Abstract
Information about helminths of Molothrus bonariensis (Gmelin, 1789) (Passeriformes: Icteridae) are scarce; in this sense the objective of this paper was to contribute to its knowledge. Five hosts of southern Brazil were examined and the helminths Prosthogonimus ovatus, Tanaisia valida (Digenea), Diplotriaena bargusinica and Synhimantus (Dispharynx) nasuta (Nematoda) were identified. The species T. valida, P. ovatus and S. (D.) nasuta are for the first time registered for the bird in Brazil. Prosthogonimus ovatus, T. valida, D. bargusinica e S. (D.) nasuta are first recorded in M. bonariensis in the southern Brazilian state Rio Grande do Sul.
Keywords:
Shiny cowbird; Trematoda; Nematoda; parasites
Resumo
Informações sobre helmintos de Molothrus bonariensis (Gmelin, 1789) (Passeriformes: Icteridae) são escassas, nesse sentido, o objetivo do trabalho foi contribuir para esse conhecimento. Foram examinados cinco hospedeiros do extremo sul do Brasil, identificou-se os helmintos Prosthogonimus ovatus, Tanaisia valida (Digenea), Diplotriaena bargusinica e Synhimantus (Dispharynx) nasuta (Nematoda). As espécies T. valida, P. ovatus e S. (D.) nasuta são pela primeira vez registradas para a ave no Brasil. Prosthogonimus ovatus, T. valida, D. bargusinica e S. (D.) nasuta são pela primeira vez registradas em M. bonariensis no estado do Rio Grande do Sul.
Palavras-chave:
Vira-bosta; Trematoda; Nematoda; parasitos
Parasites are among the most common organisms in the world (HOBERG & KUTZ, 2013Hoberg EP, Kutz SJ. Parasites. In: Bültmann H, Ims RA, Fridriksson F, Hoberg EP, Meltofte H, Ganter B, Josefson A. Artic biodiversity assessment: status and trends in Artic biodiversity [online]. Akureyri: Conservation of Arctic Flora and Fauna; 2013 [cited 2015 Sep 02]. Available from: http://www.arcticbiodiversity.is/the-report/chapters/parasites
http://www.arcticbiodiversity.is/the-rep...
). They are extremely diversified and play a key role in ecological and evolutionary processes (GOMÉZ & NICHOLS, 2013Goméz A, Nichols E. Neglected wild life: Parasitic biodiversity as a conservation target. Int J Parasito.Parasit Wildl 2013; 2: 222-227. http://dx.doi.org/10.1016/j.ijppaw.2013.07.002.
http://dx.doi.org/10.1016/j.ijppaw.2013....
). Parasitism is one of the most successful ways of living presented by living organisms (POULIN & MORAND, 2000Poulin R, Morand S. The diversity of parasites. Q Rev Biol 2000; 75(3): 277-293. http://dx.doi.org/10.1086/393500. PMid:11008700.
http://dx.doi.org/10.1086/393500...
).
Birds are commonly parasitized by trematodes, nematodes, cestodes and acanthocephalans, as well as other groups (AMATO & AMATO, 2010Amato JFR, Amato SB. Técnicas gerais para coleta e preparação de helmintos endoparasitos de aves. In: Von Matter S, Straube FC, Accordi I, Piacentini V, Cândido JF Jr. Ornitologia e conservação: ciência aplicada, técnicas de pesquisa e levantamento. Rio de Janeiro: Technical Books; 2010. p. 369-393.). Taking into account the fact that Brazil presents one of the richest avifaunas in the world, much remains to be explored regarding this subject.
Molothrus bonariensis (Gmelin, 1789) (Passeriformes: Icteridae), known as the shiny cowbird, occurs in South America, islands of Central America, United States and Canada (The IUCN Red List of Threatened Species, 2015The IUCN Red List of Threatened Species [online]. Gland: International Union for Conservation of Nature and Natural Resources; 2015 [cited 2015 Aug 20]. Available from: http://www.iucnredlist.org.
http://www.iucnredlist.org...
). In the state of Rio Grande do Sul, Brazil, they inhabit the entire state, where enormous flocks often gather in cereal crops, especially rice, during and after harvesting (BELTON, 1994Belton W. Aves do Rio Grande do Sul: distribuição e biologia. São Leopoldo: UNISINOS; 1994. 548 p.). The male is black-feathered with bluish reflexes, and the female is brown. They feed on seeds and insects, and occasionally on fruits, and often can be seen near to cattle, turning over excrement to look for undigested seeds (EFE et al., 2001Efe MA, Mohr LV, Bugoni L. Guia ilustrado das aves dos parques de Porto Alegre. Porto Alegre: PROAVES/SMAM/COPESUL/CEMAVE; 2001. 144 p.).
Information regarding the shiny cowbird is mainly focused on the fact that this species lays its eggs in other birds’ nests. In Brazil, approximately 60 species belonging to nine families of Passeriformes are affected by the action of the shiny cowbird. This species is entirely dependent on other bird species for its reproduction, because it has not only lost the ability to build its own nests but also the instinct of taking care of its offspring (BLANCO, 1995Blanco DE. Parasitismo de cria del Tordo Renegrido Molothrus bonariensis sobre , en el este de la província de Buenos Aires. Agelaius ruficapillusEl Hornero 1995; 14(1-2): 44-45.; SICK, 1997Sick H. Ornitologia brasileira. Rio de Janeiro: Editora Nova Fronteira; 1997. 862 p.; EFE et al., 2001Efe MA, Mohr LV, Bugoni L. Guia ilustrado das aves dos parques de Porto Alegre. Porto Alegre: PROAVES/SMAM/COPESUL/CEMAVE; 2001. 144 p.; MERMOZ & REBOREDA, 2003Mermoz ME, Reboreda JC. Reproductive sucess of shiny cowbird () parasitizing the larger brown-and-yellow marshbird (Molothrus bonariensisPseudoleistes virescens) in Argentina. Auk 2003; 120(4): 1128-1139. http://dx.doi.org/10.1642/0004-8038(2003)120[1128:RSOSCM]2.0.CO;2.
http://dx.doi.org/10.1642/0004-8038(2003...
; ASTIÉ & REBOREDA, 2006Astié AA, Reboreda JC. Costs of egg punctures and parasitism by shiny cowbirds () at creamy-bellied thrush (Molothrus bonariensisTurdus amaurochalinus) nests. Auk 2006; 123(1): 23-32. http://dx.doi.org/10.1642/0004-8038(2006)123[0023:COEPAP]2.0.CO;2.
http://dx.doi.org/10.1642/0004-8038(2006...
).
