Abstracts
The present study examined occurrences of ectoparasites and identified them on dogs in rural regions in Brazil, and assessed the influence of climate on these parasites. Ectoparasites were randomly collected from 194 dogs living on farms located in Lavras (n = 92) and Nanuque (n = 102) during the dry season. During the subsequent rainy season, the same dogs in Lavras (n = 71) and Nanuque (n = 66) were resampled. During the experiment, fleas, ticks, lice and fly larvae were collected. The flea species Ctenocephalides felis was the most common ectoparasite collected from these dogs. The main tick species that infested the dogs in rural areas of Nanuque and Lavras was Amblyomma cajennense. In Lavras, the dogs had high levels of flea infestation (80.4 and 88.7% in the dry and rainy seasons, respectively) and low levels of tick infestation (19.6 and 28.2% in the dry and rainy seasons, respectively), without any significant differences in infestation rates between the seasons. In Nanuque, moderate levels of flea infestation (68.6 and 43.9% in the dry and rainy seasons, respectively) and A. cajennense (65.7 and 47.0% in the dry and rainy seasons, respectively) were observed, with significantly lower prevalence in the rainy season (p < 0.05). The presence of ectoparasites was evident at both times of the year, but the different temperatures may have influenced the occurrences of parasites in Lavras and Nanuque.
Ectoparasites; tick; fleas; dog; rural regions; Brazil
O presente estudo examinou a ocorrência e identificação de ectoparasitas em cães de áreas rurais no Brasil, e a influência do clima sobre esses parasitas. Ectoparasitas foram aleatoriamente coletados de 194 cães provenientes de fazendas localizadas em Lavras (n = 92) e Nanuque (n = 102) durante o período seco. No período chuvoso subsequente, os mesmos cães de Lavras (n = 71) e Nanuque (n = 66) foram re-amostrados. Durante o experimento, pulgas, carrapatos, piolhos e larvas de diptera foram coletados. A espécie de pulga Ctenocephalides felis foi o ectoparasita mais comumente encontrado desses cães. A principal espécie de carrapato que infestava os cães nas áreas rurais de Nanuque e Lavras foi Amblyomma cajennense. Em Lavras, os cães tinham altos níveis de infestação de pulgas (80,4 e 88,7% nas estações seca e chuvosa, respectivamente) e baixos níveis de infestação por carrapatos (19,6 e 28,2% nas estações seca e chuvosa, respectivamente), não sendo observada diferença significativa nas taxas de infestação entre as estações. Em Nanuque, níveis moderados de infestação por pulgas (68,6 e 43,9% nas estações seca e chuvosa, respectivamente) e A. cajennense (65,7 e 47,0% na estação seca e chuvosa, respectivamente) foram observadas, com prevalência significativamente menor na estação chuvosa (p < 0,05). A presença de ectoparasitas foi evidente em ambas as épocas do ano, mas as diferentes temperaturas poderiam ter influenciado a ocorrência de parasitas em Lavras e Nanuque.
