Abstract
Bivalve culture is of considerable economic and social interest in northeastern (NE) Brazil. The polyculture is an alternative approach to traditional monoculture for reducing the environmental impact of shrimp farming and improving oyster culture. Perkinsus marinus andPerkinsus olseni were found infecting oysters in NE Brazil and can threaten oyster production. This study evaluatedPerkinsus spp. occurrence in Crassostrea gasar during all production stages. Oyster spats were produced in a hatchery and grown in shrimp ponds in Rio Grande do Norte state.Perkinsus spp. were surveyed by Ray’s fluid thioglycollate medium and confirmed by polymerase chain reaction. Prevalence and intensity of infection were determined in oysters until they reached 7 cm. Results showed that the broodstock was already infected by Perkinsus (60%), but the derived spats were Perkinsus-free. Oyster spats acquired Perkinsus infection when transferred to ponds. The prevalence gradually increased in the seven months following placement in ponds (73%), and then decreased to 17% by the tenth month. The infections were initially mild, but intensity increased at the final growth stage. In conclusion, it is possible to produce Perkinsus-free C. gasar oyster spats from infected broodstock, and their culture in shrimp ponds is feasible.
Keywords:
Crassostrea gasar; hatchery; oyster; polyculture; Perkinsus sp.; sanitary control
Resumo
O cultivo de bivalves é de grande interesse econômico e social no Nordeste (NE) do Brasil. O policultivo é uma alternativa ao monocultivo tradicional de camarões para reduzir o impacto ambiental e melhorar a produção de ostras.Perkinsus marinus e Perkinsus olseni foram identificados infectando ostras no Nordeste do Brasil e representam uma ameaça a produção de ostras. Este estudo avaliou a ocorrência dePerkinsus spp. em Crassostrea gasardurante todas as fases de produção. Sementes de ostras foram produzidas em laboratório e cultivadas em viveiros de camarão no Rio Grande do Norte.Perkinsus spp. foram diagnosticados com o uso do meio de tioglicolato fluido de Ray e confirmado por reação em cadeia da polimerase. A prevalência e intensidade de infecção foram determinadas em ostras até atingirem 7 cm. Os resultados mostraram que os reprodutores encontravam-se infectados porPerkinsus (60%), mas as sementes produzidas estavam livres de Perkinsus. As sementes adquiriram a infecção porPerkinsus quando transferidas para os viveiros. A prevalência aumentou gradualmente nos sete meses após a colocação nos viveiros (73%) e, em seguida, diminuiu para 17% até o décimo mês. As infecções foram inicialmente leves, mas aumentaram até a fase final do crescimento. Em conclusão, é possível produzir sementes de ostras C. gasarlivres de Perkinsus a partir de reprodutores infectados e seu cultivo em viveiros de camarão é viável.
Palavras-chave:
Crassostrea gasar; unidade de reprodução; ostras; policultivo; Perkinsus sp.; controle sanitário
Introduction
The culture of mollusks is of considerable economic and social interest in a number of countries worldwide, including Brazil. The state of Santa Catarina is the largest national producer of mollusks and the second largest in Latin America, with an annual production of 21,553 tons in 2014. Nevertheless, just 3,600 tons correspond to the Japanese oyster Crassostrea gigas (EPAGRI, 2014Empresa de Pesquisa Agropecuária e Extensão Rural de Santa Catarina – EPAGRI. Síntese informativa da maricultura [online]. Florianópolis: EPAGRI; 2014 [cited 2015 June 28]. Available from: http://www.epagri.sc.gov.br/wp-content/uploads/2013/08/Sintese_informativa_da_maricultura_2014.pdf
http://www.epagri.sc.gov.br/wp-content/u...
), highlighting the potential for increasing the national production of oysters. Crassostrea gigas was introduced into Brazil in the 1970s and quickly adapted to the climate conditions of the Santa Catarina state (south Brazil). The Laboratory of Marine Mollusks (LMM) of the Federal University of Santa Catarina (UFSC) has been a pioneer in improving the technical procedures for production of C. gigas spat in Brazil and in transferring technologies for its growth to the production sector. Currently, much of the production of C. gigas oysters in the state of Santa Catarina depends on the spats produced by LMM.
The northeastern (NE) coast of Brazil is known for the presence of large estuaries inhabited by numerous species of edible bivalves, including two species of oysters,Crassostrea gasar and Crassostrea rhizophorae. Due to high primary productivity in the seawater and favorable climatic and geographic conditions in these environments, the NE region shows great potential for increasing the development of oyster culture (LAVANDER et al., 2013Lavander HD, Cardoso LO Jr, Silva LOB, Gàlvez AO. Estudo de viabilidade econômica para ostreicultura familiar em Pernambuco, Brasil. Custos e @gronegócio Online 2013; 9(2): 173-187.).
Unlike Santa Catarina, oyster production in the NE region faces many obstacles, including various economic and, particularly, technical (lack of specialized technical assistance) difficulties. The production is based primarily on oyster collection from natural estuarine populations, which does not allow for oysters with assured sanitary or productivity qualities. Therefore, the inability to select oysters with a good growth rate and appearance is one of the disadvantages of the traditional culture methods that prevail in the NE region today.
In Brazil, there are currently no hatchery facilities for native oysters because production techniques have not been fully optimized for these species. However, some efforts have been made by research institutions to understand certain biological aspects of C. gasar that may be useful in the production process (LOPES et al., 2013Lopes GR, Gomes CHAM, Tureck CR, Melo CMR. Growth of cultured in marine and estuary environments in brazilian waters. Crassostrea gasarPesq Agropec Brasil 2013; 48(8): 975-982. http://dx.doi.org/10.1590/S0100-204X2013000800024.
http://dx.doi.org/10.1590/S0100-204X2013...
; RAMOS et al., 2014Ramos CO, Gomes CHAM, Magalhães ARM, Santos AI, Melo CMR. Maturation of the mangrove oyster at diferent temperatures in the laboratory. Crassostrea gasarJ Shellfish Res 2014; 33(1): 187-194. http://dx.doi.org/10.2983/035.033.0118.
http://dx.doi.org/10.2983/035.033.0118...
; SILVEIRA et al., 2011Silveira RC, Silva FC, Gomes CHM, Ferreira JF, Melo CMR. Larval settlement and spat recovery rates of the oyster (Lamarck, 1819) using different systems to induce metamorphosis. Crassostrea brasilianaBraz J Biol 2011; 71(2): 557-562. http://dx.doi.org/10.1590/S1519-69842011000300029. PMid:21755177.
http://dx.doi.org/10.1590/S1519-69842011...
; GOMES et al., 2014Gomes CHAM, Silva FC, Lopes GR, Melo CMR. The reproductive cycle of the oyster Crassostrea gasar.Braz J Biol 2014; 74(4): 967-976. http://dx.doi.org/10.1590/1519-6984.04912. PMid:25627610.
http://dx.doi.org/10.1590/1519-6984.0491...
).
Shrimp culture is the dominant aquaculture activity in the NE region, however, this practice is known to negatively impact water quality of the surrounding estuaries (PÁEZ-OSUNA, 2001Páez-Osuna F. The environmental impact of shrimp aquaculture: causes, effects, and mitigating alternatives. Environ Manage 2001; 28(1): 131-140. http://dx.doi.org/10.1007/s002670010212. PMid:11436996.
http://dx.doi.org/10.1007/s002670010212...
). Thus, in order to minimize the environmental impact of shrimp farm effluents and promote sustainable aquaculture as well as improve oyster culture in the NE region, implementation of polycultures is a good alternative (RAMOS et al., 2008Ramos R, Vinatea L, Costa R. Tratamiento de efluentes del cultivo de Litopenaeus vannamei por sedimentación y filtración por la ostra Crassostrea rhizophorae.Lat Am J Aquat Res 2008; 36(2): 235-244. http://dx.doi.org/10.3856/vol36-issue2-fulltext-6.
http://dx.doi.org/10.3856/vol36-issue2-f...
; REYES, 2013Reyes GG. Mixed culture (shrimps, oysters and macroalgae) to improve the productivity, and reduce the environmental impact to coastal ecosystem caused by shrimp hatcheries in Mexico. Int J Fish Aquacult 2013; 5(10): 262-269.; ROSA, 2014Rosa LC. Crescimento e sobrevivência da ostra (Lamarck, 1819) mantida em um viveiro de cultivo de camarão. Crassostrea brasilianaArq. Cienc Mar 2014; 47(1): 64-68.; MARTINEZ-PORCHAS et al., 2010Martínez-Porchas M, Martínez-Córdova LR, Pórchas-Cornejo MA, López-Elías JA. Shrimp polyculture: a potentially profitable, sustainable, but uncommon aquacultural pratice. Rev Aquaculture 2010; 2(2): 73-85. http://dx.doi.org/10.1111/j.1753-5131.2010.01023.x.
http://dx.doi.org/10.1111/j.1753-5131.20...
