Abstract
This study evaluated the isotopic composition of three zooplankton species - Notodiaptomus amazonicus (Wright, 1935), Moina minuta Hansen, 1899 and Bosmina hagmanni Stingelin, 1904 - from the Upper Paraná River floodplain. We predicted that there would be isotopic variability among species in the different sampled environments. Samplings were conducted in three lakes from the Baía subsystem and in two lakes from the Paraná subsystem. At each location, some abiotic variables were measured, and zooplankton (3 to 4) and phytoplankton (3) samples were taken. The species sampled did not present significant differences between subsystems; however, they were different among the lakes, with variations of about 10.8‰ for δ13C and 5.8‰ for δ15N. The isotopic values found for phytoplankton also did not differ significantly among sampling stations, presenting variations of about 9.3‰ for δ13C and 7.4‰ for δ15N. These results indicate that the isotopic values of species varied among the lakes, and that they are dependent on isotopic signals from phytoplankton.
Food web; limnology; phytoplankton; energy; stable isotopes
ECOLOGY
Variability of δ13C and δ15N in three zooplankton species from the Upper Paraná River floodplain
Ana R. A. Santana 1 Corresponding author. E-mail: eva@nupelia.uem.br ; Fabio Amodêo Lansac-Tôha; Evanilde Benedito1 1 Corresponding author. E-mail: eva@nupelia.uem.br
Programa de Pós-graduação em Biologia Comparada, Núcleo de Pesquisas em Limnologia, Ictiologia e Aqüicultura, Universidade Estadual de Maringá. Avenida Colombo 5790, Jardim Universitário, 87020-900 Maringá, Paraná, Brasil
ABSTRACT
This study evaluated the isotopic composition of three zooplankton species - Notodiaptomus amazonicus (Wright, 1935), Moina minuta Hansen, 1899 and Bosmina hagmanni Stingelin, 1904 - from the Upper Paraná River floodplain. We predicted that there would be isotopic variability among species in the different sampled environments. Samplings were conducted in three lakes from the Baía subsystem and in two lakes from the Paraná subsystem. At each location, some abiotic variables were measured, and zooplankton (3 to 4) and phytoplankton (3) samples were taken. The species sampled did not present significant differences between subsystems; however, they were different among the lakes, with variations of about 10.8 for δ13C and 5.8 for δ15N. The isotopic values found for phytoplankton also did not differ significantly among sampling stations, presenting variations of about 9.3 for δ13C and 7.4 for δ15N. These results indicate that the isotopic values of species varied among the lakes, and that they are dependent on isotopic signals from phytoplankton.
Key words: Food web; limnology; phytoplankton; energy; stable isotopes.
Ecologists usually quantify the niche through analyses of stomach content and the feeding behavior of consumers (KLING et al. 1992). Recently, many studies have associated the use of stable isotopes (δ13C and δ15N) with the population niche (LEVESQUE et al. 2003, GREY et al. 2004) or with individual differences in diet (POST 2003, BOLNICK et al. 2003). The δ13C content may indicate the carbon sources most used by the species; this is possible because of the enrichment of about 1 δ13C by trophic level between the organism and its diet (VANDER ZANDEN & RASMUSSEN 2001, POST 2002). To add to information generated by the carbon stable isotope analysis, the 15N isotope indicates the species' trophic position because of the enrichment of 15N between the adjacent trophic levels by approximately 3.4 (VANDER-ZANDEN et al. 1997, VANDER-ZANDEN & RASMUSSEN 2001, POST 2002).
Studies using this technique with zooplankton generally consider these organisms to be at the level of higher taxonomical groups. In temperate regions, this technique has been applied to a single species (KLING et al. 1992). These authors defined the structure of the planktonic food chain through isotopic values from algae and zooplankton species in Arctic lakes and, subsequently compared this new structure with the traditional analyses, thus obtaining more accurate results when evaluating this community at the specific level.
In aquatic systems, several factors influence the signals of sources used by zooplankton, including their productivity and respiration (FRANCE et al. 1997), the allochtonous sources of dissolved inorganic carbon (ARAVENA et al. 1992), the composition of particulate organic matter (GREY et al. 2001) and algal species (ZOHARY et al. 1994).