Regarding helminths in M. bonariensis in Brazil, there have been isolated reports of Lyperosomum oswaldoi (Travassos, 1919) (Trematoda: Dicrocoeliidae) (TRAVASSOS et al., 1969Travassos L, Teixeira de Feitas JF, Kohn A. Trematódeos do Brasil. Mem Inst Oswaldo Cruz 1969; 67: 1-886.), Diplotriaena bargusinica Skrjabin, 1917 (Nematoda: Diplotriaenidae) (VICENTE et al., 1983Vicente JJ, Pinto RM, Noronha D. Estudo das espécies brasileiras do gênero Diplotriaena Henry & Ozoux, 1909 (Nematoda, Filarioidea). Mem Inst Oswaldo Cruz 1983; 78(2): 165-182. http://dx.doi.org/10.1590/S0074-02761983000200005.
http://dx.doi.org/10.1590/S0074-02761983...
) and Mediorhynchus emberizae (Rudolphi, 1819) (Acanthocephala: Gigantorhynchidae) (PETROCHENKO, 1971Petrochenko VI. Acanthocephala of domestic and wild animals. Jerusalém: Israel Program for Scientific Translations; 1971. vol. 2, 478 p.). In this context, the present study had the objective of reporting the presence of helminths associated with M. bonariensis.
Five hosts were examined (one male and four females), originating from the municipality of Rio Grande, Rio Grande do Sul, Brazil (32º 14'.37.24" S; 52°29'38.71" W) two birds in the summer and three in the winter. They were accidentally sampled in a trap that had been set to catch Chrysomus ruficapillus (Vieillot, 1819) (Passeriformes: Icteridae), in accordance with a license granted by the Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio No. 41095-3) and approval granted by the Comitê de Ética e Experimentação Animal from the Universidade Federal de Pelotas (CEEA/UFPel No. 147), covering the period of 2013-2015.
These specimes of Molothrus bonariensis were euthanasied, individually placed in identified plastic bags, and were transported to the Laboratório de Parasitologia de Animais Silvestres (LAPASIL/UFPel) and frozen until processing. To collect helminths, were necropsied and their organs (mouth, esophagus, proventriculus, gizzard, cecum, small and large intestines, trachea, lungs, heart, liver, gall bladder, pancreas, reproductive system, kidneys, cloaca and air sacs) were separated from each other, opened and rinsed with running water under a sieve of mesh size 150 µm.
The helminths were fixed in AFA for 24 hours and were stored in accordance with the protocol proposed by Amato & Amato, 2010. Trematodes were stained with Langeron’s carmine and nematodes were clarified with Aman’s lactophenol. They were mounted on permanent and semi-permanent slides and photographed through a microscope (Olympus® BX 41) with a coupled camera system. Morphological and morphometric identification were performed in accordance with Freitas (1951)Freitas JFT. Revisão da família Eucotylidae Skrjabin, 1924 (Trematoda). Mem Inst Oswaldo Cruz 1951; 49: 33-123. http://dx.doi.org/10.1590/S0074-02761951000100003. PMid:14890534.
http://dx.doi.org/10.1590/S0074-02761951...
, Kohn & Fernandes (1972)Kohn A, Fernandes BMM. Sobre a valiedade das espécies pertencentes ao gênero Luehe, 1899, da coleção helmintológica do Instituto Oswaldo Cruz. ProsthogonimusMem Inst Oswaldo Cruz 1972; 70(3): 309-325. http://dx.doi.org/10.1590/S0074-02761972000300005.
http://dx.doi.org/10.1590/S0074-02761972...
, Gibson et al. (2002)Gibson DI, Jones A, Bray RA. Keys to the Trematoda. London: CABI International/The Natural History Museum; 2002. vol. 1, 521 p., Bray et al. (2008)Bray RA, Gibson DI, Jones A. Keys to Trematoda. London: CABI International/Natural History Museum; 2008. vol. 3, 824 p., Lunaschi et al. (2015)Lunaschi LI, Drago FB, Draghi R. Redescription of Tanaisia dubia (Digenea) from the northeast region of Argentina, with a key to Neotropical species of the genus, and a key to genera of Tanaisiinae. Rev Mex Biodiv 2015; 86(4): 888-895. http://dx.doi.org/10.1016/j.rmb.2015.06.012.
http://dx.doi.org/10.1016/j.rmb.2015.06....
for Digenea, and with Vicente et al. (1983)Vicente JJ, Pinto RM, Noronha D. Estudo das espécies brasileiras do gênero Diplotriaena Henry & Ozoux, 1909 (Nematoda, Filarioidea). Mem Inst Oswaldo Cruz 1983; 78(2): 165-182. http://dx.doi.org/10.1590/S0074-02761983000200005.
http://dx.doi.org/10.1590/S0074-02761983...
, Zhang et al. (2004)Zhang L, Brooks DR, Causey D. Two species of Synhimantus (Dispharynx) Railliet, Henry and Sisoff, 1912 (Nematoda: Acuarioidea: Acuariidae) in passerine birds from the area the Conservación Guanacaste, Costa Rica. J Parasitol 2004; 90(5): 1133-1138. http://dx.doi.org/10.1645/GE-3253. PMid:15562615.
http://dx.doi.org/10.1645/GE-3253...
and Anderson et al. (2009)Anderson RC, Chabaud AG, Willmott S. Keys to the Nematode parasites of vertebrates: archival volume. London: CABI International; 2009. 463 p., for Nematoda. The parameters calculated were prevalence (P), mean abundance (MA) and mean intensity of infection (MI), in accordance with Bush et al. (1997)Bush AO, Lafferty KD, Lotz JM, Shostak AW. Parasitology meets ecology on its own terms: Margolis et al. revisited. J Parasitol 1997; 83(4): 575-583. http://dx.doi.org/10.2307/3284227. PMid:9267395.
http://dx.doi.org/10.2307/3284227...
. The voucher specimens were deposited in the “Coleção de Helmintos do Laboratório de Parasitologia de Animais Silvestres” from Instituto de Biologia, Universidade Federal de Pelotas (CHLAPASIL/UFPel) (No. 612 to 616).
Out of the five hosts Molothrus bonariensis examined four were positive for helminths (only females). Were identified two species of Trematoda Prosthogonimus ovatus (Rudolphi, 1803) (Lühe, 1899) (Prostogonimidae) (n = 5) and Tanaisia valida Freitas, 1951 (Eucotylidae) (n = 20) and two Nematoda Diplotriaena bargusinica Skrjabin, 1917 (Diplotriaenidae) (n = 37) (20 males and 17 females) and Synhimantus (Dispharynx) nasuta (Rudolphi, 1819) Chabaud, 1975 (Acuariidae) (n = 2) (one male and one female), the results for each host and the parasitological indexes are presented in Table 1. Diplotriaena bargusinica was the most prevalent parasite and the one with the highest mean infection intensity, followed by T. valida. The trematode P. ovatus and T. valida co-occurred (one host); D. bargusinica and T. valida (one host) and D. bargusinica and S. (D.) nasuta (one host). There were not analyzed pathological aspects related to the identified helminths.
Helminths and parasitological indexes of Molothrus bonariensis (Passeriformes: Icteridae) are from Rio Grande, Rio Grande do Sul, Brazil.
