Abstracts
Over recent decades, diseases have been shown to be important causes of extinctions among wild species. Greater emphasis has been given to diseases transmitted by domestic animals, which have been increasing in numbers in natural areas, along with human populations. This study had the aim of investigating the presence of intestinal helminths in wild canids (maned wolf, Chrysocyon brachyurus, and crab-eating fox, Cerdocyon thous) in the Serra do Cipó National Park (43-44º W and 19-20º S) and endo and ectoparasites of domestic dogs in the Morro da Pedreira Environmental Protection Area (an area surrounding the National Park). The Serra do Cipó is located in the state of Minas Gerais, Brazil. Among the enteroparasites found in domestic and wild canids, the following taxons were identified: Ancylostomidae, Trichuridae, Toxocara sp., Spirocerca sp., Physaloptera sp., Strongyloides sp., Cestoda, Dipylidium caninum, Diphyllobothriidae, Hymenolepidae, Anoplocephalidae, Trematoda, Acanthocephala and Isospora sp. Domestic dogs were positive for leishmaniasis and Babesia canis in serological tests. Among the ectoparasites, Rhipicephalus sanguineus, Amblyomma cajennense and Ctenocephalides felis felis were observed in domestic dogs. Variations in the chaetotaxy of the meta-episternum and posterior tibia were observed in some specimens of C. felis felis.
Parasites; Canis familiaris; Chrysocyon brachyurus; Cerdocyon thous; conservation
Nas últimas décadas, as doenças têm sido apontadas como importantes causas de extinção de espécies silvestres. Maior ênfase tem sido dada às doenças transmitidas por animais domésticos que crescem em número, bem como as populações humanas, em áreas naturais. O presente estudo objetivou verificar a presença de helmintos intestinais de canídeos silvestres (lobo-guará - Chrysocyon brachyurus e cachorro-do-mato - Cerdocyon thous) do Parque Nacional da Serra do Cipó (43-44º W e 19-20º S) e endo e ectoparasitos de cães domésticos da Área de Proteção Ambiental Morro da Pedreira (entorno do Parque Nacional). A Serra do Cipó está localizada no Estado de Minas Gerais, Brasil. Entre os enteroparasitos encontrados em canídeos domésticos e silvestres, os táxons seguintes, Ancylostomidae, Trichuridae, Toxocara sp., Spirocerca sp., Physaloptera sp., Strongyloides sp., Cestoda, Dipylidium caninum, Diphyllobothriidae, Hymenolepidae, Anoplocephalidae, Trematoda, Acanthocephala e Isospora sp. foram identificados. Cães domésticos foram positivos para leishmaniose e Babesia canis nos testes sorológicos. Entre os ectoparasitos, Rhipicephalus sanguineus, Amblyomma cajennense, Ctenocephalides felis felis foram observados em cães domésticos. Variações na quetotaxia do metepisterno e tíbia posterior foram observados em alguns espécimes de C. felis felis.
Parasitos; Canis familiaris; Chrysocyon brachyurus; Cerdocyon thous; conservação
FULL ARTICLE
Parasites of domestic and wild canids in the region of Serra do Cipó National Park, Brazil
Parasitos de canídeos domésticos e silvestres na região do Parque Nacional da Serra do Cipó - Brasil
Juliana Lúcia Costa SantosI,II; Noele Borges MagalhãesI; Hudson Andrade dos SantosI,III; Raul Rio RibeiroIV,V; Marcos Pezzi GuimarãesI
ILaboratório de Helmintologia Veterinária, Departamento de Parasitologia, Instituto de Ciências Biológicas, Universidade Federal de Minas Gerais - UFMG, Belo Horizonte, MG, Brasil
IISuperintendencia de Pesquisa, Escola de Saúde Pública de Minas Gerais - ESP-MG, Belo Horizonte, MG, Brasil
IIIUniversidade Metodista Izabela Hendrix, Belo Horizonte, MG, Brasil
IVLaboratório de Sorologia de Leishmanioses, Departamento de Parasitologia, Instituto de Ciências Biológicas, Universidade Federal de Minas Gerais - UFMG, Belo Horizonte, MG, Brasil
VCentro de Ciências Agrárias, Ambientais e Biológicas, Universidade Federal do Recôncavo da Bahia - UFRB, Cruz das Almas, BA, Brasil
Corresponding author Corresponding author: Juliana Lúcia Costa Santos Escola de Saúde Pública de Minas Gerais - ESP-MG Av. Augusto de Lima, 2061, Barro Preto CEP 30190-002, Belo Horizonte, MG, Brasil e-mail: julianalcsantos@yahoo.com.br
ABSTRACT
