Abstract
Parastacus defossus Faxon, 1898 is a fossorial crayfish species, which constructs its burrows in swampy areas in southeast Uruguay and in the state of Rio Grande do Sul, Brazil. The present field study was carried out in Lami, Porto Alegre, Rio Grande do Sul, Brazil, from May 2003 through August 2005. Environmental measurements (temperature, pH, dissolved oxygen, and water-table depth) of the water in the galleries were made monthly. Burrow morphology was analyzed by means of resin and gypsum casts. The spatial distribution and changes in the number and shape of the surface openings over time were observed in a 100 m² area. To estimate the spatial distribution of the openings, the observed distribution was compared with the expected distribution predicted by the Poisson and Negative Binomial frequency-distribution models. The adult population density was estimated by direct observation of burrows and counts in the study area. Inside the burrows of P. defossus, the water temperature ranged between 16.6°C (autumn 2004) and 23°C (spring 2003). The water was hypoxic and slightly acidic, and the dissolved oxygen content was very low (mean 1.43 mg/l) (18.2% saturation). The soil with burrows had higher percentages of coarse sand, fine sand, and silt. The spatial distribution of the openings showed a significant fit to the Negative Binomial distribution, indicating that the distribution of the openings is aggregated, as confirmed from the burrow morphology. The galleries are always formed by a central tunnel with multiple branchings that connect the underground water to the soil surface by one or more openings, which can be recognized by the chimneys. From knowledge of the burrow morphology, the population density was estimated to be about 120 individuals/100 m².
Burrow morphology; crayfish; galleries
BIOLOGY
Programa de Pós-Graduação em Biologia Animal, Departamento de Zoologia, Universidade Federal do Rio Grande do Sul. Avenida Bento Gonçalves 9500, prédio 43435, sala 214, 91540-000 Porto Alegre, Brazil. E-mail: clarissanoro@hotmail.com
ABSTRACT
Parastacus defossus Faxon, 1898 is a fossorial crayfish species, which constructs its burrows in swampy areas in southeast Uruguay and in the state of Rio Grande do Sul, Brazil. The present field study was carried out in Lami, Porto Alegre, Rio Grande do Sul, Brazil, from May 2003 through August 2005. Environmental measurements (temperature, pH, dissolved oxygen, and water-table depth) of the water in the galleries were made monthly. Burrow morphology was analyzed by means of resin and gypsum casts. The spatial distribution and changes in the number and shape of the surface openings over time were observed in a 100 m2 area. To estimate the spatial distribution of the openings, the observed distribution was compared with the expected distribution predicted by the Poisson and Negative Binomial frequency-distribution models. The adult population density was estimated by direct observation of burrows and counts in the study area. Inside the burrows of P. defossus, the water temperature ranged between 16.6°C (autumn 2004) and 23°C (spring 2003). The water was hypoxic and slightly acidic, and the dissolved oxygen content was very low (mean 1.43 mg/l) (18.2% saturation). The soil with burrows had higher percentages of coarse sand, fine sand, and silt. The spatial distribution of the openings showed a significant fit to the Negative Binomial distribution, indicating that the distribution of the openings is aggregated, as confirmed from the burrow morphology. The galleries are always formed by a central tunnel with multiple branchings that connect the underground water to the soil surface by one or more openings, which can be recognized by the chimneys. From knowledge of the burrow morphology, the population density was estimated to be about 120 individuals/100 m2.
Key words: Burrow morphology; crayfish; galleries.
Members of several groups of decapod crustaceans live in subterranean habitats, from marine intertidal zones to semiterrestrial environments. Among the diverse functions attributed to decapod burrows, the principal function in semiterrestrial species is protection from adverse environmental conditions, such as temperature and humidity extremes, and guaranteed access to water (NASH et al. 1984, ATKINSON & TAYLOR 1988, SHIMODA & TAMAKI 2004).