). It is known that shrimp pond water quality can be improved by the use of filter feeding bivalves such as oysters, which can help decrease the concentration of total suspended solids, organic and inorganic matter, nitrogen, phosphorous, chlorophyll a and bacteria (JONES et al., 2001Jones AB, Dennison WC, Preston NP. Integrated treatment of shrimp effluent by sedimentation, oyster filtration and macroalgal absorption: a laboratory scale study. Aquaculture 2001; 193(1-2): 155-178. http://dx.doi.org/10.1016/S0044-8486(00)00486-5.
http://dx.doi.org/10.1016/S0044-8486(00)...
; JONES & PRESTON, 1999Jones AB, Preston NP. Sydney rock oyster, (Iredale & Roughley), filtration of shrimp farm effluent: the effects on water quality. Saccostrea commercialisAquacult Res 1999; 30(1): 51-57. http://dx.doi.org/10.1046/j.1365-2109.1999.00299.x.
http://dx.doi.org/10.1046/j.1365-2109.19...
; MARTINEZ-CORDOVA & MARTINEZ-PORCHAS, 2006Martinez-Cordova LR, Martinez-Porchas M. Polyculture of pacific white shrimp, , giant oyster, and black clam, Chione fluctifraga in ponds in Sonora, Mexico. Litopenaeus vannameiCrassostrea gigasAquaculture 2006; 258(1-4): 321-326. http://dx.doi.org/10.1016/j.aquaculture.2006.03.026.
http://dx.doi.org/10.1016/j.aquaculture....
; RAMOS et al., 2008Ramos R, Vinatea L, Costa R. Tratamiento de efluentes del cultivo de Litopenaeus vannamei por sedimentación y filtración por la ostra Crassostrea rhizophorae.Lat Am J Aquat Res 2008; 36(2): 235-244. http://dx.doi.org/10.3856/vol36-issue2-fulltext-6.
http://dx.doi.org/10.3856/vol36-issue2-f...
). The improvement of pond water quality contributes to improved growth performance and health of the shrimp (XIE et al., 2011Xie B, Jiang W, Yang H. Growth perfomance and nutrient quality of Chinese shrimp Penaeus chinensis in organic polyculture with razor clam or hard clam Sinovacula constrictaMeretix meretrix.Bulg J Agric Sci 2011; 17(6): 851-858.; REYES, 2013Reyes GG. Mixed culture (shrimps, oysters and macroalgae) to improve the productivity, and reduce the environmental impact to coastal ecosystem caused by shrimp hatcheries in Mexico. Int J Fish Aquacult 2013; 5(10): 262-269.). Xie et al. (2011)Xie B, Jiang W, Yang H. Growth perfomance and nutrient quality of Chinese shrimp Penaeus chinensis in organic polyculture with razor clam or hard clam Sinovacula constrictaMeretix meretrix.Bulg J Agric Sci 2011; 17(6): 851-858. recorded an increase in body length and fresh and dry weight of shrimps cultured in ponds with clams. Oysters or other bivalves may benefit from the high concentration of nutrient particles available in shrimp ponds leading to improved growth (MARTINEZ-CORDOVA & MARTINEZ-PORCHAS, 2006Martinez-Cordova LR, Martinez-Porchas M. Polyculture of pacific white shrimp, , giant oyster, and black clam, Chione fluctifraga in ponds in Sonora, Mexico. Litopenaeus vannameiCrassostrea gigasAquaculture 2006; 258(1-4): 321-326. http://dx.doi.org/10.1016/j.aquaculture.2006.03.026.
http://dx.doi.org/10.1016/j.aquaculture....
).
The production of oysters is also threatened by the emergence of epizootic diseases, mainly caused by bacteria and protozoa (LAUCKNER, 1983Lauckner G. Diseases of mollusca: bivalvia. In: Kinne O. Diseases of marine animals. Hamburg: Biologische Anstalt Helgoland; 1983. p. 477-961.; OIE, 2006World Organization for Animal Health – OIE. Manual of diagnostic tests for aquatic animals. 5th ed. Paris: OIE; 2006.). Protozoan species of the genus Perkinsus are a well-known cause of mortality in bivalve populations and consequently have a negative impact on the economy (VILLALBA et al., 2004Villalba A, Reece KS, Ordás MC, Casas SM, Figueras A. Perkinsosis in molluscs: a review. Aquat Living Resour 2004; 17(4): 411-432. http://dx.doi.org/10.1051/alr:2004050.
http://dx.doi.org/10.1051/alr:2004050...
, 2011Villalba A, Gestal C, Casas SM, Figueras A. Perkinsosis en moluscos. In: Figueras A, Novoa B. Enfermedades de moluscos bilvalvos de interés en acuicultura. Madrid: Fundación Observatorio Español de Acuicultura; 2011. p. 181-242.; CHOI & PARK, 2010Choi KS, Park KI. Review on the protozoan parasite (Lester and Davis 1981) infection in Asian waters. In: Perkinsus olseniIshimatsu A, Lie HJ. Coastal environmental and ecosystem issues of the east China sea. Nagazaki: TERRAPUB, Nagasaki University; 2010. p. 269-281.). Two species, P. marinus and P. olseni, are the most pathogenic and, thus, require mandatory notification to the World Organization for Animal Health (OIE, 2015World Organization for Animal Health – OIE. OIE Listed diseases, infections and infestations in force in 2015. Paris: OIE; 2015 [cited 2015 June 28]. Available from: http://www.oie.int/animal-health-in-the-world/oie-listed-diseases-2015
http://www.oie.int/animal-health-in-the-...
). The disease can affect all ages of oysters, from spat to adult (PAYNTER et al., 2010Paynter KT, Politano V, Lane HA, Allen SM, Meritt D. Growth rates and prevalence of prevalence in restored oyster populations in Maryland. Perkinsus marinusJ Shellfish Res 2010; 29(2): 309-317. http://dx.doi.org/10.2983/035.029.0205.
http://dx.doi.org/10.2983/035.029.0205...
).
In Brazil in 2013, one parasite from this genus (P. beihaiensis) was detected for the first time infecting the oyster C. rhizophoraefrom Ceará state, with low prevalence and intensity (SABRY et al., 2009Sabry RC, Rosa RD, Magalhães ARM, Barracco MA, Gesteira TCV, Silva PM. First report of Perkinsus sp. infecting mangrove oysters from the Brazilian coast. Crassostrea rhizophoraeDis Aquat Organ 2009; 88(1): 13-23. http://dx.doi.org/10.3354/dao02136. PMid:20183961.
http://dx.doi.org/10.3354/dao02136...
, 2013Sabry RC, Gesteira TCV, Magalhães ARM, Barracco MA, Guertler C, Ferreira LP, et al. Parasitological survey of mangrove oyster, , in the Pacoti River Estuary, Ceará State, Brazil. Crassostrea rhizophoraeJ Invertebr Pathol 2013; 112(1): 24-32. http://dx.doi.org/10.1016/j.jip.2012.10.004. PMid:23147104.
http://dx.doi.org/10.1016/j.jip.2012.10....
). This species was recently identified infecting the clamAnomalocardia brasiliana, as well (FERREIRA et al., 2015Ferreira LP, Sabry RC, Silva PM, Gesteira TCV, Romão LS, Paz MP, et al. First report of Perkinsus beihaiensis in wild clams (Bivalvia: Veneridae) in Brazil. Anomalocardia brasilianaExp Parasitol 2015; 150: 67-70. http://dx.doi.org/10.1016/j.exppara.2014.07.012. PMid:25088443.
http://dx.doi.org/10.1016/j.exppara.2014...
). In contrast, P. marinus was detected in two oyster species, C. rhizophorae and C. gasar from Paraíba state with high prevalence (71% to 100%) and moderate mean intensities (SILVA et al., 2013Silva PM, Vianna RT, Guertler C, Ferreira LP, Santana LN, Fernandéz-Boo S, et al. First report of the protozoan parasite in South America, infecting mangrove oysters from the Paraíba River. Perkinsus marinusCrassostrea rhizophoraeJ Invertebr Pathol 2013; 113(1): 96-103. http://dx.doi.org/10.1016/j.jip.2013.02.002. PMid:23439264.
http://dx.doi.org/10.1016/j.jip.2013.02....
; QUEIROGA et al., 2015Queiroga FR, Vianna RT, Vieira CB, Farias ND, Silva PM. Parasites infecting the cultured oyster (Adanson, 1757) in Northeast Brazil. Crassostrea gasarParasitology 2015; 142(6): 756-766. http://dx.doi.org/10.1017/S0031182014001863. PMid:25553815.
http://dx.doi.org/10.1017/S0031182014001...
). Perkinsus marinus was also detected inC. gasar from Sergipe state, although the prevalences in oysters varied seasonally (7% in July to 80% in December) and was higher in cultured oysters (56%) than wild oysters (35%) (SILVA et al., 2014Silva PM, Scardua MP, Vianna RT, Mendonça RC, Vieira CB, Dungan CF, et al. Two Perkinsus spp. infect oysters from cultured and wild populations of the Rio São Francisco estuary, Sergipe, northeastern Brazil. Crassostrea gasarJ Invertebr Pathol 2014; 119: 62-71. http://dx.doi.org/10.1016/j.jip.2014.04.005. PMid:24780219.
http://dx.doi.org/10.1016/j.jip.2014.04....