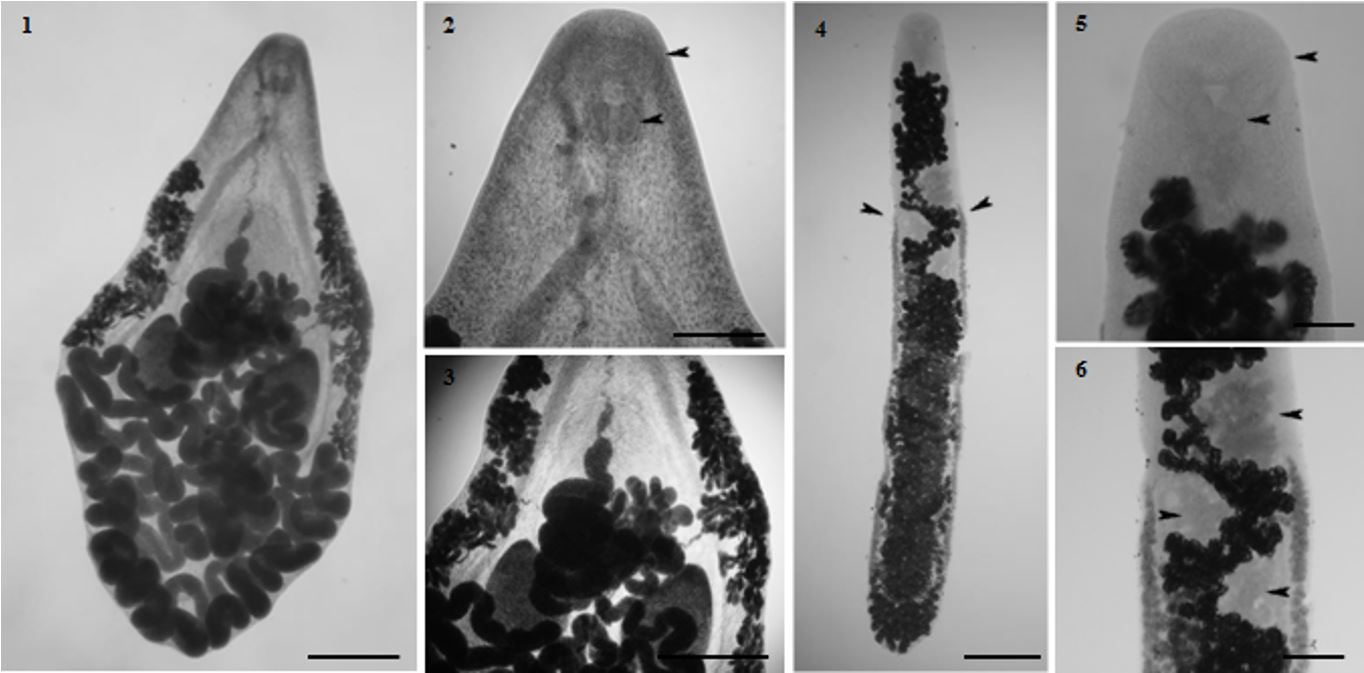
The morphological identification of each species was carried out according to the characteristics described by the previously mentioned authors. We present below the relevant aspects of each helminth. P. ovatus (Figure 1) is characterized by having the piriformis body, subterminal oral sucker, muscular pharynx present (Figure 2), simple intestinal cecum, few meandering surpassing the testicles. Acetabulum pre-equatorial, terminal genital pore located next to the oral sucker. Cirrus sac extending from the genital pore to the level of esophageal bifurcation, well-developed testicles, post-acetabular, post-ovarian, equatorial (Figure 3). Ovary pretesticular lobed, Mehlis gland situated below the ovary, uterus occupies half of the body and the excretory portion is terminal.
1-3- Prosthogonimus ovatus (Rudolphi, 1803) (Lühe, 1899) (Trematoda: Prostogonimidae) of Molothrus bonariensis (Gmelin, 1789) (Passeriformes: Icteridae) from southern Brazil. 1- ventral view of P. ovatus; bar = 0.6 mm; 2- arrows point to the oral suckers and pharynx; bar = 0.25 mm; 3- median area of the body, vitellarias, ovary and testicles; bar = 0.68 mm. 4-6 Tanaisia valida Freitas, 1951 (Trematoda: Eucotylidae) of M. bonariensis. 4- ventral view of T. valida; Arrows point out the beginning of the vitelline glands; bar = 0.39 mm; 5- anterior area, arrows point to the terminal oral sucker and pharynx; bar = 0.08 mm; 6- median area of the body, arrows point to ovary and lobate testes; bar = 0.14 mm.
Tanaisia valida has elongated body, flattened, in tegument with thorns, vitelline glands begging in post-ovarian region to the back of the body (Figure 4), oral sucker subterminal, non visible acetabulum, muscular pharynx (Figure 5), intestinal cecum is positioned dorsally in relation to the gonads fused near to end of the body. Lobate testis with diagonal layout, pre-equatorial, post-ovarian (Figure 6). Lobate ovary, uterus with a large number of eggs, these yellow-brownish color with a smooth shell.
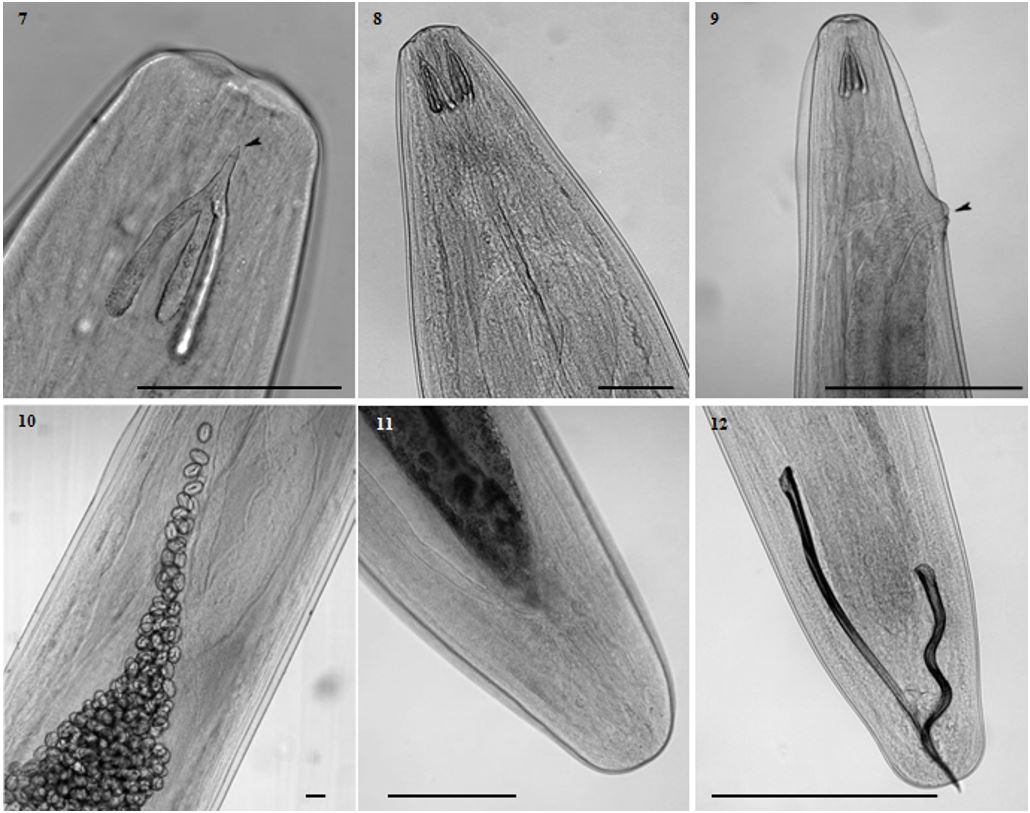
In the anterior region of D. bargusinica has a trident well developed (Figure 7), trident with tapered apex (Figure 8), females has a genital opening near to the anterior region (Figure 9), eggs with size range of 0.044 × 0.029mm (Figure 10), females with posterior rounded region (Figure 11), males with uneven spicules with small number of papillae (Figure 12).
