Abstract
The occurrence and intensity of helminth infections were evaluated in sheep from pastures shared with cattle. In 2015 and 2016, young male sheep acquired in Santana do Livramento, Rio Grande do Sul, Brazil, were finished in integrated crop-livestock system. We selected the 12 sheep that showed the highest number of nematode eggs per gram of faeces to search for worms in the gastrointestinal tract. Haemonchus contortus and Trichostrongylus colubriformis were the major parasites. H. contortus presented mean intensities of 1,159 and 257 worms in 2015 and 2016, respectively. T. colubriformis displayed mean intensities of 4,149 and 2,427 worms in 2015 and 2016, respectively. Of the 127 male specimens of Haemonchus spp. analysed by Polymerase Chain Reaction (PCR), 125 were H. contortus, one Haemonchus placei and one hybrid. Other species detected were Cooperia punctata , Cooperia pectinata, Cooperia spatulata, Cooperia curticei, Ostertagia ostertagi, Teladorsagia circumcincta , Trichostrongylus axei, Nematodirus spathiger , and Trichuris ovis. Twenty lambs presented cysts of Taenia hydatigena in the liver and mesentery. One lamb presented Coenurus cerebralis, the larval stage of Taenia multiceps, in the brain. In conclusion, sheep from pasture shared with cattle presented a high diversity of nematode species. H. contortus and H. placei co-infection occur with consequent hybridization.
Keywords:
Cooperia; Nematodirus; Ostertagia; Taenia hydatigena; Teladorsagia; Trichostrongylus
Resumo
A ocorrência de infecções helmínticas foi avaliada em ovinos que compartilhavam pastagem com bovinos. Em 2015 e em 2016, cordeiros machos foram adquiridos em Santana do Livramento, Rio Grande do Sul, para serem terminados em sistema de lavoura – pecuária (ILP). Em cada ano, 12 cordeiros que tinham maior contagem de ovos nas fezes foram abatidos para recuperação dos vermes. Haemonchus contortus e Trichostrongylus colubriformis foram as principais espécies registradas. H. contortus apresentou intensidade média de 1159 e 257 vermes em 2015 e 2016, respectivamente. T. colubriformis apresentou intensidade média de 4149 e 2427 parasitas em 2015 e 2016, respectivamente. De 127 machos de Haemonchus spp. analisados por “Polymerase Chain Reaction” (PCR), 125 foram identificados como H. contortus, um como Haemonchus placei e um como híbrido. Cooperia punctata, Cooperia pectinata, Cooperia spatulata, Cooperia curticei, Ostertagia ostertagi, Teladorsagia circumcincta , Trichostrongylus axei, Nematodirus spathiger e Trichuris ovis foram as outras espécies de nematódeos registradas. Vinte cordeiros apresentaram cistos de Taenia hydatigena no mesentério e no fígado. Um cordeiro apresentou no cérebro Coenurus cerebralis, o estágio larval de Taenia multiceps . Em conclusão, ovinos criados com bovinos apresentam grande diversidade de nematódeos. A co-infecção de H. contortus e H. placei favorece a produção de híbridos.
Palavras-chave:
Cooperia; Nematodirus; Ostertagia; Taenia hydatigena; Teladorsagia; Trichostrongylus
Introduction
Rio Grande do Sul State (RS) has the largest sheep population in Brazil, with an estimated 3,957,275 head ( IBGE, 2015 Instituto Brasileiro de Geografia e Estatística – IBGE. Tabela 3939: efetivo dos rebanhos, por tipo de rebanho [online]. Rio de Janeiro; 2015 [cited 2017 June 10]. Available from: https://sidra.ibge.gov.br/tabela/3939#resultado
https://sidra.ibge.gov.br/tabela/3939#r...
). In this State, most sheep are raised by continuous grazing on grassland vegetation (native pasture) that is usually shared with cattle in the biome of the Pampa, located in the southern half of RS. Southern Brazil occupies a transitional zone between tropical and temperate climates, with hot summers, cool winters, and no dry season. The southern half of RS and adjacent areas of Uruguay and Argentina have annual precipitation in the range of 1200-1600 mm and mean annual temperatures of 13-17 °C. Grass-dominated vegetation types prevail, with many herb, shrub, and tree let species co-occurring within the grass matrix ( OVERBECK et al., 2007 Overbeck GE, Müller SC, Fidelis A, Pfadenhauer J, Pillar VD, Blanco CC, et al. Brazil’s neglected biome: The South Brazilian Campos. Perspect Plant Ecol Evol Syst 2007; 9(2): 101-116. http://dx.doi.org/10.1016/j.ppees.2007.07.005.
http://dx.doi.org/10.1016/j.ppees.2007....
). In this environment, sheep production faces gastrointestinal nematode (GIN) infections, which are a significant cause of productivity losses and mortality. This problem has become worse due to the widespread emergence of nematode populations with resistance to anthelmintics ( ECHEVARRIA et al., 1996 Echevarria F, Borba MFS, Pinheiro AC, Waller PJ, Hansen JW. The prevalence of anthelmintic resistance in nematode parasites of sheep in Southern Latin America: Brazil. Vet Parasitol 1996; 62(3-4): 199-206. http://dx.doi.org/10.1016/0304-4017(95)00906-X. PMid:8686165.
http://dx.doi.org/10.1016/0304-4017(95)...
).
Environmental conditions greatly influence the distribution of different nematode species and the risk of massive infections. Santiago et al. (1975) Santiago MAM, Costa UC, Benevenga SF. Estudo comparativo da prevalência de helmintos em ovinos e bovinos criados na mesma pastagem. Pesqui Agropecu Bras 1975; 10(8): 51-56. published the first report on the prevalence of helminths in sheep and cattle sharing the same pasture in RS, reporting that Trichostrongylus colubriformis, Teladorsagia circumcincta, Oesophagostomum columbianum, Oesophagostomum venulosum and Nematodirus filicollis were specifically found in sheep, while Cooperia spp. infected both sheep and cattle. Later, in a detailed study of GIN epizootiology in sheep in Itaqui, RS, Santiago et al. (1976) Santiago MAM, Benevenga SF, Costa UC. Epidemiologia e controle da helmintose ovina no município de Itaqui, Rio Grande do Sul. Pesqui Agropecu Bras 1976; 11(9): 1-7. reported nine species of GINs. Among them, Haemonchus contortus, Trichostrongylus axei, Trichostrongylus colubriformis, and Oesophagostomum columbianum were the main causes of economic losses in sheep in that region. Regarding Haemonchus species, Santiago (1968) Santiago MAM. Haemonchus Cobb, 1898 (Nematoda: Trichostrongylidae) contribuição ao estudo da morfologia, biologia e distribuição geográfica das espécies parasitas de ovinos e bovinos, no Rio Grande do Sul [thesis]. Santa Maria: Universidade Federal de Santa Maria; 1968. postulated that natural cross-infection of cattle and sheep with Haemonchus spp. in RS was negligible. Studies in São Paulo State demonstrated that cattle were the preferential host of Haemonchus similis and Haemonchus placei and that sheep were the preferential host of Haemonchus contortus ( AMARANTE et al., 1997 Amarante AFT, Bagnola J Jr, Amarante MRV, Barbosa MA. Host specificity of sheep and cattle nematodes in São Paulo State, Brazil. Vet Parasitol 1997; 73(1-2): 89-104. http://dx.doi.org/10.1016/S0304-4017(97)00036-8. PMid:9477496.
http://dx.doi.org/10.1016/S0304-4017(97...