Over recent decades, diseases have been shown to be important causes of extinctions among wild species. Greater emphasis has been given to diseases transmitted by domestic animals, which have been increasing in numbers in natural areas, along with human populations. This study had the aim of investigating the presence of intestinal helminths in wild canids (maned wolf, Chrysocyon brachyurus, and crab-eating fox, Cerdocyon thous) in the Serra do Cipó National Park (43-44º W and 19-20º S) and endo and ectoparasites of domestic dogs in the Morro da Pedreira Environmental Protection Area (an area surrounding the National Park). The Serra do Cipó is located in the state of Minas Gerais, Brazil. Among the enteroparasites found in domestic and wild canids, the following taxons were identified: Ancylostomidae, Trichuridae, Toxocara sp., Spirocerca sp., Physaloptera sp., Strongyloides sp., Cestoda, Dipylidium caninum, Diphyllobothriidae, Hymenolepidae, Anoplocephalidae, Trematoda, Acanthocephala and Isospora sp. Domestic dogs were positive for leishmaniasis and Babesia canis in serological tests. Among the ectoparasites, Rhipicephalus sanguineus, Amblyomma cajennense and Ctenocephalides felis felis were observed in domestic dogs. Variations in the chaetotaxy of the meta-episternum and posterior tibia were observed in some specimens of C. felis felis.
Keywords: Parasites, Canis familiaris, Chrysocyon brachyurus, Cerdocyon thous, conservation.
RESUMO
Nas últimas décadas, as doenças têm sido apontadas como importantes causas de extinção de espécies silvestres. Maior ênfase tem sido dada às doenças transmitidas por animais domésticos que crescem em número, bem como as populações humanas, em áreas naturais. O presente estudo objetivou verificar a presença de helmintos intestinais de canídeos silvestres (lobo-guará - Chrysocyon brachyurus e cachorro-do-mato - Cerdocyon thous) do Parque Nacional da Serra do Cipó (43-44º W e 19-20º S) e endo e ectoparasitos de cães domésticos da Área de Proteção Ambiental Morro da Pedreira (entorno do Parque Nacional). A Serra do Cipó está localizada no Estado de Minas Gerais, Brasil. Entre os enteroparasitos encontrados em canídeos domésticos e silvestres, os táxons seguintes, Ancylostomidae, Trichuridae, Toxocara sp., Spirocerca sp., Physaloptera sp., Strongyloides sp., Cestoda, Dipylidium caninum, Diphyllobothriidae, Hymenolepidae, Anoplocephalidae, Trematoda, Acanthocephala e Isospora sp. foram identificados. Cães domésticos foram positivos para leishmaniose e Babesia canis nos testes sorológicos. Entre os ectoparasitos, Rhipicephalus sanguineus, Amblyomma cajennense, Ctenocephalides felis felis foram observados em cães domésticos. Variações na quetotaxia do metepisterno e tíbia posterior foram observados em alguns espécimes de C. felis felis.
Palavras-chave: Parasitos, Canis familiaris, Chrysocyon brachyurus, Cerdocyon thous, conservação.
Introduction
Over recent decades, diseases have been shown to be important causes of extinctions among wild species. Greater emphasis has been given to diseases transmitted by domestic animals, which have been increasing in numbers in natural areas, along with human populations.
With regard to conservation of carnivores, diseases are particularly important since many species or populations are already severely threatened due to loss and fragmentation of habitats, hunting and traffic (MURRAY et al. 1999).
Serra do Cipó National Park and Morro da Pedreira Environmental Protected Area (EPA) form a home for populations of three wild canids: the maned wolf (Chrysocyon brachyurus), hoary fox (Lycalopex vetulus) and crab-eating fox (Cerdocyon thous) (CÂMARA; MURTA, 2003). They also form a home for human communities consisting of owners of domestic animals, including dogs. Concerning the threat of extinction, C. thous is not endangered but C. brachyurus is regarded as close to being threatened. In relation to L. vetulus, the data available is too poor to be able to assess its risk of extinction (SILLERO-ZUBIRI et al., 2004).
In a study carried out in this region, Curi (2005) reported that helminths like Acanthocephala, Trematoda, Ancylostomidae, Trichuridae, Hymenolepidae, Physaloptera, Toxocara, Spirometra and Platynosomum were present in the feces of wild canids. Positive serological tests for Leishmania sp., both in domestic and in wild canids, have also been reported, as well as the presence of ectoparasites like fleas and ticks of the genus Ctenocephalides and genus Amblyomma in wild canids.