Diplotriaena bargusinica Skrjabin, 1917 (Nematoda: Diplotriaenidae) of Molothrus bonariensis (Gmelin, 1789) (Passeriformes: Icteridae) from southern Brazil. 7- dorsal view, smooth trident highlighted, arrow points to the apex of the tapered trident; bar = 0.14 mm; 8- dorsal view of the anterior area; bar = 0.14 mm; 9- anterior area of the female, arrow points to genital opening; bar = 0.35 mm; 10- posterior area of the male, detail of bigger and smaller spicules; bar = 0.5 mm; 11- posterior area of the female; bar = 0.25 mm; 12- median area of the female, detail of the eggs; bar = 0.05 mm.
Synhimantus (D.) nasuta (Figure 13) has a filiform body, cuticle striated crosswise, small and conic lips, four cords recurrent non anastomosing beginning on surface dorsal and ventral of the oral opening extending to the posterior portion of the muscle esophagus and bifurcated cervical papillae (Figure 14) Females presented well-developed uterine, vulvar opening near the posterior end of the body (Figure 15). Males presented curved caudal end, nine pairs of caudal papillae, four pairs pre-cloacal and five post-cloacal (Figure 16), unequal and dissimilar spicules (Figure 17).
Synhimantus (Dispharynx) nasuta (Rudolphi, 1819) Chabaud, 1975 (Nematoda: Acuariidae) of Molothrus bonariensis (Gmelin, 1789) (Passeriformes: Icteridae) from southern Brazil. 13- recurrent cordons of S. (D.) nasuta, arrow points to the papilla; bar = 0.07 mm; 14- detain of the bifurcated cervical papilla; bar = 0.01 mm; 15- genital opening of the female; bar = 0.03 mm; 16- posterior area of the male, detail of pre and post-cloacal papillae; bar = 0.17 mm; 17- lateral view, arrows point to uneven spicules; BAR = 0.8 mm.
The trematodes Prosthogonimidae (Lühe, 1909) have worldwide distribution. They are parasites of the bursa of Fabricius, oviduct, cloaca or nictitating membrane of birds, in mammals are found in the intestine, liver and body cavity (BRAY et al., 2008Bray RA, Gibson DI, Jones A. Keys to Trematoda. London: CABI International/Natural History Museum; 2008. vol. 3, 824 p.).
In Brazil, P. ovatus were registered in the bursa of Fabricius of Gallus gallus domesticus (Linnaeus, 1758) (Galliformes: Phasianidae) (n = 17) P = 17.6% and in wild birds: Theristicus caerulescens (Vieillot, 1817) (n = 6) P = 16.6%; Mesembrinibis cayennensis (Gmelin, 1789) (n = 3) P = 33.3% (Pelecaniformes: Threskiornithidae); Piaya cayana (Linnaeus, 1766) (Cuculiformes: Cuculidae) (n = 3) P = 33.3%; Monasa nigrifrons (Spix, 1824) (Galbuliformes: Bucconidae) (n = 10) P = 10.0% Cyanocorax cyanomelas (Vieillot, 1818) (Passeriformes: Corvidae) (n = 4) P = 25.0% Xiphorhynchus guttatus (Lichtenstein, 1820) (Passeriformes: Dendrocolaptidae) (n = 1) P = 100% (TRAVASSOS, 1928Travassos L. Fauna helminthologica de Matto Grosso (Trematódeos -1ª parte). Mem Inst Oswaldo Cruz 1928; 21(2): 323-338. http://dx.doi.org/10.1590/S0074-02761928000200002.
http://dx.doi.org/10.1590/S0074-02761928...
; TRAVASSOS et al., 1969Travassos L, Teixeira de Feitas JF, Kohn A. Trematódeos do Brasil. Mem Inst Oswaldo Cruz 1969; 67: 1-886.).
Kohn & Fernandes (1972)Kohn A, Fernandes BMM. Sobre a valiedade das espécies pertencentes ao gênero Luehe, 1899, da coleção helmintológica do Instituto Oswaldo Cruz. ProsthogonimusMem Inst Oswaldo Cruz 1972; 70(3): 309-325. http://dx.doi.org/10.1590/S0074-02761972000300005.
http://dx.doi.org/10.1590/S0074-02761972...
reported the ocurrence of P. ovatus in Pteroglossus aracari (Linnaeus, 1758) (Piciformes: Ramphastidae), Cairina moschata (Linnaeus, 1758) and Cairina moschata domesticus (Anseriformes: Anatidae), Crotophaga major Gmelin, 1788 (Cuculiformes: Cuculidae), Theresticus caerulescens (Vieillot, 1817) (Pelecaniformes: Threskiornithidae), Hydropsalis Wagler, 1832 (Caprimulgiformes: Caprimulgidae), Leptoptila verreauxi Bonaparte, 1855 (Columbiformes: Columbidae), in the Charadriiformes: Thalasseus maximus (Boddaert, 1783) (Sternidae), Gallinago paraguaiae (Vieillot, 1816) (Scolopacidae), Larus Linnaeus, 1758 and Larus dominicanus (Laridae) in Passeriformes: Tangara sayaca (Linnaeus, 1766) and Lanio melanops (Vieillot, 1818) (Thraupidae), Turdus amaurochalinus Cabanis, 1850 (Turdidae), Taraba major Gmelin, 1788 (Thamnophilidae), Cyanocorax chrysops (Vieillot, 1818) (Corvidae), Icterus croconotus (Wagler, 1829) and Sturnella superciliaris (Bonaparte, 1840) (Icteridae).