; SILVA et al., 2015 Silva MRL, Amarante MRV, Bresciani KDS, Amarante AFT. Host-specificity and morphometrics of female Haemonchus contortus, H. placei and H. similis (Nematoda: Trichostrongylidae) in cattle and sheep from shared pastures in São Paulo State, Brazil. J Helminthol 2015; 89(3): 302-306. http://dx.doi.org/10.1017/S0022149X14000078. PMid:24589375.
http://dx.doi.org/10.1017/S0022149X1400...
).
Most studies on GIN epidemiology were performed decades ago, before the development of molecular techniques that can be used to distinguish nematode species. In studies that aim to evaluate the prevalence and intensity of infection, correct identification of parasite species is essential. The use of molecular tools is particularly important in Haemonchus spp. diagnosis because morphological measurements of spicules may overlap, raising issues about the precise differentiation among species ( SANTOS et al., 2014 Santos MC, Amarante MRV, Silva MRL, Amarante AFT. Differentiation of Haemonchus placei from Haemonchus contortus by PCR and by morphometrics of adult parasites and third stage larvae. Rev Bras Parasitol Vet 2014; 23(4): 495-500. http://dx.doi.org/10.1590/S1984-29612014085. PMid:25517528.
http://dx.doi.org/10.1590/S1984-2961201...
, 2017 Santos MC, Redman E, Amarante MRV, Gilleard JS, Amarante AFT. A panel of microsatellite markers to discriminate and study interactions between Haemonchus contortus and Haemonchus placei. Vet Parasitol 2017; 244: 71-75. http://dx.doi.org/10.1016/j.vetpar.2017.07.011. PMid:28917321.
http://dx.doi.org/10.1016/j.vetpar.2017...
). In addition, co-infection with H. contortus and H. placei can result in hybrids ( HUSSAIN et al., 2014 Hussain T, Periasamy K, Nadeem A, Ellahi Babar M, Pichler R, Diallo A. Sympatric species distribution, genetic diversity and population structure of Haemonchus isolates from domestic ruminants in Pakistan. Vet Parasitol 2014; 206(3-4): 188-199. http://dx.doi.org/10.1016/j.vetpar.2014.10.026. PMid:25468018.
http://dx.doi.org/10.1016/j.vetpar.2014...
; AMARANTE et al., 2017 Amarante MRV, Santos MC, Bassetto CC, Amarante AFT. PCR primers for straightforward differentiation of Haemonchus contortus, Haemonchus placei and their hybrids. J Helminthol 2017; 91(6): 757-761. http://dx.doi.org/10.1017/S0022149X16000882. PMid:28069083.
http://dx.doi.org/10.1017/S0022149X1600...
). For this reason, the accurate identification of both species and hybrids is imperative in epidemiological studies where the species are sympatric, especially when small ruminants and cattle share the same pastures ( AMARANTE, 2011 Amarante AFT. Why is it important to correctly identify Haemonchus species? Rev Bras Parasitol Vet 2011; 20(4): 263-268. http://dx.doi.org/10.1590/S1984-29612011000400002. PMid:22166378.
http://dx.doi.org/10.1590/S1984-2961201...
). Therefore, the aim of this study was to evaluate the occurrence and intensity of helminth infection in sheep from pastures shared with cattle in RS, with an emphasis on the identification of Haemonchus spp.
Materials and Methods
This study was conducted in accordance with the Ethics Committee on Animal Use (CEUA) of São Paulo State University (UNESP), Instituto de Biociências, Botucatu, São Paulo, Brazil (protocol number 716-CEUA).
In 2015 and 2016, young sheep were purchased in Santana do Livramento, which is located in the southern half of RS State, to be finished in an integrated crop-livestock system (ICL) in Botucatu, São Paulo State, Brazil, where they rotationally grazed paddocks for a total of 72 days. At the farm of origin lambs grazed simultaneously with cattle (Bos taurus ) of different ages.
They were allocated in a clean pasture that had been used in an ICL system in previous years. Before introduction of the experimental sheep, the area was left without animals for 300 days, for a period of crop production followed by pasture seeding and growth. This resulted in a clean pasture that was free of contamination by the free-living stages of GIN parasites. Decontamination of the area was confirmed by the use of worm-free tracer lambs that grazed the pasture and remained free of helminth infection (data not publish). We then analysed the worm population acquired by the sheep on their farm of origin in Santana do Livramento.
Experimental animals
In the first year (2015), we used 64 Poll Dorset x Corriedale (crossbred) uncastrated male sheep of 8 months of age on average and with a mean body weight of 24.4 ± 3.4 kg. In the second year (2016), we used 48 Texel x Corriedale (crossbred) male young sheep of the same age as the animals from the previous year and with a body weight of 26.4 ± 3.5 kg.
At the end of 72 days of grazing, we selected in each year the 12 sheep with the highest numbers of eggs per gram of faeces (EPG) based on the average of six EPG counts of faecal samples taken every two weeks. The selected lambs were slaughtered to count and characterize the worms in the gastrointestinal tract. At necropsy, the abomasum and small and large intestines were removed and opened and the contents were placed in graduated buckets. A 10% aliquot was frozen at -18 °C for subsequent enumeration and identification of the helminths ( UENO & GONÇALVES, 1998 Ueno H, Gonçalves PC. Manual para diagnóstico das helmintoses de ruminantes. 4th ed. Tokyo: Japan International Cooperation Agency; 1998. ). The small intestine was digested in saline solution (0.85% NaCl) for 4 h at 37 °C, and a 10% sample of the digested material was collected and frozen.
All internal organs were examined to search for cysts of Taenia hydatigena and Echinococcus granulosus, and the brain was examined to search for cysts of Taenia multiceps.
Nematodes were counted and sexed, and 20 male specimens of each genus were examined per lamb (or all males when fewer than 20 specimens were recovered) for the identification of species. Identification down to the species level of Cooperia, Trichostrongylus , Nematodirus, and Trichuris was based on the morphology of the spicules of male specimens ( LEVINE, 1978 Levine ND. Textbook of veterinary parasitology. Minneapolis: Burgess Publishing Company; 1978. ; LICHTENFELS & PILITT, 1983 Lichtenfels JR, Pilitt PA. Cuticular ridge patterns of Nematodirus (Nematoda: Trichostrongyloidea) parasitic in domestic ruminants of North America, with a key to species. Proc Helminthol Soc Wash 1983; 50(2): 261-274. ; VICENTE et al., 1997 Vicente JJ, Rodrigues HO, Gomes DC, Pinto RM. Nematóides do Brasil. Parte V: nematóides de mamíferos. Rev Bras Zool 1997;14(Suppl 1): 1-452. http://dx.doi.org/10.1590/S0101-81751997000500001.
http://dx.doi.org/10.1590/S0101-8175199...
; UENO & GONÇALVES, 1998 Ueno H, Gonçalves PC. Manual para diagnóstico das helmintoses de ruminantes. 4th ed. Tokyo: Japan International Cooperation Agency; 1998. ). Identification of Ostertagia spp. and Teladorsagia spp. was conducted according to Lichtenfels & Hoberg (1993) Lichtenfels JR, Hoberg EP. The systematics of nematodes that cause ostertagiasis in domestic and wild ruminants in North America: an update and key to species. Vet Parasitol 1993; 46(1-4): 33-53. http://dx.doi.org/10.1016/0304-4017(93)90046-P. PMid:8484224.
http://dx.doi.org/10.1016/0304-4017(93)...
