Abstracts
The present study details the pathological and parasitological findings of parasitic ventriculitis and nematode infections in the large intestines of two female Rhea americana americana birds. The birds were housed in captivity, and both exhibited poor body condition and lethargy. The rheas were sent to the Veterinary Hospital of the Veterinary School, Universidade Federal de Minas Gerais (UFMG) and, despite medical care, the clinical condition of the birds did not improve. The birds died two days after admission, and were submitted to necropsy. Gross, histopathology and parasitological analysis resulted in the identification of Sicarius uncinipenis, which is associated with parasitic ventriculitis, while Deletrocephalus cesarpintoi was identified in the large intestine of both rheas. The apparent clinical indications, including loss of appetite and death, combined with the discovery of numerous parasites and other pathology changes, supported the conclusion that the death of the birds was caused by the parasitic infection. Further investigations of these infections in free-living and captive rheas are required, such that accurate data on the incidence and pathogenicity of these parasites can be obtained.
Rhea americana americana ; endoparasites; histopathology; Sicarius uncinipenis ; ventriculitis; Deletrocephalus cesarpintoi
O presente estudo relata os achados patológicos e parasitológicos de ventriculite parasitária e da infecção por nematódeo no intestino grosso em duas fêmeas Rhea americana americana. As aves eram mantidas em cativeiro e ambas apresentaram condição corporal ruim e inapetência. As emas foram encaminhadas para o Hospital Veterinário da Escola de Veterinária da UFMG e, apesar dos cuidados médicos, não houve melhora na condição clínica. As aves morreram dois dias após a internação e foram encaminhadas para a necropsia. Ao exame macroscópico, histopatológico e parasitológico, Sicarius uncinipenis foi identificado e associado com ventriculite parasitária, enquanto Deletrocephalus cesarpintoi foi identificado no intestino grosso. Possivelmente, o quadro de inapetência e morte foi causado pela infecção parasitária, pois os parasitos eram numerosos. Mais investigações dessa infecção são necessárias em emas de vida livre e cativeiro, para a obtenção de informações mais precisas da incidência e patogenicidade desses parasitos.
Rhea americana americana ; endoparasitas; histopatologia; Sicarius uncinipenis ; ventriculite; Deletrocephalus cesarpintoi
Introduction
The Greater rhea is native to South America, and belongs to the order Struthioniforme, family Rheidae, and species Rhea americana (Linnaeus, 1758). There are five subspecies of rheas distributed across different regions (ALMEIDA, 2007Almeida MA. Struthioniformes (Ema, Avestruz). In: Cubas ZS, Silva JCR, Catão-Dias JL. Tratado de Animais Selvagens. São Paulo: Roca; 2007. p. 136-157. PMid:17963762). The subspecies americana inhabits the northeast and southeast regions of Brazil, but is concentrated in the states of Mato Grosso and Goiás, including the savannah of the west and northeast regions of Minas Gerais State (ALMEIDA, 2007Almeida MA. Struthioniformes (Ema, Avestruz). In: Cubas ZS, Silva JCR, Catão-Dias JL. Tratado de Animais Selvagens. São Paulo: Roca; 2007. p. 136-157. PMid:17963762). The adult Greater rhea is the largest and heaviest Brazilian bird, with heights ranging between 134-170 cm. Further, the male can reach up to 34.4 kg, while the females can weigh as much as 32 kg (SICK, 1997Sick H. Ornitologia Brasileira. Rio de Janeiro: Nova Fronteira; 1997. PMCid:PMC191379). The plumage of the rhea is grey; the feathers of the abdomen and lower dorsal region are white. The male is distinguished only by black feathers on the head, neck, and on the middle part of the upper dorsum (SICK, 1997Sick H. Ornitologia Brasileira. Rio de Janeiro: Nova Fronteira; 1997. PMCid:PMC191379; ALMEIDA, 2007Almeida MA. Struthioniformes (Ema, Avestruz). In: Cubas ZS, Silva JCR, Catão-Dias JL. Tratado de Animais Selvagens. São Paulo: Roca; 2007. p. 136-157. PMid:17963762). Free-living rheas usually consume leaves, fruit, seeds, insects, and small mammals (SICK, 1997Sick H. Ornitologia Brasileira. Rio de Janeiro: Nova Fronteira; 1997. PMCid:PMC191379). However, the population has declined drastically because of human encroachment, and the rhea is currently listed as near threatened to extinction, according to the Red List of Threatened Species from International Union for the Conservation of Nature and Natural Resources (IUCN) (BIRDLIFE INTERNATIONAL, 2012).