Research performed by LANSAC-TÔHA et al. (2004, 2009) has contributed to the knowledge of the zooplankton fauna from the Upper Paraná River floodplain. Their studies analyzed the composition, richness and abundance of principal plankton species and their relationships to abiotic environmental factors. The most recent studies highlight the variability of isotopic values from the main primary producers (LOPES et al. 2006) and ichthyofauna (LOPES et al. 2007) in this floodplain. However, considering that zooplankton is the principal link in energy transfer to the other trophic levels, it is extremely important to study it using isotopic variability. Besides their high productivity, the floodplains are characterized by the input of organic matter due to the high levels of sedimentation in these environments (JUNK et al. 1989). This input hinders the determination of energetic sources through traditional methods for the organisms dependent on this resource (ARAÚJO-LIMA et al. 1986, ALMEIDA et al. 1993, GERKING 1994).
Therefore, this study investigates the variability of δ13C and δ15N for three zooplankton species, Notodiaptomus amazonicus (Wright, 1935), Moina minuta Hansen, 1899 and Bosmina hagmanni Stingelin, 1904, and for the phytoplankton community occurring in five lakes from the Upper Paraná River floodplain. According to LANSAC-TÔHA et al. (2004), the cladocerans B. hagmanni and M. minuta are species that occur in high densities, whereas the copepod N. amazonicus is representative in its biomass. These species presents a wide range of feeding habits and body sizes, from a few micrometers to some millimeters, which contributes to the structural diversity of the environment (BOZELLI & HUSZAR 2003). The zooplankton are filter-feeding organisms, with a short life cycle and quick reproduction, (especially in the case of cladocerans). Moreover, they are very sensitive to environmental changes. In this paper we test the hypothesis that there is isotopic variability among zooplankton species, in keeping with the isotopic variation observed in phytoplankton in the different environments from the Upper Paraná River floodplain.
MATERIAL AND METHODS
Sampling was undertaken during May 2007, which is the rainy period in the Upper Paraná River floodplain (22º40'-22º50'S, 53º10'-53º40'W). In this season, the highest number of zooplankton taxa is recorded (LANSAC-TÔHA et al. 2004, 2009). Also during this period there is significant input of allochtonous material from the flooded adjacent area, influencing the energy flow of the food chains in floodplains (LANSAC-TÔHA et al. 2004).
The zooplankton was sampled at two subsystems, comprising five sampling stations. For the Baía subsystem, samplings were performed in Lake Porcos, Guaraná and Maria Luiza. In the Paraná subsystem, the Lake Garças and Leopoldo Backwater were sampled (Fig. 1). These environments have distinct features that may influence the structure of the local communities (ROBERTO et al. 2009).
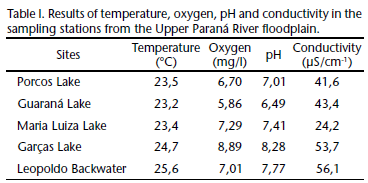
From each sampled location, some abiotic variables were measured; pH (Digimed DM-2 pHmeter), conductivity (Digimed DM-3condutivimeter), temperature (ºC) and dissolved oxygen (YSI oximeter) (Tab. I). At each location, three zooplankton samples were taken using 68-µm plankton nets. Three phytoplankton samples were also taken using 15-µm plankton nets for each location. Samples were filtered in a Kitasato system and retained in sterilized glass fiber filters (Whatman SeS GF 52-C) in a muffle at 450ºC for five hours.
All sampled material was taken to the Laboratories of Zooplankton and Energetic Ecology from the Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (Nupélia) of the Universidade Estadual de Maringá (UEM), where the three zooplankton species were sorted from each sample. From each sample, we separated three to four subsamples of each of the three zooplankton species sorted. The number of individuals by species varied according to the body size, from 100 to 130 individuals for N. amazonicus, 150 to 200 for M. minuta and from 250 to 300 for B. hagmanni (Tab. II).