. Ostertagia ostertagi and Ostertagia lyrata were considered as two morphotypes of a single species ( LICHTENFELS et al., 1988 Lichtenfels JR, Pilitt PA, Lancaster MB. Cuticular ridge patterns of seven species of Ostertagiinae (Nematoda) parasitic in domestic ruminants. Proc Helminthol Soc Wash 1988; 55(1): 77-86. ; WYROBISZ et al., 2016 Wyrobisz A, Kowal J, Nosal P. Insight into species diversity of the Trichostrongylidae Leiper, 1912 (Nematoda: Strongylida) in ruminants. J Helminthol 2016; 90(6): 639-646. http://dx.doi.org/10.1017/S0022149X15001017. PMid:26643224.
http://dx.doi.org/10.1017/S0022149X1500...
).
Measurements of the spicules and barbs of Haemonchus spp. were used to calculate a discriminate function (DF) using the formula (DF = 0.0016TL + 0.128THr + 0.152THl − 9.97) described by Jacquiet et al. (1996) Jacquiet P, Cabaret J, Cheikh D, Thiam E. Identification of Haemonchus species in domestic ruminants based on morphometrics of spicules. Parasitol Res 1996; 83(1): 82-86. http://dx.doi.org/10.1007/s004360050213. PMid:9000240.
http://dx.doi.org/10.1007/s004360050213...
and Achi et al. (2003) Achi YL, Zinsstag J, Yao K, Yeo N, Dorchies P, Jacquiet P. Host specificity of Haemonchus spp. for domestic ruminants in the savanna in northern Ivory Coast. Vet Parasitol 2003; 116(2): 151-158. http://dx.doi.org/10.1016/S0304-4017(03)00258-9. PMid:14519319.
http://dx.doi.org/10.1016/S0304-4017(03...
, where TL is the total length of the spicule, THr is the distance from the tip to the barb of the right spicule and THl is the distance from the tip to the barb of the left spicule. Species identification was established as follows:
-
DF < 0.63: H. contortus;
-
0.63 < DF < 3: H. placei.
The number of females of each species was calculated based on the percentage of male species identified for each genus.
Molecular evaluation of Haemonchus spp. by Polymerase Chain Reaction (PCR)
In the second year (2016), we collected 127 Haemonchus adult male worms from 12 lambs. The spicule measurement and a DNA sample were obtained from each specimen according to the protocol described by Santos et al. (2014) Santos MC, Amarante MRV, Silva MRL, Amarante AFT. Differentiation of Haemonchus placei from Haemonchus contortus by PCR and by morphometrics of adult parasites and third stage larvae. Rev Bras Parasitol Vet 2014; 23(4): 495-500. http://dx.doi.org/10.1590/S1984-29612014085. PMid:25517528.
http://dx.doi.org/10.1590/S1984-2961201...
.
Genomic DNA was obtained from cattle and sheep blood samples (hosts) and from previously identified specimens of H. contortus and H. placei (based on morphological analysis) and these were used as positive and negative PCR controls.
DNA samples were extracted from single specimens of Haemonchus spp. with the QIAamp DNA Mini Kit (QIAGEN, Hilden, Germany) according to the manufacturer’s instructions.
Molecular identification of Haemonchus spp. was performed by PCR using the species-specific primer pairs HcBotuF1/R2 and HpBotuF/R ( AMARANTE et al., 2017 Amarante MRV, Santos MC, Bassetto CC, Amarante AFT. PCR primers for straightforward differentiation of Haemonchus contortus, Haemonchus placei and their hybrids. J Helminthol 2017; 91(6): 757-761. http://dx.doi.org/10.1017/S0022149X16000882. PMid:28069083.
http://dx.doi.org/10.1017/S0022149X1600...
). Each primer pair amplified a single distinct band for each species. The primer pair HcBotuF1/R2 amplified an approximately 260 base pair (260 bp) band from H. contortus samples and HpBotuF/R amplified a PCR product of approximately 459 bp from H. placei samples. The PCR conditions were previously described by Amarante et al. (2017) Amarante MRV, Santos MC, Bassetto CC, Amarante AFT. PCR primers for straightforward differentiation of Haemonchus contortus, Haemonchus placei and their hybrids. J Helminthol 2017; 91(6): 757-761. http://dx.doi.org/10.1017/S0022149X16000882. PMid:28069083.
http://dx.doi.org/10.1017/S0022149X1600...
.
The PCR products were electrophoresed on a 2% agarose gel in 1% TAE buffer containing ethidium bromide and photographed under a UV light using a Sony Cyber-shot DSC-HX1 camera (Sony Electronics, San Diego, CA, USA). All PCR reactions were performed at least in duplicate.
Data analysis
Descriptive statistical analyses were used to summarise the data, as proposed by Bush et al. (1997) Bush AO, Lafferty KD, Lotz JM, Shostak AW. Parasitology meets ecology on its own terms: Margolis et al. revisited. J Parasitol 1997; 83(4): 575-583. http://dx.doi.org/10.2307/3284227. PMid:9267395.
http://dx.doi.org/10.2307/3284227 ...
, using the following terms:
-
Prevalence: the number of hosts infected with each nematode species divided by the number of hosts examined;
-
Intensity (of infection): the number of each nematode species in a single infected host;
-
Mean intensity: the total number of parasites of a particular species divided by the number of hosts infected with that parasite.
Results
The selected lambs presented a mean of 4,242 EPG in the first year and 1,781 EPG in the second year. The following species of nematodes were recovered from their gastrointestinal tracts: H. contortus, H. placei, T. axei, T. circumcincta, Ostertagia ostertagi, T. colubriformis , Cooperia curticei, Cooperia pectinata , Cooperia punctata, Cooperia spatulata, Nematodirus spathiger, Strongyloides papillosus and Trichuris ovis.
Haemonchus spp.
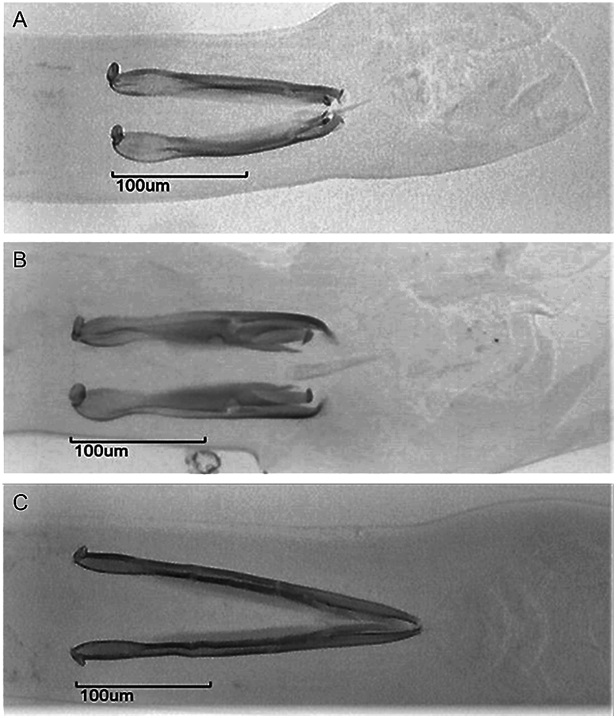
In 2015, we performed a morphological evaluation of 178 male Haemonchus specimens. All of them showed measurements consistent with H. contortus: a spicule length of 418 ± 3 µm, a right spicule barb length of 41 ± 3 µm, and a left spicule barb length of 21 ± 2 µm ( Figure 1 A). In 2016, in addition to the spicule measurements, we performed a molecular identification by PCR ( Table 1 ). We found 125 H. contortus, one H. placei ( Figure 1 C) and one hybrid specimen by PCR ( Figure 2 ; Figure 1 E; Table 1 ). Four specimens showed morphometrics consistent with H. placei; however, only one of these worms proved to be H. placei in the PCR analysis. The other three specimens were H. contortus with longer spicules and hooks. The prevalence of H. contortus was 100% in 2015 (mean intensity of 1,159 worms) and 92% in 2016 (mean intensity of 257 worms) ( Table 2 ).