In freshwater crayfishes, the burrowing habit is known in the superfamilies Astacoidea and Parastacoidea. Crayfishes belonging to Parastacidae occur in Oceania (Australia, Tasmania, New Zealand, and New Guinea), Madagascar, and South America. They live in a wide variety of habitats and are among the best-adapted members of the group to adverse environmental conditions (HOGGER 1988, RICHARDSON 1983, RUDOLPH 1997, BUCKUP 1999). Although the majority of species are confined to the margins of streams and lakes, some are found in peat bogs and clayey soils at long distances from surface water bodies (RICHARDSON 1983, BUCKUP 1999).
Different species of Parastacidae may burrow to a greater or lesser extent, and their burrows can vary in structure, from simple and unbranched to complex systems of burrows (LAKE & NEWCOMBE 1975, SUTER & RICHARDSON 1977, RICHARDSON & SWAIN 1980, HORWITZ & KNOOT 1983, HORWITZ et al. 1985a,b, HORWITZ & RICHARDSON 1986, GROWNS & RICHARDSON 1988).
According to RIEK (1972), the species of Parastacidae can be separated into two distinct ecological groups. Primary burrowers permanently inhabit subterranean waters, live in complex burrows, and show a moderate to strong burrowing habit. Secondary burrowers inhabit lotic or lentic waters and show weak burrowing activity.
In South America, there are representatives of two of the ecological groups proposed by RIEK (1972). Samastacus spinifrons (Philippi, 1882) inhabits rivers and lakes in Chile and Lake Nahuel-Huapi in Argentina (MANNING & HOBBS 1977), and is a secondary burrower. Virilastacus araucanius (Faxon, 1914) and the eight species of Parastacus that inhabit subterranean waters in southern Brazil, Uruguay, and south-central Chile (BUCKUP & ROSSI 1980, 1993, RUDOLPH 1997, BUCKUP 1999) and Virilastacus rucapihuelensis Rudolph & Crandall, 2005, a recently described species collected in burrows in the province of Osorno, Chile (RUDOLPH & CRANDALL 2005), are primary burrowers.
The sparse information on subterranean habitations of species in South America is limited to the work of KILIAN (1959) and RUDOLPH (1997) on the Chilean species Parastacus nicoleti (Philippi, 1882). For the Brazilian species, BUCKUP & ROSSI (1980) described the morphology of a gallery of P. pilimanus (von Martens, 1869), but for the other species there are only references to the localities where specimens were collected.
Parastacus defossus Faxon, 1898 is a small species that spends its entire life cycle in burrows and has been recorded only in Brazil (eastern and southeastern Rio Grande do Sul) (BUCKUP & ROSSI 1980) and in southeastern Uruguay (ZOLESSI & PHILIPPI 1995). It generally lives far from open water, preferring muddy fields, where it constructs its burrows, which reach the level of the water table (BUCKUP 1999, BUCKUP & ROSSI 1980, 1993). The breeding season of the species occurs in spring. The maximum size recorded by NORO et al. (2008) was 32.42 mm cephalothorax length.
Because of the small number of studies on bio-ecological characteristics of the Brazilian species, the present investigation describes the subterranean habitations and the environmental conditions in which the fossorial species P. defossus lives.
MATERIAL AND METHODS
The field study was carried out on a privately owned farm located in Lami (30°11'41"S, 51°06'00"W), municipality of Porto Alegre, state of Rio Grande do Sul, Brazil, from May 2003 through August 2005. In this area, P. defossus constructs subterranean tunnels, not necessarily near surface water, preferring swampy areas where the water table remains near the surface nearly year-round.
Each month, three burrow systems were randomly chosen in the study area, and temperature, pH, and dissolved oxygen were measured (with a portable thermo-oximeter and a pH meter) in the burrow water. The measurements were made 10 cm below the burrow openings. During the summer months (January, February, March) of 2004 and 2005, measurements could not be made because the extreme drought in this period sharply lowered the groundwater level,and the measuring instruments could not be used. To monitor the variation of the water table in the soil, a 1.5 m-deep piezometer was set up. Soil samples were collected in the crayfish burrows and adjacent areas, once during the field study, for granulometric analysis.