). Perkinsus olseni was also identified in the oyster C. gasar from Sergipe. However, it seems that this protozoan species occurs with a very low prevalence because few nucleotide sequences are obtained from host tissues (SILVA et al., 2014Silva PM, Scardua MP, Vianna RT, Mendonça RC, Vieira CB, Dungan CF, et al. Two Perkinsus spp. infect oysters from cultured and wild populations of the Rio São Francisco estuary, Sergipe, northeastern Brazil. Crassostrea gasarJ Invertebr Pathol 2014; 119: 62-71. http://dx.doi.org/10.1016/j.jip.2014.04.005. PMid:24780219.
http://dx.doi.org/10.1016/j.jip.2014.04....
). In Bahia state, an unidentified Perkinsus sp. was reported in C. rhizophorae with variable prevalence (21% to 92%) and moderate intensity. Despite the high prevalence and intensity ofPerkinsus spp. infection in Brazilian oysters, a massive mortality event has not yet been reported.
In the current study, Perkinsus sp. infection was detected for the first time in C. gasar oysters from the state of Rio Grande do Norte, and the prevalence and intensity of this protozoan was evaluated in oysters produced in the hatchery and cultured in shrimp ponds.
Materials and Methods
Collection of oysters
The PRIMAR farm located in Tibau do Sul, state of Rio Grande do Norte, RN (6° 13' 31.06” S; 35° 8' 19.92” W) produces organic shrimps (3-4 animals per m2, without supplementary feeding) and oysters in shrimp ponds. The farm has recently implemented (2014) a hatchery facility for oyster spats production. Crassostrea gasar oyster spats were produced in a hatchery and grown in shrimp ponds at the PRIMAR farm. The water temperature and salinity of the ponds were monitored daily throughout the oyster growth period (July 2014 to May 2015).
Table 1 lists the dates, number of oysters sampled at each production stage and type of analysis performed.
Oyster samplings performed during the production stages at the PRIMAR farm and analyses performed. RFTM: Ray’s fluid thioglycolate medium analysis; PCR-ITS: used for the identification of thePerkinsus genus; PCR-RFLP: used for the identification of the oyster species. Spats: ≤ 2 cm; Juveniles: > 2 < 5 cm; Adults ≥ 5 cm.
The first sampling of oysters (n = 10) was conducted in February 2013 for a preliminary evaluation of the presence of Perkinsus spp. in adult oysters cultured for more than three years at the PRIMAR farm to use as breeders.
On April 12, 2014, 160 adult oysters (breeders from PRIMAR) were divided into two polyethylene tanks (1000 L; 80 individuals/tank) to undergo mass spawning. Spawning occurred on April 13 and resulted in 180 million D larvae. At this time, 12 breeder oysters were sampled to detect Perkinsusspp.
The D larvae were placed in 150 L cylindrical-conical tanks with a continuous flow-through system with treated seawater (25 psu), i.e. chlorinated with sodium hypochlorite solution at 20 mg per L of free-chlorine, filtrated up to 1 µm, and sterilized by ultraviolet (UV)-C lamps (95W. 50/60 Hz, wavelength of 254 nm) arranged in series, resulting in a dose around 30,000 µW/cm2. Settlement occurred in shell powder on April 27. The spats were subsequently transferred to tanks in a down-welling system, inside the hatchery. On May 2014, after 45 days of spawning, spats were sampled before being introduced into the shrimp culture ponds. Nine pools of spats were prepared, each weighing 400 mg.
In May 2014, spats were transferred to box-type tanks of 1-mm-mesh, hanging from cords supported by stakes stuck into the bottom of the pond. In July 2014, spats were sampled (n = 50). After the July sampling, oysters were separated by size. This is a common practice among oyster farmers, in order to separate oysters with different growth rates. The larger oysters were placed in plastic bags with a 9-mm mesh, and the smaller oysters were placed in plastic bags with a 4-mm mesh. Because of their rapid growth, the larger oysters were separated by the producer for use as breeders in the future.
In October 2014 (n = 50) and February 2015 (n = 30) juvenile oysters were sampled. In March (n = 10) and May 2015 (n = 100) adult oysters were sampled. Sampling performed in March corresponded to the larger oysters separated in July 2014.
Diagnosis of Perkinsus spp. by Ray’s fluid thioglycollate medium
Oysters (n = 262; Table 1) were opened by cutting the adductor muscle to remove the demibranches, which were placed in tubes containing Ray’s fluid thioglycollate medium (RFTM) supplemented with antibiotics, penicillin/streptomycin (final concentration 100 U/mL and 0.1 mg/mL, respectively), and nystatin (100 U/mL). The tubes were placed in the dark at room temperature (25 °C) for seven days. After this period, demibranches were chopped into small pieces, stained with Lugol’s solution (4%) and examined by light microscopy. The samples were analyzed to determine the presence and the number of Perkinsus spp. hypnospores. The animals were classified according to Ray’s scale (1954) adapted by Silva et al. (2013)Silva PM, Vianna RT, Guertler C, Ferreira LP, Santana LN, Fernandéz-Boo S, et al. First report of the protozoan parasite in South America, infecting mangrove oysters from the Paraíba River. Perkinsus marinusCrassostrea rhizophoraeJ Invertebr Pathol 2013; 113(1): 96-103. http://dx.doi.org/10.1016/j.jip.2013.02.002. PMid:23439264.
http://dx.doi.org/10.1016/j.jip.2013.02....
as described below:
-
Null infection (0): No hypnospores observed on the whole slide (100x);
-
Very light infection (1): Up to 10 hypnospores observed in the whole slide (100x);
-
Light infection (2): 11-100 hypnospores observed in the whole slide (100x);
-
Moderate infection (3): Up to 40 hypnospores observed in a total of 10 random fields (400x), scattered throughout the slide;
-
Heavy infection (4): More than 40 hypnospores observed in a total of 10 random fields (400x), scattered throughout the slide.
The mean intensity of infection was calculated as the mean intensity considering only the infected oysters (1-4). The prevalence of infection was calculated as the percentage of infected oysters in each sample.
DNA extraction
A demibranch fragment or the entire oyster was sampled and immediately preserved in 95% ethanol for molecular analyses. For adult and juvenile oysters, a small fragment of the gill was removed from each animal (30-40 mg; n = 40) and individually analyzed. For spats from the hatchery, due to their small size, groups of whole spats were sampled by weight (400 mg; n = 9). For spats from the nursery, gill fragments from 10 oysters were grouped together (30-40 mg; n = 5); in this case, the oysters were not the same as those used for the diagnosis ofPerkinsus spp. by RFTM.
Deoxyribonucleic acid (DNA) was extracted from each sample using the Wizard® Genomic DNA Purification Kit (Promega) following the manufacturer’s instructions and used for Perkinsus spp. confirmation and oyster species identification.
PCR analysis to confirm the genus Perkinsus
To confirm the presence of Perkinsus spp. (n = 54; Table 1), polymerase chain reaction (PCR) assay was prepared using a pair of Perkinsus genus-specific primers, PerkITS85 / PerkITS750 (CASAS et al., 2002Casas SM, Villalba A, Reece KS. Study of perkinsosis in the carpet shell clam Tapes decussatus in Galicia (NW Spain). I. Identification of the aetiological agent and in vitro modulation of zoosporulation by temperature and salinity. Dis Aquat Organ 2002; 50(1): 51-65. http://dx.doi.org/10.3354/dao050051. PMid:12152905.
http://dx.doi.org/10.3354/dao050051...
). These primers specifically hybridize to the conserved regions of the ribosomal ribonucleic acid (RNA) gene, including internal transcribed spacer 1 (ITS), the 5.8S gene and ITS2 (ITS rDNA). The reactions were performed in a total volume of 25 µL containing 1 µL of genomic DNA (50 - 200 ng), 1x PCR buffer, 1.5 mM MgCl2, 0.2 mM nucleotides, 0.4 µM primers and 0.04 U/µL Taq DNA polymerase. The reaction conditions for amplification included DNA denaturation at 94 °C for 10 min, followed by 40 cycles of 94 °C for 1 min, 58 °C for 1 min and 72 °C for 1 min, with a final 10 min elongation step at 72 °C. The expected product size was 703 bp. Deoxyribonucleic acid samples isolated from oysters infected with P. marinus (SILVA et al., 2013Silva PM, Vianna RT, Guertler C, Ferreira LP, Santana LN, Fernandéz-Boo S, et al. First report of the protozoan parasite in South America, infecting mangrove oysters from the Paraíba River. Perkinsus marinusCrassostrea rhizophoraeJ Invertebr Pathol 2013; 113(1): 96-103. http://dx.doi.org/10.1016/j.jip.2013.02.002. PMid:23439264.
http://dx.doi.org/10.1016/j.jip.2013.02....
