Abstract
A new species of Goeldia Keyserling, 1891 is described. Goeldia is the only genus of Titanoecidae with species in the Neotropical region and includes eight species diagnosed by the presence of a patellar process in the male palp. The new species, Goeldia zyngierae sp. nov., was found living inside the Casa de Pedra cave, Ribeira, Itabaiana, Sergipe, Brazil. Goeldia zyngierae sp. nov. is diagnosed by the tegular process with a small constriction near the base of the dorsal part and by a distal narrowing, more visible in the dorsal side of the patellar process of the male palp. The female epigynum shows a pair of sinuous depressions and a large mid field that does not close the copulatory openings as in Goeldia luteipes Keyserling, 1878. The vulva presents a narrow and straight copulatory duct in comparison with those of Goeldia mexicana Pickard-Cambridge, 1896 and a narrow spermathecae. The description includes illustrations of the genital organs, measurements of the body and legs and leg spination. Notes on the natural history show that inside the cave the spiders live farther than the first 40 m, in the aphotic zone, under stones or on the wall. They were never observed alone or walking on the ground, and the webs contained two to thirty spiders of different life stages, living together, suggesting a possible cooperative behavior. Further field work is needed to confirm and understand the degree of sociability of this new species, if this species is restricted to caves, and if this behavior occurs only inside the caves.
Cave; Neotropical; social behavior; spider; taxonomy
TAXONOMY AND NOMENCLATURE
A new species of Goeldia (Araneae: Titanoecidae) with notes on its natural history
Lina M. Almeida-SilvaI, 1 1 Corresponding author ; Antonio D. BrescovitI; Sidclay Calaça DiasII
ILaboratório de Artrópodes, Instituto Butantan. Avenida Vital Brazil 1500, Butantã, 05503-900 São Paulo, São Paulo, Brasil. E-mail: linamas@gmail.com; adbresc@terra.com.br
IILaboratório de Aracnologia, Museu Paraense Emílio Goeldi. Avenida Perimetral 1901, Caixa Postal 399, 66017-970 Belém, Pará, Brasil. E-mail: pachistopelma@hotmail.com
ABSTRACT
A new species of Goeldia Keyserling, 1891 is described. Goeldia is the only genus of Titanoecidae with species in the Neotropical region and includes eight species diagnosed by the presence of a patellar process in the male palp. The new species, Goeldia zyngierae sp. nov., was found living inside the Casa de Pedra cave, Ribeira, Itabaiana, Sergipe, Brazil. Goeldia zyngierae sp. nov. is diagnosed by the tegular process with a small constriction near the base of the dorsal part and by a distal narrowing, more visible in the dorsal side of the patellar process of the male palp. The female epigynum shows a pair of sinuous depressions and a large mid field that does not close the copulatory openings as in Goeldia luteipes Keyserling, 1878. The vulva presents a narrow and straight copulatory duct in comparison with those of Goeldia mexicana Pickard-Cambridge, 1896 and a narrow spermathecae. The description includes illustrations of the genital organs, measurements of the body and legs and leg spination. Notes on the natural history show that inside the cave the spiders live farther than the first 40 m, in the aphotic zone, under stones or on the wall. They were never observed alone or walking on the ground, and the webs contained two to thirty spiders of different life stages, living together, suggesting a possible cooperative behavior. Further field work is needed to confirm and understand the degree of sociability of this new species, if this species is restricted to caves, and if this behavior occurs only inside the caves.
Key words: Cave; Neotropical; social behavior; spider; taxonomy.
The spider family Titanoecidae was proposed by LEHTINEN (1967) to include the new genera Anuvinda and Pandava as well as Titanoeca Thorell, 1870, Nurscia Simon, 1874 and Goeldia Keyserling, 1891, which were transferred from Amaurobiidae Thorell, 1870. All Titanoecidae genera have a divided cribellum, a complex dorso-apical fold in the tibia of the male palpus, a tegular process near the base of the embolus and a median apophysis that is a flexible projection attached to the middle of the tegulum (LEHTINEN 1967, LEECH 1972, GRISWOLD et al. 2005).