To investigate the morphology of the burrow systems, we used three different materials to obtain burrow casts: gypsum, polyurethane foam, and polyester resin. Gypsum was used in four galleries. After approximately 48 hours, the casts were carefully excavated. Gypsum produces good casts but is also fragile and easily breakable, so that it was impossible to dig out complete casts of the burrows. Polyurethane foam was tested in one burrow, but did not give good results. The reaction with the chemical reagent in this material is so rapid that the hardened foam is produced before the primary liquid can completely fill the burrow system, resulting in an incomplete cast. Polyester resin produced far superior casts. We used non-saturated polyester resin (Polyte) with its appropriate catalyst (Methyl-Ethyl-Acetone Peroxide) (Brasnox DM50) in three galleries. The resulting casts were very robust and could be removed in one piece.
Spatial distribution and changes in the number and shape of the surface burrow openings were monitored seasonally, from August 2003 through August 2004. An area of 100 m2 was delimited and divided into 100 quadrats of 1 m2, with a nylon rope. In each season, all openings in each quadrat were located and plotted, and the status (sealed or unsealed) was recorded. The dispersion pattern of the openings was examined by comparing the observed frequency distribution of openings with the expected distribution, as described by the Poisson Distribution and Negative Binomial Distribution. Frequencies of burrows per quadrat predicted by the Poisson and Negative Binomial distributions were calculated following POOLE (1974). Goodness of fit was tested using chi-square.
The adult population density was calculated by direct observation, in a 100 m2 area. In this area, the number of burrows was estimated by directly counting the openings. From previous knowledge of the structure of the burrows and the number of individuals that inhabit each system (maximum two adults), we could estimate the mean number of individuals per burrow, and from this the population density.
RESULTS
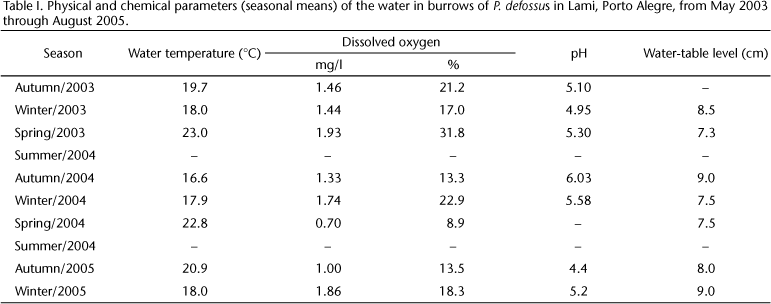
In the burrows of P. defossus, the water temperature ranged between 16.6°C (autumn 2004) and 23°C (spring 2003). The water was hypoxic and slightly acidic. The dissolved-oxygen content was very low (mean 1.43 mg/l) (18.2%), minimum 0.7 mg/l, maximum 1.93 mg/l. The pH varied little during the year, averaging 5.2 (Tab. I).
In autumn and winter, the soil became completely saturated or flooded (9 cm above the soil surface). In summer, the depth of the water table sank to as much as 1.5 m. Analysis of the soil of burrowed and non-burrowed areas showed no differences in the percentages of coarse sand, fine sand, and silt (Tab. II).
In the study area (100 m2), the mean number of burrow openings was 2.37/m2. The Poisson distribution model did not fit the observed frequencies, and therefore the hypothesis that the burrows are randomly distributed was rejected. The Negative Binomial distribution had a significant fit (α = 0.01) (winter 2003: χ2 = 18.34; spring 2003: χ2 = 20.92; summer 2004: χ2 = 23.47; autumn 2004: χ2 = 27.0; winter 2004: χ2 = 20.74), indicating that the distribution of the openings is aggregated, as confirmed from the burrow morphology. A burrow is always formed by a central tunnel with branches that open onto the surface.
A high proportion of burrow openings were sealed during summer and autumn (96.7 and 95.3% respectively). Unsealed openings were more frequent in winter (26.7%) and spring (11.6%). The construction of new chimneys by the crayfish was more frequent in spring 2003, when 118 new chimneys were recorded. Construction of new chimneys was positively correlated with the rainy season.
