Abstract
Ergasilus trygonophilus sp. nov. is described from freshwater stingrays (Potamotrygon spp. and Plesiotrygon iwamae Rosa, Castello & Thorson, 1987) from the state of Pará, Brazil. The new species differs from all known species of Ergasilus Nordman, 1832 from Brazilian waters by possessing: (1) an elongate bullet-shaped cephalosome; (2) antennule setal formula 1: 10: 4: 4: 2 + 1 ae: 6 + 1 ae; (3) maxillule bearing two distal setae; and (4) terminal endopodal segment of leg 1 with rosette-like array of blunt spinules. This is the first species of a freshwater stingray Ergasilus reported from Brazil.
Copepod parasite; Potamotrygon spp; Plesiotrygon iwamae; Neotropical Region
TAXONOMY AND NOMENCLATURE
Ergasilus trygonophilus sp. nov. (Copepoda: Ergasilidae) a branchial parasite of freshwater stingrays (Potamotrygonidae) from state of Pará, Brazil
Marcus V. DominguesI; Taísa M. MarquesII
IInstituto de Estudos Costeiros, Universidade Federal do Pará. Campus Universitário de Bragança, Alameda Leandro Ribeiro, Bairro Aldeia, 68600-000 Bragança, PA, Brazil. E-mail: mvdomingues71@gmail.com
IILaboratório de Ecologia Molecular e Parasitologia Evolutiva, Departamento de Zoologia, Universidade Federal do Paraná. Caixa Postal 19073, 81531-980 Curitiba, PR, Brazil. E-mail: ta_mendes@yahoo.com.br
ABSTRACT
Ergasilus trygonophilussp. nov. is described from freshwater stingrays (Potamotrygon spp. and Plesiotrygon iwamae Rosa, Castello & Thorson, 1987) from the state of Pará, Brazil. The new species differs from all known species of Ergasilus Nordman, 1832 from Brazilian waters by possessing: (1) an elongate bullet-shaped cephalosome; (2) antennule setal formula 1: 10: 4: 4: 2 + 1 ae: 6 + 1 ae; (3) maxillule bearing two distal setae; and (4) terminal endopodal segment of leg 1 with rosette-like array of blunt spinules. This is the first species of a freshwater stingray Ergasilus reported from Brazil.
Key words: Copepod parasite; Potamotrygon spp.; Plesiotrygon iwamae; Neotropical Region.
Species of Ergasilus Nordman, 1832 are parasitic copepods found world-wide in aquatic environments, and are considered an important plague of pisciculture (THATCHER 2006). Only females are found on fish hosts. Males are free-living in the zooplankton. Most species are found on freshwater fishes, but a few infect marine fishes of coastal waters (AMADO et al. 1995, BOXSHALL & HALSEY 2004). LUQUE & TAVARES (2007) reported 26 valid species plus 10 unidentified species of Ergasilus from Brazilian waters, of which 23 are from freshwater hosts and 13 are from brackish or marine hosts. Later, THATCHER & BRASIL-SATO (2008) described Ergasilus salmini Thatcher & Brasil-Sato, 2008 from Salminus franciscanus Lima & Britsky, 2007 (Characidae) from the upper São Francisco River, Brazil.
There are only a few parasites known from Potamotrygo-nidae (DOMINGUES & MARQUES 2007, DOMINGUES et al. 2007, LUCHETTI et al. 2008), limited to seven groups: Acanthocephala, Branchiura, Digenea, Eucestoda, Monogenoidea, Nematoda and Pentasto-mida. According to, the parasite fauna of Potamotrygonidae is unusual because it is represented by typically marine taxa - (Acanthobothrium van Beneden, 1849, Anindobothrium Marques, Brooks & Lasso, 2001, Echinocephalus Molin, 1858, Rhinebothrium Linton, 1890 -; some are found only in Potamotrygonidae (Megapriapus Golvan, 1957, Paraheteronchocotyle Mayes, Brooks & Thorson, 1981, Paraoncomegas Campbell, Marques, & Ivanov, 1999, Potamo-trygonocestus Brooks & Thorson, 1976, Rinebothroides Mayes, Brooks & Thorson, 1981); whereas others are also found in teleosteans (Argulus Mueller, 1785, Dolops Marshall, 1889, Paravitellotrema Watson, 1976, and Terranova Leiper & Atkinson, 1914) and crocodilians (Leiperia Sambon, 1922 and Brevimulti-caecum Mozgovoy, 1952). The fact that potamo-trygonids are infested both by marine and freshwater parasites makes them excellent models for evolutionary and biogeographical studies. The present paper describes the first species of Ergasilus known from Brazilian freshwater stingrays.