Ectoparasitas; carrapato; pulga; cães; área rural; Brasil
FULL ARTICLE
Occurrence of ectoparasites on dogs in rural regions of the state of Minas Gerais, Brazil
Ocorrência de ectoparasitos de cães de área rural do estado de Minas Gerais, Brasil
Livio Martins Costa-JuniorI; Karina RembeckII; Filipe Lucas de Melo MendonçaIII; Sandro Coelho AzevedoIII; Lygia Maria Friche PassosII,III; Mucio Flavio Barbosa RibeiroIV
ICentro de Ciências Agrárias e Ambientais, CCAA, Universidade Federal do Maranhão - UFMA, Chapadinha, MA, Brasil
IIInstitute for Comparative Tropical Medicine and Parasitology, Ludwig Maximilian University, Munich, Germany
IIIEscola de Veterinária, Universidade Federal de Minas Gerais - UFMG, Belo Horizonte, MG, Brasil
IVDepartamento de Parasitologia, ICB, Universidade Federal de Minas Gerais - UFMG, Belo Horizonte, MG, Brasil
Corresponding author Corresponding author: Mucio Flavio Barbosa Ribeiro Departamento de Parasitologia, Universidade Federal de Minas Gerais - UFMG Belo Horizonte, MG, Brasil e-mail: muciobr@icb.ufmg.br
ABSTRACT
The present study examined occurrences of ectoparasites and identified them on dogs in rural regions in Brazil, and assessed the influence of climate on these parasites. Ectoparasites were randomly collected from 194 dogs living on farms located in Lavras (n = 92) and Nanuque (n = 102) during the dry season. During the subsequent rainy season, the same dogs in Lavras (n = 71) and Nanuque (n = 66) were resampled. During the experiment, fleas, ticks, lice and fly larvae were collected. The flea species Ctenocephalides felis was the most common ectoparasite collected from these dogs. The main tick species that infested the dogs in rural areas of Nanuque and Lavras was Amblyomma cajennense. In Lavras, the dogs had high levels of flea infestation (80.4 and 88.7% in the dry and rainy seasons, respectively) and low levels of tick infestation (19.6 and 28.2% in the dry and rainy seasons, respectively), without any significant differences in infestation rates between the seasons. In Nanuque, moderate levels of flea infestation (68.6 and 43.9% in the dry and rainy seasons, respectively) and A. cajennense (65.7 and 47.0% in the dry and rainy seasons, respectively) were observed, with significantly lower prevalence in the rainy season (p < 0.05). The presence of ectoparasites was evident at both times of the year, but the different temperatures may have influenced the occurrences of parasites in Lavras and Nanuque.
Keywords: Ectoparasites, tick, fleas, dog, rural regions, Brazil.
RESUMO
O presente estudo examinou a ocorrência e identificação de ectoparasitas em cães de áreas rurais no Brasil, e a influência do clima sobre esses parasitas. Ectoparasitas foram aleatoriamente coletados de 194 cães provenientes de fazendas localizadas em Lavras (n = 92) e Nanuque (n = 102) durante o período seco. No período chuvoso subsequente, os mesmos cães de Lavras (n = 71) e Nanuque (n = 66) foram re-amostrados. Durante o experimento, pulgas, carrapatos, piolhos e larvas de diptera foram coletados. A espécie de pulga Ctenocephalides felis foi o ectoparasita mais comumente encontrado desses cães. A principal espécie de carrapato que infestava os cães nas áreas rurais de Nanuque e Lavras foi Amblyomma cajennense. Em Lavras, os cães tinham altos níveis de infestação de pulgas (80,4 e 88,7% nas estações seca e chuvosa, respectivamente) e baixos níveis de infestação por carrapatos (19,6 e 28,2% nas estações seca e chuvosa, respectivamente), não sendo observada diferença significativa nas taxas de infestação entre as estações. Em Nanuque, níveis moderados de infestação por pulgas (68,6 e 43,9% nas estações seca e chuvosa, respectivamente) e A. cajennense (65,7 e 47,0% na estação seca e chuvosa, respectivamente) foram observadas, com prevalência significativamente menor na estação chuvosa (p < 0,05). A presença de ectoparasitas foi evidente em ambas as épocas do ano, mas as diferentes temperaturas poderiam ter influenciado a ocorrência de parasitas em Lavras e Nanuque.
Palavras chaves: Ectoparasitas, carrapato, pulga, cães, área rural, Brasil.
Introduction
Ectoparasites have a close relationship with their hosts and are important in spreading various diseases among animals and humans. Parasite feeding habits cause direct harm to their hosts in the form of irritation, itching and blood infection and can even induce allergic reactions and dermatitis. Ectoparasites, including fleas, ticks and lice, which infest domestic dogs, are also vectors for several pathogens, such as Rickettsia, Borrelia and Babesia, as well as intermediate hosts for tapeworms and other protozoa (GONZÁLES et al., 2004).
The diversity and abundance of ectoparasites differ between regions according to climatic variation and seasonality. Brazil is a continental country that presents marked differences in climate between several states. However, studies on occurrences of external parasitic infestation, particularly in relation to dogs in rural areas, are scarce. A previous study indicated that Amblyomma ticks are found more frequently than is Rhipicephalus sanguineus (LABRUNA; PEREIRA, 2001). However, additional information about the parasite burden in these areas is important with regard to choosing and implementing prevention and control methodologies.