Since both the National Park and the EPA are fragmented areas, and domestic animals are present within their limits, this study aimed to investigate occurrences of endo and ectoparasites in dogs belonging to residents of the region, and also gastrointestinal helminths in wild canids that form part of the fauna of the park.
Materials and Methods
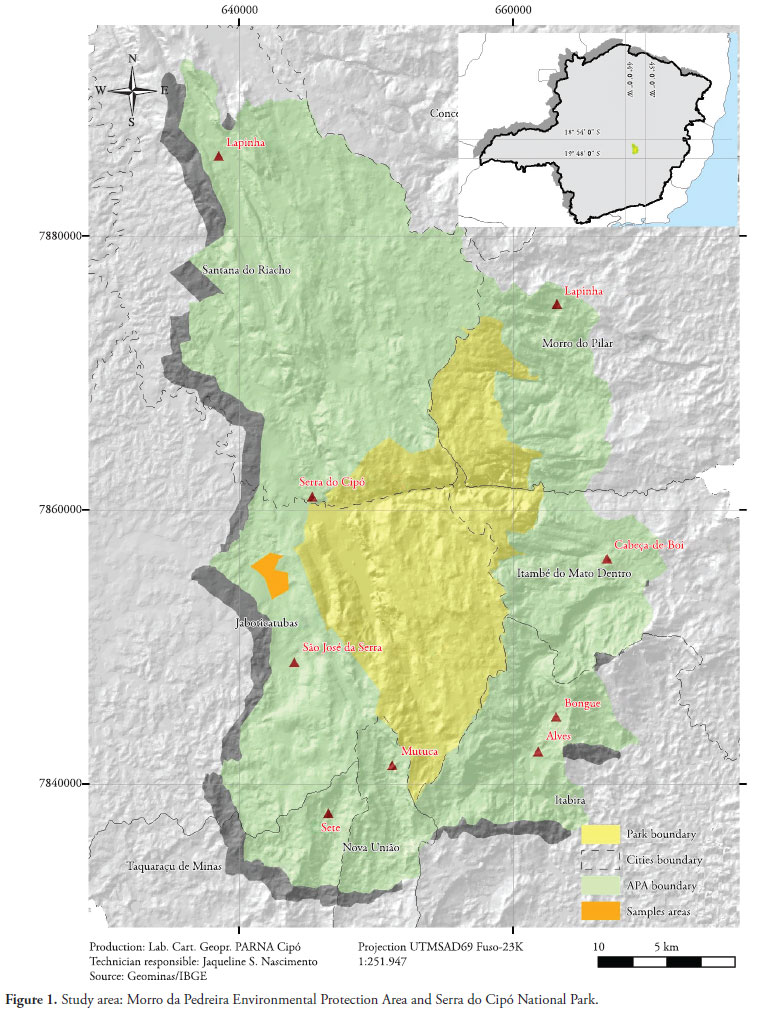
This study was conducted in the Serra do Cipó National Park and the Morro da Pedreira EPA, in three rural districts of Jaboticatubas, between January 2006 and October 2007. The Serra do Cipó National Park has an area of approximately 33,800 ha within a perimeter of 154 km, while the Morro da Pedreira EPA has an area of 66,200 ha within a perimeter of 400 km. Both areas are located in the central portion of the state of Minas Gerais, in the southern part of the Espinhaço mountain range (Figure 1).
Through stimulation of the rectum, feces were obtained from 45 dogs belonging to people living in the surrounding areas. This material was then packed in plastic bags and taken for examination. The methods used for finding parasites in the samples were sedimentation (HOFFMAN et al., 1934), floatation in saturated sodium chloride solution (WILLIS, 1921) and formalin-ether centrifugation (RITCHIE, 1948). Morphometric analysis and identification of parasites were done under an optical microscope.
Necropsies were performed on seven dogs donated by local residents. In order to deal with the dogs, xylazine (Kensol®; 1.1 to 2.2 mg/kg) and ketamine (Vetanarcol®; 10 mg/kg were administered intramuscularly as a preanesthetic. The animals were then sacrificed by means of an intravenous overdose of sodium thiopental (Thiopentax®).
The nematodes collected from the necropsy were fixed in 10% formalin at 60 ºC and then placed between a slide and coverslip and cleared in lactophenol for identification.
The cestodes were stretched out on glass plates and placed under compression between the plates with a 10% formalin solution. Alum acetocarmine staining was applied and the specimens were cleared in creosote.
The helminths were identified by means of optical microscopy (SKRJABIN, 1969; YAMAGUTI, 1961, 1963).