After this procedure, the subsamples of the three zooplankton species and the filtered phytoplankton samples were dried at 50ºC in a muffle for about 48 hours, and sent to Centro de Isótopos Estáveis, of the Instituto de Biociências from the Universidade Estadual Paulista (Unesp/Botucatu, State of São Paulo), to determine the ratio δ13C/a12C and δ15N/a14N using a mass spectrometer.
Considering that the assumptions of the Analysis of Variance were not violated, the spatial differences in the mean isotopic values () from zooplankton and phytoplankton were tested using the Analysis of Variance of Null Model (IO) with the ECOSIM 7.0 software (GOTELLI & ENTSMINGER 2003). In this analysis, the sampling sites were considered categorical variables. The significance level adopted was 5% probability. The graphical representations were created using STATISTICA 6.0 software.
RESULTS
Abiotic variables
The abiotic parameters showed variations among the sampling stations (Tab. II). The minimum water temperature was registered in the Guaraná Lake, 23.2ºC, and the maximum, 25.6ºC, in Leopoldo Backwater. The values of dissolved oxygen were higher in the Garças Lake (8.89 mg l-1) than in the Guaraná Lake (5.86 mg l-1); in the same way, the pH was slightly acidic in the Guaraná Lake (6.49) and basic in the Garças Lake (8.28). The values of electric conductivity ranged from 24.2 ¼S cm-1 in Maria Luiza Lake to 56.1 ¼S cm-1 in Leopoldo Backwater.
Spatial variation
Grouping the results from the lakes belonging to the Paraná subsystem (Garças Lake and Leopoldo Backwater) and Baía subsystem (Porcos, Guaraná and Maria Luiza lakes), there was no significant difference among the isotopic means of carbon (IO = 1.54, p = 0.69) or nitrogen (IO = 0.0003, p = 1.0) from the three zooplankton species (Fig. 2). The individuals from the three zooplankton species taken from sampling stations belonging to the Paraná subsystems showed the lowest mean both for δ13C (-33.4 ± 4.64) and δ15N (10.7 ± 1.74). Those belonging to the Baía subsystem showed both the highest mean of δ13C (-32.4 ± 3.02) and δ15N (11.2 ± 2.64).
The mean isotopic values observed for the three species showed high variability among the sampling stations, for both carbon and nitrogen (Fig. 3). Nevertheless, for the Baía subsystem, we verified a sequence of isotopic arrangement from the three species. The lower values were observed for N. amazonicus, whereas higher values were recorded for M. minuta. For these subsystems, significantly different isotopic values were also registered (Guaraná Lake: IO = 85.5, p = 0.1; and Maria Luiza Lake: IO = 11.1, p = 0.04).
Intraspecific and interspecific variations of the three zooplankton species
Notodiaptomus amazonicus showed the highest δ13C values in the Garças Lake (-30.5 ± 0.4) (Fig. 3). Meanwhile, in the Leopoldo Backwater, the highest mean was registered for B. hagmanni (-30.4), and the lowest for M. minuta (-39.1 ± 0.45). In the Guaraná Lake, the highest isotopic value (δ13C) was verified for M. minuta (-28.2), and the lowest one for N. amazonicus (-39.0 ± 0.45). Moreover, in the Maria Luiza Lake, the highest isotopic value was also observed for M. minuta (-29.4 ± 0.86), and the lowest one for N. amazonicus (-32.1 ± 0.94), although the highest isotopic value for B. hagmanni (-29.4 ± 0.57) was found in this sampling station.
In general, the higher values of carbon isotope were verified for M. minuta in all locations (-28.2) except for the Leopoldo Backwater (-39.1 ± 0.45) where the lowest mean for this species was registered, whereas the lower values were found for N. amazonicus in all locations (the lowest was -39.0 ± 0.45), except for Garças Lake (-30.4 ± 0.49), where the highest isotopic value was recorded.
Regarding the δ15N, there was no significant differences among species in the Garças Lake (IO = 3.0, p = 0.26) and in the Porcos Lake (IO = 0.92, p = 0.66). However, a significant difference between M. minuta and N. amazonicus was verified in the Leopoldo Backwater (IO = 7.11, p = 0.09) (Fig. 4).