This study aimed to determine the frequency and intensity of ectoparasite infestation in these regions and to examine the environmental influences on the ectoparasite population among domestic dogs in rural areas of different regions of the state of Minas Gerais, Brazil.
Materials and Methods
1. Location
This study was conducted in two distinct rural areas (Lavras and Nanuque) in the state of Minas Gerais, Brazil. The climatic conditions in these areas differ, although both have two well-defined seasons during the year: a dry season (from April to September) and a rainy season (from October to March). The climatic and geographic data for each location are shown in Table 1.
2. Sampling procedure
From June to August 2004 (dry season), ectoparasites were randomly collected from 194 dogs that lived on farms located in Lavras (n = 92) and Nanuque (n = 102). During the subsequent rainy season, the same dogs were scheduled to be resampled; however, only some of the dogs were sampled in each region: Lavras (n = 71) and Nanuque (n = 66). Each dog was thoroughly examined and the levels of flea and tick infestation were recorded as follows: one to three specimens = low infestation; four to ten = moderate infestation and over ten specimens = high infestation. Samples of the ectoparasites were collected and placed in tubes containing 70% alcohol. Ticks, fleas, lice and fly larvae were identified using proper taxonomic keys (ARAGÃO; FONSECA, 1961; FURMAN; CATTS, 1982; LINARDI; GUIMARÃES, 2000; BARBIERI et al., 2007; MARTINS et al., 2010).
3. Climatic conditions
During the dry season, Lavras had lower temperatures (minimum of 13.5 ºC) and lower humidity than seen in Nanuque, where the minimum temperatures and humidity were higher. A similar comparison could be made during the rainy season, when Nanuque showed higher temperatures and humidity than seen in Lavras (Table 1).
4. Statistical analyses
Statistical analyses were performed using the BioEstat 4.0 software (AYRES et al., 2005). The chi-square test was used to evaluate associations between the ectoparasite infestation rates, seasons and regions. A p value of less than 0.05 was considered to be statistically significant.
Results
1. Collection of ectoparasites
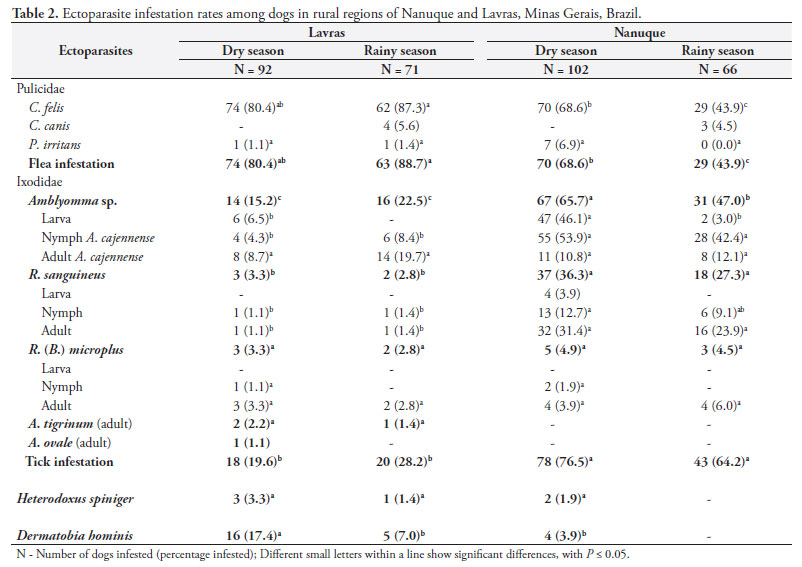
During the experiment, 2407 specimens were collected and identified as fleas, ticks, lice and fly larvae. The sex ratio among the ectoparasites did not change between the regions and seasons. The female-to-male ratio among the parasites was 2.6:1 for fleas and 1:1 for ticks.