Five milliliters of blood were collected from 56 dogs through puncturing the cephalic or external jugular vein. The samples were centrifuged to obtain serum, which was frozen at -20 ºC. To detect antibodies to Babesia canis and Leishmania sp., the technique used was the indirect immunofluorescence test (IFAT). To detect antibodies to Leishmania sp., the enzyme-linked immunosorbent assay (ELISA) technique was also used. Furthermore, smears were made using peripheral blood obtained from the ears, in order to detect Babesia. The smears were stained using Giemsa. Ectoparasites were collected from 33 dogs living in homes around the Park, including the necropsied dogs. Samples were collected using forceps from various regions of the body. The ectoparasites were placed in vials containing glycerin alcohol (70º GL). Ticks were identified under a stereoscope, at genus and species level, following the criteria described by Aragão and Fonseca (1961). Fleas were identified under an optical microscope, at species level, following the dichotomous key developed by Hopkins and Rothschild (1953). Preparation of the fleas and mounting them on slides followed the techniques used by Neves and Vieira-Filho (1976).
Thirty-eight fecal samples were collected from wild canids on the trails of the Park and the surrounding area. Thirty-three fecal samples were collected from maned wolves and five samples were collected from crab-eating foxes. The samples were identified in terms of size, shape, appearance and smell. The sampling points were marked using the global positioning system (GPS) and the software used to construct the position maps was Arcgis 9.1. The sample packaging, sample processing, methods used for finding parasites in the samples and subsequent identification were carried out in accordance with the same protocol used for domestic dogs. Fecal culturing techniques and the modified Baermann technique were also used (MORAES, 1948).
This study was authorized by the Brazilian Institute for the Environment and Renewable Resources (IBAMA), under research license no. 129/07/DIPI. The study was also approved by the Ethics Committee for Animal Research (CETEA) under protocol no. 18/2007.
Results
Out of the 45 dogs from which stools were obtained, 28 (62.2%) were positive for some type of worm. The most frequent family was Ancylostomidae, which was found in 19 (42.2%) samples. All five samples (100%) collected from crab-eating foxes were positive for some type of worm. Ancylostomidae and Diphyllobothriidae occurred in three samples (60%). Out of the 33 fecal samples collected from maned wolves, 31 (93.9%) were positive for helminths, and Trichuridae occurred in 25 (75.8%) (Table 1).
The map (Figure 2) shows the points where feces were collected from wild canids. The red, gray and blue points indicate the presence of more than one type of parasite.
All seven dogs that were necropsied were found to be infected with helminths. Ancylostoma caninum and Dipylidium caninum were found in four animals (57.1%), while Spirocerca lupi was found in three animals (42.8%) (Table 2).
Out of the 56 dogs from which blood was obtained, two (3.57%) showed positive serological tests for Leishmania sp. Only one showed clinical signs suggestive of canine visceral leishmaniasis, such as: skin disorders (alopecia, skin ulcers or hyperkeratosis), onychogryphosis, weight loss or ophthalmopathies.
Forty-one (73.2%) were positive for Babesia canis. However, in the direct search for parasites on blood smears, none of the dogs was positive.
All 33 dogs sampled for ectoparasites were positive for fleas or ticks. C. felis felis was present in 33 dogs (100%) and, among these, hybrid specimens with characteristics of both C. felis felis and C. canis were found in 15 animals (45.5%). Among the ticks, the species R. sanguineus was found in 29 dogs (87.9%). A. cajennense was found in 27 dogs (81.8%), while 23 dogs (69.7%) were infected with both types of ticks.
Discussion
Endoparasite surveys among dogs in Brazil can be divided into two basic types. One type is based on postmortem examination of street dogs caught by the public health services. The other type is based on fecal examinations of pet dogs at veterinary clinics (LABRUNA et al., 2006). The present study used both approaches on domestic dogs. However, only fecal sample examinations were performed in relation to wild canids, since we did not intend to sacrifice vulnerable or endangered wild species. Labruna et al. (2006) presented a review on occurrences of various gastrointestinal helminths in dogs in Brazilian cities. The helminths most commonly found in fecal examinations were in the Ancylostomidae family (46.3%). This finding is consistent with the results from the present study, in which 42.2% of the domestic dogs were infected by these helminths. In necropsied dogs, Labruna et al. (2006) indicated that A. caninum predominated, with an average occurrence of almost 100%, thus diverging from what was found in this study (57.1%) (Table 2). The Ancylostomidae family was also significantly present in the wild canids (Table 1). We believe that the eggs found in maned wolf feces belonged to A. caninum, since this is the species most usually found in dogs. This species was also previously observed in necropsy results and found in maned wolves in Minas Gerais (MUNDIM et al., 1991).