In Rio Grande do Sul, Monteiro et al. (2007)Monteiro CM, Amato JFR, Amato SB. (Rudolphi) (Digenea, Prosthogonimidae) em três espécies de aves aquáticas da Região Sul do Brasil. Prosthogonimus ovatusRev Bras Zool 2007; 24(1): 253-257. http://dx.doi.org/10.1590/S0101-81752007000100035.
http://dx.doi.org/10.1590/S0101-81752007...
identified P. ovatus in waterbirds, Dendrocygna bicolor (Vieillot, 1816) (n = 33), P = 3.0% and MI = 1; and Netta peposaca (Vieillot, 1816) (n = 20), P = 15.0% and MI = 4.3 (Anseriformes: Anatidae); and in Phalacrocorax brasilianus (Gmelin, 1789) (n = 47), P = 2.1% and MI = 1 (Suliformes: Phalacrocoracidae). Mascarenhas et al. (2009)Mascarenhas CS, Krüger C, Müller G. The helminth fauna of the red-crested cardinal (Paroaria coronata) Passeriformes: Emberizidae in Brazil. Parasitol Res 2009; 105(5): 1359-1363. http://dx.doi.org/10.1007/s00436-009-1569-8. PMid:19636590.
http://dx.doi.org/10.1007/s00436-009-156...
identified it in Paroaria coronata (Miller, 1776) (n = 40), P = 7.5% and MI = 3 (Passeriformes: Thraupidae).
According to Boddeke (1960)Boddeke R. The life history of Rudolphi II. The intermediate hosts. Prosthogonimus ovatusTrop Geogr Med 1960; 12: 363-377. the life cycle of P. ovatus involves two intermediate hosts, the first is a mollusk and the second are young forms of Odonata. The bird infection occurs through ingestion of young or adult dragonflies containing the encysted metacercariae (BODDEKE, 1960Boddeke R. The life history of Rudolphi II. The intermediate hosts. Prosthogonimus ovatusTrop Geogr Med 1960; 12: 363-377.). In laying birds, they affect egg production, through causing a decline in egg formation, or non-formation (OLSEN, 1974Olsen OW. Animal parasites: their life cycles and ecology. Baltimore: University Park Press; 1974. 562 p.).
Tanaisia Skrjabin, 1924, parasitizes the renal tubules and kidneys of birds. In Brazil, it has mainly been reported in Passeriformes, although there have been records of occurrences in Cuculiformes, Charadriiformes, Gruiformes and Strigiformes. The Eucotylidae taxonomy is a complex one due to morphological structures that characterize the genera and species (integument, position and shape of the testis and ovaries, etc.).
Lunaschi et al. (2015)Lunaschi LI, Drago FB, Draghi R. Redescription of Tanaisia dubia (Digenea) from the northeast region of Argentina, with a key to Neotropical species of the genus, and a key to genera of Tanaisiinae. Rev Mex Biodiv 2015; 86(4): 888-895. http://dx.doi.org/10.1016/j.rmb.2015.06.012.
http://dx.doi.org/10.1016/j.rmb.2015.06....
redescribed Tanaisia dubia Freitas, in 1951, had created a key to the Neotropical species Tanaisia and a key to the Tanaisiinae genres that gives support in species identification. Those authors pointed out the extent of vitelline glands as an important morphological characteristic to distinguish three genres of Tanaisiinae, and in the species of Tanaisia the vitelline glands extend up from ovarian or testicular region.
Tanaisia valida Freitas, 1951 was described in Himantopus melanurus Vieillot, 1817 (Charadriiformes: Recurvirostridae) in the state of Rio de Janeiro and, later on, was reported by Mascarenhas et al. (2009)Mascarenhas CS, Krüger C, Müller G. The helminth fauna of the red-crested cardinal (Paroaria coronata) Passeriformes: Emberizidae in Brazil. Parasitol Res 2009; 105(5): 1359-1363. http://dx.doi.org/10.1007/s00436-009-1569-8. PMid:19636590.
http://dx.doi.org/10.1007/s00436-009-156...
, in Paroaria coronata (Miller, 1776) (Passeriformes: Thraupidae) (n = 40), with P = 2.5%, MA = 0.05 and MI = 2, in the state of Rio Grande do Sul.
In South America, other authors have had identified T. valida in Charadriiformes. In Peru, Ibañez-Herrera (1998)Ibañez-Herrera, N. Mención de algunos tremátodos en la fauna helmintológica peruana. Rev Per Parasit 1998; 13: 90-96. recorded T. valida parasitizing kidneys in Himantopus melanurus Vieillot, 1817 (Recurvirostridae) without presenting parasitological and sample size indexes, because it is a list of species for the fauna of Peru, later Tantalean et al. (1992)Tantalean M, Sarmiento LB, Huiza AF. Digeneos (Trematoda) del Perú. Bol Lima 1992; 80: 47-84. recorded in Phalaropus tricolor (Vieillot, 1819) (Scolopacidae) in the same country. Tanaisia valida was identified in Charadrius wilsonia Ord, 1814 (Charadriidae) (n = 1) (P = 100%) (MI = 4) in Colombia (LUNASCHI et al. 2015Lunaschi LI, Drago FB, Draghi R. Redescription of Tanaisia dubia (Digenea) from the northeast region of Argentina, with a key to Neotropical species of the genus, and a key to genera of Tanaisiinae. Rev Mex Biodiv 2015; 86(4): 888-895. http://dx.doi.org/10.1016/j.rmb.2015.06.012.
http://dx.doi.org/10.1016/j.rmb.2015.06....
).
About the life cycle of T. valida does not exist information, however considering the life cycle of Tanaisiinae, the birds become infected by ingesting (Mollusca: Gastropoda) containing metacercariae (LUNASCHI et al. 2015Lunaschi LI, Drago FB, Draghi R. Redescription of Tanaisia dubia (Digenea) from the northeast region of Argentina, with a key to Neotropical species of the genus, and a key to genera of Tanaisiinae. Rev Mex Biodiv 2015; 86(4): 888-895. http://dx.doi.org/10.1016/j.rmb.2015.06.012.
http://dx.doi.org/10.1016/j.rmb.2015.06....
).
Diplotriaena Railliet & Henry, 1909, has parasitic specificity restricted to birds and is distributed among Anseriformes, Apodiformes, Galliformes, Charadriiformes, Columbiformes, Piciformes and Passeriformes, with wide geographic distribution (VICENTE et al., 1983Vicente JJ, Pinto RM, Noronha D. Estudo das espécies brasileiras do gênero Diplotriaena Henry & Ozoux, 1909 (Nematoda, Filarioidea). Mem Inst Oswaldo Cruz 1983; 78(2): 165-182. http://dx.doi.org/10.1590/S0074-02761983000200005.
http://dx.doi.org/10.1590/S0074-02761983...
; ATKINSON et al., 2009Atkinson CT, Thomas NJ, Hunter DB. Parasitic diseases of wild birds. Ames: Wiley-Blackwell; 2009. 592 p.).