(A) Spicules and (B) spicule tips of Haemonchus contortus; (C) spicules and (D) spicule tips of Haemonchus placei; and (E) spicules and (F) spicule tips of a hybrid.
Number of Haemonchus male specimens identified by PCR with its respective morphometrics (means followed by size range in parentheses).
PCR with genomic DNA samples to identify Haemonchus contortus, H. placei and their hybrid. (A) Species-specific primer pair HcBotuF1/R2 employed to amplify H. contortus (lane 3) and the hybrid (lane 5); (B) Species-specific primer pair HpBotuF/R employed to amplify H. placei (lane 4) and the hybrid (lane 5). Each photograph also shows reactions with: the control sample from ovine host (lane 2) and reagents without DNA in lane 6. Molecular marker (100 bp; GE Healthcare) were run in lanes 1 and 7 (A and B).
Prevalence and mean intensity of infection of gastrointestinal nematodes in lambs from Santana do Livramento – RS, Brazil, in 2015 and 2016 (12 lambs in each year).
Ostertagia spp. and Teladorsagia circumcincta
Ostertagia spp. was found in 2015 and 2016, with a low prevalence and intensity of infection. We found 14 male specimens with the O. ostertagi morphotype ( Figure 3 A) and 8 with the Ostertagia lyrata morphotype ( Figure 3 B). Both were considered to be a single species, O. ostertagi, with different morphotypes. Teladorsagia circumcincta ( Figure 3 C) was only recovered in 2016, with a prevalence of 25% and a mean intensity of 27 worms ( Table 2 ).
Spicules of Ostertagia ostertagi morphotype (A), Ostertagia lyrata morphotype (B) and Teladorsagia circumcincta (C).
Trichostrongylus spp.
Among the two species of Trichostrongylus found, T. colubriformis ( Figure 4 A) was by far the predominant species, with prevalence rates of 100% and 83% and mean intensities of 4,149 and 2,427 worms in 2015 and 2016, respectively ( Table 2 ). Trichostrongylus axei ( Figure 4 B) was only recorded in 2016, with a prevalence of 17% and a mean intensity of 750 worms.
Cooperia spp.
Four species of Cooperia were recovered ( Table 2 ). In 2015, C. punctata ( Figure 5 A) was the predominant species (prevalence, 92%; mean intensity, 810 worms) followed by C. pectinata (prevalence, 58%; mean intensity, 178; Figure 5 B) and C. spatulata (prevalence, 17%; mean intensity, 191; Figure 5 C). In 2016, Cooperia spp. was recovered in lower numbers than in 2015. The predominant species was C. curticei (prevalence, 67%; mean intensity, 260; Figure 5 D) followed by C. punctata (prevalence, 17%; mean intensity, 375) and C. pectinata (prevalence, 17%; mean intensity, 65).
Spicules of Cooperia punctata (A), Cooperia pectinata (B), Cooperia spatulata (C) and Cooperia curticei (D).
Other helminths
In both 2015 and 2016, N. spathiger ( Figure 6 ) was recovered, with prevalence rates of 83% in 2015 and 58% in 2016, but with a low mean intensity of infection ( Table 2 ). In 2016, S. papillosus was detected in the small intestine, with a high prevalence (92%) and mean intensity (1033 worms; Table 2 ). Few specimens of Trichuris were recovered from the large intestine. In 2015, only three adult females were recovered; therefore, it was not possible to identify the species. In 2016, two adult T. ovis males were recovered. In 2015, the prevalence was 25% and the mean intensity was 10 worms, whereas in 2016, the prevalence was 8% and the mean intensity was 50 worms ( Table 2 ).
In addition to the nematodes, 20 lambs (83.3%) presented cysts of Taenia hydatigena in the liver and mesentery. In 2015, one lamb presented Coenurus cerebralis , the larval stage of Taenia multiceps, in the brain. This animal did not show any clinical sign of a nervous disorder.
Discussion
In the present study, H. contortus and T. colubriformis were the major gastrointestinal parasites in sheep. These were also the main species, with the highest prevalence rates and infection intensities, in sheep from other Brazilian States, such as Santa Catarina ( RAMOS et al., 2004 Ramos CI, Bellato V, Souza AP, Avila VS, Coutinho GC, Dalagnol CA. Epidemiologia das helmintoses gastrintestinais de ovinos no Planalto Catarinense. Cienc Rural 2004; 34(6): 1889-1895. http://dx.doi.org/10.1590/S0103-84782004000600034.
http://dx.doi.org/10.1590/S0103-8478200...
) and São Paulo ( WILMSEN et al., 2014 Wilmsen MO, Silva BF, Bassetto CC, Amarante AFT. Gastrointestinal nematode infections in sheep raised in Botucatu, State of São Paulo, Brazil. Rev Bras Parasitol Vet 2014; 23(3): 348-354. http://dx.doi.org/10.1590/S1984-29612014058. PMid:25271455.
http://dx.doi.org/10.1590/S1984-2961201...
). In other important sheep-producing areas of South America, such as Uruguay, H. contortus is also the most important parasite in almost all seasons except winter, when T. colubriformis, and to a lesser extent, T. axei, T. circumcincta and Cooperia spp. are more important ( NARI & CARDOZO, 1987 Nari A, Cardozo H. Enfermedades causadas por parasitos internos: nematodes gastrointestinales. In: Bonino J, Durfin dei Campo A, Mad LJ, editors. Enfermedades de los Lanares . Montevideo: Hemisferio Sur; 1987. p. 1-57. ). In Argentina, H. contortus is also the primary nematode in summer and autumn, while other parasites (i.e., Nematodirus, Teladorsagia and Trichostrongylus ) are dominant in winter and spring ( SUAREZ & BUSETTI, 1995 Suarez VH, Busetti MR. The epidemiology of helminth infections of growing sheep in Argentina’s Western Pampas. Int J Parasitol 1995; 25(4): 489-494. http://dx.doi.org/10.1016/0020-7519(94)00122-5. PMid:7635625.
http://dx.doi.org/10.1016/0020-7519(94)...
; EDDI et al., 1996 Eddi C, Caracostantogolo J, Peña M, Schapiro J, Marangunich L, Waller PJ, et al. The prevalence of anthelmintic resistance in nematode parasites of sheep in Southern Latin America: Argentina. Vet Parasitol 1996; 62(3-4): 189-197. http://dx.doi.org/10.1016/0304-4017(95)00905-1. PMid:8686164.
http://dx.doi.org/10.1016/0304-4017(95)...
).
In our study, Nematodirus and Teladorsagia, which are typical parasites in temperate areas, occurred in small numbers. These nematodes have been reported at low infection intensities in sheep in RS ( SANTIAGO et al., 1975 Santiago MAM, Costa UC, Benevenga SF. Estudo comparativo da prevalência de helmintos em ovinos e bovinos criados na mesma pastagem. Pesqui Agropecu Bras 1975; 10(8): 51-56. , 1976 Santiago MAM, Benevenga SF, Costa UC. Epidemiologia e controle da helmintose ovina no município de Itaqui, Rio Grande do Sul. Pesqui Agropecu Bras 1976; 11(9): 1-7. ) and Santa Catarina ( RAMOS et al., 2004 Ramos CI, Bellato V, Souza AP, Avila VS, Coutinho GC, Dalagnol CA. Epidemiologia das helmintoses gastrintestinais de ovinos no Planalto Catarinense. Cienc Rural 2004; 34(6): 1889-1895. http://dx.doi.org/10.1590/S0103-84782004000600034.
http://dx.doi.org/10.1590/S0103-8478200...