The Trichuridae family presented high occurrence among the wild canids (75.8% in maned wolves and 20% in crab-eating foxes), in contrast with the domestic dogs, among which only 13.3% showed the parasite (Table 1). The average measurements on the eggs found in stool samples were a length of 64 microns and a width of 35 microns. The morphometric data were consistent with what has been described for C. hepatica, a whipworm species that is widespread in rodents. This is probably the kind of egg found in maned wolf and crab-eating fox feces, since rodents are important items in their diet (JÁCOMO et al., 2004; SILVEIRA, 1999).
The phylum Acanthocephala was significantly present in wild canids but not in domestic dogs (Table 1). Labruna et al. (2006) did not mention occurrences of any species of this phylum in dogs in the Brazilian cities studied. The low prevalence of this parasite in dogs may be related to their diet. The larval forms of Acanthocephala are found in mollusks, crustaceans, insects, fish, reptiles, amphibians and rodents. These items occur only infrequently in dogs' diet, especially those that live in close proximity to humans, as in the study area. Despite the low frequency, Travassos (1917) reported that Moniliformis moniliformis was present in dogs and Prosthenorchis pachyacanthus in wild canids. Since 51.5% of the maned wolves and 40% of the crab-eating foxes presented eggs of these parasites in fecal samples, and considering what the diet of these canids consists of, the infection may have occurred due to predation of rodents. This is more evident in the analysis presented in Figure 2, which shows that the fecal samples containing eggs of Acanthocephala were those that had eggs of C. hepatica. However, further studies are necessary to elucidate the dynamics of infection of these parasites.
Among the 45 dogs that underwent fecal examinations, 15.6% were positive for S. lupi. This proportion was higher than the average of 0.6% average quoted by Labruna et al. (2006). The discrepant results are probably due to the different methods used in each paper, plus the fact that dogs in rural environments have different diets from those in urbanized environments.
Spirocercosis is a disease that affects carnivores, especially canids (VAN DER MERWE et al., 2008). Occurrences of S. lupi and Acanthocephala should follow the same pattern among the canids, since their intermediate and parathenic host overlap. However, domestic dogs showed higher occurrence of S. lupi than seen maned wolves, while the opposite was seen in relation to Acanthocephala (Table 1). This may be due to different eating habits and the areas occupied by these canids in the study region. It was observed that 20% of the samples from crab-eating foxes were positive for S. lupi and 40% for Acanthocephala. This leads us to believe that the species share common areas.
Although all the dogs sampled were infested by fleas of the Ctenocephalides genus, which is the main intermediate host for D. caninum, only 8.9% had ovigerous capsules in the feces. Although small, this value was much greater than the average in Brazilian cities (2.5%) that was recorded by Labruna et al. (2006). However, the average occurrence in these cities in necropsied dogs was 35.8%. In the present study, 57.1% of the dogs had D. caninum. This difference can be explained by the fact that this parasite does not lay eggs. Hence, rupturing of proglottids is necessary for eggs to be released. Most the time, proglottids are actively released by hosts, and so they are not easily found in feces. The absence of this helminth in maned wolves was perhaps for this reason, or because of low flea infestation rates, although Curi (2005) reported that Pulex irritans was present in this animal. Regarding crab-eating foxes, this parasite was present (Table 1). This reinforces the idea that this canid probably has access to places visited by domestic dogs and is therefore contaminated by their ectoparasites.
The Diphyllobothriidae family was significantly present in crab-eating foxes (Table 1). This comprises two genera, Diphyllobothrium and Spirometra, whose eggs are very similar morphologically, thus making accurate identification difficult. Crab-eating foxes have been reported as hosts for D. mansonoides (SCHMIDT, 1986) and D. mansoni was reported by Santos et al. (2004) in this canid. No record of this family was found in the maned wolves or domestic dogs of the present study.
Out of the 56 dogs from which blood was obtained for serologically diagnosing leishmaniasis, only two were reactive, and one of them showed signs consistent with visceral leishmaniasis characteristics. Symptomatic animals have greater potential to be sources of infection for sandflies (SILVA et al., 2005). However, it has been shown that asymptomatic infected dogs are also a source of infection for these insects, thus presenting an active role in the transmission (GONTIJO; MELO, 2004). This is even more significant when apparently healthy dogs are transported from endemic areas to areas where foci have not become established. This may have occurred in the study area of the present study, since many of the dogs sampled were from closed endemic areas. Curi et al. (2006) took the view that a municipal district located a short distance from the present study area, with high epidemiological risk, since canids (both domestic and wild) tested positive for Leishmania sp.