In Brazil D. bargusinica was reported by Vicente et al. (1983)Vicente JJ, Pinto RM, Noronha D. Estudo das espécies brasileiras do gênero Diplotriaena Henry & Ozoux, 1909 (Nematoda, Filarioidea). Mem Inst Oswaldo Cruz 1983; 78(2): 165-182. http://dx.doi.org/10.1590/S0074-02761983000200005.
http://dx.doi.org/10.1590/S0074-02761983...
parasitizing some Passeriformes in different areas of the country: Cacicus cela (Linnaeus, 1758), Cacicus haemorrhous (Linnaeus, 1766), Gnorimopsar chopi (Vieillot, 1819), Icterus croconotus (Wagler, 1829), Icterus sp., Psarocolius decumanus maculosus (Chapman, 1920), Molothrus bonariensis (Gmelin, 1789) (Icteridae) and Xiphocolaptes major castaneus Ridgway, 1890 (Dendrocolaptidae), in the states of Mato Grosso do Sul, São Paulo and Pará and in Paraguay, without informing the number of birds examined or their indexes.
Pinto et al. (1997)Pinto RM, Vicente JJ, Noronha D. Nematode Parasites of Brazilian Corvid Birds (Passeriformes): a general survey with a description of Viktorocara brasiliensis n. sp. (Acuariidae, Schistorophinae). Mem Inst Oswaldo Cruz 1997; 92(2): 209-214. http://dx.doi.org/10.1590/S0074-02761997000200013.
http://dx.doi.org/10.1590/S0074-02761997...
reported the presence of D. bargusinica for the first time in Cyanocorax cyanomelas (Vieillot, 1818) (Corvidae) in the state of Mato Grosso do Sul, from samples collected between 1921 and 1925 in scientific expeditions conducted in Brazil, without informing the number of birds examined or their indexes. Gonçalves et al. (2002)Gonçalves AQ, Vicente JJ, Pinto RM. Nematodes of Amazonian vertebrates deposited in the Helminthological Collection of the Oswaldo Cruz Institute with new records. Rev Bras Zool 2002; 19(2): 453-465. http://dx.doi.org/10.1590/S0101-81752002000200011.
http://dx.doi.org/10.1590/S0101-81752002...
found D. bargusinica in the body cavity of three individuals of Psarocolius bifasciatus (Spix, 1824) (Icteridae) in the state of Amazonas and Carvalho et al. (2007)Carvalho AR, Daemon E, Souza-Lima S. Relação entre o peso do baço e infecção por helmintos em galo de campina Paroaria dominicana (Linnaeus, 1758) (Passeriformes, Emberizidae) do estado da Bahia, Brasil. Rev Bras Zooc 2007; 9(2): 219-224. in air sacs of Paroaria dominicana (Linnaeus, 1758) (Emberizidae) (n = 34), P = 8.8%, MA = 0.32 and MI = 3.66, in the state of Bahia.
Anderson (1962)Anderson RC. On the development, morphology, and experimental transmission of (Filaroidea: Diplotriaenidae). Diplotriaena bargusinicaCan J Zool 1962; 40(7): 1175-1186. http://dx.doi.org/10.1139/z62-093.
http://dx.doi.org/10.1139/z62-093...
, performed experimental infection with D. bargusinica in wild birds (Turdidae and Icteridae) and described details of its biological cycle which uses grasshoppers (Orthoptera) as intermediate hosts. Birds become infected by ingesting grasshoppers that contain third-stage larvae, and the adult nematodes become established in the air sacs of the host (ANDERSON, 2000Anderson RC. Nematode parasites of vertebrates: their development and transmission. 2nd ed. London: CABI International; 2000. 650 p.). The clinical signs common to parasitized birds are lethargy, labored breathing, size and weight below average and little plumage (ATKINSON et al., 2009Atkinson CT, Thomas NJ, Hunter DB. Parasitic diseases of wild birds. Ames: Wiley-Blackwell; 2009. 592 p.).
Synhimantus (D.) nasuta has worldwide distribution and parasitizes the proventriculus and gizzard of birds. In Brazil, it has been found in Galliformes of economic importance, and also in Columbiformes, Cuculiformes, Strigiformes, Falconiformes, Charadriiformes and Passeriformes (BARTMANN & AMATO, 2009Bartmann A, Amato SB. Dispharynx nasuta (Nematoda: Acuariidae) em Guira guira e (Cuculiformes: Cuculidae) no Estado do Rio Grande do Sul, Brasil. Crotophaga aniCienc Rural 2009; 39(4): 1152-1158. http://dx.doi.org/10.1590/S0103-84782009005000059.
http://dx.doi.org/10.1590/S0103-84782009...
).
In Rio Grande do Sul, S. (D.) nasuta was reported in Paroaria coronata (Miller, 1776) (Passeriformes: Thraupidae) (n = 40), P = 2.5%, MA = 0.1 and MI = 4, by Mascarenhas et al., 2009; in Guira guira (Gmelin, 1788) (n = 120), P = 28.3%, MA = 2.28 and MI = 8.8; in Crotophaga ani Linnaeus, 1758 (Cuculiformes: Cuculidae) (n = 120), P = 26.7%, MA = 1.32 and MI = 5.1, by Bartmann & Amato (2009)Bartmann A, Amato SB. Dispharynx nasuta (Nematoda: Acuariidae) em Guira guira e (Cuculiformes: Cuculidae) no Estado do Rio Grande do Sul, Brasil. Crotophaga aniCienc Rural 2009; 39(4): 1152-1158. http://dx.doi.org/10.1590/S0103-84782009005000059.
http://dx.doi.org/10.1590/S0103-84782009...
; in Columbina picui (Columbiformes: Columbidae) (n = 34), P = 5.9%, MA = 1.1 and MI = 19.5, by Coimbra et al. (2009)Coimbra MAA, Mascarenhas CS, Krüger C, Muller G. Helminths parasitizing (Columbiformes: Columbidae) in Brazil. Columbina picuiJ Parasitol 2009; 95(4): 1011-1012. http://dx.doi.org/10.1645/GE-1948.1. PMid:20050007.
http://dx.doi.org/10.1645/GE-1948.1...
; in Vanellus chilensis (Molina, 1782) (Charadriiformes: Charadriidae) (n = 28), P = 28.6%, MA = 4.11 and MI = 14.38, by Avancini (2009)Avancini LF. Helmintos e artrópodes de Vanellus chilensis (Molina, 1782) quero-quero, da região sul do Rio Grande do Sul [Dissertação]. Pelotas: Universidade Federal de Pelotas; 2009.; in Passer domesticus (Linnaeus, 1758) (Passeriformes: Passeridae) (n = 160), P = 0.63%, MA = 0.11 and MI = 18, by Calegaro-Marques & Amato (2010)Calegaro-Marques C, Amato SB. Helminths of introduced house sparrows (Passer domesticusIheringia) in Brazil: does population age affect parasite richness? . Série Zool 2010; 100(1): 73-78.; and in Pitangus sulphuratus (Passeriformes: Tyrannidae) (n = 78), P = 3.85%, MA = 0.10 and MI = 2.66, by Mendes (2011)Mendes MM. Helmintos e ácaros nasais parasitos de Pitangus sulphuratus (Passeriformes: Tyrannidae), bem-te-vi, no Rio Grande do Sul [Dissertação]. Pelotas: Universidade Federal de Pelotas; 2011..