), especially during the winter. The free-living stages of Nematodirus and Teladorsagia are adapted to develop and survive in cold climates ( MEDEROS et al., 2010 Mederos A, Fernández S, VanLeeuwen J, Peregrine AS, Kelton D, Menzies P, et al. Prevalence and distribution of gastrointestinal nematodes on 32 organic and conventional commercial sheep farms in Ontario and Quebec, Canada (2006-2008). Vet Parasitol 2010; 170(3-4): 244-252. http://dx.doi.org/10.1016/j.vetpar.2010.02.018. PMid:20236769.
http://dx.doi.org/10.1016/j.vetpar.2010...
).
We found a relatively high number of nematode species (13 total species) infecting the sheep. In contrast, Amarante et al. (2004) Amarante AFT, Bricarello PA, Rocha RA, Gennari SM. Resistance of Santa Ines, Suffolk and Ile de France sheep to naturally acquired gastrointestinal nematode infections. Vet Parasitol 2004; 120(1-2): 91-106. http://dx.doi.org/10.1016/j.vetpar.2003.12.004. PMid:15019147.
http://dx.doi.org/10.1016/j.vetpar.2003...
and Wilmsen et al. (2014) Wilmsen MO, Silva BF, Bassetto CC, Amarante AFT. Gastrointestinal nematode infections in sheep raised in Botucatu, State of São Paulo, Brazil. Rev Bras Parasitol Vet 2014; 23(3): 348-354. http://dx.doi.org/10.1590/S1984-29612014058. PMid:25271455.
http://dx.doi.org/10.1590/S1984-2961201...
reported only five nematode species in sheep that grazed alone in São Paulo State. When sheep share pasture with cattle, which was the case in the present study, the helminth species diversity is higher. A similar situation was observed by Giudici et al. (1999) Giudici C, Aumont G, Mahieu M, Saulai M, Cabaret J. Changes in gastro-intestinal helminth species diversity in lambs under mixed grazing on irrigated pastures in the tropics (French West Indies). Vet Res 1999; 30(6): 573-581. PMid:10596405. in the French West Indies.
Among the four species of Cooperia recorded in the present study, only Cooperia curticei was reported in sheep raised in the absence of cattle in São Paulo State ( AMARANTE et al., 2004 Amarante AFT, Bricarello PA, Rocha RA, Gennari SM. Resistance of Santa Ines, Suffolk and Ile de France sheep to naturally acquired gastrointestinal nematode infections. Vet Parasitol 2004; 120(1-2): 91-106. http://dx.doi.org/10.1016/j.vetpar.2003.12.004. PMid:15019147.
http://dx.doi.org/10.1016/j.vetpar.2003...
, 2009 Amarante AFT, Susin I, Rocha RA, Silva MB, Mendes CQ, Pires AV. Resistance of Santa Ines and crossbred ewes to naturally acquired gastrointestinal nematode infections. Vet Parasitol 2009; 165(3-4): 273-280. http://dx.doi.org/10.1016/j.vetpar.2009.07.009. PMid:19656629.
http://dx.doi.org/10.1016/j.vetpar.2009...
; WILMSEN et al., 2014 Wilmsen MO, Silva BF, Bassetto CC, Amarante AFT. Gastrointestinal nematode infections in sheep raised in Botucatu, State of São Paulo, Brazil. Rev Bras Parasitol Vet 2014; 23(3): 348-354. http://dx.doi.org/10.1590/S1984-29612014058. PMid:25271455.
http://dx.doi.org/10.1590/S1984-2961201...
). The other three species detected, C. punctata, C. pectinata and C. spatulata, are commonly found in cattle in Brazil ( LIMA, 1998 Lima WS. Seasonal infection pattern of gastrointestinal nematodes of beef cattle in Minas Gerais State - Brazil. Vet Parasitol 1998; 74(2-4): 203-214. http://dx.doi.org/10.1016/S0304-4017(97)00164-7. PMid:9561708.
http://dx.doi.org/10.1016/S0304-4017(97...
; BRICARELLO et al., 2007 Bricarello PA, Zaros LG, Coutinho LL, Rocha RA, Kooyman FN, De Vries E, et al. Field study on nematode resistance in Nelore-breed cattle. Vet Parasitol 2007; 148(3-4): 272-278. http://dx.doi.org/10.1016/j.vetpar.2007.06.013. PMid:17659839.
http://dx.doi.org/10.1016/j.vetpar.2007...
; SANTOS et al., 2010 Santos TR, Lopes WDZ, Buzulini C, Borges FA, Sakamoto CAM, Lima RCA, et al. Helminth fauna of bovines from the Central-Western region, Minas Gerais State, Brazil. Cienc Rural 2010; 40(4): 934-938. http://dx.doi.org/10.1590/S0103-84782010005000040.
http://dx.doi.org/10.1590/S0103-8478201...
; BASSETTO et al., 2014 Bassetto CC, Silva MRL, Newlands GFJ, Smith WD, Ratti J Jr, Martins CL, et al. Vaccination of grazing calves with antigens from the intestinal membranes of Haemonchus contortus: effects against natural challenge with Haemonchus placei and Haemonchus similis. Int J Parasitol 2014; 44(10): 697-702. http://dx.doi.org/10.1016/j.ijpara.2014.04.010. PMid:24960373.
http://dx.doi.org/10.1016/j.ijpara.2014...
). In the case of these last three species, bovine appears to be the source for sheep infection. A similar situation may have occurred with Ostertagia ostertagi and H. placei, two typical parasites of cattle ( WILLIAMS et al., 1987 Williams JC, Knox JW, Marbury KS, Kimball MD, Baumann BA, Snider TG. The epidemiology of Ostertagia ostertagi and other gastrointestinal nematodes of cattle in Louisiana. Parasitology 1987; 95(1): 135-153. http://dx.doi.org/10.1017/S0031182000057619. PMid:3670895.
http://dx.doi.org/10.1017/S003118200005...
; CARDOSO et al., 2013 Cardoso CP, Silva BF, Trinca LA, Amarante AFT. Resistance against gastrointestinal nematodes in Crioulo Lageano and crossbred Angus cattle in southern Brazil. Vet Parasitol 2013; 192(1-3): 183-191. http://dx.doi.org/10.1016/j.vetpar.2012.10.018. PMid:23177359.
http://dx.doi.org/10.1016/j.vetpar.2012...
) that were also detected in the present study. Taken together, these findings demonstrate that the high diversity of nematode species results from cross infections in sheep-cattle mixed grazing systems. What is the economical consequence of such cross infections? Apparently, they are not relevant. Santiago et al. (1975) Santiago MAM, Costa UC, Benevenga SF. Estudo comparativo da prevalência de helmintos em ovinos e bovinos criados na mesma pastagem. Pesqui Agropecu Bras 1975; 10(8): 51-56. , Amarante et al. (1997) Amarante AFT, Bagnola J Jr, Amarante MRV, Barbosa MA. Host specificity of sheep and cattle nematodes in São Paulo State, Brazil. Vet Parasitol 1997; 73(1-2): 89-104. http://dx.doi.org/10.1016/S0304-4017(97)00036-8. PMid:9477496.
http://dx.doi.org/10.1016/S0304-4017(97...
and Rocha et al. (2008) Rocha RA, Bresciani KDS, Barros TFM, Fernandes LH, Silva MB, Amarante AFT. Sheep and cattle grazing alternately: Nematode parasitism and pasture decontamination. Small Rumin Res 2008; 75(2-3): 135-143. http://dx.doi.org/10.1016/j.smallrumres.2007.09.001.
http://dx.doi.org/10.1016/j.smallrumres...
considered nematode cross infections to be of low epidemiological importance when sheep and cattle share pastures, because of the low infection intensities, suggesting that integrated grazing systems using cattle and sheep could be used for pasture decontamination. However, the H. contortus and H. placei co-infections detected in the present study and others in which sheep and cattle share the same pasture ( AMARANTE et al., 1997 Amarante AFT, Bagnola J Jr, Amarante MRV, Barbosa MA. Host specificity of sheep and cattle nematodes in São Paulo State, Brazil. Vet Parasitol 1997; 73(1-2): 89-104. http://dx.doi.org/10.1016/S0304-4017(97)00036-8. PMid:9477496.
http://dx.doi.org/10.1016/S0304-4017(97...