) and P. beihaiensis(SABRY et al., 2009Sabry RC, Rosa RD, Magalhães ARM, Barracco MA, Gesteira TCV, Silva PM. First report of Perkinsus sp. infecting mangrove oysters from the Brazilian coast. Crassostrea rhizophoraeDis Aquat Organ 2009; 88(1): 13-23. http://dx.doi.org/10.3354/dao02136. PMid:20183961.
http://dx.doi.org/10.3354/dao02136...
) were used as controls. The PCR amplification products were stained with Blue Green Loading Dye I (LGC Biotecnologia) and resolved via electrophoresis in 1% agarose gels.
Identification of oyster species by PCR-RFLP
Oysters show high levels of morphologic phenotypic plasticity, which makes differentiation among species extremely difficult (AMARAL & SIMONE, 2014Amaral VS, Simone LRL. Revision of genus (Bivalvia: Ostreidae) of Brazil. CrassostreaJ Mar Biol Assoc UK 2014; 94(4): 811-836. http://dx.doi.org/10.1017/S0025315414000058.
http://dx.doi.org/10.1017/S0025315414000...
). In this study, to identify theCrassostrea species (n = 25; Table 1), the mitochondrial gene encoding the small ribosomal subunit (16S) was amplified using the primers 16SAR/16SBR (KESSING et al., 1989Kessing B, Croom H, Martin A, McIntosh C, Owen M, Palumbi S. A simple guide to PCR. Honolulu: Department of Zoology, University of Hawaii; 1989.). The PCR assays were prepared in a total volume of 25 µL containing 1 µL of genomic DNA (50 - 200 ng), 1x PCR buffer, 1.5 mM MgCl2, 0.2 mM nucleotides, 0.4 µM primers and 0.04 U/µL Taq DNA polymerase. The conditions for the amplification reaction included DNA denaturation at 94 °C for 10 min, followed by 30 cycles of 94 °C for 1 min, 50 °C for 1 min and 72 °C for 1 min, with a final 10 min elongation step at 72 °C. The expected product size was 560 bp. Samples of DNA fromC. gasar (SILVA et al., 2014Silva PM, Scardua MP, Vianna RT, Mendonça RC, Vieira CB, Dungan CF, et al. Two Perkinsus spp. infect oysters from cultured and wild populations of the Rio São Francisco estuary, Sergipe, northeastern Brazil. Crassostrea gasarJ Invertebr Pathol 2014; 119: 62-71. http://dx.doi.org/10.1016/j.jip.2014.04.005. PMid:24780219.
http://dx.doi.org/10.1016/j.jip.2014.04....
), C. rhizophorae (SILVA et al., 2013Silva PM, Vianna RT, Guertler C, Ferreira LP, Santana LN, Fernandéz-Boo S, et al. First report of the protozoan parasite in South America, infecting mangrove oysters from the Paraíba River. Perkinsus marinusCrassostrea rhizophoraeJ Invertebr Pathol 2013; 113(1): 96-103. http://dx.doi.org/10.1016/j.jip.2013.02.002. PMid:23439264.
http://dx.doi.org/10.1016/j.jip.2013.02....
) and C. gigas(provided by the Laboratory of Immunology Applied to Aquaculture, UFSC), were used as controls. The PCR amplification products were stained with Blue Green Loading Dye I (LGC Biotecnologia) and resolved via electrophoresis in 1% agarose gels.
The 16S amplification products generated by PCR were digested with the enzymeAluI according to the manufacturer’s instructions (Promega), as described by Lazoski (2004)Lazoski CVS. Sistemática molecular e genética populacional de ostras brasileiras (Crassostrea spp.) [Thesis] Rio de Janeiro: Universidade Federal do Rio de Janeiro; 2004.. The digestion reactions were prepared in a final volume of 20 µL, containing 2 µg of the PCR product, 2 µL of reaction buffer, 0.2 µL of BSA and 0.5 µL of the restriction enzyme diluted in ultrapure water. The obtained Restriction Fragment Length Polymorphism (RFLP) patterns were compared with those observed following the digestion of DNA from the C. gigas, C. gasar and C. rhizophorae controls.
Aliquots of 10 µL of the digestion products were resolved on 8% polyacrylamide non-denaturing gels, which were stained with the Gel Red™ Nucleic Acid Gel Stain (Biotium) for 20 min.
Results
Identification of oyster species
Oysters from the different production stages were analyzed by PCR, and a fragment of the expected size of 560 bp was produced. The analysis of these fragments digested with endonuclease AluI showed a pattern consistent with the control corresponding to the species C. gasar. This pattern is shown in Figure 1, consisting of a large 250 bp fragment and a group of small 100 bp fragments.
Fragmentation profile of the 16S mitochondrial gene from oysters sampled at different stages of culture, after digestion with the endonuclease AluI, resolved on an 8% polyacrylamide gel. Samples of breeder oysters (R1, R2 and R3) and spats (S1, S2 and S3). DNA controls for C. gasar (Cga),C. rhizophorae (Cr) and C. gigas (Cgi). C: negative control for PCR assays. M: 50 bp molecular weight marker (Thermo).
Monitoring of Perkinsus spp. on the PRIMAR farm
Perkinsus spp. infection was evaluated in oysters during all production stages, from broodstock to commercial size. The parasitic indices determined for the oysters analyzed in this study are shown in Table 2. Perkinsus spp. infections were confirmed by PCR for all sampling dates (Figure 2).
Parasitic indices [prevalence (Prev.) and intensity of infection byPerkinsus sp. (Int.)] obtained by Ray’s fluid thioglycolate medium analysis (RFTM) in Crassostrea gasar oysters sampled during the production stages at the PRIMAR farm and confirmation of the genus Perkinsus by PCR. N = number of samples tested by RFTM or PCR.
Agarose gel electrophoresis (1%) of the ITS rDNA fragments from genusPerkinsus amplified from DNA from C. gasar oysters sampled at different stages of culture.Perkinsus-infected oysters (I1, I2, I3) and non-infected oysters (NI1, NI2). Positive controls with P. marinus (Pm) and P. beihaiensis (Pb). M: 1 Kb molecular weight marker (Thermo).
The broodstock analyzed in 2013 showed a high prevalence (60%) but a low-intensity of Perkinsus spp. infection (1.17 ± 0.41). In contrast, the breeder oysters used for spawning in 2014 presented a high prevalence (75%) and moderate intensity of infection (2.78 ± 0.44). However, the spats obtained from the spawning of these breeders were negative forPerkinsus spp. infection during the period in which they were kept inside the hatchery. When the spats were transferred to the pond, where they came into contact with water from the shrimp pond, they acquired the infection (46%), but at a low intensity (1.08 ± 0.28). The growth of spats in ponds increased the prevalence of Perkinsus spp. to 56% three months after placement in the ponds (October 2014), and to 73% at seven months after placement (February 2015) (Table 2). Then, the prevalence of Perkinsus spp. decreased in both groups of oysters: in the larger ones sampled in March (50%) and in the smaller ones sampled in May (17%). Interestingly, the intensity of infection in the larger oysters was higher (2.8 ± 1.64) than in the smaller oysters, sampled in February (1.27 ± 0.46), but not as high as that observed in oysters at the final stage of growth (3.94 ± 0.24; Table 2).