Analysis of the gypsum and resin casts indicated that the burrows of P. defossus may reach 1.15 m in depth, with a length of 1.50 m. Horizontally, the area occupied by a burrow system may reach 1.44 m2. The burrows usually consist of several inclined tunnels that connect the underground water to the soil surface (Figs 1-5). The galleries open on the surface by one or more openings that can be recognized by the conical towers (chimneys) formed by deposition of sediment removed by the crayfish during the burrowing process. The chimneys can reach 20 cm high and number 5 to 12. From the surface, inclined tunnels with a mean diameter of 8 to 12 cm descend. At 30 cm beneath the surface, these secondary tunnels connect to the larger main tunnel, which may be 11 to 16 cm in diameter, and this tunnel descends vertically or obliquely until it reaches the water table. The burrow ends in an enlarged portion, known as the habitation chamber, which is always at the level of the water table (Figs 1-5).
During the field study, a maximum of two adult individuals of P. defossus were found per burrow (not including the reproductive season, when juveniles were present together with adults in the same burrow). From knowledge of the burrow morphology, the population density could be estimated at 120 adult specimens/100 m2.
DISCUSSION
The area inhabited by P. defossus consists, in geological terms, of recent sedimentary deposits, mainly sand with some fine sand and clay (PRINTES 2002). In this area, P. defossus, as noted by other investigators (FAXON 1898, BUCKUP & ROSSI 1980), construct their burrows far from open water bodies, preferring muddy fields where the underground water remains close to the surface for most of the year.
In the present study, the soil-texture analysis showed no apparent differences between soils where crayfish were burrowing and adjacent areas. The Chilean species Parastacus nicoleti occurs in areas with high percentages of fine sand (RUDOLPH 1997) and the Australian Engaeus tuberculatus Clark, 1936 and E. urostrictus Riek, 1969 prefer soils with high percentages of sand (40.6 and 47.6%), clay (34.8 and 29.5%), and silt (21.3 and 18.1%), respectively (HORWITZ et al. 1985a). In contrast. in the study area, the most important factors in the distribution of P. defossus seem to be soil moisture and the level of the underground water, rather than the soil texture.
The water in the burrows of P. defossus is hypoxic and slightly acidic, and with a marked seasonal oscillation in the water-table level. The mean annual pH in the environment of the Chilean species P. nicoleti is about 6.1 (RUDOLPH 1997), and an acidic environment seems to be common for many freshwater crayfishes. In Tasmania, NEWCOMBE (1975) observed that a pH of 4.5 is typical in the galleries of Parastacoides tasmanicus (Erichson, 1846), although pH values below 2.75 are lethal for this species.
Some of the physical and chemical factors from the burrow water seem to be directly related to precipitation. During the autumn and spring, the water table remained near the surface, or the soil became completely saturated or flooded. In this period, the water temperature inside the burrows varied little, and these oscillations may be even milder in deeper regions of the burrow. During the dry months in summer and early autumn, the level of the water table declined sharply, making it impossible to access the water inside the burrows. In these dry conditions, we presume that dissolved oxygen and pH reached minimum values, as observed elsewhere by LAKE & NEWCOMBE (1975) and RUDOLPH (1997). Because of this, the summer season seems to be a period of stress for the crayfish. The burrows serve an essential function in the biology of many crayfishes, mainly in species that live far from open water bodies, because the burrows function as a refuge against extreme environmental conditions and provide the only access to water.
In spite of the advantages of the burrows, the low oxygen concentration in the water seems to be a common problem for all burrowing crayfishes. As in the present study with P. defossus, low oxygen concentrations have also been recorded in burrows of Australian parastacids (LAKE & NEWCOMBE 1975, SUTER & RICHARDSON 1977, HORWITZ et al. 1985a,b), Chilean parastacids (RUDOLPH 1997), and the North American cambarid Fallicambarus devastator Hobbs & Whiteman,, 1987 (HOBBS & WHITMAN 1991).