In the Garças Lake and in the Leopoldo Backwater, the highest value δ15N was found in N. amazonicus (11.18 ± 0.91 and 12.78 ± 1.90 , respectively), whereas in the Porcos Lake, the highest isotopic value was found for M. minuta (13.71).
Therefore, the results for δ13C and δ15N did not show a pattern of spatial variation among the three species of zooplankton, simply considering the range of isotopic variation for the sampling stations.
Spatial variation of phytoplankton
The isotopic analyses evidenced that the phytoplankton was significantly different among the sampling stations, considering the mean values of δ13C (IO = 33, p < 0.05). The highest mean was registered in Garças Lake (-27.1 ± 1.80), while the lowest one was observed in the Leopoldo Backwater (-36.4 ± 1.3).
For the phytoplankton, the δ15N results by sampling station were significantly different among sampled environments (IO = 21.4, p = 0.001). The highest mean value was registered in Leopoldo Backwater (11.1 ± 1.13), followed by Garças Lake (10.4 ± 1.39). The lowest value (3.7 ± 0.75) was recorded in the Guaraná Lake (Fig. 4).
DISCUSSION
The limnological features of floodplain lakes are determined by local and regional processes (THOMAZ et al. 2004). In the present study, there was physical and chemical variation in all factors tested among the sampled lakes except the temperature, a fact previously found by THOMAZ et al. (1997). The isotopic composition of the three zooplankton species analyzed here was significantly different among the five studied environments. Nevertheless, for the lakes, which belonged to Baía and Paraná subsystems, there were no differences for either δ13C or δ15N among the zooplankton species. The zooplankton community may change both its food resources, through interactions with abiotic gradients and ecological processes (MATTHEWS & MAZUMDER 2005), and the selection and spatial exploitation of habitats (MATTHEWS & MAZUMDER 2006).
Variables such as pH and dissolved oxygen may be limiting factors in some environments. Research carried out by BONECKER et al. (1998) in Guaraná Lake found that some taxa were not found in adverse conditions - for example in acidic and hypoxic conditions. In the present study, the Guaraná Lake also showed similar low pH and low levels of dissolved oxygen (Tab. II). It is possible that these factors were part of the reason for the low number of individuals observed in this sampling station, resulting in few isotopic determinations.
Among such environmental factors, there is evidence that the temperature may influence isotopic fractioning (BOSLEY et
al. 2002, OLIVE et al. 2003). A study performed with Daphnia magna Straus, 1820 and Hyalella sp. indicated a negative relationship between δ13C and δ15N through the temperature gradient. In this way, when an increase in temperature was observed, there was an increase in the values of the δ13C and a decrease in the values of the δ15N (POWER et al. 2003). In the present study, although the temperature has been similar in the different sampling sites the set of abiotic factors may have influenced the isotopic variability and fractioning found among the species in the different environments. In this sense, the mean isotopic signals ranged from -39.1 to -27.1 for carbon and from 3.7 to 13.7 for nitrogen.
The zooplankton communities in the lakes suffer from the constant changes in the environment, e.g., with hydrological cycles, since the different seasons may increase or decrease primary production, influence nutrient input from the sediment and from the water column mixture, and affect the depths of these lakes (TRAIN & RODRIGUES 1998, THOMAZ et al. 2004). Consequently, the zooplankton assemblages recorded in these environments also are different (LANSAC-TÔHA et al. 2004).
We must also consider that in this study, the species sampled showed a great range of isotopic variation among the environments, reaching -10.9 for M. minuta. In the latter case, it is possible that the species is using a variety of food resources. MATHEWS & MAZUMDER (2006) stated that the interactions between the abiotic gradients and the ecological processes may cause changes in relation to food resources for zooplankton, since these organisms may explore both autochthonous and allochtonous resources, according to their adaptability to the habitat. On the other hand, it is possible that species is using the same resource, but obtaining different nutritional value. It is very complicated to find these isotopic values due to the difficulties of separating the algae from the other components associated with phytoplankton. This hinders the determination of an actual isotopic signal for organisms dependent on this resource (MICHENER & SCHELL 1994, FINLAY 2001, HESSEN et al. 2003). The omnivore N. amazonicus is a selective filter-feeder. It ingests mainly phytoplankton and occasionally detritus, whereas M. minuta and B. hagmanni, also filter-feeding species, ingest particles of varied sizes, preferentially nanoplankton, in addition to detritus (MARGALEF 1983, REYNOLDS 1984).