Gastrointestinal nematodes have been described in the genus Rhea, including Sicarius uncinipenis, Molin, 1860, which is a parasite of the ventricle (NEMEJC; LUKESOVA, 2012Nemejc K, Lukesova D. Parasite fauna of ostriches, emus and rheas. Agricul Trop Subtrop 2012; 45(1): 45-50.). Likewise, Deletrocephalus dimidiatus, Diesing, 1851, Deletrocephalus cesarpintoi, Vaz, 1936, and Paradeletrocephalus minor, Molin, 1861 are Strongyles of the small and large intestine of rheas (FREITAS; LENT, 1947bFreitas JFT, Lent H. Revisão da subfamília “Deletrocephalinae” Railliet, 1916 (Nematoda, Strongyloidea). Rev Brasil Biol 1947b; 7(1): 73-100.; VICENTE et al., 1995; TAYLOR et al., 2010Taylor MA, Coop RL, Wall RL. Parasitologia Veterinária. 3rd ed. Rio de Janeiro: uanabara Koogan; 2010.; NEMEJC; LUKESOVA, 2012Nemejc K, Lukesova D. Parasite fauna of ostriches, emus and rheas. Agricul Trop Subtrop 2012; 45(1): 45-50.). Deletrocephalus dimidiatus (Strongyloidea: Deletrocephalidae) is the most common nematode found in the intestinal tract of the Greater rhea (ZETTERMANN et al., 2005Zettermann CD, Nascimento AA, Tebaldi JA, Szabó MJP. Observations on helminth infections of free-living and captive rheas (Rhea americana) in Brazil. Vet Parasitol 2005; 129(1-2): 169-172. PMid:15817218. http://dx.doi.org/10.1016/j.vetpar.2004.12.015
http://dx.doi.org/10.1016/j.vetpar.2004....
). The life cycle of the parasite is directly related to the ingestion of larvae by the bird (NEMEJC; LUKESOVA, 2012Nemejc K, Lukesova D. Parasite fauna of ostriches, emus and rheas. Agricul Trop Subtrop 2012; 45(1): 45-50.). Sicarius uncinipenis (Nematoda, Habronematidae) is located in the submucosa of the ventricle of the genus Rhea, and is the nematode found most frequently in the ventricle of these birds (FREITAS; LENT, 1947aFreitas JFT, Lent H. “Spiruroidea” parasitos de “Rheiformes” (Nematoda). Mem Inst Oswaldo Cruz 1947a; 45(4): 743-760. http://dx.doi.org/10.1590/S0074-02761947000400002
http://dx.doi.org/10.1590/S0074-02761947...
; ZETTERMANN et al., 2005Zettermann CD, Nascimento AA, Tebaldi JA, Szabó MJP. Observations on helminth infections of free-living and captive rheas (Rhea americana) in Brazil. Vet Parasitol 2005; 129(1-2): 169-172. PMid:15817218. http://dx.doi.org/10.1016/j.vetpar.2004.12.015
http://dx.doi.org/10.1016/j.vetpar.2004....
).
The aim of the current study was to characterize the histopathological and parasitological aspects of the endoparasites Sicarius uncinipenis and Deletrocephalus cesarpintoi in two specimens of captive Rhea americana americana in Minas Gerais State, Brazil.
Materials and Methods
In June 2013, two female Rhea americana americana birds (Greater 1 and Greater 2) from the farm of the Universidade Federal de Minas Gerais (UFMG), in the city of Pedro Leopoldo (19° 37′ 02.6″ S and 44° 02′ 22.5″ W), died one to two days after hospitalization at the Veterinary Hospital of UFMG. These birds were born in captivity at Baldim city, 40 km away from the city of Belo Horizonte. Greater 1 and 2 were both two years old, and were transported, together with 16 other birds, to a paddock at the University farm. The staff of the hatchery reported that birds were not eating normally there are two weeks. During transport, Greater 1 suffered a fracture in the right femur. At the new farm, the birds were kept inside a wide paddock, with a less dense population of birds. During hospitalization, the food intake of Greater 1 and 2 decreased dramatically, and no clinical improvements were observed. After two days, the birds died and the carcasses of the birds were refrigerated and later necropsied at the Veterinary School. The remaining 16 birds were treated with Febantel (Panacur®, 40 mg/kg) at day 1 after the death of Greater 1 and Greater 2, as well as 14 days later. Following the administration of Febantel, all of the birds displayed normal eating behavior, and improved body condition.
After gross evaluation of the organs of the deceased rheas, samples including brain, lung, trachea, heart, esophagus, ventricle, proventriculus, intestines, and kidneys were collected for histopathological analysis. The samples were fixed in 10% buffered formalin for 52 h, dehydrated in ethanol, diaphanized in xylene, and embedded in paraffin for histological sectioning. Serial sections of 4 µm were obtained, and prepared for routine histological evaluation under light microscopy (LUNA, 1968).