2. Flea population
Out of the 92 dogs examined during the dry season in the rural areas of Lavras, 80.4% were infested with fleas and the majority had a low level of infestation (57.6%) (Tables 2 and 3). Ctenocephalides felis was the species most frequently found (80.4%), whereas Pulex irritans was observed on only one dog (1.1%). In the rainy season, the dogs maintained high rates of infestation (88.7%) (p > 0.05), and 50.7% had moderate levels of infestation. At this time, the presence of Ctenocephalides canis was confirmed on 5.6% of the dogs examined.
During the dry season in Nanuque, 68.6% of the dogs examined were infested by fleas and 43.1% had low levels of infestation (Tables 2 and 3). In the rainy season, a significant reduction (p < 0.05) in the infestation rate (43.9%) was observed and the level of infestation was also low. C. felis was the predominant species infesting the animals (43.9%), and C. canis was found on 4.5% of the dogs.
3. Tick population
In the rural areas of Lavras, the dogs had a lower rate (p < 0.05) of infestation by ticks than shown by the dogs in Nanuque. In the dry season (19.6%) and the rainy season (28.2%), the level of infestation did not differ significantly between the dry season (19.6%) and the rainy season (28.2%). The main species that infested the dogs in the Lavras region was A. cajennense, whereas R. sanguineus, Rhipicephalus (Boophilus) microplus, Amblyomma ovale and Amblyomma tigrinum were found sporadically.
In the Nanuque region, the animals showed high rates of tick infestation in the dry season (76.5%) and rainy season (64.2%), with moderate to high intensities of infestation. The main species observed was A. cajennense, which presented higher infestation rates (p < 0.05) in the dry season. R. sanguineus was the second most frequently observed species, with low infestation rates in both seasons. The highest infestation rates for Amblyomma sp. and A. cajennense were, respectively, in relation to larvae (46.1%) and nymphs (53.9%) found in the dry season and nymphs of A. cajennense (42.4%) in the rainy season. Adults of R. sanguineus predominated in both the dry and the rainy season (31.4 and 23.9%, respectively).
4. Other ectoparasites
Chewing lice (Heterodoxus spiniger) were observed on dogs at low infestation rates in the two regions, with higher levels observed in the dry season (3.3%). Dermatobia hominis larvae were observed at higher levels (p < 0.05) among dogs in Lavras, mainly in the dry season (17.4%). In Nanuque, the observed prevalence of these parasites was low (3.9%), and they were not found in any dogs during the rainy season.
Discussion
Ticks and fleas are the principal ectoparasites on dogs from rural areas in Minas Gerais state, with differences observed between seasons and regions. The highest rate of flea infestation was among dogs in rural areas of Lavras, whereas ticks were more prevalent in the Nanuque region. The predominant species of fleas in both regions was C. felis, whereas P. irritans and C. canis were rarely observed. C. felis has been reported as the most prevalent flea species on domestic dogs in several studies in Brazil (LINARDI; NAGEM, 1973; COSTA et al., 1990; RODRIGUES et al., 2001; BELLATO et al., 2003; DANTAS-TORRES et al., 2009). The reason for the high prevalence of C. felis is unclear; however, it is suspected that this species is of African origin and may be better adapted to the hot climate that is typical of tropical countries, compared with C. canis, which is of Palearctic origin (BECK et al., 2006). C. canis has been reported in Brazil, particularly in cold regions, such as the states of Santa Catarina (BELLATTO et al., 2003) and Rio Grande do Sul (OLIVEIRA; RIBEIRO, 1982-1983). In Minas Gerais, Linardi and Nagem (1973) and Rodrigues et al. (2001) found C. canis on dogs in urban areas of municipalities with low average temperatures. Guimarães et al. (2011) found that this species predominated on dogs in an urban area of Lavras, thus indicating that C. canis is adapted to the climatic conditions of this region, where the average temperature is below the state average.
Dogs in Lavras showed higher rates of flea infestation and higher intensities of parasitism during the rainy season, while dogs in Nanuque had higher levels in the dry season. The different climatic conditions in these areas, particularly with regard to temperature and humidity, may interfere with the biological cycles of the fleas in the two regions. Temperatures above 35 ºC and below 3 ºC and relative humidity less than 33% reduce the population of fleas in the environment (SILVERMANN et al., 1981; SILVERMANN; RUST, 1983). During the rainy season in Nanuque, the temperature is high (31 to 34 ºC) and may not benefit the fleas, which are more prevalent in the dry season. In the Lavras region, where the temperature during the dry season is low (13.5 ºC), the fleas are able to develop better during the rainy season.