MATERIAL AND METHODS
Potamotrygonids were captured with throw nets and long lines in the Igarapé do Urubu, municipality of Cachoeira do Arari (1º00'16.22"S, 48º57'28.8"W) and Marajo Bay, municipality of Colares (0º55'47.22"S, 48º17'30.68"W), state of Pará, Brazil. Copepod parasites were removed from the gill filaments with dissecting needles and fixed in 70% alcohol. They were transported to a laboratory of the Universidade Federal do Paraná, Curitiba, PR, where permanent slide preparations were made using the phenol-balsam method explained in THATCHER (2006). Drawings were prepared with the help of an Olympus BX 50 microscope with DIC optics and a camera lucida. Measurements were made utilizing a measuring ocular and are expressed in micrometers (µm). Basins and sub-basin nomenclature following the Agência Nacional de Águas, Ministério do Meio Ambiente, Brasil (http://hidroweb.ana.gov.br). Type specimens and vouchers were deposited in the Crustacean Collection of the Instituto de Pesquisas da Amazônia (INPA), Manaus, AM, Brazil. Hosts have been deposited at the Museu de Zoologia, Universidade de São Paulo, São Paulo, SP, Brazil (MZUSP).
TAXONOMY
Ergasilus trygonophilussp. nov.
Diagnosis. Based on 10 females studied and measured; measurements in tables I and II. Cephalosome elongate, round anteriorly, longer than wide (bullet-shaped); antennules and antenna visible in dorsal view. Four free pedigers decreasing in width posteriorly; fifth pediger reduced (Fig. 1). Genital complex as wide as long, with a row of spinules on posteroventral margin (Fig. 5). Three free abdominal somites naked (Fig. 5), with a row of spinules on posteriorventral margin; anal somite with posteriorventral row of spinules, slightly indented medially. Caudal rami longer than wide, without ornamentation, each ramus with one long, one medium, and two short setae; principal seta plumose.
Antennule (Fig. 2) with six segments, tapering distally, provided with simple setae, setal formula from proximal to distal segments: 1: 10: 4: 4: 2 + 1 ae: 6 + 1 ae.
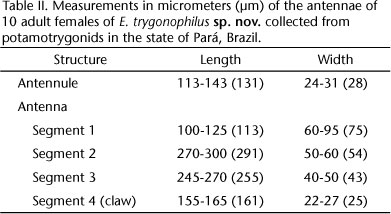
Antenna (Fig. 3) long, slender 4-segmented, comprising a short coxobasis, 3-segmented endopod and slightly recurved claw. Coxobasis without ornamentation; articulation membrane not inflated. Proximal endopodal segment slightly curved, longer than medial and distal endopodal segments combined, armed with short seta near mid-point on projection of inner margin. Medial endopodal segment with short seta near proximal portion on projection of inner margin and one short seta in distal portion of inner margin. Distal endopodal segment incomplete, vestigial, with one proximal seta on anterior surface.
Mandible (Fig. 4) with three blades: anterior delicate with bristles on anterior margin; median blade robust with large teeth on posterior margin; posterior blade falciform with teeth on posterior margin; Maxillule trapezoid, lobate, bearing two outer setae. Maxilla consisting of large syncoxa, with proximal projection, unarmed, tapering distally; and distal projection (small basis), armed distally with sharp teeth.
Legs (Figs 6-8). Legs 1-4 biramous, with rami 3-segmented, except for the first endopod and the fourth exopod, which are 2-segmented. Legs 1-4 with outer margins of both rami ornamented with rows of spinules of variable length outer margin of first and second endopodal segments of leg 4 pilose; inner margin of first exopodal segment of legs 1-4 pilose. Basis of legs 1-3 bearing outer naked setae on posterior surface, near the exopod (Figs 6-7); basis of leg 4 with plumose seta (Fig. 8). Interpodal plates of legs 1-3 with two rows of spinules. Leg 1 (Fig. 6) with first exopodal segment bearing a single spine on outer distal margin; second exopodal segment with a single, median, inner plumose seta; first seta of third exopodal segment falciform, semipinnate, four plumose setae on inner margin, two short plumose setae on outer margin, apical spine serrated on outer margin, plumose on inner margin; first endopodal segment elongate with long plumose seta near distal portion; terminal endopodal segment with a rosette-like array of blunt spinules, first and second setae falciform, semipinnate, five plumose setae on inner margin. Legs 2 and 3 (Fig. 7) similar, first exopodal segment with distal spine on outer margin; second exopodial segment with inner medial plumose seta; third exopodial segment with outer small plumose seta, six plumose setae; first endopodal segment with inner plumose seta near distal portion; second endopodal segment with two inner plumose setae; third endopodal segment with apical spine serrated on outer margin only, four plumose setae. Leg 4 (Fig. 8) without seta or spine on first exopodal segment; terminal exopodal segment with outer short plumose seta, five plumose setae; first endopodal segment with inner plumose seta; second endopodal segment with two inner plumose setae; third endopodal segment with apical spine serrated on outer margin only, three plumose setae (spine and seta formula on table III). Egg sac elongate with 20-30 eggs.