The digestive tract (esophagus, proventriculus, ventricle, and intestines) of the birds were opened with a longitudinal incision, and the contents were transferred to a clear glass container. After mucosal scraping, the contents were washed in a sieve with 0.297-mm openings and examined under a stereo microscope. The nematodes found in the ventricle and large intestine were collected and washed with 0.85% NaCl solution. For identification purposes, the nematodes were fixed in 10% formalin at 80°C, diaphanized in lactophenol Amann solution, mounted between a glass slide and cover slip, and observed under both a light and stereo microscope.
Results and Discussion
Clinically, Greater 1 and 2 demonstrated lethargy and were unable to stand up and eat. Upon physical examination, poor corporal condition was noted in both birds. After death, necropsy revealed that subcutaneous tissue, blood clots, and muscles were pale red characterizing moderate anemia. Several firm, whitish nodules, measuring approximately 0.5 cm in diameter were apparent in the lungs of the birds, and indicated mild, multifocal granulomatous bronchopneumonia. The main change was observed in the ventricle of both birds, and was marked in Greater 1, and moderate in the Greater 2. The koilin membrane in the ventricle of both birds was ulcerated and detached easily. As well, numerous reddish cylindrical parasites were inserted between the koilin and submucosal layer (Figure 1). Under the koilin membrane, the mucosal surface exhibited many suffusions, and parasites were observed in the proventriculus and ventricle transition, and in the initial portion of duodenum. Adult parasites had apparently migrated through the mucosa and submucosa, and larval forms penetrated the submucosa of the organ, causing traumatic lesions. Ecchymosis was noted on the mucosal surface of the large intestine, as well as numerous white cylindrical parasites. Adjacent to the pituitary gland, a firm white-yellowish nodule (1.0 cm in diameter) was observed in Greater 1.
Sicarius uncinipenis in the ventricle of Rhea americana americana There are numerous reddish and cylindrical parasites measuring 20 mm on average under the koilin membrane or inserted in the koilin membrane with the anterior end facing the lumen.
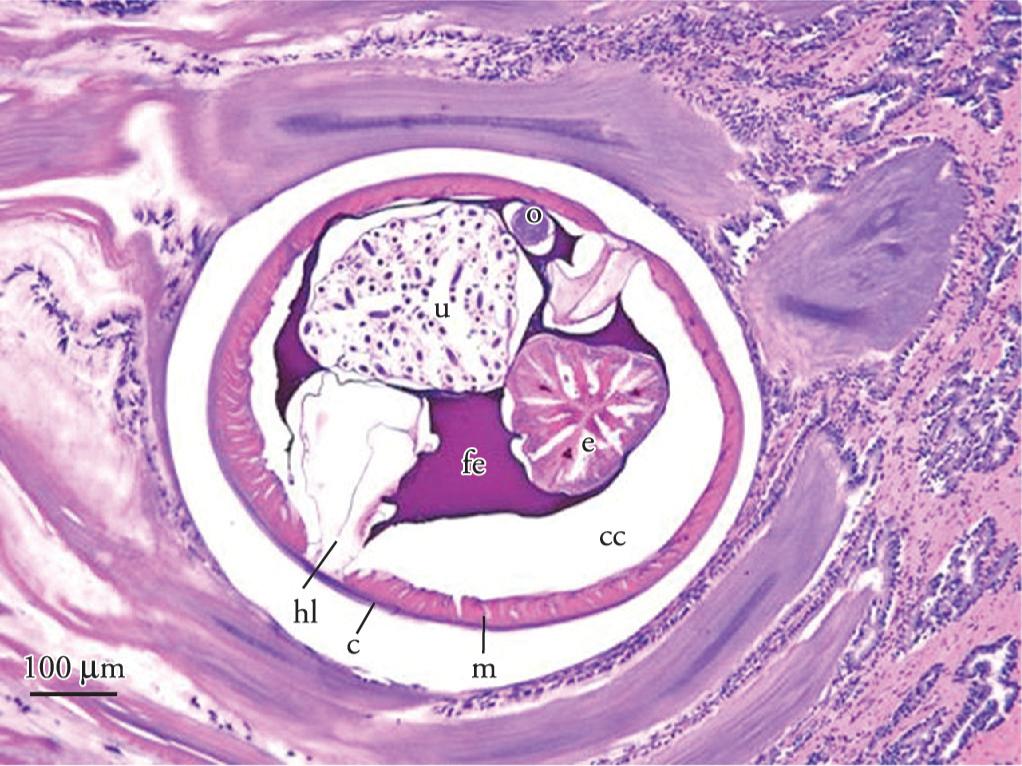
Histopathology revealed caseous necrosis in the lungs, surrounded by heterophils, macrophages, and multinucleate giant cells. In the central area of necrosis, many septa and branching filamentous hyphae compatible with Aspergillus spp. were observed. In the ventricles, there was detachment of the koilin membrane and, mild to moderate infiltration of lymphocytes, histiocytes and heterophils in the submucosa. The inflammatory infiltrate was more intense surrounding free eggs inside the mucosa. In addition, several transversal and longitudinal sections of adult parasites with body cavity, inserted in the koilin and mucosal layer were observed. The parasites had a wall composed of cuticle without projections, hypodermis with two lateral halos, and coelomyarian muscles. Most cross sections of female nematodes revealed ovaries, and a uterus filled with embryonated