food Research International, 44(8), 2515-2523. http://dx.doi.org/10.1016/j.foodres.2011.03.012
» http://dx.doi.org/10.1016/j.foodres.2011.03.012 - Wiriyaphan, C., Chitsomboon, B., & Yongsawadigul, J. (2012). Antioxidant activity of protein hydrolysates derived from threadfin bream surimi byproducts. Food Chemistry, 132(1), 104-111. PMid:26434269. http://dx.doi.org/10.1016/j.foodchem.2011.10.040
» http://dx.doi.org/10.1016/j.foodchem.2011.10.040 - Zare, F., Champagne, C. P., Simpson, B. K., Orsat, V., & Boye, J. I. (2012). Effect of the addition of pulse ingredients to milk on acid production by probiotic and yoghurt starter cultures. Food Science and Technology, 45, 155-160. http://dx.doi.org/10.1016/j.lwt.2011.08.012
» http://dx.doi.org/10.1016/j.lwt.2011.08.012 - Zhang, B., Deng, Z., Ramdath, D. D., Tang, Y., Chen, P. X., Liu, R., Liu, Q., & Tsao, R. (2015). Phenolic profiles of 20 Canadian lentil cultivars and their contribution to antioxidant activity and inhibitory effects on α-glucosidase and pancreatic lipase. Food Chemistry, 172, 862-872. PMid:25442631. http://dx.doi.org/10.1016/j.foodchem.2014.09.144
» http://dx.doi.org/10.1016/j.foodchem.2014.09.144
Publication Dates
-
Publication in this collection
30 Mar 2020 -
Date of issue
2020
History
-
Received
29 Apr 2019 -
Accepted
23 Sept 2019