The biological cycle of S. (D.) nasuta is heteroxenous, using terrestrial isopods, orthopterans, beetles or millipedes (GOBLE & KUTZ, 1945Goble FC, Kutz HL. The genus (Nematoda: Acuariidae) in Galliform and Passariform birds. DispharynxJ Parasitol 1945; 31(5): 323-331. http://dx.doi.org/10.2307/3273088.
http://dx.doi.org/10.2307/3273088...
). The larvae develop in non-specific tissues of isopods, in which they become encapsulated and are later on ingested by birds (ANDERSON, 2000Anderson RC. Nematode parasites of vertebrates: their development and transmission. 2nd ed. London: CABI International; 2000. 650 p.). The effects on the definitive host are related to the parasitic load, usually comprising inflammations, granulomas and lesions in the proventriculus. In wild birds, these macroscopic and histopathological alterations were described by Bartmann & Amato (2009)Bartmann A, Amato SB. Dispharynx nasuta (Nematoda: Acuariidae) em Guira guira e (Cuculiformes: Cuculidae) no Estado do Rio Grande do Sul, Brasil. Crotophaga aniCienc Rural 2009; 39(4): 1152-1158. http://dx.doi.org/10.1590/S0103-84782009005000059.
http://dx.doi.org/10.1590/S0103-84782009...
.
In general, the parasitological indexes of M. bonariensis are similar to those found by those previously mentioned authors, considering that the previous studies had sample sizes close to the one of the present study future research is needed to complement existing information in order to contribute to the understanding of the biology and ecology of these organisms.
In Brazil, for the first time the species T. valida, P. ovatus e S. (D.) nasuta are recorded parasitizing M. bonariensis. Prosthogonimus ovatus, T. valida, D. bargusinica e S. (D.) nasuta characterize first record in M. bonariensis in Rio Grande do Sul. Whereas the helminthological information for the specie are scarce, this work expands the knowledge to Icteridae and Molothrus bonariensis.
Acknowledgements
The authors thank the contributions of the reviewers of the Brazilian Journal of Veterinary Parasitology, at the Coordenação de Aperfeiçoamento do Pessoal de Nível Superior (CAPES) for the doctoral scholarship of the first author, for the financial support provided through announcement no. 2010/032 and Granjas 4 Irmãos S.A. for its assistance in collecting the birds on its property.
References
- Amato JFR, Amato SB. Técnicas gerais para coleta e preparação de helmintos endoparasitos de aves. In: Von Matter S, Straube FC, Accordi I, Piacentini V, Cândido JF Jr. Ornitologia e conservação: ciência aplicada, técnicas de pesquisa e levantamento. Rio de Janeiro: Technical Books; 2010. p. 369-393.
- Anderson RC, Chabaud AG, Willmott S. Keys to the Nematode parasites of vertebrates: archival volume. London: CABI International; 2009. 463 p.
- Anderson RC. Nematode parasites of vertebrates: their development and transmission. 2nd ed. London: CABI International; 2000. 650 p.
- Anderson RC. On the development, morphology, and experimental transmission of (Filaroidea: Diplotriaenidae). Diplotriaena bargusinicaCan J Zool 1962; 40(7): 1175-1186. http://dx.doi.org/10.1139/z62-093
» http://dx.doi.org/10.1139/z62-093 - Astié AA, Reboreda JC. Costs of egg punctures and parasitism by shiny cowbirds () at creamy-bellied thrush (Molothrus bonariensisTurdus amaurochalinus) nests. Auk 2006; 123(1): 23-32. http://dx.doi.org/10.1642/0004-8038(2006)123[0023:COEPAP]2.0.CO;2
» http://dx.doi.org/10.1642/0004-8038(2006)123[0023:COEPAP]2.0.CO;2 - Atkinson CT, Thomas NJ, Hunter DB. Parasitic diseases of wild birds. Ames: Wiley-Blackwell; 2009. 592 p.
- Avancini LF. Helmintos e artrópodes de Vanellus chilensis (Molina, 1782) quero-quero, da região sul do Rio Grande do Sul [Dissertação]. Pelotas: Universidade Federal de Pelotas; 2009.
- Bartmann A, Amato SB. Dispharynx nasuta (Nematoda: Acuariidae) em Guira guira e (Cuculiformes: Cuculidae) no Estado do Rio Grande do Sul, Brasil. Crotophaga aniCienc Rural 2009; 39(4): 1152-1158. http://dx.doi.org/10.1590/S0103-84782009005000059
» http://dx.doi.org/10.1590/S0103-84782009005000059 - Belton W. Aves do Rio Grande do Sul: distribuição e biologia. São Leopoldo: UNISINOS; 1994. 548 p.
- Blanco DE. Parasitismo de cria del Tordo Renegrido Molothrus bonariensis sobre , en el este de la província de Buenos Aires. Agelaius ruficapillusEl Hornero 1995; 14(1-2): 44-45.
- Boddeke R. The life history of Rudolphi II. The intermediate hosts. Prosthogonimus ovatusTrop Geogr Med 1960; 12: 363-377.
- Bray RA, Gibson DI, Jones A. Keys to Trematoda. London: CABI International/Natural History Museum; 2008. vol. 3, 824 p.
- Bush AO, Lafferty KD, Lotz JM, Shostak AW. Parasitology meets ecology on its own terms: Margolis et al. revisited. J Parasitol 1997; 83(4): 575-583. http://dx.doi.org/10.2307/3284227 PMid:9267395.
» http://dx.doi.org/10.2307/3284227 - Calegaro-Marques C, Amato SB. Helminths of introduced house sparrows (Passer domesticusIheringia) in Brazil: does population age affect parasite richness? . Série Zool 2010; 100(1): 73-78.
- Carvalho AR, Daemon E, Souza-Lima S. Relação entre o peso do baço e infecção por helmintos em galo de campina Paroaria dominicana (Linnaeus, 1758) (Passeriformes, Emberizidae) do estado da Bahia, Brasil. Rev Bras Zooc 2007; 9(2): 219-224.
- Coimbra MAA, Mascarenhas CS, Krüger C, Muller G. Helminths parasitizing (Columbiformes: Columbidae) in Brazil. Columbina picuiJ Parasitol 2009; 95(4): 1011-1012. http://dx.doi.org/10.1645/GE-1948.1 PMid:20050007.
» http://dx.doi.org/10.1645/GE-1948.1 - Efe MA, Mohr LV, Bugoni L. Guia ilustrado das aves dos parques de Porto Alegre. Porto Alegre: PROAVES/SMAM/COPESUL/CEMAVE; 2001. 144 p.
- Freitas JFT. Revisão da família Eucotylidae Skrjabin, 1924 (Trematoda). Mem Inst Oswaldo Cruz 1951; 49: 33-123. http://dx.doi.org/10.1590/S0074-02761951000100003 PMid:14890534.
» http://dx.doi.org/10.1590/S0074-02761951000100003 - Gibson DI, Jones A, Bray RA. Keys to the Trematoda. London: CABI International/The Natural History Museum; 2002. vol. 1, 521 p.