To date, the family includes 46 species in five genera distributed in Holartic, Oriental and Neotropical zones. The least speciose genus is Anuvinda, with only one species from India, followed by Pandava, with two Oriental species, Nurscia with four species from the Paleartic and Oriental regions and Titanoeca with 31 Holartic species (PLATNICK 2008).
Goeldia, the only genus occurring in the Neotropical region, includes eight species (PLATNICK 2008) and is diagnosed by the combination of the following characters: presence of a patellar process; a fold with two plates at the dorso-prolateral distal part of the tibia; the position of the tegular process, anterior to the base of the embolus in the male palp; and by the lateral position of the copulatory openings at the posterior border of the female epigynum (LEECH 1972). The genus is currently being revised by the first author, which enabled the proposition of Goeldia zyngierae sp. nov. as a new species.
Titanoecid spiders build cribellate webs, which can be attached to the substrate and can include sheet-like components (GRISWOLD et al. 2005). No species of this family is known to display social behaviors such as building and maintaining communal nests, cooperative hunting and parental care of the spiderlings (D'ANDREA 1987). The cooperative behavior or sociality seems to appear independently in a small fraction of spider species (AVILÉS et al. 2006, MILLER 2006). The two attributes commonly used to classify degree of sociality of the species are permanence and territoriality (D'ANDREA 1987, AVILÉS 1997). Subsociality consists in the formation of periodically territorial or non-territorial colonies of individuals frequently related to the extension of the maternal care period (AVILÉS 1997, AVILÉS & Bucowisk 2006, MILLER 2006). Quasisociality (WILSON 1971), also labeled non-territorial permanently-social (D'ANDREA 1987), is marked by a high degree of inbreeding and strongly female-biased sex-ratios (AVILÉS 1997, AGNARSSON et al. 2006). On the other hand, the territorial permanent-social, labeled "communal", is characterized by a cluster of individuals in connected webs in a colony (AVILÉS 1997, MILLER 2006). In this paper, we describe a new species of Goeldia that displays subsocial behavior, inside a cave, in the state of Sergipe, Brazil. This represents the first case of cooperative behavior recorded for Titanoecidae.
MATERIAL AND METHODS
Taxonomy: the material examined is deposited in the following institutions (Abbreviation and curators in parenthesis): Instituto Butantan, São Paulo (IBSP, A.D. Brescovit) and Museu Paraense Emílio Goeldi, Belém (MPEG, A.B. Bonaldo). All measurements are in millimeters. Male palp was illustrated in ventral, dorsal and retrolateral views. Female epigyne were dissected and submerged in clove oil to study internal structures. Illustrations were made using stereoscopic microscope LEICA (MZ12s) with camera lucida. Only articles with spines are mentioned on the leg spination.
Biological observations: most specimens were collected inside the Casa de Pedra cave (10º50'S, 38º27'W), formerly known as Ribeira Cave located in the Ribeira region, Itabaiana, Sergipe, Brazil, where ad libitum observations on the natural history were made. This cave is located in an ecotone between Atlantic Rain forest and Brazilian "Caatinga". It is a small and dry cave, located 100 m above sea level, with a horizontal development of 210 m and a maximum depth of 8.62 m. The cave has the shape of a "T" with an opening to the outside at the right side of the "T" (Fig. 1). The entrance of the cave is 5.20 m wide and 1.55 m high. Temperature data taken inside and outside the cave showed a difference of 10 degrees Celsius during the day.
Spiders were found in two saloons in the aphotic zone, almost 40 m from the cave entrance (Fig. 1). Samplings were carried out in March, May, June and September 2001. In order to determine if this species was living exclusively inside the cave, fifty pitfall-traps were installed close to the entrance of the cave for five nights.
TAXONOMY
Goeldia zyngierae sp. nov.
Type material. Male holotype and female paratype from Casa de Pedra Cave (10º50'S, 38º27'W), Ribera region, Itabaiana, Sergipe, Brazil, (Fig. 1), 28.III.2001, N. Zyngier & S.C. Dias leg., deposited in IBSP 31856. Paratypes: 1 male and 1 female with the same data as holotype, deposited in MPEG 13808.
