Abstract
This study describes the diet and assesses potential overlap in the feeding habits of Trichiurus lepturus Linnaeus, 1758 and Pontoporia blainvilleiGervais & D'Orbigny, 1844 in northern Rio de Janeiro, southeastern Brazil. Fishes were numerically dominant in both diets, followed by cephalopods for P. blainvillei and crustaceans for T. lepturus. Both predators move along similar coastal feeding areas in northern Rio de Janeiro, but our results indicate differences in their resource exploitation, what allows for their coexistence.
Cannibalism; diet; feeding; food habits; predators
SHORT COMMUNICATION
Diet and potential feeding overlap between Trichiurus lepturus (Osteichthyes: Perciformes) and Pontoporia blainvillei (Mammalia: Cetacea) in Northern Rio de Janeiro, Brazil
Vanessa T. Bittar1 1 Corresponding autor. E-mail: vatrindade@gmail.com ; Ana Paula M. Di Beneditto
Laboratório de Ciências Ambientais, Centro de Biociências e Biotecnologia, Universidade Estadual do Norte Fluminense. Avenida Alberto Lamego 2000, 28013-602 Campos dos Goytacazes, Rio de Janeiro, Brasil
ABSTRACT
This study describes the diet and assesses potential overlap in the feeding habits of Trichiurus lepturus Linnaeus, 1758 and Pontoporia blainvilleiGervais & D'Orbigny, 1844 in northern Rio de Janeiro, southeastern Brazil. Fishes were numerically dominant in both diets, followed by cephalopods for P. blainvillei and crustaceans for T. lepturus. Both predators move along similar coastal feeding areas in northern Rio de Janeiro, but our results indicate differences in their resource exploitation, what allows for their coexistence.
Key words: Cannibalism; diet; feeding; food habits; predators.
Trichiurus lepturus Linnaeus, 1758 (Perciformes: Trichiuriidae) and Pontoporia blainvillei Gervais & D'Orbigny, 1844 (Cetacea: Pontoporiidae) are carnivore species that coexist along the Brazilian coast (DI BENEDITTO et al. 2001, BITTAR et al. 2008). There are no comparatives studies on the feeding habits of these species, but their diet has been studied separately (SHUOZENG 1995, DI BENEDITTO & RAMOS 2001, DI BENEDITTO et al. 2001, DANILEWICZ et al. 2002, RODRIGUEZ et al. 2002, MARTINS et al. 2005, CHIOU et al. 2006, BITTAR et al. 2008).
This study compares the feeding habits of these predator species, since both coexist in northern Rio de Janeiro and could be trophic competitors. We endavour to contribute to a better understanding of the marine food chain along the Brazilian coastal waters.
All specimens analyzed were collected in northern Rio de Janeiro through gillnet fisheries carried out at Atafona fishing village, between 21º18'S and 22º25'S, from 0.02 to 42.1 nautical miles from shore and in waters 5-70 m deep. Between August 2004 and July 2006, 350 individuals of T. lepturus were collected, and 343 were included in the diet analysis. Only adult specimens, measuring between 100 and 163 cm of total length, were analyzed, due to their hightest likelyood of being potential trophic competitors of P. blainvillei (DI BENEDITTO et al. 2001). Ninety-nine specimens of P. blainvillei collected from September 1989 to December 2005 were analyzed. The total length of the P. blainvillei specimens ranged from 78.2 to 148.0 cm.
Stomachs were separated and their contents rinsed in a sieve of 400 µm to recover the contents. Undigested prey were measured and weighted. The Index of Relative Importance (IRI) (PINKAS et al. 1971) was applied to determine the representative prey species and considered teleosts, cephalopods, and crustaceans as independent prey to reduce under or overestimation of their importance (CLARKE 1986). A non-parametric descriptive statistic, with the median as a central tendency measure, was used for the size, biomass and density (number of individuals) comparison considering the main teleost and cephalopod items consumed by both carnivores. The crustaceans were not considered in our comparisons due to their scarcity in the diet of P. blainvillei. The diets of T. lepturus and P. blainvillei were also compared through the diversity and dominance indexes of Simpson and the Jaccard (qualitative), and Morisita-Horn (quantitative) similarity indexes, described in MAGURRAN (1988).