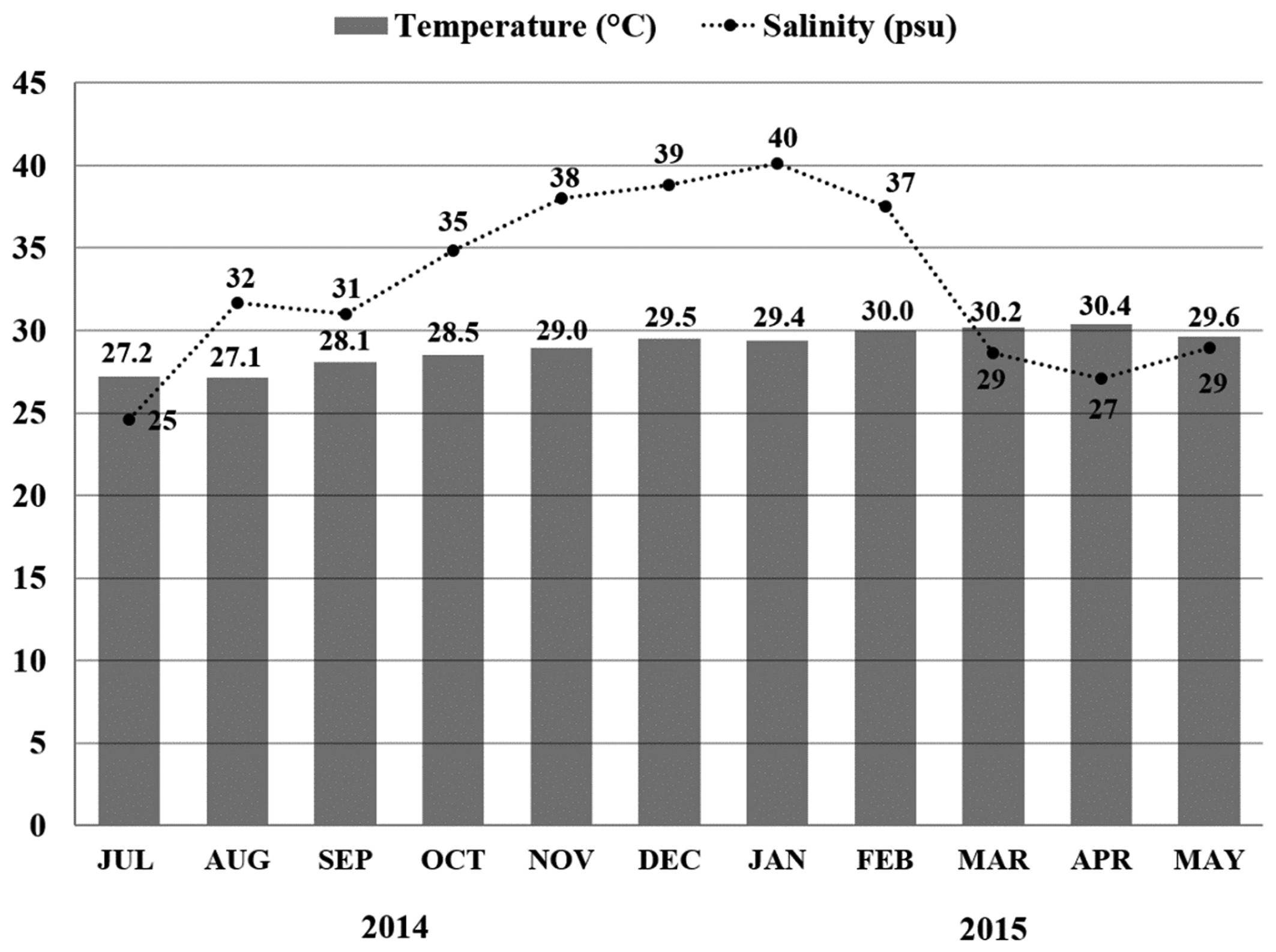
Figure 3 shows the temperature and salinity of the shrimp pond from July 2014 to May 2015. Temperature did not vary (mean ± SD: 29.0 ± 1.1 °C), while salinity ranged from 25 to 40 psu (mean ± SD: 32.8 ± 5.3 psu).
Water temperature (°C) and salinity (psu) in the shrimp pond during the oysters’ growing stage (July 2014 to May 2015).
Oyster productivity traits
Oysters cultured in shrimp ponds showed different growth rates. One group grew quicker and corresponded to the oysters selected and separated in July 2014 because of their larger size. Those oysters reached commercial size (7.1 cm) eight months after placement in the ponds. The other group grew slower and reached a small commercial size (5.3 cm) 10 months after placement in the ponds. In NE Brazil, oysters are commercialized starting at 5 cm in size. The growth rates of these two groups corresponded to 6.79 and 3.6 mm per month, respectively.
Although no sampling was specifically done to estimate oyster mortality during the growing stage in shrimp ponds, no abnormal mortality was reported during this period.
Discussion
This study analyzed the dynamics of Perkinsus spp. infection in a complete production system for native oysters for the first time in Brazil. The prevalence and intensity of Perkinsus spp. infection were determined in all stages of production, including (1) broodstock oysters, (2) spats in a hatchery, (3) spats in the shrimp ponds, (4) juvenile oysters, and (5) adult oysters until they reached 7 cm in a total of 13 months of growth.
The native oysters inhabiting estuaries in NE Brazil belong to at least two different species. Oysters of the species C. gasar show better growth under culture than those of the species C. rhizophorae (Dr. Carlos Gomes; personal communication), which has been previously reported for these species during the larval growth stage (CHRISTO et al., 2010Christo SW, Absher TM, Boehs G. Morphology of the larval shell of three oyster species of the genus Crassostrea Sacco, 1897 (Bivalvia: Ostreidae). Braz J Biol 2010; 70(3): 645-650. http://dx.doi.org/10.1590/S1519-69842010000300023. PMid:20730353.
http://dx.doi.org/10.1590/S1519-69842010...
). Therefore, in regions where both species can be found, C. gasar has garnered more interest in regard to oyster farming. In this study, all of the breeder oysters and, thus, their offspring belonged to a single species, C. gasar.
The results of this study indicate that broodstock oysters maintained for several years in the shrimp ponds at the PRIMAR farm were previously infected byPerkinsus spp. It is possible that the broodstock oysters had acquired the infection by direct contact with the estuary water that supplies the shrimp ponds from the Guaraíras lagoon, where there are also natural oyster populations. This hypothesis should be further investigated, as there are currently no studies on the prevalence of Perkinsus spp. in oysters at this location. This is therefore the first report of Perkinsus spp. in the state of Rio Grande do Norte.
The transmission of Perkinsus spp. parasites is known to occur through direct contact of healthy individuals (susceptible hosts) with water containing parasite cells released from infected individuals. There are no intermediate hosts and all stages of the parasite’s lifecycle are infective (VILLALBA et al., 2004Villalba A, Reece KS, Ordás MC, Casas SM, Figueras A. Perkinsosis in molluscs: a review. Aquat Living Resour 2004; 17(4): 411-432. http://dx.doi.org/10.1051/alr:2004050.
http://dx.doi.org/10.1051/alr:2004050...
). Infection is thought to occur through at least two mechanisms: 1) via ingestion of the parasite into the gastrointestinal tract (TASUMI & VASTA, 2007Tasumi S, Vasta GR. A Galectin of unique domain organization from hemocytes of the eastern oyster (Crassostrea virginicaPerkinsus marinus.) is a receptor for the protistan parasite J Immunol 2007; 179(5): 3086-3098. http://dx.doi.org/10.4049/jimmunol.179.5.3086. PMid:17709523.
http://dx.doi.org/10.4049/jimmunol.179.5...
; CHU, 1996Chu FLE. Laboratory investigations of susceptibility, infectivity, and transmission of in oysters. Perkinsus marinusJ Shellfish Res 1996; 15: 57-66.; BUSHEK et al., 2002Bushek D, Scarpa J, Laramore SE. Susceptibility of the Caribbean oyster to Crassostrea rhizophoraePerkinsus marinus.J Shellfish Res 2002; 21: 371-372.), and 2) in the act of feeding through the oyster mantle during the rejection of pseudofeces at the labial palps (ALLAM et al., 2013Allam B, Carden WE, Ward JE, Ralph G, Winnick S, Espinosa EP. Early host-pathogen interactions in marine bivalves: evidence that the alveolate parasite Perkinsus marinus infects through the oyster mantle during rejection of pseudofeces. J Invertebr Pathol 2013; 113(1): 26-34. http://dx.doi.org/10.1016/j.jip.2012.12.011. PMid:23274079.
http://dx.doi.org/10.1016/j.jip.2012.12....
). However, there are no existing studies on vertical transmission of Perkinsus spp., even though in cases with a severe intensity of infection, the gonad follicles can contain numerous parasite cells (CASAS & VILLALBA, 2012Casas SM, Villalba A. Study of perkinsosis in the grooved carpet shell clam in Galicia (NW Spain). III. The effects of infection on clam reproduction. Ruditapes decussatusPerkinsus olseniAquaculture 2012; 356-357: 40-47. http://dx.doi.org/10.1016/j.aquaculture.2012.05.038.
http://dx.doi.org/10.1016/j.aquaculture....
; SILVA et al., 2015Silva PM, Scardua MP, Vieira CB, Alves AC, Dungan CF. Survey of pathologies in (Adanson, 1757) oysters from cultured and wild populations in the São Francisco estuary, Sergipe, northeast Brazil. Crassostrea gasarJ Shellfish Res 2015; 34(2): 289-296. http://dx.doi.org/10.2983/035.034.0210.
http://dx.doi.org/10.2983/035.034.0210...
).
In the current study, transmission of Perkinsus spp. from parents to the offspring was not observed, as all of the spats samples (n = 9; totaling 3.6 g) were negative when analyzed by PCR. Studies conducted to evaluate different methods of eliminating Perkinsus spp. have indicated that low-to-moderate levels of UV irradiation (4,000 to 14,000 µW.s/cm2) inhibit the proliferation of the parasite and that high levels (> 28,000 µW.s/cm2) kill P. marinus cells (BUSHEK & HOWELL, 2000Bushek D, Howell TL. The effect of UV irradiation on perkinsus marinus and its potential use to reduce transmission via shellfish effluents [online]. North Dartmouth: Northeastern Regional Aquaculture Center; 2000. 4 p. Publication No. 00-008. [cited 2015 June 28]. Available from: https://agresearch.umd.edu/sites/default/files/_docs/00-008%20Shellfish.pdf
https://agresearch.umd.edu/sites/default...