The sex ratio of the fleas sampled showed that females occurred more frequently during all the periods surveyed. The female-to-male ratio of C. felis found in this study is consistent with previous data from Linardi and Nagem (1973), Oliveira and Ribeiro (1982-1983), Raszl et al. (1998), Rodrigues et al. (2001) and Rinaldi et al. (2007). Male C. canis and P. irritans parasites not were observed on any of the dogs studied.
The dogs in the two areas studied also showed differences in the diversity of tick species and their infestation rates. The most frequent species found in both areas was A. cajennense, which is consistent with the observations of O'Dwyer et al. (2001), who found that Amblyomma ticks were the species most often found infesting dogs in rural areas of Brazil. In the dry season, the presence of larvae of Amblyomma sp. and nymphs of A. cajennense was observed, and in the rainy season, the nymph stage predominated. This finding is consistent with data in the literature, since A. cajennense in the state of Minas Gerais only has one generation per year: the period corresponding to presence of larvae is from April to September (dry season); and nymphs from May to December (rainy season) (OLIVEIRA et al., 2000; LABRUNA et al., 2002).
Dogs in rural areas have constant contact with other farm animals and are reared in continual contact with forest areas, thus suggesting that Amblyomma ticks should be frequently found on these dogs (LABRUNA; PEREIRA 2001; LABRUNA et al., 2001). In this study, we were able to confirm this hypothesis and found a greater number of dogs infested with Amblyomma, in comparison with other tick species.
In a survey on 282 dogs in the metropolitan region of Belo Horizonte, state of Minas Gerais, 32.9% of the dogs were infested with R. sanguineus, and 5.3% were infested with A. cajennense (LINARDI; NAGEM, 1973). In the city of Porto Alegre, southern Brazil, among 236 stray dogs infested with ticks, 93.2% were infested with R. sanguineus and 3.8% with Amblyomma aureolatum (RIBEIRO et al., 1997). Among 102 dogs examined in the urban area of Franca, state of São Paulo, 27.5% were infested only with R. sanguineus (SZABÓ et al., 2001). In these three studies, the dogs were mainly in urban areas, which explains the predominance of R. sanguineus. However, the dogs in rural areas of Nanuque had a high rate of infection with R. sanguineus, which was an unexpected finding. R. sanguineus is a tick with three hosts, using dogs as a primary host for all stages of its development. The presence of dogs is sufficient to maintain the parasite population at a particular site, provided that the environmental conditions are favorable for their development (SILVEIRA et al., 2009). Dantas-Torres et al. (2009) also found high levels of R. sanguineus infestation among dogs in rural areas of São Vicente, state of Pernambuco, Brazil. Dogs in rural areas are often not only close to homes but also have free access to forest areas, which would explain the observations of other tick species.
Climatic conditions are an important factor influencing the dynamics of tick populations. At temperatures below 17 ºC, ticks present delayed oviposition, which influences egg hatching and increases the duration of the parasitic phase, thereby resulting in a reduction in the number of generations within one year (SONENSHINE, 1993). The Lavras region, where the lowest annual mean temperature was observed in the dry season, showed the lowest percentage of tick-infested animals (p < 0.05).
In contrast, the Nanuque region was seen to present an optimal temperature for tick development throughout most of year, and showed higher percentages of infested animals than observed in Lavras. In the present study, there was no direct association between temperature and intensity of tick infestation. Even in Nanuque, the highest intensity of infestation was observed in the dry season, when the temperature is milder and favors the biological cycle of R. sanguineus.
In the tick population sampled, the female-to-male ratio was 1:1, which differed from the data published by Linardi and Nagem (1973) and Dantas-Torres et al. (2009).
The dogs examined in the present study showed low rates of head louse infestation, although the rate was higher during the dry season. This can be explained by the negative influence of rainfall and humidity on the nymph stage of Mallophaga (COSTA; VIEIRA, 1984, SANTOS et al., 2006).