eggs. The intestine was composed of cylindrical cells, and other sections revealed an esophageal and eosinophilic fluid in the pseudocoelom (Figure 2). Histological characteristics of the parasites allowed the classification as a nematode belonging to the superfamily Spiruroidea (GARDINER; POYNTON, 1999). As additional findings in both rheas, the skeletal muscle showed areas of mild, multifocal coagulative necrosis, which suggested metabolic myopathy post physical effort. The nodule in the pituitary gland of Greater 1 was diagnosed as a keratin-filled cyst. Keratin-filled cysts, which originate from agranular cells of the adenohypophysis, contain macerated keratin and are lined with stratified squamous epithelium (SHIMADA; NAKAMURA, 1988Shimada T, Nakamura F. Dome formation of keratin-containing agranular cells from rat anterior pituitary gland in vitro. In Vitro Cell Dev Biol. 1988; 24(12): 1193-200. http://dx.doi.org/10.1007/BF02624189
http://dx.doi.org/10.1007/BF02624189...
).
Histological cross section of Sicarius uncinipenis inserted in the koilin membrane of the Rhea americana americana. The structures included the cuticle (c), celomyarian muscles (m), lateral halo (hl), body cavity (cc), esophagus (e), uterus filled with embryonated eggs (u), ovary (o) and eosinophilic fluid in the pseudocoelom (fe). BAR = 100 µm.
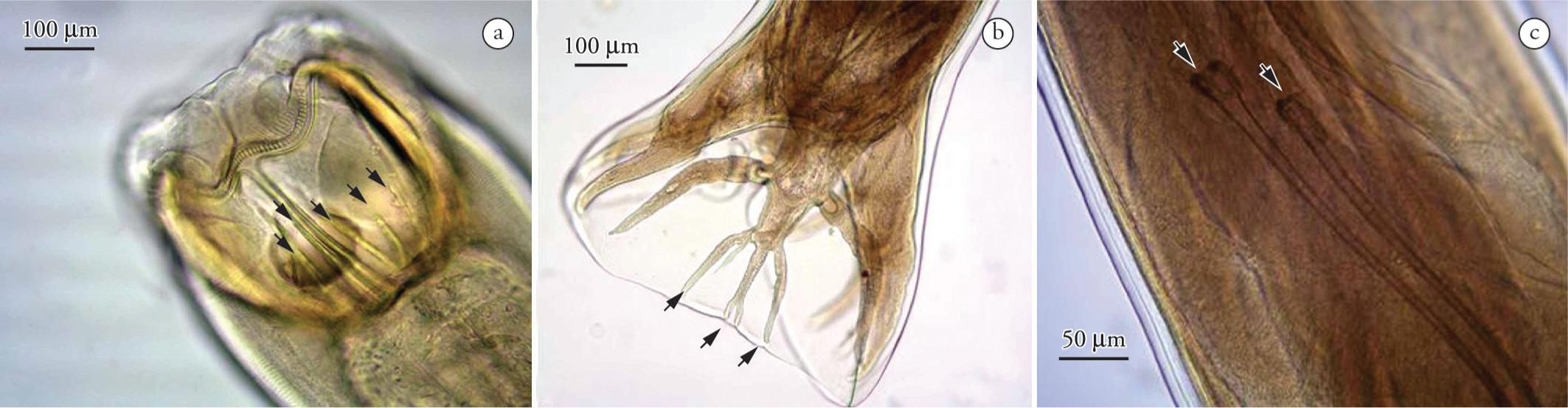
For parasitological evaluation and measurements, five male and five female nematode specimens were used. The male specimens measured 21.57 ± 2.14 mm in length, and 0.63 ± 0.05 mm in width, and females measured 27.53 mm ± 2.75 in length and 0.68 ± 0.07 mm in width. Both genders had a mouth surrounded by two lateral lips and two inner lips, one dorsal and one ventral. The oral capsule was present and contained a chitinized wall (Figure 3a). The male oral capsule measured 0.059 ± 0.005 mm in length, and 0.076 ± 0.005 mm in width, while the female oral capsule measured 0.004 ± 0.056 mm by 0.081 mm ± 0.003, respectively. The esophagus was long and divided into an anterior glandular sections measuring 0.33 ± 0.02 mm in length by 0.07 ± 0.003 mm in width in males. The esophagus in the females measured 0.38 ± 0.02 mm by 0.1 mm ± 0.007. The posterior muscular section measured 3.33 ± 0.23 mm in length and 0.23 ± 0.03 in width in the males, and 3.67 ± 0.17 mm and 0.22 ± 0.01 mm, respectively, in females. The cervical papillae were very small and located 0.31± 0.02 mm from the anterior end of the males, and 0.31 ± 0.03 mm in the females. The vulvar opening was rounded, and was located 1.7 ± 1.83 mm from the posterior end. The female specimens were ovoviviparous, and presented mature larval eggs with a smooth, thick shell, measuring 0.046 mm in length and 0.026 mm in width. The anus was located 0.23 ± 0.01 mm from the tip of the tail. The males had unequal and dissimilar spicules, with the largest measuring 3.47 ± 0.21 mm with a tapered tip and widened base, and the smallest measuring 0.76 ± 0.11 mm, including the width base and rounded tip. The gubernaculum was ring-shaped, and the posterior end was wrapped in a spiral with thickened caudal wings that lacked a copulation bursa (Figure 3b). The morphological and morphometric characteristics of the parasite structures were consistent with Sicarius uncinipenis, as described by Freitas and Lent (1947a)Freitas JFT, Lent H. “Spiruroidea” parasitos de “Rheiformes” (Nematoda). Mem Inst Oswaldo Cruz 1947a; 45(4): 743-760. http://dx.doi.org/10.1590/S0074-02761947000400002