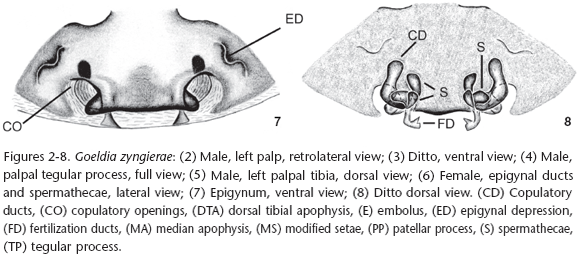
Diagnosis. Males of Goeldia zyngierae sp. nov. differ from those of the remaining species of the genus by the shape of the tegular process (Figs 2-5) which has a small constriction near the base of the dorsal part (Fig. 3) and by a distal narrowing at the patellar process, more visible dorsally (Figs 3 and 4), in the male palp. The female epigynum (Fig. 7) resembles that of G. mexicana (O. Pickard-Cambridge, 1896) by the straight board of the mid field but differs by the large median plate that projects laterally the copulatory openings as in G. mexicana. The vulva (Figs 6 and 8) presents a narrow and straight copulatory duct when compared to those of Goeldia mexicana (LEECH 1972: 385 fig. 384) and a narrow spermathecae (Figs 6 and 8).
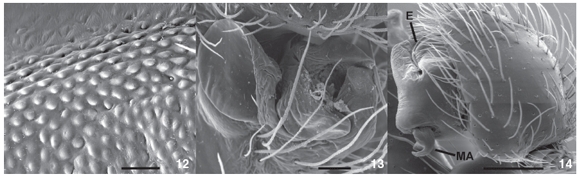
Description. Male (IBSP 31856). Coloration: carapace and sternum brown, chelicerae, labium and endites dark brown. Legs brown to yellow except femur I dark brown. Abdomen brown, dorsally with pale brown lateral spots, converging to center. Total length 6.44. Carapace 2.92 long, 2.12 wide, 1.04 high. Clypeus 0.19 high. Abdomen 3.52 long, 2.28 wide. Eye diameters and interdistances: AME 0.11, ALE 0.13, PME 0.09 PLE 0.14; AME-AME 0.12, AME-ALE 0.14, PME-PME 0.16, PME-PLE 0.24, AME-PME 0.11, ALE-PLE 0.06. Chelicerae 1.4 long, with a promarginal tooth between two denticles and two retromarginal denticles (Fig. 9). Leg measurements: I: femur 2.33/patella 0.9/tibia 2.15/metatarsus 2.05/tarsus 0.93/total 8.35; II: 2.28/0.96/2.04/1.96/0.83/8.07; III: 2.04/0.83/1.58/1.70/0.70/6.84; IV: 2.40/0.78/2.24/2.08/0.70/8.20. Leg formula 1423. Leg spination: femur I p0-0-1m, II p0-0-1m; tibia I v1r-0-2-0 or v0-2-2, r2-1-0 or r1-1-0; III p0-0-1m; IV r0-0-1m; metatarsus I v1r-1r-1m or v0-2-1m, p0-0-1m, r0-0-1m; II v1r-2-1m or v0-2-1m, p0-0-1m, r0-0-1m; III d0-0-2, v1r-1r-2-1m or v0-2-1m, p0-0-1m, r0-0-1m; IV d0-0-2 or 0-0-0, v1p-2-2-1m or v1r-1r-2-1m, p0-0-1m, r0-0-1m. Cribellum present, bipartite; Calamistrum inconspicuous; Chelicerae with stridulatory files at the retrolateral side (Fig. 10). Palpal femur with stridulatory files (Figs 11 and 12). Palp with modified setae at cymbium border (Figs 2, 3); spermatic duct narrow, bent in the middle of tegulum (Fig. 3). Dorsal tibial apophysis with median lobe partially sclerotized (Figs 5 and 14). Median apophysis bifid (Figs 2, 3 and 14), with two regions: a pointed one bent over an enlarged one.