). Moreover, high levels of UV irradiation (60,000 µW.s/cm2) are required to kill P. olseni hypnospores (LESTER & HAYWARD, 2005Lester RJG, Hayward CJ. Control of Perkinsus disease in abalone. Brisbane: University of Queensland, Fisheries Research and Development Corporation; 2005. Project 2000/151 Final Report.). Other methods, such as chlorination, are also effective in inactivating P. marinus and P. olseni cells (BUSHEK & HOWELL, 2000Bushek D, Howell TL. The effect of UV irradiation on perkinsus marinus and its potential use to reduce transmission via shellfish effluents [online]. North Dartmouth: Northeastern Regional Aquaculture Center; 2000. 4 p. Publication No. 00-008. [cited 2015 June 28]. Available from: https://agresearch.umd.edu/sites/default/files/_docs/00-008%20Shellfish.pdf
https://agresearch.umd.edu/sites/default...
; GOGGIN et al., 1990Goggin CL, Sewel KB, Lester RJG. Tolerances of spp. (Protozoa, Apicomplexa) to temperature, chlorine and salinity. PerkinsusJ Shellfish Res 1990; 9: 145-148.; BUSHEK et al., 1997Bushek D, Holley RA, Kelly MC. Treatment of Perkinsus marinus: contaminated materials. J Shellfish Res 1997; 16: 330.). Both methods (chlorination and UV-C irradiation of water) are commonly used in bivalve hatcheries and were also employed at the PRIMAR facility, in addition to 1-µm seawater filtration. In the present study, ifPerkinsus spp. cells had been released from breeders during spawning, which lasted two days, the applied disinfection procedures would ultimately have led to the complete elimination of parasite cells. Therefore, this result clearly demonstrates the feasibility of producingPerkinsus-free oyster spats from infected broodstock and the possibility of further commercialization.
Currently, Brazilian law prevents the exportation of any bivalve species from a state with confirmed cases of P. marinus and P. olseni(notifiable species to the OIE). This situation recently occurred in the state of Paraíba (Ordinance No. 4 of the MPA, 31 January 2013; SILVA et al., 2013Silva PM, Vianna RT, Guertler C, Ferreira LP, Santana LN, Fernandéz-Boo S, et al. First report of the protozoan parasite in South America, infecting mangrove oysters from the Paraíba River. Perkinsus marinusCrassostrea rhizophoraeJ Invertebr Pathol 2013; 113(1): 96-103. http://dx.doi.org/10.1016/j.jip.2013.02.002. PMid:23439264.
http://dx.doi.org/10.1016/j.jip.2013.02....
). These measurements, therefore, have negative consequences for oyster farming in NE Brazil. In this context, generatingPerkinsus-free oysters is a promising alternative to sanitary controls in bivalves.
The results of the present study also showed that when oyster spats (free ofPerkinsus spp.) were exposed to water from the ponds, they were quickly infected by Perkinsus spp. (after two months). These infections likely occurred for at least two reasons: 1) spats were transferred to the same pond where broodstock oysters were kept, and 2) Perkinsusspp. were already present in the ponds and/or seawater estuary that supplied the shrimp ponds.
In this study, we observed a gradual increase in the prevalence ofPerkinsus spp. infection (46%, 56% and 73%) up to seven months after the spats had been transferred to the ponds, although the intensity was mild. This information indicates that individuals in the population acquired the infection slowly. However, 10 months after placement in the ponds, there was a sharp drop in the prevalence (17%), and a significant increase in the intensity (3.94), of infection, the latter of which almost reached the highest possible level (4.0) at that time. Experimental infections of C. virginica oysters withP. marinus have indicated that there are two phases of infection: first, low-intensity infections associated with low mortality (after three weeks), and second, very heavy-intensity infections, with high mortality (after six weeks; FORD et al., 2002Ford SE, Chintala MM, Bushek D. Comparison of in vitro-cultured and wild-type Perkinsus marinus. I. Pathogen virulence. Dis Aquat Organ 2002; 51(3): 187-201. http://dx.doi.org/10.3354/dao051187. PMid:12465877.
http://dx.doi.org/10.3354/dao051187...
). It is possible that a similar pattern occurs in natural infections of P. marinus in Brazil, but with a longer duration. Despite the high prevalence (73%; 7th month) and intensity (3.94; 10th month) of Perkinsus spp. infection, both conditions did not occur together, which might have contributed to the lack of mortality events during the growing stage of oysters. To date, massive mortality events have not been reported in cultured oysters from NE Brazil (Farmers, personal communication; SILVA et al., 2013Silva PM, Vianna RT, Guertler C, Ferreira LP, Santana LN, Fernandéz-Boo S, et al. First report of the protozoan parasite in South America, infecting mangrove oysters from the Paraíba River. Perkinsus marinusCrassostrea rhizophoraeJ Invertebr Pathol 2013; 113(1): 96-103. http://dx.doi.org/10.1016/j.jip.2013.02.002. PMid:23439264.
http://dx.doi.org/10.1016/j.jip.2013.02....
, 2014Silva PM, Scardua MP, Vianna RT, Mendonça RC, Vieira CB, Dungan CF, et al. Two Perkinsus spp. infect oysters from cultured and wild populations of the Rio São Francisco estuary, Sergipe, northeastern Brazil. Crassostrea gasarJ Invertebr Pathol 2014; 119: 62-71. http://dx.doi.org/10.1016/j.jip.2014.04.005. PMid:24780219.
http://dx.doi.org/10.1016/j.jip.2014.04....
; QUEIROGA et al., 2015Queiroga FR, Vianna RT, Vieira CB, Farias ND, Silva PM. Parasites infecting the cultured oyster (Adanson, 1757) in Northeast Brazil. Crassostrea gasarParasitology 2015; 142(6): 756-766. http://dx.doi.org/10.1017/S0031182014001863. PMid:25553815.
http://dx.doi.org/10.1017/S0031182014001...
).
In NE Brazil, the pattern of infection by Perkinsus spp. in oysters seems to be associated with salinity, i.e. the prevalence and parasite burden decrease at low salinities, which usually occurs in the rainy season (BRANDÃO et al., 2013Brandão RP, Boehs G, Sabry RC, Ceuta LO, Luz MSA, Queiroga FR, et al. Perkinsus sp. infecting oyster (Guilding, 1828) on the coast of Bahia, Brazil. Crassostrea rhizophoraeJ Invertebr Pathol 2013; 112(2): 138-141. http://dx.doi.org/10.1016/j.jip.2012.11.003. PMid:23201453.
http://dx.doi.org/10.1016/j.jip.2012.11....
; SILVA et al., 2014Silva PM, Scardua MP, Vianna RT, Mendonça RC, Vieira CB, Dungan CF, et al. Two Perkinsus spp. infect oysters from cultured and wild populations of the Rio São Francisco estuary, Sergipe, northeastern Brazil. Crassostrea gasarJ Invertebr Pathol 2014; 119: 62-71. http://dx.doi.org/10.1016/j.jip.2014.04.005. PMid:24780219.
http://dx.doi.org/10.1016/j.jip.2014.04....
; QUEIROGA et al., 2015Queiroga FR, Vianna RT, Vieira CB, Farias ND, Silva PM. Parasites infecting the cultured oyster (Adanson, 1757) in Northeast Brazil. Crassostrea gasarParasitology 2015; 142(6): 756-766. http://dx.doi.org/10.1017/S0031182014001863. PMid:25553815.
http://dx.doi.org/10.1017/S0031182014001...
). Accordingly, in the shrimp pond, high salinity (37 to 40 psu) accumulated during November 2014 to February 2015, could have contributed to the highest prevalence of Perkinsus spp. (73%) observed in February 2015. However, studies linking physical parameters of the seawater,Perkinsus spp. infection dynamics, and productive traits (growth and mortality) are missing and must be explored in future experiments to fully understand the impact of Perkinsus spp. disease in cultured oysters in Brazil.
Another interesting observation made in the present study was that the larger oysters that reached a commercial size in March (8th month of culture in the ponds) showed a lower prevalence of infection than the smaller ones (50% vs. 73%), but their intensity of infection was higher (2.80 vs. 1.27). It is possible that their larger size and, therefore, larger gill area led to a higher filtration rate (EHRICH & HARRIS, 2015Ehrich MK, Harris LA. A review of existing eastern oyster filtration rate models. Ecol Modell 2015; 297: 201-212. http://dx.doi.org/10.1016/j.ecolmodel.2014.11.023.
http://dx.doi.org/10.1016/j.ecolmodel.20...
), which would result in increased ingestion of parasite cells, thus favoring infection. Paynter et al. (2010)Paynter KT, Politano V, Lane HA, Allen SM, Meritt D. Growth rates and prevalence of prevalence in restored oyster populations in Maryland. Perkinsus marinusJ Shellfish Res 2010; 29(2): 309-317. http://dx.doi.org/10.2983/035.029.0205.
http://dx.doi.org/10.2983/035.029.0205...
showed that prevalence and intensity of P. marinus infection increased with the age ofC. virginica oysters, which was highly correlated with oyster shell height and dry tissue weight. In C. gasar, despite the lowest prevalence, the parasite burden was higher in tissues of larger oysters.