Out of the 33 dogs sampled for ectoparasites, 29 (87.9%) were infested with Rhipicephalus sanguineus, which is the main vector of Babesia canis. Although more than 70% of dogs showed positive results from indirectly surveying B. canis by means of IFAT, none of the dogs were positive in direct searches in peripheral blood smears. This result suggests that the parasitemia level was low, which is a characteristic of subclinical or chronic infection. Canine babesiosis can affect wild canids, since these are also subject to infestation by ticks.
Rhipicephalus sanguineus is found with high prevalence in urban areas, while the opposite occurs in rural environments (LABRUNA et al., 2001). The high occurrence of this species in the study area may have been because most of the dogs were from nearby urban areas, with breeding styles similar to urban environments, with conditions favoring colonization by R. sanguineus (LABRUNA, 2004).
The high occurrence of Amblyomma cajennense that was sampled among the dogs was consistent with other studies conducted in rural areas (LABRUNA et al., 2000, 2001; O'DWYER et al., 2001). This species was found concurrently with R. sanguineus in 23 animals (69.7%). This was probably because dogs spend part of the day outside the home environment, with access to forest remnants (LABRUNA et al., 2000). Findings of R. sanguineus in L. vetulus, C. brachyurus and C. thous have been reported, including in one specimen of each of the first two species kept in captivity, a situation that provides ideal conditions for establishment of R. sanguineus. The other L. vetulus and C. thous specimens were in the wild, but close to domestic dogs in rural areas (LABRUNA et al., 2005). Nonetheless, despite these records, the assertion that these animals act as primary hosts for R. sanguineus should be treated with more caution, since their habits differ substantially from those of dogs.
With regard to fleas, all the dogs were infested by C. felis felis. This is the most common Canidae flea in Brazil, and it has been found in several cities (LINARDI; NAGEM, 1973; RODRIGUES et al., 2001; BELLATO et al., 2003; CASTRO; RAFAEL, 2006). On the other hand, C. canis occurs at lower prevalence, mainly in areas with high or low temperatures (LINARDI; NAGEM, 1973). Although the mean annual temperature in the Serra do Cipó Natural Park remains around 21.2 ºC, and even though no specimen of C. canis has been found, this species may also occur in the study area. This can be concluded because some specimens of C. f. felis have presented altered chaetotaxy on the meta-episternum and metatibia. This suggests that hybridization may occur between the two species, as previously proposed by Holland (1949), Fox (1953), Amin et al. (1974) and Amin (1976). However more studies are needed in order to confirm this suggestion. In Brazil, similar results were also observed by Nagem (1977), in examining fleas from dogs in the municipality of São João d'El Rei, state of Minas Gerais and also by Fernandes et al. (1996), in examining dogs and cats in the municipality of Rio de Janeiro, state of Rio de Janeiro.
Although the presence of domestic dogs inside the Serra do Cipó National Park had already been reported, no signs of these animals traveling along trails (droppings and footprints) were found at the sites where feces of wild canids were collected in the present study. Adjacent to the park, where human communities are still present, dogs and feces were found. Interestingly, on this side of the park, there were no fecal samples from wild canids. Local residents reported, not long ago, seeing these animals in the vicinity. It seems that overlapping between areas used by domestic and wild canids occurs more frequently around the Park, especially in localities where "foxes" are spotted by the locals. It is not known whether transmission of parasites occurs among the canids in the study area. Further studies are needed in order to determine the rate of contact between these animals and their exposure to pathogens, combined with parasitological monitoring through various tests. Furthermore, it is important to learn more, not only about the disease, but also about the ecology of domestic dogs living near protected areas. This will help in foreseeing and avoiding impacts caused by these animals on wild populations. Likewise, it is imperative to adopt preventive measures, including vaccination, deworming and controlling of ectoparasites of dogs. Education for the people inhabiting these sites is also needed in order to keep these animals healthy and limit their access only to the home environment.
Acknowledgements
To the staff of the Brazilian Institute for the Environment and Renewable Resources (IBAMA); and especially to Jaqueline, Celso and Sandro, at the Veterinary Protozoology Laboratory and the Leishmaniasis Serological Diagnosis Laboratory (ICB-UFMG), and to Nelson Curi for fieldwork help.
Received November 23, 2011
Accepted April 26, 2012
- Amin OM, Wells TR, Gately HL. Comb variations the cat flea, Ctenocephalides f. felis (Bouché). Ann Entomol Soc Am 1974;67(6):831-834.
