Abstract
The freshwater snail Gundlachia ticaga (Marcus & Marcus, 1962) is widely distributed in Brazil, but its morphology has been poorly studied. We compared the shell morphology of samples from four sites (Vila do Abraão, Vila de Provetá, Parnaioca and Praia do Sul) in Ilha Grande (Angra dos Reis, state of Rio de Janeiro) in order to evaluate differences within and among four populations. We used nine morphometric characters representing shell size and shape. To analyze shell morphology we considered aperture shape, sculpture of teleoconch, apex carving and position. The resulting dataset was correlated by Pearson´s linear correlation and shell differences among populations were tested using ANOVA and Discriminant Function Analysis. The results showed that there is significant variation among populations concerning shell shape and morphology. Shells from preserved localities (Praia do Sul and Parnaioca) and shells from highly modified environments (Vila do Abraão and Vila de Provetá) were the most similar to each other. Results from the visual assessment and from the discriminant analysis were almost identical. The shell indices are the most important variables in the differentiation of samples. The observed variation corroborates the hypothesis that G. ticaga displays phenotypic plasticity, which may lead to wrong identifications. Narrower shells with an elongate aperture could be misidentified as Ferrissia Walker, 1903 and, broader shells with a roundish aperture could be wrongly identified as Burnupia Walker, 1912. We confirmed that the absence of radial lines is not a good diagnostic character for G. ticaga. The analysis of the apical micro-sculpture and soft parts is essential for a correct identification.
Phenotypic plasticity; freshwater snails; Mollusca; shell morphology
BIOLOGY
Shell morphology of the freshwater snail Gundlachia ticaga (Gastropoda: Ancylidae) from four sites in Ilha Grande, southeastern Brazil
Luiz Eduardo M. Lacerda; Igor C. Miyahira; Sonia B. Santos* * Corresponding author, E-mail: gundlachia@yahoo.com.br
Laboratório de Malacologia Límnica e Terrestre, Programa de Pós-Graduação em Ecologia e Evolução, Departamento de Zoologia, Universidade do Estado do Rio de Janeiro. Rua São Francisco Xavier 524, PHLC, sala 525/2, Maracanã, 20550-900 Rio de Janeiro, RJ, Brazil
ABSTRACT
The freshwater snail Gundlachia ticaga (Marcus & Marcus, 1962) is widely distributed in Brazil, but its morphology has been poorly studied. We compared the shell morphology of samples from four sites (Vila do Abraão, Vila de Provetá, Parnaioca and Praia do Sul) in Ilha Grande (Angra dos Reis, state of Rio de Janeiro) in order to evaluate differences within and among four populations. We used nine morphometric characters representing shell size and shape. To analyze shell morphology we considered aperture shape, sculpture of teleoconch, apex carving and position. The resulting dataset was correlated by Pearson´s linear correlation and shell differences among populations were tested using ANOVA and Discriminant Function Analysis. The results showed that there is significant variation among populations concerning shell shape and morphology. Shells from preserved localities (Praia do Sul and Parnaioca) and shells from highly modified environments (Vila do Abraão and Vila de Provetá) were the most similar to each other. Results from the visual assessment and from the discriminant analysis were almost identical. The shell indices are the most important variables in the differentiation of samples. The observed variation corroborates the hypothesis that G. ticaga displays phenotypic plasticity, which may lead to wrong identifications. Narrower shells with an elongate aperture could be misidentified as Ferrissia Walker, 1903 and, broader shells with a roundish aperture could be wrongly identified as Burnupia Walker, 1912. We confirmed that the absence of radial lines is not a good diagnostic character for G. ticaga. The analysis of the apical micro-sculpture and soft parts is essential for a correct identification.
Key words: Phenotypic plasticity; freshwater snails; Mollusca; shell morphology.
doi: 10.1590/S1984-46702011000300007
Shell morphometry is a useful tool in mollusc taxonomy and ecology (CIPRIANI 2007). In studies of freshwater snails, it has been used to discriminate between species (WULLSCHLEGER & JOKELA 2002), to recognize intraspecific morphological variation (CHIU et al. 2002), and to associate shell variations with environmental conditions (MACMAHON & WHITEHEAD 1987, WULLSCHLEGER & JOKELA 2002) and geographical distribution (PFENNINGER et al. 2003). The well known uncertainty of freshwater mollusc taxonomy owes to inappropriate descriptions, either of genera or species (BASCH 1963), and to phenotypic plasticity in shell morphology (BASCH 1963, DURRANT 1977, MCMAHON & WHITEHEAD 1987, WULLSCHLEGER & JOKELA 2002), leading to taxonomic inflation (STRONG et al. 2008). Conversely, the absence of shell variability in cryptic species also results in underestimation of true species richness (WALTHER et al. 2006).
Gundlachia ticaga (Marcus & Marcus, 1962) is a basommatophoran without a coiled shell (HUBENDICK 1978), in the family Ancylidae sensu lato, whose members are limpet-shaped and small (3 to 15 mm in length), occurring mainly in lentic environments adhered to aquatic plants, branches and leaves of decayed riparian vegetation, or rocks (LANZER & VEITENHEIMER-MENDES 1985, SANTOS 2003). Gundlachia ticaga is widely distributed, mainly in southeastern (SANTOS 2003) and central-western regions of Brazil (THIENGO et al. 2005). It is the most frequent ancylid in freshwater ecosystems of the state of Rio de Janeiro (THIENGO et al. 1998, 2001, 2002a,b, 2004a,b, 2006), being recently recorded for Praia do Sul Biological Reserve, Ilha Grande (SANTOS et al. 2009).