We must emphasize that there was no pattern of minimum and maximum values in the variation of δ13C and δ15N afound in the phytoplankton relative to their environments. KLING et al. (1992) observed that, although the species composition was similar in eight Arctic lakes, the structure of the food chain determined by the stable isotopes technique also varied among the environments. Moreover, they verified that the absolute isotopic value is dependent on the bottom of the food chain. The authors mention the difficulty of estimating this value, coming as it does from the combination of food resources from several origins. Thus, it is possible that temperature is not a limiting factor for the isotopic variation pattern of zooplankton in distinct ecosystems. Furthermore, BONN & BUNN (1994) and FINLAY (2001) state that the spatial variations in the isotopic composition from primary producers must be always considered in order to avoid misinterpretations about aquatic trophic structures. The high variability in δ13C values from algae and consumers confounds the need for reliable values to use for accurate application of isotopic methods in food chains (FINLAY 2001).
In the sampling sites of the present study, the null hypothesis that isotopic variability among three zooplankton species and the phytoplankton community varies isotopically in the different environments from the Upper Paraná River floodplain can be accepted. Therefore, studies on trophic dynamics of zooplankton communities must consider spatial variations of the different species because each one has distinct ecological requirements in different environments.
In summary, the results of this study emphasize the need to investigate the complexity of zooplankton dynamics through stable isotopes. Meanwhile, our study evidenced high isotopic variation for zooplankton and phytoplankton, highlighting that research related to trophic structure in environments subjected to constant changes should be carefully developed.
ACKNOWLEDGEMENTS
We thank to CNPq/Peld, and UEM/Nupelia/PGB for financial and logistic support.
LITERATURE CITED
Submitted: 14.I.2009; Accepted: 11.XII.2009.
Editorial responsibility: Paulo da Cunha Lana
- ALMEIDA, V.L.L.; E.K. RESENDE; M.S. LIMA & C.J.A. FERREIRA. 1993. Dieta e atividade alimentar de Prochilodus lineatus (Characiformes, Prochilodontidae) no Pantanal do Miranda-Aquidauana, Mato Grosso do Sul, Brasil. Revista Unimar 15: 125-141.
- ARAÚJO-LIMA, C.A.R.M.; B.R. FORSBERG; R. VICTORIA, R. & L.A. MARTINELLI. 1986. Energy sources for detritivorous fishes in the Amazon. Science 234: 1256-1258.
- ARAVENA R.; S.L. SCHIFF; S.E. TRUMBORE; P.J. DILLON & R. ELGOOD. 1992. Evaluating dissolved inorganic carbon cycling in a forested lake watershed using carbon isotopes. Radiocarbon 34: 636-645.
- BOLNICK, D.; R. SVANBACK; J. FORDYCE; L. YANG; J. DAVIS; C. HULSEY & C. FORISTER. 2003. The ecology of individuals: incidence and implications of individual specialization. The American Naturalist 161: 1-28.
- BONECKER, C. C.; F. A. LANSAC-TÔHA & D. C. ROSSA. 1998. Planktonic and non-planktonic rotifers in two environments of the Upper Paraná River Floodplain, State of Mato Grosso do Sul, Brazil. Brazilian Archives of Biology and Technology, 41: 447-456.
- BONN, P.I. & S.E. BUNN. 1994. Variations in the stable isotope composition of aquatic plants and their implications for food webs analysis. Aquatic Botany 48: 99-108.