; ACHI et al., 2003 Achi YL, Zinsstag J, Yao K, Yeo N, Dorchies P, Jacquiet P. Host specificity of Haemonchus spp. for domestic ruminants in the savanna in northern Ivory Coast. Vet Parasitol 2003; 116(2): 151-158. http://dx.doi.org/10.1016/S0304-4017(03)00258-9. PMid:14519319.
http://dx.doi.org/10.1016/S0304-4017(03...
) raise the possibility of interspecies hybridization ( CHAUDHRY et al., 2015 Chaudhry U, Redman EM, Abbas M, Muthusamy R, Ashraf K, Gilleard JS. Genetic evidence for hybridisation between Haemonchus contortus and Haemonchus placei in natural field populations and its implications for interspecies transmission of anthelmintic resistance. Int J Parasitol 2015; 45(2-3): 149-159. http://dx.doi.org/10.1016/j.ijpara.2014.09.002. PMid:25449043.
http://dx.doi.org/10.1016/j.ijpara.2014...
). By PCR, we detected one hybrid specimen among 127 Haemonchus individuals. Brasil et al. (2012) Brasil BS, Nunes RL, Bastianetto E, Drummond MG, Carvalho DC, Leite RC, et al. Genetic diversity patterns of Haemonchus placei and Haemonchus contortus populations isolated from domestic ruminants in Brazil. Int J Parasitol 2012; 42(5): 469-479. http://dx.doi.org/10.1016/j.ijpara.2012.03.003. PMid:22787588.
http://dx.doi.org/10.1016/j.ijpara.2012...
also reported the presence of 1.28% hybrids in a population of 156 H. contortus and H. placei recovered from sheep, cattle, goat, and buffalo. This hybridization could lead to interspecies introgression of genes and provide a mechanism for the transmission of anthelmintic resistance between parasite species ( CHAUDHRY et al., 2015 Chaudhry U, Redman EM, Abbas M, Muthusamy R, Ashraf K, Gilleard JS. Genetic evidence for hybridisation between Haemonchus contortus and Haemonchus placei in natural field populations and its implications for interspecies transmission of anthelmintic resistance. Int J Parasitol 2015; 45(2-3): 149-159. http://dx.doi.org/10.1016/j.ijpara.2014.09.002. PMid:25449043.
http://dx.doi.org/10.1016/j.ijpara.2014...
). This would be a major concern given the high prevalence of anthelmintic resistance in H. contortus. Molecular tools are very useful for elucidating the dynamics of H. contortus and H. placei infections when the two species are sympatric, because identification based on morphological analysis has limitations for differentiating species and detecting hybrids within a population ( SANTOS et al., 2014 Santos MC, Amarante MRV, Silva MRL, Amarante AFT. Differentiation of Haemonchus placei from Haemonchus contortus by PCR and by morphometrics of adult parasites and third stage larvae. Rev Bras Parasitol Vet 2014; 23(4): 495-500. http://dx.doi.org/10.1590/S1984-29612014085. PMid:25517528.
http://dx.doi.org/10.1590/S1984-2961201...
; AMARANTE et al., 2017 Amarante MRV, Santos MC, Bassetto CC, Amarante AFT. PCR primers for straightforward differentiation of Haemonchus contortus, Haemonchus placei and their hybrids. J Helminthol 2017; 91(6): 757-761. http://dx.doi.org/10.1017/S0022149X16000882. PMid:28069083.
http://dx.doi.org/10.1017/S0022149X1600...
).
Twenty of the 24 lambs (83.3%) presented Cysticercus tenuicollis, the larval stage of Taenia hydatigena, and one lamb presented Coenurus larvae (Taenia multiceps) in the brain. Sheep acting as an intermediate host of dog cestodes is a relatively common problem in Brazil ( RISSI et al., 2008 Rissi DR, Rech RR, Pierezan F, Gabriel AL, Trost ME, Barros CSL. Cenurose em ovinos no sul do Brasil: 16 casos. Cienc Rural 2008; 38(4): 1044-1049. http://dx.doi.org/10.1590/S0103-84782008000400021.
http://dx.doi.org/10.1590/S0103-8478200...
; DE LA RUE et al., 2011 De la Rue ML, Takano K, Brochado JF, Costa CV, Soares AG, Yamano K, et al. Infection of humans and animals with Echinococcus granulosus (G1 and G3 strains) and E. ortleppi in Southern Brazil. Vet Parasitol 2011; 177(1-2): 97-103. http://dx.doi.org/10.1016/j.vetpar.2010.11.018. PMid:21273000.
http://dx.doi.org/10.1016/j.vetpar.2010...
; MORAIS et al., 2017 Morais DF, Vilela VLR, Feitosa TF, Santos VM, Gouveia VR, Athayde ACR, et al. Prevalence and risk factors for Cysticercus tenuicollis in goats and sheep in Paraíba, northeastern Brazil. Rev Bras Parasitol Vet 2017; 26(2): 235-238. http://dx.doi.org/10.1590/s1984-29612016092. PMid:28146156.
http://dx.doi.org/10.1590/s1984-2961201...
). Our results demonstrate that dogs are still being fed raw sheep offal after home slaughtering and/or have access to dead sheep carcasses on farms. The high occurrence of these cestodes, reported in the present study, demonstrates the need for immediate action by veterinary authorities, due to the serious implications of dog cestodes on animal and public health.
In conclusion, sheep from pastures shared with cattle presented a high diversity of nematode species. Among them, H. contortus and T. colubriformis showed the highest prevalence and infection intensities. Co-infection with H. contortus and H. placei was also observed, with consequent hybridization.
Acknowledgements
The authors would like to thank Professor Ciniro Costa and Cristiano M. Pariz by allowing animals to be used and Marina L.S.T. Piza for help in field activities. Fabiana A. Almeida received a scholarship from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), grant #502234/2014-8 and São Paulo Research Foundation (FAPESP), grant #2015/25718-7.
References
- Achi YL, Zinsstag J, Yao K, Yeo N, Dorchies P, Jacquiet P. Host specificity of Haemonchus spp. for domestic ruminants in the savanna in northern Ivory Coast. Vet Parasitol 2003; 116(2): 151-158. http://dx.doi.org/10.1016/S0304-4017(03)00258-9. PMid:14519319.
» http://dx.doi.org/10.1016/S0304-4017(03)00258-9 - Amarante AFT, Bagnola J Jr, Amarante MRV, Barbosa MA. Host specificity of sheep and cattle nematodes in São Paulo State, Brazil. Vet Parasitol 1997; 73(1-2): 89-104. http://dx.doi.org/10.1016/S0304-4017(97)00036-8. PMid:9477496.
» http://dx.doi.org/10.1016/S0304-4017(97)00036-8 - Amarante AFT, Bricarello PA, Rocha RA, Gennari SM. Resistance of Santa Ines, Suffolk and Ile de France sheep to naturally acquired gastrointestinal nematode infections. Vet Parasitol 2004; 120(1-2): 91-106. http://dx.doi.org/10.1016/j.vetpar.2003.12.004. PMid:15019147.