Despite the wide geographical range of G. ticaga, the regional morphological variation of the species is poorly known. The morphology of G. ticaga has only been described in the original description of the species (MARCUS & MARCUS 1962), and subsequently by OHLWEILER & LANZER (1993), LANZER (1996) and SANTOS (2003). Shell variability may explain the original combination, Uncancylus ticagus Marcus & Marcus, 1962, because a projected apex that curves somewhat to the right is found in some specimens; it may also explain, at least in part, the classification of G. ticaga as a synonym of G. obliqua (Broderip & Sowerby, 1832) (= Anisancylus obliquus) (HUBENDICK 1964). OHLWEILER & LANZER (1993) did not accept this synonymy and revalidated G. ticaga. OHLWEILER & LANZER (1994), LANZER (1996) and SANTOS (2003) also considered G. ticaga as valid, based on characteristics of the shell, soft parts, radula and jaw.
Here we present a characterization of shell variation in G. ticaga. We conducted morphometric analyzes and compared samples from four different freshwater environments in Ilha Grande, southeastern Brazil, to better determine the range of variation and thereby improve species delineation.
MATERIAL AND METHODS
Ilha Grande (23º05'-23º15'S, 44º06'-44º23'W) is a continental island located in Ilha Grande Bay, southeastern Rio de Janeiro, within the Atlantic Rain Forest, a highly diverse and important diversity hot-spot (MYERS et al. 2000). The island has 32 micro-watersheds, most of which have streams flowing through rocky shores into the sea, without areas that favor colonization by mollusks. Ilha Grande island has a long history of human occupation and environmental modifications, beginning in the pre-historical times (PRADO 2006), with increased tourism during the last decade (PRADO 2003).
We sampled between December 2005 and March 2007 in four places: Vila do Abraão (23º08'31"S, 44º10'10"W), Vila de Provetá (23º10'39"S, 44º20'33"W), Parnaioca (23º11'36"S, 44º15'00"W) and Praia do Sul (23º10'30"S, 44º17'55"W, Fig. 1). The lower part of these streams flow through lowlands (< 10 m above sea level) and provide conditions for colonization by freshwater mollusks.
Vila do Abraão, Vila de Provetá and Parnaioca are the most altered localities in Ilha Grande, due to continuous human activities, especially deforestation for subsistence agriculture, constructions, modification of riverbanks and organic pollution. Although Vila de Provetá is a densely populated settlement, it does not have the touristic appeal of Vila do Abraão, the main harbor in Ilha Grande. The latter has many guest houses, small hotels, camping areas, restaurants and pubs, which greatly impact the water bodies of Vila do Abraão. Parnaioca corresponds to the area of the old Parnaioca Farm, which had been gradually deactivated since the establishment of the Ilha Grande Prison in Dois Rios Village, in 1903 (SANTOS 2009). Nowadays, the region has only six inhabitants, and its environment is recovering. In Parnaioca we found ancylids in the small stream next to the access to the Parnaioca-Vila Dois Rios trail. The only three houses in Parnaioca are not very close to the riverbanks, and apparently they do not release sewage into the stream. All studied streams are 1-2 m wide and are in average 10-50 cm deep. They have sand-rocky bottom with mud deposits in some spots and reduced riparian vegetation with a predominance of exotic species (banana, jackfruit and bamboo trees). In Vila do Abraão, walls made of rock or concrete restrain the stream. There are houses very close to the riverbanks, sewage is released into the streams, and sand has been removed in some sites. In Vila de Provetá the riverbanks are not restrained. Downstream there are no houses close to the stream margin and vegetation is sparse, allowing stronger illumination of the riverbed. Sewage is also directly released into the water.
The most preserved stream is located at the Praia do Sul Biological Reserve, one of the last well preserved arboreal restingas (coastal sandy plain vegetation) in the state of Rio de Janeiro (MACIEL et al. 1984, AMADOR 1988), where the integrity of the riparian vegetation has been maintained. The studied stream flows into a lagoon known as Lagoa do Sul; it is 2.5 m wide and 30 cm deep on average, being fairly shaded and having a sand-muddy bottom covered with decomposing leaves.
The specimens studied are housed at the Malacological Collection of the Universidade do Estado do Rio de Janeiro (Col. Mol. UERJ).
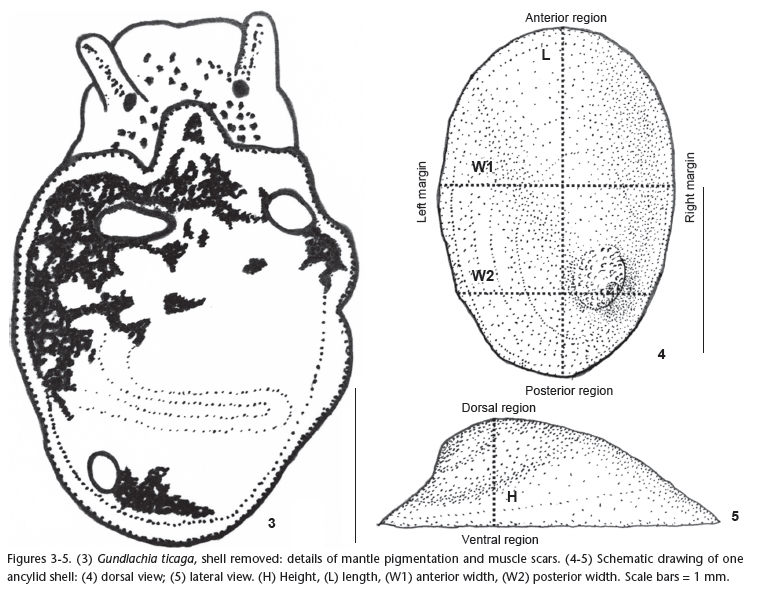
The identification of specimens was carried out using shell teleoconch and protoconch morphology, with emphasis on the apical micro-sculpture (Fig. 2), mantle pigmentation and shape of muscle scars (Fig. 3), according to MARCUS & MARCUS (1962), OHLWEILER & LANZER (1993) and SANTOS (2003).