Type-Hosts and type-locality: Holotype and five paratypes, INPA nº 1859, 1860 a-e Potamotrygon scobina Garman, 1913 (Raspy river stingray) Marajo Bay, Municipality Colares, state of Pará, Brazil (0º55'47.22"S, 48º17'30.68"W), 20.VIII.2007 (Host n° MZUSP PA07-05, PA07-26).
Other hosts and localities: five vouchers, INPA nº 1861 a-b, 1862 a-b, 1863, Potamotrygon orbignyi (Castelnau, 1855) (smooth back river stingray) from Marajo Bay, Municipality of Colares, Pará, Brazil, 16.VIII.2007 (host n° MZUSP PA07-10), Plesiotrygon iwamae Rosa, Castello & Thorson, 1987 (long-tailed river stingray) from Marajo Bay, Municipality of Colares, Pará, Brazil, 21.VIII.2007 (host n° MZUSP PA07-38, PA07-47, PA07-48); and Potamotrygon sp. from Igarapé do Urubu, Municipality of Cachoeira do Arari, state of Pará, Brazil (1º00'16.22"S, 48º57'28.8"W), 29.VIII.2007 (host n° MZUSP PA07-86).
Male: Unknown.
Site of infestation: Gill filaments.
Type and voucher specimens: Holotype female and 9 paratype females deposited in the Crustacea Collection of the Instituto Nacional de Pesquisas da Amazônia (INPA), Manaus, AM, Brazil.
Etymology. The specific name comes from potamotrygon = ray and philos = likes, indicating a copepod that likes rays.
Remarks. Ergasilus trygonophilus sp. nov. is similar to E. versicolor Wilson, 1911 (Siluriformes: Ictaliridae) (see ROBERT 1969), E. foresti Boxshall, Araújo & Montú, 2002 (undefined host) (BOXSHALL et al. 2002) and E. youngi Tavares & Luque, 2005 (Siluriformes: Arridae) (TAVARES & LUQUE 2005), in the segmentation and setation formula of the swimming legs (Tab. III), and by having only one seta on the first segment of the antennule. Ergasilus versicolor differs from the new species by having the terminal endopodal segment of leg 1 without a rosette-like array of blunt spinules. Ergasilus trygonophilus sp. nov. seems to be closely related to E. foresti Boxshall, Araújo & Montú, 2002 and E. youngi Tavares & Luque, 2005 by having leg 1 with a terminal endopodal segment with a rosette-like array of blunt spinules. Ergasilus trygonophilus can be distinguished from these two species by having 1) a bullet-shaped cephalosome which is fused to the head and the first two thoracic segments; 2) antennule setal formula 1: 10: 4: 4: 2 + 1 ae: 6 + 1 ae (1: 11: 5: 4: 2 + ae: 7 + ae in E. foresti and 1: 11: 3: 3: 1 + 1 ae.: 6 + 1 ae. in E. youngi); and 3) basis of legs 1-3 naked and leg 4 plumose (basis of legs 1-4 plumose in E. foresti; basis of legs 1-4 naked in E. youngi). OGAWA (1991) reported for the first time a species of Ergasilus from an elasmobranch host, Pristis microdon Latham, 1794 (Pristidae) from Daly river, Australia. However, this species has also been reported from Lates calcifer (Bloch, 1790) (Latidae) collected in the same locality, suggesting low host-specificity for this species. LUQUE & TAVARES (2007) reported 26 species of Ergasilus occurring in freshwater and brackish fishes (Atheriniformes, Characiformes, Perciformes, Siluriformes, Tetraodontiformes) in Brazil. All these species seem to be related to a specific environment (e.g. freshwater or brackish) and host (e.g. host species or at least host family). This pattern can be observed for E. trygonophallus n. sp. which is restricted to brackish waters and is found parasitizing different species of Potamotrygonidae.