http://dx.doi.org/10.1590/S0074-02761947...
.
Sicarius uncinipenis from the ventricle of Rhea americana americana (a) Bucal capsula (arrow) and cervical papillae (black arrow) of the anterior end of a female specimen. BAR = 100 µm. (b) Posterior end of a male wrapped in spiral and containing a smaller spicule (black arrow) and a major spicule (white arrow). BAR = 200 µm. The spicule ratio of 1:4 is characteristic of this species.
Sicarius uncinipenis is a parasite typically found in the ventricle of rheas, that can also be found in the initial portion of the duodenum (FREITAS; LENT, 1947aFreitas JFT, Lent H. “Spiruroidea” parasitos de “Rheiformes” (Nematoda). Mem Inst Oswaldo Cruz 1947a; 45(4): 743-760. http://dx.doi.org/10.1590/S0074-02761947000400002
http://dx.doi.org/10.1590/S0074-02761947...
). In both birds included on this study, the parasites were found in the ventricle, as well as a few specimens in the lumen of the duodenum. However, post-mortem migration of the parasites cannot be excluded. A study of the parasitic fauna in free-living rheas from Pantanal of Mato Grosso do Sul, and captive rheas from Jaboticabal, São Paulo state, Brazil, reported that S. uncinipenis was the most prevalent helminth in the ventricle. Further, infection is believed to originate from the introduction of naturally infected animals (ZETTERMANN et al., 2005Zettermann CD, Nascimento AA, Tebaldi JA, Szabó MJP. Observations on helminth infections of free-living and captive rheas (Rhea americana) in Brazil. Vet Parasitol 2005; 129(1-2): 169-172. PMid:15817218. http://dx.doi.org/10.1016/j.vetpar.2004.12.015
http://dx.doi.org/10.1016/j.vetpar.2004....
). In the present study, the infection likely occurred in captivity, and the stress during capture and transport may have contributed to development and progression in the birds with more severe parasitic infections. Studies pertaining to the intermediate host are limited, but Schimidt and Roberts (1977)Schimidt GD, Roberts LS. Foundation of Parasitology. Missouri: CV Mosby Company St Louis; 1977. p. 604., as cited by Zettermann et al. (2005)Zettermann CD, Nascimento AA, Tebaldi JA, Szabó MJP. Observations on helminth infections of free-living and captive rheas (Rhea americana) in Brazil. Vet Parasitol 2005; 129(1-2): 169-172. PMid:15817218. http://dx.doi.org/10.1016/j.vetpar.2004.12.015
http://dx.doi.org/10.1016/j.vetpar.2004....
have considered the association with an arthropod. Further, the gross and histopathology changes observed were in accord with those reported by Zettermann et al. (2005)Zettermann CD, Nascimento AA, Tebaldi JA, Szabó MJP. Observations on helminth infections of free-living and captive rheas (Rhea americana) in Brazil. Vet Parasitol 2005; 129(1-2): 169-172. PMid:15817218. http://dx.doi.org/10.1016/j.vetpar.2004.12.015
http://dx.doi.org/10.1016/j.vetpar.2004....