All shells were drawn using a camera lucida, and classified according to shape of the shell's aperture (oval-elongated or rounded), presence of radial lines, and position of the salient apex in relation to the shell margin.
We measured 171 shells and obtained four linear measurements under a dissecting microscope using a caliper to the nearest
0.05 mm: length (L), height (H), anterior width (W1) and posterior width (W2). We also obtained five morphometric indices of shell shape: W1/L, W2/L, H/L, H/W1 and W2/W1 (Figs 4 and 5).
Measurements were normalized and standardized (KREBS 1998, ZAR 1999). Normality was tested and all variables were loge transformed, except W2/L that was already normal. Morphometric differences among populations were tested by analysis of variance (ANOVA) followed by Tukey pairwise comparison test (significance level = 0.05). Variables were analyzed by Pearson's correlation test and those strongly correlated (r > 0.92) were excluded, in order to ensure independence among variables (KLECKA 1982, ENGELMAN 1997). The Discriminant Function Analysis was carried out in order to test for the level of discrimination of samples, the statistic package SYSTAT 10.2 was used in all statistical procedures.
Studied specimens: Gundlachia ticaga: Brazil, Rio de Janeiro: Angra dos Reis (Ilha Grande, Parnaioca) 11 animals, 10.XII. 2005, Col. Mol. UERJ 7035; (Provetá), 24 animals, 02.V.2006, Col. Mol. UERJ 7055, 36 animals, 03.V.2006, Col. Mol. UERJ 7056; (Praia do Sul), 28 animals, 11.XI.2006, Col. Mol. UERJ 7058, 23 animals, 03.III.2007, Col. Mol. UERJ 7060; (Vila do Abraão), 05 animals, 25.III.2006, Col. Mol. UERJ 7049, 22 animals, 24.III.2006, Col. Mol. UERJ 7050, 22 animals, 28.X.2005, Col. Mol. UERJ 7054. All specimens were collected by L.E.M. Lacerda, I.C. Miyahira & S.B. Santos.
RESULTS
All specimens were identified as G. ticaga based on the following characters: elliptical shell aperture; projected rounded apex on the posterior right quadrant of the shell, flexed to the right, sometimes extended beyond the right margin; protoconch with an apical depression, with a short smooth and flat area followed by an area with irregular punctuations; teleoconch with concentric growth lines; radial lines absent or present; mantle with dark pigmentation, sparse, tending to concentrate in the left side; anterior right elliptical muscle scar, tear drop-shaped; anterior left muscle scar and posterior muscle scar rounded (Fig. 3).
Variation within population
Excluding differences in shell size, which could reflect differences in shell growth, each of the four populations of G. ticaga from Ilha Grande varied in shell aperture shape, shell carving and position and shape of the apex (Figs 6-16, Tab. I). In each sample, we observed a predominant shell pattern comprising 69% of specimens from Vila do Abraão, 70% from Provetá, 88% from Praia do Sul and 55% from Parnaioca (Tab. I). Protoconch sculpture is invariable (Fig. 2), reflecting a consistent taxonomic character.
Variation among populations
The shells from Praia do Sul and Parnaioca (except one) had radial lines on the teleoconch; by contrast, those from Vila do Abraão and Provetá lacked radial lines (Tab. I).
The largest specimens were from Provetá (Tab. II), where one reached 5.7 mm in length, 4.20 mm in width and 1.5 mm in height. Specimens from Provetá and Vila do Abraão were longer than those from Praia do Sul and Parnaioca (Tab. II).
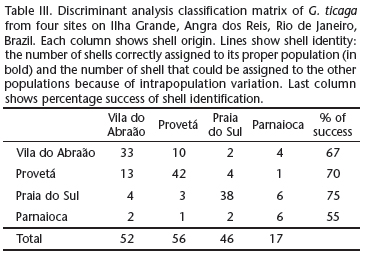
Morphometrical variables differed significantly among the four populations studied, as showed by the ANOVA and the Tukey test (Tab. II). Variables L and W1 (r = 0.983), W1 and W2 (r = 0.947), W2 and L (0,936), and H and L (0,933) were much correlated and therefore the variables L and W1 were removed from the discriminant analysis. The ANOVA results were confirmed by the DA (Wilks' Lambda = 0. 2848; F15,450 = 17.2830; df = 3; p = 0.000). The classification matrix (Tab. III) also corroborated that each population had its proper morphological identity. The F-matrix among groups shows that samples from Vila do Abraão vs Provetá (8.552), Vila do Abraão vs Parnaioca (6.034) and Provetá vs Parnaioca (8.552) are more similar to each other when compared with those from Praia do Sul, the most isolated locality, with low anthropic impacts: Praia do Sul vs Provetá (39.327), Praia do Sul vs Vila do Abraão (33.840) and Praia do Sul vs Parnaioca (6.756).