- BOSLEY, K.L.; D. A. WITTING; R.C. CHAMBERS & S.C. WAINRIGHT. 2002. Estimating turnover rates of carbon and nitrogen in recently metamorphosed winter flounder Pseudopleuronectes americanus with stable isotopes. Marine Ecology Progress Series 236: 233-240.
- BOZELLI, R.L., HUSZAR, V.L.M. 2003. Comunidades fito e zooplanctônicas continentais em tempo de avaliação. Limnotemas Sociedade Brasileira de Limnologia. Available online at: http://ecologia.icb.ufmg.br/~rpcoelho/RioDoce/website/pt_fabricia.htm [Acessed: 29/I/2008]
- FINLAY, J.C. 2001. Stable-Carbon-isotope ratios of river biota: implications for energy flow in lotic food webs. Ecology 82: 1052-1064.
- FRANCE R.L.; P.A. DEL GIORGIO & K.A. WESTCOTT. 1997. Productivity and heterotrophy influences on zooplankton δ13C in northern temperate lakes. Aquatic Microbial Ecology 12: 85-93.
- GERKING, S.D. 1994. Feeding ecology of fish. San Diego, Academic Press, 416p.
- GOTELLI, N.J. & G.L. ENTSMINGER. 2003. EcoSim: Null models software for ecology. Available online at: http://homepages.together.net/~gentsmin/ecosim.htm [Acessed: 09/X/2007]
- GREY J.; R.I. JONES & D. SLEEP. 2001. Seasonal changes in the importance of the source of organic matter to the diet of zooplankton in Loch Ness, as indicated by stable isotope analysis. Limnology and Oceanography 46: 505-513.
- GREY, J.A.; A. KELLY; S. WARD; N. SOMMERWERK & R.I. JONES. 2004. Seasonal changes in the stable isotope values of lake-dwelling chironomid larvae in relation to feeding and life cycle variability. Freshwater Biology 49: 681-689.
- HESSEN, D.O.; T.P. ANDERSEN; P. BRETTUM & B.A. FAAFENG. 2003. Phytoplankton contribution to sestonic mass and elemental ratios in lakes: implications for zooplankton nutrition. Limnology and Oceanography 48: 1289-1296.
- JUNK, W.J.; P.B. BAYLEY & R.E. SPARKS. 1989. The flood pulse concept in River Foodplains Systems. Fisheries and Aquatic Sciences 106: 110-127.
- KLING, W.G.; B. FRY & W.J. O'BRIEN. 1992. Stable isotopes and planktonic trophic structure in arctic lakes. Ecology 73: 561-566.
- LANSAC-TÔHA, F.A.; C.C. BONECKER & L.F.M. VELHO. 2004. Composition, Species Richness and Abundance of the Zooplankton Community, p. 145-190. In: S.M. THOMAZ; A.A. AGOSTINHO & N.S. HAHN (Eds). The Upper Paraná River and its floodplain: physical aspects, ecology and conservation. Leiden, Backhuys Publishers, XXII+393p.
- LANSAC-TÔHA, F.A.; C.C. BONECKER; L.F.M. VELHO; N.R. SIMÕES; J.D. DIAS; G.M. ALVES & E.M. TAKAHASHI. 2009. Biodiversity of zooplankton communities in the Upper Paraná River floodplain: interannual variation from long-term studies. Brazilian Journal of Biology 69: 539-549.
- LEVESQUE, C.; S.K. JUNIPER & J. MARCUS. 2003. Food resource partitioning and competition among alveinellid polychaetes of Juan de Fuca Ridge hydrothermal vents. Marine Ecology Progress Series 246: 173-182.
- LOPES, C.A., E. BENEDITO-CECILIO; L.A. MARTINELLI & M.C. SOUZA. 2006. Variability of δ13C and δ15N in terrestrial and aquatic sources in the upper Paraná river basin. Acta Limnologica Brasiliensia 18: 281-292.
- LOPES, C.A.; E. BENEDITO-CECILIO & L. A. MARTINELLI. 2007. Variability in the carbon isotope signature of Prochilodus lineatus (Prochilodontidae, Characiformes) a bottom-feeding fish of the Neotropical region. Journal of Fish Biology 70: 1649-1659.