Many freshwater crayfishes are able to survive dry periods by remaining in the water retained in the habitation chamber of their burrow; when the water table is very deep, they are also able to remain completely exposed to the air. To deal with these extreme conditions, most burrowing crayfishes have adaptations that permit them to utilize aerial respiration to survive the dry conditions inside the burrow (SUTER & RICHARDSON 1977, GROW & MERCHANT 1980, ATKINSON & TAYLOR 1988, MCMAHON 2002). Both Cherax sp. and Procambarus clarkia (Girard, 1852), although only temporary burrowers, can survive periods in excess of 28 days without any access to water, in conditions of relatively humid air (HUNER 1989).
The small number of open chimneys during the dry summer is likely a strategy to maintain higher humidity inside the burrows and maintain favorable conditions for survival during the dry season. The larger number of newly constructed chimneys observed in spring may be related to a period of greater activity of the animals, which coincides with the reproductive season in October through December (NORO et al. 2008) and with the rainy season.
Parastacus defossus is a strong burrowing species, which constructs burrows with multiple branches. These structures are very similar to the burrow morphology described for other species of parastacids from Oceania and South America (LAKE & NEWCOMBE 1975, SUTER & RICHARDSON 1977, BUCKUP & ROSSI 1980, HORWITZ et al. 1985a,b, RUDOLPH 1997).
Besides the similarity in the burrow morphology, parastacids may show variations in their galleries, depending on the area where they occur. Complex systems have been reported for Cherax destructor Clark, 1936 and for species of Engaeus Erichson, 1846 (SUTER & RICHARDSON 1977, HORWITZ et al. 1985a,b, HOGGER 1988). The burrows of P. tasmanicus can vary from simple structures to many-branched systems (LAKE & NEWCOMBE 1975, HAMR & RICHARDSON 1994). Other species, such as Cherax plebejus (Hess, 1865), construct very simple galleries (HORWITZ & KNOTT 1983). Of the South American species, P. nicoleti (see KILIAN 1959, RUDOLPH 1997) and P. pilimanus (see BUCKUP & ROSSI 1980) construct burrows that are organized similarly to those of P. defossus.
The depth of parastacid burrows is also determined by the water-table depth, and consequently the depth can vary with locality. Galleries 3 m deep have been recorded for C. destructor and E. tuberculatus (HOGGER 1988).
Many authors, in their descriptions of parastacid burrows, have mentioned the presence of "blind chambers" that end among the roots of vegetation, assumed that these areas are used for feeding, and termed them "feeding chambers" (LAKE & NEWCOMBE 1975, RICHARDSON & SWAIN 1980, HORWITZ & KNOTT 1983, HORWITZ et al. 1985a, HAMR & RICHARDSON 1994, RUDOLPH 1997). However, such chambers were not observed in the galleries of P. defossus.
The models proposed by HOBBS (1942) to classify North American crayfishes, and by RIEK (1972) to classify the species of Parastacidae, are both based on their burrowing behavior. In Australia, however, HORWITZ & RICHARDSON (1986) found a group of crayfish that do not conform to any of Hobbs' and Riek's categories. Therefore they proposed a new classification for Australian crayfish burrows: 1) burrows in, or connected to open water; 2) burrows connected to the water table; 3) burrows independent of the water table. However, according to HOBBS (1942) and HORWITZ & RICHARDSON (1986), rarely will a crayfish conform to the definition of a single class of burrowers, because their habits can vary depending on the area where they live. WELCH & EVERSOLE (2006), observing burrowing crayfishes in the southeastern United States, proposed a classification that identifies ecological groups of crayfishes based on their distribution in the landscape in relation to surface water, and within landscape patches in relation to the water table and with respect to the life-history strategies independently of the water table.
According to the classification of HOBBS (1942), P. defossus can be classified as a primary burrower, because it constructs complex burrow systems consisting of an elaborate system of tunnels and one tunnel that extends downward below the water table, and in this burrow the animal spends its entire life. In the classification of HORWITZ & RICHARDSON (1986), the burrows of P. defossus can be classified as Type 2, because its burrows are connected to the underground water. Following the classification scheme of WELCH & EVERSOLE (2006), P. defossus fits the characteristics of a Type 2 primary burrower, by being independent of surface water but limited by requiring a connection to the water table.