The first discriminant function (FD1) was the most important to explain group discrimination (proportion of explanation was 0.76 for the FD1, 0.49 for the FD2 and 0.31 for FD3). In decreasing order of importance, the variables that most contributed to FD1 were: W2/W1 (-4.428), W2/L (3.239), W1/L (-2.336), W2 (0.824) and H (0.047). Thus, morphometric indices are better for group samples, that is, shell indices better reflects shell geometrics.
DISCUSSION
The original description of G. ticaga describes shell sculpture with concentric lines and slight radial lines present only on the inner surface of some shells (MARCUS & MARCUS 1962). OHLWEILER & LANZER (1993), when revalidating the species, also considered the absence of radial lines on the teleoconch as a diagnostic character for G. ticaga.
In the present study, the four sampled populations were considered to belong to G. ticaga based on homogeneity of muscle scar shape (Fig. 3) and apex microsculpture (Fig. 2). In natural populations radial lines may range from presence in nearly entire populations (as seen to specimens from Provetá and Praia do Sul) to absence (Vila do Abraão and Parnaioca). In this study 36% of all the studied shells had radial lines. Thus, absence of radial lines is not a good diagnostic character for this species.
In the original description, MARCUS & MARCUS (1962) provided measurements of some specimens, one of which was 6.5 mm long, 5.0 mm wide and 1.3 mm high. LANZER (1996) reported a maximum length of 4.5 mm, width of 2.9 mm and height of 1.3 mm, for specimens from many localities in the state of Rio Grande do Sul, southern Brazil. The specimens analyzed by us are similar in size to those studied by LANZER (1996), but smaller than those studied by MARCUS & MARCUS (1962) with two exceptions: two specimens from Provetá that reached more than 5 mm. It is important to consider that the specimens used in the original description came from a single aquarium population from the Instituto de Biociências da Universidade de São Paulo (MARCUS & MARCUS 1962). It is possible that those specimens were larger and taller than those observed in natural populations (S.B. Santos, personal observation). This would explain, at least in part, the original description of G. ticaga as Uncancylus ticagus.
Extrinsic factors such as a greater input of organic matter and light in streams seem to affect shell size, resulting in populations with larger individuals, as found in Provetá, followed by Vila do Abraão. The absence of riparian vegetation, which results in increased light on the stream bed, associated with greater input of organic matter, calcium availability and alkalinity (DELAY & PONTIER 1997, LANZER 2001), result in a greater availability of food (periphytic algae) (DILLON 2000) and may result in more pronounced growth. SANTOS (2003) observed that, in impacted streams in the state of Rio de Janeiro, larger specimens of G. ticaga are frequent.
In fresh water ecosystems, the alteration and destruction of habitats through the removal of the riparian forest and pollution by domestic sewage create conditions that may cause or accelerate intraspecific morphological variations (DURRANT 1977, LANZER 1996). Shell variability influenced by the environment has been reported for an unidentified Laevapecinae, as demonstrated by the long-time study of MACMAHON (2004). Ecophenotypic plasticity in shell morphology is common to many freshwater limpet Ancylidae species, being in part responsible for nominal species (HUBENDICK 1964, WALTHER et al. 2006) and taxa identified to sub-familial or to generic categories only (MCMAHON 2004).
The population from Praia do Sul was the most different, according to the F-matrix among groups. The difference could be explained by a combination of geographic isolation and possible environmental differences, such as a better conserved habitat and sedimentary deposit formation, contrasting with the other places, where granitic rocks prevail (AMADOR 1988). Otherwise, the morphological similarity observed between the populations of Praia do Sul and Parnaioca could be explained by a greater geographic proximity of both localities in the ocean side of Ilha Grande (Fig. 1) Although Provetá is closer to Praia do Sul than Parnaioca, and also located on the ocean side, its streams show marked organic pollution by domestic sewage, probably affecting shell morphology. These hypotheses can be tested in the future by genetic and molecular analyses.
Within population variation was observed mainly in the samples coming from altered environments, i.e. Vila do Abraão, Provetá and Parnaioca, which had three different shell patterns each. The samples from Praia do Sul, a well preserved environment, showed only two patterns.
Despite within population shell variation, a "morphological identity" was recognized in each locality. (Tabs I and II). The morphological variation of the shells in the ancylid populations of Ilha Grande is consistent with the morphological identities suggested in the classification matrix created from shells measurement data (Tab. III). It is probable that variation among populations is influenced by environmental factors. PFENNINGER et al. (2003) showed an ecological differentiation of Ancylus fluviatilis Müller, 1774 in Europe and MCMAHON (2004) showed the same for Laevapex fuscus (Adams, 1841) in the United States.
The shells from Praia do Sul and Parnaioca are elliptical-rounded, whereas those from Vila do Abraão and Provetá are elliptical-elongated and narrower at the posterior region. These two sites had the most similar shells, although they are the most distant from each other (Fig. 1). It is important to notice that the morphological groups identified in the visual analysis for each population (Tab. I) was very congruent with the percentage of successful identifications calculated in the DA (Tab. III) for each population. This congruence highlights the importance of the participation of a competent morphologist in systematic studies, someone who is able to discriminate among intra and interpopulation differences or cryptic species and avoid misidentifications. Although the simplicity of the pateliform shell would suggest the opposite, its variability among individuals highlights the need to study more than one population to better define species.