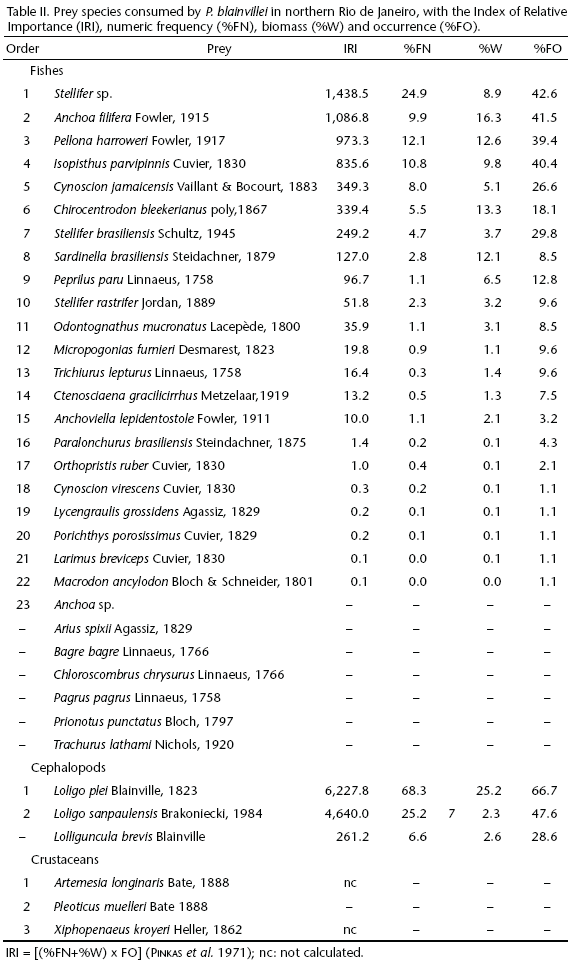
Trichiurus lepturus and P. blainvillei prey upon neritic species, both pelagic and demersal (Tabs I and II). Teleosts species of Scianidae, Engraulidae and Clupeidae are the most representative items in the diet of both species (Tabs I and II). Cephalopods are more frequent in the diet of P. blainvillei, while the crustaceans dominate in stomach contents of T. lepturus. All favoured fish (DI BENEDITTO et al. 2001), cephalopod (ROPER et al. 1984) and crustacean (DI BENEDITTO et al. 1998, COSTA et al. 2003) species occur all year round in northern Rio de Janeiro.
Results from the non-parametric descriptive statistics are presented on table III. In general, prey species ingested by T. lepturus are larger than those ingested by P. blainvillei. However, the total biomass and density recorded in the stomach contents are lower in T. lepturus. The greater prey selectivity by T. lepturus is possibly associated with its voracity, as reported by MARTINS et al. (2005) and CHIOU et al. (2006). The values of biomass and density (number of individuals), in turn, may reflect differences in the digestion rate, the ability to fill the stomach and/or the nutritional needs of the predators.
The diet of P. blainvillei is more diverse than that of T. lepturus. Although prey richness is similar, the high abundance of prey consumed for P. blainvillei may be responsible for this difference. The prey diversity is influenced by the equitable distribution of the fishes consumed by P. blainvillei, resulting in a lower dominance value. For T. lepturus, the opposite pattern is recorded. The two e species show 60% of similarity in their diet. However, the quantitative comparison (abundance) indicates a low diet similarity between them (Tab. IV).
Diet overlap is expected between sympatric species of carnivores that have similar sizes and food preferences (ZAVALA-CAMIN 1996). In this study, the lower diversity of prey and the higher dominance may indicate that the diet of T. lepturus is more selective than the diet of P. blainvillei. However, we need to be careful when applying such ecological indexes to diet studies. Differences in food assimilation rates between the predators, for instance, can under or over estimate their food preferences.
In northern Rio de Janeiro, the adult specimens of T. lepturus and P. blainvillei exploit coastal waters to obtain their food resources. However, the former feeds preferentially on pelagic species, while the latter feeds along the water column. In general, the prey consumed by both species in northern Rio de Janeiro have low commercial value or are treated as by-catch by the local fisheries. When the preys are commercially valuable, as I. parvipinnis and L. plei, the fisheries targets larger specimens than those consumed by the species studied (COSTA & HAIMOVICI 1990, DI BENEDITTO et al. 1998). This indicates that the prey species can be at pre-recruitment sizes of the fishery grounds.
Despite some overlap in their feeding habits, quantitative variations in size, biomass and density of prey consumed indicate differences in their exploitation of resources, allowing for the coexistence of T. lepturus and P. blainvillei in the region. However, additional studies on their feeding habits are still needed in the areas where both species are sympatric in order to confirm this pattern.