- Amin OM. Host associations and seasonal occurrence of fleas from Southeastern Wisconsin mammals with observations on morphologic variations. J Med Entomol 1976;13(2):179-192. PMid:978691.
- Aragão H, Fonseca F. Notas de Ixodologia. VIII. Lista e chave para os representantes da Fauna Ixodológica brasileira. Mem Inst Oswaldo Cruz 1961;59(2):115-130. PMid:13861962.
- Bellato V, Sartor AA, Souza A, Ramos BC. Ectoparasitos em caninos do município de Lages, Santa Catarina, Brasil. Rev Bras Parasitol Vet 2003;12(3):95-98.
- Câmara T, Murta R. Mamíferos da Serra do Cipó Belo Horizonte: Puc-Minas, Museu de Ciências Naturais; 2003.
- Castro MCM, Rafael JA. Ectoparasitos de cães e gatos da cidade de Manaus, Amazonas, Brasil. Acta Amaz 2006;36(4):535-538. http://dx.doi.org/10.1590/S0044-59672006000400015
- Curi NHA. Avaliação do estado de saúde e do risco de transmissão de doenças entre canídeos (Mammalia, Carnívora) silvestres e domésticos na região da Serra do Cipó, Minas Gerais: implicações para a conservação [Dissertação]. Belo Horizonte: Pontifícia Universidade Católica de Minas Gerais; 2005.
- Curi NHA, Miranda I, Talamoni SA. Serologic evidence of Leishmania infection in free-ranging wild and domestic canids around a Brazilian National Park. Mem Inst Oswaldo Cruz 2006;101(1):99-101. http://dx.doi.org/10.1590/S0074-02762006000100019
- Fernandes CGN, Linardi PM, Faccine JLH, Moura ST. Pulicídeos de cães e gatos da cidade do Rio de Janeiro (RJ, Brasil) e municípios vizinhos. Rev Univ Rural Ser Cienc Vida 1996;18(1-2):115-118.
- Fox I. Notes on the cat flea in Puerto Rico. Am J Trop Med Hyg 1953;2(2):337-342.
- Gontijo CMF, Melo MN. Leishmaniose Visceral no Brasil: quadro atual, desafios e perspectivas. Rev Bras Epidemiol 2004;7(3):338-349. http://dx.doi.org/10.1590/S1415-790X2004000300011
- Hoffman WA, Pons JA, Janer JL. The sedimentation-concentration method in schistosomiasis mansoni. Puerto Rico J Public Health Trop Med 1934;9:283-291.
- Holland GP. The Siphonaptera of Canada Department of Agriculture Technical Bulletin; 1949. vol. 70.
- Hopkins GHE, Rothschild M. An illustrated catalogue of the Rothschild collection of fleas (Siphonaptera) in the British Museum (Natural History):with keys and short descriptions for the identification of families, genera, species and subspecies. London: Cambridge; 1953. vol. 1.
- Jácomo ATA, Silveira L, Diniz-Filho JAF. Niche separation betwen the maned wolf (Chrysocyon brachyurus), the crab-eating fox (Dusicyon thou) and hoary fox (Dusicyon vetulus) in Central Brazil. J Zool 2004;262(1):99-106. http://dx.doi.org/10.1017/S0952836903004473
- Labruna MB. Biológica-ecologia de Rhipicephalus sanguineus (Acari: Ixodidae). Rev Bras Parasitol Vet 2004;13(S1):123-124.
- Labruna MB, Homem VSF, Heinemann MB, Ferreira Neto JS. Ticks (Acari: Ixodidae) Associated with Rural Dogs in Uruará, Eastern Amazon, Brazil. J Med Entomol 2000;37(5):774-776. http://dx.doi.org/10.1603/0022-2585-37.5.774
- Labruna MB, Jorge RSP, Sana DA, Jácomo ATA, Kashivakura CK, Furtado MM, et al. Ticks (Acari: Ixodida) on wild carnivores in Brazil. Exp Appl Acarol 2005;36(1-2):149-163. http://dx.doi.org/10.1007/s10493-005-2563-1
- Labruna MB, Pena HFJ, Souza SLP, Pinter A, Silva JCR, Ragozo AMA, et al. Prevalência de endoparasitas em cães da área urbana do município de Monte Negro, Rondônia. Arq Inst Biol São Paulo 2006;73(2):183-193.