LITERATURE CITED
Submitted: 06.X.2009; Accepted: 22.III.2010
Editorial responsibility: Maria Lúcia Negreiros-Fransozo
- ATKINSON, R.J.A. & A.C. TAYLOR. 1988. Physiological ecology of burrowing decapods. Symposium of the Zoological Society of London 59: 201-226.
- BUCKUP, L. 1999. Família Parastacidae, p. 319-327. In: L. BUCKUP & G. BOND-BUCKUP (Eds). Os Crustáceos do Rio Grande do Sul. Porto Alegre, Editora UFRGS, 503p.
- BUCKUP, L. & A. ROSSI. 1980. O gênero Parastacus no Brasil (Crustacea, Decapoda, Parastacidae). Revista Brasileira de Biologia 40 (4): 663-681.
- BUCKUP, L. & A. ROSSI. 1993. Os Parastacidae do espaço maridional andino (Crustacea,Astacidea). Revista Brasileira de Biologia 53 (2): 167-176.
- GROW, L. & H. MERCHANT. 1980. The burrow habitat of the crayfish, Cambarus diogenes diogenes (Girard). The American Midland Naturalist 103: 231-237.
- GROWNS, I.O. & A.M.M. RICHARDSON. 1988. Diet and burrowing habits of the freshwater crayfish, Parastacoides tasmanicus tasmanicus Clark (Decapoda:Parastacidae). Australian Journal of Marine and Freshwater Research 39: 525-534.
- HAMR, P. & A.M.M. RICHARDSON. 1994. Life history of Parastacoides tasmanicus tasmanicus Clark, a burrowing freshwater crayfish from South-western Tasmania. Australian Journal of Marine and Freshwater Research 45: 455-470.
- HOBBS JR, H.H. 1942. The crayfishes of Florida. University of Florida Publications, Biological Science Series 3: 1-79.
- HOBBS, H.H. & M. WHITEMAN. 1991. Notes on the burrow, behavior, and color of the crayfish Fallicambarus devastator (Decapoda: Cambaridae). The Southwestern Naturalist 36 (1): 128-135.
- HOGGER, J.B. 1988. Ecology, Population Biology and Behaviour, p. 114-144. In: D.M. HOLDICH & R.S. LOWERY (Eds). Freshwater Crayfish, Biology, Management and Exploitation. Portland, Timber Press, 685p.
- HORWITZ, P. H. & B. KNOTT. 1983. The burrowing habitat of the koonac Cherax plebeus (Decapoda: Parastacidae). Western Australian Naturalist: 113-117.
- HORWITZ, P.H. & A.M.M. RICHARDSON. 1986. An ecological classification of the burrows of Australian freshwater crayfish. Australian Journal of Marine and Freshwater Research 37: 237-242.
- HORWITZ, P.H.; A.M.M. RICHARDSON & A. BOULTON. 1985a. The burrow habit of two sympatric species of land crayfish, Engaeus urostrictus and E. tuberculatus (Decapoda: Parastacidae). Victoria Naturalist 102: 188-197.
- HORWITZ, P.H.J.; A.M.M. RICHARDSON & P.M. CRAMP. 1985b. Aspects of the life history of the burrowing crayfish, Engaeus leptorhynchus, at Rattrays Marsh, North East Tasmania. Tasmanian Naturalist 82: 1-5.
- HUNER, J.V. 1989. Survival of the red swamp and white river crayfish under simulated burrow conditions. Crayfish Tales 8 (1): 29.
- KILIAN, E.F. 1959. La construcción de los tubos habitacionales del Parastacus nicoleti (Philippi, 1882). Valdivia Facultad de Estudios Generales, Universidad Austral de Chile, 36p.