- Goble FC, Kutz HL. The genus (Nematoda: Acuariidae) in Galliform and Passariform birds. DispharynxJ Parasitol 1945; 31(5): 323-331. http://dx.doi.org/10.2307/3273088
» http://dx.doi.org/10.2307/3273088 - Goméz A, Nichols E. Neglected wild life: Parasitic biodiversity as a conservation target. Int J Parasito.Parasit Wildl 2013; 2: 222-227. http://dx.doi.org/10.1016/j.ijppaw.2013.07.002
» http://dx.doi.org/10.1016/j.ijppaw.2013.07.002 - Gonçalves AQ, Vicente JJ, Pinto RM. Nematodes of Amazonian vertebrates deposited in the Helminthological Collection of the Oswaldo Cruz Institute with new records. Rev Bras Zool 2002; 19(2): 453-465. http://dx.doi.org/10.1590/S0101-81752002000200011
» http://dx.doi.org/10.1590/S0101-81752002000200011 - Hoberg EP, Kutz SJ. Parasites. In: Bültmann H, Ims RA, Fridriksson F, Hoberg EP, Meltofte H, Ganter B, Josefson A. Artic biodiversity assessment: status and trends in Artic biodiversity [online]. Akureyri: Conservation of Arctic Flora and Fauna; 2013 [cited 2015 Sep 02]. Available from: http://www.arcticbiodiversity.is/the-report/chapters/parasites
» http://www.arcticbiodiversity.is/the-report/chapters/parasites - Ibañez-Herrera, N. Mención de algunos tremátodos en la fauna helmintológica peruana. Rev Per Parasit 1998; 13: 90-96.
- Kohn A, Fernandes BMM. Sobre a valiedade das espécies pertencentes ao gênero Luehe, 1899, da coleção helmintológica do Instituto Oswaldo Cruz. ProsthogonimusMem Inst Oswaldo Cruz 1972; 70(3): 309-325. http://dx.doi.org/10.1590/S0074-02761972000300005
» http://dx.doi.org/10.1590/S0074-02761972000300005 - Lunaschi LI, Drago FB, Draghi R. Redescription of Tanaisia dubia (Digenea) from the northeast region of Argentina, with a key to Neotropical species of the genus, and a key to genera of Tanaisiinae. Rev Mex Biodiv 2015; 86(4): 888-895. http://dx.doi.org/10.1016/j.rmb.2015.06.012
» http://dx.doi.org/10.1016/j.rmb.2015.06.012 - Mascarenhas CS, Krüger C, Müller G. The helminth fauna of the red-crested cardinal (Paroaria coronata) Passeriformes: Emberizidae in Brazil. Parasitol Res 2009; 105(5): 1359-1363. http://dx.doi.org/10.1007/s00436-009-1569-8 PMid:19636590.
» http://dx.doi.org/10.1007/s00436-009-1569-8 - Mendes MM. Helmintos e ácaros nasais parasitos de Pitangus sulphuratus (Passeriformes: Tyrannidae), bem-te-vi, no Rio Grande do Sul [Dissertação]. Pelotas: Universidade Federal de Pelotas; 2011.
- Mermoz ME, Reboreda JC. Reproductive sucess of shiny cowbird () parasitizing the larger brown-and-yellow marshbird (Molothrus bonariensisPseudoleistes virescens) in Argentina. Auk 2003; 120(4): 1128-1139. http://dx.doi.org/10.1642/0004-8038(2003)120[1128:RSOSCM]2.0.CO;2
» http://dx.doi.org/10.1642/0004-8038(2003)120[1128:RSOSCM]2.0.CO;2 - Monteiro CM, Amato JFR, Amato SB. (Rudolphi) (Digenea, Prosthogonimidae) em três espécies de aves aquáticas da Região Sul do Brasil. Prosthogonimus ovatusRev Bras Zool 2007; 24(1): 253-257. http://dx.doi.org/10.1590/S0101-81752007000100035
» http://dx.doi.org/10.1590/S0101-81752007000100035 - Olsen OW. Animal parasites: their life cycles and ecology. Baltimore: University Park Press; 1974. 562 p.
- Petrochenko VI. Acanthocephala of domestic and wild animals. Jerusalém: Israel Program for Scientific Translations; 1971. vol. 2, 478 p.
- Pinto RM, Vicente JJ, Noronha D. Nematode Parasites of Brazilian Corvid Birds (Passeriformes): a general survey with a description of Viktorocara brasiliensis n. sp. (Acuariidae, Schistorophinae). Mem Inst Oswaldo Cruz 1997; 92(2): 209-214. http://dx.doi.org/10.1590/S0074-02761997000200013
» http://dx.doi.org/10.1590/S0074-02761997000200013 - Poulin R, Morand S. The diversity of parasites. Q Rev Biol 2000; 75(3): 277-293. http://dx.doi.org/10.1086/393500 PMid:11008700.
» http://dx.doi.org/10.1086/393500 - Sick H. Ornitologia brasileira. Rio de Janeiro: Editora Nova Fronteira; 1997. 862 p.
- Tantalean M, Sarmiento LB, Huiza AF. Digeneos (Trematoda) del Perú. Bol Lima 1992; 80: 47-84.
- The IUCN Red List of Threatened Species [online]. Gland: International Union for Conservation of Nature and Natural Resources; 2015 [cited 2015 Aug 20]. Available from: http://www.iucnredlist.org
» http://www.iucnredlist.org - Travassos L, Teixeira de Feitas JF, Kohn A. Trematódeos do Brasil. Mem Inst Oswaldo Cruz 1969; 67: 1-886.
- Travassos L. Fauna helminthologica de Matto Grosso (Trematódeos -1ª parte). Mem Inst Oswaldo Cruz 1928; 21(2): 323-338. http://dx.doi.org/10.1590/S0074-02761928000200002
» http://dx.doi.org/10.1590/S0074-02761928000200002 - Vicente JJ, Pinto RM, Noronha D. Estudo das espécies brasileiras do gênero Diplotriaena Henry & Ozoux, 1909 (Nematoda, Filarioidea). Mem Inst Oswaldo Cruz 1983; 78(2): 165-182. http://dx.doi.org/10.1590/S0074-02761983000200005
» http://dx.doi.org/10.1590/S0074-02761983000200005 - Zhang L, Brooks DR, Causey D. Two species of Synhimantus (Dispharynx) Railliet, Henry and Sisoff, 1912 (Nematoda: Acuarioidea: Acuariidae) in passerine birds from the area the Conservación Guanacaste, Costa Rica. J Parasitol 2004; 90(5): 1133-1138. http://dx.doi.org/10.1645/GE-3253 PMid:15562615.
» http://dx.doi.org/10.1645/GE-3253
Publication Dates
-
Publication in this collection
25 Aug 2016 -
Date of issue
Jul-Sep 2016
History
-
Received
15 Oct 2015 -
Accepted
11 May 2016