» http://dx.doi.org/10.1016/j.vetpar.2003.12.004 - Amarante AFT, Susin I, Rocha RA, Silva MB, Mendes CQ, Pires AV. Resistance of Santa Ines and crossbred ewes to naturally acquired gastrointestinal nematode infections. Vet Parasitol 2009; 165(3-4): 273-280. http://dx.doi.org/10.1016/j.vetpar.2009.07.009. PMid:19656629.
» http://dx.doi.org/10.1016/j.vetpar.2009.07.009 - Amarante AFT. Why is it important to correctly identify Haemonchus species? Rev Bras Parasitol Vet 2011; 20(4): 263-268. http://dx.doi.org/10.1590/S1984-29612011000400002. PMid:22166378.
» http://dx.doi.org/10.1590/S1984-29612011000400002 - Amarante MRV, Santos MC, Bassetto CC, Amarante AFT. PCR primers for straightforward differentiation of Haemonchus contortus, Haemonchus placei and their hybrids. J Helminthol 2017; 91(6): 757-761. http://dx.doi.org/10.1017/S0022149X16000882. PMid:28069083.
» http://dx.doi.org/10.1017/S0022149X16000882 - Bassetto CC, Silva MRL, Newlands GFJ, Smith WD, Ratti J Jr, Martins CL, et al. Vaccination of grazing calves with antigens from the intestinal membranes of Haemonchus contortus: effects against natural challenge with Haemonchus placei and Haemonchus similis. Int J Parasitol 2014; 44(10): 697-702. http://dx.doi.org/10.1016/j.ijpara.2014.04.010. PMid:24960373.
» http://dx.doi.org/10.1016/j.ijpara.2014.04.010 - Brasil BS, Nunes RL, Bastianetto E, Drummond MG, Carvalho DC, Leite RC, et al. Genetic diversity patterns of Haemonchus placei and Haemonchus contortus populations isolated from domestic ruminants in Brazil. Int J Parasitol 2012; 42(5): 469-479. http://dx.doi.org/10.1016/j.ijpara.2012.03.003. PMid:22787588.
» http://dx.doi.org/10.1016/j.ijpara.2012.03.003 - Bricarello PA, Zaros LG, Coutinho LL, Rocha RA, Kooyman FN, De Vries E, et al. Field study on nematode resistance in Nelore-breed cattle. Vet Parasitol 2007; 148(3-4): 272-278. http://dx.doi.org/10.1016/j.vetpar.2007.06.013. PMid:17659839.
» http://dx.doi.org/10.1016/j.vetpar.2007.06.013 - Bush AO, Lafferty KD, Lotz JM, Shostak AW. Parasitology meets ecology on its own terms: Margolis et al. revisited. J Parasitol 1997; 83(4): 575-583. http://dx.doi.org/10.2307/3284227. PMid:9267395.
» http://dx.doi.org/10.2307/3284227 - Cardoso CP, Silva BF, Trinca LA, Amarante AFT. Resistance against gastrointestinal nematodes in Crioulo Lageano and crossbred Angus cattle in southern Brazil. Vet Parasitol 2013; 192(1-3): 183-191. http://dx.doi.org/10.1016/j.vetpar.2012.10.018. PMid:23177359.
» http://dx.doi.org/10.1016/j.vetpar.2012.10.018 - Chaudhry U, Redman EM, Abbas M, Muthusamy R, Ashraf K, Gilleard JS. Genetic evidence for hybridisation between Haemonchus contortus and Haemonchus placei in natural field populations and its implications for interspecies transmission of anthelmintic resistance. Int J Parasitol 2015; 45(2-3): 149-159. http://dx.doi.org/10.1016/j.ijpara.2014.09.002. PMid:25449043.
» http://dx.doi.org/10.1016/j.ijpara.2014.09.002 - De la Rue ML, Takano K, Brochado JF, Costa CV, Soares AG, Yamano K, et al. Infection of humans and animals with Echinococcus granulosus (G1 and G3 strains) and E. ortleppi in Southern Brazil. Vet Parasitol 2011; 177(1-2): 97-103. http://dx.doi.org/10.1016/j.vetpar.2010.11.018. PMid:21273000.
» http://dx.doi.org/10.1016/j.vetpar.2010.11.018 - Echevarria F, Borba MFS, Pinheiro AC, Waller PJ, Hansen JW. The prevalence of anthelmintic resistance in nematode parasites of sheep in Southern Latin America: Brazil. Vet Parasitol 1996; 62(3-4): 199-206. http://dx.doi.org/10.1016/0304-4017(95)00906-X. PMid:8686165.
» http://dx.doi.org/10.1016/0304-4017(95)00906-X - Eddi C, Caracostantogolo J, Peña M, Schapiro J, Marangunich L, Waller PJ, et al. The prevalence of anthelmintic resistance in nematode parasites of sheep in Southern Latin America: Argentina. Vet Parasitol 1996; 62(3-4): 189-197. http://dx.doi.org/10.1016/0304-4017(95)00905-1. PMid:8686164.
» http://dx.doi.org/10.1016/0304-4017(95)00905-1 - Giudici C, Aumont G, Mahieu M, Saulai M, Cabaret J. Changes in gastro-intestinal helminth species diversity in lambs under mixed grazing on irrigated pastures in the tropics (French West Indies). Vet Res 1999; 30(6): 573-581. PMid:10596405.
- Hussain T, Periasamy K, Nadeem A, Ellahi Babar M, Pichler R, Diallo A. Sympatric species distribution, genetic diversity and population structure of Haemonchus isolates from domestic ruminants in Pakistan. Vet Parasitol 2014; 206(3-4): 188-199. http://dx.doi.org/10.1016/j.vetpar.2014.10.026. PMid:25468018.
» http://dx.doi.org/10.1016/j.vetpar.2014.10.026 - Instituto Brasileiro de Geografia e Estatística – IBGE. Tabela 3939: efetivo dos rebanhos, por tipo de rebanho [online]. Rio de Janeiro; 2015 [cited 2017 June 10]. Available from: https://sidra.ibge.gov.br/tabela/3939#resultado
» https://sidra.ibge.gov.br/tabela/3939#resultado - Jacquiet P, Cabaret J, Cheikh D, Thiam E. Identification of Haemonchus species in domestic ruminants based on morphometrics of spicules. Parasitol Res 1996; 83(1): 82-86. http://dx.doi.org/10.1007/s004360050213. PMid:9000240.
» http://dx.doi.org/10.1007/s004360050213 - Levine ND. Textbook of veterinary parasitology Minneapolis: Burgess Publishing Company; 1978.
- Lichtenfels JR, Hoberg EP. The systematics of nematodes that cause ostertagiasis in domestic and wild ruminants in North America: an update and key to species. Vet Parasitol 1993; 46(1-4): 33-53. http://dx.doi.org/10.1016/0304-4017(93)90046-P. PMid:8484224.
» http://dx.doi.org/10.1016/0304-4017(93)90046-P - Lichtenfels JR, Pilitt PA, Lancaster MB. Cuticular ridge patterns of seven species of Ostertagiinae (Nematoda) parasitic in domestic ruminants. Proc Helminthol Soc Wash 1988; 55(1): 77-86.
- Lichtenfels JR, Pilitt PA. Cuticular ridge patterns of Nematodirus (Nematoda: Trichostrongyloidea) parasitic in domestic ruminants of North America, with a key to species. Proc Helminthol Soc Wash 1983; 50(2): 261-274.