- LAKE. P.S. & K.J. NEWCOMBE. 1975. Observations on the ecology of the crayfish Parastacoides tasmanicus (Decapoda:Parastacidae) from south-western Tasmania. Australian Zoologist 18: 197-214
- MANNING, R.B. & H.H. HOBBS JR. 1977. Decapoda, p. 157-162. In: S.H. HULBERT (Ed.). Biota Acuática de Sudamérica Austral. San Diego, San Diego State University, 267p.
- MCMAHON, B.R. 2002. Physiological Adaptation to Environment, p. 327-369. In: D.M. HOLDICH (Ed.). Biology of Freshwater Crayfish. Oxford, Blackwell Science, 568p.
- NASH, R.D.M.; C.J. CHAPMAN; R.J.A. ATKINSON & P.J. MORGAN. 1984. Observations on the burrows and burrowing behavior of Calocaris macandreae (Crustacea: Decapoda: Thalassinoidea). Journal of Zoology 202: 425-439.
- NEWCOMBE, K.J. 1975. The pH tolerance of the crayfish Parastacoides tasmanicus (Erichson) (Decapoda, Parastacidae). Crustaceana 29 (3): 231-234.
- NORO, C.K.; L.S. LÓPEZ-GRECO & L. BUCKUP. 2008. Gonad morphology and type of sexuality in Parastacus defossus Faxon 1898, a burrowing, intersexed crayfish from southern Brazil (Decapoda, Parastacidae). Acta Zoologica 88: 59-67.
- POOLE, R.W. 1974. An Introduction to Quantitative Ecology. New York, International Student Edition. 532p.
- PRINTES, R.C. 2002. Plano de Manejo Participativo da Reserva Biológica do Lami. Porto Alegre, SMAM, 102p.
- RICHARDSON, A.M.M. 1983. The effect of the burrows of a crayfish on the respiration of the surrounding soil. Soil Biology and Biochemistry 15: 239-242.
- RICHARDSON, A.M.M. & R. SWAIN. 1980. Habitat requirements and distribution of Engaeus cisternarius and three subspecies of Parastacoides tasmanicus (Decapoda: Parastacidae), burrowing crayfish from an area of south-western Tasmania. Australian Journal of Marine and Freshwater Research 31: 475-484.
- RIEK, E.F. 1972. The phylogeny of the Parastacidae (Crustacea: Astacoidea) and description of a new genus of Australian fresh-water crayfishes. Australian Journal of Zoology 20: 369-389.
- RUDOLPH, E. 1997. Aspectos fisicoquímicos del habitat y morfología de las galerias del camaron excavador Parastacus nicoleti (Philippi, 1882) (Decapoda, Parastacidae) en el sur de Chile. Gayana, Zoología, 61 (2): 97-108.
- RUDOLPH, E. & K.A. CRANDALL. 2005. A new species of burrowing crayfish, Virilastacus rucapihuelensis (Crustacea: Decapoda: Parastacidae), from southern Chile. Proceedings of the Biological Society of Washington 118 (4): 765-776.
- SHIMODA, K. & A. TAMAKI. 2004. Burrow morphology of the ghost shrimp Nihonotrypaea petalura (Decapoda: Thalassinidea: Callianassidae) from western Kyushu, Japan. Marine Biology 44: 723-734.
- SUTER, P.J. & M. RICHARDSON. 1977. The biology of two species of Engaeus (Decapoda: Parastacidae) in Tasmania III. Habitat, food, associated fauna and distribution. Australian Journal of Marine and Freshwater Research 28: 95-103.
- WELCH, S.M. & A.G. EVERSOLE. 2006. The occurrence of primary burrowing crayfish in terrestrial habitat. Biological Conservation 130 (3): 458-464.
- ZOLESSI, L.C. & M.E. PHILIPPI. 1995. Lista sistemática de Decapoda del Uruguay (Arthropoda: Crustacea). Comunicaciones Zoológicas del Museo de Historia Natural de Montevideo 12: 1-24.
The burrows of Parastacus defossus (Decapoda: Parastacidae), a fossorial freshwater crayfish from southern Brazil
Publication Dates
-
Publication in this collection
12 July 2010 -
Date of issue
June 2010
History
-
Received
06 Oct 2009 -
Accepted
22 Mar 2010