Female (IBSP 31856) Coloration: same pattern as in male but lighter (Fig. 18). Total length: 6.44. Carapace 2.8 long, 1.87 wide, 0.96 high, clypeus 0.16 high. Abdomen 3.64 long, 2.60 wide. Eye diameters and interdistances: AME 0.09, ALE 0.14, PME 0.12, PLE 0.15; AME-AME 0.11, AME-ALE 0.13, PME-PME 0.18, PME-PLE 0.24, AME-PME 0.13, ALE-PLE 0.06, Chelicerae 1.20 long, with a promarginal tooth between two denticles and two retromarginal denticles. Leg measurements: leg I: femur 2.08/patella 0.88/tibia 1.75/metatarsus 1.45/tarsus 0.80/total 6.96; leg II: 1.68/0.78/1.45/1.35/0.70/5.96; leg III: 1.66/0.68/1.28/1.05/0.57/5.23; leg IV: 2.00/0.85/1.88/1.52/0.63/6.89. Leg formula 1423. Leg spination: femur I p 0-0-1m, metatarsus I p 0-0-1m, v 0-0-1m, r 0-0-1m; metatarsus II-IV p 0-0-1m, v 0-0-1m, r 0-0-1m. Cribellum as in male; Calamistrum uniseriate, occupying all metatarsus IV. Stridulatory ridges as a line of spines (Fig. 15) over the mid dorsal side of the chelicerae, with enlarged bases (Fig. 16). Stridulatory files of the palpal femur as in male. Metatarsal trichobothrium with transversal ridges (Fig. 17). Walls of the copulatory duct and part of the fertilization ducts visible by transparence (Fig. 7). Copulatory ducts "L" shaped with curved bases (Fig. 8). Spermathecae bifid, laterally attached at the end of the copulatory duct.
Variation. Coloration can be darker or lighter but always displays the same pattern. Nine males: total length: 4.3-7.31; carapace length: 2.1-3.35; femur I: 1,85-2,28. Ten females: total length: 5.1-8; carapace length 2.4-3.06; femur I: 1.84-2.91.
Additional Material Examined. Brazil, Sergipe: Itabaiana (Ribeira area, Casa da Pedra Cave, 10º50'S, 38º27'W), 2 females, 28.II.2001 (IBSP 31834, 31836); 4 males, 3 females, 5 imm, 28.III.2001, (IBSP 31850, 31851, 31853 31856); 1 female, 5 eggsacs, 15.VI.2001 (IBSP 31843); 3 female, 17.VI.2001 (IBSP 31854); 1 male, 10 females, 16 eggsacs, 19.VI.2001 (IBSP 31832, 31835, 31844, 31845, 31846, 31847, 31849, 31857); 1 male, 14 female, 1 imm. 2 eggsacs, 4.V.2001 (IBSP 31833, 31838-31842, 31852, 31855); 1 male, 3 females, 1 imm. 05.IX.2001 (IBSP 31848) all collected by S.C. Dias & N. Zyngier leg.; 2 males, 17/XI/2002, N. Zyngier leg. (IBSP 36799).
Distribution. Only known from the Ribeira area, Itabaiana, state of Sergipe, Brazil
Natural History. Home range: spiders inside the cave were never found walking on the ground. Webs were usually found on the cave walls (Fig. 18), but small (Fig. 19) webs of approximately 30 cm, with females and eggsacs/spiderlings, were found close to the ground. The largest web was fixed to the wall and was five meters long and 60 cm high. Due to the authors personal observations that Goeldia males can walk on the ground during the day we decided to install pitfall traps outside the cave in order to verify if they wander outside the cave. Two adult males were collected, suggesting that this species is not exclusively cavernicolous and occurs in the surrounding epigeic region as well.
Feeding and prey capture: the most frequent prey remains found in webs were Lepidoptera (Fig. 21), Coleoptera, Diptera (Insecta) and Acari (Arachnida). We did not observe the spiders feeding together but when the web was disturbed a group of spiders would move quickly towards the disturbed area.
Body size, life cycle and egg sacs: females are the same size of the males (mean carapace length: female 2.71, male 2.71) but males have longer legs (femur I: female 1.86, male 2.54). Analyzing the samples and comparing the data, males were more abundant than females in March, decreasing in abundance in May and no males were found in June. These data suggest that March is probably the mating month. Twenty-three egg sacs were collected between May and June and the average size was 5.56 ± 0.92 mm. Twelve of them had only molts and three to eight unhatched eggs. In one case, spiderlings and ecdysis were found in the same egg sac. Of the remaining ten egg sacs, six had eggs, the number varying from 42 to 101 and four had from 57 to 82 spiderlings, supporting the suggestion that March is the mating month and May and June are the egg laying months.