ACKNOWLEDGEMENTS
The authors would like to thank to Marcos C. de O. Santos, Ilana R. Zalmon, Leandro R. Monteiro and Carlos E.V. de Carvalho for reviewing the early draft of this manuscript. We also thank the fishermen from Atafona fishing village that provided us with the studied specimens and Silvana R. Gomes for helping the collection of specimens. This study was supported by FAPERJ (E-26/170670/2004) and A.P.M. DI BENEDITTO was supported by CNPq (305160/2006-0).
LITERATURE CITED
Submitted: 03.IX.2008; Accepted: 16.VI.2009
Editorial responsability: Paulo da Cunha Lana
- BITTAR, V.T.; B.F.L. CASTELLO & A.P.M. DI BENEDITTO. 2008. Hábito alimentar do peixe-espada adulto, Trichiurus lepturus, na costa norte do Rio de Janeiro, sudeste do Brasil. Biotemas 21 (2): 83-90.
- CHIOU, W.D.; C.Y. CHEN; C.M. WANG & C.T. CHEN. 2006. Food and feeding habits of ribbonfish Trichiurus lepturus in coastal waters of south-western Taiwan. Fisheries Science 72: 373-381.
- CLARKE, M.R. 1986. A handbook for the identification of cephalopod beaks. Claredon Press, Oxford, 273p.
- COSTA, P.A. & M. HAIMOVICI. 1990. A pesca de polvos e lulas no litoral do Rio de Janeiro. Ciência e Cultura 42 (12): 1124-1130.
- COSTA, R.C.; A. FRANSOZO; G.A.S. MELO & F.A.M. FREIRE. 2003. Chave ilustrada para identificação dos camarões Dendrobranchiata do litoral norte do Estado de São Paulo, Brasil. Biota Neotropica 3 (1): BN01503012003. Available online at: http://www.biotaneotropica.org.br [Accessed: 10.VI.2007]
- DANILEWICZ, D.; F. ROSAS; R. BASTIDA; J. MARIGO; M. MUELBERT; D. RODRIGUEZ; J. LAILSON BRITO; V. RUOPOLLO; R. RAMOS; M. BASSOI; P.H. OTT; G. CAON; A.M. ROCHA; J.L. CATAO-DIAS & E. SECCHI. 2002. Report of the working group on biollogy and ecology. The Latin American Journal of Aquatic Mammals (Special Issue 1) 1: 25-42.
- DI BENEDITTO, A.P.M. & R.M.A. RAMOS. 2001. Biology and conservation of the franciscana (Pontoporia blainvillei) in the north of Rio de Janeiro State, Brazil. Journal of Cetacean Research and Management 3 (2): 185-192.
- DI BENEDITTO, A.P.M.; R. RAMOS & N.R.W. LIMA. 1998. Fishing activity on Northern Rio de Janeiro State (Brazil) and its relation with small cetaceans. Brazilian Archives of Biolology and Technology 41 (3): 296-302.
- DI BENEDITTO, A.P.M.; R.M.A. RAMOS & N.R.W. LIMA. 2001. Os golfinhos: origem, classificação, captura acidental, hábito alimentar. Porto Alegre, Ed. Cinco Continentes, 152p.
- MAGURRAN, A.E. 1988. Ecological diversity and its meansurement. Princeton University Press, Princeton, 179p.
- MARTINS, A.S.; M.HAIMOVICI & R. PALACIOS. 2005. Diet and feeding of the cutlassfish Trichiurus lepturus in the Subtropical Convergence Ecosystem of southern Brazil. Journal of the Marine Biological Association of the United Kingdom 85: 1223-1229.
- PINKAS, L.; M.S. OLIPHANT & I.L.K. IVERSON. 1971. Food habits of albacore, bluefin tuna and bonito in Californian waters. Californian of Fish and Game 152: 1-105.
- RODRIGUEZ, D.; L. RIVERO & R. BASTIDA. 2002. Feeding ecology of the franciscana (Pontoporia blainvillei) in marine and estuarine waters of Argentina. The Latin American Journal of Aquatic Mammals 1 (Special Issue 1): 77-94.
- ROPER, C.F.E.; M.J. SWEENEY & C.E. NAUEN. 1984. FAO species catalogue cephalopods of the world. Rome, FAO, vol. 3, 277p.
- SHUOZENG, D. 1995. Food utilization of adult flatfishes co-occurring in the Bohai Sea of China. Netherlands Journal of Sea Research 34 (1-3): 183-193.
- ZAVALA-CAMIN, L. A. 1996. Introdução sobre alimentação natural em peixes. Maringá, Editora da UEM.129p.
Publication Dates
-
Publication in this collection
28 July 2009 -
Date of issue
June 2009
History
-
Received
03 Sept 2008 -
Accepted
16 June 2009