- Labruna MB, Souza SLP, Guimarães Junior JS, Pacheco RC, Pinter A, Gennari SM. Prevalência de carrapatos em cães de áreas rurais da região norte do Estado do Paraná. Arq Bras Med Vet Zoo 2001;53(5):553-556. PMid:4804478. http://dx.doi.org/10.1590/S0102-09352001000500007
- Linard PM, Nagem RL. Pulicídeos e outros ectoparasitos de cães de Belo Horizonte e municípios vizinhos. Rev Bras Biol 1973;33(4):529-537.
- Van der Merwe LL, Kirberger RM, Clift S, Williams M, Keller N, Naidoo V. Spirocerca lupi infection in the dog: a review. Vet. J 2008;176(3):294-309. http://dx.doi.org/10.1016/j.tvjl.2007.02.032
- Moraes RG. Contribuição para o estudo do Strongyloides stercoralis e da estrongiloidíase no Brasil. Rev Serv Espec Saúde Pública 1948;1:507-524.
- Mundim MJS, Machado MI, Bevilaqua E, Mundim AV, Maywald PG, Oliveira MG. Ocorrência e identificação de Ancilostomatídeos em lobo-guará (Chrysocyon brachyurus Illiger, 1811) da região do Triângulo Mineiro, Minas Gerais, Brasil. Braz J Vet Res Anim Sci 1991;28(1):39-43.
- Murray DL, Kapke CA, Evermann JF, Fuller TK. Infectious disease and the conservation of free-ranging large carnivores. Anim Conserv 1999;2:241-254. http://dx.doi.org/10.1111/j.1469-1795.1999.tb00070.x
- Nagem RL. Sifonápteros da Coleção UFMG: sistemática, relações estruturais, adaptativas e entre hospedeiro/parasito [Tese]. Belo Horizonte: Universidade Federal de Minas Gerais; 1977.
- Neves DP, Vieira-Filho M. Biotécnica: técnicas básicas do laboratório biomédico. Belo Horizonte; 1976.
- O'Dwyer LH, Massard C, Souza JCP. Hepatozoon canis infection associated with dog ticks of rural areas of Rio de Janeiro State, Brazil. Vet Parasitol 2001;94(3):143-150. http://dx.doi.org/10.1016/S0304-4017(00)00378-2
- Ritchie LS. An ether sedimentation technique for routine stool examinations. Bull U S Army Med Dep 1948;8(4):326.
- Rodrigues AFSF, Daemon E, D'Agosto M. Investigação sobre alguns ectoparasitos em cães de rua no município de Juiz de Fora, Minas Gerais. Rev Bras Parasitol Vet 2001;10(1):13-19.
- Santos KR, Catenacci LS, Pestelli MM, Takahira RK, Silva RJ. First report of Diphyllobothrium mansoni (Cestoda, Diphyllobothridae) infecting Cerdocyon thous (Mammalia, Canidae) in Brazil. Arq Bras Med Vet Zootec 2004;56(6):796-798. http://dx.doi.org/10.1590/S0102-09352004000600016
- Schmidt GD. Handbook of tapeworm identification Boca Raton: CRC Press; 1986.
- Sillero-Zubiri C, Hoffmann M, Macdonald DW. Canids: Foxes, wolves, jackals and dogs: Status survey and conservation action plan. Switzerland, Cambridge: IUCN/SSC Canid Specialist Group; 2004.
- Silva AVM, Paula AA, Cabrera MAA, Carreira JCA. Leishmaniose em cães domésticos: aspectos epidemiológicos. Cad Saúde Pública 2005;21(1):324-328. http://dx.doi.org/10.1590/S0102-311X2005000100036
- Silveira L. Ecologia e conservação dos mamíferos carnívoros do Parque Nacional das Emas, Goiás [Dissertação]. Goiânia: Universidade Federal de Goiás, 1999.
- Skrjabin KI. Key to parasitic nematodes. Vol I. Spirurata and filariata Jerusalem: IPST Press Binding; 1969;1:497.
- Travassos L. Contribuições para o conhecimento da fauna helmintolójica brazileira: revisão dos acantocéfalos brazileiros. Mem. Inst. Oswaldo Cruz 1917;19:5-62.
- Willis HH. A simple levitation method for the detection of hookworm ova. Med J Aust 1921;11:375-376.
- Yamaguti S. Systema Helminthum: The Nematodes of Vertebrates. New York: Interscience; 1961. vol. 3(I e II).
- Yamaguti S. Systema Helminthum: Acanthocephala. New York: Interscience; 1963. vol. 5.
Corresponding author:
Publication Dates
-
Publication in this collection
11 Oct 2012 -
Date of issue
Sept 2012
History
-
Received
23 Nov 2011 -
Accepted
26 Apr 2012