. The authors of the prior report noted reddish parasites inserted between the koilin layer and the mucosa of the ventricle. Hemorrhagic depressions in the mucosa, associated with necrosis and macrophages, could be related to the development of larval forms tracking to the submucosa and mucosa layers. However, the pathogenicity is poorly understood, and there are no reports regarding the clinical indications. No erythrocytes or iron pigment was observed inside the adult parasites of birds analyzed by Zettermann et al. (2005)Zettermann CD, Nascimento AA, Tebaldi JA, Szabó MJP. Observations on helminth infections of free-living and captive rheas (Rhea americana) in Brazil. Vet Parasitol 2005; 129(1-2): 169-172. PMid:15817218. http://dx.doi.org/10.1016/j.vetpar.2004.12.015
http://dx.doi.org/10.1016/j.vetpar.2004....
. In contrast, adult parasites found in the birds of the current study were red, and contain brown pigment within intestine suggesting iron from hematophagic activity. Possibly, reduced nutritional absorption and interference with the ventricle function in the host also contributed to the pathogenicity of this parasite.
For parasitological evaluation and measurement of the intestinal parasites, five male and five female specimens were also utilized. The male specimens measured 14.67 ± 0.63 mm in length and 0.48 ± 0.008 mm in width, and females measured 20.7 ± 1.7 mm and ± 0.59 0.025 mm, respectively. Both genders had a wide mouth capsule measuring 0.18 ± 0.009 mm in length and 0.22 ± 0.01 mm in width for males, and 0.25 ± 0.015 mm and 0.29 ± 0.019 mm for females. Additionally, the parasites possessed an external capsule corona radiata, consisting of six striated lobes, and several internal chitinized denticles (Figure 4a). The walls of the capsule of corona radiata were comprised of serrated ridges that varied in length. The inner corona radiata was sinuous. The parasite had a claviforme esophagus that was dilated anteriorly and posteriorly, and measured 1.42 ± 0.018 mm in length by 0.19 ± 0.016 mm in width in males, and 1.65 ± 0.056 mm by 0.24 ± 0.008 mm in females. The papillae were very small, and were located at 0.88 ± 0.12 mm from the cervical anterior end of the males, and 0.98 ± 0.063 mm in females. The nervous ring was situated 0.53 ± 0.023 mm from the anterior end in males, and 0.57 ± 0.018 mm in females. The vulvar opening in females was located in a cuticular salience 0.75 ± 0.044 mm from the posterior end of the body. The females possessed large eggs with thin shells, measuring 0.157 mm in length by 0.081 mm in width. The anal opening was located 0.44 ± 0.019 mm from the tapered tail tip. The males had small and non-striated bursa, which were closed ventrally. The ventral rays had a common core (Figure 4b), as well as lateral and dorsal rays. The dorsal ray split into two external branches that bifurcated apically. The spicules were thin, with a wider proximal end and a tapered distal end that measured 4.81 ± 0.27 mm (Figure 4c). The gubernaculum measured 0.038 ± 0.005 mm in length, and was slightly chitinized, and difficult to visualize. The morphological and morphometric characteristics of the structures of the parasites were consistent with Deletrocephalus cesarpintoi, as described by Freitas and Lent (1947b)Freitas JFT, Lent H. Revisão da subfamília “Deletrocephalinae” Railliet, 1916 (Nematoda, Strongyloidea). Rev Brasil Biol 1947b; 7(1): 73-100..
Deletrocephalus cesarpintoi from the large intestine (cecum) of Rhea americana americana (a) Buccal cavity of the anterior end of a female highlighting the chitinized denticles (arrows). BAR = 100 µm. (b) Copulatory bursa presenting the dorsal ray with three divisions (arrows) at the posterior end of a male. BAR = 100 µm. (c) Spicules (arrows) of the posterior end of a male specimen. BAR = 50 µm.
Deletrocephalus cesarpintoi has been described in the large intestine and rectum of Rhea americana americana and Rhea americana intermedia (FREITAS; LENT, 1947bFreitas JFT, Lent H. Revisão da subfamília “Deletrocephalinae” Railliet, 1916 (Nematoda, Strongyloidea). Rev Brasil Biol 1947b; 7(1): 73-100.; VICENTE et al., 1995; ZETTERMANN et al., 2005Zettermann CD, Nascimento AA, Tebaldi JA, Szabó MJP. Observations on helminth infections of free-living and captive rheas (Rhea americana) in Brazil. Vet Parasitol 2005; 129(1-2): 169-172. PMid:15817218. http://dx.doi.org/10.1016/j.vetpar.2004.12.015
http://dx.doi.org/10.1016/j.vetpar.2004....