- MATTHEWS, B. & A. MAZUMDER. 2005. Temporal variation in body composition (C:N) helps explain seasonal patterns of zooplankton δ13C. Freshwater Biology 50: 502-515.
- MATTHEWS, B. & A. MAZUMDER. 2006. Habitat specialization and the exploitation of allochthonous carbon by zooplankton. Ecology 87: 2800-2812.
- MARGALEF, R. 1983. Limnología. Barcelona, Ediciones Omega, 1010p.
- MICHENER, R.H. & D.M. SCHELL. 1994. Stable isotope ratios as tracers in marine aquatic food webs, p. 138-157. In: H. LAJTHA & R.H. MICHENER (Eds). Stable Isotopes in Ecology and Environmental Science. Oxford, Blackwell Scientific, 316p.
- OLIVE, P.J.W.; J.K.PINNEGAR; N.V.C. POLUNIN; G. RICHARDS & R.WELCH. 2003. Isotope trophic-step fractionation: a dynamic equilibrium model. Journal of Animal Ecology 72: 608-617.
- POST, D.M. 2002. Using stable isotopes to estimate trophic position: Models, methods, and assumptions. Ecology 83: 703-71.
- POST, D.M. 2003. Individual variation in the timing of ontogenetic niche shifts in largemouth bass. Ecology 84: 1298-1310.
- POWER, M.; K. GUIGUER & D.R. BARTON. 2003. Effects of temperature on isotopic enrichment in Daphnia magna: implications for aquatic food-web studies. Rapid Communications in Mass Spectrometry 17: 1619-1625.
- REYNOLDS, C.S. 1984. The ecology of freshwater phytoplankton. Cambridge, Cambridge University Press, 384p.
- ROBERTO, M.C.; N.F. SANTANA & S.M. THOMAZ. 2009. Limnology in the upper Paraná River floodplain: large - scale spatial and temporal and influence of reservoirs. Brazilian Journal of Biology 69: 717-725.
- THOMAZ, S.M.; M.C. ROBERTO & L.M. BINI. 1997. Caracterização limnológica dos ambientes aquáticos e influências dos níveis fluviométricos, p. 73-102. In: A.E.A.M. VAZZOLER; A.A. AGOSTINHO & N.S. HAHN (Eds). A planície de inundação do alto rio Paraná. Maringá, Eduem, 460p.
- THOMAZ, S.M.; T.A. PAGIORO; L.M. BINI; M.C. ROBERTO & R.R.A. ROCHA. 2004. Limnological characterization of the aquatic environments and the influence of hydrometric levels, p. 75-102. In: S.M. THOMAZ; A.A. AGOSTINHO & N.S. HAHN (Eds). The Upper Paraná River and its Floodplain: Physical aspects, Ecology and Conservation. Leiden, Backhuys Publishers, XXII+393p.
- TRAIN, S. & L.C. RODRIGUES. 1998. Temporal fluctuations of the phytoplankton community of the Baía River, in the upper Paraná River floodplain, Mato Grosso do Sul, Brazil. Hydrobiologia 361: 125-134.
- VANDER ZANDEN, M.J. & J.B. RASMUSSEN. 2001. Variation in δ15Nand δ13C trophic fractionation: Implications for aquatic food web studies. Limnology and Oceanography 46 (8): 2061-2066.
- VANDER-ZANDEN, M.J.; G. CABANA & J.B. RASMUSSEM. 1997. Comparing trophic position of freshwater fish calculated using stable nitrogen isotope ratios (δ15N) and literature dietary data. Canadian Journal Fisheries and Aquatic Sciences 54: 1142-1158.
- ZOHARY T.; J. EREZ; M. GOPHEN; I. BERMAN-FRANK & M. STILLER. 1994. Seasonality of stable carbon isotopes within the pelagic food web of Lake Kinneret. Limnology and Oceanography 39: 1030-1043.
Publication Dates
-
Publication in this collection
15 Jan 2010 -
Date of issue
Dec 2009
History
-
Accepted
11 Dec 2009 -
Received
14 Jan 2009