A single study evaluated the feasibility of C. gasar oysters growing and surviving in shrimp ponds in the Sergipe state (NE, Brazil) (ROSA, 2014Rosa LC. Crescimento e sobrevivência da ostra (Lamarck, 1819) mantida em um viveiro de cultivo de camarão. Crassostrea brasilianaArq. Cienc Mar 2014; 47(1): 64-68.). Crassostrea gasar oysters with an initial size of 4 cm were growth for just four months; the results showed reduced growth rate (2.37 mm per month). In the current study, growth rates were more variable and higher (3.6 and 6.79 mm per month) than the previous study, and even higher than those (2.16-2.64 mm per month) found byPereira et al. (2001)Pereira OM, Machado IC, Henriques MB, Yamanaka N. Crescimento da ostra semeada sobre tabuleiro em diferentes densidades na região estuarino-lagunar de Cananéia-SP (25º S, 48º W). Crassostrea brasilianaBol Inst Pesca SP 2001; 27(2): 163-174. for C. gasar cultured by traditional rack methods in São Paulo state.
In conclusion, this is the first study in Brazil to evaluate the presence ofPerkinsus spp. at all stages of C. gasaroyster production, and the case described herein is also the first reported in the state of Rio Grande do Norte (NE Brazil). The results of this study also showed that it is possible to produce Perkinsus-free C. gasaroyster spats from infected broodstock, and that their production in shrimp ponds is feasible.
Acknowledgements
We thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq for financial support to the project CNPq/MPA No. 406170/2012-6. We thank to Universidade Federal da Paraíba for English editing service. We thank to Sâmia Sousa Duarte for the technical assistance.
References
- Allam B, Carden WE, Ward JE, Ralph G, Winnick S, Espinosa EP. Early host-pathogen interactions in marine bivalves: evidence that the alveolate parasite Perkinsus marinus infects through the oyster mantle during rejection of pseudofeces. J Invertebr Pathol 2013; 113(1): 26-34. http://dx.doi.org/10.1016/j.jip.2012.12.011. PMid:23274079.
» http://dx.doi.org/10.1016/j.jip.2012.12.011 - Amaral VS, Simone LRL. Revision of genus (Bivalvia: Ostreidae) of Brazil. CrassostreaJ Mar Biol Assoc UK 2014; 94(4): 811-836. http://dx.doi.org/10.1017/S0025315414000058.
» http://dx.doi.org/10.1017/S0025315414000058 - Brandão RP, Boehs G, Sabry RC, Ceuta LO, Luz MSA, Queiroga FR, et al. Perkinsus sp. infecting oyster (Guilding, 1828) on the coast of Bahia, Brazil. Crassostrea rhizophoraeJ Invertebr Pathol 2013; 112(2): 138-141. http://dx.doi.org/10.1016/j.jip.2012.11.003. PMid:23201453.
» http://dx.doi.org/10.1016/j.jip.2012.11.003 - Bushek D, Holley RA, Kelly MC. Treatment of Perkinsus marinus: contaminated materials. J Shellfish Res 1997; 16: 330.
- Bushek D, Howell TL. The effect of UV irradiation on perkinsus marinus and its potential use to reduce transmission via shellfish effluents [online]. North Dartmouth: Northeastern Regional Aquaculture Center; 2000. 4 p. Publication No. 00-008. [cited 2015 June 28]. Available from: https://agresearch.umd.edu/sites/default/files/_docs/00-008%20Shellfish.pdf
» https://agresearch.umd.edu/sites/default/files/_docs/00-008%20Shellfish.pdf - Bushek D, Scarpa J, Laramore SE. Susceptibility of the Caribbean oyster to Crassostrea rhizophoraePerkinsus marinus.J Shellfish Res 2002; 21: 371-372.
- Casas SM, Villalba A, Reece KS. Study of perkinsosis in the carpet shell clam Tapes decussatus in Galicia (NW Spain). I. Identification of the aetiological agent and in vitro modulation of zoosporulation by temperature and salinity. Dis Aquat Organ 2002; 50(1): 51-65. http://dx.doi.org/10.3354/dao050051. PMid:12152905.
» http://dx.doi.org/10.3354/dao050051 - Casas SM, Villalba A. Study of perkinsosis in the grooved carpet shell clam in Galicia (NW Spain). III. The effects of infection on clam reproduction. Ruditapes decussatusPerkinsus olseniAquaculture 2012; 356-357: 40-47. http://dx.doi.org/10.1016/j.aquaculture.2012.05.038.
» http://dx.doi.org/10.1016/j.aquaculture.2012.05.038 - Choi KS, Park KI. Review on the protozoan parasite (Lester and Davis 1981) infection in Asian waters. In: Perkinsus olseniIshimatsu A, Lie HJ. Coastal environmental and ecosystem issues of the east China sea. Nagazaki: TERRAPUB, Nagasaki University; 2010. p. 269-281.
- Christo SW, Absher TM, Boehs G. Morphology of the larval shell of three oyster species of the genus Crassostrea Sacco, 1897 (Bivalvia: Ostreidae). Braz J Biol 2010; 70(3): 645-650. http://dx.doi.org/10.1590/S1519-69842010000300023. PMid:20730353.
» http://dx.doi.org/10.1590/S1519-69842010000300023 - Chu FLE. Laboratory investigations of susceptibility, infectivity, and transmission of in oysters. Perkinsus marinusJ Shellfish Res 1996; 15: 57-66.
- Ehrich MK, Harris LA. A review of existing eastern oyster filtration rate models. Ecol Modell 2015; 297: 201-212. http://dx.doi.org/10.1016/j.ecolmodel.2014.11.023.
» http://dx.doi.org/10.1016/j.ecolmodel.2014.11.023 - Empresa de Pesquisa Agropecuária e Extensão Rural de Santa Catarina – EPAGRI. Síntese informativa da maricultura [online]. Florianópolis: EPAGRI; 2014 [cited 2015 June 28]. Available from: http://www.epagri.sc.gov.br/wp-content/uploads/2013/08/Sintese_informativa_da_maricultura_2014.pdf
» http://www.epagri.sc.gov.br/wp-content/uploads/2013/08/Sintese_informativa_da_maricultura_2014.pdf - Ferreira LP, Sabry RC, Silva PM, Gesteira TCV, Romão LS, Paz MP, et al. First report of Perkinsus beihaiensis in wild clams (Bivalvia: Veneridae) in Brazil. Anomalocardia brasilianaExp Parasitol 2015; 150: 67-70. http://dx.doi.org/10.1016/j.exppara.2014.07.012. PMid:25088443.
» http://dx.doi.org/10.1016/j.exppara.2014.07.012 - Ford SE, Chintala MM, Bushek D. Comparison of in vitro-cultured and wild-type Perkinsus marinus. I. Pathogen virulence. Dis Aquat Organ 2002; 51(3): 187-201. http://dx.doi.org/10.3354/dao051187. PMid:12465877.
» http://dx.doi.org/10.3354/dao051187 - Goggin CL, Sewel KB, Lester RJG. Tolerances of spp. (Protozoa, Apicomplexa) to temperature, chlorine and salinity. PerkinsusJ Shellfish Res 1990; 9: 145-148.
- Gomes CHAM, Silva FC, Lopes GR, Melo CMR. The reproductive cycle of the oyster Crassostrea gasar.Braz J Biol 2014; 74(4): 967-976. http://dx.doi.org/10.1590/1519-6984.04912. PMid:25627610.
» http://dx.doi.org/10.1590/1519-6984.04912 - Jones AB, Dennison WC, Preston NP. Integrated treatment of shrimp effluent by sedimentation, oyster filtration and macroalgal absorption: a laboratory scale study. Aquaculture 2001; 193(1-2): 155-178. http://dx.doi.org/10.1016/S0044-8486(00)00486-5.
» http://dx.doi.org/10.1016/S0044-8486(00)00486-5 - Jones AB, Preston NP. Sydney rock oyster, (Iredale & Roughley), filtration of shrimp farm effluent: the effects on water quality. Saccostrea commercialisAquacult Res 1999; 30(1): 51-57. http://dx.doi.org/10.1046/j.1365-2109.1999.00299.x.
» http://dx.doi.org/10.1046/j.1365-2109.1999.00299.x - Kessing B, Croom H, Martin A, McIntosh C, Owen M, Palumbi S. A simple guide to PCR. Honolulu: Department of Zoology, University of Hawaii; 1989.
- Lauckner G. Diseases of mollusca: bivalvia. In: Kinne O. Diseases of marine animals. Hamburg: Biologische Anstalt Helgoland; 1983. p. 477-961.