), which was in accord with discoveries in the birds in the present study. D. dimidiatus has been described in the small and large intestine and rectum (VICENTE et al., 1995), but only in moderate numbers in the large intestine (ZETTERMANN et al., 2005Zettermann CD, Nascimento AA, Tebaldi JA, Szabó MJP. Observations on helminth infections of free-living and captive rheas (Rhea americana) in Brazil. Vet Parasitol 2005; 129(1-2): 169-172. PMid:15817218. http://dx.doi.org/10.1016/j.vetpar.2004.12.015
http://dx.doi.org/10.1016/j.vetpar.2004....
). There are no reports regarding the pathogenesis and clinical signs of D. cesarpintoi infection in rheas. All studies are related to D. dimidiatus, which was observed in association with hemorrhage in the mucosa, and dark fluid feces (MONTEIRO et al., 2002Monteiro SG, Flores ML, Segabinazi SD, Lagaggio VRA. Ocorrência de Deletrocephalus dimidiatus (Diesing, 1851) Nematoda em ema (Rhea amaricana) criada em cativeiro no RS. Rev FZVA 2002; 9(1): 100-103.). Similar changes in the mucosa were observed in the birds in the current study, but only for D. cesarpintoi. It is believed that severe D. dimidiatus infections can cause weakness and diarrhea in young birds (EWING et al., 1995Ewing ML, Yonzon ME, Page RK, Brown TP, Davidson WR. Deletrocephalus dimidiatus infestation in an adult rhea (Pterocnemia pennata). Avian Dis 1995; 39(2): 441-443. PMid:7677670. http://dx.doi.org/10.2307/1591892
http://dx.doi.org/10.2307/1591892...
; TAYLOR et al., 2010Taylor MA, Coop RL, Wall RL. Parasitologia Veterinária. 3rd ed. Rio de Janeiro: uanabara Koogan; 2010.). In addition, Craig and Diamond (1996)Craig TM, Diamond PL. Parasites of ratites. In: Tully Jr TN, Shane SM. Ratite Management Medicine and Surgery. Malabar: Krieger Publishing Company; 1996. p. 115-126. considered the species relevant, due to hematophagous habits that can lead to anemia. The pathological findings, combined with moderate to high parasitic infections in the ventricle and intestine, could be associated with the death of the rheas in this study. Other changes that could explain the death of the birds were not found.
The identification of the third-stage larvae by coproculture provided evidence for a direct life cycle, which indicated that the bird could become infected by ingesting the larvae during foraging (TAYLOR et al., 2000Taylor MA, Hunt KR, Smith G, Otter A. Deletrocephalus dimidiatus in greater rheas (Rhea americana) in the UK. Vet Rec 2000; 146(1): 19-20. PMid:10661457. http://dx.doi.org/10.1136/vr.146.1.19
http://dx.doi.org/10.1136/vr.146.1.19...
). However, larval migration of the Deletrocephalus to adulthood is unknown (EWING et al., 1995Ewing ML, Yonzon ME, Page RK, Brown TP, Davidson WR. Deletrocephalus dimidiatus infestation in an adult rhea (Pterocnemia pennata). Avian Dis 1995; 39(2): 441-443. PMid:7677670. http://dx.doi.org/10.2307/1591892
http://dx.doi.org/10.2307/1591892...
).
Despite the lack of experimental studies regarding the treatment of this nematode in rheas, benzimidazoles and ivermectin have been used to control nematodes in ostriches (TAYLOR et al., 2010Taylor MA, Coop RL, Wall RL. Parasitologia Veterinária. 3rd ed. Rio de Janeiro: uanabara Koogan; 2010.). Artificial incubation, separation of chicks from adults, and allocation of chicks in a decontaminated area (EWING et al., 1995Ewing ML, Yonzon ME, Page RK, Brown TP, Davidson WR. Deletrocephalus dimidiatus infestation in an adult rhea (Pterocnemia pennata). Avian Dis 1995; 39(2): 441-443. PMid:7677670. http://dx.doi.org/10.2307/1591892
http://dx.doi.org/10.2307/1591892...
) are important for decreasing or preventing the transmission of D. dimidiatus. Furthermore, anthelmintic drugs tests and strategies for parasite control should be adopted.
The histopathological examination is very useful for identification of the genera of the parasites and association with the lesions in the infected tissues. However, only the parasitological analysis allowed the identification of the species. Finally, further investigations of these infections in free-living and captive rheas are required, to obtain more accurate data of the incidence and pathogenicity of these parasites. Moreover, it is important to adopt periodic examinations of feces, and to use anthelmintic strategies to control these parasites in captive rheas.