Gundachia ticaga show ample phenotypic plasticity in shell morphology. As the radial lines were present in at least 30% of the studied shells, we cannot consider absence of radial lines as a good diagnostic character.
ACKNOWLEDGEMENTS
We thank Conselho Nacional para o Desenvolvimento Científico e Tecnológico (CNPq) for S.B. Santos grant (47668204) and for scholarships to L.E.M. de Lacerda (2007-2008) and I.C. Miyahira (2005-2007); IBAMA for license SISBIO 10812-1; Instituto Estadual de Florestas (IEF) for license 18/2007; Reserva Biológica da Praia do Sul for licence 2/2006; Centro de Estudos Ambientais e Desenvolvimento Sustentável da Universidade do Estado do Rio de Janeiro (Ceads/UERJ) for logistic support; the Laboratory of Malacology staff for helping with field work and the anonymous reviewers for their valuable suggestions.
LITERATURE CITED
AMADOR, E.S. 1988. Geologia e geomorfologia da planície costeira da Praia do sul - Ilha Grande - Uma contribuição à elaboração do plano diretor da Reserva Biológica. Anuário do Instituto de Geociências/UFRJ 11: 35-58. BASCH, P. 1963. A review of the recent freshwater limpet snails of North America. Bulletin of the Museum of Comparative Zoology 129: 399-461.
CHIU, Y.; H. CHEN; S. LEE & C.A. CHEN. 2002. Morphometric analysis of shell and operculum variations in the Viviparid snail, Cipangopaludina chinensis (Mollusca: Gastropoda), in Taiwan. Zoological Studies 41 (3): 321-331.
CIPRIANI, R. 2007. Modelando las conchas de los moluscos, o la búsqueda de la espiral perfecta, p. 3-11. In: S.B. SANTOS; A.D. PIMENTA; S.C. THIENGO; M.A. FERNANDEZ & R.S. ABSALÃO (Eds). Tópicos em Malacologia - Ecos do XVIII Encontro Brasileiro de Malacologia. Rio de Janeiro, Ed. Sociedade Brasileira de Malacologia, XIV+365p.
DELAY, B. & J.P. POINTIER. 1997. Factors affecting the distribution and abundance of two Prosobranch snails in a thermal spring. Journal of Freshwater Ecology 12 (1): 75-79.
DILLON, R.T. 2000. The ecology of freshwater mollusks. Cambridge, Cambridge University Press, 449p.
DURRANT, P.M. 1977. Some factors that affect the distribution of Ancylus fluviatilus (Müller) in the rivers systems of Great Britain. Journal of Molluscan Studies 43: 67-78.
ENGELMAN, K. 1997. SYSTAT 7.0. Chicago, SPSS Inc Press, 7th ed., 421p.
HUBENDICK, B. 1964. Studies on Ancylidae. The subgroups. Göteborgs Kungl Vetenskap-Och Vitterhets- Samhalles Handdlinger 9B (6): 1-72.
HUBENDICK, B. 1978. Systematics and comparative morphology of the Basommatophora, p. 1-48. In: V. FRETTER & J. PEAKE (Eds). Pulmonates. London, Academic Press, vol. 2A, XI + 540p.
KLECKA, W.R. 1982. Discriminant Analysis. Sage University Paper Series on Quantitative Applications in the Social Sciences, 07-0119. Beverly Hills, California Sage Publications, 72p.
KREBS, J.C. 1998. Ecological Methodology. New York, Benjamin Cummings, XII+620p.
LANZER, R.M. 1996. Ancylidae (Gastropoda: Basommatophora) na América do Sul: sistemática e distribuição. Revista Brasileira de Zoologia 13: 175-210.
LANZER, R.M. 2001. Distribuição, fatores históricos e dispersão de moluscos límnicos em lagoas do sul do Brasil. Biociências 9: 63-84.
LANZER, R.M. & I.L. VEITENHEIMER-MENDES. 1985. Aspectos morfológicos e biológicos de uma população de G. concentrica (Orbigny, 1835) (Mollusca: Ancylidae) de um açude do sul do Brasil. Iheringia, Série Zoologia, 65: 41-56.
MACIEL, N.C.; D.S.D. ARAUJO & A. MAGNANINI. 1984. Reserva Biológica Estadual da Praia do Sul (Ilha Grande, Angra dos Reis, RJ): Contribuição para o conhecimento da fauna e flora. Fundação Brasileira para a Conservação da Natureza 19: 126-148.
MARCUS, E. & E. MARCUS. 1962. On Uncancylus ticagus. Boletim da Faculdade de Filosofia Ciências e Letras da Universidade de São Paulo, Zoologia 24: 217-245.
MCMAHON, R.F. 2004. A 15-year study of interannual shell shape variation in a population of freshwater limpets (Pulmonata: Basommatophora: Ancylidae). American Malacological Bulletin 19: 101-109.
MCMAHON, R.F. & B.E. WHITEHEAD. 1987. Environmental induction of shell morphometric variation in the European stream limpet, Ancylus fluviatilis (Müller) (Pulmonata: Basommatophora). American Malacological Bulletin 5: 105-124.