- Lavander HD, Cardoso LO Jr, Silva LOB, Gàlvez AO. Estudo de viabilidade econômica para ostreicultura familiar em Pernambuco, Brasil. Custos e @gronegócio Online 2013; 9(2): 173-187.
- Lazoski CVS. Sistemática molecular e genética populacional de ostras brasileiras (Crassostrea spp.) [Thesis] Rio de Janeiro: Universidade Federal do Rio de Janeiro; 2004.
- Lester RJG, Hayward CJ. Control of Perkinsus disease in abalone. Brisbane: University of Queensland, Fisheries Research and Development Corporation; 2005. Project 2000/151 Final Report.
- Lopes GR, Gomes CHAM, Tureck CR, Melo CMR. Growth of cultured in marine and estuary environments in brazilian waters. Crassostrea gasarPesq Agropec Brasil 2013; 48(8): 975-982. http://dx.doi.org/10.1590/S0100-204X2013000800024.
» http://dx.doi.org/10.1590/S0100-204X2013000800024 - Martinez-Cordova LR, Martinez-Porchas M. Polyculture of pacific white shrimp, , giant oyster, and black clam, Chione fluctifraga in ponds in Sonora, Mexico. Litopenaeus vannameiCrassostrea gigasAquaculture 2006; 258(1-4): 321-326. http://dx.doi.org/10.1016/j.aquaculture.2006.03.026.
» http://dx.doi.org/10.1016/j.aquaculture.2006.03.026 - Martínez-Porchas M, Martínez-Córdova LR, Pórchas-Cornejo MA, López-Elías JA. Shrimp polyculture: a potentially profitable, sustainable, but uncommon aquacultural pratice. Rev Aquaculture 2010; 2(2): 73-85. http://dx.doi.org/10.1111/j.1753-5131.2010.01023.x.
» http://dx.doi.org/10.1111/j.1753-5131.2010.01023.x - Páez-Osuna F. The environmental impact of shrimp aquaculture: causes, effects, and mitigating alternatives. Environ Manage 2001; 28(1): 131-140. http://dx.doi.org/10.1007/s002670010212. PMid:11436996.
» http://dx.doi.org/10.1007/s002670010212 - Paynter KT, Politano V, Lane HA, Allen SM, Meritt D. Growth rates and prevalence of prevalence in restored oyster populations in Maryland. Perkinsus marinusJ Shellfish Res 2010; 29(2): 309-317. http://dx.doi.org/10.2983/035.029.0205.
» http://dx.doi.org/10.2983/035.029.0205 - Pereira OM, Machado IC, Henriques MB, Yamanaka N. Crescimento da ostra semeada sobre tabuleiro em diferentes densidades na região estuarino-lagunar de Cananéia-SP (25º S, 48º W). Crassostrea brasilianaBol Inst Pesca SP 2001; 27(2): 163-174.
- Queiroga FR, Vianna RT, Vieira CB, Farias ND, Silva PM. Parasites infecting the cultured oyster (Adanson, 1757) in Northeast Brazil. Crassostrea gasarParasitology 2015; 142(6): 756-766. http://dx.doi.org/10.1017/S0031182014001863. PMid:25553815.
» http://dx.doi.org/10.1017/S0031182014001863 - Ramos CO, Gomes CHAM, Magalhães ARM, Santos AI, Melo CMR. Maturation of the mangrove oyster at diferent temperatures in the laboratory. Crassostrea gasarJ Shellfish Res 2014; 33(1): 187-194. http://dx.doi.org/10.2983/035.033.0118.
» http://dx.doi.org/10.2983/035.033.0118 - Ramos R, Vinatea L, Costa R. Tratamiento de efluentes del cultivo de Litopenaeus vannamei por sedimentación y filtración por la ostra Crassostrea rhizophorae.Lat Am J Aquat Res 2008; 36(2): 235-244. http://dx.doi.org/10.3856/vol36-issue2-fulltext-6.
» http://dx.doi.org/10.3856/vol36-issue2-fulltext-6 - Reyes GG. Mixed culture (shrimps, oysters and macroalgae) to improve the productivity, and reduce the environmental impact to coastal ecosystem caused by shrimp hatcheries in Mexico. Int J Fish Aquacult 2013; 5(10): 262-269.
- Rosa LC. Crescimento e sobrevivência da ostra (Lamarck, 1819) mantida em um viveiro de cultivo de camarão. Crassostrea brasilianaArq. Cienc Mar 2014; 47(1): 64-68.
- Sabry RC, Gesteira TCV, Magalhães ARM, Barracco MA, Guertler C, Ferreira LP, et al. Parasitological survey of mangrove oyster, , in the Pacoti River Estuary, Ceará State, Brazil. Crassostrea rhizophoraeJ Invertebr Pathol 2013; 112(1): 24-32. http://dx.doi.org/10.1016/j.jip.2012.10.004. PMid:23147104.
» http://dx.doi.org/10.1016/j.jip.2012.10.004 - Sabry RC, Rosa RD, Magalhães ARM, Barracco MA, Gesteira TCV, Silva PM. First report of Perkinsus sp. infecting mangrove oysters from the Brazilian coast. Crassostrea rhizophoraeDis Aquat Organ 2009; 88(1): 13-23. http://dx.doi.org/10.3354/dao02136. PMid:20183961.
» http://dx.doi.org/10.3354/dao02136 - Silva PM, Vianna RT, Guertler C, Ferreira LP, Santana LN, Fernandéz-Boo S, et al. First report of the protozoan parasite in South America, infecting mangrove oysters from the Paraíba River. Perkinsus marinusCrassostrea rhizophoraeJ Invertebr Pathol 2013; 113(1): 96-103. http://dx.doi.org/10.1016/j.jip.2013.02.002. PMid:23439264.
» http://dx.doi.org/10.1016/j.jip.2013.02.002 - Silva PM, Scardua MP, Vianna RT, Mendonça RC, Vieira CB, Dungan CF, et al. Two Perkinsus spp. infect oysters from cultured and wild populations of the Rio São Francisco estuary, Sergipe, northeastern Brazil. Crassostrea gasarJ Invertebr Pathol 2014; 119: 62-71. http://dx.doi.org/10.1016/j.jip.2014.04.005. PMid:24780219.
» http://dx.doi.org/10.1016/j.jip.2014.04.005 - Silva PM, Scardua MP, Vieira CB, Alves AC, Dungan CF. Survey of pathologies in (Adanson, 1757) oysters from cultured and wild populations in the São Francisco estuary, Sergipe, northeast Brazil. Crassostrea gasarJ Shellfish Res 2015; 34(2): 289-296. http://dx.doi.org/10.2983/035.034.0210.
» http://dx.doi.org/10.2983/035.034.0210 - Silveira RC, Silva FC, Gomes CHM, Ferreira JF, Melo CMR. Larval settlement and spat recovery rates of the oyster (Lamarck, 1819) using different systems to induce metamorphosis. Crassostrea brasilianaBraz J Biol 2011; 71(2): 557-562. http://dx.doi.org/10.1590/S1519-69842011000300029. PMid:21755177.
» http://dx.doi.org/10.1590/S1519-69842011000300029 - Tasumi S, Vasta GR. A Galectin of unique domain organization from hemocytes of the eastern oyster (Crassostrea virginicaPerkinsus marinus.) is a receptor for the protistan parasite J Immunol 2007; 179(5): 3086-3098. http://dx.doi.org/10.4049/jimmunol.179.5.3086. PMid:17709523.
» http://dx.doi.org/10.4049/jimmunol.179.5.3086 - Villalba A, Gestal C, Casas SM, Figueras A. Perkinsosis en moluscos. In: Figueras A, Novoa B. Enfermedades de moluscos bilvalvos de interés en acuicultura. Madrid: Fundación Observatorio Español de Acuicultura; 2011. p. 181-242.
- Villalba A, Reece KS, Ordás MC, Casas SM, Figueras A. Perkinsosis in molluscs: a review. Aquat Living Resour 2004; 17(4): 411-432. http://dx.doi.org/10.1051/alr:2004050.
» http://dx.doi.org/10.1051/alr:2004050 - World Organization for Animal Health – OIE. Manual of diagnostic tests for aquatic animals. 5th ed. Paris: OIE; 2006.
- World Organization for Animal Health – OIE. OIE Listed diseases, infections and infestations in force in 2015. Paris: OIE; 2015 [cited 2015 June 28]. Available from: http://www.oie.int/animal-health-in-the-world/oie-listed-diseases-2015
» http://www.oie.int/animal-health-in-the-world/oie-listed-diseases-2015 - Xie B, Jiang W, Yang H. Growth perfomance and nutrient quality of Chinese shrimp Penaeus chinensis in organic polyculture with razor clam or hard clam Sinovacula constrictaMeretix meretrix.Bulg J Agric Sci 2011; 17(6): 851-858.
Publication Dates
-
Publication in this collection
18 Mar 2016 -
Date of issue
Jan-Mar 2016
History
-
Received
21 Aug 2015 -
Accepted
13 Jan 2016