Acknowledgements
Pró-Reitoria de Pesquisa da Universidade Federal de Minas Gerais and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
References
- Almeida MA. Struthioniformes (Ema, Avestruz). In: Cubas ZS, Silva JCR, Catão-Dias JL. Tratado de Animais Selvagens. São Paulo: Roca; 2007. p. 136-157. PMid:17963762
- BirdLife International. Rhea americana. 2012 [cited 2014 Jan 8]. In: Union for Conservation of Nature - IUCN. IUCN Red List of Threatened Species. Version 2013.2. 2013. Available from: http://www.iucnredlist.org.
» http://www.iucnredlist.org - Craig TM, Diamond PL. Parasites of ratites. In: Tully Jr TN, Shane SM. Ratite Management Medicine and Surgery. Malabar: Krieger Publishing Company; 1996. p. 115-126.
- Ewing ML, Yonzon ME, Page RK, Brown TP, Davidson WR. Deletrocephalus dimidiatus infestation in an adult rhea (Pterocnemia pennata). Avian Dis 1995; 39(2): 441-443. PMid:7677670. http://dx.doi.org/10.2307/1591892
» http://dx.doi.org/10.2307/1591892 - Freitas JFT, Lent H. “Spiruroidea” parasitos de “Rheiformes” (Nematoda). Mem Inst Oswaldo Cruz 1947a; 45(4): 743-760. http://dx.doi.org/10.1590/S0074-02761947000400002
» http://dx.doi.org/10.1590/S0074-02761947000400002 - Freitas JFT, Lent H. Revisão da subfamília “Deletrocephalinae” Railliet, 1916 (Nematoda, Strongyloidea). Rev Brasil Biol 1947b; 7(1): 73-100.
- Gardiner CH, Poynton SL. An Atlas of Metazoan Parasites in Animal Tissues. Washington: Armed Forces Institute of Pathology; 1999. 64 p.
- Luna LG. Manual of histologic staining methods of the Armed Forces Institute of Pathology. New York: McGraw-Hill; 1968. 258 p.
- Monteiro SG, Flores ML, Segabinazi SD, Lagaggio VRA. Ocorrência de Deletrocephalus dimidiatus (Diesing, 1851) Nematoda em ema (Rhea amaricana) criada em cativeiro no RS. Rev FZVA 2002; 9(1): 100-103.
- Nemejc K, Lukesova D. Parasite fauna of ostriches, emus and rheas. Agricul Trop Subtrop 2012; 45(1): 45-50.
- Schimidt GD, Roberts LS. Foundation of Parasitology. Missouri: CV Mosby Company St Louis; 1977. p. 604.
- Shimada T, Nakamura F. Dome formation of keratin-containing agranular cells from rat anterior pituitary gland in vitro. In Vitro Cell Dev Biol. 1988; 24(12): 1193-200. http://dx.doi.org/10.1007/BF02624189
» http://dx.doi.org/10.1007/BF02624189 - Sick H. Ornitologia Brasileira. Rio de Janeiro: Nova Fronteira; 1997. PMCid:PMC191379
- Taylor MA, Coop RL, Wall RL. Parasitologia Veterinária. 3rd ed. Rio de Janeiro: uanabara Koogan; 2010.
- Taylor MA, Hunt KR, Smith G, Otter A. Deletrocephalus dimidiatus in greater rheas (Rhea americana) in the UK. Vet Rec 2000; 146(1): 19-20. PMid:10661457. http://dx.doi.org/10.1136/vr.146.1.19
» http://dx.doi.org/10.1136/vr.146.1.19 - Vicente JJ, Rodrigues HO, Gomes DC, Pinto MR. Nematóides do Brasil. Parte IV: Nematóides de aves. Rev Bras Zool 1995; 12(S1): 1-273.
- Zettermann CD, Nascimento AA, Tebaldi JA, Szabó MJP. Observations on helminth infections of free-living and captive rheas (Rhea americana) in Brazil. Vet Parasitol 2005; 129(1-2): 169-172. PMid:15817218. http://dx.doi.org/10.1016/j.vetpar.2004.12.015
» http://dx.doi.org/10.1016/j.vetpar.2004.12.015
Data availability
Data citations
BirdLife International. Rhea americana. 2012 [cited 2014 Jan 8]. In: Union for Conservation of Nature - IUCN. IUCN Red List of Threatened Species. Version 2013.2. 2013. Available from: http://www.iucnredlist.org.
Publication Dates
-
Publication in this collection
Jul-Sep 2014
History
-
Received
5 May 2014 -
Accepted
25 June 2014