The presence of ectoparasites was evident during both periods of the year, but the different temperatures over the course of the year influenced the occurrences of parasites in Lavras and Nanuque. C. felis was the most common species on the dogs in the present study. The main tick species that infested the dogs in rural areas in Nanuque and Lavras was A. cajennense.
Acknowledgements
We thank the Brazilian National Council for Scientific and Technological Development (CNPq) for the fellowship for L.M. Costa-Júnior and M.F.B. Ribeiro, the Brazilian agency CAPES (Project 182/04) and the German agency DAAD for the financial support for exchange scientists and students.
Received October 27, 2011
Accepted April 14, 2012
- Aragão HB, Fonseca F. Notas de Ixodologia. VIII. Lista e chave para os representantes da fauna Ixodológica Brasileira. Mem Inst Oswaldo Cruz 1961;59(2):115-129.
- Ayres M, Ayres Junior M, Ayres Dl, Santos AS. BioEstat 4. 0: Aplicações estatísticas nas áreas das ciências biológicas e médicas. Belém: Sociedade Civil Mamirauá. 2005.
- Barbieri FS, Chácon SC, Labruna MB, Battesti DMB, Faccini JLH, Famadas KM. Topographical and numerical study of the idiosomal integumentary structures of the larva of four neotropical species of Amblyomma Koch, 1844 (Acari: Ixodidae). Syst Parasitol 2007;68(1):57-70. http://dx.doi.org/10.1007/s11230-006-9078-y
- Beck W, Boch K, Mackensen H, Wiegand B, Pfister K. Qualitative and quantitative observations on the flea population dynamics of dogs and cats in several areas of Germany. Vet Parasitol 2006;137(1-2):130-136. http://dx.doi.org/10.1016/j.vetpar.2005.12.021
- Bellato V, Sartor AA, Souza AP, Ramos BC. Ectoparasitos em caninos do município de Lages, Santa Catarina, Brasil. Rev Bras Parasitol Vet 2003;12(3):95-98.
- Costa AFC, Vieira LS. Ectoparasitos permanentes de caprinos e ovinos em Sobral-CE. Pesq Agropec Bras 1984;19:639-646.
- Costa JO, Guimarães MP, Lima WS, Antunes E. Freqüência de endo e ectoparasitos de cães capturados nas ruas de Vitória- ES, Brasil. Arq Bras Med Vet Zootec 1990;42(5):451-452.
- Dantas-Torres F, Melo MF, Figueredo LA, Brandão-Filho SP. Ectoparasite infestation on rural dogs in the municipality of São Vicente Férrer, Pernambuco, Northeastern Brazil. Rev Bras ParasitolVet 2009;18(3):75-77. http://dx.doi.org/10.4322/rbpv.01803014
- Furman C, Catts DM. Manual of Medical Entomology Cambridge University Press; 1982.
- Gonzáles A, Del-Castro C, Gonzáles S. Ectoparasitic species from Canis familiaris (Linné) in Buenos Aires province, Argentina. Vet Parasitol 2004;120(1-2):123-129. http://dx.doi.org/10.1016/j.vetpar.2003.12.001
- Guimarães AM, Lima BS, Rocha CMBM. Ectofauna parasitária de cães urbanos domiciliados atendidos em clínicas veterinárias particulares na cidade de Lavras, MG. Cien Anim Bras 2011;12(1):172-177.
- Labruna MB, Pereira MC. Carrapatos em cães do Brasil. Clin Vet 2001;30:24-32.
- Labruna MB, Souza SLP, Guimarães Junior JS, Pacheco RC, Pinter A, Gennari SM. Prevalência de carrapatos em cães de áreas rurais da região norte do Estado do Paraná. Arq Bras Med Vet Zootec 2001;53(5):553-556. http://dx.doi.org/10.1590/S0102-09352001000500007
- Labruna MB, Kasai N, Ferreira F, Faccini JLH, Gennari SM. Seasonal dynamics of ticks (Acari: Ixodidae) on horses in the state of São Paulo, Brazil. Vet Parasitol 2002;105(1):65-77. http://dx.doi.org/10.1016/S0304-4017(01)00649-5
- Linardi PM, Guimarães LR. Siphonápteros do Brasil São Paulo: Museu de Zoologia da Universidade de São Paulo; 2000.