MYERS, N.; R.A. MITTERMEIER; C.G. MITTERMEIER; G.A.B. FONSECA & J. KENT. 2000. Biodiversity hotspots for conservation priorities. Nature 403: 853-858.
OHLWEILER, F.P. & R.M. LANZER. 1993. Morfologia da concha, rádula e mandíbula de Gundlachia obliqua (Broderip & Sowerby, 1832) como uma contribuição à sistemática de Ancylidae. Biociências 1: 121-149.
OHLWEILER, F.P. & R.M. LANZER. 1994. Morfologia de Gundlachia obliqua (Broderip & Sowerby, 1832) (Mollusca, Gastropoda, Ancylidae). Iheringia, Série Zoologia, 77: 113-127.
PFENNINGER, M.; S. STAUBACH; C.ALBRECHT; B. STREIT & K. SCHWENK. 2003. Ecological and morphological differentiation among cryptic evolutionary lineages in freshwater limpets of the nominal form-group Ancylus fluviatilis (O.F. Müller, 1774). Molecular Ecology 12: 2731-2745.
PRADO, R.M. 2003. Tensão no paraíso: aspectos da intensificação do turismo na Ilha Grande. Caderno Virtual de Turismo 3 (1):1-9.
PRADO, R.M. 2006. Ilha Grande - do sambaqui ao turismo. Rio de Janeiro, Garamond, 288p.
SANTOS, M.S. 2009. Os porões da República. A barbárie nas prisões da Ilha Grande, 1894-1945. Rio de Janeiro, Garamond. 336p.
SANTOS, S.B. 2003. Estado atual do conhecimento dos ancilídeos na América do Sul. (Mollusca, Gastropoda, Pulmonata, Basommatophora). Revista de Biologia Tropical 50 (Supl. 3): 191-224.
SANTOS, S.B.; L.E.M. LACERDA & I.C. MIYAHIRA. 2009. Uncancylus concentricus (Mollusca, Gastropoda, Ancylidae): New ocurrence in state of Rio de Janeiro, Brazil. Check List 5 (3): 513-517.
STRONG, E.E.; O. GARGOMINY; W.F. PONDER & P. BOUCHET. 2008. Global diversity of gastropods (Gastropoda; Mollusca) in freshwater. Hydrobiologia 595: 149-166.
THIENGO S.C.; M.A. FERNANDEZ; M.F. BOAVENTURA & M.A. STORTTI. 1998. A survey of gastropods in the Microrregião Serrana of the Rio de Janeiro, Brazil. Memórias do Instituto Oswaldo Cruz 93 (Suppl. 1): 233-234.
THIENGO S.C.; M.A. FERNANDEZ; M.F. BOAVENTURA; C.E. GRAULT; H.F.R. SILVA; A.C. MATTOS & S.B. SANTOS. 2001. Freshwater snails and schistosomiasis mansoni in the state of Rio de Janeiro, Brazil: I - Metropolitan mesoregion. Memórias do Instituto Oswaldo Cruz 96 (Suppl.): 177-184.
THIENGO S.C.; M.A. FERNANDEZ; M.F. BOAVENTURA; S.B. SANTOS & A.C MATTOS. 2002a. Freshwater snails and schistosomiasis mansoni in the state of Rio de Janeiro, Brazil: II - Centro Fluminense Mesoregion. Memórias do Instituto Oswaldo Cruz 97: 621-626.
THIENGO S.C.; M.A. FERNANDEZ; M.F. BOAVENTURA; M.G. MAGALHÃES & S.B. SANTOS. 2002b. Freshwater snails and schistosomiasis mansoni in the state of Rio de Janeiro, Brazil: III - Baixadas Mesoregion. Memórias do Instituto Oswaldo Cruz 97 (Suppl. 1): 43-46.
THIENGO S.C.; A.C. MATTOS; M.F. BOAVENTURA & M.A. FERNANDEZ. 2004a. Freshwater snails and schistosomiasis mansoni in the state of Rio de Janeiro, Brazil: IV - Sul Fluminense mesoregion. Memórias do Instituto Oswaldo Cruz 99: 275-280.
THIENGO S.C.; A.C. MATTOS; M.F. BOAVENTURA; M.S. LOUREIRO; S.B. SANTOS & M.A. FERNANDEZ. 2004b. Freshwater snails and schistosomiasis mansoni in the state of Rio de Janeiro, Brazil: V - Norte Fluminense mesoregion. Memórias do Instituto Oswaldo Cruz 99 (Suppl.1): 99-103.
THIENGO S.C.; S.B. SANTOS & M.A. FERNANDEZ. 2005. Malacofauna límnica da área de influência do lago da usina hidrelétrica de Serra da Mesa, Goiás, Brasil. I Estudo qualitativo. Revista Brasileira de Zoologia 22 (4): 867-874.
THIENGO S.C.; A.C. MATTOS; M.A. FERNANDEZ & S.B. SANTOS. 2006. Freshwater snails and schistosomiasis mansoni in the state of Rio de Janeiro, Brazil: VI - Noroeste Fluminense Mesoregion. Memórias do Instituto Oswaldo Cruz 101 (Suppl. 1): 239-245.
WALTHER A.C.; T. LEE, J.B. BURCH & D.Ó. FOIGHIL. 2006. Confirmation that the North American ancylid Ferrissia fragilis (Tryon, 1863) is a cryptic invader of European and East Asian freshwater ecosystems. Journal of Molluscan Studies 72 (3): 318-321.