Courtship behavior: an aborted courtship event was observed in the laboratory. The event took place during the day and lasted almost an hour. The male placed himself in front of the female, drumming with the pedipalps and shaking its body. He twice tried to raise the female with his leg I unsuccessfully. After these attempts, he gave up and ran away.
Sociality: spiders of different sexes and development stages were found living on webs that were fixed to the walls of the cave, at approximately one meter from the ground. These webs presented multiple openings (Fig. 20) and we were able to observe free movement of the spiders inside the webs. These interconnected webs plus the free movement of the spiders inside and between them, points towards a non-territorial sociality (AVILÉS 1997, MILLER 2006). Although there is no good evidence of quasisocial behavior, such as, biased sex ratio or communal brood, the possibility of "subsocial" behavior is not discarded. Thus, further field work is necessary to confirm and understand the degree of sociality, if this behavior occurs only inside caves and if so, what kinds of features of the cave impose this behavior on this species.
Etymology. The specific name honors Nicole Zyngier who collected the spiders and took notes on their behavior.
ACKNOWLEDGMENTS
We wish to thank FAPESP (LMAS MS grant 06/05453-0) and CNPq (ADB PQ 301776/2004-0 and SCD PhD grant) for financial support. We also thank Cristina A. Rheims for revision of the English version and helpful suggestions on the manuscript, and Adalberto José dos Santos for helpful suggestions on the manuscript.
LITERATURE CITED
Submitted: 27.VIII.2008; Accepted: 15.VI.2009.
Editorial responsability: Claudio J.B. de Carvalho
- AGNARSSON, I.; L. AVILÉS; J. A. CODDINGTON & W.P. MADDISON. 2006. Sociality in Theridiidae spiders: Repeated origins for an evolutionary dead end. Evolution 60 (11): 2342-2351
- AVILÉS, L. 1997, Causes and consequences of cooperation and permanent sociality in spiders, p. 476-497. In: J. CHOE & B. CRESPI (Eds). The Evolution of Social Behavior in Insects and Arachnids. Cambridge Cambridge University Press, 561p.
- AVILÉS, L & T.C. BUCOWSKI. 2006. Group living and inbreeding depression in a subsocial spider. Proceedings of the Royal Society 270: 157-163.
- AVILÉS, L.; W.P. MADDISON & I. AGNARSSON. 2006. A new independently derived social spider with explosive colony proliferation and a female size dimorphism. Biotropica 38: 743-753.
- D'ANDREA, M. 1987. Social behavior in spiders (Arachnida; Araneae). Italian Journal of Zoology Monograph 3: 1-156.
- GRISWOLD, C.E.; M.J. Ramírez; J.A. CODDINGTON & N.I. PLATNICK. 2005. Atlas of phylogenetic data for entelegyne spiders (Araneae: Araneomorphae: Entelegynae) with comments on their phylogeny. Proceedings of the California Academy of Sciences 56 (Suppl. 2): 1-324.
- KEYSERLING, E. 1891. Die Spinnen Amerikas. Brasilianische Spinnen. Nürnberg, 3: 1-278.
- LEECH, R.E. 1972. A revision of the Nearctic Amaurobiidae (Arachnida: Araneida). Memoirs of the Entomological Society of Canada 84: 1-182.
- LEHTINEN, P.T. 1967. Classification of the cribellate spiders and some allied families, with notes on the evolution of the suborder Araneomorpha. Annales Zoologici Fennici 4: 199-468.
- MILLER, J.A. 2006. Web-sharing sociality and cooperative prey capture in a Malagasy spintting spider (Araneae: Scytodidae). Proceedings of the California Academy of Sciences 25 (57): 739-750.
- PLATNICK, N. I. 2008. The World Spider Catalog, version 8.5. Available online at: http://research.amnh.org/entomology/spiders/catalog81-87/INTRO1.html (Accessed: 09/VII/2008).
- WILSON, E.O. 1971. The Insect Societies. Cambridge, Belknap Press, 548p.
Publication Dates
-
Publication in this collection
27 July 2009 -
Date of issue
June 2009
History
-
Received
27 Aug 2008 -
Accepted
15 June 2009