- Linardi PM, Nagem RL. Pulicídeos e outros ectoparasitos de cães de Belo Horizonte e municípios vizinhos. Rev Bras Biol 1973;33(4):529-537.
- Martins TF, Onofrio VC, Barros-Battesti DM, Labruna MB. Nymphs of the genus Amblyomma (Acari: Ixodidae) of Brazil: descriptions, redescriptions, and identification key. Ticks Tick Borne Dis 2010;1(2):75-99. http://dx.doi.org/10.1016/j.ttbdis.2010.03.002
- O'Dwyer LH, Massard CL, Pereira de Souza JC. Hepatozoon canis infection associated with dog ticks of rural areas of Rio de Janeiro state, Brazil. Vet Parasitol 2001;94(3):143-150. http://dx.doi.org/10.1016/S0304-4017(00)00378-2
- Oliveira PR, Borges LM, Lopes CM , Leite RC. Population dynamics of the free-living stages of Amblyomma cajennense (Fabricius, 1787) (Acari: Ixodidae) on pastures of Pedro Leopoldo, Minas Gerais State, Brazil. Vet Parasitol 2000;92(4):295-301. http://dx.doi.org/10.1016/S0304-4017(00)00322-8
- Oliveira CMB, Ribeiro PB. Espécies de pulgas que parasitam cães em Porto Alegre e suas prevalências mensais. Arq Fac Vet UFRGS 1982-83;10-11:29-33.
- Raszl SM, Cabral DD, Linardi PM. Xenopsylla cheopis em cães do Brasil: Primeiro relato. Arq Bras Med Vet Zootec 1998;50(2):211-212.
- Ribeiro VLS, Weber MA, Fetzer LO, Vargas CRB. Espécies e prevalências das infestações por carrapatos em cães de rua da cidade de Porto Alegre, RS, Brasil. Ciênc Rural 1997;27(2):285-289. http://dx.doi.org/10.1590/S0103-84781997000200019
- Rinaldi L, Spera G, Musella V, Carbone S, Veneziano V, Iori A, et al. A survey of fleas on dogs in Southern Italy. Vet Parasitol 2007;148(3-4):375-378. http://dx.doi.org/10.1016/j.vetpar.2007.06.036
- Rodrigues AFSF, Daemon E, D'Agosto M. Investigação sobre alguns ectoparasitos em cães de rua no município de Juiz de Fora, Minas Gerais. Rev Bras Parasitol Vet 2001;10(1):13-19.
- Santos SB, Faccini JLH, Santos ACG. Variação estacional de Bovicola caprae parasitando caprinos no Estado da Paraíba. Pesq Vet Bras 2006;26(4):249-253. http://dx.doi.org/10.1590/S0100-736X2006000400010
- Silveira JAG, Passos LMF, Ribeiro MFB. Population dynamics of Rhipicephalus sanguineus (Latrielle, 1806) in Belo Horizonte, Minas Gerais state, Brazil. Vet Parasitol 2009;161(3-4):270-275. http://dx.doi.org/10.1016/j.vetpar.2009.01.028
- Silvermann J, Rust MK. Some abiotic factors affecting the survival of the cat flea, Ctenocephalides felis (Siphonaptera: Pulicidae). Environ Entomol 1983;12(2):490-492.
- Silvermann J, Rust MK, Reierson DA. Influence of temperature and humidity on survival and development of the cat flea, Ctenocephalides felis (Siphonaptera: Pulicidae). J Med Entomol 1981;18:78-83.
- Sonenshine DE. Ecology, behavior, and host-parasite interactions. In: Sonenshine DE. Biology of ticks. New York: Oxford University Press; 1993.
- Szabó MPJ, Cunha TM, Pinter A, Vicentini F. Ticks (Acari: Ixodidae) associated with domestic dogs in Franca region, São Paulo, Brazil. Exp Appl Acarol 2001;25(10-11):909-916.
Corresponding author:
Publication Dates
-
Publication in this collection
11 Oct 2012 -
Date of issue
Sept 2012
History
-
Received
27 Oct 2011 -
Accepted
14 Apr 2012