WULLSCHLEGER, E.B. & J. JOKELA. 2002. Morphological plasticity and divergence in life-history traits between two closely related freshwater snails; Lymnea ovata and Lymnaea peregra. Journal of Molluscan Studies 68: 1-5.
ZAR J.H. 1999. Biostatistical Analysis. New Jersey, Prentice-Hall, 663p.
Submitted: 20.V.2010;
Accepted: 21.03.2011.
Editorial responsibility: Rosana M. da Rocha
- AMADOR, E.S. 1988. Geologia e geomorfologia da planície costeira da Praia do sul - Ilha Grande - Uma contribuição à elaboração do plano diretor da Reserva Biológica. Anuário do Instituto de Geociências/UFRJ 11: 35-58.
- BASCH, P. 1963. A review of the recent freshwater limpet snails of North America. Bulletin of the Museum of Comparative Zoology 129: 399-461.
- CHIU, Y.; H. CHEN; S. LEE & C.A. CHEN. 2002. Morphometric analysis of shell and operculum variations in the Viviparid snail, Cipangopaludina chinensis (Mollusca: Gastropoda), in Taiwan. Zoological Studies 41 (3): 321-331.
- CIPRIANI, R. 2007. Modelando las conchas de los moluscos, o la búsqueda de la espiral perfecta, p. 3-11. In: S.B. SANTOS; A.D. PIMENTA; S.C. THIENGO; M.A. FERNANDEZ & R.S. ABSALÃO (Eds). Tópicos em Malacologia - Ecos do XVIII Encontro Brasileiro de Malacologia. Rio de Janeiro, Ed. Sociedade Brasileira de Malacologia, XIV+365p.
- DELAY, B. & J.P. POINTIER. 1997. Factors affecting the distribution and abundance of two Prosobranch snails in a thermal spring. Journal of Freshwater Ecology 12 (1): 75-79.
- DILLON, R.T. 2000. The ecology of freshwater mollusks. Cambridge, Cambridge University Press, 449p.
- DURRANT, P.M. 1977. Some factors that affect the distribution of Ancylus fluviatilus (Müller) in the rivers systems of Great Britain. Journal of Molluscan Studies 43: 67-78.
- ENGELMAN, K. 1997. SYSTAT 7.0. Chicago, SPSS Inc Press, 7th ed., 421p.
- HUBENDICK, B. 1964. Studies on Ancylidae. The subgroups. Göteborgs Kungl Vetenskap-Och Vitterhets- Samhalles Handdlinger 9B (6): 1-72.
- HUBENDICK, B. 1978. Systematics and comparative morphology of the Basommatophora, p. 1-48. In: V. FRETTER & J. PEAKE (Eds). Pulmonates. London, Academic Press, vol. 2A, XI + 540p.
- KLECKA, W.R. 1982. Discriminant Analysis. Sage University Paper Series on Quantitative Applications in the Social Sciences, 07-0119. Beverly Hills, California Sage Publications, 72p.
- KREBS, J.C. 1998. Ecological Methodology. New York, Benjamin Cummings, XII+620p.
- LANZER, R.M. 1996. Ancylidae (Gastropoda: Basommatophora) na América do Sul: sistemática e distribuição. Revista Brasileira de Zoologia 13: 175-210.
- LANZER, R.M. 2001. Distribuição, fatores históricos e dispersão de moluscos límnicos em lagoas do sul do Brasil. Biociências 9: 63-84.
- LANZER, R.M. & I.L. VEITENHEIMER-MENDES. 1985. Aspectos morfológicos e biológicos de uma população de G. concentrica (Orbigny, 1835) (Mollusca: Ancylidae) de um açude do sul do Brasil. Iheringia, Série Zoologia, 65: 41-56.
- MACIEL, N.C.; D.S.D. ARAUJO & A. MAGNANINI. 1984. Reserva Biológica Estadual da Praia do Sul (Ilha Grande, Angra dos Reis, RJ): Contribuição para o conhecimento da fauna e flora. Fundação Brasileira para a Conservação da Natureza 19: 126-148.
- MARCUS, E. & E. MARCUS. 1962. On Uncancylus ticagus. Boletim da Faculdade de Filosofia Ciências e Letras da Universidade de São Paulo, Zoologia 24: 217-245.
- MCMAHON, R.F. 2004. A 15-year study of interannual shell shape variation in a population of freshwater limpets (Pulmonata: Basommatophora: Ancylidae). American Malacological Bulletin 19: 101-109.
- MCMAHON, R.F. & B.E. WHITEHEAD. 1987. Environmental induction of shell morphometric variation in the European stream limpet, Ancylus fluviatilis (Müller) (Pulmonata: Basommatophora). American Malacological Bulletin 5: 105-124.
- MYERS, N.; R.A. MITTERMEIER; C.G. MITTERMEIER; G.A.B. FONSECA & J. KENT. 2000. Biodiversity hotspots for conservation priorities. Nature 403: 853-858.
- OHLWEILER, F.P. & R.M. LANZER. 1993. Morfologia da concha, rádula e mandíbula de Gundlachia obliqua (Broderip & Sowerby, 1832) como uma contribuição à sistemática de Ancylidae. Biociências 1: 121-149.
- OHLWEILER, F.P. & R.M. LANZER. 1994. Morfologia de Gundlachia obliqua (Broderip & Sowerby, 1832) (Mollusca, Gastropoda, Ancylidae). Iheringia, Série Zoologia, 77: 113-127.