- Lima WS. Seasonal infection pattern of gastrointestinal nematodes of beef cattle in Minas Gerais State - Brazil. Vet Parasitol 1998; 74(2-4): 203-214. http://dx.doi.org/10.1016/S0304-4017(97)00164-7. PMid:9561708.
» http://dx.doi.org/10.1016/S0304-4017(97)00164-7 - Mederos A, Fernández S, VanLeeuwen J, Peregrine AS, Kelton D, Menzies P, et al. Prevalence and distribution of gastrointestinal nematodes on 32 organic and conventional commercial sheep farms in Ontario and Quebec, Canada (2006-2008). Vet Parasitol 2010; 170(3-4): 244-252. http://dx.doi.org/10.1016/j.vetpar.2010.02.018. PMid:20236769.
» http://dx.doi.org/10.1016/j.vetpar.2010.02.018 - Morais DF, Vilela VLR, Feitosa TF, Santos VM, Gouveia VR, Athayde ACR, et al. Prevalence and risk factors for Cysticercus tenuicollis in goats and sheep in Paraíba, northeastern Brazil. Rev Bras Parasitol Vet 2017; 26(2): 235-238. http://dx.doi.org/10.1590/s1984-29612016092. PMid:28146156.
» http://dx.doi.org/10.1590/s1984-29612016092 - Nari A, Cardozo H. Enfermedades causadas por parasitos internos: nematodes gastrointestinales. In: Bonino J, Durfin dei Campo A, Mad LJ, editors. Enfermedades de los Lanares . Montevideo: Hemisferio Sur; 1987. p. 1-57.
- Overbeck GE, Müller SC, Fidelis A, Pfadenhauer J, Pillar VD, Blanco CC, et al. Brazil’s neglected biome: The South Brazilian Campos. Perspect Plant Ecol Evol Syst 2007; 9(2): 101-116. http://dx.doi.org/10.1016/j.ppees.2007.07.005.
» http://dx.doi.org/10.1016/j.ppees.2007.07.005 - Ramos CI, Bellato V, Souza AP, Avila VS, Coutinho GC, Dalagnol CA. Epidemiologia das helmintoses gastrintestinais de ovinos no Planalto Catarinense. Cienc Rural 2004; 34(6): 1889-1895. http://dx.doi.org/10.1590/S0103-84782004000600034.
» http://dx.doi.org/10.1590/S0103-84782004000600034 - Rissi DR, Rech RR, Pierezan F, Gabriel AL, Trost ME, Barros CSL. Cenurose em ovinos no sul do Brasil: 16 casos. Cienc Rural 2008; 38(4): 1044-1049. http://dx.doi.org/10.1590/S0103-84782008000400021.
» http://dx.doi.org/10.1590/S0103-84782008000400021 - Rocha RA, Bresciani KDS, Barros TFM, Fernandes LH, Silva MB, Amarante AFT. Sheep and cattle grazing alternately: Nematode parasitism and pasture decontamination. Small Rumin Res 2008; 75(2-3): 135-143. http://dx.doi.org/10.1016/j.smallrumres.2007.09.001.
» http://dx.doi.org/10.1016/j.smallrumres.2007.09.001 - Santiago MAM, Benevenga SF, Costa UC. Epidemiologia e controle da helmintose ovina no município de Itaqui, Rio Grande do Sul. Pesqui Agropecu Bras 1976; 11(9): 1-7.
- Santiago MAM, Costa UC, Benevenga SF. Estudo comparativo da prevalência de helmintos em ovinos e bovinos criados na mesma pastagem. Pesqui Agropecu Bras 1975; 10(8): 51-56.
- Santiago MAM. Haemonchus Cobb, 1898 (Nematoda: Trichostrongylidae) contribuição ao estudo da morfologia, biologia e distribuição geográfica das espécies parasitas de ovinos e bovinos, no Rio Grande do Sul [thesis]. Santa Maria: Universidade Federal de Santa Maria; 1968.
- Santos MC, Amarante MRV, Silva MRL, Amarante AFT. Differentiation of Haemonchus placei from Haemonchus contortus by PCR and by morphometrics of adult parasites and third stage larvae. Rev Bras Parasitol Vet 2014; 23(4): 495-500. http://dx.doi.org/10.1590/S1984-29612014085. PMid:25517528.
» http://dx.doi.org/10.1590/S1984-29612014085 - Santos MC, Redman E, Amarante MRV, Gilleard JS, Amarante AFT. A panel of microsatellite markers to discriminate and study interactions between Haemonchus contortus and Haemonchus placei. Vet Parasitol 2017; 244: 71-75. http://dx.doi.org/10.1016/j.vetpar.2017.07.011. PMid:28917321.
» http://dx.doi.org/10.1016/j.vetpar.2017.07.011 - Santos TR, Lopes WDZ, Buzulini C, Borges FA, Sakamoto CAM, Lima RCA, et al. Helminth fauna of bovines from the Central-Western region, Minas Gerais State, Brazil. Cienc Rural 2010; 40(4): 934-938. http://dx.doi.org/10.1590/S0103-84782010005000040.
» http://dx.doi.org/10.1590/S0103-84782010005000040 - Silva MRL, Amarante MRV, Bresciani KDS, Amarante AFT. Host-specificity and morphometrics of female Haemonchus contortus, H. placei and H. similis (Nematoda: Trichostrongylidae) in cattle and sheep from shared pastures in São Paulo State, Brazil. J Helminthol 2015; 89(3): 302-306. http://dx.doi.org/10.1017/S0022149X14000078. PMid:24589375.
» http://dx.doi.org/10.1017/S0022149X14000078 - Suarez VH, Busetti MR. The epidemiology of helminth infections of growing sheep in Argentina’s Western Pampas. Int J Parasitol 1995; 25(4): 489-494. http://dx.doi.org/10.1016/0020-7519(94)00122-5. PMid:7635625.
» http://dx.doi.org/10.1016/0020-7519(94)00122-5 - Ueno H, Gonçalves PC. Manual para diagnóstico das helmintoses de ruminantes 4th ed. Tokyo: Japan International Cooperation Agency; 1998.
- Vicente JJ, Rodrigues HO, Gomes DC, Pinto RM. Nematóides do Brasil. Parte V: nematóides de mamíferos. Rev Bras Zool 1997;14(Suppl 1): 1-452. http://dx.doi.org/10.1590/S0101-81751997000500001.
» http://dx.doi.org/10.1590/S0101-81751997000500001 - Williams JC, Knox JW, Marbury KS, Kimball MD, Baumann BA, Snider TG. The epidemiology of Ostertagia ostertagi and other gastrointestinal nematodes of cattle in Louisiana. Parasitology 1987; 95(1): 135-153. http://dx.doi.org/10.1017/S0031182000057619. PMid:3670895.
» http://dx.doi.org/10.1017/S0031182000057619 - Wilmsen MO, Silva BF, Bassetto CC, Amarante AFT. Gastrointestinal nematode infections in sheep raised in Botucatu, State of São Paulo, Brazil. Rev Bras Parasitol Vet 2014; 23(3): 348-354. http://dx.doi.org/10.1590/S1984-29612014058. PMid:25271455.
» http://dx.doi.org/10.1590/S1984-29612014058 - Wyrobisz A, Kowal J, Nosal P. Insight into species diversity of the Trichostrongylidae Leiper, 1912 (Nematoda: Strongylida) in ruminants. J Helminthol 2016; 90(6): 639-646. http://dx.doi.org/10.1017/S0022149X15001017. PMid:26643224.
» http://dx.doi.org/10.1017/S0022149X15001017
Publication Dates
-
Publication in this collection
16 Aug 2018 -
Date of issue
Jul-Sep 2018
History
-
Received
10 Jan 2018 -
Accepted
09 May 2018