ACKNOWLEDGEMENTS
We would like to thank F.P.L. Marques, M. Cardoso Jr and V.M. Bueno (IB-USP) for collecting the specimens on the collecting trip to Marajó; V.E. Thatcher (UFPR) provided important comments and a pre-submission review of this manuscript. This work was supported by a research grant from the Fundação de Amparo à Pesquisa do Estado de São Paulo(FAPESP 03/01816-2 and 05/01299-3) to F.P.L. Marques (IB-USP) and by a post doctoral fellowship to the senior author (FAPESP 04/09267-0).
LITERATURE CITED
Submitted: 11.IX.2009; Accepted: 10.VII.2010.
Editorial responsibility: Marcos D.S. Tavares
- AMADO, M.A.P.; J.S. HO & C.E.F. DA ROCHA. 1995. Phylogeny and biogeography of the Ergasilidae (Copepoda, Poecilostoma-toida), with reconsideration of the taxonomic status of the Vaigamidae. Contributions to Zoology 65: 233-243.
- BOXSHALL, G.A. & S.H. HALSEY. 2004. An introduction to copepod diversity. London, The Ray Society, 966p.
- BOXSHALL, G.A.: H M.P. ARAUJO & M. MONTÚ. 2002. A new species of Ergasilus Nordmann, 1832 (Copepoda, Ergasilidae) from Brazil. Crustaceana 75: 269-276.
- DOMINGUES, M.V. & F.P.L. MARQUES. 2007. Revision of Potamotrygo-nocotyle Mayes, Brooks & Thorson, 1981 (Platyhelminthes: Monogenoidea: Monocotylidae), with descriptions of four new species from the gills of the freshwater stingrays Potamotrygon spp. (Rajiformes: Potamotrygonidae) from the La Plata river basin. Systematic Parasitology 67: 157-174.
- DOMINGUES, M.V.; N.C.M. PANCERA & F.P.L. MARQUES. 2007. Monogenoidean parasites of freshwater stingrays (Rajifor-mes: Potamotrygonidae) from the Negro River, Amazon, Brazil: species of Potamotrygonocotyle (Monocotylidae) and Paraheteronchocotyle (Hexabothriidae). Folia Parasitologica 54: 177-190.
- LUCHETTI, N.M; F.P.L. MARQUES & P. CHARVET-ALMEIDA. 2008. A new species of Potamotrygonocestus Brooks and Thorson, 1976 (Eucestoda: Tetraphyllidea) of Plesiotrygon iwamae Rosa, Castello and Thorson, 1987 (Myliobatoidea: Potamotrygo-nidae) and redescription of Potamotrygonocestus chaoi Marques, Brooks and Araujo, 2003. Systematic Parasitology 70: 131-45.
- LUQUE, J.L. & L.E.R TAVARES. 2007. Checklist of Copepoda associated with fishes from Brazil. Zootaxa 1579: 1-39.
- OGAWA, K. 1991. Ectoparasites of sawfish, Pristis microdon, caught in freshwaters of Australia and Paupa New Guinea. University Museum, the University of Tokyo, Nature and Culture 3: 91-101.
- ROBERT, L. 1969. Ergasilus arthrosis n. sp. (Copepoda: Cyclopoida) and the taxonomic status of Ergasilus versicolor Wilson,1911, Ergasilus elegans Wilson, 1916 and Ergasilus celestas Mueller, 1936, from North American fishes. Journal of Fisheries Research Board of Canada 26: 997-1011.
- TAVARES, L.E.R. & J.L. LUQUE. 2005. Ergasilus youngi sp. nov. (Copepoda, Poecilostomatoida, Ergasilidae) parasitic on Aspistor luniscutis (Actinopterygii, Ariidae) from off the State of Rio de Janeiro, Brazil. Acta Parasitologica 50: 150-155.
- THATCHER, V.E. 2006. Amazon Fish Parasites. Sofia, Pensoft Publishers, 2nd ed., 508p.
- THATCHER, V.E. & M.C. BRASIL-SATO. 2008. Ergasilus salmini sp. nov. (Copepoda: Ergasilidae) a branchial parasite of "dourado", Salminus franciscanus from the upper São Francisco River, Brazil. Revista Brasileira de Zoologia 25: 555-557.
Publication Dates
-
Publication in this collection
23 Nov 2010 -
Date of issue
Oct 2010
History
-
Accepted
10 July 2010 -
Received
11 Sept 2009