- PFENNINGER, M.; S. STAUBACH; C.ALBRECHT; B. STREIT & K. SCHWENK. 2003. Ecological and morphological differentiation among cryptic evolutionary lineages in freshwater limpets of the nominal form-group Ancylus fluviatilis (O.F. Müller, 1774). Molecular Ecology 12: 2731-2745.
- PRADO, R.M. 2003. Tensão no paraíso: aspectos da intensificação do turismo na Ilha Grande. Caderno Virtual de Turismo 3 (1):1-9.
- PRADO, R.M. 2006. Ilha Grande - do sambaqui ao turismo. Rio de Janeiro, Garamond, 288p.
- SANTOS, M.S. 2009. Os porões da República. A barbárie nas prisões da Ilha Grande, 1894-1945. Rio de Janeiro, Garamond. 336p.
- SANTOS, S.B. 2003. Estado atual do conhecimento dos ancilídeos na América do Sul. (Mollusca, Gastropoda, Pulmonata, Basommatophora). Revista de Biologia Tropical 50 (Supl. 3): 191-224.
- SANTOS, S.B.; L.E.M. LACERDA & I.C. MIYAHIRA. 2009. Uncancylus concentricus (Mollusca, Gastropoda, Ancylidae): New ocurrence in state of Rio de Janeiro, Brazil. Check List 5 (3): 513-517.
- STRONG, E.E.; O. GARGOMINY; W.F. PONDER & P. BOUCHET. 2008. Global diversity of gastropods (Gastropoda; Mollusca) in freshwater. Hydrobiologia 595: 149-166.
- THIENGO S.C.; M.A. FERNANDEZ; M.F. BOAVENTURA & M.A. STORTTI. 1998. A survey of gastropods in the Microrregião Serrana of the Rio de Janeiro, Brazil. Memórias do Instituto Oswaldo Cruz 93 (Suppl. 1): 233-234.
- THIENGO S.C.; M.A. FERNANDEZ; M.F. BOAVENTURA; C.E. GRAULT; H.F.R. SILVA; A.C. MATTOS & S.B. SANTOS. 2001. Freshwater snails and schistosomiasis mansoni in the state of Rio de Janeiro, Brazil: I - Metropolitan mesoregion. Memórias do Instituto Oswaldo Cruz 96 (Suppl.): 177-184.
- THIENGO S.C.; M.A. FERNANDEZ; M.F. BOAVENTURA; S.B. SANTOS & A.C MATTOS. 2002a. Freshwater snails and schistosomiasis mansoni in the state of Rio de Janeiro, Brazil: II - Centro Fluminense Mesoregion. Memórias do Instituto Oswaldo Cruz 97: 621-626.
- THIENGO S.C.; M.A. FERNANDEZ; M.F. BOAVENTURA; M.G. MAGALHÃES & S.B. SANTOS. 2002b. Freshwater snails and schistosomiasis mansoni in the state of Rio de Janeiro, Brazil: III - Baixadas Mesoregion. Memórias do Instituto Oswaldo Cruz 97 (Suppl. 1): 43-46.
- THIENGO S.C.; A.C. MATTOS; M.F. BOAVENTURA & M.A. FERNANDEZ. 2004a. Freshwater snails and schistosomiasis mansoni in the state of Rio de Janeiro, Brazil: IV - Sul Fluminense mesoregion. Memórias do Instituto Oswaldo Cruz 99: 275-280.
- THIENGO S.C.; A.C. MATTOS; M.F. BOAVENTURA; M.S. LOUREIRO; S.B. SANTOS & M.A. FERNANDEZ. 2004b. Freshwater snails and schistosomiasis mansoni in the state of Rio de Janeiro, Brazil: V - Norte Fluminense mesoregion. Memórias do Instituto Oswaldo Cruz 99 (Suppl.1): 99-103.
- THIENGO S.C.; S.B. SANTOS & M.A. FERNANDEZ. 2005. Malacofauna límnica da área de influência do lago da usina hidrelétrica de Serra da Mesa, Goiás, Brasil. I Estudo qualitativo. Revista Brasileira de Zoologia 22 (4): 867-874.
- THIENGO S.C.; A.C. MATTOS; M.A. FERNANDEZ & S.B. SANTOS. 2006. Freshwater snails and schistosomiasis mansoni in the state of Rio de Janeiro, Brazil: VI - Noroeste Fluminense Mesoregion. Memórias do Instituto Oswaldo Cruz 101 (Suppl. 1): 239-245.
- WALTHER A.C.; T. LEE, J.B. BURCH & D.Ó. FOIGHIL. 2006. Confirmation that the North American ancylid Ferrissia fragilis (Tryon, 1863) is a cryptic invader of European and East Asian freshwater ecosystems. Journal of Molluscan Studies 72 (3): 318-321.
- WULLSCHLEGER, E.B. & J. JOKELA. 2002. Morphological plasticity and divergence in life-history traits between two closely related freshwater snails; Lymnea ovata and Lymnaea peregra Journal of Molluscan Studies 68: 1-5.
- ZAR J.H. 1999. Biostatistical Analysis. New Jersey, Prentice-Hall, 663p.
Publication Dates
-
Publication in this collection
25 July 2011 -
Date of issue
June 2011
History
-
Received
20 May 2010 -
Accepted
21 Mar 2011